Water Oxidation Catalysts: The Quest for New Oxide-Based Materials
Abstract
1. Introduction
2. Recent Developments of Metal Oxide-Based WOCs
2.1. Ru-, Ir-, and Cu-based Metal Oxides
2.2. Noble Metal-Free Oxide-Related WOCs
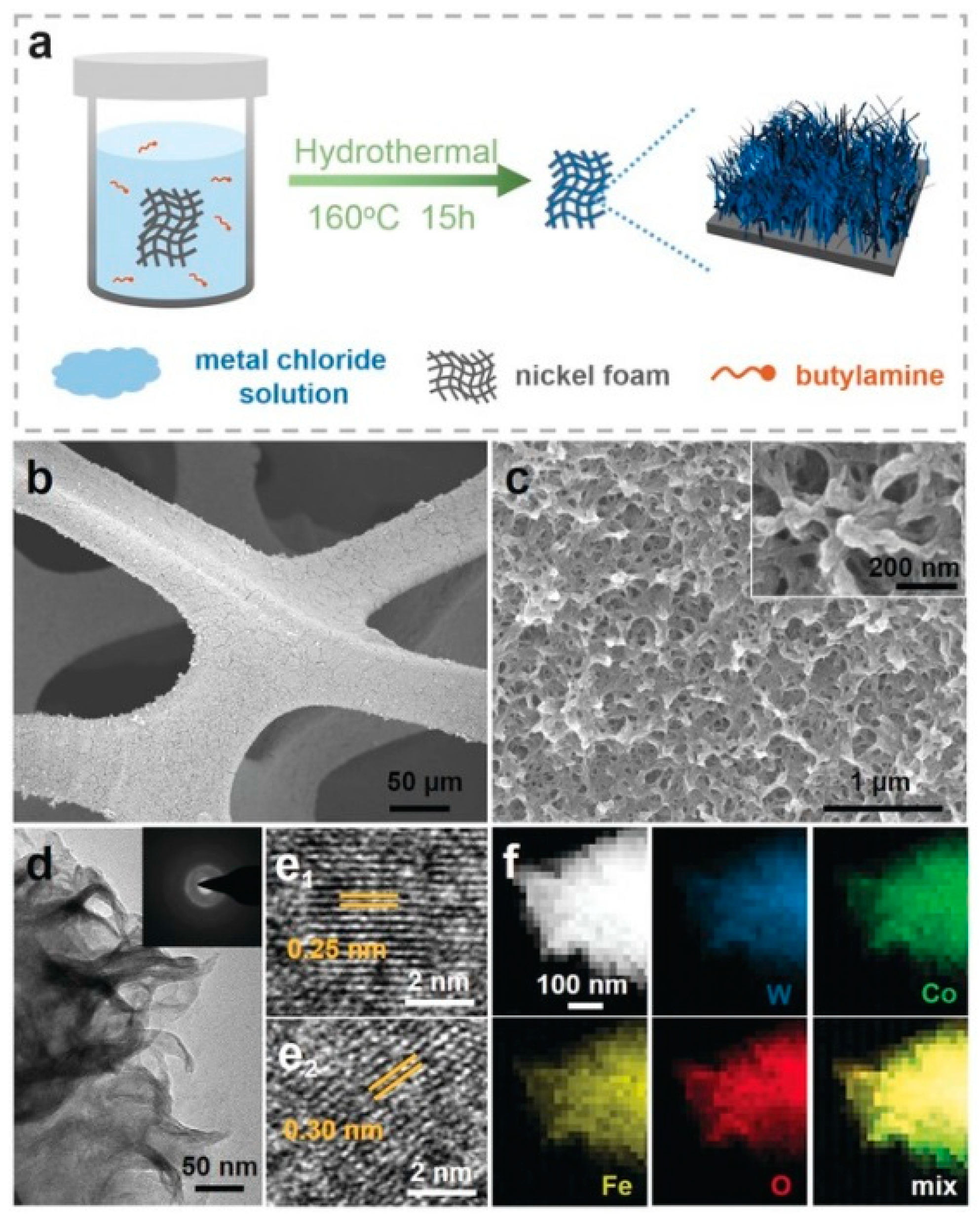
2.2.1. Layered Hydroxides and Related Ni–Fe-Based Materials
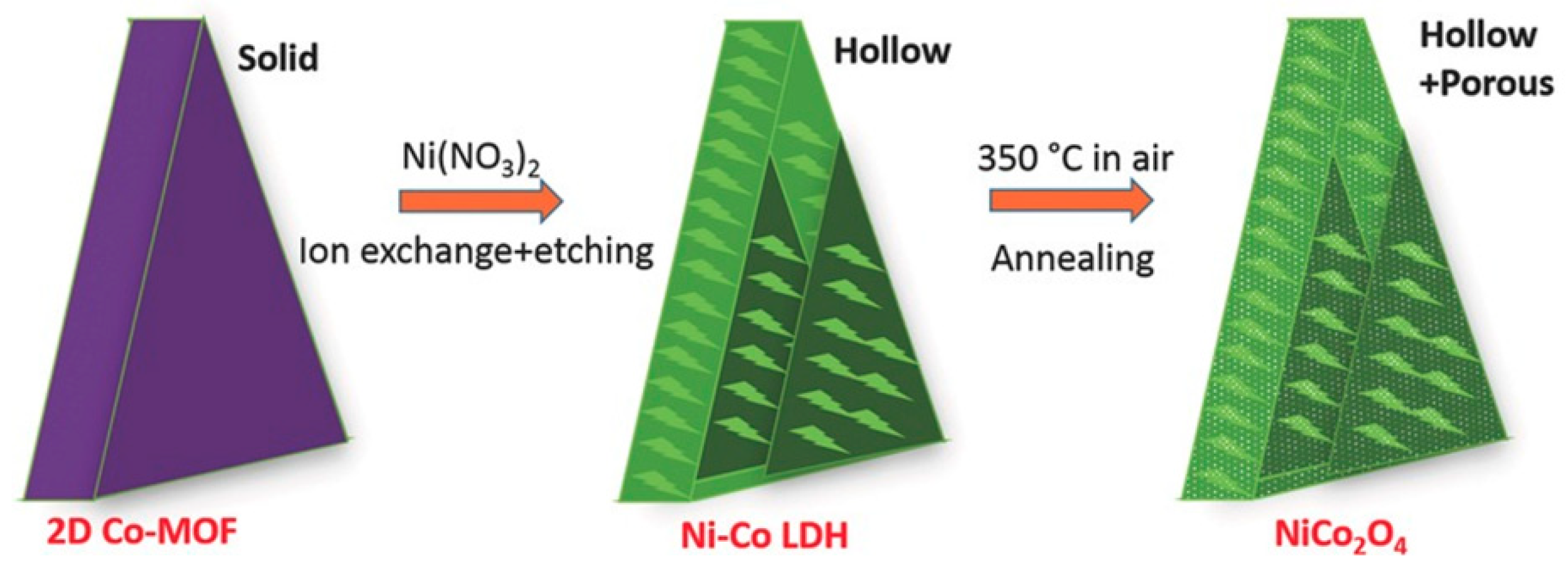
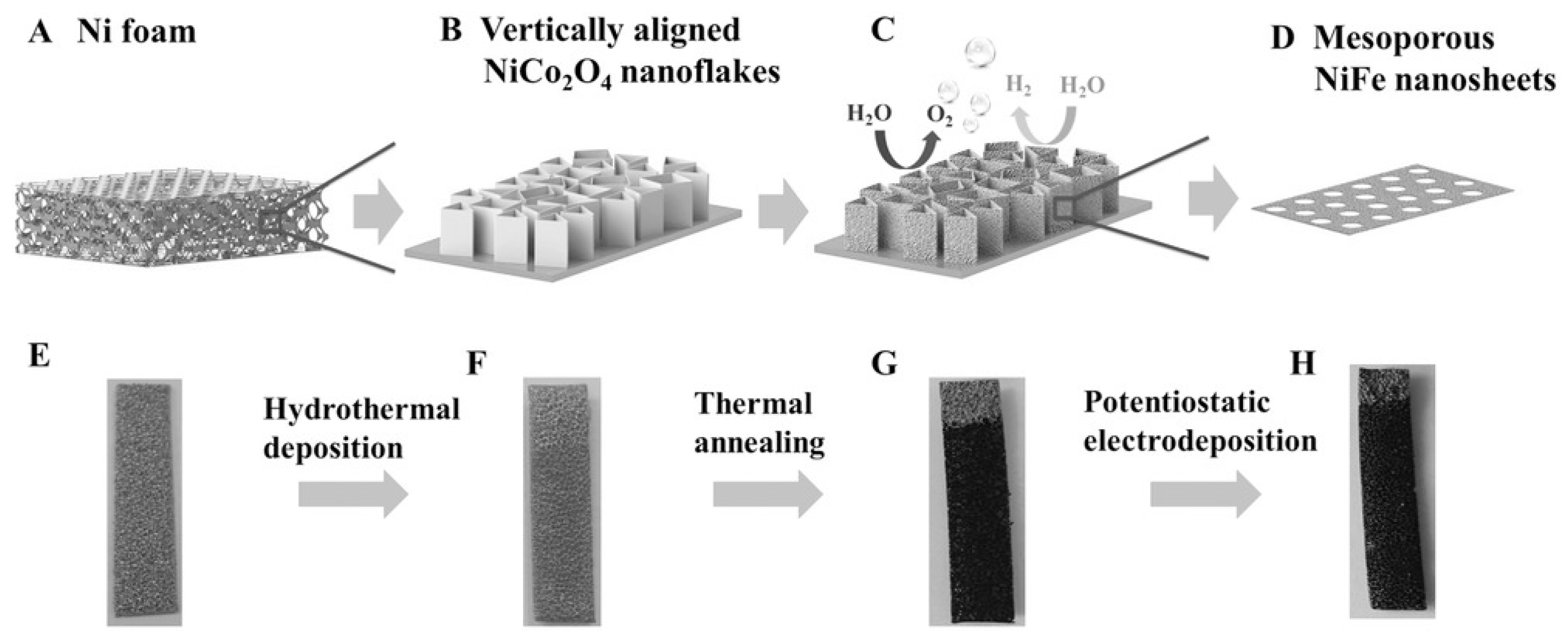
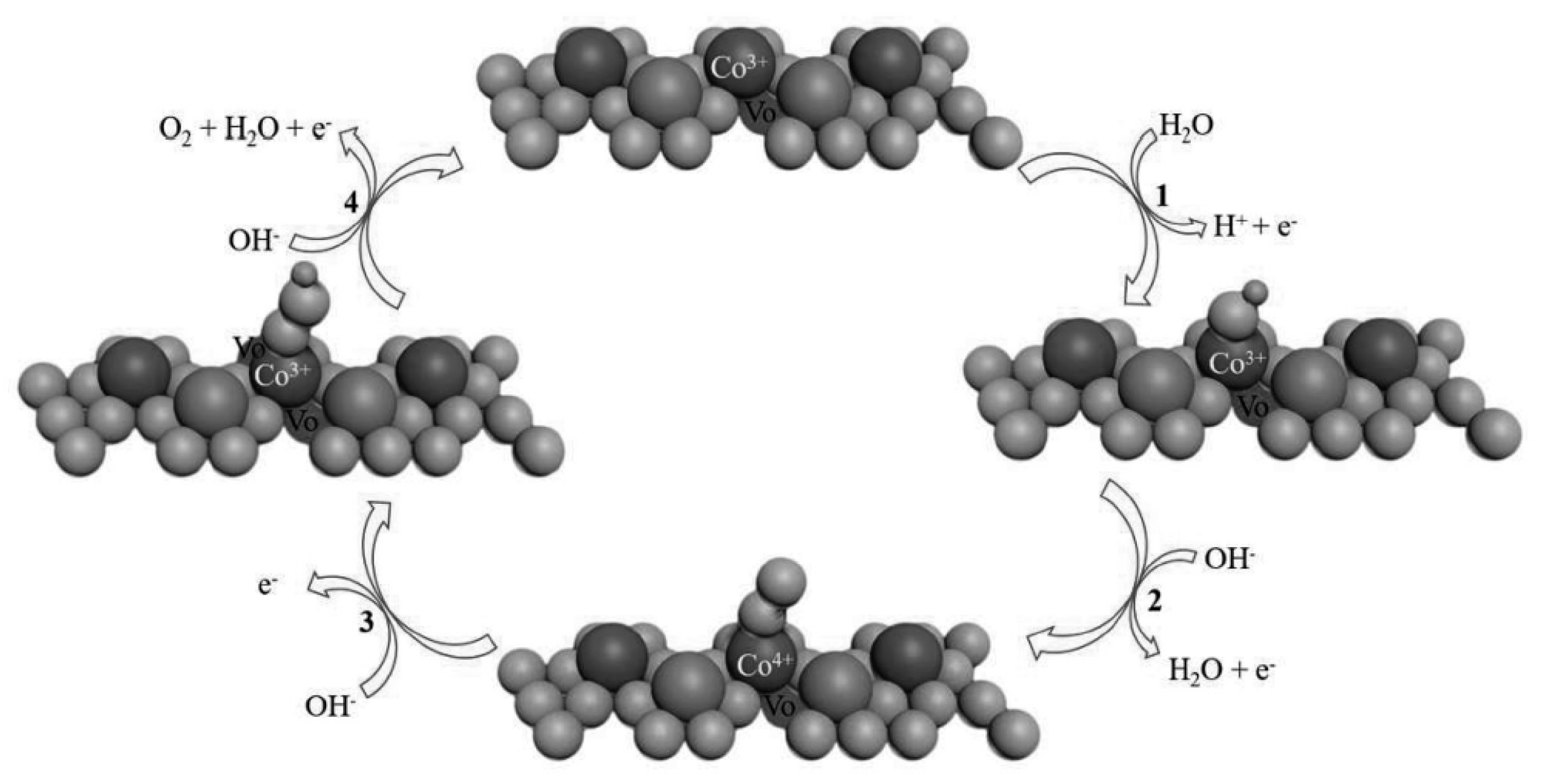
2.2.2. Spinels
| Catalyst | Preparation Method | Onset Potential (V) | η (at 10 mV/cm2) (V) | Tafel Slope (mV dec−1) | Durability (h) | Stability | Electrolyte | Ref. |
|---|---|---|---|---|---|---|---|---|
| Co3O4/N-graphene | Hydrothermal | 1.49 | 0.96 | 121.8 | 4 | Good * | 0.1 M KOH | [50] |
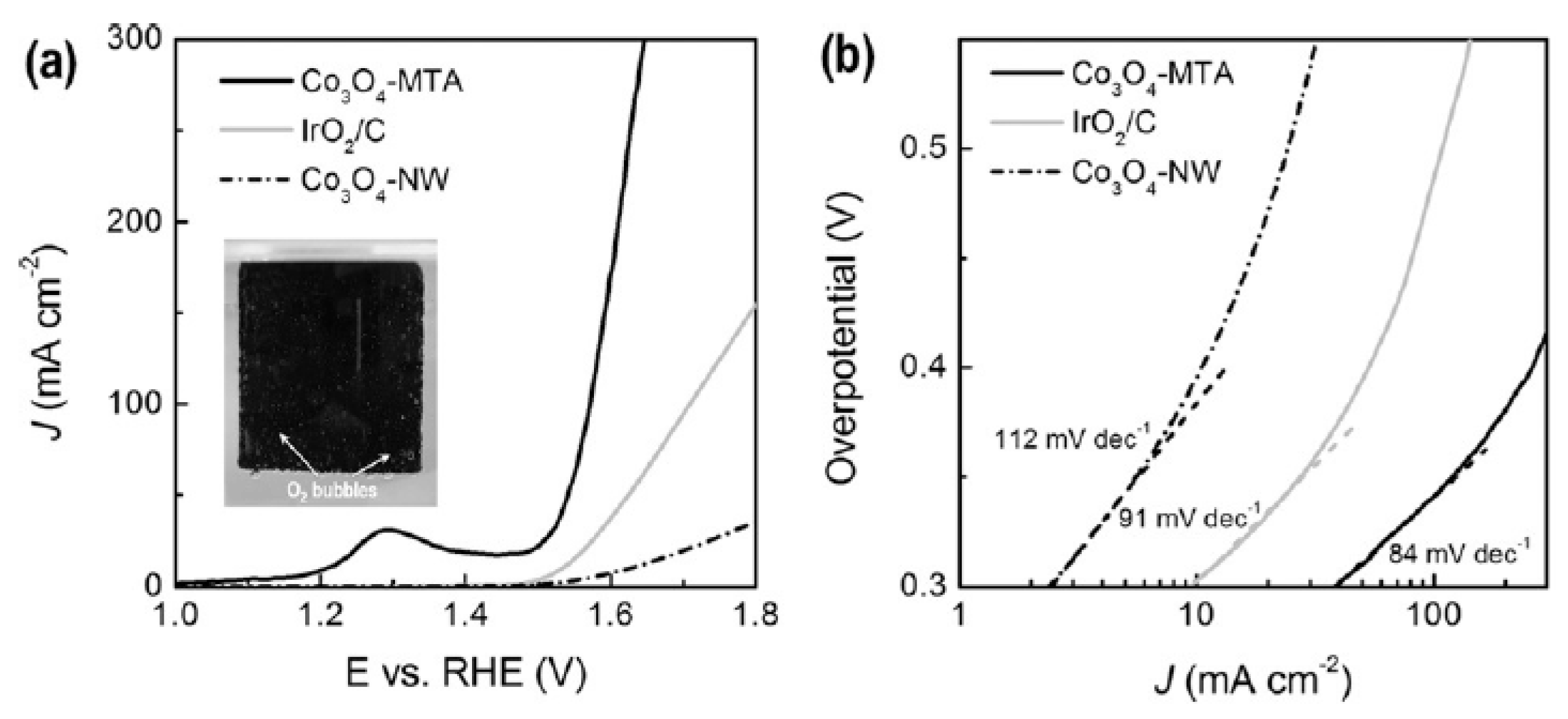
| Co3O4 assembled hollow spheres | Solvothermal & calcination | 1.43 | 0.29 | 86 | 10 | Prominent | 1 M KOH | [46] |
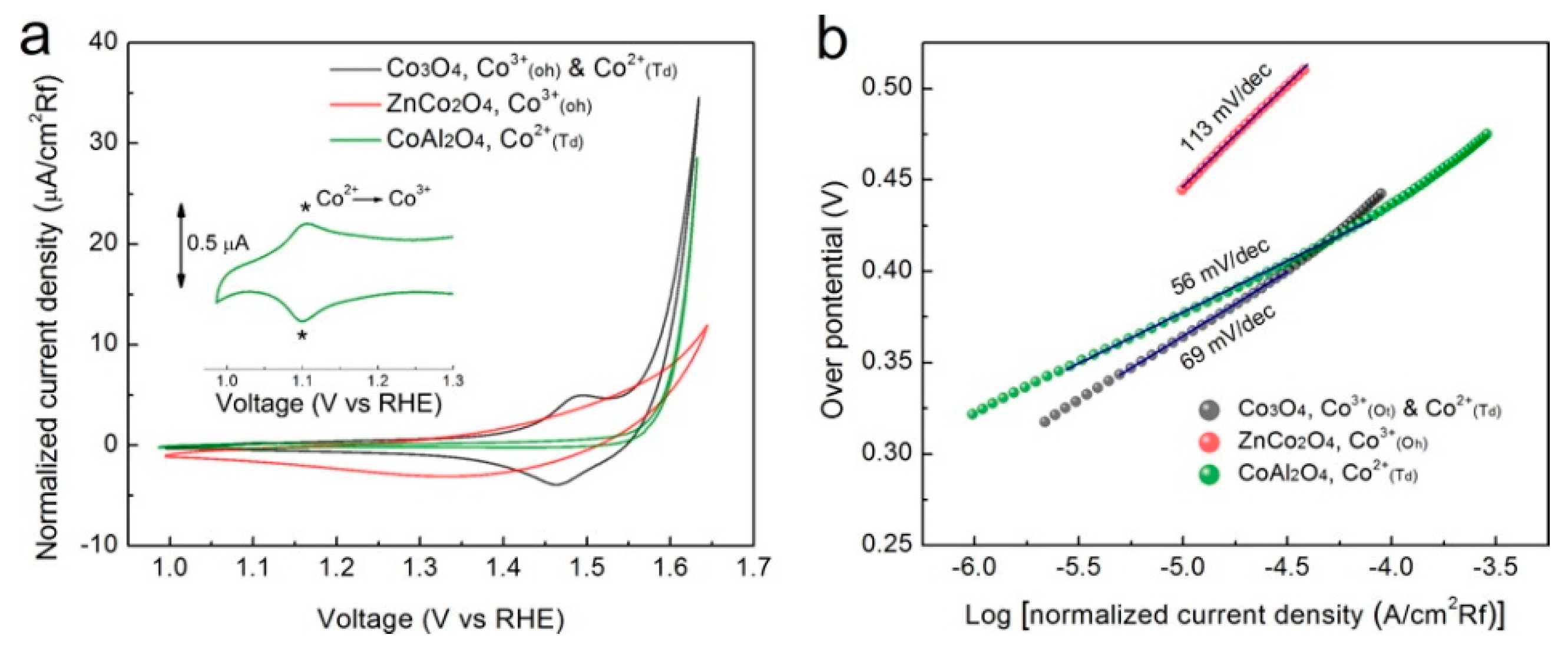
| Co3O4, ZnCo2O4 & CoAl2O4 | Sol-gel | - | - | 69, 113 & 56 | - | - | 0.1 M KOH | [47] |
| Co@Co3O4/N-CNT | Pyrolysis in H2 atm. & oxidative calcination | 1.62 | 0.39 | 54.3 | 45 | Good * | 0.1 M KOH | [51] |
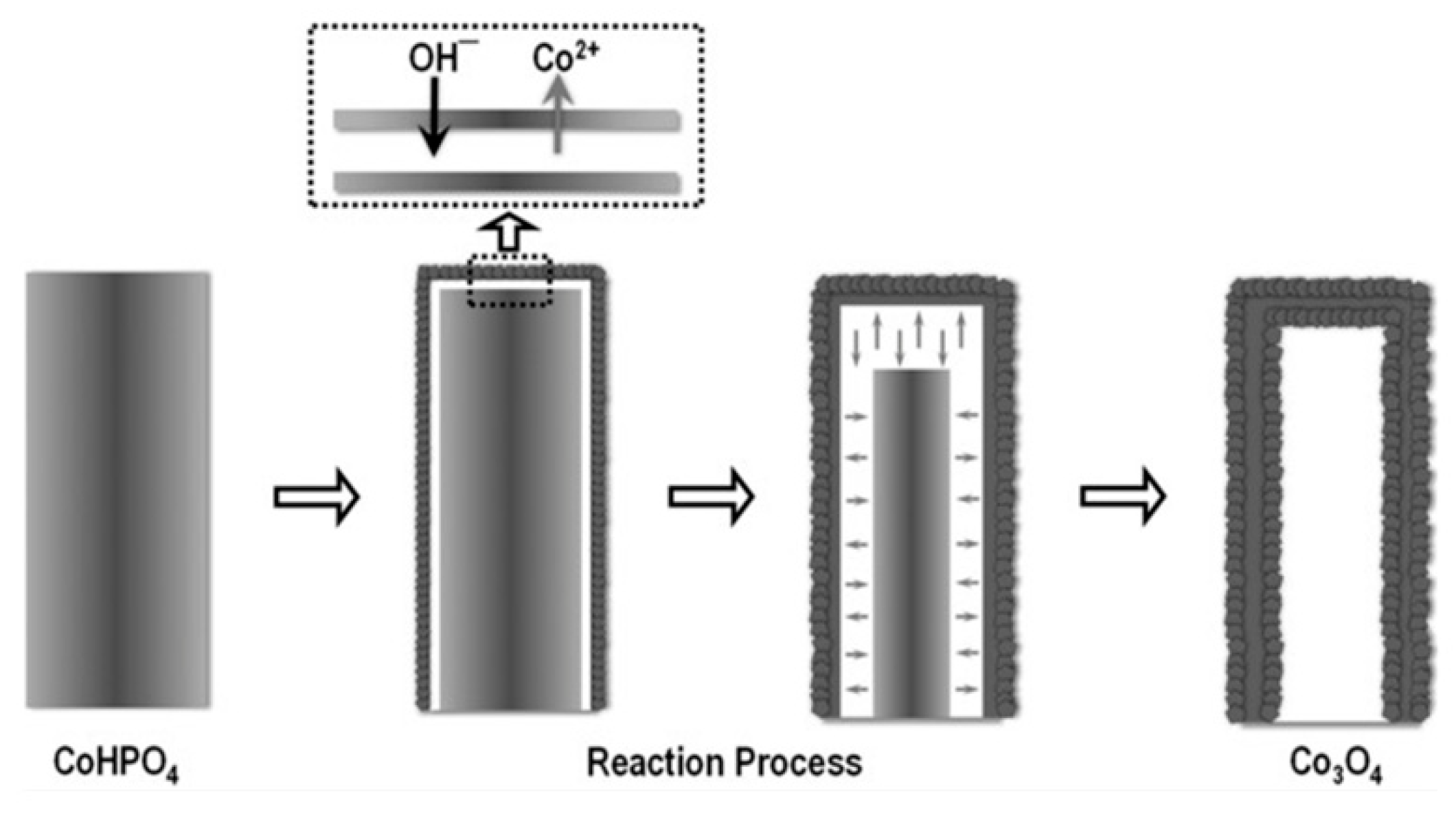
| Hollow Co3O4 microtube arrays | Hydrothermal & electrochemical treatment | 1.52 | 0.151 | 84 | 12 | High | 1 M KOH | [33] |
| ZnCo2O4 QDs/N-CNT | Hydrothermal | 1.56 | 0.43 | 70.6 | 10 | High | 0.1 M KOH | [52] |
| NiFe/Ni Co2O4 | Hydrothermal & electrodeposition | 1.47 | 0.34 | 38.8 | 10 | Excellent | 1 M KOH | [53] |
| NiCo2O4 NPs | Two-step solution method | 1.49 | 0.157 | 75 | 20 | Excellent | 1 M KOH | [54] |
| CoFeOx, CoFeNiOx & FeNiOx | Electrodeposition | 1.43 | 0.24 | 37 | - | - | 1 M KOH | [55] |
| CoFe2O4@Co-Fe-Bi | Hydrothermal | - | 0.46 | 127 | 20 | Good | 0.1 M K2B4O8 | [56] |
| Co3O4 | Hydrothermal | 143 | 0.35 | 69 | - | - | 1 M KOH | [57] |
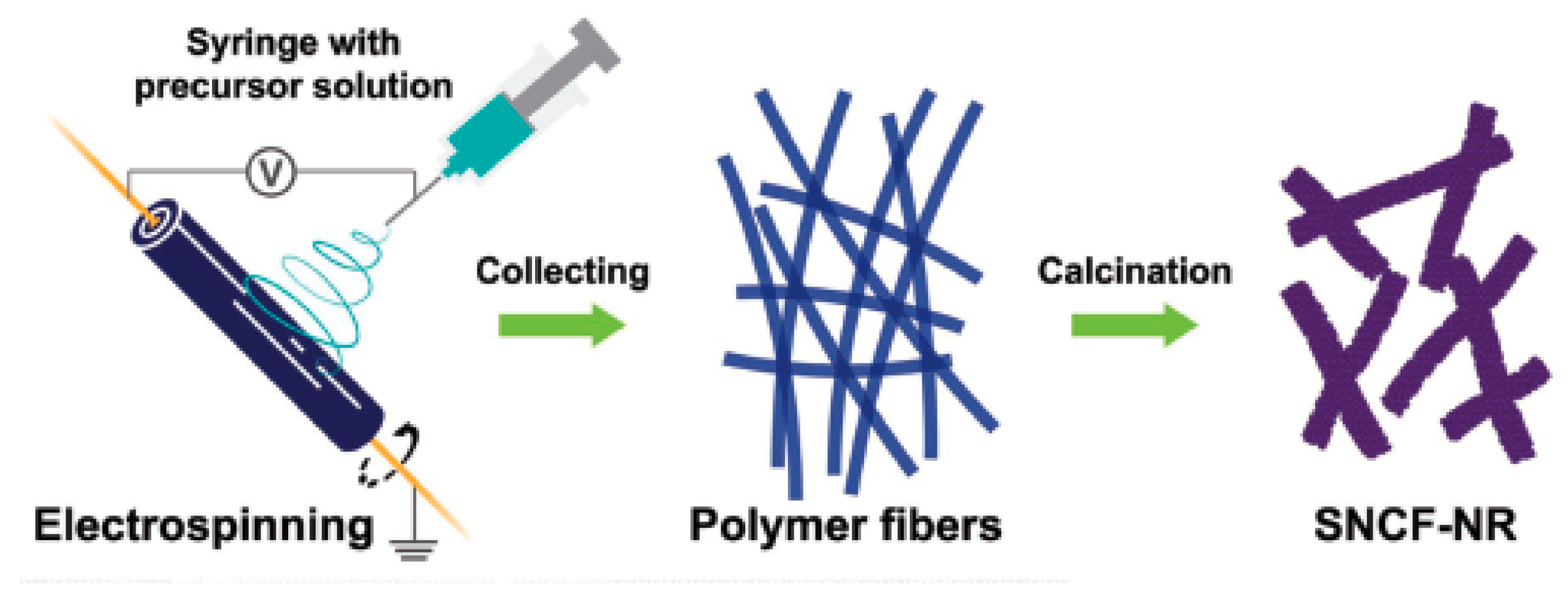
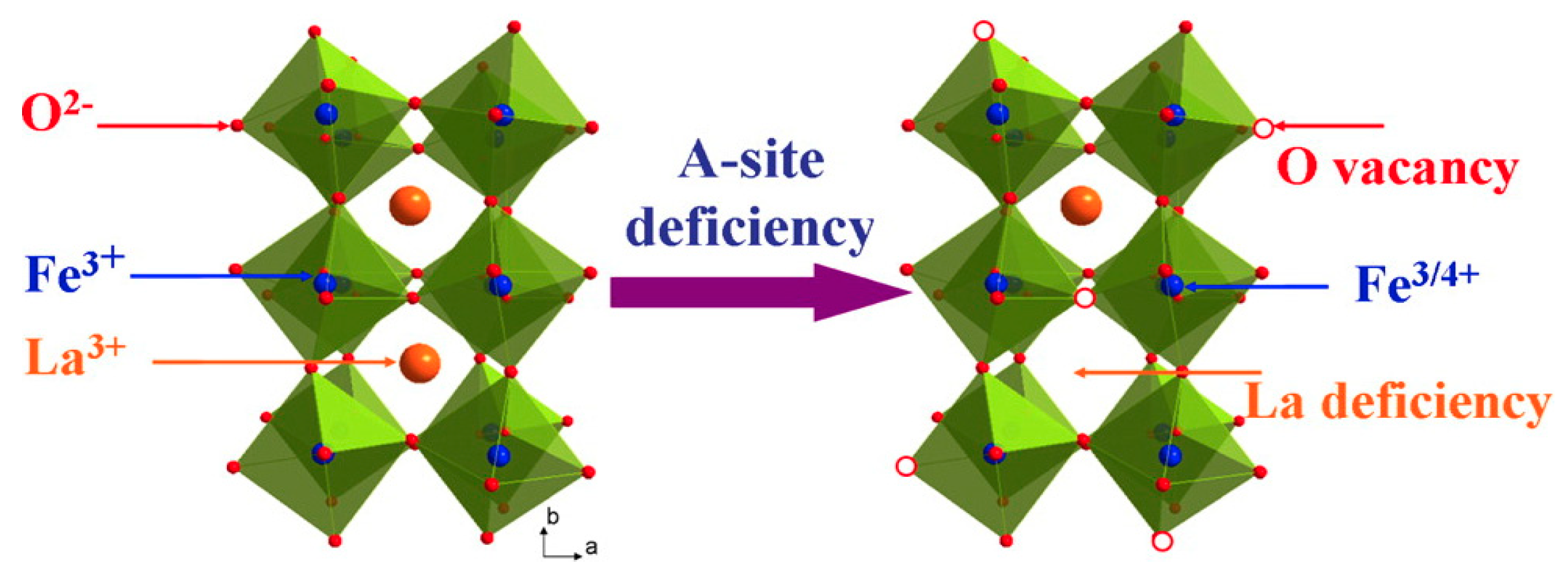
2.2.3. Perovskite-Based WOCs
2.2.4. Mixed Oxides
| Catalyst | Preparation Method | Onset Potential (V) | η (at 10 mV/cm2) (V) | Tafel Slope (mV dec−1) | Durability (h) | Stability | Electrolyte | Ref. |
|---|---|---|---|---|---|---|---|---|
| NiCeOx | Electrodeposition | - | 0.27 | - | >24 | Excellent | 1 M KOH | [68] |
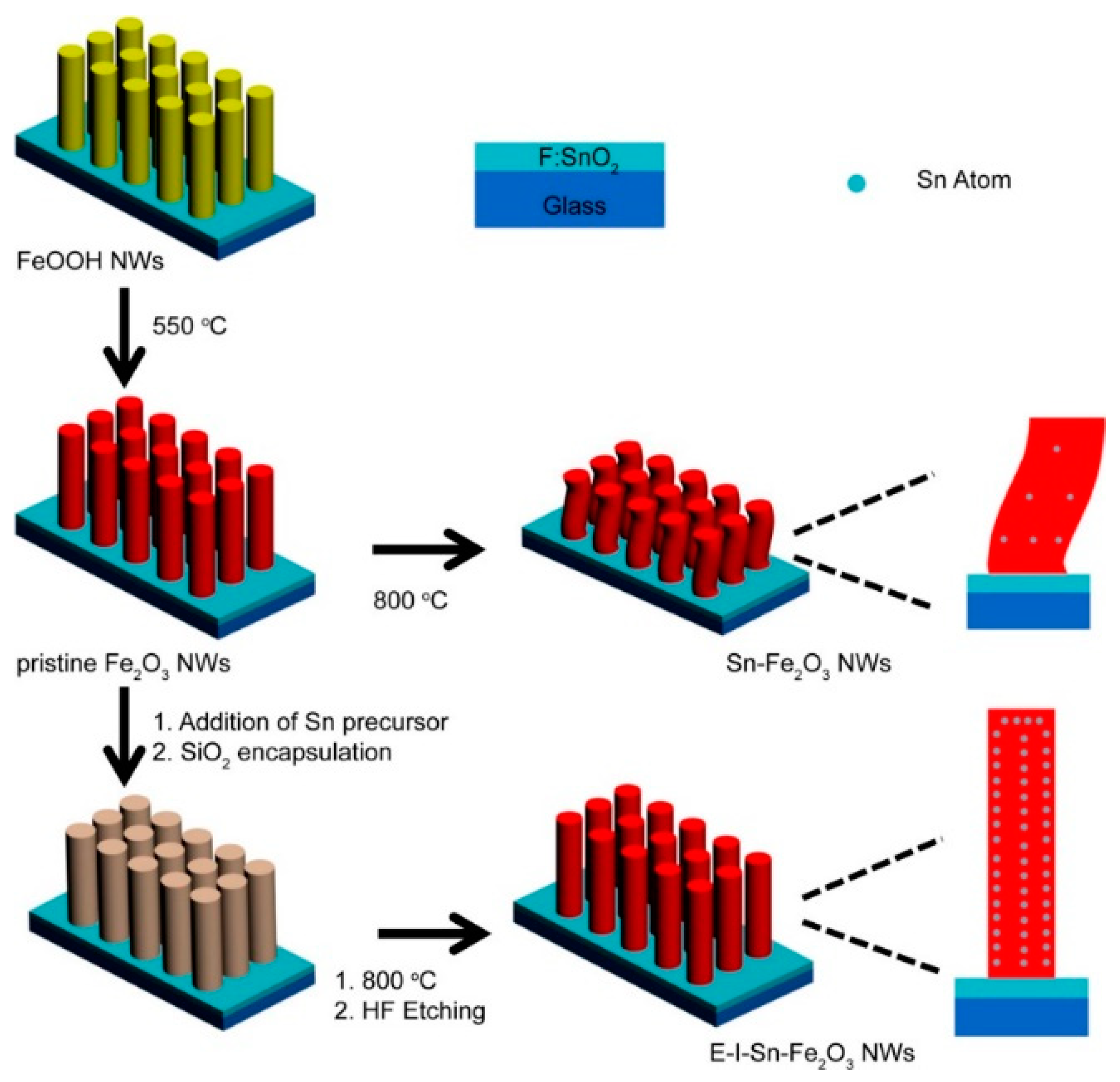
| Sn-Fe2O3 NWs | Hydrothermal | 0.1 | - | - | 10 | Excellent | 1 M KOH | [69] |
| Ni-FeOx & Ni-Fe-CoOx NPs | Impregnation in the presence of aniline | - | 0.2 | - | 1 | Good * | 0.1 M KOH | [70] |
| CoNiOx/rGO | Precipitation | - | 0.28 | 45 | 20 | Advanced | 0.1 M & 1 M KOH | [71] |
| FexCoyO | Precipitation | - | 0.35 | 36.8 | 2.5 | Good | 0.1 M KOH | [72] |
| Fe3O4-Co9S8 NPs/rGO | Two solvothermal steps | 1.48 | 0.34 | 82.8 | 6 | High | 0.1 M KOH | [75] |
| Na1−xNiyFe1−yO2 | Hydrothermal | 1.35 | 0.26 | 44 | 12 | Excellent | 1 M KOH | [76] |
| CoNiO, MnNiO & FeNiO | Precipitation reaction with urea | CoNiO: 1.60, MnNiO: 1.61 & FeNiO: 1.54 | - | CoNiO: 39, MnNiO: 43 & FeNiO: 18 | - | - | 0.1 M KOH | [77] |
| Fe2−xCrxO3 | Coprecipitation | <1.3 | 0.45 | - | 1.5 | Good * | 100 mM PBS | [78] |
| MnVOx@N-rGO | Hydrothermal | 1.32 | 0.39 | 271 | 4 | Excellent | 0.1 M KOH | [79] |
| MnOx/N-CNT | Hydrothermal | ca. 1.4 | 0.36 | 75 | 2 | Catalyst detachment | 1 M KOH | [80] |
| MnxSb1−xOx | Physical Vapor Deposition (PVD) | 1.5 | 0.3 | 75 | 25 | High * | 1 M H2SO4 | [81] |
| Ho2O3/MnOx | Hydrothermal | Varied according to calcination | - | - | 0.5 | High | 0.25 M LiClO4 | [82] |
2.2.5. Other Hydroxides and (Oxy)hydroxides
| Catalyst | Preparation Method | Onset Potential (V) | η (at 10 mV/cm2) (V) | Tafel Slope (mV dec−1) | Durability (h) | Stability | Electrolyte | Ref. |
|---|---|---|---|---|---|---|---|---|
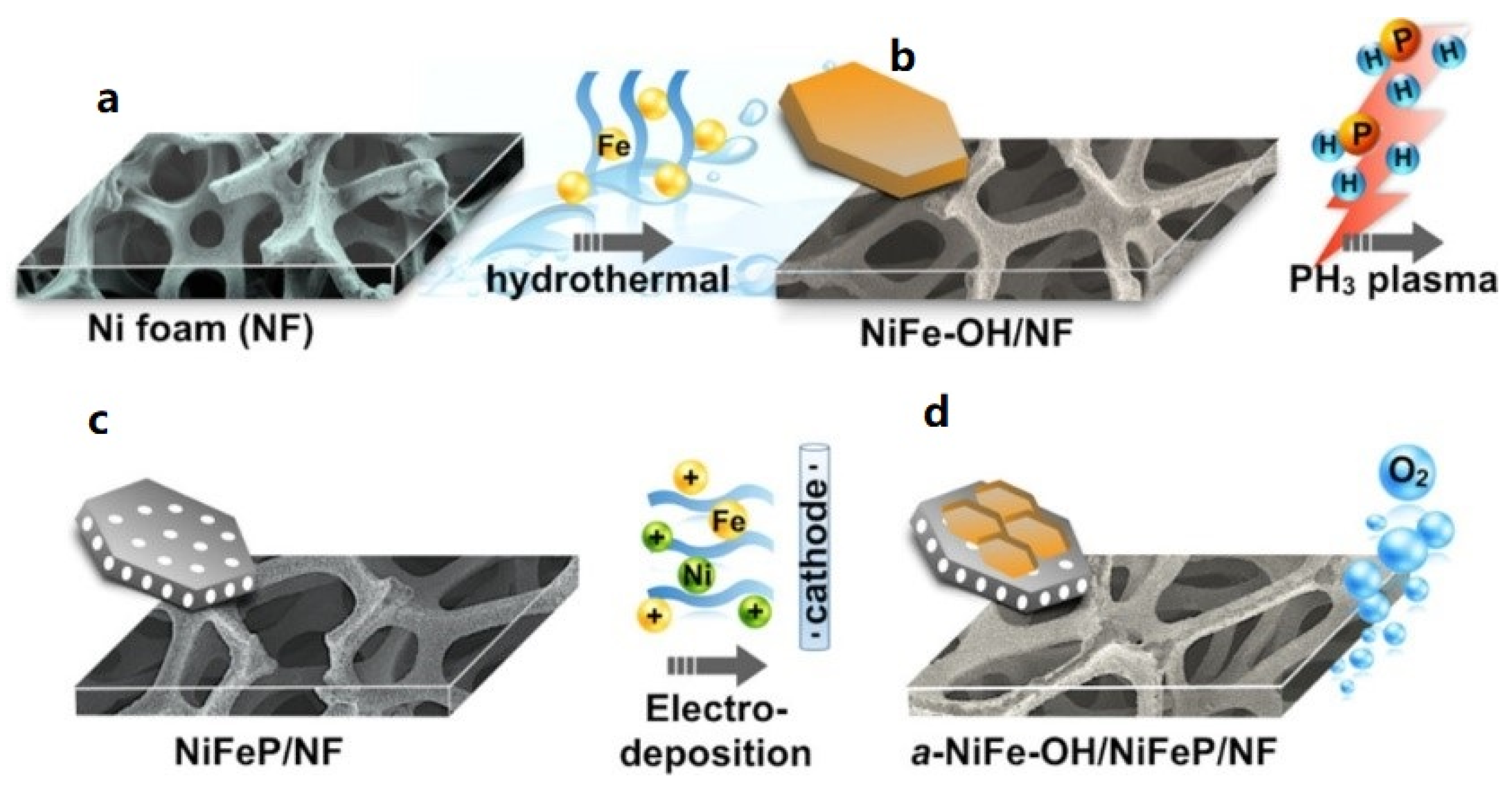
| NiFeOH/NiFeP | Hydrothermal & PH3 plasma treatment & electrodeposition | - | 0.258 | 39 | 24 | High * | 1 M KOH | [85] |
| NiFe-OH | Stepwise electrodeposition | - | 0.24 | 38.9 | 5 | Good * | 1 M KOH | [86] |
| W0.5Co0.4Fe0.1 oxyhydroxide coralloids | Hydrothermal | 1.53 | 0.25 | 32 | >500 | Superior | 1 M KOH | [87] |
| CoFe-H hydroxide & CoFe-H/BiVO4 | Electrodeposition | 0.23 | 0.28 | 28 | 45 | High * | 1 M KOH | [88] |
| CeO2/FeOOH | Electrodeposition | 0.21 | 0.25 | 32 | 50 | High | 1 M NaOH | [89] |
| FeOOH/Co/FeOOH | Electrodeposition | 0.22 | 0.25 | 32 | 50 | High | 1 M NaOH | [90] |
| FeCoW oxyhydroxides | Modified sol-gel | - | 0.191 | 37 | 550 | High | 1 M KOH | [91] |
