Spatial-Frequency Domain Imaging: An Emerging Depth-Varying and Wide-Field Technique for Optical Property Measurement of Biological Tissues
Abstract
1. Introduction
2. Principles and Methods
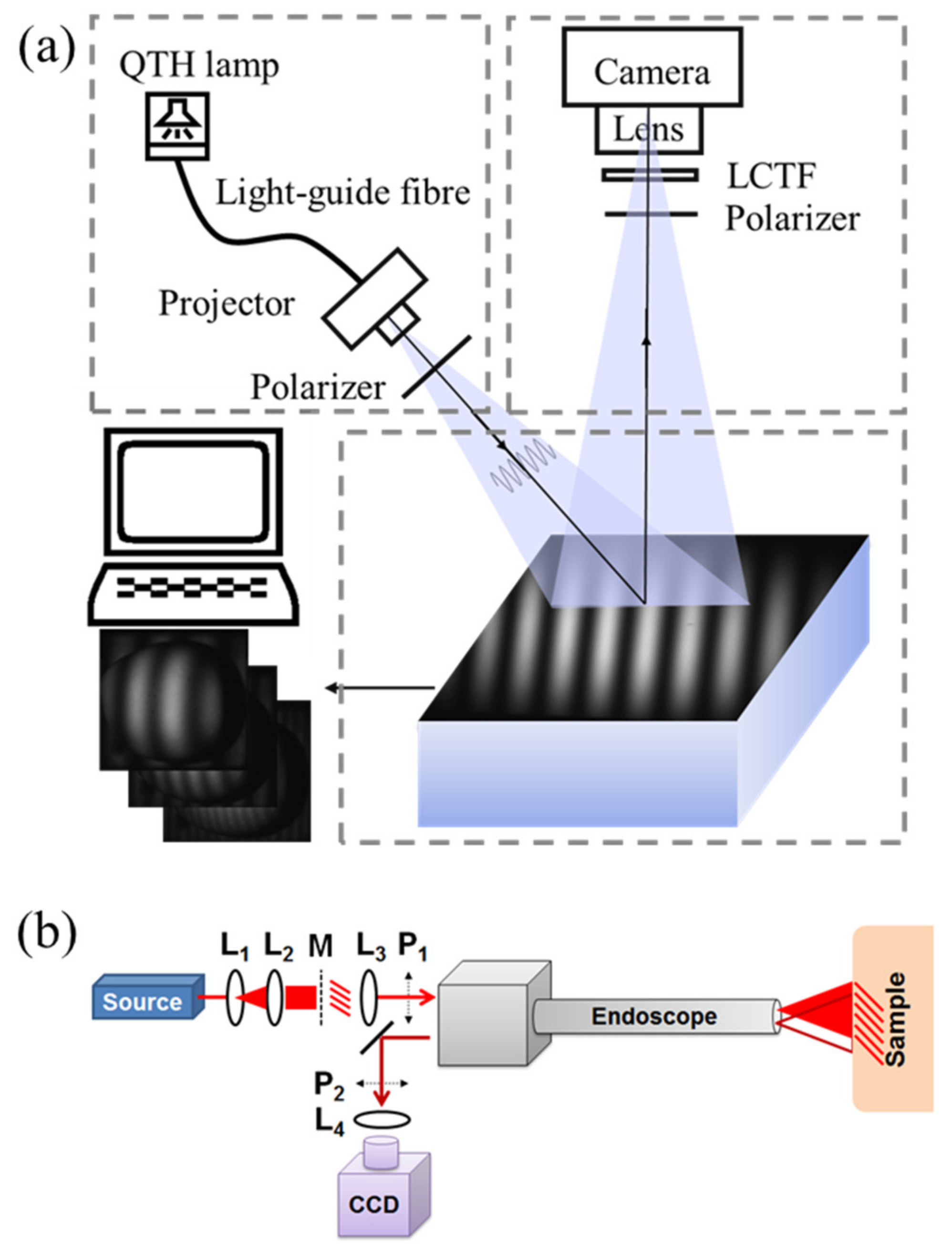
2.1. Typical SFDI System
2.2. Principle of SFDI for Estimating Optical Properties
3. Applications
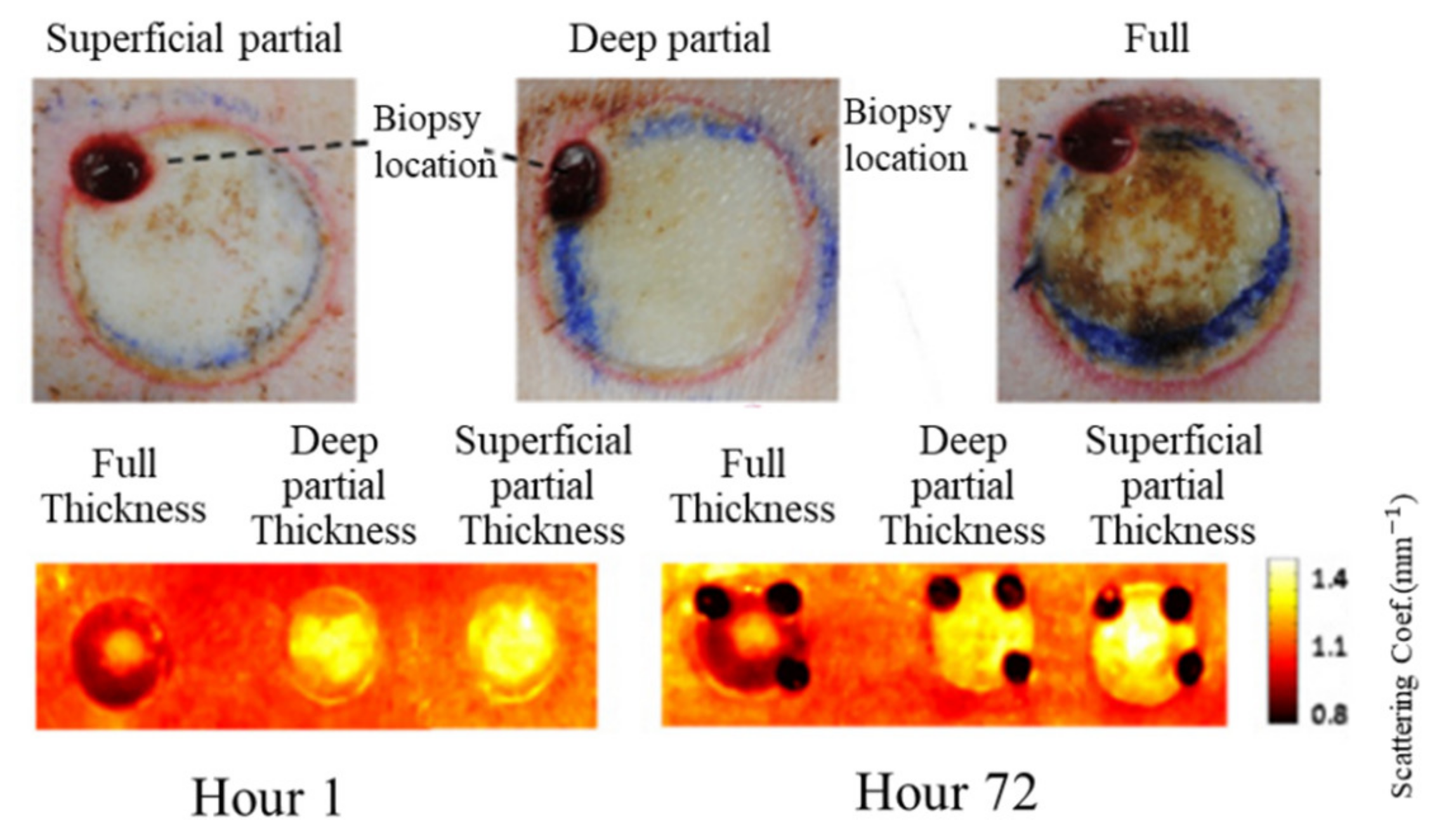
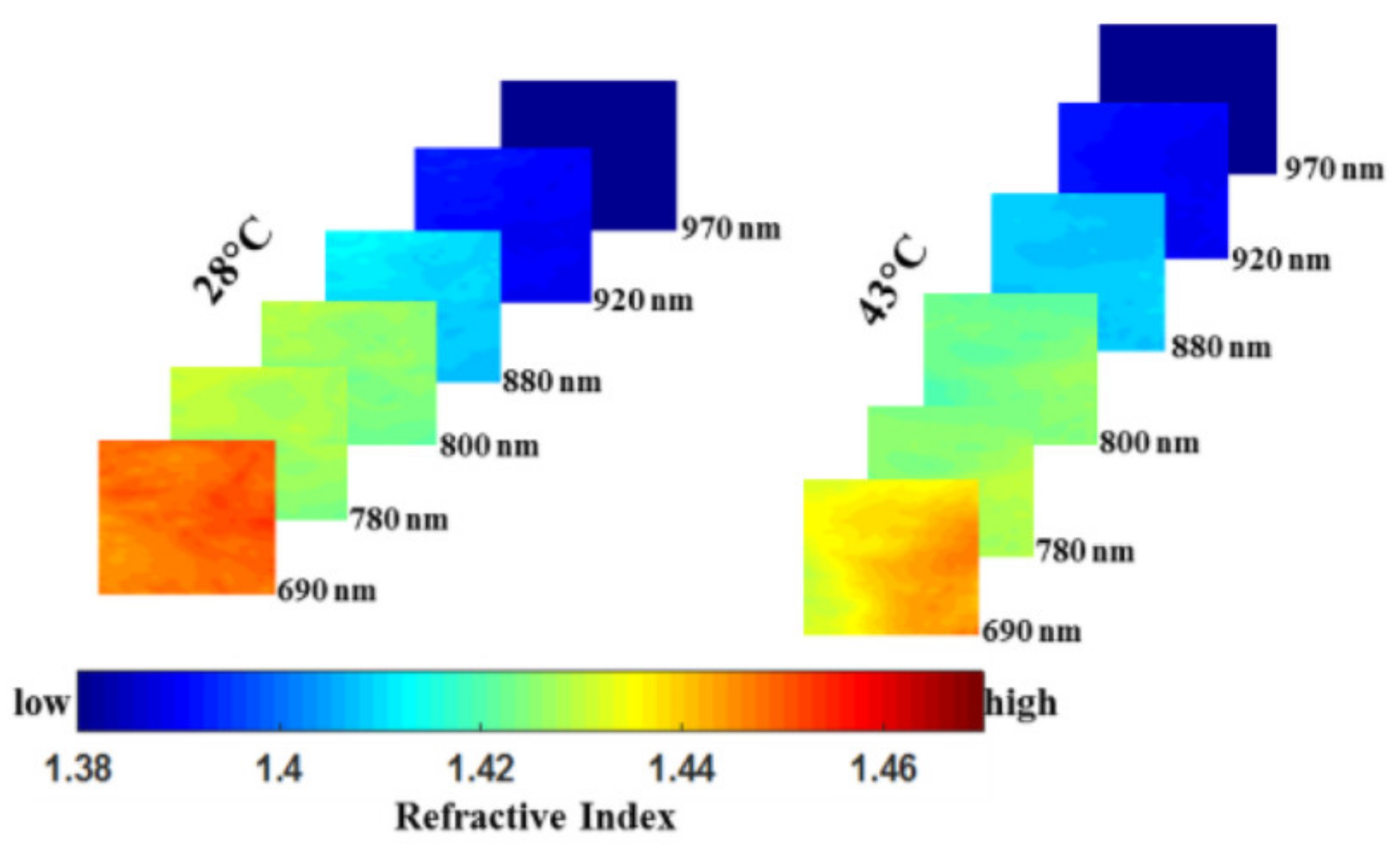
3.1. Burn Assessment
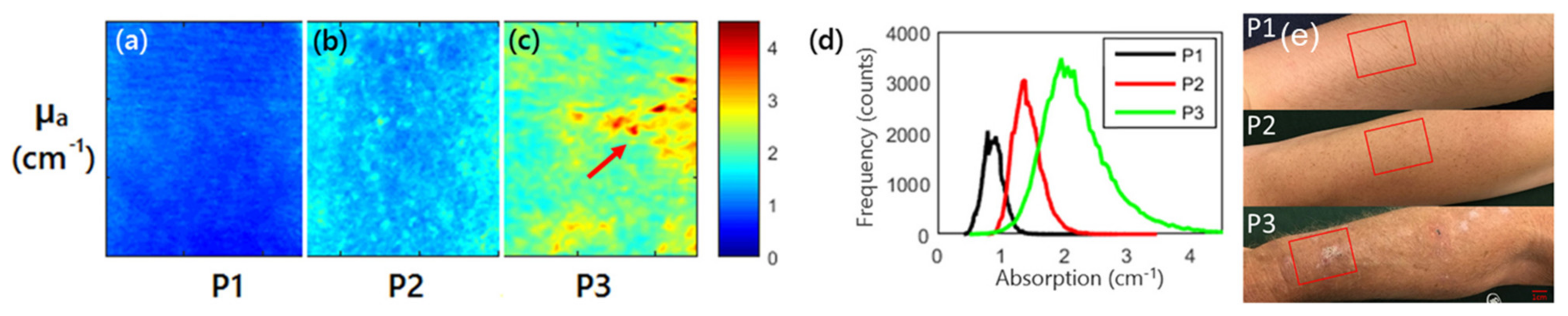
3.2. Skin Tissue Evaluation
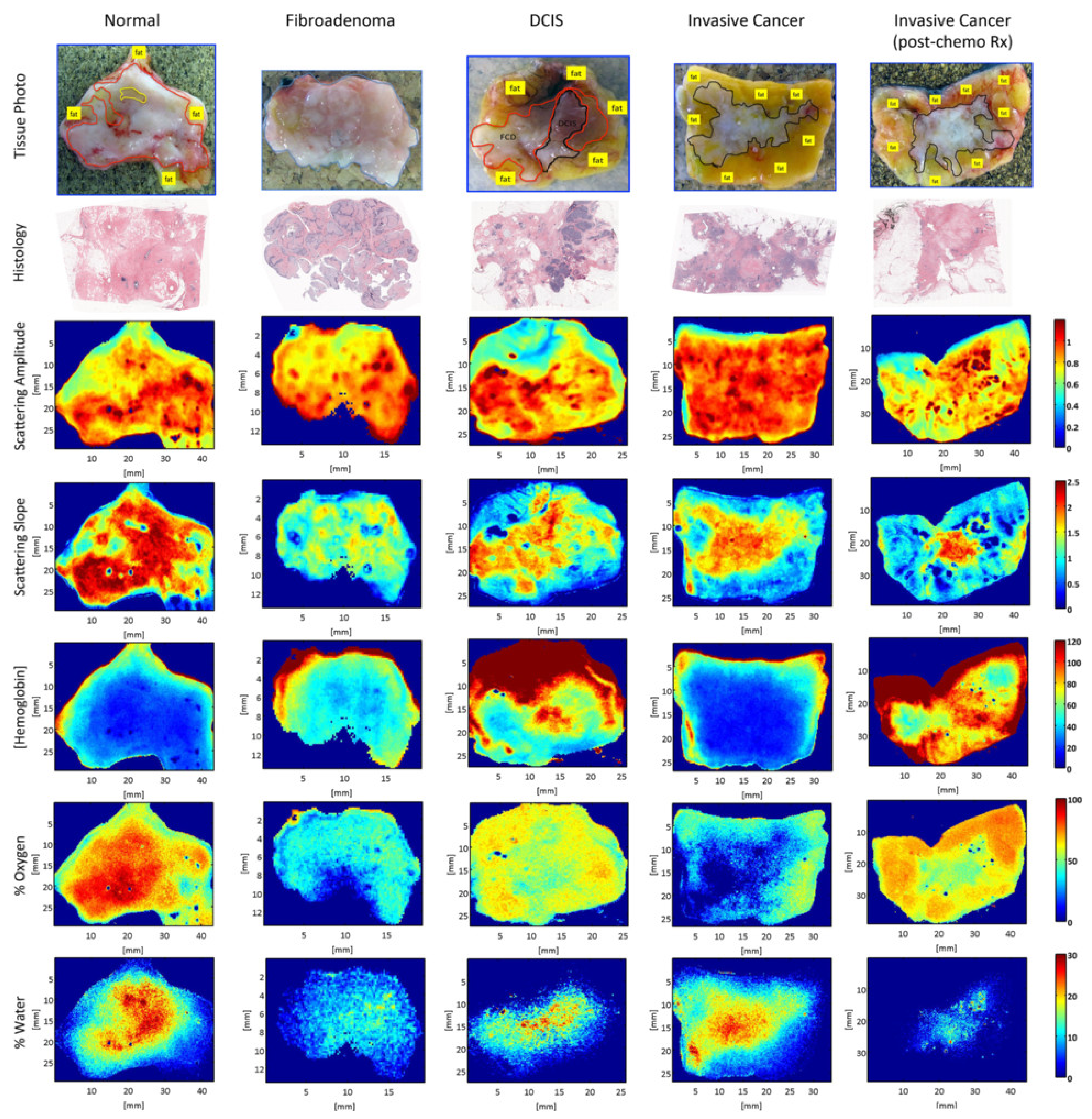
3.3. Tumor Tissue Detection
3.4. Brain Tissue Monitoring
3.5. Quality Evaluation of Agro-Products
4. Challenges and Future Perspectives
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Chen, B.; Tong, C.M. Modified physical optics algorithm for near field scattering. Chin. Phys. B 2018, 27, 114102–114106. [Google Scholar] [CrossRef]
- Shi, R.; Hellmann, C.; Wyrowski, F. Physics optics propagation through curved surfaces. J. Opt. Soc. Am. A 2019, 36, 1252–1260. [Google Scholar] [CrossRef] [PubMed]
- Liemert, A.; Reitzle, D.; Kienle, A. Analytical solutions of the radiative transport equation for turbid and fluorescent layered media. Sci. Rep. 2017, 7, 3819. [Google Scholar] [CrossRef]
- Liemert, A.; Kienle, A. Green’s function of the time-dependent radiative transport equation in terms of rotated spherical harmonics. Phys. Rev. E 2012, 86, 036603. [Google Scholar] [CrossRef]
- Liemert, A.; Kienle, A. Spatially modulated light source obliquely incident on a semi-infinite scattering medium. Opt. Lett. 2012, 37, 4158–4160. [Google Scholar] [CrossRef] [PubMed]
- Liemert, A.; Kienle, A. Exact and efficient solution of the radiative transport equation for the semi-infinite medium. Sci. Rep. 2013, 3, 2018. [Google Scholar] [CrossRef] [PubMed]
- Liemert, A.; Geiger, S.; Kienle, A. Solutions for single-scattered radiance in the semi-infinite medium based on radiative transport theory. J. Opt. Soc. Am. A 2021, 38, 405–411. [Google Scholar] [CrossRef] [PubMed]
- Ostermeyer, M.R.; Jacques, S.L. Perturbation theory for diffuse light transport in complex biological tissues. J. Opt. Soc. Am. A 1997, 14, 255–261. [Google Scholar] [CrossRef]
- Fan, S.X.; Li, C.Y.; Huang, W.Q.; Chen, L.P. Detection of blueberry internal bruising over time using NIR hyperspectral reflectance imaging with optimum wavelengths. Postharvest Biol. Technol. 2017, 134, 55–66. [Google Scholar] [CrossRef]
- Anderson, E.R.; Cuccia, D.J.; Durkin, A.J. Detection of bruises on golden delicious apples using spatial- frequency-domain imaging. Proc. SPIE Int. Soc. Opt. Eng. 2007, 36, 6430. [Google Scholar] [CrossRef]
- Vanoli, M.; Van Beers, R.; Sadar, N.; Rizzolo, A.; Buccheri, M.; Grassi, M.; Lovati, F.; Nicolaï, B.; Aernouts, B.; Watté, R.; et al. Time- and spatially-resolved spectroscopy to determine the bulk optical properties of ‘Braeburn’ apples after ripening in shelf life. Postharvest Biol. Technol. 2020, 168. [Google Scholar] [CrossRef]
- Wang, L.V.; Wu, H.-I.; Masters, B.R. Biomedical Optics, Principles and Imaging. J. Biomed. Opt. 2008. [Google Scholar] [CrossRef]
- Wilson, B.C.; Patterson, M.S.; Flock, S.T. Indirect versus direct techniques for the measurement of the optical properties of tissues. Photochem. Photobiol. 1987, 46, 601–608. [Google Scholar] [CrossRef]
- Bashkatov, A.N.; Genina, E.A.; Kochubey, V.I.; Tuchin, V.V. Optical properties of human skin, subcutaneous and mucous tissues in the wavelength range from 400 to 2000 nm. J. Phys. D Appl. Phys. 2005, 38, 2543. [Google Scholar] [CrossRef]
- Cheong, W.F.; Prahl, S.A.; Welch, A.J. A review of the optical properties of biological tissues. IEEE J. Quantum. Elect. 1990, 26, 2166–2185. [Google Scholar] [CrossRef]
- Rohrbach, D.J.; Muffoletto, D.; Huihui, J.; Saager, R.; Keymel, K.; Paquette, A.; Morgan, J.; Zeitouni, N.; Sunar, U. Preoperative Mapping of Nonmelanoma Skin Cancer Using Spatial Frequency Domain and Ultrasound Imaging. Acad. Radiol. 2014, 21, 263–270. [Google Scholar] [CrossRef]
