Effect of Nitric Acid Modification on Characteristics and Adsorption Properties of Lignite
Abstract
:1. Introduction
2. Experimental Parameters
2.1. Raw Materials and Experimental Coal Sample Preparation
2.2. Coal Sample Mineral Composition Analysis
2.3. Coal Sample Pore Structure Characterization
2.4. Coal Sample Surface Morphology Analysis
2.5. Coal Sample Surface Functional Group Analysis
2.6. Coal Surface Potential Test
2.7. Analysis of the Existence of the Types of Coal Surface Elements
2.8. Determination of the Adsorption Capacity of Pb2+ by Coal Samples
3. Results and Discussion
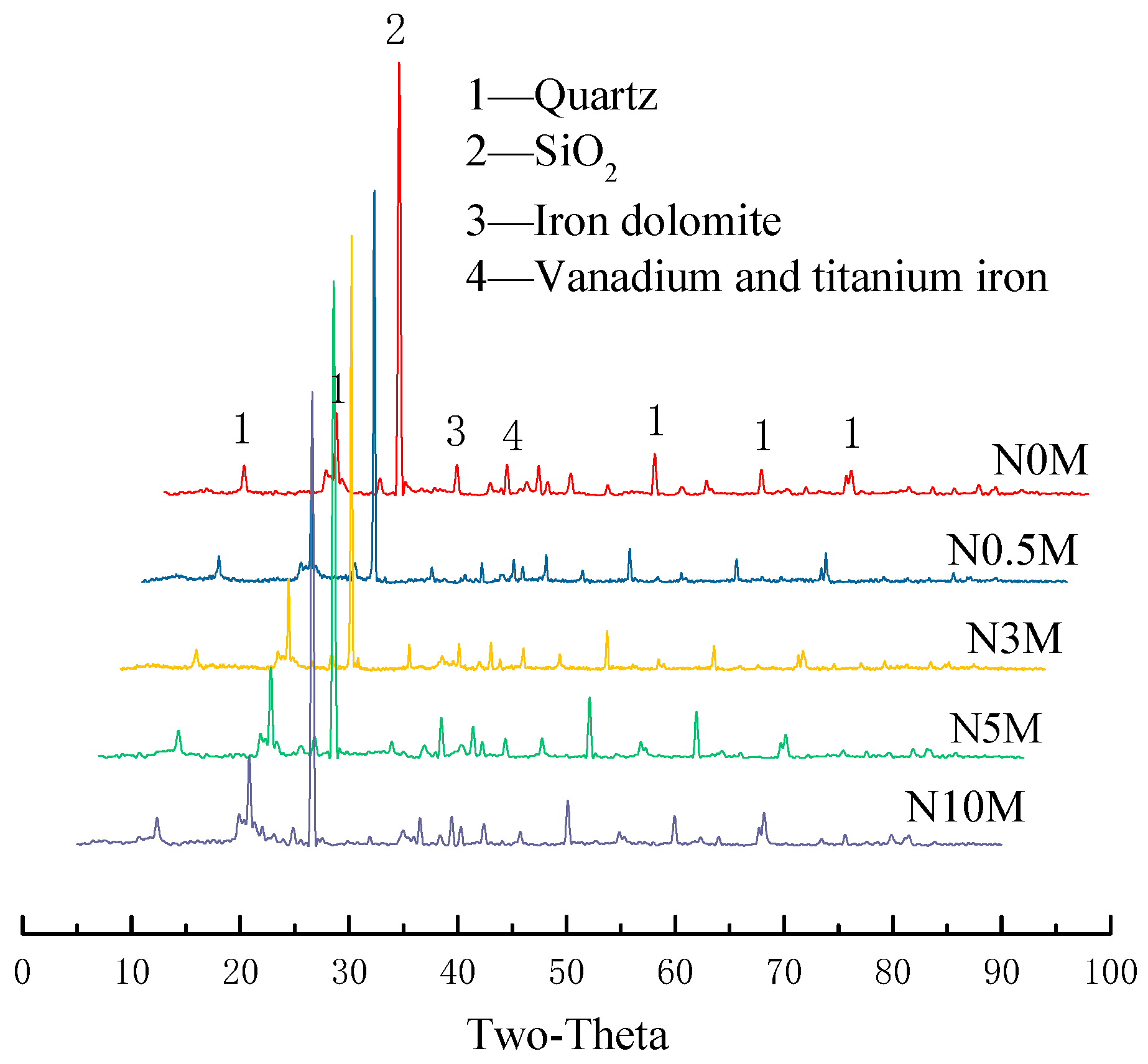
3.1. Effects of Different Modified Concentrations of Nitric Acid on the Mineral Composition of Lignite
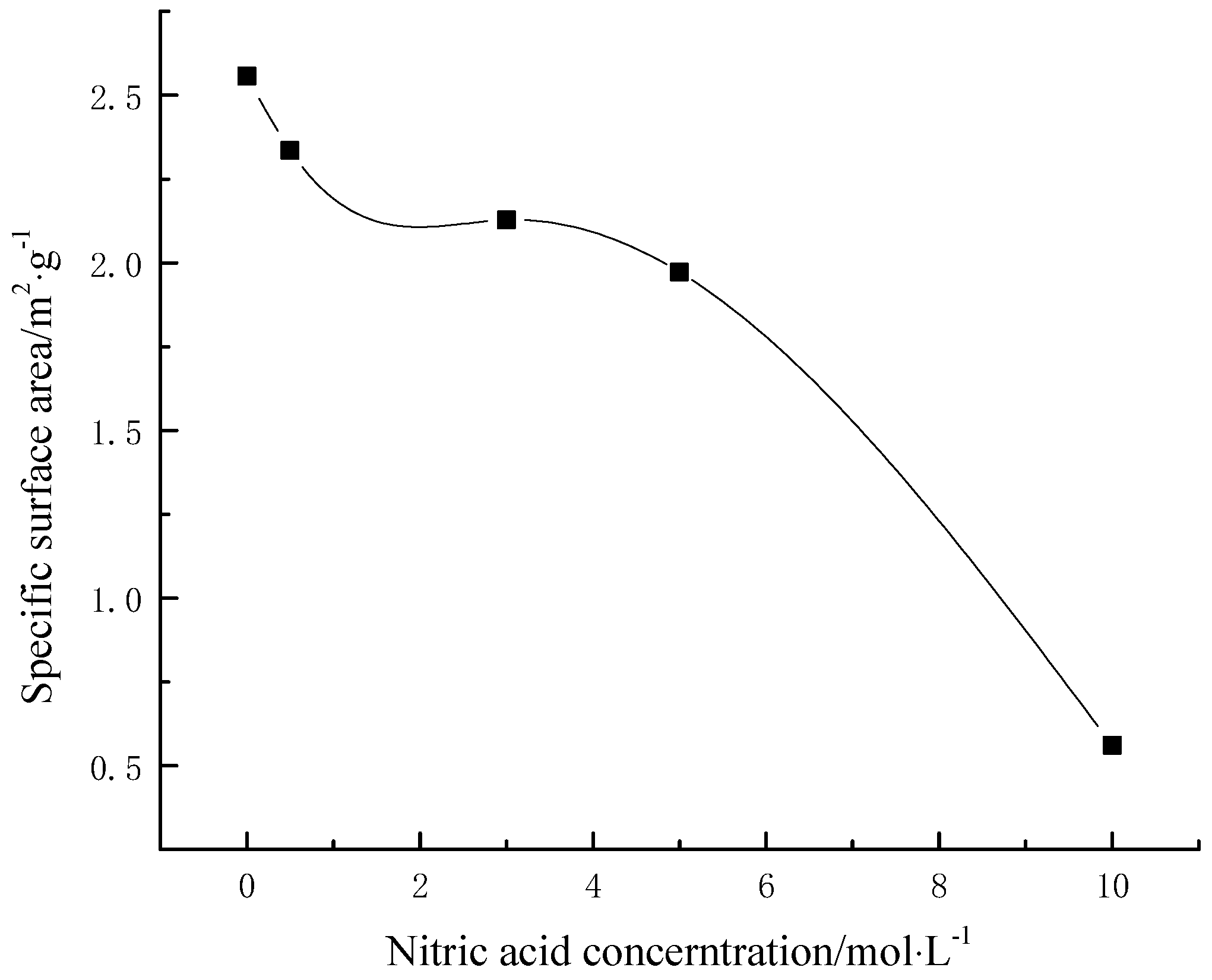
3.2. Effects of Different Modified Concentrations of Nitric Acid on the Specific Surface Area of Lignite
3.3. Effects of Different Modified Concentrations of Nitric Acid on the Pore Shape of Lignite
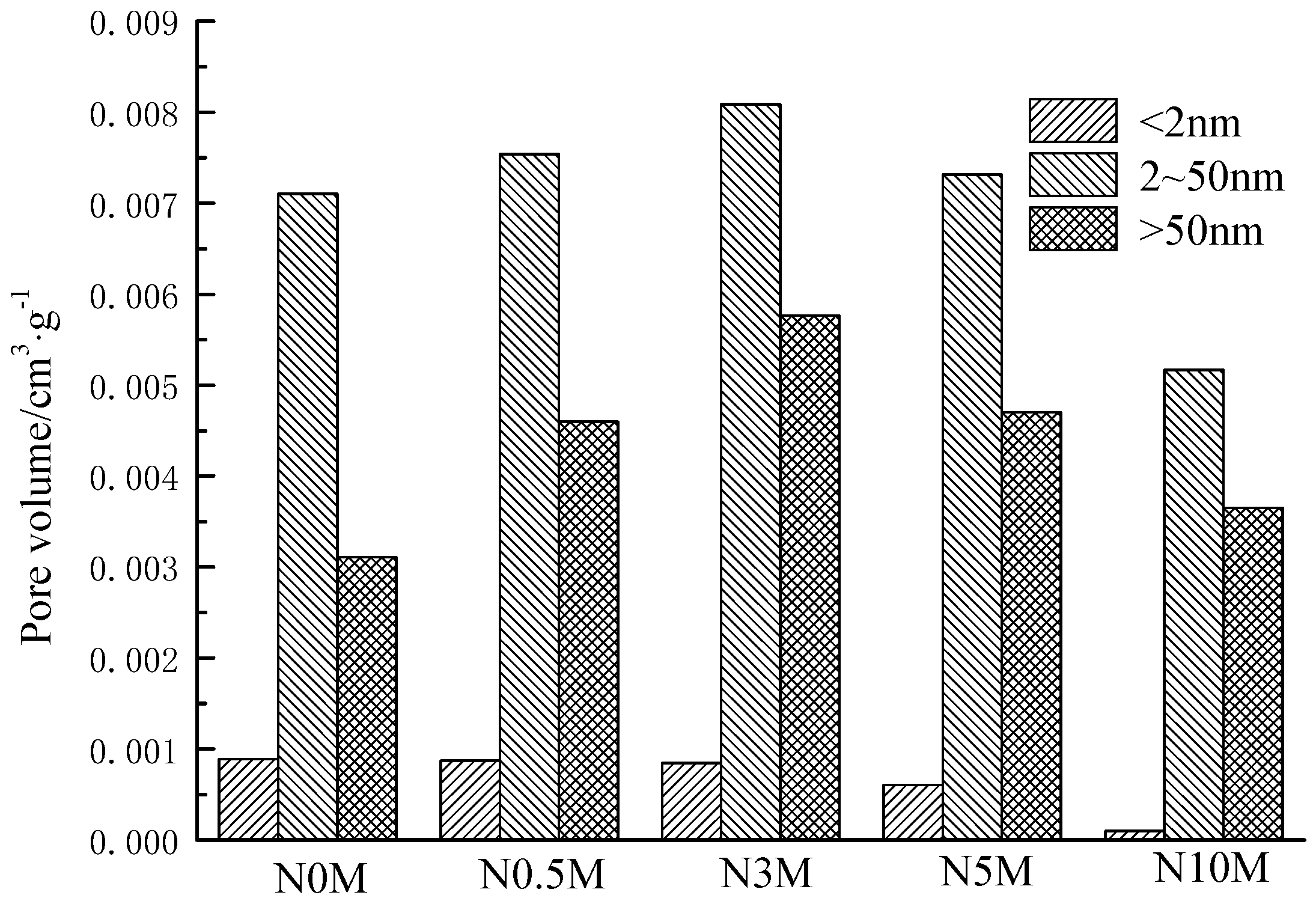
3.4. Effects of Different Modified Concentrations of Nitric Acid on the Structural Characteristics of Lignite Pores
