The Interconnection Between 3D and 4D Printing and Rheology: From Extrusion and Nozzle Deposition to Final Product Functionality
Abstract
1. Introduction
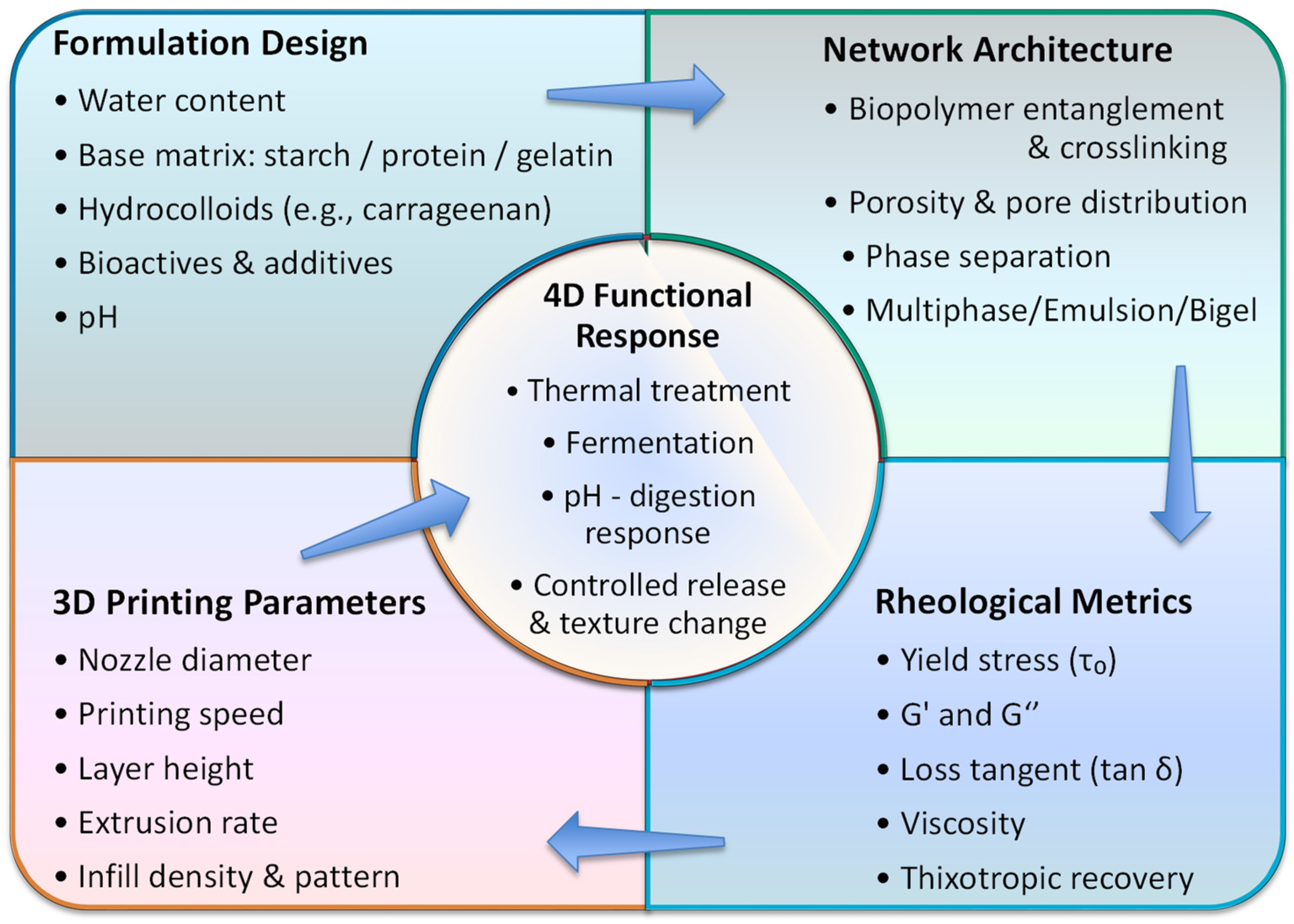
2. Network Architecture in Edible Ink Formulations
2.1. Gel Network Formation and Molecular Architecture
2.2. Composition-Dependent Network Properties
2.3. Microstructural Characterization and Spatial Organization
3. Critical Rheological Properties Governing Printability
3.1. Yield Stress and Extrusion Stability
3.2. Viscoelasticity and the Storage–Loss Moduli Balance
3.3. Loss Tangent and Network Elasticity Dominance
3.4. Apparent Viscosity and Filament Formation
3.5. Thixotropy, Recovery Kinetics, and Structural Integrity
4. Process Characteristics and Parameter Optimization
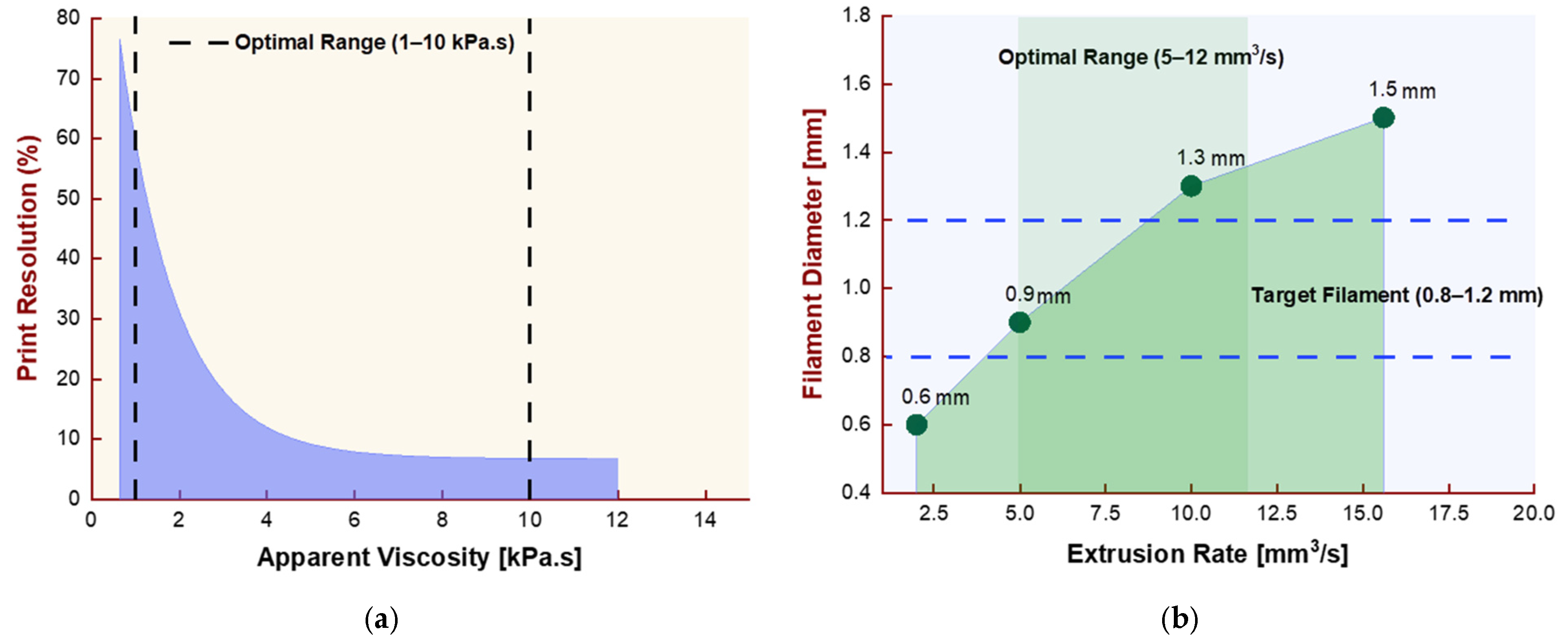
4.1. Nozzle Diameter and Printing Resolution Trade-Off
4.2. Printing Speed, Layer Height, and Dimensional Accuracy
4.3. Temperature Control and Thermal Stability
4.4. Extrusion Parameters and Filament Uniformity
4.5. Multi-Parameter Optimization and Interaction Effects
4.6. Printing Accuracy and Fidelity Metrics
5. 4D: Post-Printing Treatment and Temporal Activation of Printed Structures
5.1. Thermal Post-Processing as Activation: Structure Consolidation and Bioactive Preservation
5.2. Fermentation-Driven Activation: Time-Programmed Softening, Porosity Effects, and Nutritional Evolution
5.3. Digestive Environment Activation: pH, Ionic Strength, Hydration, and Staged Release
