We present the synthesis of [Zn
2(NH
3)
2(NH
2)
3]Cl and [Zn(NH
3)
2(NH
2)]Br, two ammoniates of zinc halide amides, both with tetrahedral coordination by ammonia and amide ligands at the Zn central atom. Similar simultaneous coordination by amide ions and ammonia molecules was previously reported in, for example, KNH
2·2NH
3 [
5], or more relevant for the discussion, [Cr
2(NH
2)
3(NH
3)
6]I
2 [
6] and InF
2(NH
2)(NH
3) [
7], both with octahedral coordination at the metal atom and obtained from the respective halides under ammonothermal conditions.
Both title compounds were obtained in the colder zone of the autoclave, while additional Zn(NH
2)
2 was observed in the hot zone. According to literature, Zn(NH
2)
2 is insoluble in liquid ammonia at ambient conditions [
8]; however, we have frequently observed that Zn reacts under various ammonoacidic as well as ammonobasic conditions to form the diamide and crystallizes in the colder zone of the autoclave in large crystals, indicating the ammonoamphoteric character of Zn and an enhanced solubility of the binary amide at elevated temperatures and pressures. However, it is apparently possible to adjust the solubility, respectively its temperature dependence, by the addition of halide ions and thus favor the formation of the supposedly less soluble title compounds in the zone with lower temperature. The deposition at the hotter or colder zone in the reaction vessel is usually dictated by the temperature dependence of the solubility. This temperature dependence can fundamentally change with the type of mineralizer and therefore with the nature of the dissolved species, as is well known, for example, for the ammonothermal synthesis of GaN [
1].
It is interesting to note that we were only able to synthesize the title compounds in the presence of platinum used as liner material to minimize contact of the solution with the autoclave wall and thus minimize corrosion, known to be severe in ammonoacidic solutions at elevated temperatures [
10]. A catalytic action of both NH
