The Role of Heterotrophic Microalgae in Waste Conversion to Biofuels and Bioproducts
Abstract
1. Introduction
2. Autotrophic Versus Heterotrophic Mode for Microalgal Waste Stream Treatment
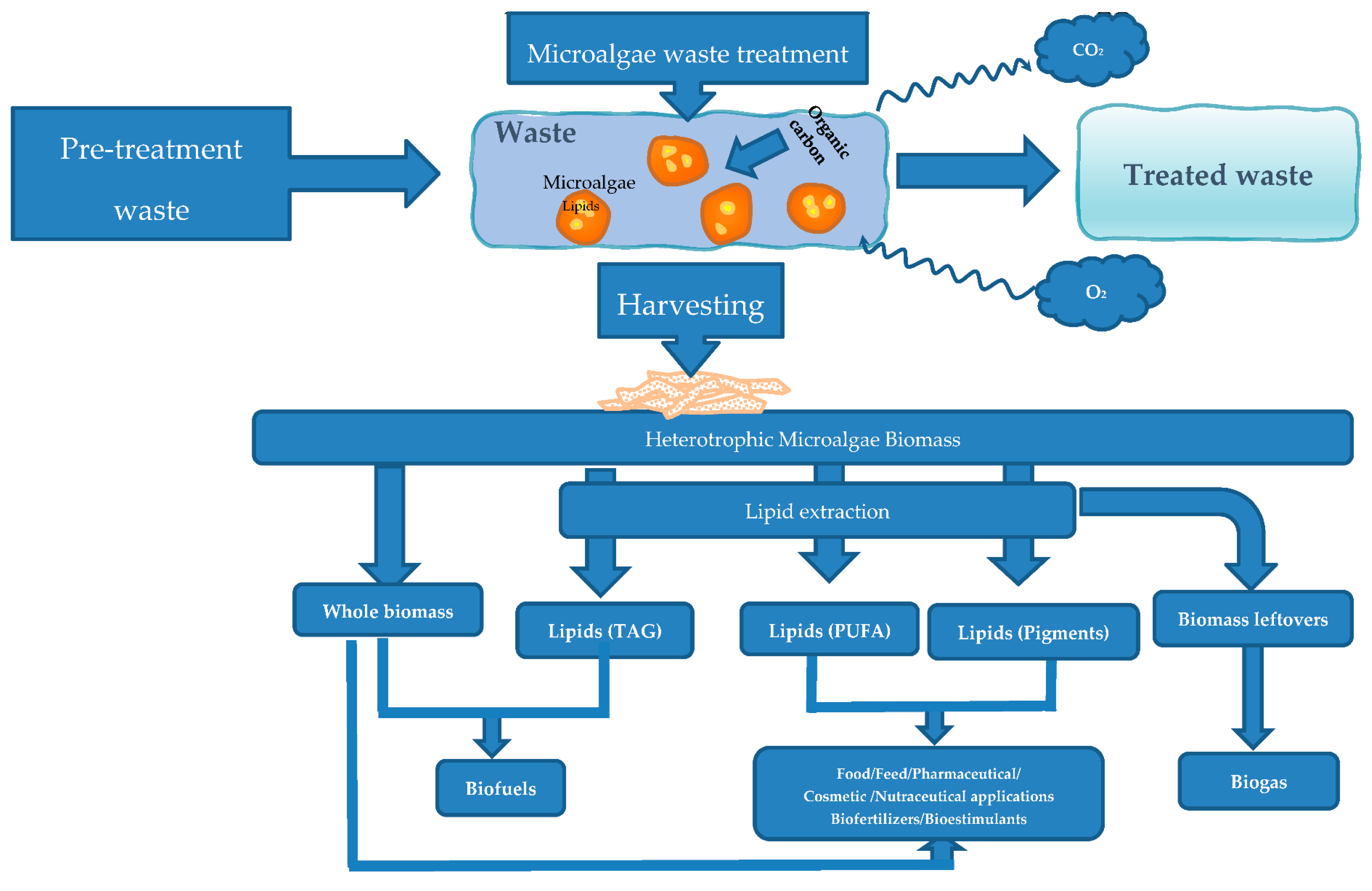
3. Heterotrophic Microalgae Waste Treatment
3.1. Heterotrophic Metabolism
3.2. Waste Pre-Treatment Step
3.3. Strains
3.4. Media Supplementation
3.5. Microalgae Heterotrophic Wastewater Treatment Efficiency
4. Microalgal Products Obtained from Heterotrophic Growth
4.1. Products Extraction
4.2. Microalgal Products
5. Heterotrophic Microalgae Waste Treatment Process Monitoring
6. Drawbacks and Bottlenecks
7. Heterotrophic/Autotrophic Microalgae Waste Treatment Economic Evaluation
8. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Waste Collection Systems Assessed and Good Practices Identified. Available online: https://www.collectors2020.eu/ (accessed on 19 April 2021).
- Gray, N.F. Biology of Wastewater Treatment, 2nd ed.; Imperial College Press: London, UK, 2004; pp. 1–936. [Google Scholar]
- Grady, L.; Daigger, G.; Love, N.; Filipe, C. Biological Wastewater Treatment, 3rd ed.; CRC Press: Abingdon, UK, 2011; pp. 1–1395. [Google Scholar]
- UN-Water Annual Report 2015. Available online: www.unwater.org (accessed on 19 April 2021).
