Capture and Reuse of Carbon Dioxide (CO2) for a Plastics Circular Economy: A Review
Abstract
1. Introduction
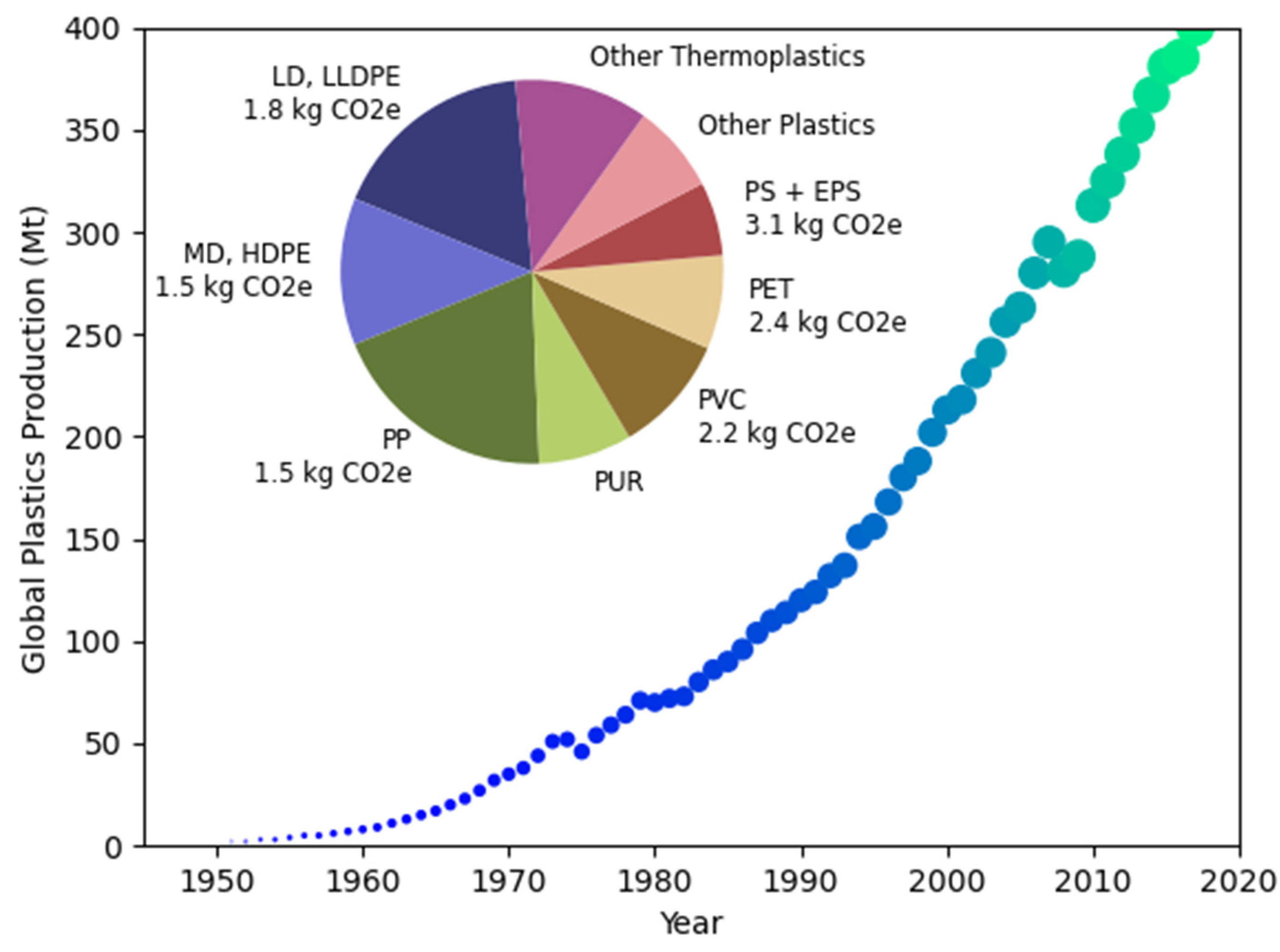
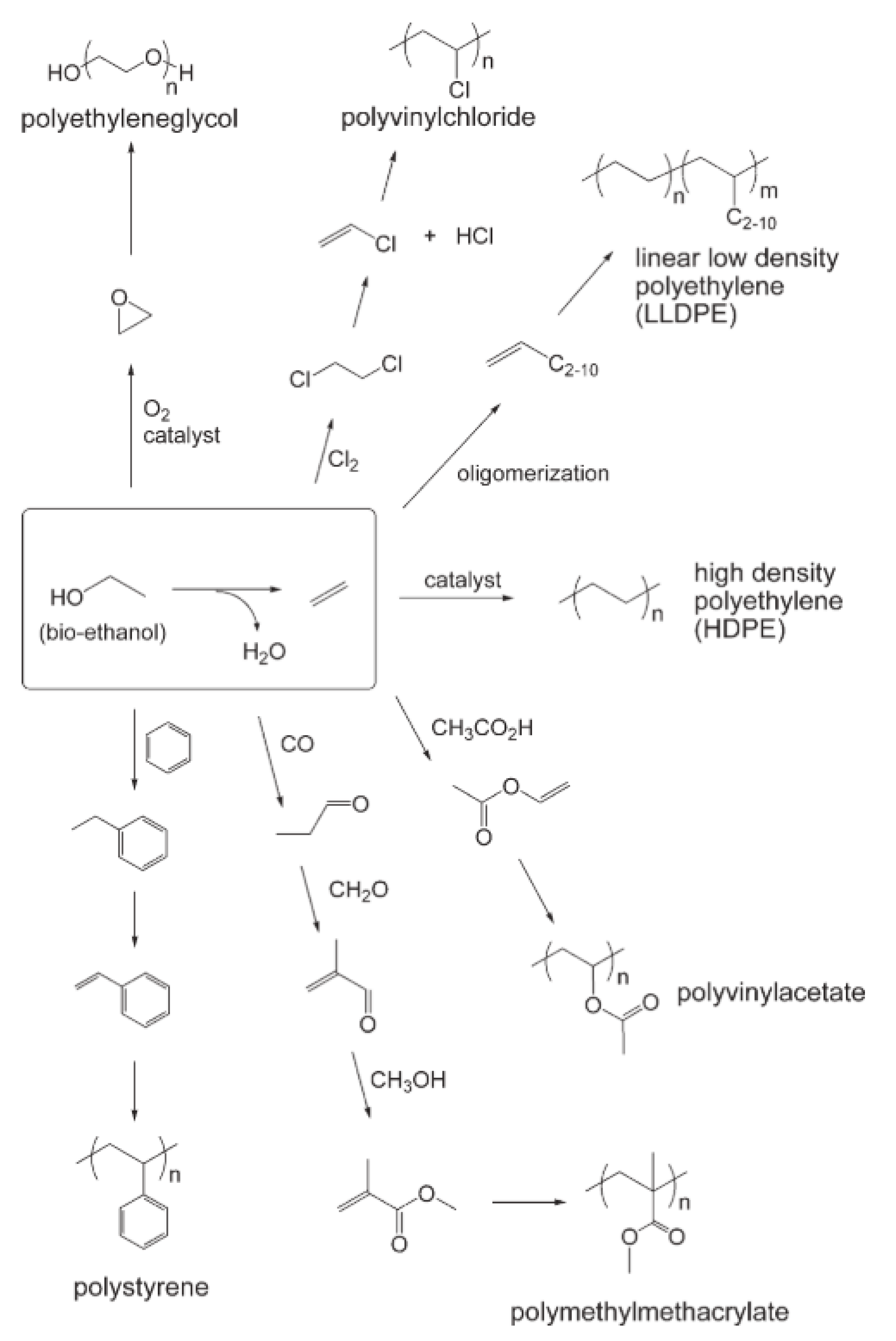
1.1. Carbon Footprint and the Plastics Industries
1.2. The Role of Carbon Capture
- response to society demands;
- development of innovative processes and products;
- attainment of circular economy conditions through chemical recycling of plastic wastes and carbon;
- production of liquid fuels and integration within the existing refining infrastructure;
- attainment of the United Nations Sustainability Development Goals (SDGs), in order to ensure access to affordable, reliable, sustainable, and modern energy; build resilient infrastructure; promote inclusive and sustainable industrialization and foster innovation; make cities and human settlements inclusive, safe, resilient, and sustainable; take urgent action to combat climate change and its impacts;
- improvement of the public image of plastic products;
- obtainment of emission reduction credits.
2. Methodology
2.1. Search Methods and Procedures (Phase 1)
2.2. Search Methods and Procedures (Phase 2)
3. Results
3.1. Bibliometric Analysis of Scientific Review Papers: General Information
3.1.1. The Annual Distribution
3.1.2. The Scientific Journals Distribution
3.1.3. The Country and Institutions Distribution
3.1.4. The Most Cited Review Papers
3.2. Bibliometric Analysis of Scientific Review Papers: Technical Information
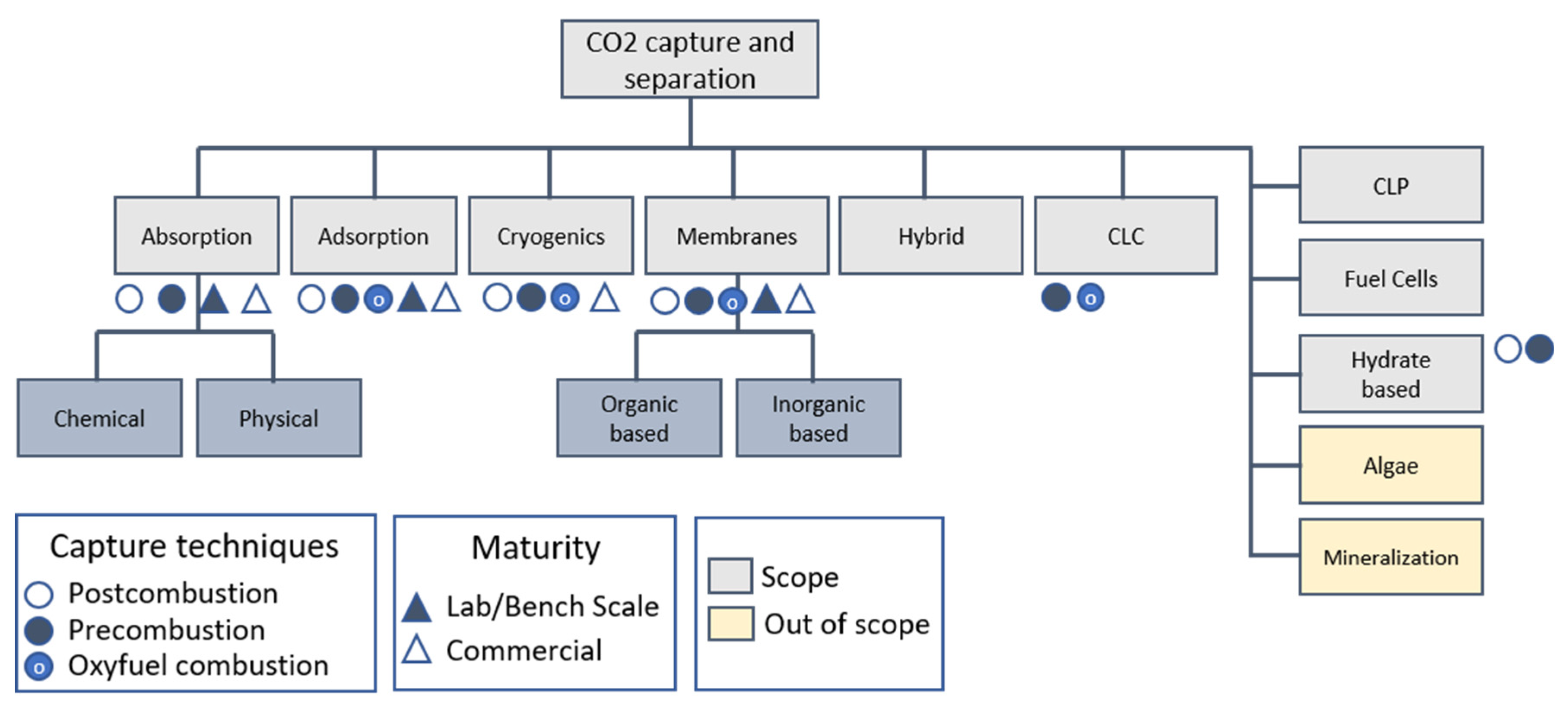
3.2.1. Carbon Capture
- CO2 Source: Fuel-Fire Power and Heat Generation Stage
3.2.2. CO2 Separation Techniques
- Absorption
- Adsorption
- Biofixation
- Membrane separations
- Hybrid technologies
- Cryogenic separation
- Other separation techniques
- Additional comments
3.3. Bibliometric Analysis of Patents Literature and Technological Players
| Code | Priority Year | Status | Name | Technology | Product | Ref. |
|---|---|---|---|---|---|---|
| WO2007117157A1 | 2006 | Application (2007) Grant in USA | Microbial fermentation of gaseous substrates to produce alcohols | Bioconversion | Alcohols | [370] |
| WO2015058011A1 | 2014 | Application (2015) Grant in CA | Carbon capture in fermentation | Bioconversion | Ethanol, acetate, and/or 2,3-butanediol | [371] |
| Code | Priority Year | Status | Name | Technology | Product | Ref. |
|---|---|---|---|---|---|---|
| WO2019133726A1 | 2018 | Application | Genetic modulation of photosynthetic organisms for improved growth | Bioconversion | Biofuel | [372] |
| WO2017095960A1 | 2016 | Application Grant in USA | Compositions and methods for expressing genes in algae | Bioconversion | Biofuel | [373] |
| WO2017070404A2 | 2016 | Application Grant in USA | Enhanced productivity by attenuation of chlorophyll binding protein genes | Bioconversion | Biofuel | [374] |
| WO2017041048A1 | 2016 | Application Grant in USA | Microorganisms engineered for increased productivity | Bioconversion | Biofuel | [375] |
| WO2017011707A1 | 2016 | Application Grant in USA | Microorganisms having increased lipid productivity | Bioconversion | Biofuel | [375] |
| WO2015103307A1 | 2014 | Application Grant in USA and EP. | Biomass productivity regulator | Bioconversion | Biofuel | [376] |
| WO2015051342A2 | 2014 | Application Grant in USA | Compositions and methods for modulating biomass productivity | Bioconversion | Biofuel | [377] |
| WO2009098089A2 | 2009 | Application Grant in USA | Genetically modified cyanobacteria for the production of ethanol | Bioconversion | Biofuel | [378] |
| Code | Priority Year | Status | Name | Technology | Ref. |
|---|---|---|---|---|---|
| US7682821B2 | 2006 | Grant (2010) | Closed photobioreactor system for continued daily in situ production, separation, collection, and removal of ethanol from genetically enhanced photosynthetic organisms | Conversion by cyanobacteria | [379] |
| US8691538B1 | 2012 | Grant (2014) | Biofilm photobioreactor system and method of use | Conversion by microorganisms | [380] |
| US9896652B2 | 2014 | Grant (2018) | Photobioreactor, system and method of use | Conversion by microorganisms | [381] |
| US89121012B2 | 2013 | Grant (2015) | Staged inoculation of multiple cyanobacterial photobioreactors | Conversion by cyanobacteria | [382] |
| WO2014145185A1 | 2008 | Application (2010) | Process for inoculating closed photobioreactors with cyanobacteria | Conversion by cyanobacteria | [383] |
| US8846369B2 | 2012 | Grant (2014) | Cyanobacterium sp. host cell and vector for production of chemical compounds in cyanobacterial cultures | Conversion by cyanobacteria | [384] |
| WO2014100799A3 | 2012 | Application (2014) Grant in USA and EP | Cyanobacterium sp. for production of compounds | Conversion by cyanobacteria | [385] |
| WO2007084477A1 | 2006 | Application (2007) Grant in USA, EP, JP, CA, DE, and ES | Methods and compositions for ethanol producing cyanobacteria | Conversion by cyanobacteria | [386] |
| WO2011072122A1 | 2009 | Application (2011) Grant in USA | Water/carbonate stripping for CO2 capture adsorber regeneration and CO2 delivery to photoautotrophs | Air capture and separation by adsorption | [387] |
Patents Regarding CO2 Capture Deposited by Plastic and Resin Manufacturers
| Company | Country | Technology | Technology Description | Scale | Patents in CO2 Capture |
|---|---|---|---|---|---|
| BASF | Germany | Chemical absorption | Chemical absorption mainly with amines (especially methyldiethanolamine), but also ammonia. | Large scale | 24 |
| ExxonMobil | USA | Adsorption; MCFCs | Amine or zeolite-based materials for adsorption of CO2. Desorption may be performed by a swing adsorption process. Development of MCFCs is also occurring in partnership with FuelCell Energy, Inc. | ֍ | 23 |
| Sumitomo Chemical | Japan | Membranes | Acid gas membrane separation to remove CO2 from synthesis gas, natural gas, exhaust gas, and gas streams that contain N2 and O2. Membranes contain at least one hydrophilic polymer layer. | Pilot unit | 9 |
| Reliance Industries | India | Absorption; adsorption | Chemical absorption with ionic liquid. Adsorption with oxides (temperature swing). | ֍ | 8 |
| ENI | Italy | Algae | Cultivation of microalgae for posterior biomass bioconversion into ethanol, butanol, and diesel. | Pilot | 5 |
| Dow Chemical | USA | Adsorption | Utilization of amine/alkanolamine for CO2 removal. | Pilot unit | 3 |
| Mitsubishi Chemical ▲ | Japan | Membrane | Zeolite membrane for removal from methane. | ֍ | 3 |
| Sabic | Saudi Arabia | Cryogenic separation; Chemical absorption | Removal of CO2 from syngas using aqueous solvents or cryogenic separation. | ֍ | 2 |
| Chevron Phillips | USA | Physical absorption | Use of ionic liquids to separate CO2. | ֍ | 1 [406] |
| Ineos | UK | Chemical absorption | Chemical absorption with amines. | Bench | 1 |
| Lanxess | Germany | Chemical absorption | Absorption using polystyrene-based resins that contain primary amines and are crosslinked with divinyl aromatics. | Pilot unit | 1 |
3.4. Bibliometric Analysis of Carbon Utilization: Technical Information
3.4.1. Electrochemical Cells
| Thermodynamic Electrochemical Half-Reactions | Electrode Potentials (V VS. SHE) |
|---|---|
3.4.2. Dry Reforming of Methane (DRM)
3.4.3. Hydrogenation
- Methane decomposition into hydrogen and solid carbon through thermal pyrolysis, avoiding the production of CO2 as a byproduct (although applications of the solid carbon byproduct are not fully developed and heat transfer problems must still be solved in the moving carbon bed reactor) [68];
- Biomass thermochemical decomposition in presence (gasification) or absence (pyrolysis) of oxygen at elevated temperatures, although the technological development of the field is still immature in many aspects and the process operation and blends of obtained products are highly dependent on the quality of available feedstocks [495], justifying the chemical looping gasification as an alternative strategy [430].
3.4.4. Methanation
3.4.5. Methanol-To-Olefin (MTO) Reaction
3.4.6. Polymerization (Polycarbonates and Polyurethanes)
3.4.7. Oxidative Dehydrogenation (ODH) of Light Alkanes to Alkenes with CO2
3.4.8. Overview of Olefin Production and Carbon Emissions
3.5. Bibliometric Analysis of Carbon Utilization: Patents and Players
| Company | Country | Technology | Technology Description | Product | Scale | No. of Patents in the Field |
|---|---|---|---|---|---|---|
| AGG Biofuel | USA | Boudouard reaction; WGS | Reaction of CO2 with C and steam at 1330 °C | Syngas | Pilot | 3 |
| Breathe | India | Hydrogenation | Hydrogenation of CO2 from flue gas using alloys/intermetallic/bimetallic/core-shell materials based on Cu, Ni, Fe catalysts | Methanol | Bench | 0 |
| BSE Engineering | USA | Hydrogenation | Process development to use excess electrical current and off-gas CO2 in parallel with catalysts development for CO2 hydrogenation to methanol | Methanol | Bench | 0 |
| C4X | Canada/China | Hydrogenation of cyclic carbonates | Hydrogenation of cyclic carbonates using metal–silica catalysts | Ethylene carbonate (EC), ethylene glycol (EG), methanol | Pilot | 0 |
| Carbon Engineering | Canada | Hydrogenation | Combination of H2 (split from water using renewable electricity) with captured atmospheric CO2 | Fuels (gasoline, diesel, jet-A), hydrocarbons, syngas | Bench | 1 |
| Carbon Recycling International | Iceland | Hydrogenation | Production of methanol from CO2, H2, and renewable electricity | Methanol | Large | 4 |
| CERT (University of Toronto) | Canada | Electrochemical reduction | Development of catalysts for the reduction of CO2 into CO in membrane electrode assembly (MEA) electrochemical cells, using a copper catalyst [544] | Ethylene and others | Bench | 0 |
| Covestro (Bayer spin-off) | Germany | Polymerization | Development of catalyst for producing polyether polyols and polyurethane from polyether carbonate polyol | Polyurethane | Large | 34 |
| Dimensional Energy (Cornell University startup) | USA | Photocatalytic hydrogenation | Use a photocatalyst to react CO2 with H2 using sunlight. | Syngas or methanol | Bench | 0 |
| Dioxide Materials | USA | Electrochemical reduction | Development of CO2 electrolyzers (the Sustainion® anion exchange membranes, AEMs, based on imidazolium functionalized polystyrene) [545], and a bifunctional catalyst: combination of silver nanoparticles and ionic liquid | Syngas, formic acid, hydrocarbons | Bench | 10 |
| Econic Technologies | UK | Polymerization | Development of catalyst technologies to build polyols from CO2 with epoxides and, further, polyurethane | Polyols and polyurethanes | Large | 6 |
| Hago energetics | USA | Boudouard reaction, RWGS | Conversion of CO2 with C, H2 (from CH4), and energy [546] | Char and syngas, further processed to methanol | Pilot | 1 |
| Newlight Technologies | USA | Bio(polymerization) | Conversion of air and CH4/CO2 into biodegradable plastics by bacteria | Polyhydroxyalkanoates (PHA) | Large | 5 |
| Novomer | USA | Polymerization | Reaction between CO2 with epoxides using a proprietary catalyst | Polyurethanes | Large | 13 |
| Sandia National Laboratories (SNL) (National Nuclear Security Administration, NNSA) | USA | Electrochemical reduction; | Electrochemical cell using an organometallic Zn; “solar reactor” (photovoltaics with an electrochemical cell) (Sandia’s Sunshine to Petrol, S2P), containing reduced cobalt-doped ferrite (FeO) that absorbs oxygen, leaving behind CO and ferrite, which is recycled [547,548]. | “Liquid fuels”: methanol, gasoline, jet fuel | Bench | 0 |
| Sunfire | Germany | Electrochemical reduction (power-to-methane) | Fuel cells; solid oxide electrolysis cell (SOEC) | Hydrogen, syngas, methane, methanol | Bench | 5 |
| OPUS 12 | USA | Electrochemical reduction | Membrane stack including a polymer electrolyte membrane (PEM) and catalysts | Syngas, methane, methanol, ethylene and others | Large | 2 |
Patents from Plastic and Resin Manufacturers Regarding CO2 Use
4. Conclusions, Challenges, and Future Trends
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Appendix A
References
- Andrady, A.L. Plastics and Environmental Sustainability, 1st ed.; John Wiley & Sons, Inc: Hoboken, NJ, USA, 2015; ISBN 9781119009405. [Google Scholar]
- Keller, F.; Lee, R.P.; Meyer, B. Life Cycle Assessment of Global Warming Potential, Resource Depletion and Acidification Potential of Fossil, Renewable and Secondary Feedstock for Olefin Production in Germany. J. Clean. Prod. 2020, 250, 119484. [Google Scholar] [CrossRef]
- Plastics and the Environment. Available online: https://www.planete-energies.com/en/medias/close/plastics-and-environment (accessed on 20 May 2020).
- The Ellen MacArthur Foundation. The New Plastics Economy: Rethinking the Future of Plastics & Catalysing Action. Available online: https://www.ellenmacarthurfoundation.org/publications/the-new-plastics-economy-rethinking-the-future-of-plastics-catalysing-action (accessed on 21 May 2020).
- Geyer, R.; Jambeck, J.R.; Law, K.L. Production, Use, and Fate of All Plastics Ever Made. Sci. Adv. 2017, 3, 5. [Google Scholar] [CrossRef]
- CIEL Plastics & Climate—The Hidden Costs of a Plastic Planet. Available online: www.ciel.org/plasticandclimate (accessed on 21 May 2020).
- Zheng, J.; Suh, S. Strategies to Reduce the Global Carbon Footprint of Plastics. Nat. Clim. Chang. 2019, 9, 374–378. [Google Scholar] [CrossRef]
- IEA The Future of Petrochemicals. Available online: https://www.iea.org/reports/the-future-of-petrochemicals (accessed on 3 November 2020).
- Malik, D.; Manchanda, P.; Jan Simons, T.; Wallach, J. The Impact of COVID-19 on the Global Petrochemical Industry. Available online: https://www.mckinsey.com/industries/chemicals/our-insights/the-impact-of-covid-19-on-the-global-petrochemical-industry (accessed on 20 May 2020).
- Glaun, D. The Plastic Industry Is Growing During COVID. Recycling? Not So Much. Available online: https://www.pbs.org/wgbh/frontline/article/the-plastic-industry-is-growing-during-covid-recycling-not-so-much/ (accessed on 1 March 2021).
- Parashar, N.; Hait, S. Plastics in the Time of COVID-19 Pandemic: Protector or Polluter? Sci. Total Environ. 2021, 759, 144274. [Google Scholar] [CrossRef]
- IEA Oil 2020. Available online: https://www.iea.org/reports/oil-2020 (accessed on 21 May 2020).
- IEA CO2 Emissions from Fuel Combustion: Overview. Available online: https://www.iea.org/reports/co2-emissions-from-fuel-combustion-overview (accessed on 9 May 2020).
- Elliott, D. Renewables; IOP Publishing: Philadelphia, PA, USA, 2013; ISBN 978-0-750-31040-6. [Google Scholar]
- Radu, A.L.; Scrieciu, M.A.; Caracota, D.M. Carbon Footprint Analysis: Towards a Projects Evaluation Model for Promoting Sustainable Development. Procedia Econ. Financ. 2013, 6, 353–363. [Google Scholar] [CrossRef]
- Mackay, A. Climate Change 2007: Impacts, Adaptation and Vulnerability. Contribution of Working Group II to the Fourth Assessment Report of the Intergovernmental Panel on Climate Change. J. Environ. Qual. 2008. [Google Scholar] [CrossRef]
- Eriksson, O.; Finnveden, G. Plastic Waste as a Fuel - CO2-Neutral or Not? Energy Environ. Sci. 2009, 2, 907. [Google Scholar] [CrossRef]
- Intergovernmental Panel on Climate Change. Climate Change 2014 Mitigation of Climate Change; Intergovernmental Panel on Climate Change: Geneva, Switzerland, 2014. [Google Scholar]
- PlasticsEurope Plastics—The Facts 2019. Available online: https://www.plasticseurope.org/application/files/9715/7129/9584/FINAL_web_version_Plastics_the_facts2019_14102019.pdf (accessed on 5 June 2020).
- PlasticsEurope Plastics—The Facts 2018. Available online: https://www.plasticseurope.org/application/files/6315/4510/9658/Plastics_the_facts_2018_AF_web.pdf (accessed on 20 May 2020).
- PlasticsEurope Plastics—The Facts 2017. Available online: https://www.plasticseurope.org/application/files/5715/1717/4180/Plastics_the_facts_2017_FINAL_for_website_one_page.pdf (accessed on 20 May 2020).
- PlasticsEurope Plastics—The Facts 2014/2015. Available online: https://www.plasticseurope.org/application/files/5515/1689/9220/2014plastics_the_facts_PubFeb2015.pdf (accessed on 20 May 2020).
- PlasticsEurope Plastics—The Facts 2020. Available online: https://www.plasticseurope.org/en/resources/publications/4312-plastics-facts-2020 (accessed on 20 February 2021).
- Stahel, W.R. The Circular Economy. Nature 2016, 531, 435–438. [Google Scholar] [CrossRef]
- Bruyn, S.D.; Jongsma, C.; Kampman, B.; Görlach, B.; Thie, J. Energy-Intensive Industries—Challenges and Opportunities in Energy Transition. Available online: https://cedelft.eu/publications/2475/energy-intensive-industries-challenges-and-opportunities-in-the-energy-transition (accessed on 3 February 2020).
- Worrell, E.; Phylipsen, D.; Einstein, D.; Martin, N. Energy Use and Energy Intensity of the U.S. Chemical Industry. Available online: https://www.osti.gov/servlets/purl/773773 (accessed on 5 May 2020).
- Gutowski, T.G.; Sahni, S.; Allwood, J.M.; Ashby, M.F.; Worrell, E. The Energy Required to Produce Materials: Constraints on Energy-Intensity Improvements, Parameters of Demand. Philos. Trans. R. Soc. A Math. Phys. Eng. Sci. 2013, 371, 20120003. [Google Scholar] [CrossRef]
- Sinha, R.K.; Chaturvedi, N.D. A Review on Carbon Emission Reduction in Industries and Planning Emission Limits. Renew. Sustain. Energy Rev. 2019, 114, 109304. [Google Scholar] [CrossRef]
- North, M.; Styring, P. Perspectives and Visions on CO2 Capture and Utilisation. Faraday Discuss. 2015, 183, 489–502. [Google Scholar] [CrossRef] [PubMed]
- Griffin, P.W. Radical Change in Energy Intensive UK Industry. Ph.D. Thesis, University of Bath, Bath, UK, May 2015. [Google Scholar]
- Griffin, P.W.; Hammond, G.P.; Norman, J.B. Opportunities for Energy Demand and Carbon Emissions Reduction in the Chemicals Sector. Energy Procedia 2017, 105, 4347–4356. [Google Scholar] [CrossRef]
- Nkwachukwu, O.; Chima, C.; Ikenna, A.; Albert, L. Focus on Potential Environmental Issues on Plastic World towards a Sustainable Plastic Recycling in Developing Countries. Int. J. Ind. Chem. 2013, 4, 34. [Google Scholar] [CrossRef]
- Franklin Associates; The American Chemistry Council (ACC); The Canadian Plastics Industry Association (CPIA). (CPIA) Impact of Plastics Packaging on Life Cycle Energy Consumption & Greenhouse Gas Emission in the United States and Canada: Substitution Analysis. Available online: https://plastics.americanchemistry.com/Education-Resources/Life-Cycle-Assessment-Study/Executive-Summary-Impact-of-Plastics-Packaging-on-Life-Cycle-Energy-Consumption.pdf (accessed on 9 October 2020).
- Turnau, S.; Mignot, C.S.; Mostert, C.; Bringezu, S. Material or Fuel: Comparative Cradle-to-Grave Climate and Material Footprint Analysis for the Use of Methanol from Recycled CO2. Green Chem. 2020. [Google Scholar] [CrossRef]
- Murcia Valderrama, M.A.; van Putten, R.J.; Gruter, G.J.M. The Potential of Oxalic—and Glycolic Acid Based Polyesters (Review). Towards CO2 as a Feedstock (Carbon Capture and Utilization—CCU). Eur. Polym. J. 2019, 119, 445–468. [Google Scholar] [CrossRef]
- Thunman, H.; Berdugo Vilches, T.; Seemann, M.; Maric, J.; Vela, I.C.; Pissot, S.; Nguyen, H.N.T. Circular Use of Plastics-Transformation of Existing Petrochemical Clusters into Thermochemical Recycling Plants with 100% Plastics Recovery. Sustain. Mater. Technol. 2019, 22, e00124. [Google Scholar] [CrossRef]
- Machado, C.F.R.; Araújo, O.D.Q.F.; de Medeiros, J.L.; de Brito Alves, R.M. Carbon Dioxide and Ethanol from Sugarcane Biorefinery as Renewable Feedstocks to Environment-Oriented Integrated Chemical Plants. J. Clean. Prod. 2018, 172, 1232–1242. [Google Scholar] [CrossRef]
- Amghizar, I.; Vandewalle, L.A.; Van Geem, K.M.; Marin, G.B. New Trends in Olefin Production. Engineering 2017, 3, 171–178. [Google Scholar] [CrossRef]
- Dean, C. Naphtha Catalytic Cracking for Propylene Production. Pet. Technol. Q. 2013, 18, 33–37. [Google Scholar]
- Sadrameli, S.M. Thermal/Catalytic Cracking of Hydrocarbons for the Production of Olefins: A State-of-the-Art Review I: Thermal Cracking Review. Fuel 2015, 140, 102–115. [Google Scholar] [CrossRef]
- United States Department of Energy Natural Gas Liquids Primer—With a Focus on the Appalachian Region. Available online: https://www.energy.gov/sites/prod/files/2018/07/f54/NGL_Primer.pdf (accessed on 8 October 2020).
- Ren, T.; Patel, M.; Blok, K. Olefins from Conventional and Heavy Feedstocks: Energy Use in Steam Cracking and Alternative Processes. Energy 2006, 31, 425–451. [Google Scholar] [CrossRef]
- Amghizar, I.; Dedeyne, J.N.; Brown, D.J.; Marin, G.B.; Van Geem, K.M. Sustainable Innovations in Steam Cracking: CO2 Neutral Olefin Production. React. Chem. Eng. 2020, 5, 239–257. [Google Scholar] [CrossRef]
- Johansson, D.; Sjöblom, J.; Berntsson, T. Heat Supply Alternatives for CO2 Capture in the Process Industry. Int. J. Greenh. Gas Control 2012, 8, 217–232. [Google Scholar] [CrossRef]
- Capture, C. Technology Roadmap: Carbon Capture and Storage in Industrial Applications. Encycl. Prod. Manuf. Manag. 2006, 781–782. [Google Scholar] [CrossRef]
- Weiss, M.; Neelis, M.; Blok, K.; Patel, M. Non-Energy Use of Fossil Fuels and Resulting Carbon Dioxide Emissions: Bottom-up Estimates for the World as a Whole and for Major Developing Countries. Clim. Chang. 2009, 95, 369–394. [Google Scholar] [CrossRef]
- Neelis, M.L.; Patel, M.; Gielen, D.J.; Blok, K. Modelling CO2 Emissions from Non-Energy Use with the Non-Energy Use Emission Accounting Tables (NEAT) Model. Resour. Conserv. Recycl. 2005, 45, 226–250. [Google Scholar] [CrossRef]
- Griffin, P.W.; Hammond, G.P.; Norman, J.B. Industrial Energy Use and Carbon Emissions Reduction in the Chemicals Sector: A UK Perspective. Appl. Energy 2018, 227, 587–602. [Google Scholar] [CrossRef]
- Neelis, M.; Patel, M.; Blok, K.; Haije, W.; Bach, P. Approximation of Theoretical Energy-Saving Potentials for the Petrochemical Industry Using Energy Balances for 68 Key Processes. Energy 2007, 32, 1104–1123. [Google Scholar] [CrossRef]
- Rethink Plastic; Zero Waste Europe; Bellona Counting Carbon—A Lifecycle Assessment Guide for Plastic Fuels. Available online: https://zerowasteeurope.eu/library/counting-carbon-a-lifecycle-assessment-guide-for-plastic-fuels/ (accessed on 2 March 2021).
