1. Introduction
Cake filtration is important in many processes. However, the analysis and design of filtration operations is still largely dependent on heuristics and experimentation. In industry, constant rate filtration (CRF) is a widely practiced and mature technology to recover both the solids and the filtrate, or only one of these [
1,
2]. There are a number of advantages to use CRF over the alternative of constant pressure filtration (CPF). For example, constant rate filtration operates at low pressures at the start of the filtration cycle, and thus any fine particles present are less likely to penetrate the filter medium leading to filter medium blinding [
3,
4]. In addition, the CRF provides more reliable data at the start of the filtration that can help to determine a value for the filter medium resistance (
Rm). The reliability of this data is greater than what is normally obtained from the alternative CPF, where a negative intercept on the classical
t/
V plot used for data analysis is often found [
5,
6].
The common objective in many laboratory filtration experiments include determination of the filter cake permeability, or specific cake resistance to filtration, and how it varies with filtration pressure, as well as information on the filter medium resistance, after it has stabilised [
7,
8]. It has been proposed that a homogeneous constant growth rate of cake, using a constant rate, provides a better packing structure of particles that is independent of cake thickness [
6,
9,
10]. In addition, reports by many authors [
11,
12,
13,
14,
15,
16,
17] indicate that, for the cake and the medium, the measured resistances are influenced by the migration of the fine particles, which increases the interactions between the particles and reduces fluid flow rates through them. When filtering suspension with low initial concentrations, the ability of fine particles to migrate into the filter medium is greater. Therefore, during the initial stages of filtration, the medium resistance will rapidly increase to what is assumed to be a constant value, owing to clogging by fine particles, but is often orders of magnitude greater than the medium resistance in the absence of solids [
11,
12,
13,
14,
15].
However, it is worth noting that the most noticeable variation of specific resistance with pressure is found within the lower pressure range. As pressure increases, the change in resistance becomes less pronounced, as would be expected of a power law relationship, such as that shown in Equation (1). Hence, the most relevant region to investigate is the lower pressure one [
5] which is possible to do using a CRF system, as utilised in this work.
It is usual to define an average cake concentration and average specific resistance, both of which may be a function of the applied pressure over the cake at any instance in time. Increasing the applied pressure increases the cake density by reducing the spaces between particles. The common equations used to correlate both the average specific resistance (
αav) and the average concentration (
Cav) respectively with pressure, are given in Equations (1) and (2)
where ∆
Pc is the pressure drop across the cake (Pa),
α0 is the specific cake resistance (m/kg) for zero applied pressure, while
C0 is the bed concentration (m
3/m
3) for the same conditions. The power law exponents
n and
m are other constants for the material, called its compressibility coefficients (relating to the cake resistance and concentration, respectively). Values for
αav can be determined experimentally by performing a series of filtrations at different pressures, allowing the resistance exponent n to be evaluated in a relative straight forward manner. While the same is possible for the cake concentration, to find the exponent
m, it is more difficult to determine accurately experimentally. However, it can be normally assessed to the required degree, simply from measuring the change in cake height.
Although the cake filtration behaviour of both incompressible and compressible filter cakes is now more understood, there still remains many fundamental issues, such as the difficulty of changing the cake structure with different applied pressures or cake thicknesses. Additionally, the influence of sedimentation during the initial stage of filtration is another problem that needs to be better understood. According to Rietema [
18,
19], the so-called retarded packing compressibility (RPC) can be found in both constant pressure and constant rate filtration with almost any kind of material (compressible and incompressible). This phenomenon occurs only after some time after the initial filtration stage and for a critical cake height. The phenomenon is represented by two different upper and lower formed cake layers. The upper layer retains its original packing structure and porosity, while the lower is much denser packing with increasing resistance. He also found that, in the case of observing this phenomenon during constant rate filtration, the rate of pressure increase suddenly jumps to a higher and constant value, which is due to the specific resistance increase at the bottom of the cake. As a result, the measured pressure and porosity distribution as well as the filtration rate with cake thickness are affected by this phenomenon. Similar behaviour to an RPC has also been noted by other authors [
20,
21,
22].
On the other hand, the retarded packing compressibility (RPC) phenomena has only limited support, and there are a number of researchers including Shirato et al. [
23,
24] and Murase et al. [
25] who do not accept Rietema’s results, while it has still not fitted fully into existing and developing theories of cake filtration. They suggested that this behaviour was related to the presence of pin electrodes that did not allow normal compression to occur within the experimental equipment. Moreover, Tarleton et al. [
2] and Sorensen et al. [
26] claim that the maximum solid concentration of the formed cake may be reached away from the medium at a position into the cake. Furthermore, in some cases, there are temporary increases in filtrate flow for a short time, which is accompanied by a sudden increase in the turbidity of the filtrate, whereupon the filtration rate returned to the original level. Tarleton et al. [
27] concluded that this behaviour is more likely to occur during the filtration of suspensions containing structured or more loosely bound particles. Recognising the previous aspects, it is not easy to obtain values of average permeability or specific resistance for all materials used in filtration. The permeability (
k) of a filter cake is related to its cake specific resistance by Equation (3).
where
k is the hydraulic permeability (m
2),
C is the solid concentration by volume (m
3/m
3) and
is the density of solids (kg/m
3).
