Anticancer Properties of Strobilanthes crispus: A Review
Abstract
1. Introduction
2. Anticancer Properties of Strobilanthes crispus against Various Cancers
2.1. Breast Cancer
2.1.1. In Vitro Studies
2.1.2. In Vivo Studies
2.2. Liver Cancer
2.2.1. In Vitro Studies
2.2.2. In Vivo Studies
2.3. Colon Cancer
2.3.1. In Vitro Studies
2.3.2. In Vivo Studies
2.4. Prostate Cancer
In Vitro Studies
2.5. Cervical Cancer
In Vitro Studies
2.6. Nasopharyngeal Cancer
In Vitro Studies
2.7. Lung Cancer
In Vitro Studies
3. Toxicology Studies
3.1. In Vitro Studies
3.2. In Vivo Studies
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Abbas, Z.; Rehman, S. An Overview of Cancer Treatment Modalities. In Neoplasm; InTech: London, UK, 2018. [Google Scholar]
- Hanahan, D.; Weinberg, R.A. Hallmarks of Cancer: The Next Generation. Cell 2011, 144, 646–674. [Google Scholar] [CrossRef]
- World Health Organization. Available online: https://www.who.int/health-topics/cancer (accessed on 17 July 2020).
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA. Cancer J. Clin. 2021, 71, 209–2491. [Google Scholar] [CrossRef]
- Lasley, I. What Cancer Treatments May Be Used. In 21st Century Cancer Treatment; CreateSpace Independent Publishing Platform: Scotts Valley, CA, USA, 2010; pp. 16–55. [Google Scholar]
- Centers for Disease Control and Prevention. Available online: https://www.cdc.gov/cancer/survivors/patients/side-effects-of-treatment.htm (accessed on 17 July 2020).
- National Cancer Institute. Available online: https://www.cancer.gov/about-cancer/treatment/side-effects (accessed on 17 July 2020).
- Vickers, A. Herbal medicine. West. J. Med. 2001, 175, 125–128. [Google Scholar] [CrossRef]
- Falzon, C.C.; Balabanova, A. Phytotherapy. Prim. Care Clin. Off. Pract. 2017, 44, 217–227. [Google Scholar] [CrossRef]
- Ghosh, D. Seed to Patient in Clinically Proven Natural Medicines**Partly adapted from Zangara and Ghosh (2014), with permission from CCR Press. In Nutraceuticals; Elsevier: Amsterdam, The Netherlands, 2016; pp. 925–931. [Google Scholar]
- Zhan, W.; Liu, Y.; Li, D.; Liu, Y. Advancing insights on the anti-obesity biochemical mechanism of (−)-epigallocatechin gallate (EGCG) by inhibiting α-amylase activity. RSC Adv. 2016, 6, 96918–96927. [Google Scholar] [CrossRef]
- Lee, S.; Al Razqan, G.S.; Kwon, D.H. Antibacterial activity of epigallocatechin-3-gallate (EGCG) and its synergism with β-lactam antibiotics sensitizing carbapenem-associated multidrug resistant clinical isolates of Acinetobacter baumannii. Phytomedicine 2017, 24, 49–55. [Google Scholar] [CrossRef]
- Zhu, C.; Xu, Y.; Liu, Z.-H.; Wan, X.-C.; Li, D.-X.; Tai, L.-L. The anti-hyperuricemic effect of epigallocatechin-3-gallate (EGCG) on hyperuricemic mice. Biomed. Pharmacother. 2018, 97, 168–173. [Google Scholar] [CrossRef]
- Steinmann, J.; Buer, J.; Pietschmann, T.; Steinmann, E. Anti-infective properties of epigallocatechin-3-gallate (EGCG), a component of green tea. Br. J. Pharmacol. 2013, 168, 1059–1073. [Google Scholar] [CrossRef]
