Development of a Chemiluminescent Method in a Microfluidic Device for Ultrasensitive Determination of Okadaic Acid with Highly Efficient Aptamer-Based Isolation
Abstract
1. Introduction
2. Materials and Methods
2.1. Material
2.2. Coupling of Okadaic Acid with Horseradish Peroxidase
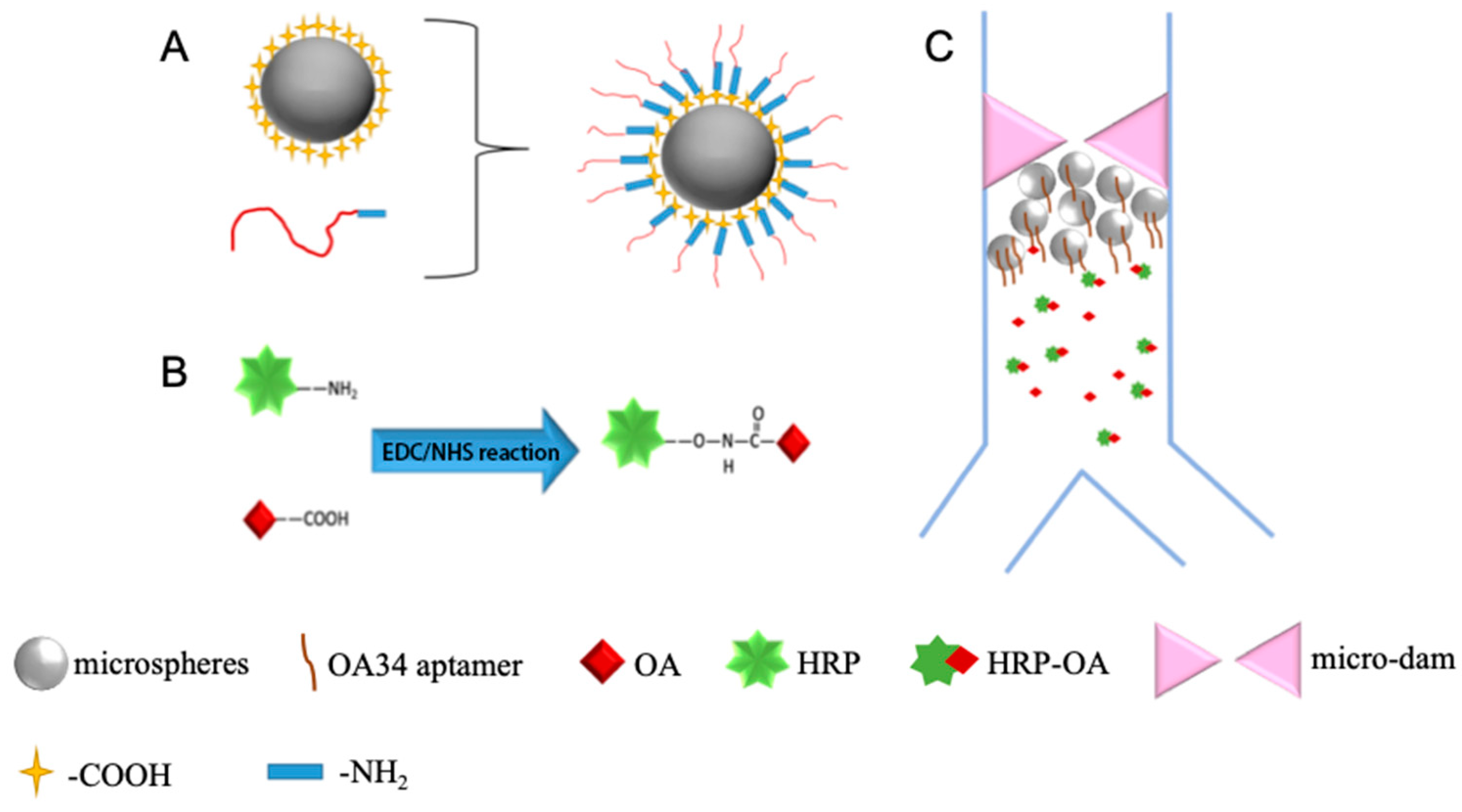
2.3. Immobilization of Aptamers on Polystyrene Microspheres
2.4. The Competitive Chemiluminescent Method Developed in the Microfluidic Device
2.5. Real Sample Preparation for OA Detection
3. Results and Discussion
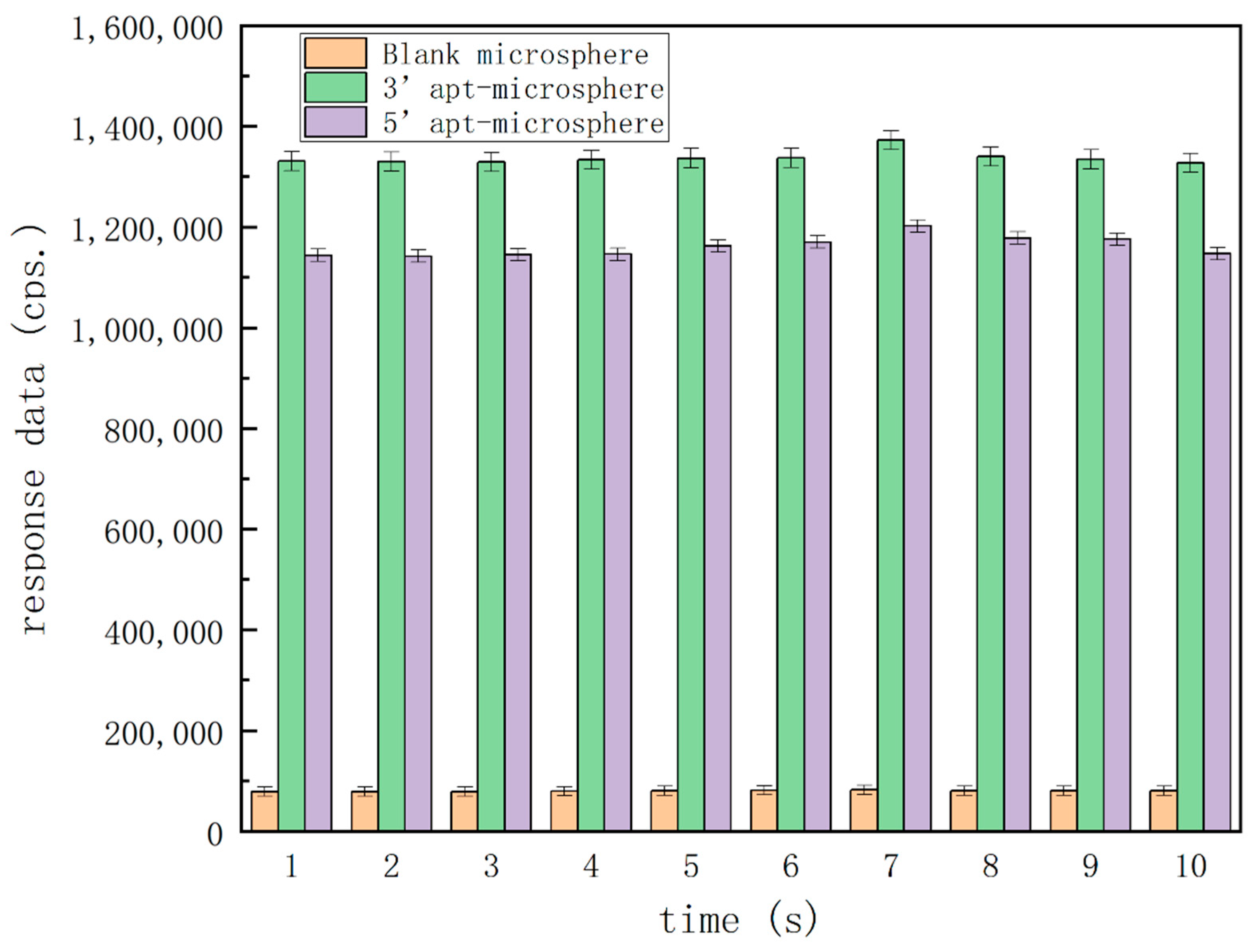
3.1. Characterization of the Immobilization of Aptamers
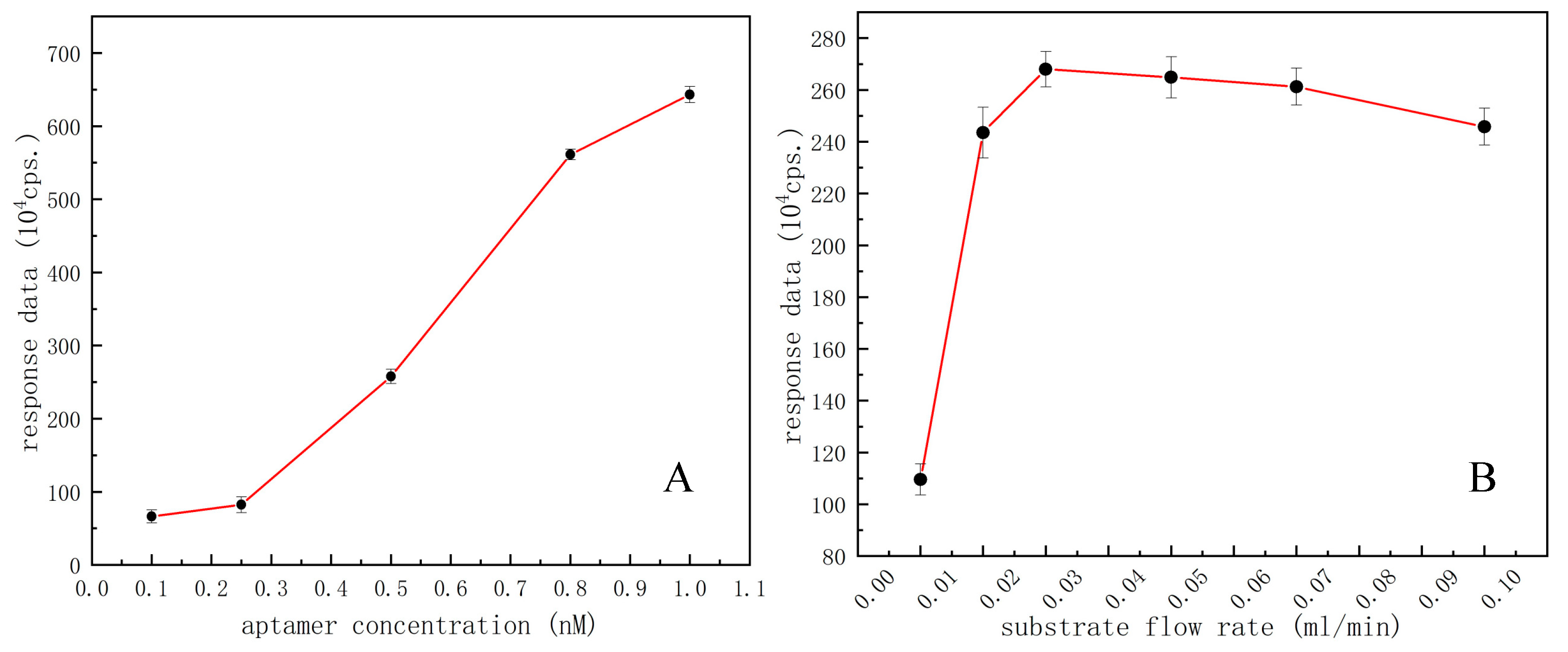
3.2. Optimizing of Key Experimental Parameters
3.3. Development of Calibration Curve for Quantitation
3.4. Real Samples OA Detection and Method Accuracy Characterization
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Falconer, I.R. Algal Toxins in Seafood and Drinking Water; Academic Press: Waltham, MA, USA, 1993. [Google Scholar]
- Yasumoto, T.; Oshima, Y.; Sugawara, W.; Fukuyo, Y.; Oguri, H.; Igarashi, T.; Fujita, N. Identification of Dinophysis fortii as the Causative Organism of Diarrhetic Shellfish Poisoning. Nippon. Suisan Gakkaishi 1980, 46, 1405–1411. [Google Scholar] [CrossRef]
- Murata, M.; Shimatani, M.; Sugitani, H.; Oshima, Y.; Yasumoto, T. Isolation and structural elucidation of the causative toxin of the diarrhetic shellfish poisoning. Nihon-Suisan-Gakkai-Shi 1982, 48, 549–552. [Google Scholar] [CrossRef]
- Traoré, A.; Baudrimont, I.; Ambaliou, S.; Dano, S.D.; Creppy, E.E. DNA breaks and cell cycle arrest induced by okadaic acid in Caco-2 cells, a human colonic epithelial cell line. Arch. Toxicol. 2001, 75, 110–117. [Google Scholar] [CrossRef] [PubMed]
