A Novel Manganese-Rich Pokeweed Biochar for Highly Efficient Adsorption of Heavy Metals from Wastewater: Performance, Mechanisms, and Potential Risk Analysis
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals and Raw Materials
2.2. Preparation of the Biochar
2.3. Characterization
2.4. Adsorption Experiments
2.5. Potential Risk Analysis
3. Results
3.1. Characterization of the Materials
3.1.1. FTIR Analyses
3.1.2. SEM-EDS Analyses
3.1.3. BET Results and Elemental Analyses
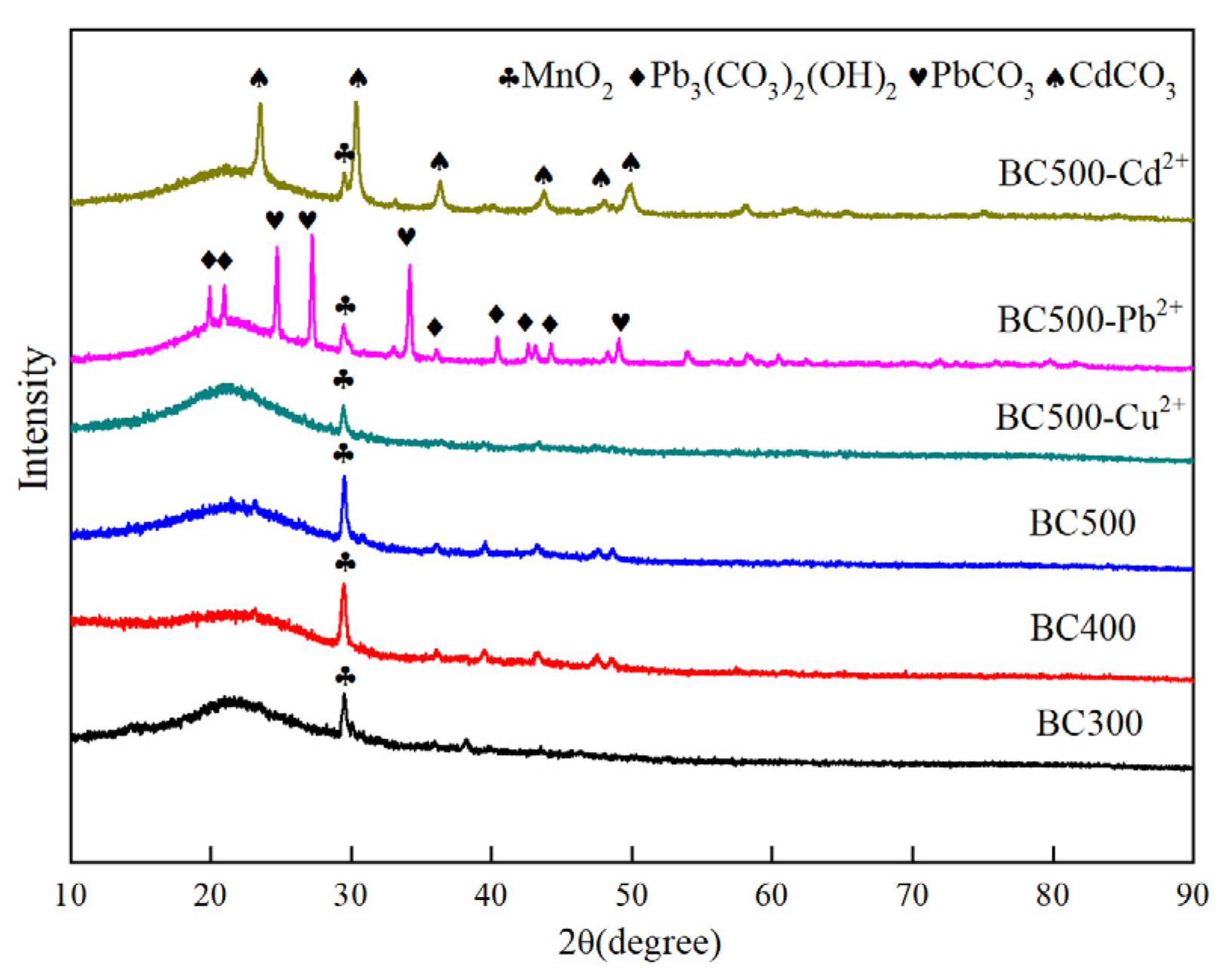
3.1.4. XRD Analyses
3.1.5. XPS Analyses
3.2. Adsorption Experiments
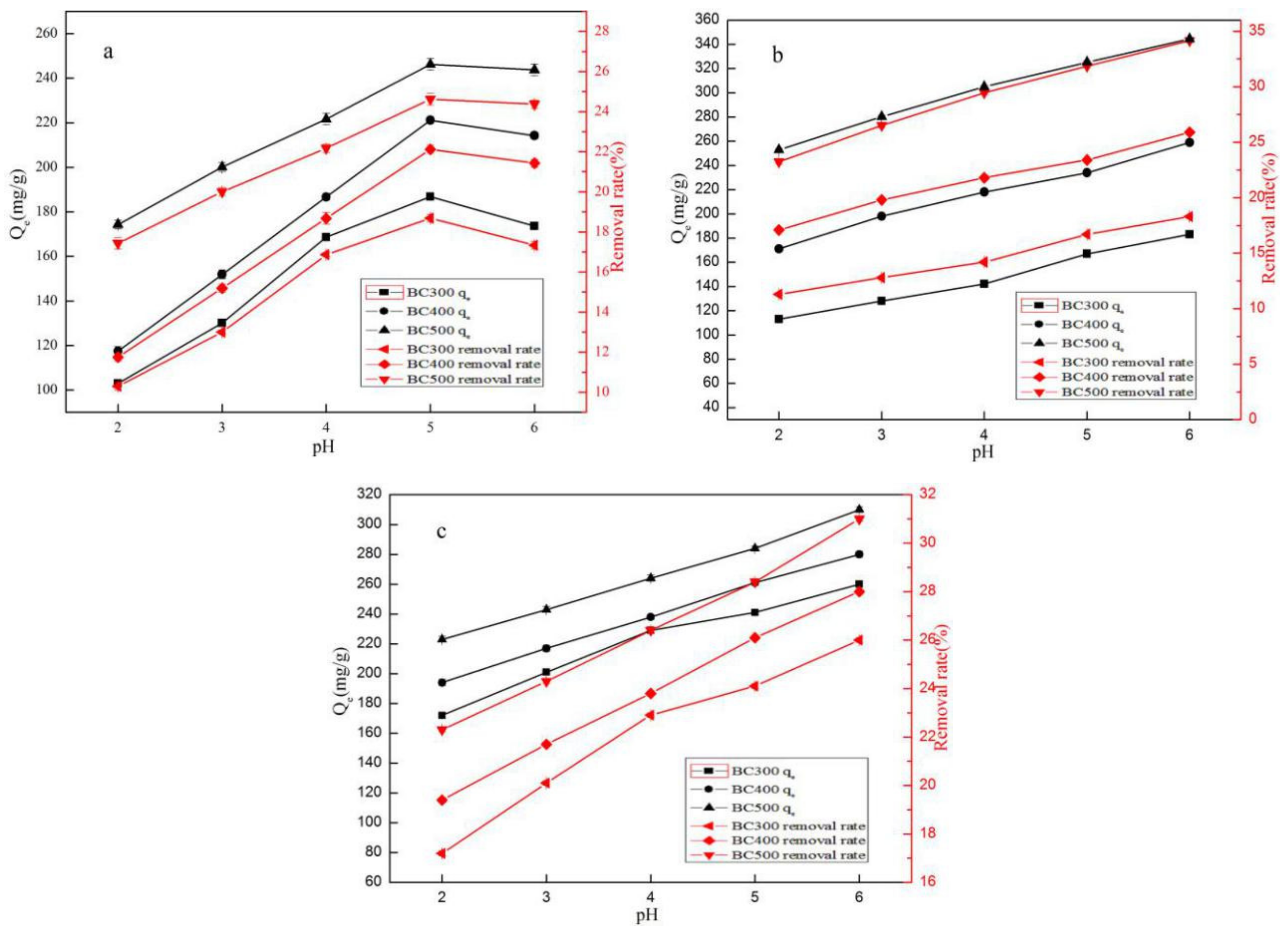
3.2.1. The Effect of pH
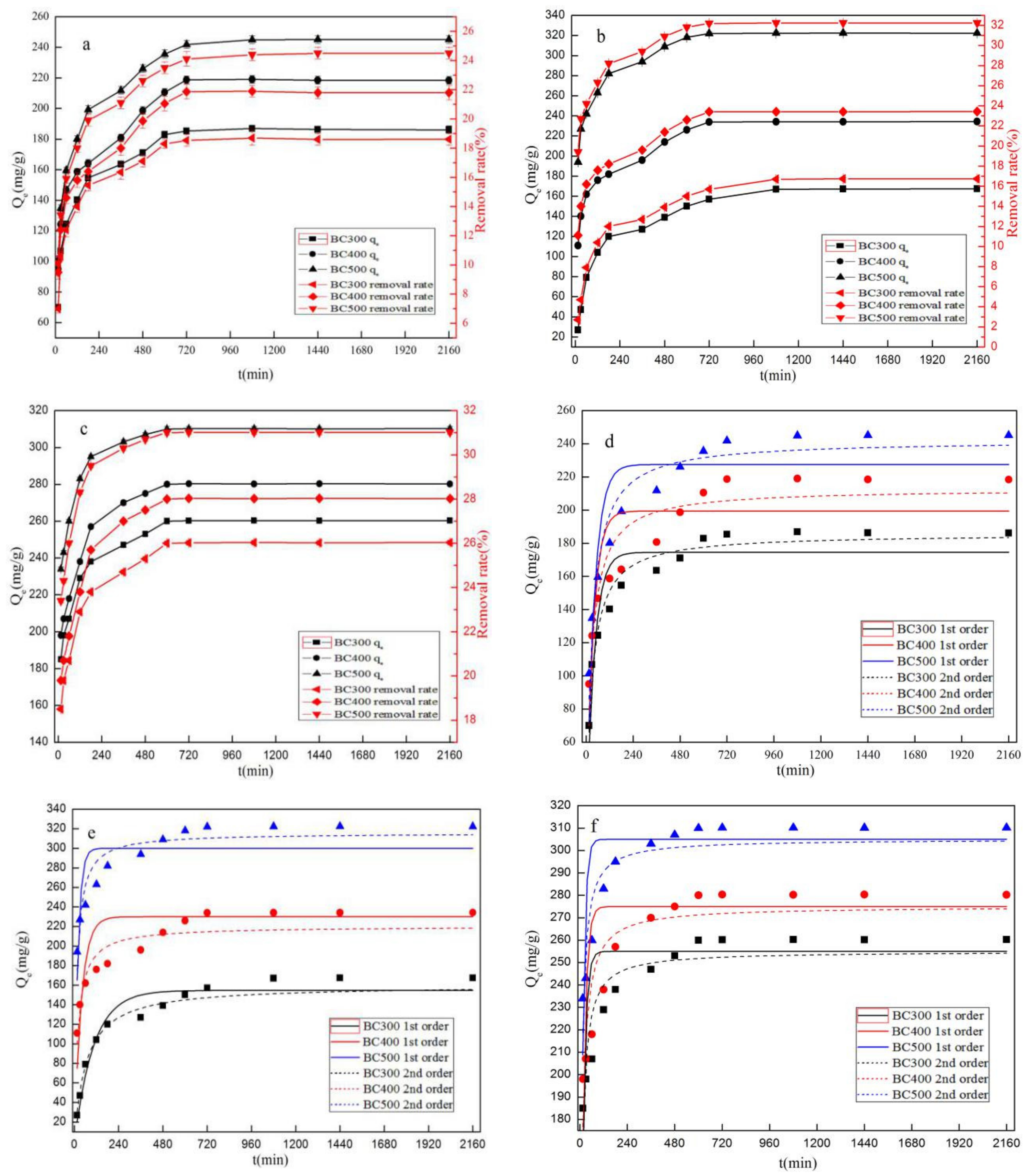
3.2.2. Adsorption Kinetics
3.2.3. The Effect of Temperature
3.2.4. The Effect of Dosages
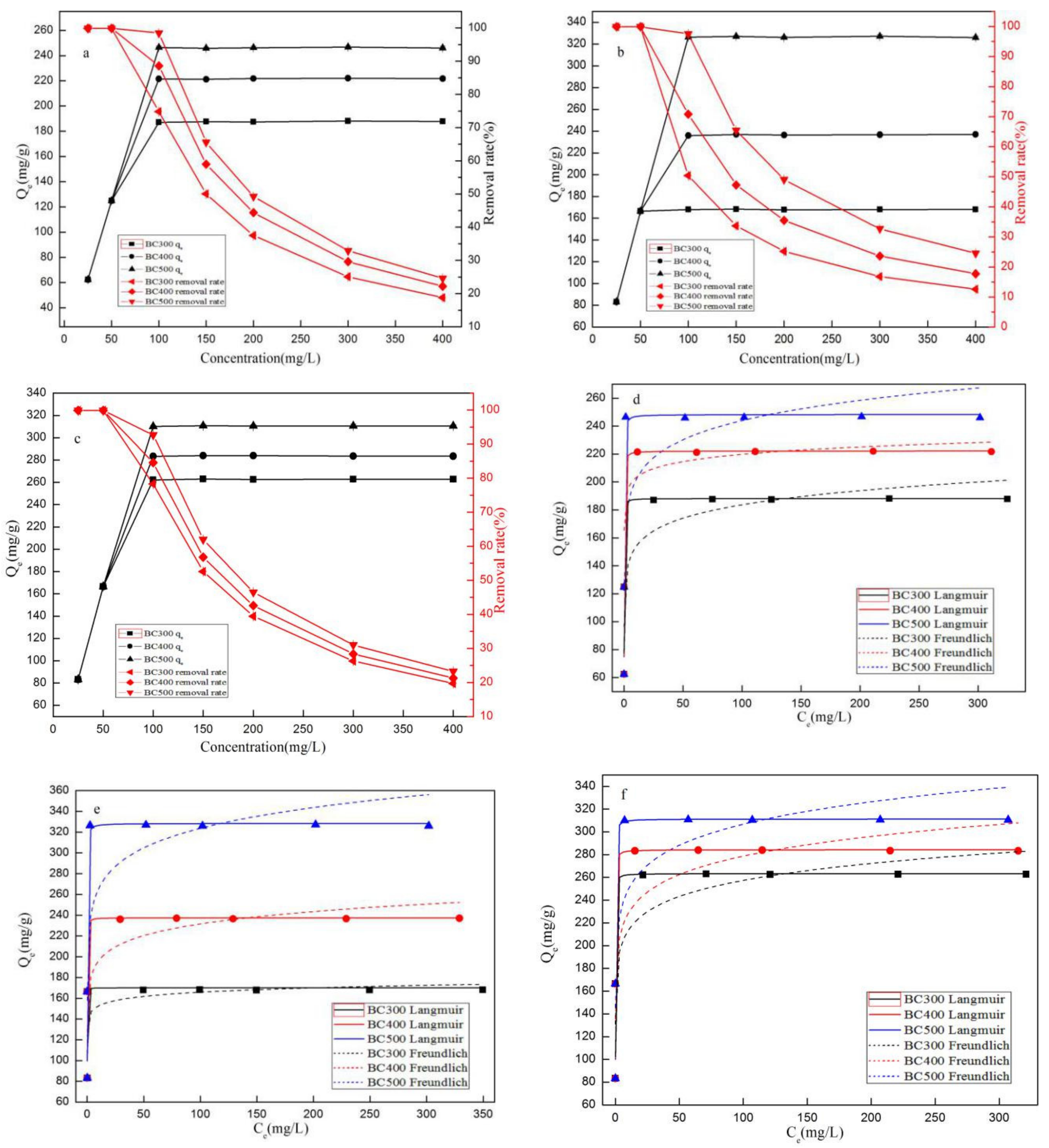
3.2.5. Adsorption Isotherms
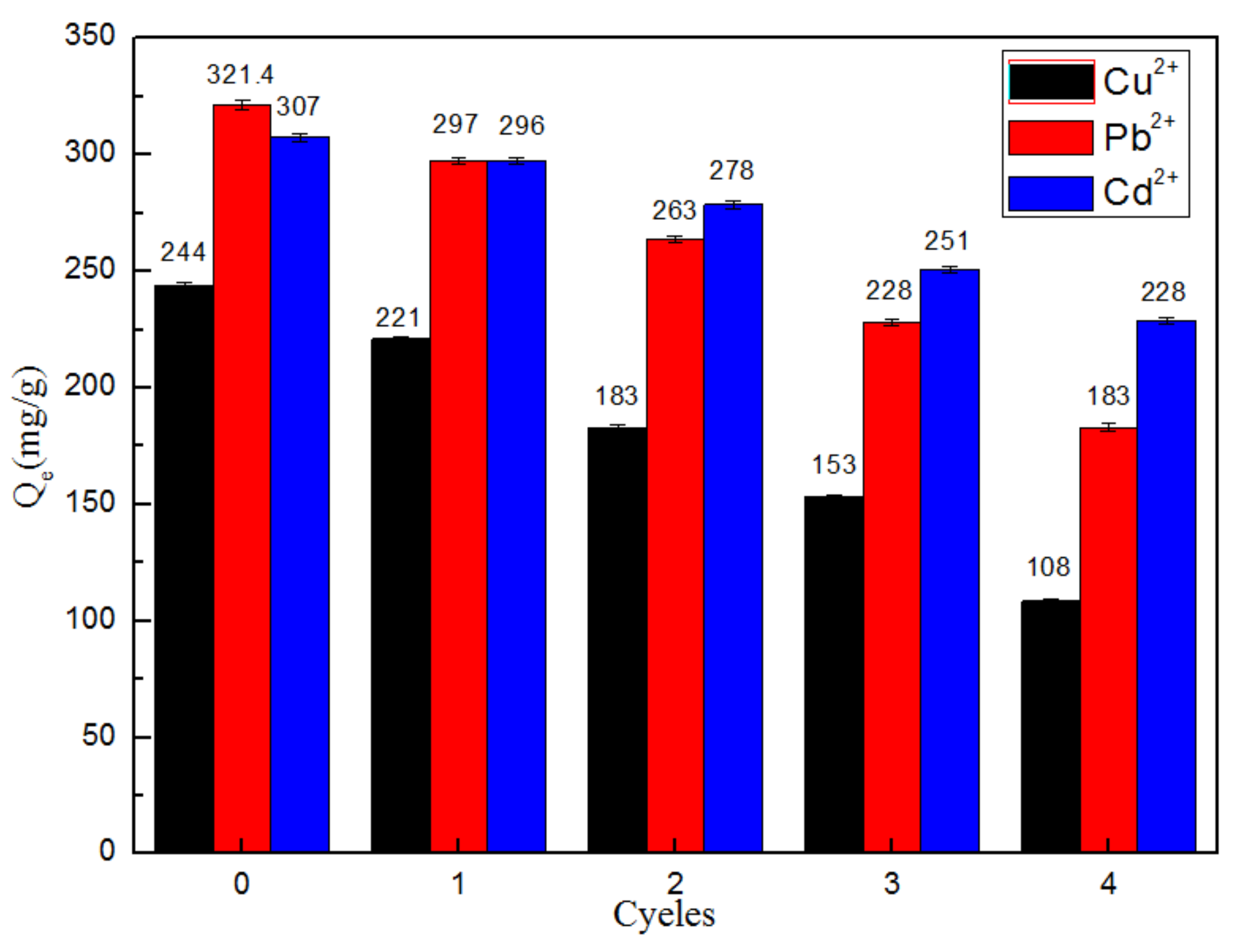
3.2.6. Effects of Repeated Adsorption Cycles
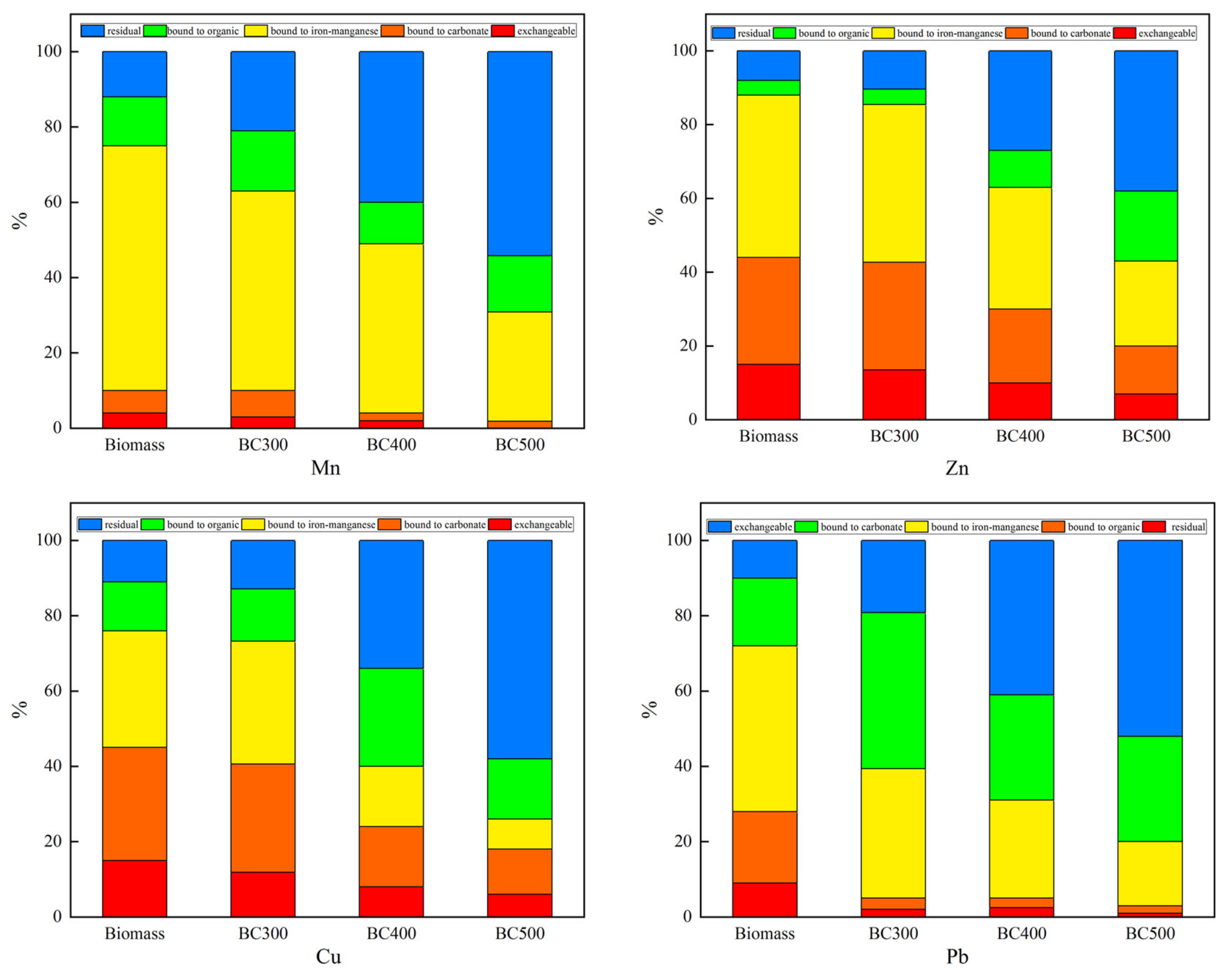
3.3. Potential Risk Analysis
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Chen, Y.; Jiang, X.; Wang, Y.; Zhuang, D. Spatical characteristics of heavy metal pollution and the potential ecological risk of a typical mining area: A case study in China. Process Saf. Environ. 2018, 113, 204–219. [Google Scholar] [CrossRef]
- Kranthi, K.; Sardar, R.; Bhargavi, E.; Devi, I.; Bhunia, B.; Tiwari, N. Advances in exopolysaccharides based bioremediation of heavy metals in soil and water: A critical review. Carbohydr. Polym. 2018, 199, 353–364. [Google Scholar]
- Fu, L.; Wang, Q. Removal of heavy metal ions from wastewaters: A review. J. Environ. Manag. 2011, 92, 407–418. [Google Scholar] [CrossRef]
- Li, N.; Kang, Y.; Pan, W.; Zeng, L.; Zhang, Q.; Luo, J. Concentration and transportation of heavy metals in vegetables and risk assessment of human exposure to bioaccessible heavy metals in soil near a waste incinerator site. Sci. Total Environ. 2015, 521, 144–151. [Google Scholar] [CrossRef] [PubMed]
- Kumar, R.; Rani, M.; Gupta, H.; Gupta, B. Trace metal fractionation in water and sediments of an urban river stretch. Chem. Speciat. Bioavailab. 2014, 26, 200–209. [Google Scholar] [CrossRef] [Green Version]
- Yu, F.; Liu, K.; Ye, P.; Zhou, Z.; Chen, S.; Li, Y. Manganese tolerance and accumulation characteristics of a woody accumulator Camellia oleifera. Environ. Sci. Pollut. Res. 2019, 26, 21329–21339. [Google Scholar] [CrossRef]
- Moreno, L.; Neretnieks, I. Long-term environmental impact of tailings deposits. Hydrometallurgy 2006, 83, 176–183. [Google Scholar] [CrossRef]
