Enhanced Biosensing Activity of Bimetallic Surface Plasmon Resonance Sensor
Abstract
1. Introduction
2. Experimental Methods
2.1. Chemicals and Materials
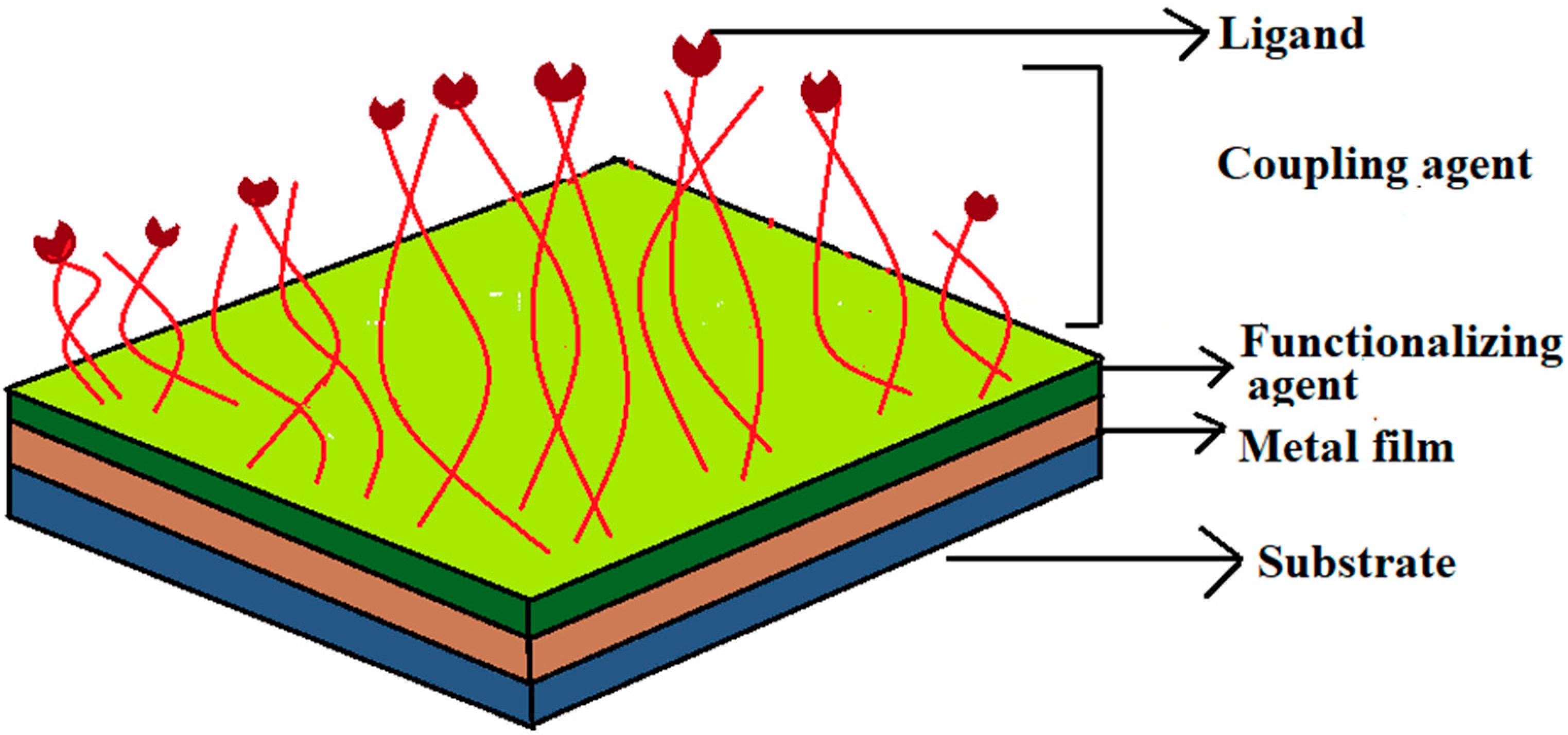
2.2. Sensor Fabrication
2.3. Method of Elemental Characterization
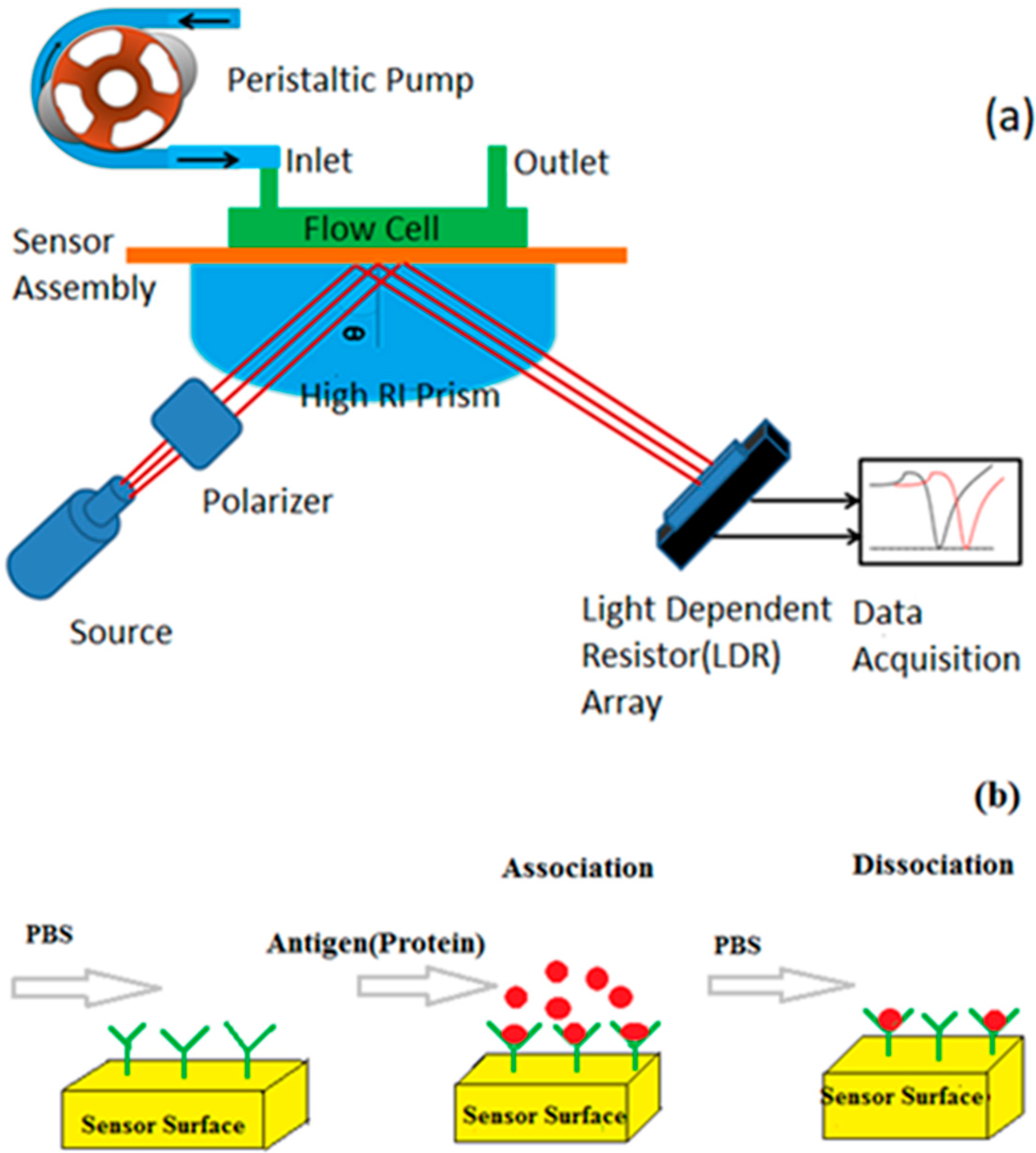
2.4. SPR Device and Measurements
3. Results
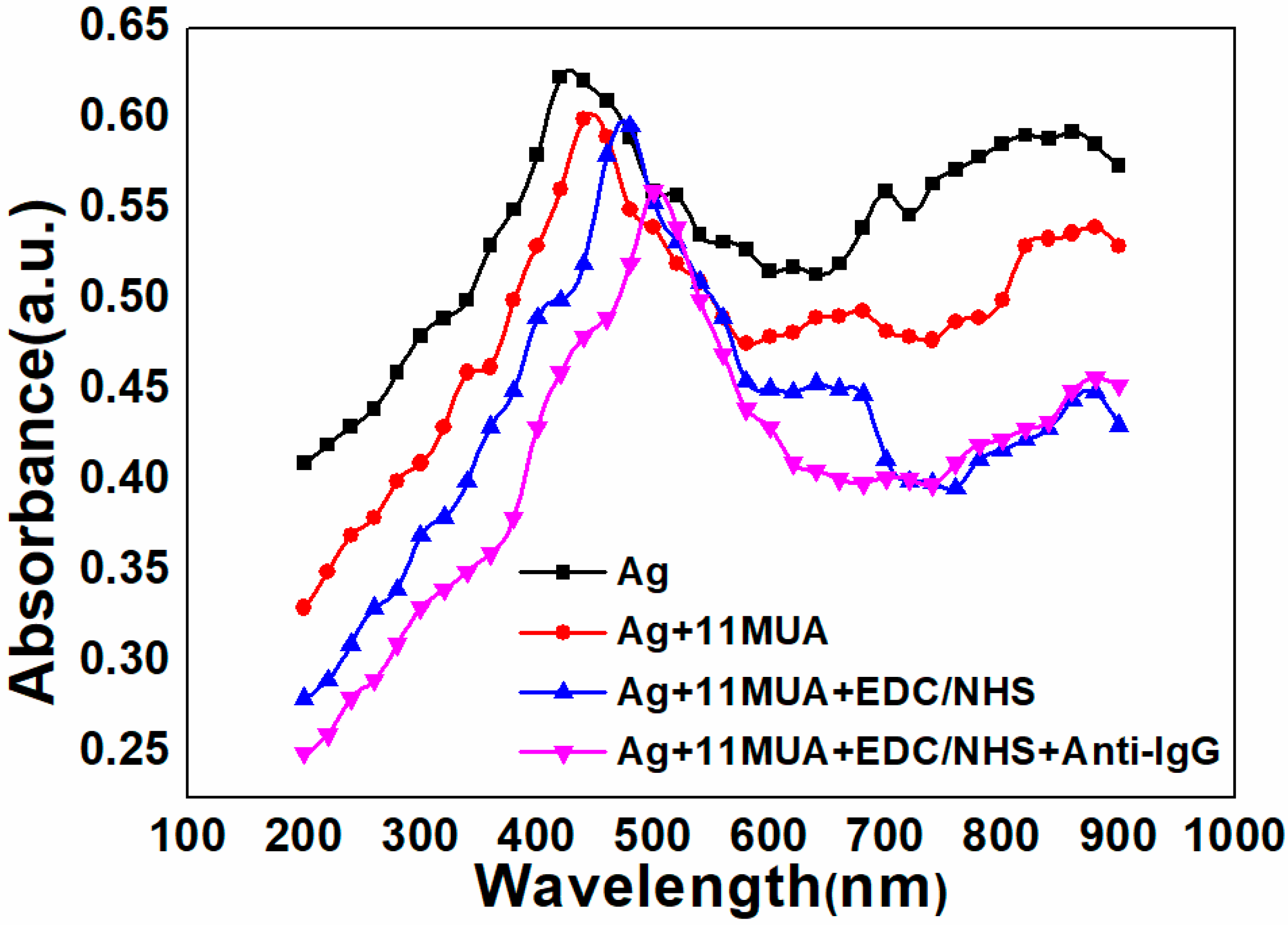
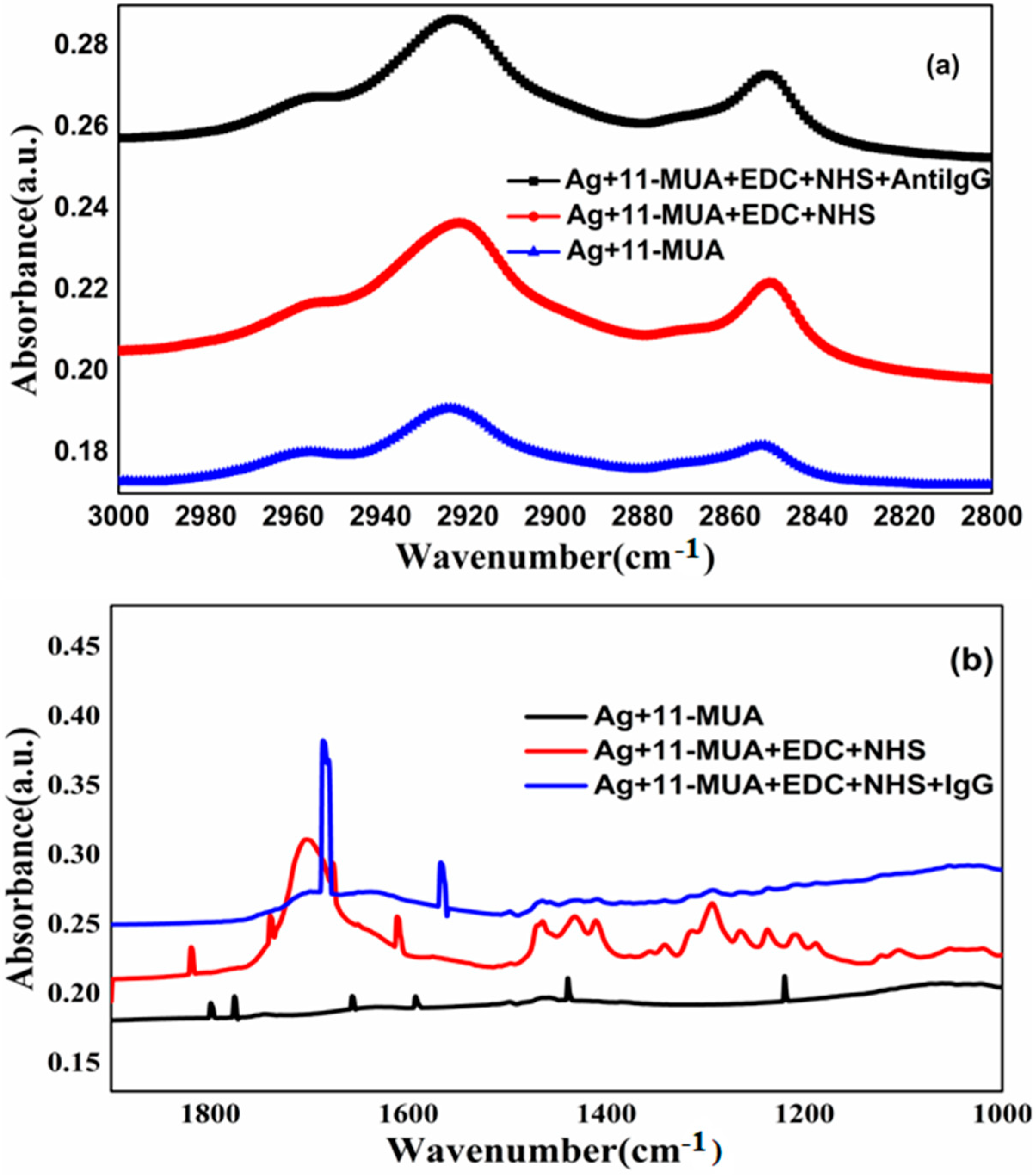
3.1. Elemental and Structural Characterization
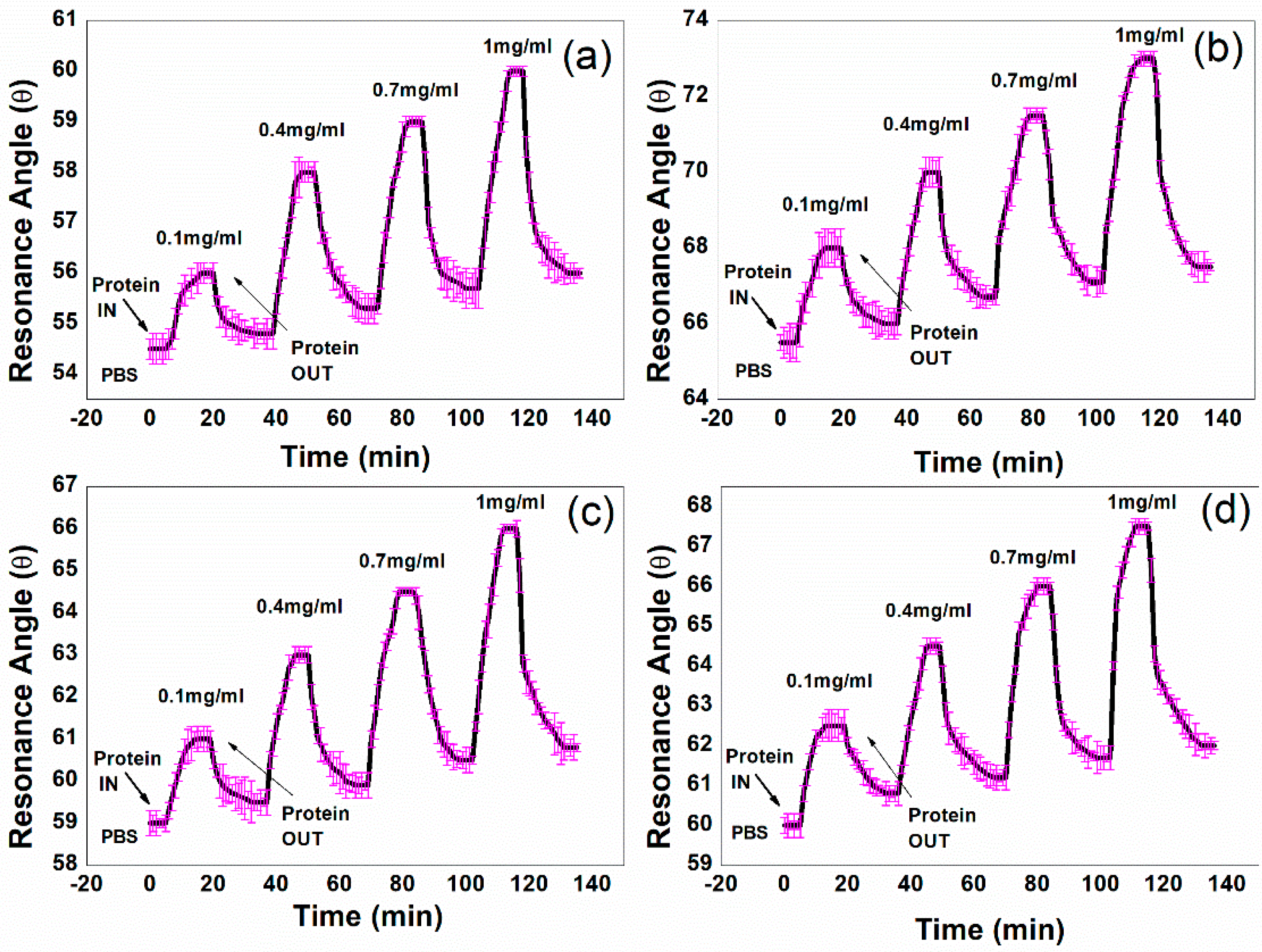
3.2. Sensor Response
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Gordon, J.G., II; Ernst, S. Surface Plasmons as a Probe of the Electrochemical Interface. Surf. Sci. 1980, 101, 499–506. [Google Scholar] [CrossRef]
- Nylander, C.; Liedberg, B.; Lind, T. Gas Detection by Means of Surface Plasmon Resonance. Sens. Actuators 1982, 3, 79–88. [Google Scholar] [CrossRef]
- Liedberg, B.; Nylander, C.; Lundstrom, I. Surface Plasmon Resonance for Gas Detection and Biosensing. Sens. Actuators B Chem. 1983, 4, 299–304. [Google Scholar] [CrossRef]
- Liedberg, B.; Nylander, C.; Lundstrom, I. Biosensing with Surface Plasmon Resonance-How it all Started. Biosens. Bioelectron. 1995, 10, 1–9. [Google Scholar] [CrossRef]