- Lopes da Silva, T.; Moniz, P.; Silva, C.; Reis, A. The Dark Side of Microalgae Biotechnology: A Heterotrophic Biorefinery Platform Directed to omega-3 Rich Lipid Production. Microorganisms 2019, 7, 670. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.T.; Yang, Y.; Keller, A.A.; Li, X.; Feng, S.J.; Dong, Y.N.; Li, F.T. Comparative analysis of energy intensity and carbon emissions in wastewater treatment in USA, Germany, China and South Africa. Appl. Energy 2016, 148, 873–881. [Google Scholar] [CrossRef]
- Mohsenpour, S.F.; Hennige, S.; Willoughby, N.; Adeloye, A.; Gutierrez, T. Integrating micro-algae into wastewater treatment: A review. Sci. Total Environ. 2021, 752, 1–25. [Google Scholar] [CrossRef]
- Mehariya, S.; Goswami, R.K.; Karthikeysan, O.P.; Verma, P. Microalgae for high-value products: A way towards green nutraceutical and pharmaceutical compounds. Chemosphere 2021, 280, 1–23. [Google Scholar] [CrossRef] [PubMed]
- Mehariya, S.; Goswami, R.K.; Verma, P.; Lavecchia, R.; Zuorro, A. Integrated Approach for Wastewater Treatment and Biofuel Production in Microalgae Biorefineries. Energies 2021, 14, 2282. [Google Scholar] [CrossRef]
- Goswami, R.K.; Mehariya, S.; Karthikeyan, O.P.; Verma, P. Advanced microalgae-based renewable biohydrogen production systems: A review. Bioresour. Technol. 2020, 320, 1–15. [Google Scholar]
- Goswami, R.K.; Mehariya, S.; Verma, P.; Lavecchia, R.; Zuorro, A. Microalgae-based biorefineries for sustainable resource recovery from wastewater. J. Water Process. Eng. 2021, 40, 1447–1463. [Google Scholar] [CrossRef]
- Bhatia, S.K.; Mehariya, S.; Bhatia, R.K.; Kumar, M.; Pugazhendhi, A.; Awasthi, M.K.; Atabani, A.E.; Kumar, G.; Kim, W.; Seo, S.O.; et al. Wastewater based microalgal biorefinery for bioenergy production. Prog. Chall. Sci. Total Environ. 2021, 751, 141599. [Google Scholar] [CrossRef] [PubMed]
- Perez-Garcia, O.; Escalante, F.M.E.; de-Bashan, L.E.; Bashan, Y. Heterotrophic cultures of microalgae: Metabolism and potential products. Water Res. 2011, 45, 11–36. [Google Scholar] [CrossRef]
- Girard, J.M.; Tremblay, R.; Faucheux, N.; Heitz, M.; Deschenes, J.S. Phycoremediation of cheese whey permeate using directed commensalism between Scenedesmus obliquus and Chlorella protothecoides. Algal Res. 2017, 22, 122–126. [Google Scholar] [CrossRef]
- Shen, L.; Ndayambaje, J.D.; Murwanashyaka, T.; Cui, W.Q.; Manirafasha, E.; Chen, C.X.; Wang, Y.P.; Lu, Y.H. Assessment upon heterotrophic microalgae screened from wastewater microbiota for concurrent pollutants removal and biofuel production. Bioresour. Technol. 2017, 245, 386–393. [Google Scholar] [CrossRef]
- Abdel-Raouf, N.; Al-Homaidan, A.A.; Ibraheem, I.B.M. Microalgae and wastewater treatment. Saudi J. Biol. Sci. 2012, 19, 257–275. [Google Scholar] [CrossRef] [PubMed]
- Quijano, G.; Arcila, J.S.; Buitron, G. Microalgal-bacterial aggregates: Applications and perspectives for wastewater treatment. Biotechnol. Adv. 2017, 35, 772–781. [Google Scholar] [CrossRef]
- Debowski, M.; Zielinski, M.; Kazimierowicz, J.; Kujawska, N.; Talbierz, S. Microalgae Cultivation Technologies as an Opportunity for Bioenergetic System Development-Advantages and Limitations. Sustainability 2020, 12, 9980. [Google Scholar] [CrossRef]
- Perez-Garcia, O.; Bashan, Y.; Puente, M.E. Organic Carbon Supplementation of Sterilized Municipal Wastewater Is Essential for Heterotrophic Growth and Removing Ammonium by the Microalga Chlorella Vulgaris. J. Phycol. 2011, 47, 190–199. [Google Scholar] [CrossRef] [PubMed]
- Morales-Sanchez, D.; Martinez-Rodriguez, O.A.; Martinez, A. Heterotrophic cultivation of microalgae: Production of metabolites of commercial interest. J. Chem. Technol. Biotechnol. 2017, 92, 925–936. [Google Scholar] [CrossRef]
- Ende, S.S.W.; Noke, A. Heterotrophic microalgae production on food waste and by-products. J. Appl. Phycol. 2019, 31, 1565–1571. [Google Scholar] [CrossRef]
