Potential Use of Papaya Waste as a Fuel for Bioelectricity Generation
Abstract
:1. Introduction
2. Materials and Methods
2.1. Construction of Single-Chamber Microbial Fuel Cells
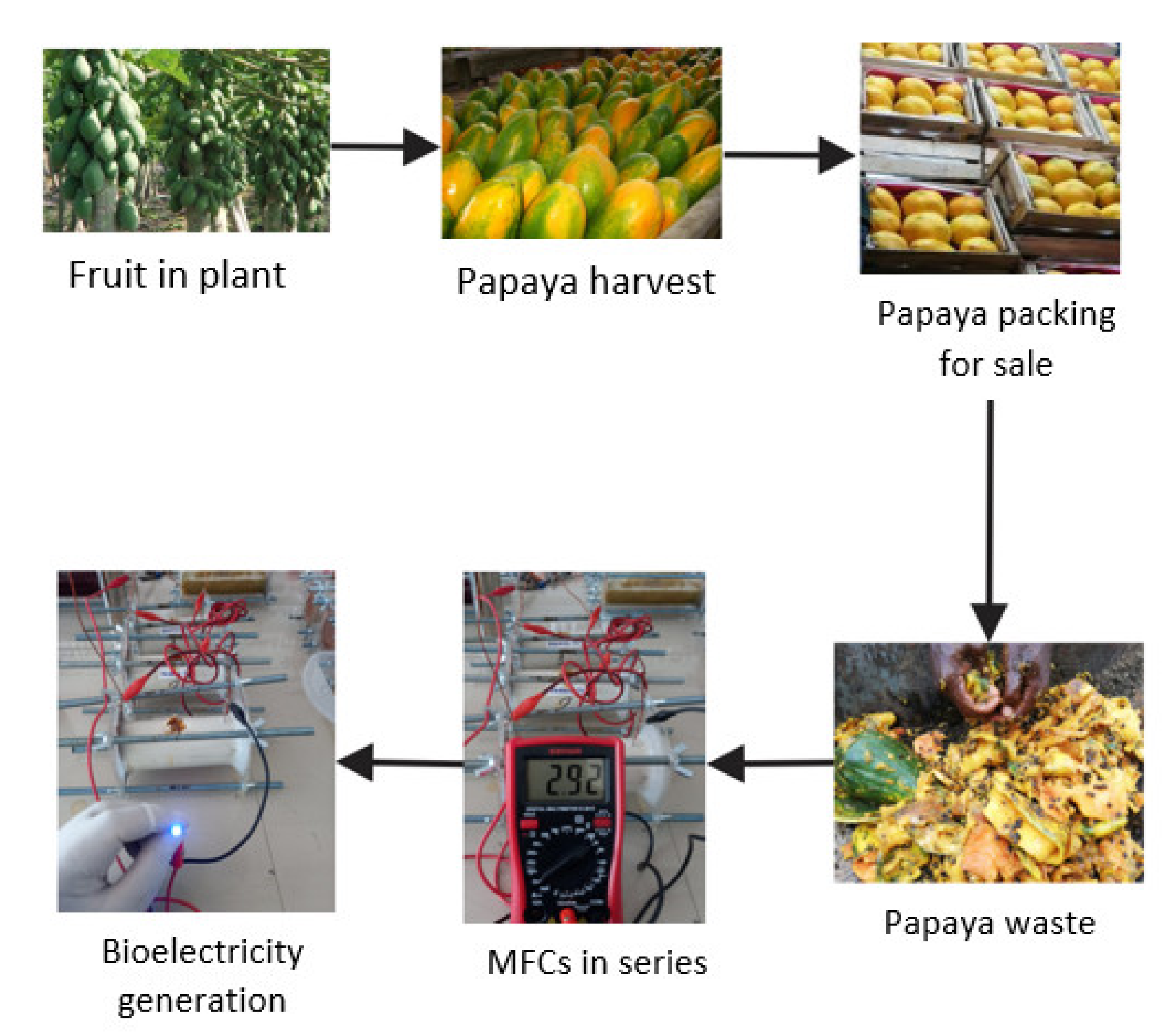
2.2. Collection and Preparation of Papaya Waste
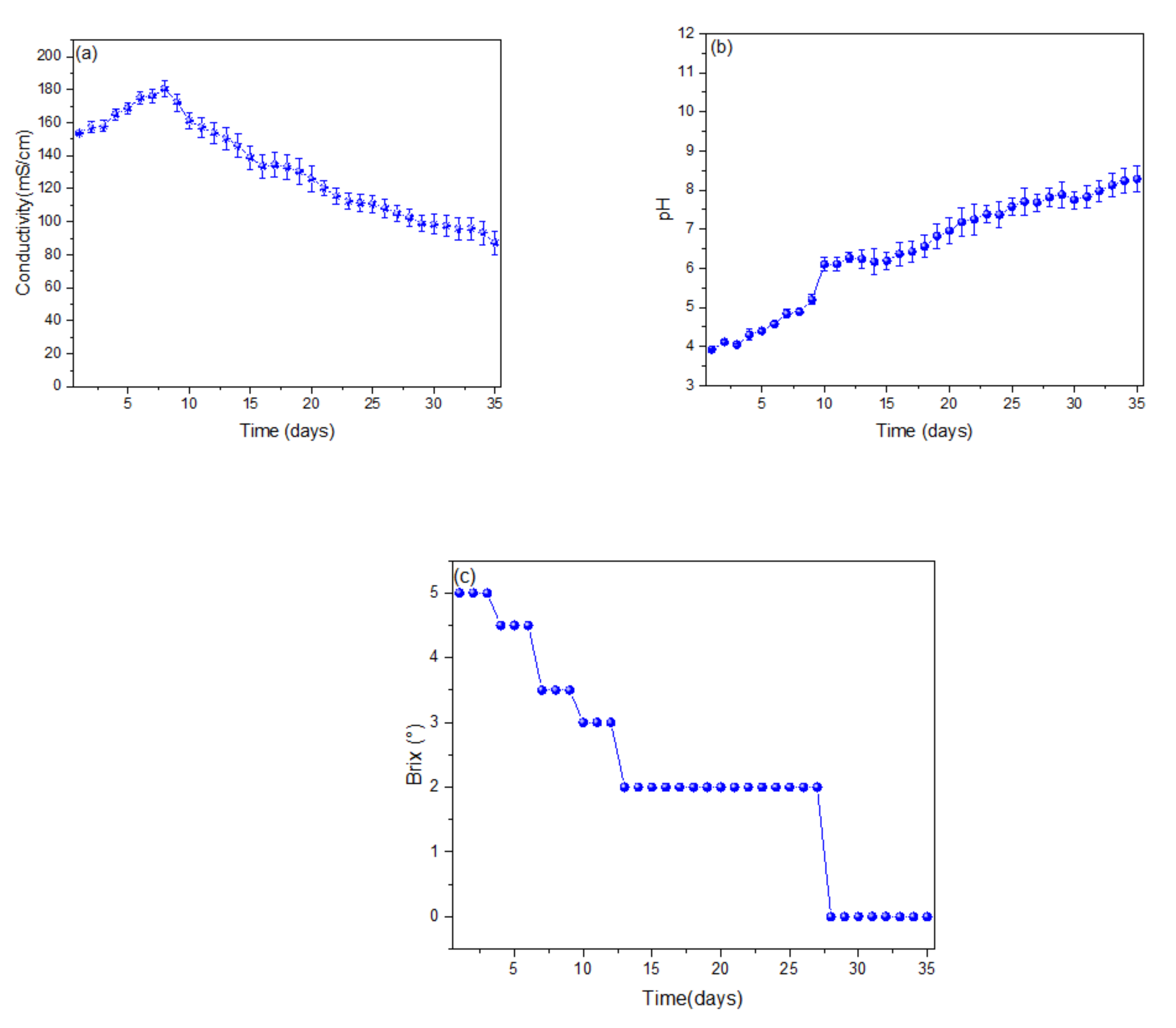
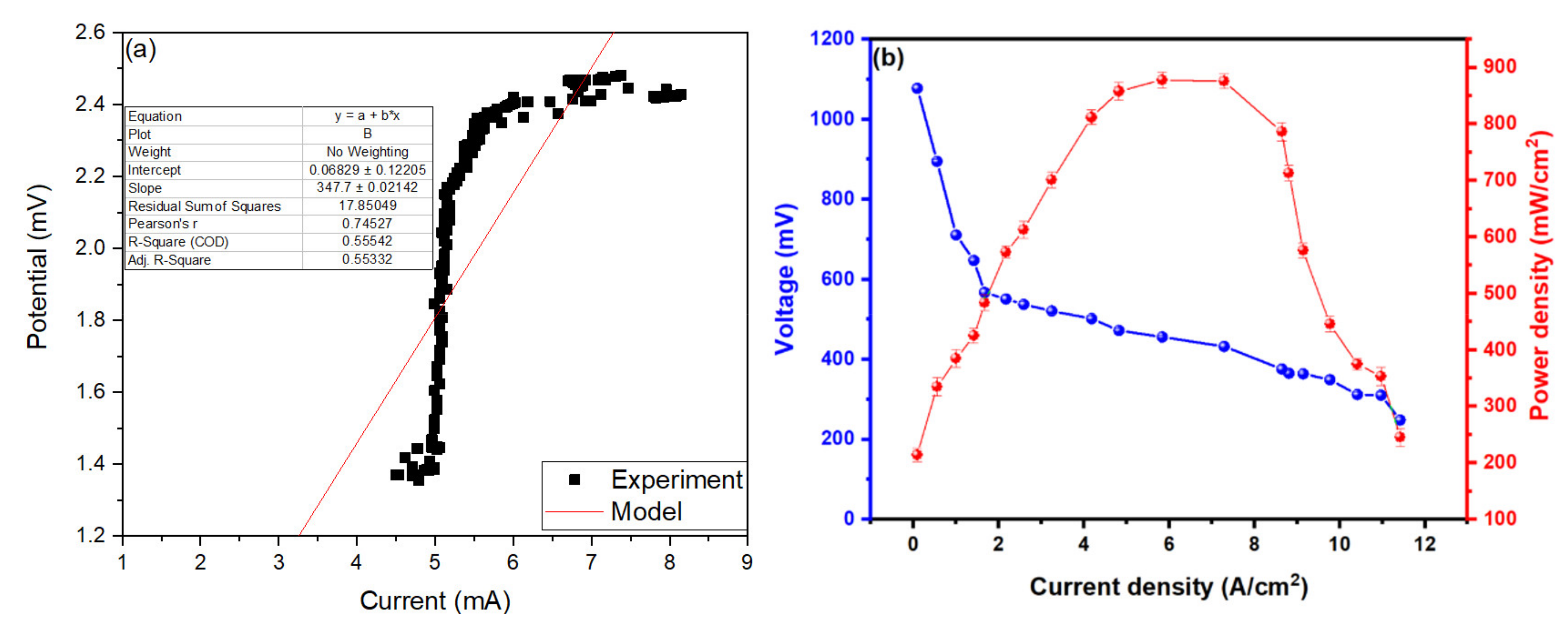
2.3. Characterisation of Microbial Fuel Cells
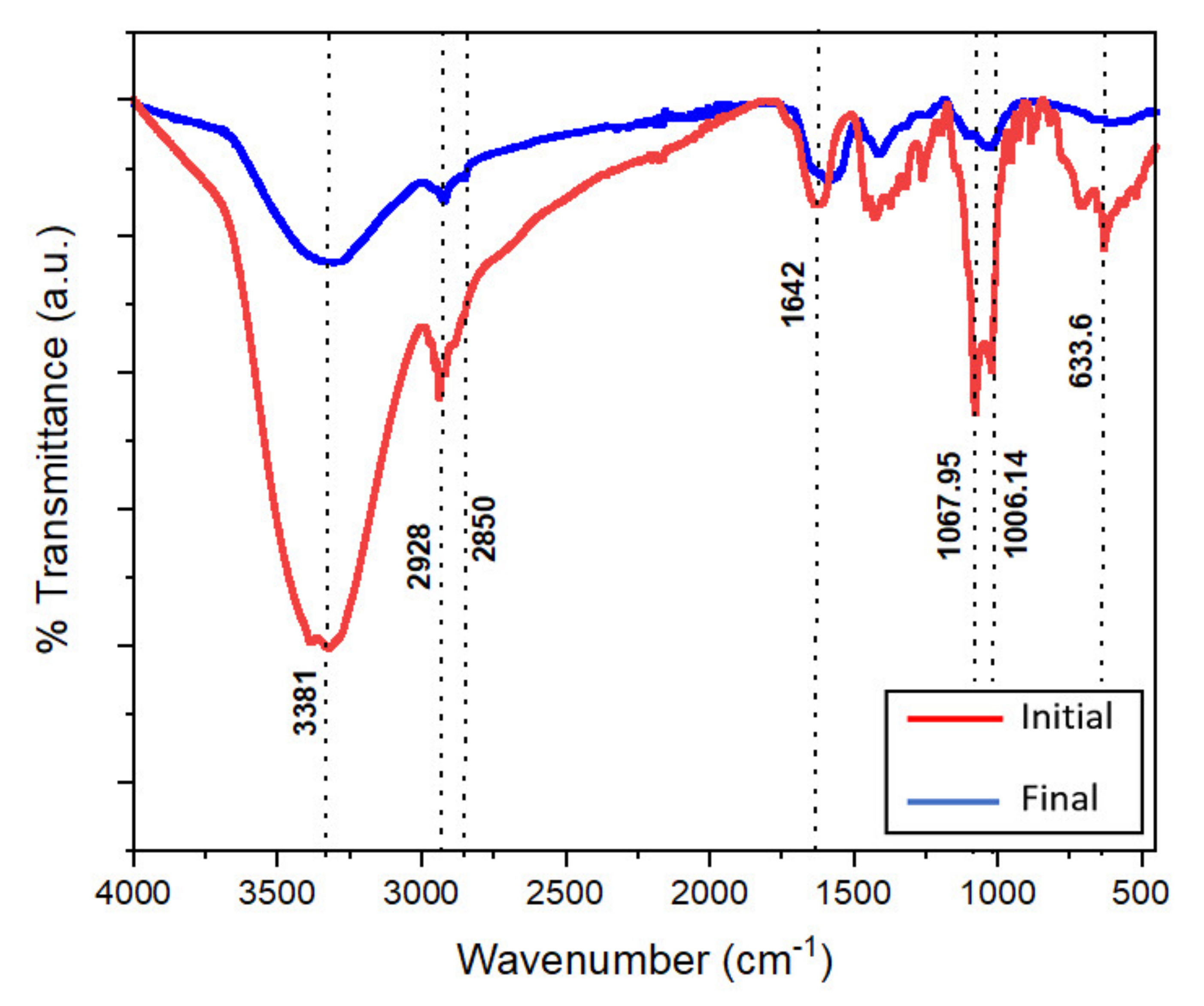
2.4. Isolation of Electrogenic Microorganisms in Anodic Chamber
3. Results
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Hindatu, Y.; Annuar, M.S.M.; Gumel, A.M. Mini-review: Anode modification for improved performance of microbial fuel cell. Renew. Sustain. Energy Rev. 2017, 73, 236–248. [Google Scholar] [CrossRef]
- Abdallah, M.; Feroz, S.; Alani, S.; Sayed, E.T.; Shanableh, A. Continuous and scalable applications of microbial fuel cells: A critical review. Rev. Environ. Sci. Bio/Technol. 2019, 18, 543–578. [Google Scholar] [CrossRef]
- Beyene, H.D.; Werkneh, A.A.; Ambaye, T.G. Current updates on waste to energy (WtE) technologies: A review. Renew. Energy Focus 2018, 24, 1–11. [Google Scholar] [CrossRef]
- Goel, S. From waste to watts in micro-devices: Review on development of Membraned and Membraneless Microfluidic Microbial Fuel Cell. Appl. Mater. Today 2018, 11, 270–279. [Google Scholar] [CrossRef]
- Chatterjee, P.; Ghangrekar, M.M.; Leech, D. A brief review on recent advances in air-cathode microbial fuel cells. Environ. Eng. Manag. J. EEMJ 2018, 17, 1531–1544. [Google Scholar]
- Dwivedi, M.K.; Sonter, S.; Mishra, S.; Patel, D.K.; Singh, P.K. Antioxidant, antibacterial activity, and phytochemical characterization of Carica papaya flowers. Beni-Suef Univ. J. Basic Appl. Sci. 2020, 9, 1–11. [Google Scholar] [CrossRef]
