Effect of Physical and Mechanical Activation on the Physicochemical Structure of Coal-Based Activated Carbons for SO2 Adsorption
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
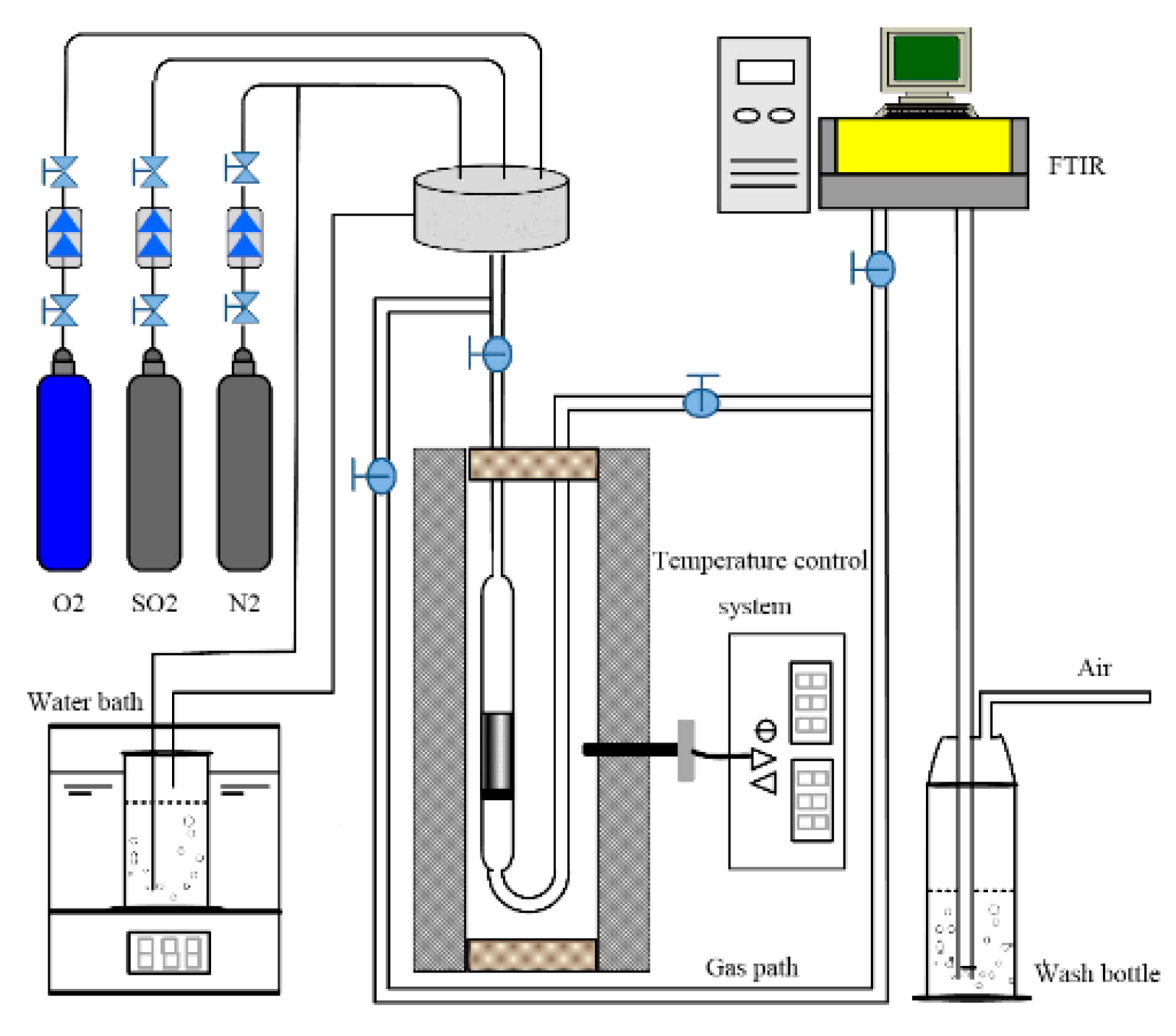
2.2. Experimental Process
2.2.1. CO2 activation
2.2.2. Mechanical activation
2.3. Measurement Analysis
2.4. SO2 Adsorption Test
2.5. Thermal Regeneration
3. Results and Discussion
3.1. Microstructure and Surface Morphology Analysis by HRTEM and SEM
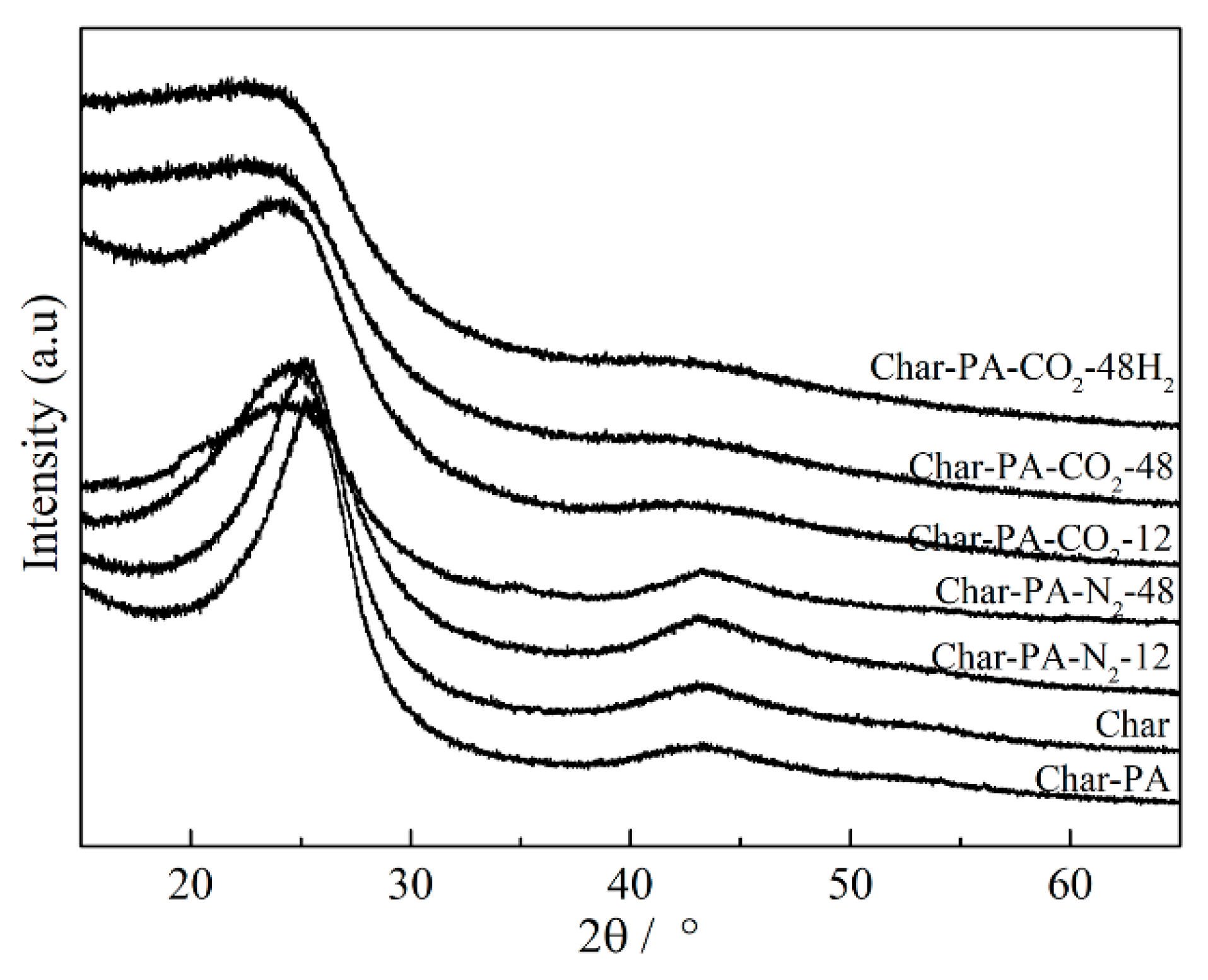
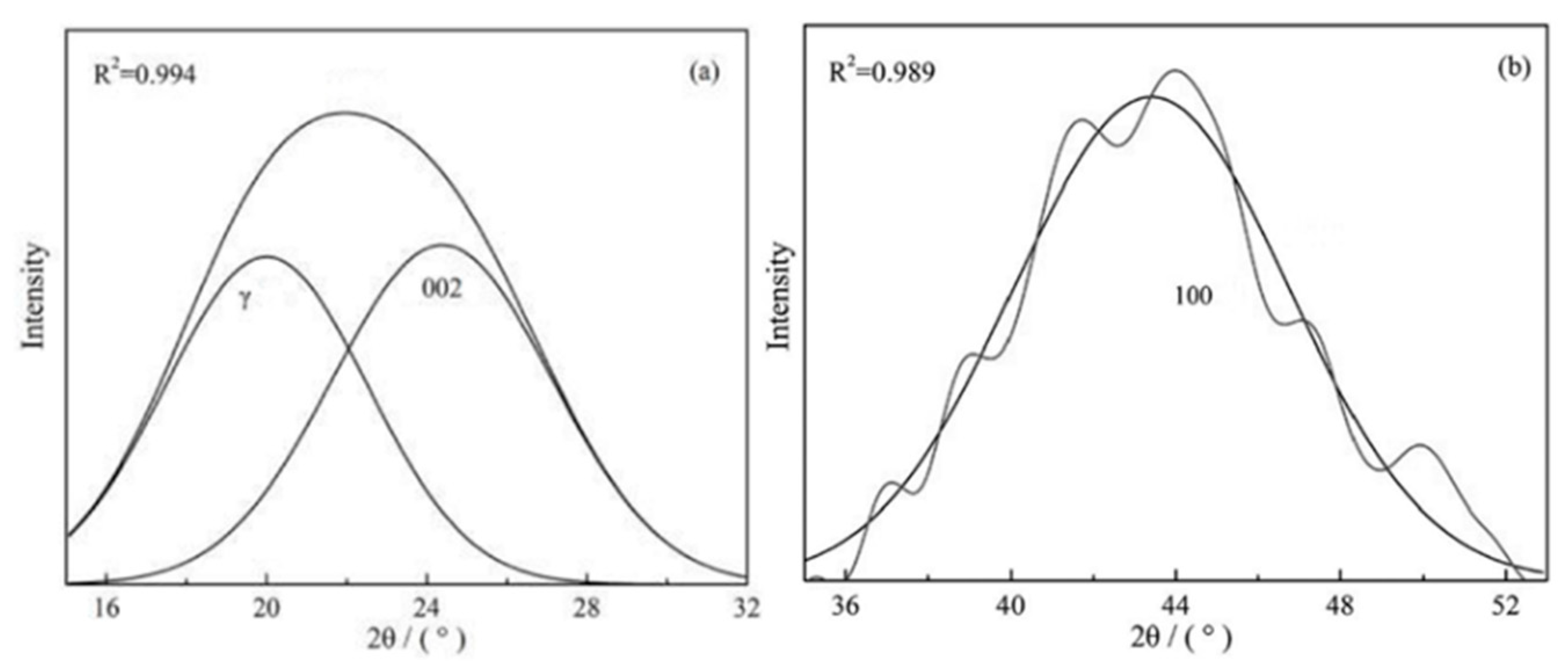
3.2. Crystal Structure Analysis by XRD
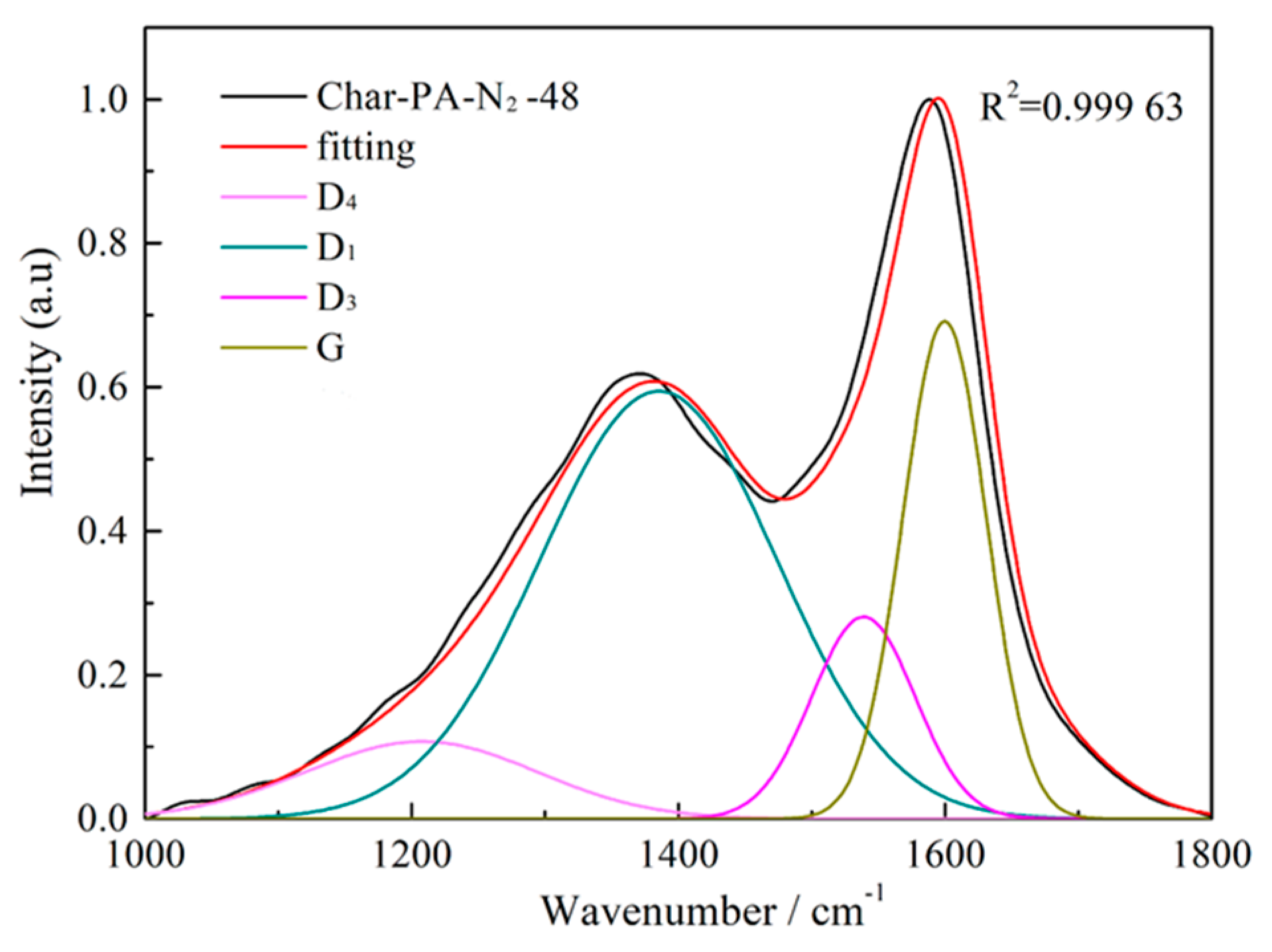
3.3. Carbon Structure Analysis by Raman
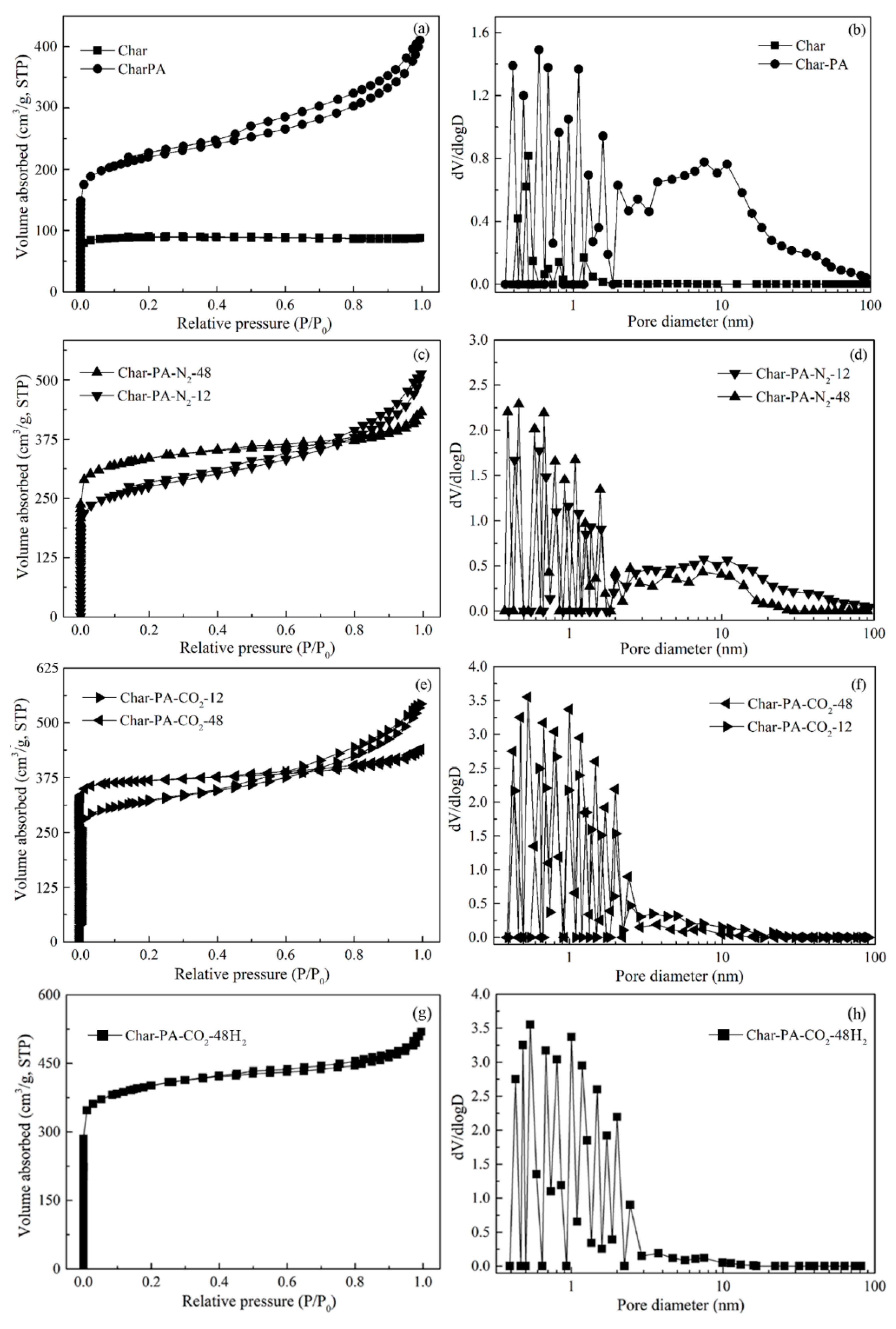
3.4. Pore Structure Analysis by N2 Adsorption
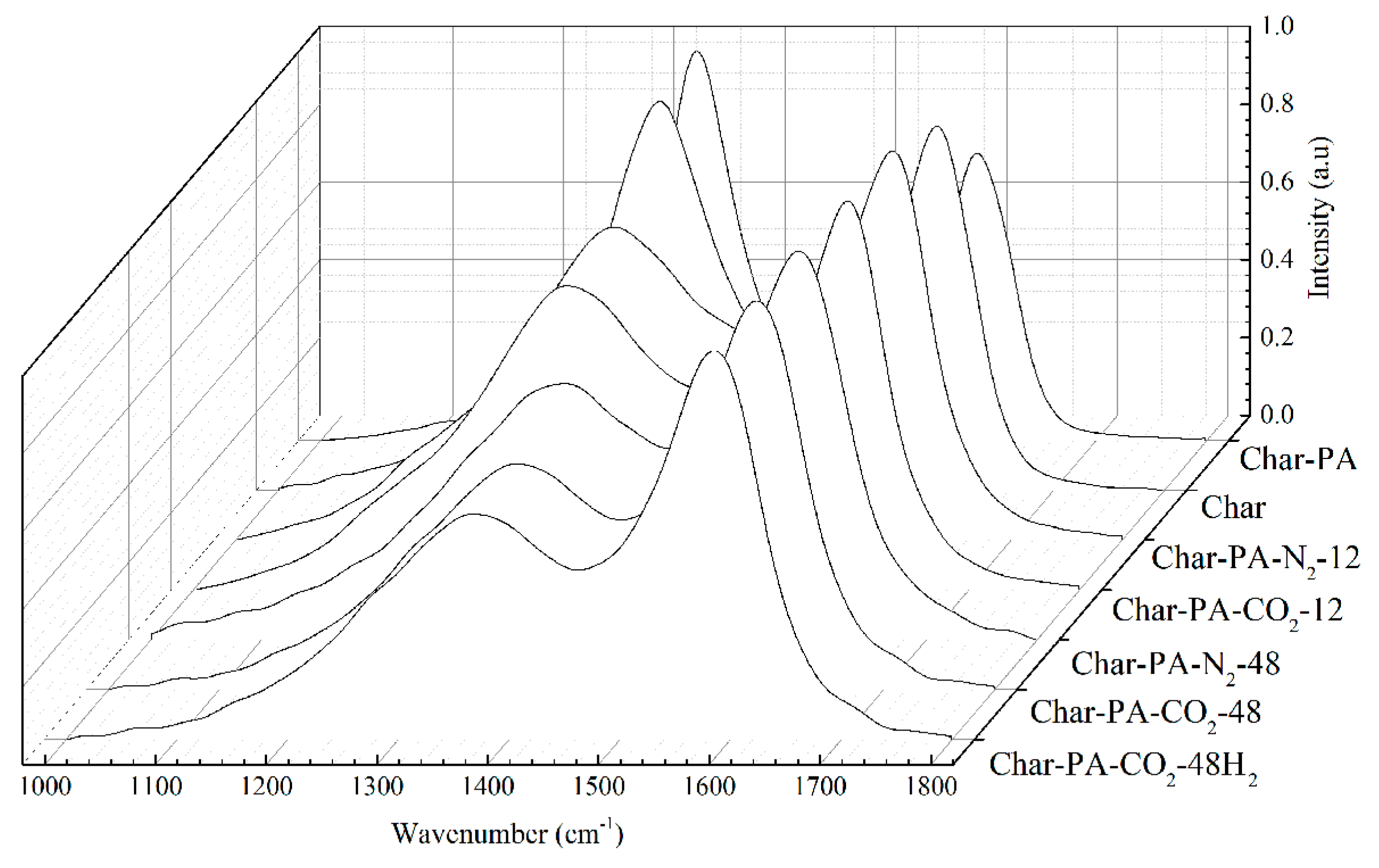
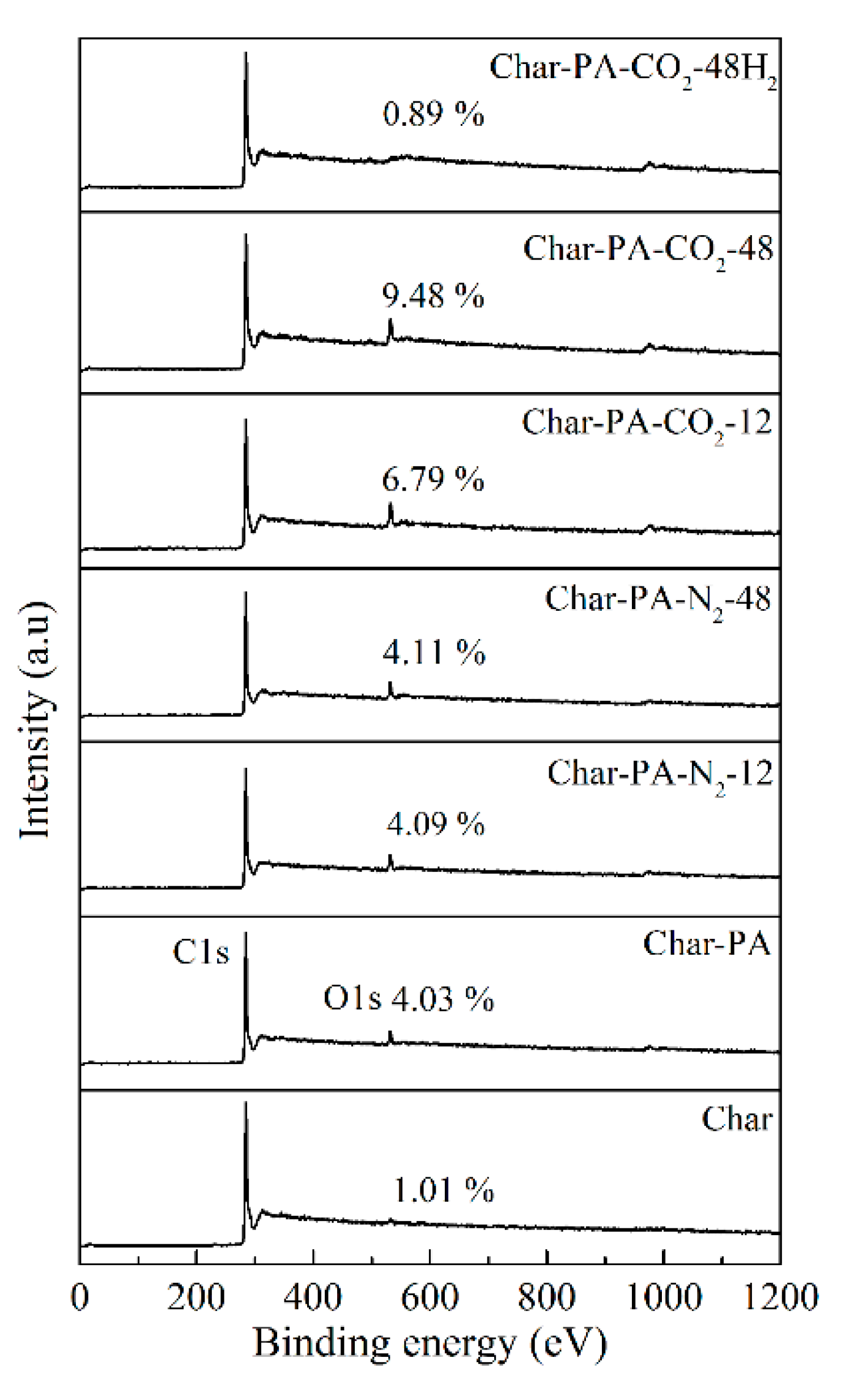
3.5. Surface Chemical Structure Analysis of XPS
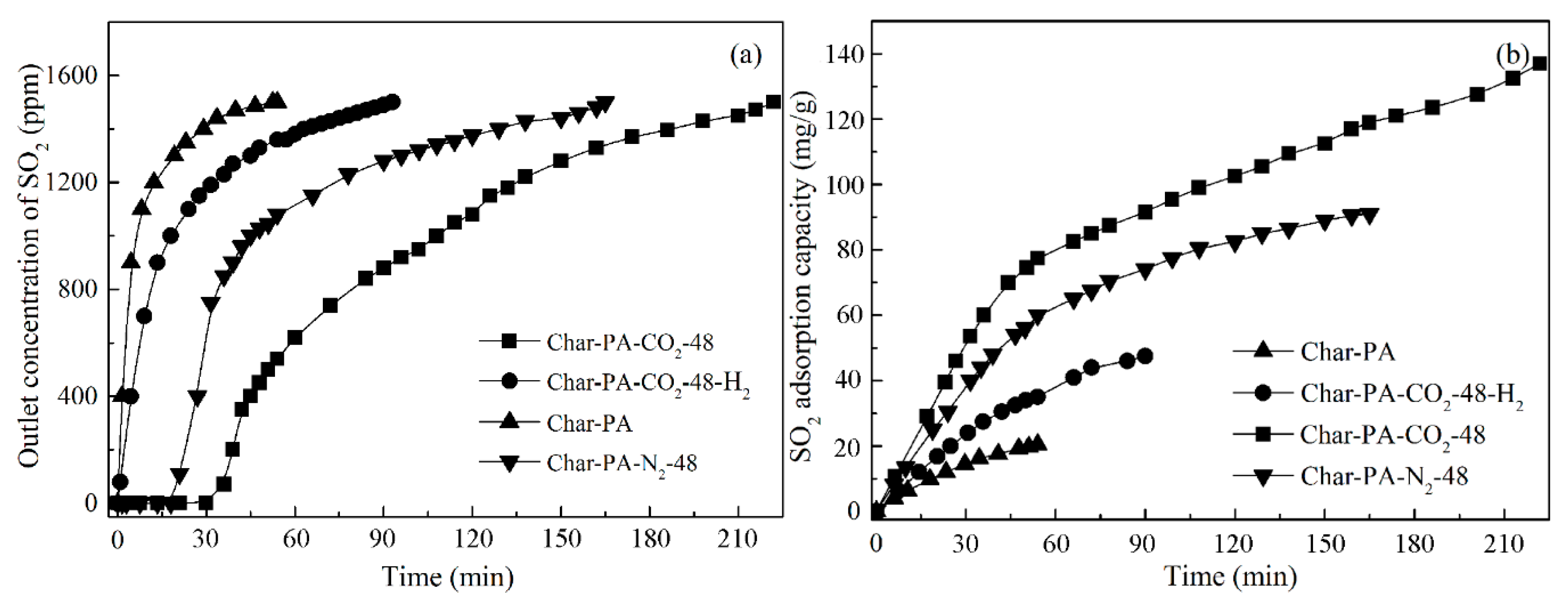
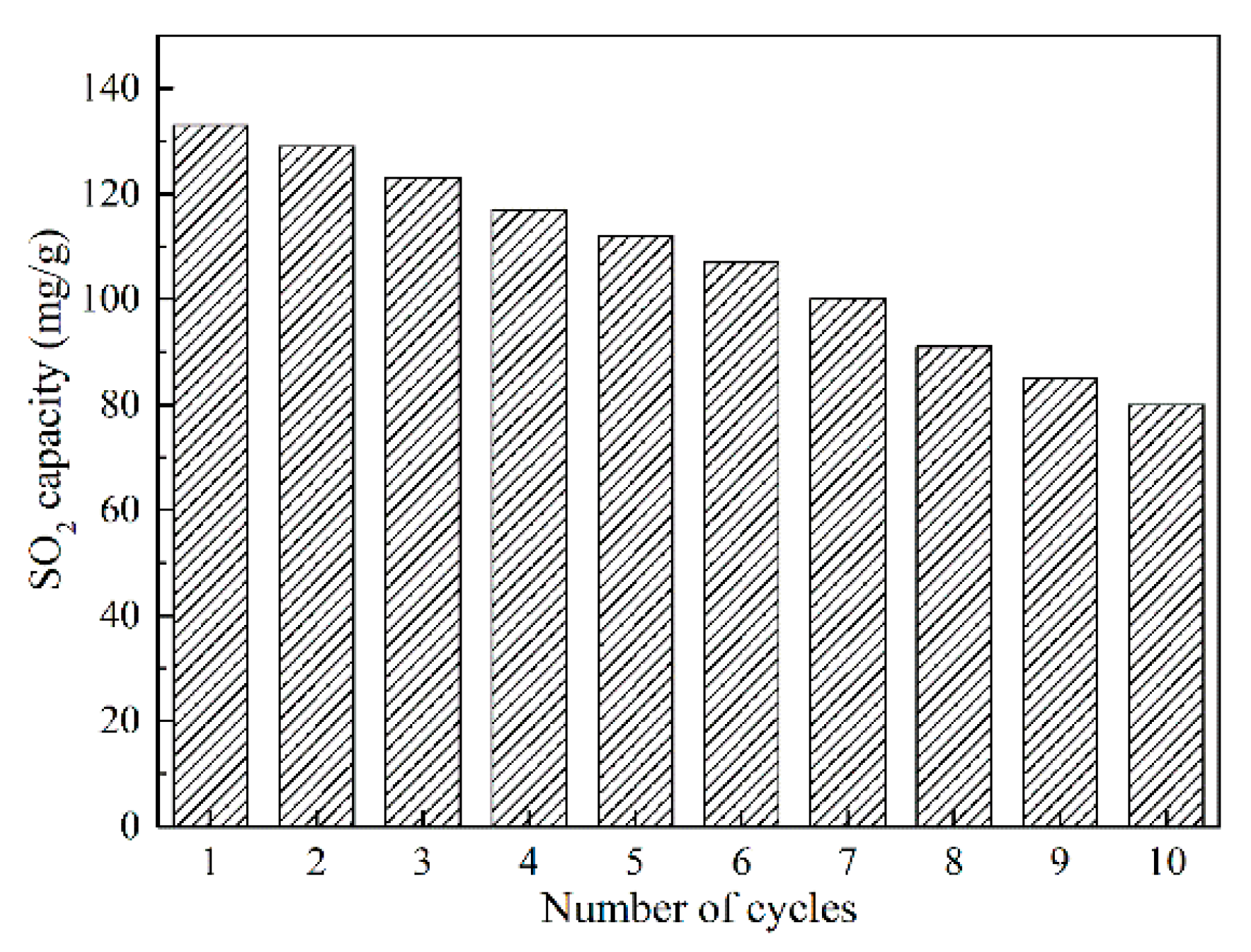
3.6. Study of SO2 Adsorption
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Liu, D.D.; Gao, J.H.; Cao, Q.X.; Wu, S.H.; Qin, Y.K. Improvement of activated carbon from Jixi bituminous coal by air peroxidation. Energy Fuels 2017, 31, 1406–1415. [Google Scholar] [CrossRef]
- Sumathi, S.; Bhatia, K.S.; Lee, K.T.; Mohamed, A.R. Adsorption isotherm models and properties of SO2 and NO removal by palm shell activated carbon supported with cerium (Ce/PSAC). Chem. Eng. J. 2010, 162, 194–200. [Google Scholar] [CrossRef]
- Bahamon, D.; Vega, L.F. Systematic evaluation of materials for post-combustion CO2 capture in a temperature swing adsorption process. Chem. Eng. J. 2016, 284, 438–447. [Google Scholar] [CrossRef]
- He, X.J.; Zhang, H.B.; Zhang, H. Direct synthesis of 3D hollow porous graphene balls from coal tar pitch for high performance supercapacitors. J. Mater. Chem. A 2014, 46, 19633–19640. [Google Scholar] [CrossRef]