- Nawaz, Z. Light Alkane Dehydrogenation to Light Olefin Technologies: A Comprehensive Review. Rev. Chem. Eng. 2015, 31, 413–436. [Google Scholar] [CrossRef]
- Wood, D.A.; Nwaoha, C.; Towler, B.F. Gas-to-Liquids (GTL): A Review of an Industry Offering Several Routes for Monetizing Natural Gas. J. Nat. Gas Sci. Eng. 2012, 9, 196–208. [Google Scholar] [CrossRef]
- Rodríguez-Vallejo, D.F.; Guillén-Gosálbez, G.; Chachuat, B. What Is the True Cost of Producing Propylene from Methanol? The Role of Externalities. ACS Sustain. Chem. Eng. 2020. [Google Scholar] [CrossRef]
- Corma, A.; Corresa, E.; Mathieu, Y.; Sauvanaud, L.; Al-Bogami, S.; Al-Ghrami, M.S.; Bourane, A. Crude Oil to Chemicals: Light Olefins from Crude Oil. Catal. Sci. Technol. 2017. [Google Scholar] [CrossRef]
- Rahim, R.; Abdul Raman, A.A. Carbon Dioxide Emission Reduction through Cleaner Production Strategies in a Recycled Plastic Resins Producing Plant. J. Clean. Prod. 2017, 141, 1067–1073. [Google Scholar] [CrossRef]
- Wernet, G.; Bauer, C.; Steubing, B.; Reinhard, J.; Moreno-Ruiz, E.; Weidema, B. The Ecoinvent Database Version 3 (Part I): Overview and Methodology. Int. J. Life Cycle Assess. 2016, 21, 1218–1230. [Google Scholar] [CrossRef]
- Hackl, R. Power Production with Organic Rankine Cycle Technology Utilizing Waste Heat from a Cracker and Three Polyethylene Units. MSc Thesis, Chalmers University of Technology, Göteborg, Sweden, 2009. [Google Scholar]
- Daniel Posen, I.; Jaramillo, P.; Landis, A.E.; Michael Griffin, W. Greenhouse Gas Mitigation for U.S. Plastics Production: Energy First, Feedstocks Later. Environ. Res. Lett. 2017, 12. [Google Scholar] [CrossRef]
- SABIC Highlights Innovation with Details of World’s Largest CO2 Purification Plant in Sustainability Report. Available online: https://www.sabic.com/en/news/4055-sabic-highlights-innovation-with-details-of-world-s-largest-co2-purification-plant-in-sustainability-report (accessed on 3 May 2020).
- Schiffer, Z.J.; Manthiram, K. Electrification and Decarbonization of the Chemical Industry. Joule 2017, 1, 10–14. [Google Scholar] [CrossRef]
- Levi, P.G.; Cullen, J.M. Mapping Global Flows of Chemicals: From Fossil Fuel Feedstocks to Chemical Products. Environ. Sci. Technol. 2018, 52, 1725–1734. [Google Scholar] [CrossRef] [PubMed]
- Griffin, P.W.; Hammond, G.P.; Norman, J.B. Industrial Energy Use and Carbon Emissions Reduction: A UK Perspective. Wiley Interdiscip. Rev. Energy Environ. 2016, 5, 684–714. [Google Scholar] [CrossRef]
- Chu, S.; Cui, Y.; Liu, N. The Path towards Sustainable Energy. Nat. Mater. 2016, 16, 16–22. [Google Scholar] [CrossRef]
- Hassiba, R.J.; Al-Mohannadi, D.M.; Linke, P. Carbon Dioxide and Heat Integration of Industrial Parks. J. Clean. Prod. 2017, 155, 47–56. [Google Scholar] [CrossRef]
- Raj, N.T.; Iniyan, S.; Goic, R. A Review of Renewable Energy Based Cogeneration Technologies. Renew. Sustain. Energy Rev. 2011, 15, 3640–3648. [Google Scholar] [CrossRef]
- Broberg Viklund, S.; Karlsson, M. Industrial Excess Heat Use: Systems Analysis and CO2 Emissions Reduction. Appl. Energy 2015, 152, 189–197. [Google Scholar] [CrossRef]
- Broberg Viklund, S.; Johansson, M.T. Technologies for Utilization of Industrial Excess Heat: Potentials for Energy Recovery and CO2 Emission Reduction. Energy Convers. Manag. 2014, 77, 369–379. [Google Scholar] [CrossRef]
- Thiruvenkataswamy, P.; Eljack, F.T.; Roy, N.; Mannan, M.S.; El-Halwagi, M.M. Safety and Techno-Economic Analysis of Ethylene Technologies. J. Loss Prev. Process Ind. 2016, 39, 74–84. [Google Scholar] [CrossRef]
- Schepp, C.; Tabrizi, M.; Tucker, T.; Nicol, J.; English, B.; Kent, R. Plastics Energy Best Practice Guidebook. Available online: https://focusonenergy.com/sites/default/files/plastics_guidebook.pdf (accessed on 4 June 2020).
- Scott, A. European Chemical Makers Plan “Cracker of the Future”. Available online: https://cen.acs.org/business/petrochemicals/European-chemical-makers-plan-cracker/97/i35 (accessed on 7 February 2020).
- Posen, I.D.; Jaramillo, P.; Griffin, W.M. Uncertainty in the Life Cycle Greenhouse Gas Emissions from U.S. Production of Three Biobased Polymer Families. Environ. Sci. Technol. 2016, 50, 2846–2858. [Google Scholar] [CrossRef] [PubMed]
- Posen, I.D.; Griffin, W.M.; Matthews, H.S.; Azevedo, I.L. Changing the Renewable Fuel Standard to a Renewable Material Standard: Bioethylene Case Study. Environ. Sci. Technol. 2015, 49, 93–102. [Google Scholar] [CrossRef]
- Hunt, A.J.; Sin, E.H.K.; Marriott, R.; Clark, J.H. Generation, Capture, and Utilization of Industrial Carbon Dioxide. ChemSusChem 2010, 3, 306–322. [Google Scholar] [CrossRef]
- Ghanta, M.; Fahey, D.; Subramaniam, B. Environmental Impacts of Ethylene Production from Diverse Feedstocks and Energy Sources. Appl. Petrochemical Res. 2014, 4, 167–179. [Google Scholar] [CrossRef]
- Eckert, C.; Xu, W.; Xiong, W.; Lynch, S.; Ungerer, J.; Tao, L.; Gill, R.; Maness, P.-C.; Yu, J. Ethylene-Forming Enzyme and Bioethylene Production. Biotechnol. Biofuels 2014, 7, 33. [Google Scholar] [CrossRef]
- Mohsenzadeh, A.; Zamani, A.; Taherzadeh, M.J. Bioethylene Production from Ethanol: A Review and Techno-Economical Evaluation. ChemBioEng Rev. 2017, 4, 75–91. [Google Scholar] [CrossRef]
- Hahladakis, J.N.; Iacovidou, E. Closing the Loop on Plastic Packaging Materials: What Is Quality and How Does It Affect Their Circularity? Sci. Total Environ. 2018, 630, 1394–1400. [Google Scholar] [CrossRef]
- Rahimi, A.R.; Garciá, J.M. Chemical Recycling of Waste Plastics for New Materials Production. Nat. Rev. Chem. 2017, 1, 1–11. [Google Scholar] [CrossRef]
- CE Delft Exploratory Study on Chemical Recycling. Update 2019. Available online: https://cedelft.eu/publications/exploratory-study-on-chemical-recycling-update-2019/ (accessed on 23 March 2021).
- Meys, R.; Frick, F.; Westhues, S.; Sternberg, A.; Klankermayer, J.; Bardow, A. Towards a Circular Economy for Plastic Packaging Wastes—The Environmental Potential of Chemical Recycling. Resour. Conserv. Recycl. 2020, 162, 105010. [Google Scholar] [CrossRef]
- BASF Life Cycle Assessment (LCA) for ChemCyclingTM. Available online: https://www.basf.com/global/en/who-we-are/sustainability/we-drive-sustainable-solutions/circular-economy/mass-balance-approach/chemcycling/lca-for-chemcycling.html (accessed on 23 April 2021).
- Jeswani, H.; Krüger, C.; Russ, M.; Horlacher, M.; Antony, F.; Hann, S.; Azapagic, A. Life Cycle Environmental Impacts of Chemical Recycling via Pyrolysis of Mixed Plastic Waste in Comparison with Mechanical Recycling and Energy Recovery. Sci. Total Environ. 2021, 769, 144483. [Google Scholar] [CrossRef] [PubMed]
- Gracida-Alvarez, U.R.; Winjobi, O.; Sacramento-Rivero, J.C.; Shonnard, D.R. System Analyses of High-Value Chemicals and Fuels from a Waste High-Density Polyethylene Refinery. Part 2: Carbon Footprint Analysis and Regional Electricity Effects. ACS Sustain. Chem. Eng. 2019, 7, 18267–18278. [Google Scholar] [CrossRef]
- Kaufman, H. Why Plastics Are Also a Climate Issue. Available online: https://www.greenbiz.com/article/why-plastics-are-also-climate-issue (accessed on 23 March 2021).
- Leyman, P.; Vanhoucke, M. Greenhouse Gas Emissions from New Petrochemical Plants Background Information Paper for the Elaboration of Technical Notes and Guidelines for IDB Projects. Int. J. Prod. Res. 2015, 53, 2771–2786. [Google Scholar] [CrossRef]
- PlasticsEurope Annual Review 2017–2018. Available online: https://www.plasticseurope.org/en/resources/publications/498-plasticseurope-annual-review-2017-2018 (accessed on 6 July 2020).
- Raheem, A.; Sikarwar, V.S.; He, J.; Dastyar, W.; Dionysiou, D.D.; Wang, W.; Zhao, M. Opportunities and Challenges in Sustainable Treatment and Resource Reuse of Sewage Sludge: A Review. Chem. Eng. J. 2018, 337, 616–641. [Google Scholar] [CrossRef]
- Sindelar, A.J.; Schmer, M.R.; Gesch, R.W.; Forcella, F.; Eberle, C.A.; Thom, M.D.; Archer, D.W. Winter Oilseed Production for Biofuel in the US Corn Belt: Opportunities and Limitations. GCB Bioenergy 2017, 9, 508–524. [Google Scholar] [CrossRef]
- Tuffi, R.; D’Abramo, S.; Cafiero, L.M.; Trinca, E.; Vecchio Ciprioti, S. Thermal Behavior and Pyrolytic Degradation Kinetics of Polymeric Mixtures from Waste Packaging Plastics. Express Polym. Lett. 2018, 12, 82–99. [Google Scholar] [CrossRef]
- Kunwar, B.; Cheng, H.N.; Chandrashekaran, S.R.; Sharma, B.K. Plastics to Fuel: A Review. Renew. Sustain. Energy Rev. 2016, 54, 421–428. [Google Scholar] [CrossRef]
- Czajczyńska, D.; Nannou, T.; Anguilano, L.; Krzyzyńska, R.; Ghazal, H.; Spencer, N.; Jouhara, H. Potentials of Pyrolysis Processes in the Waste Management Sector. Energy Procedia 2017, 123, 387–394. [Google Scholar] [CrossRef]
- Centi, G.; Perathoner, S. Opportunities and Prospects in the Chemical Recycling of Carbon Dioxide to Fuels. Catal. Today 2009, 148, 191–205. [Google Scholar] [CrossRef]
- Kätelhön, A.; Meys, R.; Deutz, S.; Suh, S.; Bardow, A. Climate Change Mitigation Potential of Carbon Capture and Utilization in the Chemical Industry. Proc. Natl. Acad. Sci. USA 2019, 166, 11187–11194. [Google Scholar] [CrossRef] [PubMed]
- Koytsoumpa, E.I.; Bergins, C.; Kakaras, E. The CO2 Economy: Review of CO2 Capture and Reuse Technologies. J. Supercrit. Fluids 2018, 132, 3–16. [Google Scholar] [CrossRef]
- Norhasyima, R.S.; Mahlia, T.M.I. Advances in CO2 Utilization Technology: A Patent Landscape Review. J. CO2 Util. 2018, 26, 323–335. [Google Scholar] [CrossRef]
- Carus, M.; Dammer, L.; Raschka, A.; Skoczinski, P. Renewable Carbon: Key to a Sustainable and Future-oriented Chemical and Plastic Industry: Definition, Strategy, Measures and Potential. Greenh. Gases Sci. Technol. 2020, 10, 488–505. [Google Scholar] [CrossRef]
- Bertini, M.; Buehler, S.; Halbheer, D.; Lehmann, D.R. Carbon Footprinting and Pricing Under Climate Concerns. J. Mark. 2020. [Google Scholar] [CrossRef]
- Ren, T.; Daniëls, B.; Patel, M.K.; Blok, K. Petrochemicals from Oil, Natural Gas, Coal and Biomass: Production Costs in 2030–2050. Resour. Conserv. Recycl. 2009, 53, 653–663. [Google Scholar] [CrossRef]
- Analytics, C. It’s Time to Get the Facts. Available online: https://clarivate.com/wp-content/uploads/2017/05/d6b7faae-3cc2-4186-8985-a6ecc8cce1ee_Crv_WoS_Upsell_Factbook_A4_FA_LR_edits.pdf (accessed on 9 August 2019).
- Polymer Properties Database Crow’s Top 10 Plastics and Resins Producers. Available online: https://polymerdatabase.com/PolymerBrands/PlasticManufacturers.html (accessed on 23 March 2021).
- World’s 10 Largest Petrochemicals Companies. Available online: https://www.oilandgasmiddleeast.com/article-6235-worlds-10-largest-petrochemicals-companies (accessed on 23 March 2021).
- Simpson, T.B. Limiting Emissions of the Greenhouse Gas, CO2. Environ. Prog. 1991, 10, 248–250. [Google Scholar] [CrossRef]
- Godec, M.L.; Kuuskraa, V.A.; Dipietro, P. Opportunities for Using Anthropogenic CO2 for Enhanced Oil Recovery and CO2 Storage. Energy Fuels 2013, 27, 4183–4189. [Google Scholar] [CrossRef]
- Herzog, H.J.; Drake, E.M. Carbon Dioxide Recovery and Disposal from Large Energy Systems. Annu. Rev. Energy Environ. 1996, 21, 145–166. [Google Scholar] [CrossRef]
- Leung, D.Y.C.; Caramanna, G.; Maroto-Valer, M.M. An Overview of Current Status of Carbon Dioxide Capture and Storage Technologies. Renew. Sustain. Energy Rev. 2014, 39, 426–443. [Google Scholar] [CrossRef]
- MacDowell, N.; Florin, N.; Buchard, A.; Hallett, J.; Galindo, A.; Jackson, G.; Adjiman, C.S.; Williams, C.K.; Shah, N.; Fennell, P. An Overview of CO2 Capture Technologies. Energy Environ. Sci. 2010, 3, 1645–1669. [Google Scholar] [CrossRef]
- White, C.M.; Smith, D.H.; Jones, K.L.; Goodman, A.L.; Jikich, S.A.; LaCount, R.B.; DuBose, S.B.; Ozdemir, E.; Morsi, B.I.; Schroeder, K.T. Sequestration of Carbon Dioxide in Coal with Enhanced Coalbed Methane Recovery—A Review. Energy Fuels 2005, 19, 659–724. [Google Scholar] [CrossRef]
- Olah, G.A.; Prakash, G.K.S.; Goeppert, A. Anthropogenic Chemical Carbon Cycle for a Sustainable Future. J. Am. Chem. Soc. 2011, 133, 12881–12898. [Google Scholar] [CrossRef] [PubMed]
- Graves, C.; Ebbesen, S.D.; Mogensen, M.; Lackner, K.S. Sustainable Hydrocarbon Fuels by Recycling CO2 and H2O with Renewable or Nuclear Energy. Renew. Sustain. Energy Rev. 2011, 15, 1–23. [Google Scholar] [CrossRef]
- Ghaib, K.; Ben-Fares, F.Z. Power-to-Methane: A State-of-the-Art Review. Renew. Sustain. Energy Rev. 2018, 81, 433–446. [Google Scholar] [CrossRef]
- Patel, H.A.; Byun, J.; Yavuz, C.T. Carbon Dioxide Capture Adsorbents: Chemistry and Methods. ChemSusChem 2017, 10, 1303–1317. [Google Scholar] [CrossRef]
- Nemitallah, M.A.; Habib, M.A.; Badr, H.M.; Said, S.A.; Jamal, A.; Ben-Mansour, R.; Mokheimer, E.M.A.; Mezghani, K. Oxy-Fuel Combustion Technology: Current Status, Applications, and Trends. Int. J. Energy Res. 2017, 41, 1670–1708. [Google Scholar] [CrossRef]
- Pieri, T.; Nikitas, A.; Castillo-Castillo, A.; Angelis-Dimakis, A. Holistic Assessment of Carbon Capture and Utilization Value Chains. Environments 2018, 5, 108. [Google Scholar] [CrossRef]
- Al-Mamoori, A.; Krishnamurthy, A.; Rownaghi, A.A.; Rezaei, F. Carbon Capture and Utilization Update. Energy Technol. 2017, 5, 834–849. [Google Scholar] [CrossRef]
- Andersson, V.; Franck, P.Å.; Berntsson, T. Techno-Economic Analysis of Excess Heat Driven Post-Combustion CCS at an Oil Refinery. Int. J. Greenh. Gas Control 2016, 45, 130–138. [Google Scholar] [CrossRef]
- Sanna, A.; Uibu, M.; Caramanna, G.; Kuusik, R.; Maroto-Valer, M.M. A Review of Mineral Carbonation Technologies to Sequester CO2. Chem. Soc. Rev. 2014, 43, 8049–8080. [Google Scholar] [CrossRef] [PubMed]
- Vitillo, J.G. Magnesium-Based Systems for Carbon Dioxide Capture, Storage and Recycling: From Leaves to Synthetic Nanostructured Materials. RSC Adv. 2015, 5, 36192–36239. [Google Scholar] [CrossRef]
- Rahman, F.A.; Aziz, M.M.A.; Saidur, R.; Bakar, W.A.W.A.; Hainin, M.R.; Putrajaya, R.; Hassan, N.A. Pollution to Solution: Capture and Sequestration of Carbon Dioxide (CO2) and Its Utilization as a Renewable Energy Source for a Sustainable Future. Renew. Sustain. Energy Rev. 2017, 71, 112–126. [Google Scholar] [CrossRef]
- Álvarez, A.; Bansode, A.; Urakawa, A.; Bavykina, A.V.; Wezendonk, T.A.; Makkee, M.; Gascon, J.; Kapteijn, F. Challenges in the Greener Production of Formates/Formic Acid, Methanol, and DME by Heterogeneously Catalyzed CO2 Hydrogenation Processes. Chem. Rev. 2017, 117, 9804–9838. [Google Scholar] [CrossRef]
- Global CCS Institute Carbon Capture and Storage Images. Available online: https://www.globalccsinstitute.com/resources/ccs-image-library/ (accessed on 24 December 2019).
- MIT Carbon Capture and Sequestration Project Database. Available online: http://sequestration.mit.edu/tools/projects/index.html (accessed on 10 February 2019).
- Mikulčić, H.; Ridjan Skov, I.; Dominković, D.F.; Wan Alwi, S.R.; Manan, Z.A.; Tan, R.; Duić, N.; Hidayah Mohamad, S.N.; Wang, X. Flexible Carbon Capture and Utilization Technologies in Future Energy Systems and the Utilization Pathways of Captured CO2. Renew. Sustain. Energy Rev. 2019, 114, 109338. [Google Scholar] [CrossRef]
- Abanades, J.C.; Arias, B.; Lyngfelt, A.; Mattisson, T.; Wiley, D.E.; Li, H.; Ho, M.T.; Mangano, E.; Brandani, S. Emerging CO2 Capture Systems. Int. J. Greenh. Gas Control 2015, 40, 126–166. [Google Scholar] [CrossRef]
- Kuramochi, T.; Ramírez, A.; Turkenburg, W.; Faaij, A. Techno-Economic Prospects for CO2 Capture from Distributed Energy Systems. Renew. Sustain. Energy Rev. 2013, 19, 328–347. [Google Scholar] [CrossRef]
- Fennell, P.S.; Florin, N.; Napp, T.; Hills, T. CCS from Industrial Sources. Sustain. Technol. Syst. Policies 2012, 17. [Google Scholar] [CrossRef]
- Azapagic, A.; Cue, R.M. Carbon Capture, Storage and Utilisation Technologies: A Critical Analysis and Comparison of Their Life Cycle Environmental Impacts. J. CO2 Util. 2015, 9, 82–102. [Google Scholar] [CrossRef]
- Metz, B.; Davidson, O.; Coninck, H.; Loos, M.; Meyer, L. Carbon Dioxide Capture and Storage. Available online: https://www.ipcc.ch/report/carbon-dioxide-capture-and-storage/ (accessed on 4 May 2020).
- Wang, M.; Oko, E. Special Issue on Carbon Capture in the Context of Carbon Capture, Utilisation and Storage (CCUS). Int. J. Coal Sci. Technol. 2017, 4, 1–4. [Google Scholar] [CrossRef]
- Rubin, E.S.; Mantripragada, H.; Marks, A.; Versteeg, P.; Kitchin, J. The Outlook for Improved Carbon Capture Technology. Prog. Energy Combust. Sci. 2012, 38, 630–671. [Google Scholar] [CrossRef]
- Hossain, M.M.; de Lasa, H.I. Chemical-Looping Combustion (CLC) for Inherent CO2 Separations—A Review. Chem. Eng. Sci. 2008, 63, 4433–4451. [Google Scholar] [CrossRef]
- Porrazzo, R.; White, G.; Ocone, R. Techno-Economic Investigation of a Chemical Looping Combustion Based Power Plant. Faraday Discuss. 2016, 192, 437–457. [Google Scholar] [CrossRef] [PubMed]
- Thallam Thattai, A.; Oldenbroek, V.; Schoenmakers, L.; Woudstra, T.; Aravind, P.V. Towards Retrofitting Integrated Gasification Combined Cycle (IGCC) Power Plants with Solid Oxide Fuel Cells (SOFC) and CO2 Capture—A Thermodynamic Case Study. Appl. Therm. Eng. 2017, 114, 170–185. [Google Scholar] [CrossRef]
- Chisti, Y. Biodiesel from Microalgae. Biotechnol. Adv. 2007, 25, 294–306. [Google Scholar] [CrossRef] [PubMed]
- Ho, S.-H.; Chen, C.-Y.; Lee, D.-J.; Chang, J.-S. Perspectives on Microalgal CO2-Emission Mitigation Systems—A Review. Biotechnol. Adv. 2011, 29, 189–198. [Google Scholar] [CrossRef]
- Anguselvi, V.; Ebhin Masto, R.; Mukherjee, A.; Kumar Singh, P. CO2 Capture for Industries by Algae. In Algae; Wong, Y.K., Ed.; IntechOpen: London, UK, 2019; ISBN 978-1-83880-563-0. [Google Scholar]
- Zhang, Q.; Cheng, C.L.; Nagarajan, D.; Chang, J.S.; Hu, J.; Lee, D.J. Carbon Capture and Utilization of Fermentation CO2: Integrated Ethanol Fermentation and Succinic Acid Production as an Efficient Platform. Appl. Energy 2017, 206, 364–371. [Google Scholar] [CrossRef]
- Campos, J.L.; Valenzuela-Heredia, D.; Pedrouso, A.; Val del Río, A.; Belmonte, M.; Mosquera-Corral, A. Greenhouse Gases Emissions from Wastewater Treatment Plants: Minimization, Treatment, and Prevention. J. Chem. 2016, 2016, 1–12. [Google Scholar] [CrossRef]
- Alqaheem, Y.; Alomair, A.; Vinoba, M.; Pérez, A. Polymeric Gas-Separation Membranes for Petroleum Refining. Int. J. Polym. Sci. 2017, 2017. [Google Scholar] [CrossRef]
- Lü, F.; Hua, Z.; Shao, L.; He, P. Loop Bioenergy Production and Carbon Sequestration of Polymeric Waste by Integrating Biochemical and Thermochemical Conversion Processes: A Conceptual Framework and Recent Advances. Renew. Energy 2018, 124, 202–211. [Google Scholar] [CrossRef]
- Lee, C.; Lee, S.; Park, J.; Lee, D.; Hwang, K.; Ryi, S.; Kim, S. Long-Term CO2 Capture Tests of Pd-Based Composite Membranes with Module Configuration. Int. J. Hydrogen Energy 2013, 38, 7896–7903. [Google Scholar] [CrossRef]
- Trazzi, P.A.; Higa, A.R.; Dieckow, J.; Mangrich, A.S.; Higa, R.C.V. Biochar: Reality and Potential Use in Forestry. Ciência Florest. 2018, 28, 875–887. [Google Scholar] [CrossRef]
- Budzianowski, W.M. Negative Carbon Intensity of Renewable Energy Technologies Involving Biomass or Carbon Dioxide as Inputs. Renew. Sustain. Energy Rev. 2012, 16, 6507–6521. [Google Scholar] [CrossRef]
- Tan, X.; Liu, S.; Liu, Y.; Gu, Y.; Zeng, G.; Hu, X.; Wang, X.; Liu, S.; Jiang, L. Biochar as Potential Sustainable Precursors for Activated Carbon Production: Multiple Applications in Environmental Protection and Energy Storage. Bioresour. Technol. 2017, 227, 359–372. [Google Scholar] [CrossRef] [PubMed]
- Liang, J.; Zhang, C.; Liu, D.; Zhang, L.; Ling, G.; Yang, Q.-H. From Trash to Treasure: Turning Air Pollutants into Materials for Energy Storage. ChemNanoMat 2017, 3, 392–400. [Google Scholar] [CrossRef]
- Katuri, K.P.; Kalathil, S.; Ragab, A.; Bian, B.; Alqahtani, M.F.; Pant, D.; Saikaly, P.E. Dual-Function Electrocatalytic and Macroporous Hollow-Fiber Cathode for Converting Waste Streams to Valuable Resources Using Microbial Electrochemical Systems. Adv. Mater. 2018, 1707072, 1–18. [Google Scholar] [CrossRef]
- Beltrán-Rocha, J.C.; Guajardo-Barbosa, C.; Barceló-Quintal, I.D.; López-Chuken, U.J. Biotratamiento de Efluentes Secundarios Municipales Utilizando Microalgas: Efecto Del PH, Nutrientes (C, N y P) y Enriquecimiento Con CO2. Rev. Biol. Mar. Oceanogr. 2017, 52, 417–427. [Google Scholar] [CrossRef]
- Su, T.; Qin, Z.; Ji, H.; Jiang, Y.; Huang, G. Recent Advances in the Photocatalytic Reduction of Carbon Dioxide. Environ. Chem. Lett. 2016, 14, 99–112. [Google Scholar] [CrossRef]
- Vilarrasa, V.; Rutqvist, J. Thermal Effects on Geologic Carbon Storage. Earth-Sci. Rev. 2017, 165, 245–256. [Google Scholar] [CrossRef]
- Shi, R.; Waterhouse, G.I.N.; Zhang, T. Recent Progress in Photocatalytic CO2 Reduction Over Perovskite Oxides. Sol. RRL 2017, 1, 1700126. [Google Scholar] [CrossRef]
- Chang, R.; Kim, S.; Lee, S.; Choi, S.; Kim, M.; Park, Y. Calcium Carbonate Precipitation for CO2 Storage and Utilization: A Review of the Carbonate Crystallization and Polymorphism. Front. Energy Res. 2017, 5, 1–12. [Google Scholar] [CrossRef]
- Quina, M.J.; Bontempi, E.; Bogush, A.; Schlumberger, S.; Weibel, G.; Braga, R.; Funari, V.; Hyks, J.; Rasmussen, E.; Lederer, J. Technologies for the Management of MSW Incineration Ashes from Gas Cleaning: New Perspectives on Recovery of Secondary Raw Materials and Circular Economy. Sci. Total Environ. 2018, 635, 526–542. [Google Scholar] [CrossRef]
- Wassie, S.A.; Medrano, J.A.; Zaabout, A.; Cloete, S.; Melendez, J.; Tanaka, D.A.P.; Amini, S.; van Sint Annaland, M.; Gallucci, F. Hydrogen Production with Integrated CO2 Capture in a Membrane Assisted Gas Switching Reforming Reactor: Proof-of-Concept. Int. J. Hydrogen Energy 2018, 43, 6177–6190. [Google Scholar] [CrossRef]
- Sun, N.; Tang, Z.; Wei, W.; Snape, C.E.; Sun, Y. Solid Adsorbents for Low-Temperature CO2 Capture with Low-Energy Penalties Leading to More Effective Integrated Solutions for Power Generation and Industrial Processes. Front. Energy Res. 2015, 3, 1–16. [Google Scholar] [CrossRef]
- Li, J.; Hitch, M.; Power, I.; Pan, Y. Integrated Mineral Carbonation of Ultramafic Mine Deposits—A Review. Minerals 2018, 8, 147. [Google Scholar] [CrossRef]
- Jacobs, L. Sustainability @ Sabic Carbon Capture & Utilization. In Proceedings of the Conference—When Electrons Power Molecules, Geleen, The Netherlands, 13 December 2018. [Google Scholar]
- Ramírez-Santos, Á.A.; Castel, C.; Favre, E. A Review of Gas Separation Technologies within Emission Reduction Programs in the Iron and Steel Sector: Current Application and Development Perspectives. Sep. Purif. Technol. 2018, 194, 425–442. [Google Scholar] [CrossRef]
- Araújo, O. de Q.F.; Medeiros, J.L. Carbon Capture and Storage Technologies: Present Scenario and Drivers of Innovation. Curr. Opin. Chem. Eng. 2017, 2, 22–34. [Google Scholar] [CrossRef]
- Peters, M.; Köhler, B.; Kuckshinrichs, W.; Leitner, W.; Markewitz, P.; Müller, T.E. Chemical Technologies for Exploiting and Recycling Carbon Dioxide into the Value Chain. ChemSusChem 2011, 4, 1216–1240. [Google Scholar] [CrossRef]
- Bains, P.; Psarras, P.; Wilcox, J. CO2 Capture from the Industry Sector. Prog. Energy Combust. Sci. 2017, 63, 146–172. [Google Scholar] [CrossRef]
- Li, B.; Duan, Y.; Luebke, D.; Morreale, B. Advances in CO2 Capture Technology: A Patent Review. Appl. Energy 2013, 102, 1439–1447. [Google Scholar] [CrossRef]
- Salvi, B.L.; Jindal, S. Recent Developments and Challenges Ahead in Carbon Capture and Sequestration Technologies. SN Appl. Sci. 2019, 1, 885. [Google Scholar] [CrossRef]
- Balarini, C.M.; Cruz, J.C.; Alves, J.L.B.; França-Silva, M.S.; Braga, V.A. Developing New Organic Nitrates for Treating Hypertension. In Nitric Oxide Donors; Elsevier: Amsterdam, The Netherlands, 2017; pp. 243–262. ISBN 9780128134597. [Google Scholar]
- Khalilpour, R.; Mumford, K.; Zhai, H.; Abbas, A.; Stevens, G.; Rubin, E.S. Membrane-Based Carbon Capture from Flue Gas: A Review. J. Clean. Prod. 2015, 103, 286–300. [Google Scholar] [CrossRef]
- Li, A.; Wang, J.; Bao, B. High-efficiency CO2 Capture and Separation Based on Hydrate Technology: A Review. Greenh. Gases Sci. Technol. 2019, 9, 175–193. [Google Scholar] [CrossRef]
- Wang, B.; Lan, C.Q. Biofixation of carbon dioxide (CO2) by microorganisms. In Developments and Innovation in Carbon Dioxide (CO2) Capture and Storage Technology; Elsevier: Amsterdam, The Netherlands, 2010; pp. 411–432. ISBN 9781845697976. [Google Scholar]
- Wu, M.; Zhang, W.; Ji, Y.; Yi, X.; Ma, J.; Wu, H.; Jiang, M. Coupled CO2 Fixation from Ethylene Oxide Off-Gas with Bio-Based Succinic Acid Production by Engineered Recombinant Escherichia Coli. Biochem. Eng. J. 2017, 117, 1–6. [Google Scholar] [CrossRef]
- Zevenhoven, R.; Fagerlund, J. Mineralisation of carbon dioxide (CO2). In Developments and Innovation in Carbon Dioxide (CO2); Woodhead Publishing: Cambridge, UK, 2010; ISBN 9781845697976. [Google Scholar]
- Wheeldon, J.M.; Thimsen, D. Economic evaluation of circulating fluidized bed combustion (CFBC) power generation plants. In Fluidized Bed Technologies for Near-Zero Emission Combustion and Gasification; Woodhead Publishing: Cambridge, UK, 2013; ISBN 9780857095411. [Google Scholar]
- Dincer, I.; Rosen, M.A. Exergy Analysis of Fuel Cell Systems. In Exergy; Elsevier: Amsterdam, The Netherlands, 2013; pp. 363–382. [Google Scholar]
- Wang, M.; Hariharan, S.; Shaw, R.A.; Hatton, T.A. Energetics of Electrochemically Mediated Amine Regeneration Process for Flue Gas CO2 Capture. Int. J. Greenh. Gas Control 2019. [Google Scholar] [CrossRef]
- Hahladakis, J.N.; Velis, C.A.; Weber, R.; Iacovidou, E.; Purnell, P. An Overview of Chemical Additives Present in Plastics: Migration, Release, Fate and Environmental Impact during Their Use, Disposal and Recycling. J. Hazard. Mater. 2018, 344, 179–199. [Google Scholar] [CrossRef]
- Carneiro, M.L.N.M.; Pradelle, F.; Braga, S.L.; Gomes, M.S.P.; Martins, A.R.F.A.; Turkovics, F.; Pradelle, R.N.C. Potential of Biofuels from Algae: Comparison with Fossil Fuels, Ethanol and Biodiesel in Europe and Brazil through Life Cycle Assessment (LCA). Renew. Sustain. Energy Rev. 2017, 73, 632–653. [Google Scholar] [CrossRef]
- Tu, Q.; Eckelman, M.; Zimmerman, J. Meta-Analysis and Harmonization of Life Cycle Assessment Studies for Algae Biofuels. Environ. Sci. Technol. 2017, 51, 9419–9432. [Google Scholar] [CrossRef]
- Vikrant, K.; Kumar, V.; Kim, K.H.; Kukkar, D. Technical and Economic Aspects of Carbon Capture an Storage—A Review. Energy Explor. Exploit. 2008, 5, 22877–22896. [Google Scholar]
- Rees, N.V.; Compton, R.G. Electrochemical CO2 Sequestration in Ionic Liquids; a Perspective. Energy Environ. Sci. 2011, 4, 403–408. [Google Scholar] [CrossRef]
- Yang, Z.Z.; Zhao, Y.N.; He, L.N. CO2 Chemistry: Task-Specific Ionic Liquids for CO2 Capture/Activation and Subsequent Conversion. RSC Adv. 2011, 1, 545–567. [Google Scholar] [CrossRef]