4Cl and Pt, for example, for the formation of alkali- and alkaline-earth metal amides from liquid ammonia is well established [
11].
2.1. Crystal Structure Description
[Zn
2(NH
3)
2(NH
2)
3]Cl crystallizes in the chiral space group
P2
12
12 with two formula units in the unit cell. A Flack parameter close to ½ indicates the presence of a racemic inversion twin.
Table 1 gives further selected information on the crystal structure and its determination.
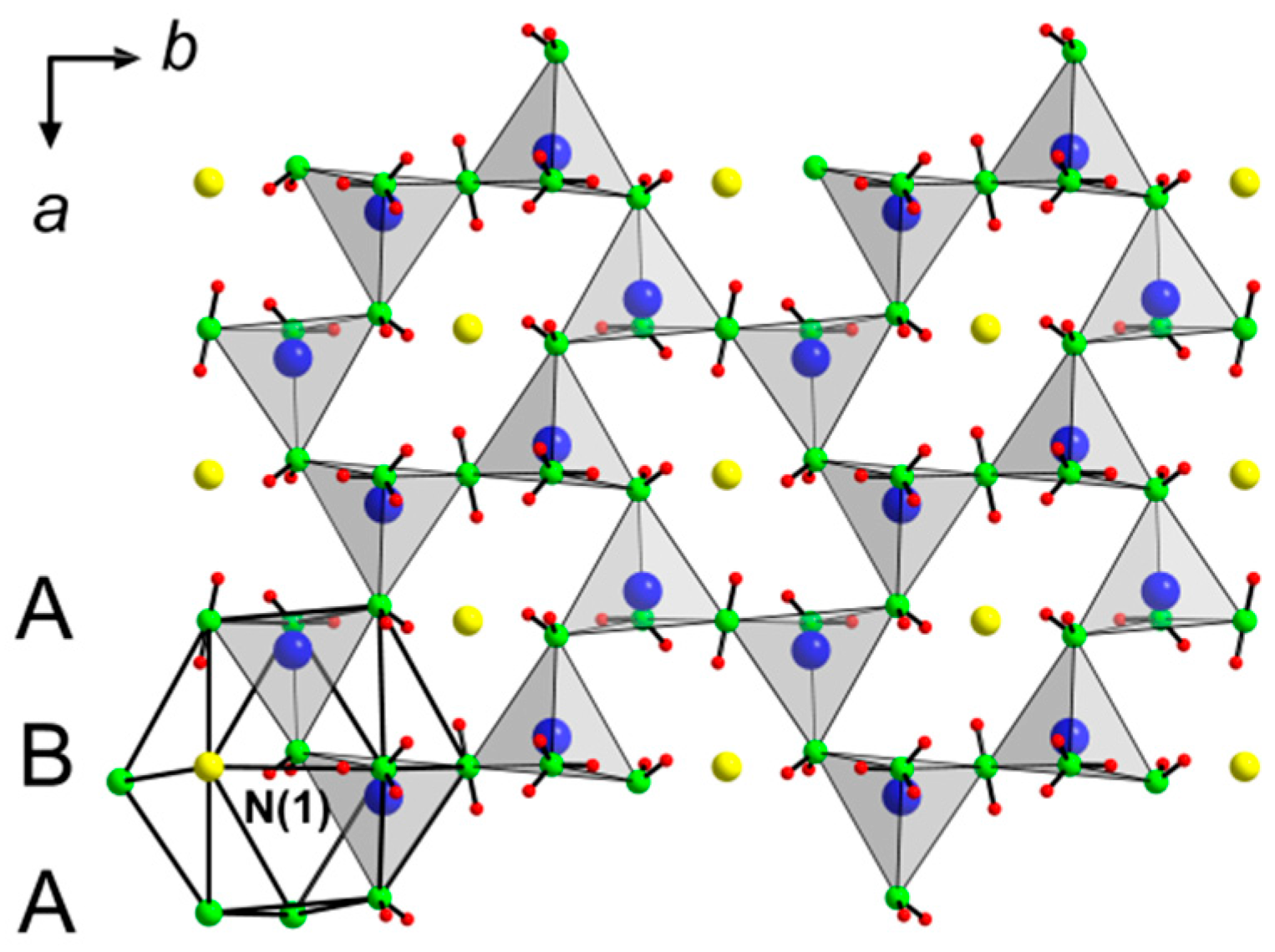
Table 2 presents positional parameters and
Table A1 anisotropic displacement parameters. In the crystal structure, the ammonia molecules, the amide and the chloride ions together form the motif of a hexagonal closed packing with stacking of hexagonal layers along [100]. In addition, 1/6 of the tetrahedral voids exclusively built by ammonia molecules and amide groups are occupied by Zn. These tetrahedra are linked via amide vertices to layers
orientated within the
a/
b plane (
Figure 1). As may be expected, the distances Zn–N to the amide groups with 199.1(5) pm and 201.2(6) pm are significantly shorter as compared to the distance to the NH
3 ligand (213.5(7) pm). Similar distance relations were earlier observed, e.g., in KNH
2·2NH
3 [
5] or [Cr
2(NH
2)
3(NH
3)
6]I
3 [
6]. Angles around Zn are in the range of 101.8° to 116.4°, thus close to the ideal tetrahedral angle. Further selected distances and angles are summarized in
Table 3.
The arrangement of tetrahedra within the layer
is reminiscent of the very similar interconnection within the structure of binary Zn(NH
2)
2, where six tetrahedra are condensed to rings, however, interconnected within a three-dimensional framework. According to the nomenclature after Liebau, developed to classify oxosilicate structures,
represents an unbranched
vierer single-layer with a molar ratio
n(Zn):
n(N) of 2:5 [
12].
Hydrogen atoms of the amide groups point towards the chloride ions located between the layers and form hydrogen bonds (
Figure 2). Every chloride ion connects to twelve hydrogen atoms, where half of the hydrogen atoms belong to each neighboring layer.