- Parimal, C.B.; Aola, S.; Rituparna, K.; Mridushmita, B.; Chubaakum, P.; Dipak, S. Activated carbon synthesized from biomass material using single-step KOH activation for adsorption of fluoride: Experimental and theoretical investigation. Korean J. Chem. Eng. 2019, 36, 551–562. [Google Scholar] [CrossRef]
- Li, Y.T.; Pi, Y.T.; Lu, L.M. Hierarchical porous active carbon from fallen leaves by synergy of K2CO3, and their supercapacitor performance. J. Power Sources 2015, 299, 519–528. [Google Scholar] [CrossRef]
- Foo, K.Y.; Hameed, B.H. Preparation and characterization of activated carbon from sunflower seed oil residue via microwave assisted K2CO3 activation. Bioresour. Technol. 2010, 102, 9794–9799. [Google Scholar] [CrossRef]
- Ma, G.F.; Yang, Q.; Sun, K.J. Nitrogen-doped porous carbon derived from biomass waste for high-performance supercapacitor. Bioresour. Technol. 2015, 197, 137–142. [Google Scholar] [CrossRef]
- Elmouwahidi, A.; Bailón-García, E.; Pérez-Cadenas, A.F. Activated carbons from KOH and H3PO4-activation of olive residues and its application as supercapacitor electrodes. Electrochim. Acta 2017, 229, 219–228. [Google Scholar] [CrossRef]
- Rodríguez-Reinoso, F.; Molina-Sabio, M.; González, M.T. The use of steam and CO2 as activating agents in the preparation of activated carbons. Carbon 1995, 33, 15–23. [Google Scholar] [CrossRef]
- Yang, L.; Huang, T.; Jiang, X.; Jiang, W.J. Effect of steam and CO2activation on characteristics and desulfurization performance of pyrolusite modified activated carbon. Adsorption 2016, 22, 1099–1107. [Google Scholar] [CrossRef]
- Valente Nabais, J.M.; Nunes, P.; Carrott, P.J.M.; Carrott, M.M.L.R.; García, A.M.; Díaz-Díezb, M.A. Production of activated carbons from coffee endocarp by CO2 and steam activation. Fuel Process. Technol. 2008, 89, 262–268. [Google Scholar] [CrossRef]
