Investigation of CO2-CH4-H2O Diffusion in Gas Reservoirs: Combining Experimental Measurement and Molecular Dynamics Simulation
Abstract
1. Introduction
2. Experiment and Simulation Preparation
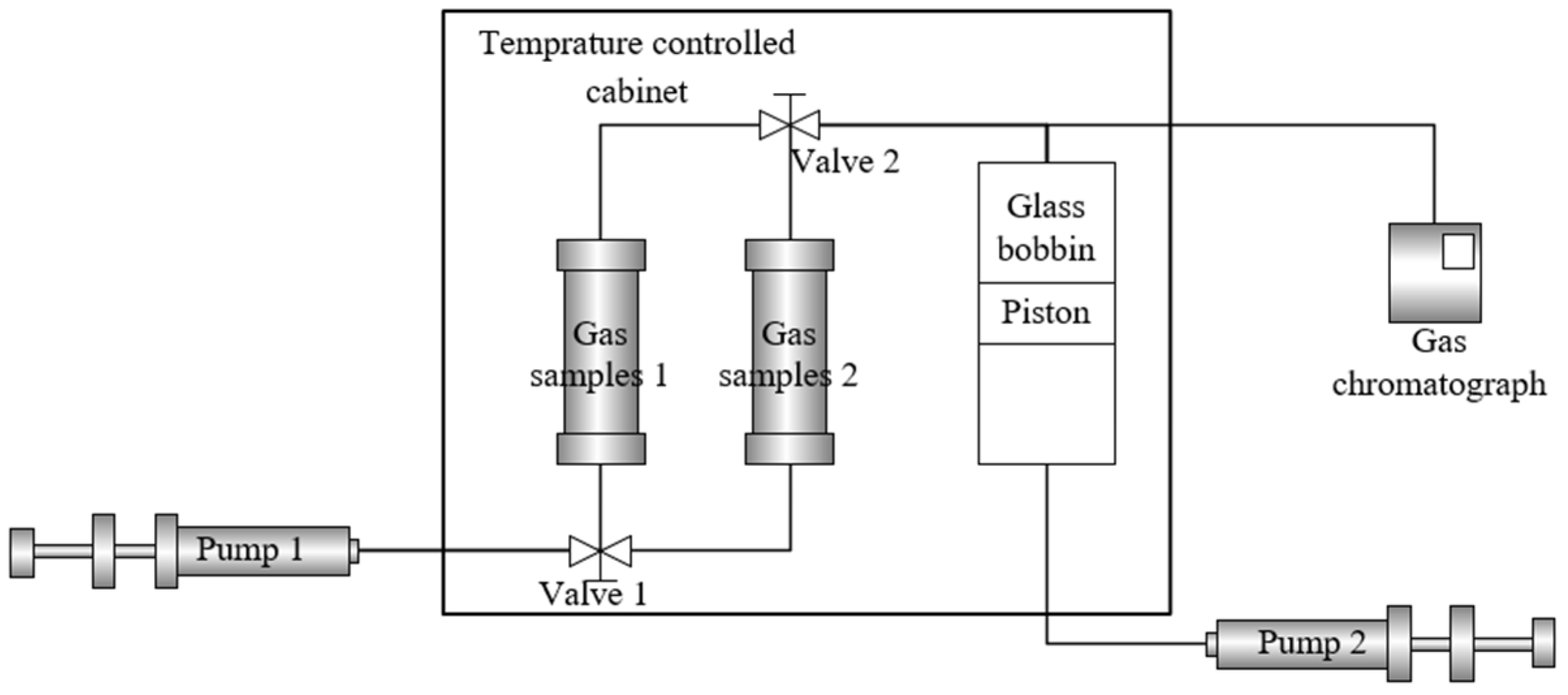
2.1. Experiment Methods
- Sample preparation: injection gas and formation gas samples were prepared. The purity of all gas samples was ensured to meet experimental requirements.
- System purging: the air inside the diffusion cylinder was removed by flushing the system via Pump 2, and the piston was pushed to its uppermost position to eliminate residual gases.
- Sample injection: the gas sample was connected to the top of the diffusion cylinder. CO2 was slowly injected using the high-pressure displacement Pump 1, followed by the injection of the prepared formation gas after system stabilization. To minimize nonlinear effects, the injection rate was controlled at 0.05 mL/min.
- Component transfer and detection: the gas sample entered the diffusion column, where axial diffusion and radial dispersion occurred simultaneously during flow. Gas samples were collected from the diffusion cylinder at 2, 4, 8, and 16 h for gas chromatographic analysis. The concentration changes of the upper gas phase were monitored, and chromatographic peaks were recorded.
- Data recording and reproducibility: the retention time, peak shape, and peak width of chromatographic signals were accurately recorded. All experiments were performed in triplicate to ensure data reliability and reproducibility.
- Diffusion coefficient calculation: the molecular diffusion coefficient D of each component was calculated based on known parameters, including carrier gas velocity, diffusion column length, and inner diameter.