| NiFe(OH)/NiFe:Pi | Electrodeposition & phosphorylation | 1.43 | 0.29 | 38 | 10 | Excellent | 1 M KOH | [92] |
| Ag-decorated Co(OH)2 | Selective reduction-oxidation | - | 0.27 | 67–109 (depending on the amount of Ag) | 10 | Good | 1 M KOH | [94] |
| NiFeOOH/TiO2 core-shell | Hydrothermal | 0.273 | 86.9 | 24 | High * | 0.01 M Na2SO4 | [95] | |
| α-NiCo(OH)2 | Electrodeposition | 1.43 | 0.26 | 25-30 | 6 | High * | 1 M KOH | [96] |
| NiPS3@NiOOH | Chemical vapor transport & liquid exfoliation | 1.48 | 0.35 | 80 | >160 | Excellent | 0.1 M KOH | [97] |
3. Brief Discussion of the Unresolved Bottlenecks
4. Directions to New Composite Materials
5. Conclusions and Perspectives
Funding
Acknowledgments
Conflicts of Interest
References
- Takanabe, K. Photocatalytic Water Splitting: Quantitative Approaches toward Photocatalyst by Design. ACS Catal. 2017, 7, 8006–8022. [Google Scholar] [CrossRef]
- Roger, I.; Shipman, M.A.; Symes, M.D. Earth-abundant catalysts for electrochemical and photoelectrochemical water splitting. Nat. Rev. Chem. 2017, 1, 3. [Google Scholar] [CrossRef]
- Doyle, R.L.; Lyons, M.E.G. The Oxygen Evolution Reaction: Mechanistic Concepts and Catalyst Design. In Photoelectrochemical Solar Fuel Production; Giménez, S., Bisquert, J., Eds.; Springer International Publishing: Cham, Switzerland, 2016; pp. 41–104. [Google Scholar]
- Turner, J. Sustainable hydrogen production. Science 2004, 305, 973–974. [Google Scholar] [CrossRef] [PubMed]
- Nocera, D.G. The artificial leaf. Acc. Chem. Res. 2012, 45, 767–776. [Google Scholar] [CrossRef] [PubMed]
- Joya, K.S.; Joya, Y.F.; Ocakoglu, K.; van de Krol, R. Water-splitting catalysis and solar fuel devices: Artificial leaves on the move. Angew. Chem. 2013, 52, 10426–10437. [Google Scholar] [CrossRef] [PubMed]
- Si, W.; Pergolesi, D.; Haydous, F.; Fluri, A.; Wokaun, A.; Lippert, T. Investigating the behavior of various cocatalysts on LaTaON2 photoanode for visible light water splitting. Phys. Chem. Chem. Phys. 2016, 19, 656–662. [Google Scholar] [CrossRef] [PubMed]
- Ardo, S.; Fernandez Rivas, D.; Modestino, M.A.; Schulze Greiving, V.; Abdi, F.F.; Alarcon Llado, E.; Artero, V.; Ayers, K.; Battaglia, C.; Becker, J.-P.; et al. Pathways to electrochemical solar-hydrogen technologies. Energy Environ. Sci. 2018, 103, 15729. [Google Scholar] [CrossRef]
- Turan, B.; Becker, J.-P.; Urbain, F.; Finger, F.; Rau, U.; Haas, S. Upscaling of integrated photoelectrochemical water-splitting devices to large areas. Nat. Commun. 2016, 7, 12681. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Güttinger, R.; Moré, R.; Song, F.; Wan, W.; Patzke, G.R. Frontiers of water oxidation: The quest for true catalysts. Chem. Soc. Rev. 2017, 46, 6124–6147. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Wang, L. Photoelectrodes for water splitting: Materials, fabrication and characterization. Sci. China Mater. 2018, 61, 806–821. [Google Scholar] [CrossRef]
- Lee, Y.; Suntivich, J.; May, K.J.; Perry, E.E.; Shao-Horn, Y. Synthesis and Activities of Rutile IrO2 and RuO2 Nanoparticles for Oxygen Evolution in Acid and Alkaline Solutions. J. Phys. Chem. Lett. 2012, 3, 399–404. [Google Scholar] [CrossRef] [PubMed]
- Walter, M.G.; Warren, E.L.; McKone, J.R.; Boettcher, S.W.; Mi, Q.; Santori, E.A.; Lewis, N.S. Solar water splitting cells. Chem. Rev. 2010, 110, 6446–6473. [Google Scholar] [CrossRef] [PubMed]
- Védrine, J. Heterogeneous Catalysis on Metal Oxides. Catalysts 2017, 7, 341. [Google Scholar] [CrossRef]
- Weber, D.; Schoop, L.M.; Wurmbrand, D.; Laha, S.; Duppel, V.; Podjaski, F.; Mueller, K.; Starke, U.; Lotsch, B.V. IrOOH nanosheets as acid stable electrocatalysts for the oxygen evolution reaction. J. Mater. Chem. A 2018. [Google Scholar] [CrossRef]
- Seitz, L.C.; Dickens, C.F.; Nishio, K.; Hikita, Y.; Montoya, J.; Doyle, A.; Kirk, C.; Vojvodic, A.; Hwang, H.Y.; Norskov, J.K.; et al. A highly active and stable IrOx/SrIrO3 catalyst for the oxygen evolution reaction. Science 2016, 353, 1011–1014. [Google Scholar] [CrossRef] [PubMed]
- Deng, Y.; Handoko, A.D.; Du, Y.; Xi, S.; Yeo, B.S. In Situ Raman Spectroscopy of Copper and Copper Oxide Surfaces during Electrochemical Oxygen Evolution Reaction: Identification of Cu(III) Oxides as Catalytically Active Species. ACS Catal. 2016, 6, 2473–2481. [Google Scholar] [CrossRef]
