Evaluation of a Full-Scale Suspended Sludge Deammonification Technology Coupled with an Hydrocyclone to Treat Thermal Hydrolysis Dewatering Liquors
Abstract
1. Introduction
2. Materials and Methods
2.1. Influent Characteristics
2.2. Reactor Configurations
2.3. Reactor Operation and Process Evaluation
2.4. Sample Collection and Analysis
3. Results and Discussion
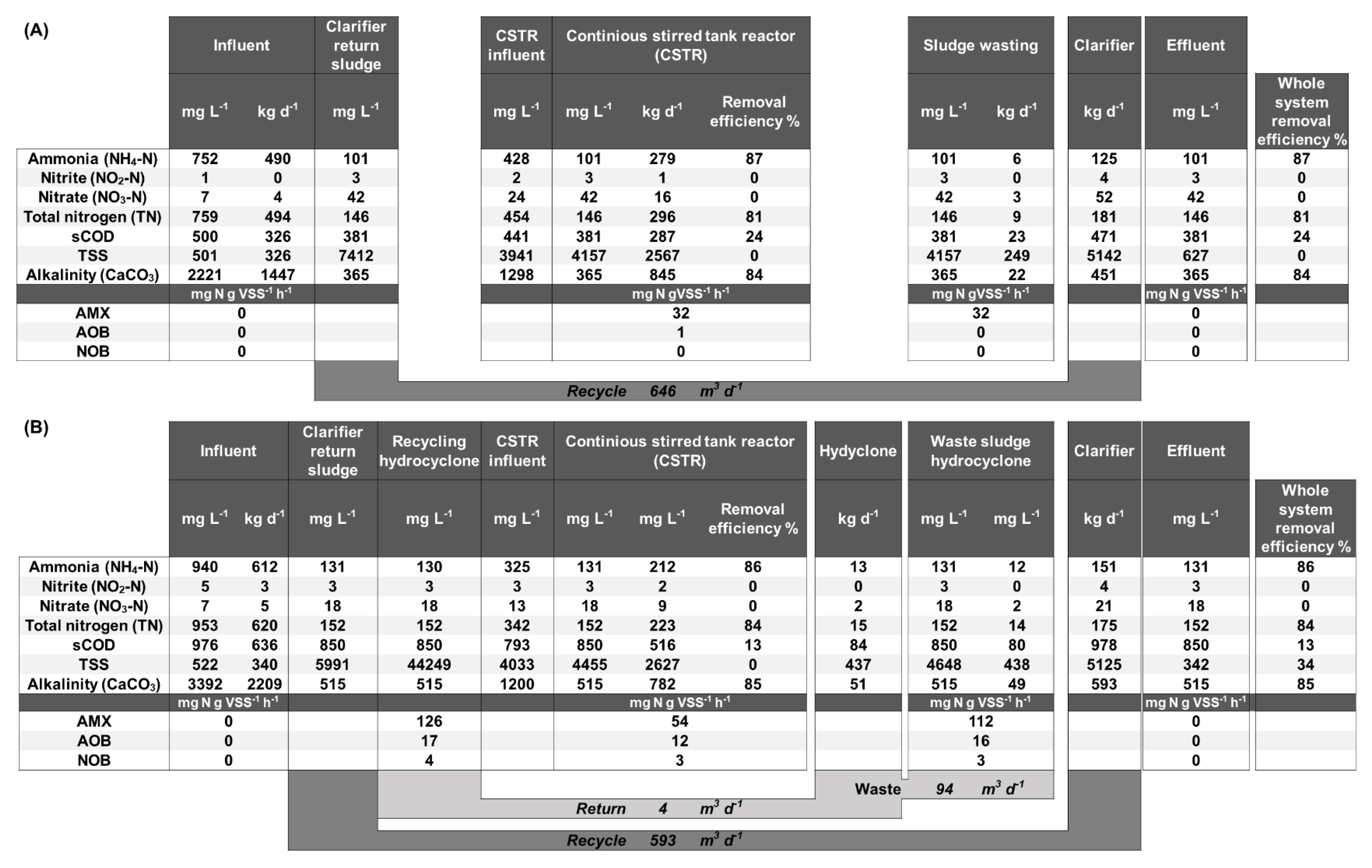
3.1. Reactor Performance
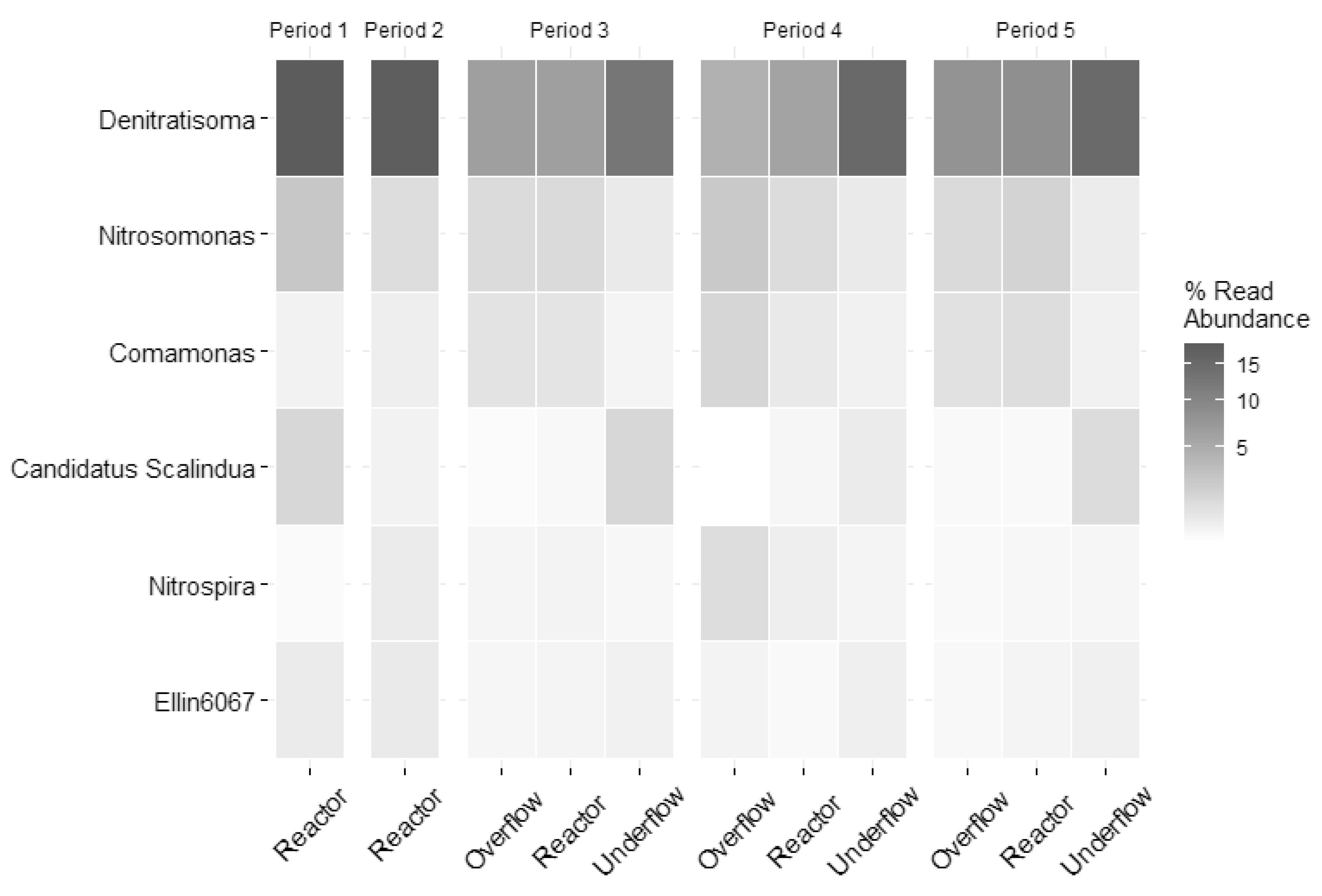
3.2. Biomass Retention
4. Conclusions
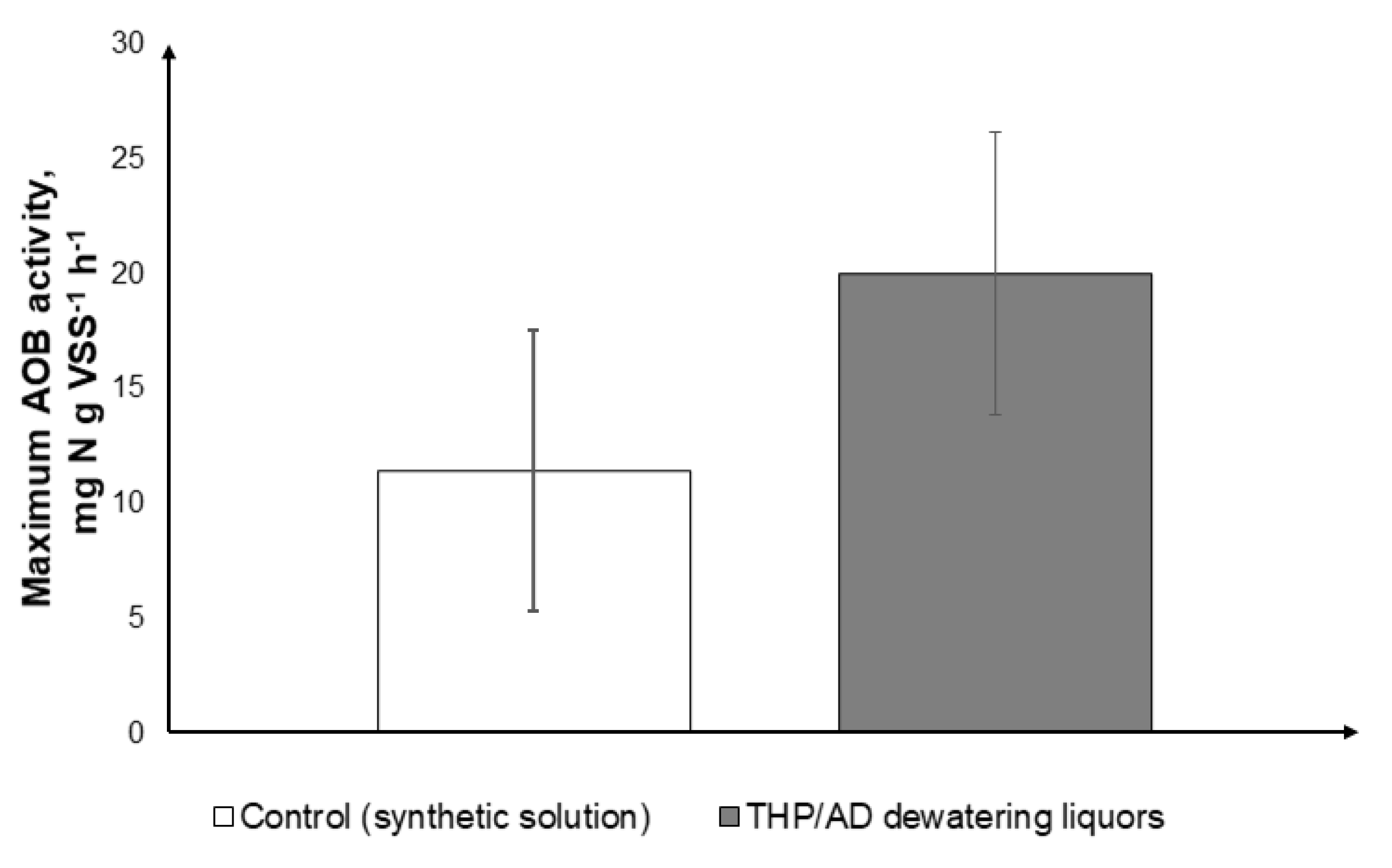
- The S-CSTR achieved successful ammonia removal from THP/AD dewatering liquors with efficiencies >85%. Ex-situ AOB activity tests indicated that THP/AD dewatering liquors did not impact the ammonia conversion.
- Sodium hydroxide dosing enabled the greater ammonia removal efficiencies of 95% but led to the undesired NOB overgrowth, and nitrate accumulation.
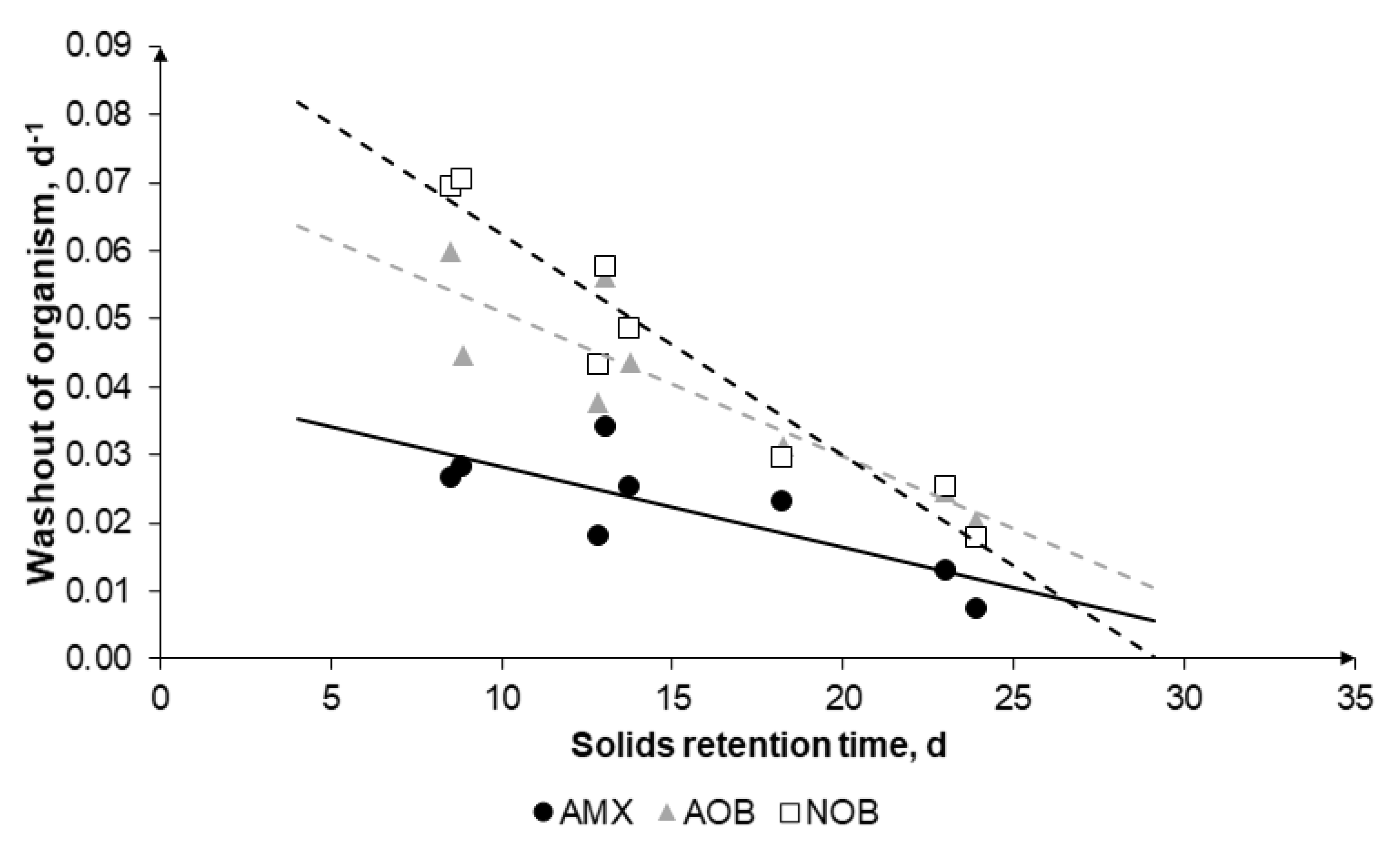
- The hydrocyclone was key to retain 56–83% AMX biomass. Additionally, the AMX washout of the hydrocyclone based on mass-balance was 0.01–0.03 d−1 while the washout of AOB and NOB was 0.3–0.7 d−1.
- SRTs of shorter than 13 days lead to NOB washout, but below 11 days AOB will also be wash-out.
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Barber, W.P.F. Thermal hydrolysis for sewage treatment: A critical review. Water Res. 2016, 104, 53–71. [Google Scholar] [CrossRef]
- Winter, P.; Ng, B.; Smith, S.R.; Strange, G.; Jarvis, S.; Macedo, F.; Shen, X. Impact of Sludge Liquors on the Hydraulic, Nutrient and Solids Load of Wastewater Treatment. In Proceedings of the IWA Specialist Conference on Sludge Management 17; IWA Publishing: London, UK, 2017; pp. 1–17. [Google Scholar]
- Strous, M.; Heijnen, J.; Kuenen, J.; Jetten, M. The sequencing batch reactor as a powerful tool for the study of slowly growing anaerobic ammonium-oxidizing microorganisms. Appl. Microbiol. Biotechnol. 1998, 50, 589–596. [Google Scholar] [CrossRef]
- Jetten, M.S.M.; Strous, M.; Fuerst, J.A.; Kramer, E.H.M.; Logemann, S.; Muyzer, G.; van de Pas-Schoonen, K.T.; Webb, R.; Kuenen, J.G.; Jhon, A.F.; et al. Missing lithotroph identified as new planctomycete. Nature 1999, 400, 446–449. [Google Scholar] [CrossRef]