2.2. Modelling
2.3. Simulation Details
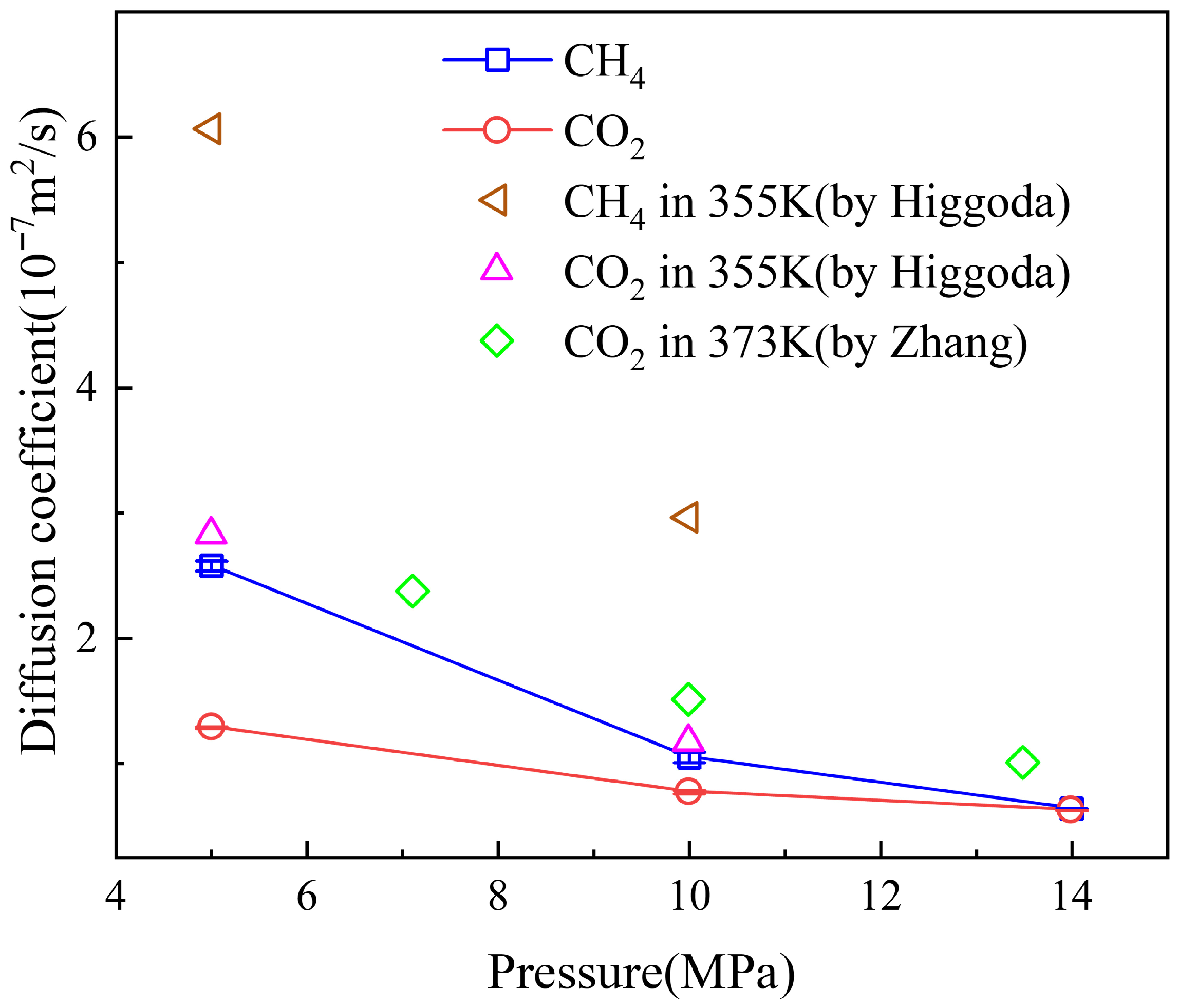
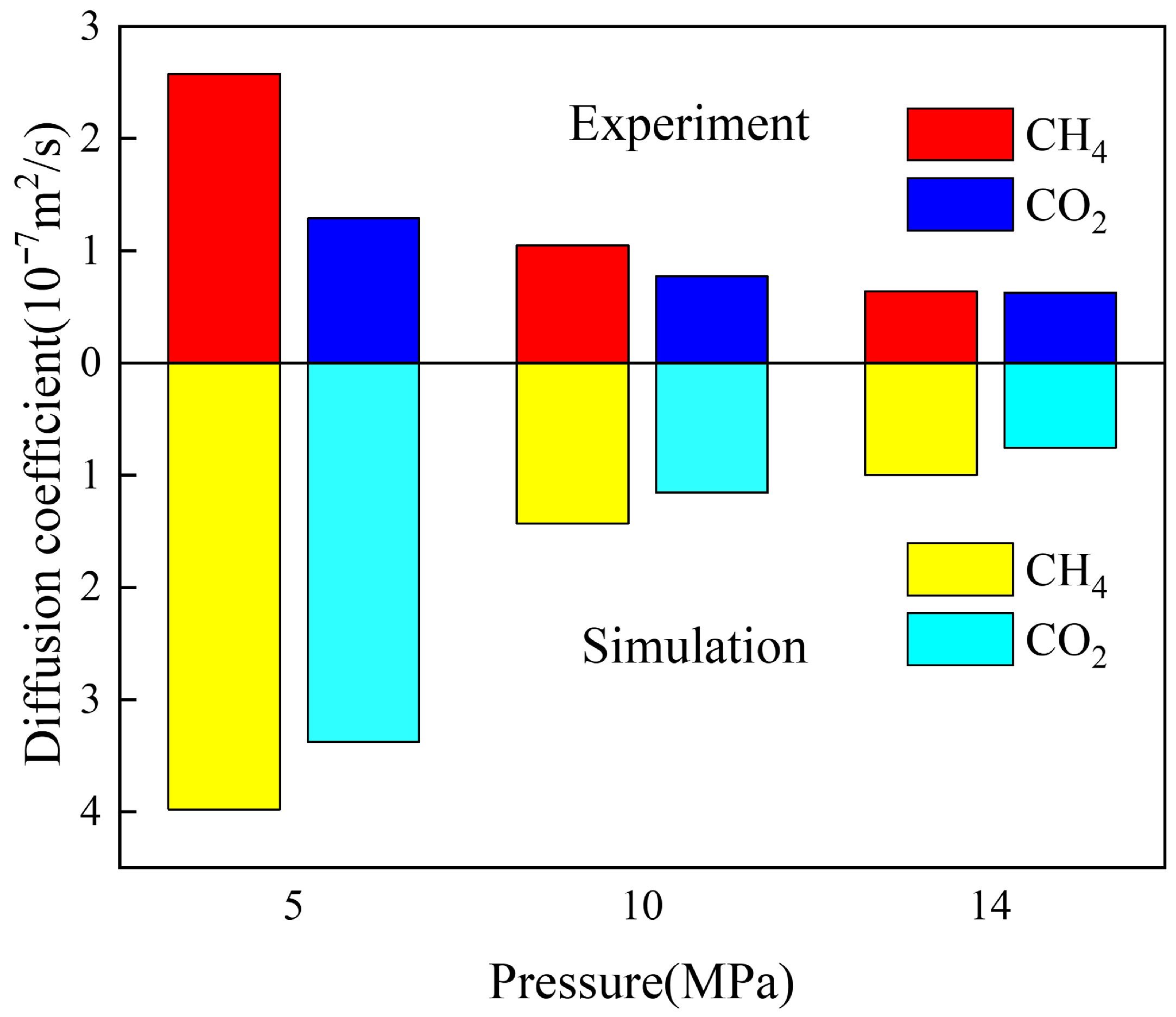
2.4. Model Verification
3. Result and Discussion
3.1. Experimental Results
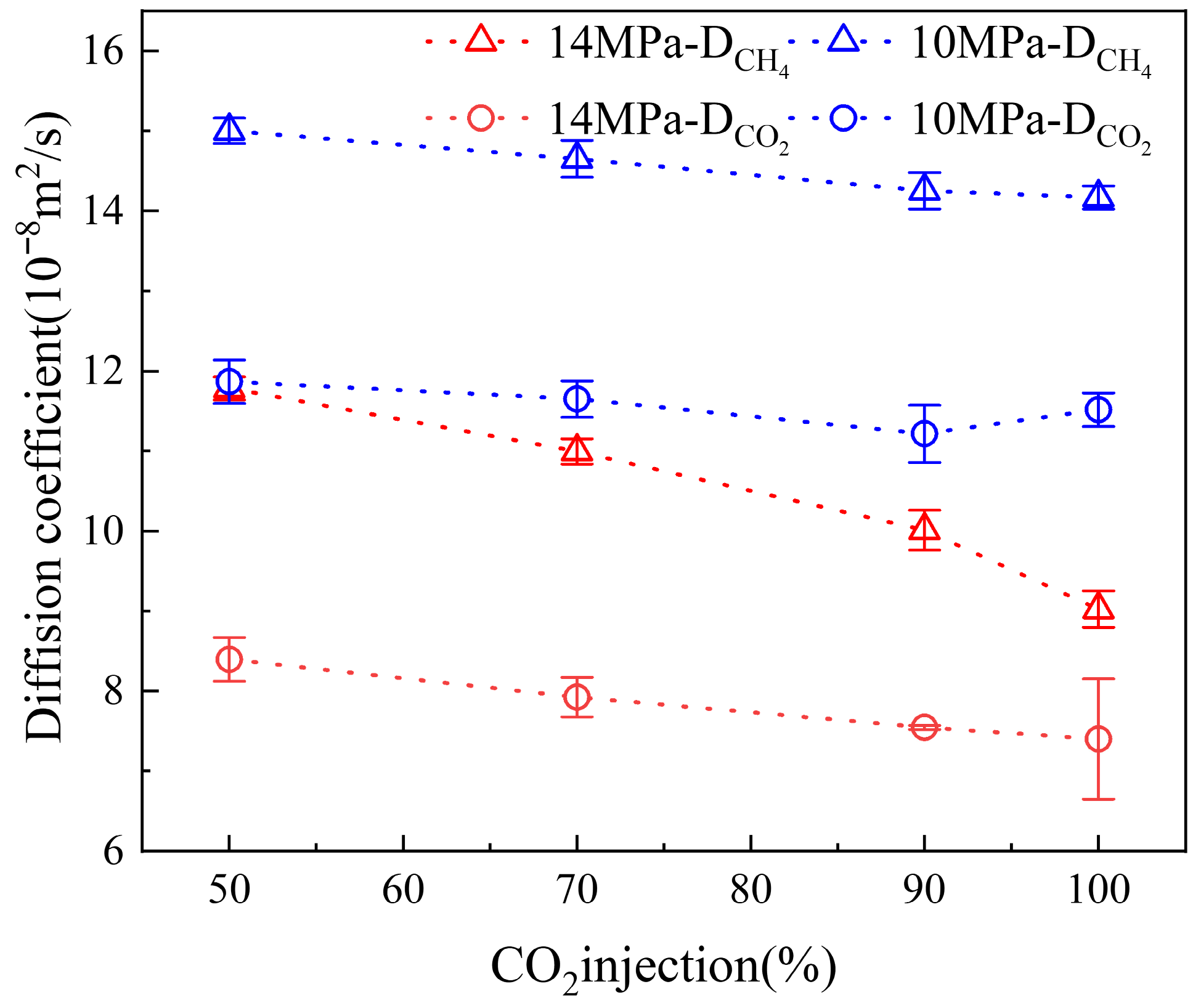
3.2. Diffusion Coefficient of Binary Fluid
3.3. Thermodynamic Factor Γ
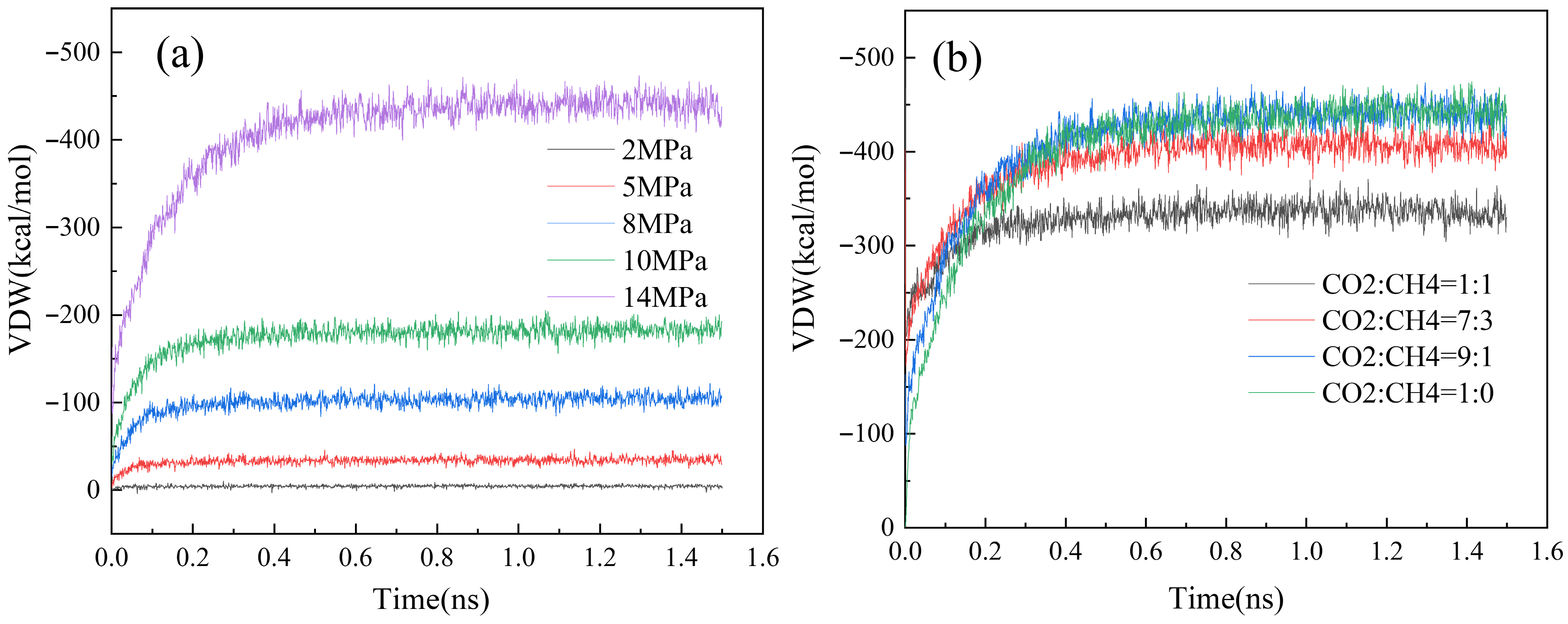
3.4. Characterization of CO2-CH4 Interaction
3.5. Ternary Fluid Diffusion Coefficient
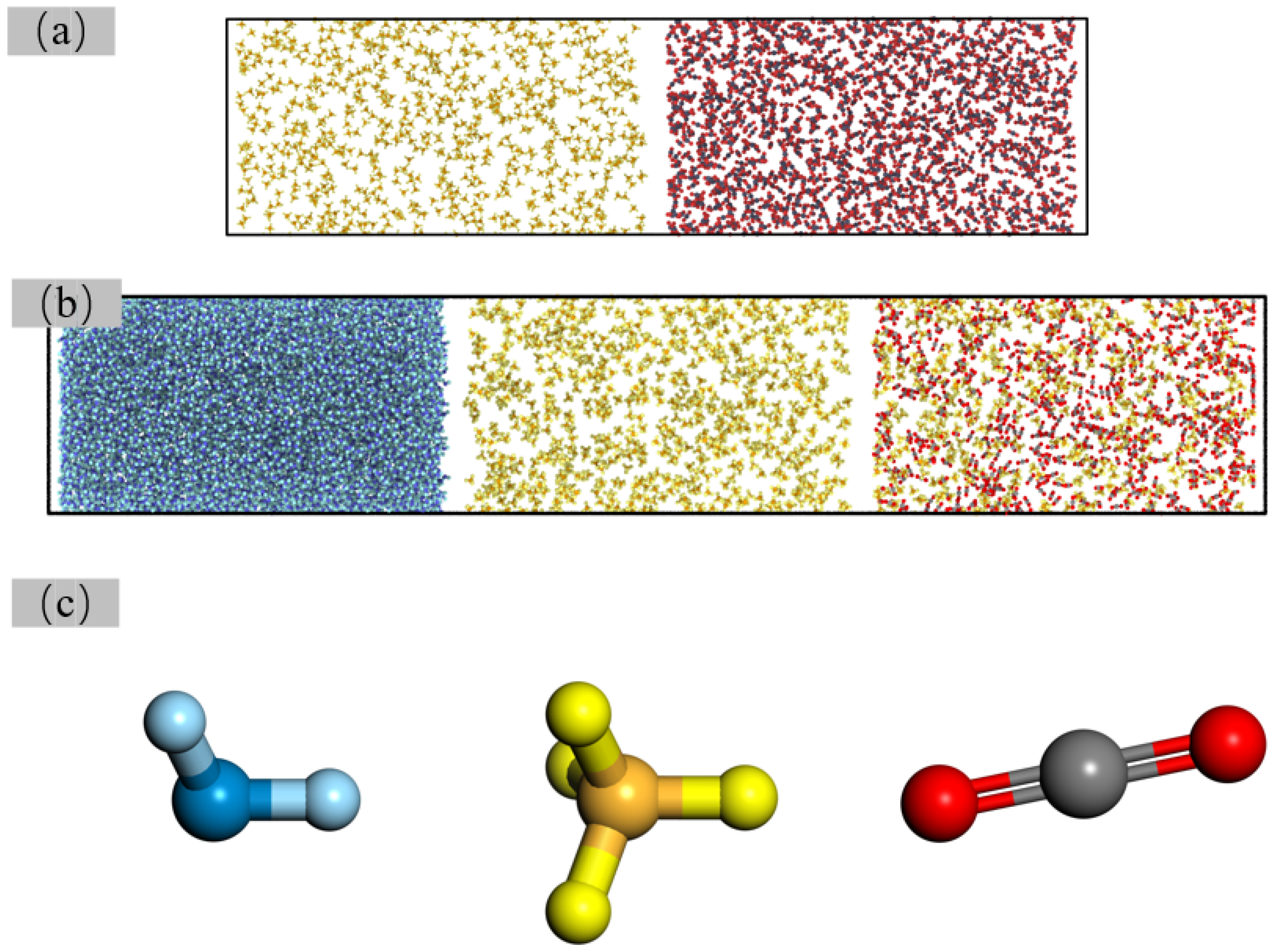
3.6. Molecular Spatial Distribution Characteristics
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| CCUS | Carbon capture, utilization and storage |
| EGR | Enhanced oil and gas recovery |
| QENS | Quasi-Elastic Neutron Scattering |
| PFG-NMR | Pulsed Field Gradient Nuclear Magnetic Resonance |
| HPGC | High-pressure gas chromatography |
| MD | Molecular Dynamics |
| GCMC | Grand canonical Monte Carlo |
References
- Liu, E.; Lu, X.; Wang, D. A Systematic Review of Carbon Capture, Utilization and Storage: Status, Progress and Challenges. Energies 2023, 16, 2865. [Google Scholar] [CrossRef]
- Vitillo, J.G.; Eisaman, M.D.; Aradóttir, E.S.P.; Passarini, F.; Wang, T.; Sheehan, S.W. The role of carbon capture, utilization, and storage for economic pathways that limit global warming to below 1.5°C. iScience 2022, 25, 104237. [Google Scholar] [CrossRef] [PubMed]
- Liang, B.; Chen, C.; Jia, C.; Wang, C.; Wang, X.; Zha, Y.; Meng, Z.; Wang, H. Carbon capture, utilization and storage (CCUS) in oil and gas reservoirs in China: Status, opportunities and challenges. Fuel 2024, 375, 132353. [Google Scholar] [CrossRef]
- Liu, Z.; Gao, M.; Zhang, X.; Liang, Y.; Guo, Y.; Liu, W.; Bao, J. CCUS and CO2 injection field application in abroad and China: Status and progress. Geoenergy Sci. Eng. 2023, 229, 212011. [Google Scholar] [CrossRef]
- Rui, Z.; Liu, T.; Wen, X.; Meng, S.; Li, Y.; Dindoruk, B. Investigating the Synergistic Impact of CCUS-EOR. Engineering 2025, 48, 16–40. [Google Scholar] [CrossRef]
- Al Ghafri, S.Z.S.; Forte, E.; Maitland, G.C.; Rodriguez-Henríquez, J.J.; Martin Trusler, J.P. Experimental and Modeling Study of the Phase Behavior of (Methane + CO2 + Water) Mixtures. J. Phys. Chem. B 2014, 118, 14461–14478. [Google Scholar] [CrossRef]