3.5. Effects of Different Modified Concentrations of Nitric Acid on the Surface Morphology of Lignite
3.6. Effects of Different Concentrations of Nitric Acid on the Surface Functional Groups of Lignite
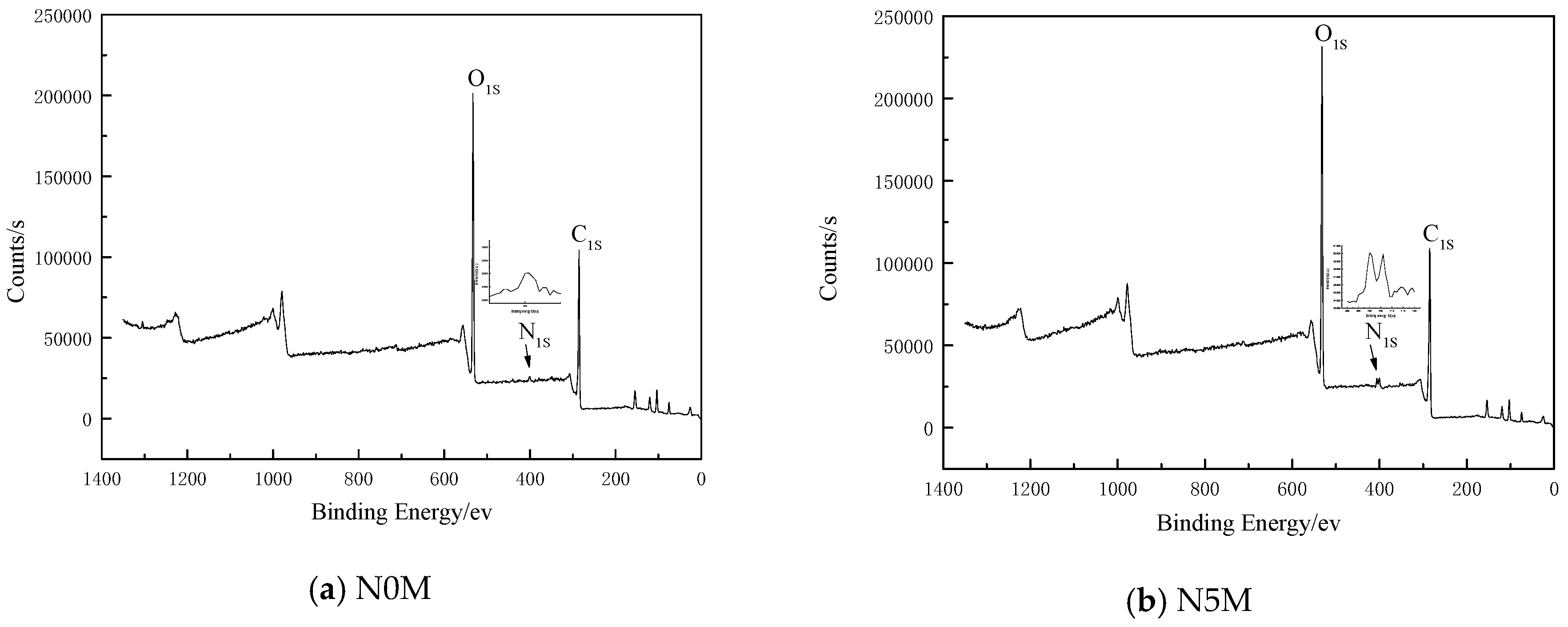
3.7. Effects of Nitric Acid Modification on the Types of Elements Existing on Coal Surfaces
3.7.1. Effects of Nitric Acid Modification on the Distribution of Chemical Functional Groups on the Lignite Surface
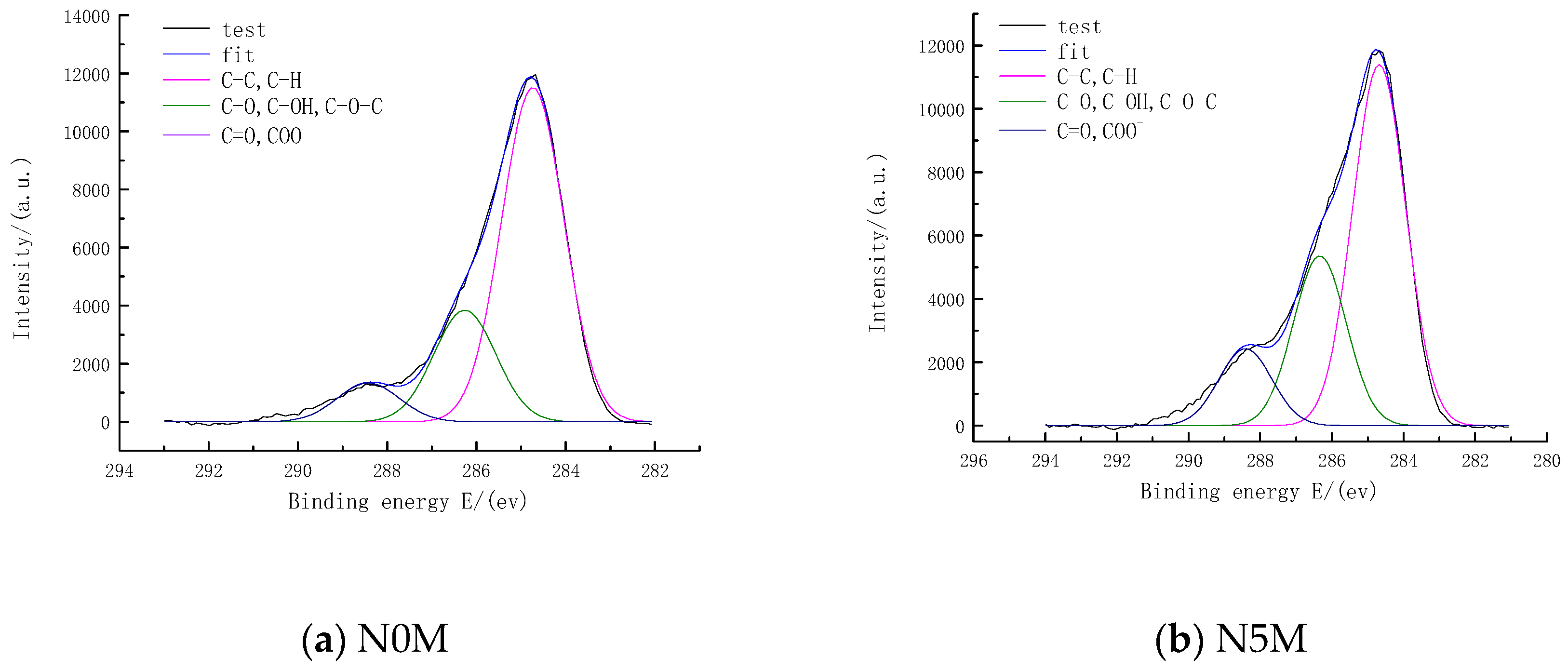
3.7.2. Distribution of Carbon Chemical Functional Groups of Lignite and Modified Lignite Surfaces