- Figdore, B.; Wett, B.; Hell, M.; Murthy, S. Deammonification of Dewatering Sidestream from Thermal Hydrolysis-Mesophilic Anaerobic Digestion Process. Proc. Water Environ. Fed. 2011, 2011, 1037–1052. [Google Scholar] [CrossRef][Green Version]
- Zhang, Q.; de Clippeleir, H.; Su, C.; Omari, A.A.; Wett, B.; Vlaeminck, S.E.; Murthy, S.; Figdore, B.; Wett, B.; Hell, M.; et al. Deammonification for digester supernatant pretreated with thermal hydrolysis: Overcoming inhibition through process optimization. Appl. Microbiol. Biotechnol. 2016, 100, 5595–5606. [Google Scholar] [CrossRef]
- Driessen, W.; van Veldhoven, J.T.A.; Janssen, M.; van Loosdrecht, M.C.M. Treatment of sidestream dewatering liquors from thermally hydrolised and anaerobically digested biosolids. Water Pract. Technol. 2020, 15, 142–150. [Google Scholar] [CrossRef]
- Ochs, P.; Martin, B.D.; Germain, E.; Stephenson, T.; van Loosdrecht, M.; Soares, A. Ammonia removal from thermal hydrolysis dewatering liquors via three different deammonification technologies. Sci. Total Environ. 2021, 755, 142684. [Google Scholar] [CrossRef]
- Lackner, S.; Gilbert, E.M.; Vlaeminck, S.E.; Joss, A.; Horn, H.; van Loosdrecht, M.C.M. Full-scale partial nitritation/anammox experiences—An application survey. Water Res. 2014, 55, 292–303. [Google Scholar] [CrossRef]
- Daigger, G.T.; Sanjines, P.; Pallansch, K.; Sizemore, J.; Wett, B. Implementation of a full-scale anammox-based facility to treat an anaerobic digestion sidestream at the alexandria sanitation authority water resource facility. Water Pract. Technol. 2011, 6. [Google Scholar] [CrossRef]
- Tchobanoglous, G.; Burton, F.L.; Tsuchihashi, R.; Stensel, H.D. Wastewater Engineering: Treatment and Resource Recovery, 5th ed.; Abu-Orf, M., Bowden, G., Pfrang, W., Tchobanoglous, G., Stensel, D.H., Tsuchihashi, R.B.F., Eds.; McGraw-Hill: New York, NY, USA, 2014; ISBN 0073401188. [Google Scholar]
