Infrared Radiation in the Management of Musculoskeletal Conditions and Chronic Pain: A Systematic Review
Abstract
:1. Introduction
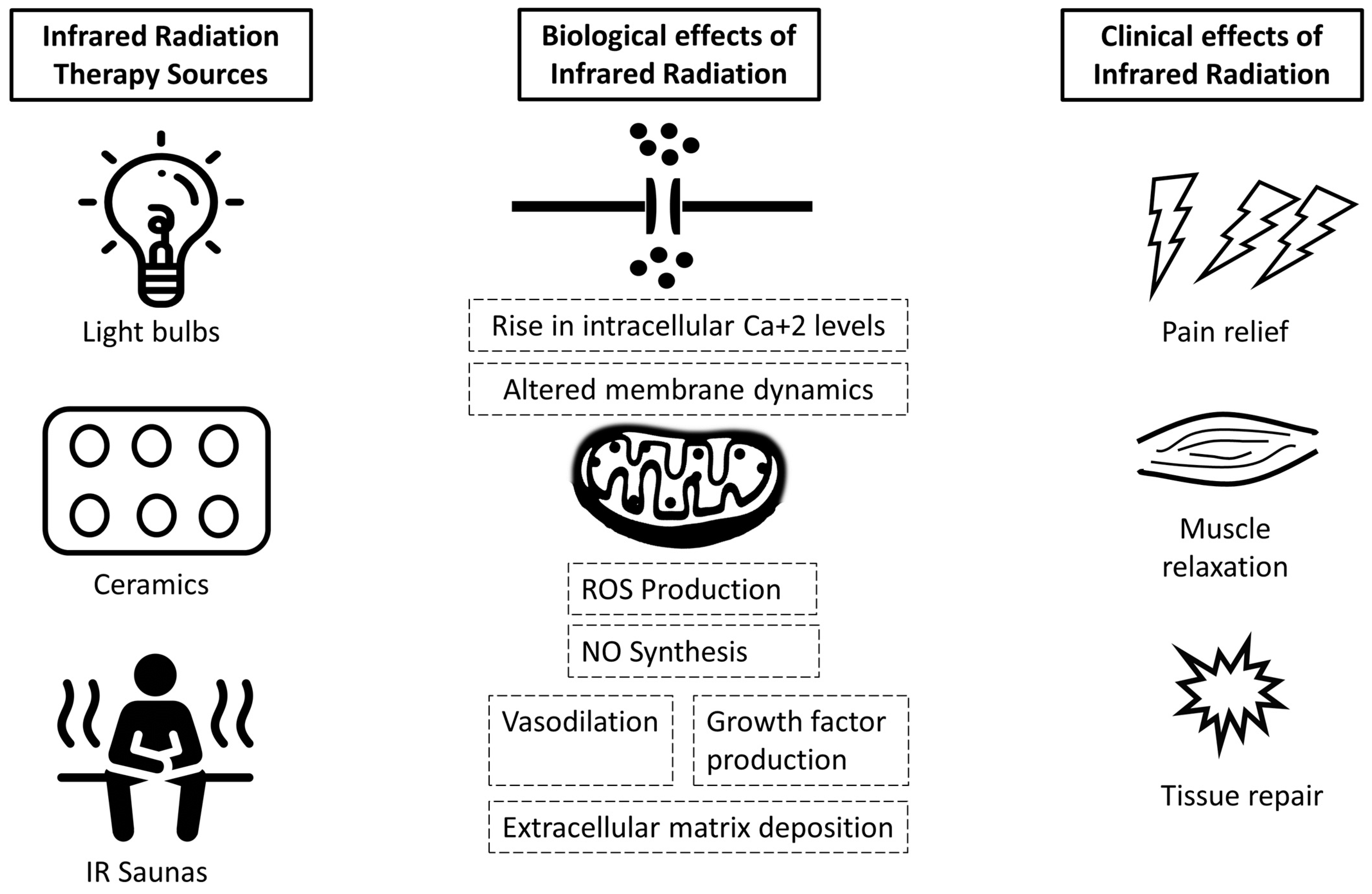
1.1. Production and Provision of IR in Clinical Settings
1.2. Effects of Infrared Light on Health
2. Materials and Methods
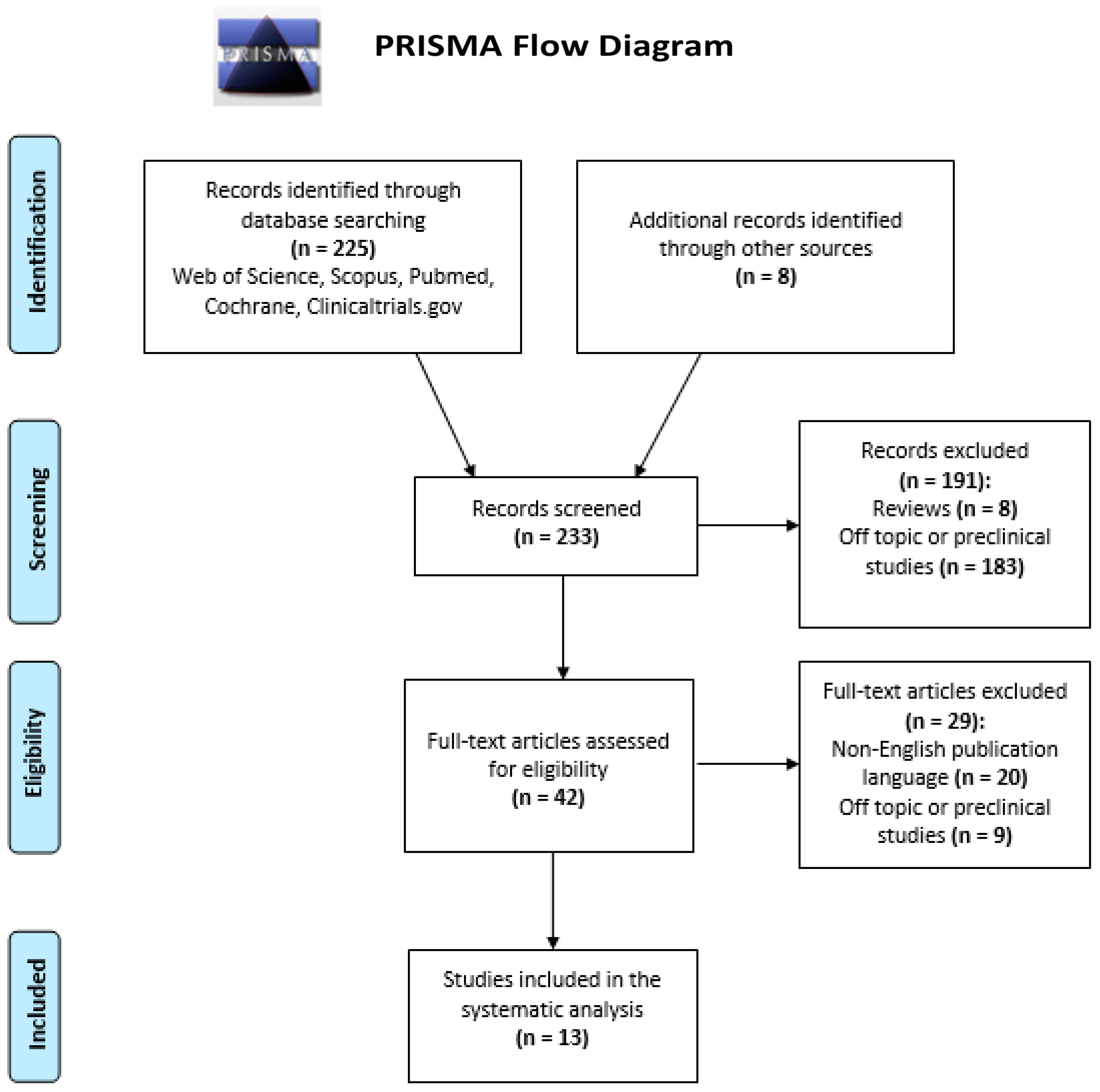
2.1. Literature Search
2.2. Data Synthesis and Analysis
3. Results
3.1. Qualitative Outcomes
3.1.1. Osteoarthritis
3.1.2. Fibromyalgia
3.1.3. Other Conditions
4. Discussion
4.1. Limitations
4.2. Future Research
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Tsagkaris, C.; Corriero, A.C.; Moysidis, D.V.; Loudovikou, A.; Papazoglou, A.S. Knowing When Not to Operate: Following the Footsteps of Demokidis of Crotone in Orthopaedics. Surg. Innov. 2021, 13, 15533506211041903. [Google Scholar] [CrossRef] [PubMed]
- NASA, Infrared Light. 2018. Available online: https://www.nasa.gov/audience/forstudents/5-8/features/F_Infrared_Light_5-8.html (accessed on 1 October 2021).
