Ameliorative Effect of Heat-Killed Lactobacillus plantarum L.137 and/or Aloe vera against Colitis in Mice
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Experimental Animals
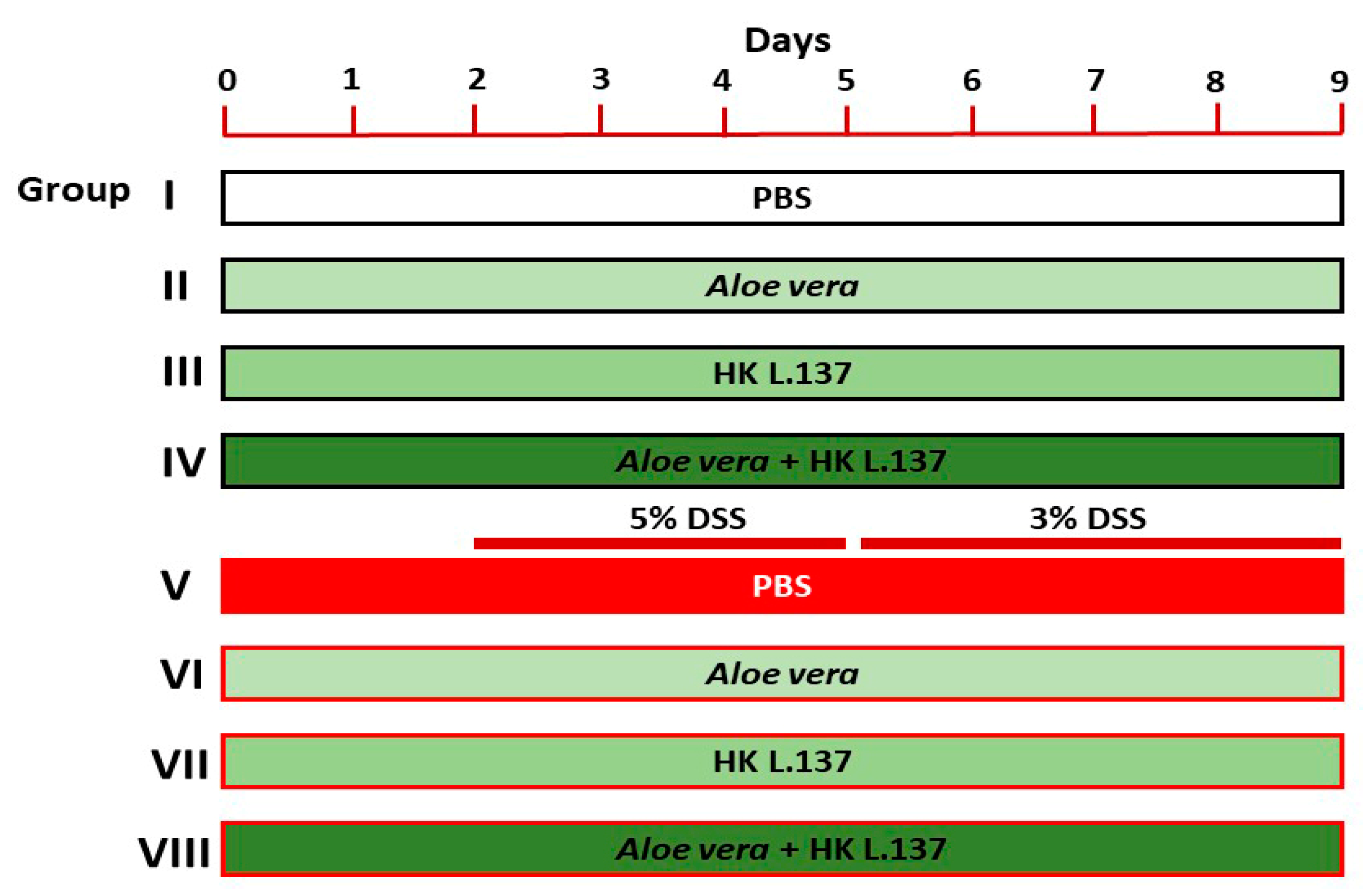
2.3. Experimental Design
- (1)
- Group I (Control): mice received 0.5 mL of phosphate buffer saline (PBS) orally for 9 days.
- (2)
- Group II (Aloe): mice received 200 mg/kg Aloe [24] dissolved in PBS orally for 9 days.
- (3)
- Group III (HK L.137): mice received 100 mg/kg HK L.137 [25] dissolved in PBS orally for 9 days.
- (4)
- Group IV (Aloe/HK L.137): mice received 200 mg/kg Aloe and 100 mg/kg HK L.137 in PBS orally for 9 days.
- (5)
- Group V (DSS): mice received 5% DSS for 3 days and 3% DSS for 4 days.
- (6)
- Group VI (Aloe + DSS): mice received Aloe for 9 days and DSS as in group V.
- (7)
- Group VII (HK L.137 + DSS): mice received HK L.137 for 9 days and DSS as in group V.
- (8)
- Group VIII (Aloe/HK L.137 + DSS): mice received Aloe and HK L.137 for 9 days and DSS as in group V.