- Mehrdad, M.; Park, H.; Ramalingam, K.; Fillos, J.; Beckmann, K.; Deur, A.; Chandran, K. Anammox moving bed biofilmreactor pilot at the 26th Ward wastewater treatment plants in Brooklyn, New York: Start-up, biofilm population diversity and performance optimization. Water Sci. Technol. 2014, 70, 1448–1455. [Google Scholar] [CrossRef]
- Lotti, T.; Kleerebezem, R.; Hu, Z.; Kartal, B.; Jetten, M.S.M.; van Loosdrecht, M.C.M. Simultaneous partial nitritation and anammox at low temperature with granular sludge. Water Res. 2014, 66, 111–121. [Google Scholar] [CrossRef]
- Fux, C.; Siegrist, H. Nitrogen removal from sludge digester liquids by nitrification/denitrification or partial nitritation/anammox: Environmental and economical considerations. Water Sci. Technol. 2004, 50, 19–26. [Google Scholar] [CrossRef]
- van der Star, W.R.L.; Abma, W.R.; Blommers, D.; Mulder, J.W.; Tokutomi, T.; Strous, M.; Picioreanu, C.; van Loosdrecht, M.C.M. Startup of reactors for anoxic ammonium oxidation: Experiences from the first full-scale anammox reactor in Rotterdam. Water Res. 2007, 41, 4149–4163. [Google Scholar] [CrossRef] [PubMed]
- Wett, B. Development and implementation of a robust deammonification process. Water Sci. Technol. 2007, 56, 81–88. [Google Scholar] [CrossRef] [PubMed]
- Wett, B.; Hell, M.; Nyhuis, G.; Puempel, T.; Takacs, I.; Murthy, S. Syntrophy of aerobic and anaerobic ammonia oxidisers. Water Sci. Technol. 2010, 61, 1915–1922. [Google Scholar] [CrossRef] [PubMed]
- van Winckel, T.; Vlaeminck, S.E.; Omari, A.A.; Bachmann, B.; Sturm, B.; Wett, B.; Takács, I.; Bott, C.; Murthy, S.N.; de Clippeleir, H. Screen versus cyclone for improved capacity and robustness for sidestream and mainstream deammonification. Environ. Sci. Water Res. Technol. 2019, 5, 1769–1781. [Google Scholar] [CrossRef]
- Shi, Y.; Wells, G.; Morgenroth, E. Microbial activity balance in size fractionated suspended growth biomass from full-scale sidestream combined nitritation-anammox reactors. Bioresour. Technol. 2016, 218, 38–45. [Google Scholar] [CrossRef]
- Wett, B.; Nyhuis, G.; Takács, I.; Murthy, S. Development of Enhanced Deammonification Selector. Proc. Water Environ. Fed. 2010, 2010, 5917–5926. [Google Scholar] [CrossRef]