- Singh, S.P.; Kumar, S.; Mathan, S.V.; Tomar, M.S.; Singh, R.K.; Verma, P.K.; Acharya, A. Therapeutic application of Carica papaya leaf extract in the management of human diseases. DARU J. Pharm. Sci. 2020, 28, 735–744. [Google Scholar] [CrossRef]
- Zainal-Abidin, R.A.; Ruhaizat-Ooi, I.H.; Harun, S. A Review of Omics Technologies and Bioinformatics to Accelerate Improvement of Papaya Traits. Agronomy 2021, 11, 1356. [Google Scholar] [CrossRef]
- Zhou, Z.; Ford, R.; Bar, I.; Kanchana-Udomkan, C. Papaya (Carica papaya L.) Flavour Profiling. Genes 2021, 12, 1416. [Google Scholar] [CrossRef]
- Joymak, W.; Ngamukote, S.; Chantarasinlapin, P.; Adisakwattana, S. Unripe Papaya By-Product: From Food Wastes to Functional Ingredients in Pancakes. Foods 2021, 10, 615. [Google Scholar] [CrossRef]
- Oladipo, B.; Ojumu, T.V.; Latinwo, L.M.; Betiku, E. Pawpaw (Carica papaya) Peel Waste as a Novel Green Heterogeneous Catalyst for Moringa Oil Methyl Esters Synthesis: Process Optimization and Kinetic Study. Energies 2020, 13, 5834. [Google Scholar] [CrossRef]
- Pap, S.; Knudsen, T.Š.; Radonić, J.; Maletić, S.; Igić, S.M.; Sekulić, M. Utilization of fruit processing industry waste as green activated carbon for the treatment of heavy metals and chlorophenols contaminated water. J. Clean. Prod. 2017, 162, 958–972. [Google Scholar] [CrossRef] [Green Version]
- Rahman, W.; Yusup, S.; Mohammad, S.A. Screening of fruit waste as substrate for microbial fuel cell (MFC). In AIP Conference Proceedings; American Institute of Physics Inc.: College Park, MD, USA, 2021; Volume 2332. [Google Scholar] [CrossRef]
- Ghazali, N.F.; Mahmood, N.A.N.; Abu Bakar, N.F.; Ibrahim, K.A. Temperature dependence of power generation of empty fruit bunch (EFB) based microbial fuel cell. Malays. J. Fundam. Appl. Sci. 2019, 15, 489–491. [Google Scholar] [CrossRef]