- Legoix, L.; Ruffine, L.; Donval, J.P.; Haeckel, M. Phase Equilibria of the CH4-CO2 Binary and the CH4-CO2-H2O Ternary Mixtures in the Presence of a CO2-Rich Liquid Phase. Energies 2017, 10, 2034. [Google Scholar] [CrossRef]
- Kastanidis, P.; Michalis, V.K.; Romanos, G.E.; Stubos, A.K.; Economou, I.G.; Tsimpanogiannis, I.N. Solubility of Methane and Carbon Dioxide in the Aqueous Phase of the Ternary (Methane + Carbon Dioxide + Water) Mixture: Experimental Measurements and Molecular Dynamics Simulations. J. Chem. Eng. Data 2018, 63, 1027–1035. [Google Scholar] [CrossRef]
- Liu, Y.; Larson, T.E.; Nicot, J.P. Theoretical and Experimental Study of Controls on CO2 Dissolution and CH4 Outgassing Rates. Energy Procedia 2014, 63, 4773–4781. [Google Scholar] [CrossRef][Green Version]
- Loring, J.S.; Bacon, D.H.; Springer, R.D.; Anderko, A.; Gopinath, S.; Yonkofski, C.M.; Thompson, C.J.; McGrail, B.P.; Rosso, K.M.; Schaef, H.T. Water Solubility at Saturation for CO2–CH4 Mixtures at 323.2 K and 9.000 MPa. J. Chem. Eng. Data 2017, 62, 1608–1614. [Google Scholar] [CrossRef]
- Wang, W.; Xie, Q.; An, S.; Bakhshian, S.; Kang, Q.; Wang, H.; Xu, X.; Su, Y.; Cai, J.; Yuan, B. Pore-scale simulation of multiphase flow and reactive transport processes involved in geologic carbon sequestration. Earth-Sci. Rev. 2023, 247, 104602. [Google Scholar] [CrossRef]
- Li, H.; Yang, D. Determination of Individual Diffusion Coefficients of Solvent/CO2 Mixture in Heavy Oil with Pressure-Decay Method. SPE J. 2016, 21, 131–143. [Google Scholar] [CrossRef]
- Wang, Z.; Hou, J. Measurement of CO2 diffusion coefficients in both bulk liquids and carven filling porous media of fractured-vuggy carbonate reservoirs at 50 MPa and 393 K. RSC Adv. 2021, 11, 19712–19722. [Google Scholar] [CrossRef]
- Liu, F.; Gao, X.; Du, J.; Lin, L.; Hou, D.; Luo, J.; Zhao, J. Microscopic mechanism of enhancing shale oil recovery through CO2 flooding- insights from molecular dynamics simulations. J. Mol. Liq. 2024, 410, 125593. [Google Scholar] [CrossRef]
