1. Introduction
Coastal lagoons are unique but fragile ecosystems with significant ecological, social, and economic value [
1]. These ecosystems are represented in the European Directive 92/43/EEC (Habitat Directive) (habitat code 1150*), but at the same time are considered endangered with a high risk of disappearance [
2], especially by morphological and hydrological changes [
1] and overall inconsistency to the favorable status of habitats [
3]. Lagoons are ecosystems that are partially or completely separated from the sea with strips of sand or stones, or less frequently, with rocks and bedrock. Such dynamic conditions create a variety of enclosed or semi-enclosed shallow water bodies that vary in size, water salinity, sediment accumulation, biodiversity, and ecological status and are greatly influenced by precipitation, evaporation, temporary floods, and storms [
3]. Since the Baltic Sea consists of low-saline brackish water with the addition of freshwater sources, lagoons can represent freshwater with their corresponding species or brackish water ecosystems [
4], all of which depend on local peculiar conditions and structures, such as natural/human-made channels, isolated pools, and oligohaline areas [
1].
High variability of natural and anthropogenic conditions forms the uniqueness of lagoon habitats, as well as their role in the preservation of biological diversity [
5,
6,
7,
8,
9,
10]. Assessing the ecological state, studies on long-term changes have, so far, been carried out only in the largest coastal lagoons in the Baltic Sea, as well as river inlets [
2,
11,
12,
13,
14,
15].
Therefore, the ecological aspects of smaller lagoons, affected by various anthropogenic influences and morphological changes both natural and human-induced, remain unclear; the high potential variation in their size and conditions is also a factor. Due to the sensitivity of these ecosystems, it has been concluded that settlement expansion and inadequate management greatly affect the ecological quality and even the existence of coastal lagoons [
16].
The lagoon lakes of the coastal area of Latvia were formed in the latest stages of the Littorina Sea and are relatively large waterbodies, whereas the lagoons formed much more recently and are typically small in size [
17]. Lagoon areas can contain several separate waterbodies that are either completely isolated or partially connected with one another and/or the sea. Dynamic coastal processes prevent long-lasting conditions and, in turn, the subsequent creation of permanent objects in the coastal areas of Latvia, yet there are three areas where long-lasting lagoons have formed: the eastern shore of the Gulf of Riga between Salacgrīva and Ainaži, as well as between the Svētupe estuary and Salacgrīva; on the western shore of the Gulf of Riga, where some small lagoons have formed in Mērsrags; and in the southern part of the Gulf, on the island of Daugavgrīva [
16]. The ecological status and development of these lagoons are significantly affected by the ecological status of the Baltic Sea, as well as influencing factors from the adjacent land areas, such as water, nutrient and suspended material flows (including pollution) from ditches, roads, and other anthropogenic areas. These factors contribute to the overgrowth of lagoons (mainly with reeds), and, together with sediment accumulation, are some of the main reasons for the decline of lagoon areas. Until the end of the 20th century, the shores of long-lasting lagoons and the surrounding areas were grazed or mowed, thus maintaining favorable conditions for coastal grasslands and lagoons. Nowadays, the management of these lagoon areas has significantly decreased or even stopped altogether, leading to the formation of dense continuous reed stands in the lagoons and large areas around them [
4].
The aim of our work was to study two Latvian lagoons and characterize their current conditions and ecological parameters because currently there is a lack of information on the quality of such ecosystems and their evaluation indicators (except overall biodiversity), which is necessary in order to support their assessment and develop sustainable management measures. Regarding these two lagoons, we analyzed sediment thickness and composition, benthic macroinvertebrate species, phytoplankton and zooplankton species, vegetation, and the seasonal changes in water chemistry.
2. Materials and Methods
The studied lagoons (Mērsrags and the “Randu pļavas” (Randu meadows) nature reserve) are located in the coastal area of Latvia (
Figure 1) and were studied in the period of 2020–2021. Monitoring data of the SLLC “Latvian Environment, Geology and Meteorology Centre” [
18] in stations “Mērsrags” and “Ainaži” show that in the study period, the annual average temperature varied between 7 and 9 °C, with the warmest months being between June and August when the monthly average temperature reached up to 21.18 °C in July of 2021. Seasonal temperature differences determine the length of the productive season, which starts in the 2nd part of April and lasts until October. The location in the coastal area of the Baltic Sea also determines that stable snow covers are formed only periodically. In 2020, there was no snow cover at all, while in 2021, snow cover formed at the beginning of January with a thickness of 13 and 19 cm in the middle of the month on the Randu pļavas and Mērsrags lagoons, respectively, although they melted during next 5 days. This was followed by a new period of snow accumulation, which melted at the end of February. The next snow event occurred only at the end of November 2021, which completely melted after a few days. Significant differences between lagoons were demonstrated by precipitation rates reaching 435 and 638 mm in 2020 and 2021 in the Mērsrags lagoon and 799 and 730 mm in 2020 and 2021 in the Randu pļavas lagoon. It is important to mention that there can be high monthly variability of precipitation; for example, less than 1 mm in March 2021 in the Mērsarags lagoon and 205 mm in August of the same year. Another aspect of the big differences between months is the occurrence of intensive precipitation over short periods, reaching 58 mm in June 30 in 2020 and 60 mm in July 13 in 2021 in Randu pļavas lagoon, where these events occur with higher recurrence rates.
2.1. Characterization of Lagoons
The Randu pļavas lagoon has been less affected by morphological changes due to anthropogenic activities; however, in the last decades, management measures, such as grazing and mowing, have stopped, which has led to the overgrowing of the lagoon. Sediment transport along the shore and coastal processes are slowly isolating the lagoon from the sea with sandy sediments, yet in stormy conditions, the lagoon is still flooded with seawater. In the coastal areas of the lagoon, there is an increased pressure from tourism activities, but since this lagoon has been included in the list of protected areas, other anthropogenic influences have been significantly reduced. The main body of the existing lagoon is located at its northern end and is connected to the other parts of the lagoon by a culvert. The lagoon is shallow, and the deepest parts are closer to the shore with depths up to 0.45 m; in colder winters, this creates the prerequisites for the water to freeze all the way down to the sediments, thus affecting all biological constituents. This lagoon has great marine influence because of its shallow and narrow connection to the sea; it is also directly influenced by the small tides. The parts of the lagoon lying south of the main body consist of several isolated water bodies; one of these smaller bodies still has a connection to the main body, although with reduced water exchange capacity. The partially closed and completely closed lagoons do not have a direct connection to the sea, and their average water depth does not exceed 0.4 m; in the smallest isolated water bodies, the depth of water in some cases is only 5–10 cm, and water exchange with the sea happens through filtration and occasional flooding affected by gusts of wind.
