Efficiency of the Macrophyte Azolla filiculoides in Phytoremediation of Wastewater in the Central Region of Peru
Abstract
1. Introduction
2. Materials and Methods
2.1. Description of the Study Site
2.2. Design and Construction of the Treatment System
2.3. Phytoremediating Species
2.4. Wastewater Collection
2.5. Sampling
2.6. Physicochemical and Microbiological Analysis
2.7. Statistical Analysis
3. Results & Discussion
3.1. Efficiency of Physical Parameters
3.2. Efficiency of Chemical Parameters
3.3. Efficiency of Microbiological Parameters of Azolla filiculoides
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- UNESCO. The United Nations World Water Development Report 2022, Groundwater Making the Invisible Visible; UNESCO: Paris, France, 2022. [Google Scholar]
- SUNASS. Benchmarking Regulatorio 2025 de Empresas Prestadoras; SUNASS: Lima, Peru, 2025. [Google Scholar]
- Lopez, J.; Barrera, J. Fitodepuración Con Cuatro Especies de Macrófitas Flotantes Mediante El Sistema de Aireación a Escala Piloto de La Laguna “Mansión” de La Universidad Peruana Unión. 2021. Available online: https://repositorio.upeu.edu.pe/items/fb53009e-5880-4c88-b750-44899ef0200c (accessed on 17 December 2025).
- Marín-Muñiz, J.L.; Sandoval Herazo, L.C.; López-Méndez, M.C.; Sandoval-Herazo, M.; Meléndez-Armenta, R.Á.; González-Moreno, H.R.; Zamora, S. Treatment Wetlands in Mexico for Control of Wastewater Contaminants: A Review of Experiences during the Last Twenty-Two Years. Processes 2023, 11, 359. [Google Scholar] [CrossRef]
- Angelakis, A.N.; Capodaglio, A.G.; Passchier, C.W.; Valipour, M.; Krasilnikoff, J.; Tzanakakis, V.A.; Sürmelihindi, G.; Baba, A.; Kumar, R.; Haut, B.; et al. Sustainability of Water, Sanitation, and Hygiene: From Prehistoric Times to the Present Times and the Future. Water 2023, 15, 1614. [Google Scholar] [CrossRef]
- Amare, E.; Kebede, F.; Mulat, W. Wastewater Treatment by Lemna Minor and Azolla filiculoides in Tropical Semi-Arid Regions of Ethiopia. Ecol. Eng. 2018, 120, 464–473. [Google Scholar] [CrossRef]
- Mustafa, H.; Hayder, G. Recent Studies on Applications of Aquatic Weed Plants in Phytoremediation of Wastewater: A Review Article. Ain Shams Eng. J. 2021, 12, 355–365. [Google Scholar] [CrossRef]
- Khan, A.U.; Khan, A.N.; Waris, A.; Ilyas, M.; Zamel, D. Phytoremediation of Pollutants from Wastewater: A Concise Review. Open Life Sci. 2022, 17, 488–496. [Google Scholar] [CrossRef]
- González-Méndez, L.M.; Martínez-Amador, S.Y.; Ríos-González, L.J.; Pérez-Rodríguez, P.; Perez-Rodríguez, M.A.; Reyes-Acosta, A.V.; Rodríguez-De la Garza, J.A. A Review on Anatomical and Physiological Traits of Aquatic Macrophytes Coupled to a Bioelectrochemical System: Comparative Wastewater Treatment Performance. Processes 2025, 13, 1545. [Google Scholar] [CrossRef]
- Benguennouna, N.; Benabdelmoumene, D.; Dahmouni, S.; Bengharbi, Z.; Qadi, W.S.M.; Hamezah, H.S.; Mediani, A. Eco-Phytoremediation Using Azolla microphylla Enhances Heavy Metal Removal, Water Quality, and Biomass Valorization in Semi-Arid Wastewater Treatment. Desalination Water Treat. 2025, 324, 101437. [Google Scholar] [CrossRef]
- Rezooqi, A.M.; Mouhamad, R.S.; Jasim, K.A. The Potential of Azolla filiculoides for in Vitro Phytoremediation of Wastewater. J. Phys. Conf. Ser. 2021, 1853, 012014. [Google Scholar] [CrossRef]
- Al-Baldawi, I.A.; Yasin, S.R.; Jasim, S.S.; Abdullah, S.R.S.; Almansoory, A.F.; Ismail, N.’I. Removal of Copper by Azolla filiculoides and Lemna Minor: Phytoremediation Potential, Adsorption Kinetics and Isotherms. Heliyon 2022, 8, e11456. [Google Scholar] [CrossRef]