- Kebaili, H.; Kameche, M.; Innocent, C.; Ziane, F.Z.; Sabeur, S.A.; Sahraoui, T.; Charef, M.A. Treatment of fruit waste leachate using microbial fuel cell: Preservation of agricultural environment. Acta Ecol. Sin. 2021, 41, 97–105. [Google Scholar] [CrossRef]
- Rojas-Flores, S.; Noriega, M.D.L.C.; Benites, S.M.; Gonzales, G.A.; Salinas, A.S.; Palacios, F.S. Generation of bioelectricity from fruit waste. Energy Rep. 2020, 6, 37–42. [Google Scholar] [CrossRef]
- Rojas-Flores, S.; Benites, S.M.; La Cruz-Noriega, D.; Cabanillas-Chirinos, L.; Valdiviezo-Dominguez, F.; Álvarez, M.A.Q.; Angelats-Silva, L. Bioelectricity Production from Blueberry Waste. Processes 2021, 9, 1301. [Google Scholar] [CrossRef]
- Gustincich, S.; Manfioletti, G.; Del Sal, G.; Schneider, C.; Carninci, P. A fast method for high-quality genomic DNA extraction from whole human blood. Biotechniques 1991, 11, 298–300. [Google Scholar]
- Valenzuela-González, F.; Casillas-Hernández, R.; Villalpando, E.; Vargas-Albores, F. El gen ARNr 16S en el estudio de comunidades microbianas marinas. Cienc. Mar. 2015, 41, 297–313. [Google Scholar] [CrossRef] [Green Version]
- Programa Online de Identificación de Levadura. Available online: https://apiweb.biomerieux.com/servlet/Identify (accessed on 1 August 2021).
- Asefi, B.; Li, S.L.; Moreno, H.A.; Sanchez-Torres, V.; Hu, A.; Li, J.; Yu, C.P. Characterization of electricity production and microbial community of food waste-fed microbial fuel cells. Process Saf. Environ. Prot. 2019, 125, 83–91. [Google Scholar] [CrossRef]
- Don, C.D.Y.A.; Babel, S. Effects of organic loading on bioelectricity and micro-algal biomass production in microbial fuel cells using synthetic wastewater. J. Water Process. Eng. 2021, 39, 101699. [Google Scholar] [CrossRef]
- Liu, X.; Zhu, F.; Zhang, R.; Zhao, L.; Qi, J. Recent progress on biodiesel production from municipal sewage sludge. Renew. Sustain. Energy Rev. 2021, 135, 110260. [Google Scholar] [CrossRef]