2.4. Assessment of Disease Activity Index (DAI)
2.5. Histology
2.6. Histological Score Assessment of Colitis
2.7. Immunohistochemical Expression of NF-κB p65 and iNOS
2.8. Determination of Myeloperoxidase (MPO) and Cytokines
2.9. Statistical Analysis
3. Results
3.1. Effect of Aloe and/or HK L.137 on DAI and Colon Length in DSS-Induced Mice
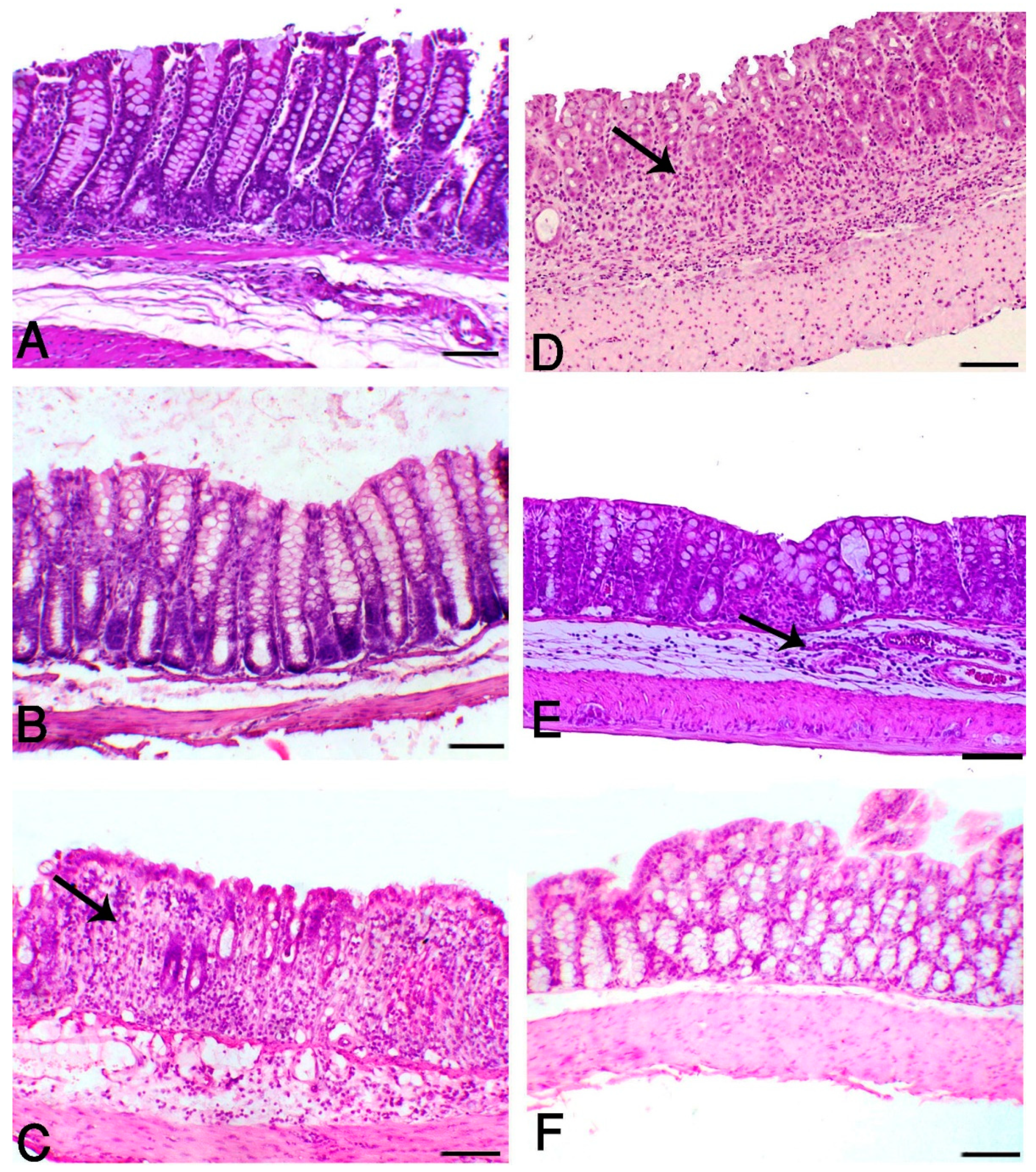
3.2. Aloe and/or HK L.137 Suppress Histopathological Alterations in DSS-Induced Mice
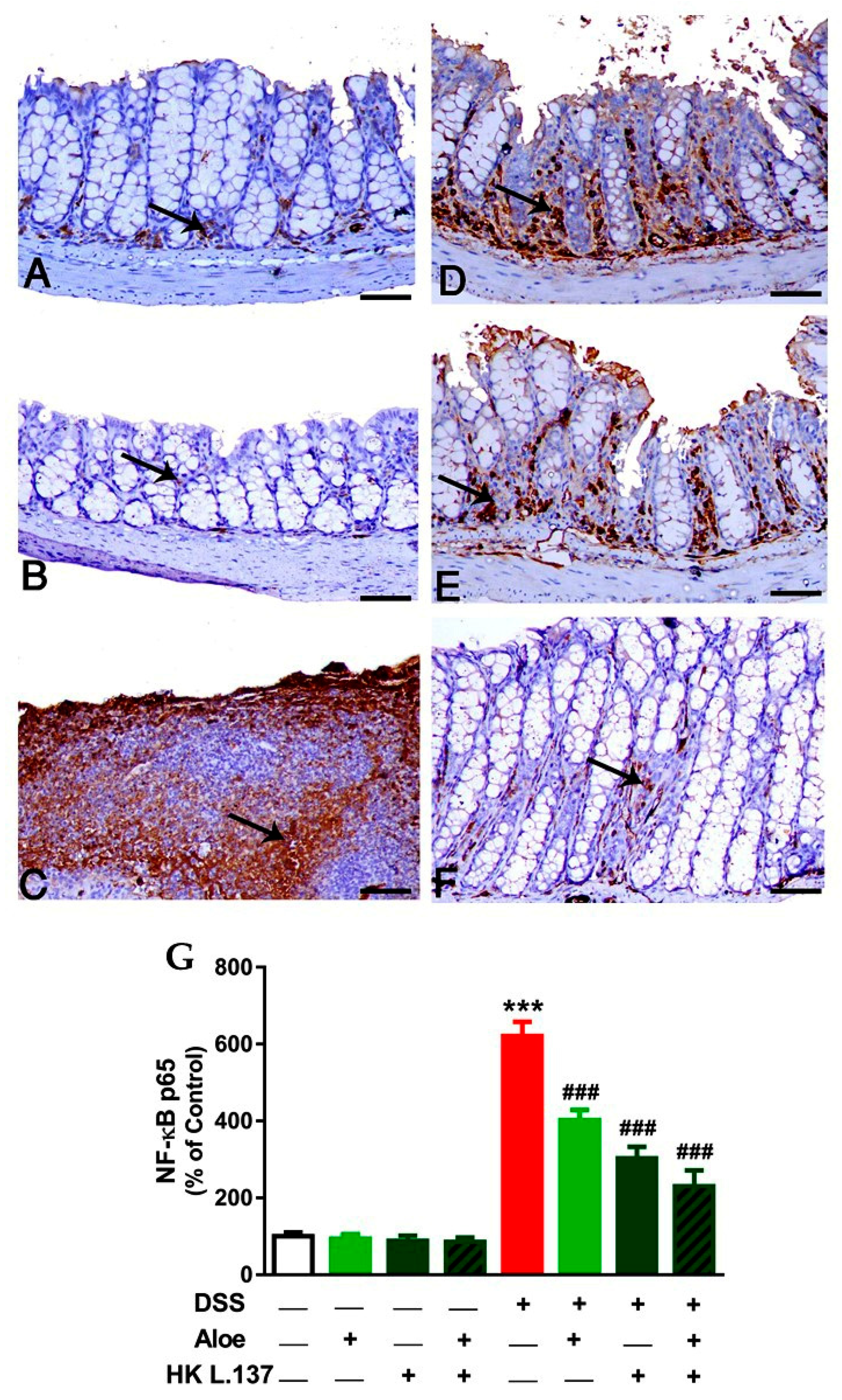
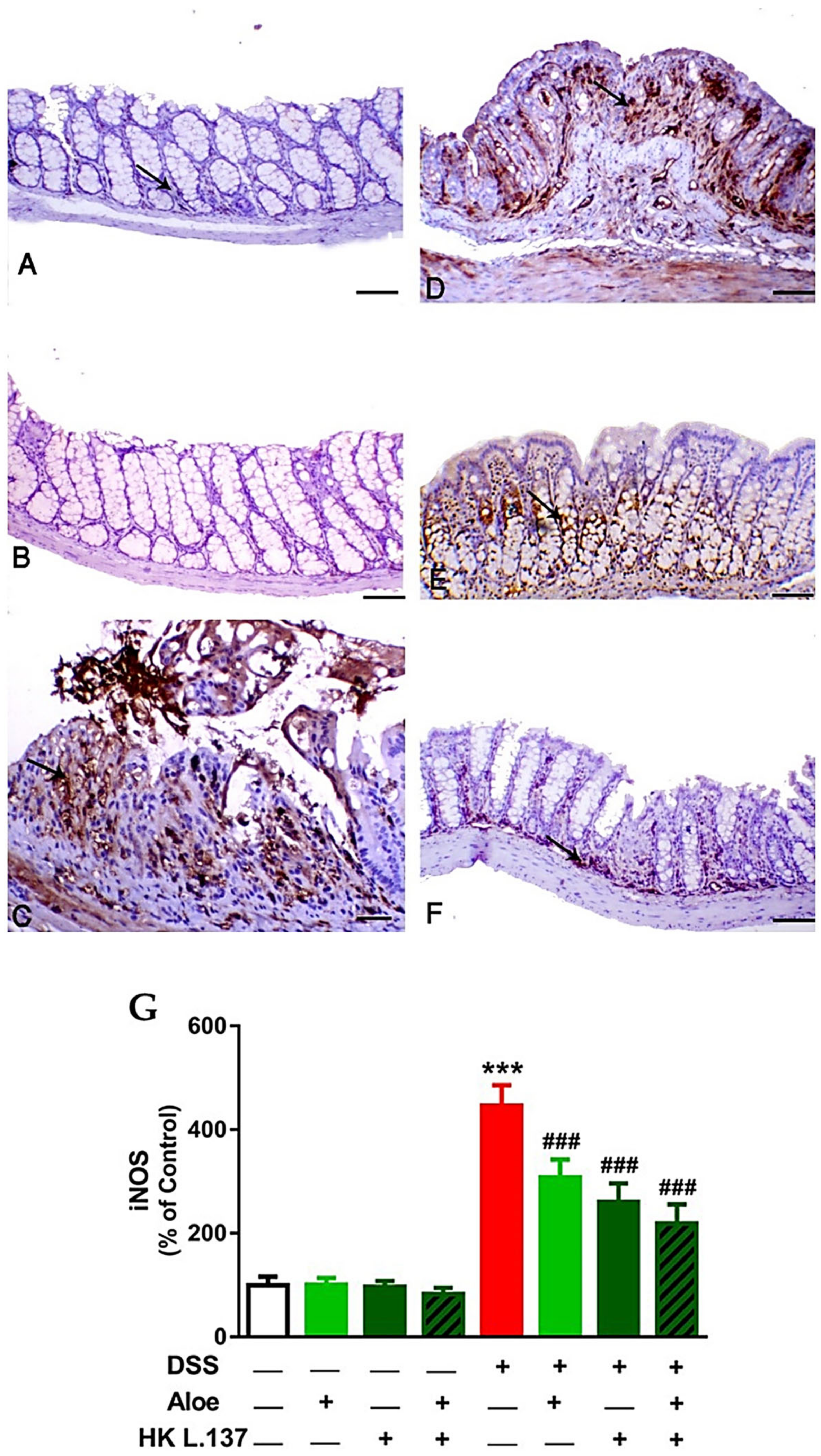
3.3. Aloe and/or HK L.137 Suppress NF-κB and iNOS Expression in the Colon of DSS-Induced Mice
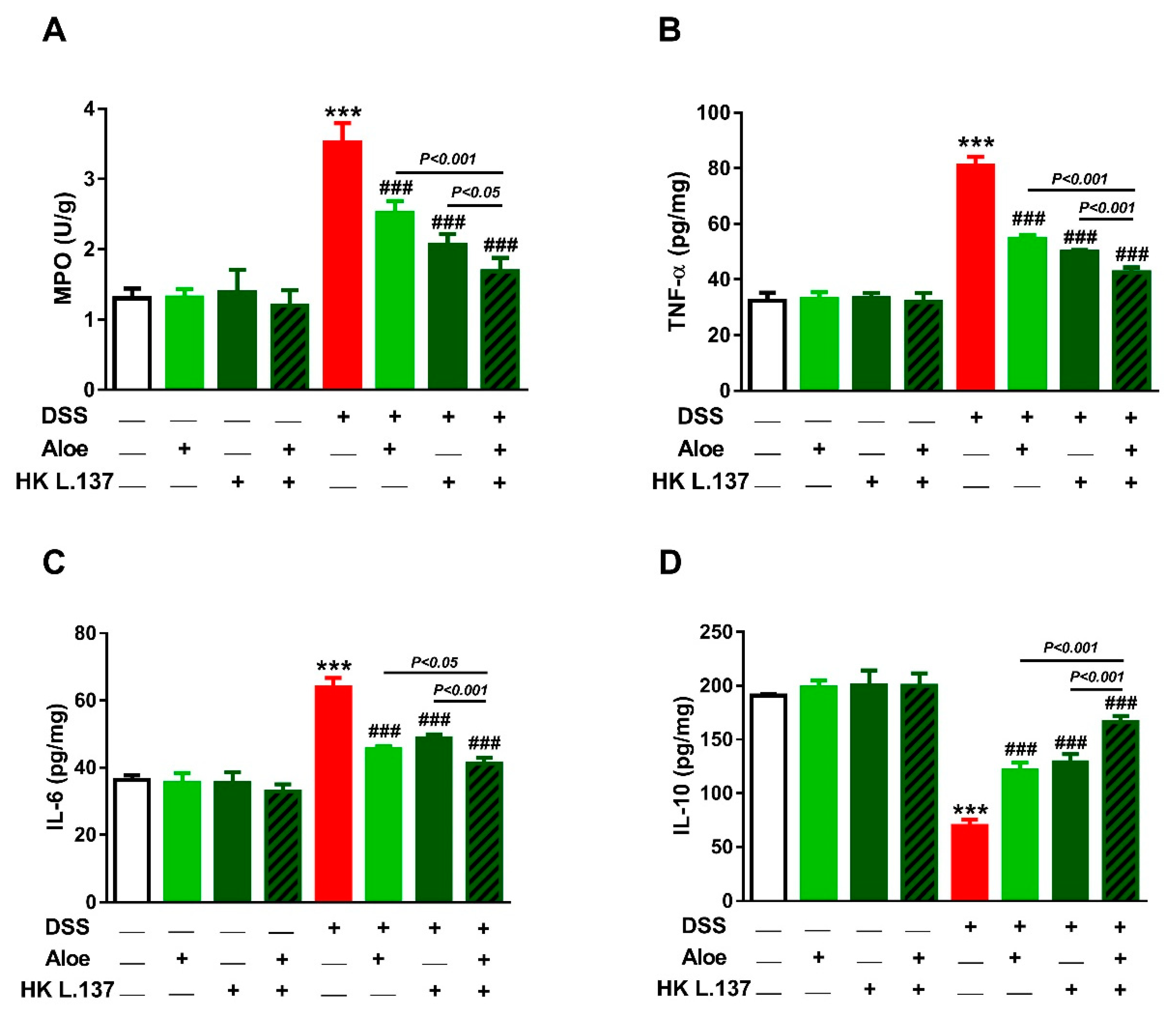
3.4. Aloe and/or HK L.137 Reduce MPO Activity and Attenuate Inflammation in DSS-Induced Mice
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Abraham, C.; Cho, J.H. Inflammatory bowel disease. N. Engl. J. Med. 2009, 361, 2066–2078. [Google Scholar] [CrossRef] [PubMed]
- Low, D.; Nguyen, D.D.; Mizoguchi, E. Animal models of ulcerative colitis and their application in drug research. Drug Des. Dev. Ther. 2013, 7, 1341–1357. [Google Scholar]
- Guan, Q.; Zhang, J. Recent advances: The imbalance of cytokines in the pathogenesis of inflammatory bowel disease. Mediat. Inflamm. 2017, 2017, 4810258. [Google Scholar] [CrossRef]
- Frolkis, A.D.; Dykeman, J.; Negron, M.E.; Debruyn, J.; Jette, N.; Fiest, K.M.; Frolkis, T.; Barkema, H.W.; Rioux, K.P.; Panaccione, R.; et al. Risk of surgery for inflammatory bowel diseases has decreased over time: A systematic review and meta-analysis of population-based studies. Gastroenterology 2013, 145, 996–1006. [Google Scholar] [CrossRef]
- Giuffrida, P.; Corazza, G.R.; Di Sabatino, A. Old and new lymphocyte players in inflammatory bowel disease. Dig. Dis. Sci. 2018, 63, 277–288. [Google Scholar] [CrossRef]
- Nagao-Kitamoto, H.; Kamada, N. Host-microbial cross-talk in inflammatory bowel disease. Immune Netw. 2017, 17, 1–12. [Google Scholar] [CrossRef]
- Ohkusa, T. Production of experimental ulcerative colitis in hamsters by dextran sulfate sodium and changes in intestinal microflora. Nihon Shokakibyo Gakkai Zasshi Jpn. J. Gastro Enterol. 1985, 82, 1327–1336. [Google Scholar]
- Bauer, C.; Duewell, P.; Mayer, C.; Lehr, H.A.; Fitzgerald, K.A.; Dauer, M.; Tschopp, J.; Endres, S.; Latz, E.; Schnurr, M. Colitis induced in mice with dextran sulfate sodium (dss) is mediated by the nlrp3 inflammasome. Gut 2010, 59, 1192–1199. [Google Scholar] [CrossRef]