- Sundararaman, S.; Kumar, P.S.; Deivasigamani, P.; Jagadeesan, A.K.; Devaerakkam, M.; Al-Hashimi, A.; Choi, D. Assessing the Plant Phytoremediation Efficacy for Azolla filiculoides in the Treatment of Textile Effluent and Redemption of Congo Red Dye onto Azolla Biomass. Sustainability 2021, 13, 9588. [Google Scholar] [CrossRef]
- Adabembe, B.A.; Fasinmirin, J.T.; Olanrewaju, O.O.; Dada, A.A.; Faloye, O.T. Phytoremediation of Aquaculture Wastewater Using Azolla pinnata and Evaluation of Its Suitability for Irrigation Purpose. Sustain. Water Resour. Manag. 2022, 8, 166. [Google Scholar] [CrossRef]
- Jonoubi, P.; Karimi Limanjoubi, S.L. Impacts of Heavy Metal and Nutrient Accumulation on the Vegetative, Anatomical and Reproductive Characteristics and Phytoremediation Efficiency of Azolla filiculoides L. Biol. Environ. Proc. R. Ir. Acad. 2024, 124, 75–90. [Google Scholar] [CrossRef]
- Ayu, N.; Hanafiah, M. Phytoremediation of Livestock Wastewater Using Azolla filiculoides and Lemna Minor. Environ. Ecosyst. Sci. 2018, 2, 13–16. [Google Scholar] [CrossRef]
- Herazo, L.C.S.; Marín-Muñiz, J.L.; Alvarado-Lassman, A.; Zurita, F.; Marín-Peña, O.; Sandoval-Herazo, M. Full-Scale Constructed Wetlands Planted with Ornamental Species and PET as a Substitute for Filter Media for Municipal Wastewater Treatment: An Experience in a Mexican Rural Community. Water 2023, 15, 2280. [Google Scholar] [CrossRef]
- SENAMHI. Climas Del Perú, Mapa de Clasificación Climática Nacional; Servicio Nacional de Meteorología e Hidrología del Perú: Lima, Peru, 2021. [Google Scholar]
- Akinbile, C.; Ogunrinde, T.; Man, H.C.b.; Aziz, H. Phytoremediation of Domestic Wastewaters in Free Water Surface Constructed Wetlands Using Azolla pinnata. Int. J. Phytoremediat. 2015, 18, 54–61. [Google Scholar] [CrossRef]
- Kumar, V.; Kumar, P.; Singh, J.; Kumar, P. Potential of Water Fern (Azolla pinnata R.Br.) in Phytoremediation of Integrated Industrial Effluent of SIIDCUL, Haridwar, India: Removal of Physicochemical and Heavy Metal Pollutants. Int. J. Phytoremediat. 2019, 22, 392–403. [Google Scholar] [CrossRef] [PubMed]
- Garcia, Z. Comparación y Evaluación de Tres Plantas Acuáticas Para Determinar La Eficiencia de Remoción de Nutrientes En El Tratamiento de Aguas Residuales Domésticas. 2012. Available online: https://repositorio.uni.edu.pe/handle/20.500.14076/1292 (accessed on 31 December 2025).
- Ballón, K. Fitorremediación de Aguas Residuales Domésticas Con La Especie Eichhornia Crassipes En El Distrito de Yarabamba, Arequipa—2021. Bachelor’s Thesis, Escuela Académico Profesional de Ingeniería Ambiental, Universidad Continental, Arequipa, Peru, 2021. [Google Scholar]
- ANA. Protocolo Nacional Para El Monitoreo de La Calidad de Los Recursos Hídricos Superficiales. 2016. Available online: https://sinia.minam.gob.pe/documentos/protocolo-nacional-monitoreo-calidad-recursos-hidricos-superficiales (accessed on 17 December 2025).
- APHA; AWWA; WEF. Standard Methods for the Examination of Water and Wastewater, 24th ed.; APHA: Washington, DC, USA, 2023. [Google Scholar]
- Aprueban Estándares de Calidad Ambiental (ECA) Para Agua y Establecen Disposiciones Complementarias. DECRETO SUPREMO N° 004-2017-MINAM. 2017. Available online: https://www.minam.gob.pe/wp-content/uploads/2017/06/DS-004-2017-MINAM.pdf (accessed on 17 December 2025).
- Ayuque, J.; Poma, K. Tratamiento de Aguas Residuales Mediante El Método Hidropónico de Mhenta Piperita y Chamaemelum Nobile En El Puesto de Salud de Chacarilla, Yauli–Huancavelica. 2022. Available online: https://repositorio.unh.edu.pe/items/fa0caec1-98e2-4e68-b5a2-dd3fe639bc91 (accessed on 19 December 2025).
