Abstract
The aim of our research was to perform a comprehensive study of ecdysterone (ECD)-containing dietary supplements (DSs). Two analytical methods were optimised according to the expected concentration of the target compounds: quantitation of ECD by liquid chromatography (LC) coupled to diode array detector (DAD), and limit test for 47 World Anti-Doping Agency prohibited by LC coupled with tandem mass spectrometer (MS/MS). For quantitation of ECD, the method was fully validated with outstanding performance characteristics (LOD: 35 µg·g−1, LOQ: 115 µg·g−1, CV% < 5%), resulting in significantly lower LOD and shorter runtime than published previously. For limit tests, a chromatographic method was developed to obtain excellent separation, while MS/MS parameters were optimised to allow the lowest possible reporting limit (RL: 0.6–10 ng·g−1 or mL−1). Twenty-one ecdysterone-containing DSs from ten brands were analysed. In all cases, the measured ECD content was much lower than labelled, and 20% of the samples contained a prohibited substance. The concentration of ecdysterone and contaminations varied randomly from batch to batch. The developed methods help to prevent the use of contaminated or useless DSs.
1. Introduction
Substances in the World Anti-Doping Agency (WADA) monitoring program are not prohibited, but they are monitored by WADA in order to detect potential patterns of misuse in sports. Since 2020, ecdysterone (20-hydroxyecdysone (ECD)) has been included in the monitoring program [1] and since that time, the results found in urine samples have been evaluated [2]. ECD is a naturally-occurring steroid hormone with a structure similar to anabolic androgenic steroids (AASs). It is primarily derived from herbal sources (Spinacia oleracea, Cyanotis arachnoidea, Leuzea carthamoides, Rhaponticum carthamoides) [3,4]. Even ECD is not produced in mammalian tissues and has several physiological effects in mammals when taken externally [5]. Its impact on the anabolic pathways, without any androgenic side-effect, is the most desirable one for athletes. Haupt et al. hypothesised that ECD presents an anabolic effect mediated by an estrogenic receptor pathway [6]. Parr et al. clearly confirmed the anabolic activity of ECD, comparing its effect to those of well-known AASs and selective androgen receptor modulators (SARMs) [7]. Later, Isenmann et al. performed a detailed investigation regarding the effect of ECD, and the results demonstrated a notable performance-enhancing effect. Thus, authors strongly suggested the inclusion of ECD in class S1.2 of the WADA prohibited list [8]. Surprisingly, another quite similar study found the opposite result, namely, ECD supplementation had no effect on training adaptation and anabolic/catabolic hormone status [9]. The significant difference can presumably be explained by incorrect labeling, namely, that the real ECD content was much lower than those indicated on the product, as found by Ambrosio et al. [10]. Webshops advertise ECD-containing DSs that promise “higher muscle mass and better performance” within a short period of time. Consequently, these products are very popular among athletes, who try to find “legal” performance enhancing products [11]. Nevertheless, ECD-containing products might be contaminated with WADA-prohibited substances, thus, their use may result in a positive doping test [12,13,14,15]. Therefore, an analysis of DSs with appropriate techniques is very important [14].
Correctly choosing the most suitable analytical method according to the scope and the order of magnitude of the target compounds is crucial. Traditionally, gas chromatography coupled with mass spectrometry (GC-MS) has been the most commonly used method for the detection of steroids [16,17,18]. Over the past few decades, GC-MS methods have developed dynamically, resulting in a significant decrease in the limit of detection (LODs) from mg·g−1 to ng·g−1 range [18,19]. Triple quadrupole MS detectors (MS/MS) operating in multiple reaction monitoring (MRM) mode allow for a more reliable and sensitive identification of prohibited steroids. Taking into account the complexity of DSs, their sample preparation typically differs considerably from those of urine or blood samples, and need to be optimised [19]. Although GC-MS/MS methods are widely used to perform steroid analysis, liquid chromatographic (LC) techniques [20], in particular multicomponent LC-MS/MS techniques, are now increasingly becoming widespread in the literature as well [21,22,23]. Numerous publications have already investigated the ECD content of some DSs and different ECD-containing plant materials. Some of them have used LC-MS/MS methods for quantifications [10,24,25,26], while reversed-phase (RP) LC coupled with diode-array detector (DAD) methods are also applicable [27]. Considering that ECD is not yet prohibited but put under the monitoring program, there has been no need to use more sensitive methods. Based on the results reported so far, the composition of many ECD-containing DSs are doubtful [10], and thus, comprehensive analytical studies are needed to obtain a better knowledge of these kinds of DSs.
Hence, our aim was to quantify the still-permitted but monitored ECD in the expected order of magnitude of mg·g−1 by means of LC-DAD, and detect prohibited compounds at ng·g−1 concentration with LC-MS/MS.
