Recent Novel Hybrid Pd–Fe3O4 Nanoparticles as Catalysts for Various C–C Coupling Reactions
Abstract
:1. Introduction
2. Urchin-like FePd–Fe3O4: Nanocomposite Magnets
2.1. Synthesis of Urchin-Like Pd–Fe3O4 and L10-FePd–Fe Nanocomposite Magnets
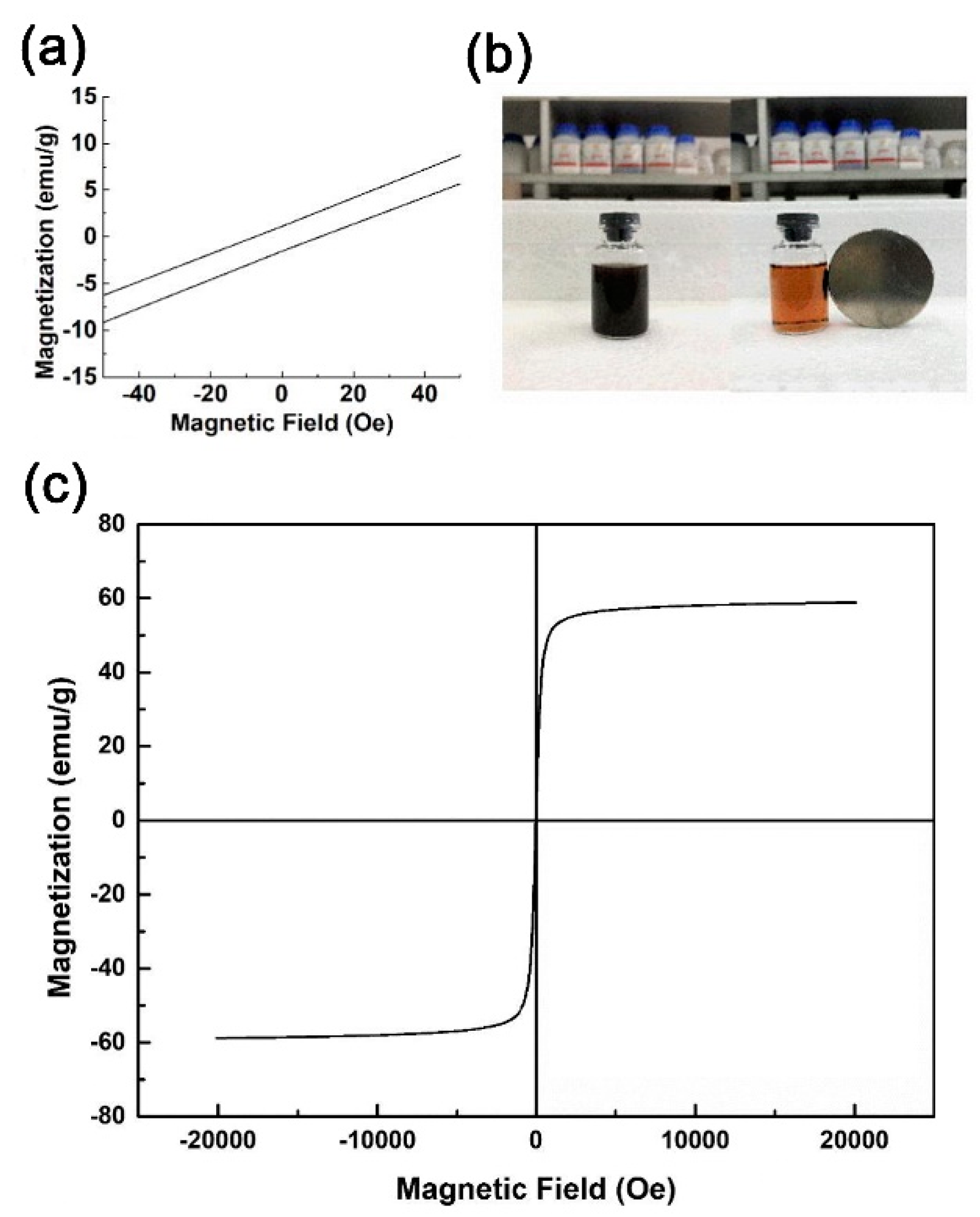
2.2. Magnetic Properties of L10-FePd–Fe Nanocomposites Magnets
3. Magnetically Recyclable Pd–Fe3O4 Hybrid Nanocatalyst: Application in Mizoroki–Heck Reaction
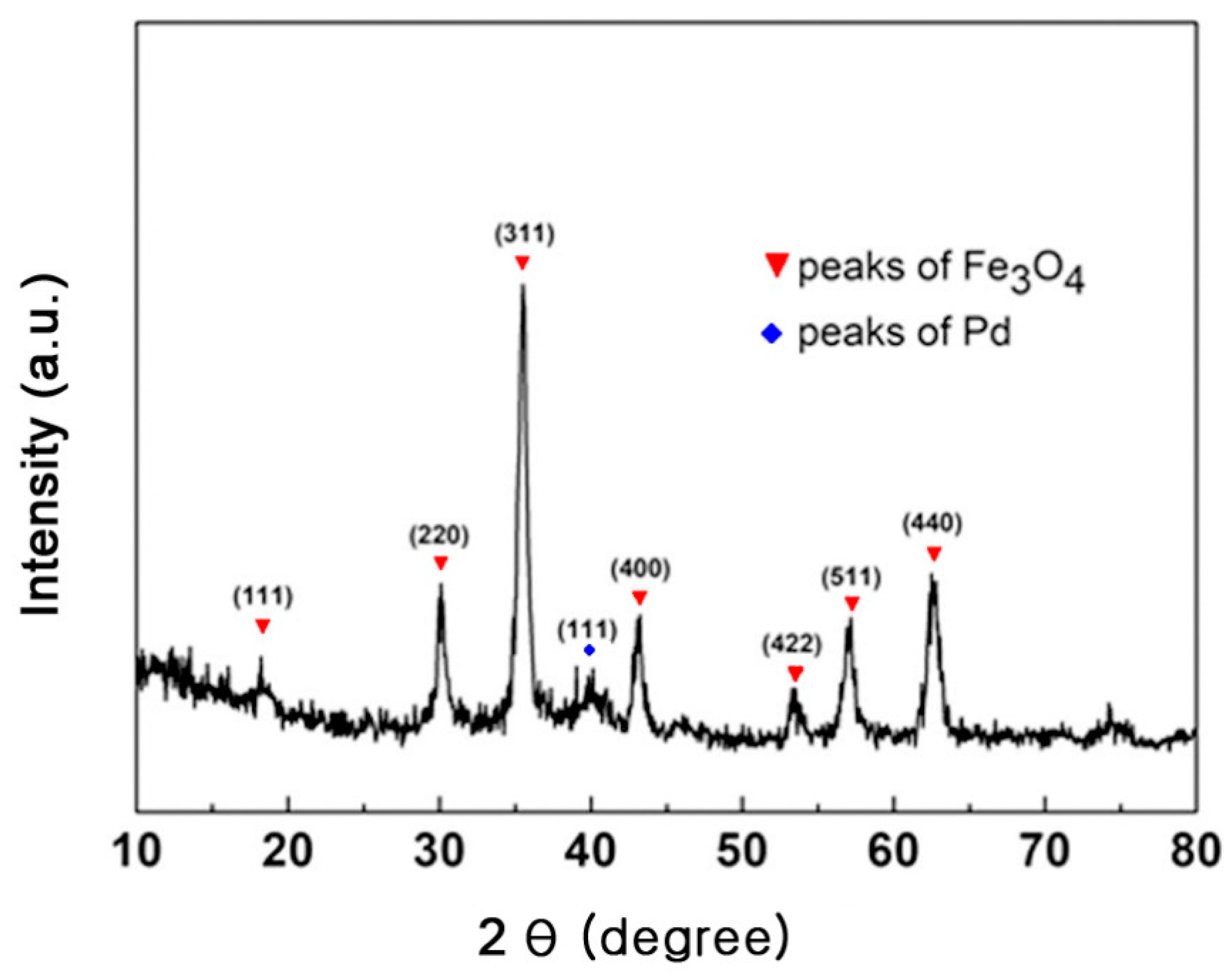
3.1. Synthesis and Characterization of Pd–Fe3O4 Hybrid Nanocatalyst
3.2. Pd–Fe3O4 Hybrid Nanocatalyst: Application in Mizoroki–Heck Reaction
4. Bifunctional Catalyst of Pd/Fe3O4/C: High content of Nanoparticles
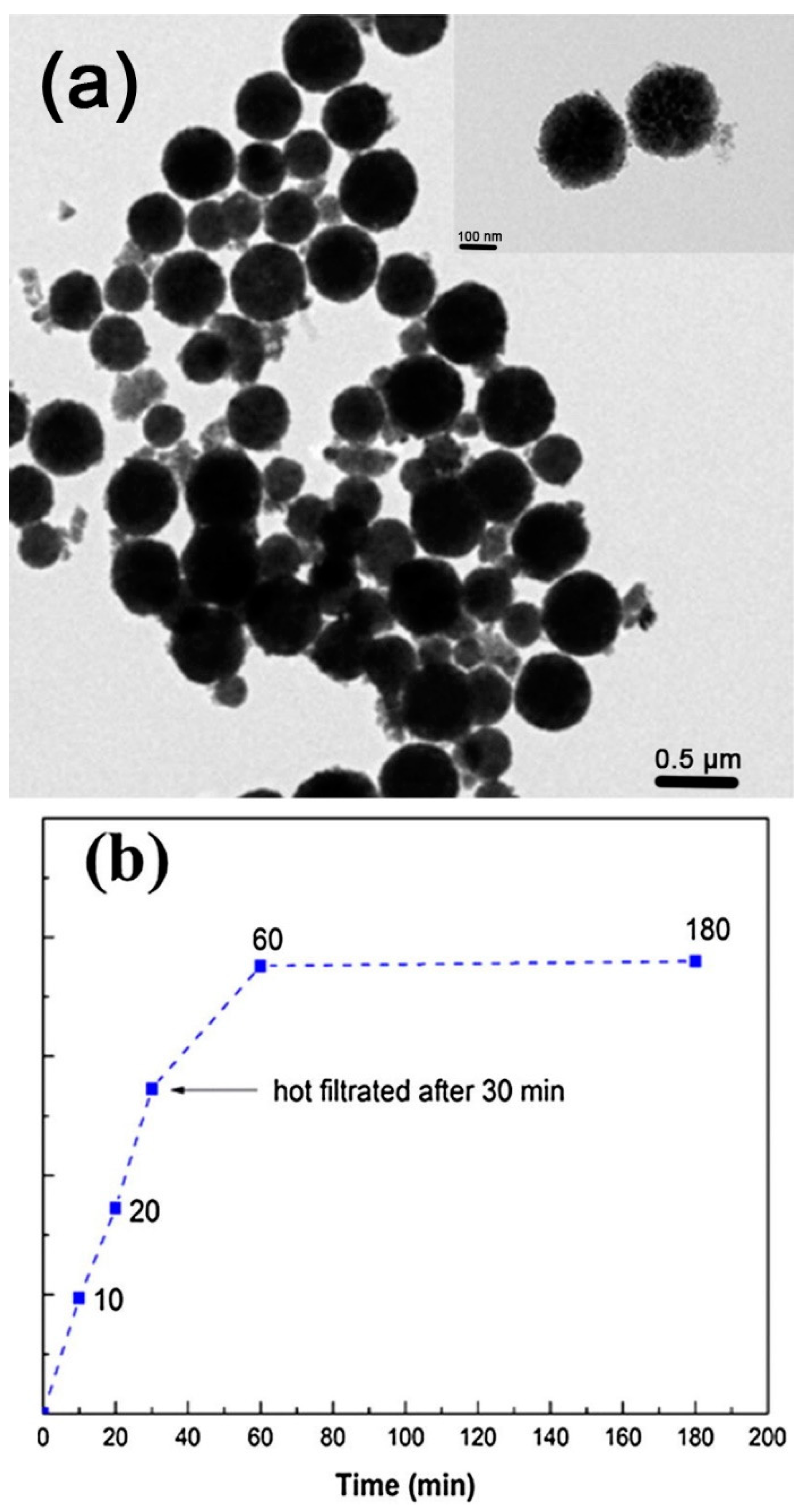
4.1. Melt Infiltration Method
4.1.1. Synthesis of Pd/Fe3O4/Charcoal Catalyst and Suzuki–Miyaura Coupling Reaction
4.1.2. Recycling and Pd Leaching Test
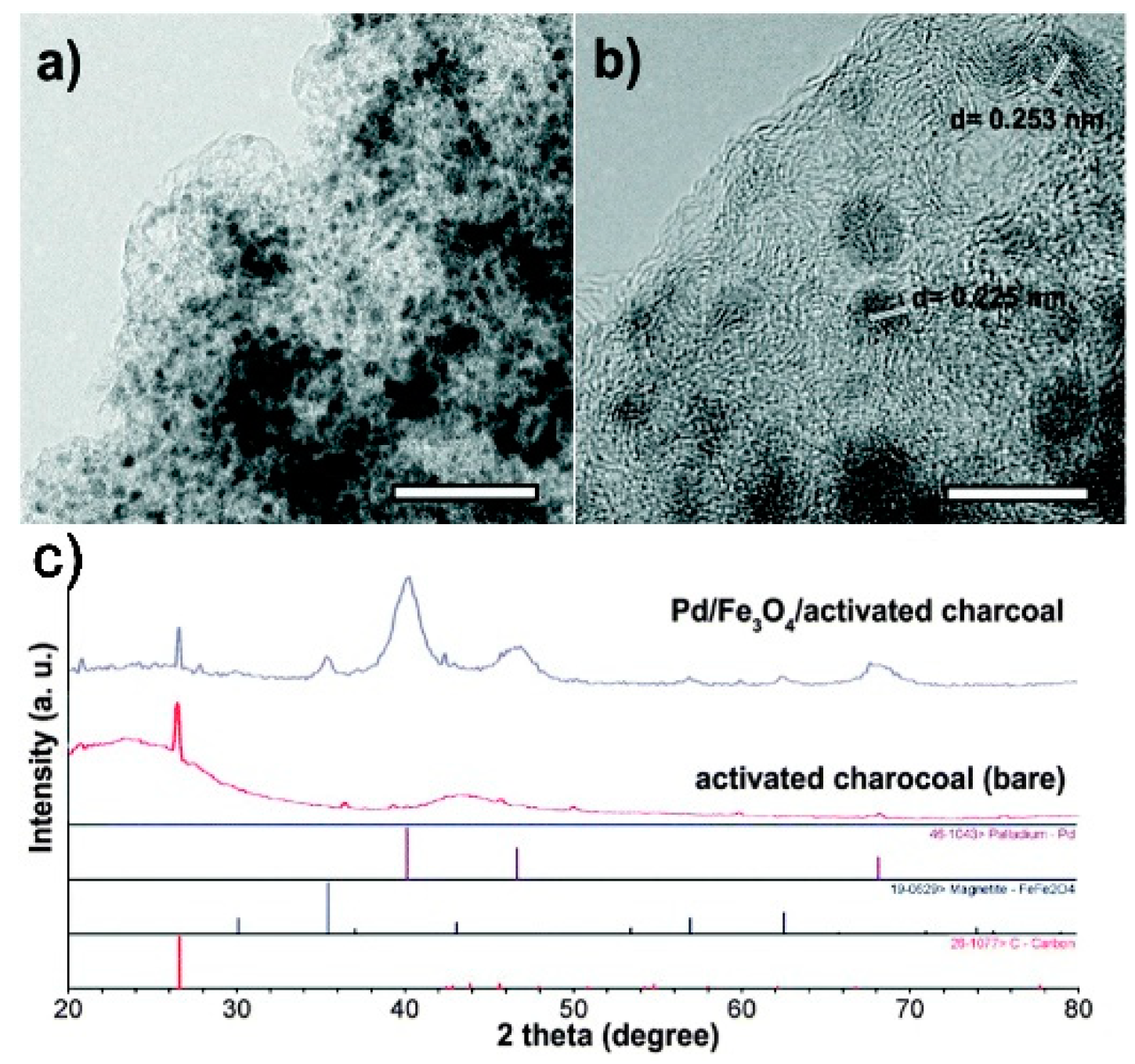
4.2. Stöber Method
4.2.1. Synthesis of Fe3O4@C–Pd Catalyst and Suzuki–Miyaura Coupling Reaction
4.2.2. Catalytic Efficiency of Fe3O4@C–Pd-550 Nanocomposite
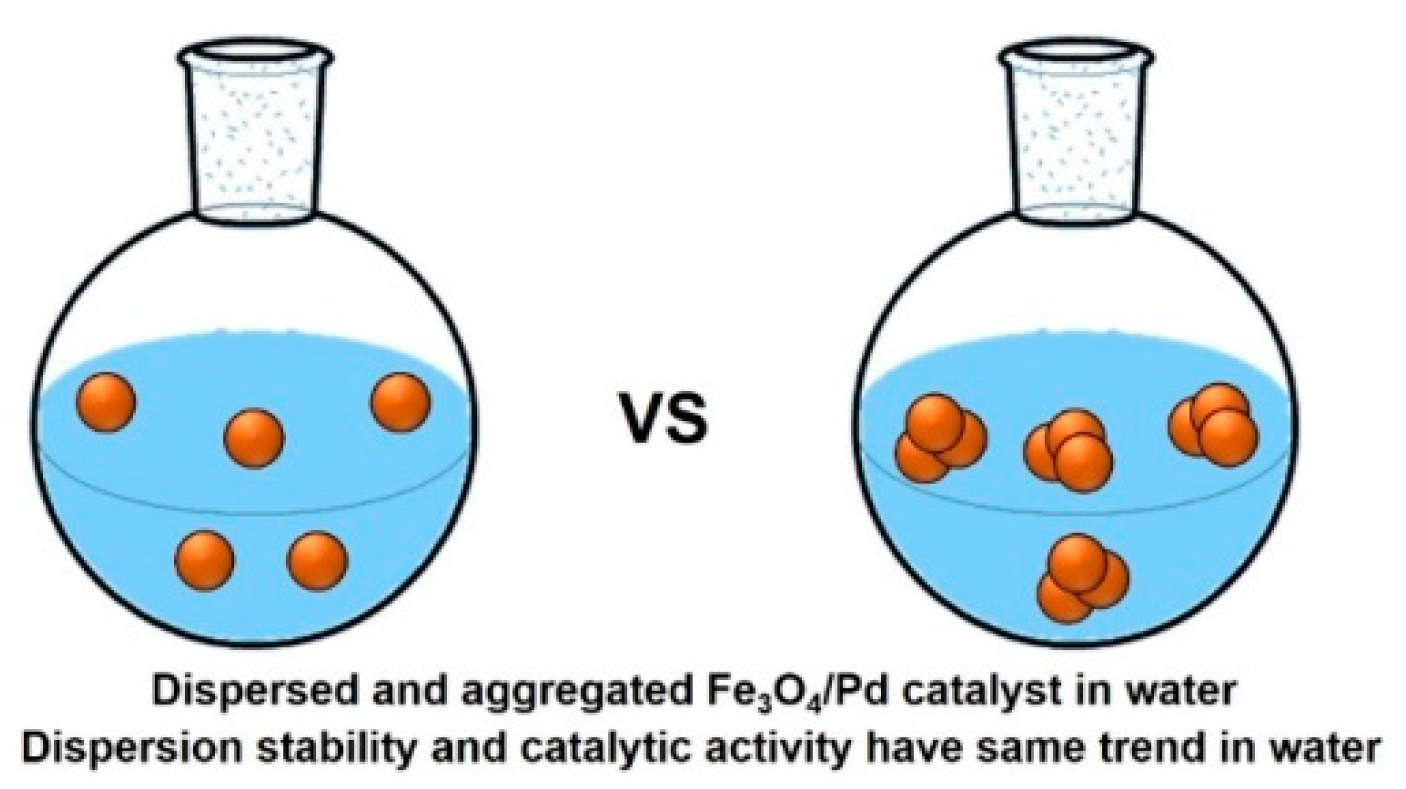
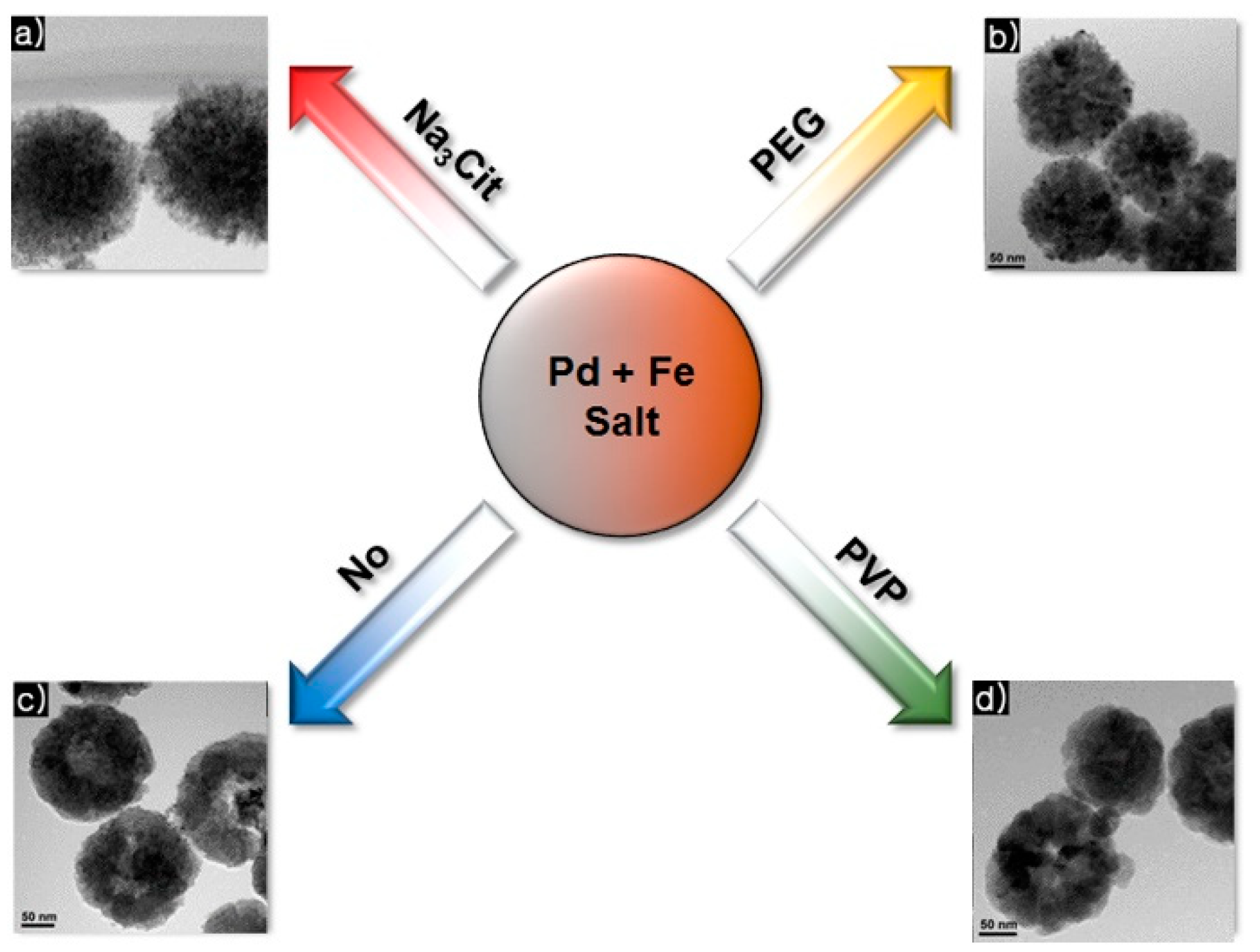
5. Hybrid Fe3O4/Pd Catalysts: Impact of Organic Capping Agents
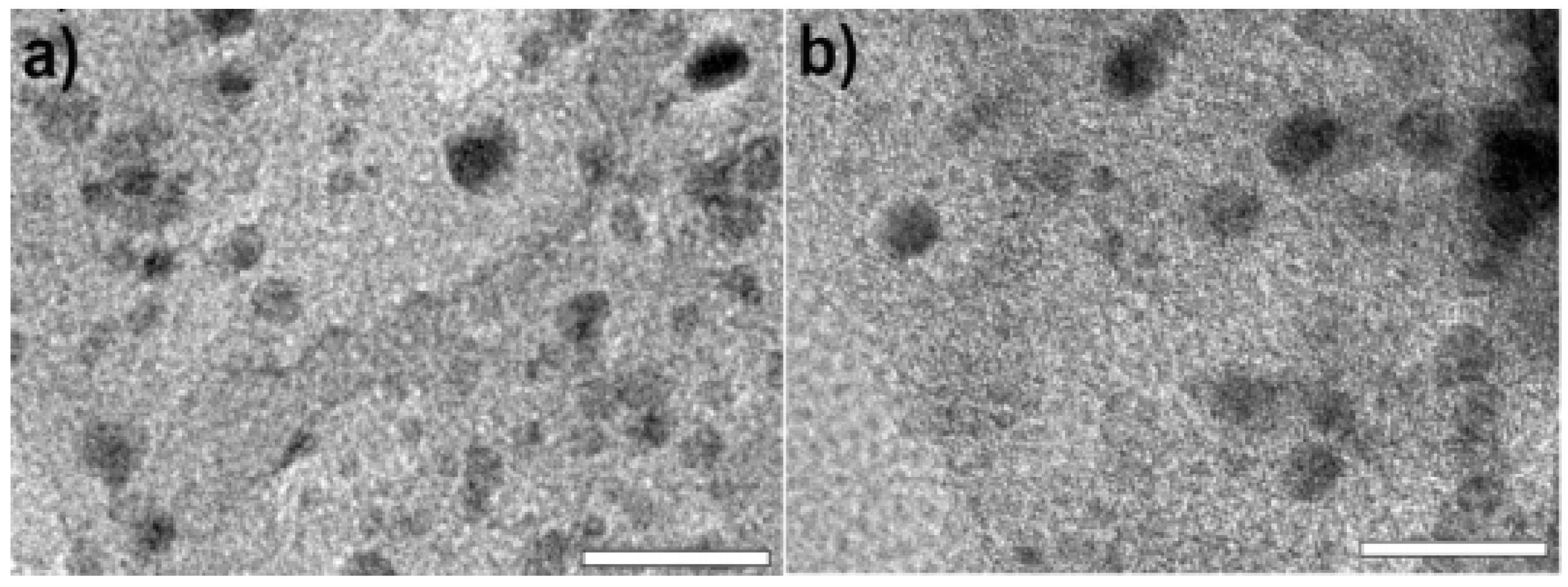
5.1. Immobilization of Pd NPs onto Each Fe3O4 Microsphere
5.2. Suzuki Coupling Reaction Using Pd/Fe3O4 Nanoparticles with Various Capping Agents
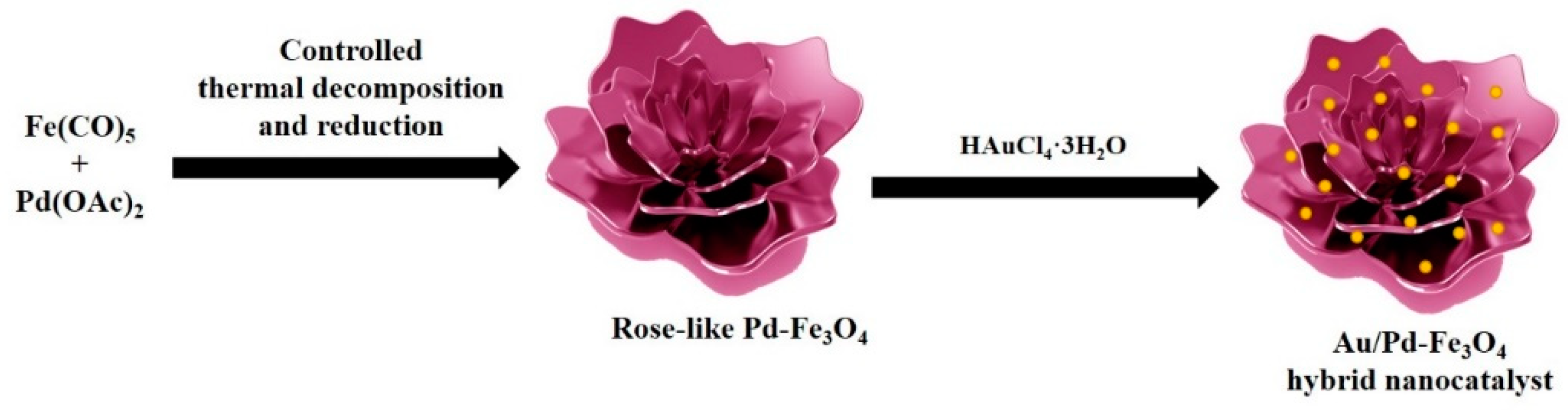
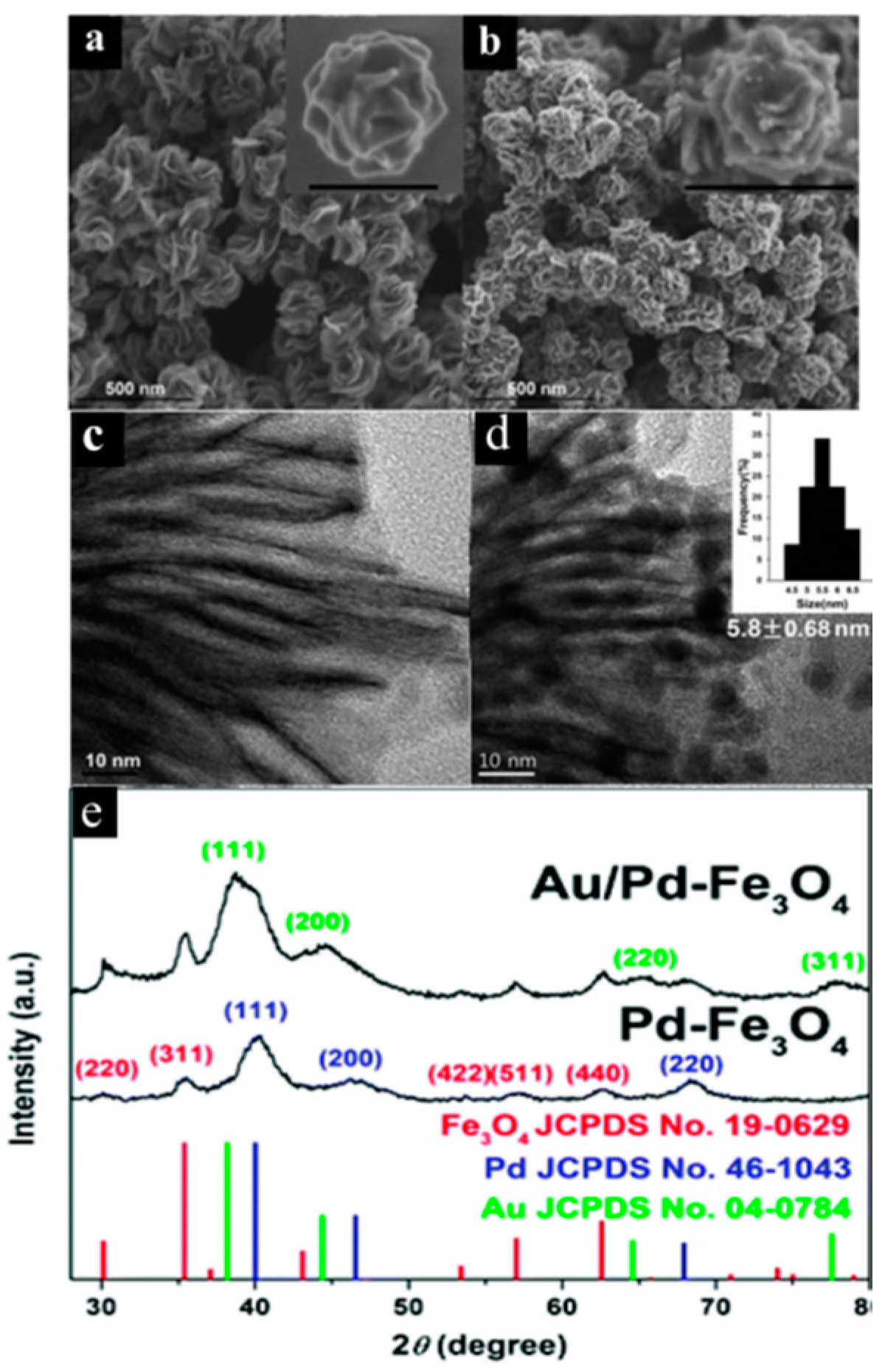
6. Flower-Like Pd–Fe3O4 and Pd–Fe3O4 Hybrid Nanocatalyst-Embedded Au Nanoparticles
6.1. Synthesis of Pd–Fe3O4 and Au/Pd–Fe3O4 Nanocomposites
6.2. Flower-Like Pd–Fe3O4: Application in Sonogashira Coupling Reactions
6.3. Pd–Fe3O4 Supported Au Nanocatalyst: Applications for Tandem Synthesis of 2-Phenylindoles
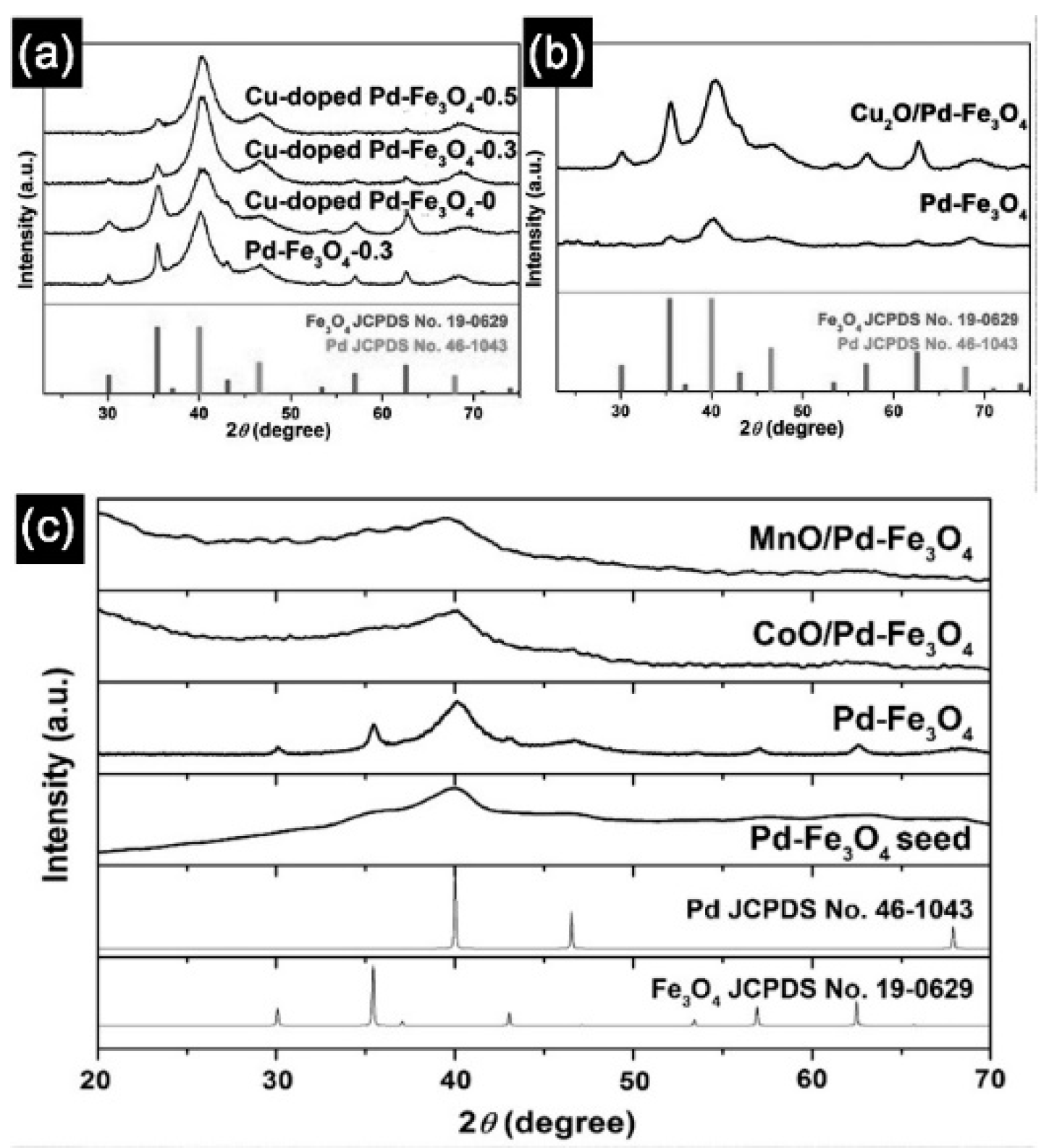
7. Transition Metal Loading Pd–Fe3O4 Heterobimetallic Nanoparticles
7.1. Synthesis of Hybrid Cu-Doped Pd–Fe3O4 Nanocatalyst
7.2. Synthesize Cu2O/Pd–Fe3O4 Nanocatalyst
7.3. Hybrid MnO and CoO/Pd–Fe3O4 Nanocomplexes
7.4. Synthesis of Hybrid Ni–Pd–Fe3O4 Nanocomposites
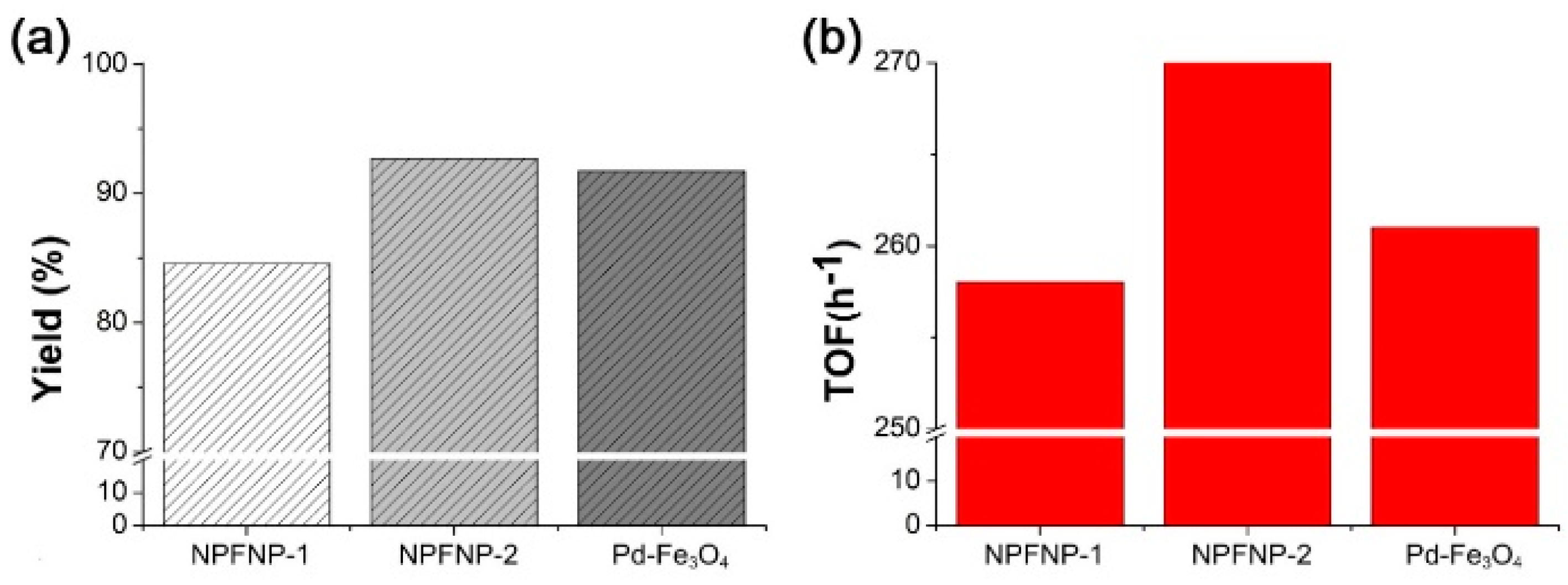
7.5. Applications of Transition Metal-Loaded Pd–Fe3O4 Heterobimetallic Nanoparticles in Organic Reactions
7.5.1. Tandem Synthesis of 2-Phenylbenzofurans
7.5.2. C–H Arylation of 1-Butyl-4-Nitro-1H-Imidazoles
7.5.3. Synthesis of Alkylboronates from Styrene
7.5.4. Suzuki–Miyaura Coupling Reaction
8. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Tan, L.; Wu, X.; Chen, D.; Liu, H.; Meng, X.; Tang, F. Confining alloy or core-shell Au-Pd bimetallic nanocrystals in silica nanorattles for enhanced catalytic performance. J. Mater. Chem. A 2013, 1, 10382–10388. [Google Scholar] [CrossRef]
- Moussa, S.; Siamaki, A.R.; Gupton, B.F.; El-Shall, M.S. Pd-partially reduced graphene oxide catalysts (Pd/PRGO): Laser synthesis of pd nanoparticles supported on PRGO nanosheets for carboncarbon cross coupling reactions. ACS Catal. 2012, 2, 145–154. [Google Scholar] [CrossRef]
- Elazab, H.A.; Siamaki, A.R.; Moussa, S.; Gupton, B.F.; El-Shall, M.S. Highly efficient and magnetically recyclable graphene-supported Pd/Fe3O4 nanoparticle catalysts for Suzuki and Heck cross-coupling reactions. Appl. Catal. A Gen. 2015, 491, 58–69. [Google Scholar] [CrossRef]
- Nasrollahzadeh, M.; Mohammad Sajadi, S.; Rostami-Vartooni, A.; Khalaj, M. Green synthesis of Pd/Fe3O4 nanoparticles using Euphorbia condylocarpa M. bieb root extract and their catalytic applications as magnetically recoverable and stable recyclable catalysts for the phosphine-free Sonogashira and Suzuki coupling reactions. J. Mol. Catal. A Chem. 2015, 396, 31–39. [Google Scholar] [CrossRef]
- Siamaki, A.R.; Khder, A.E.R.S.; Abdelsayed, V.; El-Shall, M.S.; Gupton, B.F. Microwave-assisted synthesis of palladium nanoparticles supported on graphene: A highly active and recyclable catalyst for carbon-carbon cross-coupling reactions. J. Catal. 2011, 279, 1–11. [Google Scholar] [CrossRef]
- Truong, Q.D.; Le, T.S.; Ling, Y.C. Pt deposited TiO2 catalyst fabricated by thermal decomposition of titanium complex for solar hydrogen production. Solid State Sci. 2014, 38, 18–24. [Google Scholar] [CrossRef]