Efforts to predict the permeability (or resistance) to filtration from particle size data are notoriously inaccurate for many systems, especially for irregular shaped or cohesive particle beds. Hence, there is still need for experimental empiricism to explain behaviour, for the purpose of scale up and design, and critical uncertainty in the analysis of filtration results due to the above reviewed phenomena. Well known models of permeability (and hence specific resistance) contain terms representing the specific surface area per unit volume of the particles (and thereby the particle size) and the solids volume fraction. The porous media permeability can be deduced from the Kozeny-Carman equation by using Equation (4) or Equation (5).
or in terms of a mean size (Sauter diameter):
where
C is the volume fraction of solids present,
Sv is the specific surface area per unit volume of the particles,
(1/m) and
K is the Kozeny coefficient, where a value of 5 is used as is conventionally assumed in text book for packed beds [
28,
29]. The permeability may be converted to specific resistance using Equation (3), but attempts to predict specific resistance from particle size analysis data usually fail by at least one order of magnitude [
5] with the resistance being greater than that predicted. The choice of any particular diameter for characterisation of an irregular particle depends on the intended application in most cases [
30,
31]. When the particle is non-spherical, the determination of a single particle size is not a simple task. Either particle orientation or direction affects the measurement of common statistical diameters [
32]. Rhodes [
33] reported a number of different geometrical sizes as well as different dynamic ones together with the statistical diameters. In this work, the Sauter mean diameter (
) being the diameter of a sphere with the same average specific surface area per unit volume was used. This diameter is applicable for systems where the surface area is the determining factor and particle samples have relatively narrow size distributions.
In this research, the effect of the initial filtration period on the cake specific resistance and permeability will be investigated in detail. To study this period for different materials, two particulate materials are reported, with similar particle size distributions: one mildly compressible (talc) the other substantially incompressible (calcium carbonate). Constant rate filtration will be used in this system because of two main reasons: (a) Upper and lower limits on the variation of solids pressure in the filter cake can easily be obtained and (b) the particle packing structure is expected to be independent of cake thickness, so that uniform compacts are formed [
5]. This will enable some critical understanding on the differences between predicted and permeabilities from known analytical expressions and the measured values.
3. Results and Discussion
Figure 4 illustrates three different starting suspension concentrations; 0.01, 0.03 and 0.05 by volume of talc, using the fastest pump speed (30 rpm). Overall, the figure appears to be reasonable; with the greatest suspension of solids showing the highest required filtration pressure. There was no observable evidence of significant gas bubbles being formed in the filtrate line even at the highest differential pressure (vacuum) of 0.7 bar. However, the shape of the figure is slightly unexpected in that there is a small point of inflection near the origin followed by a rapid rise in pressure, before the rate of pressure increase reduces in the latter part of the experiments. This issue will be studied in detail later in this work.
Cake concentration was determined based on the volume of the starting concentration and the volume of filtered slurry, together with the measured filter cake heights and the filter area, using material balances. The relative standard deviation (RSD) of the height measurements is obtained from the standard deviation divided by the average value. The measured heights, calculated cake concentrations, and RSD for both solid materials are shown in
Table 3a,b where the RSD of the cake concentration is determined from the variation in cake height measurements over the five measured data points. The RSD, in general, decreases with increasing initial slurry concentration: from about ±12% (0.01
v/
v) to less than ±4% (0.05
v/
v) feed suspension for talc, an expected consequence of a greater cake height being formed. However, when using calcium carbonate, the RSD varies from about ±2.4% (0.09
v/
v) to less than ±1% (0.21
v/
v). It is assumed that the cake is of uniform concentration throughout its height as cake volume is small (see
Table 3).
As would be expected, a low pump speed (5 rpm) provides a low pressure resulting in a lower specific resistance, as shown in
Table 3. Moreover, it is worth noting that the most noticeable variation of specific resistance with pressure is found within the lower pressure range; as pressure increases, the change in resistance becomes less pronounced as would be expected of a power law equation such as that shown in Equation (1). Hence, the most relevant region to investigate is the lower pressure one, as discussed in reference [
5]. For that reason, the discussion will focus on lower pump speed (5 rpm).