2. Materials and Methods
2.1. Chemicals
Ultrapure deionised water (H2O) (<18 mΩ) was purified and provided on a daily basis by a SUEZ Water Purification Systems (Pro Analitika, Hungary, Budapest). HPLC-grade acetonitrile (ACN) and LC-MS-grade methanol (MeOH) and 2-propanol (IPA) were obtained from Thermo Fisher Scientific (Budapest, Hungary). Further solvents and reagents, namely, diethyl ether (DETE), ethyl acetate (EtAc), n-pentane, and formic acid (FA), were purchased from Merck KGaA (Darmstadt, Germany). HPLC-grade ethanol (EtOH) was purchased from Molar Chemicals (Halásztelek, Hungary). Reference standards with all details are listed in Supplementary Materials Table S1.
2.2. Samples
DSs analysed in our study were purchased online or received from the National Food Chain Safety Office ((NFCSO) Hungary, Budapest)).
2.3. Standard Solutions
Stock solutions of target compounds for both scopes and deuterated internal standard (ISTD) for the MS/MS analysis were prepared according to the format of reference standards. Certified reference materials were purchased in three formats. In the case of neat (solventless solid) standards, 10–10 mg of each reference material was weighted individually by a certified analytical balance (M5-HPB2285 Di, Bel Engineering, Monza, Italy, d = 0.1 mg) into 10-mL volumetric flasks, solubilised with the appropriate solvent and completed the volume properly, resulting in a stock solution with a concentration of 1000 mg·L−1. Stock solutions of “dilute and shot” (D&S) (certified amount of standard, approximately 1 mg) reference materials were prepared through dissolving in 1 mL of an appropriate solvent, according to the certificate of analysis and measured by a Hamilton syringe. All the solutions were sonicated until the complete transparency of the solution. Certified reference material solutions (concentration of 1000 mg·L−1) in ampules were ready for use immediately. ISTD (testosterone-d3) had a certified nominal concentration of 100·mg L−1, which was taken into account during the preparation of the diluted working solutions. All the solutions were sonicated until the complete transparency of the solution.
2.4. Sample Preparation for ECD Quantitation
According to the optimised method, homogenised samples (100 mg) were weighted into a centrifuge tube, 10 mL MeOH was added to each sample, and the tubes were shaken with an orbital open-air shaker (Lab Companion, Billerica, MA, USA) for 25 min. The tubes were then centrifuged at 3000 rpm (Andreas Hettich GmbH & Co., Tuttlingen, Germany) and finally filtrated by using a cellulose acetate syringe filter (0.45 µm, (Gen-Lab Kft, Budapest, Hungary)). Taking into account the indicated ECD content, the samples were diluted further with MeOH if needed. In some cases, finding the suitable dilution posed a challenge because the real amount differed significantly from the labeled value. Samples were prepared in duplicate.
2.5. LC-DAD Quantitative ECD Analysis
Quantitative ECD analyses were carried out on an Agilent 1290 Infinity II LC system (Agilent Technologies, Palo Alto, CA, USA), which consisted of a model G7167B injector, G7120A high-speed pump, and G7117A DAD. For the separation, a Kinetex C18 RP column (4.6 mm × 100 mm × 2.6 µm (Gen-Lab Kft, Budapest, Hungary)) was applied. The injection volume was 3 µL. The following gradient program was applied with ultrapure H2O (mobile phase (A)) and ACN (mobile phase (B)): 0 min 10% B, 7 min 95% B, 9 min 95%, 9.1 min 10% B. The re-equilibration time with the starting conditions was 3 min. The flow rate was 0.5 mL·min−1, and the column was kept at 40 °C. ECD was monitored at 254 nm. Instrument control, data acquisition, and qualitative/quantitative data analysis were provided by MassHunter software ver. B.08 (Agilent Technologies, Palo Alto, CA, USA).
2.6. Calibration and Quantification
External daily calibration was used for the quantitation of ECD from dietary supplements. Five different concentrations (in the range of 1–50 µg·mL−1) were prepared from the ECD stock solution using a volumetric flask and MeOH as a diluent. Solutions of the prepared calibration series were injected into the LC-DAD system. The calibration curves were constructed by plotting the peak area versus concentration of ECD expressed in µg·mL−1, determining the linear regression equation and coefficient. For quantitative purpose, the concentration of ECD was calculated considering the actual dilution, without any further correction.
2.7. Sample Preparation of Prohibited Substances
Extraction steps for the prohibited compound analysis were carried out as published in our previous study [19].