- Xie, L.; Tang, C.; Wang, K.; Du, G.; Asiri, A.M.; Sun, X. Cu(OH)2@CoCO3(OH)2·nH2O Core–Shell Heterostructure Nanowire Array: An Efficient 3D Anodic Catalyst for Oxygen Evolution and Methanol Electrooxidation. Small 2017, 13. [Google Scholar] [CrossRef] [PubMed]
- Cherevko, S.; Geiger, S.; Kasian, O.; Kulyk, N.; Grote, J.-P.; Savan, A.; Shrestha, B.R.; Merzlikin, S.; Breitbach, B.; Ludwig, A.; et al. Oxygen and hydrogen evolution reactions on Ru, RuO2, Ir, and IrO2 thin film electrodes in acidic and alkaline electrolytes: A comparative study on activity and stability. Catal. Today 2016, 262, 170–180. [Google Scholar] [CrossRef]
- Amiri, M.; Fallahi, M.; Bezaatpour, A.; Jijie, R.; Nozari-asbmarz, M.; Rouhi, M.; Boukherroub, R.; Szunerits, S. Solution Processable Cu(II) macrocycle for the Formation of Cu2O Thin Film on Indium Tin Oxide and Its Application for Water Oxidation. J. Phys. Chem. C 2018, 122, 16510–16518. [Google Scholar] [CrossRef]
- Roy, C.; Rao, R.R.; Stoerzinger, K.A.; Hwang, J.; Rossmeisl, J.; Chorkendorff, I.; Shao-Horn, Y.; Stephens, I.E.L. Trends in Activity and Dissolution on RuO2 under Oxygen Evolution Conditions: Particles versus Well-Defined Extended Surfaces. ACS Energy Lett. 2018, 3, 2045–2051. [Google Scholar] [CrossRef]
- Oh, H.-S.; Nong, H.N.; Reier, T.; Bergmann, A.; Gliech, M.; Ferreira de Araújo, J.; Willinger, E.; Schlögl, R.; Teschner, D.; Strasser, P. Electrochemical Catalyst-Support Effects and Their Stabilizing Role for IrOx Nanoparticle Catalysts during the Oxygen Evolution Reaction. J. Am. Chem. Soc. 2016, 138, 12552–12563. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.T.; Liu, N. Visualising Battery Reactions and Processes by Using In Situ and In Operando Microscopies. Chem 2018, 4, 438–465. [Google Scholar] [CrossRef]
- Zhang, M.; Frei, H. Water Oxidation Mechanisms of Metal Oxide Catalysts by Vibrational Spectroscopy of Transient Intermediates. Annu. Rev. Phys. Chem. 2017, 68, 209–231. [Google Scholar] [CrossRef] [PubMed]
- Conway, B.E.; Bai, L.; Sattar, M.A. Role of the transfer coefficient in electrocatalysis: Applications to the H2 and O2 evolution reactions and the characterization of participating adsorbed intermediates. Int. J. Hydrogen Energy 1987, 12, 607–621. [Google Scholar] [CrossRef]
- Atabaev, T.S.; Lee, D.H.; Hong, N.H. Fabrication of TiO2/CuO photoelectrode with enhanced solar water splitting activity. Funct. Mater. Lett. 2017, 10, 1750084. [Google Scholar] [CrossRef]
- Mishra, G.; Dash, B.; Pandey, S. Layered double hydroxides: A brief review from fundamentals to application as evolving biomaterials. Appl. Clay Sci. 2018, 153, 172–186. [Google Scholar] [CrossRef]
- Han, L.; Dong, S.; Wang, E. Transition-Metal (Co, Ni, and Fe)-Based Electrocatalysts for the Water Oxidation Reaction. Adv. Mater. 2016, 28, 9266–9291. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Ren, Z.; Du, S.; Kong, L.; Liu, B.; Xi, W.; Zhu, J.; Fu, H. A highly active oxygen evolution electrocatalyst: Ultrathin CoNi double hydroxide/CoO nanosheets synthesized via interface-directed assembly. Nano Res. 2016, 9, 713–725. [Google Scholar] [CrossRef]
- Yang, F.; Sliozberg, K.; Sinev, I.; Antoni, H.; Bähr, A.; Ollegott, K.; Xia, W.; Masa, J.; Grünert, W.; Cuenya, B.R.; et al. Synergistic Effect of Cobalt and Iron in Layered Double Hydroxide Catalysts for the Oxygen Evolution Reaction. ChemSusChem 2017, 10, 156–165. [Google Scholar] [CrossRef] [PubMed]
- You, C.; Ji, Y.; Liu, Z.; Xiong, X.; Sun, X. Ultrathin CoFe-Borate Layer Coated CoFe-Layered Double Hydroxide Nanosheets Array: A Non-Noble-Metal 3D Catalyst Electrode for Efficient and Durable Water Oxidation in Potassium Borate. ACS Sustain. Chem. Eng. 2018, 6, 1527–1531. [Google Scholar] [CrossRef]
- Fan, K.; Chen, H.; Ji, Y.; Huang, H.; Claesson, P.M.; Daniel, Q.; Philippe, B.; Rensmo, H.; Li, F.; Luo, Y.; et al. Nickel-vanadium monolayer double hydroxide for efficient electrochemical water oxidation. Nat. Commun. 2016, 7, 11981. [Google Scholar] [CrossRef] [PubMed]
- Ping, J.; Wang, Y.; Lu, Q.; Chen, B.; Chen, J.; Huang, Y.; Ma, Q.; Tan, C.; Yang, J.; Cao, X.; et al. Self-Assembly of Single-Layer CoAl-Layered Double Hydroxide Nanosheets on 3D Graphene Network Used as Highly Efficient Electrocatalyst for Oxygen Evolution Reaction. Adv. Mater. 2016, 28, 7640–7645. [Google Scholar] [CrossRef] [PubMed]