- Han, M.; Vlaeminck, S.E.; Omari, A.A.; Wett, B.; Bott, C.; Murthy, S.; De Clippeleir, H. Uncoupling the solids retention times of flocs and granules in mainstream deammonification: A screen as effective out-selection tool for nitrite oxidizing bacteria. Bioresour. Technol. 2016, 221, 195–204. [Google Scholar] [CrossRef]
- Kwon, K.; Kim, H.; Kim, W.; Lee, J. Efficient nitrogen removal of reject water generated from anaerobic digester treating sewage sludge and livestock manure by combining anammox and autotrophic Sulfur denitrification processes. Water (Switzerland) 2019, 11, 204. [Google Scholar] [CrossRef]
- Lotti, T.; Kleerebezem, R.; Lubello, C.; van Loosdrecht, M.C.M. Physiological and kinetic characterization of a suspended cell anammox culture. Water Res. 2014, 60, 1–14. [Google Scholar] [CrossRef] [PubMed]
- APHA. Standard Methods for the Examination of Water and Wastewater, 23rd ed.; American Public Health Association, American Water Works Association, and Water Environment Federation: Washington, WA, USA, 2017; ISBN 9780875532875. [Google Scholar]
- Lotti, T.; van der Star, W.R.L.; Kleerebezem, R.; Lubello, C.; van Loosdrecht, M.C.M.; Van der Star, W.; Kleerebezem, R.; Research, C.L.-W. The effect of nitrite inhibition on the anammox process. Water Res. 2012, 46, 2559–2569. [Google Scholar] [CrossRef]
- van Loosdrecht, M.C.M.; Nielsen, P.H.; Vazquez, C.M.L.; Brdjanovic, D. Experimental Methods in Wastewater Treamtent, 1st ed.; IWA Publishing: London, UK, 2016; ISBN 9781780404745. [Google Scholar]
- Klindworth, A.; Pruesse, E.; Schweer, T.; Peplies, J.; Quast, C.; Horn, M.; Glöckner, F.O. Evaluation of general 16S ribosomal RNA gene PCR primers for classical and next-generation sequencing-based diversity studies. Nucleic Acids Res. 2013, 41, e1. [Google Scholar] [CrossRef]
- Schloss, P.D.; Westcott, S.L.; Ryabin, T.; Hall, J.R.; Hartmann, M.; Hollister, E.B.; Lesniewski, R.A.; Oakley, B.B.; Parks, D.H.; Robinson, C.J.; et al. Introducing mothur: Open-source, platform-independent, community-supported software for describing and comparing microbial communities. Appl. Environ. Microbiol. 2009, 75, 7537–7541. [Google Scholar] [CrossRef] [PubMed]
