Abstract
In previous studies, complex hydrides LiBH4 and Mg2FeH6 have been reported to undergo simultaneous dehydrogenation when ball-milled as composite materials (1 − x)LiBH4 + xMg2FeH6. The simultaneous hydrogen release led to a decrease of the dehydrogenation temperature by as much as 150 K when compared to that of LiBH4. It also led to the modified dehydrogenation properties of Mg2FeH6. The simultaneous dehydrogenation behavior between stoichiometric ratios of LiBH4 and Mg2FeH6 is not yet understood. Therefore, in the present work, we used the molar ratio x = 0.25, 0.5, and 0.75, and studied the isothermal dehydrogenation processes via pressure–composition–isothermal (PCT) measurements. The results indicated that the same stoichiometric reaction occurred in all of these composite materials, and x = 0.5 was the molar ratio between LiBH4 and Mg2FeH6 in the reaction. Due to the optimal composition ratio, the composite material exhibited enhanced rehydrogenation and reversibility properties: the temperature and pressure of 673 K and 20 MPa of H2, respectively, for the full rehydrogenation of x = 0.5 composite, were much lower than those required for the partial rehydrogenation of LiBH4. Moreover, the x = 0.5 composite could be reversibly hydrogenated for more than four cycles without degradation of its H2 capacity.
1. Introduction
Boron-based complex hydrides MBH4 (M = Li, Na, and K), which consist of an M+ cation and a [BH4]− complex anion, have high gravimetric H2 densities (7.7–18.4 mass %); therefore, these materials have the potential to be used as hydrogen storage materials [1,2,3]. However, MBH4 are thermodynamically stable and the hydrogenation is difficult to achieve under mild temperatures and pressures. For example, when undergoing the following Reaction (1), the dehydrogenation temperature of LiBH4 at 0.1 MPa H2 has been estimated to be 683 K on the basis of enthalpy ΔH and entropy ΔS changes of 66.6 kJ/(mole of H2) and 97.4 J/K(mole of H2), respectively [4]. In practice, significant dehydrogenation only occurs at temperatures greater than 700 K. In addition, partial rehydrogenation of LiBH4 requires a much higher pressure and temperature of 35 MPa and 873 K.
LiBH4 ⇄ LiH + B + (3/2)H2
Combining borohydrides with metallic hydrides (e.g., MgH2) or complex hydrides (e.g., Mg2NiH4) has been proven to be effective at modifying the dehydrogenation properties of MBH4. The reactions between MBH4 and the partner hydrides in these composite materials can produce the stable boride MgB2 or MgNi2.5B2 that results in a decrease of the dehydrogenation temperature of LiBH4 [5,6,7,8,9,10,11]. Therefore, in a previous study, we have tried to bring down the dehydrogenation temperature of LiBH4 by combining it with a transition-metal-based complex hydride, Mg2FeH6. We ball-milled the two complex hydrides over a large composition range: (1 − x)LiBH4 + xMg2FeH6 (0.25 ≤ x ≤ 0.9) [12,13,14,15]. The dehydrogenation properties were investigated using a dynamic measurement: thermogravimetry–mass spectrometry (TG–MS). It was observed that although the thermodynamic stabilities of LiBH4 and Mg2FeH6 differed substantially, they underwent dehydrogenation simultaneously when heated up during the TG–MS analysis, by which the dehydrogenation temperature of the composite materials was lowered by, at most, 150 K when compared to that of LiBH4. In addition to the dehydrogenation properties of LiBH4, those of Mg2FeH6 were also modified; Mg2FeH6 no longer underwent independent dehydrogenation, and the temperature of the simultaneous dehydrogenation shifted closer to that of pure Mg2FeH6, both continuously and with an increasing ratio x.
The dehydrogenation process of (1 − x)LiBH4 + xMg2FeH6 is special when compared to the processes of other composite materials, e.g., 2LiBH4 + MgH2 [16]. The dehydrogenation behavior of MgH2 was unaffected by its combination with LiBH4; furthermore, a stoichiometric reaction between LiBH4 and MgH2 existed, the dehydrogenation temperature of which would not be affected by the composition ratio in the composite materials. However, in the case of (1 − x)LiBH4 + xMg2FeH6, the properties of both LiBH4 and Mg2FeH6 were modified and the stoichiometric reaction between these two complex hydrides at a specific composition ratio was not yet understood. We considered that the stoichiometric reaction was hidden in the continuous hydrogen releasing events in the dynamic TG–MS measurements, where the thermodynamic and kinetic factors both affected the dehydrogenation process.
Besides our study, the research on LiBH4 + Mg2FeH6 composite materials has been conducted on LiBH4-rich compositions only. The reaction processes investigated by various dynamic measurement methods (for example, differential scanning calorimetry (DSC)) has been explained as Mg2FeH6 dehydrogenating to form elemental Mg and Fe, followed by the Mg and Fe reacting with LiBH4 and thereby destabilizing it thermodynamically. Because of the absence of studies on any Mg2FeH6-rich compositions, the simultaneous dehydrogenation was not discovered [17,18]. In addition, Ghaani et al. used an isothermal method (i.e., the pressure–composition–isothermal (PCT) measurement) to study the dehydrogenation process of 2LiBH4 + Mg2FeH6. Here, it was found that LiBH4 and Mg2FeH6 react with each other; however, unreacted LiBH4 was apparent using this composition ratio [19]. Whether the reaction between LiBH4 and Mg2FeH6 changes with variations in composition ratio is unclear, and whether an optimal composition ratio exists at which a stoichiometric reaction occurs without independent dehydrogenation of the parent complex hydrides remains unknown.
In this study, to confirm the stoichiometric reaction between Mg2FeH6 and LiBH4, and the optimal composition ratios for it, we used PCT measurements to evaluate the isothermal dehydrogenation processes of (1 − x)LiBH4 + xMg2FeH6. Among a large range of composition ratios, the focus was on three in the present study: x = 0.25, 0.5, and 0.75. TG–MS measurements done in our previous study revealed that x = 0.5 was a critical composition. Composites with x ≥ 0.5 exhibited a single dehydrogenation event, whereas composites with x < 0.5 underwent multiple events involving both simultaneous dehydrogenation of LiBH4 and Mg2FeH6, and individual dehydrogenation of LiBH4. We considered that the optimal ratio for the stoichiometric reaction could be deduced from these representative compositions. After deciding on the optimal ratio, its effect on the modification of the reversible hydrogenation properties of the composite materials were investigated.