- Louzao, M.C.; Fernández, D.A.; Abal, P.; Fraga, M.; Vilariño, N.; Vieytes, M.R.; Botana, L.M. Diarrhetic effect of okadaic acid could be related with its neuronal action: Changes in neuropeptide Y. Toxicol. Lett. 2015, 237, 151–160. [Google Scholar] [CrossRef]
- Louzao, M.C.; Costas, C.; Abal, P.; Suzuki, T.; Watanabe, R.; Vilariño, N.; Carrera, C.; Boente-Juncal, A.; Vale, C.; Vieytes, M.R.; et al. Serotonin involvement in okadaic acid-induced diarrhoea in vivo. Arch. Toxicol. 2021, 95, 2797–2813. [Google Scholar] [CrossRef]
- Munday, R. Is protein phosphatase inhibition responsible for the toxic effects of okadaic Acid in animals? Toxins 2013, 5, 267–285. [Google Scholar] [CrossRef] [PubMed]
- Vilariño, N.; Louzao, M.C.; Abal, P.; Cagide, E.; Carrera, C.; Vieytes, M.R.; Botana, L.M. Human Poisoning from Marine Toxins: Unknowns for Optimal Consumer Protection. Toxins 2018, 10, 324. [Google Scholar] [CrossRef]
- Tsagkaris, A.S.; Nelis, J.L.D.; Ross, G.M.S.; Jafari, S.; Guercetti, J.; Kopper, K.; Zhao, Y.; Rafferty, K.; Salvador, J.P.; Migliorelli, D.; et al. Critical assessment of recent trends related to screening and confirmatory analytical methods for selected food contaminants and allergens. TrAC Trends Anal. Chem. 2019, 121, 115688. [Google Scholar] [CrossRef]
- Ball, H.J.; Finlay, D.; Reilly, G.A. Sandwich ELISA detection of Mycoplasma bovis in pneumonic calf lungs and nasal swabs. Vet. Rec. 1994, 135, 531–532. [Google Scholar] [CrossRef]
- Rossetti, B.C.; Frey, J.; Pilo, P. Direct detection of Mycoplasma bovis in milk and tissue samples by real-time PCR. Mol. Cell. Probes 2010, 24, 321–323. [Google Scholar] [CrossRef]
