Connecting Main-Group Metals (Al, Ga, In) and Tungsten(0) Carbonyls via the N2S2 Metallo-Ligand Strategy
Abstract
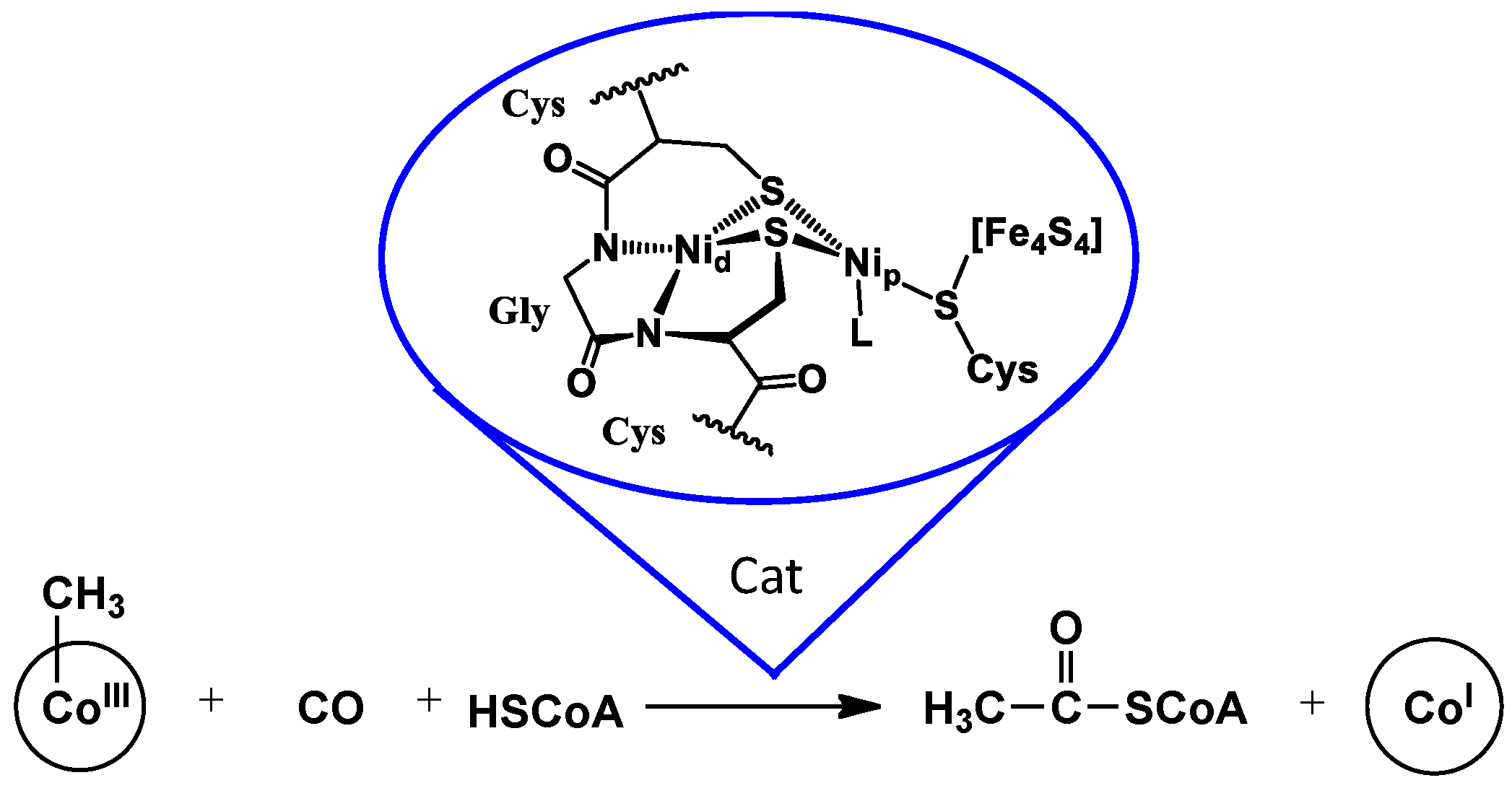
1. Introduction
2. Results
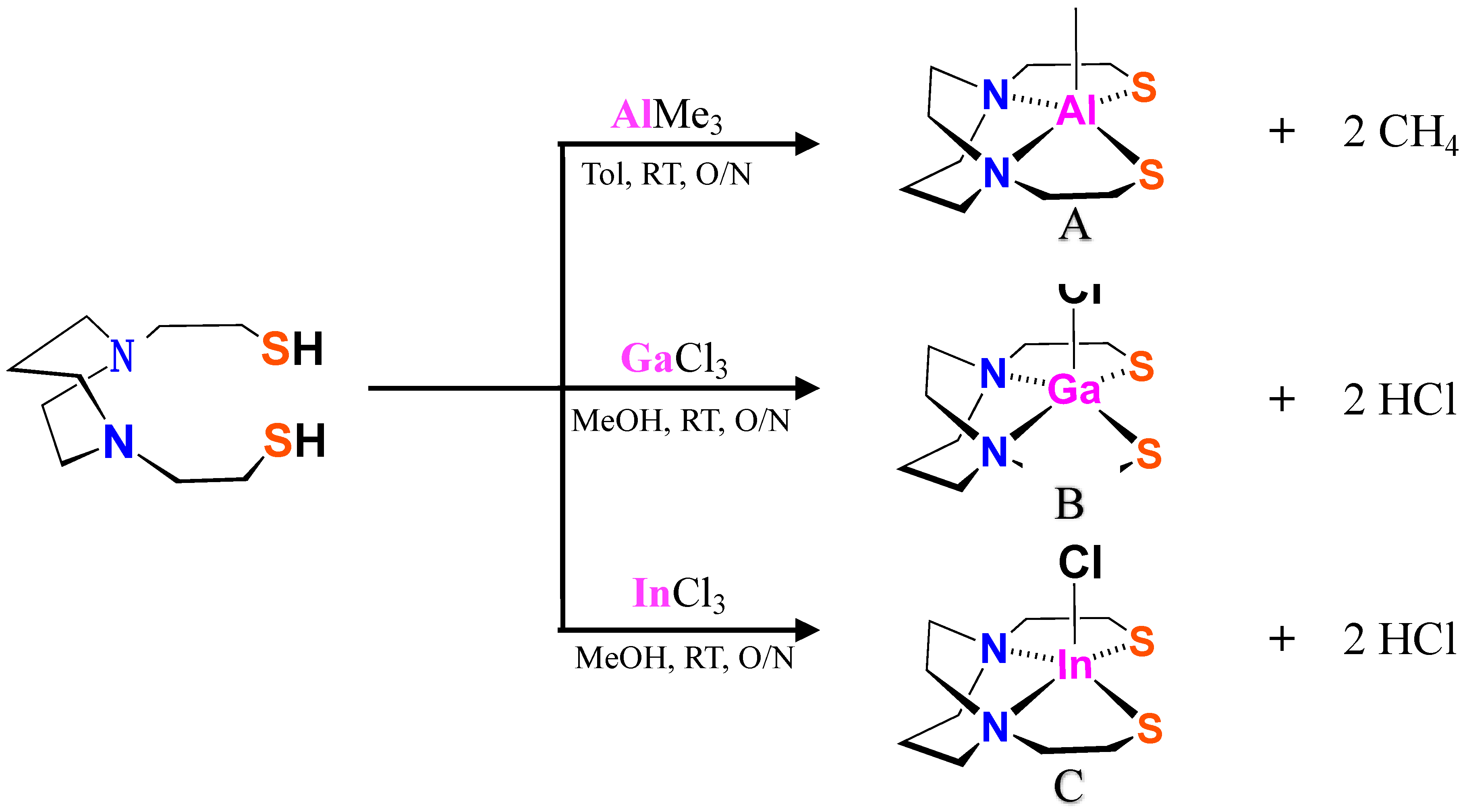
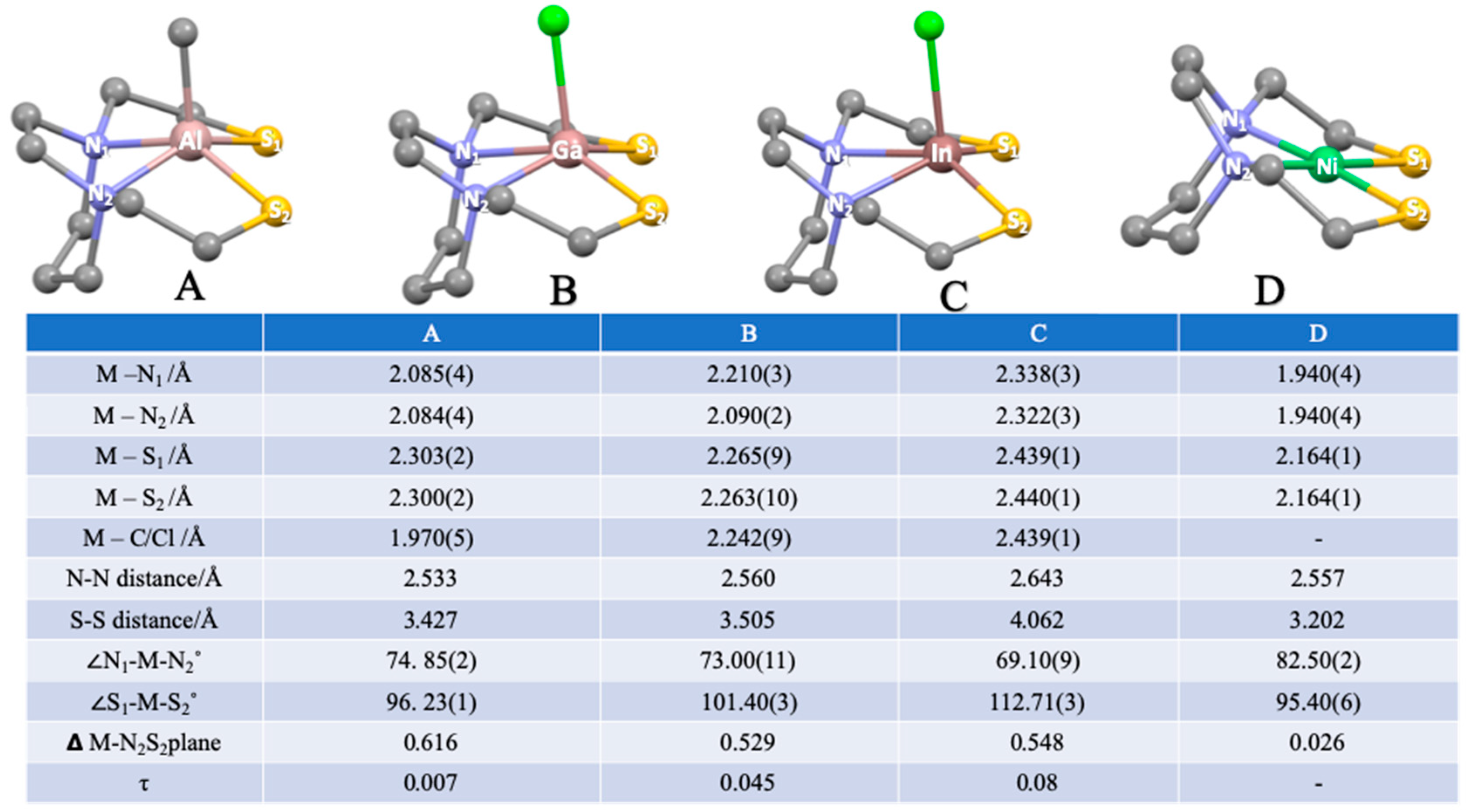
2.1. Synthesis and Characterizations of XMN2S2 Complexes
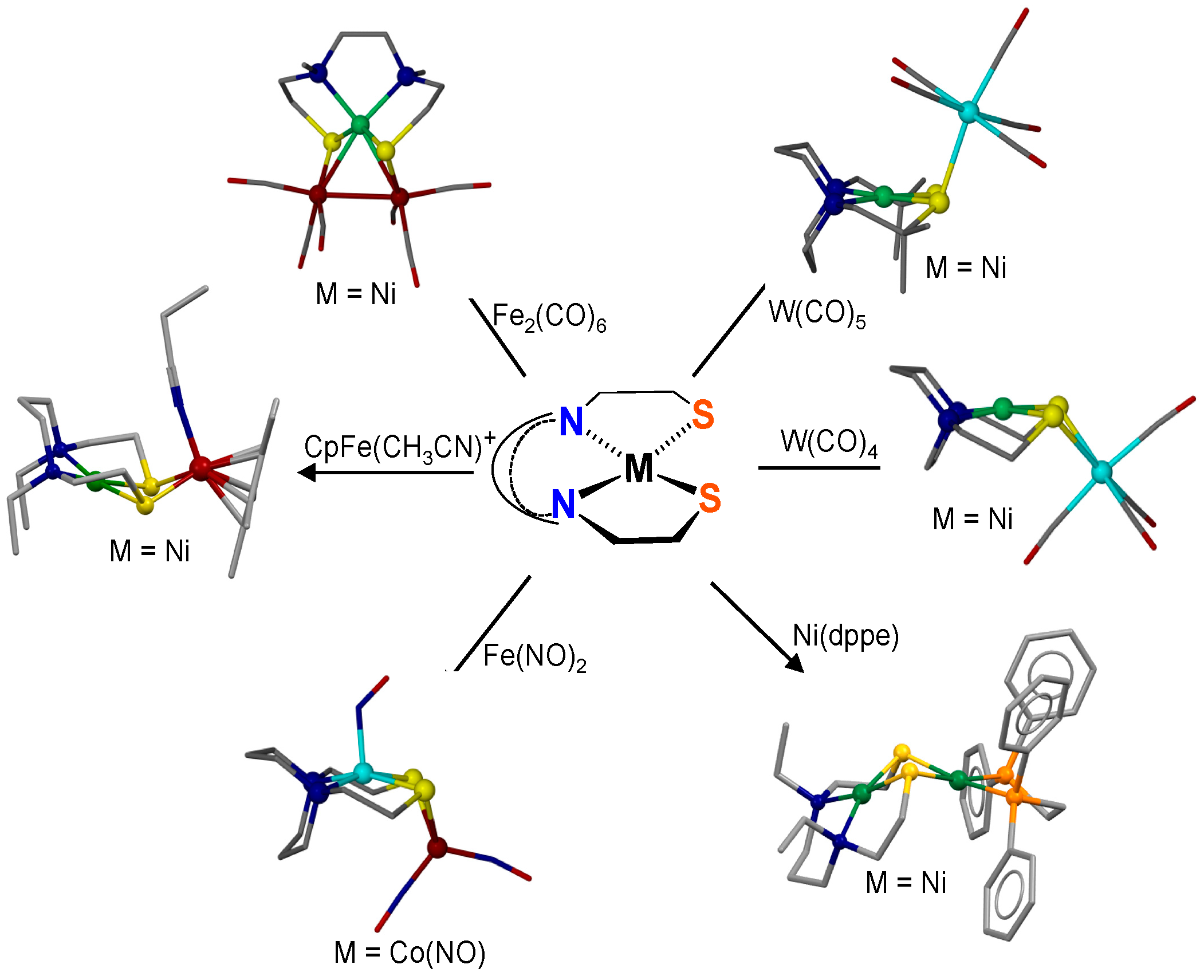
2.2. Reactions of XMN2S2 with Ni(II) Sources
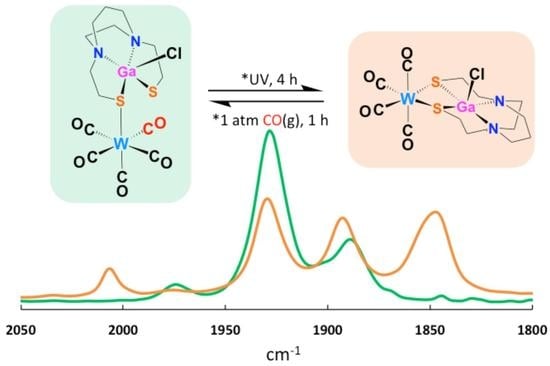
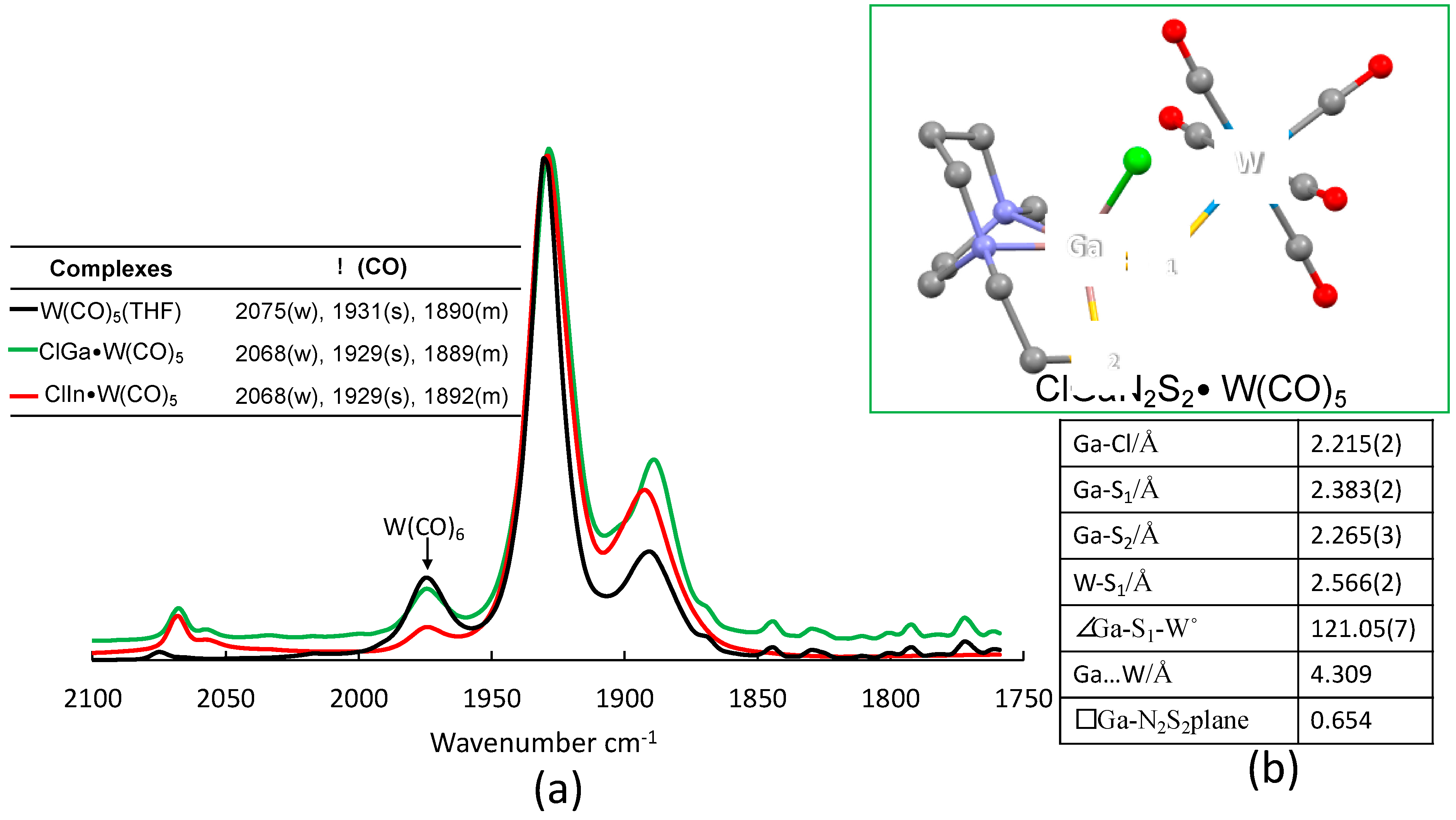
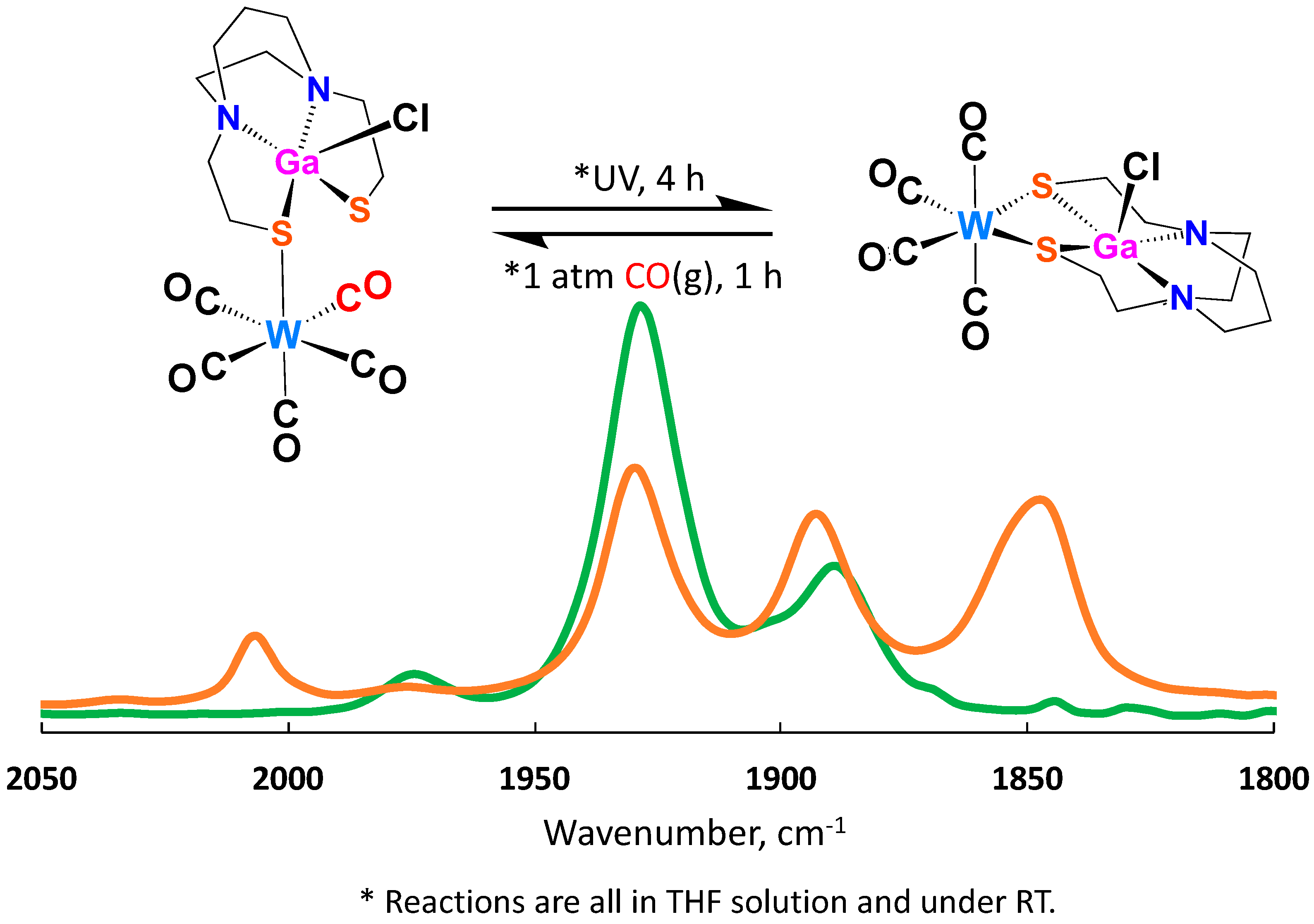
2.3. Nucleophilicity of S Lone Pairs on XMN2S2 Towards the Soft Receiver W(CO)5
3. Materials and Methods
3.1. Synthesis of XMN2S2
3.2. Reactions of XMN2S2 with Ni(II)
3.3. Synthesis of ClMW(CO)5
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Busch, D.H.; Jicha, D.C.; Thompson, M.C.; Wrathall, J.W.; Blinn, E. Reactions of Coordinated Ligands. VIII. The Reactions of Alkyl Halides with Mercapto Groups in Transition Metal Complexes of Mercaptoamines. J. Am. Chem. Soc. 1964, 86, 3642–3650. [Google Scholar] [CrossRef]