2. Results
2.1. Optimal Composition Ratio for the Stoichiometric Reaction between LiBH4 and Mg2FeH6
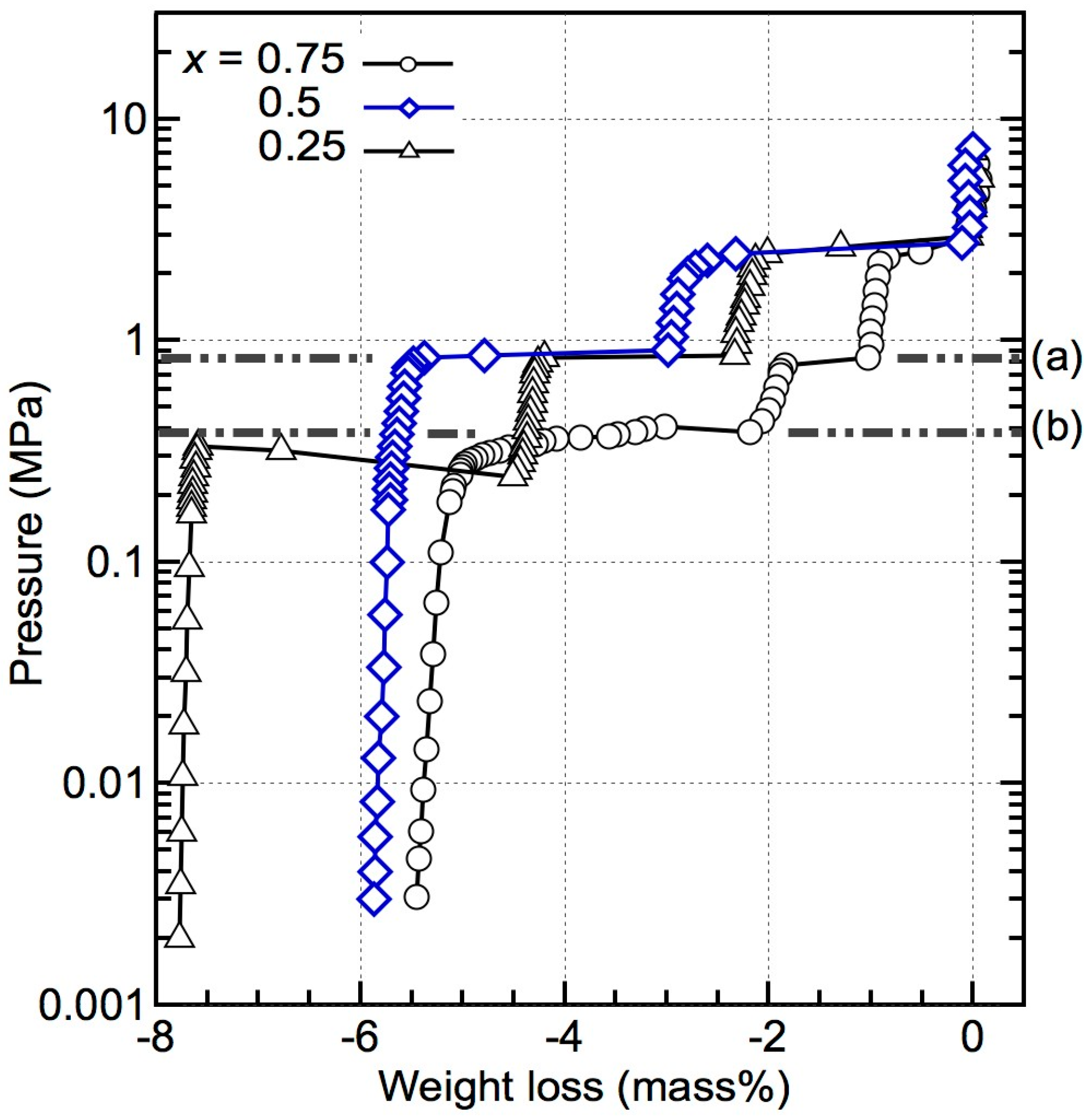
The PCT results obtained at 643 K for the composites (x = 0.25, 0.5, and 0.75; temperatures were slightly different within each measurement) are shown in Figure 1. Although only one dehydrogenation event was observed for the x = 0.5 and 0.75 composites in the dynamic TG–MS measurements, the PCT results revealed two and three thermodynamically independent reactions, respectively. For x = 0.25, three independent reactions were observed, similar to the TG–MS results in which multiple dehydrogenation events appeared. The first and second dehydrogenation reactions of all of these compositions exhibited the same equilibrium pressure of 1.85 MPa and 0.85 MPa, which indicates that these compositions have undergone similar reaction pathways.
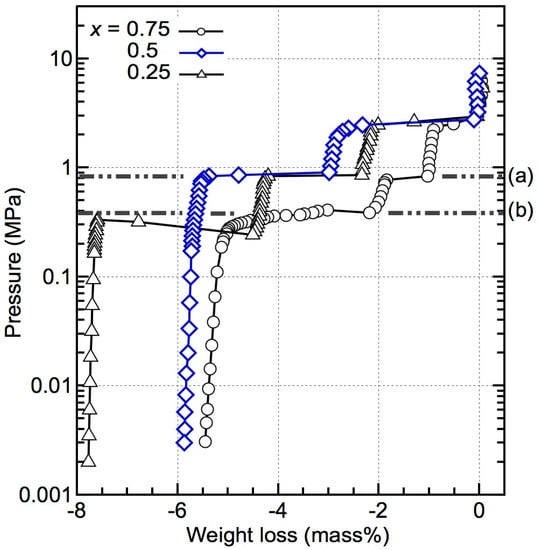
Figure 1.
Pressure–composition–isothermal (PCT) results for (1 − x)LiBH4 + xMg2FeH6 (x = 0.25, 0.5, and 0.75) at 643 K. The temperatures are slightly different within each measurement. The first and second reactions in all the compositions exhibit the same equilibrium pressures of 1.85 MPa and 0.85 MPa, respectively. Line (a) represents the equilibrium pressure of the dehydrogenation of MgH2, according to Refs. [20,21]; line (b) represents the equilibrium pressure of the dehydrogenation of Mg2FeH6, according to Refs. [22,23,24].
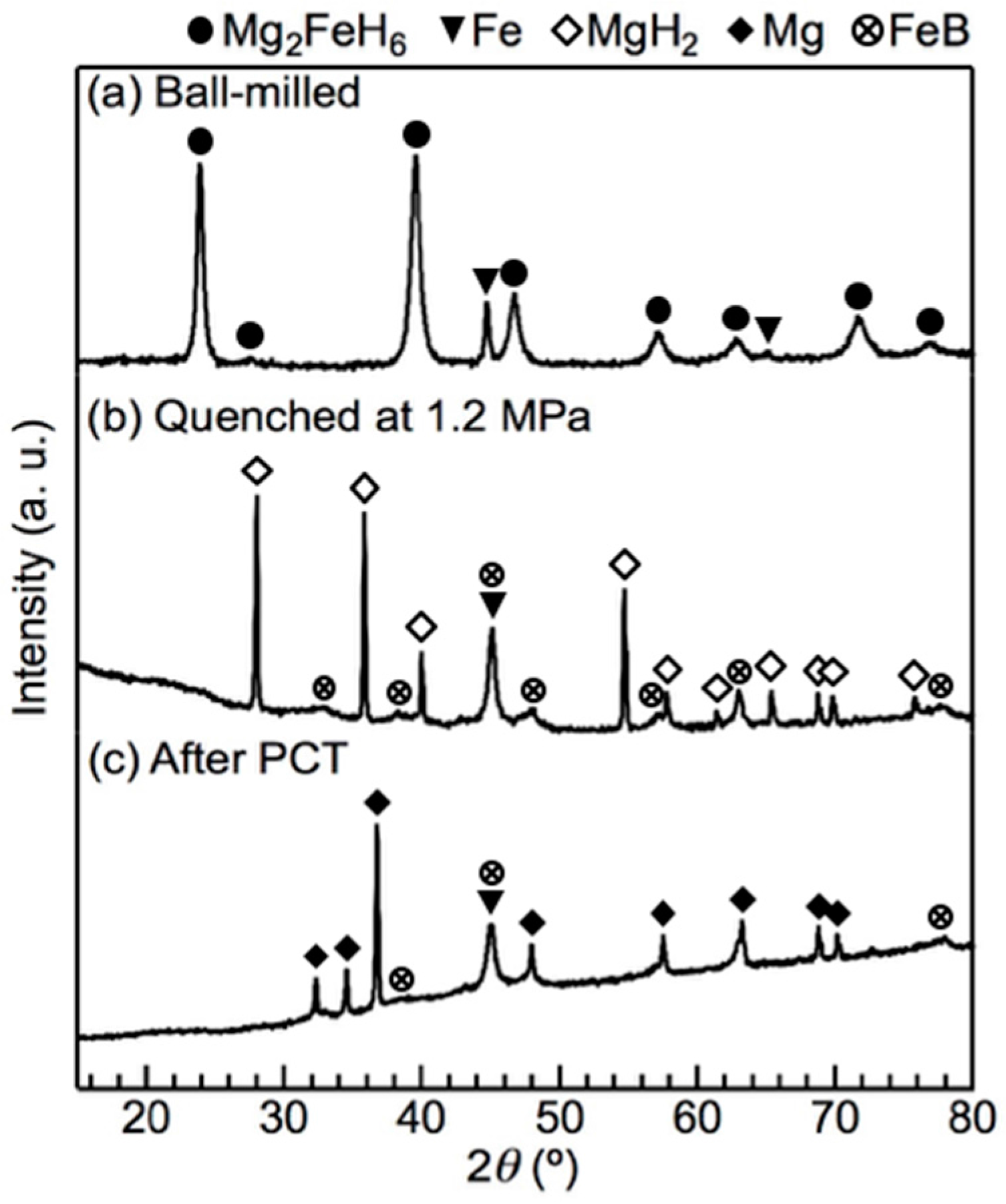
To determine the reaction pathway, the materials were quenched under H2 pressure after each thermodynamically independent reaction and each phase was identified using X-ray diffraction (XRD). The XRD results for the x = 0.5 composite after each reaction are shown in Figure 2. After the first reaction at 1.85 MPa H2, the diffraction peaks of Mg2FeH6 were no longer apparent, whilst the intensity of the diffraction peak of Fe increased. Furthermore, peaks attributable to MgH2 appeared, accompanied by several small broad peaks of an unclear phase. The results indicate that Mg2FeH6 was fully dehydrogenated. After the second reaction at 0.85 MPa H2, the diffraction peaks of MgH2 transitioned to those of Mg. The small broad unclear peaks were not prominent but were still present, indicating that this phase did not participate in the second reaction. The presence of LiBH4 was not confirmed by XRD due to its weak diffraction intensity compared to the other reactants as well as the amorphisation after ball-milling. Because direct dehydrogenation of Mg2FeH6 cannot produce MgH2; we considered that LiBH4 reacted with Mg2FeH6 in the first reaction. Also, we used scanning electron microscopy (SEM) to characterize the presence of LiBH4 before and after the first reaction.