2.8. LC-MS/MS Qualitative Prohibited Substance Analysis
LC-MS/MS analyses were carried out on the same Agilent 1290 Infinity II LC system as previously described, coupled to an Agilent 6495B triple quadrupole mass spectrometer (Agilent Technologies, Palo Alto, CA, USA). Separation of 49 steroids was achieved with a Zorbax Eclipse Plus C18, 2.1 × 50 mm × 1.8 μm column (Agilent Technologies, Palo Alto, CA, USA). Injection volume was 1 μL. Gradient elution with H2O with 0.1% FA (eluent A) and MeOH with 0.1% FA (eluent B) was applied as follows: 0 min 30% B, 2 min 30% B, 3.5 min 65% B, 8 min 65% B, 10 min 100% B, 11 min 100% B, 11.01 min 30% B, 13 min 30% B. The MS was applied in electron spray ionisation (ESI) mode. Sheet gas and auxiliary gas temperature and flow were 210 °C and 350 °C, and 14 L·min−1 and 12 L·min−1, respectively. Capillary voltage was 3000 V. Ionisation was carried out in negative mode for two SARMs (LGD4033 and ostarine). For the other target analytes, a positive ionisation mode was applied. MS/MS detection was carried out in dynamic MRM mode (dMRM). Optimised MRM transitions with collision energies (CE) and retention times (RT) of each compound are listed in Table 1. The same software was used as in the LC-DAD analysis.

Table 1.
MRM transitions and CEs (expressed as eV) of target substances for LC-MS/MS analysis. MRM 1 was used for primary identification; MRM 2 was selected as confirmatory transition. RT was expressed in min.
2.9. Method Figures of Merit
The method figures of merit were determined based on Eurachem guidelines [28]. For the quantitation of ECD by means of LC-DAD limit of detection (LOD), the limit of quantitation (LOQ), accuracy, selectivity, and repeatability were evaluated.
According to Magnusson et al. [28], the analysis of prohibited substances was considered as a limit test. Therefore, reporting limits (RL) and selectivity were assessed for all investigated compounds. RL were determined from ten replicates of a calibrating mixture at a lower concentration level (0.6–10 ng·g−1 or mL−1) from eleven different DS matrices, including liquid and solid, simple, and complex ones. Selectivity was assessed by evaluating the presence of an interfering substance from the matrix and from the spiking mixture as well.
3. Results and Discussion
3.1. Sample Preparation Optimisation for Quantitation of ECD
In order to achieve the most efficient extraction method for ECD, five types of solvents or mixtures were tried, namely, ultrapure H2O, MeOH, ethanol (EtOH), 40:60 H2O:MeOH, and 40:60 H2O:EtOH. No significant difference between the tested extraction solvents was found, thus, MeOH was chosen to facilitate the filtration step.
3.2. Method Figures of Merit for Quantitation of ECD
Method validation was performed by evaluating the main performance parameters: linear dynamic range, linearity, LOD, LOQ, selectivity, repeatability, intermediate precision, and accuracy.
Manufacturers typically use herbal extracts in DSs, with nearly-exclusively solid forms on the market, mainly in capsule form. Thus, our validation samples were all solid (three capsules, one tablet).
3.2.1. Linear Dynamic Range, Linearity, LOD, LOQ
The linear dynamic range was selected within 1–50 µg·mL−1. Linear regression analysis was used to set up a calibration curve, including five points injected in triplicate. The calibration curves showed excellent linearity over the entire range. LOD and LOQ values were calculated according to the signal intensity of the ECD peak at the lowest calibration concentration and baseline noise of a blank sample. The results are presented in Table 2.

Table 2.
Parameters of linearity (n = 3), LOD and LOQ, repeatability, intermediate precision, and accuracy in terms of recovery (%) of quantitative analysis of ECD by means of LC-DAD.
3.2.2. Selectivity
An evaluation of selectivity was performed to prove that the target component can be separated from peaks from the solvent and matrix. Solvent blank, ECD solution, and samples from different matrices were analysed. The ECD peak (RT = 3.88 min) can be clearly distinguished from possible contaminants and components from the matrices.
3.2.3. Repeatability, Intermediate Precision
After determining the suitable dilution factors, samples were analysed (6-6 replicates for each material). In order to determine inter-day precision, the samples were reanalysed 1, 2, and 3 days after sample preparation, while the samples were kept at 4 °C between each measurement. Exact ECD contents were calculated using a freshly-prepared calibration. Calculated method precisions and inter-day precisions are summarised in Table 2. According to the obtained values, the developed LC-DAD method has excellent repeatability. The CV% in every case was lower than 5% (Table 2).
3.2.4. Accuracy
In different sample preparation sets, samples were spiked at three levels, namely, 80, 100, and 120% of ECD relative to their ECD content to perform a recovery study. The 100% levels were prepared in six replicates, and all others in duplicate. Acceptance criteria of recovery was determined in the range of 70–130%. The calculated recoveries (shown in Table 2) are satisfactory for each sample, but it is notable that one of them is close to the lowest acceptance limit. This is presumably due to its high herbal content, and thus it is recommended to perform a recovery study in case of unknown or complex matrices.