- Zhu, Y.W.; Gao, J.H.; Li, Y.; Sun, F. Preparation of activated carbons for SO2 adsorption by CO2 and steam activation. J. Taiwan Inst. Chem. Eng. 2012, 43, 112–119. [Google Scholar] [CrossRef]
- Zhu, Y.W.; Gao, J.H.; Li, Y.; Sun, F.; Qin, Y.K. Preparation and characterization of activated carbons for SO2 adsorption from Taixi anthracite by physical activation with steam. Korean J. Chem. Eng. 2011, 28, 2344–2350. [Google Scholar] [CrossRef]
- Shu, S.; Guo, J.X.; Liu, X.L.; Wang, X.J.; Yin, H.Q.; Luo, D.M. Effects of pore sizes and oxygen-containing functional groups on desulfurization activity of Fe/NAC prepared by ultrasonic-assisted impregnation. Appl. Surf. Sci. 2016, 360, 684–692. [Google Scholar] [CrossRef]
- Davini, P. Adsorption and desorption of SO2 on active carbon: The effect of surface basic groups. Carbon 1990, 28, 565–571. [Google Scholar] [CrossRef]
- Shiratori, N.; Lee, K.J.; Miyawaki, J.; Hong, S.H.; Mochida, I.; An, B.; Yokogawa, K.; Jang, J.; Yoon, S.H. Pore structure analysis of activated carbon fiber by microdomain-based model. Langmuir 2009, 25, 7631–7637. [Google Scholar] [CrossRef]
- Liu, D.D.; Gao, J.H.; Wu, S.H.; Qin, Y.K. Effect of char structures caused by varying the amount of FeCl3 on the pore development during activation. RSC Adv. 2016, 6, 87478–87485. [Google Scholar] [CrossRef]
- Srinivas, G.; Yue, L.; Neal, S.; Taner, Y.; Zheng, X.G. Design of hyperporous graphene networks and their application in solid-amine based carbon capture systems. J. Mater. Chem. A 2017, 5, 17833–17840. [Google Scholar] [CrossRef]
- Hu, B.; Wang, K.; Wu, L.H.; Yu, S.H.; Antonietti, M.; Titirici, M.M. Engineering carbon materials from the hydrothermal carbonization process of biomass. Adv. Mater. 2010, 22, 813–828. [Google Scholar] [CrossRef]