- Braicu, C.; Irimie, A.I.; Zanoaga, O.; Gherman, C.; Berindan-Neagoe, I.; Campian, R.S.; Pileczki, V. Epigallocatechin-3-gallate suppresses cell proliferation and promotes apoptosis and autophagy in oral cancer SSC-4 cells. Onco. Targets. Ther. 2015, 461, 461–470. [Google Scholar] [CrossRef][Green Version]
- Liu, Y.; Wang, X.; Zeng, S.; Zhang, X.; Zhao, J.; Zhang, X.; Chen, X.; Yang, W.; Yang, Y.; Dong, Z.; et al. The natural polyphenol curcumin induces apoptosis by suppressing STAT3 signaling in esophageal squamous cell carcinoma. J. Exp. Clin. Cancer Res. 2018, 37, 303. [Google Scholar] [CrossRef] [PubMed]
- Liu, D.; You, M.; Xu, Y.; Li, F.; Zhang, D.; Li, X.; Hou, Y. Inhibition of curcumin on myeloid-derived suppressor cells is requisite for controlling lung cancer. Int. Immunopharmacol. 2016, 39, 265–272. [Google Scholar] [CrossRef]
- Samarghandian, S.; Azimi-Nezhad, M.; Farkhondeh, T.; Samini, F. Anti-oxidative effects of curcumin on immobilization-induced oxidative stress in rat brain, liver and kidney. Biomed. Pharmacother. 2017, 87, 223–229. [Google Scholar] [CrossRef] [PubMed]
- Hudson, J.B. Applications of the Phytomedicine Echinacea purpurea (Purple Coneflower) in Infectious Diseases. J. Biomed. Biotechnol. 2012, 2012, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Sharma, S.M.; Anderson, M.; Schoop, S.R.; Hudson, J.B. Bactericidal and anti-inflammatory properties of a standardized Echinacea extract (Echinaforce®): Dual actions against respiratory bacteria. Phytomedicine 2010, 17, 563–568. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Chen, W. Gambogic Acid is a Novel Anti-cancer Agent that Inhibits Cell Proliferation, Angiogenesis and Metastasis. Anticancer Agents Med. Chem. 2012, 12, 994–1000. [Google Scholar] [CrossRef]
- Cascão, R.; Vidal, B.; Raquel, H.; Neves-Costa, A.; Figueiredo, N.; Gupta, V.; Fonseca, J.E.; Ferreira Moita, L. Potent Anti-Inflammatory and Antiproliferative Effects of Gambogic Acid in a Rat Model of Antigen-Induced Arthritis. Mediators Inflamm. 2014, 2014, 1–7. [Google Scholar] [CrossRef]
- Liu, L.; Qi, X.-J.; Zhong, Z.-K.; Zhang, E.-N. Nanomedicine-based combination of gambogic acid and retinoic acid chlorochalcone for enhanced anticancer efficacy in osteosarcoma. Biomed. Pharmacother. 2016, 83, 79–84. [Google Scholar] [CrossRef]
- Lee, D.C.; Yang, C.L.; Chik, S.C.; Li, J.C.; Rong, J.; Chan, G.C.; Lau, A.S. Bioactivity-guided identification and cell signaling technology to delineate the immunomodulatory effects of Panax ginseng on human promonocytic U937 cells. J. Transl. Med. 2009, 7, 34. [Google Scholar] [CrossRef]
- Li, X.-T.; Chen, R.; Jin, L.-M.; Chen, H.-Y. Regulation on Energy Metabolism and Protection on Mitochondria of Panax Ginseng Polysaccharide. Am. J. Chin. Med. 2009, 37, 1139–1152. [Google Scholar] [CrossRef]
- Attele, A.S.; Zhou, Y.-P.; Xie, J.-T.; Wu, J.A.; Zhang, L.; Dey, L.; Pugh, W.; Rue, P.A.; Polonsky, K.S.; Yuan, C.-S. Antidiabetic Effects of Panax ginseng Berry Extract and the Identification of an Effective Component. Diabetes 2002, 51, 1851–1858. [Google Scholar] [CrossRef]
- Park, J.D.; Rhee, D.K.; Lee, Y.H. Biological Activities and Chemistry of Saponins from Panax ginseng C. A. Meyer. Phytochem. Rev. 2005, 4, 159–175. [Google Scholar] [CrossRef]