- Can, M.; Armstrong, F.A.; Ragsdale, S.W. Structure, Function, and Mechanism of the Nickel Metalloenzymes, CO Dehydrogenase, and Acetyl-CoA Synthase. Chem. Rev. 2014, 114, 4149–4174. [Google Scholar] [CrossRef] [PubMed]
- Doukov, T.I.; Blasiak, L.C.; Seravalli, J.; Ragsdale, S.W.; Drennan, C.L. Xenon in and at the End of the Tunnel of Bifunctional Carbon Monoxide Dehydrogenase/Acetyl-CoA Synthase. Biochemistry 2008, 47, 3474–3483. [Google Scholar] [CrossRef] [PubMed]
- Smee, J.J.; Miller, M.L.; Grapperhaus, C.A.; Reibenspies, J.H.; Darensbourg, M.Y. Subtle Bite-Angle Influences on N2S2Ni Complexes. Inorg. Chem. 2001, 40, 3601–3605. [Google Scholar] [CrossRef]
- Mills, D.K.; Reibenspies, J.H.; Darensbourg, M.Y. Sterically protected nickel(II) in a N2S2 donor environment: 1,5-bis(mercaptoethyl)-1,5-diazacyclooctane and its methylated derivative. Inorg. Chem. 1990, 29, 4364–4366. [Google Scholar] [CrossRef]
