Abstract
One of the most significant public health issues of the twenty-first century is antimicrobial resistance (AMR), which is responsible for thousands of deaths each year and undermines the efficacy of commonly used antibiotics. In dentistry, the frequent and sometimes inappropriate use of antibiotics, combined with the complexity of the oral microbiome, creates a high-risk environment for the development and spread of antimicrobial-resistant microorganisms. Although clinical infections have received most of the attention, dental unit waterlines, dentures, and orthodontic appliances are three reservoirs that play an important but underestimated role in this global crisis. These environments provide perfect conditions for biofilm formation and, further, the survival and growth of resistant microorganisms. This review aims to discuss the origins and mechanisms of AMR, the unrecognized role of these three reservoirs in dental settings, and their contribution to the AMR issue. It also highlights the necessity of strict infection control procedures and targeted antimicrobial stewardship strategies to overcome this growing threat.
1. Introduction
Antimicrobial resistance (AMR) has arisen as one of the most concerning public health concerns of the 21st century. AMR poses a threat to the effective treatment and prevention of a growing number of infections caused by bacteria, parasites, viruses, and fungi that are no longer susceptible to the antibiotics [1]. AMR was directly responsible for 1.27 million deaths in 2019 and was linked to approximately 5 million deaths globally [2]. According to one systematic analysis in 2022, the highest mortality rates are in western sub-Saharan Africa and South Asia, and the lowest in Australasia [3]. Escherichia coli, Staphylococcus aureus, Klebsiella pneumoniae, Streptococcus pneumoniae, Acinetobacter baumannii, and Pseudomonas aeruginosa caused nearly three-quarters of attributable deaths, mainly from lower respiratory, bloodstream, and intra-abdominal infections [3]. Resistance to β-lactams and fluoroquinolones accounted for over 70% of the burden, with the greatest impact in low-resource settings [3]. The UK government-commissioned review concluded that AMR may kill 10 million people annually by 2050 [4,5]. According to WHO and academic literature, AMR is a serious issue that must be addressed with a coordinated and worldwide plan [1,6,7,8].
In dental practice, antibiotics are typically prescribed to treat both non-odontogenic and odontogenic infections, as well as prophylactically in high-risk patients, such as those with systemic conditions such as endocarditis or congenital heart disease, to prevent focal infections. Antibiotics are also prescribed to prevent localized, systemic, and surgical site infections in patients undergoing oral surgery or dental procedures [9]. Dentistry, whether for therapeutic or prophylactic uses, accounts for almost 10% of global antibiotic prescriptions. However, inappropriate antibiotic prescribing could lead to misuse or overuse of antibiotics in dental practice [9,10]. Additionally, patient non-adherence to prescribed antimicrobial treatments further exacerbates the misuse and overuse of antibiotics in dentistry [11]. Consequently, AMR in dentistry remains a significant public health issue [12].
Several domains within dentistry contribute to the challenge of AMR. Previous systematic reviews have covered AMR in antibiotic prophylaxis in dental implants and tooth extractions, odontogenic infections, and periodontal diseases. A systematic review and meta-analysis of antibiotic prophylaxis in dental implants and extractions highlighted the risk of accelerating AMR through unnecessary prescriptions [13]. Other reviews have focused on odontogenic infections and reported resistance to antibiotics such as penicillin, clindamycin, and amoxicillin among Staphylococcus, Streptococcus, Klebsiella, and Enterococcus [14,15]. Another systematic review of AMR in periodontal diseases indicated that periodontal pathogens could develop resistance to popular antibiotics such as metronidazole, clindamycin, and amoxicillin [16].
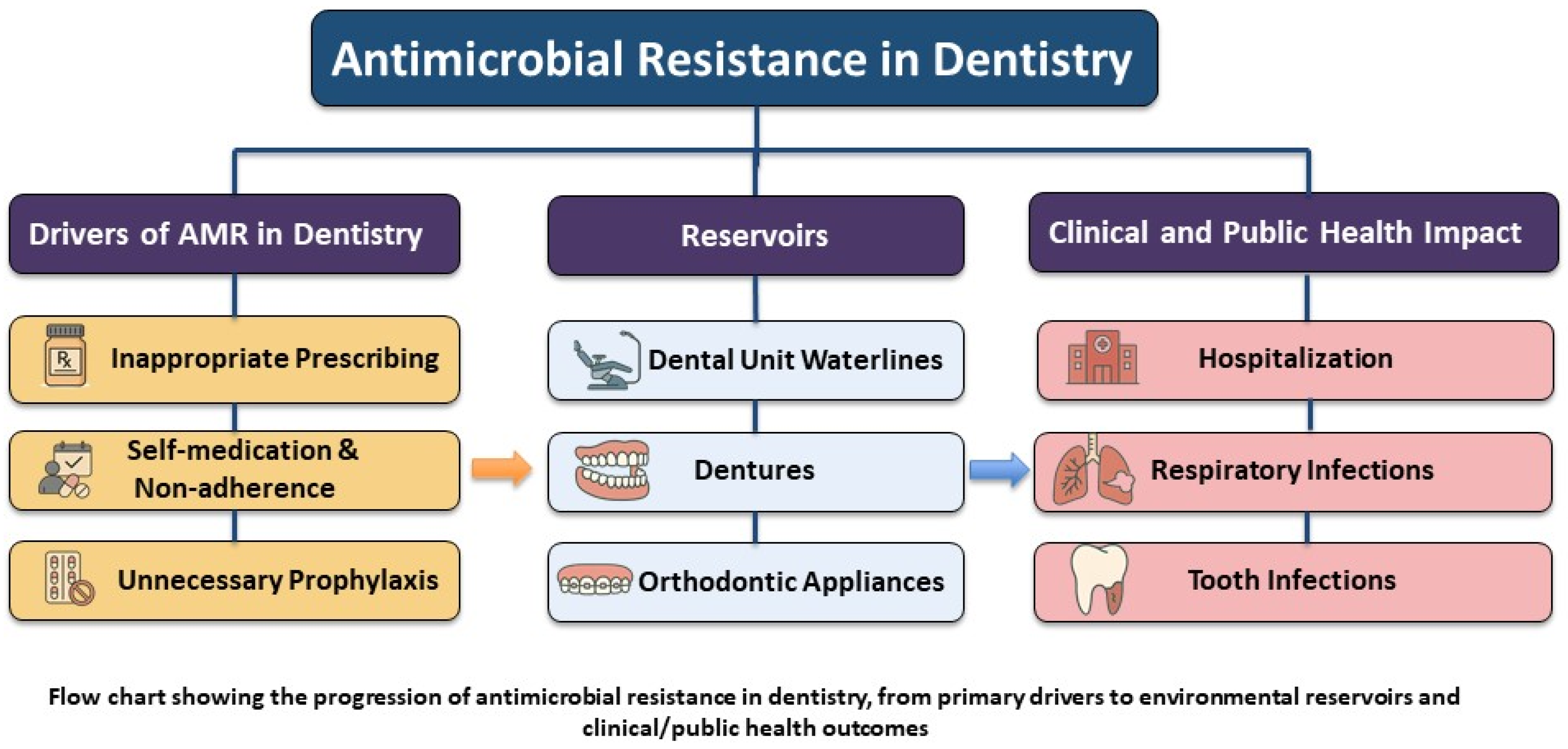
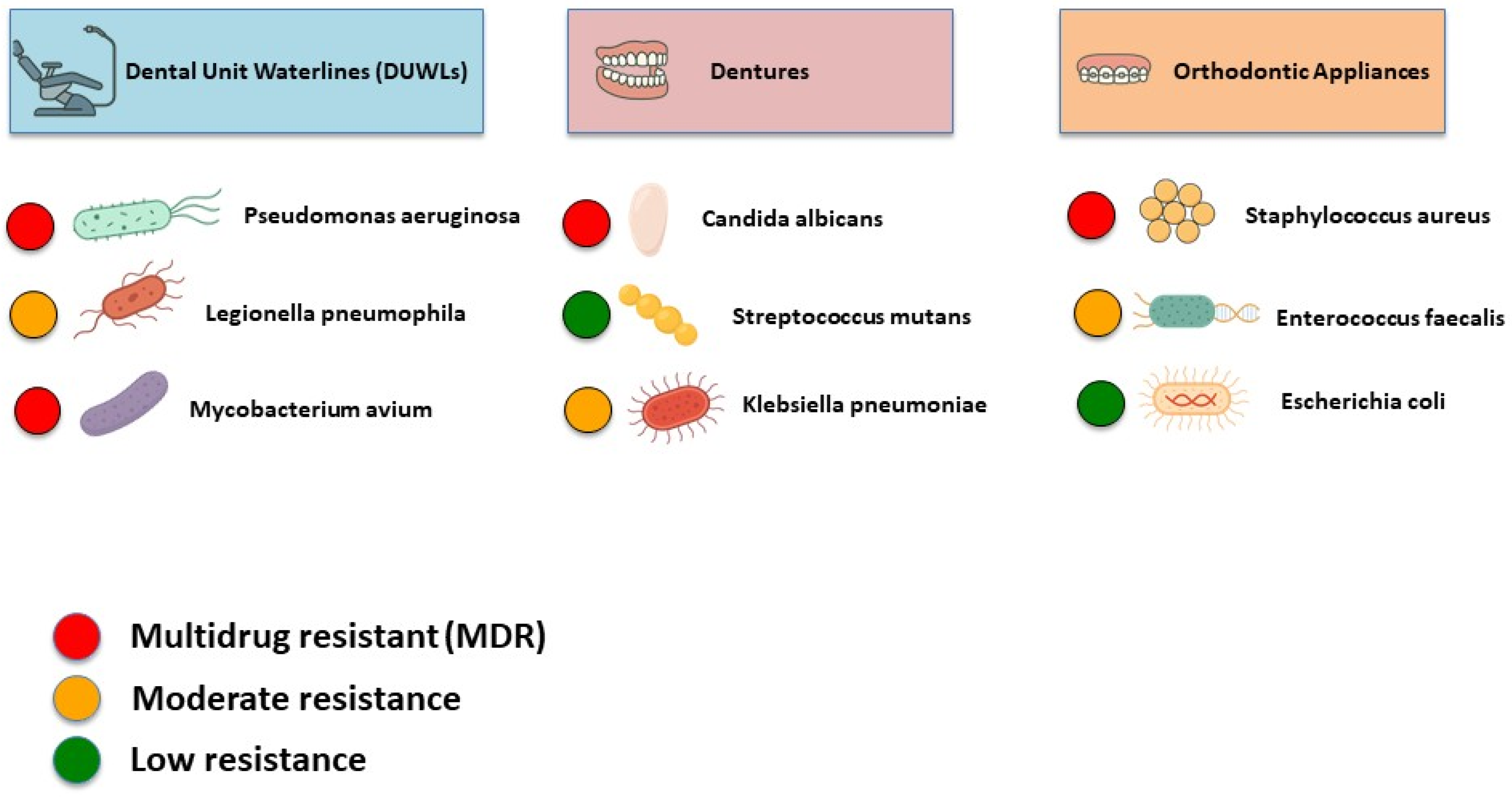
There are three overlooked but significant reservoirs of antibiotic-resistant bacteria in dental settings: dental unit waterlines (DUWLs), dentures, and orthodontic appliances (Figure 1) [17,18,19]. Persistent biofilms are maintained in these environments, offering a safe niche for resistant microorganisms to survive, evolve, and spread [17,18,19]. Such environments call for targeted infection control measures and antimicrobial stewardship (AMS) strategies to mitigate the risk of resistance transmission in dental settings. AMS in dentistry is in its early stages, with no systematic, coordinated, or consensus-driven approach [20]. Moreover, there is a gap in research, particularly in the context of non-infectious reservoirs such as DUWLs, dentures, and orthodontic appliances, which are often underestimated in current AMS strategies. Regarding the growing threat of AMR, further investigation is needed to determine how well current infection control and AMS strategies address the potential role of these reservoirs in the development and spread of resistant microorganisms.
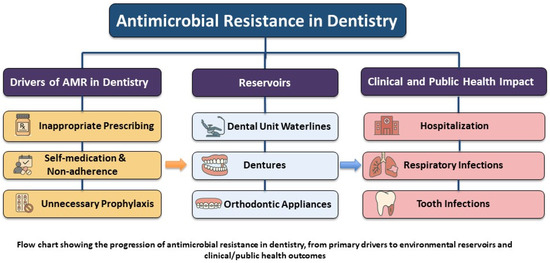
Figure 1.
Overall scope of AMR in dentistry.
Thus, this narrative review aims to highlight the role of DUWLs, dentures, and orthodontic appliances as overlooked reservoirs of antibiotic-resistant microorganisms and to determine the need for targeted infection control measures and AMS strategies to address these critical but under-researched areas in dental settings. Relevant articles were selected based on their relevance and contribution to the topic. As this is a narrative review, no formal risk of bias or quality assessment was conducted.
2. Origins of Antimicrobial Resistance
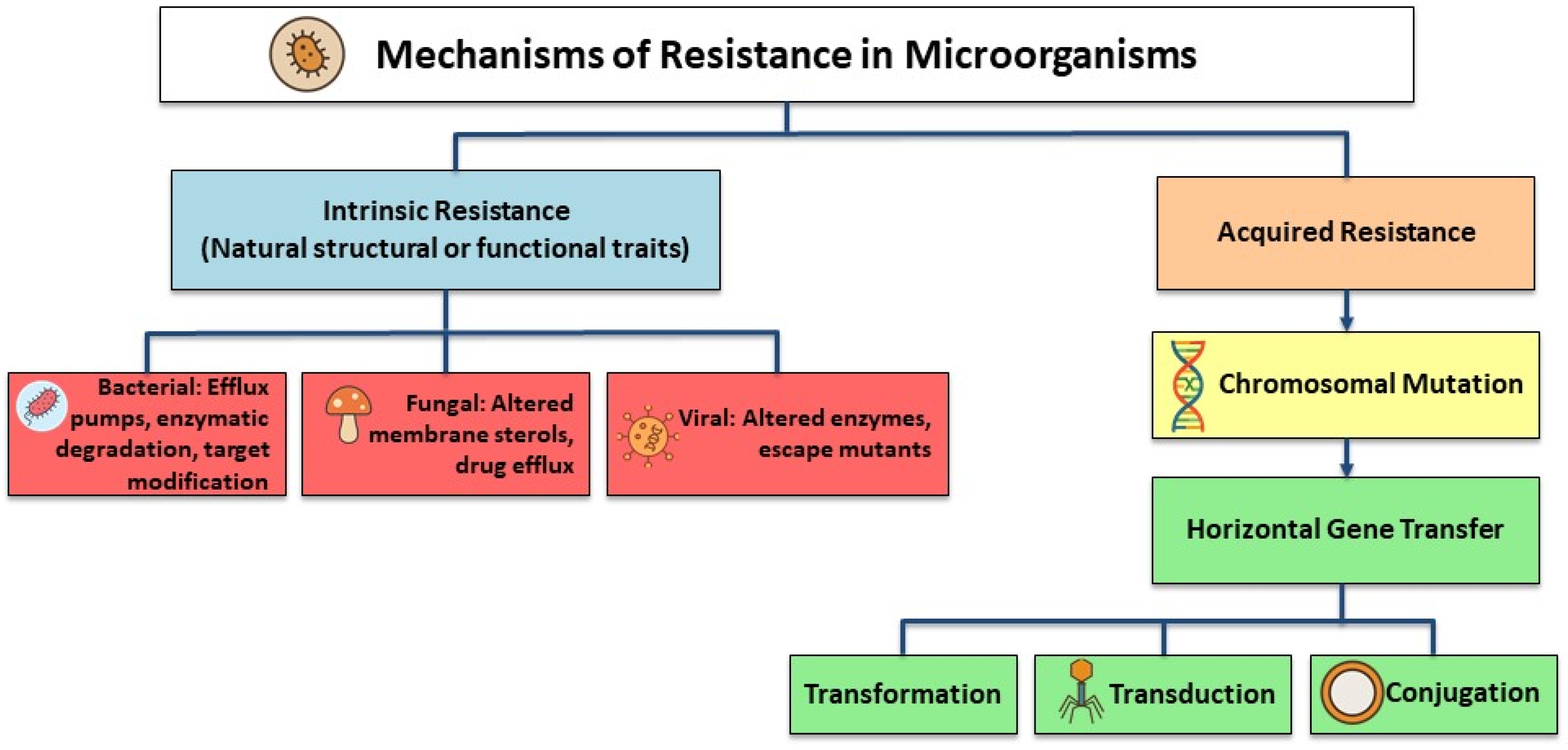
There are two types of AMR: intrinsic (natural) resistance and acquired resistance. Intrinsic resistance is a common trait shared by all strains of a particular species of microorganism, arises independently of previous antibiotic exposure, and is unrelated to horizontal gene transfer (Figure 2) [21,22]. The main bacterial mechanisms that contribute to intrinsic resistance are the outer membrane’s decreased permeability, particularly due to the presence of lipopolysaccharide (LPS) in Gram-negative bacteria, and the natural activity of efflux pumps, which actively expel antimicrobial agents from the cell [21].