The Mērsrags lagoon consists of several waterbodies and is seriously affected by morphological changes from anthropogenic activities. The body of the lagoon located north of the port has been affected by the port’s expansion, but it still has a shallow and narrow connection to the sea. It is also the deepest of the studied lagoons, reaching a depth of 2.5 m. South of the port lies another body of the lagoon that has been divided into two bodies separated by an artificial embankment designed as an access road to the sea; the waterbodies are connected to each other via a small culvert going through the embankment. The average depth of water in these bodies is approximately 0.4 m, with a maximum depth reaching 0.8–0.9 m in some parts. Although these south-of-the-port parts of the lagoon are connected to the sea via a ditch, from a hydrological perspective, there appears to be a very slight tendency of dominant water flow from the northern body to the southern body and toward the sea; seawater only flows into the lagoon during stormy conditions. It is worth mentioning that both waterbodies have a ditch next to the embankment, which provides a direct connection to the sea, but due to slight elevation differences without wind, seawater inflow is not dominant.
The most significant anthropogenic sources in Randu pļavas lagoon are surface runoff from agricultural land, nutrients from sewage water (mainly individual houses in the coastal area), and potential influences from the road, and there could be minor pressures from tourist activities. In the Mērsrags lagoon, the main anthropogenic sources can be considered the harbor, the surface runoff, and the potential presence of sewage water from the urban area of Mērsrags (more than 1500 inhabitants) [
19]. Also, the pressure of visitors at the Mērsrags lagoon is higher than at Randu pļavas lagoon.
2.2. Cartographic Analysis
To determine the overgrowth of the lagoons, an analysis of cartographic materials was performed using ArcGIS Desktop 10.8.2. As a base map layer to determine the overgrowth, orthophoto maps from different periods of time were used. The available orthophoto maps for the territory of Latvia are in 6 cycles over the period of 1994 to 2018 (1994–1999; 2003–2005; 2007–2008; 2010–2011; 2013–2015; 2016–2018) [
20]. The degree of overgrowth was determined by recognizing and marking the boundaries of the overgrown territory using visual criteria. To characterize the degree of overgrowth that has increased over time, the area of the open water was calculated for each of the used orthophoto periods.
2.3. Characterization of Water and Sediments
During the seasonal surveys of the lagoons, the following field measurements were made: the content of dissolved oxygen, pH, and electrical conductivity (EC) with a portable multimeter HACH HQ40, and water samples were collected for chemical analyses (water sampling dates—Randu pļavas: 15 July 2020; 11 August 2020; 24 November 2020; 23 February 2020; 11 May 2021; 1 July 2021; Mersrags: 30 July 2020; 25 November 2020; 25 February 2021; 12 May 2021; 29 June 2021). Water sampling (
Figure 2) was performed in 1 L PET bottles, and the samples were transported to the laboratory where the composition of the main ions was determined. Phosphate ions, total phosphorus, water color, ammonia, nitrate, and total nitrogen were determined spectrophotometrically using a Hach-Lange DR5000 UV/VIS Spectrophotometer. Phosphate ions were analyzed spectrophotometrically using the ascorbic acid reduction method, with potassium persulfate extraction for total phosphorus determination [
21]. The concentration of organic forms of phosphorus is defined as the difference between total phosphorus and the phosphate form. Nitrates were determined using the cadmium reduction method, according to the HACH water analysis handbook [
22]. Total nitrogen was determined using the Laton Total Nitrogen cuvette test (1–16 mg/L) (Hach). The concentrations of chloride were determined titrimetrically [
21]. Sulfate ions were analyzed by the turbidimetric method [
22]. Dissolved organic carbon (DOC) was measured in water samples (filtered through a 0.45 µm filter) using a Shimadzu Total Organic Carbon Analyzer TOC-VCSN.
Sediments were collected (
Figure 2) in August 2020 with a soft sediment drill, and samples were packed in plastic bags and taken to the laboratory for further analysis. Sediment moisture, organic matter, and carbonate content were analyzed using the LOI (loss on ignition) method [
23]. The pH was determined using KCl and H
2O extracts, and after shaking and filtering, was measured using a HANNA HI 2210 pH meter; the conductivity of sediment water extracts was determined using a Hanna HI 9932 Microprocessor Conductivity Meter.
The content of elements (C, N) for dried samples was determined using an Eurovector Element Analyzer EuroEA.
2.4. Characterization of Macroinvertebrates
Qualitative and semi-quantitative macroinvertebrate samples were collected using a hand net (frame size 0.25 × 0.25 m). Samples in the lagoons in Mērsrags were sampled on 30 July 2020 (
Figure 2); the lagoons at “Randu pļavas” were sampled on 12 August 2020. Larger macroinvertebrate specimens with species-characteristic features were identified during field surveys and released back into the studied lagoons. At each lagoon, 5 sub-samples were taken in proportion to the characteristic microhabitats using the sweeping technique. Samples were merged into one sample and preserved with 70% ethanol (final concentration). Preserved samples were stored in 1 l bottles. In the laboratory, samples were washed, specimens sorted, and taxa identified to the best achievable level. Macroinvertebrate sampling, sample processing, taxa identification, and the calculation of indices (LLMMI) were carried out according to the Latvian lake macroinvertebrate assessment method [
24].