- Hunter, B.M.; Winkler, J.R.; Gray, H.B. Iron Is the Active Site in Nickel/Iron Water Oxidation Electrocatalysts. Molecules 2018, 23, 903. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Moghaddam, R.B.; Brett, M.J.; Bergens, S.H. Simple Aqueous Preparation of High Activity and Stability NiFe Hydrous Oxide Catalysts for Water Oxidation. ACS Sustain. Chem. Eng. 2017, 5, 1106–1112. [Google Scholar] [CrossRef]
- Li, N.; Bediako, D.K.; Hadt, R.G.; Hayes, D.; Kempa, T.J.; von Cube, F.; Bell, D.C.; Chen, L.X.; Nocera, D.G. Influence of iron doping on tetravalent nickel content in catalytic oxygen evolving films. Proc. Natl. Acad. Sci. USA 2017, 114, 1486–1491. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Shao, M.; An, H.; Wang, Z.; Xu, S.; Wei, M.; Evans, D.G.; Duan, X. Fast electrosynthesis of Fe-containing layered double hydroxide arrays toward highly efficient electrocatalytic oxidation reactions. Chem. Sci. 2015, 6, 6624–6631. [Google Scholar] [CrossRef] [PubMed]
- Xie, C.; Wang, Y.; Hu, K.; Tao, L.; Huang, X.; Huo, J.; Wang, S. In situ confined synthesis of molybdenum oxide decorated nickel–iron alloy nanosheets from MoO42− intercalated layered double hydroxides for the oxygen evolution reaction. J. Mater. Chem. A 2017, 5, 87–91. [Google Scholar] [CrossRef]
- Görlin, M.; Ferreira de Araújo, J.; Schmies, H.; Bernsmeier, D.; Dresp, S.; Gliech, M.; Jusys, Z.; Chernev, P.; Kraehnert, R.; Dau, H.; et al. Tracking Catalyst Redox States and Reaction Dynamics in Ni–Fe Oxyhydroxide Oxygen Evolution Reaction Electrocatalysts: The Role of Catalyst Support and Electrolyte pH. J. Am. Chem. Soc. 2017, 139, 2070–2082. [Google Scholar] [CrossRef] [PubMed]
- Zhang, F.; Shi, Y.; Xue, T.; Zhang, J.; Liang, Y.; Zhang, B. In situ electrochemically converting Fe2O3-Ni(OH)2 to NiFe2O4–NiOOH: A highly efficient electrocatalyst towards water oxidation. Sci. China Mater. 2017, 60, 324–334. [Google Scholar] [CrossRef]
- Zou, X.; Liu, Y.; Li, G.-D.; Wu, Y.; Liu, D.-P.; Li, W.; Li, H.-W.; Wang, D.; Zhang, Y.; Zou, X. Ultrafast Formation of Amorphous Bimetallic Hydroxide Films on 3D Conductive Sulfide Nanoarrays for Large-Current-Density Oxygen Evolution Electrocatalysis. Adv. Mater. 2017, 29. [Google Scholar] [CrossRef] [PubMed]
- Wu, Z.; Wang, Z.; Geng, F. Radially Aligned Hierarchical Nickel/Nickel-Iron (Oxy)hydroxide Nanotubes for Efficient Electrocatalytic Water Splitting. ACS Appl. Mater. Interfaces 2018, 10, 8585–8593. [Google Scholar] [CrossRef] [PubMed]
- Hunter, B.M.; Hieringer, W.; Winkler, J.R.; Gray, H.B.; Müller, A.M. Effect of interlayer anions on [NiFe]-LDH nanosheet water oxidation activity. Energy Environ. Sci. 2016, 9, 1734–1743. [Google Scholar] [CrossRef]
- Jia, Y.; Zhang, L.; Gao, G.; Chen, H.; Wang, B.; Zhou, J.; Soo, M.T.; Hong, M.; Yan, X.; Qian, G.; et al. A Heterostructure Coupling of Exfoliated Ni–Fe Hydroxide Nanosheet and Defective Graphene as a Bifunctional Electrocatalyst for Overall Water Splitting. Adv. Mater. 2017, 29. [Google Scholar] [CrossRef] [PubMed]
- Lutterman, D.A.; Surendranath, Y.; Nocera, D.G. A self-healing oxygen-evolving catalyst. J. Am. Chem. Soc. 2009, 131, 3838–3839. [Google Scholar] [CrossRef] [PubMed]
- Xie, T.; Min, J.; Liu, J.; Chen, J.; Fu, D.; Zhang, R.; Zhu, K.; Lei, M. Synthesis of mesoporous Co3O4 nanosheet-assembled hollow spheres towards efficient electrocatalytic oxygen evolution. J. Alloy. Compd. 2018, 754, 72–77. [Google Scholar] [CrossRef]
- Wang, H.-Y.; Hung, S.-F.; Chen, H.-Y.; Chan, T.-S.; Chen, H.M.; Liu, B. In Operando Identification of Geometrical-Site-Dependent Water Oxidation Activity of Spinel Co3O4. J. Am. Chem. Soc. 2016, 138, 36–39. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.P.; Ma, T.Y.; Jaroniec, M.; Qiao, S.Z. Self-Templating Synthesis of Hollow Co3O4 Microtube Arrays for Highly Efficient Water Electrolysis. Angew. Chem. 2017, 56, 1324–1328. [Google Scholar] [CrossRef] [PubMed]
- Koroidov, S.; Anderlund, M.F.; Styring, S.; Thapper, A.; Messinger, J. First turnover analysis of water-oxidation catalyzed by Co-oxide nanoparticles. Energy Environ. Sci. 2015, 8, 2492–2503. [Google Scholar] [CrossRef]
- Wang, Q.; Hu, W.; Huang, Y. Nitrogen doped graphene anchored cobalt oxides efficiently bi-functionally catalyze both oxygen reduction reaction and oxygen revolution reaction. Int. J. Hydrogen Energy 2017, 42, 5899–5907. [Google Scholar] [CrossRef]
