Assessing the Feasibility of Repurposing the Existing Natural Gas Pipelines for Hydrogen Transport—A Comprehensive Review
Abstract
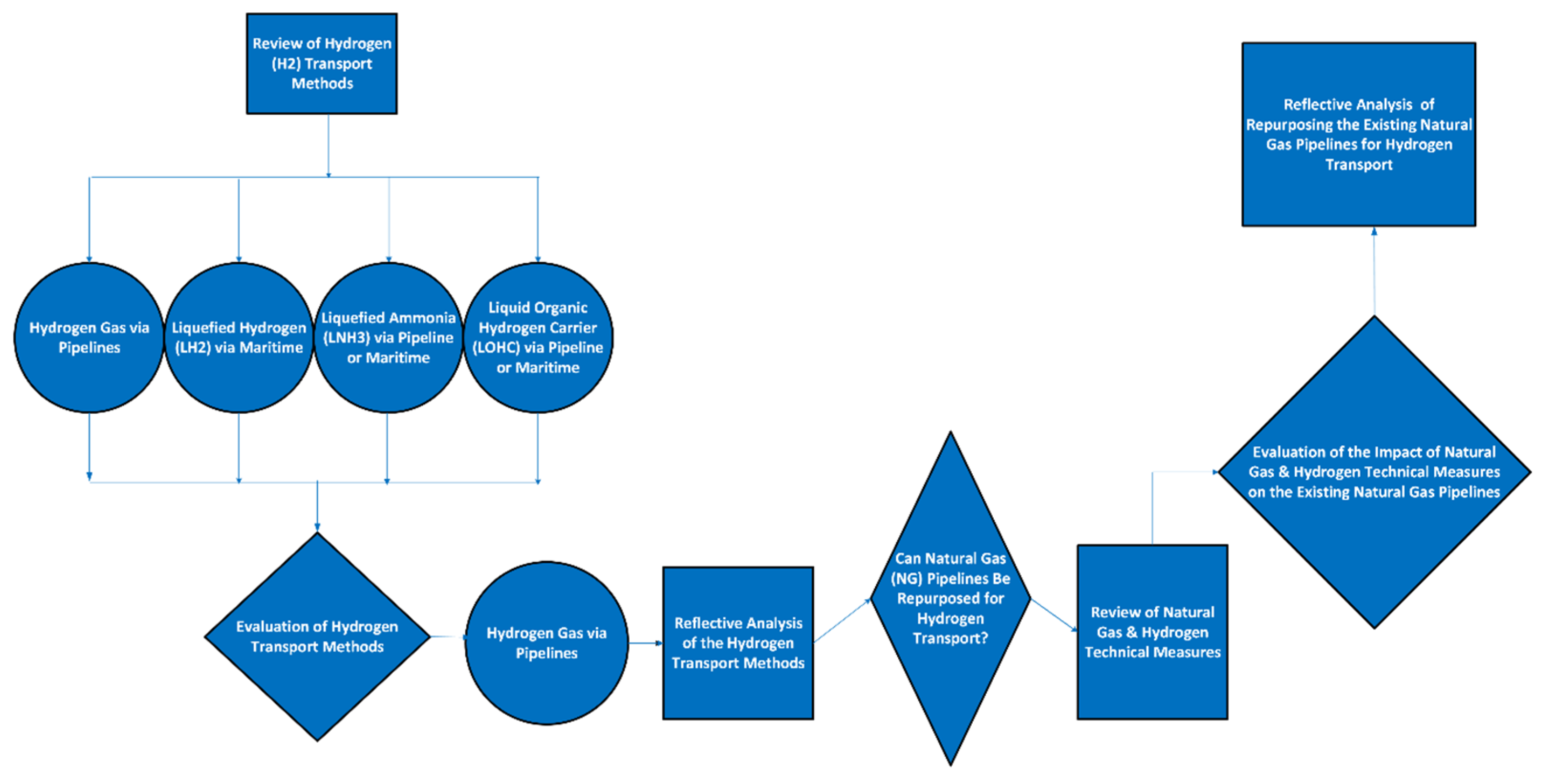
1. Introduction
2. Methodology
3. Review of Existing Hydrogen Transport Methods
3.1. Hydrogen Gas Transport
3.2. Liquefied Hydrogen (LH2) Transport
3.3. Liquefied Ammonia (NH3) Transport
3.4. Liquid Organic Hydrogen Carriers (LOHCs)
3.5. Methanol Transport
4. Evaluation of the Hydrogen Transport Methods
4.1. Reflective Analysis on Hydrogen Transport Methods
4.2. Review of the Existing Natural Gas (NG) Infrastructure
The Natural Gas Infrastructure
5. Repurposing the Existing NG Pipelines for Hydrogen Transport
5.1. Review of Technical Measures
5.1.1. Assessing the Pipeline Materials
5.1.2. Gas Composition
5.1.3. Operating Pressure
5.1.4. Operating Temperature
5.1.5. Volumetric Energy Density
- General energy equation
- AGA equation
- Weymouth equation
- Panhandle A and B
- Q = flowrate of gas, cubic feet per day at base conditions.