- Wigmans, T. Industrial aspects of production and use of activated carbons. Carbon 1989, 27, 13–22. [Google Scholar] [CrossRef]
- Dahn, J.R.; Xing, W.; Gao, Y. The “falling cards model” for the structure of microporous carbons. Carbon 1997, 35, 825–830. [Google Scholar] [CrossRef]
- Liu, D.D.; Jia, B.Y.; Liu, X.; Zhao, B.J.; Gao, J.H.; Cao, Q.X.; Wu, S.H.; Qin, Y.K. Effects of oxygen functional groups and FeCl3 on the evolution of physico-chemical structure in activated carbon obtained from Jixi bituminous coal. RSC Adv. 2018, 8, 8569–8579. [Google Scholar] [CrossRef]
- Delogu, F.; Gorrasi, G.; Sorrentino, A. Fabrication of polymer nanocomposites via ball milling: Present status and future perspectives. Prog. Mater. Sci. 2017, 86, 75–126. [Google Scholar] [CrossRef]
- He, S.H.; Qin, Y.B.; Walid, E.; Li, L.; Cui, J.; Ma, Y. Effect of ball-milling on the physicochemical properties of maize starch. Biotechnol. Rep. 2014, 3, 54–59. [Google Scholar] [CrossRef]
- Ong, T.S.; Yang, H. Effect of atmosphere on the mechanical milling of natural graphite. Carbon 2000, 38, 2077–2085. [Google Scholar] [CrossRef]
- Zhang, L.M.; Zhang, R.; Zhan, L.; Qiao, W.M.; Liang, X.Y.; Ling, L.C. Efiect of ball-milling technology on pore structure and electrochemical properties of activated carbon. J. Shanghai Univ. (Engl. Ed.) 2008, 12, 372–376. [Google Scholar] [CrossRef]
- Salver-Disma, F.; Lenain, C.; Beaudoin, B.; Aymard, L.; Tarascon, J.M. Unique effect of mechanical milling on the lithium intercalation properties of different carbons. Solid State Ion. 1997, 98, 145–158. [Google Scholar] [CrossRef]
- Salver-Disma, F.; Tarascon, J.M.; Clinard, C.; Rouzaud, J.N. Transmission electron microscopy studies on carbon materials prepared by mechanical milling. Carbon 1999, 37, 1941–1959. [Google Scholar] [CrossRef]