- Woo, H.; Lee, K.; Park, J.C.; Park, K.H. Facile synthesis of Pd/Fe3O4/charcoal bifunctional catalysts with high metal loading for high product yields in Suzuki-Miyaura coupling reactions. New J. Chem. 2014, 38, 5626–5632. [Google Scholar] [CrossRef]
- Tan, Q.; Du, C.; Sun, Y.; Yin, G.; Gao, Y. Pd-around-CeO2-x hybrid nanostructure catalyst: Three-phase-transfer synthesis, electrocatalytic properties and dual promoting mechanism. J. Mater. Chem. A 2014, 2, 1429–1435. [Google Scholar] [CrossRef]
- Hosseini, S.G.; Abazari, R.; Ghavi, A. Pure CuCr2O4 nanoparticles: Synthesis, characterization and their morphological and size effects on the catalytic thermal decomposition of ammonium perchlorate. Solid State Sci. 2015, 45, 51. [Google Scholar] [CrossRef]
- Veisi, H.; Sedrpoushan, A.; Maleki, B.; Hekmati, M.; Heidari, M.; Hemmati, S. Palladium immobilized on amidoxime-functionalized magnetic Fe3O4 nanoparticles: A highly stable and efficient magnetically recoverable nanocatalyst for sonogashira coupling reaction. Appl. Organomet. Chem. 2015, 29, 834–839. [Google Scholar] [CrossRef]
- Quek, X.Y.; Pestman, R.; vanSanten, R.A.; Hensen, E.J.M. Effect of organic capping agents on ruthenium-nanoparticle-catalyzed aqueous-phase Fischer-Tropsch synthesis. ChemCatChem 2013, 5, 3148–3155. [Google Scholar] [CrossRef]
- Tartaj, P.; del Puerto Morales, M.; Veintemillas-Verdaguer, S.; Gonzalez-Carreno, T.; Serna, C.J. The preparation of magnetic nanoparticles for applications in biomedicine. J. Phys. D Appl. Phys. 2003, 36, R182–R197. [Google Scholar] [CrossRef]
- Wang, Y.; Yang, H. Oleic acid as the capping agent in the synthesis of noble metal nanoparticles in imidazolium-based ionic liquids. Chem. Commun. 2006, 2545–2547. [Google Scholar] [CrossRef] [PubMed]
- Lévy, R.; Thanh, N.T.K.; Christopher Doty, R.; Hussain, I.; Nichols, R.J.; Schiffrin, D.J.; Brust, M.; Fernig, D.G. Rational and combinatorial design of peptide capping ligands for gold nanoparticles. J. Am. Chem. Soc. 2004, 126, 10076–10084. [Google Scholar] [CrossRef] [PubMed]
- Otsuka, H.; Nagasaki, Y.; Kataoka, K. PEGylated nanoparticles for biological and pharmaceutical applications. Adv. Drug Deliv. Rev. 2012, 64, 246–255. [Google Scholar] [CrossRef] [Green Version]
- Corbierre, M.K.; Cameron, N.S.; Sutton, M.; Laaziri, K.; Lennox, R.B. Gold nanoparticle/polymer nanocomposites: Dispersion of nanoparticles as a function of capping agent molecular weight and grafting density. Langmuir 2005, 21, 6063–6072. [Google Scholar] [CrossRef] [PubMed]
- Rajamathi, M.; Ghosh, M.; Seshadri, R. Hydrolysis and amine-capping in a glycol solvent as a route to soluble maghemite g-Fe2O3 nanoparticles water; by performing the reaction in propylene glycol under. Chem. Commun. 2002, 1152–1153. [Google Scholar] [CrossRef]
- Fang, Y.; Wang, E. Simple and direct synthesis of oxygenous carbon supported palladium nanoparticles with high catalytic activity. Nanoscale 2013, 5, 1843–1848. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.W.; Kim, M.; Kim, Z.H.; Han, S.W. One-Step Synthesis of Au@Pd Core—Shell Nanooctahedron. J. Am. Chem. Soc. 2009, 131, 17036–17037. [Google Scholar] [CrossRef]
- Linares, N.; Canlas, C.P.; Garcia-Martinez, J.; Pinnavaia, T.J. Colloidal gold immobilized on mesoporous silica as a highly active and selective catalyst for styrene epoxidation with H2O2. Catal. Commun. 2014, 44, 50–53. [Google Scholar] [CrossRef]
- Schätz, A.; Long, T.R.; Grass, R.N.; Stark, W.J.; Hanson, P.R.; Reiser, O. Immobilization on a nanomagnetic Co/C surface using ROM polymerization: Generation of a hybrid material as support for a recyclable palladium catalyst. Adv. Funct. Mater. 2010, 20, 4323–4328. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.; Zhang, B.; Tian, J.; Wang, J.; Chong, Y.; Wang, X.; Deng, Y.; Tang, M.; Li, Y.; Ge, C. Synthesis of heterodimer radionuclide nanoparticles for magnetic resonance and single-photon emission computed tomography dual-modality imaging. Nanoscale 2015, 7, 3392–3395. [Google Scholar] [CrossRef] [PubMed]
- Jang, Y.; Chung, J.; Kim, S.; Jun, S.W.; Kim, B.H.; Lee, D.W.; Kim, B.M.; Hyeon, T. Simple synthesis of Pd-Fe3O4 heterodimer nanocrystals and their application as a magnetically recyclable catalyst for Suzuki cross-coupling reactions. Phys. Chem. Chem. Phys. 2011, 13, 2512–2516. [Google Scholar] [CrossRef]