- Xu, S.; Little, P.J.; Lan, T.; Huang, Y.; Le, K.; Wu, X.; Shen, X.; Huang, H.; Cai, Y.; Tang, F.; et al. Tanshinone II-A attenuates and stabilizes atherosclerotic plaques in Apolipoprotein-E knockout mice fed a high cholesterol diet. Arch. Biochem. Biophys. 2011, 515, 72–79. [Google Scholar] [CrossRef] [PubMed]
- Gong, Y.; Li, Y.; Lu, Y.; Li, L.; Abdolmaleky, H.; Blackburn, G.L.; Zhou, J.-R. Bioactive tanshinones in Salvia miltiorrhiza inhibit the growth of prostate cancer cells in vitro and in mice. Int. J. Cancer 2011, 129, 1042–1052. [Google Scholar] [CrossRef]
- Yin, X.; Yin, Y.; Cao, F.-L.; Chen, Y.-F.; Peng, Y.; Hou, W.-G.; Sun, S.-K.; Luo, Z.-J. Tanshinone IIA Attenuates the Inflammatory Response and Apoptosis after Traumatic Injury of the Spinal Cord in Adult Rats. PLoS ONE 2012, 7, e38381. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Xu, S.; Liu, P. Salvia miltiorrhizaBurge (Danshen): A golden herbal medicine in cardiovascular therapeutics. Acta Pharmacol. Sin. 2018, 39, 802–824. [Google Scholar] [CrossRef]
- Liu, L.; Xiong, X.; Shen, M.; Ru, D.; Gao, P.; Zhang, X.; Huang, C.; Sun, Y.; Li, H.; Duan, Y. Co-Delivery of Triptolide and Curcumin for Ovarian Cancer Targeting Therapy via mPEG-DPPE/CaP Nanoparticle. J. Biomed. Nanotechnol. 2018, 14, 1761–1772. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Pan, G.; Jiang, Z.; Yang, J.; Sun, L.; Zhang, L. Triptolide inhibits human breast cancer MCF-7 cell growth via downregulation of the ERα-mediated signaling pathway. Acta Pharmacol. Sin. 2015, 36, 606–613. [Google Scholar] [CrossRef]
- Bai, S.; Hu, Z.; Yang, Y.; Yin, Y.; Li, W.; Wu, L.; Fang, M. Anti-Inflammatory and Neuroprotective Effects of Triptolide via the NF-κB Signaling Pathway in a Rat MCAO Model. Anat. Rec. 2016, 299, 256–266. [Google Scholar] [CrossRef]
- Luo, H.; Vong, C.T.; Chen, H.; Gao, Y.; Lyu, P.; Qiu, L.; Zhao, M.; Liu, Q.; Cheng, Z.; Zou, J.; et al. Naturally occurring anti-cancer compounds: Shining from Chinese herbal medicine. Chin. Med. 2019, 14, 48. [Google Scholar] [CrossRef]
- Burkill, I.H.; Birtwistle, W. A Dictionary of the Economic Products of the Malay Peninsula; Ministry of Agriculture Malaysia: Kuala Lumpur, Malaysia, 2002; ISBN 9839863347. [Google Scholar]
- Sunarto, P. Penerbit Direktorat Jenderal Pengawasan Obat dan Makanan, 1st ed.; Materia Medika Indonesia: Jakarta, Indonesia, 1977. [Google Scholar]
- Noraida, A. Penyembuhan Semula Jadi Dengan Herba; PTS Millennia Sdn. Bhd.: Selangor, Malaysia, 2005; ISBN 9833372295. [Google Scholar]
- Stone, B.C. Flora of Java (Spermatophytes Only). Volume I: Gymnospermae, Families 1-7; Angiospermae, Families 8-110. C. A. Backer, R.C. Bakhuizen van den Brink, Jr. Flora of Java (Spermatophytes Only). Volume II: Angiospermae, Families 8-110. C. A. Backer, R.C.B.Q. Rev. Biol. 1969, 44, 424–425. [Google Scholar] [CrossRef]
- Nurraihana, H.; Norfarizan-Hanoon, N.A. Phytochemistry, pharmacology and toxicology properties of Strobilanthes crispus. Int. Food Res. J. 2013, 20, 2045–2056. [Google Scholar]
- Perry, L.M.; Metzger, J. Medicinal Plants of East and Southeast Asia: Attributed Properties and Uses; MIT Press: Cambridge, MA, USA, 1980; ISBN 0262160765 9780262160766. [Google Scholar]