5.4. Multi-Stimulus Systems and Application Targets: From Delivery to Medical Nutrition
5.5. Emerging Multiphase Architectures for Functional 3D Food Printing
5.5.1. Coaxial Food Printing
5.5.2. Gels Utilizing Nanoparticles and Nanorods
5.5.3. Protein-Based Pickering Emulsion 3D Printing
6. Outlook: Multi-Parameter Interactions in 3D/4D Food Printing
- I.
- High G′ (i.e., G′ > 4000 Pa) generally increases stability and stacking accuracy, but excessive stiffness impairs extrudability unless balanced by strong shear-thinning.
- II.
- Yield stress at 100–300 Pa, which performs best for most formulations. Above 500 Pa, extrusion is difficult; below 100 Pa, spreading occurs.
- III.
- Thixotropic recovery exceeding 80% within 30 s to enable both smooth extrusion and shape fidelity.
- IV.
- Network architecture—particularly porosity and crosslink density—requires engineering not only for printability but also to control diffusion, swelling, and degradation rates during 4D activation.
- Nozzle diameter and printing speed are interdependent: smaller nozzles require slower speeds and lower viscosities; larger nozzles handle higher viscosities and faster deposition rates. The typical utilized diameter is 1.2 mm.
- Printing speeds above 400 mm/min with low-viscosity inks risk filament thinning and collapse unless elasticity is maintained. Although a wide range is observed, the usual range is between 900 (15 mm/s) and 1500 (25 mm/s).
- Layer heights below 0.3 mm can improve fidelity but require strong elastic recovery, typically from higher G′ (typically between 0.8 and 1 mm).
