Abstract
Access to and the use of irrigation water of adequate quality and targeted nutrient supplementation have become more widespread in recent years. Both crop quality and the irrigation system lifetime are affected by the quality of the water used for irrigation. Micro-irrigation (e.g., drip and sprinkle) is becoming increasingly common alongside the more typical irrigation methods, but it requires expertise and pre-treatment to ensure a proper water supply. The most significant problem is clogging, which can greatly reduce irrigation efficiency. Treatment for irrigation purposes mainly depends on the contaminants that are present in the water. The main treatment options available are biological, electromagnetic and electrostatic treatments, but these have a wide range of effectiveness levels compared to membrane separation technologies. In addition, adsorption treatments are also available, which, depending on the adsorbent used, can greatly improve the pre-treatment of irrigation water. This work provides an overview of adsorbents suitable for the treatment of irrigation water and their effectiveness. The separation of interfering components via adsorption is effective and promising for future application as the expected irrigation demands increase.
1. Introduction
1.1. The Role of Climate Change in the Necessity of Irrigation
One of the regional consequences of climate change is that weather extremes are becoming more frequent [1], so our water management needs to be reconsidered. The main problem in itself is not that the annual amount of precipitation decreases, but that the precipitation does not arrive evenly [2] and summer drought periods become more frequent [3]. Climate change and the consequent increasing dryness, forest fires, the lowering of the ground water level and the beginning of soil erosion all contribute to the process of desertification, but many factors directly related to humans are also to blame [4]. Industry and agriculture are primarily responsible for water consumption. The latter accounts for about half of the total water consumption [5]. To the best of our ability, we must do everything we can to prevent and reduce the damage caused by drought, so that we can supply water at the right time and in the right amount. Water supply plays a key role in the conditions for competitive agriculture, and the efficient use of irrigation water of adequate quality can be a key element of this supply.
1.2. The Role of Irrigation Systems
One of the biggest challenges of the 21st century is determining how to provide enough clean water for everyone (agriculture, industry and the entire living world) and not just the population [6]. Irrigation is the artificial application of water to the surface of the soil and then to deeper layers, using a variety of specialized equipment (e.g., pipes, pumps and sprinkler, dripping systems). Irregular rainfall distribution and drought make irrigation necessary in certain areas. A wide range of irrigation systems are available to distribute water evenly over the surface to be irrigated. The water used for irrigation may originate from ground water, spring water, well water, surface water, rivers, lakes, reservoirs, or even treated wastewater or desalinated water [7]. The use of irrigation and targeted nutrient supply in quality and intensive vegetable and fruit cultivation has been spreading more and more in recent years [8]. The quality of the crops, their aesthetic appearance and the life of the irrigation system are both influenced by the quality of the water used for irrigation [9]. There are many options available for irrigation water treatment. Biological treatment methods could be promising for organic and heavy metal compounds; however, they require precise operation and control. For salt removal, many studies have reported electromagnetic and electrostatic treatment as fast and quite easier treatment methods; however, the efficiency of these techniques could be lower since they are a pressure-driven membrane separation process. The use of adsorbents can be a good way to remove certain undesirable components of irrigation water. Two large groups of adsorbents are known: conventional and non-conventional adsorbents, which can achieve different efficiencies in water purification. Conventional adsorbents generally have a higher adsorption capacity compared to non-traditional adsorbents, which are readily available and therefore inexpensive, but have a lower adsorption capacity.
2. Overview of the Requirements for Irrigation Water
2.1. Legal Requirements
Nowadays, it is no longer enough to provide the physical conditions for irrigation; there are also an increasing number of legal requirements to be met. As environmental requirements are becoming more stringent due to the increasing value of clean water, no water-based activities can escape the scrutiny of the legal regulatory regime. In the following, the most important aspects and principles will be summarized; these may influence professional conclusions.
The regulatory background for irrigation covers a wide range of legislation, depending on the source, quality and agricultural area of the irrigation water.
In Hungary, the concept of water management and the related responsibilities serves to comply with the relevant EU and national legislation, i.e., it includes compliance with the requirements of Directive 91/271/EEC [10] on urban wastewater treatment, Directive 98/83/EC [11] on the quality of water intended for human consumption, the Water Framework Directive, Act LIII of 1995 [12] on the protection of the environment, Act LIII of 1996 [13] on the protection of nature, Act CLXXXV of 2012 [14] on waste, and Directive 2007/60/EC [15] on water damage.
The European Union Directive 2000/60/EC [16] is the most important and community-wide guideline for the protection of the good quality of the water bodies covered by the community.
The EU’s Water Framework Directive (WFD) aims to achieve and maintain the good status of surface, coastal, transitional and groundwater, thereby contributing to the protection of ecosystems. An important objective of the Directive is to prevent the pollution of waters, and it sets limits on anthropogenic emissions. The measures taken to achieve and maintain the objectives set out in the WFD are summarized in the river basin management plan at a national level.
Decree 90/2008 (18 July 2008) [17] of the Ministry of Agriculture and Rural Development stipulates that a soil protection plan must be drawn up for, inter alia, irrigation for agricultural purposes, on the basis of which the justification for the measure must be presented and a proposal for protection against erosion must be made after soil sampling in an accredited laboratory.
The suitability of irrigation water should also be assessed in terms of abstraction. The requirements for the suitability of irrigation water are set out in the standard MSZ-10-640:1989 [18].
In response to the challenges of climate change, to improve the competitiveness and adaptability of agriculture, to promote the wider use of irrigated agriculture and to establish irrigation communities, the National Assembly has enacted Act CXIII of 2019 [19] on Irrigation Agriculture. According to the law, “In order to carry out irrigation farming, the holder of the water right of the water facility is entitled to an irrigation easement, under which the owner or user of the property served must tolerate the establishment and operation of a linear water facility for irrigation on his property by the farmer carrying out irrigation farming, to carry out the necessary water works for the purpose of irrigation for the continuation of his agricultural activity, and to transfer the irrigation equipment, provided that it does not preclude the proper use of the property”.
It can be seen from the above that the Government is seeking to promote the competitiveness of agriculture and the protection of land and water through the appropriate use and quality of the land, in accordance with the objectives of the European Union. More modern, but more expensive irrigation technologies, can achieve the more sustainable use of water. These aspects will become increasingly important in the future.
2.2. What Water Sources Can We Work with?
The most common sources of water available for cultivation are surface water (water from lakes, canals, possibly rivers) and underground water [20]. For subsurface waters, we must separate groundwater and aquifers from a quality point of view. Groundwater is the water of the first water-tight layer, and water bodies shallower than 20 m (dug and drilled wells). Aquifers are located in layers deeper than 20 m, depending on the geological and hydrological conditions. These waters may only be extracted with pre-planned and authorized wells. The professional basis for this is to protect it from possible pollution from the surface, since it is our most valuable, cleanest, long-term water reserve [21].
From the perspective of producers and water users, the quality assessment of the three types of water is quite different. The differences are to be found in the constancy of the composition and the effects of environmental pollutants. Surface waters are constantly exposed to quality-affecting influences: inflows, water withdrawals, precipitation, washing in, dilution, and concentration, which constantly modify the composition. When using surface water for horticulture, constant caution is a good idea; at the very least, we should measure the salinity (EC) on a weekly basis, which draws our attention to major changes. Regarding groundwater wells, we can say that their waters can only be recommended for horticultural crops with greater compromises, because they can contain not only a significant amount of nutrients, but also harmful substances accumulated to a toxic level [22].
2.3. Quality Requirements of Irrigation Water
The quality of irrigation water varies from country to country. For irrigation, natural water quality would be the most suitable; however, we cannot usually ensure rainwater quality from the available water sources. Therefore, in addition to the positive effects, irrigation can also have negative effects (Table 1), which are partly due to the water quality [23].

Table 1.
The positive and negative effects of irrigation [23].
The quality of irrigation water refers to the range of properties of the water to be used that affect the soil, the life processes of the plant and the technology of water distribution. Therefore, when determining the quality indicators of irrigation water, it is advisable to examine the triple interaction of water–soil–plant [7,24]. Methods for ensuring water quality have developed continuously and basically set certain limit values based on two aspects, namely the effects on the soil–plant combination and the effects on the operation of irrigation equipment. The first group basically includes chemical parameters, and the second group includes physical, chemical and biological parameters [25].
Among the chemical requirements, the most important characteristics are as follows:
- salinity,
- indicators expressing the effect of hydrocarbons and carbonates,
- the amount of sodium ions compared to calcium–magnesium ions,
- the relative ratio of magnesium,
- electrical conductivity,
- chloride, iron and manganese content [26].
2.3.1. pH
Changes in the pH of the water during cultivation will affect the quality of the plants. A low pH inhibits the absorption of vital nutrients such as calcium, potassium, magnesium and molybdenum [27]. At the same time, a low pH increases toxicity because some trace elements are absorbed too easily [28]. At too high a pH, other substances such as phosphates and other important trace elements become unavailable to the plant [29].
2.3.2. Salinity
The sodium adsorption ratio (SAR) is the most important characteristic. It means a value representing the relative amount of sodium ions to the combined amount of calcium and magnesium ions in water using the following formula: SAR = [Na]/(([Ca] + [Mg])/2)1/2, where all concentrations are expressed as milliequivalents of charge per liter [30]. Salinity is generally dangerous for the soil, but it can also be harmful to plants. Salinity is expressed by the amount of salt dissolved in a volume unit (g/L) or by the specific electrical conductivity (EC) of the solution. When establishing the limit values, the primary consideration is that the irrigation water should not cause a level of salt accumulation in the soil that could be harmful. Therefore, it is not possible to establish a general rule regarding this, because it depends on many properties (e.g., water permeability, rainfall, irrigation method, salt tolerance of the plant [31]. It is generally accepted that if the salt content of the irrigation water is less than 500 mg/L (EC < 0.78 mS/cm), then the salt pool of the irrigated soil usually does not increase significantly [32].
As a result of certain environmental changes (e.g., concentration, dilution, increase in pH), some of the Ca and Mg ions precipitate out of waters with a high carbonate–hydrocarbonate content [33], thus increasing the proportion of Na ions in the solution, and thereby making the water salinizing [31].
2.3.3. The Amount of Sodium Ions Compared to Calcium-Magnesium Ions
Various parameters have been proposed to estimate the amount of carbonate and hydrocarbonate ions and their expected chemical interactions, e.g., phenolphthalein alkalinity, soda equivalent, saturation index, effective Ca and Mg concentration, relative hydrocarbonate and carbonate ratio [34,35]. The principle of certification in this area is that high-quality irrigation water must not contain free soda at all and must not show phenolphthalein alkalinity [36].
In terms of the cation composition of the irrigation water, a low sodium level is favorable [37]. However, in terms of the salinizing effect of water, the most important thing is not the absolute amount of Na-ions, but their proportion compared to other cations [38]. Sodium is a natural component and enters the water through the dissolution of minerals, but it can also be of municipal origin. It is the biggest problem in soil cultivation, where it has a harmful soil-destroying and salinizing effect; this is because it replaces other cations (e.g., calcium ion) on soil colloids and thus significantly changes the structure, functioning, nutrient and water management characteristics of the soil [39].
2.3.4. Manganese Content
A significant amount of magnesium ions from irrigation water with a high magnesium content can bind to soil colloids, which adversely affects the physical and water management properties of the soil. Therefore, it is advisable to take the Mg/(Ca + Mg) ratio or its percentage into account when classifying water [34,40].