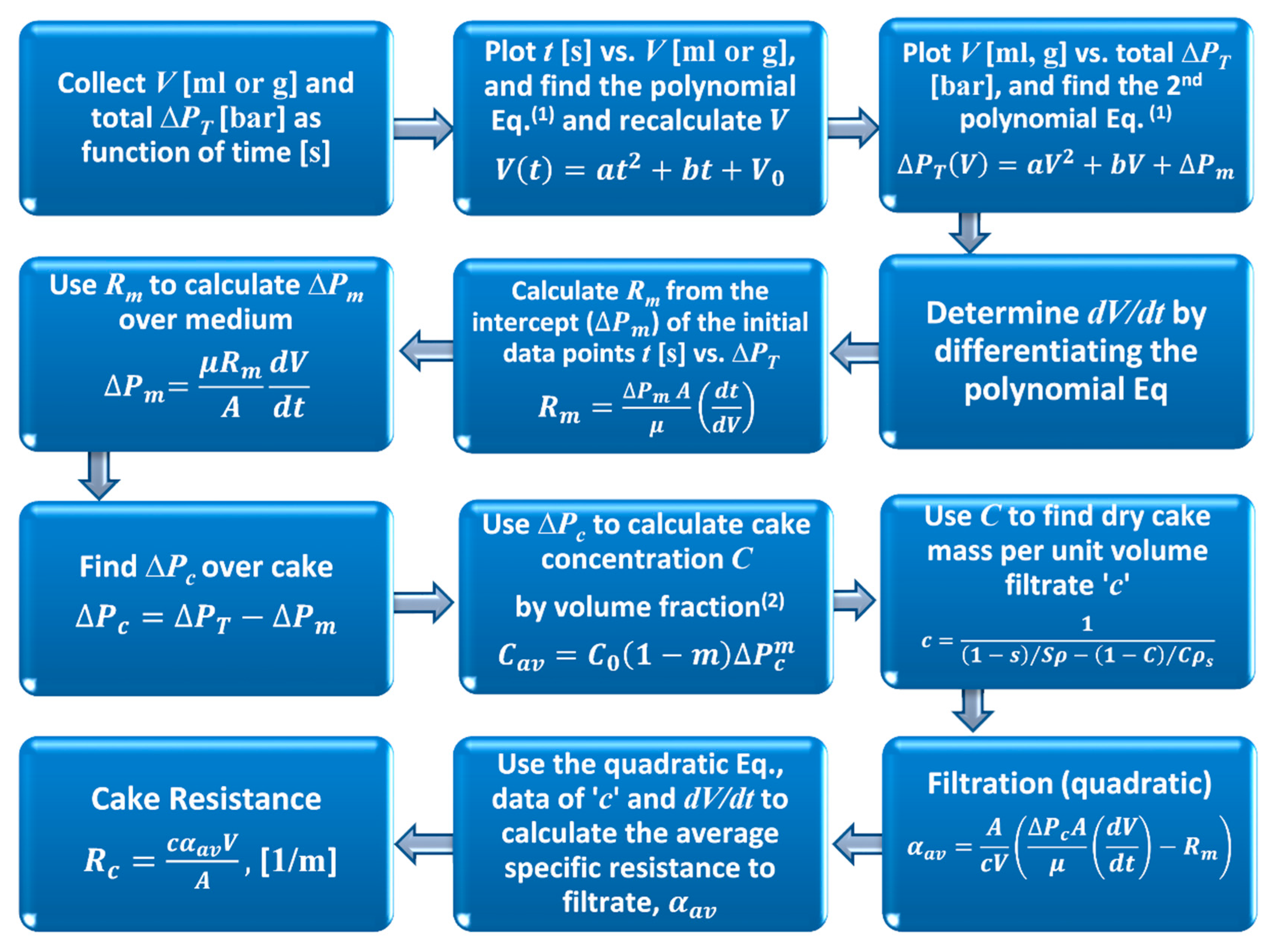
Figure 5 presents the filtrate and total applied pressure difference with time, for the talc and calcium carbonate filtration at the lowest pump speed 5 rpm (cake forming pressure). During talc filtration, the filtrate curve can be divided into two visible sections: rapid and slower filtration rates, as labelled (1) and (2) respectively, which are indicated in the figure by the two dashed vertical lines. Both the filtrate mass and pressure curves illustrate this trend. However, during calcium carbonate filtration, the filtrate curves were generally divided further into three visible sections for lower concentration (0.09
v/
v): (1) filtration, (2) permeation (after around 30 min, assumed to be due to particle settling) and (3) cake consolidation (squeezing all liquid between particles). However, only two sections are clearly evident for higher concentrations (0.21
v/
v): (1) filtration and (2) permeation. To clarify the difference between both systems (calcium carbonate and talc) the influence of particle sedimentation of both systems during the initial filtration was studied, and will be discussed herein.