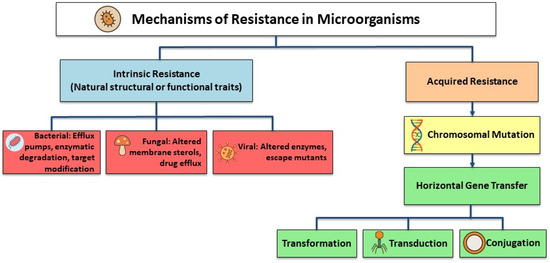
Figure 2.
Primary AMR mechanisms.
On the other hand, acquired resistance does not affect the entire species of bacteria; it only affects specific strains. There are two mechanisms for acquired resistance. The first is chromosomal mutation, where changes to the microorganism’s DNA occur spontaneously, whether the antibiotics were used or not [23]. For instance, chromosomal mutations in bacteria can alter the penicillin-binding proteins (PBPs), which are the site of action for penicillin antibiotics, giving the bacteria resistance to the antibiotic [24].
The second mechanism of acquired resistance is horizontal gene transfer, which allows resistant genes to spread between different bacterial species and strains of the same species [25]. Horizontal gene transfer is one of the most concerning mode of resistance [23], with three mechanisms: transformation, transduction, and conjugation [24]. Transformation is when bacteria with antibiotic resistance genes (ARGs) release pieces of their DNA into the environment, where they can be absorbed by nearby bacteria. Transduction follows similar steps, except that a phage particle containing the DNA segment with the resistance genes delivers the genes to the recipient bacteria. Conjugation is the direct transfer of a plasmid from a donor bacterium to a recipient through physical contact [26].
Since chromosomal mutations are limited by the bacterial life cycle, horizontal gene transfer allows bacteria to develop antibiotic resistance more rapidly. However, horizontal gene transfer enables resistance genes to proliferate among bacteria regardless of their growth rate. Conjugation is considered the most common route of horizontal gene transfer, allowing genetic material to be transferred directly between bacterial cells. Both in human populations and in the environment, this mechanism plays a major role in the spread of genes that confer resistance to antibiotics and the development of multi-drug-resistant bacteria [23].
3. Mechanisms of Resistance
The four primary categories of AMR mechanisms are (1) active drug efflux, (2) inactivating a drug, (3) limiting drug uptake, and (4) modifying the drug target.
- Active drug efflux: Energy-dependent transport proteins, such as those from the ATP-binding cassette (ABC) family or major facilitator superfamily (MFS), pump antimicrobial agents out of the cell before they reach their target concentration [27]. In Gram-negative bacteria, efflux systems like AcrAB-TolC can expel multiple classes of antibiotics, leading to multi-drug resistance [28].
- Drug inactivation: Bacteria produce enzymes that chemically modify or destroy the antimicrobial agent. Classic examples include β-lactamases, which hydrolyse the β-lactam ring in penicillins and cephalosporins, and aminoglycoside-modifying enzymes that acetylate, phosphorylate, or adenylate the drug, making it ineffective [29].
- Limiting drug uptake: Alterations in outer membrane porins or cell wall structure reduce the permeability of the bacterial cell envelope, and as a result, it can prevent antibiotics from entering. This is common in Gram-negative bacteria, where porin loss or modification limits the uptake of β-lactams and fluoroquinolones [30].
- Target modification: Genetic mutations or enzymatic alterations change the binding site of the drug so that it no longer interacts effectively [31]. For instance, mutations in PBPs confer resistance to β-lactams, while alterations in DNA gyrase or topoisomerase II lead to fluoroquinolone resistance [32].
Due to the outer membrane and efflux capabilities in Gram-negative bacteria, these bacteria can use all four main mechanisms. On the other hand, Gram-positive bacteria have fewer types of efflux pumps and less commonly limit drug uptake [33].
Mechanisms of antifungal resistance are expelling antifungal agents via efflux pumps (ABC and MFS transporters), enzymatic drug inactivation, modifying drug targets such as ergosterol biosynthesis (ERG11 mutations in azole resistance), and reduced drug uptake by altering membrane composition [34]. Viruses develop resistance to antiviral drugs and vaccines primarily through random mutations, which can change important viral enzymes, including proteases or polymerases. Furthermore, segmented viruses, such as influenza, can develop resistance through genetic reassortment, which involves exchanging gene segments with other viral strains [35]. Although the mechanism of resistance used by bacteria, fungi, and viruses differ due to structural and metabolic variations, they are all contributors to the problem of AMR.
4. Antimicrobial Resistance in Dental Settings
AMR is a growing concern within the dental field [36]. The oral cavity hosts a wide range of microorganisms, many of which exist in biofilms. Consequently, the oral cavity creates a perfect environment for the spread of antibiotic resistance through horizontal gene transfer [37].
The frequent use of prophylactic antibiotics in extraction and implant procedures is one of the root causes of AMR in dentistry. Antibiotics are prescribed for high-risk patients; however, they are frequently given to healthy people unnecessarily, which leads to oral bacterial resistance [13]. Due to selective pressure from misuse, common medicines such as amoxicillin are no longer as effective against Streptococcus spp. and anaerobic bacteria [38,39,40].
Beyond surgical prophylaxis, empirical antibiotic therapy without bacterial culture and susceptibility testing is commonly used to treat odontogenic infections, including periapical abscesses. Key pathogens such as Prevotella spp., Fusobacterium spp., and Streptococcus spp. have become more resistant, with some strains acquiring beta-lactamase-mediated resistance to amoxicillin and penicillin [40,41,42]. When once-effective antibiotics fail, dentists have to turn to second-line options, which can accelerate the resistance trends [43].
In dental plaques, a wide range of bacterial species can share and acquire resistance genes [44,45]. While antibiotics and scaling are used together to manage periodontal diseases, this approach could unintentionally promote the survival and proliferation of resistant strains such as Aggregatibacter actinomycetemcomitans and Porphyromonas gingivalis [46].
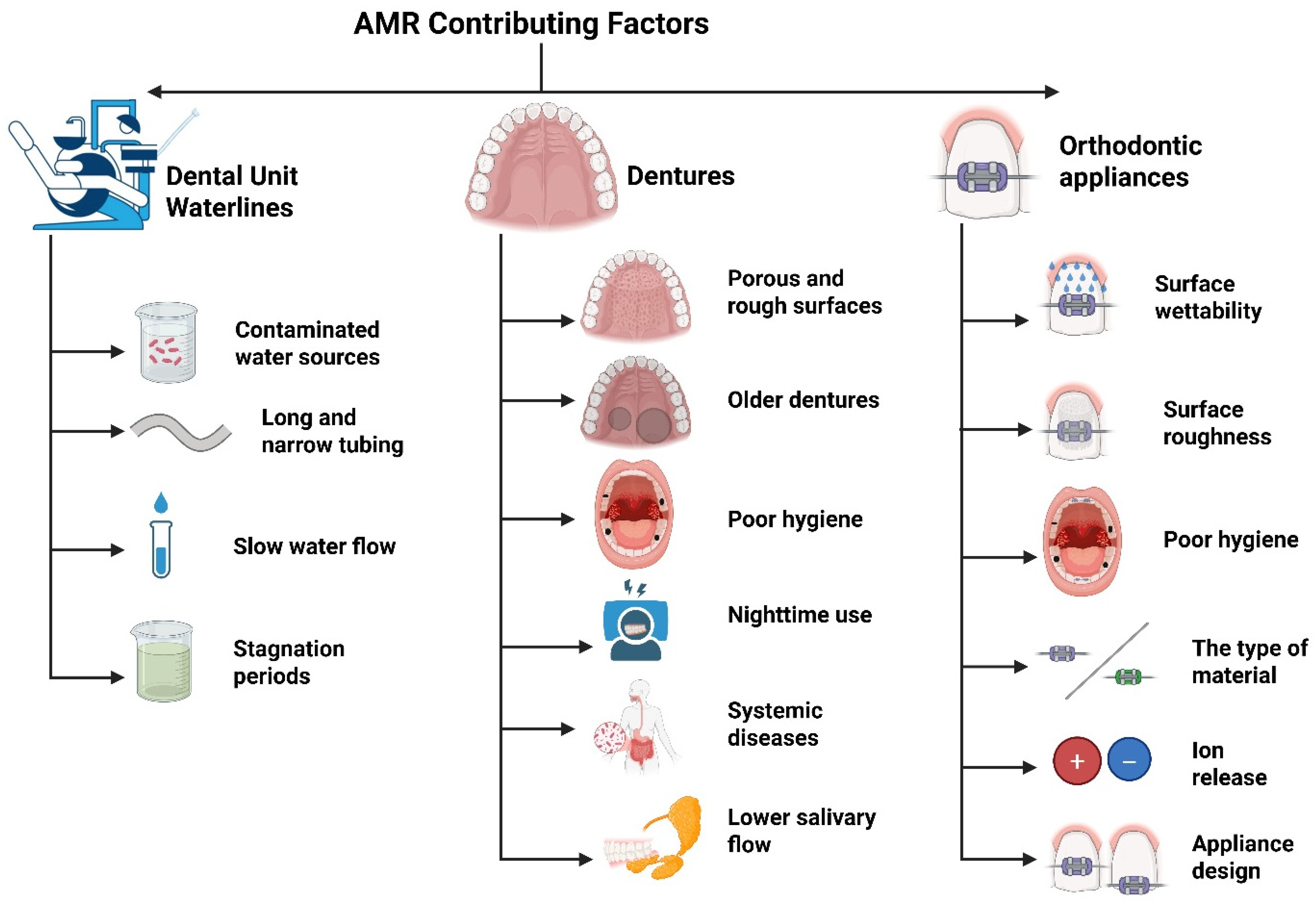
Although much of the current research on AMR in dentistry has focused on improper antibiotic use and clinical infections, other factors may also play a role. For instance, DUWLs, dentures, and orthodontic appliances create ideal conditions for biofilm formation and the spread of ARGs and contribute to the broader AMR problem. Since these areas are frequently exposed to oral microorganisms and may not be subjected to adequate, regular cleaning or maintenance protocols, resistant strains can survive and proliferate. Thus, it is crucial to investigate the role these environments play in the broader context of AMR and determine if targeted strategies are required to address them. Figure 3 summarizes the major factors contributing to AMR in these environments.
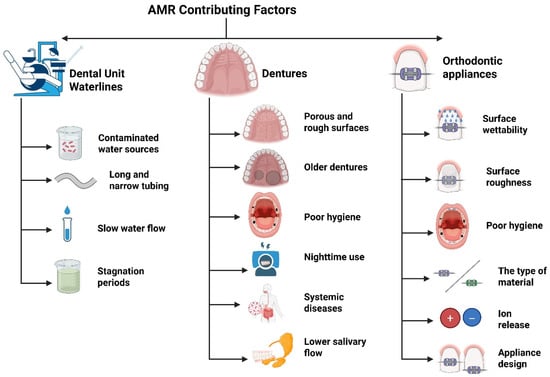
Figure 3.
Major AMR contributors in DUWLs, dentures, and orthodontic appliances.
4.1. Antimicrobial Resistance in DUWLs
4.1.1. Biofilm Formation in DUWLs and Its Role in Antimicrobial Resistance
Dental unit waterlines (DUWLs) are a complex system of tubes that supply water to rotating tools and ultrasonic scalers to cool the teeth and instruments during dental procedures and irrigate the operative site [47]. This system comprises valves, connectors, and approximately 6 m of plastic tubing usually made of polyvinyl or polyurethane, with internal diameters ranging from 0.5 to 2 mm [47]. Within this system, the maximum speed of water flow occurs at the center and gradually decreases toward the periphery and finally reaches zero at the tube walls [47]. Microorganisms contaminating DUWLs often originate from the municipal water supply [48]. Additionally, patient-derived sources, such as the retrograde flow of saliva into the waterlines, facilitate the entry of oral microbes into the system. This backflow often occurs at the end of the use of rotary dental instruments use, when the cessation or reduction of water flow creates a suction effect [49,50].
There are some contributing factors that facilitate biofilm formation inside DUWLs [51]. The tubes are long and narrow, and the slow water flow, typical of many dental procedures, creates a perfect environment for microbial buildup. Periods of stagnation, such as overnight, on weekends, or during holidays when dental clinics are closed, further promote biofilm formation [52,53]. These biofilms consist of microorganisms such as bacteria, fungi, protozoa, and viruses, all embedded in an extracellular matrix of polysaccharides and proteins [54].
Microorganisms within a biofilm are more difficult to remove than free-floating (planktonic) bacteria since they are more resistant to both antibiotics and the host’s immune system [55]. As a result, eliminating a mature biofilm often requires significantly higher doses of antimicrobial agents. Only a small number of cells in the biofilm are directly exposed to the antibiotic, since the surrounding exopolysaccharide matrix acts as a barrier, limiting the drug’s ability to penetrate the deeper layers [56]. The effective concentration of some antibiotics against biofilm-associated bacteria can be up to 100 to 1000 times higher [57]. Furthermore, the bacteria in biofilms are usually sessile, meaning they grow and divide very slowly due to their low metabolic activity [58]. Therefore, antibiotics that target actively dividing cells are often ineffective. Further, in biofilms, the close physical arrangement of bacterial cells may promote the horizontal transfer of genes, indicating that ARGs may be more easily shared in these bacterial communities [55,59,60].
4.1.2. Common Microorganisms in DUWLs
Tap water contains low levels of opportunistic pathogens such as S. aureus, P. aeruginosa, Legionella pneumophila, Mycobacterium tuberculosis, and nontuberculous mycobacteria (NTM); biofilms inside DUWLs can provide a favorable environment for these microorganisms to multiply [61,62,63]. Water samples from DUWLs have been found to contain a variety of microorganisms, including L. pneumophila, P. aeruginosa, Staphylococcus spp., Streptococcus spp., Enterobacter spp., Enterococcus spp., viruses, and fungi [50,64,65].
The water from DUWLs also forms aerosols containing these microorganisms during procedures; these aerosols can be inhaled by dental staff and patients. During other procedures, water from DUWLs comes into direct contact with surgical sites in the oral cavity. Thus, the microbiological quality of water from DUWLs is critically important [61]. Contamination of DUWLs poses a potential health risk, particularly for vulnerable groups such as the elderly, children, immunocompromised individuals, pregnant women, smokers, and individuals with chronic diseases. Additionally, dental personnel are frequently exposed to aerosols and water from DUWLs during clinical procedures, placing them at occupational risk of infection, even if they are otherwise healthy [61,66]. One of the most significant cases is the 1995 death of a dentist from pneumonia caused by Legionella dumoffii, which was isolated from a dental unit in California [67]. Furthermore, some studies have revealed that dental professionals have significantly higher antibodies against L. pneumophila than a control group [68,69]. There have also been suspected fatal cases of legionellosis to have originated in dental settings [70,71].
While some of these microorganisms show low pathogenicity, others could be opportunistic and are among the main contributors to hospital-acquired infections. The ‘ESKAPE’ pathogens (Enterococcus faecium, S. aureus, K. pneumoniae, A. baumannii, P. aeruginosa, and Enterobacter spp.) are particularly important, given their role in hospital infections and their ability to ‘escape’ the effects of antimicrobial drugs [72]. In addition to the ESKAPE pathogens, mycobacteria and NTM can contribute to persistent infections in healthcare settings [73,74].