- Ekker, M.; Akimenko, M.-A.; Bremiller, R.; Westerfield, M. Regional expression of three homeobox transcripts in the inner ear of zebrafish embryos. Neuron 1992, 9, 27–35. [Google Scholar] [CrossRef]
- Hayat, A.; Barthelmebs, L.; Sassolas, A.; Marty, J.-L. An electrochemical immunosensor based on covalent immobilization of okadaic acid onto screen printed carbon electrode via diazotization-coupling reaction. Talanta 2011, 85, 513–518. [Google Scholar] [CrossRef] [PubMed]
- Nelis, J.L.D.; Migliorelli, D.; Mühlebach, L.; Generelli, S.; Stewart, L.; Elliott, C.T.; Campbell, K. Highly sensitive electrochemical detection of the marine toxins okadaic acid and domoic acid with carbon black modified screen printed electrodes. Talanta 2021, 228, 122215. [Google Scholar] [CrossRef] [PubMed]
- Tuerk, C.; Gold, L. Systematic evolution of ligands by exponential enrichment: RNA ligands to bacteriophage T4 DNA polymerase. Science 1990, 249, 505–510. [Google Scholar] [CrossRef]
- Eissa, S.; Ng, A.; Siaj, M.; Tavares, A.C.; Zourob, M. Selection and identification of DNA aptamers against okadaic acid for biosensing application. Anal. Chem. 2013, 85, 11794–11801. [Google Scholar] [CrossRef]
- Pan, C.; Zhu, X.; Yang, Y.; Wang, H.; Liu, J.; Chen, H.; Dong, Y. Rapid visual detection of okadaic acid based on aptamer. Chin. J. Anal. Lab. 2021, 40, 686–691. [Google Scholar] [CrossRef]
- Yan, X.; Qi, X.; Zhao, Y.; Li, L.; Ma, R.; Wang, L.; Wang, S.; Mao, X. Development of a Label-Free Colorimetric Aptasensor with Rationally Utilized Aptamer for Rapid Detection of Okadaic Acid. J. Ocean. Univ. China 2022, 21, 400–408. [Google Scholar] [CrossRef]
