LC-MS/MS-Analysis and Biological Evaluation of Hop (Humulus lupulus): Antioxidant, Antidiabetic, Anticholinergic and Antiglaucoma Activities
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Preparation of Hop (H. lupulus) Extracts
2.3. Determination of Phytochemistry of Hop (H. lupulus)
2.3.1. Total Phenolic Content Determination
2.3.2. Total Flavonoid Content Determination
2.3.3. Phenolics and Flavonoids Determination by LC-MS/MS
2.4. Determination of Antioxidant Activity of Hop (H. lupulus) Extracts
2.4.1. Radical Scavenging Assays of Hop (H. lupulus) Extracts
2.4.2. Reducing Power Assays of Hop (H. lupulus) Extracts
2.5. Determination of Enzyme Inhibition Properties of Hop (H. lupulus) Extracts
2.5.1. α-Glycosidase Inhibition of Hop (H. lupulus) Extracts
2.5.2. Acetylcholinesterase Inhibition of Hop (H. lupulus) Extracts
2.5.3. Butyrylcholinesterase Inhibition of Hop (H. lupulus) Extracts
2.5.4. Human Carbonic Anhydrase I and II Inhibition of Hop (H. lupulus) Extracts
2.6. Antimicrobial Activity of Hop (H. lupulus) Extracts
2.7. IC50 Value Determination Hop (H. lupulus) Extracts
2.8. Statistics Analysis
3. Results
3.1. Antioxidant Properties of Hop (Humulus lupulus) Extracts
3.2. Estimation of Enzyme Inhibition Property of Hop (Humulus lupulus) Extracts
3.3. Phytochemical Analysis of Hop (H. lupulus) Extract
3.4. Antimicrobial Activity of Hop (H. lupulus) Extracts
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Basilio-Cortes, U.A.; Ramírez-Rodrigues, M.M.; Ramírez-Rodrigues, M.A.; González-Mendoza, D.; Tzintzun-Camacho, O.; Durán-Hernández, D.; González-Salitre, L. Phytochemical, spectroscopic analysis, and antifungal activity on bell peppers of hydrothermal bioactive metabolites of Humulus lupulus L. extracts. Nat. Prod. Res. 2024, 38, 4301–4310. [Google Scholar] [CrossRef]
- Suphiratwanich, P.; Yarangsee, C.; Lomarat, P.; Laosirisathian, N. Phytochemical screening, in vitro antioxidant, and antimicrobial efficacy of Humulus lupulus L. flowers (Newport and Comet varieties) from Thailand. Pharm. Sci. Asia 2024, 51, 391–400. [Google Scholar] [CrossRef]
- Choi, J.Y.; Desta, K.T.; Lee, S.J.; Kim, Y.; Shin, S.C.; Kim, G.; Lee, S.J.; Shim, J.; Hacımüftüoğlu, A.; El-Aty, A.M.A. LC–MS/MS profiling of polyphenol-enriched leaf, stem, and root extracts of Korean Humulus japonicus Siebold & Zucc. and determination of their antioxidant effects. Biomed. Chromatogr. 2018, 32, e4171. [Google Scholar] [CrossRef]
- Keskin, Ş.Y.; Çakir, H.E.; Keskin, M. An investigation of Humulus lupulus L.: Phenolic composition, antioxidant capacity, and inhibition properties of clinically important enzymes. S. Afr. J. Bot. 2019, 120, 170–174. [Google Scholar] [CrossRef]