In conclusion, the performance parameters of the method completely fulfill the requirements that are related to quantitative analytical purposes.
3.3. Triple Quadrupole MS Analysis Optimisation
In order to achieve the best mass spectrometric identification of the target compounds, a detailed MRM transition optimisation was carried out during LC-MS/MS method development. The solutions of all 49 pure standards, including ECD and ISTD, were individually injected in full scan acquisition mode to detect the most intensive parent ions. In general, steroids and sterane structure compounds can be easily analysed using positive ionisation mode. In contrast, in the case of some SARMs, namely, ostarine and LGD4033, negative ionisation had to be used, likely due to their own specific trifluoromethyl functional group. Parent ions were then further fragmented in product ion scan mode using different collision energies (in the range of 5–40 eV) in order to select the appropriate CEs that provide the most intensive transitions. In some special cases, mainly when the product ion had low mass, higher CEs were also tested. The optimised MS parameters are summarised in Table 1. Taking advantage of the dynamic MRM mode, the most intensive fragment was not always selected as a parent ion, but the selectivity of the transitions was also considered. Two MS/MS transitions were primarily chosen: the higher one as a quantifier transition, while the one with a lower intensity was the confirmatory qualifier transition.
3.4. Method Figures of Merit for Limit Test of Prohibited Substances
The optimised method for detecting banned compounds from S1 and S4 classes in DSs was validated as a limit test. During validation measurements, samples not spiked and samples spiked at two concentration levels with the mixture containing all target compounds were analysed (10 replicates of each). Altogether, 11 considerably diverse DSs were examined in order to demonstrate the applicability for all types of DSs. Since all the investigated steroids are structurally quite similar, the appropriate selectivity of the method was one of the key points. As a result of the well-designed chromatographic method and the optimisation of the MRM transitions, all of the critical pairs (mainly isomers, compounds with similar RT, and/or same MRM transitions) could be distinguished with a selectivity factor (α) greater than 1.5. Reporting limits were determined for each target compound in accordance with the following acceptance criteria: ΔRT < 0.1 min, α > 1.5, signal to noise ratio (S/N) > 3, transition ion ratios between 70–130%. The determined RLs mainly varied between 0.6–10 ng·g−1 or mL−1, which is lower than previously determined in DSs. Overall, the obtained results clearly show the efficiency of the developed analytical method.