- Monteiro, J.I.; Mukherji, S.; Kundu, T. Development of a Low-Cost Portable Surface Plasmon Resonance Biosensor. In Proceedings of the IEEE International Conference on Microelectronics, Communication and Renewable Energy, Kanjirapally, Kerala, India, 4–6 June 2013; IEEE: Piscataway, NJ, USA, 2013; p. 989. [Google Scholar]
- Mukherji, S.; Hussain, M.I.; Kundu, T.; Chandratre, D. Development of a Surface Plasmon Resonance-Based Biosensing System. In Micro and Smart Devices and Systems; Vinoy, K.J., Ananthasuresh, G.K., Pratap, R., Krupanidhi, S.B., Eds.; Springer: New Delhi, India, 2014; pp. 49–72. [Google Scholar]
- Otto, A. Excitation of Nonradiative Surface Plasma Waves in Silver by Method of Frustrated Total Reflection. Z. Fur. Phys. 1968, 216, 398–410. [Google Scholar] [CrossRef]
- Kretschmann, E.; Raether, H. Radiative Decay of Non-Radiative Surface Plasmons Excited by Light. Z. Fur Nat. 1968, 23, 2135–2136. [Google Scholar] [CrossRef]
- Choudhury, S.N.; Konwar, B.; Kaur, S.; Doley, R.; Mondal, B. Study on Snake Venom Protein-Antibody Interaction by Surface Plasmon Resonance Spectroscopy. Photonic Sens. 2018, 8, 193–202. [Google Scholar] [CrossRef]
- Kashyap, R.; Chakraborty, S.; Swarnakar, S.; Humbert, G.; Zeng, S.; Mondal, B. FWHM and Sensitivity Study of Bimetallic SPR Sensor Chip. In Proceedings of the 3rd IEEE International Conference on Electrical, Computer and Communication Technologies (ICECCT), SVS College of Engineering, Coimbatore, Tamil Nadu, India, 20–22 February 2019; IEEE: Piscataway, NJ, USA, 2019; Volume 3, pp. 1682–1685. [Google Scholar]