2.5. Characterization of Zooplankton
Zooplankton sampling in the Mērsrags lagoon was performed on 30 July 2020 and in the “Randu pļavas” lagoon on 11 August 2020. Zooplankton samples were collected by filtering 100 L of water through a 55 µm Apstein-type plankton net at the surface in the middle pelagic area of the shallow lagoons. Zooplankton samples were preserved in 95% ethanol. The analysis of zooplankton (identification, measurement, and counting) with at least 100× magnification was performed using a ZEISS AxioLab 5 microscope with an Axiocam 208 camera, and ZEISS Labscope software was used to measure individuals. The zooplankton were counted in a Segdwick–Rafter chamber by the subsampling method (1 mL subsample repeatedly 4×). At least 200 individuals of the dominant zooplankton taxon were counted. Zooplankton were identified to the lowest possible taxonomic level [
25,
26,
27,
28]. Where possible, the length of at least 20 individuals was measured for each taxon. The wet weight–length relationship was used for individual biomass estimation of crustaceans and rotifers [
29,
30,
31]. The abundance and biomass of zooplankton in the sample were calculated per cubic meter (m
3). The diversity of the zooplankton community was analyzed using Simpson’s reciprocal index (1/D): 1/D = 1/Σp
i2, where p
i = proportion of taxa in the community by abundance. The index varies from 1 to s, depending on it means the number of taxa in the sample [
32].
2.6. Characterization of Phytoplankton
Phytoplankton samples in lagoons were collected in the summer period of 2020 and 2021. Sampling was realized with a Rutner-type water sampler in the central part of the lagoons at a depth of 0.5 m. Samples were fixed with acidified Lugol’s solution. Samples were analyzed according to the Utermohl method [
33]. Phytoplankton counting was carried out with an inverted Leica DMIL microscope, according to Phytoplankton Counting Guidance (v1, 2007). Species composition, density, and cell dimensions were determined under a Leica DMIL microscope (200- and 400-fold magnification). Five mL and ten mL of the KC Denmark sedimentation counting chamber were used. Cell counts were converted to biovolumes (fresh weight mg/L) and calculated using measured cell dimensions applied to simple geometrical shapes. The taxonomic literature was used for phytoplankton taxonomic identification [
34,
35,
36,
37].
2.7. Characterization of Macrophytes
Macrophyte surveys were carried out in the summer of 2020 and 2021, assessing species composition and abundance of macrophytes in the entire lagoon area. Species abundance was estimated on a 7-point scale (1 (<1%, very rare), 2 (1–3%, rare), 3 (3–10%, quite rare), 4 (10–25%, infrequent), 5 (25–50%, quite common), 6 (50–75%, common), 7 (>75%, very common)). The assessment includes emergent, submerged, floating-leaved, and free-floating vegetation. A rake was used to collect plant samples for species identification. Macrophyte surveys were carried out from a boat; shallower lagoons were surveyed by wading. During the survey, the occurrence of rare and specially protected aquatic plant species was recorded.
3. Results
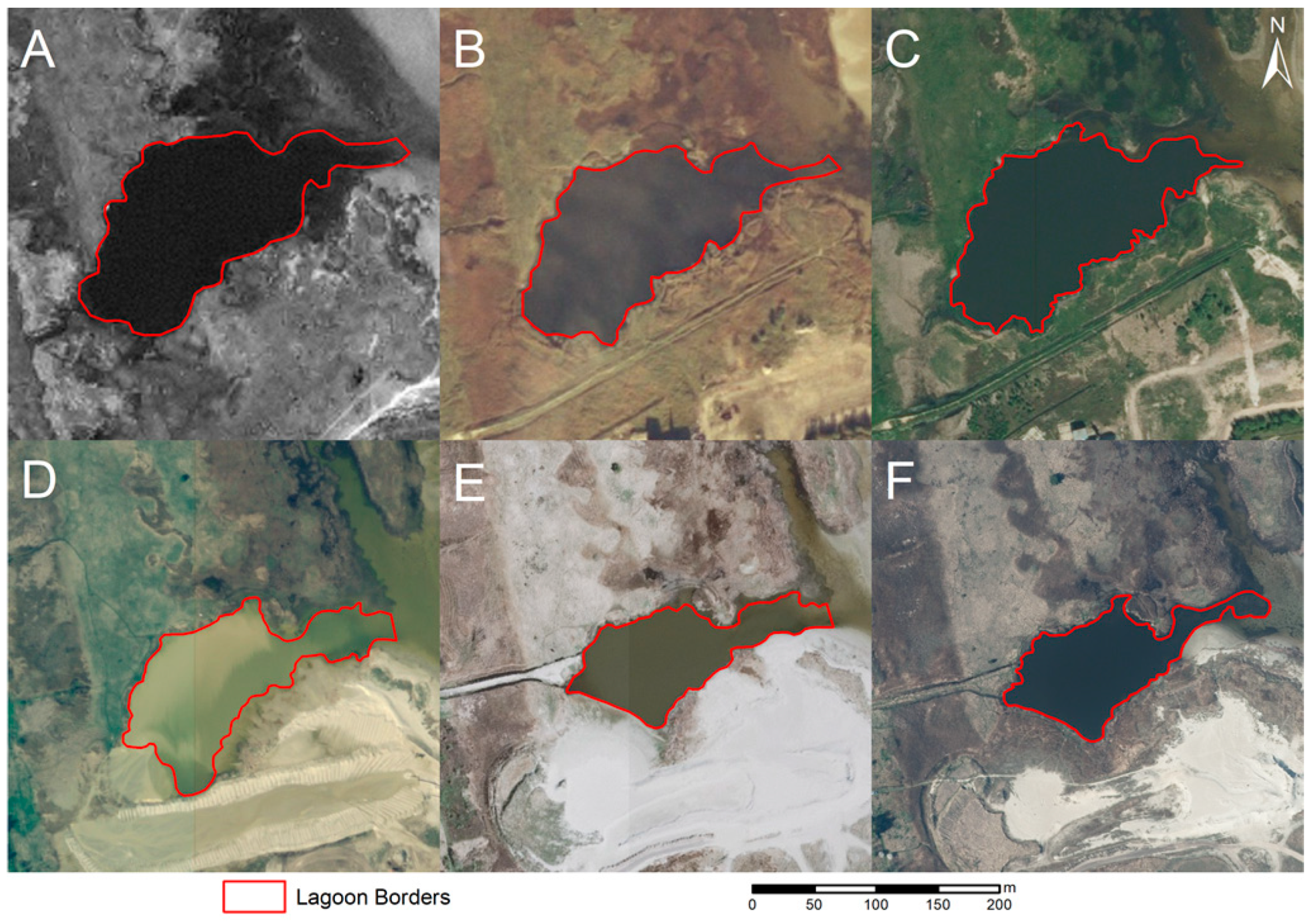
The formation of coastal lagoons depends on local coastal conditions, which can be considered very dynamic, thus limiting the existence of long-lasting objects. Although the studied lagoons have existed for decades, significant changes have occurred due to the gradual decrease in direct contact with the sea, which has led to the isolation of lagoons with sandy sediments, limited water exchange, and overgrowing, and as a result, a reduction in open water area and gradually overall depth. In the case of the “Randu pļavas” lagoon (
Figure 3), the existing water area is only 8.95 ha, although topographic maps from the middle of the last century indicate significantly larger waterbodies. Following the changes to the lagoon as seen over sequential periods of orthophoto mapping (
Figure 3), the trends of overgrowing and fragmentation of the lagoon can be clearly observed. A very similar pattern can also be seen to have taken place in the lagoons on the western shore of the Gulf of Riga, which have, in addition, also undergone significant pressure from anthropogenic activities that have led to morphological changes through disturbing the functionality of the lagoon ecosystem. Over the last 30 years, both lagoons in Mērsrags, i.e., south (3.34 ha) (
Figure 4) and north (0.98 ha) (
Figure 5) of the port, have undergone a significant reduction (2-fold) in the area covered with open water.