- Robertson, V.; Ward, A.; Low, J.; Reed, A. Infrared and visible radiations. In Electrotherapy Explained Principles and Practice, 2nd ed.; John Low & Ann Reed: Oxford, UK, 1994. [Google Scholar]
- Vatansever, F.; Hamblin, M.R. Far Infrared Radiation (FIR): Its Biological Effects and Medical Applications. Photonics Lasers Med. 2012, 4, 255–266. [Google Scholar] [CrossRef] [Green Version]
- Meng, J.; Jin, W.; Liang, J.; Ding, Y.; Gan, K.; Yuan, Y. Effects of Particle Size on Far Infrared Emission Properties of Tourmaline Superfine Powders. J. Nanosci. Nanotechnol. 2010, 10, 2083–2087. [Google Scholar] [CrossRef] [PubMed]
- Bundesamt fur Strahlenschutz [The German Federal Office for Radiation Protection], Applications of Infrared Radiation. 2020. Available online: https://www.bfs.de/EN/topics/opt/application-medicine-wellness/infrared/infrared_node.html (accessed on 16 October 2021).
- Lee, M.-S.; Baletto, F.; Kanhere, D.G.; Scandolo, S. Far-Infrared Absorption of Water Clusters by First-Principles Molecular Dynamics. J. Chem. Phys. 2008, 128, 214506. [Google Scholar] [CrossRef] [PubMed]
- Chung, H.; Dai, T.; Sharma, S.K.; Huang, Y.-Y.; Carroll, J.D.; Hamblin, M.R. The Nuts and Bolts of Low-Level Laser (Light) Therapy. Ann. Biomed. Eng. 2012, 40, 516–533. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Golovynska, I.; Golovynskyi, S.; Stepanov, Y.V.; Stepanova, L.I.; Qu, J.; Ohulchanskyy, T.Y. Red and near-infrared light evokes Ca2+ influx, endoplasmic reticulum release and membrane depolarization in neurons and cancer cells. J. Photochem. Photobiol. B 2021, 214, 112088. [Google Scholar] [CrossRef] [PubMed]
- Hamblin, M.R. Mechanisms and Mitochondrial Redox Signaling in Photobiomodulation. Photochem. Photobiol. 2018, 94, 199–212. [Google Scholar] [CrossRef] [Green Version]
- Lee, D.; Seo, Y.; Kim, Y.W.; Kim, S.; Bae, H.; Choi, J.; Lim, I.; Bang, H.; Kim, J.H.; Ko, J.H. Far-infrared radiation stimulates platelet-derived growth factor mediated skeletal muscle cell migration through extracellular matrix-integrin signaling. Korean J. Physiol. Pharmacol. 2019, 23, 141–150. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xu, J.; Deng, Y.; Yu, C.-Y.; Gao, Z.-M.; Yang, X.-R.; Zhang, Q.; Zhang, L. Efficacy of WIRA in the Treatment of Sacroiliitis in Male Patients with Ankylosing Spondylitis and Its Effect on Serum VEGF Levels. J. Orthop. Surg. Res. 2019, 14, 313. [Google Scholar] [CrossRef] [PubMed]
- Hsieh, R.-L.; Lo, M.-T.; Lee, W.-C.; Liao, W.-C. Therapeutic Effects of Short-Term Monochromatic Infrared Energy Therapy on Patients with Knee Osteoarthritis: A Double-Blind, Randomized, Placebo-Controlled Study. J. Orthop. Sports Phys. Ther. 2012, 42, 947–956. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bagnato, G.L.; Miceli, G.; Atteritano, M.; Marino, N.; Bagnato, G.F. Far Infrared Emitting Plaster in Knee Osteoarthritis: A Single Blinded, Randomised Clinical Trial. Reumatismo 2012, 64, 388–394. [Google Scholar] [CrossRef] [Green Version]