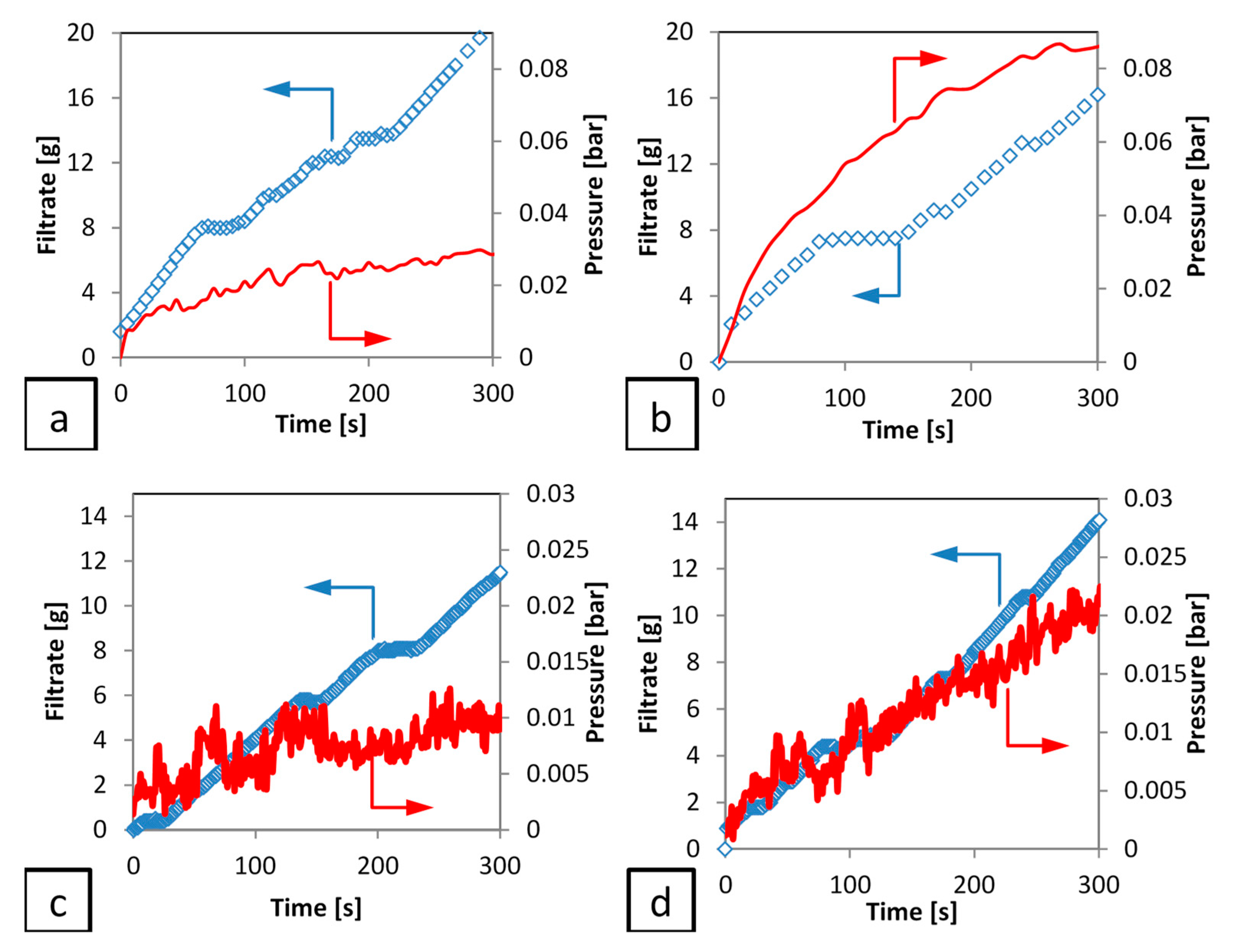
The initial filtration (period 1) is illustrated in greater detail in
Figure 6, for both talc and calcium carbonate systems, which highlights a more complex situation at these early time steps, as cake formation takes places. There appears to be an initial period of filtrate accumulation with respect to time, followed by a ‘plateau region’ during which no filtrate is formed. In the case of the more compressible material (talc) this region is longer than the less compressible material (calcium carbonate). The pressure data is somewhat ‘noisy’ in both cases, due partly to the use of a peristaltic pump to suck the filtrate through the system and the operation of the transducer at a low differential pressure. In addition, the calcium carbonate data is more noisy, due to the much lower pressures being recorded when compared to the talc filtrations, as discussed for similar systems in a previous publication [
5].
In addition, despite this experimental noise, there is no evidence from any of the runs of a differential pressure plateau corresponding to the filtrate plateau. Hence, it can be concluded that the pressure provided from the pump continues to rise, but the filtrate rate stops for a period of up to 20 s (depending on the studied system). This data shows that, for both cakes filtration (incompressible and compressible) the retarded packing compressibility (RPC) concept does exist with a significant period of zero filtrate flow, despite continued application of filtration pressure. It is suggested that the filter cake undergoes a period of rearrangement at that point, with the liquid pressed out from that rearrangement flowing upwards into the slurry rather than down through the previously formed filter cake and the filter medium. Hence, after that ‘cake collapse’ the overall resistance to filtration changed to a higher value. For that reason, pressure continues to be raised without filtrate mass, until it overcomes the new resistance due to the cake collapse.
Further effort is made to understand this period, as it is important from the point of view of attempting to determine the medium resistance, which is used later to provide a value for pressure drop over the medium and hence cake forming pressure drop. In addition, it is important to check if this behaviour is a result of equipment/process or materials influence. Two possible explanations can be suggested for these unexpected pauses in filtrate rate behaviour. The first is the influence of particle sedimentation during the initial filtration. This aspect was studied using a gravity sedimentation process to obtain the particle settling velocity. The lowest (0.09 v/v) and highest (0.21 v/v) initial concentration of calcium carbonate was used, as as plate like shape of the talc particles required longer time for settling. The results indicate that the filtration rate of calcium carbonate is much greater than the particle settling velocity by 4 to 13 times for the lowest and highest initial concentration used in this work respectively. As a result, while particle sedimentation has some contribution on the lowest concentration, the effect is very small and it can be neglected for the higher concentrations.