3.1. Sediment Analysis in Lagoons
The studied lagoons are dominated by sandy or coarse-grained sandy sediments with pebbles and an inconspicuous layer of silt; rarely does the sediment layer reach more than 5 cm. In the smaller and deeper waterbody north of the port in Mērsrags, the sediment layer reaches 105 cm and contains not only coarse-grained sandy sediments but also finely dispersed clayey material with increased organic material content (up to 18.81%).
In the area of the Mērsrags lagoon south of the port, seven sediment profiles were taken; points 1S, 3S, 6S, and 7S represent overgrown areas with less capacity for sediment movement because of dense reed stands. An accumulation of organic matter may happen (there is the presence of detritus in very tiny amounts) in these points compared to others in the waterbody. In the case of 1S, decomposed material can be seen with the naked eye as black areas in the mud; organic sediments could not be visually recognized in the other sampling points. In addition to a higher determined content of organic matter, these sampling points also had an increased content of carbonates due to shell particles. Despite the homogenous nature of this waterbody, the sediments show differences in EC and pH values (
Table 1), indicating that long-term fragmentation may affect sediment composition because of differences in evaporation rates, the amount of water flow, including freshwater flows, and the content of available oxygen in water and sediments.
The sediment profiles taken in the part of the Mērsrags lagoon located north of the port show that the accumulation of coarse-grained sand material and organic matter was more dominant in the past, indicating a higher rate of water flow or movement of sand material. In the upper 50 cm of the sediment layer, the accumulation of finer fractions of sandy, silty, and clayey sediments, including those with a higher amount of shell particles, begins to dominate, which indicates that it was formed with a lower water flow rate (or exchange rate). The depth of this northern part of the lagoon, as well as its morphology and infrequent connection with the sea, has formed conditions distinct from those in the part of the lagoon lying south of the port, the latter having higher pH values (
Table 2) and more homogeneity among its profiles.
Despite the larger amount of macrophytes and their produced biomass in the “Randu pļavas” lagoon compared to the Mērsrags lagoon, the dynamic conditions and leaching exhibited in the “Randu pļavas” lagoon have created thin sediment layers similar to those in the Mērsrags lagoon. Sampling points OL1–OL4 represent parts of the lagoon connected with the sea, whereas CL1–CL3 represent parts of the lagoon closed off to the sea; however, the results of the sediment analysis (
Table 2) are surprisingly similar. The flat terrain and low elevation allow seawater to easily flow into the lagoon, raising the water level to the point of reaching even the closed areas and, during reflux, carrying away some parts of the freshly formed sediment material. These intense water dynamics with brackish water surges and reflux in the “Randu pļavas” lagoon are intensified with higher freshwater flow from inland areas (also by higher precipitation rates), which is another factor that differentiates “Randu pļavas” from Mērsrags.
Elemental analysis of the sediments (
Table 2) shows low amounts of N, while C has higher values in areas where there was higher organic material due to distinct sediment accumulation conditions. The obtained C/N and C:N:P ratios in the Mērsrags lagoon indicate that the determined amount of organic material in the sediments has likely formed from algae as opposed to macrophytes; only a small proportion indicate macrophyte sources. Conversely, the organic material present in the sediments of the “Randu pļavas” lagoon has likely formed from reeds and other plants, and only a very small proportion has formed from algae or other various microorganisms.
3.2. Water Analysis
One of the most important abiotic factors of lagoons is the presence of brackish water, which affects the concentration of several compounds in the water, such as chlorine and sulfates (
Table S1). During the period of our study, we did not identify areas with higher sulfate or chlorine concentrations than seawater, but in closed parts of the lagoons, there were periods when some parameters could reach very high levels and even start forming H
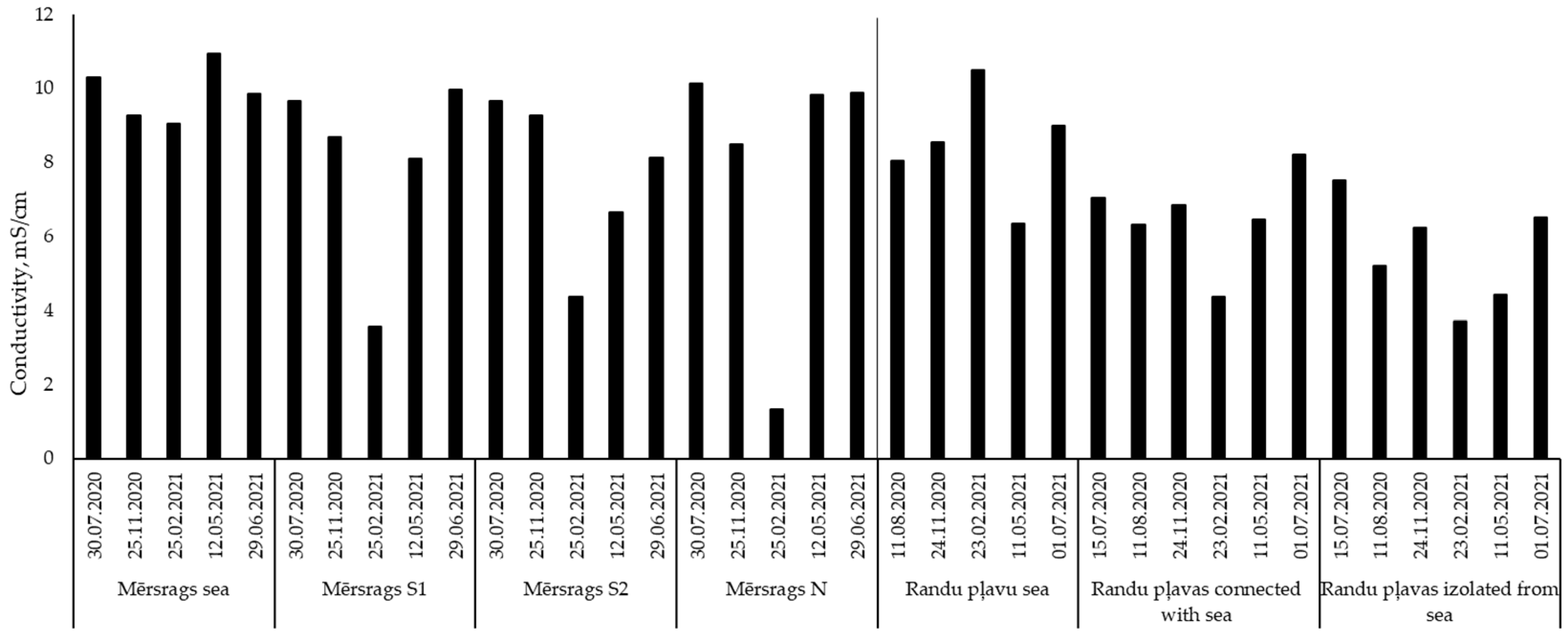
2S due to anoxic conditions. In lagoons that are still connected with the sea, the electrical conductivity of the water is usually relatively constant and corresponds with values measured in the sea (which are a bit higher in the western part of the Gulf of Riga). The results of water electrical conductivity measurements were used to characterize the amount of dissolved salts and the impact of brackish water. In the studied lagoons, however, the electrical conductivity of the parts of the lagoons that have become separated from the sea can vary significantly (3.56–9.95 µS/cm) (