- Brockow, T.; Wagner, A.; Franke, A.; Offenbächer, M.; Resch, K.L. A Randomized Controlled Trial on the Effectiveness of Mild Water-Filtered near Infrared Whole-Body Hyperthermia as an Adjunct to a Standard Multimodal Rehabilitation in the Treatment of Fibromyalgia. Clin. J. Pain 2007, 23, 67–75. [Google Scholar] [CrossRef] [PubMed]
- Salm, D.C.; Belmonte, L.A.O.; Emer, A.A.; Leonel, L.D.S.; de Brito, R.N.; da Rocha, C.C.; Martins, T.C.; Dos Reis, D.C.; Moro, A.R.P.; Mazzardo-Martins, L.; et al. Aquatic Exercise and Far Infrared (FIR) Modulates Pain and Blood Cytokines in Fibromyalgia Patients: A Double-Blind, Randomized, Placebo-Controlled Pilot Study. J. Neuroimmunol. 2019, 337, 577077. [Google Scholar] [CrossRef]
- Aparecida Santos e Campos, M.; García Pinillos, F.; Latorre Román, P.Á. Reduction in Pain after Use of Bioceramic Undershirt for Patients with Fibromyalgia. Altern. Ther. Health Med. 2017, 23, 18–22. [Google Scholar]
- Matsushita, K.; Masuda, A.; Tei, C. Efficacy of Waon Therapy for Fibromyalgia. Intern. Med. 2008, 47, 1473–1476. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nunes, R.F.H.; Cidral-Filho, F.J.; Flores, L.J.F.; Nakamura, F.Y.; Rodriguez, H.F.M.; Bobinski, F.; De Sousa, A.; Petronilho, F.; Danielski, L.G.; Martins, M.M.; et al. Effects of Far-Infrared Emitting Ceramic Materials on Recovery during 2-Week Preseason of Elite Futsal Players. J. Strength Cond. Res. 2020, 34, 235–248. [Google Scholar] [CrossRef]
- Soejima, Y.; Munemoto, T.; Masuda, A.; Uwatoko, Y.; Miyata, M.; Tei, C. Effects of Waon Therapy on Chronic Fatigue Syndrome: A Pilot Study. Intern. Med. 2015, 54, 333–338. [Google Scholar] [CrossRef] [Green Version]
- Lai, Y.-T.; Chan, H.-L.; Lin, S.-H.; Lin, C.-C.; Li, S.-Y.; Liu, C.-K.; Teng, H.-W.; Liu, W.-S. Far-Infrared Ray Patches Relieve Pain and Improve Skin Sensitivity in Myofascial Pain Syndrome: A Double-Blind Randomized Controlled Study. Complement. Ther. Med. 2017, 35, 127–132. [Google Scholar] [CrossRef] [PubMed]
- Ammar, T.A.R.A. Monochromatic Infrared Photo Energy versus Low Level Laser Therapy in Chronic Low Back Pain. J. Lasers Med. Sci. 2015, 6, 157–161. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chao, L.L. Improvements in Gulf War Illness Symptoms after Near-Infrared Transcranial and Intranasal Photobiomodulation: Two Case Reports. Mil. Med. 2019, 184, e568–e574. [Google Scholar] [CrossRef] [PubMed]
- Lin, L.; Cheng, K.; Tan, M.T.; Zhao, L.; Huang, Z.; Yao, C.; Wu, F.; Zhang, H.; Shen, X. Comparison of the Effects of 10.6-Μm Infrared Laser and Traditional Moxibustion in the Treatment of Knee Osteoarthritis. Lasers Med. Sci. 2020, 35, 823–832. [Google Scholar] [CrossRef] [Green Version]
- Tricco, A.C.; Lillie, E.; Zarin, W.; O’Brien, K.K.; Colquhoun, H.; Levac, D.; Moher, D.; Peters, M.D.J.; Horsley, T.; Weeks, L.; et al. PRISMA Extension for Scoping Reviews (PRISMA-ScR): Checklist and Explanation. Ann. Intern. Med. 2018, 169, 467–473. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G.; The PRISMA Group. Preferred Reporting Items for Systematic Reviews and Meta-Analyses: The PRISMA Statement. PLoS Med. 2009, 6, e1000097. [Google Scholar] [CrossRef] [Green Version]