- Kong, L.; Lu, X.; Bian, X.; Zhang, W.; Wang, C. Constructing carbon-coated Fe3O4 microspheres as antiacid and magnetic support for palladium nanoparticles for catalytic applications. ACS Appl. Mater. Interfaces 2011, 3, 35–42. [Google Scholar] [CrossRef] [PubMed]
- Li, R.; Zhang, P.; Huang, Y.; Zhang, P.; Zhong, H.; Chen, Q. Pd-Fe3O4@C hybrid nanoparticles: Preparation, characterization, and their high catalytic activity toward Suzuki coupling reactions. J. Mater. Chem. 2012, 22, 22750–22755. [Google Scholar] [CrossRef]
- Balanta, A.; Godard, C.; Claver, C. Pd nanoparticles for C-C coupling reactions. Chem. Soc. Rev. 2011, 40, 4973–4985. [Google Scholar] [CrossRef] [PubMed]
- Miyaura, N.; Suzuki, A. Palladium-Catalyzed Cross-Coupling Reactions of Organoboron Compounds. Chem. Rev. 1995, 95, 2457–2483. [Google Scholar] [CrossRef] [Green Version]
- Beletskaya, I.P.; Cheprakov, A.V. Heck reaction as a sharpening stone of palladium catalysis. Chem. Rev. 2000, 100, 3009–3066. [Google Scholar] [CrossRef] [PubMed]
- Zhu, M.; Diao, G. Magnetically recyclable Pd nanoparticles immobilized on magnetic Fe3O4@C nanocomposites: Preparation, characterization, and their catalytic activity toward suzuki and heck coupling reactions. J. Phys. Chem. C 2011, 115, 24743–24749. [Google Scholar] [CrossRef]
- Zhou, L.; Gao, C.; Xu, W. Robust Fe3O4/SiO2-Pt/Au/Pd magnetic nanocatalysts with multifunctional hyperbranched polyglycerol amplifiers. Langmuir 2010, 26, 11217–11225. [Google Scholar] [CrossRef]
- Yu, Y.; Sun, K.; Tian, Y.; Li, X.Z.; Kramer, M.J.; Sellmyer, D.J.; Shield, J.E.; Sun, S. One-pot synthesis of urchin-like FePd-Fe3O4 and their conversion into exchange-coupled L10-FePd-Fe nanocomposite magnets. Nano Lett. 2013, 13, 4975–4979. [Google Scholar] [CrossRef] [PubMed]
- Zhen, F.; Ran, M.; Chu, W.; Jiang, C.; Sun, W. A facile one-pot solvothermal method for synthesis of magnetically recoverable Pd-Fe3O4 hybrid nanocatalysts for the Mizoroki–Heck reaction. Chem. Phys. Lett. 2018, 695, 183–189. [Google Scholar] [CrossRef]
- Woo, H.; Lee, K.; Park, K.H. Optimized dispersion and stability of hybrid Fe3O4/Pd catalysts in water for Suzuki coupling reactions: Impact of organic capping agents. ChemCatChem 2014, 6, 1635–1640. [Google Scholar] [CrossRef]
- Woo, H.; Lee, K.; Park, J.C.; Park, K.H. One-pot synthesis of a rose-like Pd-Fe3O4 nanocatalyst for Sonogashira coupling reactions. Solid State Sci. 2016, 53, 78–82. [Google Scholar] [CrossRef]
- Woo, H.; Park, J.C.; Park, S.; Park, K.H. Rose-like Pd-Fe3O4 hybrid nanocomposite-supported Au nanocatalysts for tandem synthesis of 2-phenylindoles. Nanoscale 2015, 7, 8356–8360. [Google Scholar] [CrossRef] [PubMed]
- Woo, H.; Park, J.; Kim, J.; Park, S.; Park, K.H. Hierarchical hybrid MnO/Pd-Fe3O4 and CoO/Pd-Fe3O4 nanocomposites as efficient catalysts for hydroboration of styrene. Catal. Commun. 2017, 100, 52–56. [Google Scholar] [CrossRef]
- Woo, H.; Park, J.; Park, J.C.; Park, S.; Lee, J.M.; Park, K.H. Facile synthesis of hybrid Cu2O/Pd-Fe3O4 nanocatalysts for C-H arylation of 4-nitroimidazoles. RSC Adv. 2016, 6, 36211–36217. [Google Scholar] [CrossRef]
- Woo, H.; Kim, D.; Park, J.C.; Kim, J.W.; Park, S.; Lee, J.M.; Park, K.H. A new hybrid nanocatalyst based on Cu-doped Pd-Fe3O4 for tandem synthesis of 2-phenylbenzofurans. J. Mater. Chem. A 2015, 3, 20992–20998. [Google Scholar] [CrossRef]
- Jang, S.; Kim, T.; Park, K.H. Fabrication of Crumpled Ball-Like Nickel Doped Palladium-Iron Oxide Hybrid Nanoparticles with Controlled Morphology as Effective Catalyst for Suzuki–Miyaura Coupling Reaction. Catalysts 2017, 7, 247. [Google Scholar] [CrossRef]
- Jones, N. Materials science: The pull of stronger magnets. Nature 2011, 472, 22–23. [Google Scholar] [CrossRef] [Green Version]
- Stamps, R.L.; Breitkreutz, S.; Åkerman, J.; Chumak, A.V.; Otani, Y.; Bauer, G.E.W.; Thiele, J.U.; Bowen, M.; Majetich, S.A.; Kläui, M.; et al. The 2014 Magnetism Roadmap. J. Phys. D. Appl. Phys. 2014, 47. [Google Scholar] [CrossRef]
- Gutfleisch, O.; Willard, M.A.; Brück, E.; Chen, C.H.; Sankar, S.G.; Liu, J.P. Magnetic materials and devices for the 21st century: Stronger, lighter, and more energy efficient. Adv. Mater. 2011, 23, 821–842. [Google Scholar] [CrossRef] [PubMed]