- Xu, H.; Miao, X.; Wu, Q. High quality biodiesel production from a microalga Chlorella protothecoides by heterotrophic growth in fermenters. J. Biotechnol. 2006, 126, 499–507. [Google Scholar] [CrossRef]
- Scaife, M.A.; Merkx-Jacques, A.; Woodhall, D.L.; Armnta, R.E. Algal biofuels in Canada: Status and potential. Renew. Sustain. Energy Rev. 2015, 44, 620–642. [Google Scholar] [CrossRef]
- Chen, Y.H.; Walker, T.H. Fed-batch fermentation and supercritical fluid extraction of heterotrophic microalgal Chlorella protothecoides lipids. Bioresour. Technol. 2012, 114, 512–517. [Google Scholar] [CrossRef]
- Taborda, T.; Moniz, P.; Reis, A.; da Silva, T.L. Evaluating low-cost substrates for Crypthecodinium cohnii lipids and DHA production, by flow cytometry. J. Appl. Phycol. 2021, 33, 263–274. [Google Scholar] [CrossRef]
- Mendes, A.; Guerra, P.; Madeira, V.; Ruano, F.; da Silva, T.L.; Reis, A. Study of docosahexaenoic acid production by the heterotrophic microalga Crypthecodinium cohnii CCMP 316 using carob pulp as a promising carbon source. World J. Microbiol. Biotechnol. 2007, 23, 1209–1215. [Google Scholar] [CrossRef]
- De Swaaf, M.E.; Pronk, J.T.; Sijtsma, L. Fed-batch cultivation of the docosahexaenoic-acid-producing marine alga Crypthecodinium cohnii on ethanol. Appl. Microbiol. Biotechnol. 2003, 61, 40–43. [Google Scholar] [CrossRef] [PubMed]
- Ratledge, C.; Kanagachandran, K.; Anderson, A.J.; Grantham, D.J.; Stephenson, J.C. Production of docosahexaenoic acid by Crypthecodinium cohnii grown in a pH-auxostat culture with acetic acid as principal carbon source. Lipids 2001, 36, 1241–1246. [Google Scholar] [CrossRef] [PubMed]
- Sabeela Beevi, U.; Sukumaran, R.K. Cultivation of the fresh water microalga Chlorococcum sp. RAP13 in sea water for producing oil suitable for biodiesel. J. Appl. Phycol. 2015, 27, 141–147. [Google Scholar] [CrossRef]
- Isleten-Hosoglu, M.; Elibol, M. Improvement of medium composition and cultivation conditions for growth and lipid production by Crypthecodinium cohnii. Rom. Biotechnol. Lett. 2017, 22, 13086–13095. [Google Scholar]
- Yazdani, S.S.; Gonzalez, R. Anaerobic fermentation of glycerol: A path to economic viability for the biofuels industry. Curr. Opin. Biotechnol. 2007, 18, 213–219. [Google Scholar] [CrossRef]
- Samul, D.; Leja, K.; Grajek, W. Impurities of crude glycerol and their effect on metabolite production. Ann. Microbiol. 2014, 64, 891–898. [Google Scholar] [CrossRef]
- Zhang, T.Y.; Wu, Y.H.; Zhuang, L.L.; Wang, X.X.; Hu, H.Y. Screening heterotrophic microalgal strains by using the Biolog method for biofuel production from organic wastewater. Algal Res. 2014, 6, 175–179. [Google Scholar]
- Gaurav, K.; Srivastava, R.; Sharma, J.G.; Singh, R.; Singh, V. Molasses-based growth and lipid production by Chlorella pyrenoidosa: A potential feedstock for biodiesel. Int. J. Green Energy 2016, 13, 320–327. [Google Scholar] [CrossRef]
- Guldhe, A.; Singh, P.; Kumari, S.; Rawat, I.; Permaul, K.; Bux, F. Biodiesel synthesis from microalgae using immobilized Aspergillus niger whole cell lipase biocatalyst. Renew. Energy 2016, 85, 1002–1010. [Google Scholar] [CrossRef]
- Hena, S.; Abida, N.; Tabassum, S. Screening of facultative strains of high lipid producing microalgae for treating surfactant mediated municipal wastewater. Res. Adv. 2015, 5, 98805–98813. [Google Scholar] [CrossRef]
- Sloth, J.K.; Jensen, H.C.; Pleissner, D.; Eriksen, N.T. Growth and phycocyanin synthesis in the heterotrophic microalga Galdieria sulphuraria on substrates made of food waste from restaurants and bakeries. Bioresour. Technol. 2017, 238, 296–305. [Google Scholar] [CrossRef]
- Mendes, A.; Lopes da Silva, T.; Reis, A. DHA concentration and purification from the marine heterotrophic microalga Crypthecodinium cohnii CCMP 316 by winterization and urea complexation. Food Technol. Biotechnol. 2007, 45, 38–44. [Google Scholar]