4.1.3. DUWLs and Antimicrobial Resistance
Despite numerous studies having examined the microbial contamination of DUWLs, there is still limited research on ARGs or resistant bacterial strains. One of the few studies was conducted by Vosooghi et al., who investigated NTM in DUWLs from dental centers and offices in Tehran, Iran. Both slow- and fast-growing species, including M. chelonae, M. abscessus, M. fortuitum, and M. simiae, were identified, and the drug susceptibility testing revealed high resistance to several commonly used antibiotics: M. chelonae and M. abscessus were highly resistant to ciprofloxacin, doxycycline, trimethoprim-sulfamethoxazole, and carbapenems such as imipenem and meropenem. M. simiae isolates showed 100% resistance to multiple antibiotics, including rifampin, isoniazid, ethambutol, and streptomycin [75]. Similarly, the antibiotic resistance pattern of P. aeruginosa and other Pseudomonas spp. in DUWLs of a public dentistry clinic in Milan, Italy, was also investigated by Tesauro et al. Out of 44 dental units, P. aeruginosa was found in approximately 23% of the dental units, and Pseudomonas spp. were identified in over 52%. Thirty percent of P. aeruginosa isolates and 31.8% of Pseudomonas spp. isolates were resistant to at least one of the six tested antibiotics (ceftazidime, piperacillin/tazobactam, meropenem, and colistin) according to antibiotic susceptibility testing. Resistance to colistin was the most common finding, which affected 22% of all tested strains. Although less common, multi-drug resistance (resistance to two or more antibiotics) was also detected [47]. Gawish et al. also studied the resistance pattern of P. aeruginosa from DUWLs in Alexandria, Egypt. In contrast to the Tesauro et al. study, P. aeruginosa was detected in only 7.5% of the samples, and all isolates were fully susceptible to the tested antibiotics, including β-lactam, carbapenem, aminoglycoside, fluoroquinolone, and polymyxin [54].
In another investigation by Vincent et al., sixteen P. aeruginosa isolates were obtained from DUWLs, and their genomic characteristics were evaluated using whole-genome sequencing. Some resistance genes, including β-lactam resistance (e.g., PDC-3, PDC-5, PDC-8) and triclosan resistance (triC), were absent in the P. aeruginosa isolates. However, some isolates showed distinct gene acquisition events, especially genes involved in membrane biogenesis, which may enhance environmental adaptation rather than antibiotic resistance. Several other isolates showed insertions in key regulatory genes, including lasR and gacS, which are essential for quorum sensing and biofilm regulation [76]. The genomic profiles of the isolates showed alterations that might affect phenotypic characteristics under selective pressure, even if they were not multi-drug-resistant. Therefore, DUWLs may be an environment where P. aeruginosa develops features that support persistence and, perhaps, future resistance development [76].
Alsehlawi et al. examined the antibiotic susceptibility of L. pneumophila isolated from DUWLs in dental clinics in Najaf, Iraq. Out of 94 water samples, they recovered nine isolates. Strikingly, all of them were completely resistant to a panel of ten antibiotics, including erythromycin, lincomycin, nitrofurantoin, gentamicin, and several β-lactams such as ampicillin, cefepime, and cefotaxime, as well as rifampin. Most isolates (88.8%) also showed resistance to amoxicillin and 77.7% were resistant to tetracycline. There was moderate resistance to doxycycline and chloramphenicol. Based on these results, nearly all isolates (88.8%) were categorized as extensively drug-resistant, with one isolate (11.1%) classified as multi-drug-resistant [77].
In a study of 50 DUWLs in Istanbul, Turkey, 74% of DUWLs showed bacterial levels higher than 200 CFU/mL, which is above American Dental Association’s standard limit [78]. Most isolates were sensitive to cefoperazone, ofloxacin, gentamicin, ciprofloxacin, and piperacillin. Nevertheless, different levels of resistance were observed, particularly among Staphylococcus species. All Staphylococcus isolates showed metronidazole resistance. Staphylococcus sciuri was also resistant to methicillin, rifampicin, erythromycin, and metronidazole, while S. epidermidis was resistant to rifampicin, chloramphenicol, erythromycin, vancomycin, and metronidazole. The risk of nosocomial infection increased due to methicillin-resistant strains [79].
In another study in Izmir, Turkey, Uzel et al. found Pseudomonas, Burkholderia, Acinetobacter, and Bacillus to be the most isolated genera out of 20 DUWLs. Acinetobacter calcoaceticus and Ralstonia pickettii showed multi-drug resistance, with limited susceptibility to piperacillin (30%), gentamicin (5%), and chloramphenicol (10%). In contrast, Pseudomonas stutzerii showed complete susceptibility to all tested drugs. P. aeruginosa showed a higher susceptibility to ceftazidime (77.3%) and meropenem (90.9%) and a lower susceptibility to ampicillin (18.2%) and tetracycline (13.6%) [80].
S. aureus and S. epidermidis isolates from 160 DUWL samples were examined by Lancellotti et al. S. aureus exhibited strong resistance to oxacillin (78%) and clindamycin (78%), while being most vulnerable to ciprofloxacin (97%), amoxicillin-clavulanic acid (92%), and vancomycin (91%). Similarly, S. epidermidis showed complete susceptibility (100%) to vancomycin and ciprofloxacin; however, resistance rates for oxacillin and clindamycin were 79% and 71%, respectively. The detection of oxacillin-resistant staphylococci in DUWL samples raises concern for the presence of methicillin-resistant S. aureus (MRSA) and methicillin-resistant Staphylococcus epidermidis (MRSE) in dental settings [81].
Research on AMR in DUWLs is still relatively limited. The main aim of most research on DUWL contamination is the detection of the most common microorganisms; few studies have explored the resistance patterns and the possibility of horizontal gene transfer. Since both patients and dental staff are frequently exposed to aerosols and water from DUWLs, this knowledge gap presents a growing concern, especially as AMR continues to spread globally [3].
4.1.4. Water Quality and Disinfection Protocols
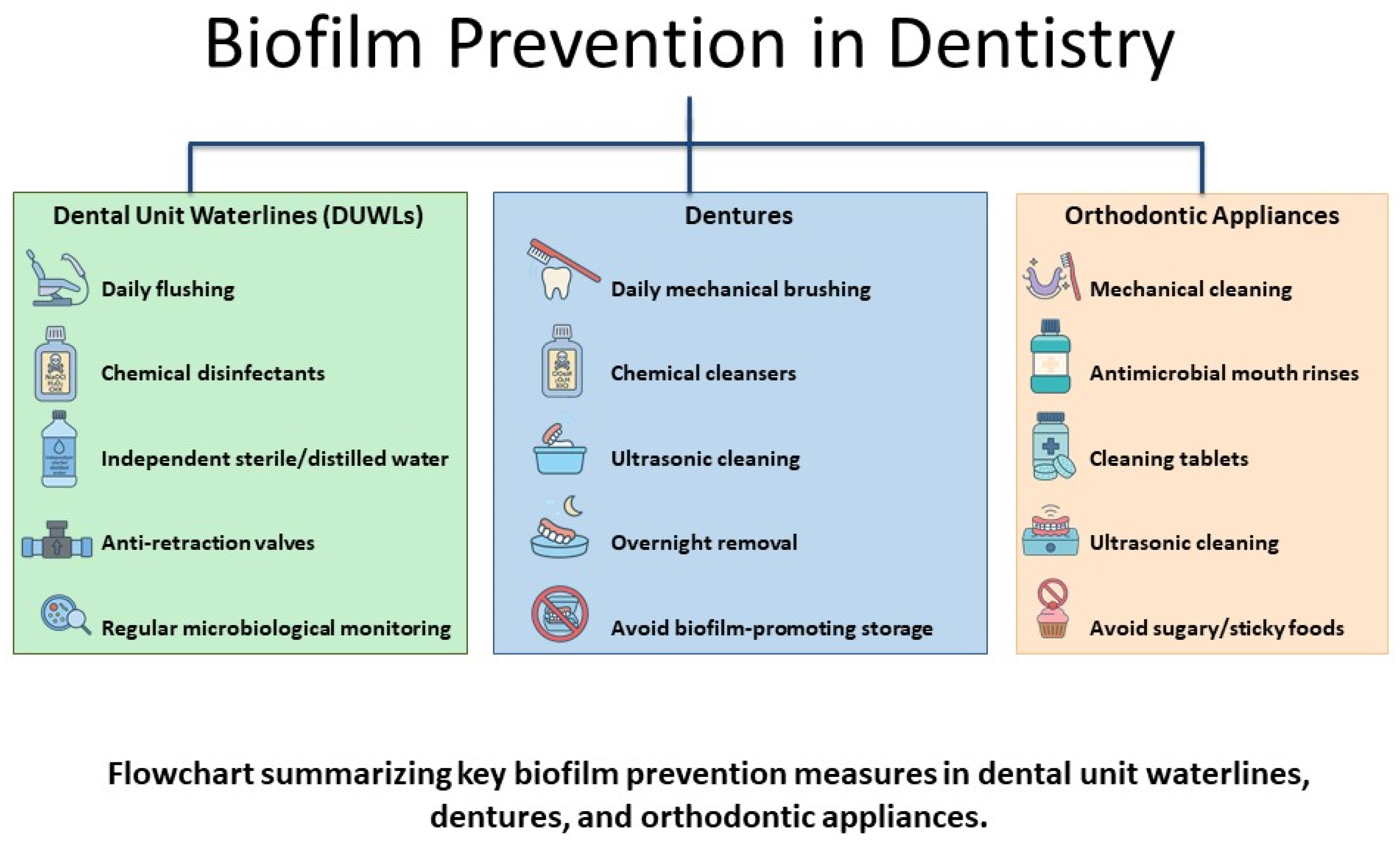
Regulatory authorities recommend that DUWLs should be purged for at least two minutes at the start of each clinic day and flushed for 20 to 30 s after each appointment to minimize biofilm development and lower the microbial load in treatment water [82,83]. Disinfectant tablets should be used regularly, and shock treatments should be administered as necessary in dental units with separate water reservoirs [84,85]. A long-term disinfection protocol using BRS® for shock treatment, Alpron® for continuous use during working hours, and Bilpron® during inactivity achieved 99.8% compliance with microbiological standards in DUWLs. Legionella spp. and P. aeruginosa were consistently undetectable [86]. Both plasma sterilization and low-concentrated ozonized water have emerged as promising alternatives to traditional chemical disinfectants to reduce microbial contamination in DUWLs. However, their current use remains largely experimental, and further long-term clinical validation is needed before they can be recommended for routine implementation in dental practice (Figure 4) [87,88].
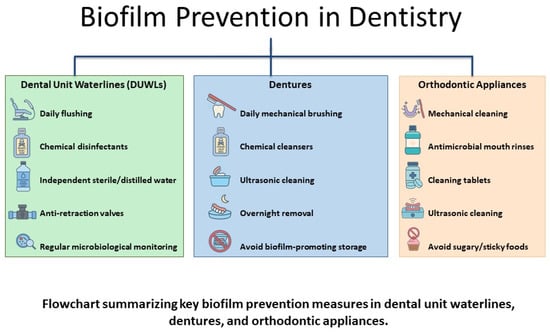
Figure 4.
Biofilm prevention strategies in dentistry.
Non-surgical operations must adhere to the guidelines of Centers for Disease Control and Prevention (CDC) recommending that water contains less than 500 colony-forming units per milliliter (CFU/mL) of heterotrophic bacteria when the output water comes into direct contact with the oral cavity [89]. DUWL testing and maintenance can be done in-house by dental offices or by external laboratory services. For instance, according to the College of Dental Surgeons of Saskatchewan (CDSS), all DUWLs in Saskatchewan, Canada, should undergo testing yearly by an external laboratory; this became mandatory in 2019 [90]. However, these testing requirements and acceptable bacterial contamination levels differ regionally [51,90,91]. Furthermore, many jurisdictions, including Saskatchewan, do not require certain actions or retesting when contaminated dental unit water is found. Additionally, the frequency of DUWL testing and retesting regulations is underreported worldwide [82].
The World Health Organization’s Water Safety Plan Manual has also provided a framework for risk-based water quality management that is directly applicable to healthcare water systems, including DUWLs. Its emphasis on hazard identification, validation of control measures, and continuous operational monitoring aligns with best practices for maintaining microbiological safety in dental waterlines [92]. Moreover, the manual’s guidance on emergency response planning and verification protocols supports the development of strategies to mitigate contamination events and reduce the selective pressure that drives AMR [92]. The European Union’s shifting regulations on drinking water quality could also raise concerns. Under Council Directive 98/83/EC, the absence of P. aeruginosa from drinking water was mandated [93]. However, the recast Directive 2020/2184, which replaces the 1998 Directive, omits P. aeruginosa from its parametric requirements and instead introduces monitoring for Legionella spp. [94]. This regulatory shift has led to a notable gap in microbial surveillance, given the role of P. aeruginosa in biofilm formation within DUWL. While the inclusion of Legionella is a welcome addition, the exclusion of P. aeruginosa may create a blind spot, particularly in dental settings where P. aeruginosa remains a well-known biofilm-forming organism and opportunistic pathogen, especially dangerous to vulnerable patients. Its absence from EU parametric values may reduce the likelihood of its routine monitoring, potentially undermining infection prevention efforts in high-risk clinical environments [95].
4.2. Antimicrobial Resistance in Oral Appliances
Denture surfaces provide optimal places for bacteria and fungi to grow [96]. It is believed that acrylic-based prostheses may be a primary origin of bacterial and fungal adherence because of their porous, rough, and hydrophobic surfaces [97]. Similar findings have emerged in the study of orthodontic appliances, where biofilm formation is also influenced by ion release from metallic components [98], irregular surfaces [99], and difficult-to-clean areas [100]. Thus, AMR and the risk of oral appliance infection are major and common problems in clinical practice [101].
4.2.1. Microbial Contamination and Biofilm Formation on Dentures
The most widely used and accessible material for making dentures and prosthetic teeth is polymethyl methacrylate (PMMA) [102]. Salivary pellicle production is the first step in biofilm formation on dentures. Bacteria and fungi attach to PMMA surfaces within minutes due to short- and long-range physicochemical interactions, Van der Waals forces, hydrophobic interactions, electrostatic interactions, and Brownian motion [102,103]. In their protected exopolysaccharide matrix, these initial microbial colonizers multiply and create multilayered cell clusters [104].
Streptococcus spp. are the most common early colonizers; their presence and metabolites change the microenvironment and create attachment sites that allow subsequent colonizers to adhere and survive. As a crucial bridge species, Fusobacterium nucleatum can connect early colonizers to subsequent, more harmful microbial colonizers, such as P. gingivalis, Treponema spp., Eubacterium spp., Prevotella intermedia, and A. actinomycetemcomitans [105]. As biofilms become more sophisticated and evolve, the oral and denture biofilm microenvironment continues to change. Obligatory anaerobic species can adhere, survive, and multiply within the biofilm when oxygen and nutrients become scarce. Advanced biofilms can contain up to 1000 different types of bacteria [106,107]. Despite the complexity of oral and denture biofilms, it has been demonstrated that Candida spp. may adhere to them at almost every stage of biofilm development [108]. Since Candida. albicans is hydrophobic, it can simply adhere to the hydrophobic PMMA [109]. According to earlier research, the level of Candida colonization and subsequent biofilm formation are correlated with the properties of denture acrylic surfaces [110,111]. The effects of surface roughness, permeability, and porosity on Candida colonization and biofilm formation on the acrylic surface of removable dentures have also been verified by some investigations [112,113].
The literature has provided clear evidence of the biofilm properties of C. albicans and Staphylococcus spp. on denture surfaces [97,114,115,116]. In one study on kidney transplant recipients wearing removable prostheses, dentures contained a substantially greater diversity and number of Staphylococcus strains than the pharyngeal mucosa, particularly S. epidermidis, S. aureus, and Staphylococcus hemolyticus [114]. Some of the pathogenic strains found in biofilm on dentures were similar to those obtained during postoperative infections [114]. In another study, Larijani et al. compared the biofilm characteristics on conventional chemical and heat-polymerized PMMA samples and CAD-CAM PMMA samples, concluding that biofilm formation and C. albicans adhesion on CAD-CAM samples were significantly lower compared to conventional PMMA. Moreover, scanning electron microscopy images showed that biofilms on CAD/CAM surfaces were sparser and morphologically disrupted [116]. However, De Foggi et al. found that although surface roughness increased hydrophobicity, there were no significant differences between the groups with different degrees of surface roughness in terms of biofilm formation of C. albicans [97].