- Zangeneh, F.T.; Sahebdelfar, S.; Ravanchi, M.T. Conversion of Carbon Dioxide to Valuable Petrochemicals: An Approach to Clean Development Mechanism. J. Nat. Gas Chem. 2011, 20, 219–231. [Google Scholar] [CrossRef]
- Olajire, A.A. CO2 Capture by Aqueous Ammonia Process in the Clean Development Mechanism for Nigerian Oil Industry. Front. Chem. Sci. Eng. 2013, 7, 366–380. [Google Scholar] [CrossRef]
- Haro, P.; Ollero, P.; Villanueva Perales, Á.L.; Vidal-Barrero, F. Potential Routes for Thermochemical Biorefineries. Biofuels Bioprod. Biorefining 2013, 7, 551–572. [Google Scholar] [CrossRef]
- Li, L.; Zhao, N.; Wei, W.; Sun, Y. A Review of Research Progress on CO2 Capture, Storage, and Utilization in Chinese Academy of Sciences. Fuel 2013, 108, 112–130. [Google Scholar] [CrossRef]
- Ma, S.; Chen, G.; Guo, M.; Zhao, L.; Han, T.; Zhu, S. Path Analysis on CO2 Resource Utilization Based on Carbon Capture Using Ammonia Method in Coal-Fired Power Plants. Renew. Sustain. Energy Rev. 2014, 37, 687–697. [Google Scholar] [CrossRef]
- Abolhasani, M.; Günther, A.; Kumacheva, E. Microfluidic Studies of Carbon Dioxide. Angew. Chem. Int. Ed. 2014, 53, 7992–8002. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; He, L.; Diao, Z.; Yang, Z. Carbon Capture with Simultaneous Activation and Its Subsequent Transformation. In CO2 Chemistry; Aresta, M., van Eldik, R., Eds.; Elsevier Inc.: Amsterdam, The Netherlands, 2014; Volume 66, pp. 289–345. ISBN 9780124202214. [Google Scholar]
- Shakerian, F.; Kim, K.H.; Szulejko, J.E.; Park, J.W. A Comparative Review between Amines and Ammonia as Sorptive Media for Post-Combustion CO2 Capture. Appl. Energy 2015, 148, 10–22. [Google Scholar] [CrossRef]
- Liang, Z. (Henry); Rongwong, W.; Liu, H.; Fu, K.; Gao, H.; Cao, F.; Zhang, R.; Sema, T.; Henni, A.; Sumon, K.; et al. Recent Progress and New Developments in Post-Combustion Carbon-Capture Technology with Amine Based Solvents. Int. J. Greenh. Gas Control 2015, 40, 26–54. [Google Scholar] [CrossRef]
- Alvarez-Guerra, M.; Albo, J.; Alvarez-Guerra, E.; Irabien, A. Ionic Liquids in the Electrochemical Valorisation of CO2. Energy Environ. Sci. 2015, 8, 2574–2599. [Google Scholar] [CrossRef]
- Quader, M.A.; Ahmed, S.; Ghazilla, R.A.R.; Ahmed, S.; Dahari, M. A Comprehensive Review on Energy Efficient CO2 Breakthrough Technologies for Sustainable Green Iron and Steel Manufacturing. Renew. Sustain. Energy Rev. 2015, 50, 594–614. [Google Scholar] [CrossRef]
- Ishak, S.A.; Hashim, H. Low Carbon Measures for Cement Plant—A Review. J. Clean. Prod. 2015, 103, 260–274. [Google Scholar] [CrossRef]
- Last, G.V.; Schmick, M.T. A Review of Major Non-Power-Related Carbon Dioxide Stream Compositions. Environ. Earth Sci. 2015, 1189–1198. [Google Scholar] [CrossRef]
- Voitic, G.; Hacker, V. Recent Advancements in Chemical Looping Water Splitting for the Production of Hydrogen. RSC Adv. 2016, 6, 98267–98296. [Google Scholar] [CrossRef]
- Shehzad, A.; Bashir, M.J.K.; Sethupathi, S. System Analysis for Synthesis Gas (Syngas) Production in Pakistan from Municipal Solid Waste Gasification Using a Circulating Fluidized Bed Gasifier. Renew. Sustain. Energy Rev. 2016, 60, 1302–1311. [Google Scholar] [CrossRef]
- Li, T.; Keener, T.C. A Review: Desorption of CO2 from Rich Solutions in Chemical Absorption Processes. Int. J. Greenh. Gas Control 2016, 51, 290–304. [Google Scholar] [CrossRef]
- Zhao, S.; Feron, P.H.M.; Deng, L.; Favre, E.; Chabanon, E.; Yan, S.; Hou, J.; Chen, V.; Qi, H. Status and Progress of Membrane Contactors in Post-Combustion Carbon Capture: A State-of-the-Art Review of New Developments. J. Memb. Sci. 2016, 511, 180–206. [Google Scholar] [CrossRef]
- Uribe-Soto, W.; Portha, J.F.; Commenge, J.M.; Falk, L. A Review of Thermochemical Processes and Technologies to Use Steelworks Off-Gases. Renew. Sustain. Energy Rev. 2017, 74, 809–823. [Google Scholar] [CrossRef]
- Salazar Duarte, G.; Schürer, B.; Voss, C.; Bathen, D. Adsorptive Separation of CO2 from Flue Gas by Temperature Swing Adsorption Processes. ChemBioEng Rev. 2017, 4, 277–288. [Google Scholar] [CrossRef]
- Adams, T.A.; Hoseinzade, L.; Madabhushi, P.B.; Okeke, I.J. Comparison of CO2 Capture Approaches for Fossil-Based Power Generation: Review and Meta-Study. Processes 2017, 5, 44. [Google Scholar] [CrossRef]
- Thakur, I.S.; Kumar, M.; Varjani, S.J.; Wu, Y.; Gnansounou, E.; Ravindran, S. Sequestration and Utilization of Carbon Dioxide by Chemical and Biological Methods for Biofuels and Biomaterials by Chemoautotrophs: Opportunities and Challenges. Bioresour. Technol. 2018, 256, 478–490. [Google Scholar] [CrossRef]
- Mukherjee, M.; Misra, S. A Review of Experimental Research on Enhanced Coal Bed Methane (ECBM) Recovery via CO2 Sequestration. Earth-Sci. Rev. 2018, 179, 392–410. [Google Scholar] [CrossRef]
- Tan, Y.; Nookuea, W.; Li, H.; Thorin, E.; Yan, J. Property Impacts on Carbon Capture and Storage (CCS) Processes: A Review. Energy Convers. Manag. 2016, 118, 204–222. [Google Scholar] [CrossRef]
- Barelli, L.; Bidini, G.; Gallorini, F.; Servili, S. Hydrogen Production through Sorption-Enhanced Steam Methane Reforming and Membrane Technology: A Review. Energy 2008, 33, 554–570. [Google Scholar] [CrossRef]
- Yang, Z.Z.; He, L.N.; Gao, J.; Liu, A.H.; Yu, B. Carbon Dioxide Utilization with C-N Bond Formation: Carbon Dioxide Capture and Subsequent Conversion. Energy Environ. Sci. 2012, 5, 6602–6639. [Google Scholar] [CrossRef]
- Wang, X.; Song, C. Carbon Capture From Flue Gas and the Atmosphere: A Perspective. Front. Energy Res. 2020, 8. [Google Scholar] [CrossRef]
- Ren, S.; Jiang, S.; Yan, X.; Chen, R.; Cui, H. Challenges and Opportunities: Porous Supports in Carbonic Anhydrase Immobilization. J. CO2 Util. 2020, 42, 101305. [Google Scholar] [CrossRef]
- Formann, S.; Hahn, A.; Janke, L.; Stinner, W.; Sträuber, H.; Logroño, W.; Nikolausz, M. Beyond Sugar and Ethanol Production: Value Generation Opportunities Through Sugarcane Residues. Front. Energy Res. 2020, 8. [Google Scholar] [CrossRef]
- Wu, Z.; Nan, Y.; Zhao, Y.; Wang, X.; Huang, S.; Shi, J. Immobilization of Carbonic Anhydrase for Facilitated CO2 Capture and Separation. Chinese J. Chem. Eng. 2020, 28, 2817–2831. [Google Scholar] [CrossRef]
- Rashid, M.I.; Benhelal, E.; Rafiq, S. Reduction of Greenhouse Gas Emissions from Gas, Oil, and Coal Power Plants in Pakistan by Carbon Capture and Storage (CCS): A Review. Chem. Eng. Technol. 2020, 43, 2140–2148. [Google Scholar] [CrossRef]
- Yamada, H. Amine-Based Capture of CO2 for Utilization and Storage. Polym. J. 2021, 53, 93–102. [Google Scholar] [CrossRef]
- Nocito, F.; Dibenedetto, A. Atmospheric CO2 Mitigation Technologies: Carbon Capture Utilization and Storage. Curr. Opin. Green Sustain. Chem. 2020, 21, 34–43. [Google Scholar] [CrossRef]
- Kar, S.; Goeppert, A.; Prakash, G.K.S. Integrated CO2 Capture and Conversion to Formate and Methanol: Connecting Two Threads. Acc. Chem. Res. 2019, 52, 2892–2903. [Google Scholar] [CrossRef] [PubMed]
- He, Q.; Shi, M.; Liang, F.; Xu, L.; Ji, L.; Yan, S. Renewable Absorbents for CO2 Capture: From Biomass to Nature. Greenh. Gases Sci. Technol. 2019, 9, 637–651. [Google Scholar] [CrossRef]
- Papadopoulos, A.I.; Tzirakis, F.; Tsivintzelis, I.; Seferlis, P. Phase-Change Solvents and Processes for Postcombustion CO2 Capture: A Detailed Review. Ind. Eng. Chem. Res. 2019, 58, 5088–5111. [Google Scholar] [CrossRef]
- Yoshimoto, M.; Walde, P. Immobilized Carbonic Anhydrase: Preparation, Characteristics and Biotechnological Applications. World J. Microbiol. Biotechnol. 2018, 34, 151. [Google Scholar] [CrossRef]
- Grande, C.A.; Ribeiro, R.P.P.L.; Rodrigues, A.E. Challenges of Electric Swing Adsorption for CO2 Capture. ChemSusChem 2010, 3, 892–898. [Google Scholar] [CrossRef] [PubMed]
- Prabu, V.; Mallick, N. Coalbed Methane with CO2 Sequestration: An Emerging Clean Coal Technology in India. Renew. Sustain. Energy Rev. 2015, 50, 229–244. [Google Scholar] [CrossRef]
- González-Salazar, M.A. Recent Developments in Carbon Dioxide Capture Technologies for Gas Turbine Power Generation. Int. J. Greenh. Gas Control 2015, 34, 106–116. [Google Scholar] [CrossRef]
- Lu, N.; Xie, D. Novel Membrane Reactor Concepts for Hydrogen Production from Hydrocarbons: A Review. Int. J. Chem. React. Eng. 2016, 14, 1–31. [Google Scholar] [CrossRef]
- Vellingiri, K.; Kumar, P.; Kim, K.-H. Coordination Polymers: Challenges and Future Scenarios for Capture and Degradation of Volatile Organic Compounds. Nano Res. 2016, 9, 3181–3208. [Google Scholar] [CrossRef]
- Brassard, P.; Godbout, S.; Raghavan, V. Soil Biochar Amendment as a Climate Change Mitigation Tool: Key Parameters and Mechanisms Involved. J. Environ. Manag. 2016, 181, 484–497. [Google Scholar] [CrossRef]
- Maleki, H. Recent Advances in Aerogels for Environmental Remediation Applications: A Review. Chem. Eng. J. 2016, 300, 98–118. [Google Scholar] [CrossRef]
- Abdul Quader, M.; Ahmed, S.; Dawal, S.Z.; Nukman, Y. Present Needs, Recent Progress and Future Trends of Energy-Efficient Ultra-Low Carbon Dioxide (CO2) Steelmaking (ULCOS) Program. Renew. Sustain. Energy Rev. 2016, 55, 537–549. [Google Scholar] [CrossRef]
- Pode, R. Potential Applications of Rice Husk Ash Waste from Rice Husk Biomass Power Plant. Renew. Sustain. Energy Rev. 2016, 53, 1468–1485. [Google Scholar] [CrossRef]
- Zhang, X.G.; Ranjith, P.G.; Perera, M.S.A.; Ranathunga, A.S.; Haque, A. Gas Transportation and Enhanced Coalbed Methane Recovery Processes in Deep Coal Seams: A Review. Energy Fuels 2016, 30, 8832–8849. [Google Scholar] [CrossRef]
- Vikrant, K.; Kumar, V.; Kim, K.H.; Kukkar, D. Metal-Organic Frameworks (MOFs): Potential and Challenges for Capture and Abatement of Ammonia. J. Mater. Chem. A 2017, 5, 22877–22896. [Google Scholar] [CrossRef]
- Moreira, M.T.; Noya, I.; Feijoo, G. The Prospective Use of Biochar as Adsorption Matrix—A Review from a Lifecycle Perspective. Bioresour. Technol. 2017, 246, 135–141. [Google Scholar] [CrossRef]
- Maity, A.; Polshettiwar, V. Dendritic Fibrous Nanosilica for Catalysis, Energy Harvesting, Carbon Dioxide Mitigation, Drug Delivery, and Sensing. ChemSusChem 2017, 10, 3866–3913. [Google Scholar] [CrossRef] [PubMed]
- Creton, B. Chemoinformatics at IFP Energies Nouvelles: Applications in the Fields of Energy, Transport, and Environment. Mol. Inform. 2017, 36, 1700028. [Google Scholar] [CrossRef]
- Zhang, H.R.; Li, S.; Kelly, K.E.; Eddings, E.G. Underground in Situ Coal Thermal Treatment for Synthetic Fuels Production. Prog. Energy Combust. Sci. 2017, 62, 1–32. [Google Scholar] [CrossRef]
- Lee, Y.R.; Soe, J.T.; Zhang, S.; Ahn, J.W.; Park, M.B.; Ahn, W.S. Synthesis of Nanoporous Materials via Recycling Coal Fly Ash and Other Solid Wastes: A Mini Review. Chem. Eng. J. 2017, 317, 821–843. [Google Scholar] [CrossRef]
- Memon, M.Z.; Zhao, X.; Sikarwar, V.S.; Vuppaladadiyam, A.K.; Milne, S.J.; Brown, A.P.; Li, J.; Zhao, M. Alkali Metal CO2 Sorbents and the Resulting Metal Carbonates: Potential for Process Intensification of Sorption-Enhanced Steam Reforming. Environ. Sci. Technol. 2017, 51, 12–27. [Google Scholar] [CrossRef]
- Chan, W.H.; Mazlee, M.N.; Ahmad, Z.A.; Ishak, M.A.M.; Shamsul, J.B. The Development of Low Cost Adsorbents from Clay and Waste Materials: A Review. J. Mater. Cycles Waste Manag. 2017, 19, 1–14. [Google Scholar] [CrossRef]
- Dhoble, Y.N.; Ahmed, S. Review on the Innovative Uses of Steel Slag for Waste Minimization. J. Mater. Cycles Waste Manag. 2018, 20, 1373–1382. [Google Scholar] [CrossRef]
- Bamdad, H.; Hawboldt, K.; MacQuarrie, S. A Review on Common Adsorbents for Acid Gases Removal: Focus on Biochar. Renew. Sustain. Energy Rev. 2018, 81, 1705–1720. [Google Scholar] [CrossRef]
- Mukhtar, A.; Saqib, S.; Mellon, N.B.; Rafiq, S.; Babar, M.; Ullah, S.; Muhammad, N.; Khan, A.L.; Ayoub, M.; Ibrahim, M.; et al. A Review on CO2 Capture via Nitrogen-Doped Porous Polymers and Catalytic Conversion as a Feedstock for Fuels. J. Clean. Prod. 2020, 277, 123999. [Google Scholar] [CrossRef]
- Alzamly, A.; Bakiro, M.; Hussein Ahmed, S.; Alnaqbi, M.A.; Nguyen, H.L. Rare-Earth Metal–Organic Frameworks as Advanced Catalytic Platforms for Organic Synthesis. Coord. Chem. Rev. 2020, 425, 213543. [Google Scholar] [CrossRef]
- Osman, A.I.; Hefny, M.; Abdel Maksoud, M.I.A.; Elgarahy, A.M.; Rooney, D.W. Recent Advances in Carbon Capture Storage and Utilisation Technologies: A Review. Environ. Chem. Lett. 2020. [Google Scholar] [CrossRef]
- Kumar, S.; Srivastava, R.; Koh, J. Utilization of Zeolites as CO2 Capturing Agents: Advances and Future Perspectives. J. CO2 Util. 2020, 41, 101251. [Google Scholar] [CrossRef]
- Singh, G.; Lee, J.; Karakoti, A.; Bahadur, R.; Yi, J.; Zhao, D.; AlBahily, K.; Vinu, A. Emerging Trends in Porous Materials for CO2 Capture and Conversion. Chem. Soc. Rev. 2020, 49, 4360–4404. [Google Scholar] [CrossRef] [PubMed]
- Pal, T.K.; De, D.; Bharadwaj, P.K. Metal–Organic Frameworks for the Chemical Fixation of CO2 into Cyclic Carbonates. Coord. Chem. Rev. 2020, 408, 213173. [Google Scholar] [CrossRef]
- Miricioiu, M.G.; Niculescu, V.-C. Fly Ash, from Recycling to Potential Raw Material for Mesoporous Silica Synthesis. Nanomaterials 2020, 10, 474. [Google Scholar] [CrossRef] [PubMed]
- Dissanayake, P.D.; You, S.; Igalavithana, A.D.; Xia, Y.; Bhatnagar, A.; Gupta, S.; Kua, H.W.; Kim, S.; Kwon, J.-H.; Tsang, D.C.W.; et al. Biochar-Based Adsorbents for Carbon Dioxide Capture: A Critical Review. Renew. Sustain. Energy Rev. 2020, 119, 109582. [Google Scholar] [CrossRef]
- Sri Shalini, S.; Palanivelu, K.; Ramachandran, A.; Raghavan, V. Biochar from Biomass Waste as a Renewable Carbon Material for Climate Change Mitigation in Reducing Greenhouse Gas Emissions—A Review. Biomass Convers. Biorefinery 2020. [Google Scholar] [CrossRef]
- Shi, Y.; Hou, S.; Qiu, X.; Zhao, B. MOFs-Based Catalysts Supported Chemical Conversion of CO2. Top. Curr. Chem. 2020, 378, 11. [Google Scholar] [CrossRef]
- Luciani, G.; Imparato, C.; Vitiello, G. Photosensitive Hybrid Nanostructured Materials: The Big Challenges for Sunlight Capture. Catalysts 2020, 10, 103. [Google Scholar] [CrossRef]
- Wang, H.; Jiang, D.; Huang, D.; Zeng, G.; Xu, P.; Lai, C.; Chen, M.; Cheng, M.; Zhang, C.; Wang, Z. Covalent Triazine Frameworks for Carbon Dioxide Capture. J. Mater. Chem. A 2019, 7, 22848–22870. [Google Scholar] [CrossRef]
- Ozdemir, J.; Mosleh, I.; Abolhassani, M.; Greenlee, L.F.; Beitle, R.R.; Beyzavi, M.H. Covalent Organic Frameworks for the Capture, Fixation, or Reduction of CO2. Front. Energy Res. 2019, 7. [Google Scholar] [CrossRef]
- Wang, J.; Wang, S. Preparation, Modification and Environmental Application of Biochar: A Review. J. Clean. Prod. 2019, 227, 1002–1022. [Google Scholar] [CrossRef]
- Fisher, L.V.; Barron, A.R. The Recycling and Reuse of Steelmaking Slags—A Review. Resour. Conserv. Recycl. 2019, 146, 244–255. [Google Scholar] [CrossRef]
- Tan, P.; Jiang, Y.; Liu, X.; Sun, L. Magnetically Responsive Porous Materials for Efficient Adsorption and Desorption Processes. Chinese J. Chem. Eng. 2019, 27, 1324–1338. [Google Scholar] [CrossRef]
- Pullumbi, P.; Brandani, F.; Brandani, S. Gas Separation by Adsorption: Technological Drivers and Opportunities for Improvement. Curr. Opin. Chem. Eng. 2019, 24, 131–142. [Google Scholar] [CrossRef]
- Huh, S. Direct Catalytic Conversion of CO2 to Cyclic Organic Carbonates under Mild Reaction Conditions by Metal—Organic Frameworks. Catalysts 2019, 9, 34. [Google Scholar] [CrossRef]
- Leng, L.; Huang, H. An Overview of the Effect of Pyrolysis Process Parameters on Biochar Stability. Bioresour. Technol. 2018, 270, 627–642. [Google Scholar] [CrossRef]
- Jie, K.; Zhou, Y.; Li, E.; Huang, F. Nonporous Adaptive Crystals of Pillararenes. Acc. Chem. Res. 2018, 51, 2064–2072. [Google Scholar] [CrossRef]
- Yen, H.-W.; Ho, S.-H.; Chen, C.-Y.; Chang, J.-S. CO2, NOx and SOx Removal from Flue Gas via Microalgae Cultivation: A Critical Review. Biotechnol. J. 2015, 10, 829–839. [Google Scholar] [CrossRef]
- Pires, J.C.M. COP21: The Algae Opportunity? Renew. Sustain. Energy Rev. 2017, 79, 867–877. [Google Scholar] [CrossRef]
- Huang, G.; Chen, F.; Kuang, Y.; He, H.; Qin, A. Current Techniques of Growing Algae Using Flue Gas from Exhaust Gas Industry: A Review. Appl. Biochem. Biotechnol. 2016, 178, 1220–1238. [Google Scholar] [CrossRef]
- Heijstra, B.D.; Leang, C.; Juminaga, A. Gas Fermentation: Cellular Engineering Possibilities and Scale Up. Microb. Cell Fact. 2017, 16, 60. [Google Scholar] [CrossRef] [PubMed]
- Liew, F.; Martin, M.E.; Tappel, R.C.; Heijstra, B.D.; Mihalcea, C.; Köpke, M. Gas Fermentation—A Flexible Platform for Commercial Scale Production of Low-Carbon-Fuels and Chemicals from Waste and Renewable Feedstocks. Front. Microbiol. 2016, 7. [Google Scholar] [CrossRef]
- Chen, Y.; Xu, C.; Vaidyanathan, S. Microalgae: A Robust “Green Bio-Bridge” between Energy and Environment. Crit. Rev. Biotechnol. 2018, 38, 351–368. [Google Scholar] [CrossRef] [PubMed]
- Patel, A.; Gami, B.; Patel, P.; Patel, B. Microalgae: Antiquity to Era of Integrated Technology. Renew. Sustain. Energy Rev. 2017, 71, 535–547. [Google Scholar] [CrossRef]
- Zhu, B.; Chen, G.; Cao, X.; Wei, D. Molecular Characterization of CO2 Sequestration and Assimilation in Microalgae and Its Biotechnological Applications. Bioresour. Technol. 2017, 244, 1207–1215. [Google Scholar] [CrossRef]
- Raslavičius, L.; Striūgas, N.; Felneris, M. New Insights into Algae Factories of the Future. Renew. Sustain. Energy Rev. 2018, 81, 643–654. [Google Scholar] [CrossRef]
- ElMekawy, A.; Hegab, H.M.; Mohanakrishna, G.; Elbaz, A.F.; Bulut, M.; Pant, D. Technological Advances in CO2 Conversion Electro-Biorefinery: A Step toward Commercialization. Bioresour. Technol. 2016, 215, 357–370. [Google Scholar] [CrossRef]
- Judd, S.J.; Al Momani, F.A.O.; Znad, H.; Al Ketife, A.M.D. The Cost Benefit of Algal Technology for Combined CO2 Mitigation and Nutrient Abatement. Renew. Sustain. Energy Rev. 2017, 71, 379–387. [Google Scholar] [CrossRef]
- Baral, S.S.; Singh, K.; Sharma, P. The Potential of Sustainable Algal Biofuel Production Using CO2 from Thermal Power Plant in India. Renew. Sustain. Energy Rev. 2015, 49, 1061–1074. [Google Scholar] [CrossRef]
- Schobert, H.H. Toward the Zero-Emission Coal-to-Liquids Plant. Technology 2015, 03, 147–153. [Google Scholar] [CrossRef]
- Olajire, A.A. Valorization of Greenhouse Carbon Dioxide Emissions into Value-Added Products by Catalytic Processes. J. CO2 Util. 2013, 3–4, 74–92. [Google Scholar] [CrossRef]
- Chiranjeevi, P.; Dahiya, S.; Kumar, N. Waste Derived Bioeconomy in India: A Perspective. N. Biotechnol. 2018, 40, 60–69. [Google Scholar] [CrossRef]
- Cheah, W.Y.; Show, P.L.; Chang, J.-S.; Ling, T.C.; Juan, J.C. Biosequestration of Atmospheric CO2 and Flue Gas-Containing CO2 by Microalgae. Bioresour. Technol. 2015, 184, 190–201. [Google Scholar] [CrossRef] [PubMed]
- Vargas-Estrada, L.; Torres-Arellano, S.; Longoria, A.; Arias, D.M.; Okoye, P.U.; Sebastian, P.J. Role of Nanoparticles on Microalgal Cultivation: A Review. Fuel 2020, 280, 118598. [Google Scholar] [CrossRef]
- Arun, S.; Sinharoy, A.; Pakshirajan, K.; Lens, P.N.L. Algae Based Microbial Fuel Cells for Wastewater Treatment and Recovery of Value-Added Products. Renew. Sustain. Energy Rev. 2020, 132, 110041. [Google Scholar] [CrossRef]
- Zhang, W.; Zhao, C.; Cao, W.; Sun, S.; Hu, C.; Liu, J.; Zhao, Y. Removal of Pollutants from Biogas Slurry and CO2 Capture in Biogas by Microalgae-Based Technology: A Systematic Review. Environ. Sci. Pollut. Res. 2020, 27, 28749–28767. [Google Scholar] [CrossRef]
- Kondaveeti, S.; Abu-Reesh, I.M.; Mohanakrishna, G.; Bulut, M.; Pant, D. Advanced Routes of Biological and Bio-Electrocatalytic Carbon Dioxide (CO2) Mitigation Toward Carbon Neutrality. Front. Energy Res. 2020, 8. [Google Scholar] [CrossRef]
- Zhang, Z.; Pan, S.-Y.; Li, H.; Cai, J.; Olabi, A.G.; Anthony, E.J.; Manovic, V. Recent Advances in Carbon Dioxide Utilization. Renew. Sustain. Energy Rev. 2020, 125, 109799. [Google Scholar] [CrossRef]
- Dineshkumar, R.; Sen, R. A Sustainable Perspective of Microalgal Biorefinery for Co-production and Recovery of High-value Carotenoid and Biofuel with CO2 Valorization. Biofuels Bioprod. Biorefining 2020, 14, 879–897. [Google Scholar] [CrossRef]
- Chen, A.Y.; Lan, E.I. Chemical Production from Methanol Using Natural and Synthetic Methylotrophs. Biotechnol. J. 2020, 15, 1900356. [Google Scholar] [CrossRef]
- Deviram, G.; Mathimani, T.; Anto, S.; Ahamed, T.S.; Ananth, D.A.; Pugazhendhi, A. Applications of Microalgal and Cyanobacterial Biomass on a Way to Safe, Cleaner and a Sustainable Environment. J. Clean. Prod. 2020, 253, 119770. [Google Scholar] [CrossRef]
- Liu, Z.; Wang, K.; Chen, Y.; Tan, T.; Nielsen, J. Third-Generation Biorefineries as the Means to Produce Fuels and Chemicals from CO2. Nat. Catal. 2020, 3, 274–288. [Google Scholar] [CrossRef]
- Patel, A.K.; Choi, Y.Y.; Sim, S.J. Emerging Prospects of Mixotrophic Microalgae: Way Forward to Sustainable Bioprocess for Environmental Remediation and Cost-Effective Biofuels. Bioresour. Technol. 2020, 300, 122741. [Google Scholar] [CrossRef] [PubMed]
- Baral, S.S.; Dionisi, D.; Maarisetty, D.; Gandhi, A.; Kothari, A.; Gupta, G.; Jain, P. Biofuel Production Potential from Wastewater in India by Integrating Anaerobic Membrane Reactor with Algal Photobioreactor. Biomass Bioenergy 2020, 133, 105445. [Google Scholar] [CrossRef]
- Jalilian, N.; Najafpour, G.D.; Khajouei, M. Macro and Micro Algae in Pollution Control and Biofuel Production—A Review. ChemBioEng Rev. 2020, 7, 18–33. [Google Scholar] [CrossRef]
- Bose, A.; Lin, R.; Rajendran, K.; O’Shea, R.; Xia, A.; Murphy, J.D. How to Optimise Photosynthetic Biogas Upgrading: A Perspective on System Design and Microalgae Selection. Biotechnol. Adv. 2019, 37, 107444. [Google Scholar] [CrossRef]
- Roy, M.; Mohanty, K. A Comprehensive Review on Microalgal Harvesting Strategies: Current Status and Future Prospects. Algal Res. 2019, 44, 101683. [Google Scholar] [CrossRef]
- Lin, W.-R.; Tan, S.-I.; Hsiang, C.-C.; Sung, P.-K.; Ng, I.-S. Challenges and Opportunity of Recent Genome Editing and Multi-Omics in Cyanobacteria and Microalgae for Biorefinery. Bioresour. Technol. 2019, 291, 121932. [Google Scholar] [CrossRef]
- Baena-Moreno, F.M.; Rodríguez-Galán, M.; Vega, F.; Alonso-Fariñas, B.; Vilches Arenas, L.F.; Navarrete, B. Carbon Capture and Utilization Technologies: A Literature Review and Recent Advances. Energy Sources Part A Recover. Util. Environ. Eff. 2019, 41, 1403–1433. [Google Scholar] [CrossRef]
- Ho, H.-J.; Iizuka, A.; Shibata, E. Carbon Capture and Utilization Technology without Carbon Dioxide Purification and Pressurization: A Review on Its Necessity and Available Technologies. Ind. Eng. Chem. Res. 2019, 58, 8941–8954. [Google Scholar] [CrossRef]
- SundarRajan, P.; Gopinath, K.P.; Greetham, D.; Antonysamy, A.J. A Review on Cleaner Production of Biofuel Feedstock from Integrated CO2 Sequestration and Wastewater Treatment System. J. Clean. Prod. 2019, 210, 445–458. [Google Scholar] [CrossRef]
- Singh, J.; Dhar, D.W. Overview of Carbon Capture Technology: Microalgal Biorefinery Concept and State-of-the-Art. Front. Mar. Sci. 2019, 6. [Google Scholar] [CrossRef]
- Mathimani, T.; Pugazhendhi, A. Utilization of Algae for Biofuel, Bio-Products and Bio-Remediation. Biocatal. Agric. Biotechnol. 2019, 17, 326–330. [Google Scholar] [CrossRef]
- Lu, L.; Guest, J.S.; Peters, C.A.; Zhu, X.; Rau, G.H.; Ren, Z.J. Wastewater Treatment for Carbon Capture and Utilization. Nat. Sustain. 2018, 1, 750–758. [Google Scholar] [CrossRef]
- Sharma, P.K.; Saharia, M.; Srivstava, R.; Kumar, S.; Sahoo, L. Tailoring Microalgae for Efficient Biofuel Production. Front. Mar. Sci. 2018, 5. [Google Scholar] [CrossRef]
- Benedetti, M.; Vecchi, V.; Barera, S.; Dall’Osto, L. Biomass from Microalgae: The Potential of Domestication towards Sustainable Biofactories. Microb. Cell Fact. 2018, 17, 173. [Google Scholar] [CrossRef]
- Teixeira, L.V.; Moutinho, L.F.; Romão-Dumaresq, A.S. Gas Fermentation of C1 Feedstocks: Commercialization Status and Future Prospects. Biofuels Bioprod. Biorefining 2018, 12, 1103–1117. [Google Scholar] [CrossRef]
- Rafiee, A.; Rajab Khalilpour, K.; Milani, D.; Panahi, M. Trends in CO2 Conversion and Utilization: A Review from Process Systems Perspective. J. Environ. Chem. Eng. 2018, 6, 5771–5794. [Google Scholar] [CrossRef]
- Vuppaladadiyam, A.K.; Yao, J.G.; Florin, N.; George, A.; Wang, X.; Labeeuw, L.; Jiang, Y.; Davis, R.W.; Abbas, A.; Ralph, P.; et al. Impact of Flue Gas Compounds on Microalgae and Mechanisms for Carbon Assimilation and Utilization. ChemSusChem 2018, 11, 334–355. [Google Scholar] [CrossRef]
- Kadir, W.N.A.; Lam, M.K.; Uemura, Y.; Lim, J.W.; Lee, K.T. Harvesting and Pre-Treatment of Microalgae Cultivated in Wastewater for Biodiesel Production: A Review. Energy Convers. Manag. 2018, 171, 1416–1429. [Google Scholar] [CrossRef]
- Laurens, L.M.L.; Lane, M.; Nelson, R.S. Sustainable Seaweed Biotechnology Solutions for Carbon Capture, Composition, and Deconstruction. Trends Biotechnol. 2020, 38, 1232–1244. [Google Scholar] [CrossRef] [PubMed]
- Xiaogang, H.; Jalalah, M.; Jingyuan, W.; Zheng, Y.; Li, X.; Salama, E.-S. Microalgal Growth Coupled with Wastewater Treatment in Open and Closed Systems for Advanced Biofuel Generation. Biomass Convers. Biorefinery 2020. [Google Scholar] [CrossRef]
- Li, P.; Pan, S.Y.; Pei, S.; Lin, Y.J.; Chiang, P.C. Challenges and Perspectives on Carbon Fixation and Utilization Technologies: An Overview. Aerosol Air Qual. Res. 2016, 16, 1327–1344. [Google Scholar] [CrossRef]
- Butti, S.K.; Mohan, S.V. Autotrophic Biorefinery: Dawn of the Gaseous Carbon Feedstock. FEMS Microbiol. Lett. 2017, 364, 1–8. [Google Scholar] [CrossRef]
- Molitor, B.; Richter, H.; Martin, M.E.; Jensen, R.O.; Juminaga, A.; Mihalcea, C.; Angenent, L.T. Carbon Recovery by Fermentation of CO-Rich off Gases—Turning Steel Mills into Biorefineries. Bioresour. Technol. 2016, 215, 386–396. [Google Scholar] [CrossRef]
- Zhang, A.; Carroll, A.L.; Atsumi, S. Carbon Recycling by Cyanobacteria: Improving CO2 Fixation through Chemical Production. FEMS Microbiol. Lett. 2017, 364. [Google Scholar] [CrossRef]
- Lim, M.; Han, G.C.; Ahn, J.W.; You, K.S. Environmental Remediation and Conversion of Carbon Dioxide (CO2) into Useful Green Products by Accelerated Carbonation Technology. Int. J. Environ. Res. Public Health 2010, 7, 203–228. [Google Scholar] [CrossRef]
- Xie, H.; Li, X.; Fang, Z.; Wang, Y.; Li, Q.; Shi, L.; Bai, B.; Wei, N.; Hou, Z. Carbon Geological Utilization and Storage in China: Current Status and Perspectives. Acta Geotech. 2014, 9, 7–27. [Google Scholar] [CrossRef]
