Phytochemical Profile of Asplenium aethiopicum (Burm. f.) Becherer Using HPTLC
Abstract
1. Introduction
2. Experimental
2.1. Collection of Plant Material
2.2. Preparation of Extracts
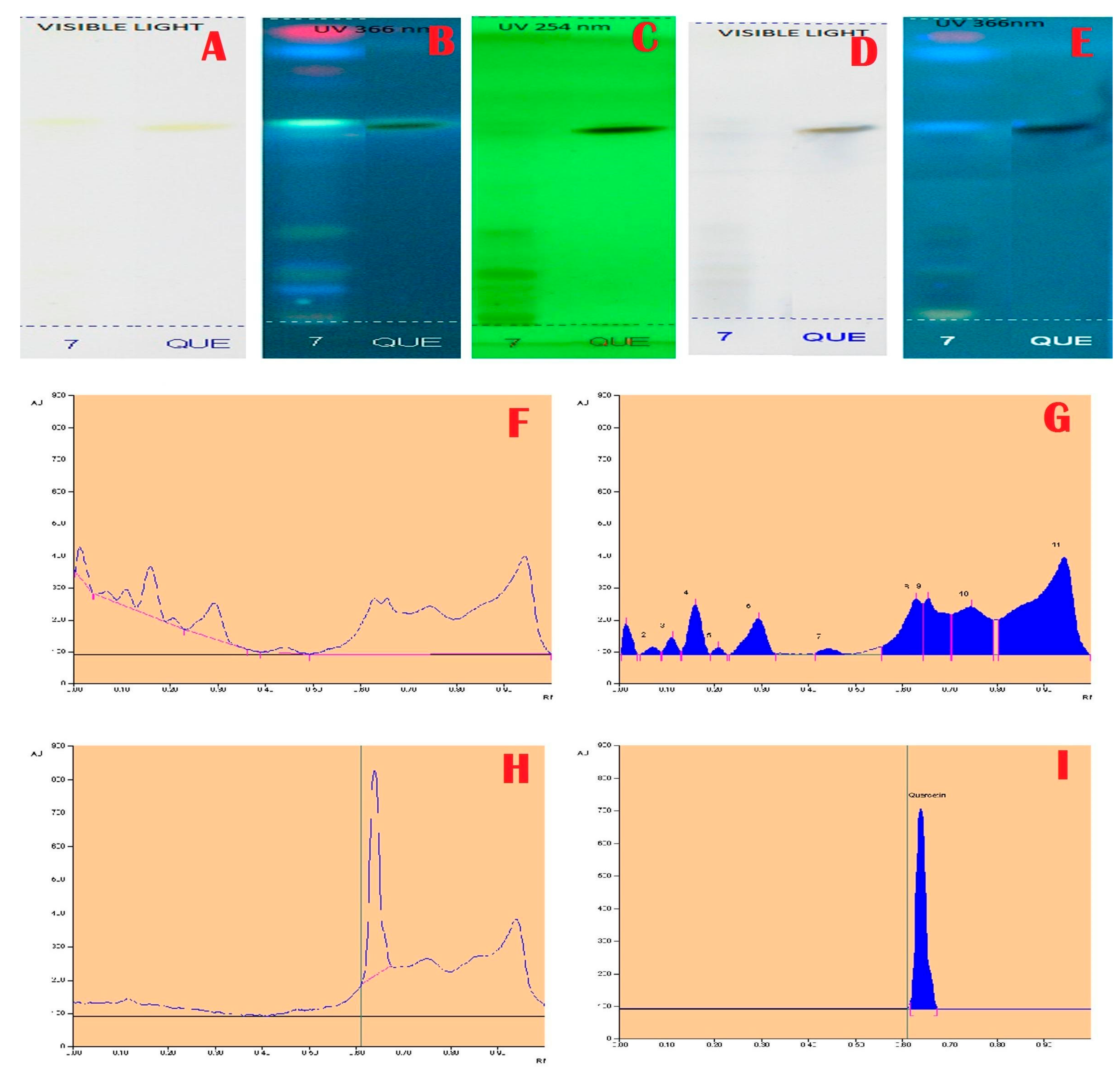
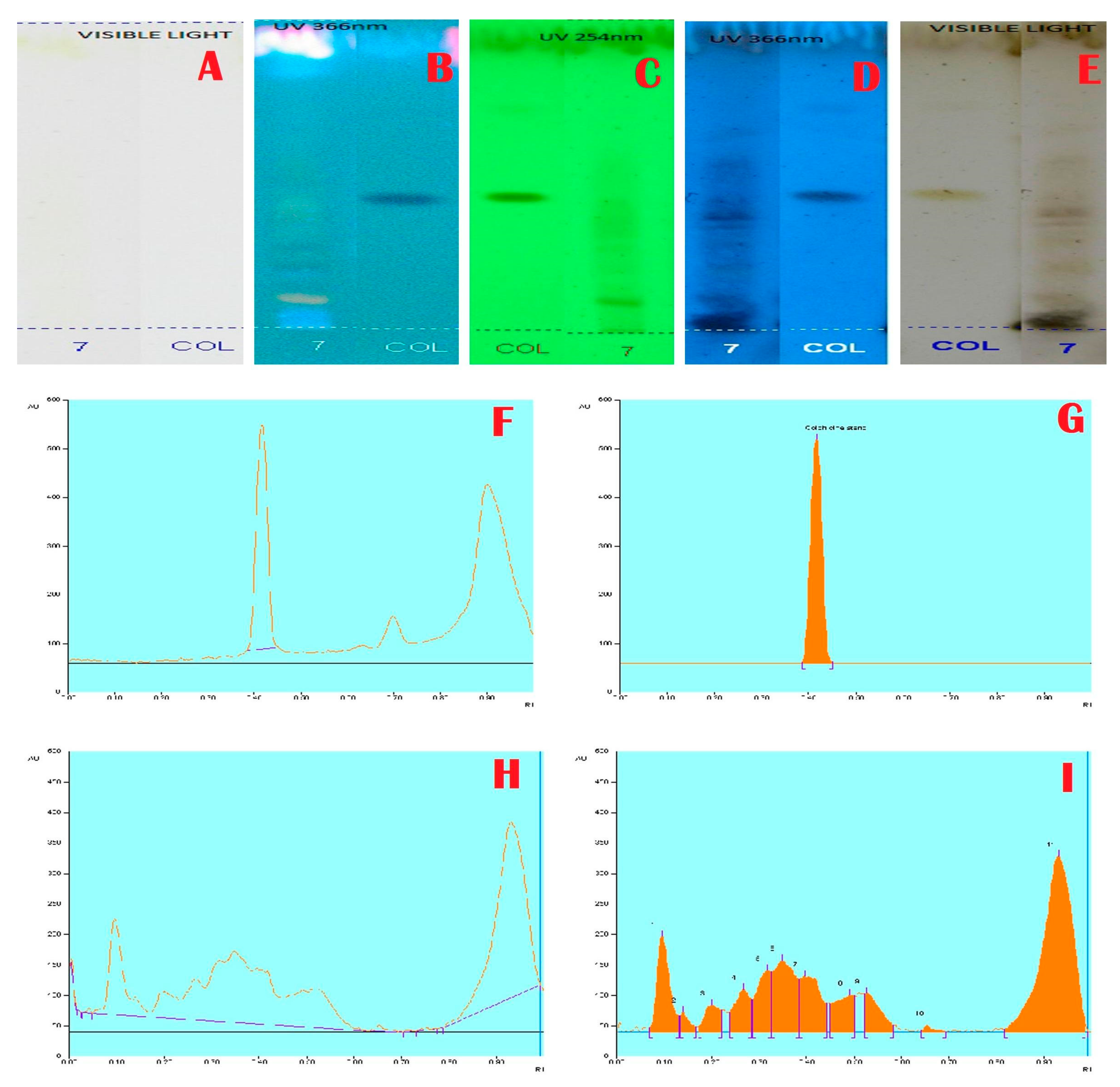
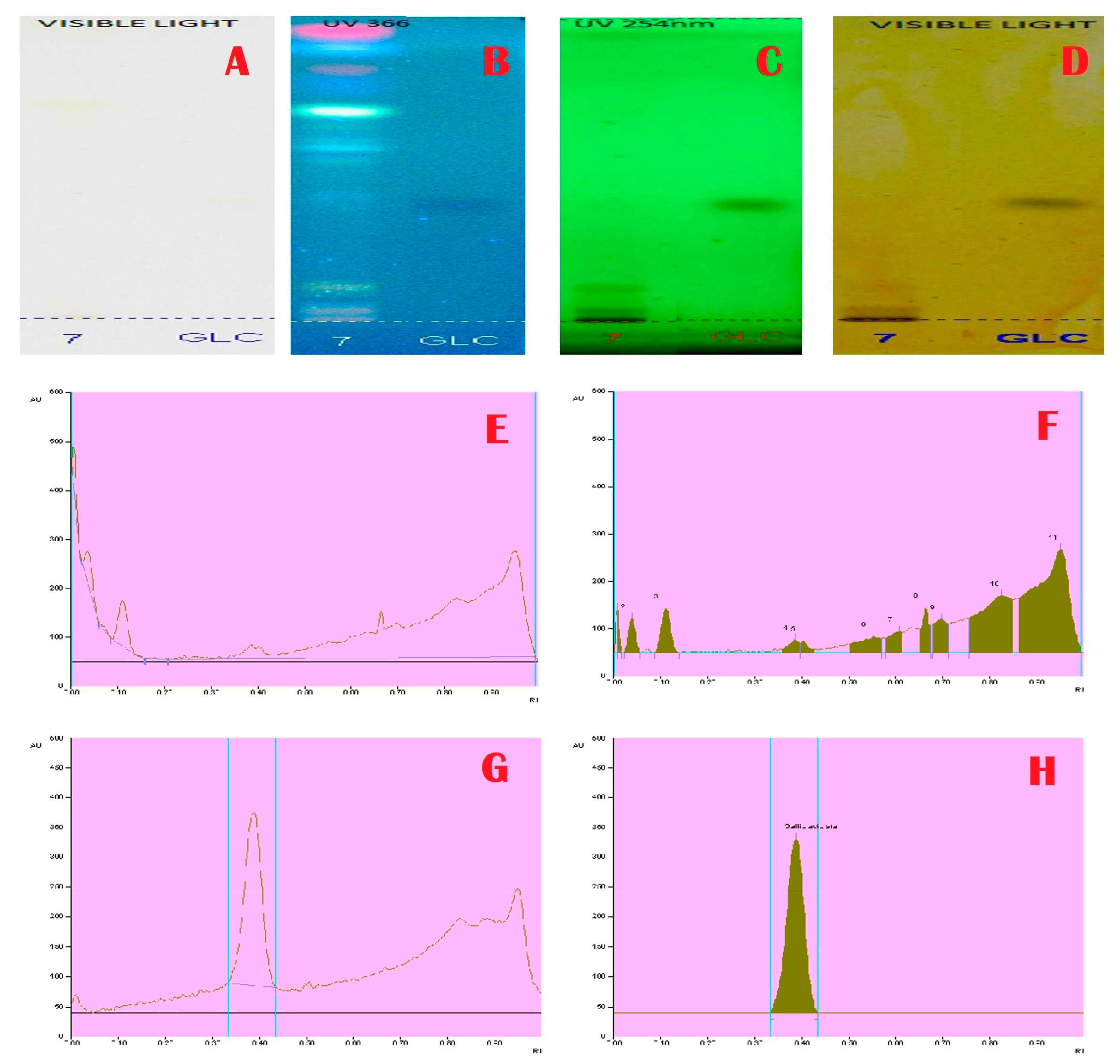
2.3. HPTLC Analysis
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Uma, B.; Prabhakar, K.; Rajendran, S. In Vitro Antimicrobial Activity and Phytochemical Analysis of Ficus religiosa L. and Ficus bengalensis L. against Diarrhoeal Enterotoxigenic E. coli. Ethnobot. Leafl. 2009, 13, 472–474. [Google Scholar]
- Aktar, M.W.; Poi, R.; Bhattacharyya, A. Status of sennosides content in various Indian herbal formulations: Method standardization by HPTLC. Bangladesh. J. Pharmacol. 2008, 3, 64–68. [Google Scholar] [CrossRef]
- Pawar, R.K.; Shivani, S.; Singh, K.C.; Rajeev, K.V.S. Physico-chemical standardization and development HPTLC method for the determination of Andrographonin in KalmghNavyasLoha. An Ayurvedic formulation. Int. J. Res. Ayurveda Pharm. 2011, 2, 295–301. [Google Scholar]
- Ram, M.; Abdin, M.Z.; Khan, M.A.; Jha, P. High-Performance Thin-Layer Chromatography (HPTLC); Springer: Berlin, Germany, 2011; p. 105. [Google Scholar]
- Mostafa, H.A.M.; el Bakry, A.A.; Alam, E.A. Evaluation of antibacterial and antioxidant activities of different plant parts of Rumex vesicarius L. (Polygonaceae). Int. J. Pharmcy Pharm. Sci. 2011, 2, 109–118. [Google Scholar]