- Kruteva, M. Dynamics studied by Quasielastic Neutron Scattering (QENS). Adsorption 2021, 27, 875–889. [Google Scholar] [CrossRef]
- Chmelik, C.; Kärger, J. In situ study on molecular diffusion phenomena in nanoporous catalytic solids. Chem. Soc. Rev. 2010, 39, 4864. [Google Scholar] [CrossRef] [PubMed]
- Schneider, D.; Mehlhorn, D.; Zeigermann, P.; Kärger, J.; Valiullin, R. Transport properties of hierarchical micro–mesoporous materials. Chem. Soc. Rev. 2016, 45, 3439–3467. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Li, S.; Wang, Y. Molecular Dynamics Simulation of Diffusion Behavior of CH4, CO2, and N2 in Mid-Rank Coal Vitrinite. Energies 2019, 12, 3744. [Google Scholar] [CrossRef]
- Long, H.; Lin, H.; Yan, M.; Bai, Y.; Tong, X.; Kong, X.; Li, S. Adsorption and diffusion characteristics of CH4, CO2, and N2 in micropores and mesopores of bituminous coal: Molecular dynamics. Fuel 2021, 292, 120268. [Google Scholar] [CrossRef]
- Gao, D.; Hong, L.; Wang, J.; Zheng, D. Molecular simulation of gas adsorption characteristics and diffusion in micropores of lignite. Fuel 2020, 269, 117443. [Google Scholar] [CrossRef]
- Hu, H.; Du, L.; Xing, Y.; Li, X. Detailed study on self- and multicomponent diffusion of CO2-CH4 gas mixture in coal by molecular simulation. Fuel 2017, 187, 220–228. [Google Scholar] [CrossRef]
- Vasileiadis, M.; Peristeras, L.D.; Papavasileiou, K.D.; Economou, I.G. Transport Properties of Shale Gas in Relation to Kerogen Porosity. J. Phys. Chem. C 2018, 122, 6166–6177. [Google Scholar] [CrossRef]
- Yuan, S.; Gang, H.Z.; Liu, Y.F.; Zhou, L.; Irfan, M.; Yang, S.Z.; Mu, B.Z. Adsorption and Diffusion Behaviors of CO2 and CH4 Mixtures in Different Types of Kerogens and Their Roles in Enhanced Energy Recovery. Sustainability 2022, 14, 14949. [Google Scholar] [CrossRef]
- Bonnaud, P.A.; Oulebsir, F.; Galliero, G.; Vermorel, R. Modeling competitive adsorption and diffusion of CH4/CO2 mixtures confined in mature type-II kerogen: Insights from molecular dynamics simulations. Fuel 2023, 352, 129020. [Google Scholar] [CrossRef]
