Formulation Optimization of GG/SS/PVA/GEL Composite Hydrogels for Extrusion-Based Bioprinting Using Response Surface Methodology
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials and Reagents
2.2. Instruments and Equipment
2.3. Experimental Methods
2.3.1. Preparation of Hydrogel
2.3.2. 3D Printing of Gel
2.3.3. Printing Fidelity Experiment
2.3.4. Mechanical Properties Testing
2.3.5. Swelling Performance Experiment
2.3.6. Enzymatic Degradation Experiment
2.3.7. Single-Factor Experiment
2.3.8. Response Surface Experiment
2.3.9. Microstructural Characterization
2.3.10. Chemical Structure Characterization
2.3.11. Rheological Property Testing
2.3.12. Scaffold Printability Evaluation
2.4. Data Analysis
3. Results
3.1. Single-Factor Experiment Results
3.1.1. GG Addition Amount
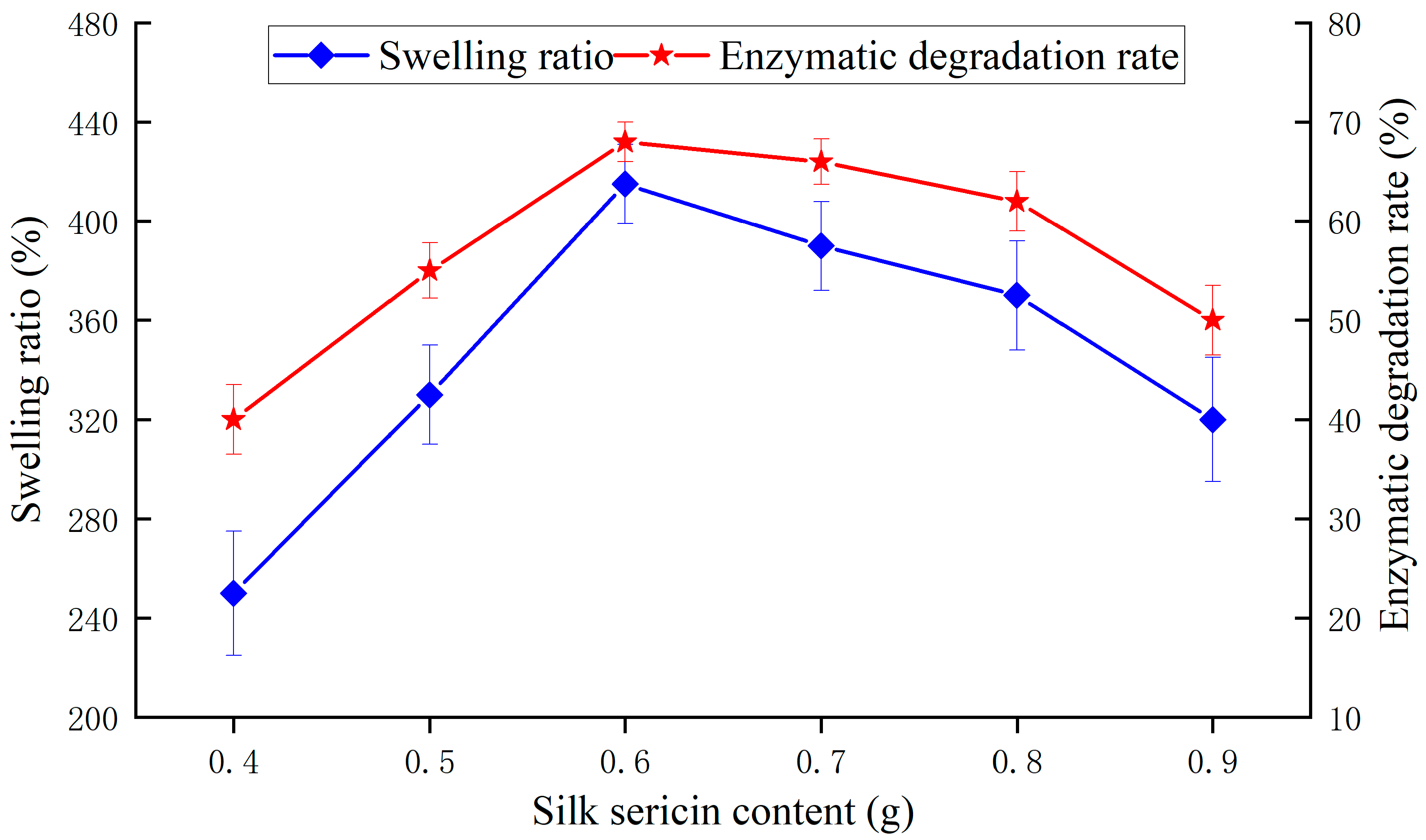
3.1.2. SS Content
3.1.3. PVA Content
3.2. Response Surface Experimental Results and Analysis
3.2.1. Optimization Results and Analysis of Response Variables
3.2.2. Analysis of the Interaction Effects Between Two Factors
3.2.3. Validation Experiment
FTIR Chemical Characterization
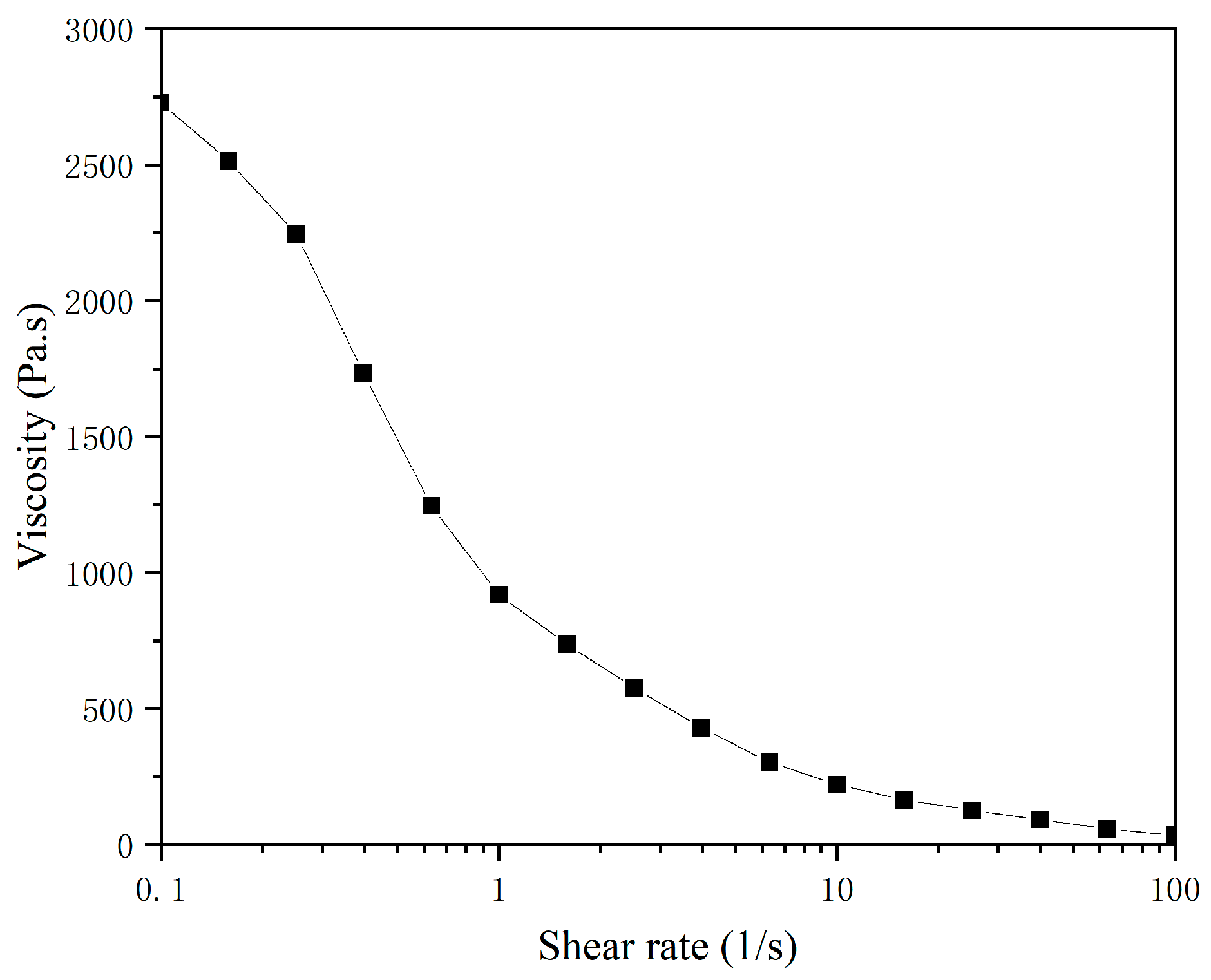
Rheological Property Testing
Scanning Electron Microscopy (SEM) Analysis
Scaffold Printing and Secondary Crosslinking Evaluation
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Kothandaraman, L.; Balasubramanian, N.K.; Palaniyappan, S.; Dalaq, A.S.; Bodaghi, M. Material extrusion techniques for biomedical applications: Biomaterials and their performance—A review. Mater. Today Commun. 2025, 49, 113707. [Google Scholar] [CrossRef]
- MacDonald, E.; Wicker, R. Multiprocess 3D printing for increasing component functionality. Science 2016, 353, aaf2093. [Google Scholar] [CrossRef] [PubMed]
- Thareja, P.; Swarupa, S.; Ahmad, S.; Jinugu, M.E. Hydrogel-based inks for extrusion 3D printing: A rheological viewpoint. Curr. Opin. Colloid Interface Sci. 2025, 77, 101918. [Google Scholar] [CrossRef]
- Lameirinhas, N.S.; Teixeira, M.C.; Carvalho, J.P.; Valente, B.F.; Pinto, R.J.; Oliveira, H.; Luís, J.L.; Pires, L.; Oliveira, J.M.; Vilela, C.; et al. Nanofibrillated cellulose/gellan gum hydrogel-based bioinks for 3D bioprinting of skin cells. Int. J. Biol. Macromol. 2023, 229, 849–860. [Google Scholar] [CrossRef]