Figure 6), depending on the intensity of water exchange with the sea, as well as evaporation and precipitation rates. Seasonal fluctuation of water EC across such a wide range affects the dominant processes in the lagoons, and, therefore, determines the uniqueness of such ecosystems. Winter conditions appear to limit water exchange between the lagoons and the sea due to ice blocking an already weak connection, as well as the lower filtration rates through sandy sediments—the initial dunes. During these periods, sources and effects of freshwater intensify. Nevertheless, even parts of the lagoon without direct connection to the sea continue to have higher EC than freshwater bodies.
In winter periods, not only is water exchange limited but gas exchange also lessens, and dissolved oxygen drops to 0.36 mg/L (
Figure 7), which, in turn, influences biological constituents and organic matter decomposition. In the Mērsrags lagoon during the winter season, such conditions led to fish death due to insufficient oxygen content. During other seasons, the oxygen level in these lagoons was better, even though in some cases oxygen content nearly exceeded 5 mg/L due to a combination of water exchange rates and local conditions. In cases where there were no major negative aspects, oxygen conditions were optimal.
Shallow, fragmented lagoons with limited water exchange can create specific local conditions, although they are not persistent/stable, and the next rainfall or water surge from the sea can change the conditions. Another aspect is the ambient temperature, which in summer 2021 ranged from 23.6 °C to 30.1 °C. This may affect the content of oxygen and the conditions for the development of biological constituents.
Increased anthropogenic pressure on lagoons can also be determined by the concentration of nutrients that contribute to eutrophication. In summer, the studied lagoons mostly have a N:P mass ratio above 17, indicating that phosphorus is the limiting element for algal growth; in some cases, the ratio is between 10 and 17, indicating that either of the nutrients may be limiting [
38]. Phosphorus concentrations typically increase sharply in winter, when organic residues decompose and mineralize and assimilation by plants and algae is absent (
Figure 8).
Since the Baltic Sea is considered eutrophic then, similar to phosphorus, the changes in nitrogen compound concentration in the lagoons also fluctuate after freshwater exchange. In periods with lower water exchange rates, noticeable saturation with nutrients occurs, at times even leading to the formation of ammonia. This pattern is highlighted in the parts of the lagoons considered closed to the sea, such as the closed part of the “Randu pļavas” lagoon, where concentrations of nitrates and ammonia can reach 11.96 and 1.27 mg/L, respectively (
Figure 9). Like the other parameters we measured in the lagoons, N and P concentrations fluctuated, but overall, the Mērsrags lagoon had higher values than the “Randu Pļavas” lagoon, reflecting a more pronounced anthropogenic impact and less freshwater input in the form of precipitation.
3.3. Macrophyte Species Composition
The studied lagoons are characterized by dense helophyte stands, where Phragmites australis and Typha angustifolia dominate. Species richness is higher in the lagoon parts connected with the sea.
In the “Randu pļavas” lagoon, the total coverage of macrophytes was 70%. Helophyte stands occur all along the shore, as well as in shallower parts of the lagoon. The dominant helophyte species were P. australis, Bolboschoenus maritimus, and Scirpus tabernaemontani. Submerged macrophytes grow in sparse stands, and the most common species are Najas marina, Zannichellia palustris, Ruppia maritima, Chara aspera, C. contraria, and Potamogeton pectinatus. The southern part of the lagoon is overgrown, and open water areas are small, and their dominating macrophyte species are P. australis, Ceratophyllum submersum, and N. marina. Species such as Z. palustris and R. maritima are rare and protected in Latvia.
In the part of the Mērsrags lagoon located south of the port, the total coverage of macrophytes was 90%. Dense P. australis and T. angustifolia stands occur all along the shore. The most common submerged macrophyte species are P. pectinatus, N. marina, and C. submersum. In some parts of the lagoon, the free-floating species Lemna minor occurs. The macrophyte species composition indicates a high nutrient content in the water.
In the Mērsrags lagoon located north of the port, the total coverage of macrophytes was 40%. The dominant species in the lagoon is P. australis; submerged species occur rarely due to higher water depths. Sparse stands of Myriophyllum spicatum, P. perfoliatus, P. pectinatus, and C. submersum have formed.