- Pu, Z.; Han, C.; Zhang, W.; Xu, M.; Wu, Z.; Liu, Y.; Wu, M.; Sun, H.; Xie, H. Systematic understanding of the mechanism and effects of arctigenin attenuates inflammation in dextran sulfate sodium-induced acute colitis through suppression of nlrp3 inflammasome by sirt1. Am. J. Transl. Res. 2019, 11, 3992–4009. [Google Scholar]
- Okayasu, I.; Hatakeyama, S.; Yamada, M.; Ohkusa, T.; Inagaki, Y.; Nakaya, R. A novel method in the induction of reliable experimental acute and chronic ulcerative colitis in mice. Gastroenterology 1990, 98, 694–702. [Google Scholar] [CrossRef]
- Melgar, S.; Karlsson, L.; Rehnstrom, E.; Karlsson, A.; Utkovic, H.; Jansson, L.; Michaelsson, E. Validation of murine dextran sulfate sodium-induced colitis using four therapeutic agents for human inflammatory bowel disease. Int. Immunopharmacol. 2008, 8, 836–844. [Google Scholar] [CrossRef] [PubMed]
- Williams, N.T. Probiotics. Am. J. Health Syst. Pharm. AJHP Off. J. Am. Soc. Health Syst. Pharm. 2010, 67, 449–458. [Google Scholar] [CrossRef] [PubMed]
- Kruis, W.; Fric, P.; Pokrotnieks, J.; Lukas, M.; Fixa, B.; Kascak, M.; Kamm, M.A.; Weismueller, J.; Beglinger, C.; Stolte, M.; et al. Maintaining remission of ulcerative colitis with the probiotic escherichia coli nissle 1917 is as effective as with standard mesalazine. Gut 2004, 53, 1617–1623. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.L.; Hsu, P.Y.; Pan, T.M. Therapeutic effects of lactobacillus paracasei subsp. Paracasei ntu 101 powder on dextran sulfate sodium-induced colitis in mice. J. Food Drug Anal. 2019, 27, 83–92. [Google Scholar] [CrossRef]
- Gamallat, Y.; Meyiah, A.; Kuugbee, E.D.; Hago, A.M.; Chiwala, G.; Awadasseid, A.; Bamba, D.; Zhang, X.; Shang, X.; Luo, F.; et al. Lactobacillus rhamnosus induced epithelial cell apoptosis, ameliorates inflammation and prevents colon cancer development in an animal model. Biomed. Pharmacother. Biomed. Pharmacother. 2016, 83, 536–541. [Google Scholar] [CrossRef]
- Kang, D.W.; Adams, J.B.; Gregory, A.C.; Borody, T.; Chittick, L.; Fasano, A.; Khoruts, A.; Geis, E.; Maldonado, J.; McDonough-Means, S.; et al. Microbiota transfer therapy alters gut ecosystem and improves gastrointestinal and autism symptoms: An open-label study. Microbiome 2017, 5, 10. [Google Scholar] [CrossRef]
- Tamboli, C.P.; Neut, C.; Desreumaux, P.; Colombel, J.F. Dysbiosis in inflammatory bowel disease. Gut 2004, 53, 1–4. [Google Scholar] [CrossRef]
- Ozsoy, N.; Candoken, E.; Akev, N. Implications for degenerative disorders: Antioxidative activity, total phenols, flavonoids, ascorbic acid, beta-carotene and beta-tocopherol in aloe vera. Oxidative Med. Cell. Longev. 2009, 2, 99–106. [Google Scholar] [CrossRef]