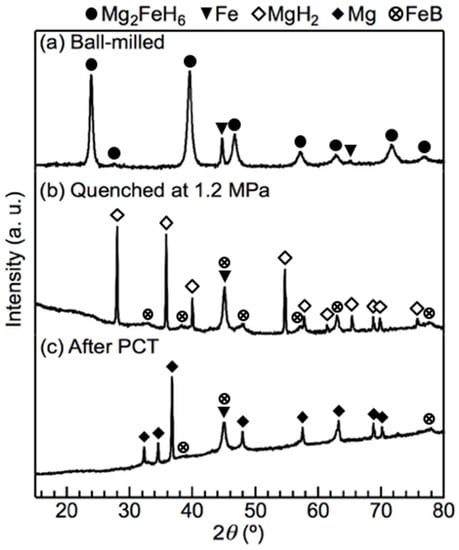
Figure 2.
X-ray diffraction (XRD) profiles of the x = 0.5 composite during the pressure–composition–isothermal (PCT) measurement at 643 K: (a) the ball-milled material before the PCT measurement; (b) the material quenched at 1.2 MPa, which is the pressure between the first isothermal reaction at 1.85 MPa and the second one at 0.85 MPa; and (c) the material after the PCT measurement. As a result of Fe in the material, the baseline tilted at high angles. The strongest diffraction peak of LiH overlapped with that of Fe at 44°.
The backscattering images (BSE) and energy-dispersive X-ray spectroscopy (EDX) analysis results obtained before the PCT measurements and after the first reaction are shown in Figure 3. Before the PCT measurement, the composite material was a uniform mixture; separate LiBH4 and Mg2FeH6 particles were not observed at about 100-nm scale. After the first isothermal reaction at 1.85 MPa, the composite became an obvious mixture. A large blank area was observed where no B, Fe, or Mg were detected, suggesting that this area was possibly composed of the dehydrogenation product of LiBH4: LiH. These results complement the XRD results, indicating that both LiBH4 and Mg2FeH6 underwent dehydrogenation in the first reaction. Element mapping of Fe and B showed significant overlap, and the particle size was of the order of several nanometers, indicating the possible formation of iron boride. A 1:1 reaction ratio between LiBH4 and Mg2FeH6 in the first isothermal reaction suggests that FeB is a possible iron boride product. The recognized diffraction patterns in the XRD results can be attributed to FeB (orthorhombic, Cmcm), but the very broad peaks made the phase identification difficult. Because of the overlapping of the strongest diffraction peaks of Fe (cubic, Im-3m) and FeB, it is hard to rule out the existence of Fe. Therefore, we still keep it in the reaction of Equation (2). The formation of FeB is crucial for the reaction between LiBH4 and Mg2FeH6. The estimated enthalpy change ΔH and entropy change ΔS for the reaction (LiBH4 + Mg2FeH6 → LiH + 2MgH2 + FeB + 5/2H2) is 64 kJ/(mol of H2) and 125 J/K(mole of H2) , respectively. According to the theoretical value, the equilibrium pressure at 643 K would be 2.1 MPa [23,25,26,27]. It is very close to the experimental data 1.85 MPa.

Figure 3.
Backscattered electron (BSE) images and energy-dispersive X-ray spectroscopy (EDX) analyses of the x = 0.5 composite: (a) the material after ball milling was a uniform mixture of LiBH4 and Mg2FeH6; (b) the material quenched at 1.2 MPa during the PCT measurement exhibited black areas, possibly LiH. These analyses indicate that LiBH4 dehydrogenated simultaneously with Mg2FeH6 during the first isothermal reaction at 1.85 MPa.
On the basis of the phase identification results and the equilibrium pressure of the dehydrogenation reaction of MgH2 calculated from previously reported thermodynamic data [20,21], Mg2FeH6 and LiBH4 clearly reacted during the first reaction (Equation (2)), whereas the second reaction (Equation (3)) was the dehydrogenation of MgH2.
LiBH4 + Mg2FeH6 → LiH + 2MgH2 + (Fe, FeB) + 5/2H2
MgH2 → Mg + H2
The theoretical weight loss due to hydrogen release of Reactions (2) and (3) is 3.8 mass % and 3.0 mass %, respectively. This correlates well with the dehydrogenation of MgH2 in the second reaction very well. However, the actual weight loss of the first reaction was only 3 mass %, and this deviation could be due to the residual Fe present from Mg2FeH6 synthesis.
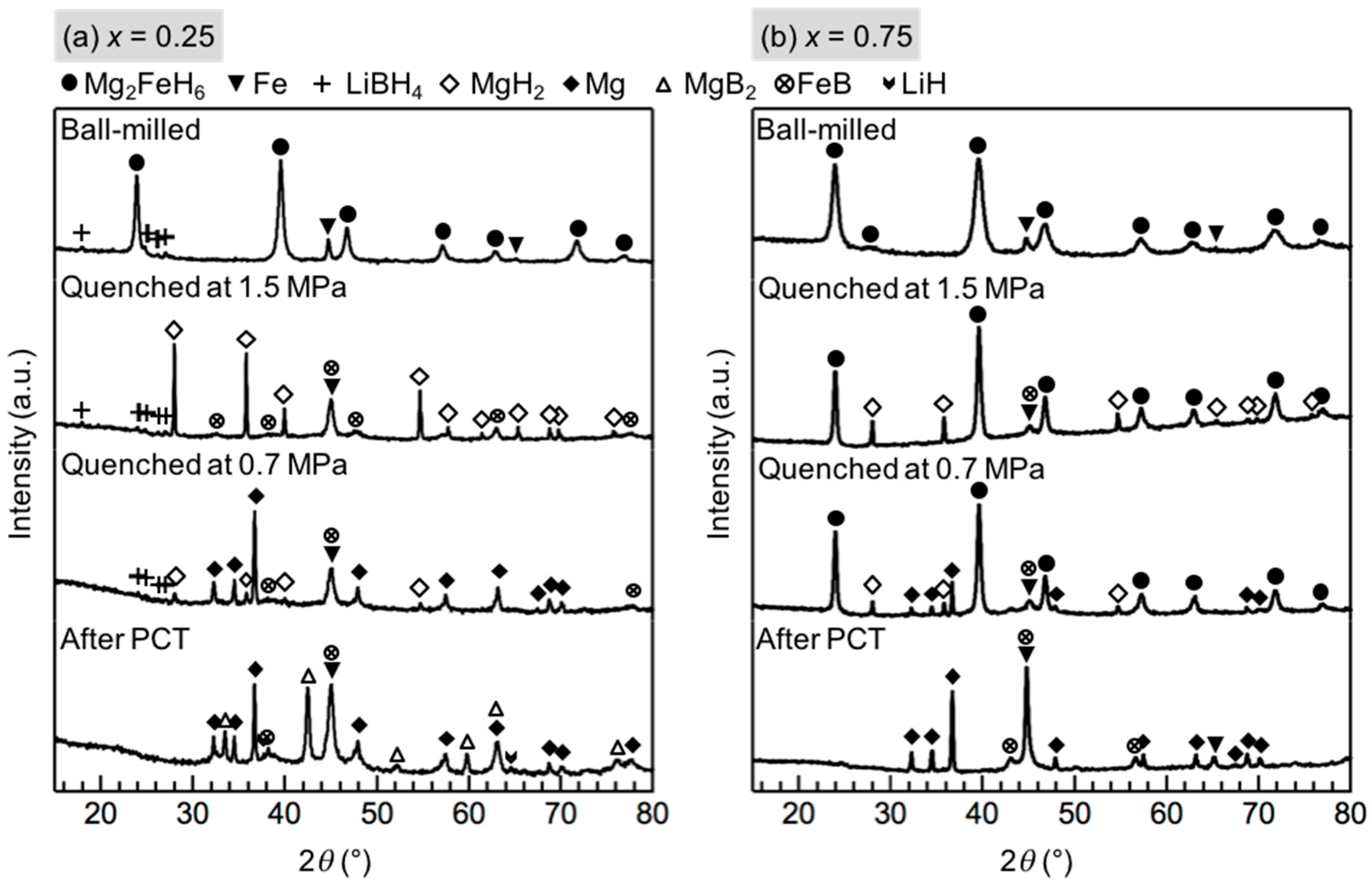
As shown in Figure 4a, for x = 0.25, the phases changes at the first and second reaction were almost the same as those of x = 0.5, except for the presented diffraction peaks of LiBH4 after the second reaction. The theoretical weight loss of Reactions (2) and (3) in x = 0.25 would be 2.9 mass % and 2.3 mass %, respectively. The actual weight loss of 2.5 mass % and 2.0 mass % at the first and second reaction, respectively, correlate with the theoretical value. The results indicate that the same stoichiometric reaction between LiBH4 and Mg2FeH6 occurred in x = 0.25, and the amount of LiBH4 in this composition is excessive for the stoichiometric reaction. An additional reaction with an incubation process occurred after the dehydrogenation of MgH2, and diffraction peaks of MgB2 appeared after this reaction. Therefore, the final isothermal reaction appears to be between LiBH4 and Mg, as shown in Equation (4).