- Kondaveeti, S.; Mohanakrishna, G.; Kumar, A.; Lai, C.; Lee, J.K.; Kalia, V.C. Exploitation of citrus peel extract as a feedstock for power generation in Microbial Fuel Cell (MFC). Indian J. Microbiol. 2019, 59, 476–481. [Google Scholar] [CrossRef]
- Mbugua, J.K.; Mbui, D.N.; Mwaniki, J.; Mwaura, F.; Sheriff, S. Influence of Substrate Proximate Properties on Voltage Production in Microbial Fuel Cells. J. Sustain. Bioenergy Syst. 2020, 10, 43. [Google Scholar] [CrossRef]
- Toding, O.S.L.; Virginia, C.; Suhartini, S. Conversion banana and orange peel waste into electricity using microbial fuel cell. In IOP Conference Series: Earth and Environmental Science; IOP Publishing: Bristol, UK, 2018; Volume 209, p. 012049. [Google Scholar] [CrossRef]
- Yang, N.; Zhan, G.; Li, D.; Wang, X.; He, X.; Liu, H. Complete nitrogen removal and electricity production in Thauera-dominated air-cathode single chambered microbial fuel cell. Chem. Eng. J. 2019, 356, 506–515. [Google Scholar] [CrossRef]
- Khan, A.M.; Hussain, M.S. Conversion of Wastes to Bioelectricity, Bioethanol, and Fertilizer: Khan and Hussain. Water Environ. Res. 2017, 89, 676–686. [Google Scholar] [CrossRef]
- Jung, S.P.; Kim, E.; Koo, B. Effects of wire-type and mesh-type anode current collectors on performance and electrochemistry of microbial fuel cells. Chemosphere 2018, 209, 542–550. [Google Scholar] [CrossRef]
- Stefanova, A.; Angelov, A.; Bratkova, S.; Genova, P.; Nikolova, K. Influence of electrical conductivity and temperature in a microbial fuel cell for treatment of mining waste water. Ann. Constantin Brâncuși Univ. Târgu Jiu Lett. Soc. Sci. Ser. 2018, 3, 18–24. [Google Scholar] [CrossRef]