- Lyu, J.I.; Ryu, J.; Seo, K.-S.; Kang, K.-Y.; Park, S.H.; Ha, T.H.; Ahn, J.-W.; Kang, S.-Y. Comparative study on phenolic compounds and antioxidant activities of hop (Humulus lupulus L.) strobile extracts. Plants 2022, 11, 135. [Google Scholar] [CrossRef]
- Zhang, J.; Zhang, Y.; Wang, J.; Xia, Y.; Zhang, J.; Chen, L. Recent advances in Alzheimer’s disease: Mechanisms, clinical trials and new drug development strategies. Sig. Transduct. Target Ther. 2024, 9, 211. [Google Scholar] [CrossRef]
- Rawat, K.; Tewari, D.; Bisht, A.; Chandra, S.; Tiruneh, Y.K.; Hassan, H.M.; Al-Emam, A.; Sindi, E.R.; Al-Dies, A.-A.M. Identification of AChE targeted therapeutic compounds for Alzheimer’s disease: An in-silico study with DFT integration. Sci. Rep. 2024, 14, 30356. [Google Scholar] [CrossRef]
- Biçer, A.; Taslimi, P.; Yakali, G.; Gülçin, İ.; Gültekin, M.S.; Turgut Cin, G. Synthesis, characterization, crystal structure of novel bis-thiomethylcyclohexanone derivatives and their inhibitory properties against some metabolic enzymes. Bioorg. Chem. 2019, 82, 393–404. [Google Scholar] [CrossRef]
- Chen, Z.R.; Huang, J.B.; Yang, S.L.; Hong, F.F. Role of cholinergic signaling in Alzheimer’s disease. Molecules 2022, 27, 1816. [Google Scholar] [CrossRef] [PubMed]
- Breijyeh, Z.; Karaman, R. Comprehensive review on Alzheimer’s disease: Causes and treatment. Molecules 2020, 25, 5789. [Google Scholar] [CrossRef]
- Nordberg, A.; Svensson, A.L. Cholinesterase inhibitors in the treatment of Alzheimer’s disease: A comparison of tolerability and pharmacology. Drug Saf. 1998, 19, 465–480. [Google Scholar] [CrossRef]
- Karagecili, H.; Yılmaz, M.A.; Ertürk, A.; Kızıltaş, H.; Güven, L.; Alwasel, S.H.; Gulcin, İ. Comprehensive metabolite profiling of Berdav propolis using LC-MS/MS: Determination of antioxidant, anticholinergic, antiglaucoma, and antidiabetic effects. Molecules 2023, 28, 1739. [Google Scholar] [CrossRef]
- Zhang, M.; Feng, R.; Yang, M.; Qian, C.; Wang, Z.; Liu, W.; Ma, J. Effects of metformin, acarbose, and sitagliptin monotherapy on gut microbiota in Zucker diabetic fatty rats. BMJ Open Diabetes Res. Care 2019, 7, e000717. [Google Scholar] [CrossRef]
- Paiva, L.; Binsack, R.; Machado, U.F. Chronic acarbose-feeding increases GLUT1 protein without changing intestinal glucose absorption function. Eur. J. Pharmacol. 2002, 434, 197–204. [Google Scholar] [CrossRef]
- Yuan, Y.; Hou, X.; Cao, X.; Ding, M.; Su, L. Acarbose-induced pneumatosis cystoides intestinalis. JCEM Case Rep. 2025, 3, luaf199. [Google Scholar] [CrossRef]
- Han, X.; Wang, P.; Zhang, J.; Lv, Y.; Zhao, Z.; Zhang, F.; Shang, M.; Liu, G.; Wang, X.; Cai, S.; et al. α-Glucosidase inhibition mechanism and anti-hyperglycemic effects of flavonoids from Astragali radix and their mixture effects. Pharmaceuticals 2025, 18, 744. [Google Scholar] [CrossRef] [PubMed]
- Supuran, C.T. Multi- and polypharmacology of carbonic anhydrase inhibitors. Pharmacol. Rev. 2025, 77, 100004. [Google Scholar] [CrossRef] [PubMed]