- Chi, Z.Y.; Hu, B.; Liu, Y.; Frear, C.; Wen, Z.Y.; Chen, S.L. Production of omega-3 polyunsaturated fatty acids from cull potato using an algae culture process. Appl. Biochem. Biotechnol. 2007, 137, 805–815. [Google Scholar] [PubMed]
- Devi, M.P.; Subhash, G.V.; Mohan, S.V. Heterotrophic cultivation of mixed microalgae for lipid accumulation and wastewater treatment during sequential growth and starvation phases: Effect of nutrient supplementation. Renew. Energy 2012, 43, 276–283. [Google Scholar] [CrossRef]
- Zheng, H.L.; Gao, Z.; Yin, F.W.; Ji, X.J.; Huang, H. Lipid production of Chlorella vulgaris from lipid-extracted microalgal biomass residues through two-step enzymatic hydrolysis. Bioresour. Technol. 2012, 117, 1–6. [Google Scholar] [CrossRef]
- Pleissner, D.; Lam, W.C.; Sun, Z.; Lin, C.S.K. Food waste as nutrient source in heterotrophic microalgae cultivation. Bioresour. Technol. 2013, 137, 139–146. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.Y.; Wu, Y.H.; Zhu, S.F.; Li, F.M.; Hu, H.Y. Isolation and heterotrophic cultivation of mixotrophic microalgae strains for domestic wastewater treatment and lipid production under dark condition. Bioresour. Technol. 2013, 149, 586–589. [Google Scholar] [CrossRef] [PubMed]
- Gong, Y.; Liu, J.; Jiang, M.; Liang, Z.; Jin, H.; Hu, X.; Wan, X.; Hu, C. Improvement of Omega-3 Docosahexaenoic Acid Production by Marine Dinoflagellate Crypthecodinium cohnii Using Rapeseed Meal Hydrolysate and Waste Molasses as Feedstock. PLoS ONE 2015, 10, 1–18. [Google Scholar] [CrossRef]
- Nguyen, H.C.; Su, C.H.; Yu, Y.K.; Huong, D.T.M. Sugarcane bagasse as a novel carbon source for heterotrophic cultivation of oleaginous microalga Schizochytrium sp. Ind. Crop. Prod. 2018, 121, 99–105. [Google Scholar] [CrossRef]
- Almeida, M.C.; Butler, D.; Friedler, E. At-source domestic wastewater quality. Urban Water J. 1999, 1, 49–55. [Google Scholar] [CrossRef]
- Sajadian, S.F.; Morowvat, M.H.; Ghasemi, Y. Investigation of autotrophic, heterotrophic, and mixotrophic modes of cultivation on lipid and biomass production in Chlorella Vulgaris. Natl. J. Physiol. Pharm. Pharmacol. 2018, 8, 594–599. [Google Scholar] [CrossRef]
- Zhang, T.-Y.; Wu, Y.-H.; Wang, J.-H.; Wang, X.-X.; Victor, M.D.-E.; Dao, G.-H.; Tong, X.; Hu, H.-Y. Heterotrophic cultivation of microalgae in straw lignocellulose hydrolysate for production of high-value biomass rich in polyunsaturated fatty acids (PUFA). Chem. Eng. J. 2019, 367, 37–44. [Google Scholar] [CrossRef]
- Couto, R.M.; Simões, P.C.; Reis, A.; Da Silva, T.L.; Martins, V.H.; Sánchez-Vicente, Y. Supercritical fluid extraction of lipids from the heterotrophic microalga Crypthecodinium cohnii. Eng. Life Sci. 2010, 10, 158–164. [Google Scholar]
- Ward, O.P.; Singh, A. Omega-3/6 fatty acids: Alternative sources of production. Process Biochem. 2005, 40, 3627–3652. [Google Scholar] [CrossRef]
- Lowrey, J.; Armenta, R.E.; Brooks, M.S. Nutrient and media recycling in heterotrophic microalgae cultures. Appl. Microbiol. Biotechnol. 2016, 100, 1061–1075. [Google Scholar] [CrossRef]
- Bohutskyi, P.; Kula, T.; Kessler, B.A.; Hong, Y.; Bouwer, E.J.; Betenbaugh, M.J.; Allnutt, F.C.T. Mixed Trophic State Production Process for Microalgal Biomass with High Lipid Content for Generating Biodiesel and Biogas. Bioenergy Res. 2014, 7, 1174–1185. [Google Scholar] [CrossRef]
- Doucha, J.; Lívanský, K. Production of high-density Chlorella culture grown in fermenters. J. Appl. Phycol. 2012, 24, 35–43. [Google Scholar] [CrossRef]
- Li, X.; Xu, H.; Wu, Q. Large-scale biodiesel production from microalga Chlorella protothecoides through heterotrophic cultivation in bioreactors. Biotechnol. Bioeng. 2007, 98, 764–771. [Google Scholar] [CrossRef] [PubMed]
- Oliver, L.; Dietrich, T.; Marañón, I.; Villarán, M.C.; Barrio, R.J. Producing Omega-3 Polyunsaturated Fatty Acids: A Review of Sustainable Sources and Future Trends for the EPA and DHA Market. Resources 2020, 9, 148. [Google Scholar] [CrossRef]