Studies that show long-term microbial survival within denture porosities, even when the prosthesis is not in active use, further support persistent colonization [117,118]. For instance, MRSA was reported to remain viable in a dormant state within biofilms on denture surfaces, and after rehydration, its growth was reactivated [119]. Such findings strongly support that denture, primarily when poorly maintained, function as long-term microbial reservoirs capable of sustaining resistant infections.
4.2.2. Common Resistant Pathogens in Dentures
As previously noted, dentures and prosthetics in the oral cavity are a significant reservoir for AMR due to biofilm formation in the porous acrylic surface [114,115,119,120,121]. Common microorganisms known for AMR include C. albicans, MRSA, Gram-negative bacteria such as Klebsiella spp. and Pseudomonas spp., and Gram-positive bacteria such as Streptococcus spp. [114,115,120,122,123,124,125,126]. Furthermore, the development of resistance to several antibiotic agents such as fluconazole, miconazole, terconazole, ketoconazole, amphotericin B, nystatin, chlorhexidine, chloramphenicol, amoxiclav, ampicillin, clindamycin, doxycycline, and methicillin has been confirmed [124,127,128,129,130]. Some risk factors have been found to contribute to AMR in denture wearers, including inadequate oral hygiene, prolonged use of dentures, systemic diseases, and prior exposure to antibiotics [5,6,11,13]. Consequently, the most effective management strategies could be antifungal/antibacterial therapies, routine disinfection of dentures, improvement of personal hygiene, and regular monitoring for microbiological infections [115,119,123,129].
Candida spp. isolated from denture biofilms could have different levels of resistance to antifungals. For example, a study reported that C. albicans had intermediate resistance to fluconazole, miconazole, and itraconazole, while non-albicans species had higher resistance [129]. In contrast to these results, Koga-Ito et al. found no resistant C. albicans on denture samples [131].
One study reported that C. albicans could become up to 1000 times more resistant to fluconazole and miconazole when grown in a biofilm rather than as free-floating planktonic cells, with minimum inhibitory concentration (MIC) values beyond clinical breakpoints [132]. Chandra et al. further concluded that biofilm-forming strains required 32–128× higher drug concentrations to be effective against Candida spp. compared to planktonic forms. Together, it could be concluded that the biofilm growth on dentures could significantly inhibit antifungal activity [130].
In several studies, C. albicans is often found as the primary fungal pathogen in patients with denture stomatitis, with its prevalence being 60% to 100% in denture wearers [133,134,135]. Even though most isolates have a normal response to the common antifungal drugs, resistance is increasingly reported. For instance, there was a drug resistance to fluconazole in 3.2% of C. albicans isolates, and the drug of choice for these patients was itraconazole in one study [124]. Aside from C. albicans, other Candida species, such as C. glabrata and C. tropicalis, were also highly resistant to fluconazole, with resistance rates approaching 18.4% [127].
In a study by Jewtuchowicz et al., the use of dental devices (both dental prostheses and orthodontic appliances) could significantly increase Candida carriage in subgingival biofilm. In this study, C. albicans was fully susceptible to fluconazole and voriconazole; however, 17% of C. dubliniensis isolates were resistant, and C. guilliermondii showed reduced fluconazole susceptibility [136].
Regarding resistant bacteria, some studies have reported MRSA and coagulase-negative staphylococci on denture surfaces [114,137,138,139]. In a study by Costa et al., all methicillin-resistant Staphylococcus were identified as S. epidermidis, and 94.4% carried the mecA gene [137]. Staphylococcus spp. resistance to amoxicillin, levofloxacin, cefoxitin, erythromycin, and clindamycin have also been reported by several studies [114,120,121]. Gram-negative bacteria such as P. aeruginosa, K. pneumoniae, Enterobacter cloacae, E. coli, and Morganella morganii were found on dentures of the elderly and showed significant resistance to ampicillin (57.4%), cephalothin (41.7%), and tetracycline (36.5%) [122].
4.2.3. Risk Factors Contributing to Antimicrobial Resistance in Denture Wearers
Colonization of dentures by resistant microorganisms is highly affected by risk factors related to the prosthesis condition, patient behavior, and systemic implications. Prosthetic-related risks, such as rough and porous denture surfaces, older dentures, poor hygiene of dentures, and denture trauma due to poor fit, are highly associated with biofilm formation and microbial persistence [137,140,141,142,143,144]. Nighttime use may significantly increase microbial load on denture surfaces and the risk of denture stomatitis, which could lead to the accumulation of resistant bacteria and fungi [141,145,146,147]. Prior exposure to antibiotic medications also plays a role in the emergence of resistant strains. Even without recent use of antibiotics, past exposure may be sufficient to develop adaptive resistance in Candida species [148,149].
Systemic diseases, including diabetes, pneumonia, and compromised immune systems, make it easier for drug-resistant microorganisms to persist [114,129,150]. Furthermore, the oral cavity of denture wearers has lower salivary flow and provides a favorable place for fungal biofilms and various bacteria to grow [151,152,153].
4.2.4. Preventing and Managing Antimicrobial Resistance in Dentures
Pharmaceutical, mechanical, and behavioral interventions are needed to address the problem of AMR among denture wearers (Figure 4).
Pharmacological Strategies
Topical antifungal agents such as nystatin, miconazole, amphotericin B, and clotrimazole are commonly recommended to control Candida overgrowth on the surfaces of dentures. These topical antifungals are typically regarded as first-line options because of their localized effect and lower risk of systemic side effects [154,155,156]. The use of systemic antifungals, such as fluconazole, can lead to resistance, and as a result, they are usually used for more severe cases. In certain situations, it may be necessary to use substitutes such as itraconazole or more recent azoles in strains resistant to fluconazole [157,158]. For biofilm-associated infections, adjuvant treatment options such as silver-based denture disinfectants, and chlorhexidine may provide additional antibacterial activity without leading to AMR [157,159,160,161].
There is growing concern over the persistence of resistant bacteria such as S. aureus, P. aeruginosa, and K. pneumoniae in denture biofilms. These bacteria could develop AMR to commonly used antibiotics due to frequent or inappropriate antibiotic use [120,121]. Therefore, antibacterial approaches often focus on using localized medicines such as chlorhexidine, sodium hypochlorite, or silver nanoparticles (AgNP) rather than systemic antibiotics [157,161]. The incorporation of antimicrobial agents into denture adhesives appears to be effective in improving prosthetic hygiene and eliminating biofilm formation [162]. These antimicrobial materials, such as antimicrobial drugs, nanomaterials, and phytochemical components, including plant extracts, were evaluated for their ability to inhibit microbial growth, particularly C. albicans [162]. Some new approaches are also being researched to target resistance mechanisms, such as the use of efflux pump inhibitors, quorum-sensing blockers, and biofilm-disrupting compounds. These emerging approaches could improve the efficacy of conventional antibiotics and prevent resistance development [163,164,165,166].
Mechanical and Behavioral Interventions
Producing new and well-fitting dentures, regularly brushing the denture, overnight soaking in disinfectants, avoiding overnight denture wear, and regular dental check-ups are much-needed steps to reduce the risk of biofilm formation and further colonization of resistant microorganisms [141,147,167]. Moreover, patients’ awareness of the consequences of self-medication such as the unsupervised and improper use of antibiotics or antifungals should be raised; otherwise, there would be the risk of AMR in denture-associated microorganisms [168].
Altogether, these methods reemphasize a wide-ranging, evidence-based management approach to controlling AMR in individuals with dentures. Preventing biofilm formation, proper medication selection based on susceptibility profiles, and complying with the highest level of prosthesis hygiene are all indispensable for successful infection control and resistance prevention in the long term [169].
4.3. Antimicrobial Resistance in Orthodontic Appliances
Orthodontic appliances, particularly fixed ones, can highly affect the oral microbiome with an increase in periodontopathic Gram-negative bacteria and cariogenic streptococci [170,171,172]. Moreover, wearing orthodontic devices can contribute to the development of new ecological niches, which are likely important in biofilm formation and further colonization by resistant microorganisms [173,174,175,176]. The biofilm formation on orthodontic devices is associated with other problems such as halitosis, periodontitis, gingivitis, dental caries, and white spot lesions [177,178,179].
4.3.1. Biofilm Formation on Orthodontic Devices
Surface wettability, surface roughness, materials used in orthodontic devices, ion release, appliance design, and tooth position can promote microbial adhesion to orthodontic appliances and biofilm formation [99,180,181,182,183].
To bond fixed orthodontic appliances to teeth, the enamel undergoes several treatments, such as acid etching, priming, and adhesive application. Changes in surface characteristics of teeth resulting from the orthodontic bonding process may have a significant impact on the development of biofilm surrounding orthodontic devices, as increased surface wettability and surface roughness of enamel promote bacterial adhesion and biofilm formation [99].
Nickel-titanium (NiTi) arch wires are commonly used because of their outstanding mechanical characteristics, but they can lead to the corrosion of nickel ions. Nickel ions may result in the adhesion and extracellular matrix formation of S. aureus, which are vital for initiating biofilm formation [181]. Additionally, it has been proven that nickel can affect bacteria by functioning as a micronutrient at lower concentrations and having bacteriostatic effects at higher concentrations [184,185]. This phenomenon, which results from exposure to heavy metals, is receiving more interest in the field of antibiotic resistance and bacterial cross-adaptation. Mutations in genes encoding metal and antibiotic targets, biofilm development, efflux pump activation, and reduced membrane permeability can contribute to bacterial adaptation and resistance to heavy metals [186,187,188].
Microbial adherence and biofilm formation may be influenced by the design and material selection of orthodontic brackets and wires. According to one study, wires are more likely than brackets to develop biofilm [180]. Another study indicated that biofilm height and coverage were substantially lower in stainless steel brackets than in ceramic and gold ones [189].
In a study of patients wearing fixed orthodontic appliances, the highest biofilm accumulation was observed on the maxillary lateral incisors and maxillary canines, particularly in the gingival area and areas behind arch wires [183]. Thus, it could be argued that biofilm formation could vary among different teeth in orthodontic patients, though further research is needed to confirm this.
4.3.2. Resistant Microorganisms Among Orthodontic Patients
Studies on AMR among patients receiving orthodontic treatment are limited. Patients with fixed orthodontic appliances have more diverse and resistant microbial populations in their oral cavity than those without, including higher levels of Streptococcus spp., Staphylococcus spp., Enterobacter spp., and Candida spp. In one study, approximately 74% of the isolated bacterial strains from orthodontic patients were resistant to at least one antimicrobial (ampicillin, ampicillin/sulbactam, benzylpenicillin, cefoxitin, cefuroxime, cefuroxime axetil, and clindamycin) [175]. These findings concurred with those of Abbas et al., who showed that S. aureus was more frequently isolated from patients with orthodontic appliances (57%) compared to non-wearers (43%). They also concluded that NiTi and stainless steel wires were associated with increased antibiotic resistance, particularly to bacitracin, ceftazidime, amoxicillin-clavulanic acid, and erythromycin [101]. Similarly, higher AMR and the increased ability for adhesion and biofilm formation on NiTi arch wires among S. aureus strains adapted to moderate nickel concentrations (250 µg/mL) were reported by Pelvic et al. [181]. In another investigation, a significant correlation between recent fixed orthodontic treatment and the presence of the agr gene in isolates of Streptococcus mutans was found. However, no link was observed between the analyzed genes (mecA, pvl, or coa) and using removable braces [190].
In one study, resistant Enterococcus and E. coli were observed in fixed orthodontic appliance wearers with poor oral hygiene, in contrast to the healthy controls without orthodontic appliances. They found resistance to erythromycin, tetracycline, and kanamycin, with some bacteria carrying multiple resistance genes, including erm(B), tet(L), and aph(3′)-IIIa [191]. Jewtuchowicz et al. reported a higher level of Candida spp. in subgingival biofilm of patients using orthodontic appliances compared to non-users. They also observed that C. dubliniensis and C. guilliermondii had some resistance to azoles, including fluconazole and voriconazole [136].
Taken together, these studies suggest that treatment with fixed orthodontic appliances can increase the resilient bacterial populations within oral biofilms (Table 1).

Table 1.
Microbial populations associated with braces and retainers.
4.3.3. Antimicrobial Resistance Prevention in Orthodontic Device Users
Regular oral hygiene practices, avoiding unnecessary antibiotic prescriptions, scheduling routine professional cleanings to disrupt biofilm maturation, and the integration of antimicrobial technologies are all important components of a multifaceted approach to prevent AMR in orthodontic patients [175,183,191,192,193,194]. Patients should be instructed on maintaining good oral hygiene, which includes regular brushing, flossing, and using antimicrobial mouthwashes (Figure 4).
Dental professionals should prescribe antibiotics only when clinically necessary and take steps toward AMS. Additionally, antimicrobial coatings, such as AgNP coatings on brackets and wires, have demonstrated potential in decreasing microbial colonization and biofilm development [195,196]. Other promising materials for orthodontic wire treatment include Teflon (PTFE), zirconium dioxide (ZrO2), and composite resins, which have been shown to reduce ion release and bacterial adhesion [197,198,199].
Table 2 and Figure 5 provide an overview of the common pathogens isolated from all three discussed reservoirs, along with their reported AMR profiles.

Table 2.
The most common pathogens found in DUWLs, dentures, and orthodontic appliances, and their resistance patterns.
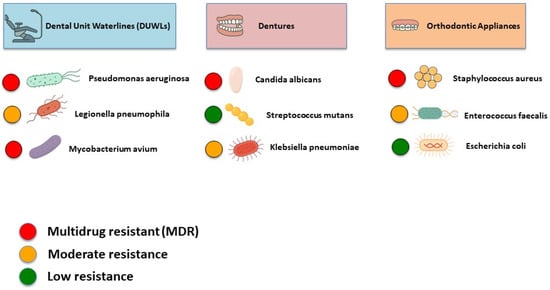
Figure 5.
AMR in key dental reservoirs: DUWLs, dentures, and orthodontic appliances.
5. Recommendations for AMS Implementation in Dentistry
Effective employment of AMS in dentistry requires a broad and context-specific approach that addresses both clinical practice and behavioral change. Recent evidence emphasizes raising awareness among dental teams and patients about the risk of unnecessary antibiotic use [20]. Antimicrobials should be prescribed in dentistry only when clinically indicated [200]. Therapeutic antibiotics must be reserved for bacterial infections with systemic involvement (such as fever, malaise, lymphadenitis, or rapidly spreading cellulitis) and avoided for localized infections manageable with operative measures alone [13,201,202]. Prophylactic use should be restricted to high-risk patients, such as those with specific cardiac conditions predisposing to infective endocarditis or certain immunocompromised states [203,204]. Prescribers should choose the narrowest effective spectrum, the shortest effective course, and appropriate dosing, and provide clear instructions to minimize unnecessary exposure and reduce AMR [205].
Integrating AMS principles into undergraduate and continuing education and promoting practices with a culture of stewardship should also be considered [20,206,207]. Distributing guidelines alone rarely leads to meaningful changes in prescribing habits. Long-lasting progress is more likely when these guidelines are combined with regular audits, constructive feedback, and targeted educational initiatives for dental professionals [208]. Collaboration with pharmacists, infectious disease specialists, and other healthcare professionals can strengthen AMS strategies, and involving educators from outside dentistry during undergraduate training has been shown to broaden perspectives and promote interprofessional teamwork [207]. To mitigate the need for antibiotics as a substitute for treatment, it is crucial to ensure that patients have access to timely definitive dental care and to adjust AMS interventions to local prescribing patterns, resources, and cultural factors [209]. Embedding AMS resources, such as prescribing recommendations, into dental software, and using patient education tools (posters, videos, and leaflets) can further reinforce best practice [206].