- Mallia, A.; Brocca, L.; Papaianni, G.G.; Banfi, C. Carbonic anhydrases inhibition in the management of cardiovascular and cardiometabolic disorders. Biomed. Pharmacother. 2025, 190, 118396. [Google Scholar] [CrossRef] [PubMed]
- Lemon, N.; Canepa, E.; Ilies, M.A.; Fossati, S. Carbonic anhydrases as potential targets against neurovascular unit dysfunction in Alzheimer’s disease and stroke. Front. Aging Neurosci. 2021, 13, 772278. [Google Scholar] [CrossRef]
- D’Ambrosio, K.; Di Fiore, A.; Alterio, V.; Langella, E.; Monti, S.M.; Supuran, C.T.; De Simone, G. Multiple binding modes of inhibitors to human carbonic anhydrases: An update on the design of isoform-specific modulators of activity. Chem. Rev. 2025, 125, 150–222. [Google Scholar] [CrossRef]
- Supuran, C.T. Carbonic anhydrase inhibitors from marine natural products. Mar. Drugs 2022, 20, 721. [Google Scholar] [CrossRef]
- Beydemir, Ş.; Gülcin, İ. Effects of melatonin on carbonic anhydrase from human erythrocytes in vitro and from rat erythrocytes in vivo. J. Enzyme Inhib. Med. Chem. 2004, 19, 193–197. [Google Scholar] [CrossRef]
- Harborne, J.B. Phytochemical Methods: A Guide to Modern Techniques of Plant Analysis, 3rd ed.; Springer: Dordrecht, The Netherlands, 1998. [Google Scholar]
- Ozbey, F.; Taslimi, P.; Gulcin, İ.; Maras, A.; Goksu, S.; Supuran, C.T. Synthesis of diaryl ethers with acetylcholinesterase, butyrylcholinesterase and carbonic anhydrase inhibitory actions. J. Enzym. Inhib. Med. Chem. 2016, 31, 79–85. [Google Scholar] [CrossRef]
- Torres, P.; Osaki, S.; Silveira, E.; dos Santos, D.Y.A.C.; Chow, F. Comprehensive evaluation of Folin-Ciocalteu assay for total phenolic quantification in algae (chlorophyta, phaeophyceae, and rhodophyta). Algal Res. 2024, 80, 103503. [Google Scholar] [CrossRef]
- Zhishen, J.; Mengcheng, T.; Jianming, W. The determination of flavonoid contents in mulberry and their scavenging effects on superoxide radicals. Food Chem. 1999, 64, 555–559. [Google Scholar] [CrossRef]
- Sultana, S.; Lawag, I.L.; Lim, L.Y.; Foster, K.J.; Locher, C. A critical exploration of the total flavonoid content assay for honey. Methods Protoc. 2024, 7, 95. [Google Scholar] [CrossRef] [PubMed]
- Akyuz, M. The determination of antidiabetic, anticholinesterase and antioxidant properties of ethanol and water extracts of blackberry (Rubus fruticosus L.) fruits at different maturity stages. S. Afr. J. Bot. 2022, 151, 1035–1048. [Google Scholar] [CrossRef]
- Güven, L.; Erturk, A.; Miloğlu, F.D.; Alwasel, S.; Gulcin, I. Screening of antiglaucoma, antidiabetic, anti-Alzheimer, and antioxidant activities of Astragalus alopecurus Pall-Analysis of phenolics profiles by LC-MS/MS. Pharmaceuticals 2023, 16, 659. [Google Scholar] [CrossRef] [PubMed]