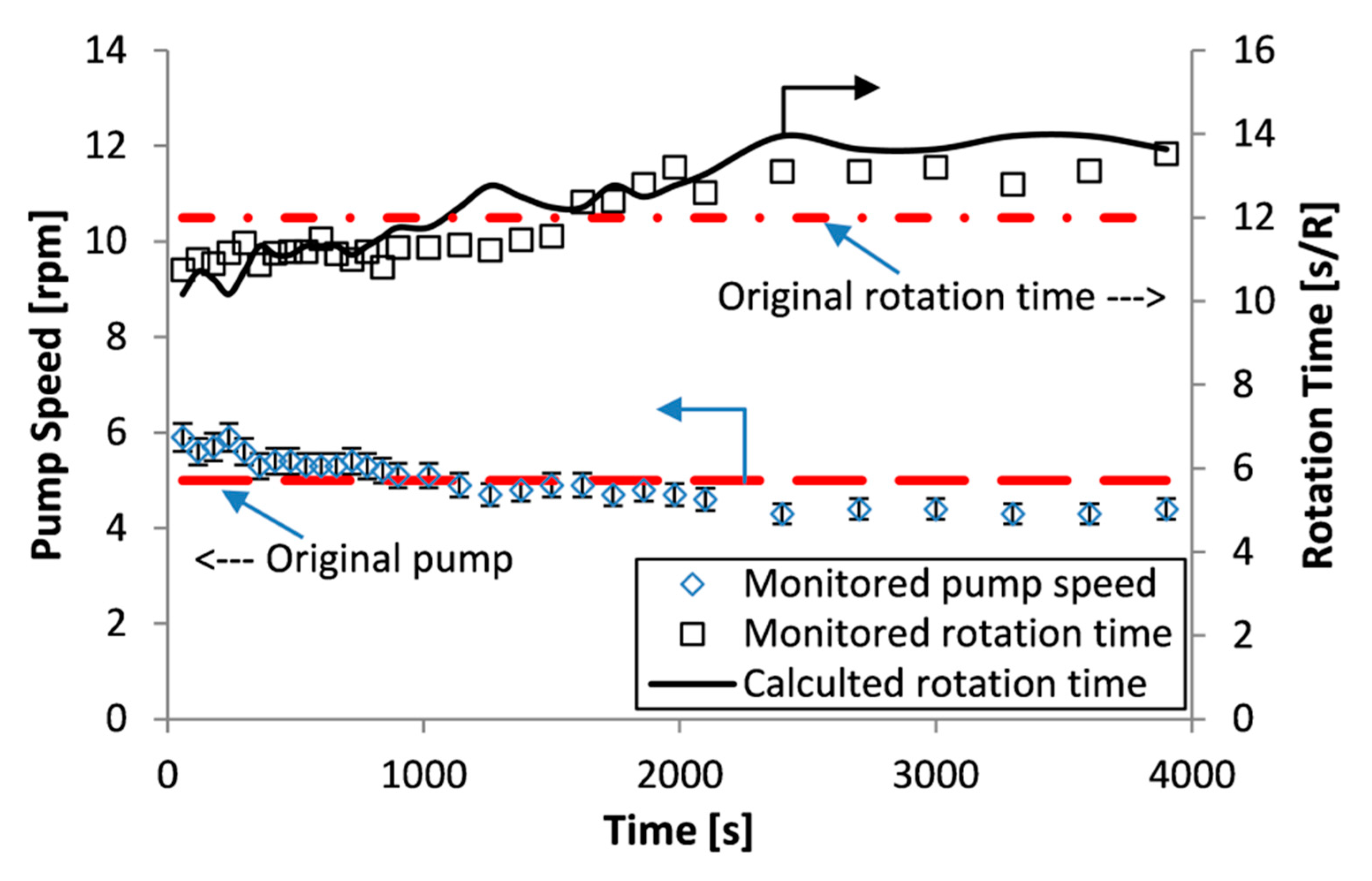
Another potential explanation is the influence of the peristaltic pump used to suck the filtrate through the filter medium. This aspect was investigated through the analysis illustrated in
Figure 7, which shows the pump speed setting and the actual rotation time during the course of the filtration. The pump is a ‘low pulsation’ peristaltic pump with four rollers, hence at a setting of 5 rpm the time period for a roller to pass over the tubing is 3 s; i.e., if filtrate discharge occurs only when a roller passes then discharge should be every 3 s. Hence, this does not explain the stoppage of filtrate for a period of 20 s. This is the lowest pump setting used and the ‘plateau effect’ was noticed for all the experiments. Furthermore, another investigation was made on the experimental procedure. Initially, experiments were performed with the outlet tube on the discharge side of the pump discharging into air in an empty beaker on the weighing scales, to investigate if the stoppage could have been due to the intermittent drainage of that discharge tube. Therefore, the experimental procedure was changed to start with water in the beaker and the discharge tube end submerged in that water to prevent any drainage occurring. This alteration made no difference to the observation of a ‘plateau region’. Hence, it was concluded that the observed behaviour is not related to the equipment, process, or methodology, while it may be due to the nature of the materials of the solids (compressible and incompressible, size distribution, and shape). Also from
Figure 7, it can be seen that the pump speed slightly decreases and the measured rotation time increases, during period (2). Hence, the pump motor is not sufficiently powerful enough to provide truly constant rate against a varying pressure formed by the filtration. This does not invalidate the filtration analysis, so long as the actual pressure and filtrate rate can be determined at any instance in time, and that is possible to assume an average cake concentration and specific resistance to filtration is valid.