- Kumar, R.A.; Sridevi, K.; Kumar, N.V.; Nanduri, S.; Rajagopal, S. Anticancer and immunostimulatory compounds from Andrographis paniculata. J. Ethnopharm. 2004, 92, 291–295. [Google Scholar] [CrossRef] [PubMed]
- Sheeja, K.; Kuttan, G. Activation of cytotoxic T lymphocyte responses and attenuation of tumor growth in vivo by Andrographis paniculata extract and andrographolide. Immunopharmacol Immunotoxicol. 2007, 29, 81–93. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, P.K.; Kumar, V.; Houghton, P.J. Screening of Indian medicinal plants for acetylcholinesterase inhibitory activity. Phytother. Res. 2007, 21, 1142–1145. [Google Scholar] [CrossRef] [PubMed]
- Johnson, M.A.; Gowtham, J.; Sivaraman, A.; Janakiraman, N.; Narayani, M. Antioxidant, Larvicidal, and Cytotoxic Studies on Asplenium aethiopicum (Burm. f.) Becherer. Int. Sch. Res. Not. 2014, 2014, 6. [Google Scholar] [CrossRef]
- Harborne, J.B. Phytochemical Methods; Chapman and Hall: London, UK, 1998. [Google Scholar]
- Wagner, H.; Baldt, S.; Zgainski, E.M. Plant Drug Analysis; Springer: Berlin, Germany, 1996. [Google Scholar]
- Janakiraman, N.; Johnson, M.A. HPTLC Fingerprint Profile (Phenolics) of Selected Cyathea Species from Western Ghats, South India. Chin. J. Biol. 2016, 2016, 7. [Google Scholar] [CrossRef]