- Tb = base absolute temperature, °R (ANSI 2530 specification: Tb = 520°R).
- Pb = base absolute pressure, psia (ANSI 2530 specification: Pb = 14.73 psia)
- E = pipeline efficiency factor (fraction).
- Ff = Fanning friction factor
- P1 = inlet pressure, psia
- P2 = outlet pressure, psia
- S = specific gravity of flowing gas (air = 1.0)
- Lm = length of line, miles.
- Tavg = average temperature, °R, [Tavg = 1/2 (Tin + Tout)]
- Zavg = average compressibility factor
- D = internal diameter of pipe, feet
5.1.6. Operating Density
- Ve = fluid erosional velocity (feet/s)
- C = empirical constant
- Qm = gas/liquid mixture at flowing pressure and temperature (lbs./ft3)
- Ve = erosional velocity (m/s)
- N = constant (1.22) to convert equation 2 to metric unit from field unit
- C = the empirical constant (varies from 100 to 250)
- ρ = gas density (kg/m3)
- Vmax is erosional velocity in m/s.
- P is gas pressure in kPa.
- T is gas temperature in K.
- Z is compressibility factor at pipeline conditions and is dimensionless.
- R is ideal gas constant in (8.314 kPa·m3/kg·mol ·K).
- G is gas gravity
5.1.7. Pressure Drop
- f = Darcy’s friction factor.
- Re = Reynolds number.
- and ε/D = relative roughness of the pipe.
- f, Re and ε/D are all dimensionless [1].
5.2. Evaluating the Feasibility of Repurposing of the Existing NG Pipelines for H2 Transport
6. Reflective Analysis on the Repurposing Existing NG Pipelines for H2 Transport
7. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
| H2 | Hydrogen |
| NG | Natural Gas |
| LH2 | Liquified Hydrogen |
| BP | Boiling Point |
| BOG | Boil-Off Gas |
| MW | Molecular Weight |
| LOHC | Liquid Organic Hydrogen Carrier |
| MCH | Methylcyclohexane |
| NH3 | Ammonia |
| CH3OH | Methanol |
| CO2 | Carbon Dioxide |
| CO | Carbon Monoxide |
| KM | Kilometer |
| DP | Pressure Drop |
| J/Scm | Joules per Standard cubic meter |
| Btu/Scf | British thermal unit per Standard cubic meter |
| CH4 | Methane |
| JT | Joule Thomson |
| PE | Polyethylene |
| ASME | American Society of Mechanical Engineers |
| O2 | Oxygen |
| SO2 | Sulphur (IV) Oxide |
| °C | Degree Celsius |
| PIP | Pipe-in-Pipe |
| MOP | Maximum Operating Pressure |
| GHG | Green House Gas |
| K | Kelvin |
| ILI | In-line Inspection |
| MJ/Sm3 | Mega-Joule per Standard cubic meter |
| Kg/m3 | Kilogram per cubic meter |
References
- Abbas, A.J.; Hassani, H.; Burby, M.; John, I.J. An Investigation into the Volumetric Flow Rate Requirement of Hydrogen Transportation in Existing Natural Gas Pipelines and Its Safety Implications. Gases 2021, 1, 156–179. [Google Scholar] [CrossRef]
- Yusaf, T.; Fernandes, L.; Abu Talib, A.R.; Altarazi, Y.S.M.; Alrefae, W.; Kadirgama, K.; Ramasamy, D.; Jayasuriya, A.; Brown, G.; Mamat, R.; et al. Sustainable Aviation—Hydrogen Is the Future. Sustainability 2022, 14, 548. [Google Scholar] [CrossRef]
- Osman, A.I.; Mehta, N.; Elgarahy, A.M.; Hefny, M.; Al Hinai, A.; Al Muhtaseb, A.H.; Rooney, D.W. Hydrogen production, storage, utilisation, and environmental impacts: A review. Environ. Chem. Lett. 2021, 20, 153–188. [Google Scholar] [CrossRef]
- Zabrzeski, Ł.; Janusz, P.; Liszka, K.; Łaciak, M.; Szurlej, A. Hydrogen—Natural Gas mixture compression in case of transporting through high-pressure gas pipelines. IOP Conf. Ser. Earth Environ. Sci. 2019, 214, 012137. [Google Scholar] [CrossRef]
- Castillo, C.A.R.; Yeter, B.; Li, S.; Brennan, F.; Collu, M. A critical review of challenges and opportunities for the design and operation of offshore structures supporting renewable hydrogen production, storage, and transport. J. Wind. Energy Sci. 2023, 9, 533–554. [Google Scholar] [CrossRef]
- Calado, G.; Castro, R. Hydrogen Production from Offshore Wind Parks: Current Situation and Future Perspectives. Appl. Sci. 2021, 11, 5561. [Google Scholar] [CrossRef]
- Lipiäinen, S.; Lipiäinen, K.; Ahola, A.; Vakkilainen, E. Use of existing gas infrastructure in European hydrogen economy. Int. J. Hydrogen Energy 2023, 48, 31317–31329. [Google Scholar] [CrossRef]
- Jens, J.; Wang, A.; van der Leun, K.; Peters, D.; Buseman, M. Extending the European Hydrogen Backbone; European Hydrogen Backbone Initiative, Guidehouse: Utrecht, The Netherlands, 2021; Available online: https://ehb.eu/files/downloads/European-Hydrogen-Backbone-April-2021-V3.pdf (accessed on 30 March 2024).