- Sumi, F.A.; Sikder, B.; Rahman, M.M.; Lubna, S.R.; Ulla, A.; Hossain, M.H.; Jahan, I.A.; Alam, M.A.; Subhan, N. Phenolic content analysis of aloe vera gel and evaluation of the effect of aloe gel supplementation on oxidative stress and fibrosis in isoprenaline-administered cardiac damage in rats. Prev. Nutr. Food Sci. 2019, 24, 254–264. [Google Scholar] [CrossRef]
- Salehi, B.; Albayrak, S.; Antolak, H.; Kregiel, D.; Pawlikowska, E.; Sharifi-Rad, M.; Uprety, Y.; Tsouh Fokou, P.V.; Yousef, Z.; Amiruddin Zakaria, Z.; et al. Aloe genus plants: From farm to food applications and phytopharmacotherapy. Int. J. Mol. Sci. 2018, 19, 2843. [Google Scholar] [CrossRef]
- Park, M.Y.; Kwon, H.J.; Sung, M.K. Dietary aloin, aloesin, or aloe-gel exerts anti-inflammatory activity in a rat colitis model. Life Sci. 2011, 88, 486–492. [Google Scholar] [CrossRef] [PubMed]
- Im, S.A.; Kim, J.W.; Kim, H.S.; Park, C.S.; Shin, E.; Do, S.G.; Park, Y.I.; Lee, C.K. Prevention of azoxymethane/dextran sodium sulfate-induced mouse colon carcinogenesis by processed aloe vera gel. Int. Immunopharmacol. 2016, 40, 428–435. [Google Scholar] [CrossRef] [PubMed]
- Chassaing, B.; Aitken, J.D.; Malleshappa, M.; Vijay-Kumar, M. Dextran sulfate sodium (dss)-induced colitis in mice. Curr. Protoc. Immunol. 2014, 104, 15–25. [Google Scholar] [CrossRef] [PubMed]
- Mojahed, L.S.; Saeb, M.; Mohammadi, M.M.; Nazifi, S. Long period starvation in rat: The effect of aloe vera gel extract on oxidative stress status. Int. Arch. Med. 2016, 9, 252. [Google Scholar]
- Dawood, M.A.O.; Magouz, F.I.; Salem, M.F.I.; Abdel-Daim, H.A. Modulation of digestive enzyme activity, blood health, oxidative responses and growth-related gene expression in gift by heat-killed lactobacillus plantarum (l-137). Aquaculture 2019, 505, 127–136. [Google Scholar] [CrossRef]
- Cooper, H.S.; Murthy, S.N.; Shah, R.S.; Sedergran, D.J. Clinicopathologic study of dextran sulfate sodium experimental murine colitis. Lab. Investig. A J. Tech. Methods Pathol. 1993, 69, 238–249. [Google Scholar]
- Camuesco, D.; Rodriguez-Cabezas, M.E.; Garrido-Mesa, N.; Cueto-Sola, M.; Bailon, E.; Comalada, M.; Arribas, B.; Merlos, M.; Balsa, D.; Zarzuelo, A.; et al. The intestinal anti-inflammatory effect of dersalazine sodium is related to a down-regulation in il-17 production in experimental models of rodent colitis. Br. J. Pharmacol. 2012, 165, 729–740. [Google Scholar] [CrossRef]