- Reportofthe U.S. Food and Drug Administration, Combating Counterfeit Drugs; FDA: Washingto DC, USA. Available online: http://www.fda.gov/oc/initiatives/counterfeit/report02_04.html (accessed on 25 July 2004).
- Deisingh, A.K. Pharmaceutical counterfeiting. Analyst 2005, 130, 271. [Google Scholar] [CrossRef] [PubMed]
- Newton, P.N.; Green, M.D.; Fernandez, F.M.; Day, N.P.; White, N.J. Counterfeit anti-infective drugs. Lancet Infect. Dis. 2006, 6, 602. [Google Scholar] [CrossRef]
- Mukhopadhyay, R. The Hunt for Counterfeit Medicine. Anal. Chem. 2007, 79, 2623. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Sherma, J. Review of advances in the thin layer chromatography of pesticides: 2004–2006. J. Environ. Sci. Health B 2007, 42, 429–440. [Google Scholar] [CrossRef] [PubMed]
- Jain, A.; Lodhi, S.; Singhai, A.K. Simultaneous estimation of quercetin and rutin in Tephrosia purpurea Pers by high performance thin-layer chromatography. Asian J. Tradit. Med. 2009, 4, 104–109. [Google Scholar]
- Sushma, G.S.; Devi, B.A.; Madhulatha, C.H.; Kumar, K.U.; Harathi, P.; Subramanian, N.S.; Ramadevi, M. Preliminary phytochemical screening and HPTLC fingerprinting of leaf extracts of Ficus nervosa Heyne ex Roth. J. Chem. Pharm. Res. 2013, 5, 98–104. [Google Scholar]

| Rf | Height | Area | Assigned Substance |
|---|---|---|---|
| 0.01 | 95.4 | 1559.4 | Unknown |
| 0.07 | 23.5 | 512.3 | Unknown |
| 0.11 | 52.8 | 1046.4 | Gallic acid |
| 0.16 | 156.2 | 3850.8 | Unknown |