2LiBH4 + Mg → 2LiH + MgB2 + 3H2
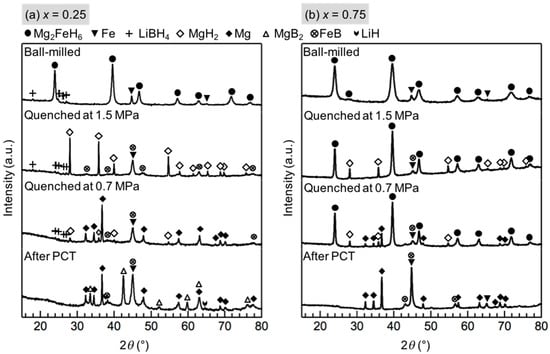
Figure 4.
XRD profiles of (a) x = 0.25 and (b) x = 0.75 composite during the PCT measurement at 643 K: the ball-milled material before the PCT measurement; the material quenched at 1.5 MPa, which is the pressure after the first isothermal reaction at 1.85 MPa; the material quenched at 0.7 MPa, which is the pressure after the second isothermal reaction at 0.85 MPa; and after the PCT measurement. The strongest diffraction peak of LiH overlapped with that of Fe at 44°.
In the case of x = 0.75, after the dehydrogenation of MgH2, diffraction peaks of Mg2FeH6 were still observed and finally disappeared after the last reaction, as shown in Figure 4b. Therefore, the last reaction can be attributed to the dehydrogenation of any residual Mg2FeH6:
Mg2FeH6 → 2Mg + Fe + 3H2
The equilibrium pressure of the dehydrogenation of Mg2FeH6, calculated using reference data, also supports this interpretation [22,23]. The same stoichiometric reaction between LiBH4 and Mg2FeH6, and the dehydrogenation of MgH2, also occurred in x = 0.75. They contributed 1.4 mass % and 1.1 mass % weight loss in theoretical, and 1.1 mass % and 1.1 mass % in practice, respectively.
2.2. Reversible Hydrogenation Due to the Optimal Composition Ratio
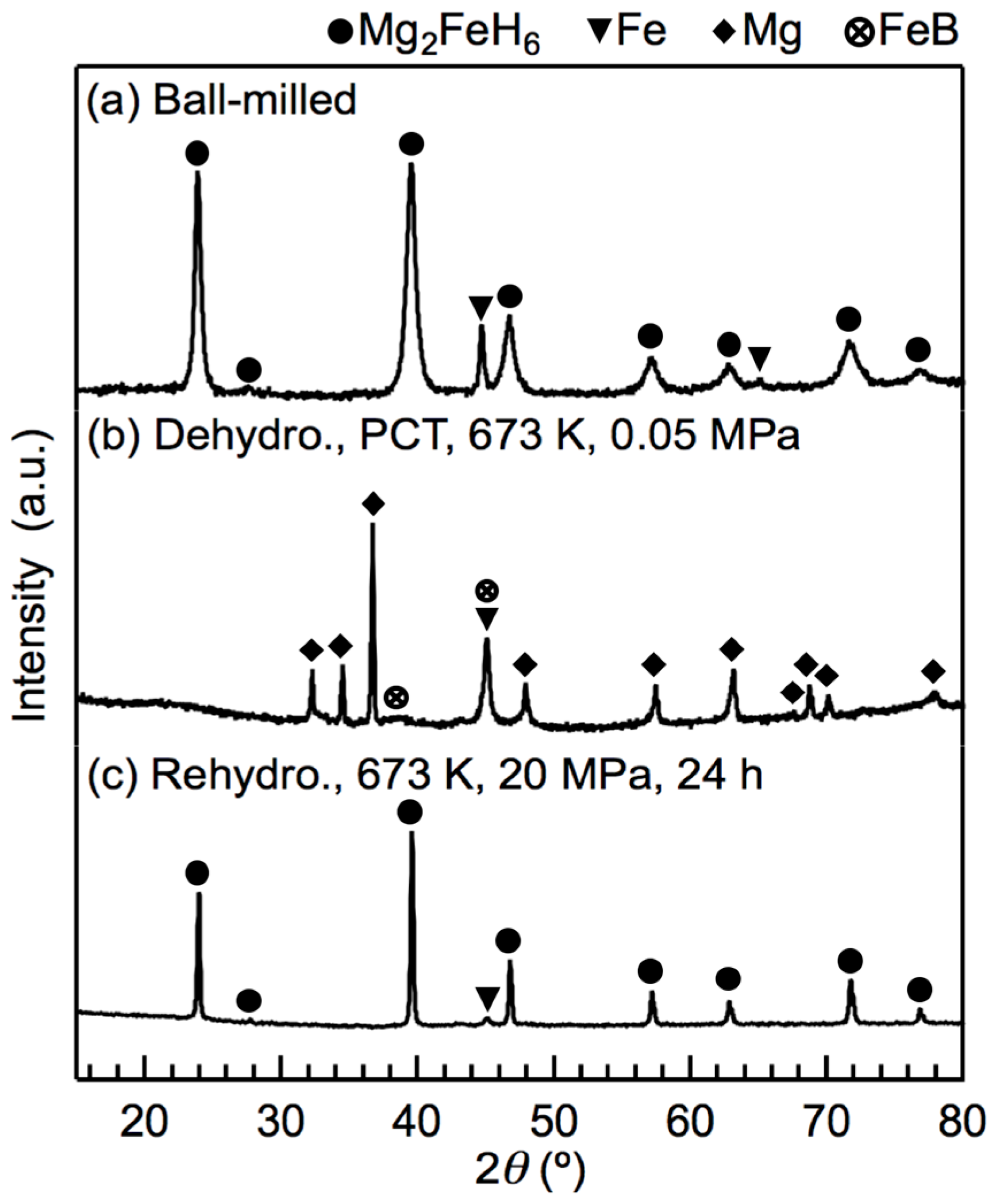
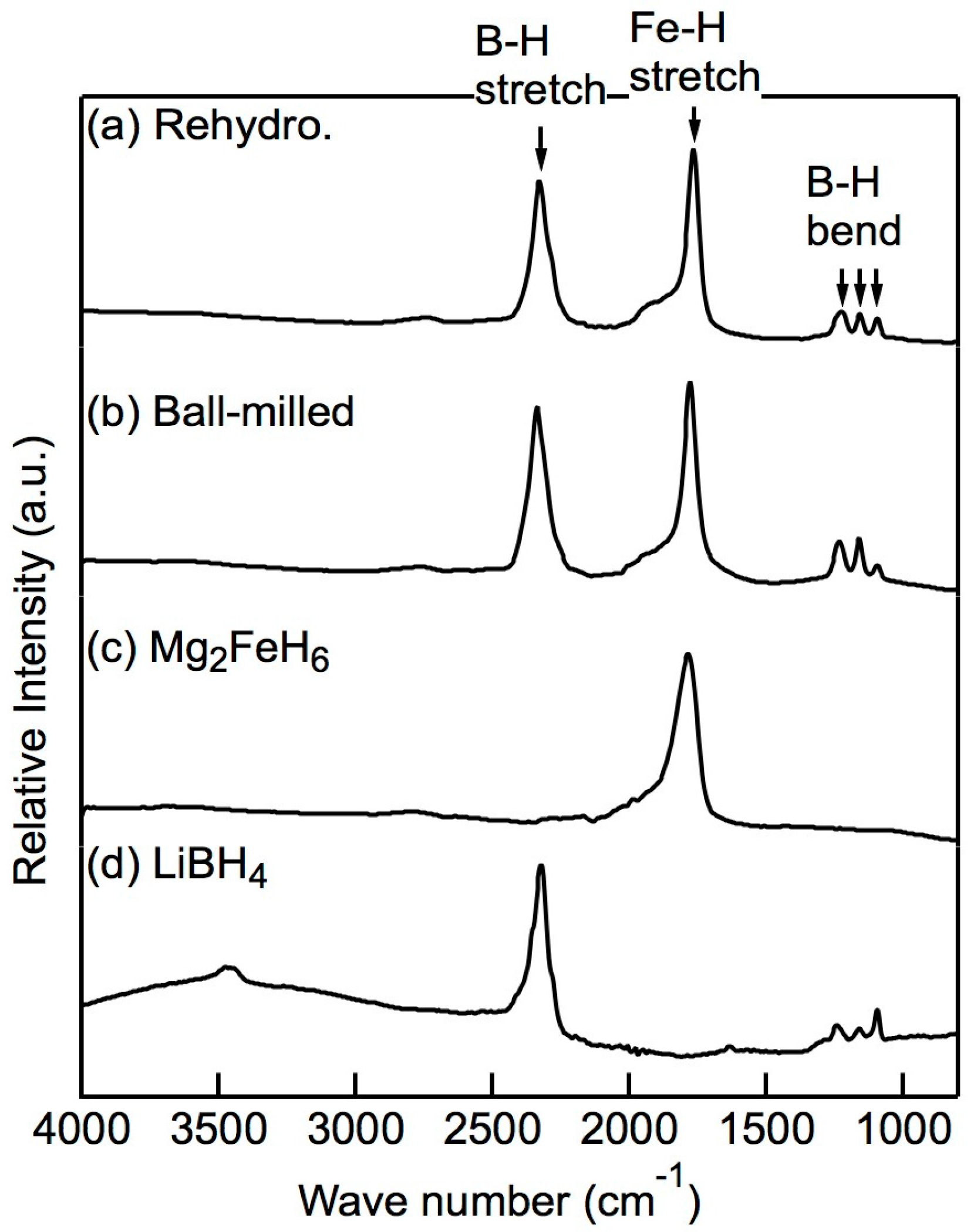
After attempting rehydrogenation at different temperatures and pressures, we confirmed that full rehydrogenation was achievable at 673 K and 20 MPa H2. The XRD profiles of the x = 0.5 composites before and after the rehydrogenation reaction at 673 K and 20 MPa H2 are shown in Figure 5. Before rehydrogenation, the material released H2 via a PCT program such that only Mg, LiH, Fe, and a small amount of FeB