- Samuel, A.J.S.J.; Kalusalingam, A.; Chellappan, D.K.; Gopinath, R.; Radhamani, S.; Husain, H.A.; Muruganandham, V.; Promwichit, P. Ethnomedical survey of plants used by the Orang Asli in Kampung Bawong, Perak, West Malaysia. J. Ethnobiol. Ethnomed. 2010, 6, 5. [Google Scholar] [CrossRef] [PubMed]
- Ismail, M.; Manickam, E.; Danial, A.M.; Rahmat, A.; Yahaya, A. Chemical composition and antioxidant activity of Strobilanthes crispus leaf extract. J. Nutr. Biochem. 2000, 11, 536–542. [Google Scholar] [CrossRef]
- Muslim, N.S.; Ng, K.W.; Itam, A.; Nassa, Z.D.; Ismail, Z.; Abdul Maji, A.M.S. Evaluation of Cytotoxic, Anti-angiogenic and Antioxidant Properties of Standardized Extracts of Strobilanthes crispus Leaves. Int. J. Pharmacol. 2010, 6, 591–599. [Google Scholar] [CrossRef]
- Qader, S.W.; Abdulla, M.A.; Chua, L.S.; Najim, N.; Zain, M.M.; Hamdan, S. Antioxidant, Total Phenolic Content and Cytotoxicity Evaluation of Selected Malaysian Plants. Molecules 2011, 16, 3433–3443. [Google Scholar] [CrossRef] [PubMed]
- Muskhazli, M.; Dirnahayu, M.; Nor Azwady, A.A.; Nurhafiza, Y.; Nor Dalilah, E.; Che Ku Nurshaira, C.K.N. Antibacterial Activity of Methanolic Crude Extracts from Selected Plant Against Bacillus cereus. Pertanika J. Trop. Agric. Sci. 2009, 32, 175–183. [Google Scholar]
- Hamad Abou Muamar, A.F. Isolation, Identification And Evaluation Of Antibacterial Activity Of The Semi-Purified Compound From Strobilanthes Crispus (L. Bremek). Master’s Thesis, Universiti Putra Malaysia, Seri Kembangan, Selangor, Malaysia, 1999. [Google Scholar]
- Norfarizan, N.A.; Asmah, R.; Rokiah, M.Y.; Fauziah, O.; Faridah, H. Antihyperglycemic, Hypolipidemic and Antioxidant Enzymes Effect of Strobilanthes crispus Juice in Normal and Streptozotocin-Induced Diabetic Male and Female Rats. Int. J. Pharmacol. 2009, 5, 200–207. [Google Scholar] [CrossRef]
- Mahmood, A.A.; Fard, A.A.; Harita, H.; Zahra, A.A.; Salmah, I. Evaluation of gastroprotective effects of Strobianthes crispus leaf extract on ethanol-induced gastric mucosal injury in rats. Sci. Res. Essays 2011, 6, 2306–2314. [Google Scholar] [CrossRef]
- Al-Henhena, N.; Mahmood, A.A.; Al-magrami, A.; Nor Syuhada, A.B.; Zahra, A.A.; Summaya, M.D.; Suzi, M.S.; Salmah, I. Histological study of wound healing potential by ethanol leaf extract of Strobilanthes crispus in rats. J. Med. Plants Res. 2011, 5, 3660–3666. [Google Scholar]
- Norfarizan, N.A.; Asmah, R.; Rokiah, M.Y.; Fauziah, O.; Faridah, H. Effects of Strobilanthes crispus Juice on Wound Healing and Antioxidant Enzymes in Normal and Streptozotocin-Induced Diabetic Rats. J. Biol. Sci. 2009, 9, 662–668. [Google Scholar] [CrossRef]
- Yaacob, N.S.; Hamzah, N.; Nik Mohamed Kamal, N.N.; Zainal Abidin, S.A.; Lai, C.S.; Navaratnam, V.; Norazmi, M.N. Anticancer activity of a sub-fraction of dichloromethane extract of Strobilanthes crispus on human breast and prostate cancer cells in vitro. BMC Complement. Altern. Med. 2010, 10, 42. [Google Scholar] [CrossRef]
- Liza, M.S.; Abdul Rahman, R.; Mandana, B.; Jinap, S.; Rahmat, A.; Zaidul, I.S.M.; Hamid, A. Supercritical carbon dioxide extraction of bioactive flavonoid from Strobilanthes crispus (Pecah Kaca). Food Bioprod. Process. 2010, 88, 319–326. [Google Scholar] [CrossRef]