The data illustrated in
Figure 5 and
Figure 6 further suggest that the pressure drop over the filter medium is negligible, with the measured pressure being close to zero at the start of the filtration. However, in order to assess the relevancy of filter medium resistance in the later calculation of pressure drop over the filter cake, the initial stages of filtration were treated as follows to provide three different possible values of the membrane filter resistance (
Rm). Firstly, it was assumed that
Rm is equal to zero (
Rm = 0 at time 0 s). Secondly, the value of
Rm was determined by applying Darcy’s law to the first data point measured, neglecting any resistance due to the filter cake (
Rm = N
1) at the first measurement of filtrate. For example, for conditions of 0.21
v/
v and a 5 rpm pump speed (representing data shown in Figure 10) N
1 is 1.07 × 10
10 m
−1 for talc and 6.8 × 10
8 m
−1 for calcium carbonate. The third approach is to consider the last time before the first ‘plateau region’ occurs and again determine
Rm by assuming that all the pressure drop is over the filter medium by applying Darcy’s law (
Rm = N
2 at the first point of the filtrate plateau/stop). In the case of the talc filtration at 0.01
v/
v solids (
Figure 6a),
Rm = N
2 = 1.4 × 10
11 m
−1, which occurs after 60 s into the filtration. While, in the case of the calcium carbonate filtration at 0.09
v/
v solids (
Figure 6c)
Rm = N
2 = 1.3 × 10
9 m
−1 would be at 10 s into the filtration. These provided three values for
Rm that could be used in the later analysis for pressure forming the filter cake. The true value of
Rm would, of course, likely be somewhere between these extreme values.
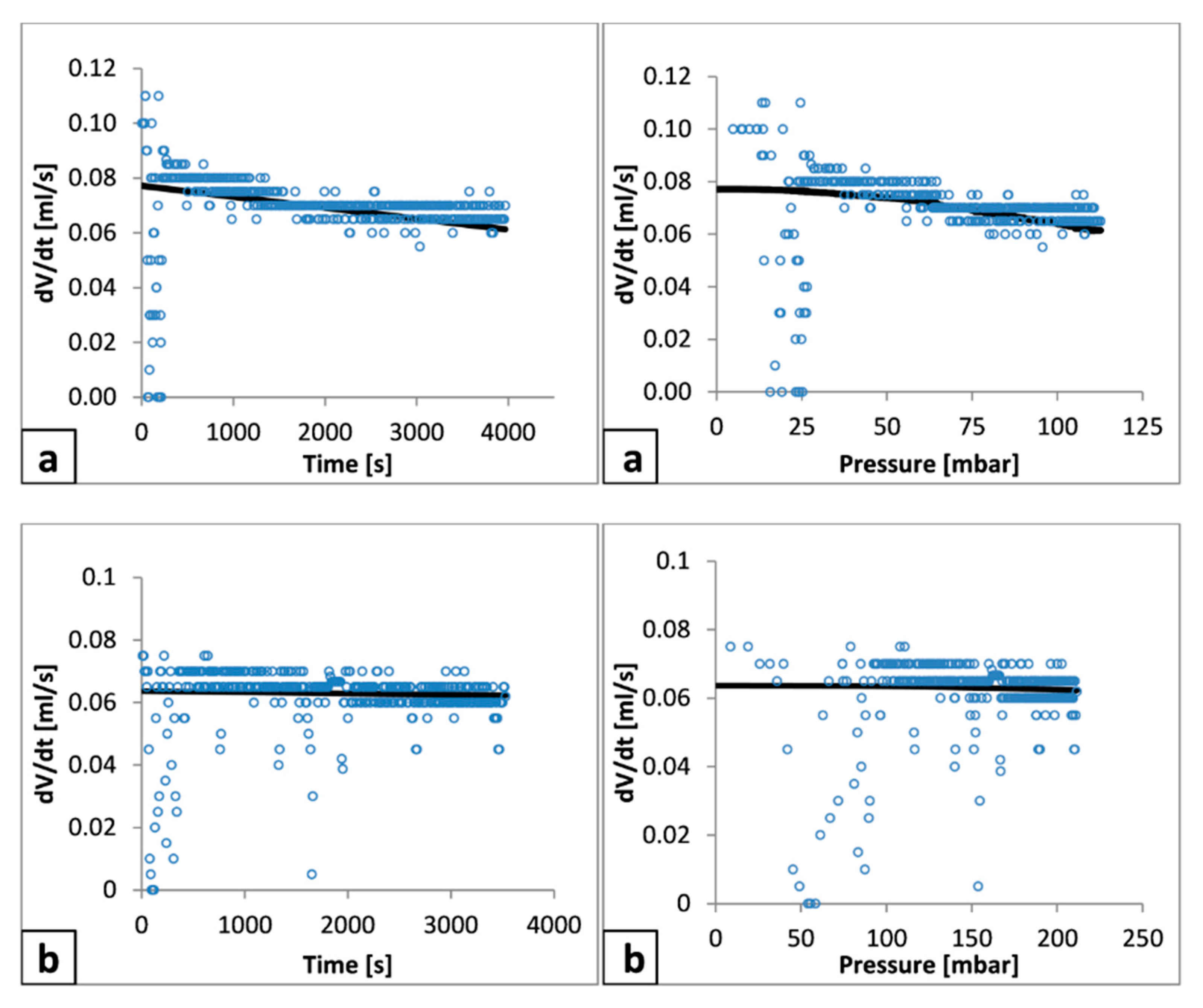
It is noted that also the general shape of the pressure versus time data shown in
Figure 5 (right hand axis) during period (2) demonstrates a reduced change with respect to time, whereas the relationship of filtrate versus time (left hand axis) time appears substantially linear, which is attributed to the pseudo constant filtrate pump. Thus, the assumed 2nd polynomial fit of the filtrate data may be a greater order than generally required, where
dV/
dt would largely be presumed to be relatively constant within each time period. To test this feature,
Figure 8 and
Figure 9 show the filtrate rate with time for talc and calcium carbonate, respectively. In addition, both the differential of the second order polynomial and the experimentally determined values are illustrated. The latter are calculated by a ‘central difference’ technique, where for the filtrate rate (