- Zhang, J.; Zheng, P.; Wang, Q. Lime Mud from Papermaking Process as a Potential Ameliorant for Pollutants at Ambient Conditions: A Review. J. Clean. Prod. 2015, 103, 828–836. [Google Scholar] [CrossRef]
- Pan, S.Y.; Chiang, A.; Chang, E.E.; Lin, Y.P.; Kim, H.; Chiang, P.C. An Innovative Approach to Integrated Carbon Mineralization and Waste Utilization: A Review. Aerosol Air Qual. Res. 2015, 15, 1072–1091. [Google Scholar] [CrossRef]
- Azdarpour, A.; Asadullah, M.; Mohammadian, E.; Hamidi, H.; Junin, R.; Karaei, M.A. A Review on Carbon Dioxide Mineral Carbonation through PH-Swing Process. Chem. Eng. J. 2015, 279, 615–630. [Google Scholar] [CrossRef]
- Sikarwar, V.S.; Zhao, M.; Clough, P.; Yao, J.; Zhong, X.; Memon, M.Z.; Shah, N.; Anthony, E.J.; Fennell, P.S. An Overview of Advances in Biomass Gasification. Energy Environ. Sci. 2016, 9, 2939–2977. [Google Scholar] [CrossRef]
- Kaliyavaradhan, S.K.; Ling, T.-C.C. Potential of CO2 Sequestration through Construction and Demolition (C&D) Waste—An Overview. J. CO2 Util. 2017, 20, 234–242. [Google Scholar] [CrossRef]
- Rai, S.; Wasewar, K.; Agnihotri, A. Treatment of Alumina Refinery Waste (Red Mud) through Neutralization Techniques: A Review. Waste Manag. Res. 2017, 35, 563–580. [Google Scholar] [CrossRef] [PubMed]
- Rakotonimaro, T.V.; Neculita, C.M.; Bussière, B.; Benzaazoua, M.; Zagury, G.J. Recovery and Reuse of Sludge from Active and Passive Treatment of Mine Drainage-Impacted Waters: A Review. Environ. Sci. Pollut. Res. 2017, 24, 73–91. [Google Scholar] [CrossRef]
- Zhang, D.; Ghouleh, Z.; Shao, Y. Review on Carbonation Curing of Cement-Based Materials. J. CO2 Util. 2017, 21, 119–131. [Google Scholar] [CrossRef]
- Wang, F.; Dreisinger, D.B.; Jarvis, M.; Hitchins, T. The Technology of CO2 Sequestration by Mineral Carbonation: Current Status and Future Prospects. Can. Metall. Q. 2018, 57, 46–58. [Google Scholar] [CrossRef]
- Liu, B.; Qin, J.; Shi, J.; Jiang, J.; Wu, X.; He, Z. New Perspectives on Utilization of CO2 Sequestration Technologies in Cement-Based Materials. Constr. Build. Mater. 2021, 272, 121660. [Google Scholar] [CrossRef]
- Yadav, S.; Mehra, A. A Review on Ex Situ Mineral Carbonation. Environ. Sci. Pollut. Res. 2021, 28, 12202–12231. [Google Scholar] [CrossRef]
- Kelemen, P.B.; McQueen, N.; Wilcox, J.; Renforth, P.; Dipple, G.; Vankeuren, A.P. Engineered Carbon Mineralization in Ultramafic Rocks for CO2 Removal from Air: Review and New Insights. Chem. Geol. 2020, 550, 119628. [Google Scholar] [CrossRef]
- Kaliyavaradhan, S.K.; Ling, T.-C.; Mo, K.H. Valorization of Waste Powders from Cement-Concrete Life Cycle: A Pathway to Circular Future. J. Clean. Prod. 2020, 268, 122358. [Google Scholar] [CrossRef]
- Hills, C.D.; Tripathi, N.; Carey, P.J. Mineralization Technology for Carbon Capture, Utilization, and Storage. Front. Energy Res. 2020, 8. [Google Scholar] [CrossRef]
- Bonfim-Rocha, L.; Silva, A.B.; de Faria, S.H.B.; Vieira, M.F.; de Souza, M. Production of Sodium Bicarbonate from CO2 Reuse Processes: A Brief Review. Int. J. Chem. React. Eng. 2019. [Google Scholar] [CrossRef]
- Liang, C.; Pan, B.; Ma, Z.; He, Z.; Duan, Z. Utilization of CO2 Curing to Enhance the Properties of Recycled Aggregate and Prepared Concrete: A Review. Cem. Concr. Compos. 2020, 105, 103446. [Google Scholar] [CrossRef]
- Woodall, C.M.; McQueen, N.; Pilorgé, H.; Wilcox, J. Utilization of Mineral Carbonation Products: Current State and Potential. Greenh. Gases Sci. Technol. 2019, 9, 1096–1113. [Google Scholar] [CrossRef]
- Azadi, M.; Edraki, M.; Farhang, F.; Ahn, J. Opportunities for Mineral Carbonation in Australia’s Mining Industry. Sustainability 2019, 11, 1250. [Google Scholar] [CrossRef]
- Ibrahim, M.; El-Naas, M.; Benamor, A.; Al-Sobhi, S.; Zhang, Z. Carbon Mineralization by Reaction with Steel-Making Waste: A Review. Processes 2019, 7, 115. [Google Scholar] [CrossRef]
- Humbert, P.S.; Castro-Gomes, J. CO2 Activated Steel Slag-Based Materials: A Review. J. Clean. Prod. 2019, 208, 448–457. [Google Scholar] [CrossRef]
- Krajewska, B. Urease-Aided Calcium Carbonate Mineralization for Engineering Applications: A Review. J. Adv. Res. 2018, 13, 59–67. [Google Scholar] [CrossRef] [PubMed]
- Pipitone, G.; Bolland, O. Power Generation with CO2 Capture: Technology for CO2 Purification. Int. J. Greenh. Gas Control 2009, 3, 528–534. [Google Scholar] [CrossRef]
- Wee, J.H. Carbon Dioxide Emission Reduction Using Molten Carbonate Fuel Cell Systems. Renew. Sustain. Energy Rev. 2014, 32, 178–191. [Google Scholar] [CrossRef]
- Selyanchyn, R.; Fujikawa, S. Membrane Thinning for Efficient CO2 Capture. Sci. Technol. Adv. Mater. 2017, 18, 816–827. [Google Scholar] [CrossRef]
- Pabby, A.K.; Swain, B.; Sastre, A.M. Recent Advances in Smart Integrated Membrane Assisted Liquid Extraction Technology. Chem. Eng. Process. Process Intensif. 2017, 120, 27–56. [Google Scholar] [CrossRef]
- Ding, Y. Perspective on Gas Separation Membrane Materials from Process Economics Point of View. Ind. Eng. Chem. Res. 2020, 59, 556–568. [Google Scholar] [CrossRef]
- Bhatia, S.K.; Bhatia, R.K.; Jeon, J.-M.; Kumar, G.; Yang, Y.-H. Carbon Dioxide Capture and Bioenergy Production Using Biological System—A Review. Renew. Sustain. Energy Rev. 2019, 110, 143–158. [Google Scholar] [CrossRef]
- Song, C.; Liu, Q.; Ji, N.; Deng, S.; Zhao, J.; Li, Y.; Song, Y.; Li, H. Alternative Pathways for Efficient CO2 Capture by Hybrid Processes—A Review. Renew. Sustain. Energy Rev. 2018, 82, 215–231. [Google Scholar] [CrossRef]
- Van Duc Long, N.; Lee, J.; Koo, K.K.; Luis, P.; Lee, M. Recent Progress and Novel Applications in Enzymatic Conversion of Carbon Dioxide. Energies 2017, 10, 473. [Google Scholar] [CrossRef]
- Habib, M.A.; Badr, H.M.; Ahmed, S.F.; Ben-Mansour, R.; Mezghani, K.; Imashuku, S.; la O’, G.J.; Shao-Horn, Y.; Mancini, N.D.; Mitsos, A.; et al. A Review of Recent Developments in Carbon Capture Utilizing Oxy-Fuel Combustion in Conventional and Ion Transport Membrane Systems. Int. J. Energy Res. 2011, 35, 741–764. [Google Scholar] [CrossRef]
- Sarbassov, Y.; Duan, L.; Manovic, V.; Anthony, E.J. Sulfur Trioxide Formation/Emissions in Coal-Fired Air- and Oxy-Fuel Combustion Processes: A Review. Greenh. Gases Sci. Technol. 2018, 8, 402–428. [Google Scholar] [CrossRef]
- Kather, A.; Scheffknecht, G. The Oxycoal Process with Cryogenic Oxygen Supply. Naturwissenschaften 2009, 96, 993–1010. [Google Scholar] [CrossRef] [PubMed]
- Varone, A.; Ferrari, M. Power to Liquid and Power to Gas: An Option for the German Energiewende. Renew. Sustain. Energy Rev. 2015, 45, 207–218. [Google Scholar] [CrossRef]
- Scheffknecht, G.; Al-Makhadmeh, L.; Schnell, U.; Maier, J. Oxy-Fuel Coal Combustion—A Review of the Current State-of-the-Art. Int. J. Greenh. Gas Control 2011, 5, S16–S35. [Google Scholar] [CrossRef]
- Bayham, S.C.; Tong, A.; Kathe, M.; Fan, L.-S. Chemical Looping Technology for Energy and Chemical Production. Wiley Interdiscip. Rev. Energy Environ. 2016, 5, 216–241. [Google Scholar] [CrossRef]
- Adánez, J.; Abad, A.; Mendiara, T.; Gayán, P.; de Diego, L.F.; García-Labiano, F. Chemical Looping Combustion of Solid Fuels. Prog. Energy Combust. Sci. 2018, 65, 6–66. [Google Scholar] [CrossRef]
- Demirel, Y.; Matzen, M.; Winters, C.; Gao, X. Capturing and Using CO2 as Feedstock with Chemical Looping and Hydrothermal Technologies. Int. J. Energy Res. 2015, 39, 1011–1047. [Google Scholar] [CrossRef]
- Blamey, J.; Anthony, E.J.; Wang, J.; Fennell, P.S. The Calcium Looping Cycle for Large-Scale CO2 Capture. Prog. Energy Combust. Sci. 2010, 36, 260–279. [Google Scholar] [CrossRef]
- Liu, W.; An, H.; Qin, C.; Yin, J.; Wang, G.; Feng, B.; Xu, M. Performance Enhancement of Calcium Oxide Sorbents for Cyclic CO2 Capture—A Review. Energy Fuels 2012, 26, 2751–2767. [Google Scholar] [CrossRef]
- Dean, C.C.; Blamey, J.; Florin, N.H.; Al-Jeboori, M.J.; Fennell, P.S. The Calcium Looping Cycle for CO2 Capture from Power Generation, Cement Manufacture and Hydrogen Production. Chem. Eng. Res. Des. 2011, 89, 836–855. [Google Scholar] [CrossRef]
- Esmaeilzadeh, F.; Hamedi, N.; Karimipourfard, D.; Rasoolzadeh, A. An Insight into the Role of the Association Equations of States in Gas Hydrate Modeling: A Review. Pet. Sci. 2020, 17, 1432–1450. [Google Scholar] [CrossRef]
- Meerman, J.C.; Ramírez, A.; Turkenburg, W.C.; Faaij, A.P.C. Performance of Simulated Flexible Integrated Gasification Polygeneration Facilities. Part A: A Technical-Energetic Assessment. Renew. Sustain. Energy Rev. 2011, 15, 2563–2587. [Google Scholar] [CrossRef]
- Muroyama, A.P.; Pătru, A.; Gubler, L. Review - CO2 Separation and Transport via Electrochemical Methods. J. Electrochem. Soc. 2020, 167, 133504. [Google Scholar] [CrossRef]
- Stern, M.C.; Simeon, F.; Herzog, H.; Hatton, T.A. Post-Combustion Carbon Dioxide Capture Using Electrochemically Mediated Amine Regeneration. Energy Environ. Sci. 2013, 6, 2505. [Google Scholar] [CrossRef]
- Oko, E.; Wang, M.; Joel, A.S. Current Status and Future Development of Solvent-Based Carbon Capture. Int. J. Coal Sci. Technol. 2017, 4, 5–14. [Google Scholar] [CrossRef]
- Takht Ravanchi, M.; Sahebdelfar, S. Carbon Dioxide Capture and Utilization in Petrochemical Industry: Potentials and Challenges. Appl. Petrochem. Res. 2014, 4, 63–77. [Google Scholar] [CrossRef]
- Pohlmann, J.; Bram, M.; Wilkner, K.; Brinkmann, T. Pilot Scale Separation of CO2 from Power Plant Flue Gases by Membrane Technology. Int. J. Greenh. Gas Control 2016, 53, 56–64. [Google Scholar] [CrossRef]
- Song, C.; Liu, Q.; Ji, N.; Deng, S.; Zhao, J.; Li, Y.; Kitamura, Y. Reducing the Energy Consumption of Membrane-Cryogenic Hybrid CO2 Capture by Process Optimization. Energy 2017, 124, 29–39. [Google Scholar] [CrossRef]
- Miranda, D.M.V.; de Dutra, L.; Way, D.; Amaral, N.; Wegenast, F.; Scaldaferri, M.C.; Jesus, N.; Pinto, J.C. A Bibliometric Survey of Paraffin/Olefin Separation Using Membranes. Membranes 2019, 9, 157. [Google Scholar] [CrossRef]
- Watkins, J.D.; Siefert, N.S.; Zhou, X.; Myers, C.R.; Kitchin, J.R.; Hopkinson, D.P.; Nulwala, H.B. Redox-Mediated Separation of Carbon Dioxide from Flue Gas. Energy Fuels 2015, 29, 7508–7515. [Google Scholar] [CrossRef]
- Zhang, P.; Tong, J.; Huang, K. Materials and Processes for CO2 Capture, Conversion, and Sequestration. In Materials and Processes for CO2 Capture, Conversion, and Sequestration; Li, L., Wong-Ng, W., Huang, K., Cook, L.P., Eds.; The American Ceramic Society: Hoboken, NJ, USA, 2018; pp. 213–266. ISBN 9781119231066. [Google Scholar]
- Pennline, H.W.; Granite, E.J.; Luebke, D.R.; Kitchin, J.R.; Landon, J.; Weiland, L.M. Separation of CO2 from Flue Gas Using Electrochemical Cells. Fuel 2010, 89, 1307–1314. [Google Scholar] [CrossRef]
- Rheinhardt, J.H.; Singh, P.; Tarakeshwar, P.; Buttry, D.A. Electrochemical Capture and Release of Carbon Dioxide. ACS Energy Lett. 2017, 2, 454–461. [Google Scholar] [CrossRef]
- Reis Machado, A.S.; Nunes da Ponte, M. CO2 Capture and Electrochemical Conversion. Curr. Opin. Green Sustain. Chem. 2018, 11, 86–90. [Google Scholar] [CrossRef]
- Kuramochi, T.; Wu, H.; Ramírez, A.; Faaij, A.; Turkenburg, W. Techno-Economic Prospects for CO2 Capture from a Solid Oxide Fuel Cell-Combined Heat and Power Plant. Preliminary Results. Energy Procedia 2009, 1, 3843–3850. [Google Scholar] [CrossRef][Green Version]
- Discepoli, G.; Cinti, G.; Desideri, U.; Penchini, D.; Proietti, S. International Journal of Greenhouse Gas Control Carbon Capture with Molten Carbonate Fuel Cells: Experimental Tests and Fuel Cell Performance Assessment. Int. J. Greenh. Gas Control 2012, 9, 372–384. [Google Scholar] [CrossRef]
- Campanari, S.; Chiesa, P.; Manzolini, G.; Bedogni, S. Economic Analysis of CO2 Capture from Natural Gas Combined Cycles Using Molten Carbonate Fuel Cells. Appl. Energy 2014, 130, 562–573. [Google Scholar] [CrossRef]
- Patricio, J.; Angelis-Dimakis, A.; Castillo-Castillo, A.; Kalmykova, Y.; Rosado, L. Method to Identify Opportunities for CCU at Regional Level - Matching Sources and Receivers. J. CO2 Util. 2017, 22, 330–345. [Google Scholar] [CrossRef]
- Marocco Stuardi, F.; MacPherson, F.; Leclaire, J. Integrated CO2 Capture and Utilization: A Priority Research Direction. Curr. Opin. Green Sustain. Chem. 2019, 16, 71–76. [Google Scholar] [CrossRef]
- Kim, H.; Lee, J.; Lee, S.; Lee, I.B.; Park, J.; Han, J. Economic Process Design for Separation of CO2 from the Off-Gas in Ironmaking and Steelmaking Plants. Energy 2015, 88, 756–764. [Google Scholar] [CrossRef]
- Zakkour, P.; Cook, G. CCS Roadmap for Industry: High-Purity CO2 Sources. Available online: https://www.researchgate.net/publication/280308375 (accessed on 7 June 2020).
- LanzaTech World’s First Commercial Waste Gas to Ethanol Plant Starts Up. Available online: https://www.lanzatech.com/2018/06/08/worlds-first-commercial-waste-gas-ethanol-plant-starts/ (accessed on 3 March 2020).
- Clifford, C. This Start-up Turns Pollution from Factories into Fuel That Powers Cars—and One Day Planes. Available online: https://www.cnbc.com/2018/07/27/lanzatech-turns-carbon-waste-into-ethanol-to-one-day-power-planes-cars.html (accessed on 19 February 2020).
- Burton, F. Novo Holdings Invests $72 Million in Sustainable Products Leader LanzaTech. Available online: https://www.lanzatech.com/2019/08/06/novo-holdings-invests-72-million-in-sustainable-products-leader-lanzatech/ (accessed on 19 February 2020).
- Global Bioenergies and LanzaTech Strengthen Cooperation. Available online: https://www.global-bioenergies.com/global-bioenergies-and-lanzatech-strengthen-cooperation/?lang=en (accessed on 26 August 2020).
- EPA Presidential Green Chemistry Challenge: 2015 Specific Environmental Benefit: Climate Change Award. Available online: https://www.epa.gov/greenchemistry/presidential-green-chemistry-challenge-2015-specific-environmental-benefit-climate (accessed on 23 March 2021).
- Simpson, S.; Forster, R.; Rowe, M. Microbial Fermentation of Gaseous Substrates to Produce Alcohols. Patent WO2007117157, 18 October 2007. [Google Scholar]
- Tizard, J.H.; Sechrist, P.A. Carbon Capture in Fermentation. Patent WO2010126382A1, 4 November 2010. [Google Scholar]
- Ajjawi, I.; Kuzminov, F.I.; Radakovits, R.R.; Verruto, J.H.; Potts, S.; Spreafico, R.; Lambert, W.F.; Greiner, J.N. Genetic Modulation of Photosynthetic Organisms for Improved Growth. Patent WO2019133726A1, 4 July 2019. [Google Scholar]
- Moellering, E.R.; Edwards, A.R.; Bauman, N. Compositions and Methods for Expressing Genes in Algae. Patent WO2017095960A1, 8 August 2017. [Google Scholar]
- Meuser, J.E.; Dipetrillo, C.G.; McCarren, J.; Bailey, S. Enhanced Productivity by Attenuation of Chlorophyll Binding Protein Genes. Patent WO2017070404A2, 27 April 2017. [Google Scholar]
- Bauman, N.; Ajjawi, I. Microorganisms Engineered for Increased Productivity. Patent WO2017041048A1, 9 March 2017. [Google Scholar]
- Ajjawi, I.; Aqui, M.; Chavez-Torres, C. Biomass Productivity Regulator. Patent WO2015103307A1, 9 July 2015. [Google Scholar]
- Ajjawi, I.; Aqui, M.; Soriaga, L. Compositions and Methods for Modulating Biomass Productivity. Patent WO2015051342A2, 9 April 2015. [Google Scholar]
- Baier, K.; DÜHRING, U.; Enke, H.; GRÜNDEL, M.; Lockau, W.; Smith, C.R.; Woods, P.; Ziegler, K.; Oesterhelt, C.; Coleman, J.R.; et al. Genetically Modified Photoautotrophic Ethanol Producing Host Cells, Method for Producing the Host Cells, Constructs for the Transformation of the Host Cells, Method for Testing a Photoautotrophic Strain for a Desired Growth Property and Method of Producin. Patent WO2009098089A2, 13 August 2009. [Google Scholar]
- Woods, R.P.; Mantecon, E.; Legere, E.; Moll, B.; Unamunzaga, C. Closed Photobioreactor System for Continued Daily in Situ Production, Separation, Collection, and Removal of Ethanol from Genetically Enhanced Photosynthetic Organisms. U.S. Patent 20080153080A1, 23 March 2008. [Google Scholar]
- Moll, B.; McCool, B.; Drake, W.; Porubsky, W.; Adams, R.; Enke, H.; Metzner, J.; Knuth, K. Biofilm Photobioreactor System and Method of Use. U.S. Patent 8691538B1, 8 April 2014. [Google Scholar]
- Ashley, O.; Drake, W.M.I.; Dalrymple, O.K.; Malkiel, E.; Woods, R.P.; Ward, J.K.; Sweeney, K.; Miller, H.L.I. Photobioreactor, System and Method of Use. Patent WO2017112835A8, 29 March 2018. [Google Scholar]
- Scott, B.; Meichel, G.; Phillips-Kress, J.; Blanks, J. Staged Inoculation of Multiple Cyanobacterial Photobioreactors. U.S. Patent 9121012B2, 1 September 2015. [Google Scholar]
- Belicka, L.; Blanks, J.; Flores, H.; Holowell, I.; Jochem, F.; Meichel, G.; Miller, H.; Phillips-Kress, J.; Scott, B.; Vasseur, C. Process for Inoculating Closed Photobioreactors with Cyanobacteria. U.S. Patent 20160024458A1, 28 January 2016. [Google Scholar]
- Piven, I.; Friedrich, A.; Diihring, U.; Uliczka, F.; Baier, K.; Inaba, M.; Shi, T.; Wang, K.; Enke, H.; Kramer, D. Cyanobacterium Sp. Host Cell and Vector for Production of Chemical Compounds in Cyanobacterial Cultures. U.S. Patent 9315832B2, 19 April 2016. [Google Scholar]
- Wang, K.; Shi, T.; Piven, I.; Suzuki, M.; Inaba, M.; Uliczka, F.; Kramer, D.; Enke, H.; Baier, K.; Friedrich, A.; et al. Cyanobacterium Sp. for Production of Compounds. U.S. Patent 009157101B2, 13 October 2015. [Google Scholar]
- Fu, P.P.; Dexter, J. Methods and Compositions for Ethanol Producing Cyanobacteria. Patent WO2007084477A1, 26 July 2007. [Google Scholar]
- Chance, R.; Koros, W.J.; McCool, B.; Noel, J. Water/Carbonate Stripping for CO2 Capture Adsorber Regeneration and CO2 Delivery to Photoautotrophs. Patent WO2011072122A1, 16 June 2011. [Google Scholar]
- Mathers, R.T. How Well Can Renewable Resources Mimic Commodity Monomers and Polymers? J. Polym. Sci. Part A Polym. Chem. 2012, 50, 1–15. [Google Scholar] [CrossRef]
- Exxonmobil and FuelCell Energy Progressing One-of-a-Kind Carbon Capture Fuel Cell Solution. Available online: https://energyfactor.exxonmobil.com/news/fuel-cell-site/ (accessed on 19 August 2020).
- Drax Secures £500,000 for Innovative Fuel Cell Carbon Capture Study. Available online: https://www.drax.com/press_release/drax-secures-500000-innovative-fuel-cell-carbon-capture-study/ (accessed on 19 August 2020).
- 8 Rivers The Allam-Fetvedt Cycle and NET Power. Available online: https://8rivers.com/portfolio/allam-fetvedt-cycle/ (accessed on 23 March 2021).
- Dubourg, S. BASF Recognized as Global Leader in Corporate Climate Action. Available online: https://www.basf.com/eg/en/who-we-are/sustainability/whats-new/sustainability-news/2019/BASF-CDP-global-climate-action-leader.html (accessed on 2 August 2020).
- ExxonMobil, FuelCell Energy Expand Agreement for Carbon Capture Technology. Available online: https://corporate.exxonmobil.com/News/Newsroom/News-releases/2019/1106_ExxonMobil-and-FuelCell-Energy-expand-agreement-for-carbon-capture-technology (accessed on 29 August 2020).
- ExxonMobil and Global Thermostat to Advance Breakthrough Atmospheric Carbon Capture Technology. Available online: https://corporate.exxonmobil.com/News/Newsroom/News-releases/2019/0627_ExxonMobil-and-Global-Thermostat-to-advance-breakthrough-atmospheric--carbon-capture-technology (accessed on 23 March 2021).
- ExxonMobil and Mosaic Materials to Explore New Carbon Capture Technology. Available online: https://corporate.exxonmobil.com/News/Newsroom/News-releases/2019/0826_ExxonMobil-and-Mosaic-Materials-to-explore-new-carbon-capture-technology (accessed on 23 March 2021).
- ExxonMobil Renews Support for MIT Energy Initiative’s Low-Carbon Research. Available online: https://corporate.exxonmobil.com/News/Newsroom/News-releases/2019/1021_ExxonMobil-renews-support-for-MIT-Energy-Initiatives-low-carbon-research (accessed on 23 March 2021).
- ExxonMobil Progresses Growth Plans and Efforts to Advance Lower Emissions Technologies. Available online: https://corporate.exxonmobil.com/News/Newsroom/News-releases/2019/0529_ExxonMobil-progresses-growth-plans-and-efforts-to-advance-lower-emissions-technologies (accessed on 23 March 2021).
- ExxonMobil Collaborates on Discovery of New Material to Enhance Carbon Capture Technology. Available online: https://corporate.exxonmobil.com/News/Newsroom/News-releases/2020/0724_ExxonMobil-collaborates-on-discovery-of-new-material-to-enhance-carbon-capture-technology (accessed on 29 August 2020).
- First Tonne of CO2 Captured in Le Havre for ADEME, Alstom and EDF. Available online: https://www.alstom.com/press-releases-news/2013/7/first-tonne-of-co2-captured-in-le-havre-for-ademe-alstom-and-edf (accessed on 18 August 2020).
- Carbon2Value. Available online: https://www.carbon2value.be/en/ (accessed on 10 August 2020).
- Lanxess to Become Climate Neutral by 2040. Available online: https://lanxess.com/en/Media/Press-Releases/2019/11/LANXESS-to-become-climate-neutral-by-2040 (accessed on 25 August 2020).
- Nesbitt, E.R. Using Waste Carbon Feedstocks to Produce Chemicals. Available online: https://www.usitc.gov/publications/332/working_papers/using_waste_carbon_feedstocks_to_produce_chemicals_0.pdf (accessed on 9 July 2020).
- CO2 Capture - Air Products. Available online: http://www.airproducts.com/Products/Technology-Solutions/Environmental-Energy-Solutions/CO2-Capture.aspx (accessed on 23 August 2020).
- CO2 Capture Plants Achievements. Available online: https://www.mhi.com/products/engineering/products/huanjingzhuangzhico2huishoufuranto.html (accessed on 24 August 2020).
- Mitsubishi Heavy Industries Engineering, Together with Kansai Electric Power Co., Have Already Established and Commercialized Its Own Post Combustion CO2 Capture Technology, so Called KM-CDR ProcessTM. Available online: https://www.mhi.com/products/engineering/co2plants.html (accessed on 26 August 2020).
- Ji, L.; Chang, A.; Driver, M.S.; Timken, H.K. Separations with Ionic Liquid Solvents. U.S. Patent 10131596B2, 20 November 2018. [Google Scholar]
- Ross, M.B.; De Luna, P.; Li, Y.; Dinh, C.-T.; Kim, D.; Yang, P.; Sargent, E.H. Designing Materials for Electrochemical Carbon Dioxide Recycling. Nat. Catal. 2019. [Google Scholar] [CrossRef]
- Alper, E.; Yuksel Orhan, O. CO2 Utilization: Developments in Conversion Processes. Petroleum 2017, 3, 109–126. [Google Scholar] [CrossRef]
- Maeda, C.; Miyazaki, Y.; Ema, T. Recent Progress in Catalytic Conversions of Carbon Dioxide. Catal. Sci. Technol. 2014, 4, 1482–1497. [Google Scholar] [CrossRef]
- Balasubramanian, P.; Bajaj, I.; Hasan, M.M.F. Simulation and Optimization of Reforming Reactors for Carbon Dioxide Utilization Using Both Rigorous and Reduced Models. J. CO2 Util. 2018, 23, 80–104. [Google Scholar] [CrossRef]
- Artz, J.; Müller, T.E.; Thenert, K.; Kleinekorte, J.; Meys, R.; Sternberg, A.; Bardow, A.; Leitner, W. Sustainable Conversion of Carbon Dioxide: An Integrated Review of Catalysis and Life Cycle Assessment. Chem. Rev. 2018, 118, 434–504. [Google Scholar] [CrossRef] [PubMed]
- Sahebdelfar, S.; Ravanchi, M.T.; Gharibi, M.; Hamidzadeh, M. Rule of 100: An Inherent Limitation or Performance Measure in Oxidative Coupling of Methane? J. Nat. Gas Chem. 2012, 21, 308–313. [Google Scholar] [CrossRef]
- Tursunov, O.; Kustov, L.; Kustov, A. A Brief Review of Carbon Dioxide Hydrogenation to Methanol Over Copper and Iron Based Catalysts. Oil Gas Sci. Technol. 2017, 72, 545–567. [Google Scholar] [CrossRef]
- Centi, G.; Iaquaniello, G.; Perathoner, S. Can We Afford to Waste Carbon Dioxide? Carbon Dioxide as a Valuable Source of Carbon for the Production of Light Olefins. ChemSusChem 2011, 4, 1265–1273. [Google Scholar] [CrossRef]
- Taheri Najafabadi, A. CO2 Chemical Conversion to Useful Products: An Engineering Insight to the Latest Advances toward Sustainability. Int. J. Energy Res. 2013, 37, 485–499. [Google Scholar] [CrossRef]
- Macelroy, J.M.D. Closing the Carbon Cycle through Rational Use of Carbon-Based Fuels. Ambio 2016, 45, 5–14. [Google Scholar] [CrossRef]
- Alaba, P.A.; Abbas, A.; Daud, W.M.W. Insight into Catalytic Reduction of CO2: Catalysis and Reactor Design. J. Clean. Prod. 2017, 140, 1298–1312. [Google Scholar] [CrossRef]
- Sarp, S.; Gonzalez Hernandez, S.; Chen, C.; Sheehan, S.W. Alcohol Production from Carbon Dioxide: Methanol as a Fuel and Chemical Feedstock. Joule 2021, 5, 59–76. [Google Scholar] [CrossRef]
- De, S.; Dokania, A.; Ramirez, A.; Gascon, J. Advances in the Design of Heterogeneous Catalysts and Thermocatalytic Processes for CO2 Utilization. ACS Catal. 2020, 10, 14147–14185. [Google Scholar] [CrossRef]
- Xie, S.; Zhang, W.; Lan, X.; Lin, H. CO2 Reduction to Methanol in the Liquid Phase: A Review. ChemSusChem 2020. [Google Scholar] [CrossRef] [PubMed]
- Chakrabortty, S.; Nayak, J.; Ruj, B.; Pal, P.; Kumar, R.; Banerjee, S.; Sardar, M.; Chakraborty, P. Photocatalytic Conversion of CO2 to Methanol Using Membrane-Integrated Green Approach: A Review on Capture, Conversion and Purification. J. Environ. Chem. Eng. 2020, 8, 103935. [Google Scholar] [CrossRef]
- Sun, Y.; Lin, Z.; Peng, S.H.; Sage, V.; Sun, Z. A Critical Perspective on CO2 Conversions into Chemicals and Fuels. J. Nanosci. Nanotechnol. 2019, 19, 3097–3109. [Google Scholar] [CrossRef]
- Brunetti, A.; Fontananova, E. CO2 Conversion by Membrane Reactors. J. Nanosci. Nanotechnol. 2019, 19, 3124–3134. [Google Scholar] [CrossRef]
- Boaro, M.; Colussi, S.; Trovarelli, A. Ceria-Based Materials in Hydrogenation and Reforming Reactions for CO2 Valorization. Front. Chem. 2019, 7. [Google Scholar] [CrossRef]
- Leonzio, G. State of Art and Perspectives about the Production of Methanol, Dimethyl Ether and Syngas by Carbon Dioxide Hydrogenation. J. CO2 Util. 2018, 27, 326–354. [Google Scholar] [CrossRef]
- Yu, B.; Diao, Z.-F.; Guo, C.-X.; He, L.-N. Carboxylation of Olefins/Alkynes with CO2 to Industrially Relevant Acrylic Acid Derivatives. J. CO2 Util. 2013, 1, 60–68. [Google Scholar] [CrossRef]
- Lv, H.; Yang, L.; Zhou, J.; Zhang, X.; Wu, W.; Li, Y.; Jiang, D. Water Resource Synergy Management in Response to Climate Change in China: From the Perspective of Urban Metabolism. Resour. Conserv. Recycl. 2020, 163, 105095. [Google Scholar] [CrossRef]
- Saini, S.; Prajapati, P.K.; Jain, S.L. Transition Metal-Catalyzed Carboxylation of Olefins with Carbon Dioxide: A Comprehensive Review. Catal. Rev. 2020, 1–47. [Google Scholar] [CrossRef]
- Parvez, A.M.; Afzal, M.T.; Victor Hebb, T.G.; Schmid, M. Utilization of CO2 in Thermochemical Conversion of Biomass for Enhanced Product Properties: A Review. J. CO2 Util. 2020, 40, 101217. [Google Scholar] [CrossRef]
- Dou, B.; Zhang, H.; Song, Y.; Zhao, L.; Jiang, B.; He, M.; Ruan, C.; Chen, H.; Xu, Y. Hydrogen Production from the Thermochemical Conversion of Biomass: Issues and Challenges. Sustain. Energy Fuels 2019, 3, 314–342. [Google Scholar] [CrossRef]
- Hamawand, I.; Yusaf, T.; Hamawand, S.G. Coal Seam Gas and Associated Water: A Review Paper. Renew. Sustain. Energy Rev. 2013, 22, 550–560. [Google Scholar] [CrossRef]
- Anon Enhanced Coalbed-Methane Recovery by Use of CO2. JPT J. Pet. Technol. 1999, 51, 1999.