- Lin, A.J.; Koike, M.A.; Green, K.N.; Kim, J.G.; Mazhar, A.; Rice, T.B.; LaFerla, F.M.; Tromberg, B.J. Spatial Frequency Domain Imaging of Intrinsic Optical Property Contrast in a Mouse Model of Alzheimer’s Disease. Ann. Biomed. Eng. 2011, 39, 1349–1357. [Google Scholar] [CrossRef] [PubMed]
- Mazhar, A.; Sharif, S.A.; Cuccia, J.D.; Nelson, J.S.; Kelly, K.M.; Durkin, A.J. Spatial frequency domain imaging of port wine stain biochemical composition in response to laser therapy: A pilot study. Laser. Surg. Med. 2012, 44, 611–621. [Google Scholar] [CrossRef] [PubMed]
- Ponticorvo, A.; Burmeister, D.M.; Rowland, R.; Baldado, M.; Kennedy, G.T.; Saager, R.; Bernal, N.; Choi, B.; Durkin, A.J. Quantitative long-term measurements of burns in a rat model using Spatial Frequency Domain Imaging (SFDI) and Laser Speckle Imaging (LSI). Laser. Surg. Med. 2017, 49, 293–304. [Google Scholar] [CrossRef] [PubMed]
- Cuccia, D.J.; Bevilacqua, F.; Durkin, A.J.; Ayers, F.R.; Tromberg, B.J. Quantitation and mapping of tissue optical properties using modulated imaging. J. Biomed. Opt. 2009, 14, 024012. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.Z.; Li, R.; Lu, R.F. Gram–Schmidt orthonormalization for retrieval of amplitude images under sinusoidal patterns of illumination. Appl. Opt. 2016, 55, 6866–6873. [Google Scholar] [CrossRef]
- Lu, Y.Z.; Li, R.; Lu, R.F. Fast demodulation of pattern images by spiral phase transform in structured-illumination reflectance imaging for detection of bruises in apples. Comput. Electron. Agric. 2016, 127, 652–658. [Google Scholar] [CrossRef]
- Bassi, A.; D’Andrea, C.; Valentini, G.; Cubeddu, R.; Arridge, S. Temporal propagation of spatial information in turbid media. Opt. Lett. 2008, 33, 2836. [Google Scholar] [CrossRef]
- Wang, L.V.; Wu, H.-I. Biomedical Optics: Principles and Imaging; John Wiley & Sons: Hoboken, NJ, USA, 2007. [Google Scholar]
- Prahl, S.A.; Vangemert, M.J.C.; Welch, A.J. Determining the optical properties of turbid media by using the adding-doubling method. Appl. Opt. 1993, 32, 559–568. [Google Scholar] [CrossRef] [PubMed]
- Patterson, M.S.; Chance, B.; Wilson, B.C. Time resolved reflectance and transmittance for the non-invasive measurement of tissue optical properties. Appl. Opt. 1989, 28, 2331–2336. [Google Scholar] [CrossRef] [PubMed]
- Patterson, M.S.; Moulton, J.D.; Wilson, B.C.; Berndt, K.W.; Lakowicz, J.R. Frequency-domain reflectance for the determination of the scattering and absorption properties of tissue. Appl. Opt. 1991, 30, 4474–4476. [Google Scholar] [CrossRef]
- Farrell, T.J.; Patterson, M.S.; Wilson, B. A diffusion theory model of spatially resolved, steady-state diffuse reflectance for the noninvasive determination of tissue optical properties in vivo. Med. Phys. 1992, 19, 879–888. [Google Scholar] [CrossRef] [PubMed]
- Kienle, A.; Patterson, M.S. Improved solutions of the steady-state and the time-resolved diffusion equations for reflectance from a semi-infinite turbid medium. J. Opt. Soc. Am. A 1997, 14, 246–254. [Google Scholar] [CrossRef]
- Cuccia, D.J.; Bevilacqua, F.; Durkin, A.J.; Tromberg, B.J. Modulated imaging: Quantitative analysis and tomography of turbid media in the spatial-frequency domain. Opt. Lett. 2005, 30, 1354–1359. [Google Scholar] [CrossRef]
- Wirth, D.; Sibai, M.; Olson, J.; Wilson, B.C.; Roberts, D.W.; Paulsen, K. Feasibility of using spatial frequency-domain imaging intraoperatively during tumor resection. J. Biomed. Opt. 2019, 24, 1–6. [Google Scholar] [CrossRef]
- Nothelfer, S.; Bergmann, F.; Liemert, A.; Reitzle, D.; Kienle, A. Spatial frequency domain imaging using an analytical model for separation of surface and volume scattering. J. Biomed. Opt. 2019, 24, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Kennedy, G.T.; Stone, R.I.; Kowalczewski, A.C.; Rowland, R.; Chen, J.H.; Baldado, M.L.; Ponticorvo, A.; Bernal, N.; Christy, R.J.; Durkin, A.J. Spatial frequency domain imaging: A quantitative, noninvasive tool for in vivo monitoring of burn wound and skin graft healing. J. Biomed. Opt. 2019, 24, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Laughney, A.M.; Krishnaswamy, V.; Rice, T.B.; Cuccia, D.J.; Barth, R.J.; Tromberg, B.J.; Paulsen, K.D.; Pogue, B.W.; Wells, W.A. System analysis of spatial frequency domain imaging for quantitative mapping of surgically resected breast tissues. J. Biomed. Opt. 2013, 18, 036012–036022. [Google Scholar] [CrossRef] [PubMed]