- Aijaz, A.; Masa, J.; Rösler, C.; Xia, W.; Weide, P.; Botz, A.J.R.; Fischer, R.A.; Schuhmann, W.; Muhler, M. Co@Co3O4 Encapsulated in Carbon Nanotube-Grafted Nitrogen-Doped Carbon Polyhedra as an Advanced Bifunctional Oxygen Electrode. Angew. Chem. 2016, 55, 4087–4091. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.-Q.; Cheng, H.; Li, N.; Ma, T.Y.; Su, Y.-Z. ZnCo2O4 Quantum Dots Anchored on Nitrogen-Doped Carbon Nanotubes as Reversible Oxygen Reduction/Evolution Electrocatalysts. Adv. Mater. 2016, 28, 3777–3784. [Google Scholar] [CrossRef] [PubMed]
- Xiao, C.; Li, Y.; Lu, X.; Zhao, C. Bifunctional Porous NiFe/NiCo2O4/Ni Foam Electrodes with Triple Hierarchy and Double Synergies for Efficient Whole Cell Water Splitting. Adv. Funct. Mater. 2016, 26, 3515–3523. [Google Scholar] [CrossRef]
- Guan, C.; Liu, X.; Ren, W.; Li, X.; Cheng, C.; Wang, J. Rational Design of Metal–Organic Framework Derived Hollow NiCo2O4 Arrays for Flexible Supercapacitor and Electrocatalysis. Adv. Energy Mater. 2017, 7, 1602391. [Google Scholar] [CrossRef]
- Morales-Guio, C.G.; Liardet, L.; Hu, X. Oxidatively Electrodeposited Thin-Film Transition Metal (Oxy)hydroxides as Oxygen Evolution Catalysts. J. Am. Chem. Soc. 2016, 138, 8946–8957. [Google Scholar] [CrossRef] [PubMed]
- Ji, X.; Hao, S.; Qu, F.; Liu, J.; Du, G.; Asiri, A.M.; Chen, L.; Sun, X. Core–shell CoFe2O4@Co-Fe-Bi nanoarray: A surface-amorphization water oxidation catalyst operating at near-neutral pH. Nanoscale 2017, 9, 7714–7718. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Chen, Y.-S.; Kamat, P.V.; Ptasinska, S. Probing Interfacial Electrochemistry on a Co3O4 Water Oxidation Catalyst Using Lab-Based Ambient Pressure X-ray Photoelectron Spectroscopy. J. Phys. Chem. C 2018, 122, 13894–13901. [Google Scholar] [CrossRef]
- Reith, L.; Lienau, K.; Cook, D.S.; Moré, R.; Walton, R.I.; Patzke, G.R. Monitoring the Hydrothermal Growth of Cobalt Spinel Water Oxidation Catalysts: From Preparative History to Catalytic Activity. Chemistry 2018. [Google Scholar] [CrossRef]
- Matsumoto, Y.; Yamada, S.; Nishida, T.; Sato, E. Oxygen evolution on La1−xSrxFe1−yCoyO3 series oxides. J. Electrochem. Sci. 1980, 127, 2360–2364. [Google Scholar] [CrossRef]
- Zhu, Y.; Zhou, W.; Yu, J.; Chen, Y.; Liu, M.; Shao, Z. Enhancing Electrocatalytic Activity of Perovskite Oxides by Tuning Cation Deficiency for Oxygen Reduction and Evolution Reactions. Chem. Mater. 2016, 28, 1691–1697. [Google Scholar] [CrossRef]
- Zhao, B.; Zhang, L.; Zhen, D.; Yoo, S.; Ding, Y.; Chen, D.; Chen, Y.; Zhang, Q.; Doyle, B.; Xiong, X.; et al. A tailored double perovskite nanofiber catalyst enables ultrafast oxygen evolution. Nat. Commun. 2017, 8, 14586. [Google Scholar] [CrossRef] [PubMed]
- Su, C.; Duan, X.; Miao, J.; Zhong, Y.; Zhou, W.; Wang, S.; Shao, Z. Mixed Conducting Perovskite Materials as Superior Catalysts for Fast Aqueous-Phase Advanced Oxidation: A Mechanistic Study. ACS Catal. 2017, 7, 388–397. [Google Scholar] [CrossRef]
- Xu, X.; Su, C.; Zhou, W.; Zhu, Y.; Chen, Y.; Shao, Z. Co-doping Strategy for Developing Perovskite Oxides as Highly Efficient Electrocatalysts for Oxygen Evolution Reaction. Adv. Sci. 2016, 3, 1500187. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Zhou, W.; Zhong, Y.; Bu, Y.; Chen, X.; Zhong, Q.; Liu, M.; Shao, Z. A Perovskite Nanorod as Bifunctional Electrocatalyst for Overall Water Splitting. Adv. Energy Mater. 2017, 7, 1602122. [Google Scholar] [CrossRef]
- Mefford, J.T.; Rong, X.; Abakumov, A.M.; Hardin, W.G.; Dai, S.; Kolpak, A.M.; Johnston, K.P.; Stevenson, K.J. Water electrolysis on La1−xSrxCoO3−δ perovskite electrocatalysts. Nat. Commun. 2016, 7, 11053. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.G.; Hwang, J.; Hwang, H.J.; Jeon, O.S.; Jang, J.; Kwon, O.; Lee, Y.; Han, B.; Shul, Y.-G. A New Family of Perovskite Catalysts for Oxygen-Evolution Reaction in Alkaline Media: BaNiO3 and BaNi0.83O2.5. J. Am. Chem. Soc. 2016, 138, 3541–3547. [Google Scholar] [CrossRef] [PubMed]
- Black, A.P.; Suzuki, H.; Higashi, M.; Frontera, C.; Ritter, C.; De, C.; Sundaresan, A.; Abe, R.; Fuertes, A. New rare earth hafnium oxynitride perovskites with photocatalytic activity in water oxidation and reduction. Chem. Commun. 2018, 54, 1525–1528. [Google Scholar] [CrossRef] [PubMed]
- Ng, J.W.D.; García-Melchor, M.; Bajdich, M.; Chakthranont, P.; Kirk, C.; Vojvodic, A.; Jaramillo, T.F. Gold-supported cerium-doped NiOx catalysts for water oxidation. Nat. Energy 2016, 1, 16053. [Google Scholar] [CrossRef]