| 0.21 | 21.5 | 366.4 | Ferulic acid/Phloroglucinol |
| 0.29 | 112.5 | 4082.7 | Unknown |
| 0.44 | 18.6 | 589.4 | Ferulic acid/Isoquercetin |
| 0.63 | 174.0 | 6797.9 | Chlorogenic acid/Resorcinol |
| 0.65 | 174.9 | 6873.2 | Resorcinol/Quercetin |
| 0.75 | 150.6 | 9503.5 | Resorcinol/Kaempferol |
| 0.94 | 303.6 | 25,474.1 | Unknown |
| Rf | Height | Area | Assigned Substance |
|---|---|---|---|
| 0.01 | 48.6 | 292.8 | Unknown |
| 0.16 | 133.3 | 8228.3 | Unknown |
| 0.26 | 30.2 | 884.2 | Ferulic acid |
| 0.36 | 47.8 | 2781.5 | Unknown |
| 0.47 | 336.0 | 13,570.6 | Rutin |
| 0.52 | 46.2 | 2147.0 | Isoquercetin |
| 0.65 | 120.4 | 4718.0 | Chlorogenic acid |
| 0.75 | 19.4 | 636.0 | Kaempferol |
| 0.83 | 52.6 | 1867.8 | Unknown |
| 0.96 | 116.7 | 3936.7 | Quercetin |
| Rf | Height | Area | Assigned Substance |
|---|---|---|---|
| 0.10 | 156.4 | 3887.9 | Unknown |
| 0.14 | 31.6 | 547.4 | Nicotine |
| 0.20 | 43.6 | 1297.2 | Colchicine |
| 0.27 | 69.3 | 2111.7 | Unknown |
| 0.32 | 100.4 | 2653.3 | Strychnine |
| 0.35 | 117.4 | 4875.9 | Colchicine |
| 0.40 | 90.7 | 3625.4 | Chelidonine |
| 0.49 | 60.3 | 2187.1 | Unknown |
| 0.53 | 63.3 | 1921.2 | Unknown |
| 0.65 | 10.5 | 171.0 | Unknown |
| 0.93 | 288.2 | 16,769.8 | Unknown |
| Rf | Height | Area | Assigned Substance |
|---|---|---|---|
| 0.01 | 124.3 | 662.4 | Tannin 1 |
| 0.04 | 70.8 | 975.2 | Tannin 2 |
| 0.11 | 92.6 | 1718.7 | Tannin 3 |
| 0.39 | 26.6 | 555.7 | Gallic acid |