3.4. Macroinvertebrate Species Composition
In the part of the Mērsrags lagoon located south of the port, thirteen macroinvertebrate taxa were recorded altogether, with nine taxa in the southern part of the lagoon and eight taxa in the northern part. In both parts of this lagoon, the most abundant taxon was Chironomidae. During fieldwork, the water bug species Ranatra linearis and Ilyocoris cimicoides were recorded in both parts of the lagoon. In addition, the legally protected dytiscid Dytiscus latissimus was observed during the sampling. This species is listed in Annex II and Annex IV of the European Council Directive 92/43/EEC. Several species of Odonata were present in the samples; only one mayfly species, Cloeon dipterum, was found.
The part of the Mērsrags lagoon located north of the port is also characterized by low taxa richness, with only nine taxa found. This part of the lagoon is distinct in terms of aquatic snail species, which were not found in the southern lagoon. The snail species collected were Radix auricularia, R. balthica, and Stagnicola palustris. The dominant taxa were chironomids and amphipods Gammarus zaddachi.
Among the studied waterbodies of the “Randu pļavas” lagoon, the greatest taxa diversity and abundance was found in the isolated waterbody. Characteristic species for eutrophic waterbodies were found, including water bugs (Ilyocoris cimicoides, Corixidae), amphipods (Gammarus spp.), isopods (Asellus aquaticus), and coleopterans (Dytiscidae, Hydrophilidae, Haliplus sp.), as well as a high abundance of Chironomidae larvae and a low abundance of Oligochaeta.
In the waterbody connected with the sea, the most abundant taxon was also Chironomidae. This waterbody had a relatively high density of Coenagrionidae larvae (including Coenagrion pulchellum), a very small abundance of Corixidae, amphipods Gammarus sp., dipterans (Tabanidae, Chrysops sp.), and snails, Radix balthica, as well as empty snail shells of the species Lymnaea stagnalis and Bithynia tentaculata and shell remains of Cirripedia, Amphibalanus improvisus.
According to the EU Water Framework directive criteria applying the LLMMI multimetric index [
24], ecological quality in the Mērsrags lagoon was bad to very bad (S1—bad, S2—bad, N—very bad, respectively) and very bad in the “Randu pļavas’’ lagoon—very bad.
3.5. Zooplankton Community Structure
The zooplankton community structure, abundance, and biomass differed between the lagoons. In total, 39 taxa were recorded. The zooplankton community in the lagoons consisted of a wide range of salinity-tolerant taxa with different feeding functional groups, mainly euryhaline Rotifera (
Table 3,
Figure S1), followed by a poor representation of Cladocera (epibenthic/benthic fine-meshed microfilter Chydoridae) and Copepoda (littoral–benthic, epibenthic
Acanthocyclops sp., euryhaline
Eurytemora affinis and Harpacticoida) taxa (
Table 3). In addition,
Brachionus plicatilis,
Keratella cochlearis var.
recurvispina,
Keratella cruciformis,
Keratella quadrata var.
platei, and
Synchaeta baltica were found among Rotifera marine species. Also found were species characteristic of eutrophic water bodies—
Brachionus angularis,
Keratella quadrata, and
Pompholyx sulcate—mainly in the Mērsrags lagoon.
The “Randu pļavas” lagoon is diverse, according to the Simpson index, and more heterogeneous compared to the Mērsrags lagoon. The Simpson reciprocal index scores for the waterbodies of the “Randu pļavas” lagoon are open waterbody—7.82; closed waterbody—4.67. The index scores for the Mērsrags lagoon are southern part 1—1.61; southern part 2—2.08; and northern part—3.06. Total zooplankton abundance in both the open and closed waterbodies of the “Randu pļavas” lagoon varied from 155,000 to 223,750 individuals m−3, while the biomass varied from 0.10 to 0.19 g m−3. In the Mērsrags lagoon, total zooplankton abundance in both southern water bodies (S1 and S2) ranged from 613,750 to 979,333 individuals m−3, while the biomass varied from 0.34 to 0.35 g m−3. The highest values of total zooplankton abundance and biomass were found in the northern waterbody of the Mērsrags lagoon, with 5,284,000 individuals m−3 and 3.30 g m−3, respectively.
3.6. Phytoplankton Community Structure
The Mērsrags lagoon S1 waterbody is characterized by small-sized cryptophytes
Cryptomonas spp., chlorophytes
Monoraphidium sp.,
Desmodesmus sp., and diatoms
Nitzschia spp. The Mērsrags S2 waterbody is characterized by small-sized cryptophytes
Cryptomonas spp.,
Rhodomonas spp., chlorophytes
Monoraphidium sp., and dinoflagellates, as well as
Woloszinskia halophila, which is typical of brackish and saline waters. The Mērsrags northern waterbody is characterized by pennate diatoms
Nitzschia acicularis, which are abundant and multiplying. Euglenophytes
Euglena sp.,
Euglena acus,
Phacus sp., and a very small size of cryptophytes were dominant. Chlorophytes
Desmodesmus sp. and dinoflagellates, as well as
Woloszinskia halophila, which is typical of brackish and saline waters, were observed. The “Randu pļavas” waterbodies isolated from the sea lagoon are characterized by large amounts of euglenophytes
Euglena sp.,
Lepocinclis sp., and cryptophytes
Cryptomonas sp., as well as diatoms
Nitzschia sp. The “Randu pļavas” waterbodies connected with the sea lagoon are characterized by a low species diversity. Seashore zone indicators of eutrophic waters include the harmful cyanobacteria
Aphanizomenon flos-aquae and the pennate diatoms
Nitzschia acicularis. In August 2020, massive phytoplankton blooms were observed in the Mērsrags northern waterbody. In 2020 and 2021, the Mērsrags lagoon exhibited comparatively high to very high phytoplankton biomass (
Figure 10 and
Figure 11), which is characteristic of eutrophic to hypertrophic waters.
3.7. Microplankton Communities
In lagoon water samples prepared for the characterization of phytoplankton community structure, we observed the presence of microplankton. Although microplankton were not estimated as biomass, we identified them as a widely represented species. In Mērsrags S1 and S2, the marine ciliate Mesodinium rubrum was found in abundance but was less abundant in the northern part of the Mērsrags lagoon, which was smaller but also much deeper. Less abundant in comparison to Mesodinium rubrum in the Mērsrags lagoon was Tintinidium sp. The marine ciliate Mesodinium rubrum and Tintinidium sp. were found in an isolated part of the Randu pļavas lagoon, but despite reduced water exchange periodically, seawater flows into this part of the lagoon. The “Randu pļavas” part of the lagoon is connected with the sea lagoon and does not have a rich diversity of phytoplankton but has a noticeable amount of microplankton with several species. The most widespread microplankton was Mesodinium rubrum but in this lagoon, Tintinidium sp., and Vorticella sp. were also observed.