- Tang, Y.; Yang, R.; Bian, X. A Review of CO2 Sequestration Projects and Application in China. Sci. World J. 2014, 2014, 1–11. [Google Scholar] [CrossRef]
- Iglauer, S.; Lebedev, M. High Pressure-Elevated Temperature x-Ray Micro-Computed Tomography for Subsurface Applications. Adv. Colloid Interface Sci. 2018, 256, 393–410. [Google Scholar] [CrossRef] [PubMed]
- Perera, M.S.A. Influences of CO2 Injection into Deep Coal Seams: A Review. Energy Fuels 2017, 31, 10324–10334. [Google Scholar] [CrossRef]
- Liu, H.J.; Were, P.; Li, Q.; Gou, Y.; Hou, Z. Worldwide Status of CCUS Technologies and Their Development and Challenges in China. Geofluids 2017, 2017, 1–25. [Google Scholar] [CrossRef]
- Perera, M.; Gamage, R.; Rathnaweera, T.; Ranathunga, A.; Koay, A.; Choi, X. A Review of CO2-Enhanced Oil Recovery with a Simulated Sensitivity Analysis. Energies 2016, 9, 481. [Google Scholar] [CrossRef]
- Cherepovitsyn, A.; Ilinova, A. Ecological, Economic and Social Issues of Implementing Carbon Dioxide Sequestration Technologies in the Oil and Gas Industry in Russia. J. Ecol. Eng. 2016, 17, 19–23. [Google Scholar] [CrossRef]
- Wang, K.; Wang, G.; Ren, T.; Cheng, Y. Methane and CO2 Sorption Hysteresis on Coal: A Critical Review. Int. J. Coal Geol. 2014, 132, 60–80. [Google Scholar] [CrossRef]
- Poordad, S.; Forutan, M.K. A Review of the Potential for CO2 Sequestration and Enhanced Gas Recovery in an Iranian Gas Condensate Reservoir from a Fluid Properties Point of View. Pet. Sci. Technol. 2013, 31, 2157–2165. [Google Scholar] [CrossRef]
- Cannone, S.F.; Lanzini, A.; Santarelli, M. A Review on CO2 Capture Technologies with Focus on CO2-Enhanced Methane Recovery from Hydrates. Energies 2021, 14, 387. [Google Scholar] [CrossRef]
- Hamza, A.; Hussein, I.A.; Al-Marri, M.J.; Mahmoud, M.; Shawabkeh, R.; Aparicio, S. CO2 Enhanced Gas Recovery and Sequestration in Depleted Gas Reservoirs: A Review. J. Pet. Sci. Eng. 2021, 196, 107685. [Google Scholar] [CrossRef]
- Teodoriu, C.; Bello, O. A Review of Cement Testing Apparatus and Methods under CO2 Environment and Their Impact on Well Integrity Prediction—Where Do We Stand? J. Pet. Sci. Eng. 2020, 187, 106736. [Google Scholar] [CrossRef]
- Cao, C.; Liu, H.; Hou, Z.; Mehmood, F.; Liao, J.; Feng, W. A Review of CO2 Storage in View of Safety and Cost-Effectiveness. Energies 2020, 13, 600. [Google Scholar] [CrossRef]
- Wang, Z.M.; Song, G.-L.; Zhang, J. Corrosion Control in CO2 Enhanced Oil Recovery From a Perspective of Multiphase Fluids. Front. Mater. 2019, 6. [Google Scholar] [CrossRef]
- Du, F.; Nojabaei, B. A Review of Gas Injection in Shale Reservoirs: Enhanced Oil/Gas Recovery Approaches and Greenhouse Gas Control. Energies 2019, 12, 2355. [Google Scholar] [CrossRef]
- Rani, S.; Padmanabhan, E.; Prusty, B.K. Review of Gas Adsorption in Shales for Enhanced Methane Recovery and CO2 Storage. J. Pet. Sci. Eng. 2019, 175, 634–643. [Google Scholar] [CrossRef]
- Goraya, N.S.; Rajpoot, N.; Marriyappan Sivagnanam, B. Coal Bed Methane Enhancement Techniques: A Review. ChemistrySelect 2019, 4, 3585–3601. [Google Scholar] [CrossRef]
- Nasirpour, N.; Mohammadpourfard, M.; Zeinali Heris, S. Ionic Liquids: Promising Compounds for Sustainable Chemical Processes and Applications. Chem. Eng. Res. Des. 2020, 160, 264–300. [Google Scholar] [CrossRef]
- Doherty, A.P.; Diaconu, L.; Marley, E.; Spedding, P.L.; Barhdadi, R.; Troupel, M. Application of Clean Technologies Using Electrochemistry in Ionic Liquids. Asia-Pacific J. Chem. Eng. 2012, 7, 14–23. [Google Scholar] [CrossRef]
- Koornneef, J.; Ramírez, A.; Turkenburg, W.; Faaij, A. The Environmental Impact and Risk Assessment of CO2 Capture, Transport and Storage—An Evaluation of the Knowledge Base. Prog. Energy Combust. Sci. 2012, 38, 62–86. [Google Scholar] [CrossRef]
- Lu, Q.; Jiao, F. Electrochemical CO2 Reduction: Electrocatalyst, Reaction Mechanism, and Process Engineering. Nano Energy 2016, 29, 439–456. [Google Scholar] [CrossRef]
- Malik, K.; Singh, S.; Basu, S.; Verma, A. Electrochemical Reduction of CO2 for Synthesis of Green Fuel. Wiley Interdiscip. Rev. Energy Environ. 2017, 6. [Google Scholar] [CrossRef]
- Du, D.; Lan, R.; Humphreys, J.; Tao, S. Progress in Inorganic Cathode Catalysts for Electrochemical Conversion of Carbon Dioxide into Formate or Formic Acid. J. Appl. Electrochem. 2017, 47, 661–678. [Google Scholar] [CrossRef]
- Vasileff, A.; Xu, C.; Jiao, Y.; Zheng, Y.; Qiao, S.-Z. Surface and Interface Engineering in Copper-Based Bimetallic Materials for Selective CO2 Electroreduction. Chem 2018, 4, 1809–1831. [Google Scholar] [CrossRef]
- Abdelkader-Fernández, V.K.; Fernandes, D.M.; Freire, C. Carbon-Based Electrocatalysts for CO2 Electroreduction Produced via MOF, Biomass, and Other Precursors Carbonization: A Review. J. CO2 Util. 2020, 42, 101350. [Google Scholar] [CrossRef]
- Ouyang, T.; Huang, S.; Wang, X.; Liu, Z. Nanostructures for Electrocatalytic CO2 Reduction. Chem. A Eur. J. 2020, 26, 14024–14035. [Google Scholar] [CrossRef] [PubMed]
- Smith, W.A.; Burdyny, T.; Vermaas, D.A.; Geerlings, H. Pathways to Industrial-Scale Fuel Out of Thin Air from CO2 Electrolysis. Joule 2019, 3, 1822–1834. [Google Scholar] [CrossRef]
- Feng, J.J.; Zeng, S.; Feng, J.J.; Dong, H.; Zhang, X. CO2 Electroreduction in Ionic Liquids: A Review. Chinese J. Chem. 2018, 36, 961–970. [Google Scholar] [CrossRef]
- Muthuraj, R.; Mekonnen, T. Recent Progress in Carbon Dioxide (CO2) as Feedstock for Sustainable Materials Development: Co-Polymers and Polymer Blends. Polymer 2018, 145, 348–373. [Google Scholar] [CrossRef]
- Tao, L.; Choksi, T.S.; Liu, W.; Pérez-Ramírez, J. Synthesizing High-Volume Chemicals from CO2 without Direct H2 Input. ChemSusChem 2020. [Google Scholar] [CrossRef]
- Luo, R.; Chen, M.; Liu, X.; Xu, W.; Li, J.; Liu, B.; Fang, Y. Recent Advances in CO2 Capture and Simultaneous Conversion into Cyclic Carbonates over Porous Organic Polymers Having Accessible Metal Sites. J. Mater. Chem. A 2020, 8, 18408–18424. [Google Scholar] [CrossRef]
- Singh Dhankhar, S.; Ugale, B.; Nagaraja, C.M. Co-Catalyst-Free Chemical Fixation of CO2 into Cyclic Carbonates by Using Metal-Organic Frameworks as Efficient Heterogeneous Catalysts. Chem. Asian J. 2020, 15, 2403–2427. [Google Scholar] [CrossRef] [PubMed]
- Kiatkittipong, K.; Mohamad Shukri, M.A.A.; Kiatkittipong, W.; Lim, J.W.; Show, P.L.; Lam, M.K.; Assabumrungrat, S. Green Pathway in Utilizing CO2 via Cycloaddition Reaction with Epoxide—A Mini Review. Processes 2020, 8, 548. [Google Scholar] [CrossRef]
- Cui, S.; Borgemenke, J.; Liu, Z.; Li, Y. Recent Advances of “Soft” Bio-Polycarbonate Plastics from Carbon Dioxide and Renewable Bio-Feedstocks via Straightforward and Innovative Routes. J. CO2 Util. 2019, 34, 40–52. [Google Scholar] [CrossRef]
- Shaikh, R.R.; Pornpraprom, S.; D’Elia, V. Catalytic Strategies for the Cycloaddition of Pure, Diluted, and Waste CO2 to Epoxides under Ambient Conditions. ACS Catal. 2018, 8, 419–450. [Google Scholar] [CrossRef]
- Skoda-Foldes, R.; Kollar, L. Synthetic Applications of Palladium Catalysed Carbonylation of Organic Halides. Curr. Org. Chem. 2002, 6, 1097–1119. [Google Scholar] [CrossRef]
- Simakov, D.S.A.; Wright, M.M.; Ahmed, S.; Mokheimer, E.M.A.; Román-Leshkov, Y. Solar Thermal Catalytic Reforming of Natural Gas: A Review on Chemistry, Catalysis and System Design. Catal. Sci. Technol. 2015, 5, 1991–2016. [Google Scholar] [CrossRef]
- Jiang, Z.; Xiao, T.; Kuznetsov, V.L.; Edwards, P.P. Turning Carbon Dioxide into Fuel. Philos. Trans. R. Soc. A Math. Phys. Eng. Sci. 2010, 368, 3343–3364. [Google Scholar] [CrossRef]
- Navarro, J.C.; Centeno, M.A.; Laguna, O.H.; Odriozola, J.A. Policies and Motivations for the CO2 Valorization through the Sabatier Reaction Using Structured Catalysts. A Review of the Most Recent Advances. Catalysts 2018, 8, 578. [Google Scholar] [CrossRef]
- Aziz, M.A.A.; Jalil, A.A.; Triwahyono, S.; Ahmad, A. CO2 Methanation over Heterogeneous Catalysts: Recent Progress and Future Prospects. Green Chem. 2015, 17, 2647–2663. [Google Scholar] [CrossRef]
- Tahir, M.; Saidina, N. Recycling of Carbon Dioxide to Renewable Fuels by Photocatalysis: Prospects and Challenges. Renew. Sustain. Energy Rev. 2013, 25, 560–579. [Google Scholar] [CrossRef]
- De Richter, R.K.; Ming, T.; Caillol, S. Fighting Global Warming by Photocatalytic Reduction of CO2 Using Giant Photocatalytic Reactors. Renew. Sustain. Energy Rev. 2013, 19, 82–106. [Google Scholar] [CrossRef]
- Izumi, Y. Recent Advances in the Photocatalytic Conversion of Carbon Dioxide to Fuels with Water and/or Hydrogen Using Solar Energy and Beyond. Coord. Chem. Rev. 2013, 257, 171–186. [Google Scholar] [CrossRef]
- Katagiri, T.; Amao, Y. Recent Advances in Light-Driven C–H Bond Activation and Building C–C Bonds with CO2 as a Feedstock for Carbon Capture and Utilization Technology. Green Chem. 2020, 22, 6682–6713. [Google Scholar] [CrossRef]
- Proppe, A.H.; Li, Y.C.; Aspuru-Guzik, A.; Berlinguette, C.P.; Chang, C.J.; Cogdell, R.; Doyle, A.G.; Flick, J.; Gabor, N.M.; van Grondelle, R.; et al. Bioinspiration in Light Harvesting and Catalysis. Nat. Rev. Mater. 2020, 5, 828–846. [Google Scholar] [CrossRef]
- Li, F.; Thevenon, A.; Rosas-Hernández, A.; Wang, Z.; Li, Y.; Gabardo, C.M.; Ozden, A.; Dinh, C.T.; Li, J.; Wang, Y.; et al. Molecular Tuning of CO2-to-Ethylene Conversion. Nature 2020, 577, 509–513. [Google Scholar] [CrossRef]
- Jones, J.P.; Prakash, G.K.S.; Olah, G.A. Electrochemical CO2 Reduction: Recent Advances and Current Trends. Isr. J. Chem. 2014, 54, 1451–1466. [Google Scholar] [CrossRef]
- Weekes, D.M.; Salvatore, D.A.; Reyes, A.; Huang, A.; Berlinguette, C.P. Electrolytic CO2 Reduction in a Flow Cell. Acc. Chem. Res. 2018, 51, 910–918. [Google Scholar] [CrossRef]
- De Luna, P.; Hahn, C.; Higgins, D.; Jaffer, S.A.; Jaramillo, T.F.; Sargent, E.H. What Would It Take for Renewably Powered Electrosynthesis to Displace Petrochemical Processes? Science 2019, 364. [Google Scholar] [CrossRef] [PubMed]
- Pappijn, C.A.R.; Ruitenbeek, M.; Reyniers, M.F.; Van Geem, K.M. Challenges and Opportunities of Carbon Capture and Utilization: Electrochemical Conversion of CO2 to Ethylene. Front. Earth Sci. 2020, 8, 1–12. [Google Scholar] [CrossRef]
- Remiro-Buenamañana, S.; García, H. Photoassisted CO2 Conversion to Fuels. ChemCatChem 2019, 11, 342–356. [Google Scholar] [CrossRef]
- Qiao, J.; Liu, Y.; Hong, F.; Zhang, J. A Review of Catalysts for the Electroreduction of Carbon Dioxide to Produce Low-Carbon Fuels. Chem. Soc. Rev. 2014, 43, 631–675. [Google Scholar] [CrossRef] [PubMed]
- Wittich, K.; Krämer, M.; Bottke, N.; Schunk, S.A. Catalytic Dry Reforming of Methane: Insights from Model Systems. ChemCatChem 2020, 12, 2130–2147. [Google Scholar] [CrossRef]
- Amin, N.A.S. A Hybrid Numerical Approach for Multi-Responses Optimization of Process Parameters and Catalyst Compositions in CO2 OCM Process over CaO-MnO/CeO2 Catalyst. Chem. Eng. J. 2005, 106, 213–227. [Google Scholar] [CrossRef][Green Version]
- Nikoo, M.K.; Amin, N.A.S. Thermodynamic Analysis of Carbon Dioxide Reforming of Methane in View of Solid Carbon Formation. Fuel Process. Technol. 2011, 92, 678–691. [Google Scholar] [CrossRef]
- Liu, M.; Yi, Y.; Wang, L.; Guo, H.; Bogaerts, A. Hydrogenation of Carbon Dioxide to Value-Added Chemicals by Heterogeneous Catalysis and Plasma Catalysis. Catalysts 2019, 19, 275. [Google Scholar] [CrossRef]
- Carbon Recycling International. Available online: https://www.carbonrecycling.is/ (accessed on 23 March 2021).
- Fazeli, H.; Panahi, M.; Rafiee, A. Investigating the Potential of Carbon Dioxide Utilization in a Gas-to-Liquids Process with Iron-Based Fischer-Tropsch Catalyst. J. Nat. Gas Sci. Eng. 2018, 52, 549–558. [Google Scholar] [CrossRef]
- Ronda-Lloret, M.; Rothenberg, G.; Shiju, N.R. A Critical Look at Direct Catalytic Hydrogenation of Carbon Dioxide to Olefins. ChemSusChem 2019, 12, 3896–3914. [Google Scholar] [CrossRef] [PubMed]
- Dimitriou, I.; García-Gutiérrez, P.; Elder, R.H.; Cuéllar-Franca, R.M.; Azapagic, A.; Allen, R.W.K. Carbon Dioxide Utilisation for Production of Transport Fuels: Process and Economic Analysis. Energy Environ. Sci. 2015, 8, 1775–1789. [Google Scholar] [CrossRef]
- El-emam, R.S.; Ozcan, H. Comprehensive Review on the Techno-Economics of Sustainable Large-Scale Clean Hydrogen Production. J. Clean. Prod. 2019, 220. [Google Scholar] [CrossRef]
- Melo Bravo, P.; Debecker, D.P. Combining CO2 Capture and Catalytic Conversion to Methane. Waste Dispos. Sustain. Energy 2019, 1, 53–65. [Google Scholar] [CrossRef]
- Jin Lee, W.; Li, C.; Prajitno, H.; Yoo, J.; Patel, J.; Yang, Y.; Lim, S. Recent Trend in Thermal Catalytic Low Temperature CO2 Methanation: A Critical Review. Catal. Today 2020, 153057. [Google Scholar] [CrossRef]
- Fahmy, T.Y.A.; Fahmy, Y.; Mobarak, F.; El-Sakhawy, M.; Abou-Zeid, R.E. Biomass Pyrolysis: Past, Present, and Future. Environ. Dev. Sustain. 2020, 22, 17–32. [Google Scholar] [CrossRef]
- Wang, S.; Wang, P.; Shi, D.; He, S.; Zhang, L.; Yan, W.; Qin, Z.; Li, J.; Dong, M.; Wang, J.; et al. Direct Conversion of Syngas into Light Olefins with Low CO2 Emission. ACS Catal. 2020, 10, 2046–2059. [Google Scholar] [CrossRef]
- Zhu, Y.; Pan, X.; Jiao, F.; Li, J.; Yang, J.; Ding, M.; Han, Y.; Liu, Z.; Bao, X. Role of Manganese Oxide in Syngas Conversion to Light Ole Fi Ns. ACS Catal. 2017, 7, 2800–2804. [Google Scholar] [CrossRef]
- Jiao, F.; Li, J.; Pan, X.; Xiao, J.; Li, H.; Ma, H.; Wei, M.; Pan, Y.; Zhou, Z.; Li, M.; et al. Selective Conversion of Syngas to Light Olefins. Science 2016, 351, 1065–1068. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Zhou, W.; Yang, Y.; Cheng, K.; Kang, J.; Zhang, L.; Zhang, G.; Min, X.; Zhang, Q.; Wang, Y. Design of Efficient Bifunctional Catalysts for Direct Conversion of Syngas into Lower Olefins via Methanol/Dimethyl Ether Intermediates. Chem. Sci. 2018, 4708–4718. [Google Scholar] [CrossRef]
- Marion, M.C.; Tasso, A.; Cornaro, U. Method for Optimizing the Operation of a Hydrocarbon Synthesis Unit Starting from Synthesis Gas, by Controlling the Partial Pressure of CO. U.S. Patent 8940802B2, 27 January 2015. [Google Scholar]
- Ail, S.S.; Dasappa, S. Biomass to Liquid Transportation Fuel via Fischer Tropsch Synthesis—Technology Review and Current Scenario. Renew. Sustain. Energy Rev. 2016, 58, 267–286. [Google Scholar] [CrossRef]
- Jalama, K. Carbon Dioxide Hydrogenation over Nickel-, Ruthenium-, and Copper-Based Catalysts: Review of Kinetics and Mechanism. Catal. Rev. Sci. Eng. 2017, 59, 95–164. [Google Scholar] [CrossRef]
- Aresta, M.; Nocito, F.; Dibenedetto, A. What Catalysis Can Do for Boosting CO2 Utilization. In Advances in Catalysis; Song, C., Ed.; Elsevier Inc.: Amsterdam, The Netherlands, 2018; Volume 62, pp. 49–111. ISBN 9780128150887. [Google Scholar]
- Garbarino, G.; Bellotti, D.; Riani, P.; Magistri, L.; Busca, G. Methanation of Carbon Dioxide on Ru/Al2O3 and Ni/Al2O3 Catalysts at Atmospheric Pressure: Catalysts Activation, Behaviour and Stability. Int. J. Hydrogen Energy 2015, 40, 9171–9182. [Google Scholar] [CrossRef]
- Stangeland, K.; Kalai, D.; Li, H.; Yu, Z. CO2 Methanation: The Effect of Catalysts and Reaction Conditions. Energy Procedia 2017, 105, 2022–2027. [Google Scholar] [CrossRef]
- Inui, T. Highly Effective Conversion of Carbon Dioxide to Valuable Compounds on Composite Catalysts. Catal. Today 1996, 29, 329–337. [Google Scholar] [CrossRef]
- Janssen, M.J.; van Egmond, C.F.; Martens, L.R.M.; Sher, J. Methanol and Fuel Alcohol Production for an Oxygenate to Olefin Reaction System. U.S. Patent 7288689B2, 30 October 2007. [Google Scholar]
- Vogt, C.; Monai, M.; Sterk, E.B.; Palle, J.; Melcherts, A.E.M.; Zijlstra, B.; Groeneveld, E.; Berben, P.H.; Boereboom, J.M.; Hensen, E.J.M.; et al. Understanding Carbon Dioxide Activation and Carbon–Carbon Coupling over Nickel. Nat. Commun. 2019, 10, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Galadima, A.; Muraza, O. From Synthesis Gas Production to Methanol Synthesis and Potential Upgrade to Gasoline Range Hydrocarbons: A Review. J. Nat. Gas Sci. Eng. 2015, 25, 303–316. [Google Scholar] [CrossRef]
- Yang, M.; Fan, D.; Wei, Y.; Tian, P.; Liu, Z. Recent Progress in Methanol-to-Olefins (MTO) Catalysts. Adv. Mater. 2019, 31, 1–15. [Google Scholar] [CrossRef]
- Masudi, A.; Jusoh, N.W.C.; Muraza, O. Opportunities for Less-Explored Zeolitic Materials in the Syngas-to-Olefins Pathway over Nanoarchitectured Catalysts: A Mini Review. Catal. Sci. Technol. 2020, 10, 1582–1596. [Google Scholar] [CrossRef]
- Wang, C.; Xu, J.; Deng, F. Mechanism of Methanol-to-Hydrocarbon Reaction over Zeolites: A Solid-State NMR Perspective. ChemCatChem 2020. [Google Scholar] [CrossRef]
- Liu, Z.; Liang, J. Methanol to Olefin Conversion Catalysts. Curr. Opin. Solid State Mater. Sci. 1999, 4, 80–84. [Google Scholar] [CrossRef]
- Chen, Q.; Lv, M.; Wang, D.; Tang, Z.; Wei, W.; Sun, Y. Eco-Efficiency Assessment for Global Warming Potential of Ethylene Production Processes: A Case Study of China. J. Clean. Prod. 2017, 142, 3109–3116. [Google Scholar] [CrossRef]
- Xiang, D.; Yang, S.; Li, X.; Qian, Y. Life Cycle Assessment of Energy Consumption and GHG Emissions of Olefins Production from Alternative Resources in China. Energy Convers. Manag. 2015, 90, 12–20. [Google Scholar] [CrossRef]
- Kleij, A.W.; North, M.; Urakawa, A. CO2 Catalysis. ChemSusChem 2017, 10, 1036–1038. [Google Scholar] [CrossRef] [PubMed]
- Mobley, P.D.; Peters, J.E.; Akunuri, N.; Hlebak, J.; Gupta, V.; Zheng, Q.; Zhou, S.J.; Lail, M. Utilization of CO2 for Ethylene Oxide. Energy Procedia 2017, 114, 7154–7161. [Google Scholar] [CrossRef]
- Covestro Greenhouse Gas Is Becoming a Useful Raw Material. Available online: https://www.covestro.de/en/projects-and-cooperations/overview (accessed on 11 August 2020).
- Covestro Cardyon®: A Polyol That Makes Environmental Sense. Available online: https://solutions.covestro.com/en/brands/cardyon (accessed on 11 August 2020).
- Haider, K.W.; Mcdaniel, K.G.; Hayes, J.E.; Shen, J. Polyether Carbonate Polyols Made via Double Metal Cyanide (DMC) Catalysis. U.S. Patent 8134022B2, 13 March 2012. [Google Scholar]
- Kamphuis, A.J.; Picchioni, F.; Pescarmona, P.P. CO2-Fixation into Cyclic and Polymeric Carbonates: Principles and Applications. Green Chem. 2019, 21, 406–448. [Google Scholar] [CrossRef]
- Bugrova, T.A.; Dutov, V.V.; Svetlichnyi, V.A.; Cortés Corberán, V.; Mamontov, G.V. Oxidative Dehydrogenation of Ethane with CO2 over CrOx Catalysts Supported on Al2O3, ZrO2, CeO2 and CexZr1-XO2. Catal. Today 2019, 71–80. [Google Scholar] [CrossRef]
- Gomez, E.; Yan, B.; Kattel, S.; Chen, J.G. Carbon Dioxide Reduction in Tandem with Light-Alkane Dehydrogenation. Nat. Rev. Chem. 2019, 3, 638–649. [Google Scholar] [CrossRef]
- Zhang, L.; Wu, Z.; Nelson, N.C.; Sadow, A.D.; Slowing, I.I.; Overbury, S.H. Role of CO2 As a Soft Oxidant for Dehydrogenation of Ethylbenzene to Styrene over a High-Surface-Area Ceria Catalyst. ACS Catal. 2015, 5, 6426–6435. [Google Scholar] [CrossRef]
- Cavani, F.; Ballarini, N.; Cericola, A. Oxidative Dehydrogenation of Ethane and Propane: How Far from Commercial Implementation? Catal. Today 2007, 127, 113–131. [Google Scholar] [CrossRef]
- Thirumala Bai, P.; Srinath, S.; Upendar, K.; Sagar, T.V.; Lingaiah, N.; Rama Rao, K.S.; Sai Prasad, P.S. Oxidative Dehydrogenation of Ethane with Carbon Dioxide over Cr2O3/SBA-15 Catalysts: The Influence of Sulfate Modification of the Support. Appl. Petrochemical Res. 2017, 7, 107–118. [Google Scholar] [CrossRef]
- Yan, B.; Yao, S.; Kattel, S.; Wu, Q.; Xie, Z.; Gomez, E.; Liu, P.; Su, D.; Chen, J.G. Active Sites for Tandem Reactions of CO2 Reduction and Ethane Dehydrogenation. Proc. Natl. Acad. Sci. USA 2018, 115, 8278–8283. [Google Scholar] [CrossRef]
- Ansari, M.B.; Park, S.E. Carbon Dioxide Utilization as a Soft Oxidant and Promoter in Catalysis. Energy Environ. Sci. 2012, 5, 9419–9437. [Google Scholar] [CrossRef]
- Palm, E.; Nilsson, L.J.; Åhman, M. Electricity-Based Plastics and Their Potential Demand for Electricity and Carbon Dioxide. J. Clean. Prod. 2016, 129, 548–555. [Google Scholar] [CrossRef]
- Nyári, J.; Magdeldin, M.; Larmi, M.; Järvinen, M.; Santasalo-Aarnio, A. Techno-Economic Barriers of an Industrial-Scale Methanol CCU-Plant. J. CO2 Util. 2020, 39, 101166. [Google Scholar] [CrossRef]
- Ren, T.; Patel, M.K.; Blok, K. Steam Cracking and Methane to Olefins: Energy Use, CO2 Emissions and Production Costs. Energy 2008, 33, 817–833. [Google Scholar] [CrossRef]
- Ren, T. Petrochemicals from Oil, Natural Gas, Coal and Biomass: Energy Use, Economics and Innovation. Ph.D. Thesis, Utrecht University, Utrecht, The Netherlands, March 2009. [Google Scholar]
- Liptow, C.; Tillman, A.-M.; Janssen, M. Life Cycle Assessment of Biomass-Based Ethylene Production in Sweden—Is Gasification or Fermentation the Environmentally Preferable Route? Int. J. Life Cycle Assess. 2015, 20, 632–644. [Google Scholar] [CrossRef]
- Brudermüller, M. Carbon Management at BASF–R&D Strategies to Reduce CO2. Available online: https://www.basf.com/global/documents/en/investor-relations/calendar-and-publications/presentations/2019/190110_BASF_RD_Webcast_Presentation_Dr_Martin_Brudermueller.pdf (accessed on 17 August 2020).
- Böhme, C. BASF Develops Process for Climate-Friendly Methanol. Available online: https://www.basf.com/global/en/media/news-releases/2019/05/p-19-218.html (accessed on 27 August 2020).
- Mass, H.-J.; Göke, V.; Machhammer, O.; Guzmann, M.; Schneider, C.; Hormuth, W.A.; Bode, A.; Klingler, D.; Kern, M.; Kolios, G. Process for production of synthesis gas. U.S. Patent 9266731B2, 23 February 2014. [Google Scholar]
- Schwab, E.; Milanov, A.; Schunk, S.; ROUSSIÈRE, T.; WASSERSCHAFF, G.; Strasser, A. Process for Producing Reforming Catalyst and Reforming of Methane. Patent WO2013068905A1, 16 June 2013. [Google Scholar]
- Yarrington, R.M.; Buchanan, W. Preparation of Normally Liquid Hydrocarbons and a Synthesis Gas to Make the Same, from a Normally Gaseous Hydrocarbon Feed 1991, U.S. Patent 5023276A, 11 June 1991. [Google Scholar]
- Bartenbach, B.; PRÖLSS, J.; Schmitz-Bäder, P.; Weichert, C.; Ehrhardt, K.R. Method for the production of olefins and synthesis gas. Patent WO2005103205A1, 3 November 2005. [Google Scholar]
- Schneider, C.; Bode, A.; Klingler, D.; Machhammer, O.; Brüggemann, P.; Kern, M.; Hormuth, W.A.; Guzmann, M.; König, R.; Bernnat, J.; et al. Process for Utilizing Blast Furnace Gases, Associated Gases and/or Biogases. Patent WO2014095661A1, 26 June 2014. [Google Scholar]
- Milanov, A.; Schwab, E.; Schunk, S.; Wasserschaff, G. High-pressure process for carbon dioxide reforming of hydrocarbons in the presence of iridium-containing active masses. Patent WO2014001423A1, 3 January 2014. [Google Scholar]
- Schunk, S.; Milanov, A.; Strasser, A.; Wasserschaff, G.; Roussiere, T. Hexaaluminate-comprising catalyst for reforming of hydrocarbons and reforming process. Patent WO2013118078A1, 15 September 2013. [Google Scholar]
- Schunk, S.; Oliveira, A.L. De Method for producing synthesis gas. Patent WO2008138899A1, 11 November 2008. [Google Scholar]
- Dinh, C.-T.; Burdyny, T.; Kibria, M.G.; Seifitokaldani, A.; Gabardo, C.M.; García de Arquer, F.P.; Kiani, A.; Edwards, J.P.; De Luna, P.; Bushuyev, O.S.; et al. CO2 Electroreduction to Ethylene via Hydroxide-Mediated Copper Catalysis at an Abrupt Interface. Science 2018, 360, 783–787. [Google Scholar] [CrossRef] [PubMed]
- Masel, R.I.; Sajjad, S.D.; Gao, Y.; Liu, Z.; Chen, Q. Ion-Conducting Membranes. Patent WO2017176599A1, 12 October 2017. [Google Scholar]
- Hago Energetics Enhancing Power Plants Profits While Reducing CO2 Emissions. Available online: http://hagoenergetics.com/wp-content/uploads/2019/07/PRESENTATION-July-2019-1-1.pdf (accessed on 19 August 2020).
- Sandia National Laboratory Sunshine to Petrol. Available online: https://energy.sandia.gov/programs/renewable-energy/solar-energy/sunshine-to-petrol/ (accessed on 20 August 2020).
- Schreier, M.; Curvat, L.; Giordano, F.; Steier, L.; Abate, A.; Zakeeruddin, S.M.; Luo, J.; Mayer, M.T.; Grätzel, M. Efficient Photosynthesis of Carbon Monoxide from CO2 Using Perovskite Photovoltaics. Nat. Commun. 2015, 6, 7326. [Google Scholar] [CrossRef]
- BASF SYNSPIRE. Available online: https://www.basf.com/global/en/who-we-are/innovation/our-innovations/energy_and_resources/synspire.html (accessed on 23 August 2020).
- Technologies That Do More with Less. Available online: https://www.linde-engineering.com/en/about-linde-engineering/success-stories/technologies-more-with-less.html (accessed on 3 September 2020).
- Carbon Capture and Utilization Project/Carbon Dioxide (CO2) Network Project. Available online: https://www.cslforum.org/cslf/Projects/SABIC (accessed on 23 March 2021).
- Synthetic Fuels (Methanol to Gasoline). Available online: https://www.exxonmobilchemical.com/en/catalysts-and-technology-licensing/synthetic-fuels (accessed on 23 August 2020).
- Schwartz, A. Dow Chemical, Algenol Biofuels Using Algae to Turn CO2 into Ethanol. Available online: https://www.fastcompany.com/1301811/dow-chemical-algenol-biofuels-using-algae-turn-co2-ethanol (accessed on 24 August 2020).
- Methanol from Natural Gas and CO2. Available online: https://www.eni.com/en-IT/operations/methanol-gas-co2.html (accessed on 4 March 2020).
- Basini, L.E.; Guarinoni, A. Short Contact Time Catalytic Partial Oxidation (SCT-CPO) for Synthesis Gas Processes and Olefins Production. Ind. Eng. Chem. Res. 2013, 52, 17023–17037. [Google Scholar] [CrossRef]
- DTP Process. Available online: https://www.jgc.com/en/business/tech-innovation/gas-petro-chemi/dtp.html (accessed on 17 August 2020).
- INOVYN to Play Vital Role in Ambitious ‘Power to Methanol’ Project to Produce Sustainable Methanol and Reduce CO2 in Antwerp. Available online: https://www.ineos.com/news/shared-news/inovyn-to-play-vital-role-in-ambitious-power-to-methanol-project-to-produce-sustainable-methanol-and-reduce-co2-in-antwerp/ (accessed on 23 August 2020).
- Converge. Available online: https://www.saudiaramco.com/en/creating-value/products/converge (accessed on 10 August 2020).
- Emissions Reduction. Available online: https://www.repsol.com/en/energy-and-innovation/technology-lab/emissions-reduction/index.cshtml (accessed on 19 August 2020).
- ReportLinker COVID-19 Impact on Packaging Market by Material Type, Application And Region—Global Forecast to 2021. Available online: https://www.prnewswire.com/news-releases/covid-19-impact-on-packaging-market-by-material-type-application-and-region---global-forecast-to-2021-301056485.html (accessed on 1 March 2021).
- Kaiser, S.; Bringezu, S. Use of Carbon Dioxide as Raw Material to Close the Carbon Cycle for the German Chemical and Polymer Industries. J. Clean. Prod. 2020, 271, 122775. [Google Scholar] [CrossRef] [PubMed]
- Pires, J.C.M.; Martins, F.G.; Alvim-Ferraz, M.C.M.; Simões, M. Recent Developments on Carbon Capture and Storage: An Overview. Chem. Eng. Res. Des. 2011, 89, 1446–1460. [Google Scholar] [CrossRef]
- Brunetti, A.; Drioli, E.; Lee, Y.M.; Barbieri, G. Engineering Evaluation of CO2 Separation by Membrane Gas Separation Systems. J. Memb. Sci. 2014, 454, 305–315. [Google Scholar] [CrossRef]
- Zhang, C.; Show, P.-L.; Ho, S.-H. Progress and Perspective on Algal Plastics—A Critical Review. Bioresour. Technol. 2019, 289, 121700. [Google Scholar] [CrossRef] [PubMed]
- Chisti, Y. Biodiesel from Microalgae Beats Bioethanol. Trends Biotechnol. 2008, 26, 126–131. [Google Scholar] [CrossRef]
- Foley, P.M.; Beach, E.S.; Zimmerman, J.B. Algae as a Source of Renewable Chemicals: Opportunities and Challenges. Green Chem. 2011, 13, 1399. [Google Scholar] [CrossRef]
- Zhang, X.; Fevre, M.; Jones, G.O.; Waymouth, R.M. Catalysis as an Enabling Science for Sustainable Polymers. Chem. Rev. 2018, 118, 839–885. [Google Scholar] [CrossRef]
- Chemical Recycling of Plastic Waste. Available online: https://www.basf.com/global/en/who-we-are/sustainability/we-drive-sustainable-solutions/circular-economy/mass-balance-approach/chemcycling.html (accessed on 1 February 2021).
- Our Carbon Management. Available online: https://www.basf.com/global/en/who-we-are/sustainability/we-produce-safely-and-efficiently/energy-and-climate-protection/carbon-management.html (accessed on 23 August 2020).
- Innovations for Climate-Friendly Chemical Production. Available online: https://www.basf.com/global/en/who-we-are/sustainability/we-produce-safely-and-efficiently/energy-and-climate-protection/carbon-management/innovations-for-a-climate-friendly-chemical-production.html (accessed on 23 August 2020).
- Macola, I.G. Q&A: How Plastics Innovation Can Spur on Renewables with SABIC. Available online: https://power.nridigital.com/future_power_technology_jul20/sabic_renewable_energy (accessed on 23 March 2021).
- John, N. Mukesh Ambani to Steer Reliance Industries to a Green Future, Cut in CO2 Emission Top Priority. Available online: https://www.businesstoday.in/current/corporate/mukesh-ambani-plans-to-cut-down-carbon-dioxide-emission-at-ril/story/410055.html (accessed on 9 August 2020).
- Ineos and Plastic Energy to Collaborate on New Advanced Plastic Recycling Facility. Available online: https://www.ineos.com/news/shared-news/ineos-and-plastic-energy-to-collaborate-on-new-advanced-plastic-recycling-facility/ (accessed on 19 August 2020).
- Leinemann, R. INEOS Styrolution: Breakthrough in Chemical Recycling of Polystyrene. Available online: https://www.ineos-styrolution.com/news/ineos-styrolution-breakthrough-in-chemical-recycling-of-polystyrene (accessed on 24 August 2020).
- Sekisui Chemical and Sumitomo Chemical to Cooperate on Circular Economy Initiative Manufacturing Polyolefin Using Waste as Raw Material. Available online: https://www.sekisuichemical.com/whatsnew/2020/1347675_36556.html (accessed on 3 September 2020).
- Waste Management. Available online: https://corporate.exxonmobil.com/Community-engagement/Sustainability-Report/Environment/Waste-management#Helpingcustomersreducetheirplasticwaste (accessed on 14 August 2020).
- Carbon Dioxide Biofixation. Available online: https://www.eni.com/en-IT/operations/carbon-dioxide-biofixation.html (accessed on 23 March 2021).
- Eni Launches a New Business Structure to Be a Leader in the Energy Transition. Available online: https://www.eni.com/en-IT/media/press-release/2020/06/eni-launches-a-new-business-structure.html (accessed on 20 March 2021).