- Mohammed, S.; Sunkara, A.K.; Walike, C.E.; Gadikota, G. The Role of Surface Hydrophobicity on the Structure and Dynamics of CO2 and CH4 Confined in Silica Nanopores. Front. Clim. 2021, 3, 713708. [Google Scholar] [CrossRef]
- Zhao, X.; Jin, H. Correlation for self-diffusion coefficients of H2, CH4, CO, O2 and CO2 in supercritical water from molecular dynamics simulation. Appl. Therm. Eng. 2020, 171, 114941. [Google Scholar] [CrossRef]
- Bai, Y.; Lin, H.F.; Li, S.G.; Yan, M.; Long, H. Molecular simulation of N2 and CO2 injection into a coal model containing adsorbed methane at different temperatures. Energy 2021, 219, 119686. [Google Scholar] [CrossRef]
- Yang, W.; Wang, L.; Yang, K.; Fu, S.; Tian, C.; Pan, R. Molecular insights on influence of CO2 on CH4 adsorption and diffusion behaviour in coal under ultrasonic excitation. Fuel 2024, 355, 129519. [Google Scholar] [CrossRef]
- Zhang, Q.; Zhu, H.; Kang, R. Influence of uniaxial strain loading on the adsorption-diffusion properties of binary components of CH4/CO2 in micropores of bituminous coal by macromolecular simulation. Powder Technol. 2023, 427, 118715. [Google Scholar] [CrossRef]
- Gamal Rezk, M.; Foroozesh, J. Uncertainty effect of CO2 molecular diffusion on oil recovery and gas storage in underground formations. Fuel 2022, 324, 124770. [Google Scholar] [CrossRef]
- Hemmat Esfe, M.; Esfandeh, S.; Hosseinizadeh, E. Nanofluid flooding in a randomized heterogeneous porous media and investigating the effect of capillary pressure and diffusion on oil recovery factor. J. Mol. Liq. 2020, 320, 113646. [Google Scholar] [CrossRef]
- Wang, S.; Zhang, M.; Zhang, Y.; Lei, Z.; Feng, Q.; Xu, S.; Zhang, J. Mechanisms of CO2 huff and puff enhanced oil recovery and storage within shale nanopores. Chem. Eng. J. 2025, 506, 160098. [Google Scholar] [CrossRef]
- Yang, M.; Huang, S.; Zhao, F.; Yang, C. A novel hybrid finite-infinite diffusion model for determining CO2 diffusion coefficient in oil-saturated porous media: Applications for enhanced oil recovery and geological carbon storage. Energy 2025, 316, 134621. [Google Scholar] [CrossRef]