- Osmałek, T.; Froelich, A.; Tasarek, S. Application of gellan gum in pharmacy and medicine. Int. J. Pharm. 2014, 466, 328–340. [Google Scholar] [CrossRef]
- Ismail, N.A.; Amin, K.A.M.; Majid, F.A.A.; Razali, M.H. Gellan gum incorporating titanium dioxide nanoparticles biofilm as wound dressing: Physicochemical, mechanical, antibacterial properties and wound healing studies. Mater. Sci. Eng. C 2019, 103, 109770. [Google Scholar] [CrossRef]
- Jin, X.; Wang, C.; Sun, Z.; Lian, Y.; Ji, Q.; Tang, J.; Ma, X. Biocompatible dually reinforced gellan gum hydrogels with selective antibacterial activity. Carbohydr. Polym. 2025, 351, 123071. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Liu, H.; Shi, X.; Qin, S.; Liu, J.; Lv, Q.; Liu, J.; Li, Q.S.; Wang, Z.; Wang, L. Development and application of an advanced biomedical material-silk sericin. Adv. Mater. 2024, 36, 2311593. [Google Scholar] [CrossRef]
- Nayak, S.; Talukdar, S.; Kundu, S.C. Potential of 2D crosslinked sericin membranes with improved biostability for skin tissue engineering. Cell Tissue Res. 2012, 347, 783–794. [Google Scholar] [CrossRef] [PubMed]
- Deng, H.; Wang, F.; Zhou, Y.; Lei, H.; Zhou, H.; Chen, S.; Meng, Z.; He, M.; Tu, D.; Wang, H. Li, X. Biosynthesis of a dual growth factors (GFs) functionalized silk sericin hydrogel to promote chronic wound healing in diabetic mice. Bioact. Mater. 2025, 52, 511–528. [Google Scholar] [CrossRef]
- Ren, C.; Chen, T.; Bao, B.; Liu, Q. Advances in Strong and Tough Gelatin Hydrogels. Polym. Bull. 2026, 39, 97–124. [Google Scholar]
- Zhao, Z.; Chen, J.; He, J.; Zhang, H.; Han, L. Study on construction and properties of bigels based on gelatin hydrogel and phytosterol/oryzanol oleogels. Cereals Oils 2025, 38, 64–71+99. [Google Scholar]
- Tordi, P.; Tamayo, A.; Jeong, Y.; Han, B.; Al Kayal, T.; Cavallo, A.; Bonini, M.; Samorì, P. Fully Bio-Based Gelatin Organohydrogels via Enzymatic Crosslinking for Sustainable Soft Strain and Temperature Sensing. Adv. Funct. Mater. 2026, 36, e20762. [Google Scholar] [CrossRef]