- Zhao, T.; Ghosh, P.; Martinez, Z.; Liu, X.; Meng, X.; Darensbourg, M.Y. Discrete Air-Stable Nickel(II)–Palladium(II) Complexes as Catalysts for Suzuki-Miyaura Reactions. Organometallics 2017, 36, 1822–1827. [Google Scholar] [CrossRef]
- Denny, J.A.; Darensbourg, M.Y. Metallodithiolates as Ligands in Coordination, Bioinorganic, and Organometallic Chemistry. Chem. Rev. 2015, 115, 5248–5273. [Google Scholar] [CrossRef]
- Yang, Z.; Yi, Y.; Zhong, M.; De, S.; Mondal, T.; Koley, D.; Ma, X.; Zhang, D.; Roesky, H.W. Addition Reactions of Me3SiCN with Aldehydes Catalyzed by Aluminum Complexes Containing in Their Coordination Sphere O, S, and N Ligands. Chem. Eur. J. 2016, 22, 6932–6938. [Google Scholar] [CrossRef]
- Chu, T.; Vyboishchikov, S.F.; Gabidullin, B.; Nikonov, G.I. Oxidative Cleavage of C=S and P=S Bonds at an AlI Center: Preparation of Terminally Bound Aluminium Sulfides. Angew. Chem. Int. Ed. 2016, 55, 13306–13311. [Google Scholar] [CrossRef]