- Liu, D.D.; Jia, B.Y.; Li, S.; Dong, L.J.; Gao, J.H.; Qin, Y.K. Effect of pyrolysis conditions on the improvement of the physicochemical structure of activated carbon obtained from Jixi bituminous coal. Asia Pac. J. Chem. Eng. 2019, 14, 1–12. [Google Scholar] [CrossRef]
- Pietrzak, R. XPS study and physico-chemical properties of nitrogen-enriched microporous activated carbon from high volatile bituminous coal. Fuel 2009, 88, 1871–1877. [Google Scholar] [CrossRef]
- Gong, X.Z.; Guo, Z.C.; Wang, Z. Variation of char structure during anthracite pyrolysis catalyzed by Fe2O3 and its influence on char combustion reactivity. Energy Fuels 2009, 23, 4547–4552. [Google Scholar] [CrossRef]
- Belhachemi, M.; Rios, R.V.; Addoun, F.; Silvestre-Albero, J.; Sepulveda-Escribano, A.; Rodrìguez-Reinoso, F. Preparation of activated carbon from date pits: Effect of the activation agent and liquid phase oxidation. J. Anal. Appl. Pyrolysis 2009, 86, 168–172. [Google Scholar] [CrossRef]
- Yang, K.B.; Peng, J.H.; Xia, H.Y.; Zhang, L.B.; Srinivasakannan, C.; Guo, S.H. Textural characteristics of activated carbon by single step CO2 activation from coconut shells. J. Taiwan Inst. Chem. Eng. 2010, 41, 367–372. [Google Scholar] [CrossRef]
- Heras, F.; Alonso-Morales, N.; Jimenez-Cordero, D.; Gilarranz, M.A.; Rodriguez, J.J. Granular mesoporous activated carbons from waste tires by cyclic oxygen chemisorption-desorption. Ind. Eng. Chem. Res. 2012, 51, 2609–2614. [Google Scholar] [CrossRef][Green Version]
- Karatepe, N.; Orbak, I.; Yavuz, R.; Özyuğuran, A. Sulfur dioxide adsorption by activated carbons having different textural and chemical properties. Fuel 2008, 87, 3207–3215. [Google Scholar] [CrossRef]