- Hong, L.; Lin, J.; Gao, D.; Zheng, D.; Wang, W. Molecular simulation of the effect of water content on CO2, CH4, and N2 adsorption characteristics of coal. Sci. Rep. 2024, 14, 18190. [Google Scholar] [CrossRef]
- Sui, H.; Zhang, F.; Wang, Z.; Wang, D.; Wang, Y. Effect of Kerogen Maturity, Water Content for Carbon Dioxide, Methane, and Their Mixture Adsorption and Diffusion in Kerogen: A Computational Investigation. Langmuir 2020, 36, 9756–9769. [Google Scholar] [CrossRef]
- Zhou, J.; Jin, Z.; Luo, K.H. Effects of Moisture Contents on Shale Gas Recovery and CO2 Sequestration. Langmuir 2019, 35, 8716–8725. [Google Scholar] [CrossRef]
- Seyyedi, M.; Mahmud, H.K.B.; Verrall, M.; Giwelli, A.; Esteban, L.; Ghasemiziarani, M.; Clennell, B. Pore Structure Changes Occur During CO2 Injection into Carbonate Reservoirs. Sci. Rep. 2020, 10, 3624. [Google Scholar] [CrossRef]
- Zhu, S.; Yu, H.; Yan, G.; Li, J.; Cao, A.; Sun, C. Miscibility Process of Hydrocarbon Mixture Gas and Crude Oil: Insights from Molecular Dynamics. Ind. Eng. Chem. Res. 2021, 60, 13710–13718. [Google Scholar] [CrossRef]
- Jorgensen, W.L.; Maxwell, D.S.; Tirado-Rives, J. Development and Testing of the OPLS All-Atom Force Field on Conformational Energetics and Properties of Organic Liquids. J. Am. Chem. Soc. 1996, 118, 11225–11236. [Google Scholar] [CrossRef]
- Higgoda, U.A.; Kankanamge, C.J.; Hellmann, R.; Koller, T.M.; Fröba, A.P. Fick diffusion coefficients of binary fluid mixtures consisting of methane, carbon dioxide, and propane via molecular dynamics simulations based on simplified pair-specific ab initio-derived force fields. Fluid Phase Equilibria 2019, 502, 112257. [Google Scholar] [CrossRef]
- Orsi, M. Comparative assessment of the ELBA coarse-grained model for water. Mol. Phys. 2014, 112, 1566–1576. [Google Scholar] [CrossRef]
- Celebi, A.T.; Jamali, S.H.; Bardow, A.; Vlugt, T.J.H.; Moultos, O.A. Finite-size effects of diffusion coefficients computed from molecular dynamics: A review of what we have learned so far. Mol. Simul. 2021, 47, 831–845. [Google Scholar] [CrossRef]