3. Physical Structure of Irrigation Systems and the Arising Problems
For the irrigation of different crops, there are different watering options, ranging from methods that have been used for decades to modern precision options that are more efficient. Agriculture is the largest water-consuming sector, and therefore the focus of attention when water management is an important point of discussion. This sector has recently been confronted with trends in water supply restrictions, making the efficient use of optimized water resources through the use of multiple irrigation systems essential. Compared to conventional irrigation methods, drip irrigation systems provide frequent and efficient water supply and also control the amount of water used. Therefore, the impact of different irrigation systems should be studied to find the best option for each case [41].
3.1. Surface Irrigation Systems
The most common irrigation method is surface irrigation, which is mainly used for crops covering large areas [42]. This type of irrigation works via gravity, using pre-dug trenches without the need for a pump. The most commonly used surface irrigation systems are border and furrow irrigations. The latter is more commonly used for the water supply of row crops and orchards; however, it is more demanding and, due to the technology, results in more losses and lower efficiency in water use. This method has different design, management factors and elements that have been thoroughly analyzed [43]. Furrow irrigation is the oldest and most widely used irrigation system. Pressure irrigation methods remain less popular due to their high energy costs and the use of automation [44]. Some researchers have made comparisons between different irrigation systems in the Jaizan Plain based on soil and land characteristics. The results showed that drip and sprinkler irrigation methods are more efficient and effective than conventional surface techniques, where drainage is the most limiting factor. The research concludes that due to the rapid decrease in and shortage of water resources, current gravity irrigation methods need to be replaced by modern (drip and sprinkler) technologies as soon as possible [45].
3.1.1. Sprinkler Irrigation
The method of using sprinkler irrigation is similar to natural rainfall. Water is delivered through a pressurized system to the pipes that release it. Eventually, sprinklers will spray it into the air, where droplets are fragmented, and fall to the ground. The pump-fed system should be designed so that each sprinkler distributes an equal amount of water [46].
The micro sprinkler technology operates at low pressure and can be used in areas in which drip irrigation is not recommended or is not efficient enough to keep the plant foliage constantly moist, or in which overhead irrigation would be required. Micro sprinklers are categorized according to several aspects, such as flow rate, wetted diameter and the method of spraying, such as producing a mist or a spray [47].
Sprinkler irrigation is preferred over subsurface drip irrigation for cotton production, but this technique is not widespread due to climatic conditions and salt accumulation [48]. Some studies compare drip and sprinkler irrigation and show that for some crops, such as grapes, sprinkler irrigation results in a greater root surface area as you move away from the trunk [49]. The study in the Jaizan Plain also showed that drip and sprinkler irrigation is more effective than conventional surface irrigation. The major limiting factor was found to be the gravel soil structure, drainage and calcium carbonate when using sprinkler irrigation [45].
3.1.2. Drip Irrigation
Drip irrigation (also known as trickle irrigation) is a very slow dripping technique (2–20 L per hour) using a small-diameter plastic system of tiny emitters placed on the outside of the pipe, which are called drippers. The water is discharged close to the plant so only the soil near the root is wetted, instead of the whole soil profile, like in the case of surface irrigation and sprinkler irrigation. With drip irrigation, treatment is much more frequent (usually every 1–3 days) than other techniques; therefore, it provides a more favorable moisture level for the plants [50]. Under-foil drip irrigation is a complex water-efficient irrigation method that can directly improve the soil water–salt conditions in the root zone of plants and affects physiological characteristics such as plant emergence, root spread and above-ground growth, therefore improving plant growth, development and yield. Numerous studies have been carried out on the characteristics of soil water and salt movement during under-film drip irrigation [51].
Some of the most convincing reasons for using drip systems are cost efficiency, easy set-up and operation, as well as low maintenance costs [52]. The drip method provides micro irrigation, which helps to reduce water use and avoid unnecessary nutrient excess. Its operation is based on direct irrigation for roots, which provides a constant flow with equal distribution while minimizing evaporation. After proper pretreatment, water is delivered through the system’s network of pipes to drippers placed close to the plants, which can be either holes in the pipes or units placed on top of them to deliver the water directly to the roots through capillary tubes. The accuracy of the system may depend on its design goals and its maintenance and operation technology [53]. The drip irrigation system will also include a slow-release device to control and optimize the flow of water to the soil. There are two different types of drip irrigation, which are subsurface and surface methods. The main difference is that the former is applied to the soil, while the latter near the roots. It is important to note that a maximum of two emitters per plant is sufficient for proper irrigation [53].
3.2. General Structure of Irrigation Systems
Traditionally used irrigation (border, basin, furrow) only requires a sufficient water surface and sometimes a pump for periodic irrigation. In contrast, precision irrigation types require the construction of a complete irrigation system. When using sprinklers or drip irrigation, the system design is almost the same, and only the method of water discharge varies.
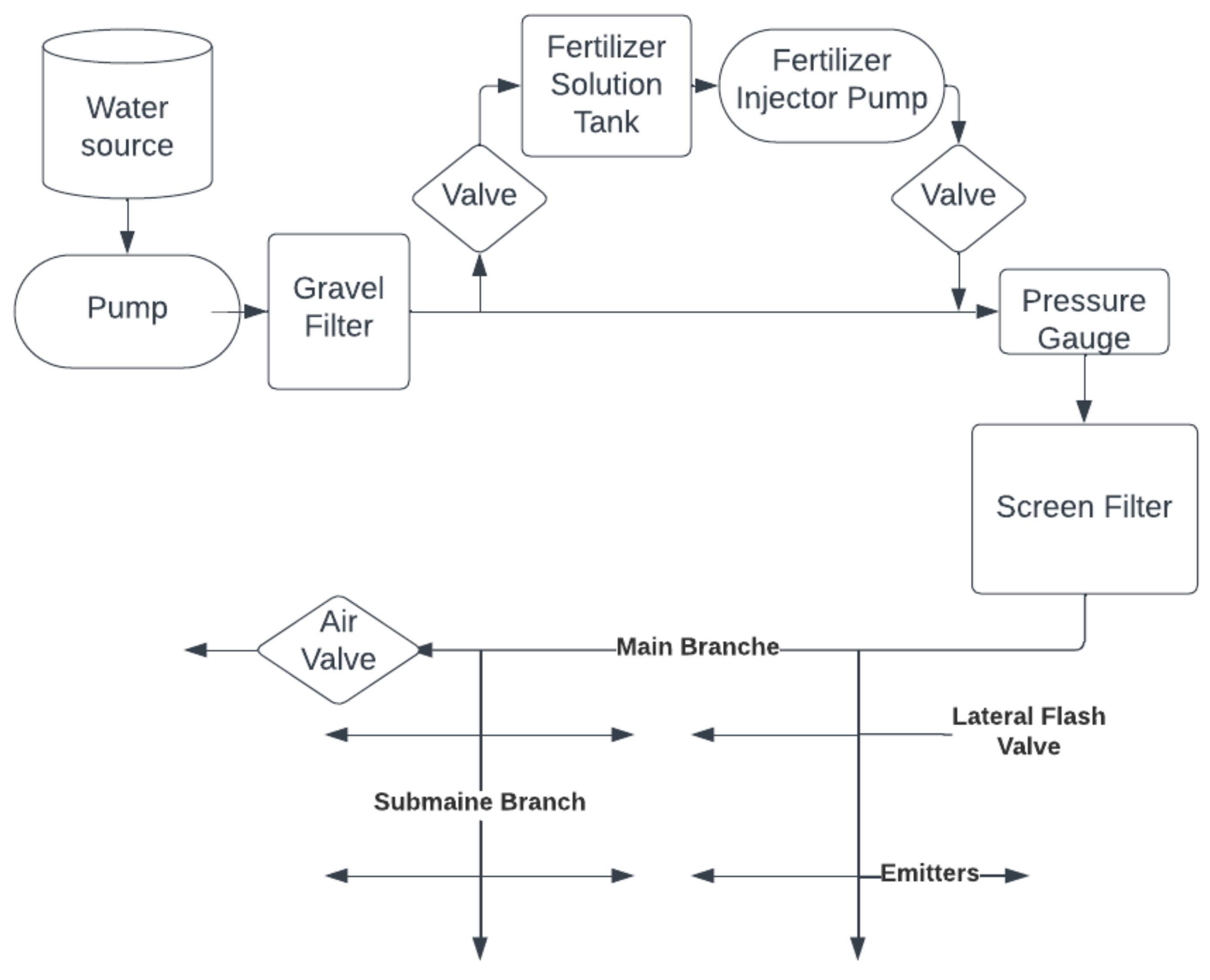
The main components used in drip irrigation include a pressure regulation valve, a filter, a system controller, injectors, gauges, flow pipes and emitters [54]. Eventually, the water is delivered to the plants via polyethylene pipes, which have smaller holes at certain distances. In the installation, it is important that the distances are properly measured so that the pressure and water flow are nearly the same even at the farthest parts of the pipes [55].
From the relevant reservoir, a pump delivers the water through filters (mostly gravel) to the pressure regulator and then to the screen filter [55]. The water is then fed to the main branch, from where it is sent to several sub-branches (Figure 1). Fertilizers can be applied through this type of system. This can result in a reduction in the fertilizer amount and costs [56].
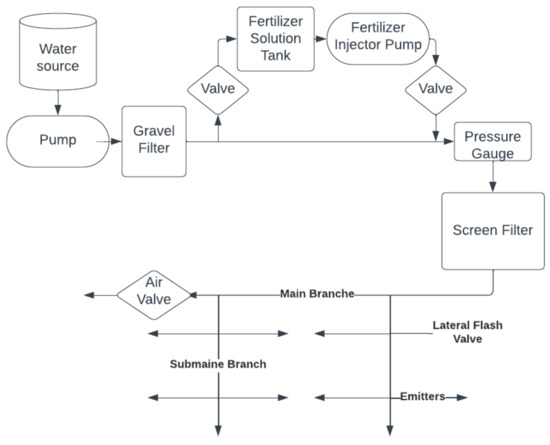
Figure 1.
The general structure of drip irrigation systems.
3.3. Clogging
Emitter clogging is the biggest limiting factor affecting the use of drip irrigation. The use of poor-quality water in micro irrigation systems greatly increases the potential for clogging, reducing the possible areas of use and increasing maintenance costs. Studying the main elements of clogging and their effects and mechanisms of accumulation have become indispensable [57]. Fluid flow in narrow pipes and emitters can easily become clogged by solid particles, chemical constituents and microorganisms during irrigation [46].
3.3.1. Physical Clogging
Physical clogging is mainly caused by various organic and inorganic materials suspended in the water [58]. These particles of clay, sand, plastic and other residues cannot be filtered out and can therefore cause the clogging of the emitter. Physical clogging caused by solid particles accounts for nearly 31% of the clogging potential, making it one of the most common causes of clogging [46].
3.3.2. Chemical Clogging
Chemical clogging is caused by substances dissolved in water, such as carbonates, phosphates, Fe2+, Ca2+ andMg2+, which under certain conditions precipitate on the inner wall of the pipe. This deposition is greatly influenced by temperature, pressure, pH and the concentration of ions [59]. In addition, the nutrients added to the water also influence clogging; for example, phosphorus added to the water greatly increases the precipitation of carbonate, quartz and silicate [60]. Fe2+ can cause serious problems in the irrigation system if its concentration exceeds 1.5 g m−3 [46], and a water hardness above 300 mg L−1 also can threaten the irrigation system [61].
3.3.3. Biological Clogging
The drip system provides a suitable habitat for algae, bacteria and other organic organisms that can form slime. This material, in combination with other contaminants, can cause biofouling [62]. However, in underground drip systems, the effect of roots can also be considered as biological clogging [63].
4. Overview of Current Irrigation Water Treatment Methods
Nowadays, irrigation water treatment methods, in which natural water sources are extensively used, are becoming more focused and based on water pollution and hectic rainfalls. The most often reported problem is the high salinity of this water, which could negatively effect agriculture production even if it is not reduced to a level that is acceptable for the different plants. The effectiveness of treatment methods is influenced by the background quality of the irrigation water and includes physical, chemical, and biological parameters. Most of those factors could impact which water treatment is efficient to achieve the required water quality needs.