- Gioux, S.; Mazhar, A.; Lee, B.T.; Lin, S.J.; Tobias, A.M.; Cuccia, D.J.; Stockdale, A.; Oketokoun, R.; Ashitate, Y.; Kelly, E.; et al. First-in-human pilot study of a spatial frequency domain oxygenation imaging system. J. Biomed. Opt. 2011, 16, 086015. [Google Scholar] [CrossRef]
- Balu, M.; Mazhar, A.; Hayakawa, C.K.; Mittal, R.; Krasieva, T.B.; Konig, K.; Venugopalan, V.; Tromberg, B.J. In vivo multiphoton NADH fluorescence reveals depth-dependent keratinocyte metabolism in human skin. Biophys. J. 2013, 104, 258–267. [Google Scholar] [CrossRef]
- Mazhar, A.; Saggese, S.; Pollins, A.C.; Cardwell, N.L.; Nanney, L.; Cuccia, D.J. Noncontact imaging of burn depth and extent in a porcine model using spatial frequency domain imaging. J. Biomed. Opt. 2014, 19, 086019–086028. [Google Scholar] [CrossRef] [PubMed]
- Gioux, S.; Mazhar, A.; Cuccia, D.J.; Durkin, A.J.; Tromberg, B.J.; Frangioni, J.V. Three-dimensional surface profile intensity correction for spatially modulated imaging. J. Biomed. Opt. 2009, 14, 034045–034055. [Google Scholar] [CrossRef]
- Angelo, J.P.; van de Giessen, M.; Gioux, S. Real-time endoscopic optical properties imaging. Biomed. Opt. Express 2017, 8, 5113–5126. [Google Scholar] [CrossRef]
- Saager, R.B.; Cuccia, D.J.; Durkin, A.J. Determination of optical properties of turbid media spanning visible and near-infrared regimes via spatially modulated quantitative spectroscopy. J. Biomed. Opt. 2015, 15, 017012. [Google Scholar] [CrossRef]
- Andrea, C.D.; Ducros, N.; Bassi, A.; Arridge, S.; Valentini, G.; Dipartimento, P.; Milano, P.; Leonardo, P. Fast 3D optical reconstruction in turbid media using spatially modulated light Abstract. Biomed. Opt. Express 2010, 1, 471–481. [Google Scholar] [CrossRef]
- BeaLanger, S.; Abran, M.; Intes, X.; Casanova, C.; Lesage, F. Real-time diffuse optical tomography based on structured illumination. J. Biomed. Opt. 2010, 15, 016006. [Google Scholar] [CrossRef] [PubMed]
- Konecky, S.D.; Owen, C.M.; Tyler, R.; Valdés, P.A.; Kolbein, K.; Wilson, B.C.; Frederic, L.; Roberts, D.W.; Paulsen, K.D.; Tromberg, B.J. Spatial frequency domain tomography of protoporphyrin IX fluorescence in preclinical glioma models. J. Biomed. Opt. 2012, 17, 056008. [Google Scholar] [CrossRef] [PubMed]
- Konecky, S.D.; Amaan, M.; Cuccia, D.; Durkin, A.J.; Schotland, J.C.; Tromberg, B.J. Quantitative optical tomography of sub-surface heterogeneities using spatially modulated structured light. Opt. Express 2009, 17, 14780–14790. [Google Scholar] [CrossRef]
- Gardner, A.R.; Vasan, V. Accurate and efficient Monte Carlo solutions to the radiative transport equation in the spatial frequency domain. Opt. Lett. 2011, 36, 2269–2271. [Google Scholar] [CrossRef] [PubMed]
- Yao, R.Y.; Intes, X.; Fang, Q.Q. Generalized mesh-based Monte Carlo for wide-field illumination and detection via mesh retessellation. Biomed. Opt. Express 2016, 7, 171–184. [Google Scholar] [CrossRef]
- He, X.M.; Jiang, X.; Fu, X.P.; Gao, Y.W.; Rao, X.Q. Least squares support vector machine regression combined with Monte Carlo simulation based on the spatial frequency domain imaging for the detection of optical properties of pear. Postharvest Biol. Technol. 2018, 145, 1–9. [Google Scholar] [CrossRef]
- Regan, C.; Hayakawa, C.K.; Choi, B. Momentum transfer Monte Carlo for the simulation of laser speckle imaging and its application in the skin. Biomed. Opt. Express 2017, 8, 5708–5723. [Google Scholar] [CrossRef]
- Kijanka, P.; Packo, P. Novel method for true guided waves spectral characteristics estimation using a logistic function fit and nonlinear least square algorithm. Struct. Control Health Monit. 2019, 26, 13. [Google Scholar] [CrossRef]