3.5. Analysis of ECD-Containing DSs
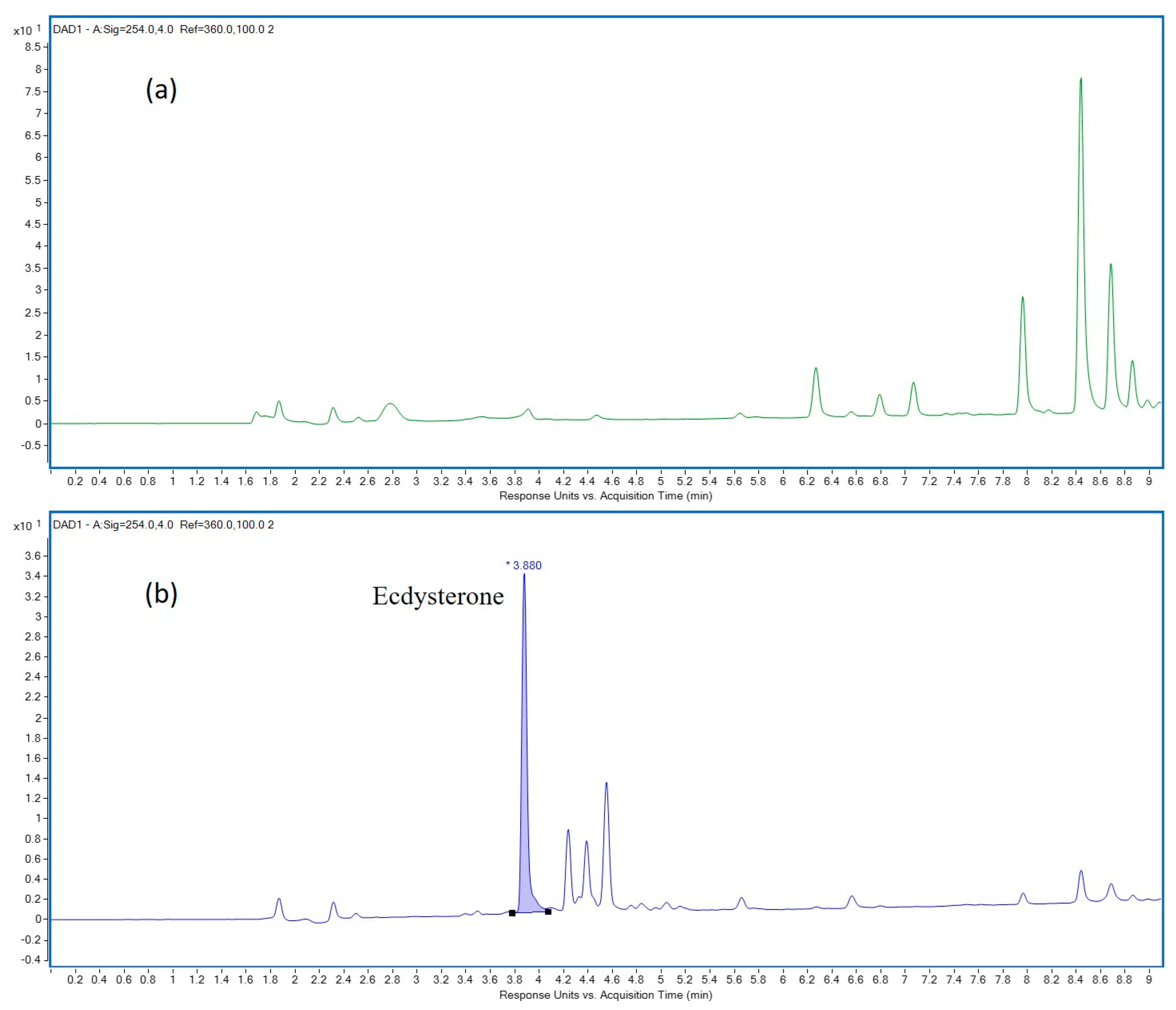
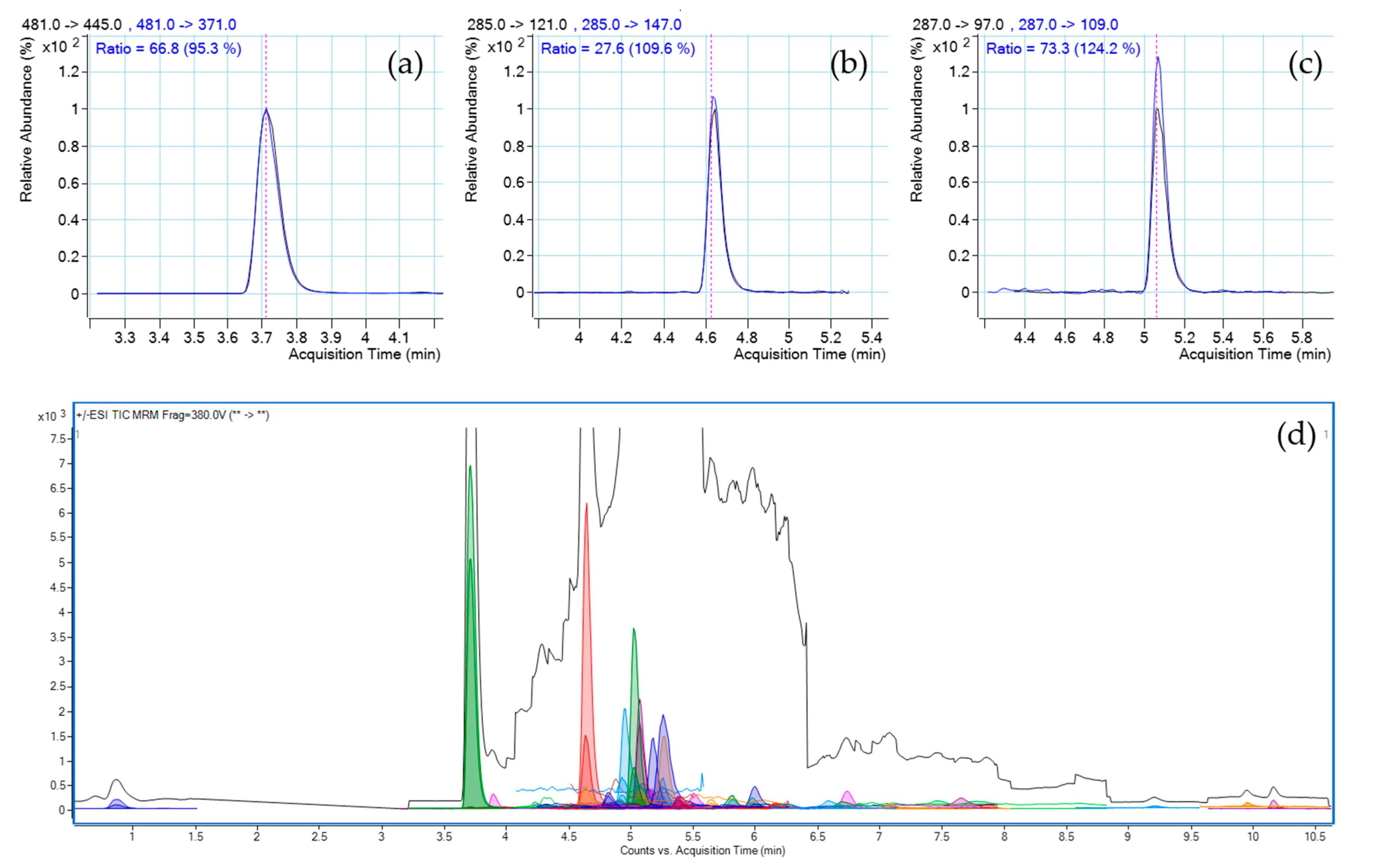
Our analytical methods were optimised according to two different scopes: exact quantitation of ECD with excellent linearity and accuracy on one hand, and limit test for prohibited AASs (S1), hormones and metabolic modulators (S4), with the lowest possible limit of detection, by means of optimised LC-MS/MS, on the other. In total, 21 different ECD-containing DSs from 10 brands were analysed. The results are shown in Table 3. In most cases, the measured ECD content was noticeably lower than as they were labeled. For one brand (E), the ECD content in both measured batches was roughly in accordance with those on the label. In most samples, the measured and labeled ECD content was not even in the same order of magnitude. Furthermore, the ECD content of different batches from the same brand differed extremely from each other. In the case of brand “A”, the coefficient of variation (CV%) of the measured ECD content was higher than 80%. In brand “I”, the target compound in one batch was not even detected, while the measured amount of ECD in the other one was barely more than half of its label (Figure 1). However, the ECD content was not defined on the label of brand “H”, while the same amount of ECD was found in the blue capsules of the two analysed batches. The highest amount of ECD in terms of mg/capsule was observed in brand “F”, precisely 34.1 mg/capsule. At the same time, 25% of the samples contained a prohibited substance from the WADA S1 class. In two batches of brand “A”, 4(5)-androstene-3,17-dione was present, while in the third one there was not any detectable prohibited substance. Unfortunately, even batches of brand “H” were identical considering the ECD content. In batch “2”, 1,4-androstene-3,17-dione was detected, while the other one was free of contamination. In the analysed batch of brand “J1” other than ECD (Figure 2a), 1,4-androstene-3,17-dione and 4(5)-androstene-3,17-dione were found (Figure 2b–d). No correlation between the source (product bought by the Laboratory or the Authority) and real composition of the product was found, including the concentration of the active compound and contaminations as well. Our findings were partially in accordance with previously-reported results [10].