- Chavan, A.; Li, H.; Scarpellini, A.; Marras, S.; Manna, L.B.; Athanassiou, A.; Fragouli, D. Elastomeric nanocomposite foams for the removal of heavy metal ions from water. ACS Appl. Mater. Interfaces 2015, 7, 14778–14784. [Google Scholar] [CrossRef]
- Gollavelli, G.; Chang, C.; Ling, C. Facile synthesis of smart magnetic graphene for safe frinking water: Heavy matal removal and disinfection control. ACS Sustain. Chem. Eng. 2013, 1, 462–472. [Google Scholar] [CrossRef]
- Song, W.; Liu, M.; Hu, R. Water-soluble polyacrylamide coated-Fe3O4 magnetic composites for high-effcient enrichment of U(IV) from radioactive wastewater. Chem. Eng. J. 2014, 246, 268–276. [Google Scholar] [CrossRef]
- Vukovic, D.; Marinkovic, D.; Colic, M.; Ristic, D.; Aleksic, R.; Grujic, P.; Uskokovic, S. Removal of cadmium from aqueous solutions solutions by oxidized and ethylenedi amine-functionalized multi-walled carbon nanotubes. Chem. Eng. J. 2010, 157, 238–248. [Google Scholar] [CrossRef]
- Li, Z.; Ma, Z.; Van, J. A review of soil heavy metal pollution from mines in China: Pollution and health risk assessment. Sci. Total Environ. 2014, 2, 843–853. [Google Scholar] [CrossRef] [PubMed]
- Zhao, T.; Ma, S.; Zheng, Y.; Han, W.; Yao, X.; Wang, Z.; Wang, S.; Feng, K. β-cyclodextrin functionalized biochars as novel sorbents for high-performance of Pb2+ removal. J. Hazard. Mater. 2019, 362, 206–216. [Google Scholar] [CrossRef] [PubMed]
- Ni, J.; Huang, S.; Wang, C.; Ni, Y.; Sun, J.; Wei, W. Competitive adsorption of heavy metals in aqueous solution onto biochar derived from anaerobically digested sludge. Chemosphere 2019, 219, 351–357. [Google Scholar] [CrossRef]
- Mahmoud, E.; Khalifa, A.; Wakeel, M.; Header, S.; Sharkawy, M.; Kumar, S.; Fattah, M. A novel nanocomposite of Liquidambar styraciflua fruit biochar-crosslinked-nanosilica for uranyl removal from water. Bioresour. Technol. 2019, 278, 124–129. [Google Scholar] [CrossRef]
- Guilhen, N.; Masek, O.; Ortiz, N.; Izidoro, C.; Fungaro, A. Pyrolytic temperature evaluation of macauba biochar for uranium adsorption from aqueous solutions. Biomass Bioenergy 2019, 122, 381–390. [Google Scholar] [CrossRef] [Green Version]
- Jiang, H.; Li, Y.; Deng, H.; Ye, H.; Wu, Q.; Lin, D.; Hang, L. Characteristics of nitrogen and phosphorus adsorption by Mg-loaded biochar from different feedstocks. Bioresour. Technol. 2019, 276, 183–189. [Google Scholar] [CrossRef]
- Kermit, W.; Hong, Y.; Chuang, W.S.; Wayne, E.M. Select metal adsorption by activated carbon made from peanut shells. Bioresour. Technol. 2006, 97, 2260–2270. [Google Scholar]
- Rao, M.; Ramana, K.; Seshaiah, K.; Wang, C.; Chien, C. Removal of some metal ions by activated carbon prepared from phaseolus aureus hulls. J. Hazard. Mater. 2009, 166, 1006–1013. [Google Scholar] [CrossRef]