- Kim, S.H.; Han, S.Y.; Kim, J.H.; Kang, Y.Y.; Lee, J.; Kim, Y. Monomeric or Dimeric Aluminium Complexes as Catalysts for Cycloaddition between CO2 and Epoxides. Eur. J. Inorg. Chem. 2015, 13, 2323–2329. [Google Scholar] [CrossRef]
- Darensbourg, D.J.; Billodeaux, D.R. Aluminum Salen Complexes and Tetrabutylammonium Salts: A Binary Catalytic System for Production of Polycarbonates from CO2 and Cyclohexene Oxide. Inorg. Chem. 2005, 44, 1433–1442. [Google Scholar] [CrossRef] [PubMed]
- Alam, I.S.; Arrowsmith, R.L.; Cortezon-Tamarit, F.; Twyman, F.; Kociok-Köhn, G.; Botchway, S.W.; Dilworth, J.R.; Carroll, L.; Aboagye, E.O.; Pascu, S.I. Microwave Gallium-68 Radiochemistry for Kinetically Stable bis(thiosemicarbazone) Complexes: Structural Investigations and Cellular Uptake under Hypoxia. Dalton Trans. 2015, 45, 144–155. [Google Scholar] [CrossRef] [PubMed]
- Zhang, N.; Tai, Y.; Li, M.; Ma, P.; Zhao, J.; Niu, J. Main Group Bismuth(III), Gallium(III) and Diorganotin(IV) Complexes derived from bis(2-acetylpyrazine) thiocarbonohydrazone: Synthesis, Crystal Structures and Biological Evaluation. Dalton Trans. 