- Song, J.W.; Lau, D.L.; Ho, Y.S.; Liu, K. Automatic look-up table based real-time phase unwrapping for phase measuring profilometry and optimal reference frequency selection. Opt. Express 2019, 27, 13357–13371. [Google Scholar] [CrossRef] [PubMed]
- Vicent Servera, J.; Alonso, L.; Martino, L.; Sabater, N.; Verrelst, J.; Camps-Valls, G.; Moreno, J. Gradient-Based Automatic Lookup Table Generator for Radiative Transfer Models. IEEE Trans. Geosci. Remote Sens. 2019, 57, 1040–1048. [Google Scholar] [CrossRef]
- Angelo, J.; Vargas, C.R.; Lee, B.T.; Bigio, I.J.; Gioux, S. Ultrafast optical property map generation using lookup tables. J. Biomed. Opt. 2016, 21, 110501. [Google Scholar] [CrossRef]
- Vervandier, J.; Gioux, S. Single snapshot imaging of optical properties. Biomed. Opt. Express 2013, 4, 2938–2944. [Google Scholar] [CrossRef]
- Aguenounon, E.; Dadouche, F.; Uhring, W.; Ducros, N.; Gioux, S. Single snapshot imaging of optical properties using a single-pixel camera: A simulation study. J. Biomed. Opt. 2019, 24, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Aguenounon, E.; Dadouche, F.; Uhring, W.; Gioux, S. Single snapshot of optical properties image quality improvement using anisotropic two-dimensional windows filtering. J. Biomed. Opt. 2019, 24, 1–21. [Google Scholar] [CrossRef]
- Dognitz, N.; Wagnieres, G. Determination of tissue optical properties by steady-state spatial frequency-domain reflectometry. Lasers Med. Sci. 1998, 13, 55–65. [Google Scholar] [CrossRef]
- Lu, R.F. Light Scattering Technology for Food Property, Quality and Safety Assessment; CRC Press: Boca Raton, FL, USA, 2017. [Google Scholar]
- Nguyen, T.T.A.; Ramella-Roman, J.C.; Moffatt, L.T.; Ortiz, R.T.; Jordan, M.H.; Shupp, J.W. Novel Application of a Spatial Frequency Domain Imaging System to Determine Signature Spectral Differences Between Infected and Noninfected Burn Wounds. J. Burn Care Res. 2013, 34, 44–50. [Google Scholar] [CrossRef][Green Version]
- Kennedy, G.T.; Stone, R.; Kowalczewski, A.C.; Chen, J.H.; Rowland, R.; Ponticorvo, A.; Christy, R.J.; Durkin, A.J. Characterization of debrided burn wounds using spatial frequency domain imaging. Photonics Dermatol. Plast. Surg. 2019, 108510. [Google Scholar] [CrossRef]
- Ponticorvo, A.; Rowland, R.; Baldado, M.; Burmeister, D.M.; Christy, R.J.; Bernal, N.P.; Durkin, A.J. Evaluating clinical observation versus Spatial Frequency Domain Imaging (SFDI), Laser Speckle Imaging (LSI) and thermal imaging for the assessment of burn depth. Burns 2019, 45, 450–460. [Google Scholar] [CrossRef]
- Rowland, R.; Ponticorvo, A.; Baldado, M.; Kennedy, G.T.; Burmeister, D.M.; Christy, R.J.; Bernal, N.P.; Durkin, A.J. A Simple Burn Wound Severity Assessment Classifier Based On Spatial Frequency Domain Imaging (SFDI) and Machine Learning. Photonics Dermatol. Plast. Surg. 2019, 1085109. [Google Scholar] [CrossRef]
- Ponticorvo, A.; Burmeister, D.M.; Yang, B.; Choi, B.; Christy, R.J.; Durkin, A.J. Quantitative assessment of graded burn wounds in a porcine model using spatial frequency domain imaging (SFDI) and laser speckle imaging (LSI). Biomed. Opt. Express 2014, 5, 3467–3481. [Google Scholar] [CrossRef]
- Poon, C.; Sunar, U.; Rohrbach, D.J.; Krishnamurthy, S.; Olsen, T.; Kent, M.; Weir, N.M.; Simman, R.; Travers, J.B. Early assessment of burn severity in human tissue ex vivo with multi-wavelength spatial frequency domain imaging. Toxicol. Vitr. 2018, 52, 251–254. [Google Scholar] [CrossRef]
- Nguyen, J.Q.; Crouzet, C.; Mai, T.; Riola, K.; Uchitel, D.; Liaw, L.H.; Bernal, N.; Ponticorvo, A.; Choi, B.; Durkin, A.J. Spatial frequency domain imaging of burn wounds in a preclinical model of graded burn severity. J. Biomed. Opt. 2013, 18, 066010. [Google Scholar] [CrossRef]
- Burmeister, D.M.; Ponticorvo, A.; Yang, B.; Becerra, S.C.; Choi, B.; Durkin, A.J.; Christy, R.J. Utility of spatial frequency domain imaging (SFDI) and laser speckle imaging (LSI) to non-invasively diagnose burn depth in a porcine model. Burns 2015, 41, 1242–1252. [Google Scholar] [CrossRef]