- He, X.F.; Jin, L.J.; Wang, D.; Zhao, Y.P.; Zhu, S.W.; Hu, H.Q. Integrated process of coal pyrolysis with CO2 reforming of methane by dielectric barrier discharge plasma. Energy Fuels 2011, 25, 4036–4042. [Google Scholar] [CrossRef]
- Li, W.; Zhu, Y.M. Structural characteristics of coal vitrinite during pyrolysis. Energy Fuels 2014, 28, 3645–3654. [Google Scholar] [CrossRef]
- Davini, P. SO2 adsorption by activated carbons with various burnoffs obtained from a bituminous coal. Carbon 2001, 39, 1387–1393. [Google Scholar] [CrossRef]
- Li, T.; Zhang, L.; Li, D. Effects of gasification atmosphere and temperature on char structural evolution during the gasification of collie sub-bituminous coal. Fuel 2014, 117, 1190–1195. [Google Scholar] [CrossRef]
- Sasezky, A.; Muckenhuber, H.; Grothe, H. Raman microspectroscopy of soot and related carbonaceous materials: Spectral analysis and structural information. Carbon 2005, 43, 1731–1742. [Google Scholar] [CrossRef]
- Sathe, C.; Pang, Y.; Li, C.Z. Effects of heating rate and ion-exchangeable cations on the pyrolysis yields from a Victorian brown coal. Energy Fuels 1999, 13, 748–755. [Google Scholar] [CrossRef]
- Guo, X.; Tay, H.L.; Li, C.Z. Changes in char structure during the gasification of a Victorian brown coal in steam and oxygen at 800 °C. Energy Fuels 2008, 22, 4034–4038. [Google Scholar] [CrossRef]
- Li, Y.; Yang, H.P.; Hu, J.H.; Wang, X.H.; Chen, H.P. Effect of catalysts on the reactivity and structure evolution of char in petroleum coke steam gasification. Fuel 2014, 117, 1174–1180. [Google Scholar] [CrossRef]
- Gaur, V.; Asthana, R.; Verma, N. Removal of SO2 by activated carbon fibers inthe presence of O2 and H2O. Carbon 2006, 44, 46–60. [Google Scholar] [CrossRef]