4.1. Overview of Contaminants
Organic contaminants are not desirable in water for irrigation based on infection risk, clogging and fouling issues. Organic matter is an organic substance that is broken down from plants, animals, microorganisms and from geological origins, and that can be found in water sources, especially in surface water in many cases [64]. These organic substances could increase the turbidity of the water supply and could affect disinfection methods, especially for UV sources. Different types of microbes may require different treatment methods, disinfection concentrations and contact times, with longer disinfection times possibly also helping to avoid infection. The pH of the irrigation water also affects the efficiency of certain treatments. For example, ozone treatments require different dosages based on pH, and chlorination is more effective in water with a lower pH [64,65].
Excess calcium and magnesium in a water source contribute to the hardness of water and can lead to the scaling of irrigation lines. This build up can decrease the effectiveness of water treatment processes such as UV, making it important to minimize scale deposits in the irrigation system [64,66].
4.2. Biological Treatment
Biological treatment is an effective method via which to reduce the organic content of irrigation water; however, one of its main challenges is keeping the microorganism alive in the treatment system and providing an equal water quality without harming the food safety or yield and the fruit or derivatives’ quality. A membrane bioreactor was tested by treating polluted surface water. The results showed that up to 99.99% of the inlet Escherichia coli and 98.52% of the total coliforms were removable, as well as some harmful compounds and heavy metals in particular, including 82% of arsenic, 82% of cadmium, 97% of chromium, 93% of copper and 99% of lead; however, boron and manganese were not removed from the permeate. The sole gravel filter and heavy metal removal device (HMR) system were not able to remove the pathogen microorganisms. However, connecting the UV treatment could solve this issue [67]. In another study, a semi-closed horizontal tubular photobioreactor (PBR) at the pilot scale was tested to remove a total of 35 target compounds, including benzotriazoles, benzophenones, antibiotics and different pharmaceuticals present in irrigation water in a peri-urban rural area. This water running through an open channel and was a mixture of reclaimed wastewater from a nearby wastewater treatment plant (WWTP) and run-off from the different agricultural fields in the area. It has been reported that the compounds studied are usually not fully eliminated during conventional wastewater treatment, which justifies the need to investigate alternative treatment strategies. A total of 21 of these compounds were detected in the irrigation water. Benzotriazoles were only partially removed after the microalgae treatment, with an elimination efficiency of conventional WWTPs. The UV filter benzophenone-3 (BP3) showed variable removals, ranging from no elimination to 51%, whereas 4-methylbenzilidenecamphor (4MBC) was completely eliminated. Regarding pharmaceuticals, the average removals were higher and in the range of 60–100%; this is with the exception of the antibiotics sulfamethoxazole (46%) and sulphapyridine, which were not removed [68]. Removing target compounds from irrigation water has a key role in protecting plants and products from unwanted changes and from accumulating these compounds in the agricultural products.
4.3. Desalination Processes
Saline groundwater irrigation is a crucial method by which to reduce the shortage of freshwater resources. However, saline water could have impact on the yield of plants and on the soil.
4.3.1. Ultrasound Treatment
Ultrasound (US) treatment is one of the most promising treatment methods and has been evaluated in long-term studies with various plant species, including some highly salinity-sensitive plants, under greenhouse conditions. The ultra sonication system provided encouraging results when used to irrigate a number of vegetable species with high-electrical-conductivity irrigation water. The results from the long-term use of ultrasound treatment increased the yield of lettuce and celery plants [69].
4.3.2. Magnetic Treatment
Another low-cost and promising operating method is applying permanent magnets for irrigation water treatment [70]. Surendran and coworkers evaluated the effects of magnetic treatment on irrigation water and the growth and yield parameters of cow pea using pot and field experiments. During their experiment, normal water and hard water at 150 and 300 ppm, respectively, and saline water at 500, 1000 and 2000 ppm of both the control and magnetic-treated solutions were used, respectively. Permanent magnets with the capacity of 1800–2000 G were applied. Their investigation indicated that the magnetic treatment of irrigation water types achieved an improvement in the crop growth and yield parameters of cow pea as well. Magnetic treatments have the potential to decrease the electrical conductivity, total dissolved solids and salinity levels of all solutions. The increase in pH was observed in all the treatments. Magnetized irrigation water increased the soil moisture compared with the control sample [71].
Furthermore, of permanent magnet electromagnetic treatment was also investigated during the irrigation of corn and potato plants. The results showed a significant increase in the germination rate of corn seedlings watered with electromagnetic-treated saline water, particularly when the water was exposed to electromagnetic fields for 15 min. A significant reduction in the soil salinity (ECe) and Na+ and Cl− contents of the soils irrigated with electromagnetic-treated saline water was detected compared to the soils irrigated with non-treated saline water. The electromagnetic treatment of saline water caused significantly higher K, N and P adsorption values in all the potato samples [72]. Wiedenfeld studied the sugarcane yield and juice quality by using an electrostatic precipitator. The study was conducted to compare the effect of water irrigation with 1.3 dS m−1 vs. 3.4 dS m−1 on sugarcane and to evaluate whether the electrostatic conditioning treatment of the water influenced the salt effects. The results showed that the cane and sugar yields were reduced approximately 17% by the 3.4 dS m−1 water compared to the 1.3 dS m−1 water, but the juice quality parameters were not affected by the soil salinity levels [73].
4.3.3. Ionization
The ability of ionizing brackish water to prevent salinity damage to cotton plant growth with an improving irrigation water productivity was investigated by Wei and coworkers, who applied deep irrigation treatments. As the results indicated, the ionization effectively reduced the mass of salt from the late seedling stage to harvest by 12.8–65%, especially for the treatment using 562.5 mm of ionized brackish water. The maximum amount of cotton was obtained in the treatment using 487.5 mm of ionized brackish water, which increased to an average of 19% compared to 487.5 mm in the non-ionized brackish water treatment experiment. The water use efficiency improved under the ionized brackish water treatments [74].
4.3.4. Membrane Separation
The membrane separation process is well known in many water and wastewater treatment applications; however, its operation and maintenance costs could decrease its popularity for use in irrigation water treatment. In special cases, geothermal water treatment is a possible economically worthwhile treatment method. In many areas of Hungary and Europe, geothermal water is used for heating purposes. However, the used/spent geothermal water rarely gets injected back into the ground. Some amount of used geothermal water ends up in rivers or lakes and causes increasing salinity levels in surface water bodies; this could have an effect on the natural vegetation and fauna. Jarma and co-workers investigated the use of treated geothermal water for the irrigation of tomato plants. Membranes (NF8040-70 as NF membrane and TM720D-400 as RO membrane) were selected for this experiment. According to the results, almost all of the minerals that are needed for plant growth were rejected by the tested membranes. Therefore, for remineralization purposes, mixed water was also tested. In the case of the NF membrane, the permeate was mixed 1:1 with non-treated geothermal water; this ratio was 2:3 in the case of the RO permeate and geothermal water, according to the higher level of salt rejected by the RO membrane. The costs of the product water were found to be 0.76 and 1.56 $/m3 for the NF and RO processes, respectively [75]. The management of saline water through desalination or blending with low-salinity canal water may be a viable strategy for potato irrigation in semi-arid regions that have saline groundwater and scarce canal water. A 4-year study was conducted to investigate the effects of four water qualities [canal water (CW), desalinated water (DSW), saline groundwater (GW) and mixed water (MW) (CW + GW)], three N–P–K fertigation levels [F100 = 100% NPK (190, 28.4 and 53.9 kg ha−1 of N, P and K, respectively), F80 = 80% NPK and F60 = 60% NPK] and their interaction on the performance of two potato varieties grown in a sandy loam alluvial soil of a semi-arid region. The electrical conductivity (EC) of CW, DSW, GW and MW ranged from 0.3 to 0.4, 0.2–0.3, 3.2–4.2 and 1.8–2.2 dS m−1, respectively. Drip irrigation with GW resulted in a 21–44% reduction in the total tuber yield compared with CW. Irrigation with MW increased the total tuber yield by 9–37% compared with GW. Irrigation with DSW effected a 41% and 14% increase in the total tuber yield compared with GW and MW, respectively. The plant growth and yield parameters reached their highest amount at F100, which reduced dramatically with a decrease in the fertigation level for all water qualities. However, these differences were slight between GW and MW, and more moderate between CW and DSW. That is the reason why, in drip-fertigated potatoes using saline or mixed water in a sandy loam soil, it is suggested that 80% NPK is used to avoid a reduction in the tuber yield and to reduce the expense of cultivation and environmental pollution [76].
4.4. Effects of Salinity
Saline water could have an effect on soil nitrogen transport, organic and inorganic content of the soil, and yield of the products. Therefore, when irrigating with saline water, all of its effects need to be considered. In order to utilize underground saltwater resources, and furthermore reduce the amount of irrigation water and nitrogen fertilizer used, Wang and coworkers carried out a field experiment in Xinjiang. The study was conducted to explore the effects of the irrigation water’s quantity, salinity, and nitrogen application rate on nitrogen fertilizer absorption and cotton yield. Three experimental factors and four levels were investigated in the experiment. Irrigation amounts of 0.7, 1.0, 1.3 and 1.6 Etc, irrigation water salinity levels of 10.9, 7.8, 4.7, 1.5 dS m−1 and nitrogen applications of 100, 200, 300 and 400 kg N ha−1 were set in the experiment. The results indicated that the salt content of the irrigation water had no effect on the soil nitrogen transport, and the soil nitrogen content only increased when more nitrogen fertilizer was used [77]. The experiment was performed by using fresh water and saline water at the seedling, jointing, heading, and filling stages. The salinity of the soil, growth, and amount of summer maize were measured by Sang and coworkers. The results showed that alternating fresh and saline water irrigation increased the salinity of the soil. The maximum quantum yield, effective quantum yield of photochemical energy conversion, photochemical quenching, and non-photochemical quenching varied greatly at the jointing stage and heading stage. Furthermore, the yield of maize that was irrigated with fresh water at the heading stage (8.53 t ha−1) was greater than that at the jointing (7.69 t ha−1) and filling stages (7.45 t ha−1). Therefore, these findings indicate that in areas in which fresh water is scarce, priority should be given to the application of fresh water at the heading stages for summer maize irrigation [78]. Saline water irrigation’s effect on the soil organic carbon (SOC) content was smaller compared to its effect on the soil inorganic carbon (SIC) content. Dong et al. irrigated soil with 1 g L−1 and 8 g L−1 of saline water in a winter wheat and summer maize rotation system. After 14 years of irrigation, the soils were analyzed in terms of their soil water, soil salt, SOC, and SIC contents. The results showed that, compared with 1 g L−1 of water irrigation, 8 g L−1 of saline water irrigation significantly increased the soil water and salt contents. Moreover, 8 g L−1 of saline water irrigation significantly decreased the SOC and SIC contents in the 0–20 cm soil layer (p < 0.05) and mainly decreased the SOC content in the >1 mm aggregates and the wheat-derived SOC content in the bulk soil. In comparison, 4 g L−1 of saline water had no significant effect on the soil water, soil salt, SOC, and SIC contents. These results indicated that a high concentration of saline water irrigation is harmful for soil carbon sequestration, while a low concentration of saline water does not affect soil carbon sequestration. Thus, using no more than 4 g L−1 of saline water irrigation for 14 years can maintain soil carbon storage in areas experiencing a water shortage [79].