- Leng, J.; Huang, J.; Li, H.; Zhou, G. Biochar stability assessment methods: A review. Bioresour. Technol. 2019, 647, 210–220. [Google Scholar] [CrossRef]
- Xu, J.; Cao, Z.; Zhang, Y.; Yuan, Z.L.; Lou, Z.; Xu, X.H.; Wang, X.K. A review of functionalized carbon nanotubes and graphene for heavy metal adsorption from water: Preparation, application, and mechanism. Chemosphere 2018, 195, 351–364. [Google Scholar] [CrossRef] [PubMed]
- Dinesh, M.; Ankur, S.; Yong, O.; Charles, P. Organic and inorganic contaminants removal from water with biochar, a renewable, low cost and sustainable adsorbent-A critical review. Bioresour. Technol. 2014, 160, 191–202. [Google Scholar]
- Ahmad, M.; Rajapaksha, A.U.; Lim, E.; Zhang, M.; Bolan, N.; Mohan, D. Biochar as a sorbent for contaminant management in soil and water: A review. Chemosphere 2014, 99, 19–33. [Google Scholar] [CrossRef]
- Yu, W.; Lian, F.; Cui, G.; Liu, Z. N-doping effectively enhances the adsorption capacity of biochar for heavy metal ions from aqueous solution. Chemosphere 2018, 193, 8–16. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Chen, G.; Chen, G.; Chen, G. Adsorption of copper and zinc by biochars produced from pyrolysis of hardwood and corn straw in aqueous solution. Bioresour. Technol. 2011, 102, 8877–8884. [Google Scholar] [CrossRef]
- Xiao, F.; Cheng, J.; Cao, W.; Yang, C.; Chen, J.; Lou, Z. Removal of heavy metals from aqueous solution using chitosan-combined magnetic biochars. J. Colloid Interface Sci. 2019, 540, 579–584. [Google Scholar] [CrossRef]
- Dilks, R.; Monette, F.; Glaus, M. The major parameters on biomass pyrolysis for hyperaccumulative plants: A review. Chemosphere 2016, 146, 385–395. [Google Scholar] [CrossRef]
- Fletcher, J.; Smith, A.; Heinemeyer, A.; Lord, R.; Ennis, J.; Hodgson, M.; Farrar, K. Production factors controlling the Physical Characteristics of biochar Derived from phytoremediation Willow for agricultural Applications. Bioenergy Res. 2013, 7, 371–380. [Google Scholar] [CrossRef]
- Chami, A.; Amer, N.; Smets, K.; Yperman, J.; Carleer, R.; Dumontet, S.; Vangronsveld, J. Evaluation of flash and slow pyrolysis applied on heavy metal contaminated Sorghum bicolor shoots resulting from phytoremediation. Biomass Bioenergy 2014, 63, 268–279. [Google Scholar] [CrossRef]
- Ying, Y.; Bin, G.; Jian, J.C.; Ming, Z.; Man, D.; Yu, C.L.; Liu, Y.Y. Engineered carbon (biochar) prepared by direct pyrolysis of Mg-accumulated tomato tissues: Characterization and phosphate removal potential. Bioresour. Technol. 2013, 138, 8–13. [Google Scholar]