- Fullerton, E.E.; Jiang, J.S.; Grimsditch, M.; Sowers, C.H.; Bader, S.D. Exchange-spring behavior in epitaxial hard/soft magnetic bilayers. Phys. Rev. B 2002, 58, 12193–12200. [Google Scholar] [CrossRef]
- Skomski, R.; Coey, J.M.D. Giant energy product in nanostructured two-phase magnets. Phys. Rev. B 1993, 48, 15812–15816. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fullerton, E.E.; Jiang, J.S.; Bader, S.D. Hard/soft magnetic heterostructures: Model exchange-spring magnets. J. Magn. Magn. Mater. 1999, 200, 392–404. [Google Scholar] [CrossRef]
- Zeng, H.; Li, J.; Liu, J.P.; Wang, Z.L.; Sun, S. Exchange-coupled nanocomposite magnets by nanoparticle self-assembly. Nature 2002, 420, 395–398. [Google Scholar] [CrossRef]
- Kneller, E.F.; Hawig, R. The Exchange-Spring Magnet: A New material Principle for Permanent Magnets. IEEE Trans. Magn. 1991, 27, 3588–3600. [Google Scholar] [CrossRef]
- Hou, B.Y.; Xu, Z.; Peng, S.; Rong, C.; Liu, J.P.; Sun, S. A facile synthesis of SmCo5 magnets from core/shell Co/Sm2O3 nanoparticles. Adv. Mater. 2007, 19, 3349–3352. [Google Scholar] [CrossRef]
- Figuerola, A.; Fiore, A.; Di Corato, R.; Falqui, A.; Giannini, C.; Micotti, E.; Lascialfari, A.; Corti, M.; Cingolani, R.; Pellegrino, T.; et al. One-pot synthesis and characterization of size-controlled bimagnetic FePt-iron oxide heterodimer nanocrystals. J. Am. Chem. Soc. 2008, 130, 1477–1487. [Google Scholar] [CrossRef]
- Sun, S.; Murray, C.B.; Weller, D.; Folks, L.; Moser, A. Monodisperse FePt Nanoparticles and Ferromagnetic FePt Nanocrystal Superlattices. Science 2000, 287, 1989–1991. [Google Scholar] [CrossRef]
- Zeng, H.; Li, J.; Wang, Z.L.; Liu, J.P.; Sun, S. Bimagnetic Core/Shell FePt/Fe3O4 Nanoparticles. Nano Lett. 2004, 4, 187–190. [Google Scholar] [CrossRef]
- Sakuma, N.; Ohshima, T.; Shoji, T.; Suzuki, Y.; Sato, R.; Wachi, A.; Kato, A.; Kawai, Y.; Manabe, A.; Teranishi, T. Exchange coupling interaction in L10 -FePd/α-Fe nanocomposite magnets with large maximum energy products. ACS Nano 2011, 5, 2806–2814. [Google Scholar] [CrossRef] [PubMed]
- Hayashi, Y. Pot economy and one-pot synthesis. Chem. Sci. 2016, 7, 866–880. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Laurent, S.; Forge, D.; Port, M.; Roch, A.; Robic, C.; Vander Elst, L.; Muller, R.N. Magnetic Iron Oxide Nanoparticles: Synthesis, Stabilization, Vectorization, Physicochemical Characterizations, and Biological Applications. Chem. Rev. 2008, 108, 2064–2110. [Google Scholar] [CrossRef] [PubMed]
- Heck, K.F.; Nolley, J.P. Palladium-Catalyzed Vinylic Hydrogen Substitution Reactions with Aryl, Benzyl, and Styryl Halides. J. Org. Chem. 1972, 37, 2320–2322. [Google Scholar] [CrossRef]
- Li, S.; Zhang, W.; Chen, F.; Chen, R. One-pot hydrothermal synthesis of Pd/Fe3O4 nanocomposite in HEPES buffer solution and catalytic activity for Suzuki reaction. Mater. Res. Bull. 2015, 66, 186–191. [Google Scholar] [CrossRef]
- Li, S.; Zhang, W.; So, M.H.; Che, C.M.; Wang, R.; Chen, R. One-pot solvothermal synthesis of Pd/Fe3O4 nanocomposite and its magnetically recyclable and efficient catalysis for Suzuki reactions. J. Mol. Catal. A Chem. 2012, 359, 81–87. [Google Scholar] [CrossRef]
- Chung, J.; Kim, J.; Jang, Y.; Byun, S.; Hyeon, T.; Kim, B.M. Heck and Sonogashira cross-coupling reactions using recyclable Pd-Fe 3O4 heterodimeric nanocrystal catalysts. Tetrahedron Lett. 2013, 54, 5192–5196. [Google Scholar] [CrossRef]
- Prasad, A.S.; Satyanarayana, B. Magnetically Recoverable Pd/Fe3O4 -Catalyzed Stille Cross-Coupling Reaction of Organostannanes with Aryl Bromides. Bull. Korean Chem. Soc. 2012, 33, 2789–2792. [Google Scholar] [CrossRef]
- Byun, S.; Chung, J.; Kwon, J.; Moon Kim, B. Mechanistic studies of magnetically recyclable Pd-Fe3O4 heterodimeric nanocrystal-catalyzed organic reactions. Chem. Asian J. 2015, 10, 982–988. [Google Scholar] [CrossRef]
- Sreedhar, B.; Kumar, A.S.; Yada, D. Magnetically recoverable Pd/Fecatalyzed hiyama cross-coupling of aryl bromides with aryl siloxanes. Synlett 2011, 8, 1081–1084. [Google Scholar] [CrossRef]
- Zhu, M.; Diao, G. Synthesis of porous Fe3O4 nanospheres and its application for the catalytic degradation of xylenol orange. J. Phys. Chem. C 2011, 115, 18923–18934. [Google Scholar] [CrossRef]