- Yılmaz, M.A. Simultaneous quantitative screening of 53 phytochemicals in 33 species of medicinal and aromatic plants: A detailed, robust and comprehensive LC–MS/MS method validation. Ind. Crop. Prod. 2020, 149, 112347. [Google Scholar] [CrossRef]
- Yuldasheva, N.; Acikyildiz, N.; Akyuz, M.; Yabo-Dambagi, L.; Aydin, T.; Cakir, A.; Kazaz, C. The synthesis of Schiff bases and new secondary amine derivatives of p-vanillin and evaluation of their neuroprotective, antidiabetic, antidepressant and antioxidant potentials. J. Mol. Struc. 2023, 1270, 133883. [Google Scholar] [CrossRef]
- Altay, A.; Degirmenci, S.; Korkmaz, M.; Cankaya, M.; Koksal, E. In vitro evaluation of antioxidant and anti-proliferative activities of Gypsophila sphaerocephala (Caryophyllaceae) extracts together with their phenolic profiles. J. Food Meas. Charact. 2018, 12, 2936–2945. [Google Scholar] [CrossRef]
- Blois, M.S. Antioxidant determinations by the use of a stable free radical. Nature 1958, 181, 1199–1200. [Google Scholar] [CrossRef]
- Re, R.; Pellegrini, N.; Proteggente, A.; Pannala, A.; Yang, M.; Rice-Evans, C. Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radic. Biol. Med. 1999, 26, 1231–1237. [Google Scholar] [CrossRef] [PubMed]
- Apak, R.; Güçlü, K.; Özyürek, M.; Karademir, S.E. Novel total antioxidant capacity index for dietary polyphenols and vitamins C and E, using their cupric ion reducing capability in the presence of neocuproine: CUPRAC method. J. Agric. Food Chem. 2004, 52, 7970–7981. [Google Scholar] [CrossRef]
- Oyaizu, M. Studies on product of browning reaction prepared from glucose amine. Jpn. J. Nutr. 1986, 44, 307. [Google Scholar] [CrossRef]
- Benzie, I.F.; Strain, J.J. The ferric reducing ability of plasma (FRAP) as a measure of “antioxidant power”: The FRAP assay. Anal. Biochem. 1996, 239, 70–76. [Google Scholar] [CrossRef]
- Adera, K.T.; Inami, Y.M.; Akamatsu, K.T.; Atsuoka, T.M. Inhibition of α-glucosidase and α-amylase by flavonoids. J. Nutr. Sci. Vitaminol. 2006, 52, 149–153. [Google Scholar] [CrossRef]
- Ellman, G.L.; Courtney, K.D.; Andres, V.; Featherstone, R.M. A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem. Pharmacol. 1961, 7, 88–95. [Google Scholar] [CrossRef]
- Wilbur, K.M.; Anderson, N.G. Electrometric and colorimetric determination of carbonic anhydrase. J. Biol. Chem. 1948, 176, 147–154. [Google Scholar] [CrossRef]
- Topal, F. Inhibition profiles of voriconazole against acetylcholinesterase, α-glycosidase, and human carbonic anhydrase I and II isoenzymes. J. Biocem. Mol. Toxicol. 1948, 33, e22385. [Google Scholar] [CrossRef]
- Gülçin, İ.; Tel, A.Z.; Kirecci, E. Antioxidant, antimicrobial, antifungal and antiradical activities of Cyclotrichium niveum (Boiss.) Manden and Scheng. Int. J. Food Proper. 2008, 11, 450–471. [Google Scholar] [CrossRef]