Critical Research Gaps and Future Directions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Qin, Z.; Yang, Y.; Zhang, Z.; Li, F.; Hou, Z.; Li, Z.; Shi, J.; Shen, T. A critical review: Gel-based edible inks for 3D food printing—Materials, rheology-geometry mapping, and control. Gels 2025, 11, 780. [Google Scholar] [CrossRef] [PubMed]
- Herrada-Manchón, H.; Fernandez, M.A.; Aguilar, E. Essential guide to hydrogel rheology in extrusion 3D printing: How to measure it and why it matters. Gels 2023, 9, 517. [Google Scholar] [CrossRef]
- Fragal, E.H.; Poirier, A.; Bleses, D.; Silva, Y.F.G.; Baccile, N.; Rharbi, Y. Microbial biosurfactant hydrogels with tunable rheology for precision 3D printing of soft scaffolds. Soft Matter 2025, 21, 4476–4487. [Google Scholar] [CrossRef]
- Fahmy, A.R.; Derossi, A.; Jekle, M. Four-Dimensional (4D) printing of dynamic foods—Definitions, considerations, and current scientific status. Foods 2023, 12, 3410. [Google Scholar] [CrossRef]
- Heckl, M.P.; Korber, M.; Jekle, M.; Becker, T. Relation between deformation and relaxation of hydrocolloids-starch based bio-inks and 3D printing accuracy. Food Hydrocoll. 2023, 137, 108326. [Google Scholar] [CrossRef]
- Liu, W.; Chen, L.; McClements, D.J.; Peng, X.; Jin, Z. Recent trends of 3D printing based on starch-hydrocolloid in food, biomedicine and environment. Crit. Rev. Food Sci. Nutr. 2024, 64, 8948–8962. [Google Scholar] [CrossRef]
- Nikolaou, E.N.; Apostolidis, E.; Nikolidaki, E.; Karvela, E.; Stergiou, A.; Kourtis, T.; Karathanos, V.T. The development and optimization of extrusion-based 3D food printing inks using composite starch gels enriched with various proteins and hydrocolloids. Gels 2025, 11, 574. [Google Scholar] [CrossRef]
- Taqdissillah, D.; Irsyad, M.; Whulanza, Y. Development of sago starch-alginate hydrogels for extrusion-based 3D food printing. East.-Eur. J. Enterp. Technol. 2025, 4, 31–41. [Google Scholar] [CrossRef]
- Chen, Y.; Zhang, M.; Bhandari, B. 3D printing of steak-like foods based on textured soybean protein. Foods 2021, 10, 2011. [Google Scholar] [CrossRef]
- Afnan, Z.; Khalid, U.; Ali, Z.; Khalid, F. Plant based ingredients in 3D food printing: A sustainable approach to personalized nutrition. Haya Saudi J. Life Sci. 2025, 10, 606–617. [Google Scholar] [CrossRef]
- Rodríguez-Herrera, V.V.; Umeda, T.; Kozu, H.; Sasaki, T.; Kobayashi, I. Printability of nixtamalized corn dough during screw-based three-dimensional food printing. Foods 2024, 13, 293. [Google Scholar] [CrossRef]
- Xu, M.; Dong, Q.; Huang, G.; Zhang, Y.; Lu, X.; Zhang, J.; Zhang, K.; Huang, Q. Physical and 3D printing properties of arrowroot starch gels. Foods 2022, 11, 2140. [Google Scholar] [CrossRef] [PubMed]
- Mishra, A.A.; Chandregowda, M.; Fai, J.; Bek, M.; Terry, A.E.; Nygård, K.; Arlov, D.; Kádár, R. Tunable elasto-viscoplastic properties of polymer blends for 3D printing applications. Macromol. Rapid Commun. 2025, 46, 2500249. [Google Scholar] [CrossRef]
- Ahmad, S.; Alam, H.; Thareja, P. 3D Printing of hydrogels: A synergistic approach of rheology and computational fluid dynamics (CFD) modeling. RSC Adv. 2025, 15, 39369–39390. [Google Scholar] [CrossRef] [PubMed]
- De Farias, P.M.; Matheus, J.R.V.; Maniglia, B.C.; Le-Bail, P.; Le-Bail, A.; Schmid, M.; Fai, A.E.C. Bibliometric mapping analysis of Pickering emulsion applied in 3D food printing. Int. J. Food Sci. Technol. 2024, 59, 2186–2196. [Google Scholar] [CrossRef]
- Ahmadzadeh, S.; Barekat, S.; Ubeyitogullari, A. Enhancing lutein and anthocyanins stability and bioaccessibility through simultaneous encapsulation using coaxial 3D food printing. npj Sci. Food 2025, 9, 96. [Google Scholar] [CrossRef] [PubMed]
- Dou, X.; Ren, J. The application of dairy products and their derivatives as edible inks in 3D printing technology: A review. Int. J. Food Sci. 2024, 59, 8630–8644. [Google Scholar] [CrossRef]
- Azman, N.Y.; Fuzi, S.; Abdul Manas, N.H. Development of cellulose based food-ink from cellulose powder. Food Res. 2024, 8, 16–21. [Google Scholar] [CrossRef]
- Yu, J. Personalized delivery of probiotics and prebiotics via 3D food printing. Metabolites 2025, 15, 744. [Google Scholar] [CrossRef]
- Domalska, Z.; Jakubczyk, E. Characteristics of food printing inks and their impact on selected product properties. Foods 2025, 14, 393. [Google Scholar] [CrossRef]