- Zhou, Q.; Liao, B.; Lin, L.; Qiu, W.W.L.; Song, Z.G. Adsorption of Cu(II) and Cd(II) from aqueous solutions by ferromanganese binary oxide-biochar composites. Sci. Total Environ. 2018, 615, 115–122. [Google Scholar] [CrossRef]
- Liu, B.; Tan, F.; Liu, G. Production of biochars from Ca impregnated ramie biomass (Boehmeria nivea (L.) Gaud.) and their phosphate removal potential. RSC Adv. 2016, 6, 5871–5880. [Google Scholar] [CrossRef]
- Zhou, L.; Liu, Y.G.; Liu, S.B.; Yin, Y.C.; Zeng, G.M.; Tan, X.F.; Hu, X.; Hu, X.J.; Jiang, L.H.; Ding, Y.; et al. Investigation of the adsorption-reduction mechanisms of hexavalent chromium by ramie biochars of different pyrolytic temperatures. Bioresour. Technol. 2016, 218, 351–359. [Google Scholar] [CrossRef]
- Liu, G.; Zhang, Z.; Zeng, M.; Huang, B.; Li, X. Heavy metal accumulation in plants on Mn mine tailings. Pedosphere 2006, 16, 131–136. [Google Scholar] [CrossRef]
- Xue, G.; Chen, X.; Reeves, D.; Baker, M.; Lin, Q.; Fernando, R. Manganese uptake and accumulation by the hyperaccumulator plant Phytolacca acinosa Roxb. (Phytolaccaeae). Environ. Pollut. 2004, 131, 393–399. [Google Scholar] [CrossRef]
- Jin, C.; Nan, Z.R.; Wang, H.C.; Jin, P. Plant growth and heavy metal bioavailability changes in a loess subsoil amended with municipal sludge compost. J. Soils Sediments 2017, 17, 2797–2809. [Google Scholar] [CrossRef]
- Tessier, A.; Campbell, P.G.C.; Bisson, M. Sequential extraction procedure for the speciation of particulate trace metals. Anal. Chem. 1979, 51, 844–851. [Google Scholar] [CrossRef]
- Chen, S.; Tong, C.; Qun, H.; Jun, W.; Sheng, L.; Jian, Y. Enhanced adsorption for Pb(II) and Cd(II) of magnetic rice husk biochar by KMnO4 modification. Environ. Sci. Pollut. R. 2019, 26, 8902–8913. [Google Scholar]
- Chen, H.; Wang, Y.Y.; Wei, Y.X.; Peng, L.; Li, G.; Yu, G.L.; Du, C.Y. Wet flue gas desulfurization wastewater treatment with reclaimed water treatment plant sludge: A case study. Water Sci. Technol. 2018, 78, 2392–2403. [Google Scholar] [CrossRef] [PubMed]
- Guang, T.; Yu, W.; Yong, L.; Dan, X. Removal of Pb(II) ions from aqueous solution by manganese oxide coated rice straw biochar-A low-cost and highly effective sorbent. J. Taiwan Inst. Chem. Eng. 2018, 7, 177–191. [Google Scholar]
- Mishra, S.; Dwivedi, J.; Kumar, A.; Sankararamakrishnan, N. Removal of antimonite (Sb(iii)) and antimonate (Sb(v)) using zerovalent iron decorated functionalized carbon nanotubes. RSC Adv. 2016, 6, 95865–95878. [Google Scholar] [CrossRef]
- Chang, L.; Lei, Z.; Yuan, G.; Aimin, L. Facile synthesis of nano ZnO/ZnS modified biochar by directly pyrolyzing of zinc contaminated corn stover for Pb(II), Cu(II) and Cr(VI) removals. Waste Manag. 2018, 79, 625–637. [Google Scholar]