- Gondal, I.A. Hydrogen Transportation by Pipelines. Compendium of Hydrogen Energy; Elsevier Ltd.: Amsterdam, The Netherlands, 2016. [Google Scholar]
- Topolski, K.; Reznicek, E.P.; Erdener, B.C.; Marchi, C.W.S.; Ronevich, J.A.; Fring, L.; Simmons, K.; Fernandez, O.J.G.; Hodge, B.-M.; Chung, M. Hydrogen Blending into Natural Gas Pipeline Infrastructure: Review of the State of Technology; National Renewable Energy Laboratory (NREL): Golden, CO, USA, 2022. [CrossRef]
- Aminul, I.; Tahrim, A.; Nathan, S.; Kara, E.; David, B.; Manuel, H. Hydrogen blending in natural gas pipelines: A comprehensive review of material compatibility and safety considerations. Int. J. Hydrogen Energy 2024, 93, 1429–1461. [Google Scholar] [CrossRef]
- Zhang, C.; Shao, Y.; Shen, W.; Li, H.; Nan, Z.; Dong, M.; Bian, J.; Cao, X. Key technologies of pure hydrogen and hydrogen-mixed natural gas pipeline transportation. ACS Omega 2023, 8, 19212–19222. [Google Scholar] [CrossRef]
- Patonia, A.; Lenivova, V.; Poudineh, R.; Nolden, C. Hydrogen Pipelines vs. HVDC Lines: Should We Transfer Green Molecules or Electrons? OIES Paper ET27; Oxford Institute for Energy Studies: Oxford, UK, 2023. [Google Scholar]
- International Energy Agency (IEA). The Future of Hydrogen Seizing Today’s Opportunities; Report prepared by the IEA for the G20, Japan; International Energy Agency (IEA): Paris, France, 2019; Available online: https://www.oecd.org/en/publications/the-future-of-hydrogen_1e0514c4-en.html#page1 (accessed on 30 March 2024).
- Patonia, A.; Poudineh, R. Global Trade of Hydrogen: What Is the Best Way to Transfer Hydrogen Over Long Distances? The Oxford Institute for Energy Studies (OIES): Oxford, UK, 2022; Available online: https://www.oxfordenergy.org/publications/global-trade-of-hydrogen-what-is-the-best-way-to-transfer-hydrogen-over-long-distances/ (accessed on 30 March 2024).
- Restelli, F.; Spatolisano, E.; Pellegrini, L.A. Hydrogen Liquefaction: A Systematic Approach to its Thermodynamic Modeling. Chem. Eng. Trans. 2023, 99, 433–438. [Google Scholar] [CrossRef]
- Parfomak, P.W. Pipeline Transportation of Hydrogen: Regulation, Research, and Policy; Congressional Research Service: Washington, DC, USA, 2021. Available online: https://crsreports.congress.gov (accessed on 30 March 2024).