- De Souza Guimarães, L.; Costa, L.d.C.M.; Araujo, A.C.; Nascimento, D.P.; Medeiros, F.C.; Avanzi, M.A.; Leal-Junior, E.C.P.; Costa, L.O.P.; Tomazoni, S.S. Photobiomodulation Therapy Is Not Better than Placebo in Patients with Chronic Nonspecific Low Back Pain: A Randomised Placebo-Controlled Trial: A Randomised Placebo-Controlled Trial. Pain 2021, 162, 1612–1620. [Google Scholar] [CrossRef] [PubMed]
- Bontemps, B.; Gruet, M.; Vercruyssen, F.; Louis, J. Utilisation of Far Infrared-Emitting Garments for Optimising Performance and Recovery in Sport: Real Potential or New Fad? A Systematic Review. PLoS ONE 2021, 16, e0251282. [Google Scholar] [CrossRef]
- Foley, J.; Vasily, D.B.; Bradle, J.; Rudio, C.; Calderhead, R.G. 830 Nm Light-Emitting Diode (Led) Phototherapy Significantly Reduced Return-to-Play in Injured University Athletes: A Pilot Study. Laser Ther. 2016, 25, 35–42. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tsai, S.-R.; Hamblin, M.R. Biological Effects and Medical Applications of Infrared Radiation. J. Photochem. Photobiol. B 2017, 170, 197–207. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wickenheisser, V.A.; Zywot, E.M.; Rabjohns, E.M.; Lee, H.H.; Lawrence, D.S.; Tarrant, T.K. Laser Light Therapy in Inflammatory, Musculoskeletal, and Autoimmune Disease. Curr. Allergy Asthma Rep. 2019, 19, 37. [Google Scholar] [CrossRef] [PubMed]
- Barros, N.D.M.; Sbroglio, L.L.; Buffara, M.D.O.; Baka, J.L.C.E.S.; Pessoa, A.D.S.; Azulay-Abulafia, L. Phototherapy. An. Bras. Dermatol. 2021, 96, 397–407. [Google Scholar] [CrossRef]
- Wang, Y.; Shui, S.; Wang, X. Molecular Mechanism of Far-infrared Therapy and Its Applications in Biomedicine. Sci. Technol. Rev. 2014, 32, 80–84. [Google Scholar] [CrossRef]
- Guessogo, W.R.; Assomo-Ndemba, P.B.; Ebal-Minye, E.; Mekoulou-Ndongo, J.; Bika-Lélé, C.B.; Mbang-Bian, W.; Djuine-Soh, E.L.; Ondoa, J.B.; Mandengue, S.H.; Temfemo, A. Effect of Schoolbag Weight on Musculoskeletal Pain among Primary School Children in Yaounde, Cameroon: A Cross-sectional Study. Int. J. Med. Stud. 2020, 8, 96–101. [Google Scholar] [CrossRef]
- Dorji, T.; Tamang, S.T.; Yoezer, S.; Wangdi, K. The Weight of Schoolbags and Musculoskeletal Pain in Children of Selected Schools in Thimphu, Bhutan: A Cross-sectional Study. Int. J. Med. Stud. 2019, 7, 29–32. [Google Scholar] [CrossRef]
| Disease | Study Type | Country | Treatment | Population (Intervention/Control) | Outcome Measurements | Key Outcomes | Reference |
|---|---|---|---|---|---|---|---|
| AS | RCT | China | wIRA | 120 (60/60) | BASDAI, pain VAS, morning stiffness VAS, RI | ↓ BASDAI ↓ pain VAS (↓ 17.5%) ↓ morning stiffness VAS ↑ RI | Xu/2019 [12] |
| Knee OA | RCT | Taiwan | MIRE Λ = 890 nm | 73 (38/38) | KOOS, LKS, HADS, OAQoL | OAQoL ↓ 14.3% | Hsieh/2012 [13] |