- Dow Signs Four Renewable Power Agreements to Achieve 2025 Goal and Lead Petrochemical Industry. Available online: https://www.dow.com/en-us/news/dow-signs-four-renewable-power-agreements-to-achieve-2025-goal.html (accessed on 20 August 2020).
- Fuenix. Available online: https://fuenix.com/ (accessed on 23 March 2021).
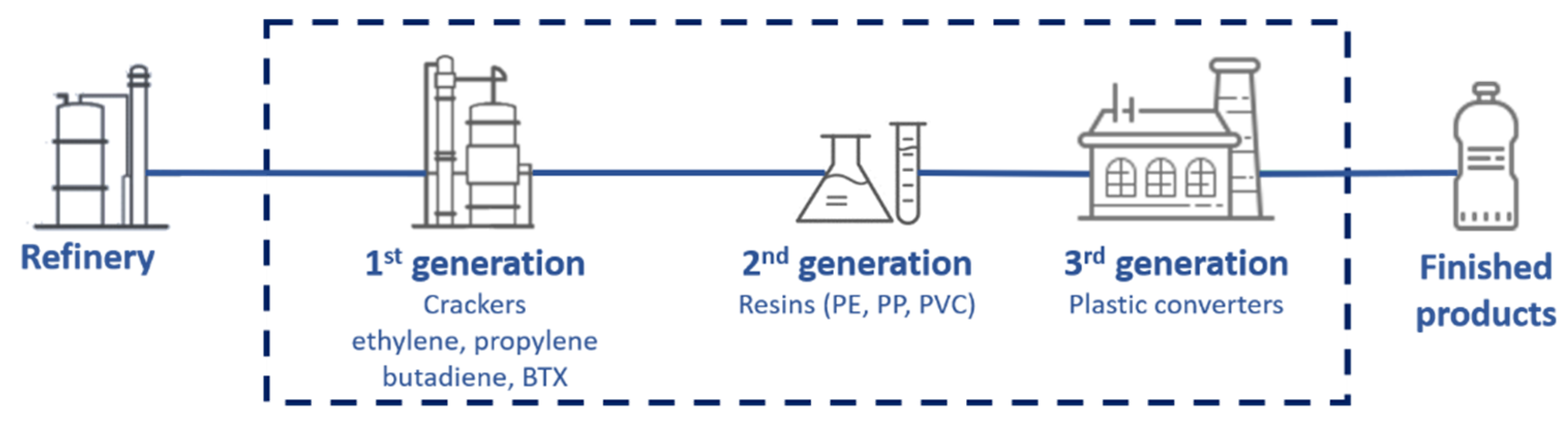
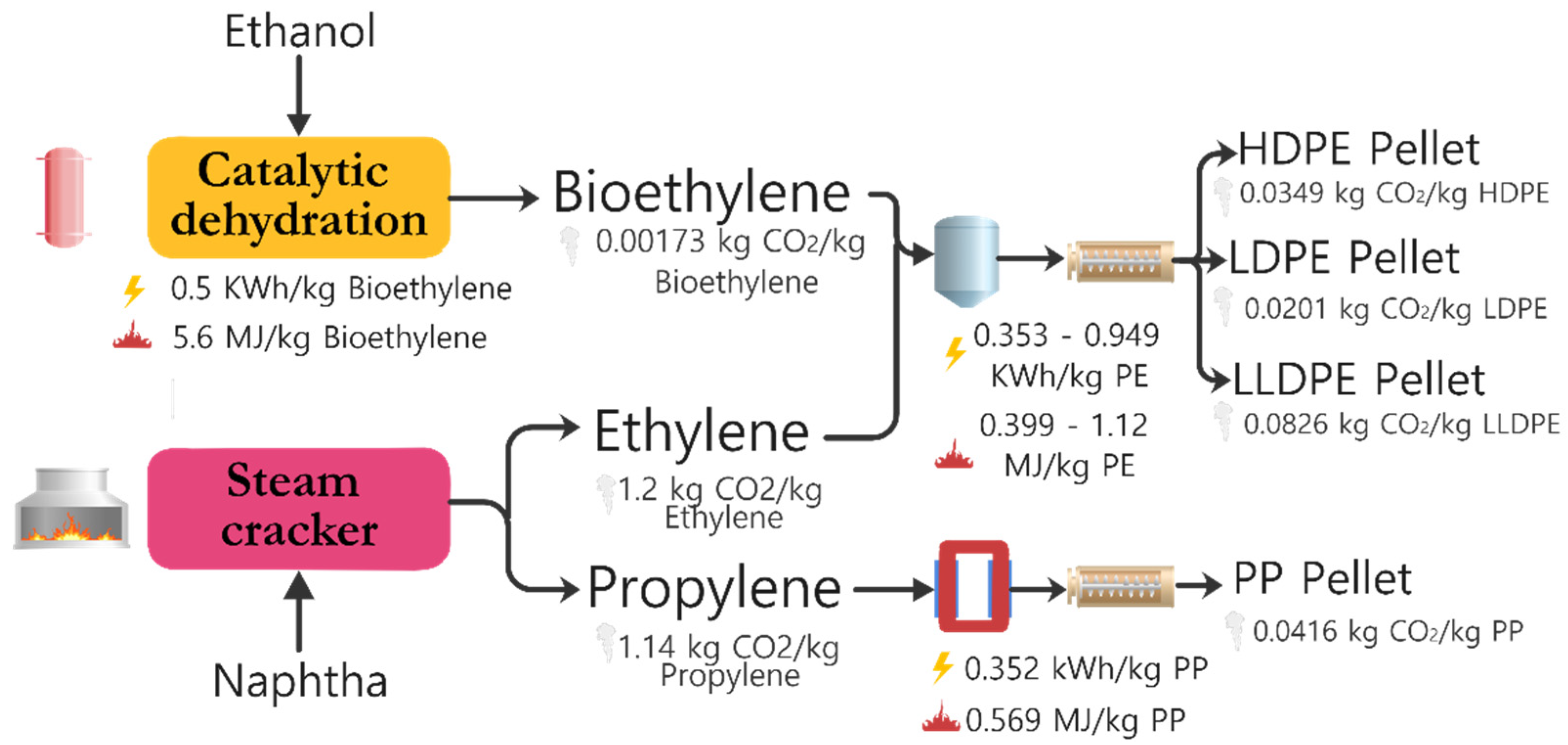
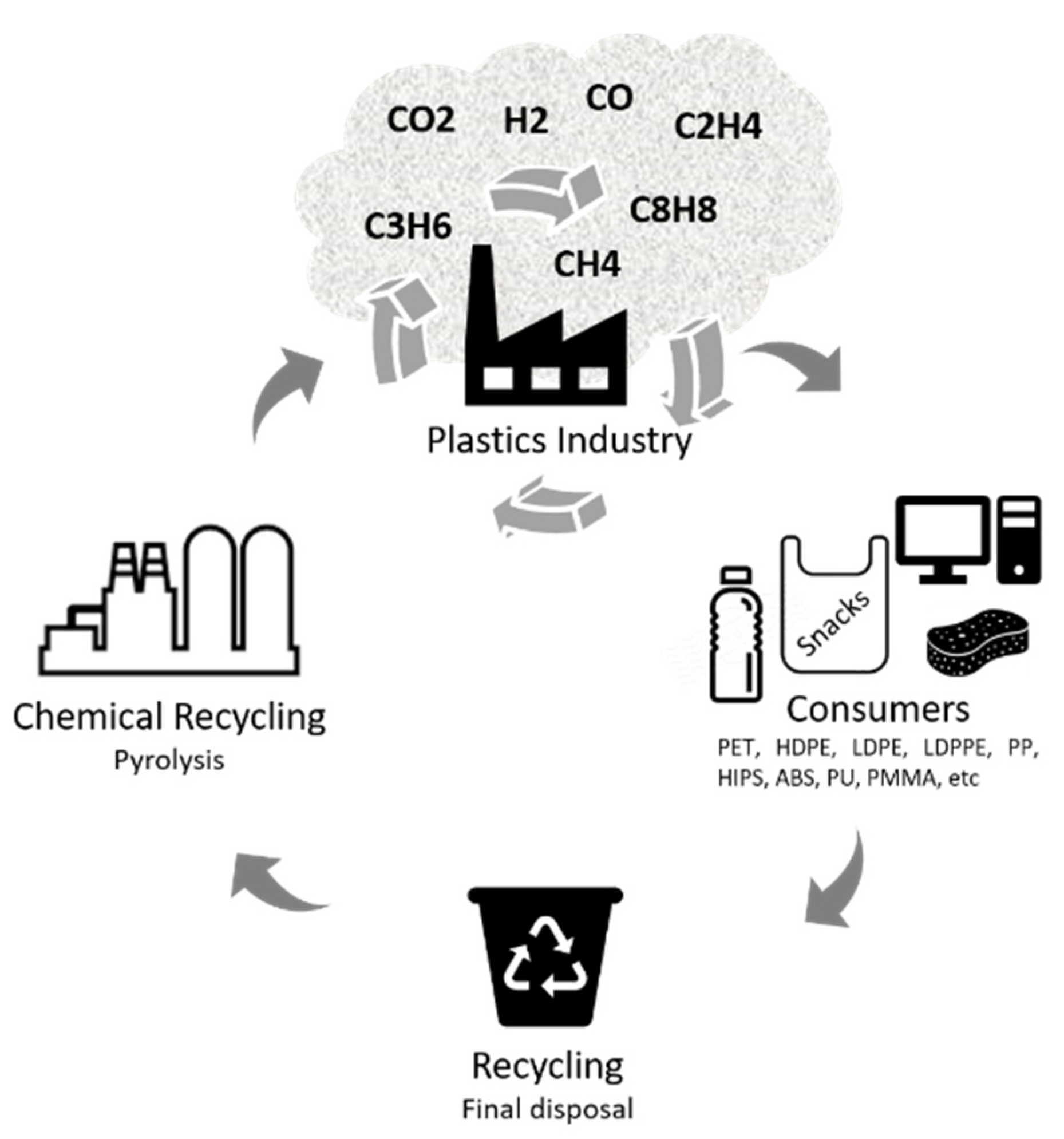
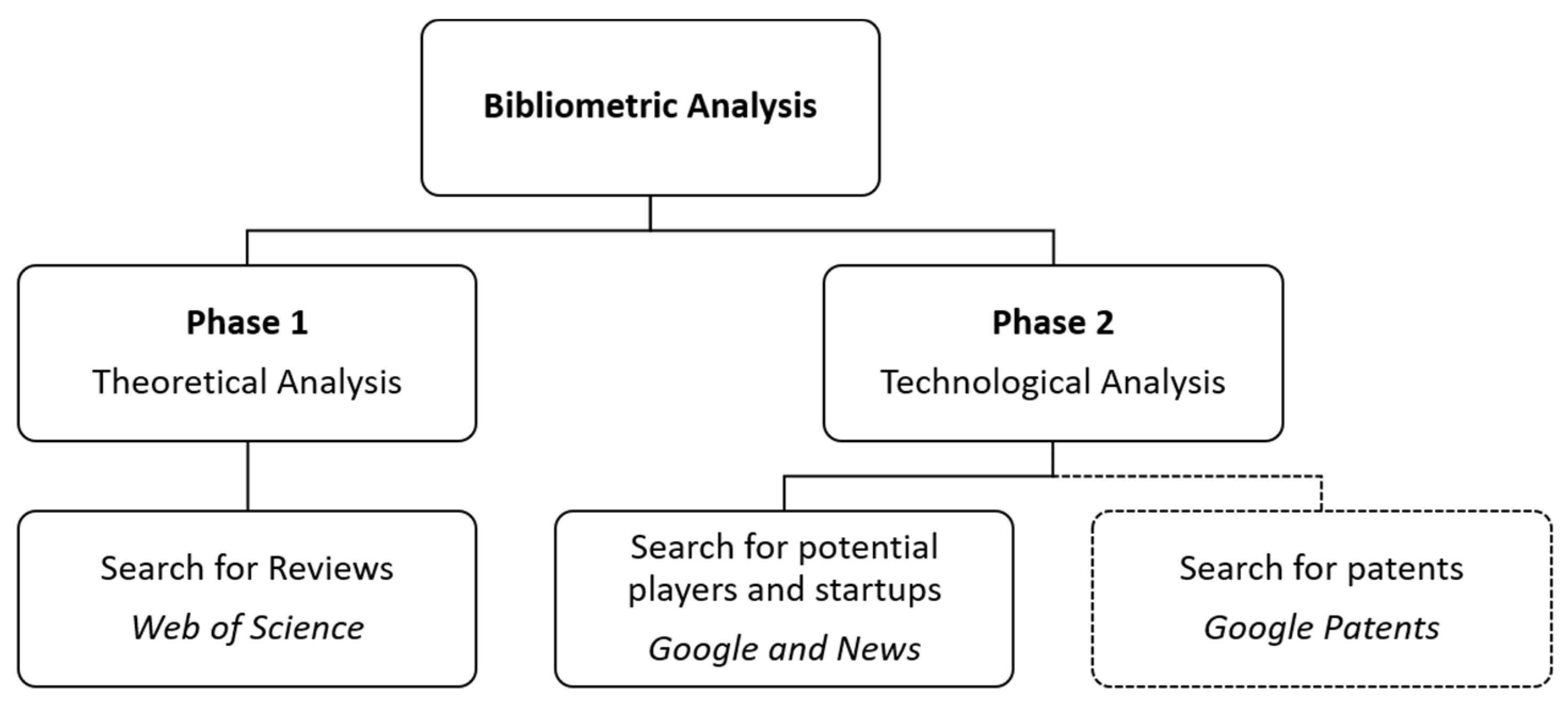
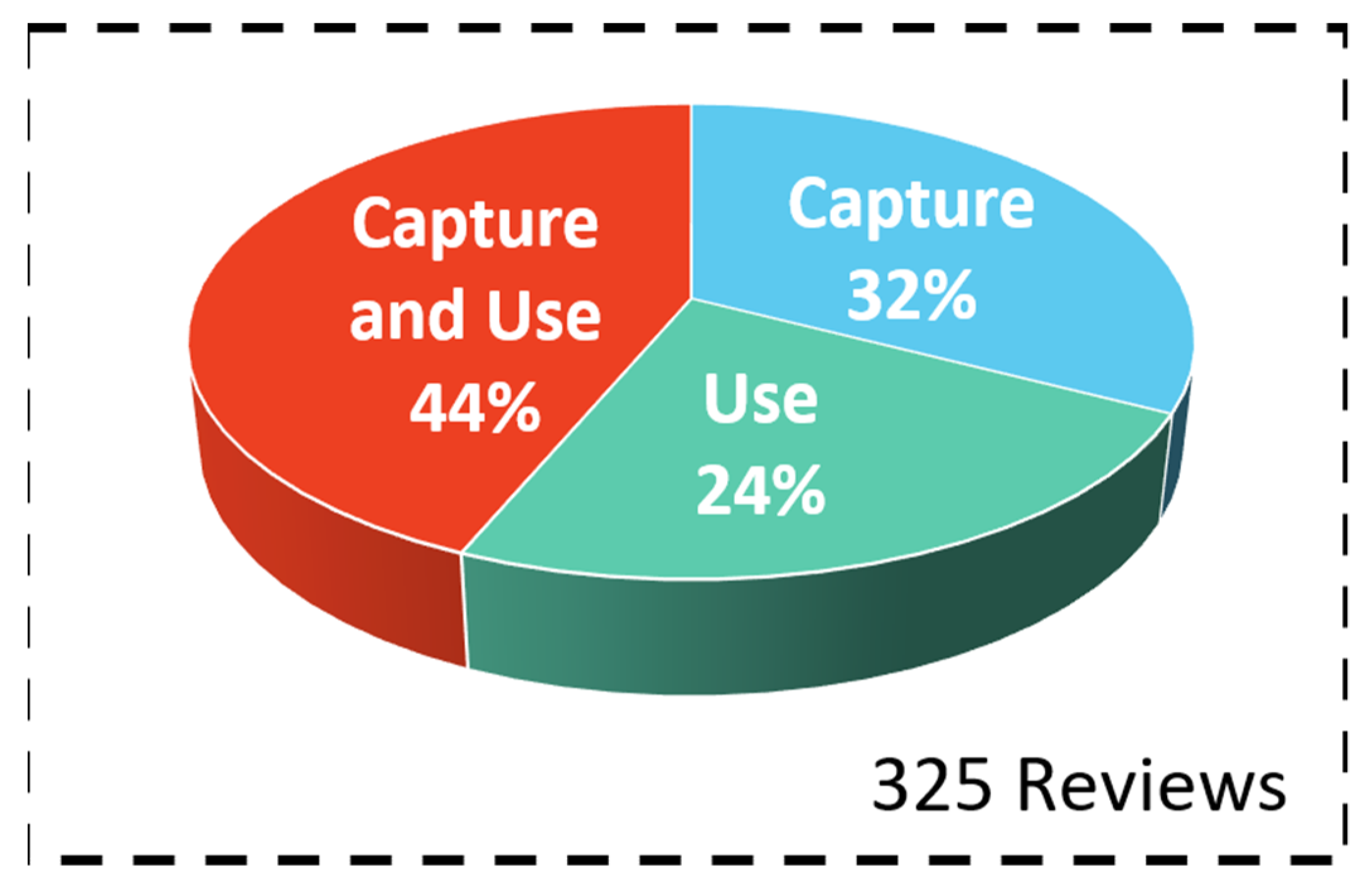
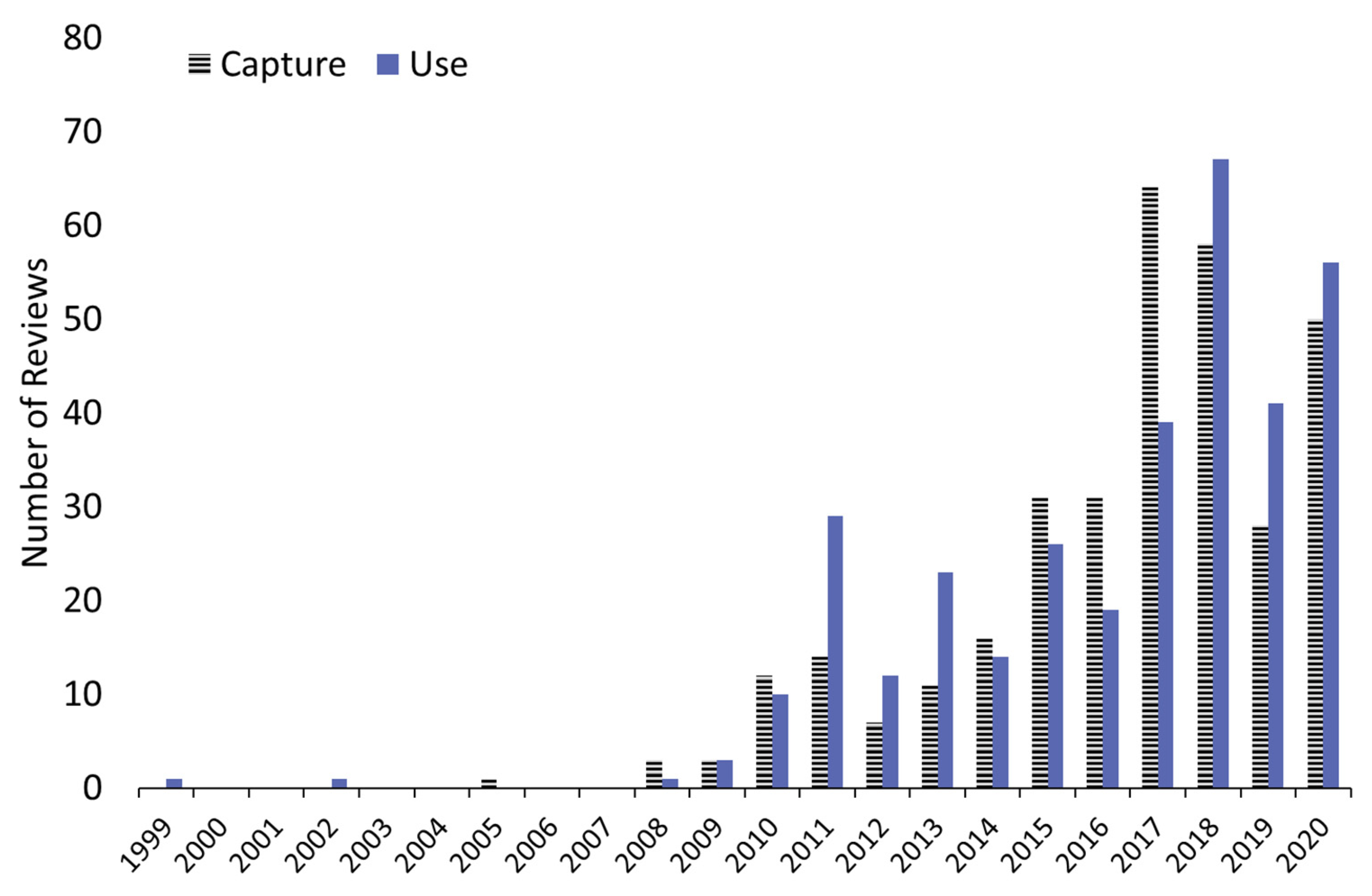
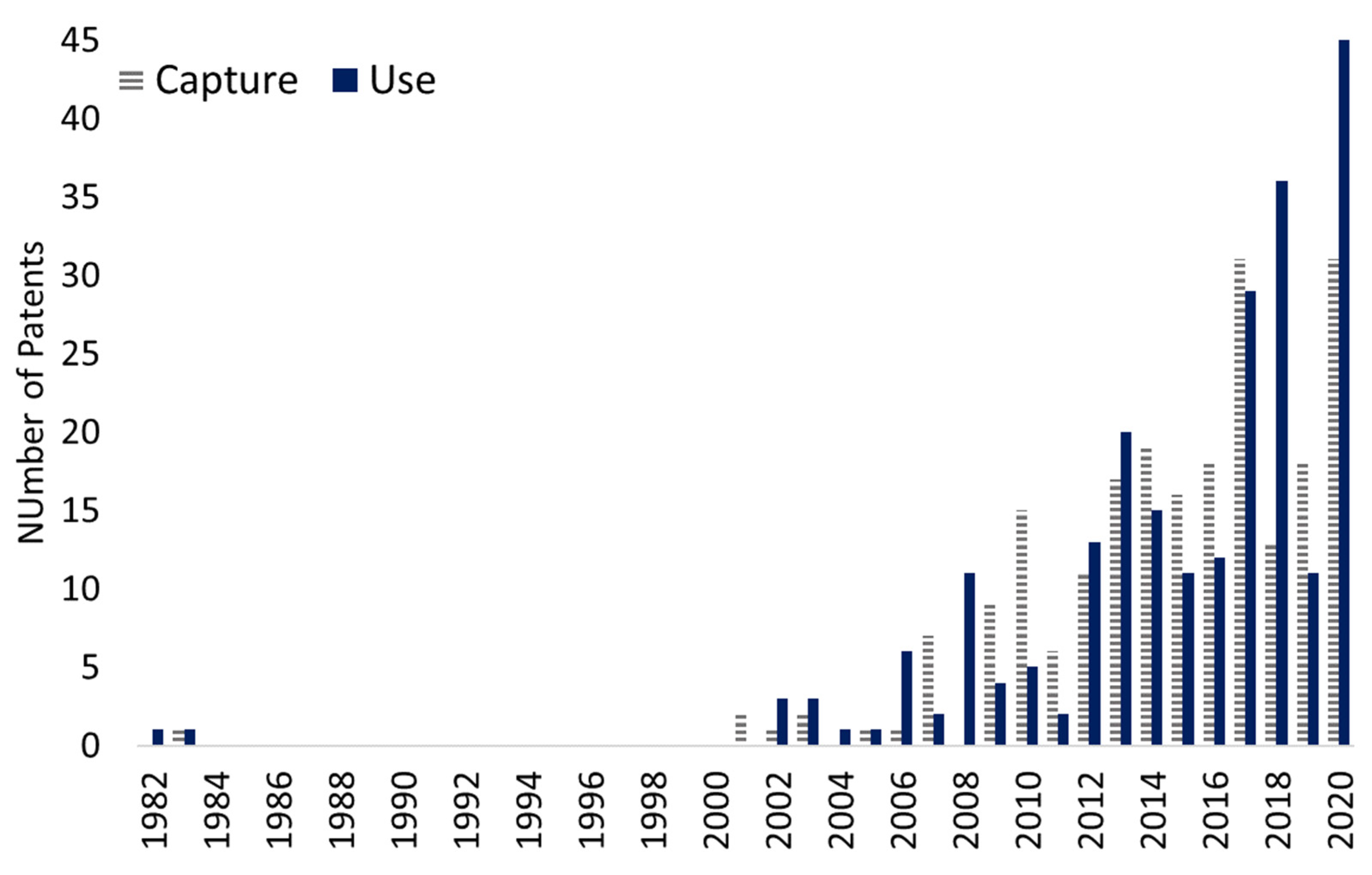
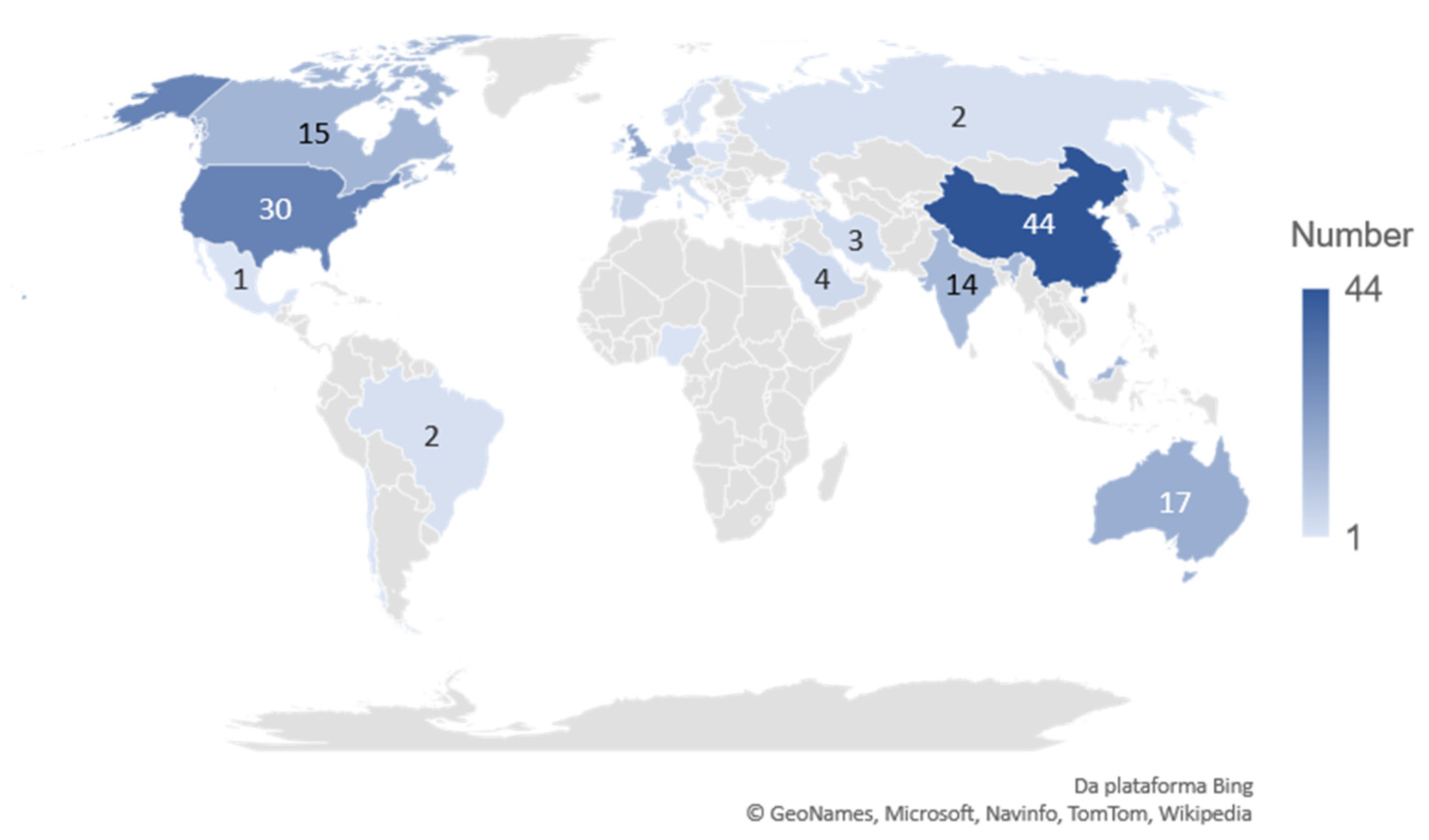
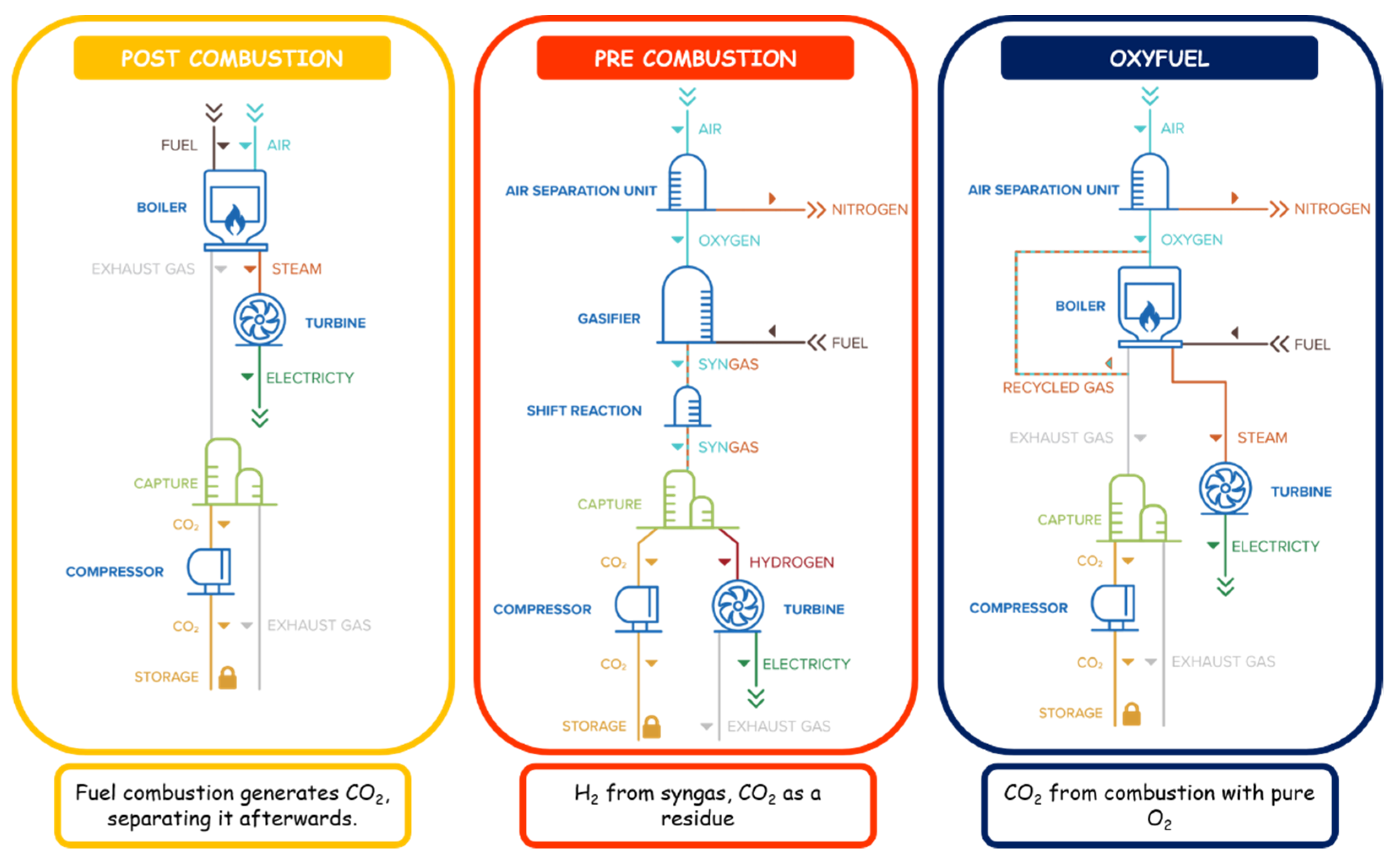
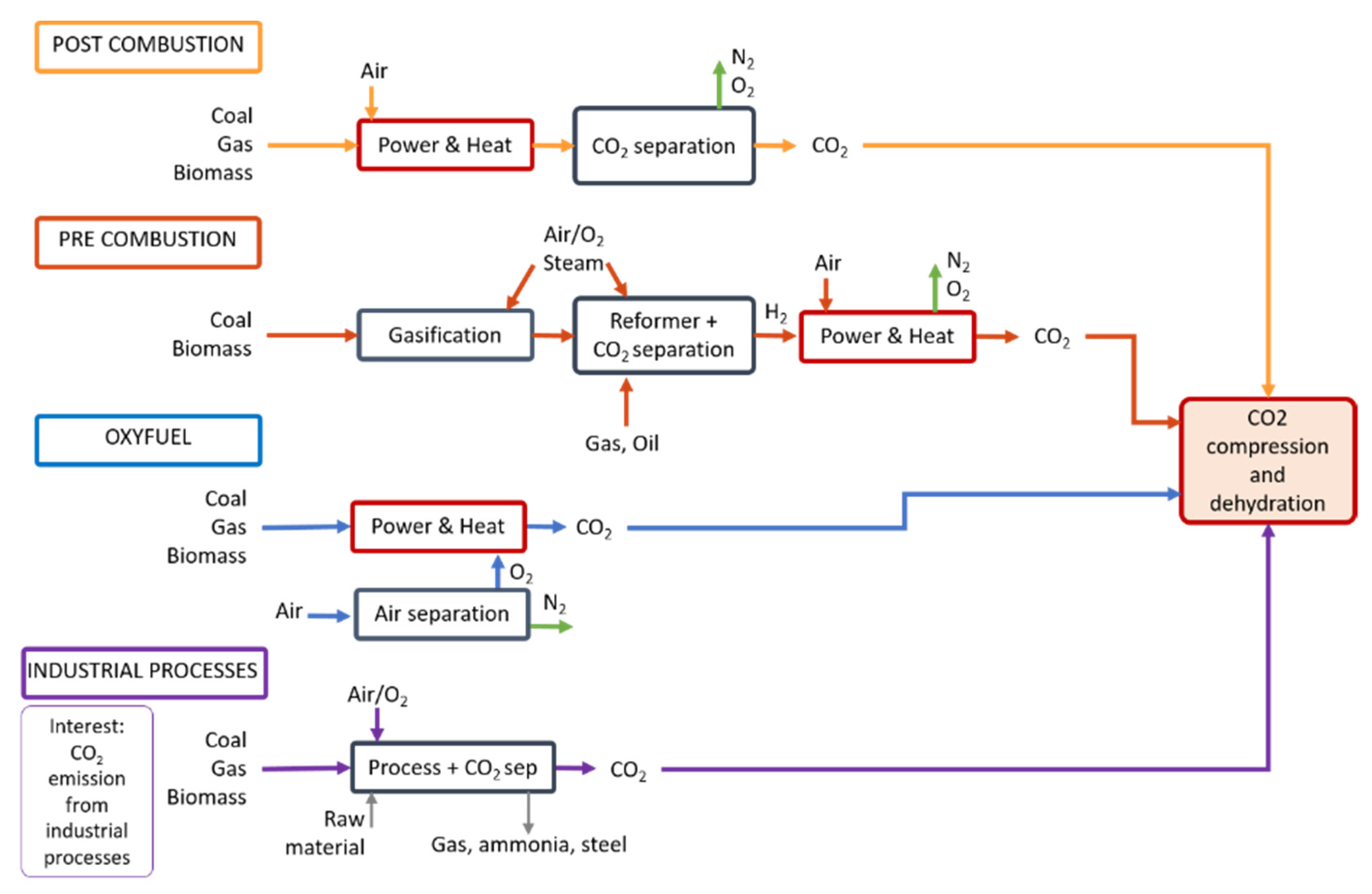
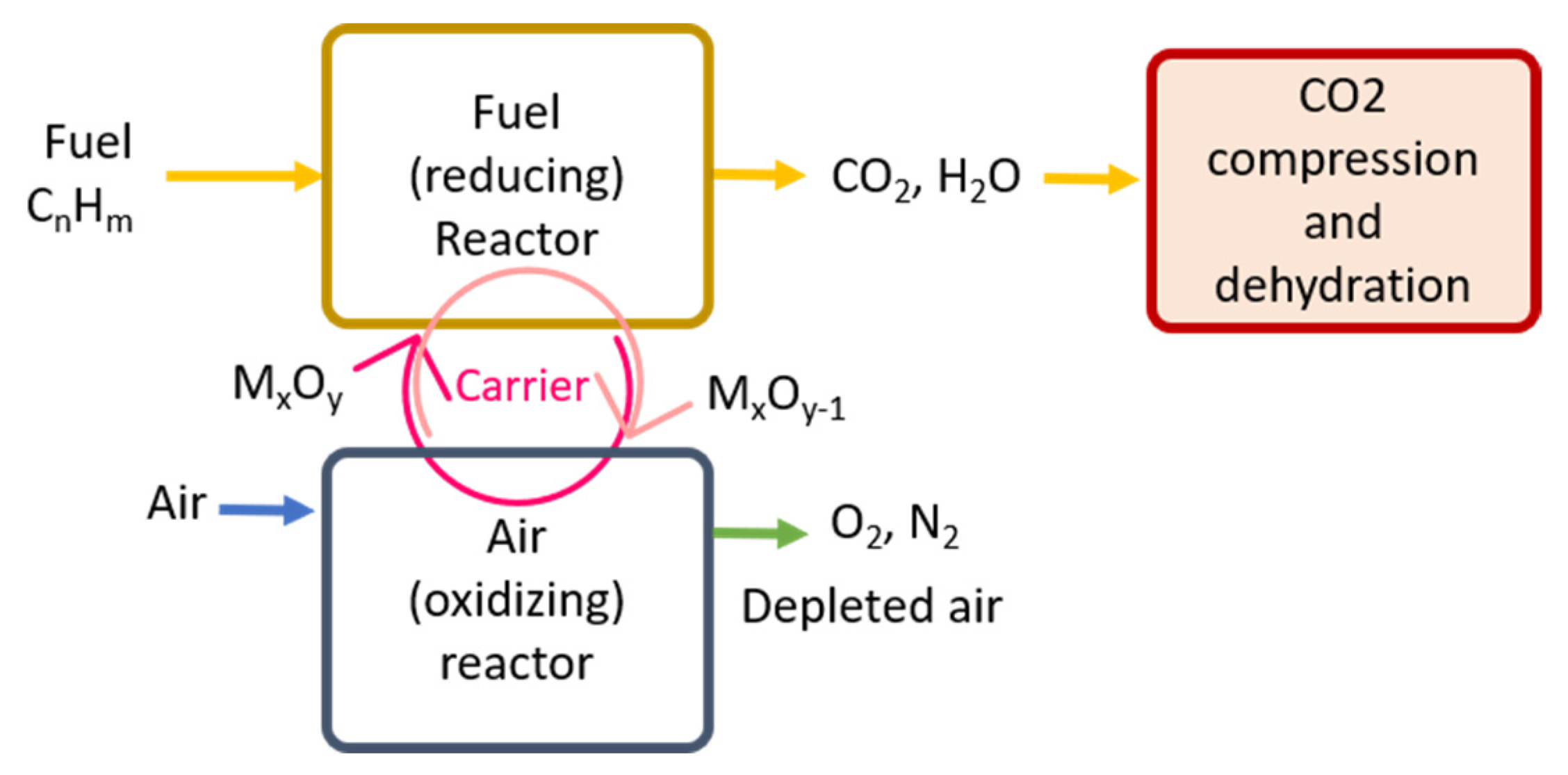
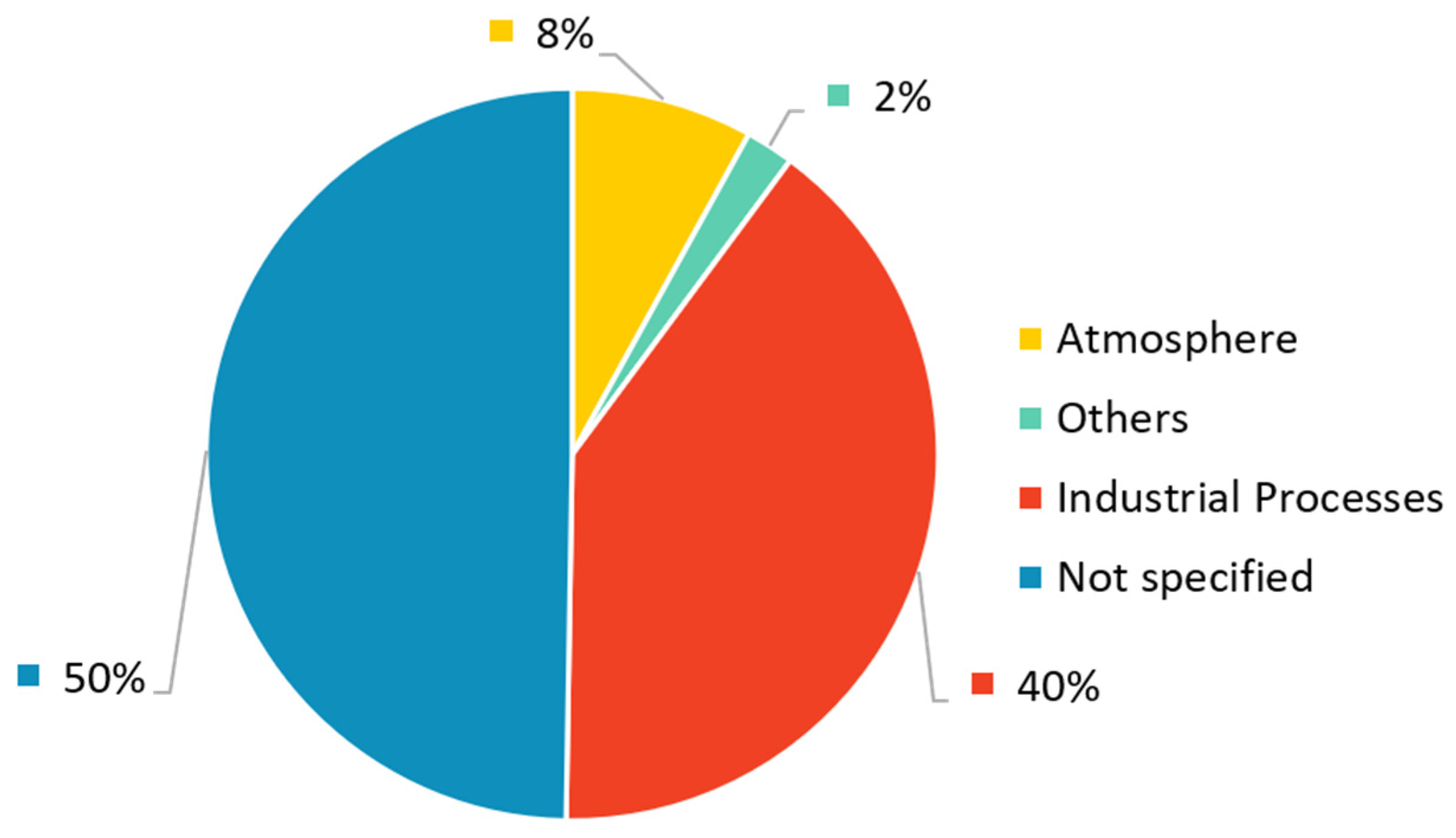
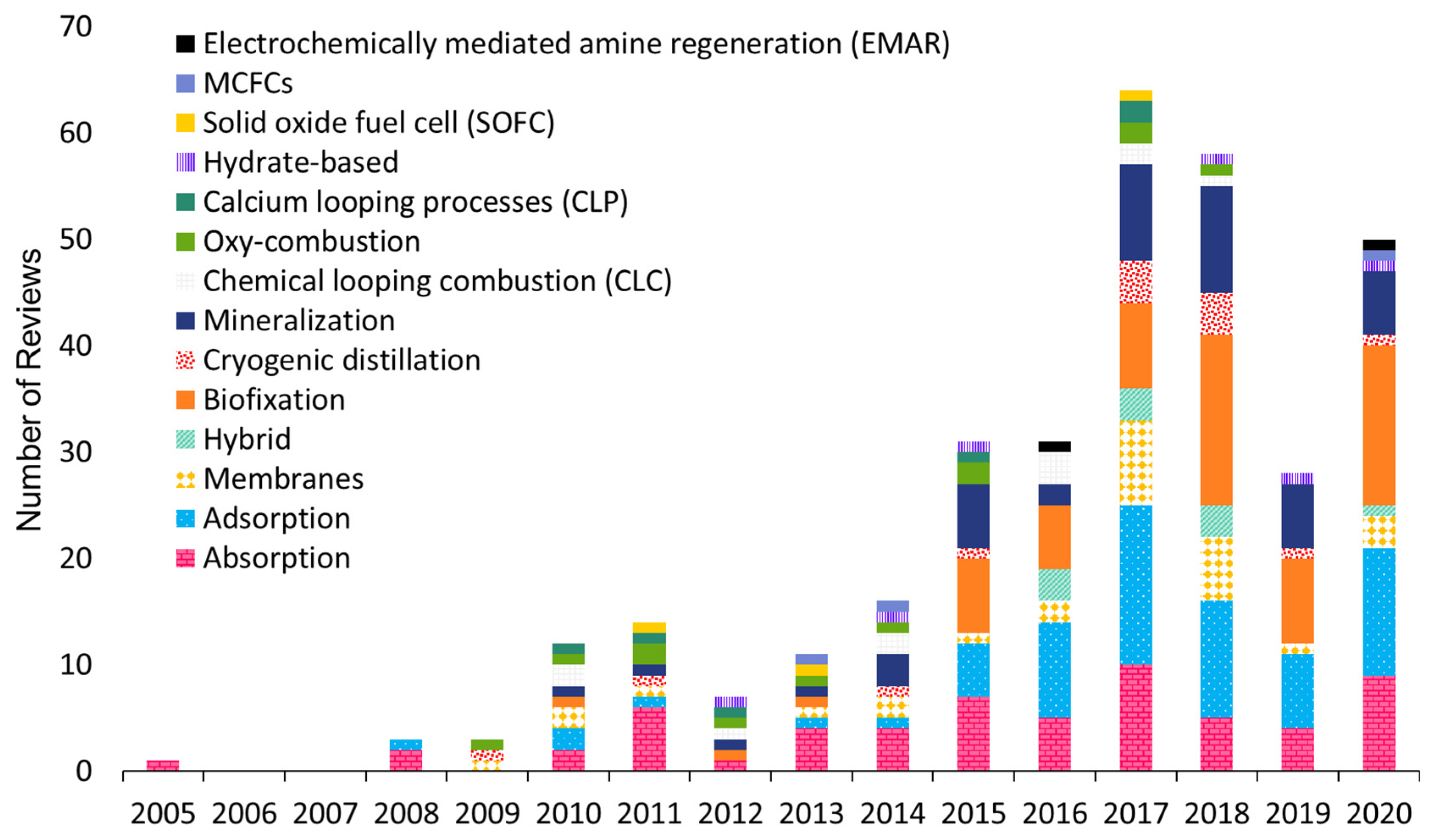
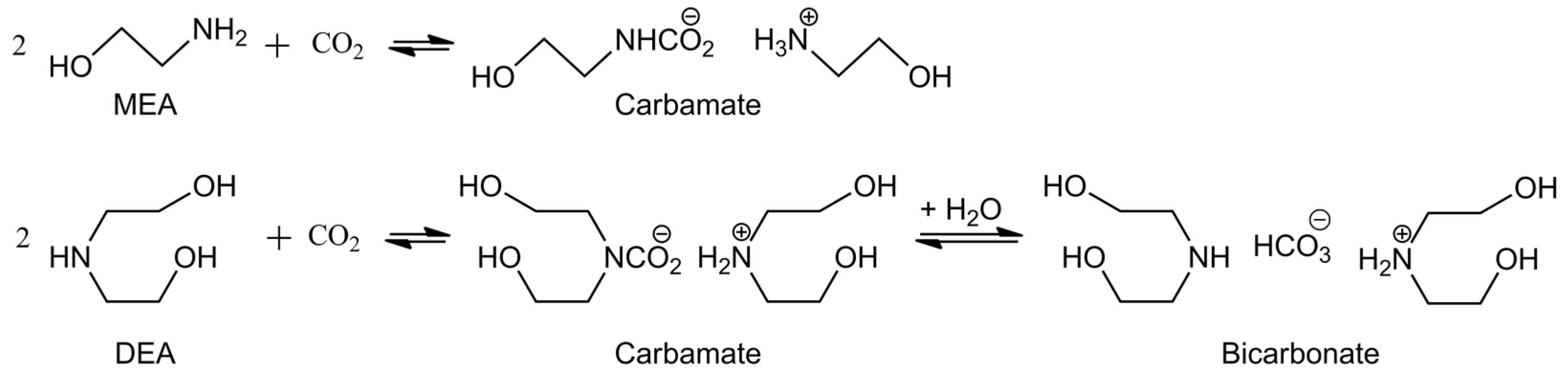
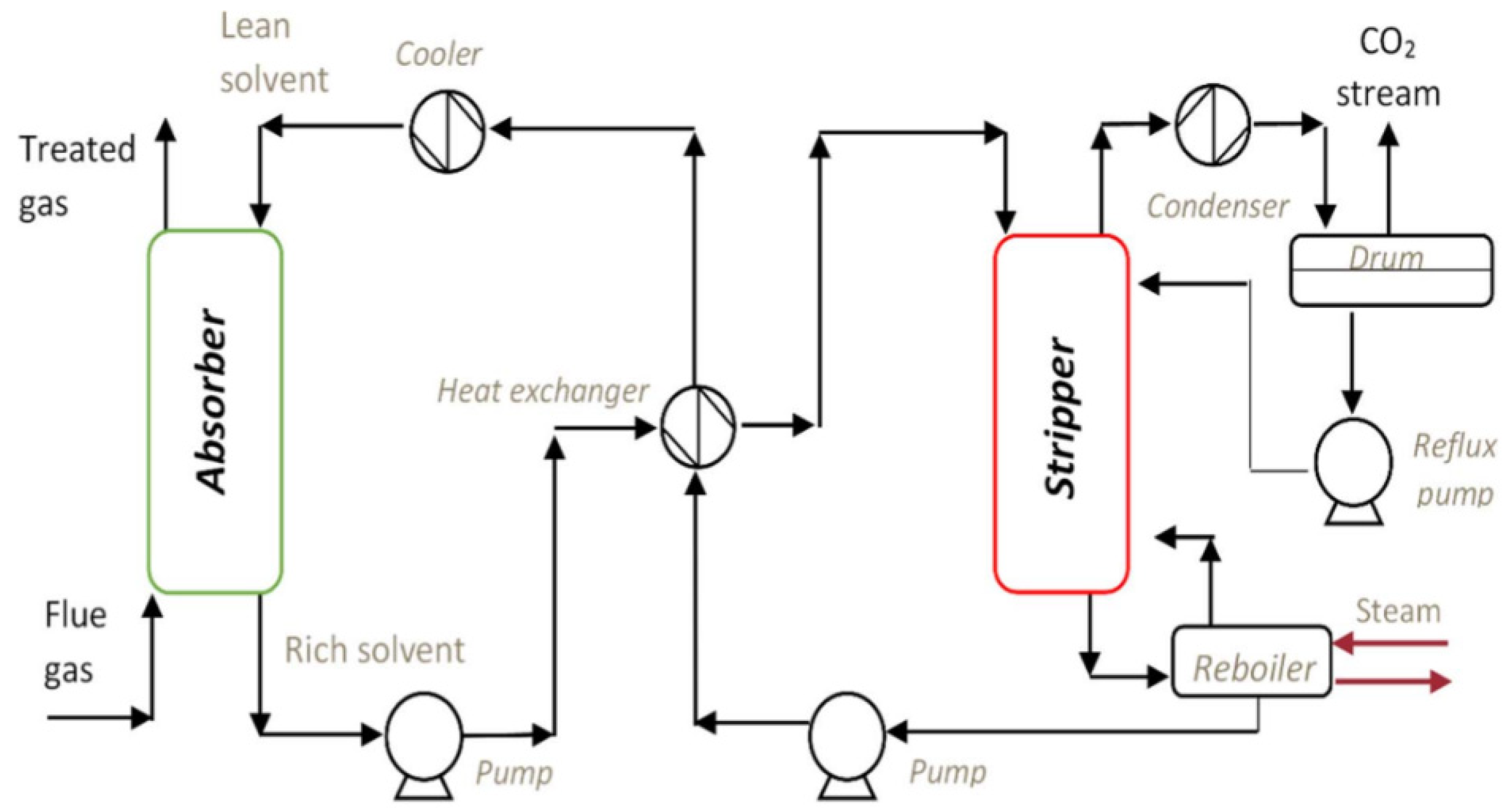

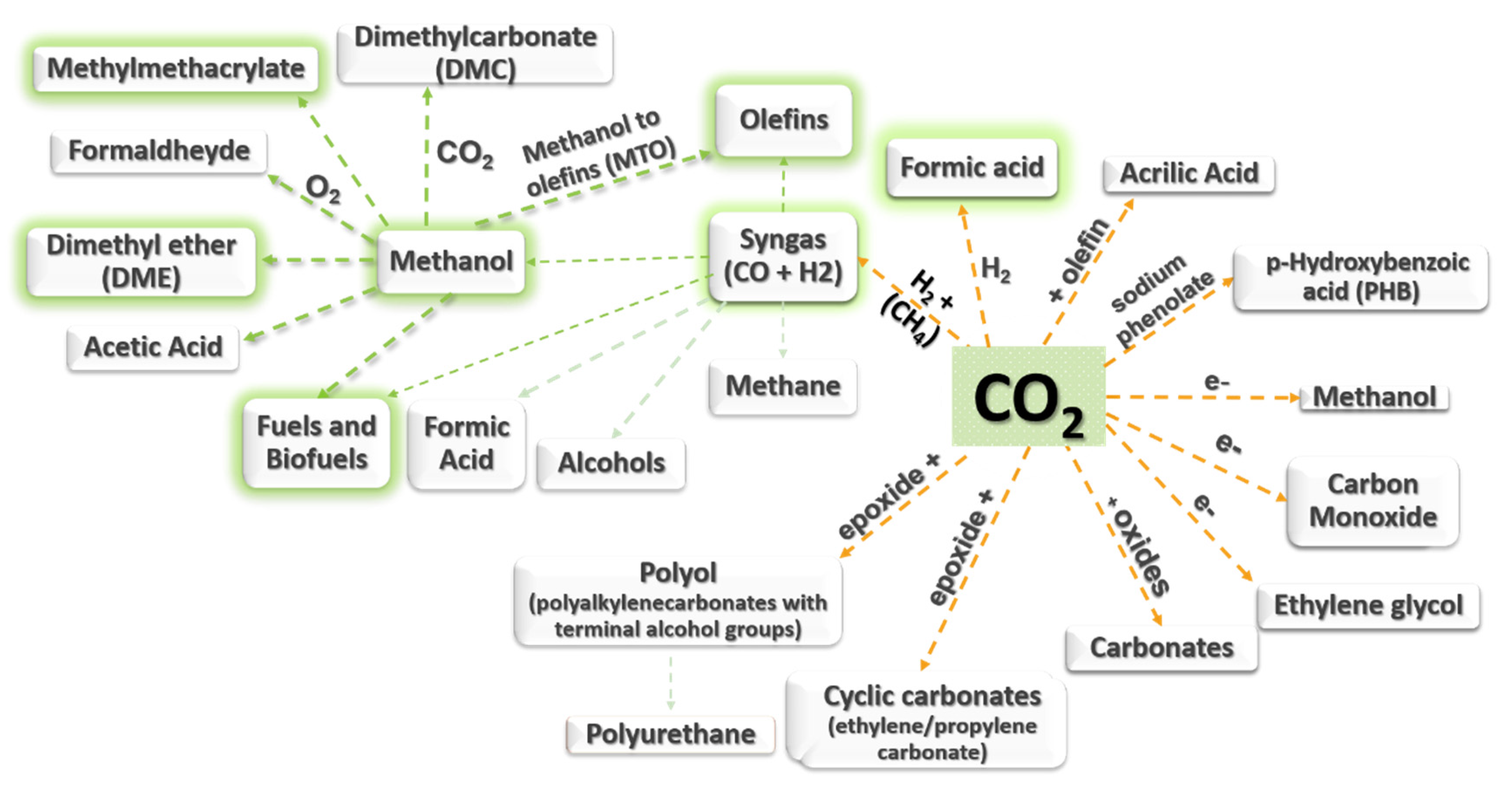
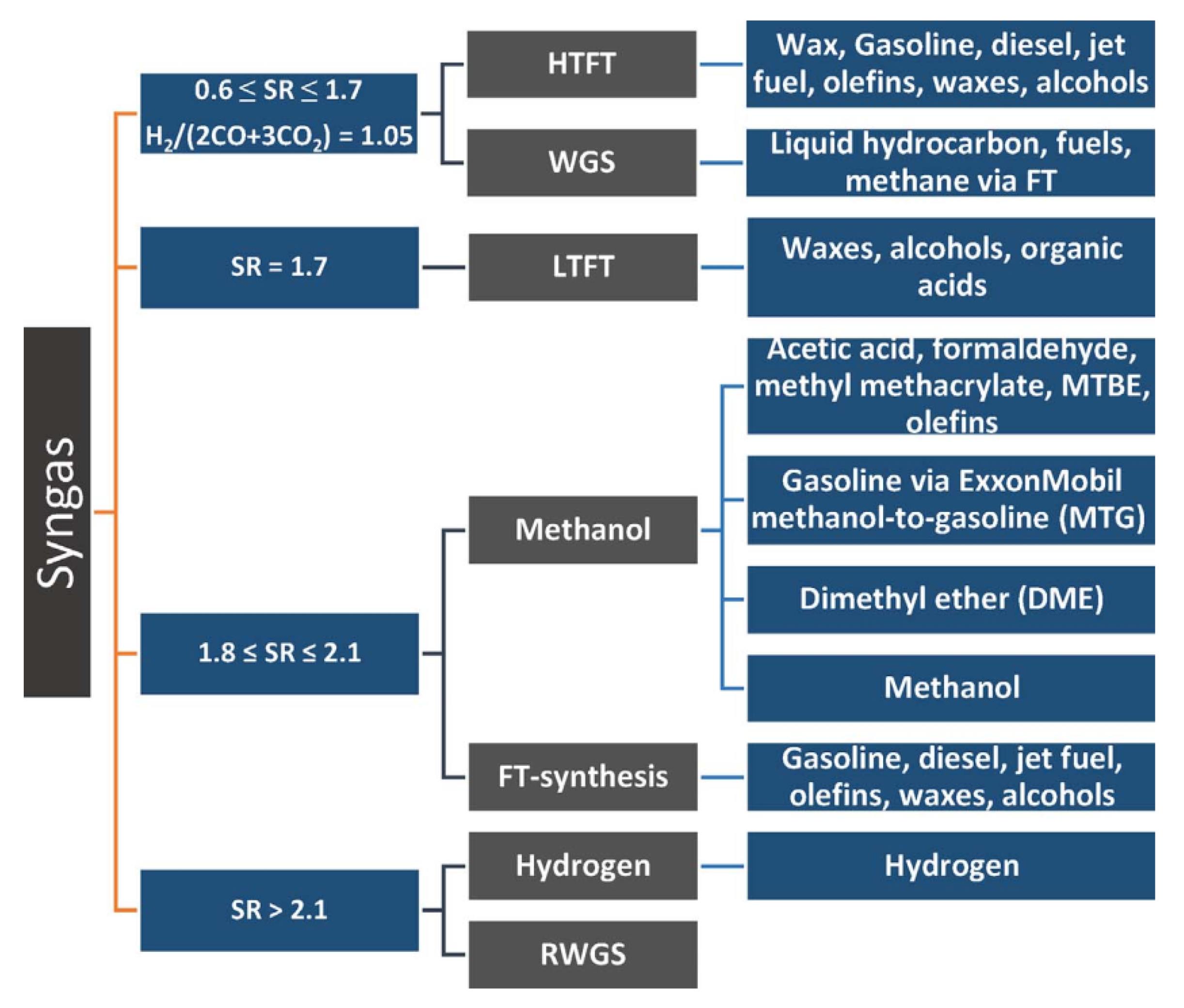
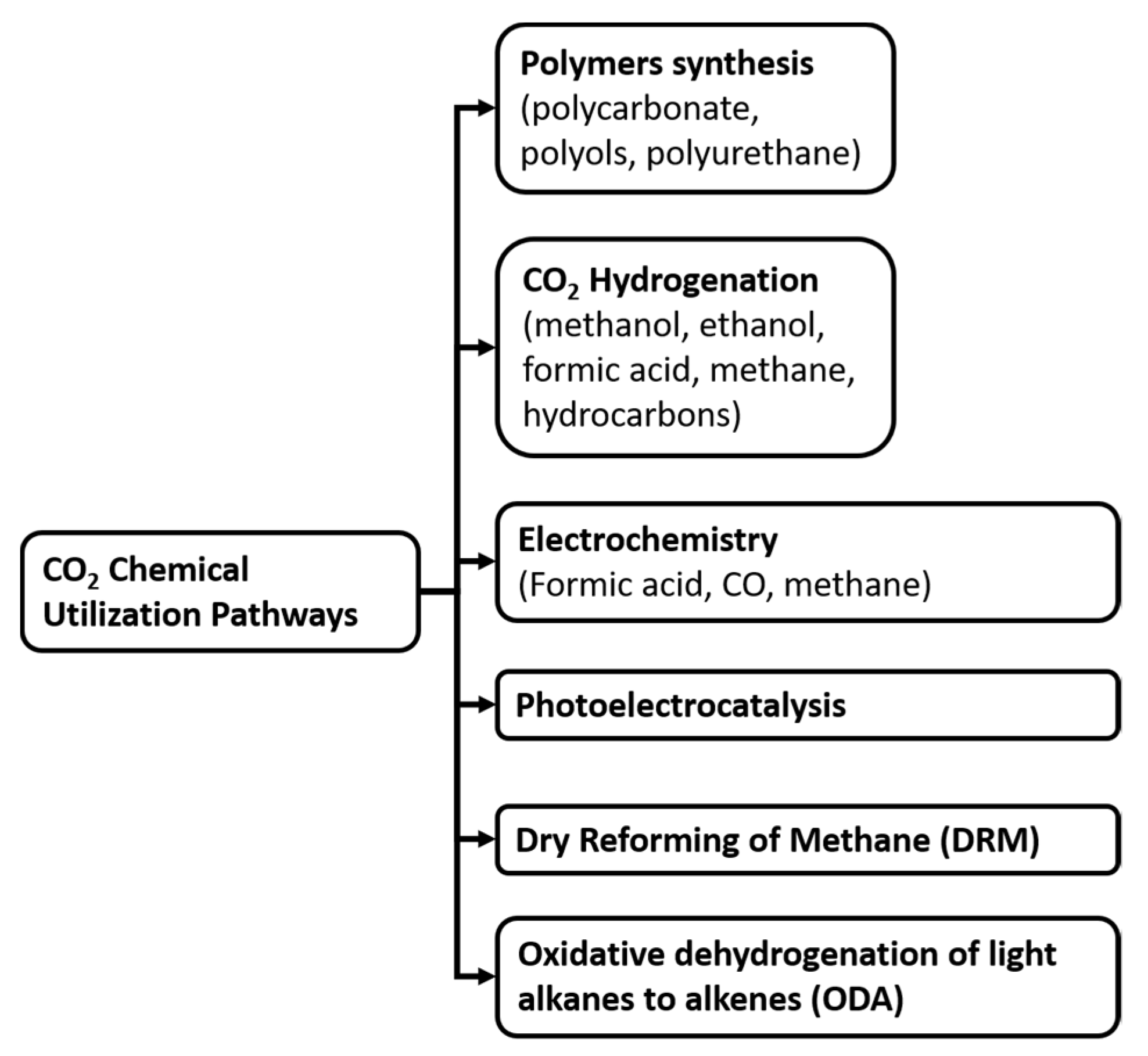
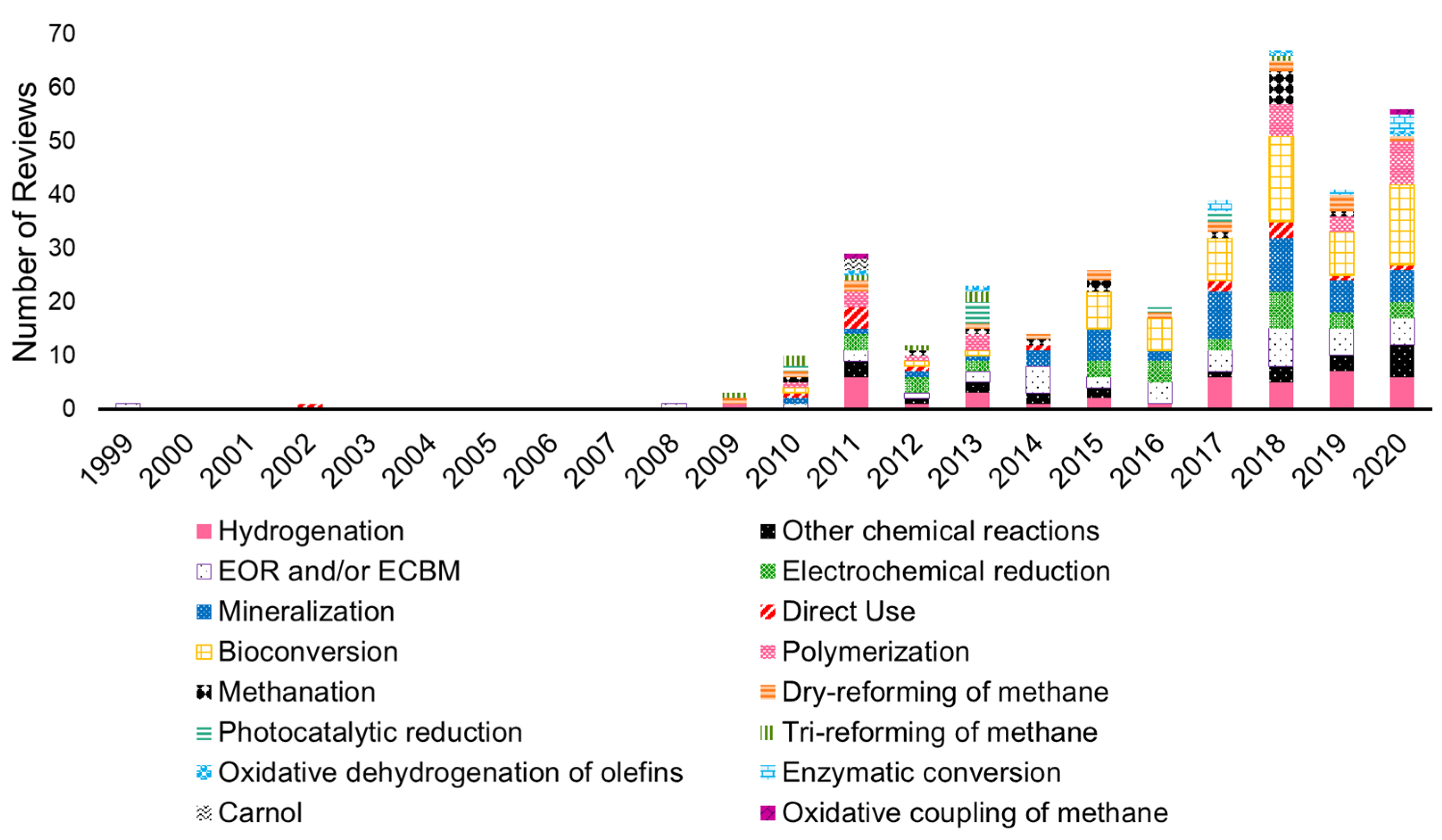
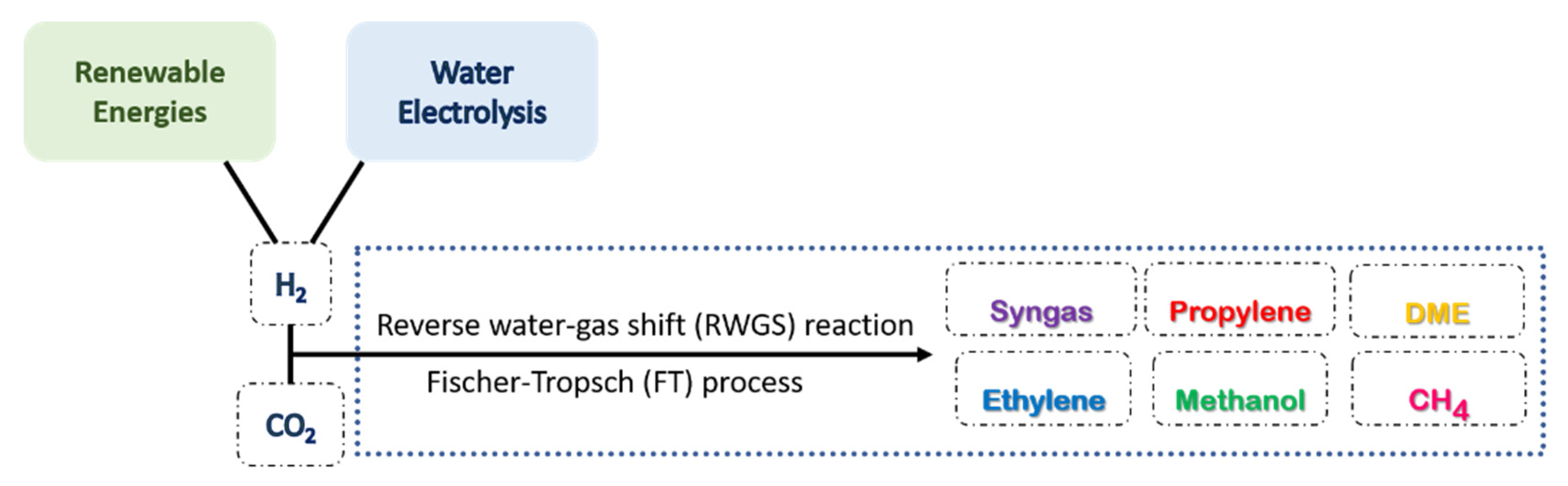
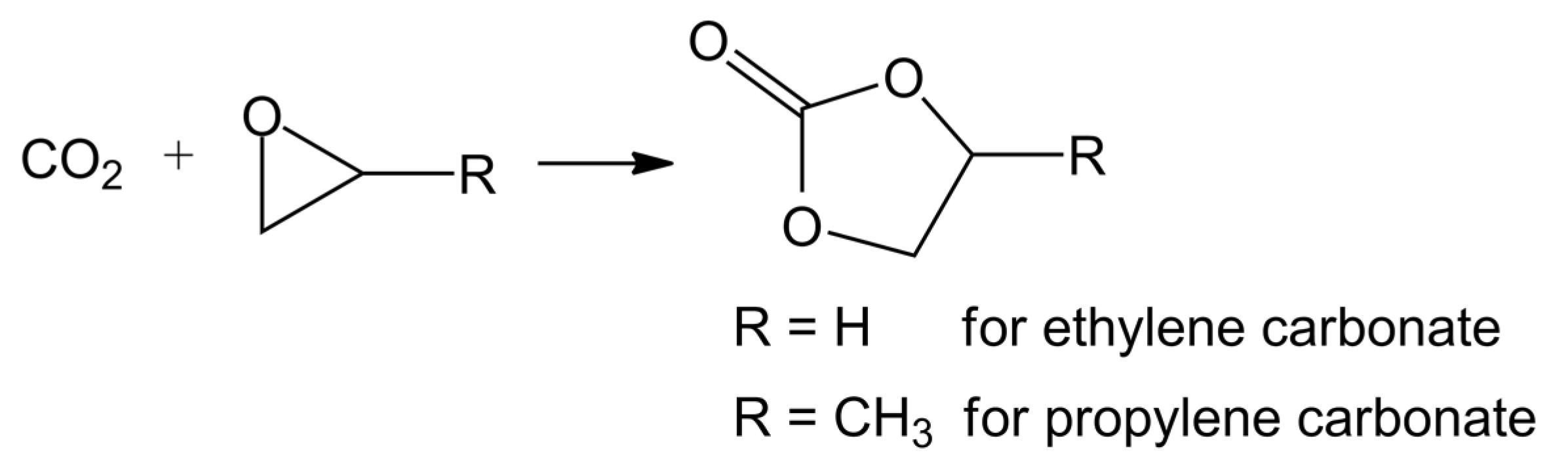
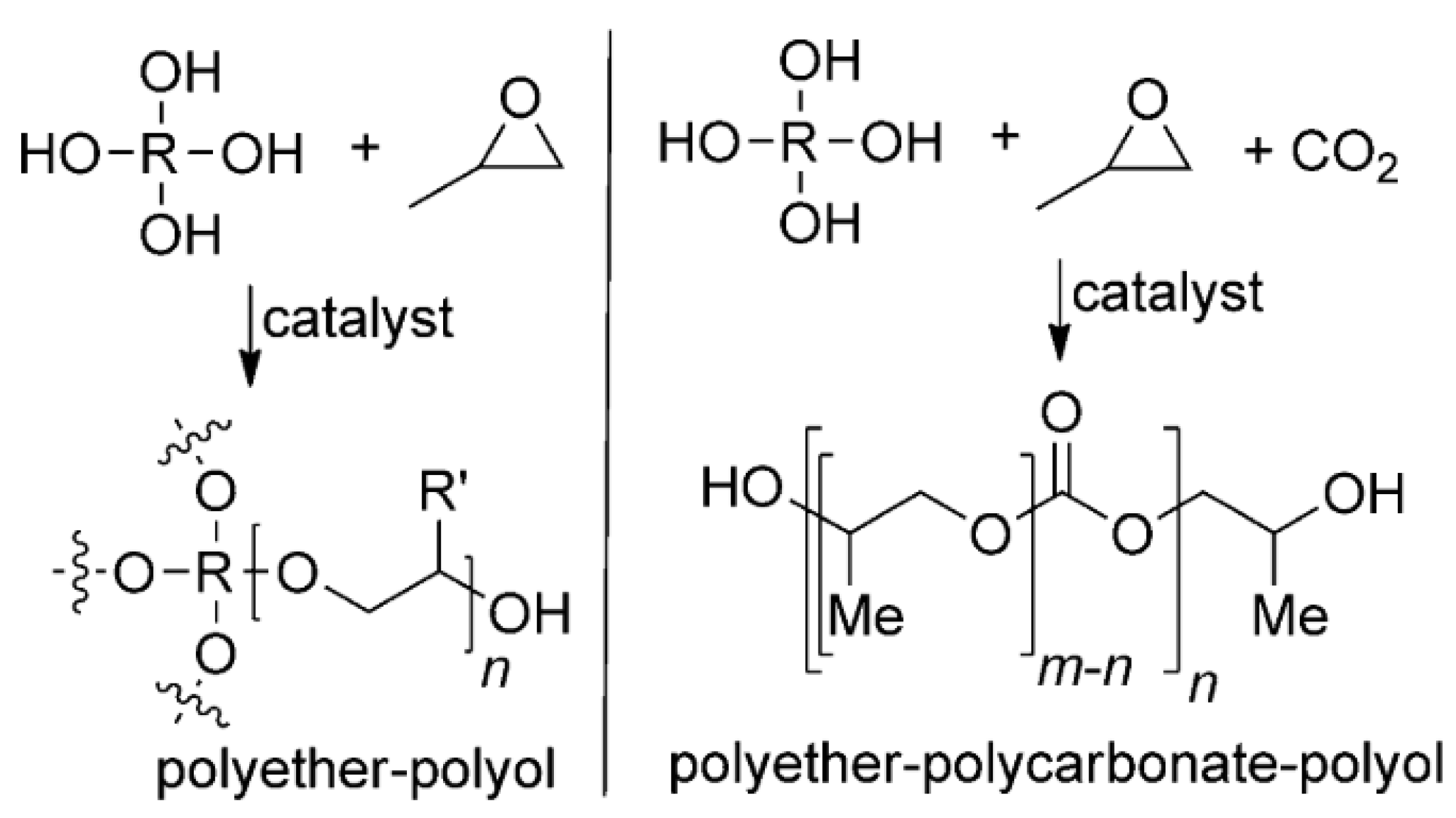
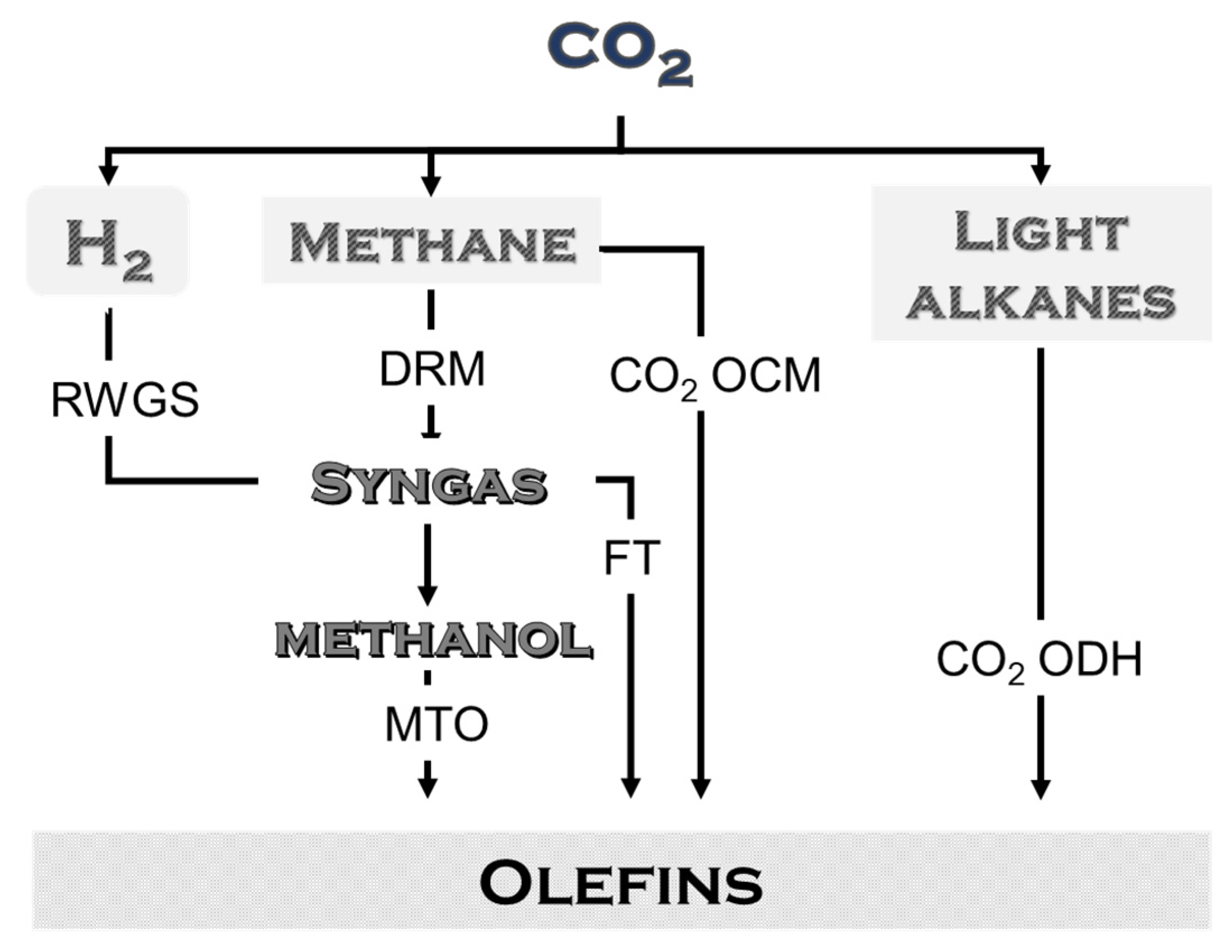
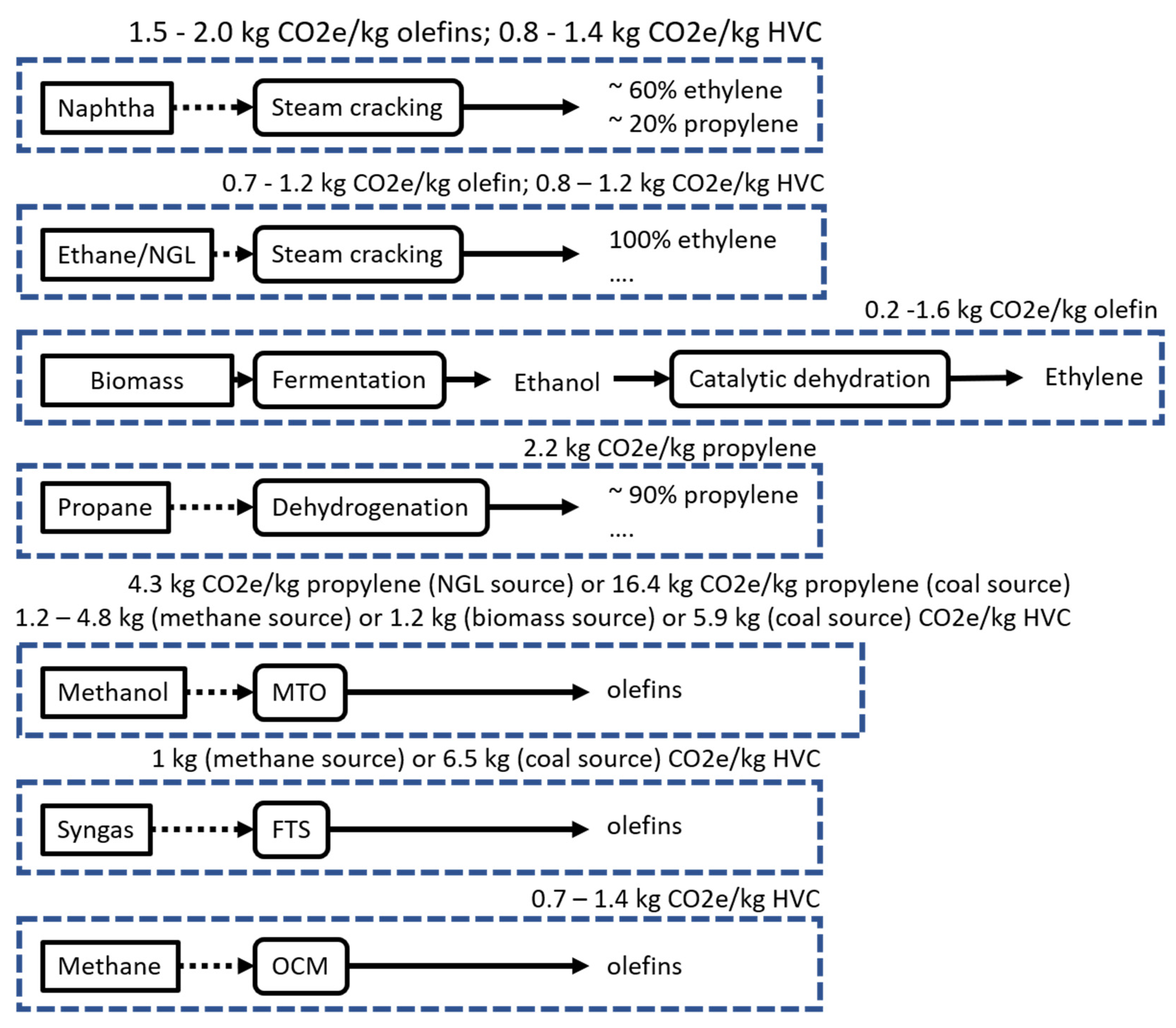

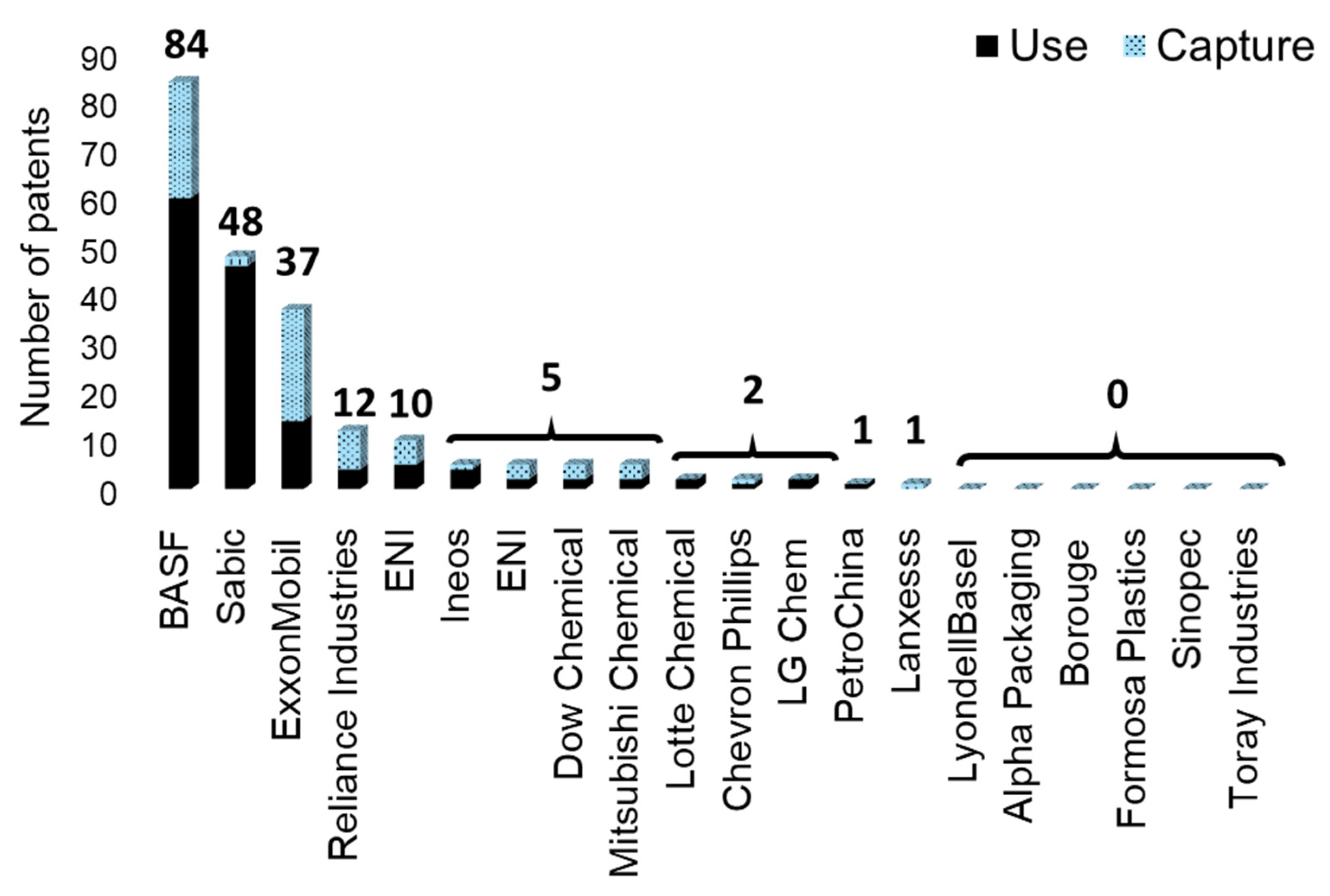
| Ethylene | Ethylene Oxide | Ethylene Glycol | Propylene | Xylene | Terephthalic Acid | |
|---|---|---|---|---|---|---|
| 1.12 | 0.21 | 0 | 1.14 | 1.3 | 0.106 | |
| Benzene | Vinyl chloride | Hydrogen | Styrene | HDPE | LDPE | |
| 1.12 | 0.187 | 0.114 | 0.114 | 0.0349 | 0.0020 | |
| LLDPE | PP | PET | PS | HIPS | PVC | |
| 0.0826 | 0.0416 | 0 | ■ | ■ | 0.0108 |
| General Information | Source | Capture | Use |
|---|---|---|---|
| Title | Source | Capture technique | Conversion technique |
| Authors | Quantity | Method | Method |
| Institution | Concentration | Material | Conversion product |
| Year | Experimental conditions | Efficiency | |
| Country | Supplier | Experimental conditions | |
| Journal | Concentration of | Application | |
| Number of citations | Cost | Material | |
| Main subject: capture/use | Volume of | Catalyst | |
| Selectivity | Reactor | ||
| Recovery | Reaction mechanisms | ||
| Operating scale | Operating scale | ||
| Relevant references | Supplier | ||
| Maturity | Maturity |
| Players (A–C) | Players (C–L) | Players (L–Z) |
|---|---|---|
| Algatec | C-Capture | LG Chem |
| A2BE Carbon Capture | CERT | LiquidLight |
| AGG Biofuel | Chevron Phillips | Lotte Chemical |
| Air Co. | Climeworks | LyondellBasel |
| Algae AquaCulture Technology | CO2 Solutions | Mitsubishi Chemical |
| Algenol Biofuels | CO2 Concrete | MOF Technologies |
| Aljadix | Cool Planet Energy Systems | Mosaic Materials |
| Alpha | Covestro | Net Power |
| BASF | Dimensional Energy | Newlight Technologies |
| Blue Planet | Dioxide Materials | Novomer |
| Bodega Algae | Dow Chemical | OPUS12 |
| Borouge | EcoEra | PetroChina |
| Breathe | Econic Technologies | Phycal |
| Bright Energy | ENI | PHYCO2 |
| bse engineering | EnobraQ | Phytonix |
| C12 Energy | Evolution Petroleum | Pond Technologies |
| C2CNT | ExxonMobil | Reliance Industries |
| C4X | Formosa Plastics Corp | RenewCO2 |
| CalciTech | FuelCell Energy | Sabic |
| Calera | Global Thermostat | Sinopec |
| CarbFix carbon mineralization | Green Minerals | Skyonic |
| Carbicrete | Grow Energy | SkyTree |
| Carbon Capture Machine | Hago energetics | Sumitomo Chemical |
| Carbon Clean Solutions | INEOS | Sustainable Energy Solutions |
| Carbon Engineering | InnoSepra | Synthetic Genomics |
| Carbon Recycling International | Innovator Energy | Tandem Technical |
| Carbon Upcycling Technologies Inc. | Jupiter Oxygen | Toray Industries |
| CarbonCure | Just BioFiber Structural Solutions Corp. | Trelys |
| Carbonfree Chemicals (Skyonic Corporation) | Lanxess | |
| Carbozyme | LanzaTech |
| Player Specification | Search Strategy | Patent Information |
|---|---|---|
| Player/assignee | Keywords search | Code |
| Official site | Number of results | Applicant/assignee |
| Activities | Author(s) | |
| Sector | Priority year | |
| Country | Status | |
| Logo | Title | |
| Main subject (capture and/or use) |
| Ranking | Journal | IF 1 | NP 2 | Percentage (%) |
|---|---|---|---|---|
| 1 | Renewable and Sustainable Energy Reviews | 10.556 | 27 | 15.0 |
| 2 | Progress in Energy and Combustion Science | 26.467 | 6 | 3.3 |
| 3 | Bioresource Technology | 6.669 | 5 | 2.8 |
| 4 | Energy and Environmental Science | 33.250 | 5 | 2.8 |
| 5 | International Journal of Greenhouse Gas Control | 3.231 | 5 | 2.8 |
| 6 | Journal of Utilization | 5.189 | 5 | 2.8 |
| 7 | Energy and Fuels | 3.021 | 4 | 2.2 |
| 8 | Journal of Cleaner Production | 6.395 | 4 | 2.2 |
| Ranking | Institution | Country | Review Publications | Percentage (%) | Subject |
|---|---|---|---|---|---|
| 1 | Nankai University | China | 4 | 2.2 | Capture and use |
| 2 | University of Malaya | Malaysia | 4 | 2.2 | Capture and use |
| 3 | Universiti Teknologi Malaysia | Malaysia | 3 | 1.7 | Capture and use |
| 4 | South China University of Technology | China | 3 | 1.7 | Capture and use |
| 5 | Tsinghua University | China | 3 | 1.7 | Capture and use |
| 6 | Monash University | Australia | 3 | 1.7 | Capture and use |
| 7 | Hunan University | China | 3 | 1.7 | Capture and use |
| 8 | Hanyang University | Republic of Korea | 3 | 1.7 | Capture and use |
| 9 | Chinese Academy of Sciences | China | 3 | 1.7 | Capture and use |
| # | Paper | Subject | Technology | Citations | Ref |
|---|---|---|---|---|---|
| 1 | An overview of current status of carbon dioxide capture and storage technologies Authors: Leung, D. Y., Caramanna, G., and Maroto-Valer, M. M. Source: Renewable and Sustainable Energy Reviews (2014) | Capture and use | Absorption Adsorption CLC 1 Membranes Gas hydrates Cryogenic distillation Chemical conversion EOR 3 and/or ECBM 2 | 1697 | [105] |
| 2 | An overview of CO2 capture technologies Authors: MacDowell, N., Florin, N., Buchard, A., et al. Source: Energy & Environmental Science (2010) | Capture | Membranes Adsorption CLC 1 | 1300 | [106] |