- Edgar, R.C.; Haas, B.J.; Clemente, J.C.; Quince, C.; Knight, R. UCHIME improves sensitivity and speed of chimera detection. Bioinformatics 2011, 27, 2194–2200. [Google Scholar] [CrossRef] [PubMed]
- Pruesse, E.; Quast, C.; Knittel, K.; Fuchs, B.M.; Ludwig, W.; Peplies, J.; Glöckner, F.O. SILVA: A comprehensive online resource for quality checked and aligned ribosomal RNA sequence data compatible with ARB. Nucleic Acids Res. 2007, 35, 7188–7196. [Google Scholar] [CrossRef] [PubMed]
- Albertsen, M.; Karst, S.M.; Ziegler, A.S.; Kirkegaard, R.H.; Nielsen, P.H. Back to basics—The influence of DNA extraction and primer choice on phylogenetic analysis of activated sludge communities. PLoS ONE 2015, 10, e0132783. [Google Scholar] [CrossRef] [PubMed]
- Nifong, A.; Nelson, A.; Johnson, C.; Bott, C.B. Performance of a Full-Scale Sidestream DEMON Deammonification Installation. Proc. Water Environ. Fed. 2013, 2013, 3686–3709. [Google Scholar] [CrossRef][Green Version]
- Vlaeminck, S.E.; van der Linden, D.; Boon, N.; Verstraete, W.; Carballa, M.; Terada, A.; Smets, B.F. Nitrogen removal from digested black water by one-stage partial nitritation and anammox. Environ. Sci. Technol. 2009, 43, 5035–5041. [Google Scholar] [CrossRef]
- Anthonisen, A.C.; Srinath, E.G.; Loehr, R.C.; Prakasam, T.B.S. Inhibition of nitrification and nitrous acid compounds. J. Water Pollut. Control Fed. 1976, 48, 835–852. [Google Scholar] [CrossRef]
- Feng, Y.; Lu, X.; Hazmi, H.A.; Mąkinia, J. An overview of the strategies for the deammonification process start-up and recovery after accidental operational failures. Rev. Environ. Sci. Biotechnol. 2017, 16, 541–568. [Google Scholar] [CrossRef]
- Chung, J.; Shim, H.; Park, S.J.; Kim, S.J.; Bae, W. Optimization of free ammonia concentration for nitrite accumulation in shortcut biological nitrogen removal process. Bioprocess Biosyst. Eng. 2006, 28, 275–282. [Google Scholar] [CrossRef] [PubMed]
- Regmi, P.; Miller, M.W.; Holgate, B.; Bunce, R.; Park, H.; Chandran, K.; Wett, B.; Murthy, S.; Bott, C.B. Control of aeration, aerobic SRT and COD input for mainstream nitritation/denitritation. Water Res. 2014, 57, 162–171. [Google Scholar] [CrossRef] [PubMed]