- Johari, N.; Adabavazeh, Z.; Baino, F. PVA-based bioinks for 3D bioprinting: A comprehensive review of their applications in tissue engineering. Bioprint 2025, 49, e00419. [Google Scholar] [CrossRef]
- Masri, S.; Maarof, M.; Abd Aziz, I.; Idrus, R.; Fauzi, M.B. Performance of hybrid gelatin-PVA bioinks integrated with genipin through extrusion-based 3D bioprinting: An in vitro evaluation using human dermal fibroblasts. Int. J. Bioprint. 2023, 9, 677. [Google Scholar] [CrossRef]
- Jergitsch, M.; Mateos-Timoneda, M.A. 3D extrusion bioprinting: Rational bioink design and advanced fabrication techniques. Trends Biotechnol. 2025, 44, 378–388. [Google Scholar] [CrossRef]
- Decante, G.; Costa, J.B.; Silva-Correia, J.; Collins, M.N.; Reis, R.L.; Oliveira, J.M. Engineering bioinks for 3D bioprinting. Biofabrication 2021, 13, 032001. [Google Scholar] [CrossRef] [PubMed]
- Shabbirahmed, A.M.; Sekar, R.; Gomez, L.A.; Sekhar, M.R.; Hiruthyaswamy, S.P.; Basavegowda, N.; Somu, P. Recent developments of silk-based scaffolds for tissue engineering and regenerative medicine applications: A special focus on the advancement of 3D printing. Biomimetics 2023, 8, 16. [Google Scholar] [CrossRef]
- Evans, S.E.; Harrington, T.; Rivero, M.C.R.; Rognin, E.; Tuladhar, T.; Daly, R. 2D and 3D inkjet printing of biopharmaceuticals–A review of trends and future perspectives in research and manufacturing. Int. J. Pharm. 2021, 599, 120443. [Google Scholar] [CrossRef] [PubMed]
- Athukorala, S.S.; Tran, T.S.; Balu, R.; Truong, V.K.; Chapman, J.; Dutta, N.K.; Roy Choudhury, N. 3D printable electrically conductive hydrogel scaffolds for biomedical applications: A review. Polymers 2021, 13, 474. [Google Scholar] [CrossRef]
- Salleh, A.; Mustafa, N.; Teow, Y.H.; Fatimah, M.N.; Khairudin, F.A.; Ahmad, I.; Fauzi, M.B. Dual-layered approach of ovine collagen-gelatin/cellulose hybrid biomatrix containing graphene oxide-silver nanoparticles for cutaneous wound healing: Fabrication, physicochemical, cytotoxicity and antibacterial characterisation. Biomedicines 2022, 10, 816. [Google Scholar] [CrossRef]
- Chen, Y.; Wu, W.; Cao, X.; Li, B. Induction of polymer-grafted cellulose nanocrystals in hydrogel nanocomposites to increase anti-swelling, mechanical properties and conductive self-recovery for underwater strain sensing. Int. J. Biol. Macromol. 2024, 274, 133410. [Google Scholar] [CrossRef]