- Eusébio, A.; Moniz, P.; Lopes da Silva, T.; Marques, I.P. Energetic valorization of Chrypthecodinium cohnii lipid production wastes by anaerobic digestion. In Proceedings of the MicroBiotec’19 Congress, Coimbra, Portugal, 5–7 December 2019. [Google Scholar]
- Lopes da Silva, T.; Reis, A. Low-Cost Feedstocks for Biofuels and High Value Added Products Production: Using Multi- Parameter Flow Cytometry as a Tool to Enhance the Process Efficiency. In Recent Advances in Analytical Techniques; Atta-ur-Rahman Sibel, A., Ozkan, U.A.E., Eds.; Bentham Science Publishers: Sharjah, United Arab Emirates, 2019; Volume 3, pp. 40–60. [Google Scholar]
- Norvill, Z.N.; Shilton, A.; Guieysse, B. Emerging contaminant degradation and removal in algal wastewater treatment ponds: Identifying the research gaps. J. Hazard. Mater. 2016, 313, 291–309. [Google Scholar] [CrossRef] [PubMed]
- Hu, W.; Gladue, R.; Hansen, J.; Wojnar, C.; Chalmers, J.J. The sensitivity of the dinoflagellate Crypthecodinium cohnii to transient hydrodynamic forces and cell-bubble interactions. Biotechnol. Prog. 2007, 23, 1355–1362. [Google Scholar] [CrossRef] [PubMed]
- Yeung, P.K.; Lam, C.M.; Ma, Z.Y.; Wong, Y.H.; Wong, J.T. Involvement of calcium mobilization from caffeine-sensitive stores in mechanically induced cell cycle arrest in the dinoflagellate Crypthecodinium cohnii. Cell Calcium 2006, 39, 259–274. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Wei, D.; Pohnert, G. Rapid Estimation of Astaxanthin and the Carotenoid-to-Chlorophyll Ratio in the Green Microalga Chromochloris zofingiensis Using Flow Cytometry. Mar. Drugs 2017, 15, 231. [Google Scholar] [CrossRef]
- Santos, C.A.; Nobre, B.; da Silva, T.L.; Pinheiro, H.M.; Reis, A. Dual-mode cultivation of Chlorella protothecoides applying inter-reactors gas transfer improves microalgae biodiesel production. J. Biotechnol. 2014, 184, 74–83. [Google Scholar] [CrossRef][Green Version]
- Ferro, L. Wastewater Treatment and Biomass Generation by Nordic Microalgae: Growth in Subarctic Climate and Microbial Interactions. Ph.D. Thesis, Umeå University, Umeå, Sweden, 2019. Comprehensive Summary. [Google Scholar]
- Yang, H.L.; Lu, C.K.; Chen, S.F.; Chen, Y.M.; Chen, Y.M. Isolation and Characterization of Taiwanese Heterotrophic Microalgae: Screening of Strains for Docosahexaenoic Acid (DHA) Production. J. Mar. Biotechnol. 2010, 12, 173–185. [Google Scholar] [CrossRef] [PubMed]
- Behrens, P. Photobioreactors and fermentors: The light and dark side of growing algae. In Algal Culturing Techniques; Andersen, R.A., Ed.; Elsevier Academic Press: Cambridge, MA, USA, 2005; pp. 189–204. [Google Scholar]
- Orfield, N.D.; Levine, R.B.; Keoleian, G.A.; Miller, S.A.; Savage, P.E. Growing Algae for Biodiesel on Direct Sunlight or Sugars: A Comparative Life Cycle Assessment. ACS Sustain. Chem. Eng. 2015, 3, 386–395. [Google Scholar] [CrossRef]
- Hu, J.J.; Nagarajan, D.; Zhang, Q.G.; Chang, J.S.; Lee, D.J. Heterotrophic cultivation of microalgae for pigment production: A review. Biotechnol. Adv. 2018, 36, 54–67. [Google Scholar] [CrossRef] [PubMed]
- Parsons, S.; Abeln, F.; McManus, M.C.; Chuck, C.J. Techno-economic analysis (TEA) of microbial oil production from waste resources as part of a biorefinery concept: Assessment at multiple scales under uncertainty. J. Chem. Technol. Biotechnol. 2019, 94, 701–711. [Google Scholar] [CrossRef]
- Fei, Q.; Chang, H.N.; Shang, L.; Choi, J.; Kim, N.; Kang, J. The effect of volatile fatty acids as a sole carbon source on lipid accumulation by Cryptococcus albidus for biodiesel production. Bioresour. Technol. 2011, 102, 2695–2701. [Google Scholar] [CrossRef] [PubMed]
- Fei, Q.; Fu, R.; Shang, L.; Brigham, C.J.; Chang, H.N. Lipid production by microalgae Chlorella protothecoides with volatile fatty acids (VFAs) as carbon sources in heterotrophic cultivation and its economic assessment. Bioprocess Biosyst. Eng. 2015, 38, 691–700. [Google Scholar] [CrossRef]