remained. After rehydrogenation, the XRD pattern showed the diffraction peaks of well-defined crystalline Mg2FeH6, indicating that Mg2FeH6 had been fully rehydrogenated. The weak diffraction peaks of α-Fe are attributed to the Fe located in the cores of the Mg2FeH6 grains, which, as reported previously [22,28], cannot be avoided. The diffraction peaks of Mg2FeH6 were sharp due to the fact that the heat treatment reduced the effect of the ball milling. LiBH4 was not observed from XRD after ball-milling nor was its presence detected after rehydrogenation. Therefore, we used infrared (IR) spectroscopy to detect the presence of LiBH4. As shown in Figure 6, the asymmetric stretching vibration mode and the bending mode of BH4 appeared in the spectra of the rehydrogenated materials at approximately 2329 cm−1 and 1234–1095 cm−1, respectively. The IR pattern correlated well with the data reported for pure LiBH4 [29], demonstrating that LiBH4 was present in the rehydrogenated material. The asymmetric stretching vibration of [FeH6]4− at 1785 cm−1 was also seen [30].
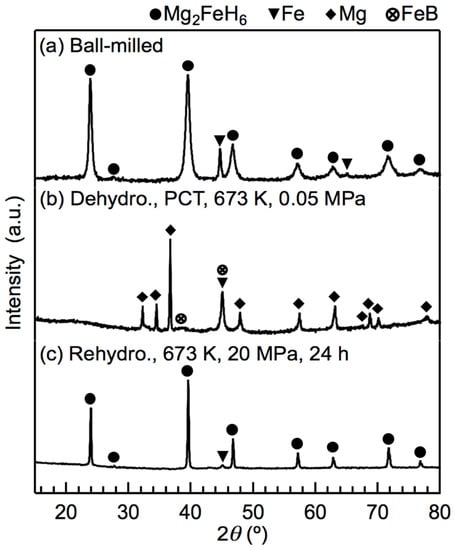
Figure 5.
XRD profiles of the x = 0.5 composite before and after rehydrogenation at 673 K and 20 MPa H2: (a) the material immediately after ball milling; (b) the material dehydrogenated at 673 K via a PCT measurement, and (c) the material rehydrogenated at 673 K and 20 MPa H2.
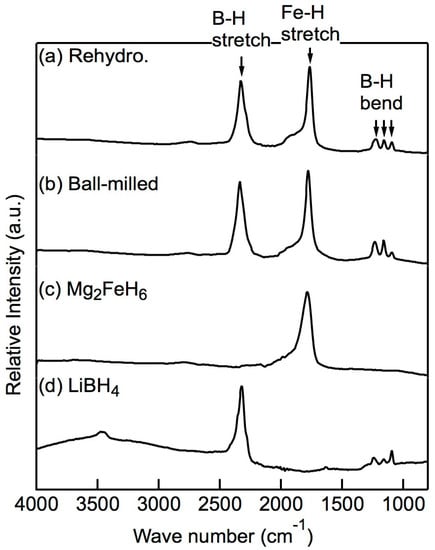
Figure 6.
Infrared (IR) spectra of the x = 0.5 composite: (a) directly after ball-milling, (b) after rehydrogenation. The IR spectra of (c) pure Mg2FeH6 and (d) pure LiBH4 are shown as references. The B–H vibration stretching at 2329 cm−1 and the bending vibration at 1234–1095 cm−1 demonstrate that LiBH4 in the composite materials was successfully rehydrogenated.