- Simonnin, P.; Noetinger, B.; Nieto-Draghi, C.; Marry, V.; Rotenberg, B. Diffusion under Confinement: Hydrodynamic Finite-Size Effects in Simulation. J. Chem. Theory Comput. 2017, 13, 2881–2889. [Google Scholar] [CrossRef]
- Yeh, I.C.; Hummer, G. System-Size Dependence of Diffusion Coefficients and Viscosities from Molecular Dynamics Simulations with Periodic Boundary Conditions. J. Phys. Chem. B 2004, 108, 15873–15879. [Google Scholar] [CrossRef]
- Higgoda, U.A.; Hellmann, R.; Koller, T.M.; Fröba, A.P. Self-diffusion coefficient and viscosity of methane and carbon dioxide via molecular dynamics simulations based on new ab initio-derived force fields. Fluid Phase Equilibria 2019, 481, 15–27. [Google Scholar] [CrossRef]
- Zhang, T.; Zhang, B.; Zhang, J.; Jin, H. Numerical Study of Self-Diffusion Coefficients for H2, O2, CO, and CH4 in Transcritical CO2 with Molecular Dynamics Simulation. Ind. Eng. Chem. Res. 2025, 64, 7587–7597. [Google Scholar] [CrossRef]
- Vella, J.R. Fick Diffusion Coefficients of the Gaseous CH4–CO2 System from Molecular Dynamics Simulations Using TraPPE Force Fields at 101.325, 506.625, 1013.25, 2533.12, and 5066.25 kPa. J. Chem. Eng. Data 2019, 64, 3672–3681. [Google Scholar] [CrossRef]
- Guevara-Carrion, G.; Ancherbak, S.; Mialdun, A.; Vrabec, J.; Shevtsova, V. Diffusion of methane in supercritical carbon dioxide across the Widom line. Sci. Rep. 2019, 9, 8466. [Google Scholar] [CrossRef]
- Mohammadi, A.; Mccague, C.; Alvar, E.N.; Kim, P.; Bahrami, M. Measuring Gas Diffusivity of Partially Water-Saturated Gas Diffusion Layers. J. Electrochem. Soc. 2025, 172, 124517. [Google Scholar] [CrossRef]








| Equipment | Parameter | Specification/Value | Uncertainty |
|---|---|---|---|
| PVT cell | inner diameter | 3.15 cm | ±0.01 cm |
| Depth | 23 cm | ±0.05 cm | |
| Maximum pressure | 70 MPa | ||
| Maximum temperature | 473 K | ||
| pump | Pressure range | 0–70 MPa | ±0.02 MPa |
| Temperature-controlled | Temperature range | 273–473 K | ±0.1 K |
| Gas chromatograph | Detector | TCD | |
| Carrier gas | Helium | ||
| Gas mixing system | CO2 mole fraction | 0.9 | ±0.5% |
| Component | Content (%) | Component | Content (%) |
|---|---|---|---|