- Tian, Y.; Zhu, P.; Chen, Y.; Bai, X.; Du, L.; Chen, W.; Wu, C.; Wang, P. Piezoelectric aptasensor with gold nanoparticle amplification for the label-free detection of okadaic acid. Sens. Actuators B Chem. 2021, 346, 130446. [Google Scholar] [CrossRef]
- Zhao, P.; Liu, H.; Zhu, P.; Ge, S.; Zhang, L.; Zhang, Y.; Yu, J. Multiple cooperative amplification paper SERS aptasensor based on AuNPs/3D succulent-like silver for okadaic acid quantization. Sens. Actuators B Chem. 2021, 344, 130174. [Google Scholar] [CrossRef]
- Ma, J.; Wang, Y.; Liu, J. Biomaterials Meet Microfluidics: From Synthesis Technologies to Biological Applications. Micromachines 2017, 8, 255. [Google Scholar] [CrossRef]
- Franke, T.A.; Wixforth, A. Microfluidics for miniaturized laboratories on a chip. Chemphyschem 2008, 9, 2140–2156. [Google Scholar] [CrossRef]
- Fu, C.; Wang, Y.; Chen, G.; Yang, L.; Xu, S.; Xu, W. Aptamer-based surface-enhanced Raman scattering-microfluidic sensor for sensitive and selective polychlorinated biphenyls detection. Anal. Chem. 2015, 87, 9555–9558. [Google Scholar] [CrossRef]
- Wang, S.; Zheng, L.; Cai, G.; Liu, N.; Liao, M.; Li, Y.; Zhang, X.; Lin, J. A microfluidic biosensor for online and sensitive detection of Salmonella typhimurium using fluorescence labeling and smartphone video processing. Biosens. Bioelectron. 2019, 140, 111333. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Zhu, L.; He, P.; Zi, F.; Hu, X.; Wang, Q. Sensitive assay of Escherichia coli in food samples by microchip capillary electrophoresis based on specific aptamer binding strategy. Talanta 2019, 197, 284–290. [Google Scholar] [CrossRef] [PubMed]
- Dong, Y.; Xu, Y.; Liu, Z.; Fu, Y.; Ohashi, T.; Tanaka, Y.; Mawatari, K.; Kitamori, T. Rapid screening swine foot-and-mouth disease virus using micro-ELISA system. Lab Chip 2011, 11, 2153–2155. [Google Scholar] [CrossRef]