- Wang, Y.Y. Hydrogen Piping and Pipelines (Pressure Boundary Integrity and Material Considerations); Engineering Mechanics Corporation of Columbus (EMC): Columbus, OH, USA, 2007. [Google Scholar]
- Risco-Bravo, A.; Varela, C.; Bartels, J.; Zondervan, E. From green hydrogen to electricity: A review on recent advances, challenges, and opportunities on power-to-hydrogen-to-power systems. Renew. Sustain. Energy Rev. 2023, 189, 113930. [Google Scholar] [CrossRef]
- Mohitpour, M.; Golsham, H.; Murray, A. Pipeline Design & Construction: A Practical Approach, 2nd ed.; American Society of Mechanical Engineers: New York, NY, USA, 2003. [Google Scholar] [CrossRef]
- Crabtree, G.W.; Dresselhaus, M.S.; Buchanan, M.V. The hydrogen economy. Phys. Today 2004, 57, 39–44. [Google Scholar] [CrossRef]
- European Union Agency for the Cooperation of Energy Regulators (EUACER). Transporting Pure Hydrogen by Repurposing Existing Gas Infrastructure: Overview of Existing Studies and Reflections on the Conditions for Repurposing; EUACER: Ljubljana, Slovenia, 2021; Available online: https://acer.europa.eu/sites/default/files/documents/Publications/Transporting%20Pure%20Hydrogen%20by%20Repurposing%20Existing%20Gas%20Infrastructure_Overview%20of%20studies.pdf (accessed on 30 March 2024).
- Gondal, I.A.; Sahir, M.H. Prospects of natural gas pipeline infrastructure in hydrogen transportation. Int. J. Energy Res. 2012, 36, 1338–1345. [Google Scholar] [CrossRef]
- Khan, M.A.; Young, C.; Layzell, D.B. Techno-Economics of Hydrogen Pipelines. Transit. Accel. Tech. Briefs 2021, 1, 1–40. [Google Scholar]
- Yang, D.; Kim, J.; Lee, S.; Park, J. Detection of hydrogen gas leak using distributed temperature sensor in green hydrogen system. Int. J. Hydrogen Energy 2024, 82, 910–922. [Google Scholar] [CrossRef]
- Xu, K. 14—Hydrogen embrittlement of carbon steels and their welds. Gaseous Hydrogen Embrittlement of Materials in Energy Technologies: The Problem, Its Characterization and Effects on Particular Alloy Classes; Woodhead Publishing: Cambridge, UK, 2012; Volume 2, pp. 526–561. [Google Scholar] [CrossRef]
- Haeseldonckx, D.; D’haeseleer, W. The Use of the Natural-Gas Pipeline Infrastructure for Hydrogen Transport in a Changing Market Structure. Int. J. Hydrogen Energy 2006, 32, 1381–1386. [Google Scholar] [CrossRef]
- Staffell, I.; Scamman, D.; Velazquez Abad, A.; Balcombe, P.; Dodds, P.E.; Ekins, P.; Shah, N.; Ward, K.R. The role of hydrogen and fuel cells in the global energy system. Energy Environ. Sci. 2019, 12, 463–491. [Google Scholar] [CrossRef]
- Oriani, R.A. A mechanistic theory of hydrogen embrittlement of steels. Berichte Bunsenges. Phys. Chem 1972, 76, 848–857. [Google Scholar] [CrossRef]
- Robertson, I.M.; Sofronis, P.; Nagao, A.; Martin, M.; Wang, S.; Gross, D.W.; Nygren, K.E. Hydrogen embrittlement understood. Metall. Mater. Trans. B 2015, 46, 1085–1103. [Google Scholar] [CrossRef]
- Olden, V.; Thaulow, C.; Johnsen, R. Modelling of hydrogen diffusion and hydrogen induced cracking in weldments. Mater. Des. 2008, 29, 1934–1948. [Google Scholar] [CrossRef]
- Long, Y.; Song, W.; Fu, A.; Xie, J.; Feng, Y.; Bai, Z.; Yin, C.; Ma, Q.; Ji, N.; Kuang, X. Combined effect of hydrogen embrittlement and corrosion on the cracking behaviour of C110 low alloy steel in O2-contaminated H2S environment. Corros. Sci. 2022, 194, 109926. [Google Scholar] [CrossRef]
- Aziz, M.; Wijayanta, A.T.; Nandiyanto, A.B.D. Ammonia as Effective Hydrogen Storage: A Review on Production, Storage and Utilization. Energies 2020, 13, 3062. [Google Scholar] [CrossRef]
- International Energy Agency (IEA). Hydrogen Production and Storage—R&D Priorities and Gap; OECD/IEA: Paris, France, 2006; Available online: https://www.iea.org/reports/hydrogen-production-and-storage (accessed on 30 March 2024).
- Serpell, O.; Hsain, Z.; Chu, A.; Johnsen, W. Ammonia’s Role in a Net-Zero Hydrogen Economy; Kleinman Center for Energy Policy: Philadelphia, PA, USA, 2023; Available online: https://kleinmanenergy.upenn.edu/research/publications/ammonias-role-in-a-net-zero-hydrogen-economy/ (accessed on 30 March 2024).