- Gandu, B.; Rozenfeld, S.; Hirsch, L.O.; Schechter, A.; Cahan, R. Enhancement of Electrochemical Activity in Bioelectrochemical Systems by Using Bacterial Anodes: An Overview. Bioelectrochem. Syst. 2020, 1, 211–238. [Google Scholar] [CrossRef]
- Margaria, V.; Tommasi, T.; Pentassuglia, S.; Agostino, V.; Sacco, A.; Armato, C.; Quaglio, M. Effects of pH variations on anodic marine consortia in a dual chamber microbial fuel cell. Int. J. Hydrogen Energy 2017, 42, 1820–1829. [Google Scholar] [CrossRef]
- Mani, P.; Keshavarz, T.; Chandra, T.S.; Kyazze, G. Decolourisation of Acid orange 7 in a microbial fuel cell with a laccase-based biocathode: Influence of mitigating pH changes in the cathode chamber. Enzym. Microb. Technol. 2017, 96, 170–176. [Google Scholar] [CrossRef] [Green Version]
- Shanmuganathan, P.; Rajasulochana, P.; Murthy, A.R. Treatment of Wastewater Using MFC. PalArch’s J. Archaeol. Egypt Egyptol. 2020, 17, 4856–4567. [Google Scholar]
- Hernández, E.; Carlos, N.; Inostroza, L.; Bautista, N.; Byrne, R.; Alencastre, A.; Sueros, S. Evaluación química y tecnológico-nutricional de “papaya de altura” (carica pubescens). Cienc. Investig. 2014, 17, 88–91. [Google Scholar] [CrossRef]
- Arkatkar, A.; Mungray, A.K.; Sharma, P. Effect of microbial growth on internal resistances in MFC: A case study. In Innovations in Infrastructure; Springer: Singapore, 2019; pp. 469–479. [Google Scholar] [CrossRef]
- Ihesinachi, A.K.; Stephen, I.A. Electricity Generation from Waste Tropical Fruits-Watermelon (Citrullus lanatus) and Paw-paw (Carica papaya) using Single Chamber Microbial Fuel Cells. Int. J. Energy Inf. Commun. 2020, 10, 1–7. [Google Scholar] [CrossRef]
- Flores, S.J.R.; Benites, S.M.; Rosa, A.L.R.A.L.; Zoilita, A.L.Z.A.L.; Luis, A.S.L. The Using Lime (Citrus× aurantiifolia), Orange (Citrus× sinensis), and Tangerine (Citrus reticulata) Waste as a Substrate for Generating Bioelectricity: Using lime (Citrus× aurantiifolia), orange (Citrus× sinensis), and tangerine (Citrus reticulata) waste as a substrate for generating bioelectricity. Environ. Res. Eng. Manag. 2020, 76, 24–34. [Google Scholar] [CrossRef]