| RCT | Italy | FIR emitting plaster | 60 (30/30) | pain VAS | ↓ pain VAS 1 week: ↓12.5% 4 weeks: ↓25% | Bagnato/2012 [14] | |
| RCT pooled data | China | 10.6 μm infrared laser vs. traditional moxibustion | 143 (55/88) | WOMAC SF-36 | pain VAS: ↓ 60.9% no significant differences | Lin/2020 [24] | |
| FM | RCT | Germany | mild water-filtered NI-WBH | 139 (69/70) | MPQ, FIQ | VAS: ↓ 37.5% FIQ: ↓ 40.4% | Brockow/2007 [15] |
| RCT | Brazil | AE, FIR | 28 (14/14) | pain VAS SF-MPQ FIQ | VAS: ↓ 41.4% FIQ: ↓ 14.7% | Salm/2019 [16] | |
| RCT | Spain | fabric coated with bio-ceramics | 39 (20/19) | pain VAS FIQ SF-12 | VAS: ↓21.76% FIQ: ↓16.46% | Campos/2017 [17] | |
| OS | Japan | Waon therapy | 13 | pain VAS FIQ | VAS: ↓52.7% FIQ: ↓34.6% | Matsushita/2008 [18] | |
| Injuries (athletes) | RCT | Brazil | cFIR | 20 (10/10) | performance biochemical markers delayed-onset muscle soreness training strain | ↑ recovery | Nunes/2020 [19] |
| OS | Japan | Waon therapy | 11 | PSS | ↓ perceived fatigue ↑ performance status PSS: ↓19.23% | Soejima/2015 [20] | |
| MPS | RCT | Taiwan | FIR patches | 125 (61/57) | pain VAS pressure pain threshold maximal pain tolerance | pain VAS: ↓8.2% ↓ pressure pain threshold ↓ maximal pain tolerance | Lai/2017 [21] |
| CLBP | RCT | Egypt | MIPE vs. LLLT | 70 (35/35) | FRI pain VAS modified Schober test | MIPE: 36% improvement LLLT: 27.87% improvement | Ammar/2015 [22] |
| Gulf War Illness | Case series | USA | NTIP | 2 | KGWMHHQ ISI BPI | pain, sleep, insomnia improvements | Chao/2019 [23] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tsagkaris, C.; Papazoglou, A.S.; Eleftheriades, A.; Tsakopoulos, S.; Alexiou, A.; Găman, M.-A.; Moysidis, D.V. Infrared Radiation in the Management of Musculoskeletal Conditions and Chronic Pain: A Systematic Review. Eur. J. Investig. Health Psychol. Educ. 2022, 12, 334-343. https://doi.org/10.3390/ejihpe12030024
Tsagkaris C, Papazoglou AS, Eleftheriades A, Tsakopoulos S, Alexiou A, Găman M-A, Moysidis DV. Infrared Radiation in the Management of Musculoskeletal Conditions and Chronic Pain: A Systematic Review. European Journal of Investigation in Health, Psychology and Education. 2022; 12(3):334-343. https://doi.org/10.3390/ejihpe12030024
Chicago/Turabian StyleTsagkaris, Christos, Andreas S. Papazoglou, Anna Eleftheriades, Stavros Tsakopoulos, Athanasios Alexiou, Mihnea-Alexandru Găman, and Dimitrios V. Moysidis. 2022. "Infrared Radiation in the Management of Musculoskeletal Conditions and Chronic Pain: A Systematic Review" European Journal of Investigation in Health, Psychology and Education 12, no. 3: 334-343. https://doi.org/10.3390/ejihpe12030024
APA StyleTsagkaris, C., Papazoglou, A. S., Eleftheriades, A., Tsakopoulos, S., Alexiou, A., Găman, M.-A., & Moysidis, D. V. (2022). Infrared Radiation in the Management of Musculoskeletal Conditions and Chronic Pain: A Systematic Review. European Journal of Investigation in Health, Psychology and Education, 12(3), 334-343. https://doi.org/10.3390/ejihpe12030024