- Sun, F.; Gao, J.; Liu, X.; Tang, X.; Wu, S. A systematic investigation of SO2 removal dynamics by coal-based activated cokes: The synergic enhancement effect of hierarchical pore configuration and gas components. Appl. Surf. Sci. 2015, 357, 1895–1901. [Google Scholar] [CrossRef]
- Pi, X.X.; Sun, F.; Gao, J.H.; Zhu, Y.W.; Wang, L.J.; Qu, Z.B.; Liu, H.; Zhao, G.B. Microwave irradiation induced high-efficiency regeneration for desulfurized activated coke: A comparative study with conventional thermal regeneration. Energy Fuels 2017, 31, 9693–9702. [Google Scholar] [CrossRef]
- Guo, Y.Y.; Li, Y.R.; Zhu, T.Y.; Ye, M.; Wang, X. Adsorption of SO2 and chlorobenzene on activated carbon. Adsorption 2013, 19, 1109–1116. [Google Scholar] [CrossRef]
- Guo, Y.Y.; Li, Y.R.; Zhu, T.Y.; Ye, M. Effects of concentration and adsorption product on the adsorption of SO2 and NO on activated carbon. Energy Fuels 2013, 27, 360–366. [Google Scholar] [CrossRef]

| Vad | FCad | Aad | Mad | Cdaf | Hdaf | Odaf * | Ndaf | Sdaf |
|---|---|---|---|---|---|---|---|---|
| 39.66 | 56.60 | 0.12 | 3.62 | 74.81 | 19.49 | 4.01 | 1.31 | 0.38 |
| Samples | La (Å) | Lc (Å) | d002 (Å) | N = Lc/d002 |
|---|---|---|---|---|
| Char | 24.81 | 13.48 | 3.50 | 3.85 |
| Char-PA | 25.73 | 13.79 | 3.45 | 3.99 |
| Char-PA-N2-12 | 24.31 | 13.15 | 3.58 | 3.67 |
| Char-PA-N2-48 | 23.19 | 12.43 | 3.72 | 3.34 |
| Char-PA-CO2-12 | 23.91 | 12.86 | 3.69 | 3.48 |
| Char-PA-CO2-48 | 22.28 | 11.79 | 3.96 | 2.98 |
| Char-PA-CO2-48H2 | 22.75 | 12.97 | 4.05 | 3.20 |
| Samples | AD1/AG | AD3/AG | AD4/AG |
|---|---|---|---|
| Char | 3.179 | 1.947 | 0.589 |
| Char-PA | 1.460 | 0.479 | 0.187 |
| Char-PA-N2-12 | 2.014 | 0.481 | 0.385 |
| Char-PA-N2-48 | 3.311 | 0.495 | 0.597 |
| Char-PA-CO2-12 | 2.187 | 0.486 | 0.418 |
| CH-PA-CO2-48 | 3.785 | 0.501 | 0.657 |
| Char-PA-CO2-48H2 | 3.785 | 0.501 | 0.657 |
| Samples | SBET (m2/g) a | Vt (m3/g) b | Vmic (m3/g) c | Non-Vmic (%) d |