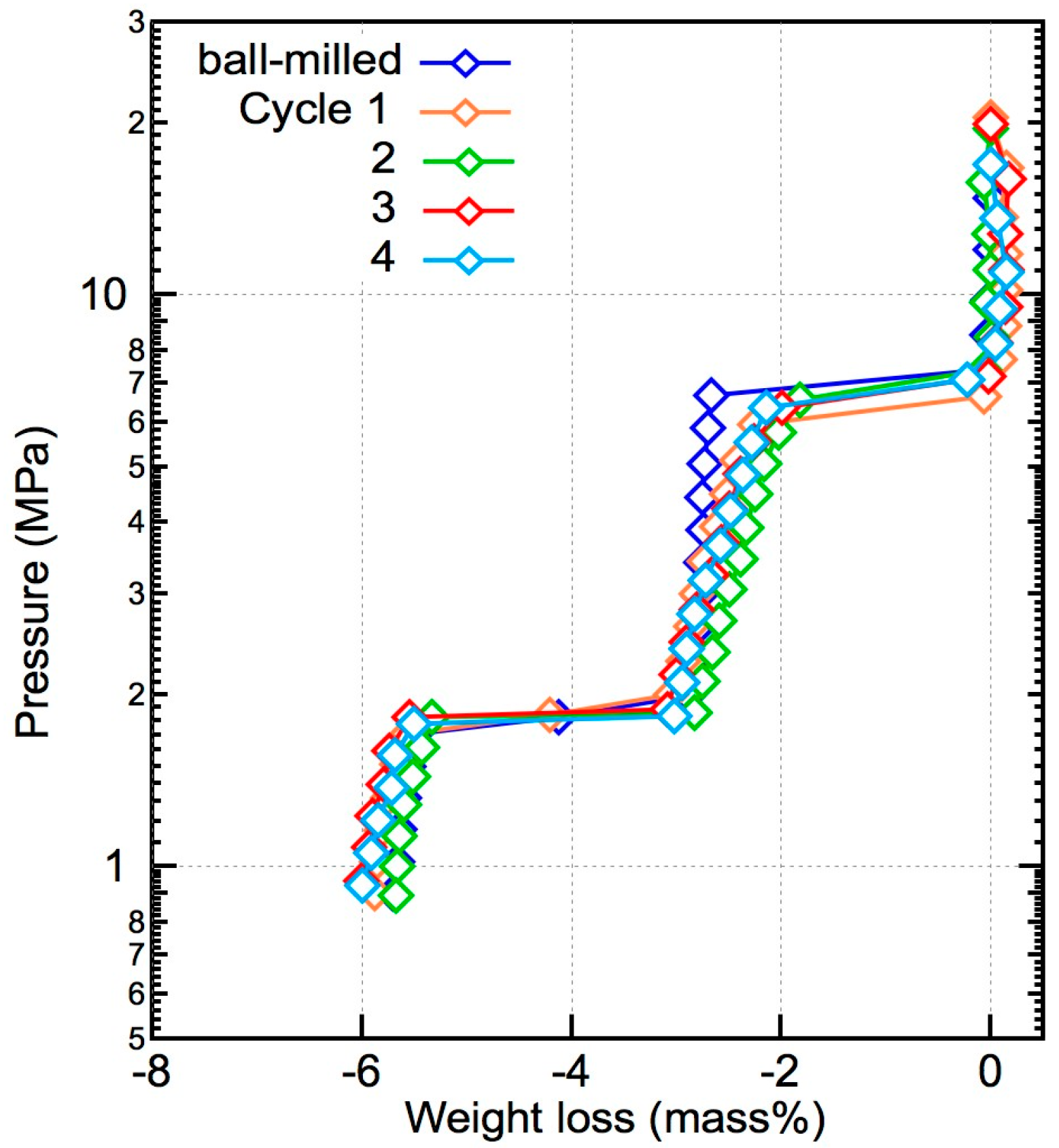
The PCT plots of the dehydrogenation part during the reversible hydrogenation tests at 673 K are shown in Figure 7. The equilibrium pressure of the reaction between LiBH4 and Mg2FeH6 at 6.8 MPa and that of the dehydrogenation reaction of MgH2 at 1.8 MPa remained stable, indicating that the properties of the composite materials did not degrade after cycling. The weight loss during each cycle did not decrease, suggesting that the composite material was fully rehydrogenated.
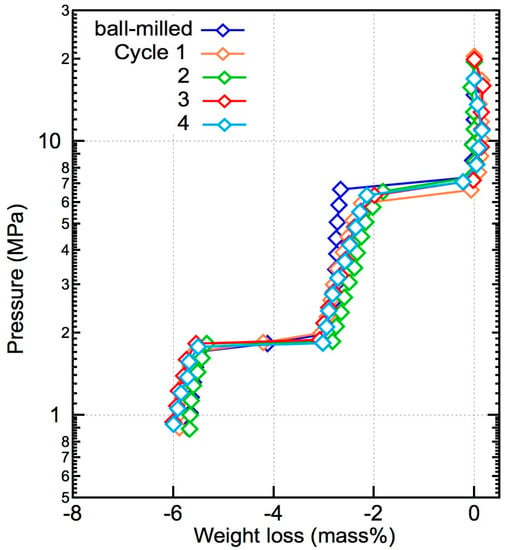
Figure 7.
De/rehydrogenation reversibility of the x = 0.5 composite. The PCT plots were constructed for the dehydrogenation process after rehydrogenation in each cycle. After four cycles, the H2 capacity of the materials remained the same.
3. Discussion
According to the PCT results, (1 − x)LiBH4 + xMg2FeH6 (x = 0.25, 0.5, and 0.75) shared the same reaction between LiBH4 and Mg2FeH6. This reaction was stoichiometric and its thermodynamic properties did not alter when excessive Mg2FeH6 or LiBH4 were present. The optimal composition for this stoichiometric reaction was x = 0.5, which can be explained by the formation of FeB. The kinetics of FeB formation was slow. For example, the reaction between LiBH4 and Mg2FeH6 in the x = 0.5 composite required more than 5 h to achieve a total hydrogen release of 3 mass %. The slow kinetics also caused the dehydrogenation process observed in the TG–MS and PCT measurements to exhibit different features. During the PCT measurements, the reaction between LiBH4 and Mg2FeH6 can be separated from the dehydrogenation of MgH2 for x = 0.5 and 0.75 because of the long reacting time. On the other hand, the high heating rate of 5 K·min−1 during the TG measurements in our previous report was not sufficient to incubate the reaction at its thermodynamically equilibrium temperature. According to theoretical data, it should be below 512 K (the equilibrium termperature at 0.1 MPa H2) [23,25,26,27], but in our previous work, the dehydrogenation of the composite materials was observed beyond 630 K. At this temperature, MgH2 was so unstable that the independent dehydrogenation of MgH2 no longer occurred. The thermodynamic property of Mg2FeH6 is similar to that of MgH2 [27]; thus, in the Mg2FeH6-rich compositions, independent dehydrogenation of Mg2FeH6 also no longer occurred and only one event appeared in the TG measurements for x ≥ 0.5, with which all of the hydrogen-containing species dehydrogenated simultaneously. LiBH4 is stable at the temperature of the simultaneous dehydrogenation. Therefore, for the x < 0.5 composite, the residue LiBH4 reacted with Mg or dehydrogenated independently after the simultaneous dehydrogenation in which several dehydrogenation events were observed. The activation energy of the reaction between LiBH4 and Mg2FeH6 should be different at the equilibrium reaction pressure and in the dynamic measurement. The detailed investigation will be carried out in a further study.
Ghaani et al. reported a composite reaction similar to that shown in Equation (2) in the LiBH4-rich composition [13,19]. Our results demonstrate that the reaction between LiBH4 and Mg2FeH6 was stoichiometric and did not degrade when excess LiBH4 or Mg2FeH6 was present. Also, an optimal composition ratio existed: x = 0.5, with which the independent dehydrogenation of the parent complex hydrides was avoided.
At the PCT experiment temperature of 643 K, the equilibrium pressure of the reaction between LiBH4 and Mg2FeH6 is 38 times or 4 times higher than that of the dehydrogenation reaction of pure LiBH4 or Mg2FeH6. This result indicates that both LiBH4 and Mg2FeH6 were thermodynamically destabilized in the composite. In other composite materials, such as that between LiBH4 and MgH2, only LiBH4 could be destabilized [3,20]. Another merit is that the H2 capacity and the dehydrogenation process remained stable even under an Ar atmosphere. For example, for x = 0.5 composite, all the H2 (6 mass %) was released in the temperature range 580 K to 630 K in the TG–MS experiments. However, an initial backpressure of at least 0.5 MPa H2 is needed for the incubation of the composite reaction between LiBH4 and MgH2 [31,32]. Also, the dehydrogenation process of the composite material of LiBH4 and Mg2NiH4 separated into more than three reactions and lasted from room temperature to beyond 673 K [6].