- Chen, X.; Lin, W.; Wang, C.; Chen, S.; Sheng, J.; Zeng, B.; Xu, M. In vivo real-time imaging of cutaneous hemoglobin concentration, oxygen saturation, scattering properties, melanin content, and epidermal thickness with visible spatially modulated light. Biomed. Opt. Express 2017, 8, 5468–5482. [Google Scholar] [CrossRef]
- Saager, R.B.; Dang, A.N.; Huang, S.S.; Kelly, K.M.; Durkin, A.J. Portable (handheld) clinical device for quantitative spectroscopy of skin, utilizing spatial frequency domain reflectance techniques. Rev. Sci. Instrum. 2017, 88, 094302. [Google Scholar] [CrossRef]
- Travers, J.B.; Poon, C.; Rohrbach, D.J.; Weir, N.M.; Cates, E.; Hager, F.; Sunar, U. Noninvasive mesoscopic imaging of actinic skin damage using spatial frequency domain imaging. Biomed. Opt. Express 2017, 8, 3045–3052. [Google Scholar] [CrossRef]
- Yafi, A.; Muakkassa, F.K.; Pasupneti, T.; Fulton, J.; Cuccia, D.J.; Mazhar, A.; Blasiole, K.N.; Mostow, E.N. Quantitative Skin Assessment Using Spatial Frequency Domain Imaging (SFDI) in Patients With or at High Risk for Pressure Ulcers. Lasers Surg. Med. 2017, 49, 827–834. [Google Scholar] [CrossRef] [PubMed]
- Gevaux, L.; Cherel, M.; Seroul, P.; Clerc, R.; Tremeau, A.; Hebert, M. Hyperspectral imaging and spatial frequency domain imaging: Combined acquisition for full face skin analysis. Imaging Manip. Anal. Biomol. Cells Tissues Xvii 2019, 10881. [Google Scholar] [CrossRef]
- Rohrbach, D.J.; Zeitouni, N.C.; Muffoletto, D.; Saager, R.; Tromberg, B.J.; Sunar, U. Characterization of nonmelanoma skin cancer for light therapy using spatial frequency domain imaging. Biomed. Opt. Express 2015, 6, 1761–1766. [Google Scholar] [CrossRef]
- Nandy, S.; Mostafa, A.; Kumavor, P.D.; Sanders, M.; Brewer, M.; Zhu, Q. Characterizing optical properties and spatial heterogeneity of human ovarian tissue using spatial frequency domain imaging. J. Biomed. Opt. 2016, 21, 101402. [Google Scholar] [CrossRef] [PubMed]
- Lin, W.H.; Zeng, B.X.; Cao, Z.L.; Chen, X.L.; Yang, K.Y.; Xu, M. Quantitative diagnosis of tissue microstructure with wide-field high spatial frequency domain imaging. Biomed. Opt. Express 2018, 9, 2905–2916. [Google Scholar] [CrossRef] [PubMed]
- Lin, W.H.; Zeng, B.X.; Cao, Z.L.; Zhu, D.F.; Xu, M. Wide-field high spatial frequency domain imaging of tissue microstructure. Prog. Biomed. Opt. Imaging Proc. SPIE 2018, 10484. [Google Scholar] [CrossRef]
- Laughney, A.M.; Krishnaswamy, V.; Rizzo, E.J.; Schwab, M.C.; Barth, R.J.; Cuccia, D.J.; Tromberg, B.J.; Paulsen, K.D.; Pogue, B.W.; Wells, W.A. Spectral discrimination of breast pathologies in situ using spatial frequency domain imaging. Breast Cancer Res. 2013, 15, R61. [Google Scholar] [CrossRef]
- Nguyen, J.T.; Lin, S.J.; Tobias, A.M.; Gioux, S.; Mazhar, A.; Cuccia, D.J.; Ashitate, Y.; Stockdale, A.; Oketokoun, R.; Durr, N.J.; et al. A Novel Pilot Study Using Spatial Frequency Domain Imaging to Assess Oxygenation of Perforator Flaps During Reconstructive Breast Surgery. Ann. Plast. Surg. 2013, 71, 308–315. [Google Scholar] [CrossRef]
- McClatchy, D.M.; Rizzo, E.J.; Wells, W.A.; Cheney, P.P.; Hwang, J.C.; Paulsen, K.D.; Pogue, B.W.; Kanick, S.C. Wide-field quantitative imaging of tissue microstructure using sub-diffuse spatial frequency domain imaging. Optica 2016, 3, 613–621. [Google Scholar] [CrossRef]
- McClatchy, D.M.; Rizzo, E.; Krishnaswamy, V.; Kanick, S.; Wells, W.; Paulsen, K.; Pogue, B. Combined multispectral spatial frequency domain imaging and computed tomography system for intraoperative breast tumor margin assessment. Prog. Biomed. Opt. Imaging Proc. SPIE 2017, 10057. [Google Scholar] [CrossRef]
- Robbins, C.M.; Antaki, J.F.; Kainerstorfer, J.M. Spatial frequency domain imaging for monitoring palpable breast lesions. Prog. Biomed. Opt. Imaging Proc. SPIE 2017, 10059. [Google Scholar] [CrossRef]
- Wei, R.L.; Leproux, A.; Laoui, S.; Kuo, J.V.; Daroui, P.; Farol, H.Y.; Ramsinghani, N.S.; Al-Ghazi, M.; Durkin, A.J.; Tromberg, B. Temporal and Spatial Quantification of Tissue Oxygen Saturation and Melanin Deposition During Whole Breast Radiation Using Noninvasive Spatial Frequency Domain Imaging. Int. J. Radiat. Oncol. 2017, 99, E54–E55. [Google Scholar] [CrossRef][Green Version]