- Fahrbach, M.; Kuever, J.; Meinke, R.; Kämpfer, P.; Hollender, J. Denitratisoma oestradiolicum gen. nov., sp. nov., a 17 β-oestradiol-degrading, denitrifying betaproteobacterium. Int. J. Syst. Evol. Microbiol. 2006, 56, 1547–1552. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.; Xu, X.; Zhou, L.; Wang, C.; Yang, F. A pilot-scale study on the start-up of partial nitrification-anammox process for anaerobic sludge digester liquor treatment. Bioresour. Technol. 2017, 241, 181–189. [Google Scholar] [CrossRef] [PubMed]
- Wu, Z.; Meng, H.; Huang, X.; Wang, Q.; Chen, W.H.; Gu, J.D.; Lee, P.H. Salinity-driven heterogeneity toward anammox distribution and growth kinetics. Appl. Microbiol. Biotechnol. 2019, 103, 1953–1960. [Google Scholar] [CrossRef]
- Daims, H.; Purkhold, U.; Bjerrum, L.; Arnold, E.; Wilderer, P.A.; Wagner, M. Nitrification in sequencing biofilm batch reactors: Lessons from molecular approaches. Water Sci. Technol. 2001, 43, 9–18. [Google Scholar] [CrossRef]
- Simoes, F.; Colston, R.; Rosa-Fernandes, C.; Vale, P.; Stephenson, T.; Soares, A. Predicting the potential of sludge dewatering liquors to recover nutrients as struvite biominerals. Environ. Sci. Ecotechnol. 2020, 3, 100052. [Google Scholar] [CrossRef]
- Zhang, L.; Okabe, S. Ecological niche differentiation among anammox bacteria. Water Res. 2020, 171, 115468. [Google Scholar] [CrossRef]
- Martinez, A.G.; Morillo, J.A.; Ruiz, M.J.G.; Lopez, J.G.; Osorio, F.; Toledo, M.V.M.; van Loosdrecht, M.C.M. Archaeal populations in full-scale autotrophic nitrogen removal bioreactors operated with different technologies: CANON, DEMON and partial nitritation/anammox. Chem. Eng. J. 2015, 277, 194–201. [Google Scholar] [CrossRef]
- Yang, Y.; Zhang, L.; Cheng, J.; Zhang, S.; Li, X.; Peng, Y. Microbial community evolution in partial nitritation/anammox process: From sidestream to mainstream. Bioresour. Technol. 2018, 251, 327–333. [Google Scholar] [CrossRef] [PubMed]


| Period 1 | Period 2 | Period 3 | Period 4 | Period 5 | |
|---|---|---|---|---|---|