- Homola, J. On the Sensitivity of Surface Plasmon Resonance Sensors with Spectral Interrogation. Sens. Actuators 1997, 41, 207–211. [Google Scholar] [CrossRef]
- Caucheteur, C.; Guo, T.; Albert, J. Review of Plasmonic Fiber Optic Biochemical Sensors: Improving the Limit of Detection. Anal. Bioanal. Chem. 2015, 407, 3883–3897. [Google Scholar] [CrossRef]
- Homola, J.; Yee, S.S.; Gauglitz, G. Surface Plasmon Resonance Sensors: Review. Sens. Actuators B Chem. 1999, 54, 3–15. [Google Scholar] [CrossRef]
- Nelson, S.G.; Johnston, K.S.; Yee, S.S. High Sensitivity Surface Plasmon Resonance Sensor Based on Phase Detection. Sens. Actuators B Chem. 1996, 35, 187–191. [Google Scholar] [CrossRef]
- Maharana, P.K.; Padhy, P.; Jha, R. On the Field Enhancement and Performance of an Ultra-Stable SPR Biosensor Based on Graphene; IEEE Photonics Technology Letters: Piscataway, NJ, USA, 2013; Volume 25, pp. 2156–2159. [Google Scholar]
- Hanken, D.G.; Jordan, C.E.; Frey, B.L.; Corn, R.M. Surface Plasmon Resonance Measurements of Ultrathin Organic Films at Electrode Surfaces. In Electroanalytical Chemistry: A Series of Advances; Marcel Dekker: New York, NJ, USA, 1998; Volume 20, pp. 141–225. [Google Scholar]