- Nandy, S.; Erfanzadeh, M.; Zhou, F.F.; Zhu, Q. Feasibility study of spatial frequency domain imaging using a handheld miniaturized projector and rigid endoscope. Prog. Biomed. Opt. Imaging Proc. SPIE 2017, 10059. [Google Scholar] [CrossRef]
- Tabassu, S.; Pera, V.; Greening, G.; Muldoon, T.J.; Roblyer, D. Two-layer inverse model for improved longitudinal preclinical tumor imaging in the spatial frequency domain. J. Biomed. Opt. 2018, 23, 076011. [Google Scholar] [CrossRef]
- Zhao, Y.; Tabassum, S.; Piracha, S.; Nandhu, M.S.; Viapiano, M.; Roblyer, D. Angle correction for small animal tumor imaging with spatial frequency domain imaging (SFDI). Biomed. Opt. Express 2016, 7, 2373–2384. [Google Scholar] [CrossRef] [PubMed]
- Burns, J.M.; Schaefer, E.; Anvari, B. Near Infrared Spatial Frequency Domain Fluorescence Imaging of Tumor Phantoms Containing Erythrocyte-Derived Optical Nanoplatforms. Prog. Biomed. Opt. Imaging Proc. SPIE 2018, 105060. [Google Scholar] [CrossRef]
- Lin, A.J.; Castello, N.A.; Lee, G.; Green, K.N.; Durkin, A.J.; Choi, B.; LaFerla, F.; Tromberg, B.J. In vivo optical signatures of neuronal death in a mouse model of Alzheimer’s disease. Lasers Surg. Med. 2014, 46, 27–33. [Google Scholar] [CrossRef] [PubMed]
- Singh-Moon, R.P.; Roblyer, D.M.; Bigio, I.J.; Joshi, S. Spatial mapping of drug delivery to brain tissue using hyperspectral spatial frequency-domain imaging. J. Biomed. Opt. 2014, 19, 096003. [Google Scholar] [CrossRef]
- Wilson, R.H.; Crouzet, C.; Torabzadeh, M.; Bazrafkan, A.; Farahabadi, M.H.; Jamasian, B.; Donga, D.; Alcocer, J.; Zaher, S.M.; Choi, B.; et al. High-speed spatial frequency domain imaging of rat cortex detects dynamic optical and physiological properties following cardiac arrest and resuscitation. Neurophotonics 2017, 4, 045008. [Google Scholar] [CrossRef] [PubMed]
- Sibai, M.; Fisher, C.; Veilleux, I.; Elliott, J.T.; Leblond, F.; Roberts, D.W.; Wilson, B.C. Preclinical evaluation of spatial frequency domain-enabled wide-field quantitative imaging for enhanced glioma resection. J. Biomed. Opt. 2017, 22, 76007. [Google Scholar] [CrossRef] [PubMed]
- Abookasis, D.; Meitav, O. Assessing mouse brain tissue refractive index in the NIR spectral range utilizing spatial frequency domain imaging technique combined with processing algorithms. Clin. Transl. Neurophotonics 2019, 10864. [Google Scholar] [CrossRef]
- Li, T.W.; He, X.M.; Fu, X.P.; Rao, X.Q. LabVIEW Base Software for Spatial Frequency Domain Imaging System. In Proceedings of the 2017 ASABE Annual International Meeting, Spokane, WA, USA, 16 July 2017. [Google Scholar]
- Hu, D.; Fu, X.P.; He, X.M.; Ying, Y.B. Noncontact and Wide-Field Characterization of the Absorption and Scattering Properties of Apple Fruit Using Spatial-Frequency Domain Imaging. Sci. Rep. 2016, 6, 37920–37930. [Google Scholar] [CrossRef]
- He, X.M.; Fu, X.P.; Rao, X.Q.; Fu, F. Nondestructive determination of optical properties of a pear using spatial frequency domain imaging combined with phase-measuring profilometry. Appl. Opt. 2017, 56, 8207–8215. [Google Scholar] [CrossRef]
- He, X.M.; Fu, X.P.; Li, T.W.; Rao, X.Q. Spatial frequency domain imaging for detecting bruises of pears. J. Food Meas. Charact. 2018, 12, 1266–1273. [Google Scholar] [CrossRef]
- Hu, D.; Lu, R.F.; Ying, Y.B. Spatial-frequency domain imaging coupled with frequency optimization for estimating optical properties of two-layered food and agricultural products. J. Food. Eng. 2020, 277, 109909–109913. [Google Scholar] [CrossRef]
- Hu, D.; Lu, R.F.; Ying, Y.B. Optimization of Spatial Frequency Domain Imaging Technique for Estimating Optical Properties of Food and Biological Materials. In Proceedings of the 2017 ASABE Annual International Meeting, Spokane, WA, USA, 16 July 2017. [Google Scholar]
- Hu, D.; Lu, R.F.; Ying, Y.B. A two-step parameter optimization algorithm for improving estimation of optical properties using spatial frequency domain imaging. J. Quant. Spectrosc. Radiat. Transf. 2018, 207, 32–40. [Google Scholar] [CrossRef]