Overall, several treatment methods are available and have been proven to reduce natural or anthropogenic organic and inorganic contaminants, microorganisms, and salt to achieve the safe irrigation of water; these do not reduce the yield of the products and do not change the soil and groundwater characteristics. However, some of the above-mentioned target compounds and contaminants could have long-term effects on the environment, but these effects are not yet well known.
4.5. Adsorbents in Irrigation Water Treatment
The use of wastewater for irrigation purposes is becoming widespread, but this raises new problems, such as the presence of heavy metals [80]. Although the heavy metal content in wastewater is low, it can accumulate in the soil and reach crops [81]. Adsorption is a suitable treatment technology for removing metal (for example Cd and Pb), or for reducing salinity, depending on the material used [82]. The use of adsorbents can be implemented using a flow-through system (similar to a sand or gravel filter) or by mixing them in a tank for a suitable period of time. Combinations of certain adsorbents are able to remove chemical oxygen demand and the total dissolved solids, and also reduce the turbidity very efficiently [83]. Adsorbents for the pre-treatment of irrigation water and their effects are discussed in more detail below.
5. Overview of the Most Important, Easily Available Adsorbents Based on Their Effectiveness
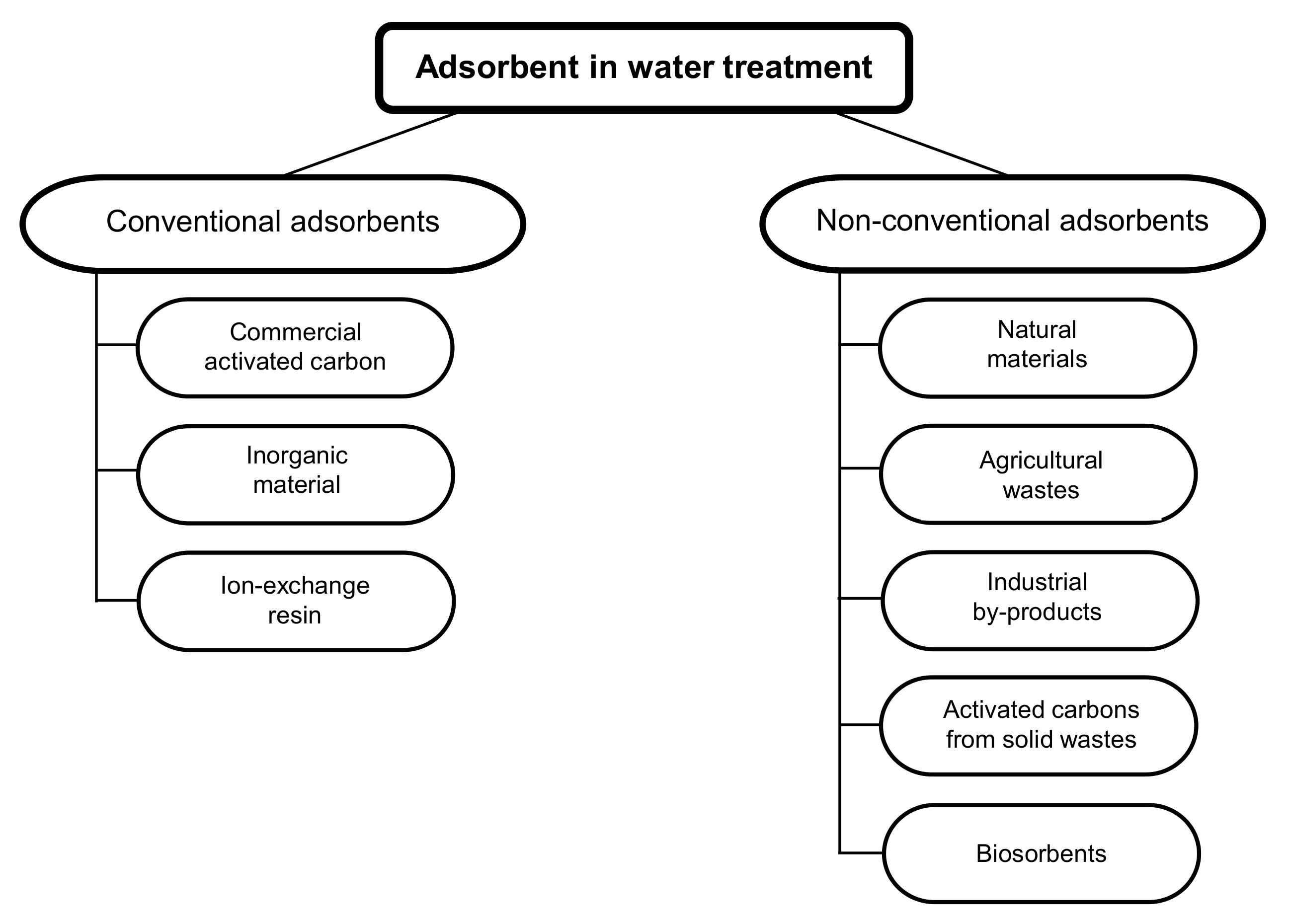
In water treatment, adsorption is an effective process for the removal of contaminants, in which ions and molecules are removed from the liquid phase via adsorption to the solid phase [84,85]. The adsorbent material can be a mineral, organic or biological material [86]. The selection of a suitable adsorbent for the removal of pollutants in water depends on the concentration and type of the pollutant, the adsorbent’s adsorbing capacity, and its efficiency [85]. The adsorption capacity of adsorbents is influenced by temperature, pH, the adsorbent dose, adsorbent particle size, agitation speed, and contact time [87]. A direct comparison of the results obtained using different adsorbents is therefore not possible since the experimental conditions are not the same during the different tests. Adsorbents can be divided into two major categories: conventional adsorbents and non-conventional adsorbents (Figure 2, Table 2) [86].
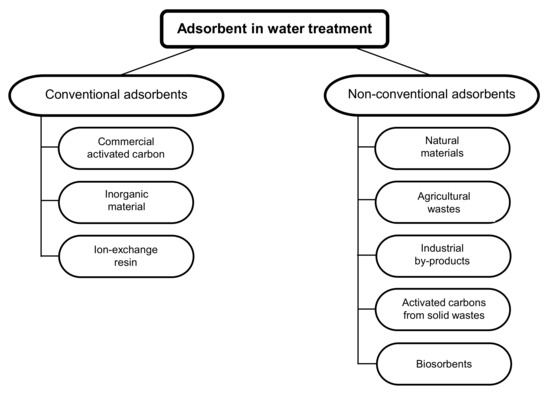
Figure 2.
Different types of adsorbents for the removal of pollutants from water [86].

Table 2.
Some examples of the most used adsorbents in water treatment, conditions and their efficiency.
5.1. Conventional Adsorbents
5.1.1. Activated Carbon
One of the most common adsorbents currently in the field of water treatment is activated carbon [85,98]. Carbon-based materials such as activated carbon (AC) can be used well as adsorbents, proving to be highly effective in removing pollutants from water [83,85,99]. Activated carbon serves as a versatile material for the removal of heavy metals and organic compounds [100,101,102] and desalination [83,88,103]. The term ‘activated carbon’ is used for carbon-containing materials with a high micropores volume, favorable pore size distribution, extensive surface area (600–4300 m2g−1) [85,104] and high adsorption capacity [83,85,98,105,106]. Almost any carbonaceous material can be used to produce activated carbon, such as coconut shells, lignite, wood, sawdust, coal, and peat [98,103,105,107,108], but its properties are influenced by the raw material and the activation method [85]. Activated carbons are generally very-broad-spectrum adsorbents [86] and their low price makes them favorable [83]. Furthermore, their favorable features include their high thermal, mechanical, and light stability [89]. AC is available in two forms: powdered activated carbon (PAC) and granular activated carbon (GAC) [98,105]. PAC generally comprises particles with a diameter of 0.2 mm, and it has a large external surface area and a high adsorption rate [105]. GAC comprises particles with a maximum diameter of 5 mm, and its external surface area is smaller than that of PAC [105]. It is generally used in fixed-bed filter systems to remove pollutants from water because the granular form is better suited to continuous contact [98,105]. The adsorption efficiency of GAC can be affected by the carbon source material, carbon particle size, influent contaminant concentration, and contact time [109]. Grant et al. [109] conducted a study using a small-scale Granular Activated Carbon (GAC) system to effectively remove a range of agrichemicals, including acephate, bifenthrin, chlorpyrifos, flurprimidol, glyphosate, hydrogen peroxide + peracetic acid, imidacloprid, paclobutrazol, didecyldimethylammonium chloride (DDAC), triclopyr, and uniconazole, from water. In laboratory conditions, a decrease in the concentration of plant protection agents was observed in all cases, and a greater percentage decrease was observed in 64 s of contact time than in 12 s. At the same time, it was pointed out that further studies are needed using the high flow rate, water pressure, volume, and level of suspended solids typically found in recirculated irrigation systems.
Ghanbarizadeh et al. [89] compared the adsorption capacity of unmodified activated carbon with activated carbons modified using HCl, NaCl and HCl + NaCl. The modified activated carbon, which was first treated with salt and then with acid, was the most effective in its total hardness removal.
Activated carbon can prove to be even more effective in water purification in combination with other adsorbents. Aghakhani et al. [88] conducted experiments on the desalination of irrigation water by combining activated carbon with other adsorbents, zeolite, peat, anion exchange resin, and cation exchange resin. The combination of AC with cationic resin worked most effectively in desalination.
5.1.2. Carbon Nanotubes
Nano-adsorbents show great promise in water treatment and water purification compared to traditional adsorbents, as they can provide greater efficiency and a faster adsorption speed due to their large surface area [85,110]. Activated carbon nanotubes are one of the most commonly used nano adsorbents in wastewater treatment [110].
Carbon nanotubes (CNTs) possess unique structural, electronic, optoelectronic, semiconductor, mechanical, chemical, and physical properties. CNTs are well suited to adsorbing heavy metals from water [110].
Stafiej and Pyrzynska [111] studied the adsorption of heavy metals on CNT as a function of the pH change. In the case of CTNs, pH plays an important influencing role in the adsorption of certain ions. According to Stafiej and Pyrzynska [111], the adsorption of cations on carbon nanotubes (CNTs) is more favorable at a pH higher than the point of zero charge of the adsorbent. Conversely, a decrease in pH results in a decrease in the adsorption of cations.
5.1.3. Zeolite
Zeolite is one of the most widely used adsorbents, and is a microporous, hydrated aluminosilicate material that has an infinite, open, three-dimensional structure [85,112,113,114,115]. Ion exchangers are widely utilized to enhance water quality due to their capacity to exchange and adsorb cations present in water. This property enables them to effectively remove undesirable cations and improve the overall quality of the water [89,93,115]. The advantage is that it is available in large quantities and is therefore cheap and easily accessible [85,116].
Natural zeolite is suitable for removing heavy metal elements and for the desalination of water; this is supported by several research studies [92,93,114,117]. Furthermore, natural zeolite and its modified forms are suitable for removing anions and organic substances from water [115]. The pH of the solution affects the cation adsorption capacity of the zeolite, as strong acids and alkalis dissolve the zeolite aluminosilicate framework. For this reason, the highest cation exchange capacity is achieved at a neutral to slightly acidic pH [113]. He et al. [90] used zeolite synthesized from fly ash to remove heavy metals. The heavy metal removal efficiency increased by increasing the adsorbent dose and increasing the pH from 1.0 to 5.0.
Ghanbarizadeh et al. [89] compared the adsorbent capacity of natural and modified zeolite for hardness removal and studied the optimal contact times. The modified zeolite showed a higher efficiency when removing hardness, and most ions were removed with a contact time of 90 min.
In their study, Neag et al. [91] employed thermally activated natural zeolite for the removal of iron (Fe) and manganese (Mn) from groundwater. They found that the zeolite activated at 200 °C demonstrated the highest effectiveness in simultaneously removing both Fe and Mn. Fe removal was more efficient than Mn removal.
