Oral Candida Carriage and Morphotype Differentiation in Chronic Periodontitis Patients with and without Diabetes in the Indian Sub-Continent
Abstract
:1. Introduction
2. Results
| Parameters | CP | CPD | p-Value (CP vs. CPD) |
|---|---|---|---|
| PI | 1.2 ± 0.34 | 1.09 ± 0.35 | NS |
| GI | 1.17 ± 0.43 | 0.92 ± 0.21 | NS |
| PD | 6.15 ± 0.84 | 6.29 ± 0.99 | NS |
| CAL | 6.36 ± 1.01 | 6.45 ± 1.18 | NS |
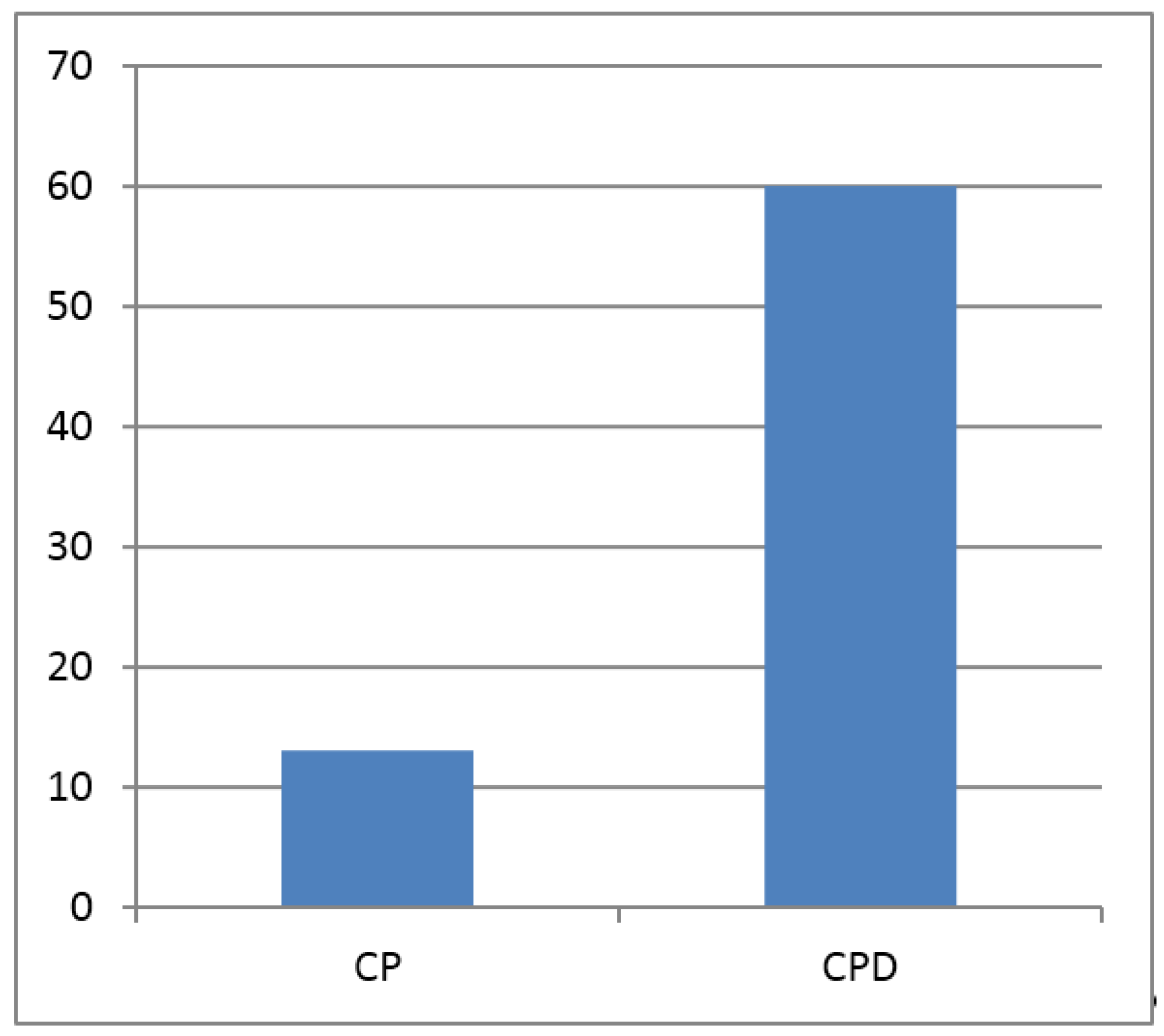
| Parameters | CP | CPD | p-Value (CP vs. CPD) |
|---|---|---|---|
| FBS | 97.33 ± 4.83 | 153.26 ± 47.37 | S |
| CFU | 0.05 ± 0.04 | 0.33 ± 0.23 | S |
3. Discussion
4. Materials and Methods
4.1. Study Population
4.2. Inclusion and Exclusion Criteria
4.3. Clinical Analysis
4.4. Fasting Blood Glucose (FBS)
4.5. Collection of Whole Saliva
4.6. Microbial Culture and Analysis
4.7. Statistical Analysis
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| CP | chronic periodontitis | CFU | colony forming units |
| CPD | chronic periodontitis with diabetes | PAS | Periodic acid-Schiff |
| PI | plaque index | OPD | out-patient department |
| GI | gingival index | PMPE | partial mouth periodontal protocols |
| PD | probing depth | BEC | buccal epithelial cells |
| CAL | clinical attachment level | PMN | polymorphonuclear neutrophils |
| FBS | fasting blood sugar |
References
- Sardi, J.C.; Duque, C.; Camargo, G.A.; Hofling, J.F.; Gonçalves, R.B. Periodontal conditions and prevalence of putative periodontopathogens and Candida spp. In insulin-dependent type 2 diabetic and non-diabetic patients with chronic periodontitis—A pilot study. Arch. Oral Biol. 2011, 56, 1098–1105. [Google Scholar] [CrossRef] [PubMed]
- Canabarro, A.; Valle, C.; Farias, M.; Santos, F.; Lazera, M.; Wanke, B. Association of subgingival colonization of Candida albicans and other yeasts with severity of chronic periodontitis. J. Periodontal Res. 2013, 48, 428–432. [Google Scholar] [CrossRef] [PubMed]
- Maddi, A.; Scannapieco, F.A. Oral biofilms, oral and periodontal infections, and systemic disease. Am. J. Dent. 2013, 26, 249–254. [Google Scholar] [PubMed]
- Cuesta, A.; Jewtuchowicz, V.; Brusca, M.; Nastri, M.; Rosa, A. Prevalence of Staphylococcus spp and Candida spp in the oral cavity and periodontal pockets of periodontal disease patients. Acta Odontol. Latinoam. 2009, 23, 20–26. [Google Scholar]
- Järvensivu, A.; Hietanen, J.; Rautemaa, R.; Sorsa, T.; Richardson, M. Candida yeasts in chronic periodontitis tissues and subgingival microbial biofilms in vivo. Oral Dis. 2004, 10, 106–112. [Google Scholar] [CrossRef] [PubMed]
- Teles, R.; Teles, F.; Frias-Lopez, J.; Paster, B.; Haffajee, A. Lessons learned and unlearned in periodontal microbiology. Periodontology 2013, 62, 95–162. [Google Scholar] [CrossRef] [PubMed]