- Li, M.; Yang, Y.; Ling, Y.; Qiu, W.; Wang, F.; Liu, T.; Song, Y.; Liu, X.; Fang, P.; Tong, Y.; et al. Morphology and Doping Engineering of Sn-Doped Hematite Nanowire Photoanodes. Nano Lett. 2017, 17, 2490–2495. [Google Scholar] [CrossRef] [PubMed]
- Bates, M.K.; Jia, Q.; Doan, H.; Liang, W.; Mukerjee, S. Charge-Transfer Effects in Ni–Fe and Ni–Fe–Co Mixed-Metal Oxides for the Alkaline Oxygen Evolution Reaction. ACS Catal. 2016, 6, 155–161. [Google Scholar] [CrossRef]
- Li, P.; Zeng, H.C. Sandwich-Like Nanocomposite of CoNiOx/Reduced Graphene Oxide for Enhanced Electrocatalytic Water Oxidation. Adv. Funct. Mater. 2017, 27, 1606325. [Google Scholar] [CrossRef]
- Zhuang, L.; Ge, L.; Yang, Y.; Li, M.; Jia, Y.; Yao, X.; Zhu, Z. Ultrathin Iron–Cobalt Oxide Nanosheets with Abundant Oxygen Vacancies for the Oxygen Evolution Reaction. Adv. Mater. 2017, 29. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Sheehan, S.W.; He, D.; He, Y.; Yao, X.; Grimm, R.L.; Brudvig, G.W.; Wang, D. Hematite-Based Solar Water Splitting in Acidic Solutions: Functionalization by Mono- and Multilayers of Iridium Oxygen-Evolution Catalysts. Angew. Chem. 2015, 54, 11428–11432. [Google Scholar] [CrossRef] [PubMed]
- Gholamrezaei, S.; Salavati-Niasari, M. Sonochemical synthesis of SrMnO3 nanoparticles as an efficient and new catalyst for O2 evolution from water splitting reaction. Ultrason. Sonochem. 2018, 40, 651–663. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Zhu, G.; Liu, Y.; Xia, J.; Ji, Z.; Shen, X.; Wu, S. Fe3O4-Decorated Co9S8 Nanoparticles In Situ Grown on Reduced Graphene Oxide: A New and Efficient Electrocatalyst for Oxygen Evolution Reaction. Adv. Funct. Mater. 2016, 26, 4712–4721. [Google Scholar] [CrossRef]
- Weng, B.; Xu, F.; Wang, C.; Meng, W.; Grice, C.R.; Yan, Y. A layered Na1−xNiyFe1−yO2 double oxide oxygen evolution reaction electrocatalyst for highly efficient water-splitting. Energy Environ. Sci. 2017, 10, 121–128. [Google Scholar] [CrossRef]
- Abbott, D.F.; Meier, M.; Meseck, G.R.; Fabbri, E.; Seeger, S.; Schmidt, T.J. Silicone Nanofilament-Supported Mixed Nickel-Metal Oxides for Alkaline Water Electrolysis. J. Electrochem. Soc. 2017, 164, F203–F208. [Google Scholar] [CrossRef]
- Kanazawa, T.; Maeda, K. Chromium-substituted hematite powder as a catalytic material for photochemical and electrochemical water oxidation. Catal. Sci. Technol. 2017, 7, 2940–2946. [Google Scholar] [CrossRef]
- Xing, X.; Liu, R.; Cao, K.; Kaiser, U.; Zhang, G.; Streb, C. Manganese vanadium oxide-N-doped reduced graphene oxide composites as oxygen reduction and oxygen evolution electrocatalysts. ACS Appl. Mater. Interfaces 2018. [Google Scholar] [CrossRef] [PubMed]
- Antoni, H.; Morales, D.M.; Fu, Q.; Chen, Y.-T.; Masa, J.; Schuhmann, W.; Muhler, M. Oxidative Deposition of Manganese Oxide Nanosheets on Nitrogen-Functionalized Carbon Nanotubes Applied in the Alkaline Oxygen Evolution Reaction. ACS Omega 2018, 3, 11216–11226. [Google Scholar] [CrossRef]
- Zhou, L.; Shinde, A.; Montoya, H.J.; Singh, A.; Gul, S.; Tano, J.; Ye, Y.; Crumlin, J.E.; Richter, M.H.; Cooper, J.K.; et al. Rutile alloys in the Mn–Sb–O system stabilize Mn3+ for OER in strong acid. ACS Catal. 2018, 8, 10938–10948. [Google Scholar] [CrossRef]
- Najafpour, M.M.; Salimi, S.; Safdari, R. Nanosized manganese oxide supported on carbon black: A new, cheap and green composite for water oxidation. Int. J. Hydrogen Energy 2017, 42, 255–264. [Google Scholar] [CrossRef]
- Najafpour, M.M.; Hołyńska, M.; Salimi, S. Applications of the “nano to bulk” Mn oxides: Mn oxide as a Swiss army knife. Coord. Chem. Rev. 2015, 285, 65–75. [Google Scholar] [CrossRef]
- Morgan Chan, Z.; Kitchaev, D.A.; Nelson Weker, J.; Schnedermann, C.; Lim, K.; Ceder, G.; Tumas, W.; Toney, M.F.; Nocera, D.G. Electrochemical trapping of metastable Mn3+ ions for activation of MnO2 oxygen evolution catalysts. Proc. Natl. Acad. Sci. USA 2018, 115, E5261–E5268. [Google Scholar] [CrossRef] [PubMed]
- Liang, H.; Gandi, A.N.; Xia, C.; Hedhili, M.N.; Anjum, D.H.; Schwingenschlögl, U.; Alshareef, H.N. Amorphous NiFe-OH/NiFeP Electrocatalyst Fabricated at Low Temperature for Water Oxidation Applications. ACS Energy Lett. 2017, 2, 1035–1042. [Google Scholar] [CrossRef]
- Zhang, W.; Wu, Y.; Qi, J.; Chen, M.; Cao, R. A Thin NiFe Hydroxide Film Formed by Stepwise Electrodeposition Strategy with Significantly Improved Catalytic Water Oxidation Efficiency. Adv. Energy Mater. 2017, 7, 1602547. [Google Scholar] [CrossRef]