- Chen, X.; Jiang, K. Effect of Aging on Optical Properties of Bimetallic Sensor Chips. Opt. Soc. Am. 2010, 18, 1105–1112. [Google Scholar] [CrossRef] [PubMed]
- Zynio, S.A.; Samoylov, A.V.; Surovtseva, E.R.; Mirsky, V.M.; Shirshov, Y.M. Bimetallic Layers Increase Sensitivity of Affinity Sensors Based on Surface Plasmon Resonance. Sensors 2002, 2, 62–70. [Google Scholar] [CrossRef]
- Yuan, X.C.; Ong, B.H.; Tan, Y.G.; Zhang, D.W.; Irawan, R.; Tjin, S.C. Sensitivity–Stability-Optimized Surface Plasmon Resonance Sensing with Double Metal Layers. J. Opt. A Pure Appl. Opt. 2006, 8, 959–963. [Google Scholar] [CrossRef]
- Ong, B.H.; Yuan, X.; Tjin, S.C.; Zhang, J.; Ng, H.M. Optimised Film Thickness for Maximum Evanescent Field Enhancement of a Bimetallic Film Surface Plasmon Resonance Biosensor. Sens. Actuators B Chem. 2006, 114, 1028–1034. [Google Scholar] [CrossRef]
- Wu, S.Y.; Ho, P.H. Sensitivity improvement of the surface plasmon resonance optical sensor by using a gold-silver transducing layer. In Proceedings of the 2002 IEEE Hong Kong Electron Devices Meeting, Hong Kong, China, 22 June 2002; pp. 63–68. [Google Scholar]
- Chen, Y.; Zheng, R.; Zhang, D.; Lu, Y.; Wang, P.; Ming, H.; Luo, Z.; Kan, Q. Bimetallic Chips for a Surface Plasmon Resonance Instrument. Appl. Opt. 2017, 50, 387–391. [Google Scholar] [CrossRef]