- Garcia-Garcia, A.; Muñana-González, S.; Lanceros-Mendez, S.; Ruiz-Rubio, L.; Alvarez, L.P.; Vilas-Vilela, J.L. Biodegradable natural hydrogels for tissue engineering, controlled release, and soil remediation. Polymers 2024, 16, 2599. [Google Scholar] [CrossRef] [PubMed]
- Fan, Z.; King, M.W. Biodegradable Polymers as the Pivotal Player in the Design of Tissue Engineering Scaffolds. Adv. Healthc. Mater. 2020, 9, E1901358. [Google Scholar] [CrossRef]
- Labus, K.; Radosinski, L.; Kotowski, P. Functional properties of two-component hydrogel systems based on gelatin and polyvinyl alcohol—Experimental studies supported by computational analysis. Int. J. Mol. Sci. 2021, 22, 9909. [Google Scholar] [CrossRef]
- Ruining, B.A.O.; Weiguo, W.U.; Luyan, L.I.A.O.; Yan, W.A.N.G. Optimization of Purification Process of Konjac Glucomannan by Response Surface Methodology. Sci. Technol. Food Ind. 2024, 45, 210–216. [Google Scholar]
- Schwab, A.; Levato, R.; D’este, M.; Piluso, S.; Eglin, D.; Malda, J. Printability and shape fidelity of bioinks in 3D bioprinting. Chem. Rev. 2020, 120, 11028–11055. [Google Scholar] [CrossRef]
- Hu, G.; Cebrero, K.G.; Venkataraman, N.; Ravichandran, D.; Lin, Y.; Jin, Z.; Gartner, Z.J.; Gu, G.X. Tuning collagen and collagen-alginate mechanics through extrusion bioprinting process parameters. RSC Adv. 2025, 15, 46591–46601. [Google Scholar] [CrossRef] [PubMed]
- da Silva, V.A.; Yu, B.; Willerth, S.M. Early-stage material properties as predictors of neural bioink performance during extrusion 3D bioprinting. Biofabrication 2026, 18, 025002. [Google Scholar] [CrossRef]
- Leanpolchareanchai, J.; Nuchtavorn, N. Wearable microneedle-based colorimetric and fluorescence sensing for transdermal diagnostics. Talanta Open 2023, 8, 100247. [Google Scholar] [CrossRef]
- Geevarghese, R.; Żur-Pińska, J.; Parisi, D.; Włodarczyk-Biegun, M.K. A comprehensive protocol for hydrogel-based bioink design: Balancing printability, stability, and biocompatibility. J. Mater. Chem. B 2025, 13, 13750–13768. [Google Scholar] [CrossRef]