- Jian, C.; Hai, Z.; Hong, C.; Yang, Y.; Ming, C.; Yan, C.; Fei, H.; Guo, Z.; Meng, Y. Spent Ganoderma lucidum substrate derived biochar as a new bio-adsorbent for Pb2+/Cd2+ removal in water. Chemosphere 2019. [CrossRef]
- Wang, H.; Gao, B.; Wang, S.; Fang, J.; Xue, Y.; Kai, Y. Removal of Pb(II), Cu(II), and Cd(II) from aqueous solutions by biochar derived from KMnO4 treated hickory wood. Bioresour. Technol. 2015, 197, 356–362. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shen, S.; Wang, L.; Tzou, M.; Yan, Y.; Kuan, W. Removal of hexavalent Cr by coconut coir and derived chars-the effect of surface functionality. Bioresour. Technol. 2012, 104, 165–172. [Google Scholar] [CrossRef] [PubMed]
- Sheng, Z.H.; Shao, L.; Chen, J.J.; Bao, W.J.; Wang, F.B.; Xia, X.H. Catalyst-Free Synthesis of Nitrogen-Doped Graphene via Thermal Annealing Graphite Oxide with Melamine and Its Excellent Electrocatalysis. ACS Nano 2011, 5, 4350–4358. [Google Scholar] [CrossRef]
- Geng, Z.B.; Wang, Y.X.; Liu, J.H.; Li, G.S.; Li, L.P.; Huang, K.K.; Yuan, L.; Feng, S.H. δ-MnO2-Mn3O4 Nanocomposite for pHotochemical Water Oxidation: Active Structure Stabilized in the Interface. ACS Appl. Mater. Interfaces 2016, 8, 27825–27831. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Wang, B.; Lee, X.; Lehmann, J.; Gao, B. Sorption and desorption of Pb(II) to biochar as affected by oxidation and pH. Sci. Total Environ. 2018, 634, 188–194. [Google Scholar] [CrossRef] [Green Version]
- Park, M.; Han, H.; Chu, H.; Her, N.; Heo, J.; Yoon, Y. Influence of solution pH, ionicstrength, and humic acid on cadmium adsorption onto activated biochar: Experiment and modeling. J. Ind. Eng. Chem. 2017, 48, 186–193. [Google Scholar] [CrossRef]
- Jiang, X.; Rui, H.; Guangcai, C. Micro-nano-engineered nitrogenous bone biochar developed with a ball-milling technique for high-efficiency removal of aquatic Cd(II), Cu(II) and Pb(II). J. Hazard. Mater. 2019. [Google Scholar] [CrossRef]
- Wan, L.; Qiu, L.; Tang, G.; Chen, Y.; Li, Y.; He, F. Ultrafast sequestration of cadmium and lead from water by manganese oxide supported on a macro-mesoporous biochar. Chem. Eng. J. 2020. [Google Scholar] [CrossRef]
- Lee, J.; Park, H.; Ahn, T.; Chung, W. Comparison of heavy metal adsorption by peat moss and peat moss-derived biochar produced under different carbonization conditions. Water Air Soil Pollut. 2015, 226, 9–20. [Google Scholar] [CrossRef]
- James, B.; Laura, R.; Robert, D. Ion exchange removal of Cu(II), Fe(II), Pb(II) and Zn(II) from acid extracted sewage sludge-resin screening in weak acid media. Water Res. 2019, 158, 257–267. [Google Scholar]