Table 3.
Detailed list of analysed products, including name sample type, description, lot number, source, daily dose, labeled ECD content, and results. Each product was encoded with an uppercase letter from “A” to “J”. In addition, consecutive numbers were assigned to different batches. In the case of product “H”, there were daily packs, containing 4 different types of capsules and tablets.
Figure 1.
LC-DAD chromatograms of sample (a) “I1” and (b) “I2” detected at 254 nm. RT of ECD is 3.88 min.
Figure 2.
LC-MS/MS chromatograms of sample “J1”. (a–c) MRM transitions and ratios of ECD, 1,4-androstene-3,17-dione and 4(5)-androstene-3,17-dione, respectively, in sample blank. (d) TIC chromatogram and dMRM chromatogram of sample spiked at RL concentration of the mixture of 47 prohibited substances and ECD.
4. Conclusions
The work aimed to scrutinise ECD-containing dietary supplements, which are widely used by both professional and amateur athletes. The reason for the great popularity of this substance is that ECD is still legally available, although included in the Monitoring Program. Hence, two analytical methods were optimised according to two different scopes. For the quantitation of ECD by means of LC-DAD, a simple and efficient sample preparation was developed, with significantly lower LOD and shorter runtime, as previously reported with the same technique [27]. The limit test for thoroughly selected 47 WADA-prohibited steroids (S1), hormones, and metabolic modulators (S4) by means of LC-MS/MS was validated with cumbersome dietary supplements, allowing lower reporting limits than previously published in DSs, which is of great importance considering the zero tolerance of these banned substances in human samples during doping control analysis. Serious discrepancies were explored on the labelling of supplements containing ECD, including the lack of active compounds and the presence of prohibited substances. The developed methods provide outstanding support for athletes regarding food-supplement safety.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/separations10040242/s1, Table S1: A detailed list of reference standards used, including compound name, WADA class, supplier, type, purity, and the applied method for detection.