- Huang, Y.; Xia, L.; Wei, W.; Chuang, C.J.; Du, C. Theoretical Investigation of Voltage Sensitivity Enhancement for Surface Plasmon Resonance Based Optical Fiber Sensor with a Bimetallic Layer. Opt. Commun. 2014, 333, 146–150. [Google Scholar] [CrossRef]
- Chen, S.; Lin, C. High-Performance Bimetallic Film Surface Plasmon Resonance Sensor Based on Film Thickness Optimization. Optik 2016, 127, 7514–7519. [Google Scholar] [CrossRef]
- Nimse, S.B.; Song, K.; Sonawane, M.D.; Sayyed, D.R.; Kim, T. Immobilization Techniques for Microarray: Challenges and Applications. Sensors 2014, 14, 22208–22229. [Google Scholar] [CrossRef]
- Rusmini, F.; Zhong, Z.; Feijen, J. Protein Immobilization Strategies for Protein Biochis. Biomacromolecules 2007, 8, 1775–1789. [Google Scholar] [CrossRef]
- Lee, J.E.; Seo, J.H.; Kim, C.S.; Kwon, Y.; Ha, J.H.; Choi, S.S.; Cha, H.J. A Comparative Study on Antibody Immobilization Strategies onto Solid Surface. Korean J. Chem. Eng. 2013, 30, 1934–1938. [Google Scholar] [CrossRef]
- Hodneland, C.D.; Lee, Y.S.; Min, D.H.; Mrksich, M. Selective Immobilization of Proteins to Self-Assembled Monolayers Presenting Active Site-Directed Capture Ligands. Proc. Natl. Acad. Sci. USA 2002, 99, 5048–5052. [Google Scholar] [CrossRef]