Natural zeolites can be modified using various methods, such as acid treatment, ion exchange, and the use of surfactants [115]; thus, they are also suitable for removing anions from water, and a higher adsorption capacity can be achieved when adsorbing organic substances and anions [113,115]. Paul et al. [92] used modified zeolite treated with Ca(OH)2 for desalination. They found that it is suitable for removing Na from salt water, while also neutralizing the acidic desalinated water. However, compared to untreated zeolite, it shows a lower efficiency during sodium removal.
Mubarak et al. [93] used TiO2-modified zeolite adsorbent to remove iron, manganese, and the total and calcium hardness from groundwater. The presence of a new active functional group resulted in the improved efficiency of the modified zeolite adsorbent compared to zeolite. This improvement was observed in terms of the removal of Mn(II), Fe(III), the total hardness, and calcium hardness. In addition, it was found that the contact time can be reduced by 29–50% if a zeolite adsorbent modified with TiO2 is used.
Gibb et al. [118] used sequentially calcined layered double hydroxide (CLDH) and acid-treated zeolite as adsorbents for aqueous solution desalination. CLDH first adsorbs Cl− and temporarily raises the pH, then the acid-treated zeolite removes Na+ and neutralizes the pH. The combined use of CLDH and acid-treated zeolite removes cations and anions (Na+, K+, Ca2+, Mg2+ and Cl−) and results in a neutral pH.
In their study, Amiri et al. [83] employed a combination of natural zeolite, activated carbon, and nano zero-valent iron to treat the gray water from a student dormitory. The treated water was subsequently utilized for irrigation purposes. This triple combination of adsorbents was found to be the most suitable for reducing COD and TDS, and almost completely removed turbidity. It proved to be more effective than using natural zeolite. The optimal contact time for the removal of COD, TDS, and turbidity was 4 h, after which no significant changes were observed. According to their research, when the three materials were used independently as adsorbents, zeolite was the most effective for reducing TDS and removing turbidity, while activated carbon proved to be the most effective for reducing COD.
5.1.4. Activated Alumina
In addition, Ghanbarizadeh et al. [89] tested activated alumina to reduce the hardness of groundwater. They found that a contact time of 90 min was the most effective; further increasing the contact time did not significantly improve the removal efficiency of the treatment process. The modification of activated alumina with salt did not increase the adsorption efficiency, while modification with acid and salt acid reduced it.
5.2. Non-Conventional Adsorbents
There are substances in nature that are available in large quantities and can remove pollutants from water and be used as adsorbents [85,119]. The raw materials of low-cost adsorbents usually do not require complicated pretreatment, which makes them economical [119].
5.2.1. Clay
Among the natural adsorbents, the use of clay is widespread, especially clays consisting of montmorillonite, bentonite, kaolinite, and zeolite [85]. Its positive properties include a high sorption capacity, high chemical and mechanical stability, a high uptake capacity, and a large surface [85,114]. Another advantage is that there are a large number of cations (Ca2+, Mg2+, H+, K+, NH4+, Na+) on their surface, which can easily be replaced by adsorbates [85].
Bentonite can also be used as an adsorbent; it is a sedimentary rock containing the clay mineral montmorillonite, a mineral mixture [114]. It is suitable for adsorbing many toxic substances and cations in water, but shows a lower affinity towards negative groups (e.g., phosphates) [114].
5.2.2. Bentonite
Bastamy et al. [114] utilized bentonite in pilot-scale bed columns constructed from various materials to investigate its effectiveness in removing pollutants from wastewater. In the experimental setup, the column was divided into three sections. The top and bottom sections were filled with a layer of gravel, each measuring 10 cm in thickness. The middle section of the column was dedicated to ion exchange beds, which were composed of a mixture of sand and bentonite. The sand-to-bentonite ratio used in the beds was 1:2. They were able to achieve a high removal efficiency, so the water could immediately be used for irrigation. The optimal removal efficiency for the tested parameters was achieved when the contact time was set at 4 h. Subsequently, the removal efficiency reached a stable state, indicating that further increasing the contact time would not result in any significant improvements in the removal process.
5.2.3. Sand
Sand is also a substance that occurs in large quantities in nature and can be used for the adsorption of heavy metals from water. Kan et al. [95] attempted to remove Mn from water using Fe and Mn oxide-coated sand. The primary objective of the study was to examine the influence of pH and temperature on the removal efficiency of manganese (Mn) from wastewater. The pH significantly affects the adsorption of Mn, and increasing the pH from 5.5 to 8 caused a significant increase in the adsorption capacity. Furthermore, the temperature is also a significant factor, and the adsorption of Mn increased as the temperature increased from 25 °C to 45 °C. In general, manganese oxides have a higher adsorption capacity for Mn than iron oxides.
5.2.4. Agricultural Wastes
Agricultural wastes can be utilized as alternative adsorbents due to their unique chemical properties and distinctive structure. The polymer chains present in these wastes often contain specific functional groups, including alcohol, phenol, aldehyde, carboxyl, and ketone. These functional groups contribute to their adsorption capabilities and make them suitable for various applications [85,120]. They are economical as they are abundantly available, renewable, environmentally friendly, and low-cost [121].
Peels of fruits and vegetables, among the agricultural waste materials, are significant as potential adsorbents in water treatment. Many of these peels are typically discarded as waste without further use, but they possess the potential to be utilized as effective adsorbents. By repurposing these peels, their adsorption properties can be harnessed to remove contaminants and improve water quality, making them a valuable resource in sustainable water treatment strategies [121].
Agricultural wastes are mostly not considered effective adsorbents, but their adsorption efficiency can be significantly increased by physical and chemical methods [122]. In bioadsorption, a simple chemically modified adsorbent can be developed from certain plant materials such as bark, leaves, husks, stems, branches, and pods [120].
Biosorbents have a relatively high biosorption capacity, strong mechanical strength, good thermal stability, and chemical resistance [123]. In recent years, biomass has mainly been used as a biosorbent, since it is difficult to meet the needs of practical applications using a single type of biological adsorbent. For the removal of heavy metals, biomass has already been combined with nanoparticles in several experiments, thereby achieving excellent adsorption properties. The creation of multi-biological adsorbents is also an effective modification, as their use can have a symbiotic and synergistic effect. Biosorbents show a lower adsorption efficiency compared to traditional adsorbents, which is why they are currently less in demand [124].
Cork is obtained from the trunk and main branches of Quercus suber L. in the form of a half-tube. Sorption using natural cork or cork modified to biochar or activated carbon can be used for a wide range of pollutants (heavy metals, organic pollutants) and in different states (liquid, gas). Cork is more selectively adsorptive for Hg than for Cd, with minimal interference of the latter, while in seawater the adsorption of Hg is much more reduced due to the formation of Hg–chlorine complexes. Anionic metals are more easily adsorbed at acidic pH levels, as electrostatic attraction may play a role in their uptake [125].
In their study, Yan et al. produced biochar from corn stalks, which can be conveniently and inexpensively used as a tool for removing various metal contaminants from wastewater. During their experiments, they investigated the removal efficiency of Cd and Pb contaminants. They conducted batch experiments while varying different factors such as pH, temperature, and background ionic strength. Increasing the pH and temperature was found to enhance the adsorption capacity, while the ionic strength showed negative effects [126].
In their study, Murtaza et al. conducted a comprehensive analysis of the mechanisms involved in the immobilization and adsorption of heavy metals and organic pollutants using biochar under varying environmental conditions. They concluded that the efficiency of biochar is influenced by factors such as pyrolysis temperature, feedstock type, residence time, and application rate. Increasing the pyrolysis temperature facilitates the production of biochars with higher porosity and a specific surface area, thereby enhancing their adsorption potential. It is important to note that the pyrolysis process generates impurities like volatile organic compounds (VOCs), dioxins, and polycyclic aromatic hydrocarbons (PAHs), which have adverse environmental effects. Hence, using unpolluted feedstock as the starting material and employing appropriate carbonization technology are imperative measures [127].
Raheem et al. [94] attempted to remove iron from groundwater using an adsorbent made from a mixture of sawdust and barley husk in different proportions. They found that a mixing ratio of sawdust and barley husk of 0.5:1 resulted in the highest adsorption efficiency at pH 6.5.
Microorganisms could be a new alternative for removing metal ions from water. Saccharomyces cerevisiae is one of the most promising bioadsorbents for the removal of metal ions from water.
Fadel et al. [96] investigated the possibility of removing manganese ions from water with S. cerevisiae. They concluded that it can be used well for this purpose, and that the pH has a significant role in the adsorption efficiency. Increasing the pH from 5 to 9 significantly increased the adsorption efficiency. Regarding the contact time, the microorganism proved to be the most effective during a contact time of 30 to 45 min.
Kamarudzaman et al. [97] used mushroom compost in a fixed-bed column as a biosorbent to remove Mn from an aqueous solution. Pleurotus mushroom compost contains lignocellulosic, which binds metal ions, and is also available in large quantities, which makes it an inexpensive bioorganic.
Peat is present in large quantities in nature and can also be used as a bioadsorbent in water purification [85]. It has a large surface area and a high adsorption capacity [85]. The main components of peat are lignin, cellulose, and humic acid [85].
6. Conclusions
Traditional surface irrigation is outdated due to scarce water reserves, but the many micro irrigation techniques that are considered modern, in addition to their efficiency and proven results, require a high level of knowledge and attention, both in their construction and application. The quality of irrigation water is affected by both chemical and physical pollutants. Chemical pollutants are important for both the soil and the irrigation system, while mechanical pollutants can mainly endanger the operational safety. With regular irrigation, special attention must be paid to water quality, as the harmful effects of irrigation often occur cumulatively (salt accumulation, leaching, etc.). The chemical treatment of irrigation water is problematic, so the best method is to choose a water source that ensures good quality. The mechanical treatment of irrigation water is determined by the sensitivity of the irrigation equipment. It is not worth filtering the water unnecessarily, because it involves a loss of energy. However, it must be taken into account that the nutrients introduced during irrigation can easily colonize microorganisms (algae, bacteria), which can cause the build-up of slime, making it easier for particles to accumulate in the system.
The use of adsorbents can be a good method for water purification, which makes it suitable for irrigation. Conventional adsorbents usually have a higher adsorption capacity but are expensive. On the other hand, non-conventional adsorbents are easily accessible and therefore cheap, but have a lower adsorption capacity. Otherwise, biological treatment methods are reported to be useful for organic contaminants and the removal of heavy metals; however, it is quite challenging to keep the microorganism alive and handle many other factors.
For salt and to avoid the formation of scaling, a couple of desalination methods have been used; these include electromagnetic field, electrostatic precipitator and even membrane separation techniques, which have a couple of limitations, such as a lower effectivity at a higher salt concentration, scaling, membrane fouling and a high energy consumption for running it. The use of adsorbents for purification could help solve many of the issues of using water for irrigation that would otherwise not be used for other purposes and become wastewater. The specific advantages of using adsorbents for wastewater treatment could be their low operation cost on the energy side and specific removal of many organic and inorganic pollutants. Therefore, the separation of interfering components via adsorption is effective and promising for future application as expected irrigation demands increase.
Author Contributions
Conceptualization, T.K. and E.T.; writing—review and editing, T.K., G.I.N., I.P. and Z.L.K.; supervision, I.G.; funding acquisition and resources: I.G. All authors have read and agreed to the published version of the manuscript.
Funding
This work was implemented by the TKP2021-NKTA-21 project with the support provided by the Ministry of Culture and Innovation of Hungary from the National Research, Development and Innovation Fund, financed under the 2021 Thematic Excellence Programme funding scheme.