Table 4 summarizes donor–acceptor distances and angles.
[Zn(NH
3)
2(NH
2)]Br suffers from twinning by partial merohedry via two-fold rotation around the face diagonal [101] in space group
P12
1/
n1 setting. Due to application of the respective twin law, the reliability factors significantly drop (see the experimental part).
Table 1 summarizes selected information for the final structure refinement,
Table 5 gives positional parameters and
Table A2 anisotropic displacement parameters.
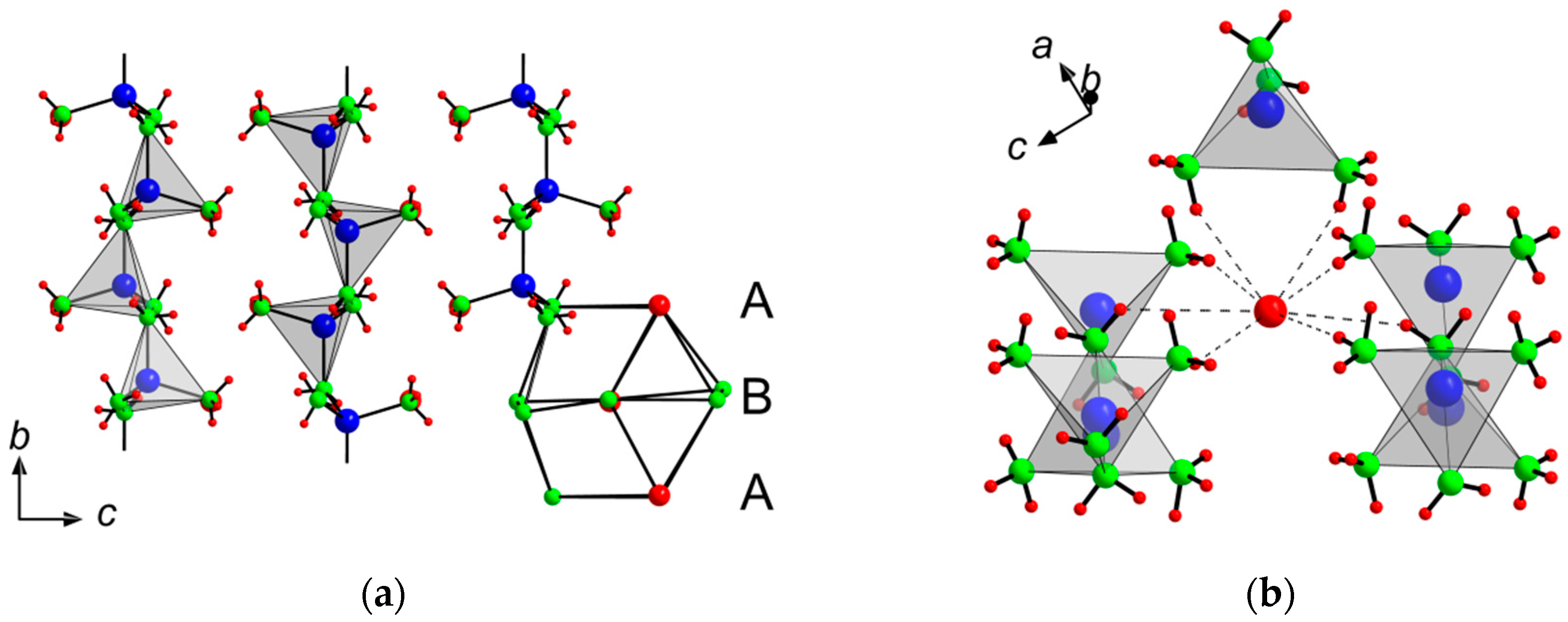
Very similar to [Zn
2(NH
3)
2(NH
2)
3]Cl, the ammonia molecules, amide and bromide ions in the crystal structure of [Zn(NH
3)
2(NH
2)]Br, form the motif of a hexagonal closed packing with stacking along [010] (
Figure 3). According to the composition, only 1/8 of the tetrahedral voids exclusively formed by two ammonia molecules and two amide ions are occupied by Zn. As discussed above for the chloride, the distances to the terminal ammonia ligands with 204(3) pm and 211(3) pm are significantly longer than those to the bridging amide ligands with 198.4(8) pm and 200.2(8) pm. Angles around Zn with 104°–123.8° deviate little from to the ideal tetrahedral angle.