- Agunbiade, A.O.; Song, L.; Agunbiade, O.J.; Ofoedu, C.E.; Chacha, J.S.; Duguma, H.T.; Hossaini, S.M.; Rasaq, W.A.; Shorstkii, I.; Osuji, C.M.; et al. Potentials of 3D extrusion-based printing in resolving food processing challenges: A perspective review. J. Food Process Eng. 2022, 45, e13996. [Google Scholar] [CrossRef]
- Kadival, A.; Kour, M.; Meena, D.; Mitra, J. Extrusion-based 3D food printing: Printability assessment and improvement techniques. Food Bioprocess Technol. 2023, 16, 987–1008. [Google Scholar] [CrossRef]
- Derossi, A.; Spence, C.; Corradini, M.G.; Jekle, M.; Fahmy, A.R.; Caporizzi, R.; Devahastin, S.; Moses, J.A.; Le-Bail, A.; Zhou, W.; et al. Personalized, digitally designed 3D printed food towards the reshaping of food manufacturing and consumption. npj Sci. Food 2024, 8, 54. [Google Scholar] [CrossRef]
- Seol, J.; Kim, J.; Hong, Y.; Cha, M.; Park, S.; Jang, K.J.; Kim, S.J.; Son, H.I. Toward intelligent 3D food printing: A review on the perspective of materials, fabrication, monitoring, and control. Crit. Rev. Food Sci. Nutr. 2025, 65, 546–562. [Google Scholar] [CrossRef]
- Chen, Y.; Bi, S.; Gu, J.; Che, Q.; Liu, R.; Li, W.; Dai, T.; Wang, D.; Zhang, X.; Zhang, Y. Achieving personalized nutrition for patients with diabetic complications via 3D food printing. Int. J. Bioprint. 2024, 10, 1862. [Google Scholar] [CrossRef]
- Nikolaou, E.N.; Karvela, E.; Apostolidis, E.; Karathanos, V.T. Effects of different mechanical processing methods on physicochemical properties of potato starch-plant protein-carrageenan composite gels. Food Meas. 2025, 19, 2926–2941. [Google Scholar] [CrossRef]
- Kaliampakou, C.; Lagopati, N.; Charitidis, C.A. Direct ink writing of alginate-gelatin hydrogel: An optimization of ink property design and printing process efficacy. Appl. Sci. 2023, 13, 8261. [Google Scholar] [CrossRef]
- Li, Y.; Cheng, Z.; Zhang, J.; Xu, S.; Cai, Y.; Ding, Y.; Lyu, F. Effect of protein-polysaccharide hybrid gelator system on the material properties and 3D extrusion printability of mashed potatoes. J. Food Sci. 2024, 89, 2347–2358. [Google Scholar] [CrossRef]
- Oyinloye, T.M.; Yoon, W.B. Impact of saturated and unsaturated oils on the nonlinear viscoelasticity, microstructure, and 3D printability of fish myofibrillar-protein-based pastes and gels. Gels 2025, 11, 295. [Google Scholar] [CrossRef] [PubMed]
- Soni, R.; Bhandarkar, V.V.; Ponappa, K.; Tandon, P. 3D extrusion printability of corn starch and optimization of process parameters for optimal food layered manufacturing. Manuf. Lett. 2025, 44, 948–957. [Google Scholar] [CrossRef]
- Dushina, E.; Popov, S.; Zlobin, A.I.; Martinson, E.; Paderin, N.; Vityazev, F.; Belova, K.; Litvinets, S. Effect of homogenized callus tissue on the rheological and mechanical properties of 3D-printed food. Gels 2024, 10, 42. [Google Scholar] [CrossRef]
- Bai, C.; Liu, R.; Shen, L.; Yu, Z.; Hu, J. Effects of konjac glucomannan and curdlan on the 3D printability and physicochemical properties of germinated brown rice gel. Foods 2025, 14, 1764. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, S.M.; Fasolin, L.H.; Vicente, A.A.; Fuciños, P.; Pastrana, L.M. Printability, microstructure, and flow dynamics of phase-separated edible 3D inks. Food Hydrocoll. 2020, 109, 106120. [Google Scholar] [CrossRef]
- Liu, R.; Yu, Z.; Song, J.; Shen, L.; Yin, Y. Evaluation of 3D printing of cereal-legume starch-based gels formulated with red adzuki bean and germinated brown rice flour. Foods 2025, 14, 1791. [Google Scholar] [CrossRef] [PubMed]
- Choudhury, D.B.; Kumari, S.; Hazarika, M.K. 3D printed sweets made with peanut chenna and milk: A new frontier in food technology. J. Food Process Eng. 2024, 47, e14528. [Google Scholar] [CrossRef]
- Agarwal, P.; Poddar, S.; Varshney, N.; Sahi, A.K.; Vajanthri, K.Y.; Yadav, K.; Parmar, A.S.; Mahto, S.K. Printability assessment of psyllium husk (isabgol)-gelatin blends using rheological and mechanical properties. J. Biomater. Appl. 2021, 35, 1132–1142. [Google Scholar] [CrossRef]
- De Salvo, M.I.D.; Palla, C.A.; Cotabarren, I.M. Development of an operational map for the 3D printing of phytosterol-enriched oleogels: Rheological insights and applications in nutraceutical design. Gels 2025, 14, 200. [Google Scholar] [CrossRef]
- Nguyen, T.; Ahmadzadeh, S.; Schöberl, H.; Ubeyitogullari, A. Optimizing printability of rice protein-based formulations using extrusion-based 3D food printing. Food Sci. Nutr. 2024, 13, e4713. [Google Scholar] [CrossRef]
- Santhoshkumar, P.; Raja, V.; Priyadarshini, S.R.; Moses, J.A. Evaluating the 3D printability of pearl millet flour with banana pulp blends. J. Sci. Food Agric. 2024, 104, 7010–7023. [Google Scholar] [CrossRef]