- Limas, C. Mejoramiento Del Humedal Artificial Del Centro de Producción Agropecuaria de “Yauris”—Piopata Con Plantas Emergentes a Escala Piloto. 2017. Available online: https://repositorio.uncp.edu.pe/handle/20.500.12894/6763 (accessed on 22 December 2025).
- Nikolić, I.; Mijić, K.; Mitrović, I. Characteristics of Food Industry Wastewaters and Their Potential Application in Biotechnological Production. Processes 2025, 13, 2401. [Google Scholar] [CrossRef]
- Palomino Seguil, Y.; Vilchez Garay, L.; Matencios Cortez, C.; Cornejo Tueros, J.; Camargo Hinostroza, S.; Canales Guerra, V. Systematic Review of the Efficiency of Aquatic Plants in the Wastewater Treatment. IOP Conf. Ser. Earth Environ. Sci. 2022, 1009, 012004. [Google Scholar] [CrossRef]
- Sun, Q.; Zhang, X.; Zhang, X. Impact of Natural Microorganisms on the Removal of COD and the Cells Activity of the Chlorella sp. in Wastewater. Water 2023, 15, 3544. [Google Scholar] [CrossRef]
- Getu, T.; Angassa, K.; Etana, R.; Abewaa, M. Enhanced Organic and Microbial Contaminant Removal Using Planted Constructed Wetland-Microbial Fuel Cells for University Wastewater Treatment. Clean. Waste Syst. 2026, 13, 100449. [Google Scholar] [CrossRef]
- Hernández-Vásquez, L.A.; Romo-Gómez, C.; Alvarado-Lassman, A.; Prieto-García, F.; Camacho-López, C.; Acevedo-Sandoval, O.A. Artificial Floating Islands for the Removal of Nutrients and Improvement of the Quality of Urban Wastewater. Water 2024, 16, 1443. [Google Scholar] [CrossRef]





| Day | ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 | 19 | 20 | 21 | 22 | 23 | 24 | 25 | 26 | 27 | 28 | |
| Light brown | ||||||||||||||||||||||||||||
| Yellowish-green | ||||||||||||||||||||||||||||
| Transparent greenish | ||||||||||||||||||||||||||||
| First Take | Second Take | Third Take | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Parameter | Unit of Measure | 12.00 p.m. | 9.00 a.m. | 12.00 p.m. | 16.00 p.m. | Average | 9.00 a.m. | 12.00 p.m. | 16.00 p.m. | Average |
| pH | pH unit | 7.9 | 8.61 | 8.75 | 8.8 | 8.72 | 8.7 | 8.89 | 8.86 | 8.82 |
| Temperature | °C | 19.1 | 13.5 | 19.1 | 19.5 | 17.37 | 15.7 | 21.8 | 20 | 19.17 |
| Total dissolved solids | ppm | 970 | 735 | 824 | 728 | 762.33 | 636 | 538 | 564 | 579.33 |
| Electrical conductivity | µS/cm | 1921 | 1461 | 1761 | 1452 | 1558 | 1172 | 1066 | 1158 | 1132 |