V) at time (
t), the rate is determined by Equation (6).
It can be seen that the polynomial approach leads to significant data smoothing, considerably reducing experimental fluctuations. Thus, while not necessarily representing the physics exactly, the methodology was maintained for all analyses. The smoothed filtrate rate from each fitting was subsequently used for the calculation of pressure drop over the filter medium and cake, thereby allowing permeability and specific resistance of the filter cake to be measured (see
Figure 3).
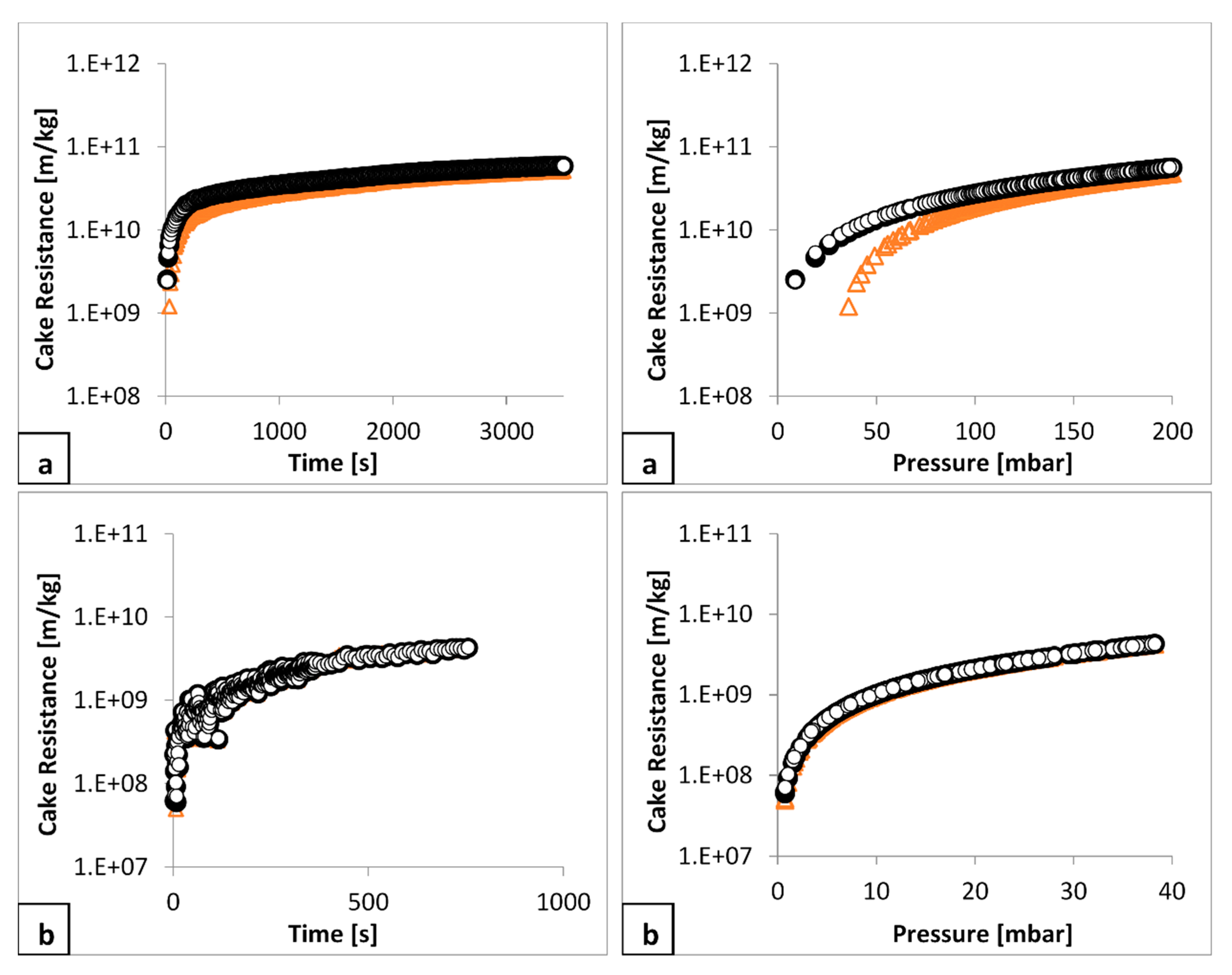
The variation of average filter cake specific resistance to filtration with time and with pressure, is illustrated for two examples filtrations in
Figure 10: for talc (a, b) and for calcium carbonate (c, d). The analysis followed the format provided in
Figure 3. One key aspect being investigated was determination of any influence of the pressure drop over the filter medium and to deduct it from the measured total pressure drop to provide that for the cake. This was achieved by considering the three different values of
Rm, hence three different values of cake forming pressure, to determine three different values of cake resistance for all the filtrations.
In general, it is demonstrated that the pressure drop over the medium is negligible as the cake resistance is the same with respect to time, regardless of the chosen value of
Rm. There is some small variation in the talc resistance with pressure curve at lower pressure, within the region of cake formation (
Figure 10a), but it is reasonably consistent after a pressure of 80 mbar has been reached. Thus, for the purpose of determining the constitutive relation between cake resistance and cake forming pressure (Equation (2)) the data illustrated in
Figure 10: (a) for talc, from 80 mbar onwards can be used and (b) for calcium carbonate all pressure values can be used.
Considering the voidage formed by the two different systems (talc and calcium carbonate) and the commonly used Kozeny approach to permeability (Equation (5)) it is possible to see why empiricism is still required for filtration modelling and scale up in design. Talc produced cakes with a concentration of between 0.18 to 0.26 (
v/
v) solids concentration; the calcium carbonate cakes were between 0.41 and 0.53 (
v/
v) in solids content (
Table 3), while the Sauter mean diameter of the two different solids were almost identical (