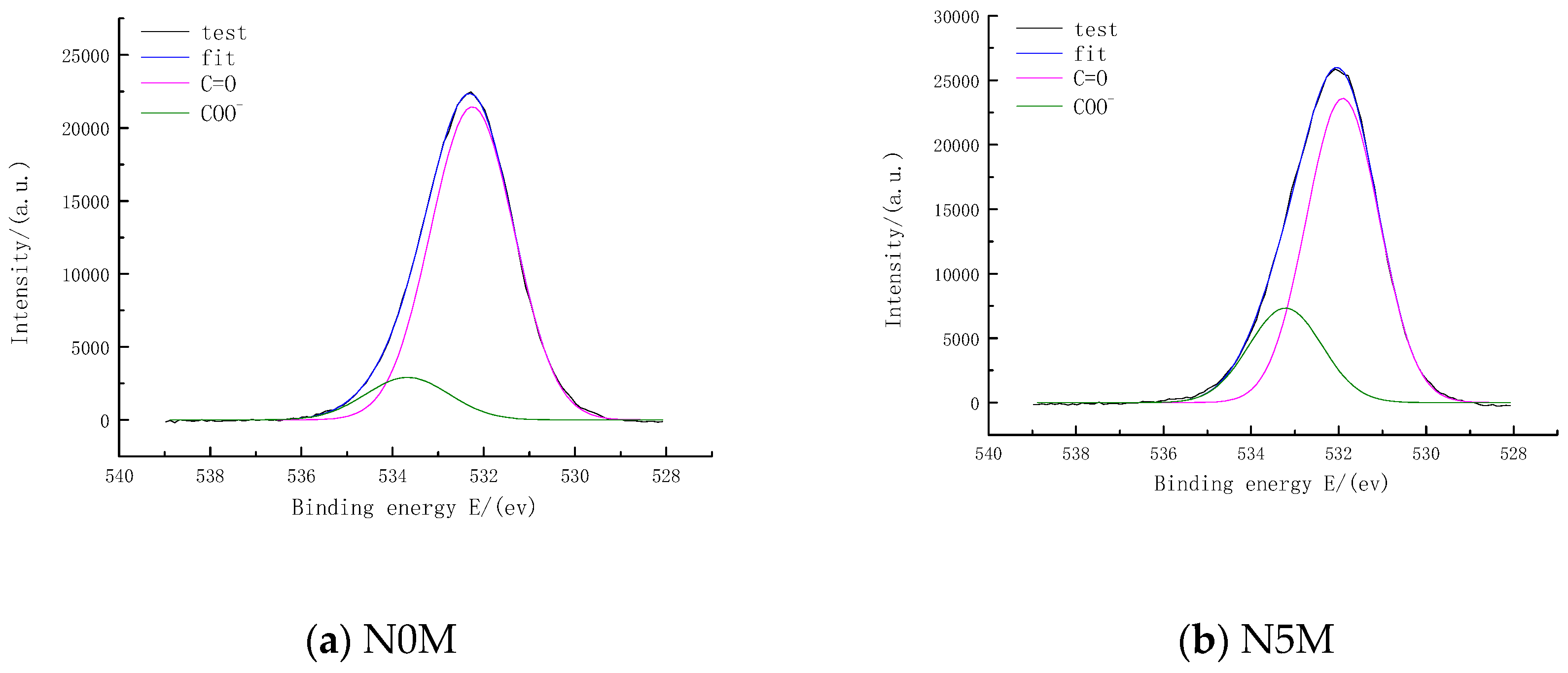
3.7.3. Distribution of Oxygen Chemical Functional Groups of Lignite and Modified Lignite Surfaces
3.7.4. Distribution of Nitrogen Chemical Functional Groups of Lignite and Modified Lignite Surfaces
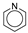 (398.0~398.1 eV), pyrrole nitrogen
(398.0~398.1 eV), pyrrole nitrogen 
 (400.0~400.4 eV), protonated pyridine
(400.0~400.4 eV), protonated pyridine 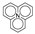 (401.6 eV), and nitrogen oxides
(401.6 eV), and nitrogen oxides  (402~405 eV) [21,22].