Table 6 collects selected interatomic distances and angles. Vertex-sharing via amide ligands leads to
zigzag chains running along [010]. According to the Liebau nomenclature, these chains are classified as unbranched
zweier single-chains with molar ratio of
n(Zn):
n(N) = 1:3 [
12]. Once again, these chains may be viewed as sections from the three-dimensional crystal structure of binary zinc amide, formally broken up by the addition of HBr.
Bromide ions interconnect three
zigzag chains each via eight H-bonds (see
Figure 3).
Table 7 gives relevant donor–acceptor distances and angles. For both title compounds, there are no indications for a rotational disorder of the ammonia ligands, prohibited by an involvement in hydrogen bonding networks.
2.2. Raman Spectroscopy
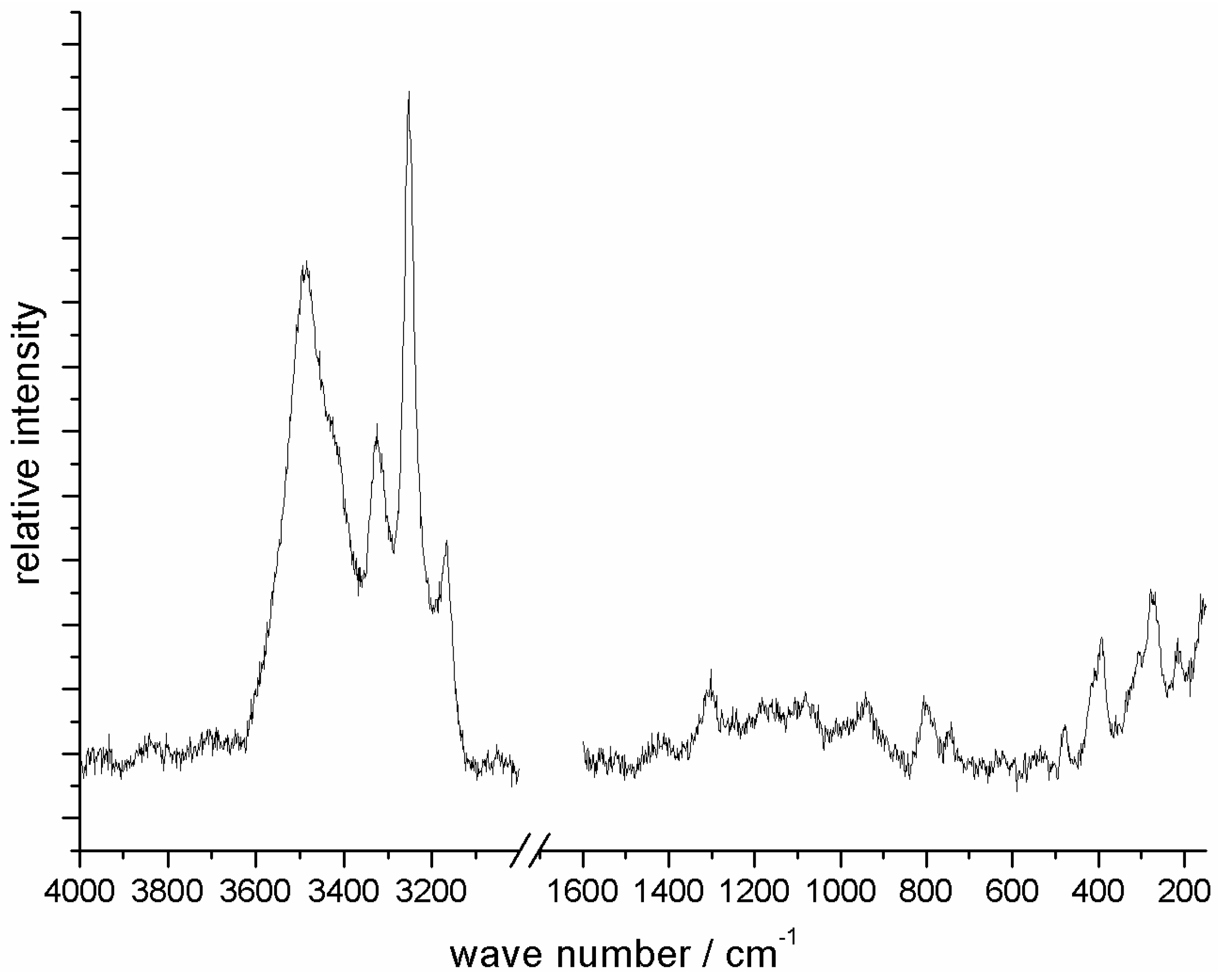
The Raman spectrum (
Figure 4) of a [Zn
2(NH
3)
2(NH
2)
3]Cl single crystal can be interpreted according to those of Zn(NH
3)
2Br
2 [
13], Zn(NH
3)
6Cl
2 [
14], [Zn(NH
3)
4]Br
2 [
4], [Zn(NH
3)
4]I
2 [
4,
15] as well as calculations for [Zn(NH
3)
4]
2+ [
16] and is in agreement with general trends for ammoniates of transition metal halides [
14,
17,
18]. Four groups of signals appear in the spectrum, of which three groups are due to modes of NH
3 and NH
2− molecules (3167–3493 cm
−1, 1301–1621 cm
−1, 737–1031 cm
−1) and one group evokes from Zn–N skeletal vibrations (134–476 cm
−1).
In the range of 3167–3493 cm
−1, three asymmetric and three symmetric valence vibrations appear in agreement with the three crystallographic different nitrogen sites of ammonia and amide ligands. The broadening of this group of signals indicates the presence of hydrogen bonds of relevant strength, which is in perfect agreement with short NH…Cl distances (see above) [