Data Availability Statement
No new data were created or analyzed in this study.
Acknowledgments
We would like to thank all of the participants in our review, who generously shared their time, experiences, and insights with us. Their willingness to engage with our research was essential to the success of this project, and we are deeply grateful for their participation. Overall, this research project would not have been possible without the support and contributions of our institute. We are deeply grateful to all of those who helped to make this review a reality, and we hope that our findings will make a meaningful contribution to the field.
Conflicts of Interest
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
References
- Zittis, G.; Almazroui, M.; Alpert, P.; Ciais, P.; Cramer, W.; Dahdal, Y.; Fnais, M.; Francis, D.; Hadjinicolaou, P.; Howari, F.; et al. Climate Change and Weather Extremes in the Eastern Mediterranean and Middle East. Rev. Geophys. 2022, 60, e2021RG000762. [Google Scholar] [CrossRef]
- Malhi, G.S.; Kaur, M.; Kaushik, P. Impact of Climate Change on Agriculture and Its Mitigation Strategies: A Review. Sustainability 2021, 13, 1318. [Google Scholar] [CrossRef]
- Tabari, H. Climate Change Impact on Flood and Extreme Precipitation Increases with Water Availability. Sci. Rep. 2020, 10, 13768. [Google Scholar] [CrossRef] [PubMed]
- Climate Change and Land: An IPCC Special Report on Climate Change, Desertification, Land Degradation, Sustainable Land Management, Food Security, and Greenhouse Gas Fluxes in Terrestrial Ecosystems; 2019; pp. 1–864.
- Akhtar, N.; Syakir Ishak, M.I.; Bhawani, S.A.; Umar, K. Various Natural and Anthropogenic Factors Responsible for Water Quality Degradation: A Review. Water 2021, 13, 2660. [Google Scholar] [CrossRef]
- Khan, S.; Tariq, R.; Yuanlai, C.; Blackwell, J. Can Irrigation Be Sustainable? Agric. Water Manag. 2006, 80, 87–99. [Google Scholar] [CrossRef]
- Hagan, R.M. Water-Soil-Plant Relations. Calif. Agric. 1957, 11, 9–12. [Google Scholar]
- Ampim, P.A.Y.; Obeng, E.; Olvera-Gonzalez, E. Indoor Vegetable Production: An Alternative Approach to Increasing Cultivation. Plants 2022, 11, 2843. [Google Scholar] [CrossRef] [PubMed]
- Gerdes, M.E.; Cruz-Cano, R.; Solaiman, S.; Ammons, S.; Allard, S.M.; Sapkota, A.R.; Micallef, S.A.; Goldstein, R.E.R. Impact of Irrigation Water Type and Sampling Frequency on Microbial Water Quality Profiles Required for Compliance with U.S. Food Safety Modernization Act Produce Safety Rule Standards. Environ. Res. 2022, 205, 112480. [Google Scholar] [CrossRef]
- European Council. 91/271/EEC Directive Concerning Urban Waste Water Treatment. Off. J. Eur. Communities II 1991, 135, 40–52. [Google Scholar]
- Council of European Communities. 98/83/EC Directive on the Quality of Water Intended for Human Consumption. Off. J. Eur. Communities 1998, 330, 32–54. [Google Scholar] [CrossRef]
- Act No. LIII of 1995 on the General Rules of Environmental Protection; Budapest, Hungary, 1995.
- Act No. LIII of 1996 on Nature Conservation; Budapest, Hungary, 1996.
- Act No. CLXXXV of 2012 on Waste; Budapest, Hungary, 2012.
- European Parliament; European Council. 2007/60/EC Directive on the Assessment and Management of Flood Risks. Off. J. Eur. Union 2007, 288, 27–34. [Google Scholar]
- European Parliament; European Council. 2000/60/EC Directive Establishing a Framework for Community Action in the Field of Water Policy. Off. J. Eur. Communities 2000, 1–72. [Google Scholar]
- Decree of the Ministry of Agriculture No. 90/2008. (VII. 18.) on Detailed Rules for Drawing Up the Soil Protection Plan. Budapest, Hungary, 2008. Available online: https://www.ecolex.org/details/legislation/decree-no-90-of-2008-vii-18-fvm-of-the-ministry-of-agriculture-and-rural-development-laying-down-detailed-rules-of-elaboration-of-soil-conservation-plans-lex-faoc124169/ (accessed on 24 July 2023).
- MSZ-10-640:1989 Water Management. Water Quality. Requirements for the Quality of Irrigation Water (Hungarian Standard). Budapest, Hungary, 1989. Available online: https://oszkdk.oszk.hu/storage/00/02/38/12/dd/1/Alkalmazkod___v__zgazd__lkod__s_ABSZTRAKT.pdf (accessed on 24 July 2023).
- Act No. CXIII on Farming Irrigation; Budapest, Hungary, 2019.
- Paudel, K.P.; Pandit, M.; Roger, H. Irrigation Water Sources and Irrigation Application Methods Used by U.S. Plant Nursery Producers. Water Resour. Res. Res. 2016, 52, 698–712. [Google Scholar] [CrossRef]
- Ma, L.; Liu, Y.; Zhang, J.; Yang, Q.; Li, G.; Zhang, D. Impacts of Irrigation Water Sources and Geochemical Conditions on Vertical Distribution of Pharmaceutical and Personal Care Products (PPCPs) in the Vadose Zone Soils. Sci. Total Environ. 2018, 626, 1148–1156. [Google Scholar] [CrossRef] [PubMed]
- Malakar, A.; Snow, D.D.; Ray, C. Irrigation Water Quality—A Contemporary Perspective. Water 2019, 11, 1482. [Google Scholar] [CrossRef]
- Stefanovics, P.; Filep, G.; Füleky, G. Talajtan; Mezőgazda Kiadó: Budapest, Hungary, 2010. [Google Scholar]
- Darab, K.; Ferencz, K. Öntözött Területek Talajtérképezése; Országos Mezőgazdasági Minőségvizsgáló Intézet: Budapest, Hungary, 1969. [Google Scholar]
- George, P.R. Agricultural Water Quality Criteria: Irrigation Aspects; Department of Agriculture: Merredin, Australia, 1983. [Google Scholar]
- Zaman, M.; Shahid, S.A.; Heng, L. Guideline for Salinity Assessment, Mitigation and Adaptation Using Nuclear and Related Techniques; Springer: Berlin, Germany, 2018; ISBN 9783319961897. [Google Scholar]
- Zhao, D.; Hao, Z.; Wang, J.; Tao, J. Effects of PH in Irrigation Water on Plant Growth and Flower Quality in Herbaceous Peony (Paeonia lactiflora Pall.). Sci. Hortic. 2013, 154, 45–53. [Google Scholar] [CrossRef]
- Guimarães, J. de J.; Sousa, F.G.G. de; Román, R.M.S.; Dal Pai, A.; Rodrigues, S.A.; Sarnighausen, V.C.R. Effect of Irrigation Water PH on the Agronomic Development of Hops in Protected Cultivation. Agric. Water Manag. 2021, 253, 106924. [Google Scholar] [CrossRef]
- Adamu, G.K. Quality of Irrigation Water and Soil Characteristics of Watari Irrigation Project. Am. J. Eng. Res. 2013, 02, 59–68. [Google Scholar]
- Seilsepour, M.; Rashidi, M.; Khabbaz, B.G. Prediction of Soil Exchangeable Sodium Ratio Based on Soil Sodium Adsorption Ratio. World Acad. Sci. Eng. Technol. 2010, 46, 255–257. [Google Scholar]
- Machado, R.M.A.; Serralheiro, R.P. Soil Salinity: Effect on Vegetable Crop Growth. Management Practices to Prevent and Mitigate Soil Salinization. Horticulturae 2017, 3, 30. [Google Scholar] [CrossRef]
- Yuan, C.; Feng, S.; Huo, Z.; Ji, Q. Effects of Deficit Irrigation with Saline Water on Soil Water-Salt Distribution and Water Use Efficiency of Maize for Seed Production in Arid Northwest China. Agric. Water Manag. 2019, 212, 424–432. [Google Scholar] [CrossRef]
- Ajay, S. Soil Salinization Management for Sustainable Development: A Review. J. Environ. Manag. 2021, 277, 111383. [Google Scholar] [CrossRef]
- Smith, C.J.; Oster, J.D.; Sposito, G. Potassium and Magnesium in Irrigation Water Quality Assessment. Agric. Water Manag. 2015, 157, 59–64. [Google Scholar] [CrossRef]
- Rácz Istvánné Nemes, Z.J. Öntőzővizek Minősége a Hazai Vízkultúrás Zöldségtermesztésben; Corvinus University of Budapest: Budapest, Hungary, 2007. [Google Scholar]
- Sarda, P.; Sadgir, P. Assessment of Multi Parameters of Water Quality in Surface Water Bodies—A Review. Int. J. Res. Appl. Sci. Eng. Technol. 2015, 3, 331–336. [Google Scholar]
- Rengasamy, P.; Olsson, K.A. Irrigation and Sodicity. Aust. J. Soil Res. 1993, 31, 821–837. [Google Scholar] [CrossRef]
- Halliwell, D.J.; Barlow, K.M.D.M.; Nash, D.M. A Review of the Effects of Wastewater Sodium on Soil Physical Properties and Their Implications for Irrigation Systems. Aust. J. Soil Res. 2001, 39, 1259–1267. [Google Scholar] [CrossRef]
- Erel, R.; Ben-Gal, A.; Dag, A.; Schwartz, A.; Yermiyahu, U. Sodium Replacement of Potassium in Physiological Processes of Olive Trees (Var. Barnea) as Affected by Drought. Tree Physiol. 2014, 34, 1102–1117. [Google Scholar] [CrossRef]
- Qadir, M.; Schubert, S.; Oster, J.D.; Sposito, G.; Minhas, P.S.; Cheraghi, S.A.M.; Murtaza, G.; Mirzabaev, A.; Saqib, M. High-magnesium Waters and Soils: Emerging Environmental and Food Security Constraints. Sci. Total Environ. 2018, 642, 1108–1117. [Google Scholar] [CrossRef]
- Martínez-Gimeno, M.A.; Bonet, L.; Provenzano, G.; Badal, E.; Intrigliolo, D.S.; Ballester, C. Assessment of Yield and Water Productivity of Clementine Trees under Surface and Subsurface Drip Irrigation. Agric. Water Manag. 2018, 206, 209–216. [Google Scholar] [CrossRef]
- Sable, R.; Kolekar, S.; Gawde, A.; Takle, S.; Pednekar, A. A Review on Different Irrigation Methods. Int. J. Appl. Agric. Res. 2019, 14, 49–60. [Google Scholar]
- Holzapfel, E.A.; Pannunzio, A.; Lorite, I.; de Oliveira, A.S.S.; Farkas, I. Design and Management of Irrigation Systems. Chil. J. Agric. Res. 2009, 69, 17–25. [Google Scholar] [CrossRef]
- Pereira, L.S. Surface Irrigation Systems. Sustain. Irrig. Agric. 1996, 269–289. [Google Scholar] [CrossRef]