| Turbidity | NTU | 144 | 5.27 | 6.15 | 6.07 | 5.83 | 2.12 | 2.02 | 2.29 | 2.14 |
| Phosphates | mg P/L | 3.48 | 1.82 | 1.91 | 1.86 | 1.86 | 0.26 | 0.17 | 0.13 | 0.19 |
| Oils and greases | mg/L | 54.20 | <5 | <5 | <5 | <5 | <5 | <5 | <5 | <5 |
| BOD5 | mg/L | 999 | 59.1 | 63.6 | 68.1 | 63.6 | 42.9 | 45.8 | 44.6 | 44.43 |
| COD | mg/L | 2149.10 | 146.5 | 156.5 | 169.8 | 157.6 | 103.2 | 109.8 | 106.5 | 106.5 |
| Ammoniacal nitrogen | mg/L | 45.39 | 23.54 | 30.83 | 31.34 | 28.57 | 2.71 | 1.36 | 1.69 | 1.92 |
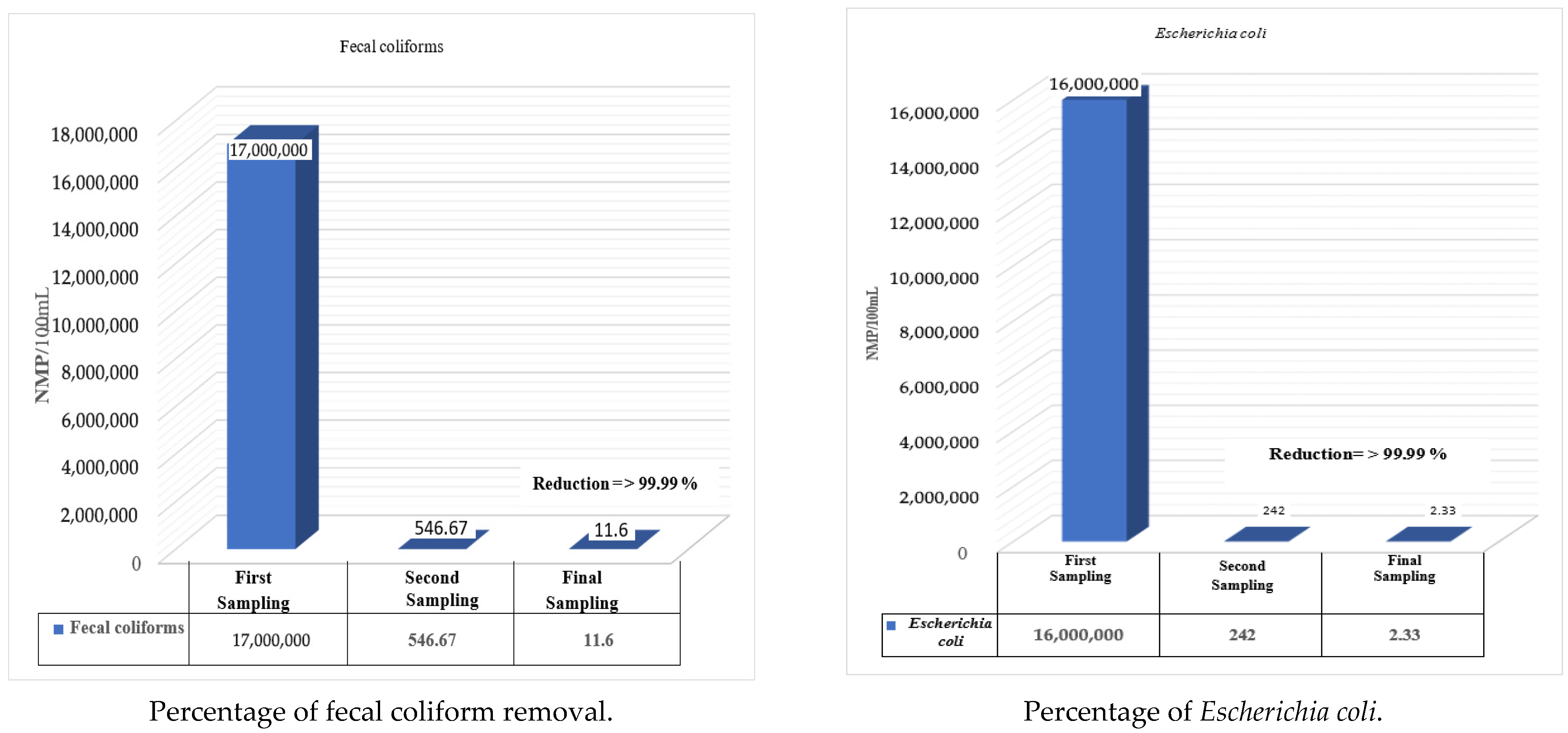
| Fecal coliforms | MPN/100 mL | 17,000,000 | 590 | 510 | 540 | 546.67 | 14 | 13 | 7.8 | 11.60 |
| Escherichia coli | MPN/100 mL | 16,000,000 | 253 | 233 | 240 | 242 | 3 | 2 | 2 | 2.33 |
| Parameter | Unit of Measurement | First Sampling Before | Second Sampling After | Third Sampling Final |
|---|---|---|---|---|
| Fecal coliforms | MPN/100 mL | 17,000,000 | 546.67 | 11.6 |
| Escherichia coli | MPN/100 mL | 16,000,000 | 242 | 2.33 |
| First Sampling | Second Sampling | Third Sampling | |||
|---|---|---|---|---|---|
| Parameter | Average | Average | Removal Efficiency (%) | Average | Removal Efficiency (%) |
| pH | 7.9 | 8.72 | - | 8.82 | - |
| Temperature | 19.1 | 17.37 | - | 19.17 | - |
| Total dissolved solids | 970 | 762.33 | 21.41 | 579.33 | 40.27 |
| electrical conductivity | 1921 | 1558 | 18.90 | 1132 | 41.07 |
| Turbidity | 144 | 5.83 | 95.95 | 2.14 | 98.51 |
| Phosphates | 3.478 | 1.86 | 46.46 | 0.19 | 94.65 |
| Oils and greases | 54.2 | <5 | >90.7 | <5 | >90.7 |
| BOD5 | 999 | 63.6 | 93.63 | 44.43 | 95.55 |
| COD | 2149.1 | 157.6 | 92.67 | 106.5 | 95.04 |
| Ammoniacal Nitrogen | 45.385 | 28.57 | 37.05 | 1.92 | 95.77 |
| Fecal coliforms | 17,000,000 | 546.67 | 99 | 11.60 | 99.99 |
| Escherichia coli | 16,000,000 | 242 | 99 | 2.33 | 99.99 |
| Statistical | Microbiological Parameter | |
|---|---|---|
| Total Coliforms | Escherichia coli | |
| Pre-treatment | 17,000,000 | 16,000,000 |
| Post-treatment | ||
| Minimum | 7.8 | 1 |
| Maximum | 14.0 | 3 |
| Mean | 11.6 | 2 |
| Standard Deviation | 3.33 | 1 |
| Skewness | −1.558 | 0 |
| Normality test | ||
| Shapiro–Wilk | 0.867 | 1 |
| p-value | 0.288 | 1 |
| is this normal? | Yes | Yes |
| Efficiency | ||
| EQSs | 1000 | 1000 |
| Student’s t-test | −514.31 | −1728.59 |