- Valatas, V.; Vakas, M.; Kolios, G. The value of experimental models of colitis in predicting efficacy of biological therapies for inflammatory bowel diseases. Am. J. Physiol. Gastrointest. Liver Physiol. 2013, 305, G763–G785. [Google Scholar] [CrossRef]
- Hoffmann, M.; Schwertassek, U.; Seydel, A.; Weber, K.; Falk, W.; Hauschildt, S.; Lehmann, J. A refined and translationally relevant model of chronic dss colitis in balb/c mice. Lab. Anim. 2018, 52, 240–252. [Google Scholar] [CrossRef]
- Yan, Y.; Kolachala, V.; Dalmasso, G.; Nguyen, H.; Laroui, H.; Sitaraman, S.V.; Merlin, D. Temporal and spatial analysis of clinical and molecular parameters in dextran sodium sulfate induced colitis. PLoS ONE 2009, 4, e6073. [Google Scholar] [CrossRef]
- Losurdo, G.; Iannone, A.; Contaldo, A.; Ierardi, E.; Di Leo, A.; Principi, M. Escherichia coli nissle 1917 in ulcerative colitis treatment: Systematic review and meta-analysis. J. Gastrointest. Liver Dis. JGLD 2015, 24, 499–505. [Google Scholar]
- Curro, D.; Ianiro, G.; Pecere, S.; Bibbo, S.; Cammarota, G. Probiotics, fibre and herbal medicinal products for functional and inflammatory bowel disorders. Br. J. Pharmacol. 2017, 174, 1426–1449. [Google Scholar] [CrossRef]
- Langmead, L.; Feakins, R.M.; Goldthorpe, S.; Holt, H.; Tsironi, E.; De Silva, A.; Jewell, D.P.; Rampton, D.S. Randomized, double-blind, placebo-controlled trial of oral aloe vera gel for active ulcerative colitis. Aliment. Pharmacol. Ther. 2004, 19, 739–747. [Google Scholar] [CrossRef] [PubMed]
- Ducrotte, P.; Sawant, P.; Jayanthi, V. Clinical trial: Lactobacillus plantarum 299v (dsm 9843) improves symptoms of irritable bowel syndrome. World J. Gastroenterol. 2012, 18, 4012–4018. [Google Scholar] [CrossRef] [PubMed]
- Stevenson, C.; Blaauw, R.; Fredericks, E.; Visser, J.; Roux, S. Randomized clinical trial: Effect of lactobacillus plantarum 299 v on symptoms of irritable bowel syndrome. Nutrition 2014, 30, 1151–1157. [Google Scholar] [CrossRef] [PubMed]
- Atiba, A.; Nishimura, M.; Kakinuma, S.; Hiraoka, T.; Goryo, M.; Shimada, Y.; Ueno, H.; Uzuka, Y. Aloe vera oral administration accelerates acute radiation-delayed wound healing by stimulating transforming growth factor-beta and fibroblast growth factor production. Am. J. Surg. 2011, 201, 809–818. [Google Scholar] [CrossRef]
- Kumar, S.; Yadav, A.; Yadav, M.; Yadav, J.P. Effect of climate change on phytochemical diversity, total phenolic content and in vitro antioxidant activity of aloe vera (l.) burm.F. BMC Res. Notes 2017, 10, 60. [Google Scholar] [CrossRef]