- Mikusheva, N.G.; Zorin, I.M.; Gubarev, A.S.; Ievlev, A.V.; Volina, O.V.; Tsvetkov, N.V. Influence of reduced molar mass of low-acyl gellan gum on weak gel formation and rheological properties. Gels 2025, 11, 398. [Google Scholar] [CrossRef] [PubMed]
- Gérardin-Charbonnier, C.; Assaker, K.; Emo, M.; Vibert, F.; Blin, J.L. Supramolecular biosourced hydrogelator: A green tool for tailoring the pore size of mesostructured silica for enzyme immobilization. New J. Chem. 2025, 49, 12931–12939. [Google Scholar] [CrossRef]
- Steier, A.; Schmieg, B.; Irtel von Brenndorff, Y.; Meier, M.; Nirschl, H.; Franzreb, M.; Lahann, J. Enzyme scaffolds with hierarchically defined properties via 3D jet writing. Macromol. Biosci. 2020, 20, 2000154. [Google Scholar] [CrossRef]
- Tanimoto, R.; Ebara, M.; Uto, K. Tunable enzymatically degradable hydrogels for controlled cargo release with dynamic mechanical properties. Soft Matter 2023, 19, 6224–6233. [Google Scholar] [CrossRef] [PubMed]
- Arango, M.C.; Jaramillo-Quiceno, N.; Badia, J.D.; Cháfer, A.; Cerisuelo, J.P.; Álvarez-López, C. The impact of green physical crosslinking methods on the development of sericin-based biohydrogels for wound healing. Biomimetics 2024, 9, 497. [Google Scholar] [CrossRef]
- Bhattacharjee, M.; Goswami, S.; Borah, P.; Saikia, M.; Barman, J.; Pramanik, N.B.; Haloi, D.J. Preparation, characterization, and antimicrobial activity of chitosan/kaolin clay biocomposite films. Macromol. Chem. Phys. 2023, 224, 2300008. [Google Scholar] [CrossRef]
- Ye, Q.M.; Jin, L.C.; Yue, C.; Ya, R.Y.; Lin, Y. 3D printing of a poly(vinyl alcohol-based nano-composite hydrogel as an artificial cartilage replacement and the improvement mechanism of printing accuracy. J. Mater. Chem. B 2019, 8, 677–690. [Google Scholar] [CrossRef]
- Zhang, B.; da Silva, M.; Johnston, I.; Aspinall, S.; Cook, M. Poly (vinyl alcohol)-Agar Double Network Hydrogels: Linking Formulation to Mechanical and Rheological Properties. Macromol. Chem. Phys. 2025, 226, e00257. [Google Scholar] [CrossRef]
- Voisin, H.; Bonnin, E.; Marquis, M.; Alvarado, C.; Lafon, S.; Lopez-Leon, T.; Jamme, F.; Capron, I. Probing the colloidal behavior of a cell wall polysaccharides-degrading enzyme in a highly constrained model system. J. Colloid Interface Sci. 2025, 694, 137685. [Google Scholar] [CrossRef] [PubMed]