- Lu, Y.Z.; Lu, R.F. Development of a Multispectral Structured Illumination Reflectance Imaging (SIRI) System and Its Application to Bruise Detection of Apples. Trans. ASABE 2017, 60, 1379–1389. [Google Scholar] [CrossRef]
- Lu, Y.Z.; Lu, R.F. Histogram-based automatic thresholding for bruise detection of apples by structured-illumination reflectance imaging. Biosyst. Eng. 2017, 160, 30–41. [Google Scholar] [CrossRef]
- Lu, Y.Z.; Huang, Y.P.; Lu, R.F. Innovative Hyperspectral Imaging-Based Techniques for Quality Evaluation of Fruits and Vegetables: A Review. Appl. Sci. 2017, 7, 189. [Google Scholar] [CrossRef]
- Lu, Y.Z.; Lu, R.F. Using composite sinusoidal patterns in structured-illumination reflectance imaging (SIRI) for enhanced detection of apple bruise. J. Food Eng. 2017, 199, 54–64. [Google Scholar] [CrossRef]
- Nguyen, T.T.A.; Le, H.N.D.; Vo, M.; Wang, Z.Y.; Luu, L.; Ramella-Roman, J.C. Three-dimensional phantoms for curvature correction in spatial frequency domain imaging. Biomed. Opt. Express 2012, 3, 1200–1214. [Google Scholar] [CrossRef] [PubMed]
- Nothelfer, S.; Liemert, A.; Reitzle, D.; Bergmann, F.; Kienle, A. A New method for correction of surface scattering in spatial frequency domain imaging for an accurate determination of volume scattering. Opt. InfoBase Conf. Pap. 2017, 10412. [Google Scholar] [CrossRef]
- Hachadorian, R.; Bruza, P.; Jermyn, M.; Mazhar, A.; Cuccia, D.; Jarvis, L.; Gladstone, D.; Pogue, B. Correcting Cherenkov light attenuation in tissue using spatial frequency domain imaging for quantitative surface dosimetry during whole breast radiation therapy. J. Biomed. Opt. 2019, 24, 1–10. [Google Scholar] [CrossRef]
- Fang, Q.Q.; Kaeli, D.R. Accelerating mesh-based Monte Carlo method on modern CPU architectures. Biomed. Opt. Express 2012, 3, 3223–3230. [Google Scholar] [CrossRef] [PubMed]
- Cai, F.H.; Lu, W. A Dynamic Accuracy Estimation for GPU-based Monte Carlo Simulation in Tissue Optics. Curr. Opt. Photonics 2017, 1, 551–555. [Google Scholar] [CrossRef]
- Ren, N.; Liang, J.; Qu, X.C.; Li, J.F.; Lu, B.J.; Tian, J. GPU-based Monte Carlo simulation for light propagation in complex heterogeneous tissues. Opt. Express 2010, 18, 6811–6823. [Google Scholar] [CrossRef] [PubMed]
- Fang, Q.Q. Mesh-based Monte Carlo method using fast ray-tracing in Plücker coordinates. Biomed. Opt. Express 2010, 1, 165–175. [Google Scholar] [CrossRef] [PubMed]
- Alerstam, E.; Svensson, T.; Andersson-Engels, S. Parallel computing with graphics processing units for high-speed Monte Carlo simulation of photon migration. J. Biomed. Opt. 2008, 13, 060504. [Google Scholar] [CrossRef]
- Cai, F.H. Using graphics processing units to accelerate perturbation Monte Carlo simulation in a turbid medium. J. Biomed. Opt. 2012, 17, 040502. [Google Scholar] [CrossRef]
- Ismail, A.; Idris, M.; Ayub, M.; Por, L. Vision-Based Apple Classification for Smart Manufacturing. Sensors 2018, 18, 4353. [Google Scholar] [CrossRef] [PubMed]
- Alerstam, E.; Andersson-Engels, S.; Svensson, T. White Monte Carlo for time-resolved photon migration. J. Biomed. Opt. 2008, 13, 10. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.Y.; Deng, Y.; Yue, S.H.; Wang, M.; Song, B.; Fan, Y.B. Direct mapping from diffuse reflectance to chromophore concentrations in multi-fx spatial frequency domain imaging (SFDI) with a deep residual network (DRN). Biomed. Opt. Express 2021, 12, 433–443. [Google Scholar] [CrossRef]
- Li, Y.; Guo, M.R.; Qian, X.F.; Lin, W.H.; Zheng, Y.; Yu, K.Y.; Zeng, B.X.; Xu, Z.; Zheng, C.; Xu, M. Single snapshot spatial frequency domain imaging for risk stratification of diabetes and diabetic foot. Biomed. Opt. Express 2020, 11, 4471–4483. [Google Scholar] [CrossRef] [PubMed]
- Nadeau, K.P.; Khoury, P.; Mazhar, A.; Cuccia, D.; Durkin, A.J. Component and system evaluation for the development of a handheld point-of-care spatial frequency domain imaging (SFDI) device. Prog. Biomed. Opt. Imaging Proc. SPIE 2013, 8573. [Google Scholar] [CrossRef]
- Valdes, P.A.; Angelo, J.P.; Choi, H.S.; Gioux, S. qF-SSOP: Real-time optical property corrected fluorescence imaging. Biomed. Opt. Express 2017, 8, 3597–3605. [Google Scholar] [CrossRef]