- Kumar, S.; Singh, B.K.; Pandey, A.K.; Kumar, A.; Sharma, S.K.; Raj, H.G.; Prasad, A.K.; Van der Eycken, E.; Parmar, V.S.; Ghosh, B. A chromone analog inhibits tnf-alpha induced expression of cell adhesion molecules on human endothelial cells via blocking nf-kappab activation. Bioorganic Med. Chem. 2007, 15, 2952–2962. [Google Scholar] [CrossRef]
- Mazzei, M.; Dondero, R.; Sottofattori, E.; Melloni, E.; Minafra, R. Inhibition of neutrophil o(2)(-) production by unsymmetrical methylene derivatives of benzopyrans: Their use as potential antiinflammatory agents. Eur. J. Med. Chem. 2001, 36, 851–861. [Google Scholar] [CrossRef]
- Liu, M.; Yokomizo, T. The role of leukotrienes in allergic diseases. Allergol. Int. Off. J. Jpn. Soc. Allergol. 2015, 64, 17–26. [Google Scholar] [CrossRef]
- Bennek, E.; Mandic, A.D.; Verdier, J.; Roubrocks, S.; Pabst, O.; Van Best, N.; Benz, I.; Kufer, T.; Trautwein, C.; Sellge, G. Subcellular antigen localization in commensal e. Coli is critical for t cell activation and induction of specific tolerance. Mucosal Immunol. 2019, 12, 97–107. [Google Scholar] [CrossRef] [PubMed]
- Wasilewski, A.; Zielinska, M.; Storr, M.; Fichna, J. Beneficial effects of probiotics, prebiotics, synbiotics, and psychobiotics in inflammatory bowel disease. Inflamm. Bowel Dis. 2015, 21, 1674–1682. [Google Scholar] [CrossRef] [PubMed]
- Imaoka, A.; Umesaki, Y. Rationale for using of bifidobacterium probiotic strains-fermented milk against colitis based on animal experiments and clinical trials. Probiotics Antimicrob. Proteins 2009, 1, 8–14. [Google Scholar] [CrossRef] [PubMed]
- Guarner, F. The intestinal flora in inflammatory bowel disease: Normal or abnormal? Curr. Opin. Gastroenterol. 2005, 21, 414–418. [Google Scholar] [PubMed]
- Ni, J.; Wu, G.D.; Albenberg, L.; Tomov, V.T. Gut microbiota and ibd: Causation or correlation? Nat. Rev. Gastroenterol. Hepatol. 2017, 14, 573–584. [Google Scholar] [CrossRef] [PubMed]
- Le, B.; Yang, S.H. Efficacy of lactobacillus plantarum in prevention of inflammatory bowel disease. Toxicol. Rep. 2018, 5, 314–317. [Google Scholar] [CrossRef] [PubMed]
- Adams, C.A. The probiotic paradox: Live and dead cells are biological response modifiers. Nutr. Res. Rev. 2010, 23, 37–46. [Google Scholar] [CrossRef]
- Kim, H.; Kim, H.; Bang, J.; Kim, Y.; Beuchat, L.R.; Ryu, J.H. Reduction of bacillus cereus spores in sikhye, a traditional korean rice beverage, by modified tyndallization processes with and without carbon dioxide injection. Lett. Appl. Microbiol. 2012, 55, 218–223. [Google Scholar] [CrossRef]
- Sarkar, A.; Mandal, S. Bifidobacteria-insight into clinical outcomes and mechanisms of its probiotic action. Microbiol. Res. 2016, 192, 159–171. [Google Scholar] [CrossRef]
