Phosphate and Ammonium Removal from Water through Electrochemical and Chemical Precipitation of Struvite
Abstract
1. Introduction
2. Materials and Methods
2.1. Water Solutions
2.2. Chemical Equilibrium Calculations
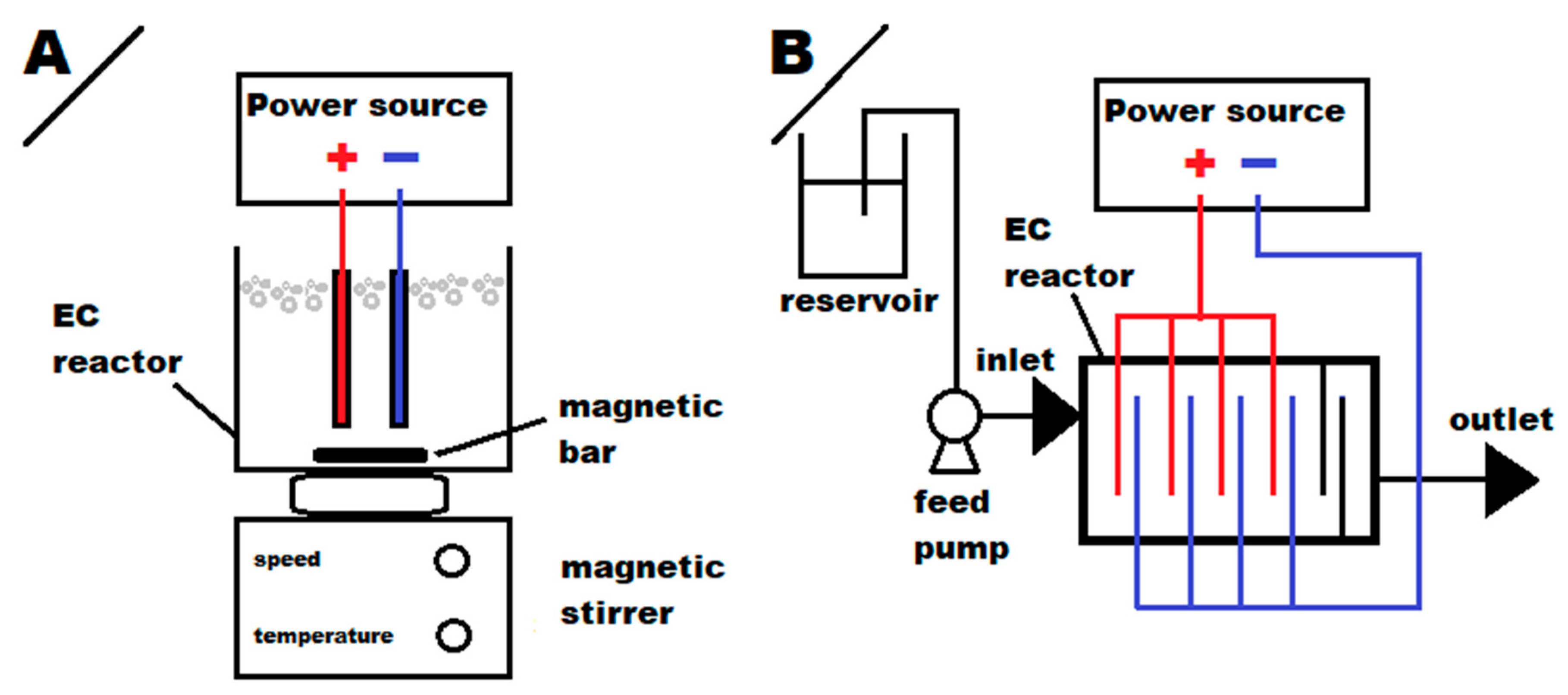