- Albaji, M.; Golabi, M.; Boroomand Nasab, S.; Zadeh, F.N. Investigation of Surface, Sprinkler and Drip Irrigation Methods Based on the Parametric Evaluation Approach in Jaizan Plain. J. Saudi Soc. Agric. Sci. 2015, 14, 1–10. [Google Scholar] [CrossRef]
- Nakayama, F.S.; Bucks, D.A. Water Quality in Drip/Trickle Irrigation: A Review. Irrig. Sci. 1991, 12, 187–192. [Google Scholar] [CrossRef]
- Yan, H.; Hui, X.; Li, M.; Xu, Y. Development in Sprinkler Irrigation Technology in China. Irrig. Drain. 2020, 69, 75–87. [Google Scholar] [CrossRef]
- Bansal, G.; Mahajan, A.; Verma, A.; Bandhu Singh, D. A Review on Materialistic Approach to Drip Irrigation System. Mater. Today Proc. 2021, 46, 10712–10717. [Google Scholar] [CrossRef]
- Henrique Bassoi, L.; Hopmans, J.W.; André de Castro Jorge, L.; Miranda de Alencar, C.; Antonio Moura Silva, J. Grapevine Root Distribution in drip and microsprinkler irrigation. Sci. Agric. 2003, 60, 377–387. [Google Scholar] [CrossRef]
- Goldberg, D.; Gornat, B.; Rimon, D. Drip Irrigation. Principles, Design and Agricultural Practices; Drip Irrigation Scientific Publications: Kfar Shmaryahu, Israel, 1976. [Google Scholar]
- Dong, R.; Liu, W.; Qu, J.; Cao, W. Accumulation of Na+ in Cotton Field under Mulched Drip Irrigation of Brackish Water in Arid Areas. Separations 2023, 10, 180. [Google Scholar] [CrossRef]
- Zapata, N.; Playán, E.; Castillo, R.; Gimeno, Y.; Oliván, I.; Jiménez, A.; Carbonell, X.; Fábregas, M.; López-Pardo, J.R.; Vicente, L.M.; et al. A Methodology to Classify Irrigated Areas: Application to the Central Ebro River Basin in Aragón (Spain). Agric. Water Manag. 2020, 241, 106365. [Google Scholar] [CrossRef]
- Arshad, I. Importance of Drip Irrigation System Installation and Management—A Review. PSM Biol. Res. 2020, 5, 22–29. [Google Scholar]
- Flores, J.H.N.; Faria, L.C.; Neto, O.R.; Diotto, A.V.; Colombo, A. Methodology for Determining the Emitter Local Head Loss in Drip Irrigation Systems. J. Irrig. Drain. Eng. 2020, 147, 06020014. [Google Scholar] [CrossRef]
- Shi, K.; Lu, T.; Zheng, W.; Zhang, X.; Zhangzhong, L. A Review of the Category, Mechanism, and Controlling Methods of Chemical Clogging in Drip Irrigation System. Agriculture 2022, 12, 1–20. [Google Scholar] [CrossRef]
- Prathyusha, K.; Chaitanya Suman, M. Design of Embedded Systems for the Automation of Drip Irrigation. Int. J. Appl. Innov. Eng. Manag. 2012, 1, 245–258. [Google Scholar]
- Li, Y.; Pan, J.; Chen, X.; Xue, S.; Feng, J.; Muhammad, T.; Zhou, B. Dynamic Effects of Chemical Precipitates on Drip Irrigation System Clogging Using Water with High Sediment and Salt Loads. Agric. Water Manag. 2019, 213, 833–842. [Google Scholar] [CrossRef]
- Bucks, D.A.; Nakayama, F.S.; Gilbert, R.G. Trickle Irrigation Water Quality and Preventive Maintenance. Agric. Water Manag. 1979, 2, 149–162. [Google Scholar] [CrossRef]
- Pitts, D.J.; Haman, D.Z.; Smajstria, A.G. Causes and Prevention of Emitter Plugging in Micro Irrigation Systems; Bulletin 2; Institute of Food and Agricultural Sciences, University of Florida: Gainesville, FL, USA, 1990. [Google Scholar]
- Muhammad, T.; Zhou, B.; Liu, Z.; Chen, X.; Li, Y. Effects of Phosphorus-Fertigation on Emitter Clogging in Drip Irrigation System with Saline Water. Agric. Water Manag. 2021, 243, 106392. [Google Scholar] [CrossRef]
- El Housse, M.; Hadfi, A.; Karmal, I.; Ben-Aazza, S.; Belattar, M.; Errami, M.; Mohareb, S.; Driouiche, A. Study of the Effect of Inorganic Inhibitor on the Calcium Carbonate Precipitation in the Localized Irrigation Systems. Nanotechnol. Environ. Eng. 2021, 6, 2–9. [Google Scholar] [CrossRef]
- Yan, D.; Bai, Z.; Rowan, M.; Gu, L.; Shumei, R.; Yang, P. Biofilm Structure and Its Influence on Clogging in Drip Irrigation Emitters Distributing Reclaimed Wastewater. J. Environ. Sci. 2009, 21, 834–841. [Google Scholar] [CrossRef]
- Ravina, I.; Paz, E.; Sofer, Z.; Marcu, A.; Schischa, A.; Sagi, G.; Yechialy, Z.; Lev, Y. Control of Clogging in Drip Irrigation with Stored Treated Municipal Sewage Effluent. Agric. Water Manag. 1997, 33, 127–137. [Google Scholar] [CrossRef]
- Dery, J.L.; Brassill, N.; Rock, C.M. Minimizing Risks: Use of Surface Water in Pre-Harvest Agricultural Irrigation Part I: Understanding Water Quality & Treatment Options Why Treat Agricultural Irrigation Water? Univ. Arizona Coop. Ext. 2019, 1793, 1–4. [Google Scholar]
- Lim, K.Y.; Foo, K.Y. Hazard Identification and Risk Assessment of the Organic, Inorganic and Microbial Contaminants in the Surface Water after the High Magnitude of Flood Event. Environ. Int. 2021, 157, 106851. [Google Scholar] [CrossRef] [PubMed]
- Jiang, D.; Ao, C.; Bailey, R.T.; Zeng, W.; Huang, J. Simulation of Water and Salt Transport in the Kaidu River Irrigation District Using the Modified SWAT-Salt. Agric. Water Manag. 2022, 272, 107845. [Google Scholar] [CrossRef]
- Battilani, A.; Steiner, M.; Andersen, M.; Back, S.N.; Lorenzen, J.; Schweitzer, A.; Dalsgaard, A.; Forslund, A.; Gola, S.; Klopmann, W.; et al. Decentralised Water and Wastewater Treatment Technologies to Produce Functional Water for Irrigation. Agric. Water Manag. 2010, 98, 385–402. [Google Scholar] [CrossRef]
- Vassalle, L.; Sunyer-Caldú, A.; Uggetti, E.; Díez-Montero, R.; Díaz-Cruz, M.S.; García, J.; García-Galán, M.J. Bioremediation of Emerging Micropollutants in Irrigation Water. The Alternative of Microalgae-Based Treatments. J. Environ. Manag. 2020, 274. [Google Scholar] [CrossRef] [PubMed]
- Gertsis, A.; Zoukidis, K. Irrigation with Highly Saline Water: A New Innovative Water Treatment System Evaluated for Vegetable Production in Greenhouse. Eur. Water 2017, 59, 331–337. [Google Scholar]
- Seyyedi, M. Magnetic Fields and Manipulation of Water Behaviors: A Viable Method for Reducing RO Specific Energy Consumption. In Proceedings of the 2022 Membrane Technology Conference and Exposition, Las Vegas, NV, USA, 21–25 February 2022. [Google Scholar]
- Surendran, U.; Sandeep, O.; Joseph, E.J. The Impacts of Magnetic Treatment of Irrigation Water on Plant, Water and Soil Characteristics. Agric. Water Manag. 2016, 178, 21–29. [Google Scholar] [CrossRef]
- Hachicha, M.; Kahlaoui, B.; Khamassi, N.; Misle, E.; Jouzdan, O. Effect of Electromagnetic Treatment of Saline Water on Soil and Crops. J. Saudi Soc. Agric. Sci. 2018, 17, 154–162. [Google Scholar] [CrossRef]
- Wiedenfeld, B. Effects of Irrigation Water Salinity and Electrostatic Water Treatment for Sugarcane Production. Agric. Water Manag. 2008, 95, 85–88. [Google Scholar] [CrossRef]
- Wei, K.; Zhang, J.; Wang, Q.; Guo, Y.; Mu, W. Irrigation with Ionized Brackish Water Affects Cotton Yield and Water Use Efficiency. Ind. Crops Prod. 2022, 175, 114244. [Google Scholar] [CrossRef]
- Jarma, Y.A.; Karaoğlu, A.; Senan, I.R.A.; Meriç, M.K.; Kukul, Y.S.; Özçakal, E.; Barlas, N.T.; Çakıcı, H.; Baba, A.; Kabay, N. Utilization of Membrane Separation Processes for Reclamation and Reuse of Geothermal Water in Agricultural Irrigation of Tomato Plants-Pilot Membrane Tests and Economic Analysis. Desalination 2022, 528, 115608. [Google Scholar] [CrossRef]
- Garg, N.; Choudhary, O.P.; Thaman, S.; Sharma, V.; Singh, H.; Vashistha, M.; Sekhon, K.S.; Sharda, R.; Dhaliwal, M.S. Effects of Irrigation Water Quality and NPK-Fertigation Levels on Plant Growth, Yield and Tuber Size of Potatoes in a Sandy Loam Alluvial Soil of Semi-Arid Region of Indian Punjab. Agric. Water Manag. 2022, 266, 107604. [Google Scholar] [CrossRef]
- Wang, T.; Wang, Z.; Zhang, J.; Ma, K. An Optimum Combination of Irrigation Amount, Irrigation Water Salinity and Nitrogen Application Rate Can Improve Cotton (for Fiber) Nitrogen Uptake and Final Yield. Ind. Crops Prod. 2022, 187, 115386. [Google Scholar] [CrossRef]
- Sang, H.; Guo, W.; Gao, Y.; Jiao, X.; Pan, X. Effects of Alternating Fresh and Saline Water Irrigation on Soil Salinity and Chlorophyll Fluorescence of Summer Maize. Water 2020, 12, 3054. [Google Scholar] [CrossRef]
- Dong, X.; Wang, J.; Zhang, X.; Dang, H.; Singh, B.P.; Liu, X.; Sun, H. Long-Term Saline Water Irrigation Decreased Soil Organic Carbon and Inorganic Carbon Contents. Agric. Water Manag. 2022, 270, 107760. [Google Scholar] [CrossRef]
- Ouoba, N.; Phung, L.D.; Sasaki, A.; Pham, D.V.; Watanabe, T. Drip Fertigation with Treated Municipal Wastewater and Soil Amendment with Composted Sewage Sludge for Sustainable Protein-Rich Rice Cultivation. Environ. Technol. Innov. 2022, 28, 102569. [Google Scholar] [CrossRef]
- Yang, S.; Feng, W.; Wang, S.; Chen, L.; Zheng, X.; Li, X.; Zhou, D. Farmland Heavy Metals Can Migrate to Deep Soil at a Regional Scale: A Case Study on a Wastewater-Irrigated Area in China. Environ. Pollut. 2021, 281, 116977. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.; Zhang, X.; Wang, Y.; Shi, E.; Lin, H.; Chen, G. Removal of Pb2+ and Cd2+ from Irrigation Water and Replenishment of Mineral Nutrients Using a Low-Cost Mineral Adsorbent Derived from Potassium-Rich Aluminum Silicates. J. Environ. Chem. Eng. 2023, 11, 109282. [Google Scholar] [CrossRef]
- Amiri, M.J.; Bahrami, M.; Badkouby, M.; Kalavrouziotis, I.K. Greywater Treatment Using Single and Combined Adsorbents for Landscape Irrigation. Environ. Process. 2019, 6, 43–63. [Google Scholar] [CrossRef]