- Bercea, M. Rheology as a tool for fine-tuning the properties of printable bioinspired gels. Molecules 2023, 28, 2766. [Google Scholar] [CrossRef]
- Liu, Z.; Bhandari, B.; Guo, C.; Zheng, W.; Cao, S.; Lu, H.; Mo, H.; Li, H. 3D printing of shiitake mushroom incorporated with gums as dysphagia diet. Foods 2021, 10, 2189. [Google Scholar] [CrossRef]
- Raja, V.; Moses, J.A.; Anandharamakrishnan, C. Effect of 3D printing conditions and post-printing fermentation on pearl millet fortified idli. J. Sci. Food Agric. 2022, 103, 2145–2154. [Google Scholar] [CrossRef]
- Lee, J.B.; Yoon, N.Y.; Bae, Y.J.; Kwon, G.Y.; Sohn, S.K.; Lee, H.R.; Kim, H.J.; Kim, M.J.; Park, H.E.; Shim, K.B. Optimizing 3D food printing of surimi via regression analysis: Physical properties and additve formulations. Foods 2025, 14, 889. [Google Scholar] [CrossRef] [PubMed]
- Theagarajan, R.; Moses, J.A.; Anandharamakrishnan, C. 3D extrusion printability of rice starch and optimization of process variables. Food Bioprocess Technol. 2020, 13, 1048–1062. [Google Scholar] [CrossRef]
- Lee, C.K.W.; Xu, Y.; Yuan, Q.; Chan, Y.H.; Poon, W.Y.; Zhong, H.; Chen, S.; Li, M.G. Advanced 3D food printing with simultaneous cooking and generative AI design. Adv. Mater. 2025, 37, 2408282. [Google Scholar] [CrossRef] [PubMed]
- Oyinloye, T.M.; Yoon, W.B. Investigation of flow field, die swelling, and residual stress in 3D printing of surimi paste using the finite element method. Innov. Food Sci. Emerg. Technol. 2022, 78, 103008. [Google Scholar] [CrossRef]
- Kong, D.; Zhang, M.; Mujumdar, A.S.; Li, J. Feasibility of hydrocolloid addition for 3D printing of Qingtuan with red bean filling as a dysphagia food. Food Res. Int. 2023, 165, 112469. [Google Scholar] [CrossRef]
- Feng, M.; Zhang, M.; Bhandari, B.; Li, C.; Mujumdar, A.S. Utilizing 3D printing to create sustainable novel food products with innovative ingredients. Innov. Food Sci. Emerg. Technol. 2025, 99, 103873. [Google Scholar] [CrossRef]
- Čukelj Mustač, N.; Pastor, K.; Kojić, J.; Voučko, B.; Ćurić, D.; Rocha, J.M.; Novotni, D. Quality assessment of 3D-printed cereal-based products. LWT 2023, 184, 115065. [Google Scholar] [CrossRef]
- Uribe-Alvarez, R.; Crofton, E.; Kilcawley, K.; Skibinska, I.; Coleman-Vaughan, C.; Murphy, C.P.; O’Shea, N. Sensory characterisation and volatile analysis of 3D-printed dairy protein-based snack structures. Int. J. Food Prop. 2025, 28, 2573182. [Google Scholar] [CrossRef]
- Liu, Z.; Zhang, M.; Bhandari, B.; Wang, Y. 3D printing: Printing precision and application in food sector. Trends Food Sci. Technol. 2017, 69, 83–94. [Google Scholar] [CrossRef]
- Mantihal, S.; Prakash, S.; Bhandari, B. Texture-modified 3D printed dark chocolate: Sensory evaluation and consumer perception study. J. Texture Stud. 2019, 50, 386–399. [Google Scholar] [CrossRef]
- Pérez-Monterroza, E.J.; Chaux-Gutiérrez, A.M.; de Moura, M.R.; Aouada, F.A. Fundamentals and functional applications of 3D and 4D printing in food manufacturing. Processes 2025, 13, 4043. [Google Scholar] [CrossRef]
- Chen, X.; Zhang, M.; Tang, T. Microwave-induced rapid shape change of 4D printed vegetable-based food. Foods 2023, 12, 2158. [Google Scholar] [CrossRef] [PubMed]
- He, C.; Zhang, M.; Fang, Z. 3D printing of food: Pretreatment and post-treatment of materials. Crit. Rev. Food Sci. Nutr. 2020, 60, 2379–2392. [Google Scholar] [CrossRef] [PubMed]
- Soni, R.; Ponappa, K.; Tandon, P. Advancing sustainable food layered manufacturing through 3D printing and post-processing of butterfly pea flower. Rapid Prototyp. J. 2026, 32, 1–8. [Google Scholar] [CrossRef]
- Zhou, Q.; Nan, X.; Zhang, S.; Zhang, L.; Chen, J.; Li, J.; Wang, H.; Ruan, Z. Effect of 3D food printing processing on polyphenol system of loaded Aronia melanocarpa and post-processing evaluation of 3D printing products. Foods 2023, 12, 2068. [Google Scholar] [CrossRef]
- Fujiwara, K.; Igeta, Y.; Toba, K.; Ogawa, J.; Furukawa, H.; Hashizume, M.; Noji, T.; Teratani, K.; Ito, N. Laser Cook Fusion: Layer-specific gelation in 3D food printing via blue laser irradiation. Food Bioprocess Technol. 2025, 18, 6265–6281. [Google Scholar] [CrossRef]
- Zhang, L.; Lou, Y.; Schutyser, M.A.I. 3D printing of cereal-based food structures containing probiotics. Food Struct. 2018, 18, 14–22. [Google Scholar] [CrossRef]