- Ihesinachi, K.A.; Lois, N.N.; Stephen, A.I. Bioenergy from Waste Paw-Paw Fruits and Peels Using Single Chamber Microbial Fuel Cells. J. Fundam. Renew. Energy Appl. 2020, 8, 1–5. [Google Scholar] [CrossRef]
- Sharma, N.; Khajuria, Y.; Singh, V.K.; Kumar, S.; Lee, Y.; Rai, P.K.; Singha, V.K. Study of Molecular and Elemental Changes in Nematode-infested Roots in Papaya Plant Using FTIR, LIBS and WDXRF Spectroscopy. At. Spectrosc. 2020, 41, 110–118. [Google Scholar] [CrossRef]
- Jain, D.; Daima, H.K.; Kachhwaha, S.; Kothari, S.L. Synthesis of plant-mediated silver nanoparticles using papaya fruit extract and evaluation of their anti microbial activities. Dig. J. Nanomater. Biostruct. 2009, 4, 557–563. [Google Scholar]
- Haq, Q.M.; Mabood, F.; Naureen, Z.; Al-Harrasi, A.; Gilani, S.A.; Hussain, J.; Al-Bahaisi, I.M. Application of reflectance spectroscopies (FTIR-ATR & FT-NIR) coupled with multivariate methods for robust in vivo detection of begomovirus infection in papaya leaves. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2018, 198, 27–32. [Google Scholar] [CrossRef]
- Moharir, P.V.; Tembhurkar, A.R. Effect of recirculation on bioelectricity generation using microbial fuel cell with food waste leachate as substrate. Int. J. Hydrogen Energy 2018, 43, 10061–10069. [Google Scholar] [CrossRef]
- Kale, R.; Barwar, S.; Kane, P.; More, S. Green synthesis of silver nanoparticles using papaya seed and its characterization. Int. J. Res. Appl. Sci. Eng. Technol. 2018, 6, 168–174. [Google Scholar] [CrossRef]