|---|---|---|---|---|
| Char | 48.45 | 0.048 | 0.034 | 29.17 |
| Char-PA | 414.78 | 0.24 | 0.10 | 58.33 |
| Char-PA-N2-12 | 715.89 | 0.33 | 0.22 | 33.33 |
| Char-PA-N2-48 | 919.74 | 0.38 | 0.30 | 21.05 |
| Char-PA-CO2-12 | 859.14 | 0.36 | 0.26 | 27.78 |
| Char-PA-CO2-48 | 1259.74 | 0.42 | 0.34 | 19.056 |
| Char-PA-CO2-48H2 | 1214.85 | 0.38 | 0.31 | 18.42 |
| Samples | C–O (%) | C=O (%) | O–C=O (%) | Chemisorbed O (or H2O) (%) |
|---|---|---|---|---|
| Char-PA | 27.3 | 39.6 | 18.4 | 14.7 |
| Char-PA-N2-12 | 29.5 | 35.4 | 25.6 | 9.5 |
| Char-PA-N2-48 | 30.4 | 31.7 | 29.7 | 8.2 |
| Char-PA-CO2-12 | 26.7 | 29.5 | 36.1 | 7.7 |
| Char-PA-CO2-48 | 23.9 | 26.4 | 41.2 | 8.5 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, D.; Hao, Z.; Zhao, X.; Su, R.; Feng, W.; Li, S.; Jia, B. Effect of Physical and Mechanical Activation on the Physicochemical Structure of Coal-Based Activated Carbons for SO2 Adsorption. Processes 2019, 7, 707. https://doi.org/10.3390/pr7100707
Liu D, Hao Z, Zhao X, Su R, Feng W, Li S, Jia B. Effect of Physical and Mechanical Activation on the Physicochemical Structure of Coal-Based Activated Carbons for SO2 Adsorption. Processes. 2019; 7(10):707. https://doi.org/10.3390/pr7100707
Chicago/Turabian StyleLiu, Dongdong, Zhengkai Hao, Xiaoman Zhao, Rui Su, Weizhi Feng, Song Li, and Boyin Jia. 2019. "Effect of Physical and Mechanical Activation on the Physicochemical Structure of Coal-Based Activated Carbons for SO2 Adsorption" Processes 7, no. 10: 707. https://doi.org/10.3390/pr7100707
APA StyleLiu, D., Hao, Z., Zhao, X., Su, R., Feng, W., Li, S., & Jia, B. (2019). Effect of Physical and Mechanical Activation on the Physicochemical Structure of Coal-Based Activated Carbons for SO2 Adsorption. Processes, 7(10), 707. https://doi.org/10.3390/pr7100707