- Lolak, N.; Akocak, S.; Topal, M.; Koçyigit, U.M.; Isik, M.; Türkes, C.; Topal, F.; Durgun, M.; Beydemir, S. Sulfonamide-bearing pyrazolone derivatives as multitarget therapeutic agents: Design, synthesis, characterization, biological evaluation, in silico ADME/T profiling and molecular docking study. Pharmacol. Res. Perspect. 2025, 13, e70088. [Google Scholar] [CrossRef]
- Kaya, E.; Aydin, T.; Saglamtas, R. Evaluation of antioxidant activities and inhibition effects of Tribulus terrestris L. extracts on some metabolic enzymes. S. Afr. J. Bot. 2024, 170, 156–162. [Google Scholar] [CrossRef]
- Sagan, B.; Czerny, B.; Stasiłowicz-Krzemień, A.; Szulc, P.; Skomra, U.; Karpiński, T.M.; Lisiecka, J.; Kamiński, A.; Kryszak, A.; Zimak-Krótkopad, O.; et al. Anticholinesterase activity and bioactive compound profiling of six hop (Humulus lupulus L.) varieties. Foods 2024, 13, 4155. [Google Scholar] [CrossRef]
- Indu, M.; Hatha, A.; Abirosh, C.; Harsha, U.; Vivekanandan, G. Antimicrobial activity of some of the South-Indian spices against serotypes of Escherichia coli, Salmonella, Listeria monocytogenes and Aeromonas hydrophila. Braz. J. Microbiol. 2006, 37, 153–158. [Google Scholar] [CrossRef]
- Khaliullina, A.; Kolesnikova, A.; Khairullina, L.; Morgatskaya, O.; Shakirova, D.; Patov, S.; Nekrasova, P.; Bogachev, M.; Kurkin, V.; Trizna, E.; et al. The antimicrobial potential of the hop (Humulus lupulus L.) extract against Staphylococcus aureus and oral streptococci. Pharmaceuticals 2024, 17, 162. [Google Scholar] [CrossRef]
- Onder, F.C.; Ay, M.; Sarker, S.D. Comparative study of antioxidant properties and total phenolic content of the extracts of Humulus lupulus L. and quantification of bioactive components by LC-MS/MS and GC-MS. J. Agric. Food Chem. 2013, 61, 10498–10506. [Google Scholar] [CrossRef]
- Tel, A.Z.; Aslan, K.; Yılmaz, M.A.; Gulcin, İ. A multidimensional study for design of phytochemical profiling, antioxidant potential, and enzyme inhibition effects of ışgın (Rheum telianum) as an edible plant. Food Chem. X 2025, 25, 102125. [Google Scholar] [CrossRef]
- Wu, C.K.; Fuh, J.L. A 2025 update on treatment strategies for the Alzheimer’s disease spectrum. J. Chin. Med. Assoc. 2025, 88, 495–502. [Google Scholar] [CrossRef] [PubMed]
- Walczak-Nowicka, L.J.; Herbet, M. Acetylcholinesterase inhibitors in the treatment of neurodegenerative diseases and the role of acetylcholinesterase in their pathogenesis. Int. J. Mol. Sci. 2021, 22, 9290. [Google Scholar] [CrossRef] [PubMed]
- Gajendra, K.; Pratap, G.K.; Poornima, D.V.; Shantaram, M.; Ranjita, G. Natural acetylcholinesterase inhibitors: A multi-targeted therapeutic potential in Alzheimer’s disease. Eur. J. Med. Chem. Rep. 2024, 11, 100154. [Google Scholar] [CrossRef]
- Supuran, C.T. Carbonic anhydrase versatility: From pH regulation to CO2 sensing and metabolism. Front. Mol. Biosci. 2023, 10, 1326633. [Google Scholar] [CrossRef]