- Hannula, J.; Dogan, B.; Slots, J.; Ökte, E.; Asikainen, S. Subgingival strains of Candida albicans in relation to geographical origin and occurrence of periodontal pathogenic bacteria. Oral Microbiol. Immunol. 2001, 16, 113–118. [Google Scholar] [CrossRef] [PubMed]
- Tapper-Jones, L.; Aldred, M.; Walker, D.; Hayes, T. Candidal infections and populations of Candida albicans in mouths of diabetics. J. Clin. Pathol. 1981, 34, 706–711. [Google Scholar] [CrossRef] [PubMed]
- Mascarenhas, P.; Fatela, B.; Barahona, I. Effect of diabetes mellitus type 2 on salivary glucose—A systematic review and meta-analysis of observational studies. PLoS ONE 2014. [Google Scholar] [CrossRef] [PubMed]
- Mealey, B.L. Periodontal disease and diabetes: A two-way street. J. Am. Dent. Assoc. 2006, 137, S26–S31. [Google Scholar] [CrossRef]
- Emrich, L.J.; Shlossman, M.; Genco, R.J. Periodontal disease in non-insulin-dependent diabetes mellitus. J. Periodontol. 1991, 62, 123–131. [Google Scholar] [CrossRef] [PubMed]
- Tran, D.T.; Gay, I.; Du, X.L.; Fu, Y.; Bebermeyer, R.D.; Neumann, A.S.; Streckfus, C.; Chan, W.; Walji, M.F. Assessing periodontitis in populations: A systematic review of the validity of partial-mouth examination protocols. J. Clin. Periodontol. 2013, 40, 1064–1071. [Google Scholar] [CrossRef] [PubMed]
- Yuan, K.; Chang, C.J.; Hsu, P.C.; Sun, H.S.; Tseng, C.C.; Wang, J.R. Detection of putative periodontal pathogens in non-insulin-dependent diabetes mellitus and non-diabetes mellitus by polymerase chain reaction. J. Periodontal Res. 2001, 36, 18–24. [Google Scholar] [CrossRef] [PubMed]
- Slutsky, B.; Buffo, J.; Soll, D.R. High-frequency switching of colony morphology in Candida albicans. Science 1985, 230, 666–669. [Google Scholar] [CrossRef] [PubMed]
- Darwazeh, A.; Lamey, P.J.; Samaranayake, L.; MacFarlane, T.; Fisher, B.; Macrury, S.; MacCuish, A. The relationship between colonisation, secretor status and in vitro adhesion of Candida albicans to buccal epithelial cells from diabetics. J. Med. Microbiol. 1990, 33, 43–49. [Google Scholar] [CrossRef] [PubMed]
- Barros, L.M.; Boriollo, M.F.; Alves, A.C.B.; Klein, M.I.; Gonçalves, R.B.; Höfling, J.F. Genetic diversity and exoenzyme activities of Candida albicans and Candida dubliniensis isolated from the oral cavity of brazilian periodontal patients. Arch. Oral Biol. 2008, 53, 1172–1178. [Google Scholar] [CrossRef] [PubMed]
- Maccarinelli, G.; Belotti, R.; Savoldi, E.; Gervasoni, M.; Cocchi, D. Phagocytosis and killing of candida albicans of polymorphonuclear cells in patients with organ transplant of periodontal disease. Miner. Stomatol. 2000, 50, 345–349. [Google Scholar]