- Ramanathan, G.; Hassan, M.; Rochev, Y. Optimising the viscoelastic properties of hyaluronic acid hydrogels through colloidal particle interactions: A response surface methodology approach. Colloids Surf. A Physicochem. Eng. Asp. 2024, 703, 135320. [Google Scholar] [CrossRef]
- Tatykhanova, G.S.; Hirvonen, S.P.; Bardadym, Y.V.; Gizatullina, N.N.; Saulimbay, M.A. Fractionation and Characterization of Commercial Low Acyl Gellan Gum. Macromol. Symp. 2024, 413, 2400001. [Google Scholar] [CrossRef]
- Arango, M.C.; Vásquez Vásquez, L.; Parra, A.C.H.; Rueda-Mira, S.; Jaramillo-Quiceno, N.; Cerisuelo, J.P.; Cháfer, A.; Álvarez-López, C. Evaluation of Sericin/Polyvinyl Alcohol Mixtures for Developing Porous and Stable Structures. Biomimetics 2025, 10, 27. [Google Scholar] [CrossRef] [PubMed]
- Hassan, H.M.; Souka, U.D.; Hassan, S.M. Differentiation and quantification of bovine and pork gelatin using UPLC-QTOF and ATR-FTIR spectroscopy: Addressing challenges in mixed gelatin analysis and detection. Food Chem. 2025, 464, 141883. [Google Scholar] [CrossRef] [PubMed]
- Mansur, H.S.; Sadahira, C.M.; Souza, A.N.; Mansur, A.A. FTIR spectroscopy characterization of poly (vinyl alcohol) hydrogel with different hydrolysis degree and chemically crosslinked with glutaraldehyde. Mater. Sci. Eng. C 2008, 28, 539–548. [Google Scholar] [CrossRef]
- Zou, L.; Li, Y.; Feng, S.; Wang, Z.; Xiao, H.; Chen, S.; Wang, Y.; He, L.; Mao, X. Innovations and Applications of Composite Hydrogels: From Polymer-Based Systems to Metal-Ion-Doped and Functional Nanomaterial-Enhanced Architectures. Small 2025, 21, 2503147. [Google Scholar] [CrossRef]
- Krishna, D.V.; Sankar, M.R. Comparative Study on the Properties of Glutaraldehyde and Glyoxal Cross-Linked Gelatin/Polyvinyl Alcohol/Guar Gum-Based Composite Hydrogel. In National Conference on Advanced and Emerging Materials for Technological Applications; Springer Nature Singapore: Singapore, 2024; pp. 519–530. [Google Scholar] [CrossRef]