2014, 43, 5182–5189. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Dodonov, V.A.; Chen, W.; Zhao, Y.; Skatova, A.A.; Fedushkin, I.L.; Roesky, P.W.; Wu, B.; Yang, X.J. Cycloaddition versus Cleavage of the C=S Bond of Isothiocyanates Promoted by Digallane Compounds with Noninnocent α-Diimine Ligands. Chem. Eur. J. 2018, 24, 14994–15002. [Google Scholar] [CrossRef] [PubMed]
- Tai, Y.; Ji, Y.; Lu, Y.; Li, M.; Wu, Y.; Han, Q. Cadmium(II) and Indium(III) Complexes Derived from 2-benzoylpyridine N(4)-cyclohexylthiosemicarbazone: Synthesis, Crystal Structures, Spectroscopic Characterization and Cytotoxicity. Synth. Met. 2016, 219, 109–114. [Google Scholar] [CrossRef]
- Arrowsmith, R.L.; Waghorn, P.A.; Jones, M.W.; Bauman, A.; Brayshaw, S.K.; Hu, Z.; Kociok-Köhn, G.; Mindt, T.L.; Tyrrell, R.M.; Botchway, S.W.; et al. Fluorescent Gallium and Indium bis(thiosemicarbazonates) and Their Radiolabelled Analogues: Synthesis, Structures and Cellular Confocal Fluorescence Imaging Investigations. Dalton Trans. 2011, 2011. 40, 6238–6252. [Google Scholar] [CrossRef]