2.3. Electrochemical Precipitation and Coagulation Experiments
2.3.1. Batch EC Process
2.3.2. Continuous EC Process
2.3.3. Chemical Precipitation
2.4. Characterization of Precipitate Samples
2.5. Economic Analysis
3. Results and Discussion
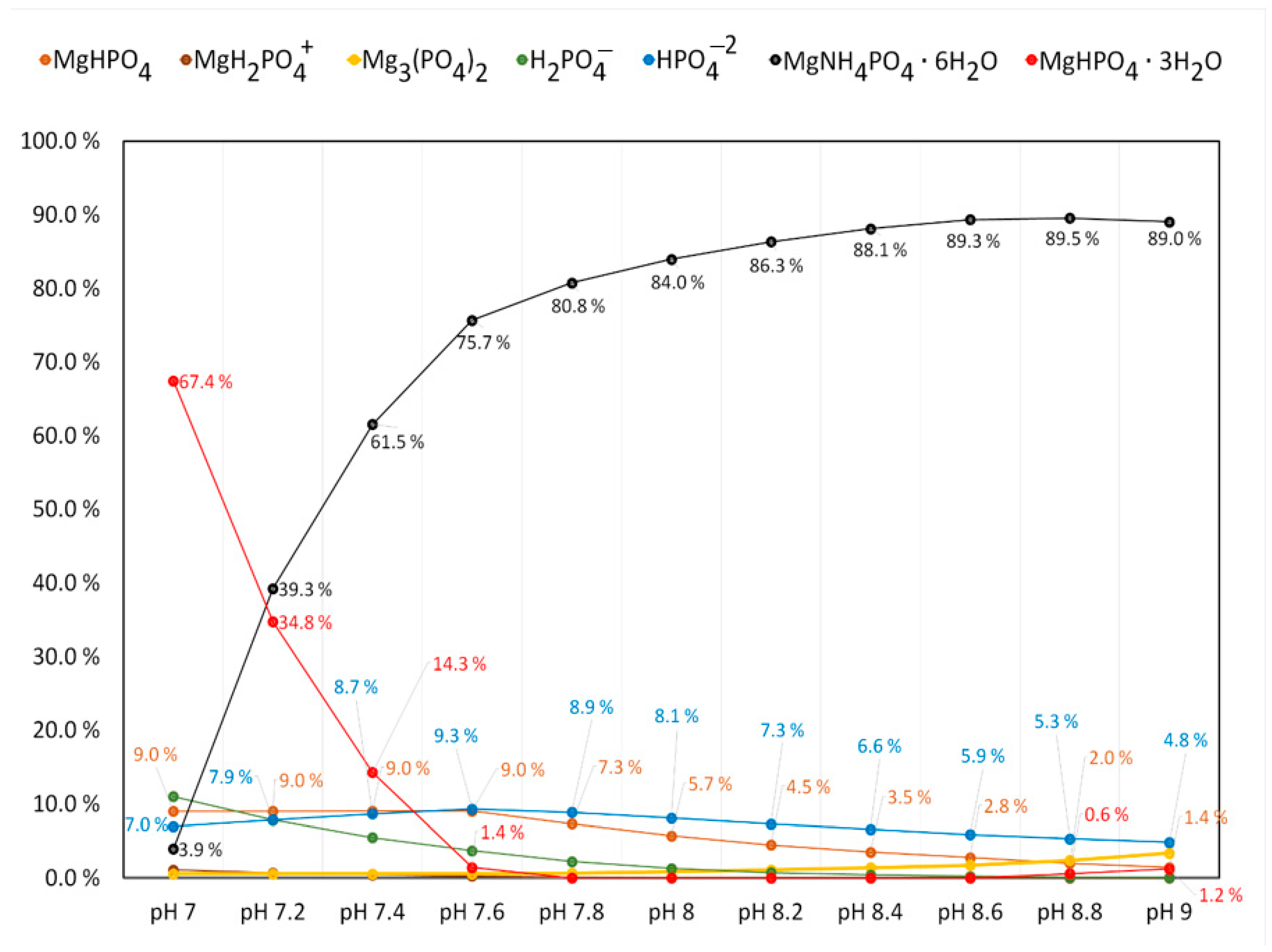
3.1. Effect of pH for Struvite Precipitation
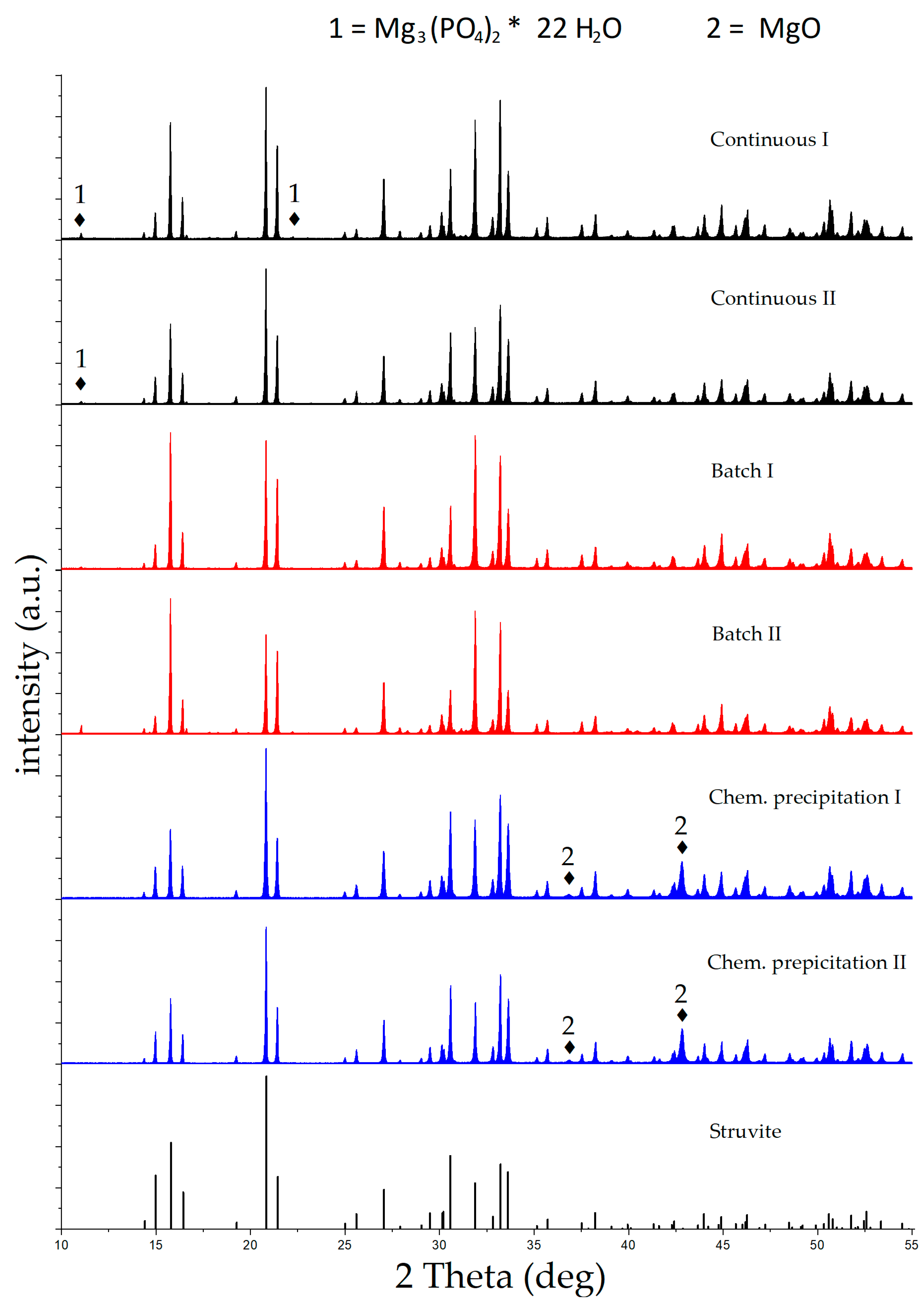
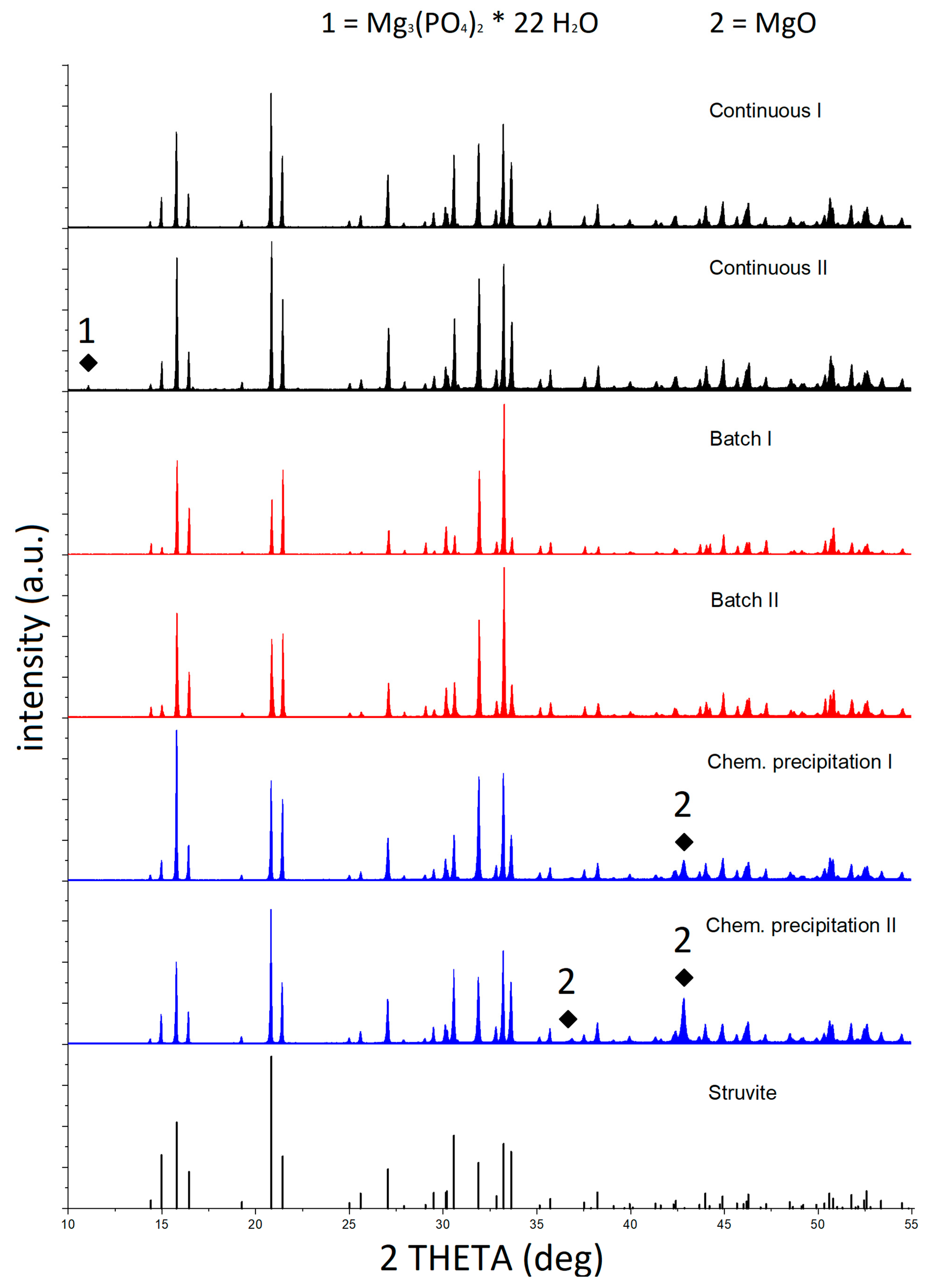
3.2. Purity of Produced Struvite