- Chinnappan, R.; AlZabn, R.; Mir, T.A.; Bader, M.; Zourob, M. Fluorometric determination of okadaic acid using a truncated aptamer. Microchim. Acta 2019, 186, 406. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Yu, X.; Zhao, J.; Shi, X.; Sun, A.; Jiao, H.; Xiao, T.; Li, D.; Chen, J. A fluorescence microplate assay based on molecularly imprinted silica coated quantum dot optosensing materials for the separation and detection of okadaic acid in shellfish. Chemosphere 2020, 246, 125622. [Google Scholar] [CrossRef]
- Castanheira, A.; Santos, M.; Rodriguez-Lorenzo, L.; Queirós, R.; Espiña, B. A novel microfluidic system for the sensitive and cost-efficient detection of okadaic acid in mussels. Analyst 2021, 146, 2638–2645. [Google Scholar] [CrossRef]



| Test Method | Recognition Element | LOD | Dynamic Range | Detection Time a/Total Analysis Time |
|---|---|---|---|---|
| Impedimetric aptasensor [16] | Aptamer | 70 pg/mL | 0.1 to 60 ng/mL | 45 min/- |
| Fluorometric determination using a truncated aptamer [27] | Aptamer | 39 pg/mL | 0.5 to 250 ng/mL | 30 min/ca. 50 min |
| Electrochemical method for immobilization of OA on screen printed carbon electrode [13] | Polyclonal antibody | 1.44 pg/mL | N/Ab | N/A b |
| Highly sensitive electrochemical detection with carbon black modified screen printed electrodes [14] | Polyclonal antibody | 150 pg/mL | 0.27 to 3.3 ng/mL | 2 min/ca. 10 min |
| A fluorescence microplate assay based on molecularly imprinted silica-coated quantum dot opto-sensing materials [28] | molecularly imprinted silica material-coated quantum dots (MIS-QDs) | 250 pg/mL | 10 to 100 ng/mL | 10 min/ca. 30 min |