- Wang, H.Q.; Wei, X.; Wang, K.X.; Chen, J.S. Controlled synthesis of magnetic Pd/Fe3O4 spheres via an ethylenediamine-assisted route. Dalt. Trans. 2012, 41, 3204–3208. [Google Scholar] [CrossRef] [PubMed]
- Opanasenko, M.; Štěpnička, P.; Čejka, J. Heterogeneous Pd catalysts supported on silica matrices. RSC Adv. 2014, 4, 65137–65162. [Google Scholar] [CrossRef] [Green Version]
- Kainz, Q.M.; Linhardt, R.; Grass, R.N.; Vilé, G.; Pérez-Ramírez, J.; Stark, W.J.; Reiser, O. Palladium nanoparticles supported on magnetic carbon-coated cobalt nanobeads: Highly active and recyclable catalysts for alkene hydrogenation. Adv. Funct. Mater. 2014, 24, 2020–2027. [Google Scholar] [CrossRef]
- Pei, G.X.; Liu, X.Y.; Wang, A.; Li, L.; Huang, Y.; Zhang, T.; Lee, J.W.; Jang, B.W.L.; Mou, C.Y. Promotional effect of Pd single atoms on Au nanoparticles supported on silica for the selective hydrogenation of acetylene in excess ethylene. New J. Chem. 2014, 38, 2043–2051. [Google Scholar] [CrossRef]
- Eggenhuisen, T.M.; Den Breejen, J.P.; Verdoes, D.; De Jongh, P.E.; De Jong, K.P. Fundamentals of melt infiltration for the preparation of supported metal catalysts. The case of Co/SiO2 for Fischer-Tropsch synthesis. J. Am. Chem. Soc. 2010, 132, 18318–18325. [Google Scholar] [CrossRef]
- Rosario-Amorin, D.; Gaboyard, M.; Clérac, R.; Vellutini, L.; Nlate, S.; Heuzé, K. Metallodendritic grafted core-shell γ-Fe2O3 nanoparticles used as recoverable catalysts in Suzuki C-C coupling reactions. Chem. A Eur. J. 2012, 18, 3305–3315. [Google Scholar] [CrossRef]
- Liao, Y.; He, L.; Huang, J.; Zhang, J.; Zhuang, L.; Shen, H.; Su, C.Y. Magnetite nanoparticle-supported coordination polymer nanofibers: Synthesis and catalytic application in suzuki-miyaura coupling. ACS Appl. Mater. Interfaces 2010, 2, 2333–2338. [Google Scholar] [CrossRef]
- Gao, X.; Yu, K.M.K.; Tam, K.Y.; Tsang, S.C. Colloidal stable silica encapsulated nano-magnetic composite as a novel bio-catalyst carrier. Chem. Commun. 2003, 2998–2999. [Google Scholar] [CrossRef]
- Gardimalla, H.M.R.; Mandal, D.; Stevens, P.D.; Yen, M.; Gao, Y. Superparamagnetic nanoparticle-supported enzymatic resolution of racemic carboxylates. Chem. Commun. 2005, 4432–4434. [Google Scholar] [CrossRef] [PubMed]
- Gleeson, O.; Tekoriute, R.; Gun’ko, Y.K.; Connon, S.J. The First Magnetic Nanoparticle-Supported Chiral DMAP Analogue: Highly Enantioselective Acylation and Excellent Recyclability. Chem. A Eur. J. 2009, 15, 5669–5673. [Google Scholar] [CrossRef] [PubMed]
- Fuertes, A.B.; Valle-Vigón, P.; Sevilla, M. One-step synthesis of silica@resorcinol-formaldehyde spheres and their application for the fabrication of polymer and carbon capsules. Chem. Commun. 2012, 48, 6124–6126. [Google Scholar] [CrossRef] [PubMed]
- Yang, T.; Liu, J.; Zheng, Y.; Monteiro, M.J.; Qiao, S.Z. Facile fabrication of core-shell-structured Ag@carbon and mesoporous yolk-shell-structured Ag@carbon@silica by an extended stöber method. Chem. A Eur. J. 2013, 19, 6942–6945. [Google Scholar] [CrossRef] [PubMed]
- Liu, R.; Qu, F.; Guo, Y.; Yao, N.; Priestley, R.D. Au@carbon yolk-shell nanostructures via one-step core-shell-shell template. Chem. Commun. 2014, 50, 478–480. [Google Scholar] [CrossRef] [PubMed]
- Sun, C.; Sun, K.; Tang, S. Extended Stöber method to synthesize core-shell magnetic composite catalyst Fe3O4@C-Pd for Suzuki coupling reactions. Mater. Chem. Phys. 2018, 207, 181–185. [Google Scholar] [CrossRef]
- Lim, C.W.; Lee, I.S. Magnetically recyclable nanocatalyst systems for the organic reactions. Nano Today 2010, 5, 412–434. [Google Scholar] [CrossRef]
- Wu, W.; He, Q.; Jiang, C. Magnetic iron oxide nanoparticles: Synthesis and surface functionalization strategies. Nanoscale Res. Lett. 2008, 3, 397–415. [Google Scholar] [CrossRef]
- Franger, S.; Berthet, P.; Berthon, J. Electrochemical synthesis of Fe3O4 nanoparticles in alkaline aqueous solutions containing complexing agents. J. Solid State Electrochem. 2004, 8, 218–223. [Google Scholar] [CrossRef]
- Nazrul, I.M.; Phong, L.V.; Jeong, J.R.; Kim, C. A facile route to sonochemical synthesis of magnetic iron oxide (Fe3O4) nanoparticles. Thin Solid Films 2011, 519, 8277–8279. [Google Scholar] [CrossRef]
- Deng, Y.; Wang, L.; Yang, W.; Fu, S.; Elaïssari, A. Preparation of magnetic polymeric particles via inverse microemulsion polymerization process. J. Magn. Magn. Mater. 2003, 257, 69–78. [Google Scholar] [CrossRef]