- Jeerh, G.; Zhang, M.; Tao, S. Recent progress in ammonia fuel cells and their potential applications. J. Mater. Chem. 2020, 9, 727–752. [Google Scholar] [CrossRef]
- Southall, E.; Lukashuk, L. Hydrogen Storage and Transportation Technologies to Enable the Hydrogen Economy: Liquid Organic Hydrogen Carriers (Overview and perspectives on liquid organic hydrogen carriers’ technology). Johns. Matthey Technol. Rev. 2022, 66, 246–258. [Google Scholar] [CrossRef]
- Melcher, B.U.; George, M.; Paetz, C. Liquid Organic Hydrogen Carriers—A Technology to Overcome Common Risks of Hydrogen Storage. In Proceedings of the International Conference on Hydrogen Safety, Edinburgh, UK, 21–23 September 2021; pp. 1–9. [Google Scholar]
- New Jersey Department of Health and Senior Services. Hazardous Substance Fact Sheet (Common Name: Methylcyclohexane); Department of Health and Services: Trenton, NJ, USA, 1997. Available online: https://nj.gov/health/eoh/rtkweb/documents/fs/1242.pdf (accessed on 30 March 2024).
- Roland Berger. Hydrogen Transportation|The Key to Unlocking the Clean Hydrogen Economy; Roland Berger GMBH: Munich, Germany, 2021; Available online: https://globalhydrogenhub.com/report-presentation/hydrogen-transportation-the-key-to-unlocking-the-clean-hydrogen-economy (accessed on 30 March 2024).
- IRENA. Global Hydrogen Trade to Meet the 1.5 °C Climate Goal: Part II—Technology Review of Hydrogen Carriers; International Renewable Energy Agency: Abu Dhabi, United Arab Emirates, 2022; Available online: https://www.irena.org/publications/2022/Apr/Global-hydrogen-trade-Part-II (accessed on 30 March 2024).
- OECD. Risk-Based Regulatory Design for the Safe Use of Hydrogen. 2023. Available online: https://www.oecd.org (accessed on 25 March 2026). [CrossRef]
- Folga, S.M. Natural Gas Pipeline Technology Overview; Argonne National Laboratory (Environmental Science Division): Oak Ridge, TN, USA, 2007. Available online: https://www.osti.gov/biblio/925391 (accessed on 30 March 2023). [CrossRef]
- Schmura, E.; Klingenberg, M.V. G 1 Existing Natural Gas Pipeline Materials and Associated Operational Characteristics; DOE: Pittsburgh, PA, USA, 2006. Available online: https://www.hydrogen.energy.gov/docs/hydrogenprogramlibraries/pdfs/progress05/v_g_1_schmura.pdf (accessed on 30 March 2024).
- ASME B31.12; Hydrogen Piping and Pipelines: ASME Code for Pressure Piping, B31 (An American National Standard). The American Society of Mechanical Engineers: New York, NY, USA, 2019.
- European Industrial Gases Association (EIGA). Gaseous Hydrogen Installations (EIGA Doc 15/21); EIGA/AISBL: Brussels, Belgium, 2021; Available online: https://www.eiga.eu/uploads/documents/DOC015.pdf (accessed on 30 March 2024).
- Melaina, M.W.; Antonia, O.; Penev, M. Blending Hydrogen into Natural Gas Pipeline Networks: A Review of Key Issues; National Renewable Energy Laboratory: Golden, CO, USA, 2013. [Google Scholar] [CrossRef]
- Mohitpour, M.; Golshan, H.; Murray, A. Pipeline Design and Construction: A Practical Approach, 4th ed.; ASME Press: New York, NY, USA, 2017. [Google Scholar]
- API. API Specification 5L: Specification for Line Pipe, 46th ed.; American Petroleum Institute: Washington, DC, USA, 2018. [Google Scholar]
- San Marchi, C.; Somerday, B.; Robinson, S. Technical Reference for Hydrogen Compatibility of Materials; Sandia National Laboratories: Livermore, CA, USA, 2017. [Google Scholar]
- American Water Works Association (AWWA). AWWA Standard for Polyethylene (PE) Pressure Pipe and Fittings, 4 in (100 mm) Through 63 in (1575 mm), for Water Distribution and Transmission, 3rd ed.; American Water Works Association (AWWA): Denver, CO, USA, 2000. [Google Scholar]
- Backman, M. Transport of Hydrogen with Polyethylene natural gas pipes [PE]. In Proceedings of the Plastic Pipes Conference & Exhibition, Vancouver, BC, Canada, 20–22 September 2010; Plastic Pipes Conference Association: Brussels, Belgium, 2010; Available online: https://www.pe100plus.com/PPCA/Transport-of-hydrogen-with-polyethylene-natural-gas-pipes-p433.html (accessed on 30 March 2024).