| N2 | 25.04 | C2-NC4 | 3.44 |
| CO2 | 5.05 | IC5-C6 | 0.81 |
| CH4 | 65.67 | C7+ | 0 |
| Pressure (MPa) | Natural Gas (g/cm3) | Injected Gas (g/cm3) | Stratum Water (g/cm3) |
|---|---|---|---|
| 2 | 0.0111 | 0.0296 | 0.8633 |
| 5 | 0.0283 | 0.0818 | 0.8645 |
| 8 | 0.0461 | 0.1478 | 0.8656 |
| 10 | 0.0581 | 0.2033 | 0.8664 |
| 14 | 0.0823 | 0.3480 | 0.8680 |
| Molecule | Type | Molar Mass | Electric Charge (e) | σ (nm) | ε (KJ/mol) |
|---|---|---|---|---|---|
| CH4 | H | 1.00797 | 0.060 | 0.250 | 0.012552 |
| C | 12.0107 | −0.240 | 0.350 | 0.276144 | |
| CO2 | C | 12.0107 | 0.700 | 0.280 | 0.224475 |
| O | 15.9994 | −0.350 | 0.305 | 0.656799 | |
| H2O | H | 1.00797 | 0.41 | 0 | 0 |
| O | 15.9994 | −0.82 | 0.3166 | 0.649984 |
| System Types | Self-Diffusion Coefficient (m2/s) | |
|---|---|---|
| CH4 | CO2 | |
| Methane system | 1.0267 × 10−7 | 7.5500 × 10−8 |
| Real system | 8.6333 × 10−8 | 6.4833 × 10−8 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Wang, Z.; Zhou, X.; Li, Y.; Zhao, J.; Fan, K.; Tu, H.; Zhao, Y.; Xia, L.; Wang, X. Investigation of CO2-CH4-H2O Diffusion in Gas Reservoirs: Combining Experimental Measurement and Molecular Dynamics Simulation. Processes 2026, 14, 1177. https://doi.org/10.3390/pr14071177
Wang Z, Zhou X, Li Y, Zhao J, Fan K, Tu H, Zhao Y, Xia L, Wang X. Investigation of CO2-CH4-H2O Diffusion in Gas Reservoirs: Combining Experimental Measurement and Molecular Dynamics Simulation. Processes. 2026; 14(7):1177. https://doi.org/10.3390/pr14071177
Chicago/Turabian StyleWang, Zhouhua, Xiaolong Zhou, Yun Li, Jianfei Zhao, Kunpeng Fan, Hanmin Tu, Yulong Zhao, Lianhua Xia, and Xin Wang. 2026. "Investigation of CO2-CH4-H2O Diffusion in Gas Reservoirs: Combining Experimental Measurement and Molecular Dynamics Simulation" Processes 14, no. 7: 1177. https://doi.org/10.3390/pr14071177
APA StyleWang, Z., Zhou, X., Li, Y., Zhao, J., Fan, K., Tu, H., Zhao, Y., Xia, L., & Wang, X. (2026). Investigation of CO2-CH4-H2O Diffusion in Gas Reservoirs: Combining Experimental Measurement and Molecular Dynamics Simulation. Processes, 14(7), 1177. https://doi.org/10.3390/pr14071177