- Wang, Z.D.; Hu, M.H.; Zhai, G.T. Application of Deep Learning Architectures for Accurate and Rapid Detection of Internal Mechanical Damage of Blueberry Using Hyperspectral Transmittance Data. Sensors 2018, 18, 1126. [Google Scholar] [CrossRef]
- Liu, R.X.; Yao, M.H.; Wang, X.B. Defects detection based on deep learning and transfer learning. Metall. Min. Ind. 2015, 7, 312–321. [Google Scholar]
- Nayeli, V.R.; Juan, G.S.; Jorge, C.P.; Juan, J.C. Early detection of mechanical damage in mango using NIR hyperspectral images and machine learning. Biosyst. Eng. 2014, 122, 91–98. [Google Scholar] [CrossRef]
- Baranowski, P.; Mazurek, W.; Pastuszka, W. Supervised classification of bruised apples with respect to the time after bruising on the basis of hyperspectral imaging data. Postharvest Biol. Technol. 2013, 86, 249–258. [Google Scholar] [CrossRef]

| Classification | Measuring Method | Light Transfer Model | Optical Property | Ref. |
|---|---|---|---|---|
| Direct method | Collimated transmittance | Beer–Lambert Law | , | [24] |
| Indirect method | Integrating sphere | Adding-doubling | , | [25] |
| Time-domain | Diffusion approximation equation, Monte Carlo or analytical solutions of radiative transfer equation | , | [26] | |
| Frequency-domain | , | [27] | ||
| Spatially resolved | , | [28,29] | ||
| Spatial-frequency domain imaging | , | [5,20,30] |
| Object | Optical Property | Indices | Frequency/mm−1 | Wavelength/nm | Ref. |
|---|---|---|---|---|---|
| Rat burn in vivo model | HbO2, Hb, HbT, StO2 | 0, 0.10 | 650–970 nm with step length of 20 nm | [64] | |
| , | StO2, Hb | 0.20 | sixteen wavelengths in 500–700 nm | [58] | |
| HbO2, Hb, H2O, StO2 | 0.20 | seventeen equally spaced wavelengths in 650–970 nm | [19] | ||
| Pig burns in vivo model | , | StO2 | 0.20 | 658, 730, 850 | [62] |
| , | StO2 | 0.20 | 658, 730, 850 | [65] | |
| - | 0, 0.05. 0.10, 0.15, 0.20 | nine wavelengths in 470–970 nm | [59] | ||
| - | 0, 0.05. 0.10, 0.15, 0.20 | eight wavelengths in 471–850 nm | [60] | ||
| calibrated reflectance | - | 0, 0.05, 0.10, 0.20 | 471, 526, 591, 621, 659, 731, 851 | [61] | |
| Heat burns skin | , | - | Eleven-frequencies in 0–0.44 | 490, 590, 660, 780 | [63] |
| Object | Wavelength/nm | Optical Property of Normal Tissue/mm−1 | Optical Property of Tumor Tissue/mm−1 | Ref. | ||
|---|---|---|---|---|---|---|
| Breast tissue | 658 | - | - | - | 0.910 | [77] |
| 750 | - | - | - | 0.750 | [78] | |
| Mouse tumor | 530 | 0.025 | 1.850 | 0.032 | 0.950 | [81] |
| 659 | - | - | 0.024 | 2.054 | [82] | |
| Non-melanoma skin cancer | 630 | 0.021 ± 0.002 | 1.497 ± 0.097 | 0.027 ± 0.003 | 1.177 ± 0.120 | [16] |
| 630 | 0.025 | 1.670 | 0.059 | 1.070 | [71] | |
| Human ovarian tissue | 730 | 0.015 | 3.370 | 0.049 | 1.050 | [72] |
| Cervical tissue | 623 | 0.018 ± 0.001 | 0.900 ± 0.062 | 0.040 ± 0.004 | 1.412 ± 0.245 | [73] |
| Bladder tumor tissue | 623 | 0.018 | 0.550 | 0.045 | 1.050 | [74] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sun, Z.; Hu, D.; Wang, Z.; Xie, L.; Ying, Y. Spatial-Frequency Domain Imaging: An Emerging Depth-Varying and Wide-Field Technique for Optical Property Measurement of Biological Tissues. Photonics 2021, 8, 162. https://doi.org/10.3390/photonics8050162
Sun Z, Hu D, Wang Z, Xie L, Ying Y. Spatial-Frequency Domain Imaging: An Emerging Depth-Varying and Wide-Field Technique for Optical Property Measurement of Biological Tissues. Photonics. 2021; 8(5):162. https://doi.org/10.3390/photonics8050162
Chicago/Turabian StyleSun, Zhizhong, Dong Hu, Zhong Wang, Lijuan Xie, and Yibin Ying. 2021. "Spatial-Frequency Domain Imaging: An Emerging Depth-Varying and Wide-Field Technique for Optical Property Measurement of Biological Tissues" Photonics 8, no. 5: 162. https://doi.org/10.3390/photonics8050162
APA StyleSun, Z., Hu, D., Wang, Z., Xie, L., & Ying, Y. (2021). Spatial-Frequency Domain Imaging: An Emerging Depth-Varying and Wide-Field Technique for Optical Property Measurement of Biological Tissues. Photonics, 8(5), 162. https://doi.org/10.3390/photonics8050162