- Torres-Ayala, L.K.; Nakamatsu, J.; Kim, S. Rheological optimization of hybrid alginate–xanthan gum hydrogels for enhanced 3D bioprinting fidelity. Polym. Bull. 2025, 82, 9947–9976. [Google Scholar] [CrossRef]















| Level | A: Gellan Gum Addition | B: Silk Sericin Addition | C: Polyvinyl Alcohol Addition |
|---|---|---|---|
| −1 | 0.06 | 0.40 | 2.1 |
| 0 | 0.10 | 0.50 | 2.3 |
| 1 | 0.14 | 0.60 | 2.5 |
| Indicator (Weight) | Evaluation Criteria | Score (Points) |
|---|---|---|
| Printing fidelity (40%) | Shape intact, smooth surface, neat edges. A 98.6% ≤ Printing fidelity ≤ 101.4% | 31~40 |
| Slight deformation, smooth and even surface. A 96.3% ≤ Printing fidelity < 98.6% or 101.4% < Printing fidelity ≤ 103% | 11~30 | |
| Incomplete shape, rough and uneven surface. Printing fidelity < 96.3% or Printing fidelity > 103% | 0~10 | |
| Mechanical Properties (20%) | High load-bearing capacity with strong resistance to deformation. Compressive stress ≥ 0.175 Mpa | 11~20 |
| Balanced support and flexibility with overall moderate performance. A 0.115 Mpa ≤ compressive stress < 0.175 Mpa | 6~10 | |
| Soft texture with low resistance to deformation. Compressive stress < 0.115 Mpa | 0~5 | |
| Bio-related Performance (40%) | Excellent swelling and degradation performance. Swelling ratio ≥ 480%, enzymatic degradation rate ≥ 72.3% | 31~40 |
| Moderate swelling and degradation performance. A 400% ≤ swelling ratio < 480% or 65.4% ≤ enzymatic degradation rate < 72.3% | 11~30 | |
| Poor swelling and degradation performance. Swelling ratio < 400%, enzymatic degradation rate < 65.4% | 0~10 |
| Run No | A(g) | B(g) | C(g) | Comprehensive Performance Evaluation of the Scaffold (Score) |
|---|---|---|---|---|
| 1 | 0.06 | 0.40 | 2.3 | 77 |
| 2 | 0.14 | 0.40 | 2.3 | 85 |
| 3 | 0.06 | 0.60 | 2.3 | 78 |
| 4 | 0.14 | 0.60 | 2.3 | 87 |
| 5 | 0.06 | 0.50 | 2.1 | 75 |
| 6 | 0.14 | 0.50 | 2.1 | 82 |
| 7 | 0.06 | 0.50 | 2.5 | 79 |
| 8 | 0.14 | 0.50 | 2.5 | 86 |
| 9 | 0.10 | 0.40 | 2.1 | 79 |
| 10 | 0.10 | 0.60 | 2.1 | 86 |
| 11 | 0.10 | 0.40 | 2.5 | 79 |
| 12 | 0.10 | 0.60 | 2.5 | 80 |
| 13 | 0.10 | 0.50 | 2.3 | 83 |
| 14 | 0.10 | 0.50 | 2.3 | 79 |
| 15 | 0.10 | 0.50 | 2.3 | 81 |
| 16 | 0.10 | 0.50 | 2.3 | 82 |
| 17 | 0.10 | 0.50 | 2.3 | 80 |