| p-value | 0 | 0 |
| Efficacy | Yes | Yes |
| Parameter | Pre-Treatment | Post-Treatment (28 d) | % Removal | ECA Cat 3 | ECA Cat 3 | Condition |
|---|---|---|---|---|---|---|
| Vegetable Irrigation | Animal Watering | |||||
| Physical | ||||||
| pH | 7.9 | 8.82 | −11.6% (Inc) | 6.5–8.5 | 6.5–8.4 | Non-compliant |
| Temperature | 19.1 | 19.17 | −0.37% | Δ 3 | Δ 3 | Compliant |
| Electric Cond. | 1921 | 1132 | 41.07% | 2500 | 5000 | Compliant |
| Turbidity | 144 | 2.14 | 98.51% | 100 | 100 | Compliant |
| Chemical | ||||||
| Phosphates | 3.478 | 0.19 | 94.54% | 0.15 | 0.15 | Non-compliant |
| Oils and Grease | 54.2 | <5 | >90.77% | 5 | 10 | Compliant |
| Total Diss. Solids | 970 | 579.33 | 40.28% | 1000 | 1000 | Compliant |
| BOD5 | 999 | 44.43 | 95.55% | 15 | 15 | Non-compliant |
| COD | 2149.1 | 106.5 | 95.04% | 40 | 40 | Non-compliant |
| Ammoniacal Nit. | 45.385 | 1.92 | 95.77% | 10 | 10 | Compliant |
| Microbiological | ||||||
| Fecal Coliforms | 1.7 × 107 | 11.6 | 99.99% | 1000 | 1000 | Compliant |
| Escherichia coli | 1.6 × 107 | 2.33 | 99.99% | 1000 | Not applicable | Compliant |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Brañes-Landeo, V.A.; Zárate-Quiñones, R.H.; Bonilla-Mancilla, H.D.; Rafaele-De La Cruz, M. Efficiency of the Macrophyte Azolla filiculoides in Phytoremediation of Wastewater in the Central Region of Peru. Processes 2026, 14, 1133. https://doi.org/10.3390/pr14071133
Brañes-Landeo VA, Zárate-Quiñones RH, Bonilla-Mancilla HD, Rafaele-De La Cruz M. Efficiency of the Macrophyte Azolla filiculoides in Phytoremediation of Wastewater in the Central Region of Peru. Processes. 2026; 14(7):1133. https://doi.org/10.3390/pr14071133
Chicago/Turabian StyleBrañes-Landeo, Victor Adriel, Rosa Haydee Zárate-Quiñones, Humberto Dax Bonilla-Mancilla, and Mauro Rafaele-De La Cruz. 2026. "Efficiency of the Macrophyte Azolla filiculoides in Phytoremediation of Wastewater in the Central Region of Peru" Processes 14, no. 7: 1133. https://doi.org/10.3390/pr14071133
APA StyleBrañes-Landeo, V. A., Zárate-Quiñones, R. H., Bonilla-Mancilla, H. D., & Rafaele-De La Cruz, M. (2026). Efficiency of the Macrophyte Azolla filiculoides in Phytoremediation of Wastewater in the Central Region of Peru. Processes, 14(7), 1133. https://doi.org/10.3390/pr14071133