Novel clinical approaches can minimize the use of unnecessary antibiotics in dentistry. For example, the use of platelet-rich fibrin (PRF) in oral surgery has been shown to enhance soft tissue healing, reduce postoperative complications, and lower infection rates, which could decrease the reliance on prophylactic or therapeutic antibiotics. Integrating such adjunctive techniques into routine practice may support AMS goals by addressing infection prevention at the procedural level [210].
Finally, effective AMS in dentistry should be embedded within broader public health strategies that aim to reduce the spread of AMR. Preventive measures, such as maintaining good oral hygiene, ensuring timely access to dental care, and enforcing rigorous infection prevention protocols, reduce the need for antibiotics and protect their long-term efficacy. By integrating public health initiatives with evidence-based prescribing, dental professionals can help prevent the emergence and transmission of resistant pathogens and contribute to global efforts to improve antibiotic effectiveness.
6. Conclusions
In conclusion, a wide range of resistant microorganisms have been detected in DUWLs, as well as on the surfaces of dentures and orthodontic appliances. Many of these pathogens are multi-drug resistant and can persist in biofilms for extended periods, increasing the risk of transmission in dental settings. Although resistance genes have been identified within these biofilm-associated communities, our understanding of how these genes are exchanged, through mechanisms such as transformation, transduction, and conjugation, remains limited. Future research should focus on elucidating these genetic transfer processes and identifying the environmental and clinical conditions that facilitate gene exchange.
Longitudinal and in situ studies are also essential to monitor resistance development over time under real-world conditions. Given the routine exposure of both patients and dental professionals to aerosolized particles, particularly during procedures involving DUWLs and contaminated appliances, the presence of resistant pathogens poses a serious clinical and public health concern. To address this, more stringent infection control protocols and AMS programs specifically designed for dental environments must be developed and consistently implemented. Standardizing AMR surveillance in dentistry, improving patient education, and integrating AMR-focused content into dental curricula and continuing professional development are also critical to reducing the spread of resistance and safeguarding both oral and systemic health.
Author Contributions
M.F.S. conceived the review topic. G.F. and Y.A. conducted the literature search and data extraction and prepared the original manuscript draft. M.F.S. supervised the work, critically revised the content, and contributed to reviewing and editing the final manuscript. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Prestinaci, F.; Pezzotti, P.; Pantosti, A. Antimicrobial resistance: A global multifaceted phenomenon. Pathog. Glob. Health 2015, 109, 309–318. [Google Scholar] [CrossRef]
- Fernandez, G. Turning the juggernaut. Lancet Planet Health 2022, 6, E75. [Google Scholar]
- Murray, C.J.L.; Ikuta, K.S.; Sharara, F.; Swetschinski, L.; Aguilar, G.R.; Gray, A.; Han, C.; Bisignano, C.; Rao, P.; Wool, E.; et al. Global burden of bacterial antimicrobial resistance in 2019: A systematic analysis. Lancet 2022, 399, 629–655. [Google Scholar] [CrossRef]
- O’Neill, J. Tackling Drug-Resistant Infections Globally: Final Report and Recommendations; Wellcome Trust: London, UK, 2016. [Google Scholar]
- O’Neill, J. Antimicrobial Resistance: Tackling a Crisis for the Health and Wealth of Nations; Review on Antimicrobial Resistance: London, UK, 2014. [Google Scholar]
- Centers for Disease Control and Prevention. Antibiotic Resistance Threats in the United States, 2019; US Department of Health and Human Services, CDC: Atlanta, GA, USA, 2019.
- WHO. Antimicrobial Resistance and the United Nations Sustainable Development Cooperation Framework; World Health Organization: Geneva, Switzerland, 2021. [Google Scholar]
- World Health Organization. Global Action Plan on Antimicrobial Resistance; World Health Organization: Geneva, Switzerland, 2015. [Google Scholar]
- Oberoi, S.S.; Dhingra, C.; Sharma, G.; Sardana, D. Antibiotics in dental practice: How justified are we. Int. Dent. J. 2020, 65, 4–10. [Google Scholar] [CrossRef] [PubMed]
- Thompson, W.; Williams, D.; Pulcini, C.; Sanderson, S.; Calfon, P.; Verma, M. Tackling antibiotic resistance: Why dentistry matters. Int. Dent. J. 2021, 71, 450–453. [Google Scholar] [CrossRef] [PubMed]
- Barone, A.; Chatelain, S.; Derchi, G.; Di Spirito, F.; Martuscelli, R.; Porzio, M.; Sbordone, L. Antibiotic’s effectiveness after erupted tooth extractions: A retrospective study. Oral. Dis. 2020, 26, 967–973. [Google Scholar] [CrossRef]
- Contaldo, M.; D’ambrosio, F.; Ferraro, G.A.; Di Stasio, D.; Di Palo, M.P.; Serpico, R.; Simeone, M. Antibiotics in dentistry: A narrative review of the evidence beyond the myth. Int. J. Environ. Res. Public Health 2023, 20, 6025. [Google Scholar] [CrossRef]
- Gill, A.S.; Morrissey, H.; Rahman, A. A systematic review and meta-analysis evaluating antibiotic prophylaxis in dental implants and extraction procedures. Medicina 2018, 54, 95. [Google Scholar] [CrossRef] [PubMed]
- Cuevas-Gonzalez, M.V.; Mungarro-Cornejo, G.A.D.; Espinosa-Cristóbal, L.F.; Donohue-Cornejo, A.; Carrillo, K.L.T.; Acuña, R.A.S.; Calderón, A.G.M.G.; Gastelum, D.A.G.; Zambrano-Galván, G.; Cuevas-Gonzalez, J.C. Antimicrobial resistance in odontogenic infections: A protocol for systematic review. Medicine 2022, 101, e31345. [Google Scholar] [CrossRef] [PubMed]
- Ardila, C.M.; Bedoya-García, J.A. Antimicrobial resistance in patients with odontogenic infections: A systematic scoping review of prospective and experimental studies. J. Clin. Exp. Dent. 2022, 14, e834. [Google Scholar] [CrossRef] [PubMed]
- Abe, F.C.; Kodaira, K.; Motta, C.d.C.B.; Barberato-Filho, S.; Silva, M.T.; Guimarães, C.C.; Martins, C.C.; Lopes, L.C. Antimicrobial resistance of microorganisms present in periodontal diseases: A systematic review and meta-analysis. Front. Microbiol. 2022, 13, 961986. [Google Scholar] [CrossRef] [PubMed]
- Dahlen, G. Biofilms in Dental Unit Water Lines. Monogr. Oral Sci. 2020, 29, 12–18. [Google Scholar]
- O’Donnell, L.E.; Smith, K.; Williams, C.; Nile, C.J.; Lappin, D.F.; Bradshaw, D.; Lambert, M.; Robertson, D.P.; Bagg, J.; Hannah, V.; et al. Dentures are a reservoir for respiratory pathogens. J. Prosthodont. 2015, 25, 99–104. [Google Scholar] [CrossRef]
- Müller, L.K.; Jungbauer, G.; Jungbauer, R.; Wolf, M.; Deschner, J. Biofilm and Orthodontic Therapy. Monogr. Oral Sci. 2021, 29, 201–213. [Google Scholar] [PubMed]
- Teoh, L.; Löffler, C.; Mun, M.; Agnihotry, A.; Kaur, H.; Born, K.; Thompson, W. A Systematic Review of Dental Antibiotic Stewardship Interventions. Community Dent. Oral Epidemiol. 2024, 53, 245–255. [Google Scholar] [CrossRef] [PubMed]
- Cox, G.; Wright, G.D. Intrinsic antibiotic resistance: Mechanisms, origins, challenges and solutions. Int. J. Med. Microbiol. 2013, 303, 287–292. [Google Scholar] [CrossRef] [PubMed]
- Martinez, J.L. General principles of antibiotic resistance in bacteria. Drug Discov. Today Technol. 2014, 11, 33–39. [Google Scholar] [CrossRef]
- Al-Haroni, M. Bacterial resistance and the dental professionals’ role to halt the problem. J. Dent. 2008, 36, 95–103. [Google Scholar] [CrossRef] [PubMed]
- Munita, J.M.; Arias, C.A. Mechanisms of antibiotic resistance. Microbiol. Spectr. 2016, 4, 481–511. [Google Scholar] [CrossRef] [PubMed]
- Burmeister, A.R. Horizontal gene transfer. Evol. Med. Public Health 2015, 2015, 193–194. [Google Scholar] [CrossRef]
- Michaelis, C.; Grohmann, E. Horizontal gene transfer of antibiotic resistance genes in biofilms. Antibiotics 2023, 12, 328. [Google Scholar] [CrossRef]
- Gaurav, A.; Bakht, P.; Saini, M.; Pandey, S.; Pathania, R. Role of bacterial efflux pumps in antibiotic resistance, virulence, and strategies to discover novel efflux pump inhibitors. Microbiology 2023, 169, 001333. [Google Scholar] [CrossRef] [PubMed]
- Jang, S. AcrAB-TolC, a major efflux pump in Gram negative bacteria: Toward understanding its operation mechanism. BMB Rep. 2023, 56, 326. [Google Scholar] [CrossRef]
- Bush, K.; Bradford, P.A. β-Lactams and β-lactamase inhibitors: An overview. Cold Spring Harb. Perspect. Med. 2016, 6, a025247. [Google Scholar] [CrossRef] [PubMed]
- Delcour, A.H. Outer membrane permeability and antibiotic resistance. Biochim. Biophys. Acta 2009, 1794, 808–816. [Google Scholar] [CrossRef] [PubMed]
- Egorov, A.; Ulyashova, M.; Rubtsova, M. Bacterial enzymes and antibiotic resistance. Acta Nat. 2018, 10, 33–48. [Google Scholar] [CrossRef]
- Pletz, M.W.R.; McGee, L.; Beall, B.; Whitney, C.G.; Klugman, K.P. Interspecies recombination in type II topoisomerase genes is not a major cause of fluoroquinolone resistance in invasive Streptococcus pneumoniae isolates in the United States. Antimicrob. Agents Chemother. 2005, 49, 779–780. [Google Scholar] [CrossRef] [PubMed]
- Reygaert, W.C. An overview of the antimicrobial resistance mechanisms of bacteria. AIMS Microbiol. 2018, 4, 482. [Google Scholar] [CrossRef]
- Hossain, C.M.; Ryan, L.K.; Gera, M.; Choudhuri, S.; Lyle, N.; Ali, K.A.; Diamond, G. Antifungals and drug resistance. Encyclopedia 2022, 2, 1722–1737. [Google Scholar] [CrossRef]
- Vere Hodge, A.; Field, H.J. General mechanisms of antiviral resistance. Genet. Evol. Infect. Dis. 2010, 24, 339–362. [Google Scholar]
- Pallasch, T.J. Global antibiotic resistance and its impact on the dental community. J. Calif. Dent. Assoc. 2000, 28, 215–231. [Google Scholar] [CrossRef]
- Anderson, A.C.; von Ohle, C.; Frese, C.; Boutin, S.; Bridson, C.; Schoilew, K.; Peikert, S.A.; Hellwig, E.; Pelz, K.; Wittmer, A.; et al. The oral microbiota is a reservoir for antimicrobial resistance: Resistome and phenotypic resistance characteristics of oral biofilm in health, caries, and periodontitis. Ann. Clin. Microbiol. Antimicrob. 2023, 22, 37. [Google Scholar] [CrossRef] [PubMed]
- Khalil, D.; Hultin, M.; Rashid, M.; Lund, B. Oral microflora and selection of resistance after a single dose of amoxicillin. Clin. Microbiol. Infect. 2016, 22, 949.e1–949.e4. [Google Scholar] [CrossRef] [PubMed]
- Masuda, K.; Nemoto, H.; Nakano, K.; Naka, S.; Nomura, R.; Ooshima, T. Amoxicillin-resistant oral streptococci identified in dental plaque specimens from healthy Japanese adults. J. Cardiol. 2012, 59, 285–290. [Google Scholar] [CrossRef] [PubMed]
- Dwivedi, D.; Kushwah, T.; Kushwah, M.; Singh, V. Antibiotic susceptibility pattern against pathogenic bacteria causing Dental Caries. S. Asian J. Exp. Biol. 2011, 1, 31–35. [Google Scholar] [CrossRef]
- Kuriyama, T.; Williams, D.W.; Yanagisawa, M.; Iwahara, K.; Shimizu, C.; Nakagawa, K.; Yamamoto, E.; Karasawa, T. Antimicrobial susceptibility of 800 anaerobic isolates from patients with dentoalveolar infection to 13 oral antibiotics. Oral Microbiol. Immunol. 2007, 22, 285–288. [Google Scholar] [CrossRef]
- Medina-Palacios, S.E.; Vitales-Noyola, M.; López-González, E.; González-Amaro, A.M.; Méndez-González, V.; Pozos-Guillén, A. Root canal microorganisms and their antibiotic susceptibility in patients with persistent endodontic infections, with and without clinical symptoms. Odontology 2021, 109, 596–604. [Google Scholar] [CrossRef] [PubMed]
- Smith, R.; Coast, J. The true cost of antimicrobial resistance. BMJ 2013, 346, f1493. [Google Scholar] [CrossRef]