Full rehydrogenation of x = 0.5 composite requires much lower temperatures and pressures than those required in the partial rehydrogenation of LiBH4 (pressures as high as 35 MPa H2 and temperatures as high as 823 K [33]). Because previous studies on LiBH4-rich compositions did not achieve full rehydrogenation, we consider that the optimal ratio contributed to the good reversible hydrogenation performance of the composite material [17,19,34].
4. Materials and Methods
4.1. Synthesis of (1 − x)LiBH4 + xMg2FeH6
The synthesis of (1 − x)LiBH4 + xMg2FeH6 (x = 0.25, 0.5, and 0.75) occurred via ball milling of commercially available LiBH4 (purity ≥ 90%, Sigma-Aldrich, St. Louis, MO, USA) with laboratory-synthesized Mg2FeH6 (purity ≥ 90%) in an Ar atmosphere. The detailed synthesis method is described in the previous paper [12].
4.2. Pressure–Composition–Isothermal (PCT) Measurements
PCT measurements were conducted using a Sievert-type apparatus (Suzuki Shokan Co. Ltd., Tokyo, Japan, High-Pressure System Co., Saitama, Japan). The composite materials were heated to the designated temperature under a high H2 pressure (>10 MPa) to prevent decomposition before the measurements. During the dehydrogenation measurements, H2 was released in increments smaller than 0.01 MPa. The pressure change was kept stable between 15 min and 5 h to allow equilibration of the reaction at each step. Hydrogen weight loss was calculated using the function related to the pressure; volume of the apparatus, container, and material; and the temperatures at each part of the apparatus. The investigated temperature range was from 623 K to 673 K. The investigation of the rehydrogenation and reversible hydrogenation property of the optimized composition was performed in two steps. First, the dehydrogenation reaction was measured via PCT measurements using the same process described previously. Second, 20 MPa H2 was added to the container, which was subsequently maintained for 20–24 h at the same temperature. This two-step process was repeated four times.
4.3. Phase Identification
Phase identification was performed using powder XRD (PANalytical X’Pert-Pro, Almelo, The Netherlands, Cu Kα radiation, λ = 1.5405 Å) at room temperature. The microstructure and element distributions of the synthesized samples and the dehydrogenated products were recorded via SEM (JSM-6009, JEOL Ltd., Tokyo, Japan) using an instrument equipped for EDS (EX-54175JMH, JEOL Ltd., Tokyo, Japan). During the synthesis and measurement processes, the samples were always handled under Ar or vacuum to avoid contamination by air or water.
5. Conclusions
The isothermal dehydrogenation processes of (1 − x)LiBH4 + xMg2FeH6 (x = 0.25, 0.5, and 0.75) composite materials were studied using PCT measurements. The PCT results suggest that a stoichiometric reaction between LiBH4 and Mg2FeH6 occurred at all of the investigated composition ratios, i.e., LiBH4 + Mg2FeH6 → LiH + 2MgH2 + (Fe, FeB) + 5/2H2. Both LiBH4 and Mg2FeH6 were thermodynamically destabilized by this reaction. x = 0.5 is the optimal ratio for the composite material, which is also the reacting ratio of the two complex hydrides in the stoichiometric reaction. With the optimal ratio, the independent dehydrogenation of the excessive LiBH4 or Mg2FeH6 was avoided. During the dynamic TG–MS experiments, only one dehydrogenation event was observed for the x = 0.5 and 0.75 composites. The difference between the PCT and TG–MS results is explained by the slow kinetics of the reaction between LiBH4 and Mg2FeH6. Beside the dehydrogenation property, the optimal ratio also contributed to the enhanced reversible hydrogenation properties of the composite materials. The x = 0.5 composites can be de/rehydrogenated completely at 673 K and 20 MPa H2 for at least four cycles without the loss of H2 capacity.
Acknowledgments
This research has been financed by the Grant-in-Aid for Young Scientists (B) (17K14830), the Grant-in-Aid for Research Fellow of Japan Society for the Promotion of Science (15J10604), the JSPS KAKENHI Grant (25220911), and the German Federal Government under the European ERA-NET CONCERT Japan scheme via the iTHEUS project (grant CONCERT-EN-015). The authors would like to acknowledge Ms. N. Warifune for providing technical support.
Author Contributions
Guanqiao Li, Motoaki Matsuo, and Shin-ichi Orimo conceived and designed the experiments; Guanqiao Li and Anna-Lisa Chaudhary performed the experiments; Toyoto Sato and Shigeyuki Takagi helped analyze the data; Anna-Lisa Chaudhary and Martin Dornheim gave advice concerning the analysis of the data; and Guanqiao Li wrote the paper.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Paskevicius, M.; Jepsen, L.H.; Schouwink, P.; Cerny, R.; Ravnsbaek, D.B.; Filinchuk, Y.; Dornheim, M.; Besenbacher, F.; Jensen, T.R. Metal Borohydrides and Derivatives—Synthesis, Structure and Properties. Chem. Soc. Rev. 2017, 46, 1565–1634. [Google Scholar] [CrossRef] [PubMed]
- Callini, E.; Atakli, Z.O.K.; Hauback, B.C.; Orimo, S.; Jensen, C.; Dornheim, M.; Grant, D.; Cho, Y.W.; Chen, P.; Hjorvarsson, B.; et al. Complex and Liquid Hydrides for Energy Storage. Appl. Phys. A 2016, 122, 353. [Google Scholar] [CrossRef]
- Orimo, S.; Nakamori, Y.; Eliseo, J.R.; Zuttel, A.; Jensen, C.M. Complex Hydrides for Hydrogen Storage. Chem. Rev. 2007, 107, 4111–4132. [Google Scholar] [CrossRef] [PubMed]
- Li, H.W.; Yan, Y.G.; Orimo, S.; Züttel, A.; Jensen, C.M. Recent Progress in Metal Borohydrides for Hydrogen Storage. Energies 2011, 4, 185–214. [Google Scholar] [CrossRef]
- Bösenberg, U.; Doppiu, S.; Mosegaard, L.; Barkhordarian, G.; Eigen, N.; Borgschulte, A.; Jensen, T.R.; Cerenius, Y.; Gutfleisch, O.; Klassen, T.; et al. Hydrogen Sorption Properties of MgH2–LiBH4 Composites. Acta Mater. 2007, 55, 3951–3958. [Google Scholar] [CrossRef]
- Vajo, J.J.; Li, W.; Liu, P. Thermodynamic and Kinetic Destabilization in LiBH4–Mg2NiH4: Promise for Borohydride-Based Hydrogen Storage. Chem. Commun. 2010, 46, 6687–6689. [Google Scholar] [CrossRef] [PubMed]
- Javadian, P.; Zlotea, C.; Ghimbeu, C.M.; Latroche, M.; Jensen, T.R. Hydrogen Storage Properties of Nanoconfined LiBH4–Mg2NiH4 Reactive Hydride Composites. J. Phys. Chem. C 2015, 119, 5819–5826. [Google Scholar] [CrossRef]