| 3 | Sequestration of carbon dioxide in coal with enhanced coalbed methane recovery: A review Authors: White, C. M., Smith, D. H., Jones, K. L., et al. Source: Energy and Fuels (2005) | Capture | Adsorption ECBM 2 | 918 | [107] |
| 4 | Anthroporgenic Chemical Carbon Cycle for a Sustainable Future Authors: Olah, G. A., Prakash, G. S., and Goeppert, A. Source: Journal of the American Chemical Society (2011) | Capture and use | Absorption Pre-combustion Hydrogenation Coal gasification Electrochemical processes | 907 | [108] |
| 5 | Sustainable hydrocarbon fuels by recycling CO2 and H2O with renewable or nuclear energy Authors: Graves, C., Ebbesen, S. D., Mogensen, M., and Lackner, K. S Source: Renewable and Sustainable Energy Reviews (2011) | Use | Electrochemical processes | 846 | [109] |
| Sector | CO2 Sources | CO2 Concentration in Exhaust Gas (%vol) |
|---|---|---|
| Biomass processes | Biomass fermentation | 15–50 |
| Biogas upgrading | ~100 | |
| Bioethanol production | ~100 | |
| Power generation plants | Natural gas combustion | 3–5 |
| Petroleum combustion | 3–8 | |
| Coal combustion | 10–15 | |
| Industrial processes | Cement production | 14–33 |
| Iron and steel production | 15–30 | |
| Ethylene oxide production | ~100 | |
| Petrochemical | 8–20 | |
| Refineries | ~3–13 | |
| Gas combustion | Vehicles | ~12–14 |
| Extraction | Gas natural mining | ~5–70 |
| Environment | Ambient air | ~0.04 |
| Volcanos | ~2–50 |
| Technique/Maturity | Type of Power Plant/Plant Efficiency (%) [112] | Advantages | Drawbacks | Separation |
|---|---|---|---|---|
| Pre-combustion [113,128] |
|
|
| |
| Post-combustion [105,119,128,129] |
|
|
|
|
| Oxyfuel [43,44,97,100] |
|
|
|
|
| Reaction | Stoichiometry |
|---|---|
| Main ethylene oxide reaction | |
| Undesired side reactions | |
| Separation Technique | Definition |
|---|---|
| Absorption | It can be classified as chemical or physical. In the first case, a chemical reaction occurs between an absorbent molecule and CO2. In the second case, the physical solvent readily incorporates and dissolves CO2, and weak binding forces between gas and solvent molecules keep CO2 in solution. Regeneration is frequently performed through pressure swing [129,160]. |
| Adsorption | A porous material with high specific surface area is used for adsorption of CO2 molecules through intermolecular forces. Selective adsorption of the gases depends on temperature, partial pressures, surface forces, and adsorbent pore size distribution. Adsorbent beds are regenerated mainly by pressure swing and temperature swing methods [160,161,162]. |
| Membranes | Semi-permeable barriers prepared with different materials (organic or inorganic) can be used to separate substances through various mechanisms, including solution/diffusion, adsorption/diffusion, molecular sieving, ionic transport, and facilitated transport [163]. |
| Cryogenic separation process | Distillation of CO2 can be performed at low temperatures and high pressures [161]. |
| Hybrid processes | Two or more separation subsystems can be combined to improve separation efficiency and reduce costs, as in the case of combined adsorption/membrane systems [114,161]. |
| Hydrate separation | Hydrates are initially formed by exposing the CO2-rich exhaust gas stream to water under high pressure. As hydrates are formed, CO2 is captured. Hydrates are then separated and dissociated, releasing CO2 in pure form [164]. |
| Biofixation | Autotrophic microorganisms fix CO2 to synthesize biological organic materials and extracellular products, capturing CO2 from the atmosphere or other sources (including flue gases) with help of different cultivation systems. As a result, CO2 is fixed in the form of organic biomass, which can be converted into different chemicals and biofuels through biorefining processes [165,166]. |
| Mineralization | Mineralization involves the reaction of CO2 with materials that contain alkaline earth oxides such as magnesium oxide (MgO) and calcium oxide (CaO). Valuable carbonate products can be obtained from industrial byproducts and wastes [167]. |
| Calcium looping processes (CLP) | The CLP technology is a special mineralization technique that removes CO2 from flue gas by carbonating calcium oxide (CaO) to calcium carbonate (CaCO3) [168]. |
| Molten carbonate fuel cell (MCFC) | The MCFC technology uses a molten carbonate salt suspended in a porous ceramic matrix as an electrolyte that is able to perform the internal reforming reaction, converting other fuels to hydrogen [169]. |
| Electrochemically mediated amine regeneration (EMAR) | The EMAR technology constitutes an alternative route for regeneration of amines for carbon capture from a flue gas source that does not make use of thermal regeneration, saving energy and increasing the CO2 capture efficiency [170]. |
| Company | Country | Technology | Technology Description | Scale | Patents Related to CO2 Capture |
|---|---|---|---|---|---|
| Bright Energy | USA | Cryogenic carbon capture | Frost Carbon Capture (FrostCC) system that cools the flue gas to separate CO2. | Bench | 0 |
| C-Capture | UK | Chemical absorption | Use of an amine-free solvent | Pilot | 2 |
| Carbon Clean Solutions | UK | Chemical absorption | Use of a proprietary solvent (APBS) that is able to capture flue gases with low CO2 concentration; enabling over 90% of CO2 capture from the feed flue gases. | Large | 8 |
| Carbon Engineering | DAC (chemical absorption) | Use of a potassium hydroxide solution | Pilot | 9 | |
| Carbozyme | USA | Enzymatically active membrane | Use of a polypropylene hollow fiber membrane with an immobilized catalyst (carbonic anhydrase, CA) | Bench | 0 |
| Climeworks | Swiss | DAC (adsorption) | Use of porous granulates modified with amines to capture of CO2 from air. Desorption is performed by temperature swing. | Large (modular) scale | 9 |
| CO2 Solutions | Canada | Enzymatically active solvent | Chemical absorption with immobilized enzymes (carbonic anhydrase, CA) | Large | 14 |
| FuelCell Energy | USA | MCFCs | MCFCs are electrochemical processes that are able to concentrate and capture CO2 | Bench | 19 a |