- Sano Coelho, R.; Cuellar, M.C.; Franco, T.T.; van der Wielen, L.A.M. Techno-economic assessment of heterotrophic microalgae biodiesel production integrated with a sugarcane bio-refinery. Biofuel Bioprod. Biorefining 2021, 15, 416–429. [Google Scholar] [CrossRef]
- Pleissner, D.; Smetana, S. Estimation of the economy of heterotrophic microalgae- and insect-based food waste utilization processes. Waste Manag. 2020, 102, 198–203. [Google Scholar] [CrossRef] [PubMed]
- Santos, A.; Roso, G.; Menezes, C.; Queiroz, M.; Zepka, L.; Jacob-Lopes, E. The bioeconomy of microalgal heterotrophic bioreactors applied to agroindustrial wastewater treatment. Desalination Water Treat. 2017, 64, 12–20. [Google Scholar] [CrossRef]
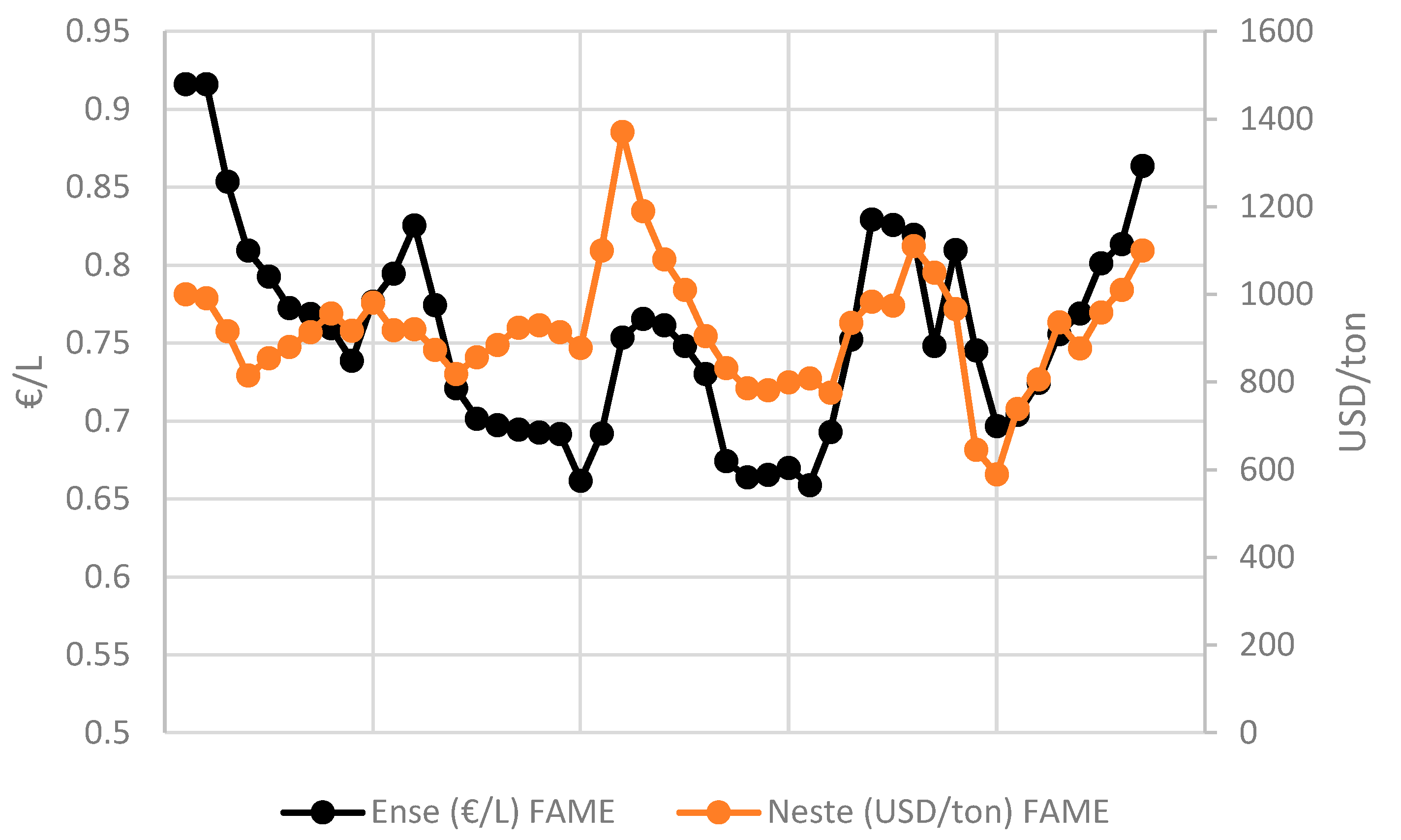
- Biodiesel Prices (SME & FAME). Available online: https://www.neste.com/investors/market-data/biodiesel-prices-sme-fame#97f21970 (accessed on 20 March 2021).
- Asselin, M.; Drogui, P.; Benmoussa, H.; Blais, J.F. Effectiveness of electrocoagulation process in removing organic compounds from slaughterhouse wastewater using monopolar and bipolar electrolytic cells. Chemosphere 2008, 72, 1727–1733. [Google Scholar] [CrossRef]
- Cristóvão, R.O.; Botelho, C.M.; Martins, R.J.E.; Loureiro, J.M.; Boaventura, R.A.R. Fish canning industry wastewater treatment for water reuse—A case study. J. Clean. Prod. 2015, 87, 603–612. [Google Scholar] [CrossRef]
- Smetana, S.; Sandmann, M.; Rohn, S.; Pleissner, D.; Heinz, V. Autotrophic and heterotrophic microalgae and cyanobacteria cultivation for food and feed: Life cycle assessment. Bioresour. Technol. 2017, 245, 162–170. [Google Scholar] [CrossRef]

| Autotrophic Cultures | Heterotrophic Cultures | |
|---|---|---|
| Advantages |
|
|
| Drawbacks |
|
|
| Waste | Pre-Treatment | System/Duration of the Experiment | Nutrients Removal | Products | Observations | Reference | |
|---|---|---|---|---|---|---|---|
| Chrypthecodinium cohnii CCMP 316 | Carob pulp syrup | Carob pulp residues were mixed with distilled water (1:2 w/w) for the syrup extraction, carried out at 150 rev/min for 6 h at 50 °C. Thereafter, the syrup was pressed and squeezed, and the supernatant was filtered and centrifuged. The liquid fraction was acidified to pH 2, in order to promote sucrose hydrolysis, and stored at −18 °C. | 2 L bioreactor, fed-batch, | Lipids: 9.2% w/w DHA: 1.99 g /L; 45.2 mg/g DCW DHA/TFA: 48% (w/w) | [38] | ||
| Schizochytrium limacinum SR 21 | Cull potato | Cull potato was boiled and minced and mixed with water in a 5-L tank with agitation. α-amylase and glucoamylase were used to hydrolyze the potato starch into glucose. | 150 mL Erlenmeyer/6 days | DHA: 5.35 g/L | 50% of hydrolyzed potato broth in the culture medium was used with 20 g/L glucose supplementation | [39] | |
| Scenedesmus sp. and Chlorella sp. | Acid rich effluent collected from a bench scale anaerobic sequencing batch reactor (AnSBR) operated with composite food waste | 250 mL conical flasks with 180 mL liquid medium/11 days | COD: 91.4 ± 0.6% | Biomass: 1.42 g/L Lipids: 26.4 (w/w) | Microalgae cultures flasks were grown at a photo period of 12 h sunlight and 12 h dark | [40] | |
| Chlorella sp. | Lipid extracted microalgae biomass residues (LMBs) + sugarcane molasses Mixture ratio: 1/4 (v/v) | LMBRs: 50 g lyophilized LMBRs dissolved in distilled water were hydrolyzed using cellulase, neutrase, and alcalase; Crude molasses: Dilution with distilled water (1:9); followed by hydrolysis with neutrase and alcalase, followed by the acidic hydrolysis (addition of H2SO4 5 M, adjusting pH to 3.5, at 60 °C for 1 h). | 500 mL Flasks with 300 mL liquid medium, 150 rpm, 25 °C/7 days | Biomass: 5.6 g/L Lipids: 43% (w/w) | [41] | ||