- Aydin, T.; Saglamtas, R.; Dogan, B.; Kostekci, E.; Durmus, R.; Cakir, A. A new specific method for isolation of tomentosin with a high yield from Inula viscosa (L.) and determination of its bioactivities. Phytochem. Anal. 2022, 33, 612–618. [Google Scholar] [CrossRef]
- Gülçin, İ.; Mutlu, M.; Bingöl, Z.; Ozden, E.M.; Mirzaee, Z.; Gören, A.C.; Koksal, E. Antioxidant activity and phytochemical profiling of flaxseed (Linum usitatissimum) oil: Therapeutic targeting against glaucoma, oxidative stress, cholinergic imbalance, and diabetes. Molecules 2025, 30, 3384. [Google Scholar] [CrossRef]
- Yagi, S.; Cetiz, M.V.; Zengin, G.; Llorent-Martínez, E.J.; Yildiztugay, E.; Bingol, Z.; Gulcin, İ.; Dall’Acqua, S. A thorough exploration of the phytochemical profile and biological potential of Epilobium parviflorum extracts using HPLC-ESI-Q-TOF-MS technique along with in vitro and in silico analysis. J. Agric. Food Res. 2025, 24, 102414. [Google Scholar] [CrossRef]
- Dursun, İ.; Sağlamtas, R.; Fettahoğlu, K.; Zor, M.; Sinan, A.; Demirci, A.; Demir, Y.; Gulçin, İ. Antioxidant and antimicrobial activities of different extracts of Tragopogon dubius and Tragopogon porrifolium L. subsp. longirostris: Determination of their phytochemical contents by UHPLC-Orbitrap®-HRMS analysis. Food Biosci. 2025, 63, 105604. [Google Scholar] [CrossRef]
- Aslan, K.; Kelle, K.; Yılmaz, M.A.; Erden Koparır, E.; Gülçin, İ. Investigation of Cuckoo-Pint’s (Arum maculatum) phytochemistry, in vitro antioxidant potential, enzyme inhibition, and antimicrobial activity. ChemistrySelect 2024, 9, e202403588. [Google Scholar] [CrossRef]
- Masi, M.; Réfregiers, M.; Pos, K.M.; Pagès, J.-M. Mechanisms of envelope permeability and antibiotic influx and efflux in Gram-negative bacteria. Nat. Microbiol. 2017, 2, 17001. [Google Scholar] [CrossRef]

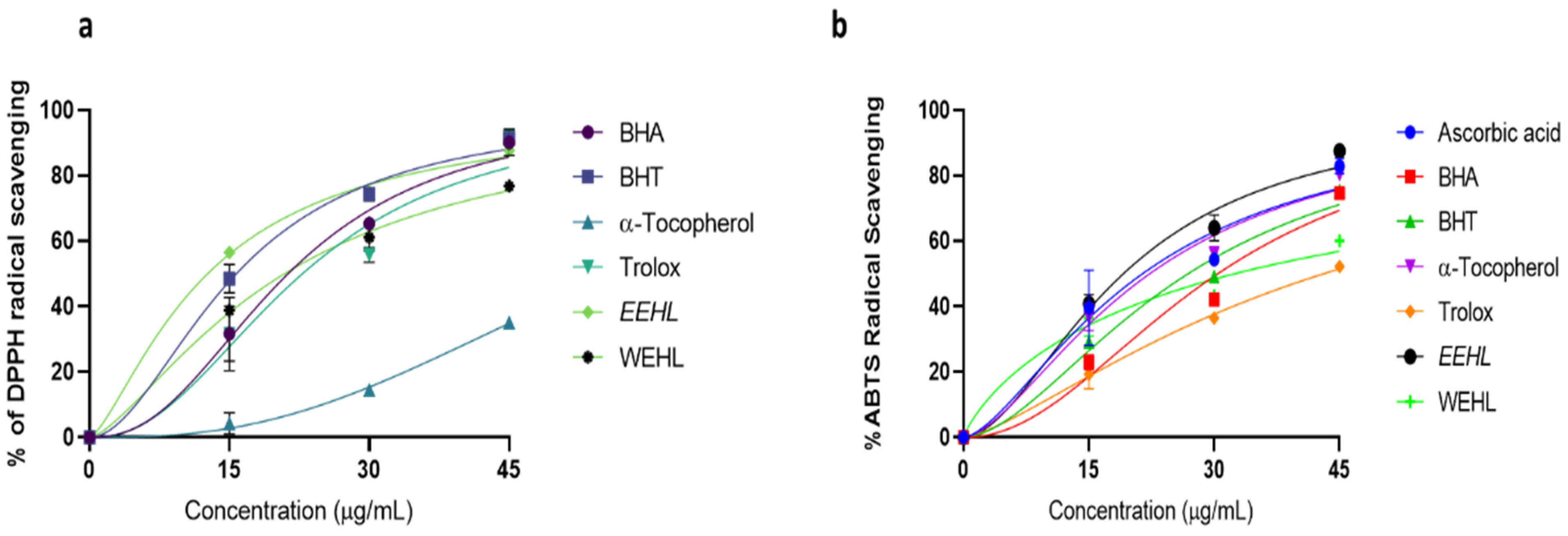


| Antioxidants | ABTS·+ Scavenging | DPPH∙ Scavenging | ||
|---|---|---|---|---|
| IC50 | r2 | IC50 | r2 | |
| Ascorbic acid | 21.42 ± 8.20 | 0.9390 | - | - |
| BHA | 30.43 ± 5.53 | 0.9660 | 21.29 ± 5.46 | 0.9789 |
| BHT | 26.87 ± 4.18 | 0.9809 | 15.74 ± 3.31 | 0.9923 |
| Trolox | 43.08 ± 2.26 | 0.9888 | 22.83 ± 9.16 | 0.9392 |
| α-Tocopherol | 22.28 ± 4.70 | 0.9788 | 56.94 ± 1.87 | 0.9843 |
| EEHL | 19.13 ± 4.66 | 0.9827 | 12.66 ± 1.94 | 0.9972 |
| WEHL | 20.90 ± 2.39 | 0.9943 | 32.41 ± 4.29 | 0.9685 |
| Antioxidants | FRAP Reducing | CUPRAC Reducing | Fe3+ Reducing | |||
|---|---|---|---|---|---|---|
| λ593 | r2 | λ450 | r2 | λ700 | r2 | |
| Ascorbic acid | - | - | - | - | - | - |
| BHA | 1.74 ± 0.04 | 0.9984 | 1.58 ± 0.02 | 0.9894 | - | - |
| BHT | 1.50 ± 0.07 | 0.9972 | 2.15 ± 0.07 | 0.9557 | 2.08 ± 0.06 | 0.9950 |
| Trolox | 0.84 ± 0.01 | 0.9990 | 1.04 ± 0.10 | 0.9829 | 0.47 ± 0.01 | 0.9829 |