- Soediro, L.; Pellecuer, J.; Andary, C.; Privat, G. Strobilanthes crispus (L) B1 I: Pemeriksaan senyawaan turunan asam kafeat verbascosid. Acta Pharm. Indones. 1983, 8, 1–10. [Google Scholar]
- Cheong, B.E.; Zakaria, N.A.; Cheng, A.Y.F.; Teoh, P.L. GC-MS Analysis of Strobilanthes crispus Plants and Callus. Trans. Sci. Technol. 2016, 3, 155–161. [Google Scholar]
- Chong, H.Z.; Rahmat, A.; Yeap, S.K.; Md Akim, A.; Alitheen, N.B.; Othman, F.; Gwendoline-Ee, C.L. In vitro cytotoxicity of Strobilanthes crispus ethanol extract on hormone dependent human breast adenocarcinoma MCF-7 cell. BMC Complement. Altern. Med. 2012, 12, 35. [Google Scholar] [CrossRef]
- Rahmat, A.; Edrini, S.; Akim, A.M.; Ismail, P.; Yap, T.Y.H.; Fadzelly, M.A.B. Anticarcinogenic Properties of Strobilanthes crispus Extracts and its Compounds in vitro. Int. J. Cancer Res. 2005, 2, 47–49. [Google Scholar] [CrossRef]
- Baraya, Y.S.; Wong, K.K.; Yaacob, N.S. Strobilanthes crispus inhibits migration, invasion and metastasis in breast cancer. J. Ethnopharmacol. 2019, 233, 13–21. [Google Scholar] [CrossRef]
- Yaacob, N.S.; Kamal, N.N.N.M.; Wong, K.K.; Norazmi, M.N. Cell Cycle Modulation of MCF-7 and MDA-MB-231 by a Sub-Fraction of Strobilanthes crispus and its Combination with Tamoxifen. Asian Pacific J. Cancer Prev. 2016, 16, 8135–8140. [Google Scholar] [CrossRef] [PubMed]
- Gordani, N.; Cheong, B.E.; Teoh, P.L. Antiproliferative Effect of Strobilanthes crispus on MCF-7 Cell Line. Trans. Sci. Technol. 2017, 4, 414–419. [Google Scholar]
- Koh, R.Y.; Lim, F.P.; Ling, L.S.Y.; Ng, C.P.L.; Liew, S.F.; Yew, M.Y.; Tiong, Y.L.; Ling, A.P.K.; Chye, S.M.; Ng, K.Y. Anticancer mechanisms of Strobilanthes crispa Blume hexane extract on liver and breast cancer cell lines. Oncol. Lett. 2017, 14, 4957–4964. [Google Scholar] [CrossRef]
- Endrini, S.; Rahmat, A.; Ismail, P.; Taufiq-Yap, Y.H. Cytotoxic effect of γ-sitosterol from Kejibeling (Strobilanthes crispus) and its mechanism of action towards c-myc gene expression and apoptotic pathway. Med. J. Indones. 2015, 23, 203–208. [Google Scholar] [CrossRef]
- Fadzelly, M.A.B.; Teh, A.H.; Rahmat, A.; Othman, F.; Hashim, N.; Fakurazi, S. Antiproliferative Properties and Antioxidant Activity of Various Types of Strobilanthes crispus Tea. Int. J. Cancer Res. 2006, 2, 152–158. [Google Scholar] [CrossRef]
- Rahmat, A.; Edrini, S.; Ismail, P.; Yap, T.Y.H.; Fadzelly, M.A.B. Chemical Constituents, Antioxidant Activity and Cytotoxic Effects of Essential Oil from Strobilanthes crispus and Lawsonia inermis. J. Biol. Sci. 2006, 6, 1005–1010. [Google Scholar] [CrossRef]
- Ng, K.-W.; Salhimi, S.; Majid, A.; Chan, K.-L. Anti-Angiogenic and Cytotoxicity Studies of Some Medicinal Plants. Planta Med. 2010, 76, 935–940. [Google Scholar] [CrossRef]
- Yaacob, N.S.; Yankuzo, H.M.; Devaraj, S.; Wong, J.K.M.; Lai, C.-S. Anti-Tumor Action, Clinical Biochemistry Profile and Phytochemical Constituents of a Pharmacologically Active Fraction of S. crispus in NMU-Induced Rat Mammary Tumour Model. PLoS ONE 2015, 10, e0126426. [Google Scholar] [CrossRef]