- Pi, Y.; Shao, Q.; Wang, P.; Lv, F.; Guo, S.; Guo, J.; Huang, X. Trimetallic Oxyhydroxide Coralloids for Efficient Oxygen Evolution Electrocatalysis. Angew. Chem. 2017, 56, 4502–4506. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Liu, H.; Dang, L.; Zhang, H.; Wu, X.; Yang, B.; Li, Z.; Zhang, X.; Lei, L.; Jin, S. Amorphous Cobalt-Iron Hydroxide Nanosheet Electrocatalyst for Efficient Electrochemical and Photo-Electrochemical Oxygen Evolution. Adv. Funct. Mater. 2017, 27, 1603904. [Google Scholar] [CrossRef]
- Feng, J.-X.; Ye, S.-H.; Xu, H.; Tong, Y.-X.; Li, G.-R. Design and Synthesis of FeOOH/CeO2 Heterolayered Nanotube Electrocatalysts for the Oxygen Evolution Reaction. Adv. Mater. 2016, 28, 4698–4703. [Google Scholar] [CrossRef] [PubMed]
- Feng, J.-X.; Xu, H.; Dong, Y.-T.; Ye, S.-H.; Tong, Y.-X.; Li, G.-R. FeOOH/Co/FeOOH Hybrid Nanotube Arrays as High-Performance Electrocatalysts for the Oxygen Evolution Reaction. Angew. Chem. 2016, 55, 3694–3698. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B.; Zheng, X.; Voznyy, O.; Comin, R.; Bajdich, M.; García-Melchor, M.; Han, L.; Xu, J.; Liu, M.; Zheng, L.; et al. Homogeneously dispersed multimetal oxygen-evolving catalysts. Science 2016, 352, 333–337. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Zhao, C. Enhancing Water Oxidation Catalysis on a Synergistic Phosphorylated NiFe Hydroxide by Adjusting Catalyst Wettability. ACS Catal. 2017, 7, 2535–2541. [Google Scholar] [CrossRef]
- Shin, H.; Xiao, H.; Goddard, W.A. In Silico Discovery of New Dopants for Fe-Doped Ni Oxyhydroxide (Ni1−xFexOOH) Catalysts for Oxygen Evolution Reaction. J. Am. Chem. Soc. 2018, 140, 6745–6748. [Google Scholar] [CrossRef] [PubMed]
- Dong, R.; Du, H.; Sun, Y.; Huang, K.; Li, W.; Geng, B. Selective Reduction–Oxidation Strategy to the Conductivity-Enhancing Ag-Decorated Co-Based 2D Hydroxides as Efficient Electrocatalyst in Oxygen Evolution Reaction. ACS Sustain. Chem. Eng. 2018, 6, 13420–13426. [Google Scholar] [CrossRef]
- Suryawanshi, M.P.; Shin, S.W.; Ghorpade, U.V.; Kim, J.; Jeong, H.W.; Kang, S.H.; Kim, J.H. A facile, one-step electroless deposition of NiFeOOH nanosheets onto photoanodes for highly durable and efficient solar water oxidation. J. Mater. Chem. A 2018, 6, 20678–20685. [Google Scholar] [CrossRef]
- Balram, A.; Zhang, H.; Santhanagopalan, S. Enhanced Oxygen Evolution Reaction Electrocatalysis via Electrodeposited Amorphous α-Phase Nickel-Cobalt Hydroxide Nanodendrite Forests. ACS Appl. Mater. Interfaces 2017, 9, 28355–28365. [Google Scholar] [CrossRef] [PubMed]
- Konkena, B.; Masa, J.; Botz, A.J.R.; Sinev, I.; Xia, W.; Koßmann, J.; Drautz, R.; Muhler, M.; Schuhmann, W. Metallic NiPS3@NiOOH Core–Shell Heterostructures as Highly Efficient and Stable Electrocatalyst for the Oxygen Evolution Reaction. ACS Catal. 2017, 7, 229–237. [Google Scholar] [CrossRef]
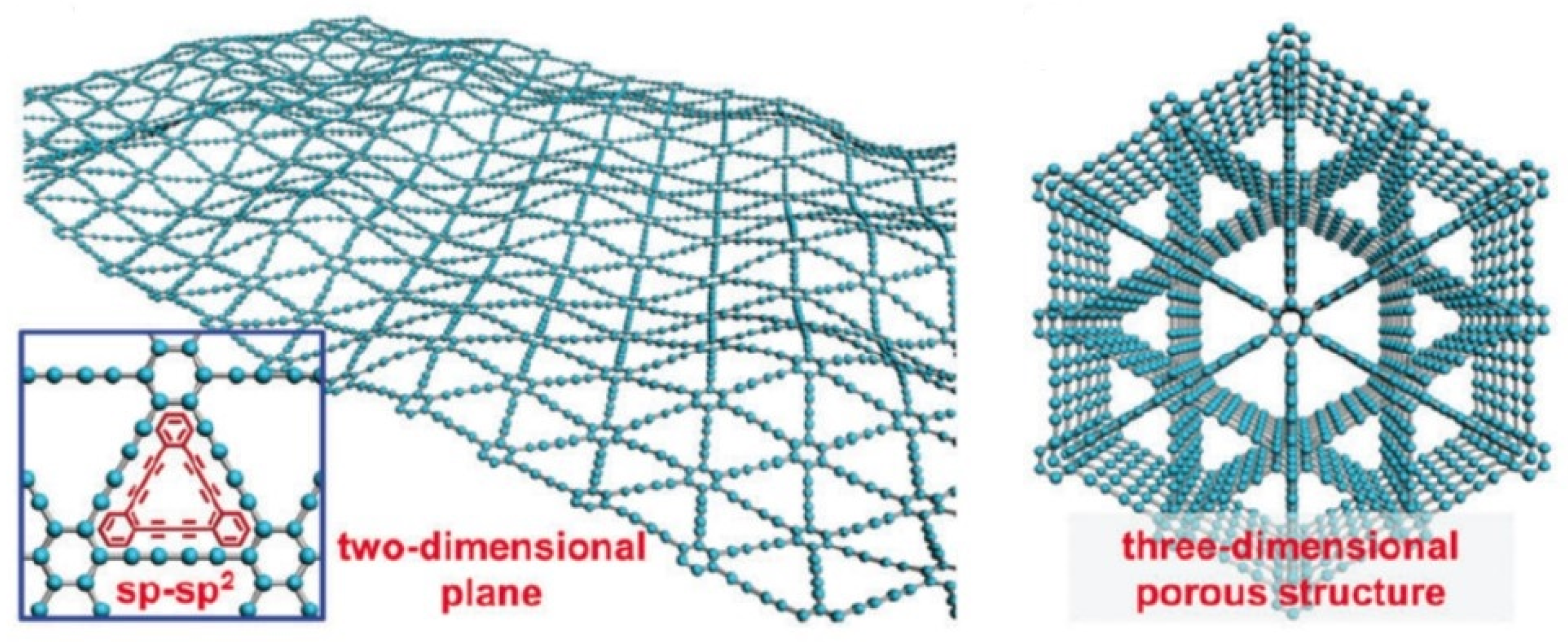
- Kuang, P.; Zhu, B.; Li, Y.; Liu, H.; Yu, J.; Fan, K. Graphdiyne: A superior carbon additive to boost the activity of water oxidation catalysts. Nanoscale Horiz. 2018, 3, 317–326. [Google Scholar] [CrossRef]
- Lu, C.; Yang, Y.; Wang, J.; Fu, R.; Zhao, X.; Zhao, L.; Ming, Y.; Hu, Y.; Lin, H.; Tao, X.; et al. High-performance graphdiyne-based electrochemical actuators. Nat. Commun. 2018, 9, 752. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Shen, X.; Pan, Y.; Liu, C.; Hwang, S.Y.; Xu, Q.; Peng, Z. Synthesis of freestanding amorphous giant carbon tubes with outstanding oil sorption and water oxidation properties. J. Mater. Chem. A 2018, 6, 3996–4002. [Google Scholar] [CrossRef]
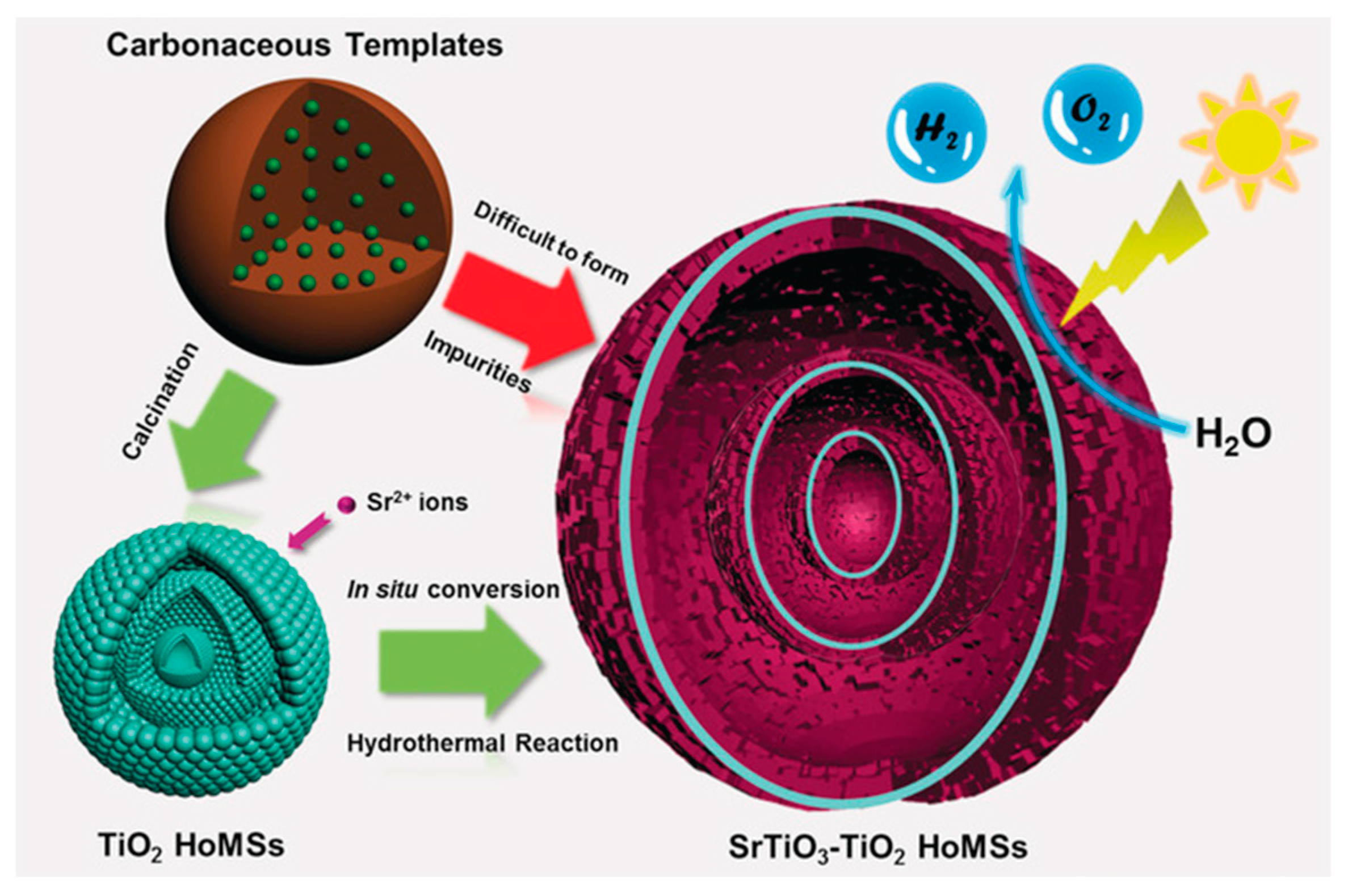
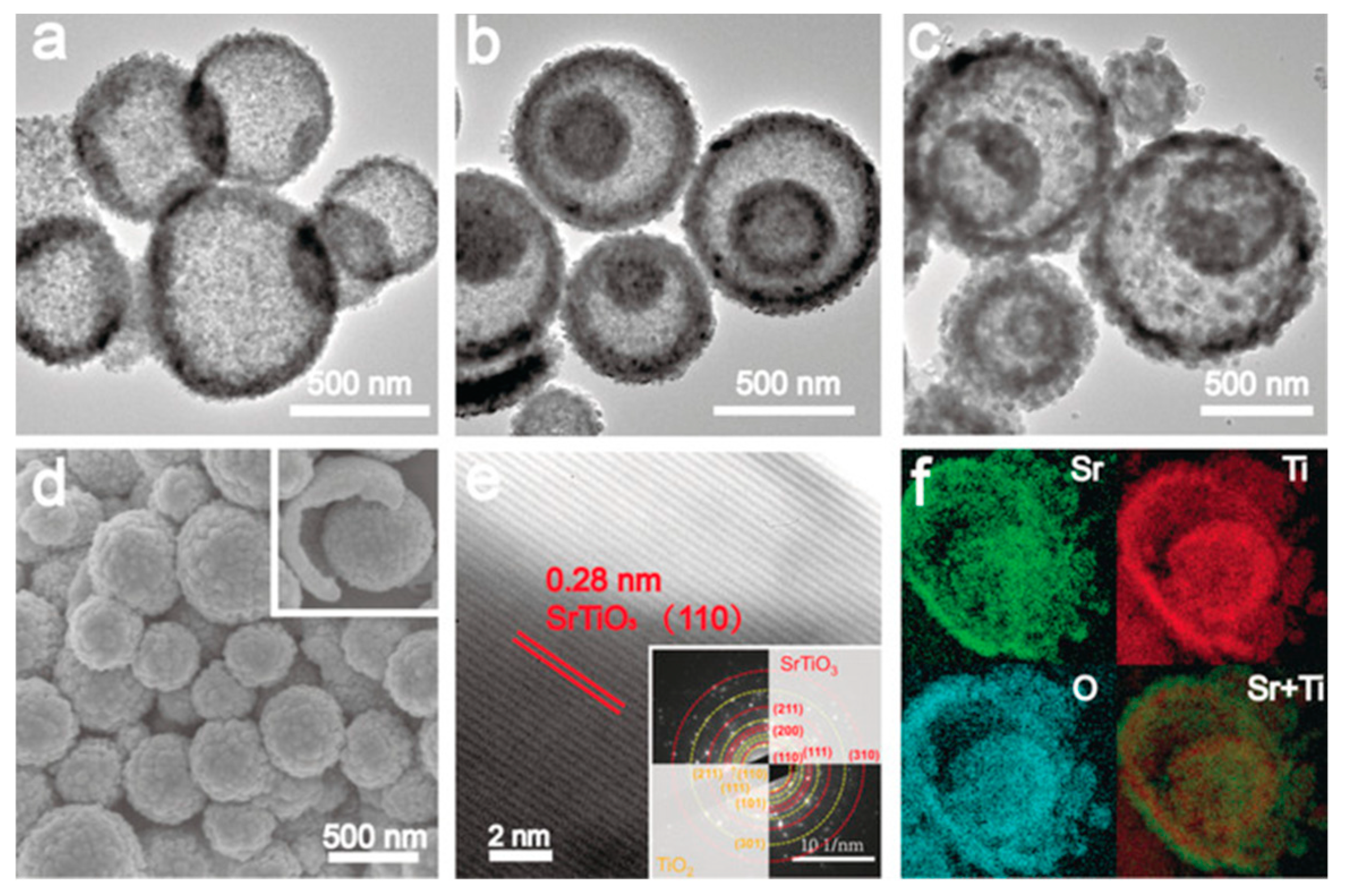
- Wei, Y.; Wang, J.; Yu, R.; Wan, J.; Wang, D. Constructing SrTiO3–TiO2 Heterogeneous Hollow Multi-shelled Structures for Enhanced Solar Water Splitting. Angew. Chem. 2019, 58, 1422–1426. [Google Scholar] [CrossRef] [PubMed]
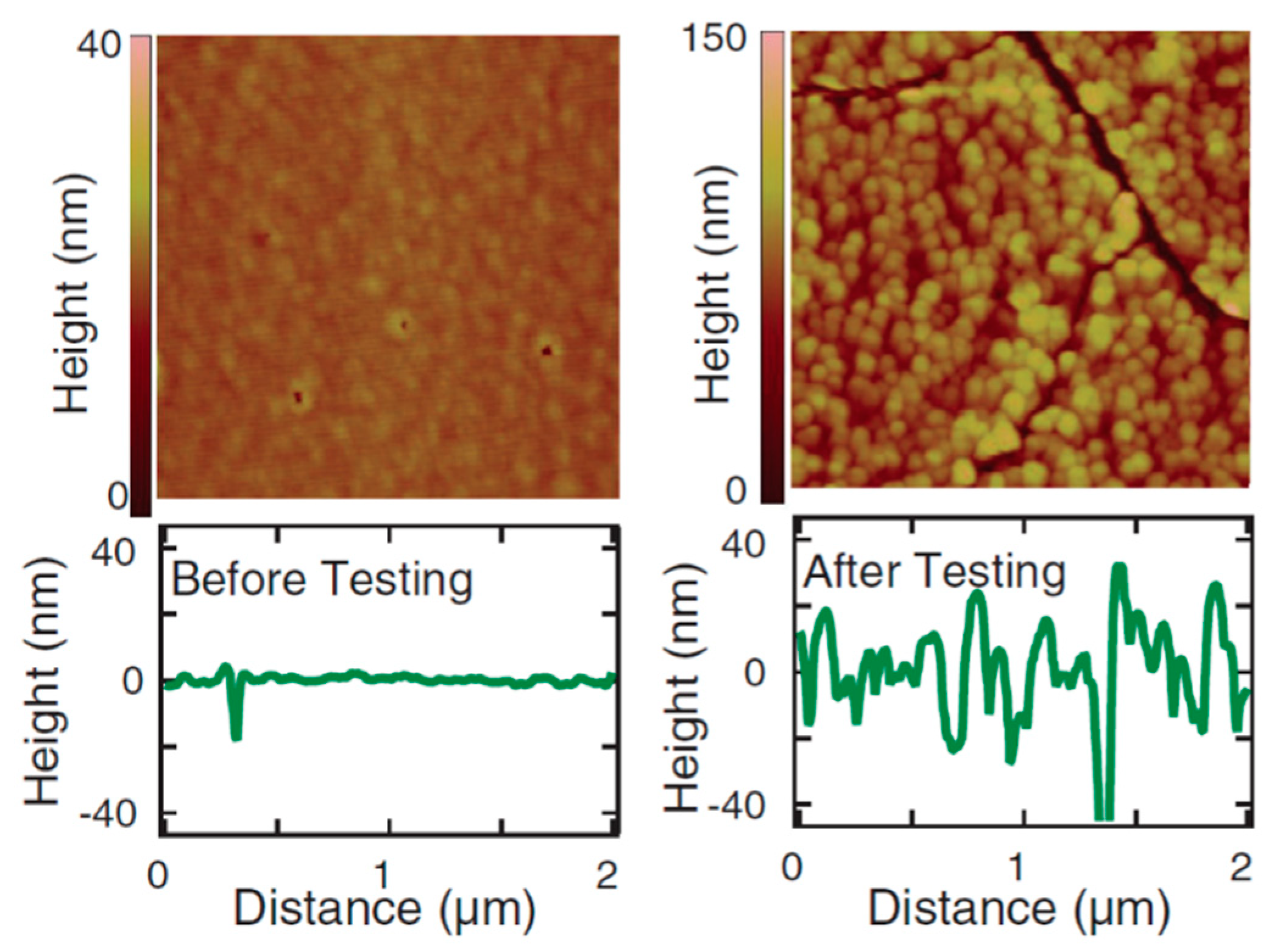
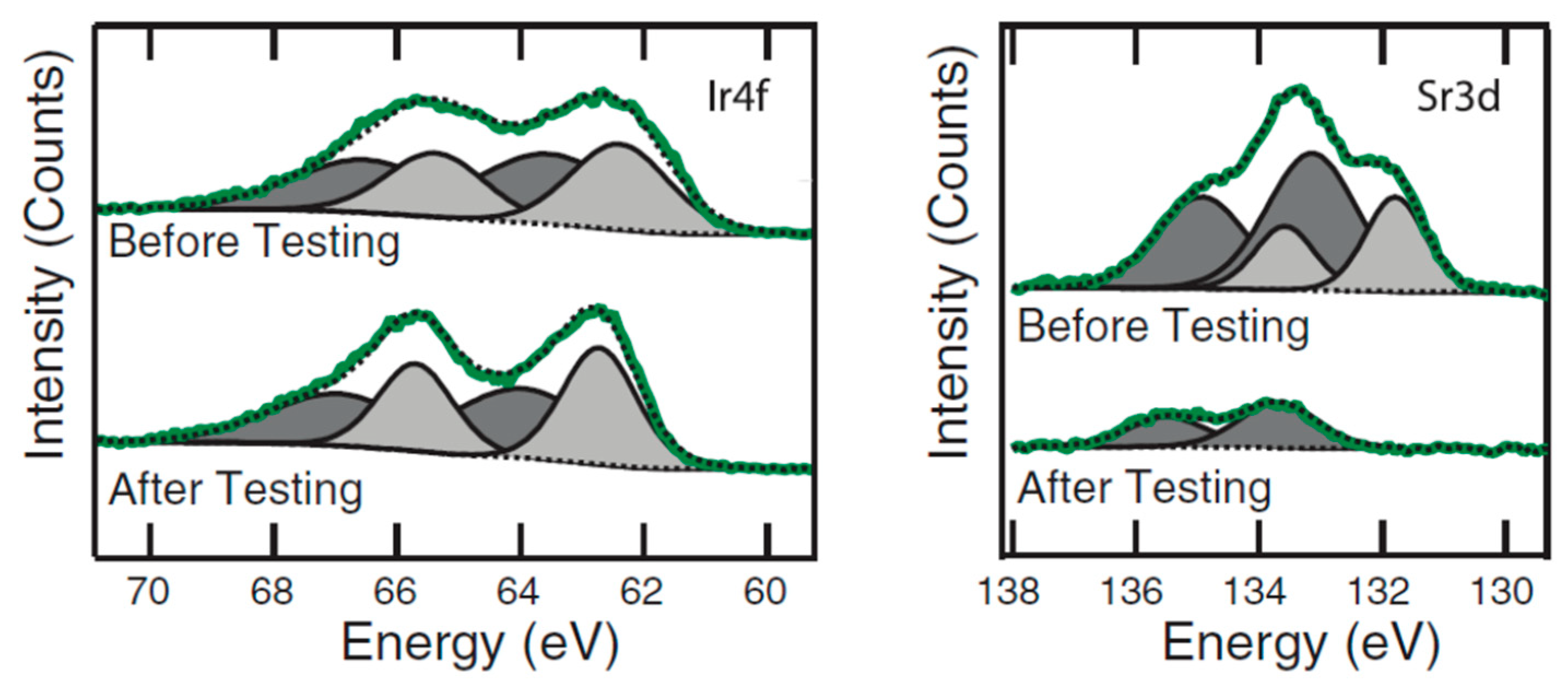
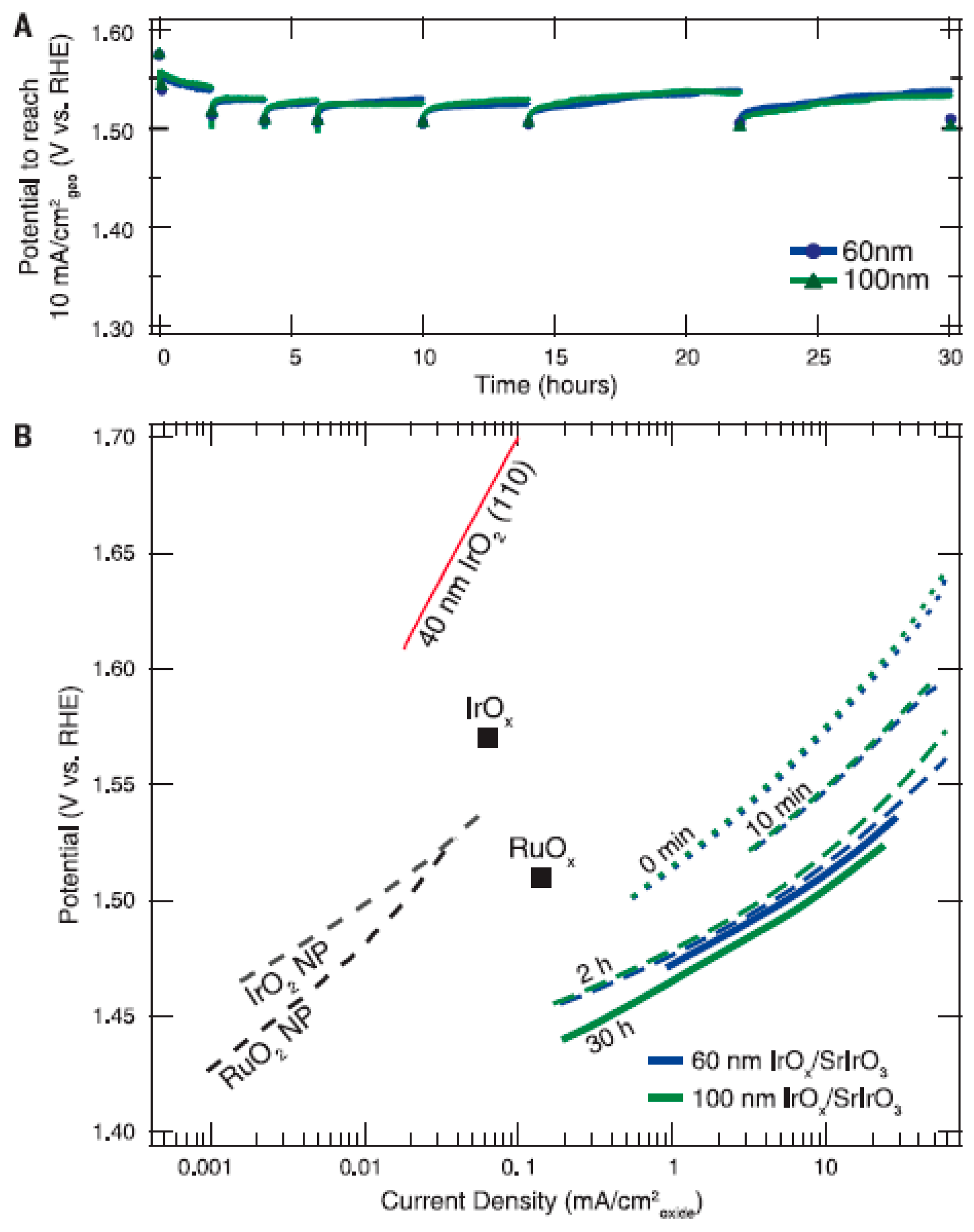
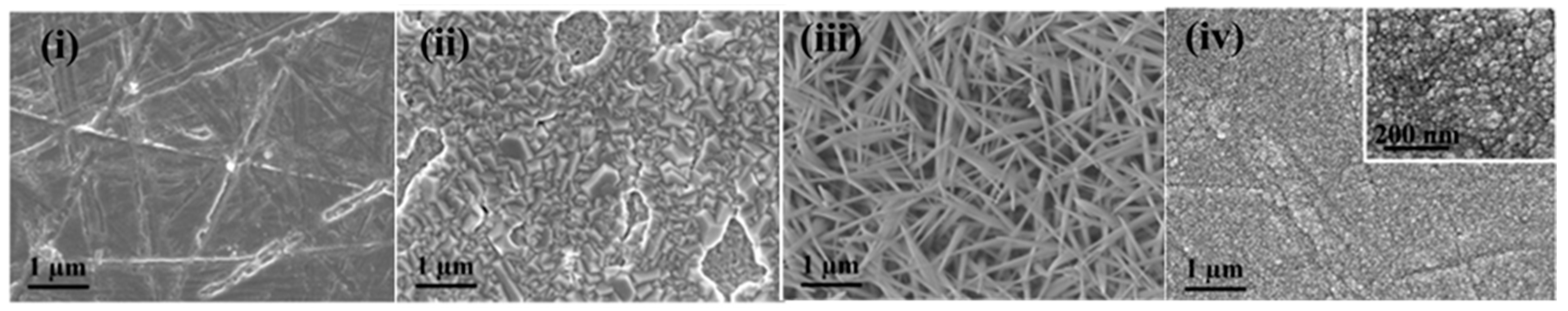







| Catalyst | Preparation Method | Onset Potential (V) | η (at 10 mV/cm2) (V) | Tafel Slope (mV dec−1) | Durability (h) | Stability | Electrolyte | Ref. |
|---|---|---|---|---|---|---|---|---|
| Ru and Ir | Sputtering & evaporation | Ru: 1.20 (both media), Ir: 1.27 & 1.43 (acidic & alkaline media, respectively) | - | 30–40 | 0.25 | Low | 0.1 M H2SO4 & 0.05 M NaOH | [19] |