- Lynch, S.P. Hydrogen embrittlement phenomena and mechanisms. Corros. Rev. 2012, 30, 105–123. [Google Scholar] [CrossRef]
- Song, J.; Curtin, W.A. A nanoscale mechanism of hydrogen embrittlement in metals. Acta Mater. 2013, 61, 6826–6843. [Google Scholar] [CrossRef]
- Wang, A.; Wang, B.; Jiang, R.; Nie, R.; Cui, G.; Liu, J.; Zhang, Y.; Zhang, H.; Xing, X. Hydrogen induced crack propagation of X80 steel under alternating pressures. Materials 2025, 18, 947. [Google Scholar] [CrossRef]
- Yu, L.; Feng, H.; Li, S.; Guo, Z.; Chi, Q. Study on hydrogen embrittlement behavior of X65 pipeline steel in gaseous hydrogen environment. Metals 2025, 15, 596. [Google Scholar] [CrossRef]
- Chalfoun, D.R.; Parker, J.; Gagliano, M.; Martínez Pañeda, E. Tailored heat treatments to characterise the fracture resistance of critical weld regions in hydrogen transmission pipelines. Int. J. Hydrogen Energy 2025, 192, 152347. [Google Scholar] [CrossRef]
- Télessy, K.; Barner, L.; Holz, F. Repurposing Natural Gas Pipelines for Hydrogen: Limits and Options from a Case Study in Germany. Int. J. Hydrogen Energy 2024, 80, 821–831. [Google Scholar] [CrossRef]
- Zhang, H.; Zhao, J.; Li, J.; Yu, B.; Wang, J.; Lyu, R.; Xi, Q. Research progress on corrosion and hydrogen embrittlement in hydrogen–natural gas pipeline transportation. Nat. Gas Ind. B 2023, 10, 570–582. [Google Scholar] [CrossRef]
- Liu, Q.; Yan, Y.; Iqbal, M.Z.; Aissa, B.; Farhat, H. Enhanced hydrogen embrittlement resistance of directed energy deposition 316L by post-manufacturing heat treatment. npj Mater. Degrad. 2025, 9, 62. [Google Scholar] [CrossRef]
- Sun, C.; Zhang, Z.; Li, Y.; Liu, X.; Wang, H.; Han, E. Effects of hydrogen blending ratios and CO2 on hydrogen embrittlement of X65 steel in high-pressure offshore hydrogen-blended natural gas pipelines. J. Mater. Res. Technol. 2024, 33, 4763–4771. [Google Scholar] [CrossRef]
- Buck, Z.; Moser, N.; Derimow, N.; Martin, M.L.; Lauria, D.; Lucon, E.; Stalheim, D.; Bradley, P.; Connolly, M. Assessing Girth Weld Quality of Pipeline Steels and Their Susceptibility to Hydrogen Embrittlement; NIST: Gaithersburg, MD, USA, 2024. [Google Scholar]
- Wang, B.; Liu, Q.; Feng, Q.; Wang, X.; Yang, Z.; Dai, L.; Huo, X.; Wang, D.; Yu, J.; Chen, J. Influence of welding defects on hydrogen embrittlement sensitivity of girth welds in X80 pipelines. Int. J. Electrochem. Sci. 2024, 19, 100661. [Google Scholar] [CrossRef]
- Mokhatab, S.; Poe, W.A. Handbook of Natural Gas Transmission and Processing; Gulf Professional Publishing: Waltham, MA, USA, 2012. [Google Scholar]
- Faucon, L.E.; Boot, T.; Riemslag, T.; Scott, S.P.; Liu, P.; Popovich, V. Hydrogen-accelerated fatigue of API X60 pipeline steel and its weld. Metals 2023, 13, 563. [Google Scholar] [CrossRef]
- Bouledroua, O.; Zelmati, D.; Hafsi, Z.; Djukic, M.B. Hydrogen embrittlement effects on remaining life and fatigue crack growth rate in API 5L X52 steel pipelines under cyclic pressure loading. Eng. Fail. Anal. 2024, 166, 108917. [Google Scholar] [CrossRef]
- Shourideh, M.; Yasseri, S.; Bahai, H. Hydrogen pipelines safety using system dynamics. Hydrogen 2025, 6, 81. [Google Scholar] [CrossRef]
- Gangloff, R.P.; Somerday, B.P. (Eds.) Gaseous Hydrogen Embrittlement of Materials in Energy Technologies: Volume 1: The Problem, Its Characterisation and Effects on Particular Alloy Classes; Woodhead Publishing: Cambridge, UK, 2012. [Google Scholar]
- Gas Processors Suppliers Association (GPSA). Engineering Databook, 12th ed.; Gas Processors Suppliers Association: Tulsa, OK, USA, 2004; Available online: http://www.gasprocessors.com/ (accessed on 30 March 2024).