- Yankuzo, H.M.; Baraya, Y.S.; Mustapha, Z.; Wong, K.K.; Yaacob, N.S. Immunomodulatory effects of a bioactive fraction of Strobilanthes crispus in NMU-induced rat mammary tumor model. J. Ethnopharmacol. 2018, 213, 31–37. [Google Scholar] [CrossRef]
- Mahmoud, S.M.A.; Paish, E.C.; Powe, D.G.; Macmillan, R.D.; Grainge, M.J.; Lee, A.H.S.; Ellis, I.O.; Green, A.R. Tumor-Infiltrating CD8 + Lymphocytes Predict Clinical Outcome in Breast Cancer. J. Clin. Oncol. 2011, 29, 1949–1955. [Google Scholar] [CrossRef]
- Ali, H.R.; Provenzano, E.; Dawson, S.-J.; Blows, F.M.; Liu, B.; Shah, M.; Earl, H.M.; Poole, C.J.; Hiller, L.; Dunn, J.A.; et al. Association between CD8+ T-cell infiltration and breast cancer survival in 12 439 patients. Ann. Oncol. 2014, 25, 1536–1543. [Google Scholar] [CrossRef]
- Loi, S.; Michiels, S.; Salgado, R.; Sirtaine, N.; Jose, V.; Fumagalli, D.; Kellokumpu-Lehtinen, P.-L.; Bono, P.; Kataja, V.; Desmedt, C.; et al. Tumor infiltrating lymphocytes are prognostic in triple negative breast cancer and predictive for trastuzumab benefit in early breast cancer: Results from the FinHER trial. Ann. Oncol. 2014, 25, 1544–1550. [Google Scholar] [CrossRef]
- Holling, T.M.; Schooten, E.; van Den Elsen, P.J. Function and regulation of MHC class II molecules in T-lymphocytes: Of mice and men. Hum. Immunol. 2004, 65, 282–290. [Google Scholar] [CrossRef]
- Shi, B.; Vinyals, A.; Alia, P.; Broceno, C.; Chen, F.; Adrover, M.; Gelpi, C.; Price, J.; Fabra, A. Differential expression of MHC class II molecules in highly metastatic breast cancer cells is mediated by the regulation of the CIITA transcriptionImplication of CIITA in tumor and metastasis development. Int. J. Biochem. Cell Biol. 2006, 38, 544–562. [Google Scholar] [CrossRef] [PubMed]
- Endrini, S.; Suherman, S.; Rahmat, A.; Ismail, P.; Taufiq-Yap, Y.; Othman, F. Effects of strobilanthes crispus extract on the apoptotic pathway of human liver carcinoma cell lines. Yars. Med. J. 2019, 15. [Google Scholar] [CrossRef]
- Hussin, F.; Eshkoor, S.A.; Rahmat, A.; Othman, F.; Akim, A.; Eshak, Z. Strobilanthes crispus Juice Concentrations and Anticancer Effects on DNA Damage, Apoptosis and Gene Expression in Hepatocellular Carcinoma Cells. Asian Pacific J. Cancer Prev. 2015, 16, 6047–6053. [Google Scholar] [CrossRef] [PubMed]
- Tan, S.-A.; Lim, S.; Law, C.; Yue, C.; Poh, T.; Saad, W.; Ismail, S.; Yusoff, K.; Loke, C. Antioxidative and Photocytotoxic Effects of Standardized Clinacanthus nutans and Strobilanthes crispus Extracts toward HepG2 Liver Cells. Pharmacogn. Mag. 2019, 15, 613. [Google Scholar] [CrossRef]
- Hanachi, P.; Fauziah, O.; Asmah, R. Lesion scoring and P450 Isoenzyme activity in liver of hepatocarcinogenesis rats treated with Strobilanthes Crispus. Int. J. Cancer Manag. 2008, 1, 11–15. [Google Scholar]
- Fauziah, O.; Hanachi, P.; Yogespiriya, S.; Asmah, R. Reducing Effect of Strobilanthes crispus Leaf Extract in Hepatocarcinogenesis Rats. Int. J. Cancer Res. 2005, 1, 109–112. [Google Scholar] [CrossRef]
- Yogespiriya, S.; Hanachi, P.; Patimah, I.; Asmah, R.; Fauziah, O. Histological Study During Hepatocarciniogenesis in Rats Treated With Strobilanthes crispus Extract. J. Biol. Sci. 2005, 5, 153–157. [Google Scholar] [CrossRef][Green Version]