- Anderson, R.C.; Cookson, A.L.; McNabb, W.C.; Park, Z.; McCann, M.J.; Kelly, W.J.; Roy, N.C. Lactobacillus plantarum mb452 enhances the function of the intestinal barrier by increasing the expression levels of genes involved in tight junction formation. BMC Microbiol. 2010, 10, 316. [Google Scholar] [CrossRef]
- Zhang, F.; Li, Y.; Wang, X.; Wang, S.; Bi, D. The impact of lactobacillus plantarum on the gut microbiota of mice with dss-induced colitis. BioMed Res. Int. 2019, 2019, 3921315. [Google Scholar] [PubMed]
- Tomusiak-Plebanek, A.; Heczko, P.; Skowron, B.; Baranowska, A.; Okon, K.; Thor, P.J.; Strus, M. Lactobacilli with superoxide dismutase-like or catalase activity are more effective in alleviating inflammation in an inflammatory bowel disease mouse model. Drug Des. Dev. Ther. 2018, 12, 3221–3233. [Google Scholar] [CrossRef] [PubMed]
- de Mattos, B.R.; Garcia, M.P.; Nogueira, J.B.; Paiatto, L.N.; Albuquerque, C.G.; Souza, C.L.; Fernandes, L.G.; Tamashiro, W.M.; Simioni, P.U. Inflammatory bowel disease: An overview of immune mechanisms and biological treatments. Mediat. Inflamm. 2015, 2015, 493012. [Google Scholar] [CrossRef] [PubMed]
- Aardoom, M.A.; Veereman, G.; de Ridder, L. A review on the use of anti-tnf in children and adolescents with inflammatory bowel disease. Int. J. Mol. Sci. 2019, 20, 2529. [Google Scholar] [CrossRef]
- Koelink, P.J.; Bloemendaal, F.M.; Li, B.; Westera, L.; Vogels, E.W.M.; van Roest, M.; Gloudemans, A.K.; van‘t Wout, A.B.; Korf, H.; Vermeire, S.; et al. Anti-tnf therapy in ibd exerts its therapeutic effect through macrophage il-10 signalling. Gut 2019. [Google Scholar] [CrossRef]
- Rafa, H.; Amri, M.; Saoula, H.; Belkhelfa, M.; Medjeber, O.; Boutaleb, A.; Aftis, S.; Nakmouche, M.; Touil-Boukoffa, C. Involvement of interferon-gamma in bowel disease pathogenesis by nitric oxide pathway: A study in algerian patients. J. Interferon Cytokine Res. Off. J. Int. Soc. Interferon Cytokine Res. 2010, 30, 691–697. [Google Scholar] [CrossRef]
- Rafa, H.; Saoula, H.; Belkhelfa, M.; Medjeber, O.; Soufli, I.; Toumi, R.; de Launoit, Y.; Morales, O.; Nakmouche, M.; Delhem, N.; et al. Il-23/il-17a axis correlates with the nitric oxide pathway in inflammatory bowel disease: Immunomodulatory effect of retinoic acid. J. Interferon Cytokine Res. Off. J. Int. Soc. Interferon Cytokine Res. 2013, 33, 355–368. [Google Scholar] [CrossRef]