| Microfluidic system to detect OA based on the protein phosphatase 1 inhibition colorimetric [29] | Inhibition of PP1 | 200 pg/mL | 1.47 to 506 ng/mL | 30 min/ca. 310 min |
| Aptamer-based chemiluminescent method in a microfluidic device (This work) | Aptamer | 12.4 pg/mL | 0.0413 to 4.02 ng/mL | 30 s/ca. 80 min |
| Spiked Concentration (nM) | Detection Concentration (nM, Mean ± SD) | Recovery (%, Mean) | CV (%) |
|---|---|---|---|
| 0.5 | 0.435 ± 2.0% | 90.6 | 4.4 |
| 1.0 | 0.952 ± 2.6% | 95.2 | 2.7 |
| 2.0 | 2.142 ± 9.1% | 107.1 | 4.2 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mao, L.; Zhao, Q.; Yang, Y.; Wang, S.; Dong, Y. Development of a Chemiluminescent Method in a Microfluidic Device for Ultrasensitive Determination of Okadaic Acid with Highly Efficient Aptamer-Based Isolation. Separations 2022, 9, 350. https://doi.org/10.3390/separations9110350
Mao L, Zhao Q, Yang Y, Wang S, Dong Y. Development of a Chemiluminescent Method in a Microfluidic Device for Ultrasensitive Determination of Okadaic Acid with Highly Efficient Aptamer-Based Isolation. Separations. 2022; 9(11):350. https://doi.org/10.3390/separations9110350
Chicago/Turabian StyleMao, Libing, Qi Zhao, Yan Yang, Sai Wang, and Yiyang Dong. 2022. "Development of a Chemiluminescent Method in a Microfluidic Device for Ultrasensitive Determination of Okadaic Acid with Highly Efficient Aptamer-Based Isolation" Separations 9, no. 11: 350. https://doi.org/10.3390/separations9110350
APA StyleMao, L., Zhao, Q., Yang, Y., Wang, S., & Dong, Y. (2022). Development of a Chemiluminescent Method in a Microfluidic Device for Ultrasensitive Determination of Okadaic Acid with Highly Efficient Aptamer-Based Isolation. Separations, 9(11), 350. https://doi.org/10.3390/separations9110350