- Suherman, J.; Asmah, R.; Fauziah, O.; Patimah, I.; Nor Haslinda, A. Effect of Strobilanthes crispus on Tumour Marker Enzymes and Glutathione During Chemical Hepatocarcinogenesis in the Rat. Pakistan J. Biol. Sci. 2004, 7, 947–951. [Google Scholar] [CrossRef][Green Version]
- Suherman, J.; Asmah, R.; Fauziah, O.; Patimah, I.; Siti Muskinah, H.M. Effect of Strobilanthes crispus on the Histology and Tumour Marker Enzymes in Rat Liver During Hepatocarcinogenesis. J. Med. Sci. 2005, 5, 130–135. [Google Scholar] [CrossRef]
- Ismail, M.; Bagalkotkar, G.; Iqbal, S.; Adamu, H.A. Anticancer Properties and Phenolic Contents of Sequentially Prepared Extracts from Different Parts of Selected Medicinal Plants Indigenous to Malaysia. Molecules 2012, 17, 5745–5756. [Google Scholar] [CrossRef] [PubMed]
- Al-Henhena, N.; Khalifa, S.A.M.; Ying, R.P.Y.; Hassandarvish, P.; Rouhollahi, E.; Al-Wajeeh, N.S.; Ali, H.M.; Abdulla, M.A.; El-Seedi, H.R. Chemopreventive effects of Strobilanthes crispus leaf extract on azoxymethane-induced aberrant crypt foci in rat colon. Sci. Rep. 2015, 5, 13312. [Google Scholar] [CrossRef]
- Chong, Y.H.; Koh, R.Y.; Ling, A.P.K.; Chye, S.M.; Yew, M.Y. Strobilanthes crispus Extract Induces Apoptosis Through Enhanced Caspases Activities in Cervical Cancer Cells. In Proceedings of the International Conference on Biological, Environment and Food Engineering (BEFE-2014), Bali, Indonesia, 4–5 August 2014; International Institute of Chemical, Biological & Environmental Engineering: Barcelona, Spain, 2014. [Google Scholar]
- Koh, R.Y.; Sim, Y.C.; Toh, H.J.; Liam, L.K.; Lynn Ong, R.S.; Yew, M.Y.; Tiong, Y.L.; Ling, A.P.K.; Chye, S.M.; Ng, K.Y. Cytotoxic and apoptogenic effects of Strobilanthes crispa Blume extracts on nasopharyngeal cancer cells. Mol. Med. Rep. 2015, 12, 6293–6299. [Google Scholar] [CrossRef]
- Norfarizan-Hanoon, N.A.; Asmah, R.; Fauziah, O.; Rokiah, M.Y.; Faridah, H. Absence of Toxicity of Strobilanthes crispa Juice in Acute Oral Toxicity Study in Sprague Dawley Rats. Sains Malays. 2012, 41, 403–409. [Google Scholar]
- Lim, K.T.; Lim, V.; Chin, J.H. Subacute oral toxicity study of ethanolic leaves extracts of Strobilanthes crispus in rats. Asian Pac. J. Trop. Biomed. 2012, 2, 948–952. [Google Scholar] [CrossRef]
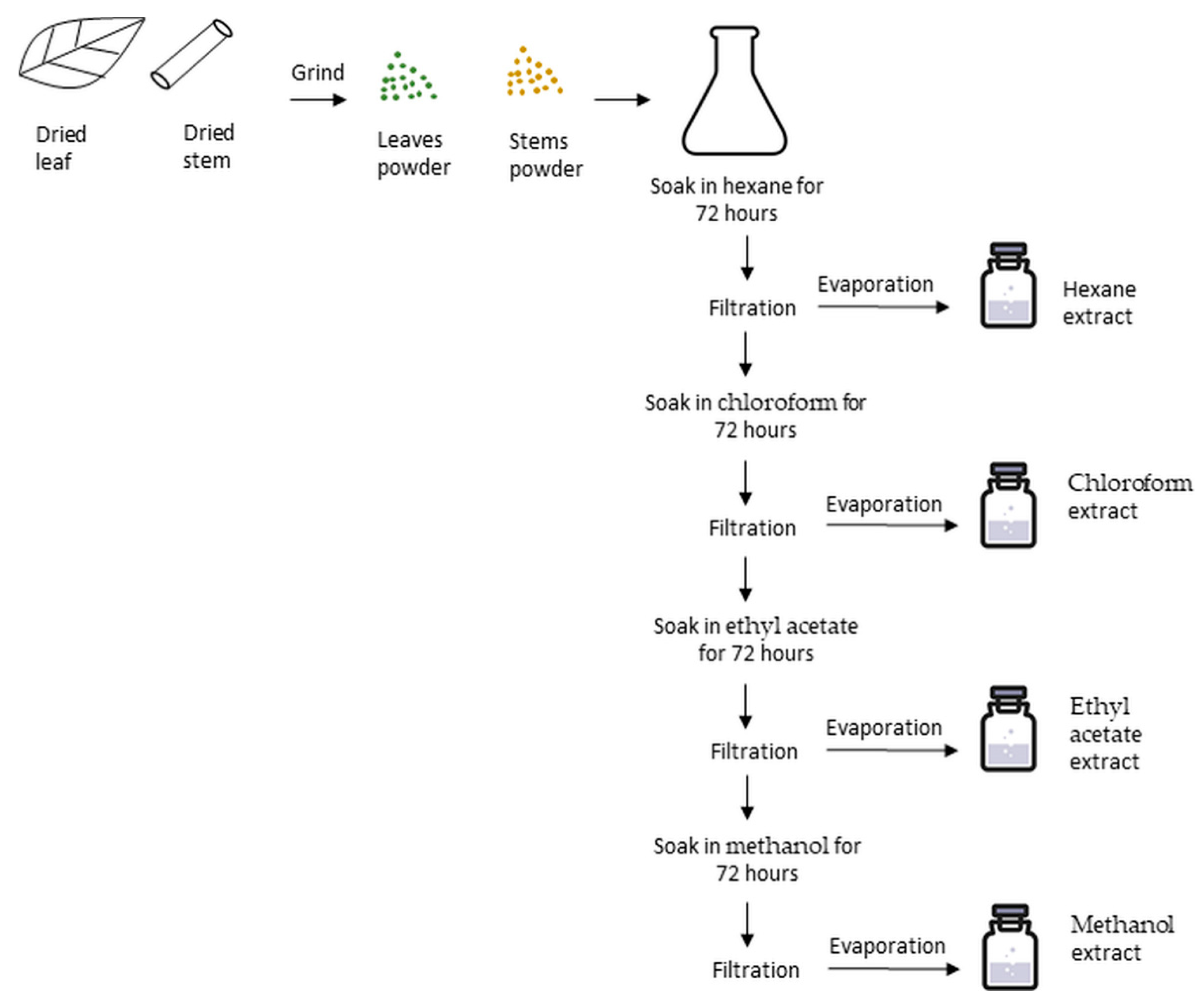
| Scientific Name & Common Name | Extracts | Therapeutic Effects |
|---|---|---|
| Camellia sinensis (Green Tea) | Epigallocatechin Gallate (EGCG) | Anti-obesity and anti-hyperuricemia, anti-oxidative, anti-viral, anti-bacterial, anti-infective, anti-angiogenic, anti-inflammation, anti-cancer [11,12,13,14,15]. |
| Curcuma longa (Turmeric) | Curcumin | Anti-cancer, anti-inflammation, anti-oxidant [8,16,17,18]. |
| Echinacea spp. (Coneflower) | Echinaforce | Anti-oxidant, anti-microbial, anti-inflammation [19,20]. |
| Garcinia spp. (Brindleberry, Kudam Puli) | Gambogic Acid (GA) | Anti-oxidative, anti-inflammation, anti-cancer [21,22,23]. |
| Panax ginseng (Asian Ginseng) | Ginsenosides | Anti-inflammation, anti-oxidant, anti-diabetic, anti-cancer [24,25,26,27]. |