- Chen, J.; Teng, X.; Zhang, M.; Bhandari, B.; Adhikari, B.; Yu, D. 5 D food printing with color change induced by probiotic growth in a starch-protein-based gel system. Food Bioprocess Technol. 2023, 16, 2304–2314. [Google Scholar] [CrossRef]
- Huang, J.; Zhang, M.; Mujumdar, A.S.; Wang, Y.; Li, C. Improvement of 3D printing age-friendly brown rice food on rough texture, swallowability, and in vitro digestibility using fermentation properties of different probiotics. Food Chem. 2024, 460, 140701. [Google Scholar] [CrossRef]
- Boukid, F.; Hassoun, A.; Zouari, A.; Tülbek, M.Ç.; Mefleh, M.; Aït-Kaddour, A.; Castellari, M. Fermentation for designing innovative plant-based meat and dairy alternatives. Foods 2023, 12, 1005. [Google Scholar] [CrossRef]
- Lombardi, L.; Gala, L.D.; Esposito, C.; Tammaro, D. Porous architecture in 3D food printing: Advances in formulation, process control, and sustainable structural design. Compr. Rev. Food Sci. Food Saf. 2025, 24, e70304. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Raza, A.; Xue, Y.-Q.; Yang, G.; Hayat, U.; Yu, J.; Liu, C.; Wang, H.-J.; Wang, J.-Y. Water-responsive 4D printing based on self-assembly of hydrophobic protein “Zein” for the control of degradation rate and drug release. Bioact. Mater. 2023, 23, 343–352. [Google Scholar] [CrossRef] [PubMed]
- Lenie, M.D.R.; Ahmadzadeh, S.; Bockstaele, F.V.; Ubeyitogullari, A. Development of a pH-responsive system based on starch and alginate-pectin hydrogels using coaxial 3D food printing. Food Hydrocoll. 2024, 153, 109989. [Google Scholar] [CrossRef]
- Said, N.S.; Olawuyi, I.F.; Lee, W.Y. Pectin hydrogels: Gel-forming behaviors, mechanisms, and food applications. Gels 2023, 9, 732. [Google Scholar] [CrossRef]
- Zhang, Y.; Wang, Y.; Dai, X.; Li, Y.; Jiang, B.; Li, D.; Liu, C.; Feng, Z. Biointerfacial supramolecular self-assembly of whey protein isolate nanofibrils on probiotic surface to enhance survival and application to 3D printing dysphagia foods. Food Chem. 2024, 460, 140720. [Google Scholar] [CrossRef]
- De Farias, B.S.; de Cunha, L.B.; Christ Ribeiro, A.; Jaeschke, D.P.; Gonçalves, J.O.; Fernandes, S.S.; Cadaval, T.R.S.A., Jr.; Pinto, L.A.D.A. Designing emulsion gels for 3D food printing: Structure, stability, and functional applications. Surfaces 2025, 8, 64. [Google Scholar] [CrossRef]
- Hashemi, B.; Jafarzadeh, S.; Mohammadi, R.; Jafari, S.M. Application of oleogels, hydrogels and bigels as novel structured materials in 3D/4D food printing. Adv. Colloid Interface Sci. 2025, 343, 103578. [Google Scholar] [CrossRef]
- Herdeiro, F.M.; Carvalho, M.O.; Nunes, M.C.; Raymundo, A. Development of healthy snacks incorporating meal from tenebrio molitor and alphitobius diaperinus using 3D printing technology. Foods 2024, 13, 179. [Google Scholar] [CrossRef]
- Ahmadzadeh, S.; Clary, T.; Rosales, A.; Ubeyitogullari, A. Upcycling imperfect broccoli and carrots into healthy snacks using an innovative 3D food printing approach. Food Sci. Nutr. 2023, 12, 84–93. [Google Scholar] [CrossRef]
- Wang, X.; Zhang, M.; Phuhongsung, P.; Mujumdar, A. Impact of internal structural design on quality and nutritional properties of 3D printed food products during post-printing: A critical review. Crit. Rev. Food Sci. Nutr. 2022, 64, 3713–3724. [Google Scholar] [CrossRef]
- Lorenz, T.; Iskandar, M.M.; Baeghbali, V.; Ngadi, M.O.; Kubow, S. 3D food printing applications related to dysphagia: A narrative review. Foods 2022, 11, 1789. [Google Scholar] [CrossRef]
- Qiu, L.; Zhang, M.; Bhandari, B.; Chitrakar, B.; Chang, L. Investigation of 3D printing of apple and edible rose blends as a dysphagia food. Food Hydrocoll. 2023, 135, 108184. [Google Scholar] [CrossRef]
- Xu, B.; Wang, X.; Chitrakar, B.; Xu, Y.; Wei, B.; Wang, B.; Lin, L.; Guo, Z.; Zhou, C.; Ma, H. Effect of various physical modifications of pea protein isolate (PPI) on 3D printing behavior and dysphagia properties of strawberry-PPI gels. Food Hydrocoll. 2025, 158, 110498. [Google Scholar] [CrossRef]
- Yang, Y.; Yang, R.; Fang, Y.; Hou, H.; Zhao, L. Interfacial thickness and roughness of sesame oil bodies by pH regulation for 3D printed dysphagia foods: Texture, rheology, and oral tribology. Food Res. Int. 2026, 229, 118501. [Google Scholar] [CrossRef] [PubMed]
- Villa, C.; Teixeira, C.; Carriço-Sá, B.; Dias, C.; Costa, J.; Mafra, I. Enzymatic hydrolysis as a strategy to reduce allergenicity in sesame (Sesamum indicum) proteins. Innov. Food Sci. Emerg. Technol. 2025, 106, 104284. [Google Scholar] [CrossRef]