- Peters, R.F.; Gutierrez-Rivera, L.; Dew, S.K.; Stepanova, M. Surface Enhanced Raman Spectroscopy Detection of Biomolecules Using EBL Fabricated Nanostructured Substrates. J. Vis. Exp. Jove 2015, 2015, e52712. [Google Scholar] [CrossRef] [PubMed]
- Mohapatra, S.; Moirangthem, R.S. Theoretical Study of Modulated Multi-Layer SPR Device for Improved Refractive Index Sensing. IOP Conf. Ser. Mater. Sci. Eng. 2018, 310, 012017. [Google Scholar] [CrossRef]
- Kuo Chih, C.; Lin, C.Y.; Chen, Y.D.; Shean Jen, C. Optimizing Silver Film for Surface Plasmon Coupled Emission Induced Two-Photon Excited Fluorescence Imaging. Opt. Express 2011, 19, 5386–5396. [Google Scholar]
- Whiteside Paul, J.D.; Chininis Jeffrey, A.; Hunt Heather, K. Techniques and Challenges for Characterizing Metal Thin Films with Applications in Photonics. Coat. Rev. 2016, 6, 35. [Google Scholar] [CrossRef]
- Aslan, K.; Luhrs, C.C.; Perez-Luna, V.H. Controlled and Reversible Aggregation of Biotinylated Gold Nanoparticles with Streptavidin. J. Phys. Chem. B 2004, 108, 15631–15639. [Google Scholar] [CrossRef]
- Weng, G.; Li, J.; Zhoa, J. Enhanced Resonance Light Scattering of Antibody Covalently Conjugated Gold Nanoparticles due to Antigen-Antibody Interaction Induced Aggregation. Nanosci. Nanotechnol. Lett. 2013, 5, 1–7. [Google Scholar] [CrossRef]