| Operation | Start-up to Steady State | No Solid Retention Time Control | Solid Retention Time Control + NaOH Dosing | Solids Retention Time Control (Load 1) | Solids Retention Time Control (Load 2) |
| Nitrogen loading rate (kg N m−3 d−1) | 0.08 ± 0.04 | 0.22 ± 0.11 | 0.30 ± 0.09 | 0.34 ± 0.12 | 0.39 ± 0.1 |
| Ammonia (mg N L−1) | 1186 ± 79 | 775.3 ± 163 | 880.7 ± 103 | 881.8 ± 273 | 963.3 ± 123 |
| Nitrite (mg N L−1) | 0.3 ± 0 | 0.6 ± 0.5 | 0.4 ± 0.6 | 4.1 ± 6.5 | 1.9 ± 3.2 |
| Nitrate (mg N L−1) | 23 ± 4.8 | 12.9 ± 7.1 | 3.1 ± 3.7 | 3.8 ± 5.5 | 5.4 ± 6.4 |
| Total nitrogen (mg N L−1) | 1209.4 ± 80 | 788.2 ± 166 | 895.5 ± 99 | 889.7 ± 267 | 970.6 ± 121 |
| Total COD (mg L−1) | 2597 ± 654 | 1976 ± 1365 | 2151 ± 534 | 2203 ± 878 | 2039 ± 509 |
| Soluble COD (mg L−1) | n/a | 882 ± 101 | 1244 ± 219 | 938 ± 399 | 1009 ± 190 |
| BOD (mg L−1) | n/a | n/a | 262 ± 76 | 178 ± 105 | 210 ± 92 |
| pH | 8.1 ± 0.1 | 8.2 ± 0.1 | 8.3 ± 0.1 | 8.4 ± 0.1 | 8.4 ± 0.1 |
| Total suspended solids (TSS) (mg L−1) | n/a | 777 ± 972 | 939 ± 1593 | 609 ± 276 | 494 ± 270 |
| Alkalinity (mg CaCO3 L−1) | 4024 ± 362 | 2768 ± 554 | 3325 ± 247 | 3266 ± 1042 | 3442 ± 427 |
| Sodium dosing (L d−1) | n/a | n/a | 917 ± 306 | n/a | n/a |
| Solids retention time (SRT) (d) | 61.3 ± 69.5 | 28.1 ± 28.9 | 17.7 ± 7 | 11.8 ± 3.5 | 13.4 ± 5.5 |
| Dissolved oxygen set-point (mg L−1) | 0.3 | 0.3 | 0.3 | 0.3 | 0.3 |
| pH set-point | 6.8 | 6.8 | 7 | 6.8 | 6.8 |
| Temperature (°C) | 33.3 ± 1.4 | 30.9 ± 2.2 | 34.2 ± 2 | 34.5 ± 1.2 | 33.6 ± 1.2 |
| Period 1 | Period 2 | Period 3 | Period 4 | Period 5 | |
|---|---|---|---|---|---|
| Operation | Start-up to Steady State | No Solid Retention Time Control | Solid Retention Time Control + NaOH Dosing | Solids Retention Time Control (Load 1) | Solids Retention Time Control (Load 2) |
| Nitrogen loading rate (kg N m−3 d−1) | 0.08 ± 0.04 | 0.22 ± 0.11 | 0.30 ± 0.09 | 0.34 ± 0.12 | 0.39 ± 0.1 |
| Nitrogen removal rate (kg N m−3 d−1) | 0.07 ± 0.03 | 0.17 ± 0.08 | 0.22 ± 0.07 | 0.29 ± 0.11 | 0.32 ± 0.1 |
| Ammonia removal efficiency (%) | 92 ± 3 | 85 ± 4 | 95 ± 2 | 87 ± 6 | 85 ± 6 |
| Nitrogen removal efficiency (%) | 89 ± 3 | 78 ± 8 | 75 ± 5 | 84 ± 6 | 82 ± 7 |
| NO3/NH4 ratio | 0.03 ± 0.02 | 0.08 ± 0.05 | 0.2 ± 0.06 | 0.02 ± 0.02 | 0.03 ± 0.01 |