- Khan, A.A.; Alsahli, M.A.; Rahmani, A.H. Myeloperoxidase as an active disease biomarker: Recent biochemical and pathological perspectives. Med. Sci. 2018, 6, 33. [Google Scholar] [CrossRef]
- Sung, M.K.; Park, M.Y. Nutritional modulators of ulcerative colitis: Clinical efficacies and mechanistic view. World J. Gastroenterol. 2013, 19, 994–1004. [Google Scholar] [CrossRef]
- Grace, O.M.; Simmonds, M.S.; Smith, G.F.; van Wyk, A.E. Therapeutic uses of aloe l. (asphodelaceae) in southern africa. J. Ethnopharmacol. 2008, 119, 604–614. [Google Scholar] [CrossRef]

| Grade | Microscopic Findings |
|---|---|
| 0 | Absence of infiltration in the lamina propria. |
| 1 | Mild and focal infiltration and crypt degeneration. |
| 2 | Moderate infiltration and crypt degeneration. |
| 3 | Marked and diffuse infiltration and marked crypt degeneration. |
| Groups | Colitis Grade (%) | |||
|---|---|---|---|---|
| G0 | G1 | G2 | G3 | |
| Control | 99.45 ± 0.74 | 0.55 ± 0.74 | ND | ND |
| Aloe | 97.90 ± 1.30 | 2.09 ± 1.32 | ND | ND |
| HK L.137 | 98.85 ± 0.77 | 1.69 ± 0.19 | ND | ND |
| Aloe/HK L.137 | 98.81 ± 0.47 | 1.46 ± 0.56 | ND | ND |
| DSS | 6.33 ± 1.89 *** | 18.16 ± 1.34 *** | 28.22 ± 1.16 | 47.52 ± 2.15 |
| Aloe + DSS | 22.89 ± 6.43 ### | 26.46 ± 1.87 ## | 27.17 ± 5.17 | 23.49 ± 3.11 ### |
| HK L.137 + DSS | 19.78 ± 0.37 ### | 30.19 ± 3.91 ### | 28.09 ± 1.25 | 21.49 ± 3.35 ### |
| Aloe/HK L.137 + DSS | 36.12 ± 3.38 ### | 27.68 ± 4.84 ## | 17.12 ± 2.62 # | 19.06 ± 1.74 ### |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ismaeil, H.; Abdo, W.; Amer, S.; Tahoun, A.; Massoud, D.; Zanaty, E.; Bin-Jumah, M.; Mahmoud, A.M. Ameliorative Effect of Heat-Killed Lactobacillus plantarum L.137 and/or Aloe vera against Colitis in Mice. Processes 2020, 8, 225. https://doi.org/10.3390/pr8020225
Ismaeil H, Abdo W, Amer S, Tahoun A, Massoud D, Zanaty E, Bin-Jumah M, Mahmoud AM. Ameliorative Effect of Heat-Killed Lactobacillus plantarum L.137 and/or Aloe vera against Colitis in Mice. Processes. 2020; 8(2):225. https://doi.org/10.3390/pr8020225
Chicago/Turabian StyleIsmaeil, Heba, Walied Abdo, Said Amer, Amin Tahoun, Diaa Massoud, Eatemad Zanaty, May Bin-Jumah, and Ayman M. Mahmoud. 2020. "Ameliorative Effect of Heat-Killed Lactobacillus plantarum L.137 and/or Aloe vera against Colitis in Mice" Processes 8, no. 2: 225. https://doi.org/10.3390/pr8020225
APA StyleIsmaeil, H., Abdo, W., Amer, S., Tahoun, A., Massoud, D., Zanaty, E., Bin-Jumah, M., & Mahmoud, A. M. (2020). Ameliorative Effect of Heat-Killed Lactobacillus plantarum L.137 and/or Aloe vera against Colitis in Mice. Processes, 8(2), 225. https://doi.org/10.3390/pr8020225