- Luo, C.; Wei, R.; Guo, D.; Zhang, S.; Yan, S. Adsorption behavior of MnO2 functionalized multi-walled carbon nanotubes for the removal of cadmium from aqueous solutions. Chem. Eng. J. 2013, 225, 406–415. [Google Scholar] [CrossRef]
- Zhang, Y.; Duan, M. Chemical precipitation of heavy metals from wastewater by using the synthetical magnesium hydroxy carbonate. Water Sci. Technol. 2020, 81, 1130–1136. [Google Scholar] [CrossRef]
- Wang, R.Z.; Huang, D.L.; Liu, Y.G.; Zhang, C.; Lai, C.; Zeng, G.M.; Cheng, M.; Gong, X.M.; Wan, J.; Luo, H. Investigating the adsorption behavior and the relative distribution of Cd(2+) sorption mechanisms on biochars by different feedstock. Bioresour. Technol. 2018, 261, 265–271. [Google Scholar] [CrossRef]
- Irene, L.; Zhang, H. EDTA-Enhanced Washing for Remediation of Pb and or Zn Contaminated Soils. J. Environ. Eng. 2006, 132, 1282–1288. [Google Scholar]
- Deng, J.; Liu, Y.; Liu, S.; Zeng, G.; Tan, X.; Huang, B.; Tang, X.; Wang, S.; Hua, Q.; Yan, Z. Competitive adsorption of Pb(II), Cd(II) and Cu(II) onto chitosan-pyromellitic dianhydride modified biochar. J. Colloid Interface Sci. 2017, 506, 355–364. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Ge, H. Adsorption of chromium(VI) from aqueous solution using a novel chitosan biguanidine. J. Dispers. Sci. Technol. 2015, 36, 1106–1114. [Google Scholar] [CrossRef]
- Hong, W.; Zhuang, Z.T.; Ling, J.; Wen, W.L.; Zheng, G.W.; Shi, Y.L.; Jing, C.; Wei, W. Highly efficient adsorption of Cr(VI) from aqueous solution by Fe3+ impregnated biochar. J. Dispers. Sci. Technol. 2017, 38, 815–825. [Google Scholar]
- Iskandar, N.; Zainudin, N.; Tan, S. Tolerance and biosorption of copper (Cu) and (Pb) by filamentous fungi ioslated from a freshwater ceosytem. J. Environ. Sci. (Engl. Ed.) 2011, 23, 824–830. [Google Scholar] [CrossRef]
- Liang, J.; Li, X.; Yu, Z.; Zeng, G.; Lou, Y.; Jiang, L.; Yang, Z.; Qian, Y.; Wu, H. Amorphous MnO2 modified biochar derived from aerobically composted swine manure for adsorption of Pb(II) and Cd(II). ACS Sustain. Chem. Eng. 2017, 5, 5049–5058. [Google Scholar] [CrossRef]
- Chen, S.; Yue, Q.; Gao, B.; Xu, X. Equilibrium and kinetic adsorption study of the adsorptive removal of Cr(VI) using modifed wheat residue. J. Colloid Interface Sci. 2010, 349, 256–264. [Google Scholar] [CrossRef] [PubMed]
- Acharya, J.; Sahu, J.; Mohanty, C.; Meikap, B. Removal of ledf(II) from wastewater activated carbon developed from Tamrind wood by zinc chloride activation. Chem. Eng. J. 2009, 149, 249–262. [Google Scholar] [CrossRef]