| Global Thermostat | USA | Chemical adsorption | Use of amine-based chemical sorbents bonded to porous ceramic monoliths. CO2 is stripped off and collected using low-temperature steam (85–100 °C). | Pilot | 5 b |
| InnoSepra | USA | Physical adsorption | Use of a zeolite-activated carbon by PSA and TSA. | Pilot | 2 |
| Innovator Energy | USA | Chemical absorption | Use of ammonia-based solution, which works at room temperature and pressure conditions. The organic solvent and CO2 absorbing solution are then regenerated using low temperature heat. | Pilot | 0 |
| Jupiter Oxygen | USA | Oxyfuel combustion | Uses 95–100% pure O2 as fuel to perform combustion. Highly concentrated CO2 streams can be obtained afterward. | Large | 5 |
| Net Power | USA | Oxyfuel combustion | Using high-pressure oxyfuel, the supercritical CO2 cycle is a CO2 capture system based on the Allam cycle. | Pilot | 13 |
| Sunfire | Germany | SOFC | Cogeneration of heat and energy with easy separation of CO2 | Large | 2 |
| Sustainable Energy Solutions | USA | Cryogenic carbon capture | CO2 is cooled to about −140 °C), separated, pressurized, melted, and delivered at pipeline pressure | Small pilot | 12 |
| Reaction. | Stoichiometry | Enthalpy |
|---|---|---|
| Steam methane reforming (SMR) | ||
| Partial oxidation reforming (POR), partial oxidation of methane (POM), or partial oxidation (POx) of methane | ||
| Dry methane reforming (DRM) |
| Reaction | Stoichiometry | Enthalpy |
|---|---|---|
| Methane thermal decomposition | ||
| Carnol process | - | |
| Methane thermal decomposition + DRM | - |
| Reaction | Stoichiometry | Enthalpy |
|---|---|---|
| OCM | ||
| CO2 OCM | ||
| Reaction | Stoichiometry | Enthalpy |
|---|---|---|
| CO hydrogenation | ||
| Methanol synthesis | ||
| rWGS |
| Reaction | Stoichiometry |
|---|---|
| Waxes | |
| Olefins | |
| Methane | |
| Alcohols | |
| Carbon deposition |
| Reaction | Stoichiometry | Enthalpy |
|---|---|---|
| Sabatier reaction | ||
| FT synthesis |
| Reaction | Stoichiometry |
|---|---|
| Dehydrogenation | |
| CO2 ODH | |
| Mars–van Krevelen mechanism | |
| Company | Country | Technology | Technology Description | Product | Scale | Patents in Use |
|---|---|---|---|---|---|---|
| BASF | Germany | Polymerization, hydrogenation, FT, DRM, methanization | Formation of polyethercarbonate polyol; copolymerization of alkylene oxides and carbon dioxide using metal cyanide catalysts; hydrogenation using metal catalysts (aluminum or copper); DRM using mixed metal oxides; methanization using a ruthenium-rhenium-based or nickel/cobalt catalyst | Polyol, polyether carbonate, methanol, DME, ethylene, propylene | 60 | |
| Sabic | Saudi Arabia | Hydrogenation, FT, DRM, reduction, oxidative dehydrogenation supercritical, carrier gas, blowing agent | Hydrogenation via RWGS (and further FT synthesis) including metal catalyst (Cr, aluminia), mixed metal oxide catalysts (CuMnAl and CuZnZr); Catofin catalyst. Reduction of CO2 with sulfur or metal-free catalyst. | CO, syngas, methanol, light olefins | 46 | |
| ExxonMobil | USA | Hydrogenation; Fischer–Tropsch, MTO, DRM | Using a molecular sieve to i) oxygenates conversion to olefins using also at least one metal oxide; MTO; production of syngas and, further, methanol; dry reforming of methane; RWGS | Syngas, methanol, ethylene, propylene | 14 | |
| ENI | Italy | RWGS, FT | RWGS and FT in the presence of a cobalt catalyst | Syngas; paraffins | 5 | |
| Ineos | UK | Hydrogenation; FT; DMR; blowing agent | Methanol synthesis from syngas, methane reforming to alcohols Fischer–Tropsch to olefins; blowing agent in expandable polystyrene | Methanol, ethylene, ethanol, propanol, butanol | 4 | |
| Reliance Industries | India | Fischer–Tropsch, methanol synthesis, MTO | Syngas to olefins; carboxylation reaction; dehydrogenation; gasifying agent | Olefins; nitric acid; butadiene; syngas | 4 | |
| Mitsubishi Chemical ▲ | Japan | Cyclic carbonates synthesis | Reaction between carbon dioxide and ethylene oxide | Ethylene carbonate | 2 | |
| Dow Chemical ▲ | USA | Hydrogenation; Fischer–Tropsch | Hydrogenation using a mixed metal oxide catalyst or a molecular sieve catalyst | Syngas | 2 | |
| Lotte Chemicals | Malaysia | Polymerization; cyclic carbonates synthesis | Copolymerization between carbon dioxide and epoxide; reaction between alkylene oxide and carbon dioxide | Polycarbonate; ethylene carbonate | 2 | |
| LG Chem | Korea | Polymerization | Polyalkylene carbonate resins by polymerization of carbon dioxide and epoxides | Polyalkylene carbonate | 2 | |
| PetroChina | China | Supercritical | Swelling polypropylene in supercritical carbon dioxide having dissolved vinyl monomer and an initiator | Solid phase graft modified polypropylene | 1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pires da Mata Costa, L.; Micheline Vaz de Miranda, D.; Couto de Oliveira, A.C.; Falcon, L.; Stella Silva Pimenta, M.; Guilherme Bessa, I.; Juarez Wouters, S.; Andrade, M.H.S.; Pinto, J.C. Capture and Reuse of Carbon Dioxide (CO2) for a Plastics Circular Economy: A Review. Processes 2021, 9, 759. https://doi.org/10.3390/pr9050759
Pires da Mata Costa L, Micheline Vaz de Miranda D, Couto de Oliveira AC, Falcon L, Stella Silva Pimenta M, Guilherme Bessa I, Juarez Wouters S, Andrade MHS, Pinto JC. Capture and Reuse of Carbon Dioxide (CO2) for a Plastics Circular Economy: A Review. Processes. 2021; 9(5):759. https://doi.org/10.3390/pr9050759
Chicago/Turabian StylePires da Mata Costa, Laura, Débora Micheline Vaz de Miranda, Ana Carolina Couto de Oliveira, Luiz Falcon, Marina Stella Silva Pimenta, Ivan Guilherme Bessa, Sílvio Juarez Wouters, Márcio Henrique S. Andrade, and José Carlos Pinto. 2021. "Capture and Reuse of Carbon Dioxide (CO2) for a Plastics Circular Economy: A Review" Processes 9, no. 5: 759. https://doi.org/10.3390/pr9050759
APA StylePires da Mata Costa, L., Micheline Vaz de Miranda, D., Couto de Oliveira, A. C., Falcon, L., Stella Silva Pimenta, M., Guilherme Bessa, I., Juarez Wouters, S., Andrade, M. H. S., & Pinto, J. C. (2021). Capture and Reuse of Carbon Dioxide (CO2) for a Plastics Circular Economy: A Review. Processes, 9(5), 759. https://doi.org/10.3390/pr9050759