- Reynaud, A.; Nygaard-Østby, B.; Bøygard, G.K.; Eribe, E.; Olsen, I.; Gjermo, P. Yeasts in periodontal pockets. J. Clin. Periodontol. 2001, 28, 860–864. [Google Scholar] [CrossRef] [PubMed]
- Hägewald, S.; Bernimoulin, J.P.; Köttgen, E.; Kage, A. Salivary iga subclasses and bacteria-reactiveiga in patients with aggressive periodontitis. J. Periodontal Res. 2002, 37, 333–339. [Google Scholar] [CrossRef] [PubMed]
- Zambon, J.J.; Reynolds, H.S.; Genco, R.J. Studies of the subgingival microflora in patients with acquired immunodeficiency syndrome*. J. Periodontol. 1990, 61, 699–704. [Google Scholar] [CrossRef] [PubMed]
- Williams, D.W.; Lewis, M.A.O. Oral microbiology: Isolation and identification of Candida from the oral cavity. Oral Dis. 2000, 6, 3–11. [Google Scholar] [CrossRef] [PubMed]
- Manfredi, M.; McCullough, M.; Al-Karaawi, Z.; Vescovi, P.; Porter, S. In vitro evaluation of virulence attributes of Candida spp. Isolated from patients affected by diabetes mellitus. Oral Microbiol. Immunol. 2006, 21, 183–189. [Google Scholar] [CrossRef] [PubMed]
- Armitage, G.C. Development of a classification system for periodontal diseases and conditions. Ann. Periodontol. 1999, 4, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Samaranayake, L.; MacFarlane, T. Host factors and oral candidosis. In Oral Candidosis; Samaranayake, L.P., MacFarlane, T.W., Eds.; Oral Candidosis: Wright, London, 1990; pp. 66–103. [Google Scholar]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Venkatesan, G.; Uppoor, A.; Naik, D.; Kadkampally, D.; Maddi, A. Oral Candida Carriage and Morphotype Differentiation in Chronic Periodontitis Patients with and without Diabetes in the Indian Sub-Continent. Dent. J. 2015, 3, 123-131. https://doi.org/10.3390/dj3040123
Venkatesan G, Uppoor A, Naik D, Kadkampally D, Maddi A. Oral Candida Carriage and Morphotype Differentiation in Chronic Periodontitis Patients with and without Diabetes in the Indian Sub-Continent. Dentistry Journal. 2015; 3(4):123-131. https://doi.org/10.3390/dj3040123
Chicago/Turabian StyleVenkatesan, Gomathinayagam, Ashita Uppoor, Dilip Naik, David Kadkampally, and Abhiram Maddi. 2015. "Oral Candida Carriage and Morphotype Differentiation in Chronic Periodontitis Patients with and without Diabetes in the Indian Sub-Continent" Dentistry Journal 3, no. 4: 123-131. https://doi.org/10.3390/dj3040123
APA StyleVenkatesan, G., Uppoor, A., Naik, D., Kadkampally, D., & Maddi, A. (2015). Oral Candida Carriage and Morphotype Differentiation in Chronic Periodontitis Patients with and without Diabetes in the Indian Sub-Continent. Dentistry Journal, 3(4), 123-131. https://doi.org/10.3390/dj3040123