4. Discussion
The studied lagoons differ in some spatial aspects, but a feature that they have in common is dynamic conditions with regular water exchange from the sea, as well as inland freshwater sources. Although these ecosystems are listed as protected and endangered, strong impacts from anthropogenic activities can, nevertheless, be recognized. One of the major factors in this case is morphological modification due to human activities [
12,
39] in the coastal area of Latvia. In terms of natural factors, the most significant ones are water cycling and coastal processes (which, together with sediment transport along the shore [
40], are causing accumulation and gradually isolating the lagoon from the sea). Sediment transport and its potential for accumulation are significantly higher on the eastern side of the Gulf of Riga [
40], where they are affecting the “Randu pļavas” lagoon. Limited water exchange with the sea causes significantly higher variability in the determined lagoon’s water electrical conductivity; the proportion of freshwater is increasing, thereby promoting overgrowth [
41] and the reduction in the size of the lagoon. The sedimentation of organic material is not among the dominant aspects reducing the size of the studied lagoons; instead, it is the accumulation of sand due to coastal processes, water flows, and the slowed speed of water cycling in overgrown areas. Overall, factors such as EC, temperature, freshwater inflow, brackish water circulation, and the enrichment of nutrients, together with organic sediments, are creating unique and variable conditions in these lagoons. The aforementioned are also affecting the diversity of plant and animal communities in the lagoons [
42]. Water exchange at the current rate of water flows has reduced the accumulation of sediments in the studied lagoons, especially if there are no deeper sections (the Mērsrags waterbody north of the port is excluded in this regard), leading to the dominance of coarse-grained sandy sediments. Even the closed parts of the “Randu pļavas” lagoon are flooded regularly, and in contrast to the Mersrags lagoon, more shells and their remains (both visually and according to analyses of their carbonate content) can be observed in the “Randu pļavas” lagoon; the higher pH values detected in the “Randu pļavas” lagoon also reflect a higher occurrence of flooding. However, a more significant influence on sediment pH levels can come from overall conditions, including oxygen content and the presence of hydrogen sulfide; among the samples taken, the lowest pH values were found in the sampling points where there was a noticeable aroma of hydrogen sulfide and stagnant water.
In our study, high water temperatures were observed in all shallow lagoons during the summer season. Although recent studies confirm average temperature rise in waterbodies [
43], processes in lagoons can create more extreme conditions and, therefore, values. Apart from morphological fragmentation, water exchange is an essential element, which is generally good in lagoons connected to the sea. These lagoons are affected by sea level, wind, and the size of the connection, but the total water level variation can be considerable, and a study of the Vistula lagoon showed a range of −0.76 m to 1.46 m [
44]. The studied lagoons are significantly smaller than the Vistula lagoon and are exposed to wind differently due to their location in the Riga Gulf. For example, in November 2020, in the Mērsrags lagoon, there were no significant water level variations, while in the “Randu pļavas”, a water level rise of 0.48 m was detected. Water inflow and outflow create movements of water masses and, therefore, sediment transport and organisms found in water can also originate from the sea and local or freshwater communities. Regarding aspects without morphology, an important aspect is exposure to wind, which is higher in coastal areas, but a significant role can be played by macrophytes, especially reeds, because they can provide shelter for the fauna and reduce the influence of water movements on macroinvertebrate fauna [
45], as well as reduce wind exposure influence on the littoral macrozoobenthic communities [
46]. In a study by Bielczyńska A. [
46], the burrowing benthic macroinvertebrates Chironomidae and Oligochaeta were less sensitive to wind exposure; these species are also abundant in the “Randu pļavas” lagoon. Although on the one hand, reeds can have positive aspects for reducing wind speed, on the other hand, we observed patterns where in reed rush patches the shading effect of reed stems resulted in the inhibition of the development of other plants and their communities. The dominance of reeds creates an abundance of local organic material, which is not always carried away in the sea but accumulates in lagoons, especially in the closed part of the “Randu pļavas” lagoon. In the long term, this also influences sediments, although their accumulation is limited. However, due to diagenetic processes and the dynamic conditions found in the studied lagoons, the results of elemental ratios can be modified and should be considered carefully [
47]. It is suggested that the microbial decay of marine particulate matter of planktonic origin produces a higher amount of N than the decay of macrophytes [
47], but since intense sediment leachate occurs in the studied lagoons, these aspects can be difficult to track through sediment analysis.
Water chemistry similar to water exchange rates is very much influenced by the sea and its annual fluctuation of water composition, which is very similar to other lagoons in the Baltic Sea [
48]. Only Curonian and Vistula lagoon’s significant source of nutrients can be from rivers [
48] but in the studied lagoons, such direct sources are relatively difficult to reveal. Nutrient input in the “Randu pļavas” lagoon from land area can be considered disperse and in the Mērsrags lagoon, there is a more direct influence from the port and city, which can induce potential sources of sewage water. During periods when water exchange with the sea does not take place, dissolved oxygen concentrations decrease and ammonia content increases, and EC, sulfate, and chloride concentrations decrease due to dilution with inflowing freshwater, despite evaporation. The productivity of these ecosystems refers to the noticeable proportion of organic N, which was also found by Stakėnienė, R [
48]. This affects other water parameters, like color, which have the highest values in the “Randu pļavas” lagoon, where, without ecosystem productivity and the content of biological constituents, can result in a higher content of organic carbon (
Table S1).