18,
19]. These hydrogen bonds are also the reason for a shift to smaller wave numbers as compared to the respective modes of the free ammonia molecule (3337 and 3450 cm
−1 [
20]). For ammoniates of zinc fluoride Zn(NH
3)
3F
2 and Zn(NH
3)
2F
2 (3093–3337 cm
−1 [
2]), a similar, but even larger shift was observed, due to the higher electronegativity of fluorine compared to chlorine, while the bromide [Zn(NH
3)
4]Br
2 (3194 cm
−1 [
4]) and the iodide [Zn(NH
3)
4]I
2 (3177 cm
−1 [
21]) exhibit smaller shifts in the symmetric valence mode.
The second range is due to the symmetric (1302 cm
−1) and asymmetric (1621 cm
−1) scissoring modes, which are not resolved for the different crystallographic sites. These vibrations also appear in the spectra of [Zn(NH
3)
4]Br
2 (1607 and 1246 cm
−1 in IR [
4]) and [Zn(NH
3)
4]I
2 (1615, 1600, 1256 and 1242 cm
−1 [
21]). In comparison to the ammoniates of zinc fluoride, these signals are shifted to lower wave numbers, again as a result of reduced hydrogen bond strength. In the third region (1031–737 cm
−1), rocking and wagging modes are present. The rocking vibrations are not present in the spectrum of free ammonia molecules, but are known to increase in intensity with increasing covalent character in complexes [
22]. The last range (476–134 cm
−1) is due to Zn–N skeleton vibrations.
The absence of signals above 3500 cm
−1 indicates the absence of significant impurities of OH
− and H
2O, which should provoke a prominent mode at around 3700 cm
−1 [
23].