| 0.40 | 24.1 | 362.5 | Caffeic Acid |
| 0.55 | 33.6 | 1414.3 | Unknown |
| 0.61 | 44.6 | 1128.6 | Unknown |
| 0.66 | 94.3 | 1294.0 | Unknown |
| 0.70 | 70.0 | 1832.5 | Unknown |
| 0.83 | 119.9 | 7703.6 | Unknown |
| 0.95 | 216.8 | 14,627.5 | Unknown |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Johnson, M.a.A.; Gowtham, J.; Janakiraman, N.; Renisheya Joy Jeba Malar, T.; Rocha, J.E.; Coutinho, H.D.M. Phytochemical Profile of Asplenium aethiopicum (Burm. f.) Becherer Using HPTLC. Separations 2020, 7, 8. https://doi.org/10.3390/separations7010008
Johnson MaA, Gowtham J, Janakiraman N, Renisheya Joy Jeba Malar T, Rocha JE, Coutinho HDM. Phytochemical Profile of Asplenium aethiopicum (Burm. f.) Becherer Using HPTLC. Separations. 2020; 7(1):8. https://doi.org/10.3390/separations7010008
Chicago/Turabian StyleJohnson, Marimuthu alias Antonysamy, Janarthanan Gowtham, Narayanan Janakiraman, Tharmaraj Renisheya Joy Jeba Malar, Janaina E. Rocha, and Henrique D. M. Coutinho. 2020. "Phytochemical Profile of Asplenium aethiopicum (Burm. f.) Becherer Using HPTLC" Separations 7, no. 1: 8. https://doi.org/10.3390/separations7010008
APA StyleJohnson, M. a. A., Gowtham, J., Janakiraman, N., Renisheya Joy Jeba Malar, T., Rocha, J. E., & Coutinho, H. D. M. (2020). Phytochemical Profile of Asplenium aethiopicum (Burm. f.) Becherer Using HPTLC. Separations, 7(1), 8. https://doi.org/10.3390/separations7010008