Table 1) which therefore would infer higher permeabilities in the talc, due to the increased porosity. The measured permeabilities for all constant rate trials were compared to estimates from the K-C equation, and are given in
Table S1, within the ESI, where a number of important trends are observed. In this calculation, the values of Sauter mean diameter for both materials are as given in
Table 1 (6 µm and 5 µm for the talc and calcium carbonate, respectively). It is firstly evident that the range of measured permeabilities for talc (e.g., 1.68 × 10
−14 and 3.6 × 10
−14 m
2 at 5 rpm pump rates) are actually similar to calcium carbonate (e.g., 1.64 × 10
−14 and 5.42 × 10
−14 m
2 at 5 pm). Hence, average specific cake resistance values (using Equation (3)) for talc are much greater, due the lower bed concentrations, giving an overall average for all conditions of 5.9 × 10
10 and 7.4 × 10
11 m/kg for calcium carbonate and talc, respectively. Even allowing for a low value of sphericity for talc (0.22) compared to calcium carbonate (0.81) [
39] would not explain the (at least) an order of magnitude higher specific resistance displayed by the talc cakes compared to the calcium carbonate. Therefore, there is clearly additional effects that are enhancing the resistance to permeation in the talc cakes compared to the calcium carbonate ones.
It is also evident that the K-C approach (using Equation (5)) considerably overestimates the permeability in both systems, but most significantly with the talc particles (see
Table S1). As such, these effects are parameters that are not recognised in the K-C equation, and they become more prevalent with the compressible talc cake. This result may be related to a number of factors, including the shape of the particles or the enhanced contribution to resistance from the fine particles with the size distribution, as discussed by number of authors [
11,
12,
13,
14,
15,
16,
17]. The particle shape and contribution of fines to the overall resistance is also being investigated using numerical models (ANN and MVR) to determine permeabilities [
40]. It may also be the case that particle agglomeration may increase the tortuosity of permeation pathways and could contribute to the overestimation in the K-C approach. It is known from previous work on similar calcium carbonate systems that some low level of agglomeration may take place, due to its small surface charge [
35], while the charge differential on the faces and sides of talc platelets may lead to complex structuring in solution, further decreasing permeability.