- Yang, R.; Cheng, Z.; Zhao, Y.; Song, Y.; Shi, X.; Yu, H.; Zhao, L. Peanut oil body as a food-grade ink for 3D printing: Preparation, characterization and performance. Food Res. Int. 2025, 212, 116486. [Google Scholar] [CrossRef]
- Chao, E.; Li, J.; Duan, Z.; Fan, L. Bigels as emerging biphasic systems: Properties, applications, and prospects in the food industry. Food Hydrocoll. 2024, 154, 110089. [Google Scholar] [CrossRef]
- Chao, E.; Yu, Q.; Li, J.; Fan, L.; Zhou, Y. Intra-phase reinforcement of chitin nanocrystals in bicontinuous bigels: A strategy for high-precision 3D food printing. Carbohydr. Polym. 2026, 379, 124948. [Google Scholar] [CrossRef]
- Cen, S.; Meng, Z. Advances of plant-based fat analogs in 3D printing: Manufacturing strategies, printabilities, and food applications. Food Res. Int. 2024, 197, 115178. [Google Scholar] [CrossRef]
- Cen, S.; Li, S.; Meng, Z. Advances of protein-based emulsion gels as fat analogues: Systematic classification, formation mechanism, and food application. Food Res. Int. 2024, 191, 114703. [Google Scholar] [CrossRef]
- Liu, X.; Cheng, Y.; Sun, T.; Lu, Y.; Huan, S.; Liu, S.; Li, W.; Li, Z.; Liu, Y.; Rojas, O.J.; et al. Recent advances in plant-based edible emulsion gels for 3D-printed foods. Annu. Rev. Food Sci. Technol. 2025, 16, 63–79. [Google Scholar] [CrossRef]
- Zhong, Y.; Wang, B.; Lv, W.; Wu, Y.; Lv, Y.; Sheng, S. Recent research and applications in lipid-based food and lipid-incorporated bioink for 3D printing. Food Chem. 2024, 458, 140294. [Google Scholar] [CrossRef]
- Johansson, L.; Badager, I.; Krona, A.; Abdollahi, M. Printability and interfacial performance of emulsion gels and bigels in multi-material dual and coaxial food 3D printing. Food Hydrocoll. 2026, 172, 111964. [Google Scholar] [CrossRef]
- Sinha, S.S.; Upadhyay, A.; Singh, A.; Mishra, S.; Pandey, N. Bigels a versatile gel composite for tailored application in food industries: A review. Food Struct. 2024, 41, 100380. [Google Scholar] [CrossRef]
- Dominguez, R.; Munekata, P.E.S.; Pateiro, M.; Lopez-Fernandez, O.; Lorenzo, J.M. Immobilization of oils using hydrogels as strategy to replace animal fats and improve the healthiness of meat products. Curr. Opin. Food Sci. 2021, 37, 135–144. [Google Scholar] [CrossRef]
- Li, X.; Fan, L.; Liu, Y.; Li, J. New insights into food O/W emulsion gels: Strategies of reinforcing mechanical properties and outlook of being applied to food 3D printing. Crit. Rev. Food Sci. Nutr. 2023, 63, 1564–1586. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.; Ma, W.; Yao, S.; Wan, B.; He, Z.; Kong, X.; Li, D.; Liu, D.; Xu, E. Morphological modulation of starch chains from nanorod to nanospindle via temperature-controlled rearrangement. Int. J. Biol. Macromol. 2025, 288, 138670. [Google Scholar] [CrossRef] [PubMed]
- Ruan, S.; Tang, J.; Qin, Y.; Wang, J.; Yan, T.; Zhou, J.; Gao, D.; Xu, E.; Liu, D. Mechanical force-induced dispersion of starch nanoparticles and nanoemulsion: Size control, dispersion behaviour, and emulsified stability. Carbohydr. Polym. 2022, 275, 118711. [Google Scholar] [CrossRef]
- Zhong, Y.; Chen, M.; Yao, S.; Zhu, Q.; Kong, X.; Zhang, H.; Liu, D.; Xu, E. 3D-printable docosahexaenoic acid-rich algal oil emulsion gels functionalized by multi-shaped starch nanorods. Carbohydr. Polym. 2026, 376, 124840. [Google Scholar] [CrossRef]
- Li, D.; Yin, H.; Wu, Y.; Feng, W.; Xu, K.-F.; Xiao, H.; Li, C. Ultrastable high internal phase Pickering emulsions: Forming mechanism, processability, and application in 3D printing. J. Agric. Food Chem. 2023, 71, 18829–18841. [Google Scholar] [CrossRef]
- Li, M.; Feng, L.; Xu, Y.; Nie, M.; Li, D.; Zhou, C.; Dai, Z.; Zhang, Z.; Zhang, M. Rheological property, β-carotene stability and 3D printing characteristic of whey protein isolate emulsion gels by adding different polysaccharides. Food Chem. 2023, 414, 135702. [Google Scholar] [CrossRef]
- Kuo, Y.-L.; Chou, Y.-J.; Hu, J.-Y.; Ting, Y. Pickering emulsion emulsified using novel cellulose nanofibers significantly lowers the lipid release rate and cellular absorption. Food Funct. 2024, 15, 4399–4408. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Cheng, L.; Hong, Y.; Li, Z.; Li, C.; Ban, X.; Gu, Z. Effects of hydrocolloids on mechanical properties, viscoelastic and microstructural properties of starch-based modeling clay. Int. J. Biol. Macromol. 2024, 266, 130963. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Liu, Z.; Shi, W. Pickering emulsion stabilized by grass carp myofibrillar protein via one-step: Study on microstructure, processing stability and stabilization mechanism. Food Chem. 2024, 447, 139014. [Google Scholar] [CrossRef]