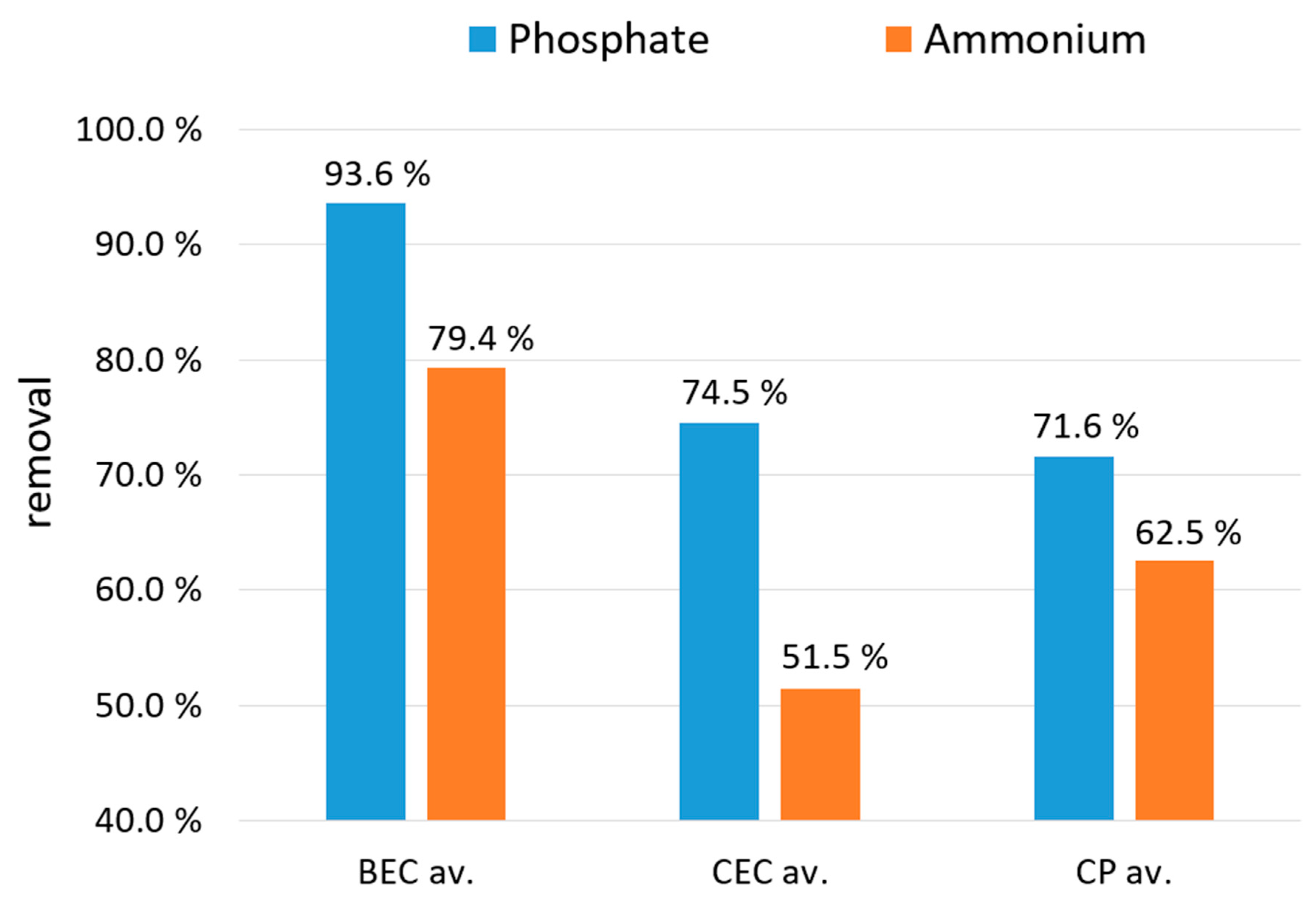
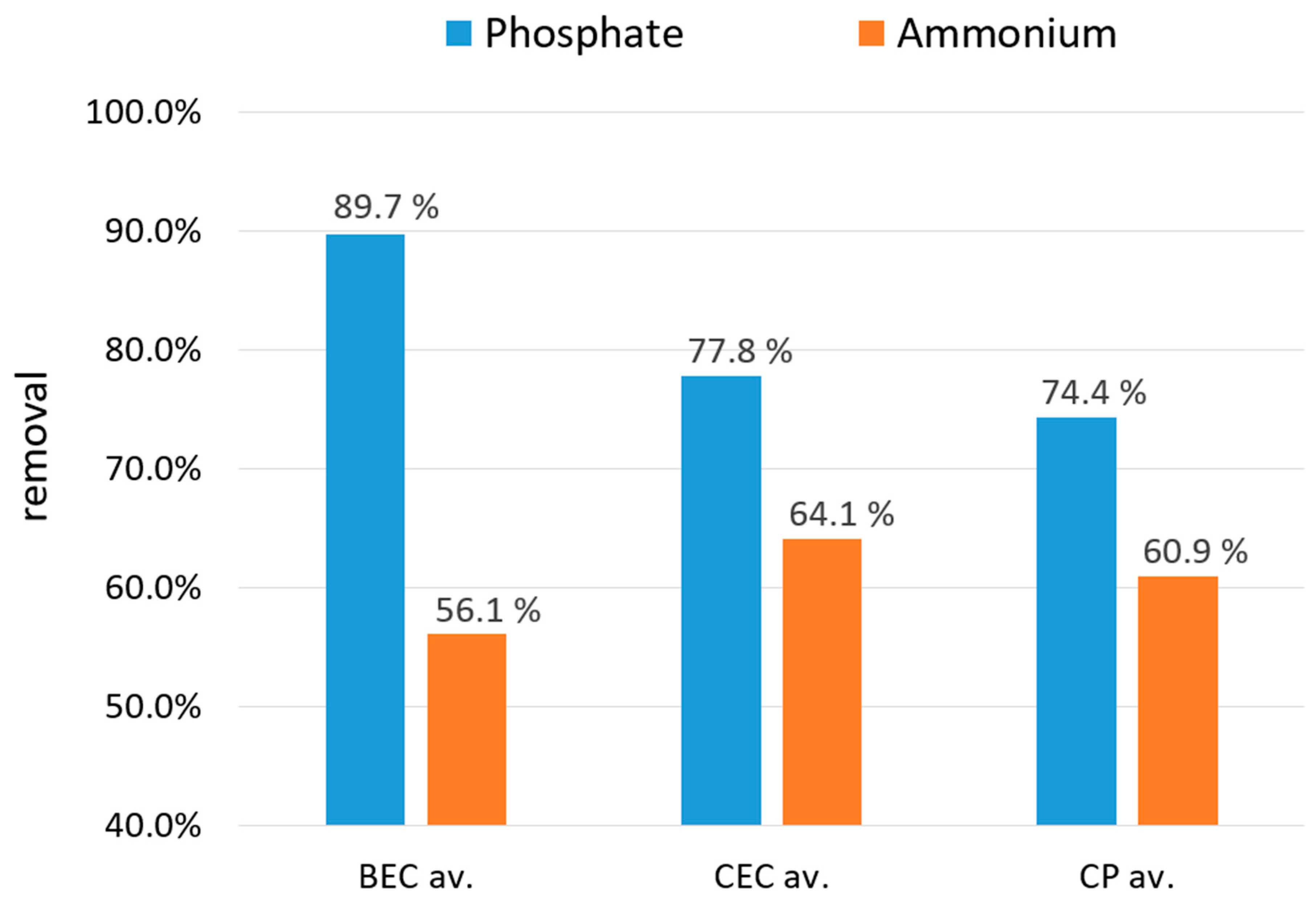
3.3. Phosphate and Ammonium Removal from Water Solutions
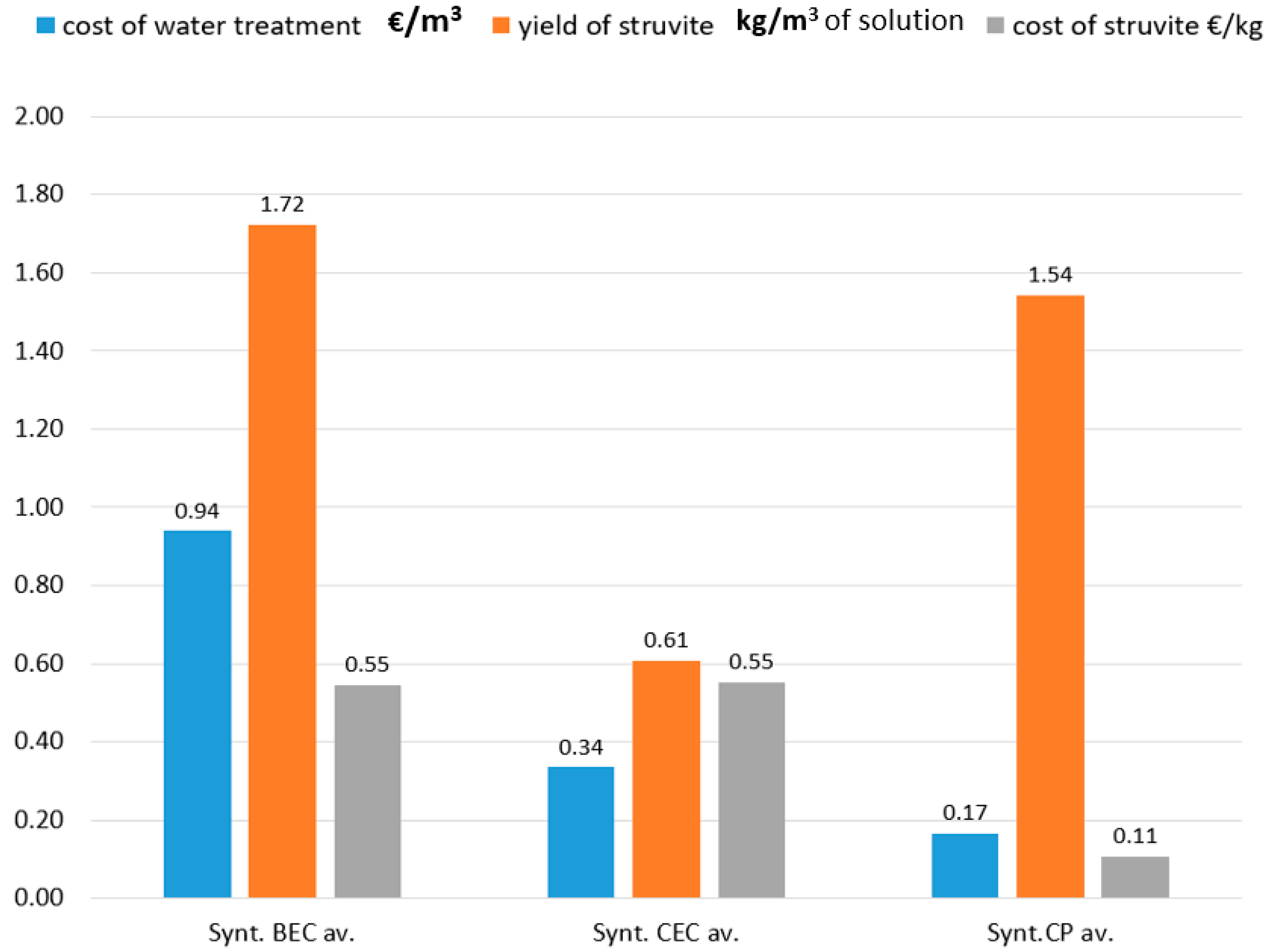
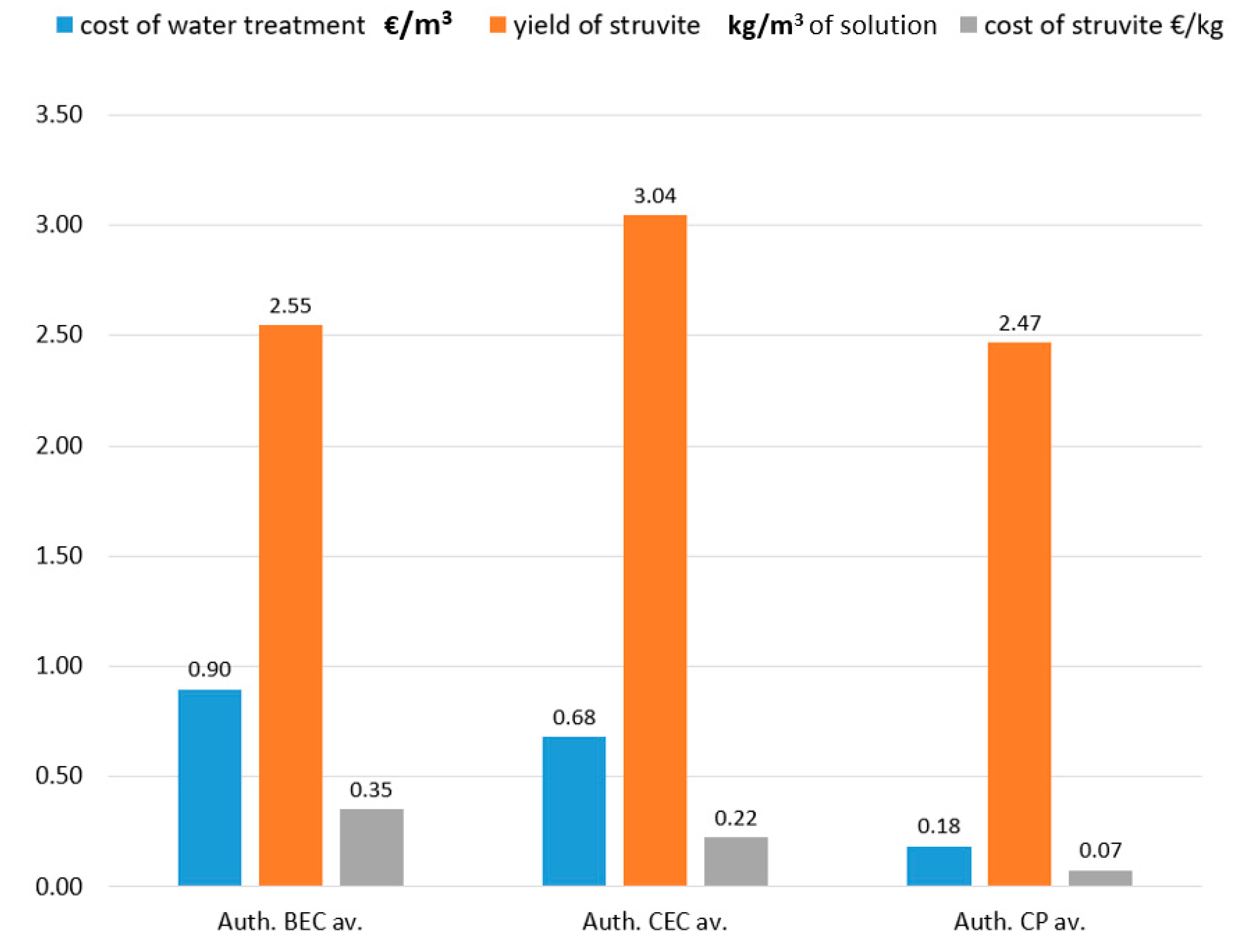
3.4. Economy Aspect of BEC, CEC, and CP
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- WHO; UNICEF. Progress on Drinking Water, Sanitation and Hygiene: 2017 Update and SDG Baselines; United Nations Children’s Fund (UNICEF): New York, NY, USA; World Health Organization (WHO): Geneva, Switzerland, 2017. [Google Scholar]
- UN GENERAL ASSEMBLY. Resolution on the Human Right to Water and Sanitation, A/RES/64/292. 2010. Available online: https://undocs.org/A/RES/64/292 (accessed on 19 April 2020).