- Kang, Y.; Sun, B.; Chen, Y.; Lou, Y.; Zheng, M.; Li, Z.; Fey, P.D. Dental plaque microbial resistomes of periodontal health and disease and their changes after scaling and root planing therapy. mSphere 2021, 6, e0016221. [Google Scholar] [CrossRef] [PubMed]
- Morales-Dorantes, V.; Domínguez-Pérez, R.A.; Pérez-Serrano, R.M.; Solís-Sainz, J.C.; García-Solís, P.; Espinosa-Cristóbal, L.F.; Cabeza-Cabrera, C.V.; Ayala-Herrera, J.L. The distribution of eight antimicrobial resistance genes in Streptococcus oralis, Streptococcus sanguinis, and Streptococcus gordonii strains isolated from dental plaque as oral commensals. Trop. Med. Infect. Dis. 2023, 8, 499. [Google Scholar] [CrossRef]
- Ardila, C.M.; Bedoya-García, J.A. Antimicrobial resistance of Aggregatibacter actinomycetemcomitans, Porphyromonas gingivalis and Tannerella forsythia in periodontitis patients. J. Glob. Antimicrob. Resist. 2020, 22, 215–218. [Google Scholar] [CrossRef] [PubMed]
- Tesauro, M.; Consonni, M.; Grappasonni, I.; Lodi, G.; Mattina, R. Dental unit water content and antibiotic resistance of Pseudomonas aeruginosa and Pseudomonas species: A case study. J. Oral Microbiol. 2022, 14, 2107316. [Google Scholar] [CrossRef] [PubMed]
- Walker, J.T.; Marsh, P.D. Microbial biofilm formation in DUWS and their control using disinfectants. J. Dent. 2007, 35, 721–730. [Google Scholar] [CrossRef] [PubMed]
- Cicciù, M. Water contamination risks at the dental clinic. Biology 2020, 9, 43. [Google Scholar] [CrossRef] [PubMed]
- Spagnolo, A.M.; Sartini, M.; Cristina, M.L. Microbial contamination of dental unit waterlines and potential risk of infection: A narrative review. Pathogens 2020, 9, 651. [Google Scholar] [CrossRef] [PubMed]
- Bayani, M.; Raisolvaezin, K.; Almasi-Hashiani, A.; Mirhoseini, S.H. Bacterial biofilm prevalence in dental unit waterlines: A systematic review and meta-analysis. BMC Oral Health 2023, 23, 158. [Google Scholar] [CrossRef]
- Singh, J.; O’Donnell, K.; Nieves, D.J.; Adler-Shohet, F.C.; Arrieta, A.C.; Ashouri, N.; Ahuja, G.; Cheung, M.; Holmes, W.N.; Huoh, K.; et al. Invasive Mycobacterium abscessus outbreak at a pediatric dental clinic. Open Forum Infect. Dis. 2021, 8, ofab165. [Google Scholar] [CrossRef]
- Pérez-Alfonzo, R.; Brito, L.E.P.; Vergara, M.S.; Damasco, A.R.; Rodríguez, P.L.M.; Quintero, C.E.K.; Martinez, C.C.; Rivera-Oliver, I.A.; Jardin, O.J.D.M.; Rodríguez-Castillo, B.A.; et al. Odontogenic cutaneous sinus tracts due to infection with nontuberculous mycobacteria: A report of three cases. BMC Infect. Dis. 2020, 20, 295. [Google Scholar] [CrossRef] [PubMed]
- Gawish, S.; Abbass, A.; Abaza, A. Occurrence and biofilm forming ability of Pseudomonas aeruginosa in the water output of dental unit waterlines in a dental center in Alexandria, Egypt. Germs 2019, 9, 71–80. [Google Scholar] [CrossRef]
- Mah, T.F. Biofilm-specific antibiotic resistance. Future Microbiol. 2012, 7, 1061–1072. [Google Scholar] [CrossRef]
- Rath, S.; Bal, S.C.B.; Dubey, D. Oral biofilm: Development mechanism, multidrug resistance, and their effective management with novel techniques. Rambam Maimonides Med. J. 2021, 12, e0004. [Google Scholar] [CrossRef] [PubMed]
- Marcinkiewicz, J.; Strus, M.; Pasich, E. Antibiotic resistance: A “dark side” of biofilm-associated chronic infections. Pol. Arch. Med. Wewn. 2013, 123, 309–313. [Google Scholar] [CrossRef]
- Sharma, S.; Mohler, J.; Mahajan, S.D.; Schwartz, S.A.; Bruggemann, L.; Aalinkeel, R. Microbial biofilm: A review on formation, infection, antibiotic resistance, control measures, and innovative treatment. Microorganisms 2023, 11, 1614. [Google Scholar] [CrossRef]
- Soto, S.M. Role of efflux pumps in the antibiotic resistance of bacteria embedded in a biofilm. Virulence 2013, 4, 223–229. [Google Scholar] [CrossRef] [PubMed]
- Van Acker, H.; Van Dijck, P.; Coenye, T. Molecular mechanisms of antimicrobial tolerance and resistance in bacterial and fungal biofilms. Trends Microbiol. 2014, 22, 326–333. [Google Scholar] [CrossRef] [PubMed]
- Barbot, V.; Robert, A.; Rodier, M.-H.; Imbert, C. Update on infectious risks associated with dental unit waterlines. FEMS Immunol. Med. Microbiol. 2012, 65, 196–204. [Google Scholar] [CrossRef]
- Dowdell, K.; Haig, S.-J.; Caverly, L.J.; Shen, Y.; LiPuma, J.J.; Raskin, L. Nontuberculous mycobacteria in drinking water systems—The challenges of characterization and risk mitigation. Curr. Opin. Biotechnol. 2019, 57, 127–136. [Google Scholar] [CrossRef] [PubMed]
- Mtetwa, H.N.; Amoah, I.D.; Kumari, S.; Bux, F.; Reddy, P. The source and fate of Mycobacterium tuberculosis complex in wastewater and possible routes of transmission. BMC Public Health 2022, 22, 145. [Google Scholar] [CrossRef]
- Alkhulaifi, M.M.; Alotaibi, D.H.; Alajlan, H.; Binshoail, T. Assessment of nosocomial bacterial contamination in dental unit waterlines: Impact of flushing. Saudi Dent. J. 2020, 32, 68–73. [Google Scholar] [CrossRef]
- Szymańska, J.; Sitkowska, J.; Dutkiewicz, J. Microbial contamination of dental unit waterlines. Ann. Agric. Environ. Med. 2008, 15, 173–179. [Google Scholar] [PubMed]
- Li, B.; Hu, Y.; Wang, Y.; Zhang, C.; Wang, Z.; Peng, X.; Feng, J. Periodic detection and disinfection maintenance of dental unit waterlines in dental simulation head model laboratories. Sci. Rep. 2025, 15, 5234. [Google Scholar] [CrossRef]
- Atlas, R.M.; Williams, J.F.; Huntington, M.K. Legionella contamination of dental-unit waters. Appl. Environ. Microbiol. 1995, 61, 1208–1213. [Google Scholar] [CrossRef] [PubMed]
- Rudbeck, M.; Viskum, S.; Mølbak, K.; Uldum, S.A. Legionella antibodies in a Danish hospital staff with known occupational exposure. J. Environ. Public Health 2009, 2009, 812829. [Google Scholar] [CrossRef]
- Kevorkyan, A.; Tomova, I.; Raycheva, R.; Stoeva, V.; Stoilova, Y.; Lalabonova, H.; Kondeva, V. Legionella pneumophila antibodies in serum samples from medical and dental personnel: A seroepidemiological survey. Biotechnol. Biotechnol. Equip. 2017, 31, 588–593. [Google Scholar] [CrossRef][Green Version]
- Schönning, C.; Jernberg, C.; Klingenberg, D.; Andersson, S.; Pääjärvi, A.; Alm, E.; Tano, E.; Lytsy, B. Legionellosis acquired through a dental unit: A case study. J. Hosp. Infect. 2017, 96, 89–92. [Google Scholar] [CrossRef]
- Ricci, M.L.; Fontana, S.; Pinci, F.; Fiumana, E.; Pedna, M.F.; Farolfi, P.; Sabattini, M.A.B.; Scaturro, M. Pneumonia associated with a dental unit waterline. Lancet 2012, 379, 684. [Google Scholar] [CrossRef] [PubMed]
- Santajit, S.; Indrawattana, N. Mechanisms of antimicrobial resistance in ESKAPE pathogens. BioMed Res. Int. 2016, 2016, 2475067. [Google Scholar] [CrossRef] [PubMed]
- Desai, A.N.; Hurtado, R.M. Infections and outbreaks of nontuberculous mycobacteria in hospital settings. Curr. Treat. Options Infect. Dis. 2018, 10, 169–181. [Google Scholar] [CrossRef] [PubMed]
- Sachan, R.S.K.; Mistry, V.; Dholaria, M.; Rana, A.; Devgon, I.; Ali, I.; Iqbal, J.; Eldin, S.M.; Al-Tawaha, A.R.M.S.; Bawazeer, S.; et al. Overcoming Mycobacterium tuberculosis drug resistance: Novel medications and repositioning strategies. ACS Omega 2023, 8, 32244–32257. [Google Scholar] [CrossRef]
- Vosooghi, K.; Larypoor, M.; Sakhaee, F.; Jajin, M.G.; Moghaddam, S.; Samieefar, N.; Keramat, R.R.; Sabotki, M.A.; Fateh, A. Distribution of nontuberculous mycobacteria in dental unit waterlines: A potential health hazard in the dental office. Microb. Pathog. 2024, 196, 106963. [Google Scholar] [CrossRef] [PubMed]
- Vincent, A.T.; Freschi, L.; Jeukens, J.; Kukavica-Ibrulj, I.; Emond-Rheault, J.-G.; Leduc, A.; Boyle, B.; Jean-Pierre, F.; Groleau, M.-C.; Déziel, E.; et al. Genomic characterisation of environmental Pseudomonas aeruginosa isolated from dental unit waterlines revealed the insertion sequence IS Pa11 as a chaotropic element. FEMS Microbiol. Ecol. 2017, 93, fix106. [Google Scholar]
- Alsehlawi, Z.S.; Al-Yasiri, I.K.; Fakhriddeen, A.J.; Taher, A.A.Y. Antibiotic susceptibility patterns of legionella pneumophila isolated from water lines of dental settings. Smile Dent. J. 2016, 11, 36–39. [Google Scholar] [CrossRef]
- American Dental Association. ADA statement on dental unit waterlines. J. Am. Dent. Assoc. 1996, 127, 185–186. [Google Scholar] [CrossRef]
- Güngör, N.D.; Kadaifçiler, D.G.; Peker, O.Ö. Investigation of the bacterial load and antibiotic susceptibility of dental units. Environ. Monit. Assess. 2013, 186, 1847–1853. [Google Scholar] [CrossRef] [PubMed]
- Uzel, A.; Cogulu, D.; Oncag, O. Microbiological evaluation and antibiotic susceptibility of dental unit water systems in general dental practice. Int. J. Dent. Hyg. 2008, 6, 43–47. [Google Scholar] [CrossRef] [PubMed]
- Lancellotti, M.; Oliveira, M.P.; Ávila, F.A. Research on Staphylococcus spp. in biofilm formation in water pipes and sensibility to antibiotics. Braz. J. Oral Sci. 2007, 6, 20. [Google Scholar]
- Buitrago, J.M.; Kolbe, R.J.; Siqueira, M.F. Dental unit waterline testing practices: An 11-Year retrospective study. BMC Oral Health 2023, 23, 867. [Google Scholar] [CrossRef]
- Kohn, W.G.; Collins, A.S.; Cleveland, J.L.; Harte, J.A.; Eklund, K.J.; Malvitz, D.M. Guidelines for infection control in dental health-care settings—2003. In MMWR Recommendations and Reports; Centers for Disease Control and Prevention (CDC): Atlanta, GA, USA, 2003; 52, pp. 1–61. [Google Scholar]
- Bowen, C.G.; Greenwood, W.; Guevara, P.; Washington, M.A. Effectiveness of a dental unit waterline treatment protocol with A-Dec ICX and Citrisil Disinfectants. Mil. Med. 2015, 180, 1098–1104. [Google Scholar] [CrossRef]
- O’Donnell, M.J.; Boyle, M.A.; Russell, R.J.; Coleman, D.C. Management of dental unit waterline biofilms in the 21st century. Future Microbiol. 2011, 6, 1209–1226. [Google Scholar] [CrossRef]
- Baudet, A.; Lizon, J.; Martrette, J.M.; Camelot, F.; Florentin, A.; Clément, C. Efficacy of BRS® and Alpron®/Bilpron® disinfectants for dental unit waterlines: A six-year study. Int. J. Environ. Res. Public Health 2020, 17, 2634. [Google Scholar] [CrossRef]
- Noopan, S.; Unchui, P.; Techotinnakorn, S.; Ampornaramveth, R.S. Plasma sterilization effectively reduces bacterial contamination in dental unit waterlines. Int. J. Dent. 2019, 2019, 5720204. [Google Scholar] [CrossRef] [PubMed]
- Okubo, K.; Ito, T.; Shiota, Y.; Kawata, Y.; Yamamoto, T.; Takashiba, S. Effectiveness and safety of low-concentrated ozonized water for the reduction of contamination in dental unit water lines. Heliyon 2019, 5, e02306. [Google Scholar] [CrossRef] [PubMed]
- Centers for Disease Control and Prevention (CDC). Best Practices for Dental Unit Water Quality. 2024. Available online: https://www.cdc.gov/dental-infection-control/hcp/dental-ipc-faqs/best-practices-dental-unit-water-quality.html (accessed on 15 May 2024).
- College of Dental Surgeons of Saskatchewan. Infection Prevention and Control Standards in the Oral Health Care Facility. Available online: https://saskdentists.com/wp-content/uploads/2023/04/Infection-Prevention-and-Control-Standard.pdf (accessed on 22 June 2025).
- Ji, X.Y.; Fei, C.N.; Zhang, Y.; Zhang, W.; Liu, J.; Dong, J. Evaluation of bacterial contamination of dental unit waterlines and use of a newly designed measurement device to assess retraction of a dental chair unit. Int. Dent. J. 2020, 66, 208–214. [Google Scholar] [CrossRef]
- World Health Organization. Water Safety Plan Manual: Step-by-Step Risk Management for Drinking-Water Suppliers; World Health Organization: Geneva, Switzerland, 2023. [Google Scholar]
- Directive, C. On the quality of water intended for human consumption. In Proceedings of the Commission of The European Communities, Brussels, Belgium, 8 July 1998. [Google Scholar]
- Directive (EU) 2020/2184 of the European Parliament and of the Council of 16 December 2020 on the Quality of Water Intended for Human Consumption; EUR-Lex: Luxembourg, 2020; Volume 435, pp. 1–62.