- Jiao, H.; Yang, H.Q. Controlled synthesis and magnetic properties of Fe3O4 walnut spherical particles and octahedral microcrystals. Sci. China Ser. E Technol. Sci. 2008, 51, 1911–1920. [Google Scholar] [CrossRef]
- Maleki, A.; Kamalzare, M.; Aghaei, M. Efficient one-pot four-component synthesis of 1,4-dihydropyridines promoted by magnetite/chitosan as a magnetically recyclable heterogeneous nanocatalyst. J. Nanostruct. Chem. 2015, 5, 95–105. [Google Scholar] [CrossRef]
- Safari, J.; Javadian, L. Chitosan decorated Fe3O4 nanoparticles as a magnetic catalyst in the synthesis of phenytoin derivatives. RSC Adv. 2014, 4, 48973–48979. [Google Scholar] [CrossRef]
- Zarnegar, Z.; Safari, J. Fe3O4@chitosan nanoparticles: A valuable heterogeneous nanocatalyst for the synthesis of 2,4,5-trisubstituted imidazoles. RSC Adv. 2014, 4, 20932–20939. [Google Scholar] [CrossRef]
- Shelke, S.N.; Bankar, S.R.; Mhaske, G.R.; Kadam, S.S.; Murade, D.K.; Bhorkade, S.B.; Rathi, A.K.; Bundaleski, N.; Teodoro, O.M.N.D.; Zboril, R.; et al. Iron oxide-supported copper oxide nanoparticles (Nanocat-Fe-CuO): Magnetically recyclable catalysts for the synthesis of pyrazole derivatives, 4-methoxyaniline, and ullmann-type condensation reactions. ACS Sustain. Chem. Eng. 2014, 2, 1699–1706. [Google Scholar] [CrossRef]
- Hui, C.; Shen, C.; Tian, J.; Bao, L.; Ding, H.; Li, C.; Tian, Y.; Shi, X.; Gao, H.-J. Core-shell Fe3O4@SiO2 nanoparticles synthesized with well-dispersed hydrophilic Fe3O4 seeds. Nanoscale 2011, 3, 701–705. [Google Scholar] [CrossRef]
- An, Q.; Yu, M.; Zhang, Y.; Ma, W.; Guo, J.; Wang, C. Fe3O4@carbon microsphere supported Ag-Au bimetallic nanocrystals with the enhanced catalytic activity and selectivity for the reduction of nitroaromatic compounds. J. Phys. Chem. C 2012, 116, 22432–22440. [Google Scholar] [CrossRef]
- Zhang, X.; Niu, C.; Wang, Y.; Zhou, S.; Liu, J. Gel-limited synthesis of dumbbell-like Fe3O4 -Ag composite microspheres and their SERS applications. Nanoscale 2014, 6, 12618–12625. [Google Scholar] [CrossRef]
- Li, W.P.; Liao, P.Y.; Su, C.H.; Yeh, C.S. Formation of oligonucleotide-gated silica shell-coated Fe3O4-Au core-shell nanotrisoctahedra for magnetically targeted and near-infrared light-responsive theranostic platform. J. Am. Chem. Soc. 2014, 136, 10062–10075. [Google Scholar] [CrossRef]
- Kirkeminde, A.; Ren, S. Interdiffusion induced exchange coupling of L10-FePd/α-Fe magnetic nanocomposites. Nano Lett. 2014, 14, 4493–4498. [Google Scholar] [CrossRef] [PubMed]
- Hoseini, S.J.; Heidari, V.; Nasrabadi, H. Magnetic Pd/Fe3O4/reduced-graphene oxide nanohybrid as an efficient and recoverable catalyst for Suzuki-Miyaura coupling reaction in water. J. Mol. Catal. A Chem. 2015, 396, 90–95. [Google Scholar] [CrossRef]
- Yeo, K.M.; Lee, S.I.; Lee, Y.T.; Chung, Y.K.; Lee, I.S. Core–Satellite Heterostruture of Fe3O4 –Pd Nanocomposite: Selective and Magnetically Recyclable Catalyst for Decarboxylative Coupling Reaction in Aqueous Media. Chem. Lett. 2007, 37, 116–117. [Google Scholar] [CrossRef]
- Barder, T.E.; Walker, S.D.; Martinelli, J.R.; Buchwald, S.L. Catalysts for Suzuki-Miyaura coupling processes: Scope and studies of the effect of ligand structure. J. Am. Chem. Soc. 2005, 127, 4685–4696. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Yi, W.B.; Cai, C. Fluorous silica gel-supported perfluoro-tagged palladium nanoparticles: An efficient and reusable catalyst for direct C-2 arylation of indoles. Chem. Commun. 2011, 47, 806–808. [Google Scholar] [CrossRef]
- Panda, B.; Sarkar, T.K. Gold and palladium combined for the Sonogashira-type cross-coupling of arenediazonium salts. Chem. Commun. 2010, 46, 3131–3133. [Google Scholar] [CrossRef]
- Woo, H.; Park, K.H. Recent developments in hybrid iron oxide–noble metal nanocatalysts for organic reactions. Catal. Today 2016, 278, 209–226. [Google Scholar] [CrossRef]
- Alayoglu, S.; Zavalij, P.; Eichhorn, B.; Wang, Q.; Frenkel, A.I.; Chupas, P. Structural and Architectural Evaluation of Bimetallic Nanoparticles: A Case Study of Pt−Ru Core−Shell and Alloy Nanoparticles. ACS Nano 2009, 3, 3127–3137. [Google Scholar] [CrossRef]