- Li, L.; Geng, M.; Tan, X.; Teng, F.; Li, Y. Insight on the interaction between soybean protein isolate and ionic/non-ionic polysaccharides: Structural analysis, oil-water interface properties investigation and double emulsion formation. Food Hydrocoll. 2024, 150, 109754. [Google Scholar] [CrossRef]
- Li, Y.; Wang, J.; Ying, R.; Huang, M.; Hayat, K. Protein-stabilized Pickering emulsions interacting with inulin, xanthan gum and chitosan: Rheology and 3D printing behaviour. Carbohydr. Polym. 2024, 326, 121658. [Google Scholar] [CrossRef] [PubMed]
- An, Z.; Hu, L.; Gong, X.; Wang, W.; Zhang, J.; Mo, H.; Xu, D.; Liu, Z. Exploring mussel adhesive protein as a natural Pickering emulsion stabilizer for 3D food printing applications. J. Food Eng. 2026, 406, 112811. [Google Scholar] [CrossRef]

| Ref. | G′ [Pa] | G″ [Pa] | η [Pa·s] | Yield Stress [Pa] | System | Note |
|---|---|---|---|---|---|---|
| [7] | 446–9707 | 93–3489 | 57–1309 | 181–4392 | starch gels + proteins + hydrocolloids | K-index [Pa·sn]: 57.3–912 |
| [11] | 200–7 × 104 | ~50–104 | ~10–105 | 9.4–3220 | nixtamalized corn dough | Flour 25–40% |
| [26] | 1150–6909 | 100–1400 | N/A | 32–455 | potato starch + plant proteins + carrageenan | - |
| [28] | ~1.00 × 104–1.22 × 104 | ~1.50 × 103–2.05 × 103 | 313.48 | 470.69 | mashed potatoes + k-carrageenan + gelatin-B | - |
| [29] | ~1800–1.3 × 104 | 200–2700 | N/A | N/A | fish myofibrillar–protein pastes with oils | for >55 °C, G′ higher for moderate lipids than the oil-free control |
| [31] | 1.34 × 104–2.28 × 104 | 1750–3380 | 9600–7.54 × 104 | N/A | agar + mashed potato + lupin | - |
| [34] | ~1000–4 × 104 | ~300–3 × 104 | ~30–8000 | N/A | cereal–legume starch-based + hydrocolloid mixture | K-index [Pa·sn]: 500–995 |
| [36] | 180–7000 | 150–800 | 49–531 | 18.6–268.7 | psyllium husk (PH)/gelatin blends | K-index [Pa·sn]: 49.3–530 |
| [37] | ~1 × 106–5 × 106 | ~7.9 × 104–4.0 × 105 | ~0.013–0.5 & ~1–1000 (*) | NR | phytosterol-enriched monoglyceride (MG) oleogels | (*) as for wt% phytosterols (up to 40%) and shear rates |
| [38] | ~1000–2 × 104 | ~200–3500 | ~0.8–5000 | N/A | rice protein–corn starch gels (high-amylose) | - |
| [41] | 3.96 × 104–6.09 × 104 | ~2000–8000 | 273–2122 | 268–1803 | shiitake mushroom paste with gums (AG/XG/KG) | - |
| [42] | 150–3.43 × 104 | 57.3–8335 | 160–3.53 × 104 | 16.6–96.3 | rice–black gram idli batter + pearl millet flour | yield stress from Herschel–Bulkley model |
| [43] | ~6000–4.5 × 104 | ~2000–8000 | 1.74 × 103–2.58 × 104 | N/A | surimi paste inks with starch–salt–water | starch enhances G′, and G″; water and salt enhance fluidity |
| Ref. | Nozzle Diameter (mm) | Printing Speed (mm/min) | Layer Height (mm) | Extrusion Rate (mm3/s) | Printing Accuracy (%) |
|---|---|---|---|---|---|
| [7] | 1.0 | 1200 (20 mm/s) | 1.0 | 2.2% | N/A |
| [11] | 2.0 | 1200 (20 mm/s) | 2.0 | N/A | N/A |
| [34] | 1.2 | 1500 (25 mm/s) | 0.84 | N/A | SSI (%) > 99 |
| [37] | 0.83 | 60–360 (1–6 mm/s) | 1.0 | N/A | N/A |
| [41] | 1.2 | 1500 (25 mm/s) | 1.2 | N/A | N/A |
| [43] | 1.94 | 900 (15 mm/s) | 1.2 | 5.75 (actual) | layer sharpness ~39.8% to 55.8% |
| [42] | 1.22 | 400–1400 | various | 7.8 to 27.3 | shape evaluation |
| [46] | 1.0 | 900 (15 mm/s) | 1.0 | 25 | N/A |
| [30] | 1, 1.5, 2, 2.5, 3 | 1800–4200 (30–70 mm/s) | N/A | N/A | N/A |
| [45] | 1.0 | 780 (13 mm/s) | 0.7 | N/A | 82.7% |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Goudoulas, T.; Varzakas, T. The Interconnection Between 3D and 4D Printing and Rheology: From Extrusion and Nozzle Deposition to Final Product Functionality. Processes 2026, 14, 1055. https://doi.org/10.3390/pr14071055
Goudoulas T, Varzakas T. The Interconnection Between 3D and 4D Printing and Rheology: From Extrusion and Nozzle Deposition to Final Product Functionality. Processes. 2026; 14(7):1055. https://doi.org/10.3390/pr14071055
Chicago/Turabian StyleGoudoulas, Thomas, and Theodoros Varzakas. 2026. "The Interconnection Between 3D and 4D Printing and Rheology: From Extrusion and Nozzle Deposition to Final Product Functionality" Processes 14, no. 7: 1055. https://doi.org/10.3390/pr14071055
APA StyleGoudoulas, T., & Varzakas, T. (2026). The Interconnection Between 3D and 4D Printing and Rheology: From Extrusion and Nozzle Deposition to Final Product Functionality. Processes, 14(7), 1055. https://doi.org/10.3390/pr14071055