Author Contributions
Conceptualisation, M.U.; methodology, Z.P.; validation, Z.P.; investigation, Z.P. and D.B.; data curation, Z.P. and D.B.; writing—original draft preparation, Z.P.; writing—review and editing, M.U.; visualisation, D.B.; supervision, M.U. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Data Availability Statement
Not applicable.
Acknowledgments
The authors thank the “State Secretariat for Sport” for the financial support.
Conflicts of Interest
The authors declare no conflict of interest.
References
- The World Anti-Doping Agency. The 2020 Monitoring Program. Available online: https://www.wada-ama.org/sites/default/files/resources/files/wada_2020_english_monitoring_program_.pdf (accessed on 9 January 2023).
- Kwiatkowska, D.; Grucza, K.; Chajewska, K.; Konarski, P.; Wojtkowiak, K.; Drapała, A.; Wicka, M. Ecdysterone: Possible sources of origin in urine. Drug Test Anal. 2022, 36480213. [Google Scholar] [CrossRef]
- Hunyadi, A.; Herke, I.; Lengyel, K.; Báthori, M.; Kele, Z.; Simon, A.; Tóth, G.; Szendrei, K. Ecdysteroid-containing food supplements from Cyanotis arachnoidea on the European market: Evidence for spinach product counterfeiting. Sci. Rep. 2016, 6, 37322. [Google Scholar] [CrossRef] [PubMed]
- Todorova, V.; Ivanov, K.; Ivanova, S. Comparison between the biological active compounds in plants with adaptogenic properties (Rhaponticum carthamoides, Lepidium meyenii, Eleutherococcus senticosus and Panax ginseng). Plants 2022, 11, 64. [Google Scholar] [CrossRef] [PubMed]
- Dinan, L.; Lafont, R. Effects and applications of arthropod steroid hormones (ecdysteroids) in mammals. J. Endocrinol. 2006, 191, 1–8. [Google Scholar] [CrossRef]
- Haupt, O.; Ngueu, S.T.; Diel, P.; Parr, M.K. Anabolic Effect of Ecdysterone Results in Hypertrophy of C2C12 Myotubes by an Estrogen Receptor Mediated Pathway. In Recent Advances in Dope Analysis, Proceedings of the Manfred-Donike-Workshop; 30th Cologne Workshop on Dope Analysis, 26th February to 2nd March 2012; Schänzer, W., Geyer, H., Gotzman, A., Mareck, U., Eds.; Sport und Buch Strauß: Cologne, Germany, 2012; pp. 221–225. [Google Scholar]
- Parr, M.K.; Botrè, F.; Naß, A.; Hengevoss, J.; Diel, P.; Wolber, G. Ecdysteroids: A novel class of anabolic agents? Biol. Sport 2015, 32, 169–173. [Google Scholar] [CrossRef]
- Isenmann, E.; Ambrosio, G.; Joseph, J.F.; Mazzarino, M.; Torre, X.; Zimmer, P.; Kazlauskas, R.; Goebel, C.; Botrè, F.; Diel, P.; et al. Ecdysteroids as non-conventional anabolic agent: Performance enhancement by ecdysterone supplementation in humans. Arch. Toxicol. 2019, 93, 1807–1816. [Google Scholar] [CrossRef] [PubMed]
- Wilborn, C.D.; Taylor, L.W.; Campbell, B.I.; Kerksick, C.; Rasmussen, C.J.; Greenwood, M.; Kreider, R.B. Effects of methoxyisoflavone, ecdysterone, and sulfo-polysaccharide supplementation on training adaptations in resistance-trained males. J. Int. Soc. Sports Nutr. 2006, 3, 19–27. [Google Scholar] [CrossRef]
- Ambrosio, G.; Wirth, D.; Joseph, J.F.; Mazzarino, M.; Torre, X.; Botrè, F.; Parr, M.K. How reliable is dietary supplement labelling?—Experiences from the analysis of ecdysterone supplements. J. Pharm. Biomed Anal. 2020, 177, 112877. [Google Scholar] [CrossRef] [PubMed]
- Garthe, I.; Maughan, R.J. Athletes and supplements: Prevalence and perspectives. Int. J. Sport Nutr. Exerc. Metab. 2018, 28, 126–138. [Google Scholar] [CrossRef]
- Kozhuharov, V.R.; Ivanov, K.; Ivanova, S. Dietary supplements as source of unintentional doping. Biomed Res. Int. 2022, 2022, 8387271. [Google Scholar] [CrossRef]
- Lauritzen, F. Dietary supplements as a major cause of anti-doping rule violations. Front. Sports Act. Living 2022, 4, 868228. [Google Scholar] [CrossRef] [PubMed]
- Geyer, H.; Parr, M.K.; Koehler, K.; Mareck, U.; Schänzer, W.; Thevis, M. Nutritional supplements cross-contaminated and faked with doping substances. J. Mass. Spectrom. 2008, 43, 892–902. [Google Scholar] [CrossRef] [PubMed]