- Clemens, B.; Xiaobo, C.; Radha, N.; Mostafa, A.E.S. Chemistry and Properties of Nanocrystals of Different Shapes. Chem. Rev. 2005, 105, 1025–1102. [Google Scholar]
- Woo, H.; Kim, E.; Kim, J.-H.; Yun, S.-W.; Park, J.C.; Kim, Y.-T.; Park, K.H. Shape and Composition Control of Monodisperse Hybrid Pt-CoO Nanocrystals by Controlling the Reaction Kinetics with Additives. Sci. Rep. 2017, 7, 3851. [Google Scholar] [CrossRef]
- Gowda, R.R.; Chen, E.Y.-X. Recyclable Earth-Abundant Metal Nanoparticle Catalysts for Selective Transfer Hydrogenation of Levulinic Acid to Produce γ-Valerolactone. ChemSusChem 2016, 9, 181–185. [Google Scholar] [CrossRef] [PubMed]
- Park, K.H.; Jung, I.G.; Kim, S.Y.; Chung, Y.K. Immobilized Cobalt/Rhodium Heterobimetallic Nanoparticle-Catalyzed Silylcarbocylization and Carbonylative Silylcarbocyclization of 1,6-Enynes. Org. Lett. 2003, 5, 4967–4970. [Google Scholar] [CrossRef] [PubMed]
- Kang, D.; Ko, J.H.; Choi, J.; Cho, K.; Lee, S.M.; Kim, H.J.; Ko, Y.-J.; Park, K.H.; Son, S.U. Dual role of Cu2O nanocubes as templates and networking catalysts for hollow and microporous Fe-porphyrin networks. Chem. Commun. 2017, 53, 2598–2601. [Google Scholar] [CrossRef] [PubMed]
- Zhan, Y.; Xu, C.; Lu, M.; Liu, Z.; Lee, J.Y. Mn and Co co-substituted Fe3O4 nanoparticles on nitrogen-doped reduced graphene oxide for oxygen electrocatalysis in alkaline solution. J. Mater. Chem. A 2014, 2, 16217–16223. [Google Scholar] [CrossRef]
- Wang, Z.; Chen, M.; Shu, J.; Li, Y. One-step solvothermal synthesis of Fe3O4@Cu@Cu2O nanocomposite as magnetically recyclable mimetic peroxidase. J. Alloys Compd. 2016, 682, 432–440. [Google Scholar] [CrossRef]
















| Entry | Pd added b | Temp (°C) | Time (h) | Yield c (%) |
|---|---|---|---|---|
| 1 | 0.054 | 110 | 3 | 93 |
| 2 | 0.108 | 110 | 3 | 99 |
| 3 | 0.162 | 110 | 3 | 99 |
| 4 | 0.108 | 60 | 3 | 19.8 |
| 5 | 0.108 | 70 | 3 | 34.5 |
| 6 | 0.108 | 80 | 3 | 51.3 |
| 7 | 0.108 | 80 | 6 | 57.1 |
| 8 | 0.108 | 80 | 24 | 91.2 |
| 9 | 0.108 | 90 | 3 | 69.2 |
| 10 | 0.108 | 100 | 3 | 85.4 |
| Entry | Catalysts | Temp (°C) | Time (h) | Solvent | Conv. a (%) | Product Time Yield (gproduct gPd−1 h−1) |
|---|---|---|---|---|---|---|
| 1 | Pd/Fe3O4/charcoal | 150 | 30 | Toluene/H2O (4:1) | 59 | 3.62 |
| 2 | Pd/Fe3O4/charcoal | 100 | 4 | Toluene/H2O (4:1) | 4 | 1.84 |
| 3 | Pd/Fe3O4/charcoal | 100 | 4 | DMSO/H2O (4:1) | 25 | 12.0 |
| 4 | Pd/Fe3O4/charcoal | 100 | 4 | THF/H2O (4:1) | 35 | 15.1 |
| 5 | Pd/Fe3O4/charcoal (Pd 0.5 mol %) | 100 | 4 | DMF/H2O (4:1) | 60 | 55.2 |
| 6 | Pd/Fe3O4/charcoal | 100 | 2 | DMF/H2O (4:1) | 60 | 55.2 |
| 7 | Pd/charcoal b | 100 | 4 | DMF/H2O (4:1) | 76 | 35.0 |
| 8 | Pd/Fe3O4/charcoal | 100 | 4 | DMF/H2O (4:1) | >99 | 36.0 |
| 9 | Fe3O4/charcoal | 100 | 4 | DMF/H2O (4:1) | No reaction | |
| 10 | Commercial Pd/charcoal | 100 | 4 | DMF/H2O (4:1) | 31 | 14.3 |
| Entry | Aryl Halide | Arylboronic Acid | Product | Yield a (%) | Product Time Yield (gproduct gPd−1 h−1) |
|---|---|---|---|---|---|
| 1 |  |  |  | >99 | 36.2 |
| 2 |  |  |  | >99 | 36.3 |
| 3 |  |  |  | >99 | 36.2 |
| 4 |  |  |  | >99 | 43.1 |
| 5 |  |  |  | 45 | 20.9 |
| 6 |  |  |  | 64 | 37.9 |
| 7 |  |  |  | 48 | 18.9 |
| 8 |  |  |  | 99 | 48.6 |
| 9 |  |  |  | 83 | 35.5 |
| 10 |  |  |  | 90 | 38.5 |
| Recycle Run | Temp (°C) | Time (h) | Solvent | Conv. a (%) | Product Yield Time (gproduct gPd−1 h−1) |
|---|---|---|---|---|---|
| 1 | 100 | 4 | DMF/H2O (4:1) | >99 | 46.0 |
| 2 | 100 | 4 | DMF/H2O (4:1) | >99 | 46.0 |
| 3 | 100 | 4 | DMF/H2O (4:1) | >99 | 46.0 |

| Entry | A | Halogen | Time (h) | Yield (%) |
|---|---|---|---|---|
| 1 | H | Br | 6 | 92.22 |
| 2 | H | Cl | 12 | 63.74 |
| 3 | NO2 | Br | 6 | 94.51 |
| 4 | OCH3 | Br | 6 | 87.12 |
| Recycle Run | Capping Agent | Average Size (nm) |
|---|---|---|
| 1 | Na3Cit | 3.3 ± 0.24 |
| 2 | PEG | 3.4 ± 0.21 |
| 3 | PVP | 4.1 ± 0.43 |
| 4 | No capping agent | 4.7 ± 0.35 |

| Entry | Cat. (mol %) | Temp (°C) | Time (h) | Base | Solvent | Yield b (%) |
|---|---|---|---|---|---|---|
| 1 | 1 (Na3Cit) | 80 | 5 | K2CO3 | DMF/H2O (4:1) | 91 |
| 2 | 1 (Na3Cit) | 50 | 5 | K2CO3 | H2O | 97 |
| 3 | 0.1 (Na3Cit) | 50 | 5 | K2CO3 | H2O | 65 |
| 4 | 0.05 (Na3Cit) | 50 | 5 | Cs2CO3 | H2O | 89 |
| 5 | 0.05 (Na3Cit) | 50 | 7 | Cs2CO3 | H2O | 98 |
| 6 | 0.05 (Na3Cit) | 50 | 5 | CsOH | H2O | 66 |