- Cholassery, S.; Krishna, V.; Sethuraj, S.; Rehina, S.S.; Ranganathan, V.; Dileep, L.C.; Chandran, R.P. Analysis of physicochemical and sensory parameters of wine produced from Carica papaya. J. Appl. Biol. Biotechnol. 2019, 7, 74–78. [Google Scholar] [CrossRef] [Green Version]
- Sakata, T.; Nishitani, S.; Kajisa, T. Molecularly imprinted polymer-based bioelectrical interfaces with intrinsic molecular charges. RSC Adv. 2020, 10, 16999–17013. [Google Scholar] [CrossRef]
- Barer, M.R.; Irving, W. Medical Microbiology E-Book: A Guide to Microbial Infections; Elsevier Health Sciences: Amsterdam, The Netherlands, 2018. [Google Scholar]
- Xue, H.; Zhou, P.; Huang, L.; Quan, X.; Yuan, J. Cathodic Cr (VI) reduction by electrochemically active bacteria sensed by fluorescent probe. Sens. Actuators B Chem. 2017, 243, 303–310. [Google Scholar] [CrossRef]
- Amoureux, L.; Bador, J.; Fardeheb, S.; Mabille, C.; Couchot, C.; Massip, C.; Neuwirth, C. Detection of Achromobacter xylosoxidans in hospital, domestic, and outdoor environmental samples and comparison with human clinical isolates. Appl. Environ. Microbiol. 2013, 79, 7142–7149. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Aftab, S.; Shah, A.; Nisar, J.; Ashiq, M.N.; Akhter, M.S.; Shah, A.H. Marketability Prospects of Microbial Fuel Cells for Sustainable Energy Generation. Energy Fuels 2020, 34, 9108–9136. [Google Scholar] [CrossRef]
- Hirose, A.; Kasai, T.; Aoki, M.; Umemura, T.; Watanabe, K.; Kouzuma, A. Electrochemically active bacteria sense electrode potentials for regulating catabolic pathways. Nat. Commun. 2018, 9, 1083. [Google Scholar] [CrossRef]
- Zafar, Z.; Ayaz, K.; Nasir, M.H.; Yousaf, S.; Sharafat, I.; Ali, N. Electrochemical performance of biocathode microbial fuel cells using petroleum-contaminated soil and hot water spring. Int. J. Environ. Sci. Technol. 2019, 16, 1487–1500. [Google Scholar] [CrossRef]


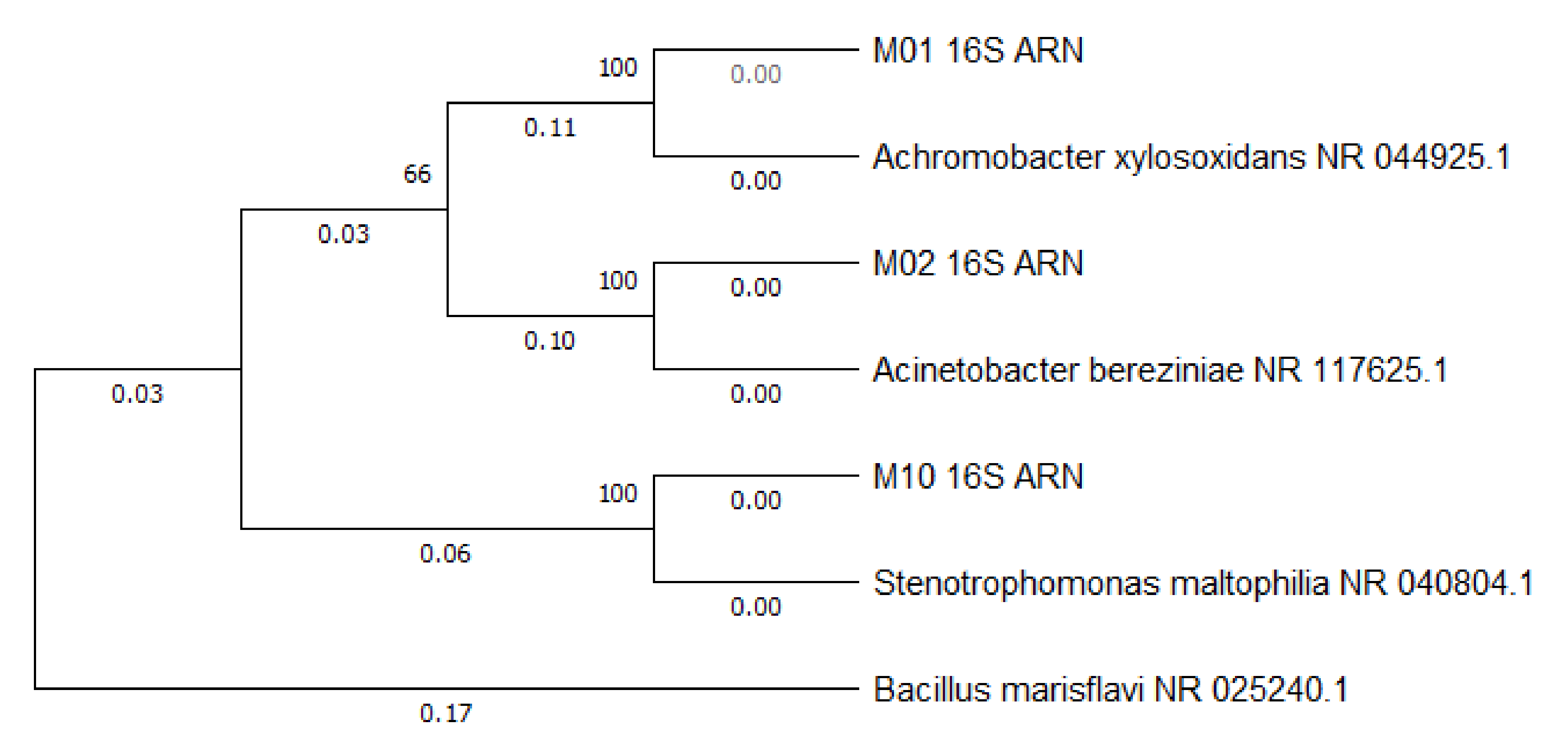