- Tuvo, B.; Totaro, M.; Cristina, M.L.; Spagnolo, A.M.; Di Cave, D.; Profeti, S.; Baggiani, A.; Privitera, G.; Casini, B. Prevention and control of Legionella and Pseudomonas spp. colonization in dental units. Pathogens 2020, 9, 305. [Google Scholar] [CrossRef]
- Vaidya, R.Y.; Hegde, V.; Sherigar, P.; Prabhu, N. Exploring microbial interactions with denture resin surface and implications for plant based plaque control strategies: A narrative review. Beni-Suef Univ. J. Basic Appl. Sci. 2024, 13, 110. [Google Scholar] [CrossRef]
- de Foggi, C.C.; Machado, A.L.; Zamperini, C.A.; Fernandes, D.; Wady, A.F.; Vergani, C.E. Effect of surface roughness on the hydrophobicity of a denture-base acrylic resin and Candida albicans colonization. J. Investig. Clin. Dent. 2016, 7, 141–148. [Google Scholar] [CrossRef] [PubMed]
- Suryawanshi, H.; Hande, A.; Dasari, A.K.; Aileni, K.R.; AlZoubi, I.; Patil, S.R. Metal ion release from orthodontic appliances: Concerns regarding potential carcinogenic effects. Oral Oncol. Rep. 2024, 10, 100309. [Google Scholar] [CrossRef]
- Jeon, D.-M.; An, J.-S.; Lim, B.-S.; Ahn, S.-J. Orthodontic bonding procedures significantly influence biofilm composition. Prog. Orthod. 2020, 21, 14. [Google Scholar] [CrossRef] [PubMed]
- Khawwam, S.I.; Al-Groosh, D.H. Effect of Different Cleaning Regimes on Biofilm Formation of Acrylic-Based Removable Orthodontic Appliance: A Randomized Clinical Trial. Sci. World J. 2023, 2023, 9920850. [Google Scholar] [CrossRef] [PubMed]
- Abbas, M.H.; Al-Yasseen, A.K.; Alhamadi, W.W. Prevalence of Staphylococcus aureus among gingivitis in patient with orthodontic wires in Kufa City/Iraq. Biotechnology 2017, 14, 91–96. [Google Scholar]
- McReynolds, D.E.; Moorthy, A.; Moneley, J.O.; Jabra-Rizk, M.A.; Sultan, A.S. Denture stomatitis—An interdisciplinary clinical review. J. Prosthodont. 2023, 32, 560–570. [Google Scholar] [CrossRef] [PubMed]
- Mayer, C.; Moritz, R.; Kirschner, C.; Borchard, W.; Maibaum, R.; Wingender, J.; Flemming, H.-C. The role of intermolecular interactions: Studies on model systems for bacterial biofilms. Int. J. Biol. Macromol. 1999, 26, 3–16. [Google Scholar] [CrossRef]
- Katsikogianni, M.; Missirlis, Y.F. Concise review of mechanisms of bacterial adhesion to biomaterials and of techniques used in estimating bacteria-material interactions. Eur. Cell Mater. 2004, 8, 37–57. [Google Scholar] [CrossRef] [PubMed]
- Sterzenbach, T.; Helbig, R.; Hannig, C.; Hannig, M. Bioadhesion in the oral cavity and approaches for biofilm management by surface modifications. Clin. Oral Investig. 2020, 24, 4237–4260. [Google Scholar] [CrossRef]
- Ten Cate, J.M. Biofilms, a new approach to the microbiology of dental plaque. Odontology 2006, 94, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Morse, D.J.; Wilson, M.J.; Wei, X.; Lewis, M.A.O.; Bradshaw, D.J.; Murdoch, C.; Williams, D.W. Denture-associated biofilm infection in three-dimensional oral mucosal tissue models. J. Med. Microbiol. 2018, 67, 364–375. [Google Scholar] [CrossRef] [PubMed]
- Sultan, A.S.; Rizk, A.M.; Vila, T.; Ji, Y.; Masri, R.; Jabra-Rizk, M.A. Digital design of a universal rat intraoral device for therapeutic evaluation of a topical formulation against Candida-associated denture stomatitis. Infect. Immun. 2019, 87, e00617-19. [Google Scholar] [CrossRef]
- Yoshizaki, T.; Akiba, N.; Inokoshi, M.; Shimada, M.; Minakuchi, S. Hydrophilic nano-silica coating agents with platinum and diamond nanoparticles for denture base materials. Dent. Mater. J. 2017, 36, 333–339. [Google Scholar] [CrossRef] [PubMed]
- Oliveira Junior, N.M.D.; Mendoza Marin, D.O.; Leite, A.R.P.; Pero, A.C.; Klein, M.I.; Compagnoni, M.A. Influence of the use of complete denture adhesives on microbial adhesion and biofilm formation by single-and mixed-species. PLoS ONE 2018, 13, e0203951. [Google Scholar] [CrossRef]
- Buranarom, N.; Komin, O.; Matangkasombut, O. Hyposalivation, oral health, and Candida colonization in independent dentate elders. PLoS ONE 2020, 15, e0242832. [Google Scholar] [CrossRef]
- Gad, M.M.; Al-Thobity, A.M.; Shahin, S.Y.; Alsaqer, B.T.; Ali, A. Inhibitory effect of zirconium oxide nanoparticles on Candida albicans adhesion to repaired polymethyl methacrylate denture bases and interim removable prostheses: A new approach for denture stomatitis prevention. Int. J. Nanomed. 2017, 12, 5409–5419. [Google Scholar] [CrossRef]
- Naji, S.A.; Kashi, T.S.J.; Pourhajibagher, M.; Behroozibakhsh, M.; Masaeli, R.; Bahador, A. Evaluation of antimicrobial properties of conventional poly(methyl methacrylate) denture base resin materials containing hydrothermally synthesised anatase TiO2 nanotubes against cariogenic bacteria and Candida albicans. Iran. J. Pharm. Res. 2018, 17, 161–172. [Google Scholar]
- Majchrzak, K.; Mierzwinska-Nastalska, E.; Chmura, A.; Kwiatkowski, A.; Paczek, L.; Mlynarczyk, G.; Szymanek-Majchrzak, K. Comparison of staphylococcal flora in denture plaque and the surface of the pharyngeal mucous membrane in kidney transplant recipients. Transplant. Proc. 2016, 48, 1590–1597. [Google Scholar] [CrossRef] [PubMed]
- Nett, J.E.; Andes, D.R. Fungal Biofilms: In vivo models for discovery of anti-biofilm drugs. Microb. Biofilms 2015, 3, 33–49. [Google Scholar] [CrossRef] [PubMed]
- Larijani, M.; Zareshahrabadi, Z.; Alhavaz, A.; Hajipour, R.; Ranjbaran, A.; Giti, R.; Soltankarimi, V.; Zomorodian, K. Evaluation of Candida albicans biofilm formation on conventional and computer-aided-design/computer-aided manufacturing (CAD/CAM) denture base materials. Curr. Med. Mycol. 2022, 8, 23–29. [Google Scholar] [CrossRef] [PubMed]
- VV, N.; Karibasappa, G.; Dodamani, A.; Prashanth, V.K. Microbial contamination of removable dental prosthesis at different interval of usage: An: In vitro: Study. J. Indian Prosthodont. Soc. 2016, 16, 346–351. [Google Scholar]
- Arbelaez, M.I.A.; Vergani, C.E.; Barbugli, P.A.; Pavarina, A.C.; Sanita, P.V.; Jorge, J.H. Long-term effect of daily chemical disinfection on surface topography and Candida albicans biofilm formation on denture base and reline acrylic resins. Oral Health Prev. Dent. 2020, 18, a45521. [Google Scholar]
- Rossi, T.; Peltonen, R.; Laine, J.; Eerola, E.; Vuopio-Varkila, J.; Kotilainen, P. Eradication of the long-term carriage of methicillin-resistant Staphylococcus aureus in patients wearing dentures: A follow-up of 10 patients. J. Hosp. Infect. 1996, 34, 311–320. [Google Scholar] [CrossRef]
- Andonissamy, L.; Karthigeyan, S.; Ali, S.A. Detection of Drug Susceptible and Resistant Viridans Streptococci sp. Staphylococcus aureus, Klebsiella pneumoniae and E. coli in Complete Denture Patients and Visualisation using Scanning Electron Microscopy. J. Clin. Diagn. Res. 2020, 14, 15. [Google Scholar]
- Gaetti-Jardim, E.C.; Marqueti, A.C.; Faverani, L.P.; Júnior, E.G.-J. Antimicrobial resistance of aerobes and facultative anaerobes isolated from the oral cavity. J. Appl. Oral Sci. 2010, 18, 551–559. [Google Scholar] [CrossRef]
- Ramos, M.M.B.; Gartti-Jardim, E.C.; Junior, E.G.-J. Resistance to tetracycline and β-lactams and distribution of resistance markers in enteric microorganisms and pseudomonads isolated from the oral cavity. J. Appl. Oral Sci. 2009, 17, 13–18. [Google Scholar] [CrossRef]
- Abu-Elteen, K.H. Candida albicans strain differentiation in complete denture wearers. New Microbiol. 2000, 23, 329–337. [Google Scholar]
- Martin-Mazuelos, E.; Aller, A.I.; Romero, M.J.; Armijo, A.R.; Gutierrez, M.J.; Bernal, S.; Montero, O. Response to fluconazole and itraconazole of Candida spp. in denture stomatitis. Mycoses 1997, 40, 283–289. [Google Scholar] [CrossRef]
- Singh, T.; Bhojaraju, N.; Vinod, V.; Anjali, K.; Shah, S.; Kumar, N. Evaluation of microbial contamination in removable dental prosthesis at different time of usage. J. Oral Maxillofac. Pathol. 2023, 27, 333–339. [Google Scholar] [CrossRef]
- Smith, A.; Brewer, A.; Kirkpatrick, P.; Jackson, M.; Young, J.; Watson, S.; Thakker, B. Staphylococcal species in the oral cavity from patients in a regional burns unit. J. Hosp. Infect. 2003, 55, 184–189. [Google Scholar] [CrossRef]
- Dorocka-Bobkowska, B.; Konopka, K. Susceptibility of Candida isolates from denture-related stomatitis to antifungal agents in vitro. Int. J. Prosthodont. 2007, 20, 504–506. [Google Scholar] [PubMed]
- Ritchie, G.; Fletcher, A.; Main, D.; Prophet, A. The etiology, exfoliative cytology, and treatment of denture stomatitis. J. Prosthet. Dent. 1969, 22, 185–200. [Google Scholar] [CrossRef] [PubMed]
- Manfredi, M.; McCullough, M.J.; Polonelli, L.; Conti, S.; Al-Karaawi, Z.M.; Vescovi, P.; Porter, S.R. In vitro antifungal susceptibility to six antifungal agents of 229 Candida isolates from patients with diabetes mellitus. Oral Microbiol. Immunol. 2006, 21, 177–182. [Google Scholar] [CrossRef] [PubMed]
- Chandra, J.; Mukherjee, P.; Leidich, S.; Faddoul, F.; Hoyer, L.; Douglas, L.; Ghannoum, M. Antifungal resistance of candidal biofilms formed on denture acrylic in vitro. J. Dent. Res. 2001, 80, 903–908. [Google Scholar] [CrossRef] [PubMed]
- Koga-Ito, C.Y.; Lyon, J.P.; Vidotto, V.; de Resende, M.A. Virulence factors and antifungal susceptibility of Candida albicans isolates from oral candidosis patients and control individuals. Mycopathologia 2006, 161, 219–223. [Google Scholar] [CrossRef] [PubMed]
- Lamfon, H.; Porter, S.R.; McCullough, M.; Pratten, J. Susceptibility of Candida albicans biofilms grown in a constant depth film fermentor to chlorhexidine, fluconazole and miconazole: A longitudinal study. J. Antimicrob. Chemother. 2004, 53, 383–385. [Google Scholar] [CrossRef] [PubMed]
- Perić, M.; Miličić, B.; Pfićer, J.K.; Živković, R.; Arsenijević, V.A. A systematic review of denture stomatitis: Predisposing factors, clinical features, etiology, and global Candida spp. distribution. J. Fungi 2024, 10, 328. [Google Scholar] [CrossRef]
- Le Bars, P.; Kouadio, A.A.; Bandiaky, O.N.; Le Guéhennec, L.; de La Cochetière, M.-F. Host’s Immunity and Candida Species Associated with Denture Stomatitis: A Narrative Review. Microorganisms 2022, 10, 1437. [Google Scholar] [CrossRef] [PubMed]
- Sartawi, S.Y.; Abu-Hammad, S.; Salim, N.A.; Al-Omoush, S. Denture stomatitis revisited: A summary of systematic reviews in the past decade and two case reports of papillary hyperplasia of unusual locations. Int. J. Dent. 2021, 2021, 7338143. [Google Scholar] [CrossRef] [PubMed]
- Jewtuchowicz, V.M.; Brusca, M.I.; Mujica, M.T.; Gliosca, L.A.; Finquelievich, J.L.; Lovannitti, C.A.; Rosa, A.C. Subgingival distribution of yeast and their antifungal susceptibility in immunocompetent subjects with and without dental devices. Acta Odontol. Latinoam. 2007, 20, 17–22. [Google Scholar]
- de Melo Costa, D.; Kipnis, A.; de Oliveira Leão-Vasconcelos, L.S.N.; Rocha-Vilefort, L.O.; Telles, S.A.; André, M.C.D.P.B.; Tipple, A.F.V.; Lima, A.B.M.; Ribeiro, N.F.G.; Pereira, M.R. Methicillin-resistant Staphylococcus sp. colonizing health care workers of a cancer hospital. Braz. J. Microbiol. 2014, 45, 799–805. [Google Scholar] [CrossRef]
- Lewis, N.; Parmar, N.; Hussain, Z.; Baker, G.; Green, I.; Howlett, J.; Kearns, A.; Cookson, B.; McDonald, A.; Wilson, M.; et al. Colonisation of dentures by Staphylococcus aureus and MRSA in out-patient and in-patient populations. Eur. J. Clin. Microbiol. Infect. Dis. 2015, 34, 1823–1826. [Google Scholar] [CrossRef] [PubMed]
- Chaulagain, R.; Thakur, S.N.; Khanal, B.; Sapkota, S.M.; Chaudhary, N.K. Microbial Spectrum of Complete Denture Wearer in Old Age People of Chitwan. J. Nepal Health Res. Counc. 2023, 21, 122–128. [Google Scholar] [CrossRef]
- Nishihama, S.; Kawada-Matsuo, M.; Le, M.N.-T.; Fujii, A.; Haruta, A.; Kajihara, T.; Hashimoto, Y.; Yoshikawa, M.; Aikawa, T.; Tsuga, K.; et al. Oral colonization of antimicrobial-resistant bacteria in home health care participants and their association with oral and systemic status. Sci. Rep. 2025, 15, 5776. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Archilla, A.; Garcia-Galan, C. Etiological factors related to denture stomatitis: A meta-analysis. Dent. Med. Res. 2020, 8, 37–42. [Google Scholar] [CrossRef]
- Martori, E.; Ayuso-Montero, R.; Martinez-Gomis, J.; Viñas, M.; Peraire, M. Risk factors for denture-related oral mucosal lesions in a geriatric population. J. Prosthet. Dent. 2014, 111, 273–279. [Google Scholar] [CrossRef]
- RB, O.; Khoder, G.; Fayed, B.; RA, K.; Elkareimi, Y.; Alharbi, N.J.P. Influence of fabrication technique on adhesion and biofilm formation of Candida albicans to conventional, milled, and 3D-printed denture base resin materials: A comparative in vitro study. Polymers 2023, 15, 1836. [Google Scholar] [CrossRef]
- Zheng, S.; Bawazir, M.; Dhall, A.; Kim, H.-E.; He, L.; Heo, J.; Hwang, G. Implication of surface properties, bacterial motility, and hydrodynamic conditions on bacterial surface sensing and their initial adhesion. Front. Bioeng. Biotechnol. 2021, 9, 643722. [Google Scholar] [CrossRef] [PubMed]
- Cankovic, M.; Bokor-Bratic, M.; Marinoski, J.; Stojanovic, D. Prevalence and possible predictors of the occurence of denture stomatitis in patients older than 60 years. Vojn. Pregl. 2017, 74, 311–316. [Google Scholar] [CrossRef]
- Emami, E.; de Grandmont, P.; Rompré, P.; Barbeau, J.; Pan, S.; Feine, J. Favoring Trauma as an Etiological Factor in Denture Stomatitis. J. Dent. Res. 2008, 87, 440–444. [Google Scholar] [CrossRef]
- Duyck, J.; Vandamme, K.; Krausch-Hofmann, S.; Boon, L.; De Keersmaecker, K.; Jalon, E.; Teughels, W.; Bencharit, S. Impact of denture cleaning method and overnight storage condition on denture biofilm mass and composition: A cross-over randomized clinical trial. PLoS ONE 2016, 11, e0145837. [Google Scholar] [CrossRef] [PubMed]
- Ben-Ami, R.; Olshtain-Pops, K.; Krieger, M.; Oren, I.; Bishara, J.; Dan, M.; Wiener-Well, Y.; Weinberger, M.; Zimhony, O.; Chowers, M.; et al. Antibiotic exposure as a risk factor for fluconazole-resistant candida bloodstream infection. Antimicrob. Agents Chemother. 2012, 56, 2518–2523. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharya, S.; Sae-Tia, S.; Fries, B.C. Candidiasis and mechanisms of antifungal resistance. Antibiotics 2020, 9, 312. [Google Scholar] [CrossRef]
- Twigg, J.A.; Smith, A.; Haury, C.; Wilson, M.J.; Lees, J.; Waters, M.; Williams, D.W. Compositional shifts within the denture-associated bacteriome in pneumonia—An analytical cross-sectional study. J. Med. Microbiol. 2023, 72, 001702. [Google Scholar] [CrossRef]
- Mhatre, S.; Srichand, R.; Sethumadhavan, J.; Mishra, P.B.; Patil, S.D.; Chavan, R.S.; Joshi, M.; Shetty, U. Dry mouth dilemma: A comprehensive review of xerostomia in complete denture wearers. Cureus 2024, 16, e58564. [Google Scholar] [CrossRef]