- Sharma Anuj, K.; JhaRajan Gupta, B.D. Fiber-OPTIC Sensors Based on Surface Plasmon Resonance: A Comprehensive Review. IEEE Sens. 2007, 7, 1118–1129. [Google Scholar] [CrossRef]
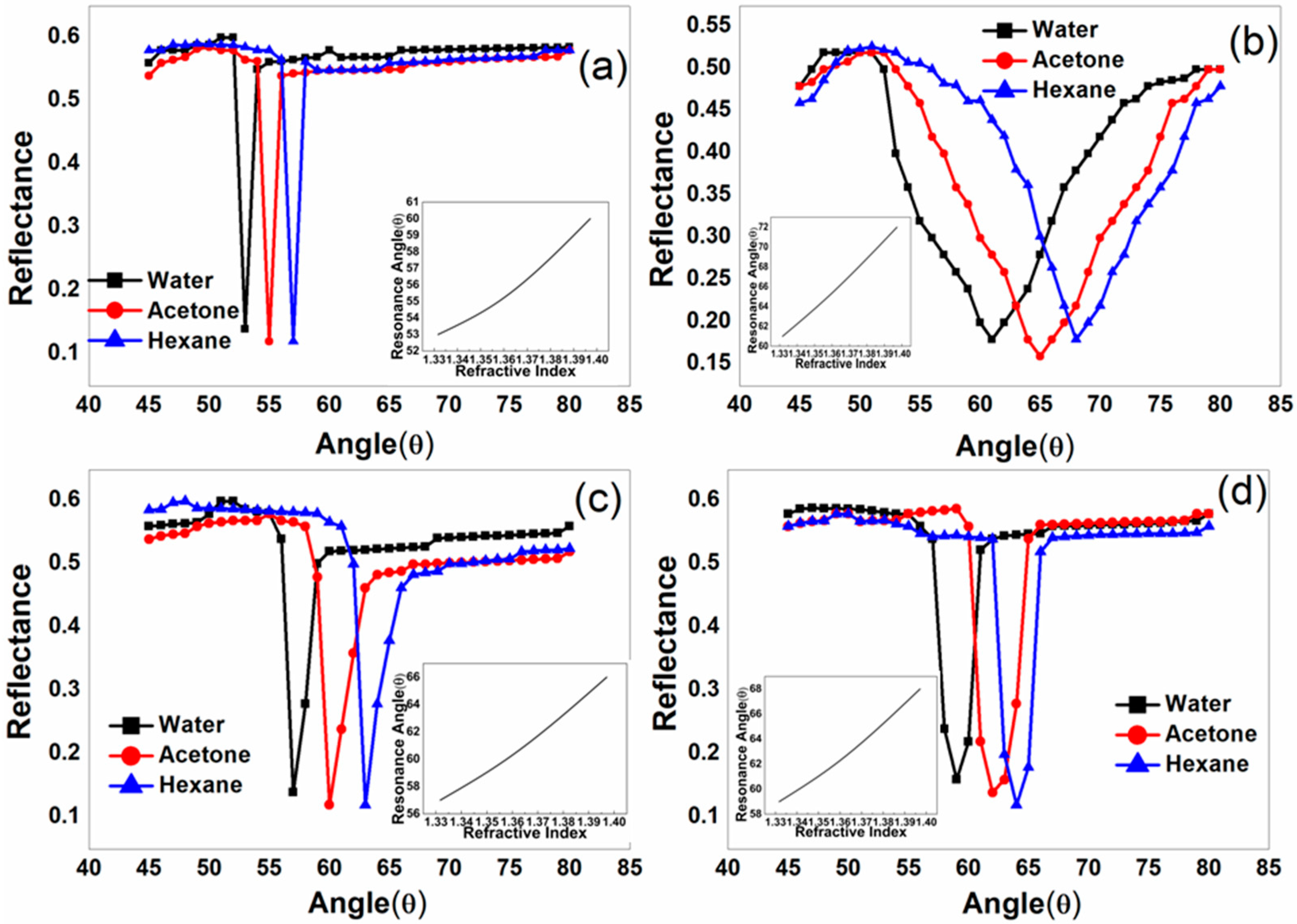
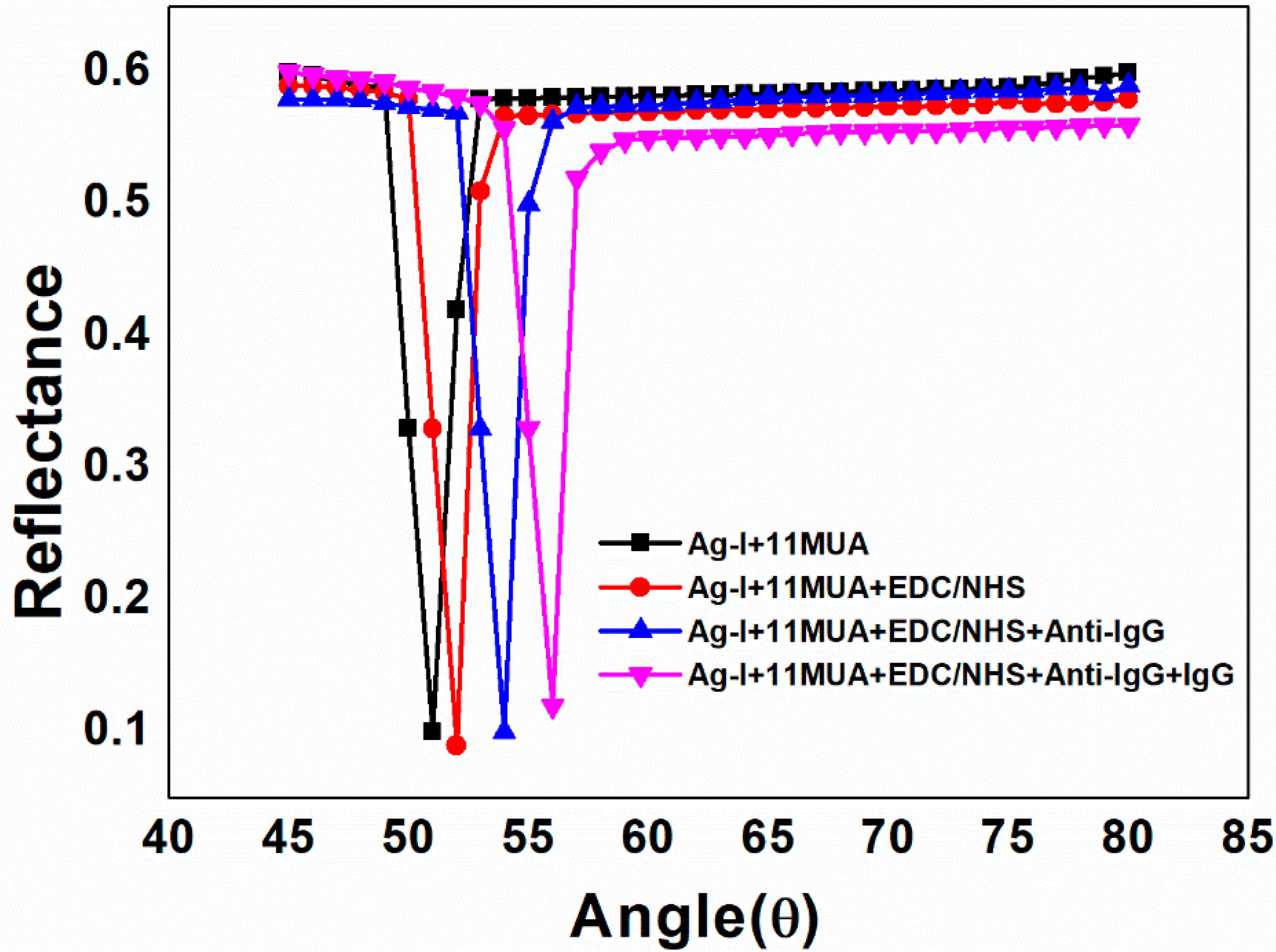
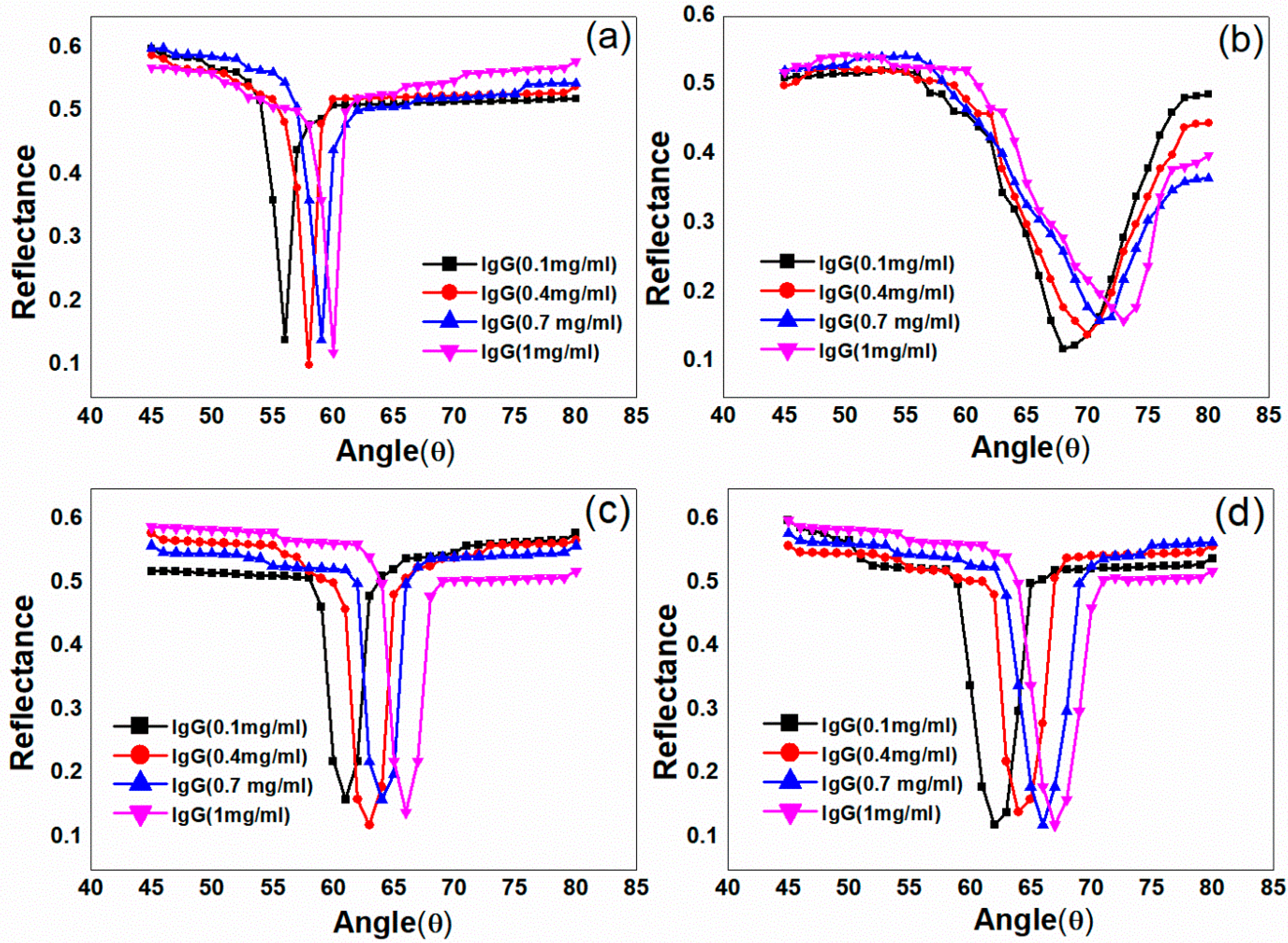
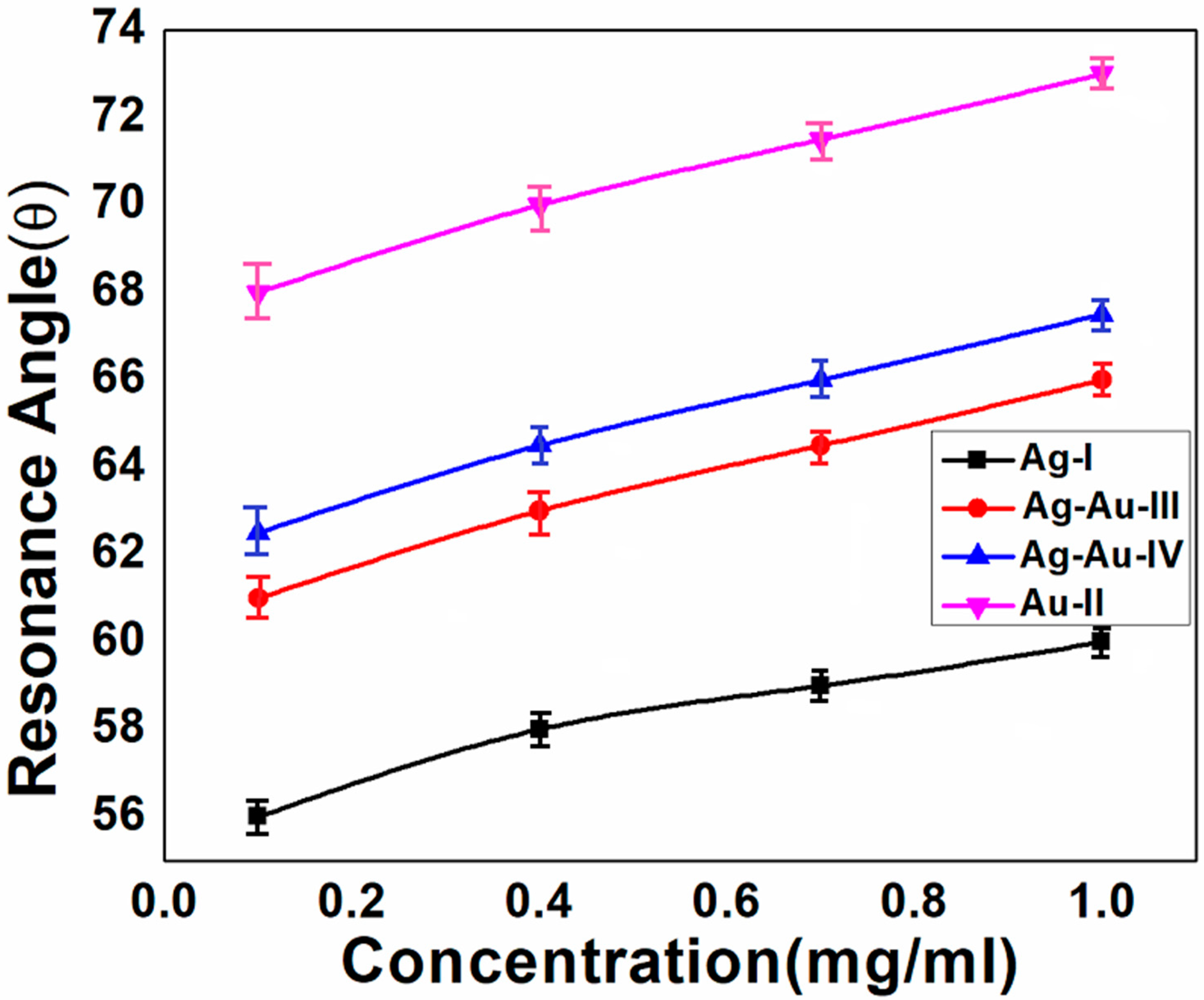
| Sample ID | Deposited Metal Film | Film Thickness (nm) | Deposition Time (s) |
|---|---|---|---|
| Ag-I | Silver (Ag) | 50 | 221 |
| Au-II | Gold (Au) | 50 | 167 |
| Ag-Au-III | Silver–Gold (Ag–Au) | 35 (Ag)–15 (Au) | Ag: 154 Au: 50 |
| Ag-Au-IV | Silver–Gold (Ag–Au) | 30 (Ag)–20 (Au) | Ag:132 Au: 67 |
| Sample | FWHM (°) | |||
|---|---|---|---|---|
| Ag-I | Au-II | Ag-Au-III | Ag-Au-IV | |
| Water | 1 ± 0.1 | 12 ± 0.3 | 2 ± 0.2 | 3 ± 0.2 |
| Acetone | 1 ± 0.2 | 12 ± 0.3 | 2.5 ± 0.1 | 3.5 ± 0.3 |
| Hexane | 1 ± 0.1 | 11 ± 0.2 | 2 ± 0.1 | 3 ± 0.2 |
| Refractive Index Unit (RIU) | Shift of the Resonance Angle (°) | |||
|---|---|---|---|---|
| Ag-I | Au-II | Ag-Au-III | Ag-Au-IV | |
| 0.0261 | 2 ± 0.1 | 4 ± 0.3 | 3.5 ± 0.1 | 3.6 ± 0.2 |
| Concentration of Protein (IgG) | FWHM (°) | Shift of the Resonance Angle (°) | ||||||
|---|---|---|---|---|---|---|---|---|
| Ag-I | Au-II | Ag-Au-III | Ag-Au-IV | Ag-I | Au-II | Ag-Au-III | Ag-Au-IV | |
| 0.1 mg/mL | 2 ± 0.2 | 10 ± 0.4 | 3 ± 0.2 | 4.5 ± 0.3 | 2 ± 0.2 | 4 ± 0.5 | 3 ± 0.3 | 3.5 ± 0.4 |