| 7 | 0.05 (Na3Cit) | 40 | 12 | Cs2CO3 | H2O | 48 |
| 8 | 0.05 (Na3Cit) | 40 | 24 | Cs2CO3 | H2O | 94 |
| 9 | 0.1 (Na3Cit) | 40 | 12 | Cs2CO3 | H2O | 76 |
| 10 | 0.05 (Na3Cit) | 100 | 1 | Cs2CO3 | H2O | 80 |
| 11 | 0.05 (Na3Cit) | 100 | 1.5 | Cs2CO3 | H2O | 98 |
| 12 | 0.05 (Na3Cit) | 25 | 24 | Cs2CO3 | H2O | 13 |
| 13 | 0.05 (PEG) | 50 | 7 | Cs2CO3 | H2O | 89 |
| 14 | 0.05 (No) | 50 | 7 | Cs2CO3 | H2O | 57 |
| 15 | 0.05 (PVP) | 50 | 7 | Cs2CO3 | H2O | 37 |

| Entry | Cat. (mol %) | Temp (°C) | Time (h) | Base | Solvent | Conv. (%) a |
|---|---|---|---|---|---|---|
| 1 | 1 | 120 | 18 | Piperidine | DMF | 72 |
| 2 | 1 | 120 | 18 | Piperidine | NMP | 76 |
| 3 | 1 | 120 | 18 | Piperidine | DMSO | 99 |
| 4 | 1 | 120 | 18 | Cs2CO3 | DMSO | 73 |
| 5 | 1 | 120 | 18 | NaOAc | DMSO | 98 |
| 6 | 1 | 120 | 18 | K2CO3 | DMSO | 78 |
| 7 | 1 | 90 | 18 | Piperidine | DMSO | 92 |
| 8 | 0.5 | 120 | 3 | Piperidine | DMSO | 99 |
| 9 | 0.25 | 120 | 3 | Piperidine | DMSO | 94 |
| 10 | 0.5 | 120 | 1 | Piperidine | DMSO | 93 |
| Entry | Aryl Halide | Arylacetylene | Product | Conversion (%) a |
|---|---|---|---|---|
| 1 |  |  |  | 72 |
| 2 |  |  |  | 76 |
| 3 |  |  |  | 99 |
| 4 |  |  |  | 73 |
| 5 |  |  |  | 98 |
| 6 |  |  |  | 78 |
| 7 |  |  |  | 92 |
| 8 |  |  |  | 99 |
| 9 |  |  |  | 94 |

| Entry | Catalyst | Temp (°C) | Time (h) | Base | Conversion (%) a |
|---|---|---|---|---|---|
| 1 | Pd–Fe3O4 | 120 | 18 | Piperidine | Trace b |
| 2 | Pd–Fe3O4 | 120 | 18 | Piperidine | 3 c |
| 3 | Pd–Fe3O4 | 120 | 18 | Piperidine | 41 |
| 4 | Pd–Fe3O4 | 120 | 18 | LiOAc | 45 |
| 5 | Pd–Fe3O4 | 120 | 18 | CsOAc | 48 |
| 6 | Au/Pd–Fe3O4 | 120 | 18 | CsOAc | 57 |
| 7 | Au/Pd–Fe3O4 | 150 | 18 | CsOAc | 97 |
| 8 | Au/Pd–Fe3O4 | 150 | 9 | CsOAc | 97 |
| 9 | Au/Pd–Fe3O4 | 150 | 6 | CsOAc | 59 |
| 10 | Au/Pd–Fe3O4 | 150 | 9 | CsOAc | 38 d |
| SI No | Catalyst | Size (nm) | Morphology | Application |
|---|---|---|---|---|
| 1 | Pd–Fe3O4 | 8.7 | Spherical | Mizoroki–Heck reaction |
| 2 | Pd–Fe3O4 | 213 | Flower-like | Sonogashira coupling reaction |
| 3 | Au/Pd–Fe3O4 | 5.8 | Flower-like | Tandem synthesis reaction |

| Entry | Catalysts | Time (h) | Temp (°C) | Conv a (%) |
|---|---|---|---|---|
| 1 | Pd–Fe3O4 | 18 | 130 | 62 |
| 2 | Cu2O/Pd–Fe3O4 | 18 | 130 | 73 |
| 3 | Cu2O/Pd–Fe3O4 | 18 | 140 | 85 b |
| 4 | Cu2O/Pd–Fe3O4 | 9 | 140 | 84 b |
| 5 | Cu2O/Pd–Fe3O4 | 4.5 | 140 | 76 b |
| 6 | Cu2O/Pd–Fe3O4 | 9 | 140 | 76 b, c |
| 7 | Pd/charcoal | 4.5 | 140 | 69 b |
| 8 | Fe3O4/charcoal | 4.5 | 140 | 0 b |
| 9 | Cu2O | 4.5 | 140 | 0 b |
| Entry | Substrate | Conv. (%) | Entry | Substrate | Conv. (%) |
|---|---|---|---|---|---|
| 1 |  | 84 | 6 |  | 70 |
| 2 |  | 79 | 7 |  | 39 |
| 3 |  | 84 | 8 |  | 71 |
| 4 |  | 85 | 9 |  | 80 |
| 5 |  | 75 | 10 |  | 69 |

| Entry | Catalysts | Solvent | Base | Time (h) | Yield (%) a |
|---|---|---|---|---|---|
| 1 | CoO/Pd–Fe3O4 | MeOH | KOtBu | 12 | Trace |
| 2 | CoO/Pd–Fe3O4 | THF | KOtBu | 12 | 33 |
| 3 | CoO/Pd–Fe3O4 | THF | NaOMe | 12 | 36 |
| 4 | CoO/Pd–Fe3O4 | THF | Cs2CO3 | 12 | 67 |
| 5 | MnO/Pd–Fe3O4 | THF | Cs2CO3 | 12 | 29 |
| 6 | Pd–Fe3O4 | THF | Cs2CO3 | 12 | 35 |
| 7 | Pd/charcoal | THF | Cs2CO3 | 12 | Trace |
| 8 | CoO/Pd–Fe3O4 | THF | Cs2CO3 | 24 | 63 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jang, S.; Hira, S.A.; Annas, D.; Song, S.; Yusuf, M.; Park, J.C.; Park, S.; Park, K.H. Recent Novel Hybrid Pd–Fe3O4 Nanoparticles as Catalysts for Various C–C Coupling Reactions. Processes 2019, 7, 422. https://doi.org/10.3390/pr7070422
Jang S, Hira SA, Annas D, Song S, Yusuf M, Park JC, Park S, Park KH. Recent Novel Hybrid Pd–Fe3O4 Nanoparticles as Catalysts for Various C–C Coupling Reactions. Processes. 2019; 7(7):422. https://doi.org/10.3390/pr7070422
Chicago/Turabian StyleJang, Sanha, Shamim Ahmed Hira, Dicky Annas, Sehwan Song, Mohammad Yusuf, Ji Chan Park, Sungkyun Park, and Kang Hyun Park. 2019. "Recent Novel Hybrid Pd–Fe3O4 Nanoparticles as Catalysts for Various C–C Coupling Reactions" Processes 7, no. 7: 422. https://doi.org/10.3390/pr7070422
APA StyleJang, S., Hira, S. A., Annas, D., Song, S., Yusuf, M., Park, J. C., Park, S., & Park, K. H. (2019). Recent Novel Hybrid Pd–Fe3O4 Nanoparticles as Catalysts for Various C–C Coupling Reactions. Processes, 7(7), 422. https://doi.org/10.3390/pr7070422