- You, D.; Min, X.; Liu, L.; Ren, Z.; Xiao, X.; Pavlostathis, S.G.; Luo, J.M.; Luo, X.B. New insight on the adsorption capacity of metallogels for antimonite and antimonate removal: From experimental to theoretical study. J. Hazard. Mater. 2018, 346, 218–225. [Google Scholar] [CrossRef]
- Devi, P.; Saroha, A. Risk analysis of pyrolyzed biochar made from paper mill effluent treatment plant sludge for bioavailability and ecotoxicity of heavy metals. Bioresour. Technol. 2014, 162, 308–315. [Google Scholar] [CrossRef]
- He, Y.D.; Zhai, Y.B.; Li, C.T.; Yang, F.; Chen, L.; Fan, X.P.; Peng, W.F.; Fu, Z.M. The fate of Cu, Zn, Pb and Cd during the pyrolysis of sewage sludge at different temperatures. Environ. Technol. 2010, 31, 567–574. [Google Scholar] [CrossRef] [PubMed]
- Yang, B.; Gong, Q.; Zhao, L.; Sun, H.; Ren, N.; Qin, J.; Xu, J.; Yang, H. Preconcentration and determination of lead and cadmium in water samples with a MnO2 coated carbon nanotubes by using ETAAS. Desalination 2011, 278, 65–69. [Google Scholar] [CrossRef]
- Hakanson, L. An ecological risk index for aquatic pollution control: A sedimentological approach. Water Res. 1980, 14, 975–1001. [Google Scholar] [CrossRef]






| Sample | Surface Area (m2·g−1) | Pore Volume (m3·g−1) |
|---|---|---|
| BC300 | 5.564 | 0.013 |
| BC400 | 9.416 | 0.011 |
| BC500 | 20.233 | 0.014 |
| Sample | C% | N% | H% | O% | H/C | O/C |
|---|---|---|---|---|---|---|
| BC300 | 61.6 | 0.93 | 4.62 | 28.724 | 0.075 | 0.46 |
| BC400 | 65.83 | 0.83 | 3.87 | 24.442 | 0.058 | 0.37 |
| BC500 | 69.49 | 0.51 | 3.5 | 21.91 | 0.05 | 0.31 |
| Adsorbent | PFO | PSO | ||||
|---|---|---|---|---|---|---|
| Qe (mg·g−1) | k1 | R2 | Qe (mg·g−1) | k2 | R2 | |
| BC300-Cu2+ | 174.52597 | 0.02544 | 0.83879 | 185.55783 | 1.97835 | 0.96389 |
| BC400-Cu2+ | 199.43948 | 0.02895 | 0.69816 | 212.80717 | 1.87106 | 0.88866 |
| BC500-Cu2+ | 227.52499 | 0.02527 | 0.79485 | 242.06866 | 1.51741 | 0.94866 |
| BC300-Pb2+ | 154.68393 | 0.00964 | 0.93444 | 160.15713 | 9.96497 | 0.96666 |
| BC400-Pb2+ | 190.51352 | 0.02642 | 0.60892 | 229.21786 | 2.50427 | 0.87913 |
| BC500-Pb2+ | 280.64762 | 0.05335 | 0.66377 | 315.52997 | 2.60974 | 0.88855 |
| BC300-Cd2+ | 214.25541 | 0.06576 | 0.59985 | 252.24123 | 5.1848 | 0.86809 |
| BC400-Cd2+ | 229.54123 | 0.06143 | 0.55518 | 275.27413 | 4.36213 | 0.83837 |
| BC500-Cd2+ | 242.57136 | 0.07693 | 0.53173 | 306.54189 | 5.54803 | 0.88314 |
| Adsorbent | Langmuir | Freundlich | ||||
|---|---|---|---|---|---|---|
| Qm (mg g−1) | KL | R2 | KF | n | R2 | |
| BC300-Cu2+ | 188.18668 | 28.02274 | 0.96239 | 128.6307 | 0.07737 | 0.79598 |
| BC400-Cu2+ | 222.20807 | 20.67084 | 0.98487 | 187.66081 | 0.0343 | 0.51224 |