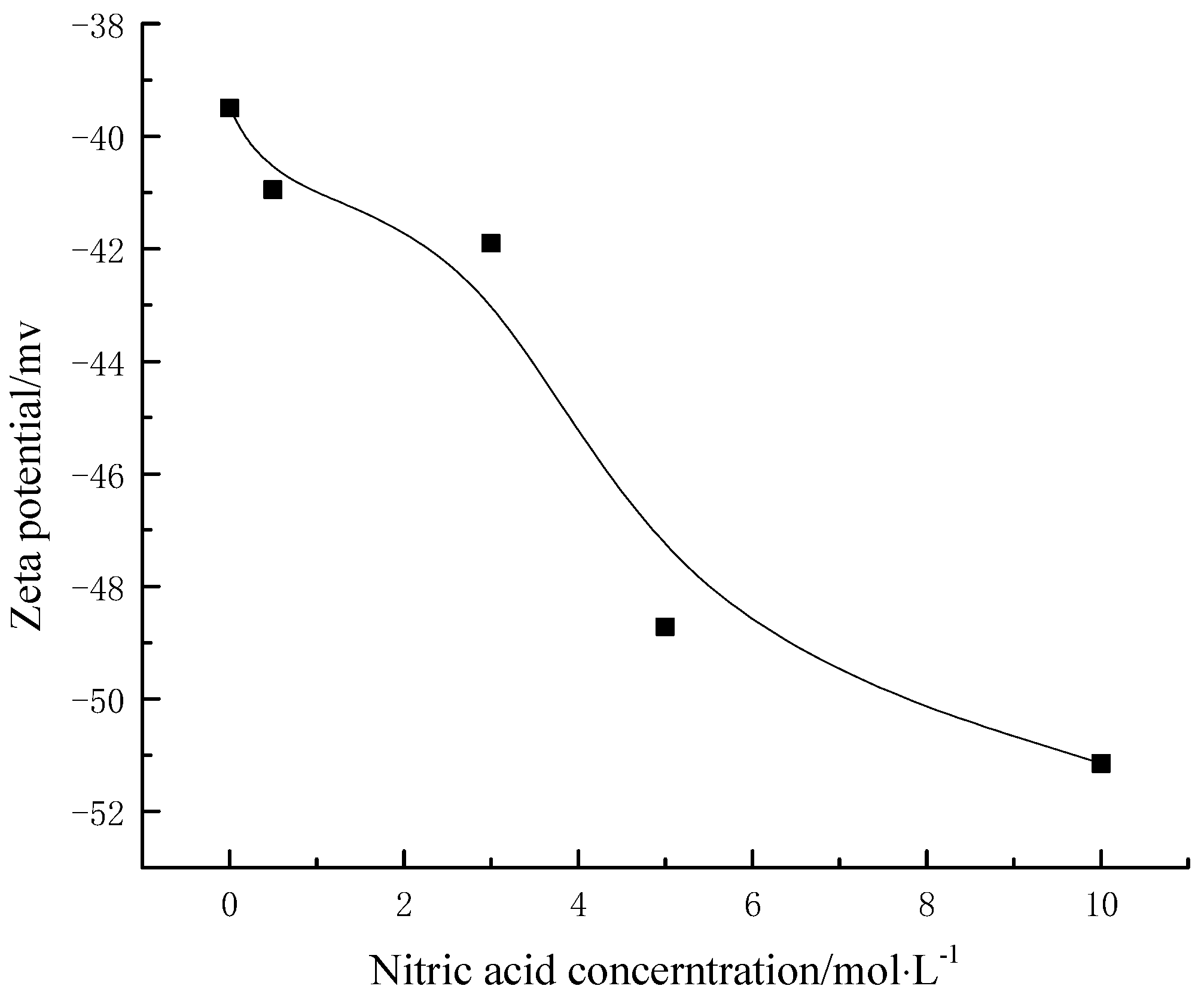
(402~405 eV) [21,22].3.8. Effects of Different Modified Concentrations of Nitric Acid on the Surface Potential of Lignite
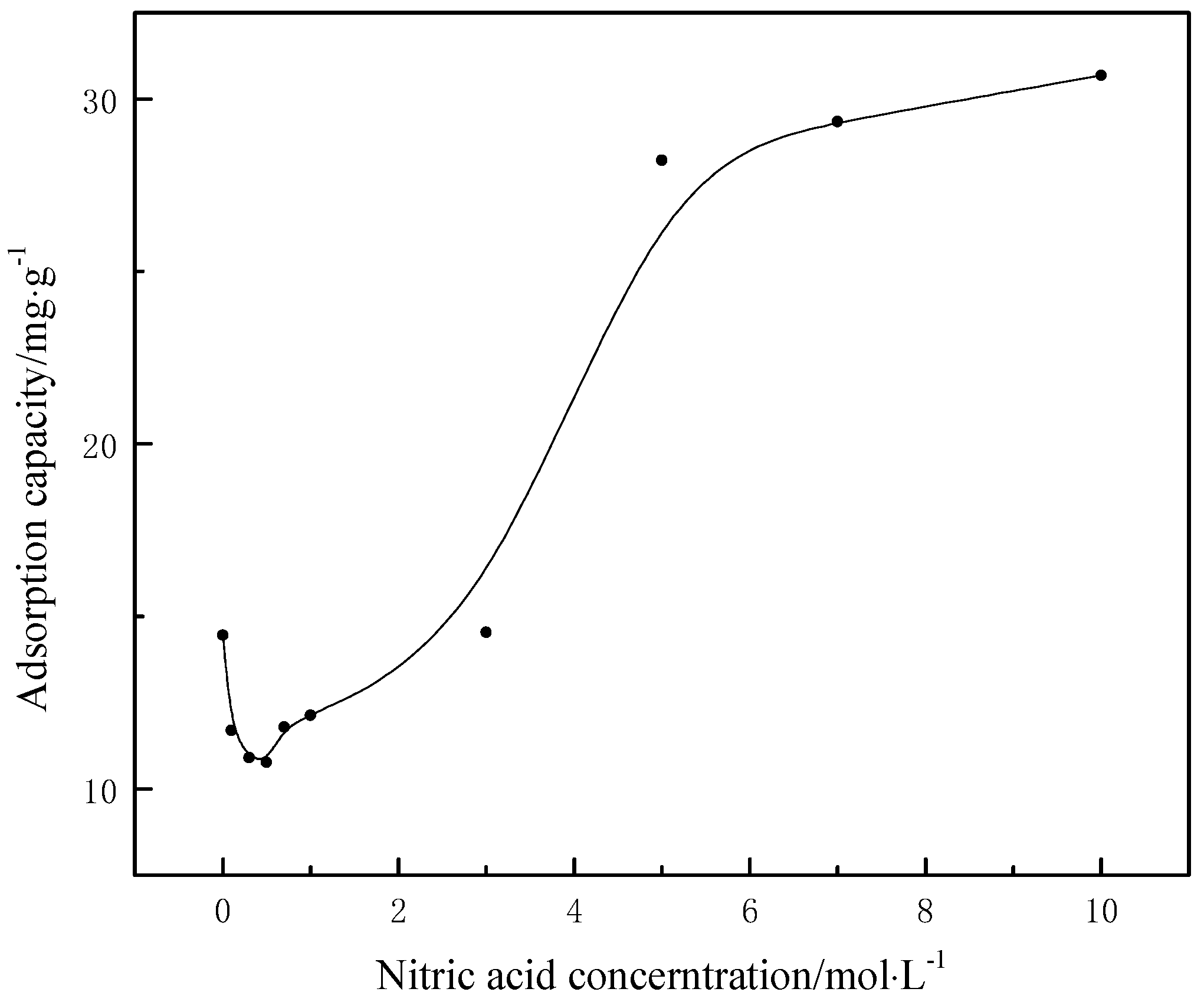
3.9. Adsorption Capacity of Pb2+ on Different Modified Coal Samples
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Ping, Z. Production and Application of Coal Humic Acid; Chemical Industry Press: Beijing, China, 1991; p. 28. [Google Scholar]
- Shuying, W.; Jinghong, L.; Yi, S. Adsorption Character of ShuLan Brown Coal for Heavy Metal Ions Such As Ni2+, Zn2+ and Pb2+. J. Northeast China Inst. Electr. Power Eng. 2004, 24, 85–91. [Google Scholar]
- Lumin, W.; Changliang, D.; Shujing, Y. Study of Longkou Lignite to Purify of Mercury-containing Waste Water. J. Yantai Univ. (Natl. Sci. Eng.) 1998, 2, 153–156. [Google Scholar]
- Polat, H.; Mlva, M.; Polat, M. Capacity and mechnism of phenol adsorption on lignite. Int. J. Miner. Proc. 2006, 79, 264–273. [Google Scholar] [CrossRef]
- DaoCheng, L.; JunFeng, L.; ZhongYuan, L. Study on the Treatment of Chromium (VI)-containing Wastewater Use Sulfonated Lignite. Coal Chem. Ind. 2010, 38, 36–38. [Google Scholar]
- Choudhury, D.; Srivastava, S.K.; Prasad, R. Removal of heavy metals in wastewater by its adsorption: On nitric acid oxidized coal. Indian J. Environ. Prot. 2013, 33, 804–815. [Google Scholar]
- Kus, J.; Misz-Kennan, M. Coal weathering and laboratory (artificial) coal oxidation. Int. J. Coal Geol. 2017, 171, 12–36. [Google Scholar] [CrossRef]
- Ward, C.R. Mineral matter in triassic and tertiary low-rank coals from South Australia. Int. J. Coal Geol. 1992, 20, 185–208. [Google Scholar] [CrossRef]
- Aparna, M.; Srivastava, S.K.; Udaybhanu, G. Removal of cadmium ions from waste water using raw and treated coals as adsorbents. Int. J. Chem. Sci. 2011, 9, 265–280. [Google Scholar]