- FAO. World Fertilizer Trends and Outlook to 2022; FAO: Rome, Italy, 2019. [Google Scholar]
- Mekonnen, M.M.; Hoekstra, A.Y. Global Anthropogenic Phosphorus Loads to Freshwater and Associated Grey Water Footprints and Water Pollution Levels: A High-Resolution Global Study. Water Resour. Res. 2018, 54, 345–358. [Google Scholar] [CrossRef]
- Talboys, P.J.; Heppell, J.; Roose, T.; Healey, J.R.; Jones, D.L.; Withers, P.J.A. Struvite: A slow-release fertiliser for sustainable phosphorus management? Plant Soil 2016, 401, 109–123. [Google Scholar] [CrossRef] [PubMed]
- Yuan, Z.; Jiang, S.; Sheng, H.; Liu, X.; Hua, H.; Liu, X.; Zhang, Y. Human Perturbation of the Global Phosphorus Cycle: Changes and Consequences. Environ. Sci. Technol. 2018, 52, 2438–2450. [Google Scholar] [CrossRef]
- Fowler, D.; Coyle, M.; Vitousek, P.; Leach, A.; Bouwman, A.F.; Butterbach-Bahl, K.; Dentener, F.; Stevenson, D.; Amann, M.; Voss, M.; et al. The global nitrogen cycle in the twenty-first century. Philos. Trans. R. Soc. B Biol. Sci. 2013, 368, 20130164. [Google Scholar] [CrossRef]
- Scholz, R.W.; Ulrich, A.E.; Eilittä, M.; Roy, A. Sustainable use of phosphorus: A finite resource. Sci. Total Environ. 2013, 461–462, 799–803. [Google Scholar] [CrossRef]
- Erisman, J.W.; Galloway, J.; Seitzinger, S.; Bleeker, A.; Butterbach-Bahl, K. Reactive nitrogen in the environment and its effect on climate change. Curr. Opin. Environ. Sustain. 2011, 3, 281–290. [Google Scholar] [CrossRef]
- Sutton, M.A.; Oenema, O.; Erisman, J.W.; Leip, A.; Van Grinsven, H.; Winiwarter, W. Too much of a good thing. Nature 2011, 472, 159–161. [Google Scholar] [CrossRef]
- UNEP AND WHRC. Reactive Nitrogen in the Environment: Too Much or too Little of a Good Thing; United Nations Environment Programme: Paris, France, 2007; ISBN 978 92 807 2783 8. [Google Scholar]
- Bolan, N.; Wong, L.; Adriano, D. Nutrient removal from farm effluents. Bioresour. Technol. 2004, 94, 251–260. [Google Scholar] [CrossRef]
- Yin, H.; Kong, M. Simultaneous removal of ammonium and phosphate from eutrophic waters using natural calcium-rich attapulgite-based versatile adsorbent. Desalination 2014, 351, 128–137. [Google Scholar] [CrossRef]
- Burgos-Castillo, R.; Sillanpää, M.; Brillas, E.; Sirés, I. Removal of metals and phosphorus recovery from urban anaerobically digested sludge by electro-Fenton treatment. Sci. Total. Environ. 2018, 644, 173–182. [Google Scholar] [CrossRef]
- Kilpimaa, S.; Runtti, H.; Kangas, T.; Lassi, U.; Kuokkanen, T. Physical activation of carbon residue from biomass gasification: Novel sorbent for the removal of phosphates and nitrates from aqueous solution. J. Ind. Eng. Chem. 2015, 21, 1354–1364. [Google Scholar] [CrossRef]
- Kuokkanen, V.; Rämö, J.; Lassi, U.; Kuokkanen, T. Electrocoagulation treatment of peat bog drainage water containing humic substances. Water Res. 2015, 79, 79–87. [Google Scholar] [CrossRef] [PubMed]
- Kuokkanen, V.; Kuokkanen, T.; Ramo, J.; Lassi, U.; Roininen, J. Removal of phosphate from wastewaters for further utilization using electrocoagulation with hybrid electrodes—Techno-economic studies. J. Water Process. Eng. 2015, 8, e50–e57. [Google Scholar] [CrossRef]
- Ghimire, U.; Jang, M.; Jung, S.P.; Park, D.; Park, S.J.; Yu, H.; Oh, S.-E. Electrochemical Removal of Ammonium Nitrogen and COD of Domestic Wastewater using Platinum Coated Titanium as an Anode Electrode. Energies 2019, 12, 883. [Google Scholar] [CrossRef]