| Salvia miltiorrhiza (Red Sage, Danshen) | Tanshinones | Anti-inflammation, anti-cancer and anti-atherosclerotic activities, cardiovascular protection [28,29,30,31]. |
| Tripterygium wilfordii (Thunder God Vine) | Triptolide | Anti-inflammation, anti-oxidative, anti-cancer [32,33,34]. |
| Part of S. crispus | Phytochemicals | References |
|---|---|---|
| Leaf | Proximate Analysis (%) Crude Fibre: 13.9 ± 0.6 Protein Content: 13.3 ± 0.9 Total Carbohydrate: 4.3 ± 0.7 Minerals (mg/100 g sample) Potassium: 10,900 ± 498 Calcium: 5185 ± 359 Sodium: 2953 ± 60 Iron: 255 ± 163 Phosphorus: 201 ± 22 Vitamins (%) Ascorbic Acid (C): 9.8 ± 1.2 Thiamin (B1): 0.14 ± 0.001 Riboflavin (B2): 0.11 ± 0.04 Other components (%) Alkaloid: 3.2 ± 0.60 Catechin: 1.18 ± 0.08 Tannin: 1.0 ± 0.30 | Maznah et al. [43] |
| Leaf | Flavonoid Content (%): 3.98 Contents of Flavonoid (mg/g) Kampferol: 19.45 Luteolin: 12.52 Rutin: 8.47 (+)-Catechin: 4.83 (–)-Epicatechin: 4.55 Myricetin: 4.10 Apigenin: 3.75 Naringenin: 3.63 | Liza et al. [53] |
| Leaf | Contents of Phenolic Acids Caffeic acid Ferulic acid Gentisic acid p-caumeric acid p-hydroxybenzoic acid Syringic acid Vanilic acid Ester glycoside: Verbascoside | Soediro et al. [54] |
| Leaf | Methanol extract Hexadecanoic acid, methylester: 12.11% 3-octadedecyne: 9.25% Stigmasterol: 7.89% α-sitosterol: 7.08% Phytol: 3.78% Lupeol: 3.60% 3,7,11,15-tetramethyl-2-hexadecen-1-ol: 3.48% And others Aqueous extract Nitrous Oxide: 21.44% Cyclobutanol: 13.56% Monoethanolamine: 5.53% n-propylacetate: 5.38% hydrazine carboxamide: 3.32% 3,5-dithiahexanol 5,5-dioxide: 3.09% And others | Muslim et al. [44] |
| Leaf | 9,12,15-Octadecatrienoic acid, (Z,Z,Z)-:26.21% Squalene:26.11% Stigmasterol:10.93% Vitamin E: 9.75% γ-sitosterol: 6.70% Campesterol: 3.57% And others | Cheong et al. [55] |
| Stem | Lupeol: 25.58% 9,12-octadecadienoic acid (Z,Z)-: 14.41% Stigmasterol: 13.23% γ-sitosterol: 8.47% Campesterol: 4.85% Vitamin E: 3.18% And others | |
| Root | β-amyrin: 49.26% Heneicosane: 5.55% Stigmasterol: 5.30% Eicosane: 2.87% 9-Octadecenamide, (Z)-: 1.75% Squalene: 1.04% Bis(2-ethylhexyl) phthalate: 0.16% | |
| Callus | β-humulene: 26.22% 4,22-Stigmastadiene-3-one: 9.08% Stigmast-4-en-3-one: 5.56% 2-methyl-Z,Z-3,13-octadecadienol: 2.03% Stigmasterol: 1.86% Cis,cis,cis-7,10,13-hexadecatriena: 1.82% And others |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ng, M.G.; Ng, C.H.; Ng, K.Y.; Chye, S.M.; Ling, A.P.K.; Koh, R.Y. Anticancer Properties of Strobilanthes crispus: A Review. Processes 2021, 9, 1370. https://doi.org/10.3390/pr9081370
Ng MG, Ng CH, Ng KY, Chye SM, Ling APK, Koh RY. Anticancer Properties of Strobilanthes crispus: A Review. Processes. 2021; 9(8):1370. https://doi.org/10.3390/pr9081370
Chicago/Turabian StyleNg, Ming Guan, Chew Hee Ng, Khuen Yen Ng, Soi Moi Chye, Anna Pick Kiong Ling, and Rhun Yian Koh. 2021. "Anticancer Properties of Strobilanthes crispus: A Review" Processes 9, no. 8: 1370. https://doi.org/10.3390/pr9081370
APA StyleNg, M. G., Ng, C. H., Ng, K. Y., Chye, S. M., Ling, A. P. K., & Koh, R. Y. (2021). Anticancer Properties of Strobilanthes crispus: A Review. Processes, 9(8), 1370. https://doi.org/10.3390/pr9081370