Winters, when ice forms that can freeze to the bottom, have a significant impact on the content of dissolved oxygen in the studied lagoons and, therefore, on the existence of organisms. In the studied lagoons, macroinvertebrate species richness is low, and ecological quality is bad to very bad, which can be explained by multiple factors, including previously described dynamic environmental conditions and a low content of dissolved oxygen. These habitats are known to harbor sea, brackish, and freshwater species [
49], although we did not observe typical sea species in the waterbodies under our study. The complexity of factors also affects the feeding conditions, e.g., Morkūnė et al. [
50] studied the Curonian lagoon and found that in the southern site, a larger proportion of sedimentary organic matter and seston dominated the diet of amphipods and gastropods, whereas in the northern site, with marine water inflow, due to less eutrophic conditions and increased water transparency, epiphytes and sedimentary organic matter were the primary food sources for the studied invertebrates. Despite the well-known fact that lagoon habitats are particularly exposed to non-native and invasive species [
10], in our study, we did not find any invasive macroinvertebrate species; widespread non-native species characteristic to the coastal zone, e.g.,
Gammarus tigrinus and others, might, however, be present [
51]. The low status of ecological quality in studied lagoons, according to the LLMMI, indicates that a specific assessment method using macroinvertebrates should be taken into consideration. However, separating natural from anthropogenic stressors might be problematic since lagoons are affected by numerous factors [
16].
Overall, the zooplankton community structure of the lagoons under study corresponds to the composition of typical planktonic and epibenthic/benthic/periphytic euryhaline and marine taxa for wide estuarine and coastal areas of the Baltic Sea [
52,
53]. The zooplankton community structure—as indicated by functional traits, taxonomic composition, diversity (Simpson’s reciprocal index), abundance, and biomass—shows distinct habitats in the Mērsrags and “Randu pļavas” lagoons along the Baltic Sea. These differences have likely been driven by environmental conditions, seeing that these sorts of dynamic processes and the eutrophication of lagoons are characteristic of Baltic Sea coastal areas (beaches, lagoons, estuaries, coastal lakes) [
54,
55,
56,
57,
58,
59,
60], as is the growth of covering macrophyte beds [
61]. The higher diversity of zooplankton and the dominance of benthic–psammic–periphytic rotifers and epibenthic harpacticoids in the “Randu pļavas” lagoon indicate dynamic conditions marked by water flow (mixing) and sediment re-suspension due to shallowness and covering macrophyte beds, which is congruent with other studies [
56,
57,
62,
63]. This could also explain the low abundance and biomass of zooplankton. Low zooplankton diversity and a high abundance and dominance of planktonic rotifers (mainly microphagous/polyphagous
Keratella spp.,
Brachionus angularis), which are typical for eutrophicated water bodies [
64], as well as the presence of marine species, indicates the occurrence of water exchange with the sea, especially in the northern waterbody of the Mērsrags lagoon. The zooplankton community structure in the Mērsrags lagoon also indicates high trophy. These observations correspond to the investigations of other lagoons and coastal lakes [
15,
54,
55,
56,
60,
65]. For example, in the Latvian eutrophic coastal lowland lakes Juglas and Ķīšezers, only one species from Rotifera,
Keratella quadrata, dominated, and the structure of zooplankton communities was characterized by a decreased species richness, numbers of dominating species, and species diversity indices [
66].
The overall phytoplankton diversity was constrained, characterized by the prevalence of a limited number of algal divisions and species. Freshwater species dominated, accompanied by the occurrence of brackish and saline water species. Undoubtedly determining the high biomass of phytoplankton provides reasonably high trophy status, which can be consumed by zooplankton, ciliates, and other consumers. Massive algae blooms can occur frequently and were detected in northern part of the Mērsrags lagoon and the “Randu pļavas” lagoon, including a bloom of cyanobacteria
Aphanizomenon. In the study of eutrophic to hypertrophic in the Curonian lagoon, a similar pattern was characteristic of a cyanobacterial, mainly
A. flos-aquae, which blooms in summer [
67]. Gasiūnaitė et al. [
68] found that the range of phytoplankton biomass was similar to our study, varying from 10.3 to 30.2 mg/L (the Mērsrags N had a higher biomass formed by diatoms in 2020) and reaching the highest values during an intense
A. flos-aquae bloom in 2002 [
69]. Compared to the lagoon-type lake Engures, which is connected to the Baltic Sea and is located near the Mērsrags lagoon, where long-term studies have been carried out [
70], differences in species composition and phytoplankton biomass are very low (0.13–0.39 mg/L). Contrary to the lagoon phytoplankton communities, lake phytoplankton community structure was more homogeneous. In the lagoons connected to the sea, dependence on phytoplankton dynamics in the Gulf of Riga has also been observed.
Additionally, ciliate species were also found in phytoplankton samples in the studied lagoons. Without two abundant ciliate species,
Tintinidium sp. and mixotrophic ciliate
Mesodinium rubrum, the presence of
Vorticella sp. was observed. Tintinnids are frequently found in shallow lakes and lagoons [
71] and were the dominant group in the ciliate community study in the Curonian lagoon, with abundance peaks in the spring and summer seasons [
72].
Mesodinium rubrum displays a wide size distribution (from 15 to 70 μm); it is abundant in the Baltic Sea and was found in other studied lagoons [
72,
73,
74,
75] in noticeable quantity. Studies suggested [
72,
76] that
Mesodinium rubrum may be negatively affected by the rapid fluctuation of salinity [
72] but due to its mixotrophic ability, it is less affected by the depletion of dissolved inorganic nutrients [
73], which can be noticed in the Mērsrags lagoon with phosphorus. Despite the dynamic hydrological regime in the studied lagoons, the frequency of brackish water inflows diluted by freshwater have longer intervals than are necessary for the development of ciliate species.
Vorticella sp. is more sensitive to abiotic factor (pH, turbidity, etc.) variability [
77], which is more intense in the closed part of the Randu pļavas lagoon; thus, the presence of these individuals is significantly lower.
The achieved results give insight into endangered ecosystems and as management measures, we recommend activities to maintain the hydrological continuum of small lagoons, which were also suggested by Ligorini, V. [
78] in studies about small Mediterranean coastal lagoons. Further steps could be reed cutting in winter periods (at least some parts of the “Randu pļavas” lagoon). A viable option could be placing stones in the lagoons because deeper areas will form around them because of the disturbance to water flow, and during winter even air pockets can form under the ice. Measures that include potential morphological changes can be considered, such as the deepening of some parts of lagoons; for example, in the form of a ditch in the sediments located perpendicular to the sea but without direct connection. This may prevent rapid sediment accumulation during water flows, provide deeper parts for organisms with potentially lower water temperatures, and provide bigger water volumes in winter. Above all, it is worth mentioning that activities limiting eutrophication can be helpful in maintaining healthy lagoon ecosystems.