- American Petroleum Institute. Recommended Practice for Design and Installation of Offshore Production Platform Piping Systems, 5th ed.; API Recommended Practice 14E (RP 14E); American Petroleum Institute: Washington, DC, USA, 1991. [Google Scholar]
- Kuczyński, S.; Łaciak, M.; Olijnyk, A.; Szurlej, A.; Włodek, T. Thermodynamic and Technical Issues of Hydrogen Mixtures Pipeline Transmission. Energies 2019, 12, 569. [Google Scholar] [CrossRef]
- European Industrial Gases Association (EIGA). Hydrogen Pipeline Systems (IGC Doc 121/14); EIGA/AISBL: Brussels, Belgium, 2014; Available online: https://www.eiga.eu/uploads/documents/DOC121.pdf (accessed on 30 March 2024).

| H2 Gas | LH2 | NH3 | LOHC (MCH) | |
|---|---|---|---|---|
| Molecular Weight (Wt.), g/mol | 2.016 | 2.016 | 17.031 | 98.186 |
| Density in normal conditions, Kg/m3 | 0.08375 | 0.08375 | 0.73 | 866.9 |
| Melting point, C | −259.16 | −259.16 | −77.73 | −126.3 |
| Explosive limit in air, vol% | 4 to 75 | 4 to 75 | 15 to 28 | 1.2 to 6.7 |
| Flame speed, cm/s | 346 | 346 | - | - |
| Liquefaction energy, MJ/Kg | - | 15.1 to 57 | >6.73 | - |
| Transport pressure, Barg | 100 | 1.013 | 1.013 | 1.013 |
| Transport temp, C | 20 | −252.87 | −33.34 | 20 to 25 |
| Density in transport conditions, kg/m3 | 7.8 | 71.1 | 686 | 866.9 |
| BOG, % day | - | 0.06 to 0.4 | 0.024 to 0.1 | 0.00416 to 0.065 |
| Gravimetric energy density, MJ/Kg | 120 | 120 | 21.18 to 22.5 | 7.35 |
| Gravimetric H2 content % | 100 | 100 | 17.8 | 6.1 |
| Volumetric energy density, MJ/Nm3 [Wh/L] | 13 [8.49] | 13 [8.49] | [12.92 to 14.4] | [5.66] |
| Volumetric H2 content kg/m3 | 100 | 70.8 | 121 | 47.1 |
| Dehydrogenation energy (MJ/Kg) | - | - | 30.67 | >43.4 |
| H2 Transport Methods | Pros | Cons |
|---|---|---|
| H2 Gas in Pipelines |
|
|
| LH2 |
|
|
| Liquid Ammonia |
|
|
| LOHC (MCH) |
|
|
| Component | Mol Fraction (%) |
|---|---|
| Methane | 93.76 |
| Ethane | 3.14 |
| Propane | 0.62 |
| Butane | 0.2 |
| Pentane | 0.07 |
| Nitrogen | 2.03 |
| Carbon dioxide | 0.18 |
| NG Transport in Pipelines | H2 Transport in Repurposed Pipelines | Implication of H2 on the Existing NG Pipeline | Possible Mitigations | |
|---|---|---|---|---|
| Gas Composition | The main component of the NG transported through the NG pipelines is methane (CH4) | If NG pipelines are repurposed to transport pure H2 gas, 100% H2 becomes the major component flowing through the existing NG pipeline | Metal pipelines can be compromised and degraded with prolonged exposure to H2 gas at high concentrations and pressure | Proactive monitoring and rigorous assessment of the pipelines’ welds and joints will help identify any possibility of embrittlement and pipeline degradation |
| Operating Pressure | The NG pipeline infrastructure can be operated up to 138 barg at operating temperatures between 6.7 °C and 60 °C. | The maximum operating pressure of a pipeline transporting H2 gas is 100 barg at about 20 °C | Pressure increase will impact embrittlement which can compromise operational safety. | H2 transport should typically be operated between 30 and 100 barg at <50 °C utilizing pipelines of low-strength material grades (<API X46) and high-strength material (X52). |
| Operating Temperature | Due to JT effect, when the pressure of NG is reduced, temperature drops by 0.5 °C for every 1 bar reduction. JT effect in NG can plug transmission materials and cause safety issues. | In H2 pipelines, when pressure is reduced, H2 temperature increases by 0.035 °C for every 1 Bar reduction, thus no JT issues. | For H2 transport, JT effect does not negatively impact the existing NG pipelines. | |