| RuO2 and IrO2 | Sputtering & evaporation | RuO2: 1.35 & 1.47 (acidic & alkaline media, respectively), IrO2: 1.47 (both media) | - | 30–40 | 0.25 | Good * | 0.1 M H2SO4 & 0.05 M NaOH | [19] |
| IrOx/SrIrO3 | Pulsed laser deposition (PLD) | 1.44 | 0.27 | - | 30 | Reasonable | 0.5 M H2SO4 | [16] |
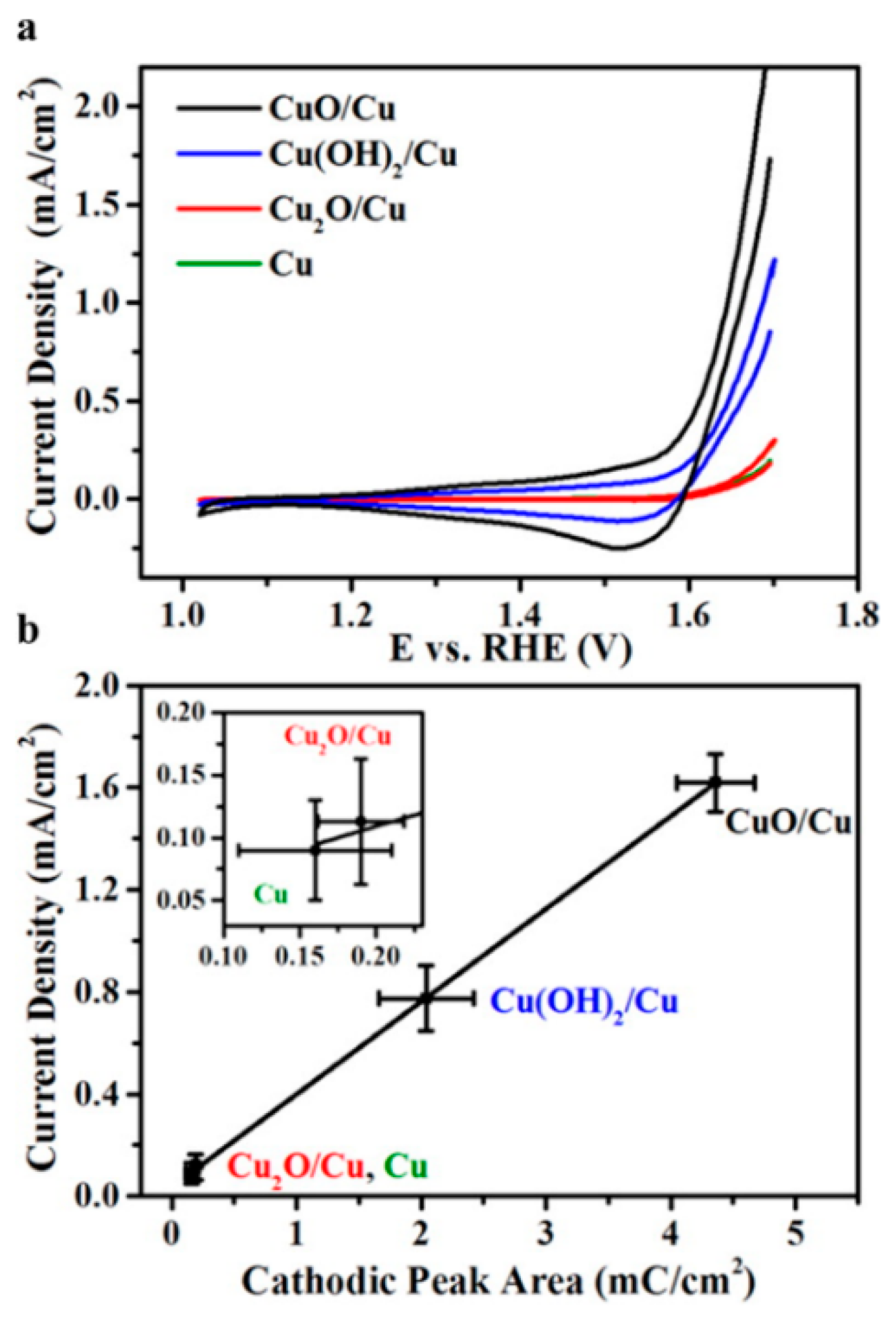
| Cu, Cu2O, Cu(OH)2 and CuO | Electrodeposition | - | 0.417 | 60–66 | 22 | Cu & Cu2O: very good *, CuO & Cu(OH)2: poor * | 0.1 M KOH | [17] |
| IrOOH nanosheets | Exfoliation in TBAOH | - | 0.344 | 58 | 14 | Extraordinary | 0.1 M HClO | [15] |
| Cu2O/ITO | Electrodeposition | <0.8 | 0.4 | 72 | 5 | High | K2B4O8, C2H3KO2 & KH2PO4 | [20] |
| Cu(OH)2@CoCO3(OH)2· nH2O | Hydrothermal | - | 0.27 | 78 | 20 | Superior | 1 M KOH | [18] |
| RuO2 (001) | Commercially available | - | - | - | 2 | - | 0.05 M H2SO4 | [21] |
| IrxO/ATO | Electrodeposition | - | 0.28 | 61.7 | 15 | High * | 0.05 M H2SO4 | [22] |
| TiO2/CuO | Spin-coating & pulsed laser deposition | - | - | - | 0.166 | Excellent | 0.1 M NaOH | [26] |
| Catalyst | Preparation Method | Onset Potential (V) | η (at 10 mV/cm2) (V) | Tafel Slope (mV dec−1) | Durability (h) | Stability | Electrolyte | Ref. |
|---|---|---|---|---|---|---|---|---|
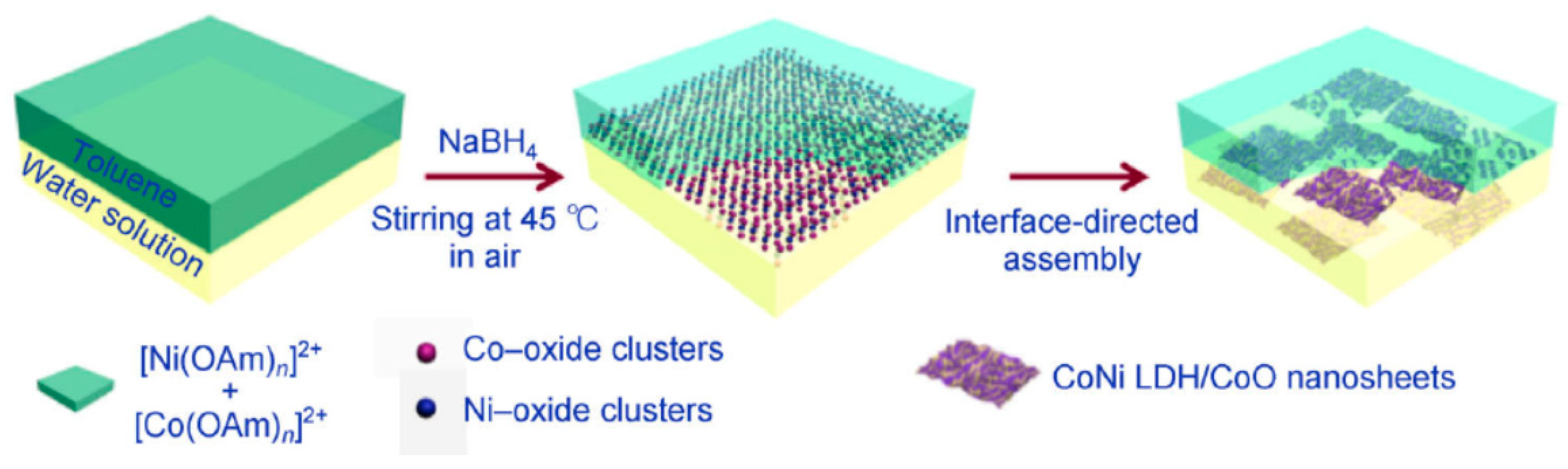
| CoNi-LDH/CoO nanosheets | In situ reduction & interface-directed assembly in air | 1.48 | 0.3 | 123 | 10 | Good | 1 M KOH | [29] |
| CoAl & CoFe LDH | Co-precipitation | 1.58 | 0.36 & 0.37 | 39 & 49 | 50 | Excellent | 0.1 M KOH | [30] |
| Co-Fe LDH nanosheets | Electrodeposition | - | 0.42 | 131 | 50 | Superior | 0.1 M K2B4O8 | [31] |
| Ni0.75–V0.25 LDH narrow nanosheets | Hydrothermal | - | 0.35 | 50 | 25 | Considerable | 1 M KOH | [32] |
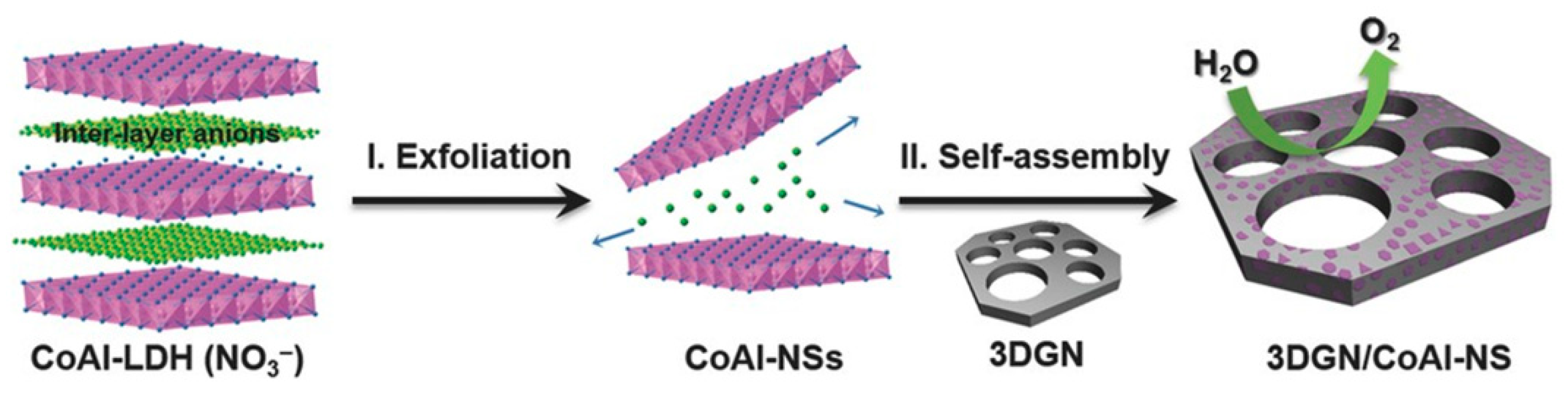
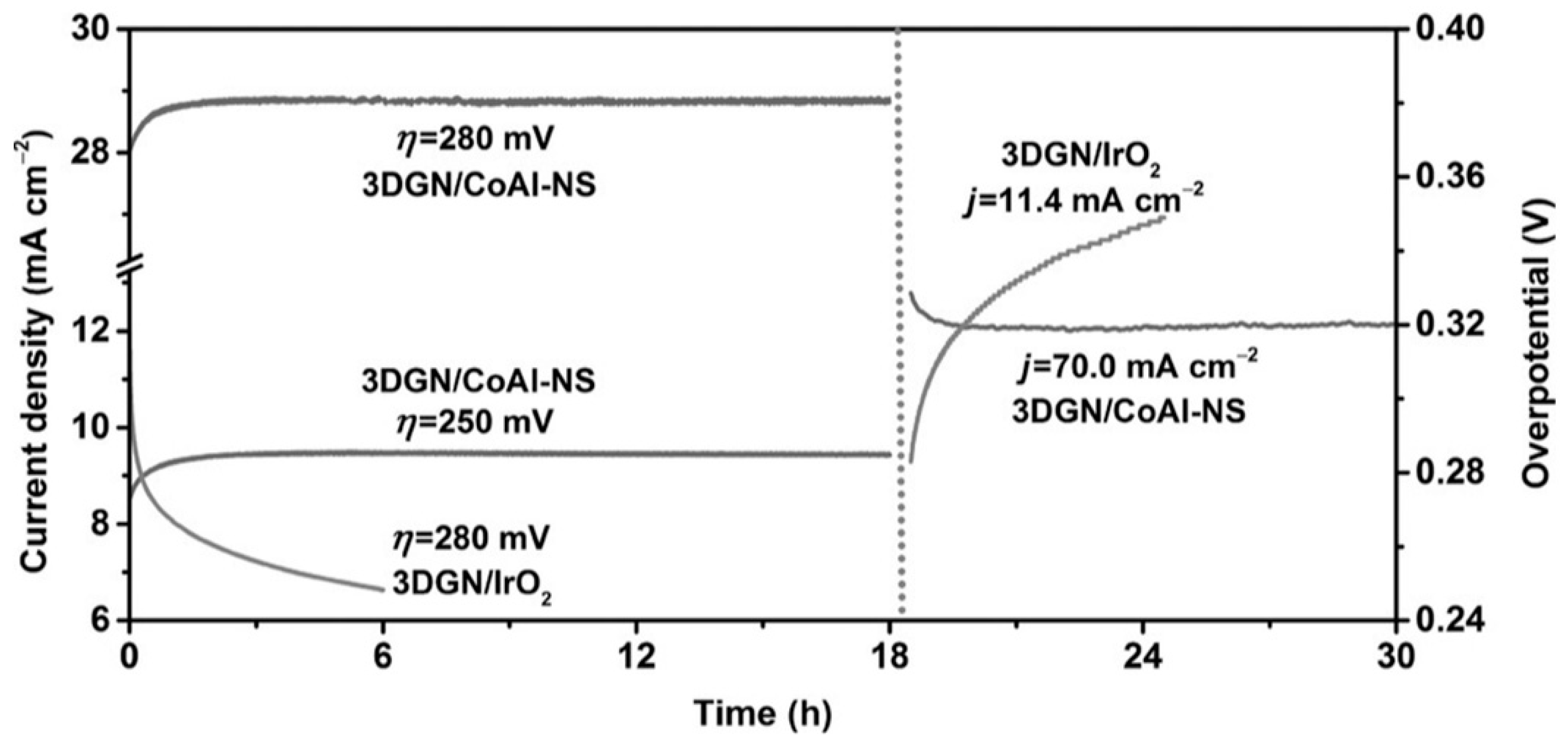
| CoAl-LDH on 3D graphene network | Hydrothermal | - | 0.28 | 36 | 30 | Excellent | 1 M KOH | [33] |
| NiFe LDHs-MoOx | Hydrothermal | - | 0.276 | 55 | 12 | Good | 1 M KOH | [38] |
| NiFe-LDH array | Electrodeposition | 1.43 | 0.224 | 52.5 | 50 | Superior | 1 M KOH | [37] |
| Fe-doped Ni(OH)2 films | Precipitation | - | 0.25 to 0.35 | - | 2 | Good * | 1 M KOH | [36] |
| Ni–FeO(OH) | Microwave-assisted autoclave synthesis under solvothermal conditions | - | 0.29 | 40 | >20 | Good | 0.1 M KOH | [39] |
| Fe2O3–Ni(OH)2 to NiFe2O4–NiOOH | Hydrothermal & anodization | - | 0.24 | 55 | 1 | High | 1 M KOH | [40] |
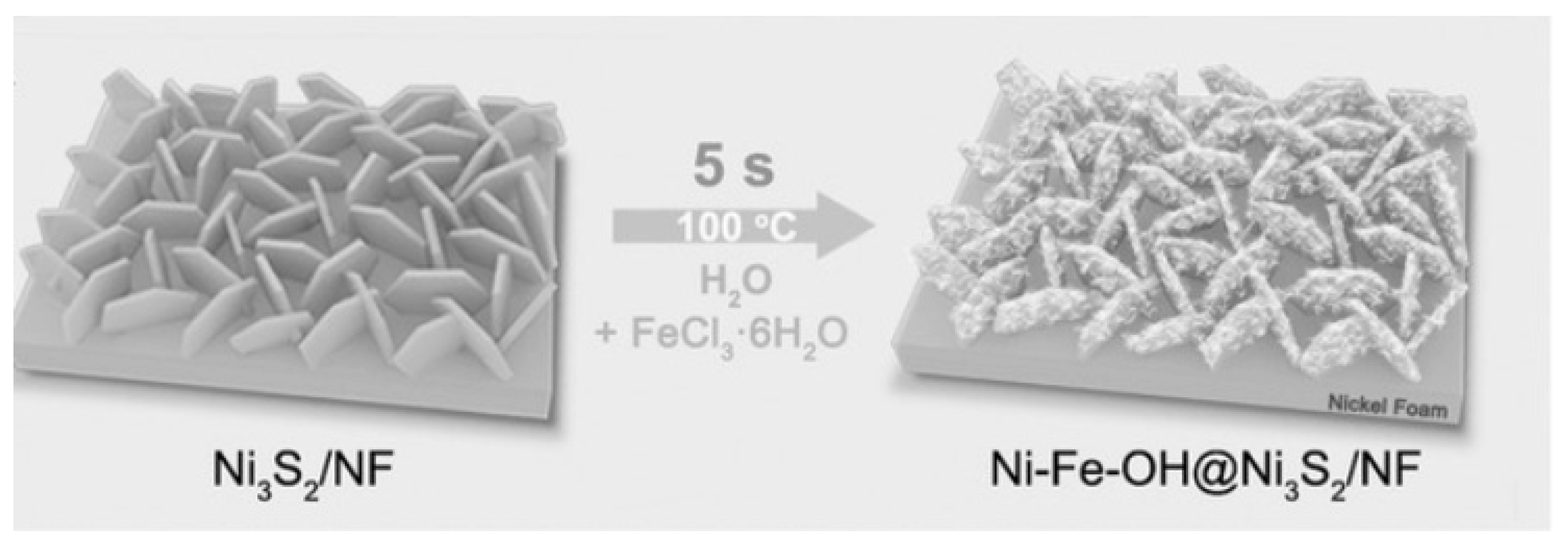
| Ni–Fe-OH@Ni3S2 | Two-step synthesis | - | 0.479 | 124 | 50 | Excellent | 1 M KOH | [41] |
| Ni/NiFe-OH | Multi-step electrodeposition | - | 0.205 | 53 | 12 | Excellent | 1 M KOH | [42] |
| NiFe-LDH nanosheets | Pulsed laser ablation in liquids | - | 0.27-0.45 | 3.5 | Remarkable | 1 M KOH | [43] | |
| NiFe LDH-NS@DG | Co-precipitation | 1.41 | 0.21 | 52 | 10 | Robust | 1 M KOH | [44] |
| Catalyst | Preparation Method | Onset Potential (V) | η (at 10 mV/cm2) (V) | Tafel Slope (mV dec−1) | Durability (h) | Stability | Electrolyte | Ref. |
|---|---|---|---|---|---|---|---|---|
| PrBa0.5Sr0.5Co1.5Fe0.5O5+δ | Electrospinning | - | 0.37 | 52 | 12 | High | 0.1 M KOH | [61] |
| SrNb0.1Co0.7Fe0.2O3−δ nanorods | Electrospinning | 1.52 | 0.37 | 61 | 30 | Remarkable | 1 M KOH | [60] |
| BaCo0.7Sn0.3O3−δ & BaCo0.7Fe0.1Sn0.2O3−δ | Ball milling-assisted solid-state reaction | 1.53 | 0.45 | 68 | 2 | Good | 0.1 M KOH | [63] |
| La0.95FeO3−δ | Sol–gel | 1.55 | 0.41 | 82 | - | - | 0.1 M KOH | [64] |
| La1−xSrxCoO3−δ | Reverse-phase hydrolysis | - | 0.4 | 31 | 24 | High | 0.1 M KOH | [65] |
| BaNiO3 & BaNi0.83O2.5 | Flux mediated crystal growth | - | - | - | 20 | Very good * | 0.1 M KOH | [66] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mavrokefalos, C.K.; Patzke, G.R. Water Oxidation Catalysts: The Quest for New Oxide-Based Materials. Inorganics 2019, 7, 29. https://doi.org/10.3390/inorganics7030029
Mavrokefalos CK, Patzke GR. Water Oxidation Catalysts: The Quest for New Oxide-Based Materials. Inorganics. 2019; 7(3):29. https://doi.org/10.3390/inorganics7030029
Chicago/Turabian StyleMavrokefalos, Christos K., and Greta R. Patzke. 2019. "Water Oxidation Catalysts: The Quest for New Oxide-Based Materials" Inorganics 7, no. 3: 29. https://doi.org/10.3390/inorganics7030029
APA StyleMavrokefalos, C. K., & Patzke, G. R. (2019). Water Oxidation Catalysts: The Quest for New Oxide-Based Materials. Inorganics, 7(3), 29. https://doi.org/10.3390/inorganics7030029