| BC500-Cu2+ | 248.30945 | 10.33169 | 0.98888 | 166.26842 | 0.08327 | 0.65712 |
| BC300-Pb2+ | 170.12479 | 70.51657 | 0.6445 | 140.66621 | 0.03573 | 0.55412 |
| BC400-Pb2+ | 237.54248 | 31.24189 | 0.9487 | 166.11347 | 0.07216 | 0.77502 |
| BC500-Pb2+ | 328.39095 | 17.43978 | 0.99035 | 217.32022 | 0.08652 | 0.69727 |
| BC300-Cd2+ | 263.34945 | 25.55479 | 0.9713 | 176.87751 | 0.08143 | 0.80421 |
| BC400-Cd2+ | 284.33611 | 22.21888 | 0.98114 | 186.82251 | 0.08687 | 0.80392 |
| BC500-Cd2+ | 311.37099 | 18.9919 | 0.98834 | 202.64684 | 0.09002 | 0.77071 |
| Er | Potential Ecological Risk | RI | Biochar Contamination |
|---|---|---|---|
| Er < 40 | Low | RI ≤ 50 | Low |
| 40 ≤ Er < 80 | Moderate | 50 < RI ≤ 100 | Moderate |
| 80 ≤ Er < 160 | Considerable | 100 ≤ RI < 200 | Considerable |
| 160 ≤ Er < 320 | High | RI > 200 | High |
| Er ≥ 320 | Very high | ||
| Samples | Parameter | Mn | Zn | Cu | Pb |
|---|---|---|---|---|---|
| Biomass | Cf | 7.33 | 11.5 | 8.09 | 9 |
| Er | 7.33 | 11.5 | 40.45 | 45 | |
| RI | 104.28 | ||||
| BC300 | Cf | 3.76 | 9 | 6.69 | 4.26 |
| Er | 3.76 | 9 | 33.45 | 21.3 | |
| RI | 67.51 | ||||
| BC400 | Cf | 1.5 | 2.7 | 1.94 | 1.43 |
| Er | 1.5 | 2.7 | 9.7 | 7.15 | |
| RI | 21.05 | ||||
| BC500 | Cf | 0.81 | 1.89 | 0.72 | 0.92 |
| Er | 0.81 | 1.89 | 3.6 | 4.6 | |
| RI | 10.9 | ||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yang, Z.; Hu, W.; Yao, B.; Shen, L.; Jiang, F.; Zhou, Y.; Núñez-Delgado, A. A Novel Manganese-Rich Pokeweed Biochar for Highly Efficient Adsorption of Heavy Metals from Wastewater: Performance, Mechanisms, and Potential Risk Analysis. Processes 2021, 9, 1209. https://doi.org/10.3390/pr9071209
Yang Z, Hu W, Yao B, Shen L, Jiang F, Zhou Y, Núñez-Delgado A. A Novel Manganese-Rich Pokeweed Biochar for Highly Efficient Adsorption of Heavy Metals from Wastewater: Performance, Mechanisms, and Potential Risk Analysis. Processes. 2021; 9(7):1209. https://doi.org/10.3390/pr9071209
Chicago/Turabian StyleYang, Zhe, Wenyong Hu, Bin Yao, Liangchen Shen, Feifeng Jiang, Yaoyu Zhou, and Avelino Núñez-Delgado. 2021. "A Novel Manganese-Rich Pokeweed Biochar for Highly Efficient Adsorption of Heavy Metals from Wastewater: Performance, Mechanisms, and Potential Risk Analysis" Processes 9, no. 7: 1209. https://doi.org/10.3390/pr9071209
APA StyleYang, Z., Hu, W., Yao, B., Shen, L., Jiang, F., Zhou, Y., & Núñez-Delgado, A. (2021). A Novel Manganese-Rich Pokeweed Biochar for Highly Efficient Adsorption of Heavy Metals from Wastewater: Performance, Mechanisms, and Potential Risk Analysis. Processes, 9(7), 1209. https://doi.org/10.3390/pr9071209