- Walpurgis, K.; Thomas, A.; Geyer, H.; Mareck, U.; Thevis, M. Dietary supplement and food contaminations and their implications for doping controls. Foods 2020, 9, 1012. [Google Scholar] [CrossRef] [PubMed]
- Pellegrini, M.; Rotolo, M.C.; Di Giovannadrea, R.; Pacifici, R.; Pichini, S. A simple toxicological analysis of anabolic steroid preparations from the black market. Ann. Toxicol. Anal. 2012, 24, 67–72. [Google Scholar] [CrossRef]
- Neves, D.B.D.J.; Calda, E.D. GC-MS quantitative analysis of black market pharmaceutical products containing anabolic androgenic steroids seized by the Brazilian Federal Police. Forensic. Sci. Int. 2017, 275, 272–281. [Google Scholar] [CrossRef]
- De Cock, K.J.S.; Delbeke, F.T.; Van Eenoo, P.; Desmet, N.; Roels, K.; De Backer, P. Detection and determination of anabolic steroids in nutritional supplements. J. Pharm. Biomed. Anal. 2001, 25, 843–852. [Google Scholar] [CrossRef]
- Micalizzi, G.; Huszti, K.; Pálinkás, Z.; Mandolfino, F.; Martos, É.; Dugo, P.; Mondello, L.; Utczás, M. Reliable identification and quantification of anabolic androgenic steroids in dietary supplements by using gas chromatography coupled to triple quadrupole mass spectrometry. Drug Test. Anal. 2021, 13, 128–139. [Google Scholar] [CrossRef]
- Kozlik, P.; Tircova, B. Development of the fast, simple and fully validated high performance liquid chromatographic method with diode array detector for quantification of testosterone esters in an oil-based injectable dosage form. Steroids 2016, 115, 34–39. [Google Scholar] [CrossRef]
- Van Poucke, C.; Detavernier, C.; Van Cauwenberghe, R.; Van Peteghem, C. Determination of anabolic steroids in dietary supplements by liquid chromatography–tandem mass spectrometry. Anal. Chim. Acta. 2017, 586, 35–42. [Google Scholar] [CrossRef]
- Leaney, A.L.; Beck, P.; Biddle, S.; Brown, P.; Grace, P.B.; Hudson, C.S.; Mawson, H.D. Analysis of supplements available to UK consumers purporting to contain selective androgen receptor. Drug Test Anal. 2021, 13, 122–127. [Google Scholar] [CrossRef]
- Tircova, B.; Bosakova, Z.; Kozlik, P. Development of an UHPLC-MS/MS method for the determination of anabolic steroids currently available on the black market in the Czech Republic and Slovakia. Drug Test Anal. 2018, 11, 355–360. [Google Scholar] [CrossRef]
- Parr, M.K.; Ambrosio, G.; Wuest, B.; Mazzarino, M.; Torre, X.; Sibilia, F.; Joseph, J.F.; Diel, P.; Botrè, F. Targeting the administration of ecdysterone in doping control samples. Forensic. Toxicol. 2020, 38, 172–184. [Google Scholar] [CrossRef]
- Grucza, K.; Wicka, M.; Drapała, A.; Kwiatkowska, D. Determination of Ecdysterone in Dietary Supplements and Spinach by Ultra-High-Performance Liquid Chromatography-Tandem Mass Spectrometry. Separations 2022, 9, 8. [Google Scholar] [CrossRef]
- Dinan, L.; Guibout, B.L.; Lafont, R. Small-scale analysis of phytoecdysteroids in seeds by HPLC-DAD-MS for the identification and quantification of specific analogues, dereplication and chemotaxonomy. Phytochem. Anal. 2020, 31, 643–661. [Google Scholar] [CrossRef] [PubMed]
- Napierała, M.; Nawrot, J.; Gornowicz-Porowska, J.; Florek, E.; Moroch, A.; Adamski, Z.; Kroma, A.; Miechowicz, I.; Nowak, G. Separation and HPLC Characterization of Active Natural Steroids in a Standardized Extract from the Serratula coronata Herb with Antiseborrheic Dermatitis Activity. Int. J. Environ. Res. Public Health 2020, 17, 6453. [Google Scholar] [CrossRef] [PubMed]
- Magnusson, B.; Örnemark, U. Eurachem Guide: The Fitness for Purpose of Analytical Method A Laboratory Guide to Method Validation and Related Topics. 2nd ed. Available online: https://www.eurachem.org/images/stories/Guides/pdf/MV_guide_2nd_ed_EN.pdf/ (accessed on 13 December 2022).
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).