- Hameed, B.H.; Tan, I.A.W.; Ahmad, A.L. Adsorption Isotherm, Kinetic Modeling and Mechanism of 2,4,6-Trichlorophenol on Coconut Husk-Based Activated Carbon. Chem. Eng. J. 2008, 144, 235–244. [Google Scholar] [CrossRef]
- Singh, N.B.; Nagpal, G.; Agrawal, S. Rachna Water Purification by Using Adsorbents: A Review. Environ. Technol. Innov. 2018, 11, 187–240. [Google Scholar] [CrossRef]
- Crini, G.; Lichtfouse, E.; Wilson, L.D.; Morin-Crini, N. Conventional and Non-Conventional Adsorbents for Wastewater Treatment. Environ. Chem. Lett. 2019, 17, 195–213. [Google Scholar] [CrossRef]
- Jain, C.K.; Malik, D.S.; Yadav, A.K. Applicability of Plant Based Biosorbents in the Removal of Heavy Metals: A Review. Environ. Process. 2016, 3, 495–523. [Google Scholar] [CrossRef]
- Aghakhani, A.; Mousavi, S.F.; Mostafazadeh-Fard, B.; Rostamian, R.; Seraji, M. Application of Some Combined Adsorbents to Remove Salinity Parameters from Drainage Water. Desalination 2011, 275, 217–223. [Google Scholar] [CrossRef]
- Ghanbarizadeh, P.; Parivazh, M.M.; Abbasi, M.; Osfouri, S.; Dianat, M.J.; Rostami, A.; Dibaj, M.; Akrami, M. Performance Enhancement of Specific Adsorbents for Hardness Reduction of Drinking Water and Groundwater. Water 2022, 14, 2749. [Google Scholar] [CrossRef]
- He, K.; Chen, Y.; Tang, Z.; Hu, Y. Removal of Heavy Metal Ions from Aqueous Solution by Zeolite Synthesized from Fly Ash. Environ. Sci. Pollut. Res. 2016, 23, 2778–2788. [Google Scholar] [CrossRef] [PubMed]
- Neag, E.; Török, A.I.; Tanaselia, C.; Aschilean, I.; Senila, M. Kinetics and Equilibrium Studies for the Removal of Mn and Fe from Binary Metal Solution Systems Using a Romanian Thermally Activated Natural Zeolite. Water 2020, 12, 1614. [Google Scholar] [CrossRef]
- Paul, B.; Dynes, J.J.; Chang, W. Modified Zeolite Adsorbents for the Remediation of Potash Brine-Impacted Groundwater: Built-in Dual Functions for Desalination and PH Neutralization. Desalination 2017, 419, 141–151. [Google Scholar] [CrossRef]
- Mubarak, M.F.; Mohamed, A.M.G.; Keshawy, M.; elMoghny, T.A.; Shehata, N. Adsorption of Heavy Metals and Hardness Ions from Groundwater onto Modified Zeolite: Batch and Column Studies. Alex. Eng. J. 2022, 61, 4189–4207. [Google Scholar] [CrossRef]
- Raheem, S.A.; Kadhim, E.J.; Abdulhasan, M.J. Comparative Study of Iron Removal from Groundwater Using Low Cost Adsorbents. J. Ecol. Eng. 2022, 23, 18–23. [Google Scholar] [CrossRef]
- Kan, C.C.; Aganon, M.C.; Futalan, C.M.; Dalida, M.L.P. Adsorption of Mn2+ from Aqueous Solution Using Fe and Mn Oxide-Coated Sand. J. Environ. Sci. 2013, 25, 1483–1491. [Google Scholar] [CrossRef]
- Fadel, M.; Hassanein, N.M.; Elshafei, M.M.; Mostafa, A.H.; Ahmed, M.A.; Khater, H.M. Biosorption of Manganese from Groundwater by Biomass of Saccharomyces Cerevisiae. HBRC J. 2017, 13, 106–113. [Google Scholar] [CrossRef]
- Kamarudzaman, A.N.; Chay, T.C.; Amir, A.; Talib, S.A. Biosorption of Mn(II) Ions from Aqueous Solution by Pleurotus Spent Mushroom Compost in a Fixed-Bed Column. Procedia—Soc. Behav. Sci. 2015, 195, 2709–2716. [Google Scholar] [CrossRef]
- Bhatnagar, A.; Minocha, A.K. Conventional and Non-Conventional Adsorbents for Removal of Pollutants from Water—A Review. Indian J. Chem. Technol. 2006, 13, 203–217. [Google Scholar]
- McKay, G.; Bino, M.J.; Altamemi, A.R. The Adsorption of Various Pollutants from Aqueous Solutions on to Activated Carbon. Water Res. 1985, 19, 491–495. [Google Scholar] [CrossRef]
- Leboda, R. Carbon-Mineral Adsorbents—New Type of Sorbents Part II. Surface Properties and Methods of Their Modification. Mater. Chem. Phys. 1993, 34, 123–141. [Google Scholar] [CrossRef]
- Gabaldón, C.; Marzal, P.; Seco, A.; Gonzalez, J.A. Cadmium and Copper Removal by a Granular Activated Carbon in Laboratory Column Systems. Sep. Sci. Technol. 2000, 35, 1039–1053. [Google Scholar] [CrossRef]
- Bhatnagar, A.; Sillanpää, M. A Review of Emerging Adsorbents for Nitrate Removal from Water. Chem. Eng. J. 2011, 168, 493–504. [Google Scholar] [CrossRef]
- Aghakhani, A.; Mousavi, S.F.; Mostafazadeh-Fard, B. Desalination of Saline Water with Single and Combined Adsorbents. Desalin. Water Treat. 2013, 51, 1928–1935. [Google Scholar] [CrossRef]
- Ahmed, A.S.; Alsultan, M.; Sabah, A.A.; Swiegers, G.F. Carbon Dioxide Adsorption by a High-Surface-Area Activated Charcoal. J. Compos. Sci. 2023, 7, 179. [Google Scholar] [CrossRef]
- Carrott, P.J.M.; Carrott, M.R. Lignin—From Natural Adsorbent to Activated Carbon: A Review. Bioresour. Technol. 2007, 98, 2301–2312. [Google Scholar] [CrossRef]
- Ahmed, M.; Chin, Y.H.; Guo, X.; Zhao, X.M. Microwave Assisted Digestion Followed by ICP-MS for Determination of Trace Metals in Atmospheric and Lake Ecosystem. J. Environ. Sci. 2017, 55, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Mattson, J.S.; Mark, H.B. Activated Carbon: Surface Chemistry and Adsorption from Solution; M. Dekker: New York, NY, USA, 1971; ISBN 0824714431. [Google Scholar]
- Chiang, Y.C.; Chiang, P.C.; Chiang, H.L.; Chang, E.E. Determination of Chemical Characterizations of Activated Carbons from Various Raw Materials. Toxicol. Environ. Chem. 1998, 67, 275–291. [Google Scholar] [CrossRef]
- Grant, G.A.; Fisher, P.R.; Barrett, J.E.; Wilson, P.C. Removal of Agrichemicals from Water Using Granular Activated Carbon Filtration. Water. Air. Soil Pollut. 2019, 230, 7. [Google Scholar] [CrossRef]
- Sadegh, H.; Ali, G.A.M.; Gupta, V.K.; Makhlouf, A.S.H.; Shahryari-ghoshekandi, R.; Nadagouda, M.N.; Sillanpää, M.; Megiel, E. The Role of Nanomaterials as Effective Adsorbents and Their Applications in Wastewater Treatment. J. Nanostruct. Chem. 2017, 7, 1–14. [Google Scholar] [CrossRef]
- Stafiej, A.; Pyrzynska, K. Adsorption of Heavy Metal Ions with Carbon Nanotubes. Sep. Purif. Technol. 2007, 58, 49–52. [Google Scholar] [CrossRef]
- Mumpton, F.A. La Roca Magica: Uses of Natural Zeolites in Agriculture and Industry. Proc. Natl. Acad. Sci. USA 1999, 96, 3463–3470. [Google Scholar] [CrossRef] [PubMed]
- Wen, J.; Dong, H.; Zeng, G. Application of Zeolite in Removing Salinity/Sodicity from Wastewater: A Review of Mechanisms, Challenges and Opportunities. J. Clean. Prod. 2018, 197, 1435–1446. [Google Scholar] [CrossRef]
- El Bastamy, E.; Ibrahim, L.A.; Ghandour, A.; Zelenakova, M.; Vranayova, Z.; Abu-Hashim, M. Efficiency of Natural Clay Mineral Adsorbent Filtration Systems in Wastewater Treatment for Potential Irrigation Purposes. Sustainability 2021, 13, 5738. [Google Scholar] [CrossRef]
- Wang, S.; Peng, Y. Natural Zeolites as Effective Adsorbents in Water and Wastewater Treatment. Chem. Eng. J. 2010, 156, 11–24. [Google Scholar] [CrossRef]
- Babel, S.; Kurniawan, T.A. Low-Cost Adsorbents for Heavy Metals Uptake from Contaminated Water: A Review. J. Hazard. Mater. 2003, 97, 219–243. [Google Scholar] [CrossRef]
- Gibb, N.P.; Dynes, J.J.; Chang, W. Synergistic Desalination of Potash Brine-Impacted Groundwater Using a Dual Adsorbent. Sci. Total Environ. 2017, 593–594, 99–108. [Google Scholar] [CrossRef] [PubMed]
- Gibb, N.P.; Dynes, J.J.; Chang, W. A Recyclable Adsorbent for Salinized Groundwater: Dual-Adsorbent Desalination and Potassium-Exchanged Zeolite Production. Chemosphere 2018, 209, 721–729. [Google Scholar] [CrossRef] [PubMed]
- Lim, A.P.; Aris, A.Z. A Review on Economically Adsorbents on Heavy Metals Removal in Water and Wastewater. Rev. Environ. Sci. Biotechnol. 2014, 13, 163–181. [Google Scholar] [CrossRef]
- Bilal, M.; Ihsanullah, I.; Younas, M.; Ul Hassan Shah, M. Recent Advances in Applications of Low-Cost Adsorbents for the Removal of Heavy Metals from Water: A Critical Review. Sep. Purif. Technol. 2022, 278, 119510. [Google Scholar] [CrossRef]
- Bhatnagar, A.; Sillanpää, M.; Witek-Krowiak, A. Agricultural Waste Peels as Versatile Biomass for Water Purification—A Review. Chem. Eng. J. 2015, 270, 244–271. [Google Scholar] [CrossRef]
- Younas, H.; Younas, F. Wastewater Application in Agriculture—A Review. Water Air Soil Pollut. 2022, 233, 329. [Google Scholar] [CrossRef]
- Beni, A.A.; Esmaeili, A. Biosorption, an Efficient Method for Removing Heavy Metals from Industrial Effluents: A Review. Environ. Technol. Innov. 2020, 17, 100503. [Google Scholar] [CrossRef]
- Qin, H.; Hu, T.; Zhai, Y.; Lu, N.; Aliyeva, J. The Improved Methods of Heavy Metals Removal by Biosorbents: A Review. Environ. Pollut. 2020, 258, 113777. [Google Scholar] [CrossRef]
- Jesus, J.; Nunes da Silva, R.; Pintor, A. Advances in Cork Use in Adsorption Applications: An Overview of the Last Decade of Research. Separations 2023, 10, 1–18. [Google Scholar] [CrossRef]
- Yan, S.; Yu, W.; Yang, T.; Li, Q.; Guo, J. The Adsorption of Corn Stalk Biochar for Pb and Cd: Preparation, Characterization, and Batch Adsorption Study. Separations 2022, 9, 22. [Google Scholar] [CrossRef]
- Murtaza, G.; Ahmed, Z.; Eldin, S.M.; Ali, I.; Usman, M.; Iqbal, R.; Rizwan, M.; Abdel-Hameed, U.K.; Haider, A.A.; Tariq, A. Biochar as a Green Sorbent for Remediation of Polluted Soils and Associated Toxicity Risks: A Critical Review. Separations 2023, 10, 197. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).