- Yan, Y.G.; Li, H.W.; Maekawa, H.; Miwa, K.; Towata, S.; Orimo, S. Formation of Intermediate Compound Li2B12H12 during The Dehydrogenation Process of The LiBH4–MgH2 System. J. Phys. Chem. C 2011, 115, 19419–19423. [Google Scholar] [CrossRef]
- Bergemann, N.; Pistidda, C.; Milanese, C.; Emmler, T.; Karimi, F.; Chaudhary, A.L.; Chierotti, M.R.; Klassen, T.; Dornheim, M. Ca(BH4)2–Mg2NiH4: On The Pathway to A Ca(BH4)2 System with A Reversible Hydrogen Cycle. Chem. Commun. 2016, 52, 4836–4839. [Google Scholar] [CrossRef] [PubMed]
- Bosenberg, U.; Kim, J.W.; Gosslar, D.; Eigen, N.; Jensen, T.R.; von Colbe, J.M.B.; Zhou, Y.; Dahms, M.; Kim, D.H.; Gunther, R.; et al. Role of Additives in LiBH4–MgH2 Reactive Hydride Composites for Sorption Kinetics. Acta Mater. 2010, 58, 3381–3389. [Google Scholar] [CrossRef]
- Yang, J.; Sudik, A.; Wolverton, C. Destabilizing LiBH4 with A Metal (M = Mg, Al, Ti, V, Cr, or Sc) or Metal Hydride (MH2, MgH2, TiH2, or CaH2). J. Phys. Chem. C 2007, 111, 19134–19140. [Google Scholar] [CrossRef]
- Li, G.; Matsuo, M.; Deledda, S.; Sato, R.; Hauback, B.C.; Orimo, S. Dehydriding Property of LiBH4 Combined with Mg2FeH6. Mater. Trans. 2013, 54, 1532–1534. [Google Scholar] [CrossRef]
- Li, G.; Matsuo, M.; Aoki, K.; Ikeshoji, T.; Orimo, S. Dehydriding Process and Hydrogen-Deuterium Exchange of LiBH4–Mg2FeD6 Composites. Energies 2015, 8, 5459–5466. [Google Scholar] [CrossRef]
- Chaudhary, A.-L.; Li, G.; Matsuo, M.; Orimo, S.; Deledda, S.; Sørby, M.H.; Hauback, B.C.; Pistidda, C.; Klassen, T.; Dornheim, M. Simultaneous Desorption Behavior of M Borohydrides and Mg2FeH6 Reactive Hydride Composites (M = Mg, then Li, Na, K, Ca). Appl. Phys. Lett. 2015, 107, 073905. [Google Scholar] [CrossRef]
- Li, G.; Matsuo, M.; Deledda, S.; Hauback, B.C.; Orimo, S. Dehydriding Property of NaBH4 Combined with Mg2FeH6. Mater. Trans. 2014, 55, 1141–1143. [Google Scholar] [CrossRef]
- Pinkerton, F.E.; Meyer, M.S.; Meisner, G.P.; Balogh, M.P.; Vajo, J.J. Phase Boundaries and Reversibility of LiBH4–MgH2 Hydrogen Storage Material. J. Phys. Chem. C 2007, 111, 12881–12885. [Google Scholar] [CrossRef]
- Deng, S.S.; Xiao, X.Z.; Han, L.Y.; Li, Y.; Li, S.Q.; Ge, H.W.; Wang, Q.D.; Chen, L.X. Hydrogen Storage Performance of 5LiBH4 + Mg2FeH6 Composite System. Int. J. Hydrog. Energy 2012, 37, 6733–6740. [Google Scholar] [CrossRef]
- Langmi, H.W.; McGrady, G.S.; Newhouse, R.; Rönnebro, E. Mg2FeH6–LiBH4 and Mg2FeH6–LiNH2 Composite Materials for Hydrogen Storage. Int. J. Hydrog. Energy 2012, 37, 6694–6699. [Google Scholar] [CrossRef]
- Ghaani, M.R.; Catti, M.; Nale, A. Thermodynamics of Dehydrogenation of the 2LiBH4–Mg2FeH6 Composite. J. Phys. Chem. C 2012, 116, 26694–26699. [Google Scholar] [CrossRef]
- Bogdanovic, B.; Bohmhammel, K.; Christ, B.; Reiser, A.; Schlichte, K.; Vehlen, R.; Wolf, U. Thermodynamic Investigation of the Magnesium–Hydrogen System. J. Alloys Compd. 1999, 282, 84–92. [Google Scholar] [CrossRef]
- Bohmhammel, K.; Wolf, U.; Wolf, G.; Konigsberger, E. Thermodynamic Optimization of the System Magnesium–Hydrogen. Thermochim. Acta 1999, 337, 195–199. [Google Scholar] [CrossRef]
- Zhang, X.; Yang, R.; Qu, J.; Zhao, W.; Xie, L.; Tian, W.; Li, X. The Synthesis and Hydrogen Storage Properties of Pure Nanostructured Mg2FeH6. Nanotechnology 2010, 21, 095706. [Google Scholar] [CrossRef] [PubMed]
- Puszkiel, J.A.; Larochette, P.A.; Gennari, F.C. Thermodynamic and Kinetic Studies of Mg–Fe–H after Mechanical Milling Followed by Sintering. J. Alloys Compd. 2008, 463, 134–142. [Google Scholar] [CrossRef]
- Wang, Y.; Cheng, F.Y.; Li, C.S.; Tao, Z.L.; Chen, J. Preparation and Characterization of Nanocrystalline Mg2FeH6. J. Alloys Compd. 2010, 508, 554–558. [Google Scholar] [CrossRef]
- Siegel, D.J.; Wolverton, C.; Ozoliņš, V. Thermodynamic Guidelines for the Prediction of Hydrogen Storage Reactions and Their Application to Destabilized Hydride Mixtures. Phys. Rev. B 2007, 76. [Google Scholar] [CrossRef]
- Price, T.E.C.; Grant, D.M.; Telepeni, I.; Yu, X.B.; Walker, G.S. The Decomposition Pathways for LiBD4–MgD2 Multicomponent Systems Investigated by In Situ Neutron Diffraction. J. Alloys Compd. 2009, 472, 559–564. [Google Scholar] [CrossRef]
- Miwa, K.; Takagi, S.; Matsuo, M.; Orimo, S. Thermodynamical Stability of Complex Transition Metal Hydrides M2FeH6. J. Phys. Chem. C 2013, 117, 8014–8019. [Google Scholar] [CrossRef]
- Bogdanovic, B.; Reiser, A.; Schlichte, K.; Spliethoff, B.; Tesche, B. Thermodynamics and Dynamics of the Mg−Fe−H System and Its Potential for Thermochemical Thermal Energy Storage. J. Alloys Compd. 2002, 345, 77–89. [Google Scholar] [CrossRef]
- Zavorotynska, O.; Corno, M.; Damin, A.; Spoto, G.; Ugliengo, P.; Baricco, M. Vibrational Properties of MBH4 and MBF4 Crystals (M = Li, Na, K): A Combined DFT, Infrared, and Raman Study. J. Phys. Chem. C 2011, 115, 18890–18900. [Google Scholar] [CrossRef]
- Parker, S.F.; Williams, K.P.J.; Bortz, M.; Yvon, K. Inelastic Neutron Scattering, Infrared, and Raman Spectroscopic Studies of Mg2FeH6 and Mg2FeD6. Inorg. Chem. 1997, 36, 5218–5221. [Google Scholar] [CrossRef]
- Kim, K.B.; Shim, J.H.; Park, S.H.; Choi, I.S.; Oh, K.H.; Cho, Y.W. Dehydrogenation Reaction Pathway of the LiBH4−MgH2 Composite under Various Pressure Conditions. J. Phys. Chem. C 2015, 119, 9714–9720. [Google Scholar] [CrossRef]
- Bosenberg, U.; Ravnsbaek, D.B.; Hagemann, H.; D’Anna, V.; Minella, C.B.; Pistidda, C.; van Beek, W.; Jensen, T.R.; Bormann, R.; Dornheim, M. Pressure and Temperature Influence on the Desorption Pathway of the LiBH4−MgH2 Composite System. J. Phys. Chem. C 2010, 114, 15212–15217. [Google Scholar] [CrossRef]
- Orimo, S.; Nakamori, Y.; Kitahara, G.; Miwa, K.; Ohba, N.; Towata, S.; Zuttel, A. Dehydriding and Rehydriding Reactions of LiBH4. J. Alloys Compd. 2005, 404, 427–430. [Google Scholar] [CrossRef]
- Gosselin, C.; Deledda, S.; Hauback, B.C.; Huot, J. Effect of Synthesis Route on the Hydrogen Storage Properties of 2MgH2–Fe Compound Doped with LiBH4. J. Alloys Compd. 2015, 645, S304–S307. [Google Scholar] [CrossRef]
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).