- Anderson, T.S.; Briand, G.G.; Brüning, R.; Decken, A.; Margeson, M.J.; Pickard, H.M.; Trevors, E.E. Synthesis, Characterization and Reactivity of (dithiolato)Indium Complexes. Polyhedron 2017, 135, 101–108. [Google Scholar] [CrossRef]
- Wadas, T.J.; Wong, E.H.; Weisman, G.R.; Anderson, C.J. Coordinating Radiometals of Copper, Gallium, Indium, Yttrium, and Zirconium for PET and SPECT Imaging of Disease. Chem. Rev. 2010, 110, 2858–2902. [Google Scholar] [CrossRef]
- Addison, A.W.; Rao, T.N.; Reedijk, J.; Jacobus, V.R.; Verschoor, G.C. Synthesis, Structure, and Spectroscopic Properties of Copper(II) Compounds Containing Nitrogen–Sulphur Donor Ligands; the Crystal and Molecular Structure of aqua [1,7-bis(N-methylbenzimidazol-2′-yl)-2,6-dithiaheptane]Copper(II) Perchlorate. J. Chem. Soc. Dalton Trans. 1984, 7, 1349–1356. [Google Scholar] [CrossRef]
- Hess, J.; Conder, H.L.; Green, K.N.; Darensbourg, M.Y. Electronic Effects of (N2S2)M(NO) Complexes (M = Fe, Co) as Metallodithiolate Ligands. Inorg. Chem. 2008, 47, 2056–2063. [Google Scholar] [CrossRef]
- Jenkins, R.; Pinder, T.A.; Hatley, M.L.; Reibenspies, J.H.; Darensbourg, M.Y. Tetradentate N2S2 Vanadyl(IV) Coordination Complexes: Synthesis, Characterization, and Reactivity Studies. Inorg. Chem. 2011, 50, 1849–1855. [Google Scholar] [CrossRef] [PubMed]
- Darensbourg, D.J.; Kump, R.L. A Convenient Synthesis of cis-Mo(CO)4L2 Derivatives (L = Group 5A Ligand) and a Qualitative Study of Their Thermal Reactivity toward Ligand Dissociation. Inorg. Chem. 1978, 17, 2680–2682. [Google Scholar] [CrossRef]
- Rampersad, M.V.; Jeffery, S.P.; Golden, M.L.; Lee, J.; Reibenspies, J.H.; Darensbourg, D.J.; Darensbourg, M.Y. Characterization of Steric and Electronic Properties of NiN2S2 Complexes as S-donor Metallodithiolate Ligands. J. Am. Chem. Soc. 2005, 127, 17323–17334. [Google Scholar] [CrossRef] [PubMed]