| Volumetric Energy Density | NG volumetric energy density (35.8 MJ/Sm3) is three times the volumetric energy density of H2 (10.8 MJ/Sm3) | H2 less volumetric energy density (10.8 MJ/Sm3) is expected to make its flowrate in the NG pipeline much greater. | Erosional velocity can be exceeded, with consequences of pipeline erosion and leakage. | Maintain the H2 transport velocity below the erosional velocity. |
| Operating Density | NG density is much greater than the H2 gas density | H2 is the lightest molecule with a very small density, 0.08375 kg/m3 | H2 low density means that it travels faster compared to NG in gas pipelines with potential safety concern if the higher velocity reaches erosional limit. | Maintain the H2 transport velocity below the erosional velocity. |
| Material Grade | Hydrogen Concentration | Operating Pressure Levels | Associated Risks | Notes |
|---|---|---|---|---|
| API 5L A, B, API 5L X42, X46 | 100% | Low (<10 barg) | Low | Higher ductility and greater resistance to hydrogen-induced cracking [48,49]. |
| API 5L A, B, API 5L X42 | 100% | Medium (30 to 60 barg) | Low | Higher ductility and greater resistance to hydrogen-induced cracking [48,49]. Failures are rare in these operating pressure conditions [48]. |
| API 5L A, B, API 5L X42, X46 | 100% | Medium (10 to 100 barg) | Low to Medium | Higher ductility and greater resistance to hydrogen-induced cracking [48,49]. Employed for H2 pipelines under normal operating conditions [24]. Normal operating pressure is between 30 barg and 60 barg [20]. |
| API 5L X42-X52 | 100% | Medium (10 to 100 barg) | Low to Medium | Good ductility when exposed to hydrogen [55]. Reference [45] declares that API grades below X42 and X52 are less affected by H2 embrittlement and are approved for hydrogen pipelines. Normal operating pressure is between 30 barg and 60 barg [20]. |
| Polyethylene (PE) | 100% | <17.5 barg | Low | Polyethylene (PE) materials are not susceptible to embrittlement [52]. Typically, they are rated 17.5 [51] barg and should not be used to transport gaseous hydrogen at elevated pressures. |
| API 5L X60, X65, X70, X80 | 100% | Medium (10 to 100 barg) | High | More dislocations and microstructural defects [30,53,54]. Vulnerable to hydrogen embrittlement, hydrogen-induced cracking and crack initiation [50]. Reduced ductility and fatigue strength with increasing hydrogen concentration and high stress [56,57]. Undergo heat treatment or reduce operating pressure to manage the risk of hydrogen embrittlement [57]. It can be re-used with major modifications such as pipe-in-pipe [58]. |
| Technical Guidelines | Implementation Measures |
|---|---|
| Material compatibility | Use of appropriate steel material grade (<X52) |
| Operating pressure | Review operating pressure and derating if necessary |
| Pipeline modification | Apply pipe-in-pipe modification technique |
| Pipeline integrity assessment | Conduct in-line inspection (ILI) prior to repurposing |
| Monitoring | Apply distributed fiber-optic sensing for real-time leak detection |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Ayodele, O.F.; Ali, D. Assessing the Feasibility of Repurposing the Existing Natural Gas Pipelines for Hydrogen Transport—A Comprehensive Review. Processes 2026, 14, 1182. https://doi.org/10.3390/pr14071182
Ayodele OF, Ali D. Assessing the Feasibility of Repurposing the Existing Natural Gas Pipelines for Hydrogen Transport—A Comprehensive Review. Processes. 2026; 14(7):1182. https://doi.org/10.3390/pr14071182
Chicago/Turabian StyleAyodele, Oluwole Foluso, and Dallia Ali. 2026. "Assessing the Feasibility of Repurposing the Existing Natural Gas Pipelines for Hydrogen Transport—A Comprehensive Review" Processes 14, no. 7: 1182. https://doi.org/10.3390/pr14071182
APA StyleAyodele, O. F., & Ali, D. (2026). Assessing the Feasibility of Repurposing the Existing Natural Gas Pipelines for Hydrogen Transport—A Comprehensive Review. Processes, 14(7), 1182. https://doi.org/10.3390/pr14071182