| NO2/NH4 ratio | 0.01 ± 0 | 0.02 ± 0.01 | 0.1 ± 0.05 | 0.04 ± 0.03 | 0.02 ± 0.01 |
| Ammonia (mg N L−1) | 97.9 ± 28.6 | 114.2 ± 39.1 | 45.9 ± 17.8 | 103.9 ± 23.7 | 144.7 ± 47.8 |
| Nitrite (mg N L−1) | 0.7 ± 0.3 | 2.6 ± 1.7 | 4.2 ± 1.9 | 3.6 ± 2.6 | 2.9 ± 1.4 |
| Nitrate (mg N L−1) | 30.3 ± 22.9 | 50.2 ± 30.9 | 172.7 ± 25.4 | 17.2 ± 11.2 | 19.6 ± 6.3 |
| Alkalinity (mg CaCO3 L−1) | 447.1 ± 134.2 | 427.8 ± 68.5 | 344.7 ± 40.2 | 480.2 ± 47.5 | 546.4 ± 159.8 |
| Free ammonia (FA) (mg N L−1) | 2.9 ± 1.3 | 3.1 ± 1.2 | 1.9 ± 1.1 | 5.0 ± 1.5 | 6.4 ± 5.2 |
| Free nitrous acid (FNA) (µg N L−1) | 0.2 ± 0.1 | 0.7 ± 0.3 | 1 ± 0.5 | 0.8 ± 0.4 | 0.6 ± 0.3 |
| Solids retention time (SRT) (d) | 61.3 ± 69.5 | 28.1 ± 28.9 | 17.7 ± 7 | 11.8 ± 3.5 | 13.4 ± 5.5 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ochs, P.; Martin, B.D.; Germain, E.; Wu, Z.; Lee, P.-H.; Stephenson, T.; van Loosdrecht, M.; Soares, A. Evaluation of a Full-Scale Suspended Sludge Deammonification Technology Coupled with an Hydrocyclone to Treat Thermal Hydrolysis Dewatering Liquors. Processes 2021, 9, 278. https://doi.org/10.3390/pr9020278
Ochs P, Martin BD, Germain E, Wu Z, Lee P-H, Stephenson T, van Loosdrecht M, Soares A. Evaluation of a Full-Scale Suspended Sludge Deammonification Technology Coupled with an Hydrocyclone to Treat Thermal Hydrolysis Dewatering Liquors. Processes. 2021; 9(2):278. https://doi.org/10.3390/pr9020278
Chicago/Turabian StyleOchs, Pascal, Benjamin D. Martin, Eve Germain, Zhuoying Wu, Po-Heng Lee, Tom Stephenson, Mark van Loosdrecht, and Ana Soares. 2021. "Evaluation of a Full-Scale Suspended Sludge Deammonification Technology Coupled with an Hydrocyclone to Treat Thermal Hydrolysis Dewatering Liquors" Processes 9, no. 2: 278. https://doi.org/10.3390/pr9020278
APA StyleOchs, P., Martin, B. D., Germain, E., Wu, Z., Lee, P.-H., Stephenson, T., van Loosdrecht, M., & Soares, A. (2021). Evaluation of a Full-Scale Suspended Sludge Deammonification Technology Coupled with an Hydrocyclone to Treat Thermal Hydrolysis Dewatering Liquors. Processes, 9(2), 278. https://doi.org/10.3390/pr9020278