- Rouquerol, J.; Avnir, D.; Fairbridge, C.W.; Everett, D.H.; Haynes, J.M.; Pernicone, N.; Ramsay, J.D.F.; Sing, K.S.W.; Unger, K.K. Physical and biophysical chemistry division commission on colloid and surface chemistry including catalysis. Pure Appl. Chem. 1994, 66, 1739–1758. [Google Scholar] [CrossRef]
- Sing, K.S.W.; Everett, D.H.; Haul, R.A.W.; Moscou, L.; Pierotti, R.A.; Rouquerol, J.; Siemieniewska, T. Reporting Physisorption Data for Gas/Solid Systems with Special Reference to the Determination of Surface Area and Porosity. Pure Appl. Chem. 1985, 57, 603–619. [Google Scholar] [CrossRef]
- Chunzhu, L. Victoria Lignite Science Progress; Chemical Industry Press: Beijing, China, 2009; p. 26. [Google Scholar]
- Yuliani, G.; Garnier, G.; Chaffee, A.L. Utilization of raw and dried Victorian brown coal in the adsorption of model dyes from solution. J. Water Proc. Eng. 2017, 15, 43–48. [Google Scholar] [CrossRef]
- Hong, Z.; Hulin, L.; Zeshen, O.; Wang, D.Z. Study on Surface Modification of Different Rank Coals by Using FTIR. J. China Univ. Min. Technol. 2001, 30, 365–369. [Google Scholar]
- Xuedong, Z.; Zibin, Z.; Chongjia, H.; Chengfang, Z. Quantitative Determination of Oxygen-containing Functional Groups in Coal by FTIR Spectroscopy. J. Fuel Chem. Technol. 1999, 4, 335–339. [Google Scholar]
- Xuqin, D.; Zune, W.; Chunbao, S. Surface Properties of Macerals from Shenfu Coal. J. China Univ. Min. Technol. 2007, 36, 630–635. [Google Scholar]
- Chen, S.; Tang, L.; Tao, X.; Chen, L.; Yang, Z.; Li, L. Effect of oxidation processing on the surface properties and floatability of Meizhiyou long-flame coal. Fuel 2017, 210, 177–186. [Google Scholar] [CrossRef]
- Xuqin, D.; Zuna, W. XPS analysis of oxygen contained functional group in coal macerals. J. Liaoning Tech. Univ. (Natl. Sci.) 2010, 29, 498–501. [Google Scholar]
- Biniak, S.; Szymański, G.; Siedlewski, J.; Świątkowski, A. The characterization of activated carbons with oxygen and nitrogen surface groups. Carbon 1997, 35, 1799–1810. [Google Scholar] [CrossRef]