| α-Tocopherol | - | - | 1.47 ± 0.04 | 0.9922 | 1.45 ± 0.01 | 0.9562 |
| EEHL | 0.77 ± 0.01 | 0.9894 | 2.09 ± 0.05 | 0.9485 | 1.95 ± 0.01 | 0.9897 |
| WEHL | 0.15 ± 0.01 | 0.9970 | 1.48 ± 0.03 | 0.9877 | 1.92 ± 0.05 | 0.9791 |
| Samples | α-Glycosidase | AChE | BChE | hCA I | hCA II | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| IC50 | r2 | IC50 | r2 | IC50 | r2 | IC50 | r2 | IC50 | r2 | |
| EEHL | 119.31 ± 0.01 | 0.9353 | 26.06 ± 0.01 | 0.9492 | 44.00 ± 0.01 | 0.9769 | 59.78 ± 0.01 | 0.9745 | 21.19 ± 0.01 | 0.9745 |
| WEHL | 121.20 ± 0.01 | 0.9927 | 30.75 ± 0.01 | 0.9510 | 69.91 ± 0.01 | 0.9775 | 155.80 ± 0.01 | 0.9993 | 77.22 ± 0.01 | 0.9070 |
| Standards * | 25.43 ± 0.01 | 0.9656 | 12.22 ± 0.01 | 0.9996 | 8.82 ± 0.01 | 0.9836 | 55.10 ± 0.01 | 0.9963 | 49.80 ± 0.01 | 0.9957 |
| Compounds | Concentration (ng/mL) | ||
|---|---|---|---|
| EEHL | WEHL | ||
| 1 | Quinic acid | 0.381 | 0.784 |
| 6 | Protocatechuic acid | 0.187 | <LOD |
| 10 | Protocatechuic aldehyde | 0.195 | <LOD |
| 17 | Caffeic acid | 0.014 | <LOD |
| 19 | Vanillin | <LOD | 0.157 |
| 24 | p-Coumaric acid | 0.619 | 0.177 |
| 29 | Salicylic acid | 0.022 | 0.053 |
| 33 | Rutin | 0.595 | <LOD |
| 34 | Isoquercitrin | 3.141 | <LOD |
| 35 | Hesperidin | 0.426 | <LOD |
| 42 | Astragalin | 0.526 | <LOD |
| 43 | Nicotiflorin | 0.129 | <LOD |
| 47 | Quercetin | 0.428 | <LOD |
| 48 | Naringenin | 0.019 | <LOD |
| 49 | Hesperetin | 0.030 | <LOD |
| 50 | Luteolin | 0.003 | <LOD |
| 52 | Kaempferol | 0.020 | <LOD |
| 54 | Amentoflavone | <LOD | 0.004 |
| 55 | Chrysin | <LOD | <LOD |
| 56 | Acacetin | <LOD | 0.004 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Aslan, K.; Gul, U.D.; Arık, M.; Yilmaz, M.A.; Cakir, O.; Gulcin, İ. LC-MS/MS-Analysis and Biological Evaluation of Hop (Humulus lupulus): Antioxidant, Antidiabetic, Anticholinergic and Antiglaucoma Activities. Processes 2026, 14, 1073. https://doi.org/10.3390/pr14071073
Aslan K, Gul UD, Arık M, Yilmaz MA, Cakir O, Gulcin İ. LC-MS/MS-Analysis and Biological Evaluation of Hop (Humulus lupulus): Antioxidant, Antidiabetic, Anticholinergic and Antiglaucoma Activities. Processes. 2026; 14(7):1073. https://doi.org/10.3390/pr14071073
Chicago/Turabian StyleAslan, Kubra, Ulkuye Dudu Gul, Mustafa Arık, Mustafa Abdullah Yilmaz, Oğuz Cakir, and İlhami Gulcin. 2026. "LC-MS/MS-Analysis and Biological Evaluation of Hop (Humulus lupulus): Antioxidant, Antidiabetic, Anticholinergic and Antiglaucoma Activities" Processes 14, no. 7: 1073. https://doi.org/10.3390/pr14071073
APA StyleAslan, K., Gul, U. D., Arık, M., Yilmaz, M. A., Cakir, O., & Gulcin, İ. (2026). LC-MS/MS-Analysis and Biological Evaluation of Hop (Humulus lupulus): Antioxidant, Antidiabetic, Anticholinergic and Antiglaucoma Activities. Processes, 14(7), 1073. https://doi.org/10.3390/pr14071073