| Schizochytrium mangrovei | Food waste (rice, noodles, meat, vegetables collected from canteens) | Food waste hydrolysis using Aspergillus awamori and Aspergillus oryzae fungal glucoamylases, proteases, and phosphatases. | 2 L bioreactor batch mode/7 days | Lipids: 3.3 g/L; 16.5% (w/w); DHA: 85.5 ± 11.2 mg/g | [42] | ||
| Chlorella pyrenoidosa | Lipids: 1.050 g/L; 20.99%(w/w) DHA: 0 | ||||||
| Scenedesmus sp. ZTY2, Scenedesmus sp. ZTY3 Chlorella sp. ZTY4 | Domestic wastewater | Centrifugation followed by sterilization | 500 mL shake flasks /11 days | Lipids Scenedesmus sp. ZTY2: 69.1% (w/w) Scenedesmus sp. ZTY3: 52.9% w/w Chlorella sp. ZTY4: 64.4% (w/w) Biomass, respectively: 0.04, 0.045, 0.054 g/L | [43] | ||
| Chlorococcum sp. RAP-13 | Dairy effluent supplemented with 6% biodiesel industry waste glycerol | Stored at 4 °C in sterilized containers before use | 500 mL with 200 mL untreated effluent /15 days | COD: 93% BOD: 82% | Biomass: 1.96 g/L Lipids: 42% w/w | [29] | |
| Chrypthecodinium cohnii ATCC 30772 | Rapeseed meal hydrolysate + crude waste molasses | Rapeseed meal: solid-state fermentation using Aspergillus oryzae LZ01, Penicillium oxalicum J1, and Neurospora crassa J2 fungal strains; Crude waste molasses: diluted with distilled water and acidified to pH 3.0, heated to 100 °C for 1 h, followed by centrifugation | 500 mL-Erlenmeyers, batch, 7 days | Lipids: 27.3% (w/w) (w/w DCW); 26.9 g/L DHA: 8.7 mg /L; DHA/TFA: 22–34% w/w | [44] | ||
| Scenedesmus sp., Chlamydomonas sp., Chlorococcum humicola, Botryococcus braunii Chlorella sp., Chlorella sp. | Surfactant mediated municipal wastewater (SMMW) | Stored at 4 °C to minimize substrate decomposition, followed by mixing for 5 min, followed by a 30 min settling, to allow the settlement of excess colloidal particles | COD > 98% PO4-P > 98% NO3-N > 99% NH4-N ~ 100% | Biomass: 0.41–0.50 g/L Lipids: 21.5–42.0% (w/w) | Previous facultative heterotrophic strains screening for the capacity to grow in SMMW | [36] | |
| Chlorella pyrenoidosa (NCIM 2738) | Treated cane molasses | Dilution 1:1 with distilled water followed by centrifugation at 2000 rpm, 30 min. The clarified liquid was passed through a strong acidic cation exchange resin for the removal of metal ions. | 250 mL flasks containing 100 mL liquid medium | Total sugars: 92.2% | Biomass: 1.2 g/L Lipids: 66% (w/w) | [34] | |
| Scenedesmus obliquus, Chlorella protothecoides | Cheese whey permeate | Filtration (0.2 µm) | Shake flasks; sequential cultivation of S. obliquus and C. protothecoides/34 days | Total lactose: 62% | Biomass Scenedesmus obliquus: 8 ± 0.2 g L−1 Chlorella protothecoides: 6.2 ± 0.4 g L−1 | S. obliquus and C. protothecoides sequential cultivation | [14] |
| Chlorella sorokiniana | Aquaculture wastewater supplemented with 400 mg/L NaNO3 | Filtration using glass fiber filter papers | 1 L Shake flasks containing 500 mL/7 days | COD: 71.9% PO4-P: 73.6% NO3 –N: 84.5% NH4-N: 75.6% | Biomass: 3.5 g/L Lipids: 30.2% (w/w) Carbohydrates: 34.7% (w/w) Proteins: 28.4% (w/w) | [35] | |
| Crypthecodinium cohnii CCMP 316 | Cheese Whey + Corn Steep Liquor | 250 mL-Erlenmeyers, | Lipids: 28.7% (w/w DCW) DHA/TFA: 8.5–27% w/w | [30] | |||
| Galdieria sulphuraria 074G | Food waste from restaurants and bakeries | Autoclaving for 30 min at 121 °C, followed by enzymatic hydrolysis for 24 h at 50 °C and pH 4.5 to produce a hydrolysate rich in sugars and amino acids/peptides | 500 mL conical flasks containing 150 mL of liquid medium | Phycocyanin: 20–22 mg/g | [37] | ||
| Schizochytrium sp. BCRC 33482 | Sugarcane bagasse with 40 g/L glucose | Alkali followed by phosphoric acid treatment, followed by enzymatic hydrolysis (cellulase) | 250 mL Flasks with 50 mL liquid medium/ 72 h for the pre-treatment step: 120 h for the microalgae cultivation | Biomass: 10. 5 g/L Lipids: 45.2% (w/w) | [45] | ||