- László, K.; Tombácz, E.; Josepovits, K. Effect of activation on the surface chemistry of carbons from polymer precursors. Carbon 2001, 39, 1217–1228. [Google Scholar] [CrossRef]
- Mei, L.; Junhe, Y.; Qifeng, Z.; Hai-zhou, C.; Hui, S. XPS study on transformation of N- and S-functional groups during pyrolysis of high sulfur New Zealand coal. J. Fuel Chem. Technol. 2013, 41, 1287–1293. [Google Scholar]
- Kelemen, S.R.; Gorbaty, M.L.; Kwiatek, P.J. Quantification of Nitrogen Forms in Argonne Premium Coals. Energy Fuels 1994, 8, 896–906. [Google Scholar] [CrossRef]
- Qihui, L.; Guoguang, W.; Zhiqiang, S.; Guangyou, W.; Hongfeng, X.; Yongjian, Z. Study on the influence of coalification degree and particle size on zeta potential of coal surface. Energy Technol. Manag. 2007, 3, 81–82. [Google Scholar]
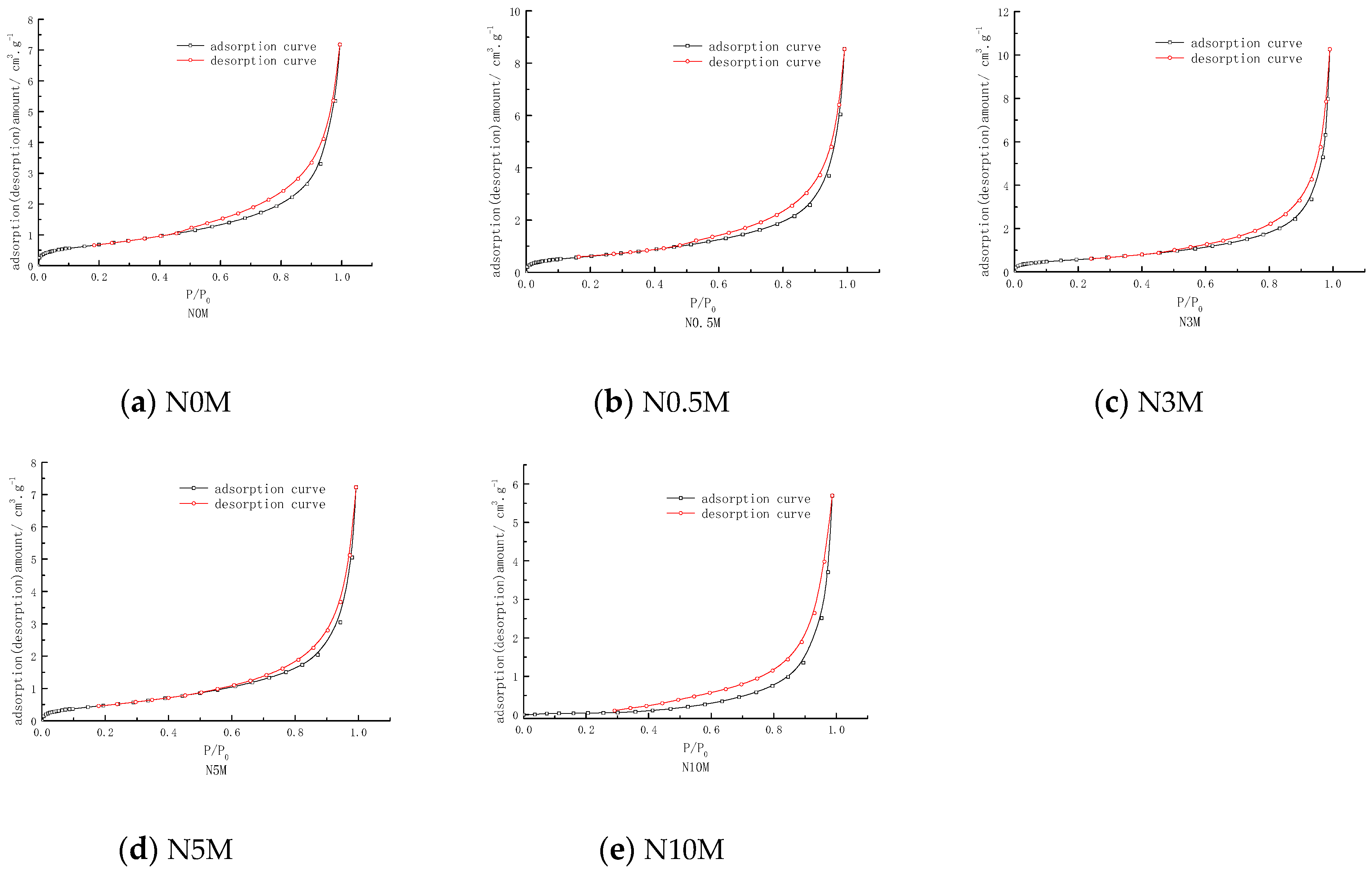



| Content | Mad | Aad | Vad | FCad |
|---|---|---|---|---|
| Wt/% | 8.72 | 10.98 | 35.65 | 44.65 |
| Samples | Total Pore Volume/(cm3/g) | Average Pore Diameter/(nm) | Most Probable Pore Size/(nm) | Microporous Volume/(cm3/g) | Microporous Most Probable Pore Size/(nm) |
|---|---|---|---|---|---|
| N0M | 0.01110 | 17.36602 | 2.856 | 0.00089 | 1.07210 |
| N0.5M | 0.01321 | 22.62922 | 3.188 | 0.00084 | 1.07615 |
| N3M | 0.01588 | 23.89515 | 3.150 | 0.00079 | 1.01992 |
| N5M | 0.01219 | 25.85776 | 2.539 | 0.00065 | 1.03273 |
| N10M | 0.00882 | 68.80520 | 6.290 | 0.000098 | 1.01285 |
| Samples | N0M | N5M | |||||
|---|---|---|---|---|---|---|---|
| Parameter | C1S | N1S | O1S | C1S | N1S | O1S | |
| Peak Height | 11,670.18 | 298.92 | 22,296.43 | 11,796.75 | 641.8 | 25,943.64 | |
| Peak Area | 29,752.88 | 1018.95 | 56,642.3 | 35,778.77 | 3648.17 | 65,990.71 | |
| Atomic % | 58.68 | 1.57 | 39.75 | 55.24 | 3.59 | 41.17 | |
| Peak | Position/eV | Area | Relative Content/% | Assignment |
|---|---|---|---|---|
| N0M 1 | 284.74 | 20,080.00 | 68.98 | C-C, C-H |