- Huang, H.; Zhang, P.; Zhang, Z.; Liu, J.; Xiao, J.; Gao, F. Simultaneous removal of ammonia nitrogen and recovery of phosphate from swine wastewater by struvite electrochemical precipitation and recycling technology. J. Clean. Prod. 2016, 127, 302–310. [Google Scholar] [CrossRef]
- Kruk, D.J.; Elektorowicz, M.; Oleszkiewicz, J.A. Struvite precipitation and phosphorus removal using magnesium sacrificial anode. Chemosphere 2014, 101, 28–33. [Google Scholar] [CrossRef]
- Mohammadi, A.; Khadir, A.; A. Tehrani, R.M. Optimization of nitrogen removal from an anaerobic digester effluent by electrocoagulation process. J. Environ. Chem. Eng. 2019, 7. [Google Scholar] [CrossRef]
- Rahman, M.; Salleh, M.A.M.; Rashid, U.; Ahsan, A.; Hossain, M.M.; Ra, C.S. Production of slow release crystal fertilizer from wastewaters through struvite crystallization—A review. Arab. J. Chem. 2014, 7, 139–155. [Google Scholar] [CrossRef]
- Song, G.; Atrens, A.; John, D.; Wu, X.; Nairn, J. The anodic dissolution of magnesium in chloride and sulphate solutions. Corros. Sci. 1997, 39, 1981–2004. [Google Scholar] [CrossRef]
- Bektaş, N. Removal of phosphate from aqueous solutions by electro-coagulation. J. Hazard. Mater. 2004, 106, 101–105. [Google Scholar] [CrossRef]
- Hug, A.; Udert, K.M. Struvite precipitation from urine with electrochemical magnesium dosage. Water Res. 2013, 47, 289–299. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.; Fu, R.; Lv, H.; Zhu, G.; Lu, B.; Zhou, Z.; Wu, X.; Chen, H. Phosphate Recovery from Swine Wastewater by a Struvite Precipitation Electrolyzer. Sci. Rep. 2019, 9, 8893. [Google Scholar] [CrossRef] [PubMed]
- Doyle, J.D.; Parsons, S.A. Struvite formation, control and recovery. Water Res. 2002, 36, 3925–3940. [Google Scholar] [CrossRef]
- Rajaniemi, K.; Metsävainio, V.-P.; Kuokkanen, V. Apparatus and Method for Treatment of Various Types of Water and Wastewater Based on Electrocoagulation. Patent FI 127889 2016. [Google Scholar]
- Taylor, A.W.; Frazier, A.W.; Gurney, E.L. Solubility products of magnesium ammonium and magnesium potassium phos-phates. Trans. Faraday Soc. 1963, 59, 1580–1584. [Google Scholar] [CrossRef]
- Rajaniemi, K.; Raulio, M.; Tuomikoski, S.; Lassi, U. Comparison of batch and novel continuous electrocoagulation processes in the treatment of paint industry wash water. DESALINATION Water Treat. 2019, 170, 394–404. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rajaniemi, K.; Hu, T.; Nurmesniemi, E.-T.; Tuomikoski, S.; Lassi, U. Phosphate and Ammonium Removal from Water through Electrochemical and Chemical Precipitation of Struvite. Processes 2021, 9, 150. https://doi.org/10.3390/pr9010150
Rajaniemi K, Hu T, Nurmesniemi E-T, Tuomikoski S, Lassi U. Phosphate and Ammonium Removal from Water through Electrochemical and Chemical Precipitation of Struvite. Processes. 2021; 9(1):150. https://doi.org/10.3390/pr9010150
Chicago/Turabian StyleRajaniemi, Kyösti, Tao Hu, Emma-Tuulia Nurmesniemi, Sari Tuomikoski, and Ulla Lassi. 2021. "Phosphate and Ammonium Removal from Water through Electrochemical and Chemical Precipitation of Struvite" Processes 9, no. 1: 150. https://doi.org/10.3390/pr9010150
APA StyleRajaniemi, K., Hu, T., Nurmesniemi, E.-T., Tuomikoski, S., & Lassi, U. (2021). Phosphate and Ammonium Removal from Water through Electrochemical and Chemical Precipitation of Struvite. Processes, 9(1), 150. https://doi.org/10.3390/pr9010150