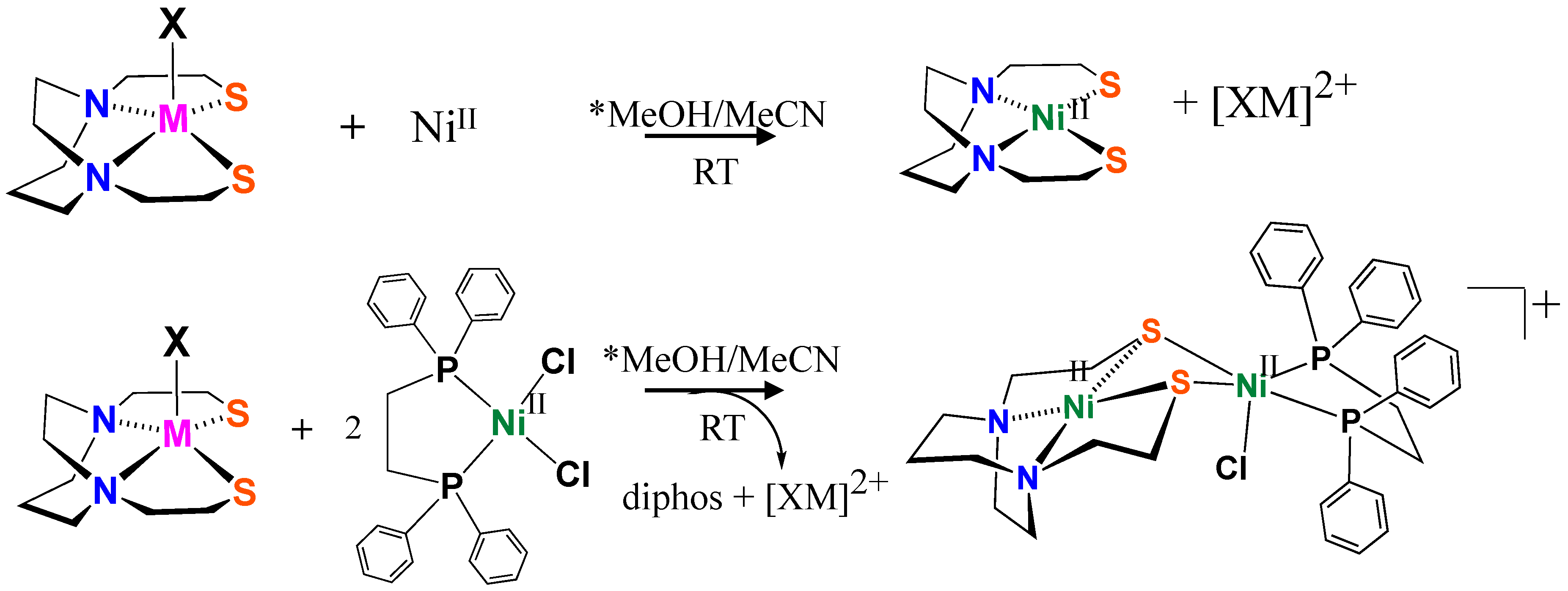
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yang, X.; Lunsford, A.; Darensbourg, M.Y. Connecting Main-Group Metals (Al, Ga, In) and Tungsten(0) Carbonyls via the N2S2 Metallo-Ligand Strategy. Inorganics 2019, 7, 115. https://doi.org/10.3390/inorganics7090115
Yang X, Lunsford A, Darensbourg MY. Connecting Main-Group Metals (Al, Ga, In) and Tungsten(0) Carbonyls via the N2S2 Metallo-Ligand Strategy. Inorganics. 2019; 7(9):115. https://doi.org/10.3390/inorganics7090115
Chicago/Turabian StyleYang, Xuemei, Allen Lunsford, and Marcetta Y. Darensbourg. 2019. "Connecting Main-Group Metals (Al, Ga, In) and Tungsten(0) Carbonyls via the N2S2 Metallo-Ligand Strategy" Inorganics 7, no. 9: 115. https://doi.org/10.3390/inorganics7090115
APA StyleYang, X., Lunsford, A., & Darensbourg, M. Y. (2019). Connecting Main-Group Metals (Al, Ga, In) and Tungsten(0) Carbonyls via the N2S2 Metallo-Ligand Strategy. Inorganics, 7(9), 115. https://doi.org/10.3390/inorganics7090115