| 0.4 mg/mL | 2 ± 0.2 | 11 ± 0.5 | 3 ± 0.2 | 4 ± 0.4 | 4 ± 0.2 | 6 ± 0.4 | 5 ± 0.2 | 5.5 ± 0.2 |
| 0.7 mg/mL | 1.5 ± 0.1 | 9.5 ± 0.3 | 3 ± 0.1 | 4.5 ± 0.4 | 5 ± 0.1 | 7.5 ±0.2 | 6.5 ± 0.1 | 7 ± 0.2 |
| 1 mg/mL | 1.5 ± 0.2 | 10 ± 0.3 | 3 ± 0.1 | 4.5 ± 0.3 | 6 ± 0.1 | 9 ± 0.2 | 8 ± 0.1 | 8.5 ± 0.2 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kashyap, R.; Chakraborty, S.; Zeng, S.; Swarnakar, S.; Kaur, S.; Doley, R.; Mondal, B. Enhanced Biosensing Activity of Bimetallic Surface Plasmon Resonance Sensor. Photonics 2019, 6, 108. https://doi.org/10.3390/photonics6040108
Kashyap R, Chakraborty S, Zeng S, Swarnakar S, Kaur S, Doley R, Mondal B. Enhanced Biosensing Activity of Bimetallic Surface Plasmon Resonance Sensor. Photonics. 2019; 6(4):108. https://doi.org/10.3390/photonics6040108
Chicago/Turabian StyleKashyap, Ritayan, Soumik Chakraborty, Shuwen Zeng, Sikha Swarnakar, Simran Kaur, Robin Doley, and Biplob Mondal. 2019. "Enhanced Biosensing Activity of Bimetallic Surface Plasmon Resonance Sensor" Photonics 6, no. 4: 108. https://doi.org/10.3390/photonics6040108
APA StyleKashyap, R., Chakraborty, S., Zeng, S., Swarnakar, S., Kaur, S., Doley, R., & Mondal, B. (2019). Enhanced Biosensing Activity of Bimetallic Surface Plasmon Resonance Sensor. Photonics, 6(4), 108. https://doi.org/10.3390/photonics6040108