| Source of Variance | Sum of Squares (SS) | Degrees of Freedom (DF) | Mean Square (MS) | F-Value | p-Value | Statistical Significance |
|---|---|---|---|---|---|---|
| Model | 123.45 | 9 | 13.72 | 1.83 | 0.0005 | Statistical significance |
| Amount of A-type GG Added | 1.13 | 1 | 1.13 | 0.1499 | 0.0002 | |
| Amount of B-type SS Protein Added | 18 | 1 | 18 | 2.4 | 0.1654 | |
| Amount of C-type PVA Added | 0.125 | 1 | 0.125 | 0.0167 | 0.0014 | |
| AB | 12.25 | 1 | 12.25 | 1.63 | 0.3422 | |
| AC | 9 | 1 | 9 | 1.2 | 0.0001 | |
| BC | 6.25 | 1 | 6.25 | 0.8325 | 0.2919 | |
| A2 | 56.09 | 1 | 56.09 | 7.47 | 0.0002 | |
| B2 | 0.6737 | 1 | 0.6737 | 0.0897 | 0.1732 | |
| C2 | 23.25 | 1 | 23.25 | 3.1 | 0.0068 | |
| Residual | 52.55 | 7 | 7.51 | |||
| Lack of Fit Error | 39.75 | 3 | 13.25 | 4.14 | 0.1018 | Not statistically significant |
| Pure Error | 12.8 | 4 | 3.2 | |||
| Total | 176 | 16 |
| Run No | A/g | B/g | C/g | Predicted Value | Experimental Value | Deviation (%) |
|---|---|---|---|---|---|---|
| 1 | 0.14 | 0.60 | 2.3 | 87 | 87 | 0 |
| 2 | 0.14 | 0.60 | 2.3 | 87 | 86 | −1.1 |
| 3 | 0.14 | 0.60 | 2.3 | 87 | 89 | +2.3 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Tang, Z.; He, J.; Cui, L.; Kang, Y.; Ni, Z. Formulation Optimization of GG/SS/PVA/GEL Composite Hydrogels for Extrusion-Based Bioprinting Using Response Surface Methodology. Processes 2026, 14, 1179. https://doi.org/10.3390/pr14071179
Tang Z, He J, Cui L, Kang Y, Ni Z. Formulation Optimization of GG/SS/PVA/GEL Composite Hydrogels for Extrusion-Based Bioprinting Using Response Surface Methodology. Processes. 2026; 14(7):1179. https://doi.org/10.3390/pr14071179
Chicago/Turabian StyleTang, Zhenhao, Jingtao He, Lujun Cui, Yingchen Kang, and Zhongjin Ni. 2026. "Formulation Optimization of GG/SS/PVA/GEL Composite Hydrogels for Extrusion-Based Bioprinting Using Response Surface Methodology" Processes 14, no. 7: 1179. https://doi.org/10.3390/pr14071179
APA StyleTang, Z., He, J., Cui, L., Kang, Y., & Ni, Z. (2026). Formulation Optimization of GG/SS/PVA/GEL Composite Hydrogels for Extrusion-Based Bioprinting Using Response Surface Methodology. Processes, 14(7), 1179. https://doi.org/10.3390/pr14071179