| BLAST Characterisation | Sequence Length (nt) | Maximum Identity % | Identification Number | Phylogeny |
|---|---|---|---|---|
| Achromobacter xylosoxidans | 1451 | 99.32% | CP053617.1 | Cellular organisms; Bacteria; Proteobacteria; Betaproteobacteria; Burkholderiales; Alcaligenaceae; Achromobacter |
| Acinetobacter bereziniae | 1468 | 99.93% | CP018259.1 | Cellular organisms; Bacteria; Proteobacteria; Gammaproteobacteria; Pseudomonadales; Moraxellaceae; Acinetobacter |
| Stenotrophomonas maltophilia | 1477 | 100.00% | NR_041577.1 | Cellular organisms; Bacteria; Proteobacteria; Gammaproteobacteria; Xanthomonadales; Xanthomonadaceae; Stenotrophomonas; Stenotrophomonas maltophilia group |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rojas-Flores, S.; Pérez-Delgado, O.; Nazario-Naveda, R.; Rojales-Alfaro, H.; Benites, S.M.; De La Cruz-Noriega, M.; Otiniano, N.M. Potential Use of Papaya Waste as a Fuel for Bioelectricity Generation. Processes 2021, 9, 1799. https://doi.org/10.3390/pr9101799
Rojas-Flores S, Pérez-Delgado O, Nazario-Naveda R, Rojales-Alfaro H, Benites SM, De La Cruz-Noriega M, Otiniano NM. Potential Use of Papaya Waste as a Fuel for Bioelectricity Generation. Processes. 2021; 9(10):1799. https://doi.org/10.3390/pr9101799
Chicago/Turabian StyleRojas-Flores, Segundo, Orlando Pérez-Delgado, Renny Nazario-Naveda, Henry Rojales-Alfaro, Santiago M. Benites, Magaly De La Cruz-Noriega, and Nélida Milly Otiniano. 2021. "Potential Use of Papaya Waste as a Fuel for Bioelectricity Generation" Processes 9, no. 10: 1799. https://doi.org/10.3390/pr9101799
APA StyleRojas-Flores, S., Pérez-Delgado, O., Nazario-Naveda, R., Rojales-Alfaro, H., Benites, S. M., De La Cruz-Noriega, M., & Otiniano, N. M. (2021). Potential Use of Papaya Waste as a Fuel for Bioelectricity Generation. Processes, 9(10), 1799. https://doi.org/10.3390/pr9101799