| Crypthecodinium cohnii ATCC 30772 | Sugarcane molasses and distilled crude glycerol | Sugarcane molasses: hydrolysis by pH drop to 3 with HCl; storage at 50 °C for 24 h; thereafter, the pH was raised to 6.5 using NaOH 50% (w/w) Crude glycerol previously distilled to remove methanol | 500 mL shake flasks with 200 mL liquid medium/192 h | Molasses: Glucose: 100% Fructose: 0% Crude Glycerol: 100% | Sugarcane molasses: Lipids: 11.2% (w/w) DHA 5.5 mg/g DCW DHA/TFA: 49.6% (w/w) Crude Glycerol: Lipids: 14.7% (w/w) DHA: 6.6 mg/g DCW DHA/TFA: 44.7% (w/w) | [25] |
| Carbon Source | System | TEA Method | Conclusions | Reference |
|---|---|---|---|---|
| Food waste | Batch; Chlorella protothecoides in heterotrophic cultures (max 48.7% lipid content; max 0.187 g/g yield) Product: biodiesel | OPEX w/carbon source cost (30$/ton), NH4Cl $150/ton, utilities cost of $0.035/kg (0.1 $/kWh−1); labor cost of $0.056/kg; general works $0.029/kg including maintenance, supervision, administration, property taxes, and insurance. CAPEX not included. w/o co-product valuation | cost of biodiesel $0.6/L compared with glucose-based biodiesel $3.79/L | [70] |
| Molasses from sugar cane processing | Scale-up simulation on SuperPro Designer™ Auxenochlorella protothecoides (50% lipid content; lipid 0.25 g/g yield) Product: Biodiesel and animal feed | Lipid productivity 8.2 gL−1 d−1, 330 working days, 10 ML bioreactors CAPEX 130 M$-160 M$ (function of equipment cost covering equipment Installation, pipping, electrical, buildings, design and engineering, Contractor’s fee and contingency) OPEX 50–60 M$/year including materials costs + utilities costs ($ 0.058/kWh) + waste treatment costs ($1.5 × 10−3 kg−1) + labor costs ($11.50 per work hour) + facility related costs 30-year net present value (NPV) and internal rate of return (IRR) Co-product animal feed ($ 0.45/kg) | Biodiesel price range Minimum 2.6–3.0 $/liter for IRR 12% | [71] |
| Food waste | Simulation of process scale-up 56.3 t of wet food waste per day 7.14 t dried biomass per day Chlorella pyrenoidosa to food and feed | 300 working days/year CAPEX (equipment) 3.3 M€ OPEX (materials, utilities, labor 8 €/h) 31.9 k€/day Dry biomass revenue 36 k€/ton | 20 years NPV w/ 5% discount rate 948 M€ In comparison with autotrophic microalgae for food/feed applications, heterotrophic microalgae cultvation operational cost is up to 50% lower | [72] |
| Agroindustrial wastewater (poultry and swine slaughterhouse wastewater) | Up-scale wastewater flow rate 16,000 m3/day from bench-scale facility data Phormidium | Working 24 h/d and 336 days/year CAPEX 71 M$ (equipment, installation, deployment, instrumentation, piping) OPEX 14 M$/year (raw materials and supplies, utilities, labor costs ($8.50/h), supervision, payroll charges, maintenance, operating supplies, general plant overheads, tax, and contingency) | 10-year lifetime facility $2.66/m3 ($ 0.70/m³ considering only operational costs) Potential revenue dry biomass production cost $ 0.03/kg (much lower than conventional heterotephic fermenters and autotrophic photobioreactors, and below theoretical target of 0.55 $/kg) Return on investment in the first year of operation | [73] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Silva, T.L.d.; Moniz, P.; Silva, C.; Reis, A. The Role of Heterotrophic Microalgae in Waste Conversion to Biofuels and Bioproducts. Processes 2021, 9, 1090. https://doi.org/10.3390/pr9071090
Silva TLd, Moniz P, Silva C, Reis A. The Role of Heterotrophic Microalgae in Waste Conversion to Biofuels and Bioproducts. Processes. 2021; 9(7):1090. https://doi.org/10.3390/pr9071090
Chicago/Turabian StyleSilva, Teresa Lopes da, Patrícia Moniz, Carla Silva, and Alberto Reis. 2021. "The Role of Heterotrophic Microalgae in Waste Conversion to Biofuels and Bioproducts" Processes 9, no. 7: 1090. https://doi.org/10.3390/pr9071090
APA StyleSilva, T. L. d., Moniz, P., Silva, C., & Reis, A. (2021). The Role of Heterotrophic Microalgae in Waste Conversion to Biofuels and Bioproducts. Processes, 9(7), 1090. https://doi.org/10.3390/pr9071090