| N0M 2 | 286.26 | 6701.38 | 23.02 | C-O, C-OH, C-O-C |
| N0M 3 | 288.42 | 2327.59 | 8.00 | C=O, COO− |
| N5M 1 | 284.68 | 21,080.00 | 59.41 | C-C, C-H |
| N5M 2 | 286.34 | 9909.68 | 27.93 | C-O, C-OH, C-O-C |
| N5M 3 | 288.41 | 4490.42 | 12.66 | C=O, COO− |
| Peak | Position/eV | Area | Relative Content/% | Assignment |
|---|---|---|---|---|
| N0M 1 | 532.25 | 49,140.00 | 88.06 | C=O |
| N0M 2 | 533.67 | 6661.53 | 11.94 | COO− |
| N5M 1 | 531.90 | 49,270.00 | 76.29 | C=O |
| N5M 2 | 533.22 | 15,310.00 | 23.71 | COO− |
| Peak | Position/eV | Area | Relative Content/% | Assignment |
|---|---|---|---|---|
| N0M 1 | 398.08 | 114.45 | 11.59 | pyridine |
| N0M 2 | 399.81 | 385.53 | 39.03 | pyrrole |
| N0M 3 | 401.07 | 278.90 | 28.24 | protonated pyridine |
| N0M 4 | 402.84 | 208.84 | 21.14 | -NO |
| N5M 1 | 399.00 | 404.37 | 10.21 | amine nitrogen |
| N5M 2 | 400.41 | 1215.78 | 30.69 | pyrrole |
| N5M 3 | 402.28 | 633.02 | 15.98 | -NO |
| N5M 4 | 405.66 | 1360.00 | 34.33 | -NO2 |
| N5M 5 | 407.55 | 348.64 | 8.80 | NO3− |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Huang, B.; Liu, G.; Wang, P.; Zhao, X.; Xu, H. Effect of Nitric Acid Modification on Characteristics and Adsorption Properties of Lignite. Processes 2019, 7, 167. https://doi.org/10.3390/pr7030167
Huang B, Liu G, Wang P, Zhao X, Xu H. Effect of Nitric Acid Modification on Characteristics and Adsorption Properties of Lignite. Processes. 2019; 7(3):167. https://doi.org/10.3390/pr7030167
Chicago/Turabian StyleHuang, Bo, Guowei Liu, Penghui Wang, Xiang Zhao, and Hongxiang Xu. 2019. "Effect of Nitric Acid Modification on Characteristics and Adsorption Properties of Lignite" Processes 7, no. 3: 167. https://doi.org/10.3390/pr7030167
APA StyleHuang, B., Liu, G., Wang, P., Zhao, X., & Xu, H. (2019). Effect of Nitric Acid Modification on Characteristics and Adsorption Properties of Lignite. Processes, 7(3), 167. https://doi.org/10.3390/pr7030167