- Ichigaya, N.; Kawanishi, N.; Adachi, T.; Sugimoto, M.; Kimoto, K.; Hoshi, N. Effects of Denture Treatment on Salivary Metabolites: A Pilot Study. Int. J. Mol. Sci. 2023, 24, 13959. [Google Scholar] [CrossRef]
- Devcic, M.K.; Simonic-Kocijan, S.; Prpic, J.; Paskovic, I.; Cabov, T.; Kovac, Z.; Glazar, I. Oral candidal colonization in patients with different prosthetic appliances. J. Fungi 2021, 7, 662. [Google Scholar] [CrossRef] [PubMed]
- Quindos, G.; Gil-Alonso, S.; Marcos-Arias, C.; Sevillano, E.; Mateo, E.; Jauregizar, N.; Eraso, E. Therapeutic tools for oral candidiasis: Current and new antifungal drugs. Med. Oral Patol. Oral Cirugia Bucal 2019, 24, e172. [Google Scholar] [CrossRef]
- Lewis, M.; Williams, D.W. Diagnosis and management of oral candidosis. Br. Dent. J. 2017, 223, 675–681. [Google Scholar] [CrossRef]
- Xiao, Y.; Yuan, P.; Sun, Y.; Xu, Y.; Deng, X.; Wang, X.; Liu, R.; Chen, Q.; Jiang, L. Comparison of topical antifungal agents for oral candidiasis treatment: A systematic review and meta-analysis. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 2022, 133, 282–291. [Google Scholar] [CrossRef]
- Abuhajar, E.; Ali, K.; Zulfiqar, G.; Al Ansari, K.; Raja, H.Z.; Bishti, S.; Anweigi, L. Management of chronic atrophic candidiasis (denture stomatitis)—A narrative review. Int. J. Environ. Res. Public Health 2023, 20, 3029. [Google Scholar] [CrossRef]
- Rai, A.; Misra, S.R.; Panda, S.; Sokolowski, G.; Mishra, L.; Das, R.; Lapinska, B. Nystatin effectiveness in Oral candidiasis treatment: A Systematic Review & Meta-Analysis of clinical trials. Life 2022, 12, 1677. [Google Scholar] [CrossRef]
- Matteis, D.V.; Cascione, M.; Toma, C.C.; Albanese, G.; De Giorgi, M.L.; Corsalini, M.; Rinaldi, R. Silver nanoparticles addition in poly(methyl methacrylate) dental matrix: Topographic and antimycotic studies. Int. J. Mol. Sci. 2019, 20, 4691. [Google Scholar] [CrossRef] [PubMed]
- Aoun, G.; Cassia, A. Effectiveness of a Chlorhexidine Digluconate 0.12% and Cetylpyridinium Chloride 0.05% Solution in eliminating Candida albicans Colonizing Dentures: A Randomized Clinical in vivo Study. J. Contemp. Dent. Pract. 2015, 16, 433–436. [Google Scholar] [PubMed]
- Procópio, A.L.F.; Lara, V.S.; Porto, V.C.; Soares, S.; Fernandes, M.H.; Urban, V.M.; Neppelenbroek, K.H. Resilient liner modified by antimicrobials for denture stomatitis treatment: A randomized controlled trial. J. Dent. 2022, 126, 104297. [Google Scholar] [CrossRef] [PubMed]
- Mendes, L.V.; Marques, M.B.e.S.; Teixeira, A.B.V.; de Castro, D.T. Incorporation of antimicrobial materials into denture adhesives: A systematic review of in vitro studies. Int. J. Adhes. Adhes. 2025, 140, 104022. [Google Scholar] [CrossRef]
- Wang, J.; Lu, X.; Wang, C.; Yue, Y.; Wei, B.; Zhang, H.; Wang, H.; Chen, J. Research progress on the combination of quorum-sensing inhibitors and antibiotics against bacterial resistance. Molecules 2024, 29, 1674. [Google Scholar] [CrossRef]
- Zhao, X.; Yu, Z.; Ding, T. Quorum-sensing regulation of antimicrobial resistance in bacteria. Microorganisms 2020, 8, 425. [Google Scholar] [CrossRef]
- Wang, S.; Zhao, Y.; Breslawec, A.P.; Liang, T.; Deng, Z.; Kuperman, L.L.; Yu, Q. Strategy to combat biofilms: A focus on biofilm dispersal enzymes. npj Biofilms Microbiomes 2023, 9, 63. [Google Scholar] [CrossRef]
- Sharma, A.; Gupta, V.K.; Pathania, R. Efflux pump inhibitors for bacterial pathogens: From bench to bedside. Indian J. Med. Res. 2019, 149, 129–145. [Google Scholar] [CrossRef]
- Compagnoni, M.A.; Souza, R.F.; Marra, J.; Pero, A.C.; Barbosa, D.B. Relationship between Candida and nocturnal denture wear: Quantitative study. J. Oral Rehabil. 2007, 34, 600–605. [Google Scholar] [CrossRef] [PubMed]
- Aslam, A.; Gajdács, M.; Zin, C.S.; Ab Rahman, N.S.; Ahmed, S.I.; Zafar, M.Z.; Jamshed, S. Evidence of the practice of self-medication with antibiotics among the lay public in low- and middle-income countries: A scoping review. Antibiotics 2020, 9, 597. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.Y.; Prentice, E.L.; Webber, M.A. Mechanisms of antimicrobial resistance in biofilms. npj Antimicrob. Resist. 2024, 2, 27. [Google Scholar] [CrossRef] [PubMed]
- AOAd, F.; Marquezan, M.; MdCG, N.; DS, A.; Maia, L.C. The influence of orthodontic fixed appliances on the oral microbiota: A systematic review. Dent. Press J. Orthod. 2014, 19, 46–55. [Google Scholar] [CrossRef] [PubMed]
- Mulimani, P.; Popowics, T. Effect of orthodontic appliances on the oral environment and microbiome. Front. Dent. Med. 2022, 3, 924835. [Google Scholar] [CrossRef]
- Lucchese, A.; Bondemark, L.; Marcolina, M.; Manuelli, M. Changes in oral microbiota due to orthodontic appliances: A systematic review. J. Oral Microbiol. 2018, 10, 1476645. [Google Scholar] [CrossRef]
- Hamdoon, S.M.; AlSamak, S.; Ahmed, M.K.; Gasgoos, S. Evaluation of biofilm formation on different clear orthodontic retainer materials. J. Orthod. Sci. 2022, 11, 34. [Google Scholar] [CrossRef] [PubMed]
- Nascimento, L.E.A.G.D.; Pithon, M.M.; dos Santos, R.L.; Freitas, A.O.A.; Alviano, D.S.; Nojima, L.I.; Nojima, M.C.G.; Ruellas, A.C.d.O. Colonization of Streptococcus mutans on esthetic brackets: Self-ligating vs. conventional. Am. J. Orthod. Dentofac. Orthop. 2013, 143, S72–S77. [Google Scholar] [CrossRef]
- Pellissari, B.A.; Sabino, G.S.P.; Lima, R.N.d.S.; Motta, R.H.L.; Suzuki, S.S.; Garcez, A.S.; Basting, R.T.; Barbosa, J.A.; Montalli, V.A.M. Antimicrobial resistance of bacterial strains in patients undergoing orthodontic treatment with and without fixed appliances. Angle Orthod. 2021, 91, 672–679. [Google Scholar] [CrossRef] [PubMed]
- Sabino, G.; Suzuki, S.; Segundo, A.G.; Czezacki, A.; Pugliesi, T.; Saba, G.; Almeida, T.; Caselato, I.; Montalli, V. Prevalence of drug-resistant microorganisms in oral cavity during orthodontic treatment. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 2019, 128, e62. [Google Scholar] [CrossRef]
- Lazar, L.; Vlasa, A.; Beresescu, L.; Bud, A.; Lazar, A.P.; Matei, L.; Bud, E. White spot lesions (WSLs)—Post-orthodontic occurrence, management and treatment alternatives: A narrative review. J. Clin. Med. 2023, 12, 1908. [Google Scholar] [CrossRef] [PubMed]
- Papageorgiou, S.N.; Papadelli, A.A.; Eliades, T. Effect of orthodontic treatment on periodontal clinical attachment: A systematic review and meta-analysis. Eur. J. Orthod. 2017, 40, 176–194. [Google Scholar] [CrossRef]
- Verrusio, C.; Iorio-Siciliano, V.; Blasi, A.; Leuci, S.; Adamo, D.; Nicolò, M. The effect of orthodontic treatment on periodontal tissue inflammation: A systematic review. Quintessence Int. 2018, 49, 69–77. [Google Scholar]
- Abutayyem, H.; Alshehhi, M.A.; Alameri, M.; Zafar, M.S. Microbial adhesion on different types of orthodontic brackets and wires: An in vitro study. Saudi Dent. J. 2024, 36, 1459–1465. [Google Scholar] [CrossRef]
- Pavlic, A.; Begic, G.; Tota, M.; Abram, M.; Spalj, S.; Gobin, I. Bacterial exposure to nickel: Influence on adhesion and biofilm formation on orthodontic archwires and sensitivity to antimicrobial agents. Materials 2021, 14, 4603. [Google Scholar] [CrossRef] [PubMed]
- Topolska, J.M.; Jagielska, A.; Motyl, S.; Kozub-Budzyń, G.A.; Kępa, L.; Wagner, B.; Wątor, K. Metal leakage from orthodontic appliances chemically alters enamel surface during experimental in vitro simulated treatment. Sci. Rep. 2024, 14, 5412. [Google Scholar] [CrossRef]
- Mei, L.; Chieng, J.; Wong, C.; Benic, G.; Farella, M. Factors affecting dental biofilm in patients wearing fixed orthodontic appliances. Prog. Orthod. 2017, 18, 4. [Google Scholar] [CrossRef] [PubMed]
- Kaluarachchi, H.; Chung, K.C.C.; Zamble, D.B. Microbial nickel proteins. Nat. Prod. Rep. 2010, 27, 681–694. [Google Scholar] [CrossRef]
- Macomber, L.; Hausinger, R.P. Mechanisms of nickel toxicity in microorganisms. Metallomics 2011, 3, 1153–1162. [Google Scholar] [CrossRef] [PubMed]
- Hu, H.-W.; Wang, J.-T.; Li, J.; Shi, X.-Z.; Ma, Y.-B.; Chen, D.; He, J.-Z. Long-term nickel contamination increases the occurrence of antibiotic resistance genes in agricultural soils. Environ. Sci. Technol. 2016, 51, 790–800. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Li, X.; Sun, G.; Zhang, Y.; Su, J.; Ye, J. Heavy metal induced antibiotic resistance in bacterium LSJC7. Int. J. Mol. Sci. 2015, 16, 23390–23404. [Google Scholar] [CrossRef]
- Abdelatey, L.M.; Khalil, W.K.; Ali, T.H.; Mahrous, K.F. Heavy Metal Resistance and Gene Expression Analysis of Metal Resistance Genes in Gram-Positive and Gram-Negative Bacteria Present in Egyptian Soils. J. Appl. Sci. Environ. Sanit. 2011, 6, 201–211. [Google Scholar]
- Dittmer, M.P.; Hellemann, C.F.; Grade, S.; Heuer, W.; Stiesch, M.; Schwestka-Polly, R.; Demling, A.P. Comparative three-dimensional analysis of initial biofilm formation on three orthodontic bracket materials. Head Face Med. 2015, 11, 10. [Google Scholar] [CrossRef] [PubMed]
- Asghar, F.; Bano, A.; Waheed, F.; Anjum, A.A.; Ejaz, H.; Javed, N. Association of exogenous factors with molecular epidemiology of Staphylococcus aureus in human oral cavity. Saudi J. Biol. Sci. 2023, 30, 103613. [Google Scholar] [CrossRef]
- Poeta, P.; Igrejas, G.; Gonçalves, A.; Martins, E.; Araújo, C.; Carvalho, C.; Rodrigues, J.; Vinué, L.; López, M.; Torres, C. Influence of oral hygiene in patients with fixed appliances in the oral carriage of antimicrobial-resistant Escherichia coli and Enterococcus isolates. Oral. Surg. Oral Med. Oral Pathol. Oral Radiol. Endodontol. 2009, 108, 557–564. [Google Scholar] [CrossRef]
- Barylyak, A.; Kisała, J.; Fornal, P.; Bobitski, Y.; Dziedzic, A.; Kus-Liśkiewicz, M. Physical, mechanical and antibacterial properties of the orthodontic adhesive doped with S-TiO2. Sci. Rep. 2024, 14, 17862. [Google Scholar] [CrossRef]
- Murphy, A.M.; Patel, U.C.; Wilson, G.M.; Suda, K.J. Prevalence of unnecessary antibiotic prescriptions among dental visits, 2019. Infect. Control Hosp. Epidemiol. 2024, 45, 890–899. [Google Scholar] [CrossRef]
- Okuwaki, S.; Hosomichi, K.; Tajima, A.; Yamaguchi, T. An Observational Study on Changes in the Oral and Gut Microbiota through Professional Mechanical Tooth Cleaning, including Tooth-Brushing Instructions in Patients with Multi-Bracket Appliances. Appl. Sci. 2023, 13, 10843. [Google Scholar] [CrossRef]
- Sycińska-Dziarnowska, M.; Szyszka-Sommerfeld, L.; Ziąbka, M.; Spagnuolo, G.; Woźniak, K. Use of antimicrobial silver coatings on fixed orthodontic appliances, including archwires, brackets, and microimplants: A systematic review. Med. Sci. Monit. 2024, 30, e944255-1. [Google Scholar] [CrossRef] [PubMed]
- Abdallah, O.M.; Sedky, Y.; Shebl, H.R. Comprehensive evaluation of the antibacterial and antibiofilm activities of NiTi orthodontic wires coated with silver nanoparticles and nanocomposites: An in vitro study. BMC Oral Health 2024, 24, 1345. [Google Scholar] [CrossRef] [PubMed]
- Kameda, T.; Sato, H.; Oka, S.; Miyazaki, A.; Ohkuma, K.; Terada, K. Low temperature polytetrafluoroethylene (PTFE) coating improves the appearance of orthodontic wires without changing their mechanical properties. Dent. Mater. J. 2020, 39, 721–734. [Google Scholar] [CrossRef] [PubMed]
- Raji, S.H.; Shojaei, H.; Ghorani, P.S.; Rafiei, E. Bacterial colonization on coated and uncoated orthodontic wires: A prospective clinical trial. Dent. Res. J. 2014, 11, 680–683. [Google Scholar]
- Selvaraj, A.; George, A.M.; Rajeshkumar, S. Efficacy of zirconium oxide nanoparticles coated on stainless steel and nickel titanium wires in orthodontic treatment. Bioinformation 2021, 17, 760. [Google Scholar] [CrossRef]
- Thabit, A.K.; Aljereb, N.M.; Khojah, O.M.; Shanab, H.; Badahdah, A. Towards Wiser Prescribing of Antibiotics in Dental Practice: What Pharmacists Want Dentists to Know. Dent. J. 2024, 12, 345. [Google Scholar] [CrossRef]
- Lockhart, P.B.; Tampi, M.P.; Abt, E.; Aminoshariae, A.; Durkin, M.J.; Fouad, A.F.; Gopal, P.; Hatten, B.W.; Kennedy, E.; Lang, M.S. Evidence-based clinical practice guideline on antibiotic use for the urgent management of pulpal-and periapical-related dental pain and intraoral swelling: A report from the American Dental Association. J. Am. Dent. Assoc. 2019, 150, 906–921.e12. [Google Scholar] [CrossRef] [PubMed]
- Bansal, R.; Jain, A.; Goyal, M.; Singh, T.; Sood, H.; Malviya, H.S. Antibiotic abuse during endodontic treatment: A contributing factor to antibiotic resistance. J. Fam. Med. Prim. Care 2019, 8, 3518–3524. [Google Scholar] [CrossRef] [PubMed]
- Sperotto, F.; France, K.; Gobbo, M.; Bindakhil, M.; Pimolbutr, K.; Holmes, H.; Monteiro, L.; Graham, L.; Hong, C.H.L.; Sollecito, T.P.; et al. Antibiotic prophylaxis and infective endocarditis incidence following invasive dental procedures: A systematic review and meta-analysis. JAMA Cardiol. 2024, 9, 599–610. [Google Scholar] [CrossRef] [PubMed]
- Thornhill, M.H.; Gibson, T.B.; Durkin, M.J.; Dayer, M.J.; Lockhart, P.B.; O’Gara, P.T.; Baddour, L.M. Prescribing of antibiotic prophylaxis to prevent infective endocarditis. J. Am. Dent. Assoc. 2020, 151, 835–845.e31. [Google Scholar] [CrossRef]
- Fluent, M.T.; Jacobsen, P.L.; Hicks, L.A. Considerations for responsible antibiotic use in dentistry. J. Am. Dent. Assoc. 2016, 147, 683–686. [Google Scholar] [CrossRef] [PubMed]
- Cooper, L.; Sneddon, J.; Thompson, W.; Guise, T.; Robertson, D.; Smith, A. Tackling antimicrobial resistance in practice: Dental students’ evaluation of university teaching supplemented by an online course. JAC-Antimicrob. Resist. 2022, 4, dlac039. [Google Scholar] [CrossRef]
- Castro-Sánchez, E.; Drumright, L.N.; Gharbi, M.; Farrell, S.; Holmes, A.H.; Panepinto, J. Mapping antimicrobial stewardship in undergraduate medical, dental, pharmacy, nursing and veterinary education in the United Kingdom. PLoS ONE 2016, 11, e0150056. [Google Scholar] [CrossRef] [PubMed]
- Teoh, L.; Thompson, W.; Suda, K. Antimicrobial stewardship in dental practice. J. Am. Dent. Assoc. 2020, 151, 589–595. [Google Scholar] [CrossRef]
- Martine, C.; Sutherland, S.; Born, K.; Thompson, W.; Teoh, L.; Singhal, S. Dental antimicrobial stewardship: A qualitative study of perspectives among Canadian dentistry sector leaders and experts in antimicrobial stewardship. JAC-Antimicrob. Resist. 2024, 6, dlae082. [Google Scholar] [CrossRef] [PubMed]
- Niemczyk, W.; Żurek, J.; Niemczyk, S.; Kępa, M.; Zięba, N.; Misiołek, M.; Wiench, R. Antibiotic-Loaded Platelet-Rich Fibrin (AL-PRF) as a New Carrier for Antimicrobials: A Systematic Review of In Vitro Studies. Int. J. Mol. Sci. 2025, 26, 2140. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).