Why Does Microalgae Biodiesel Not Work?
Abstract
1. Introduction
2. Methodological Approach
3. Microalgae Biodiesel: Current Status, Constraints, and Feasibility
4. Overview of the Microalgae Biodiesel Production Chain
4.1. Is Productivity the Breaking Point?
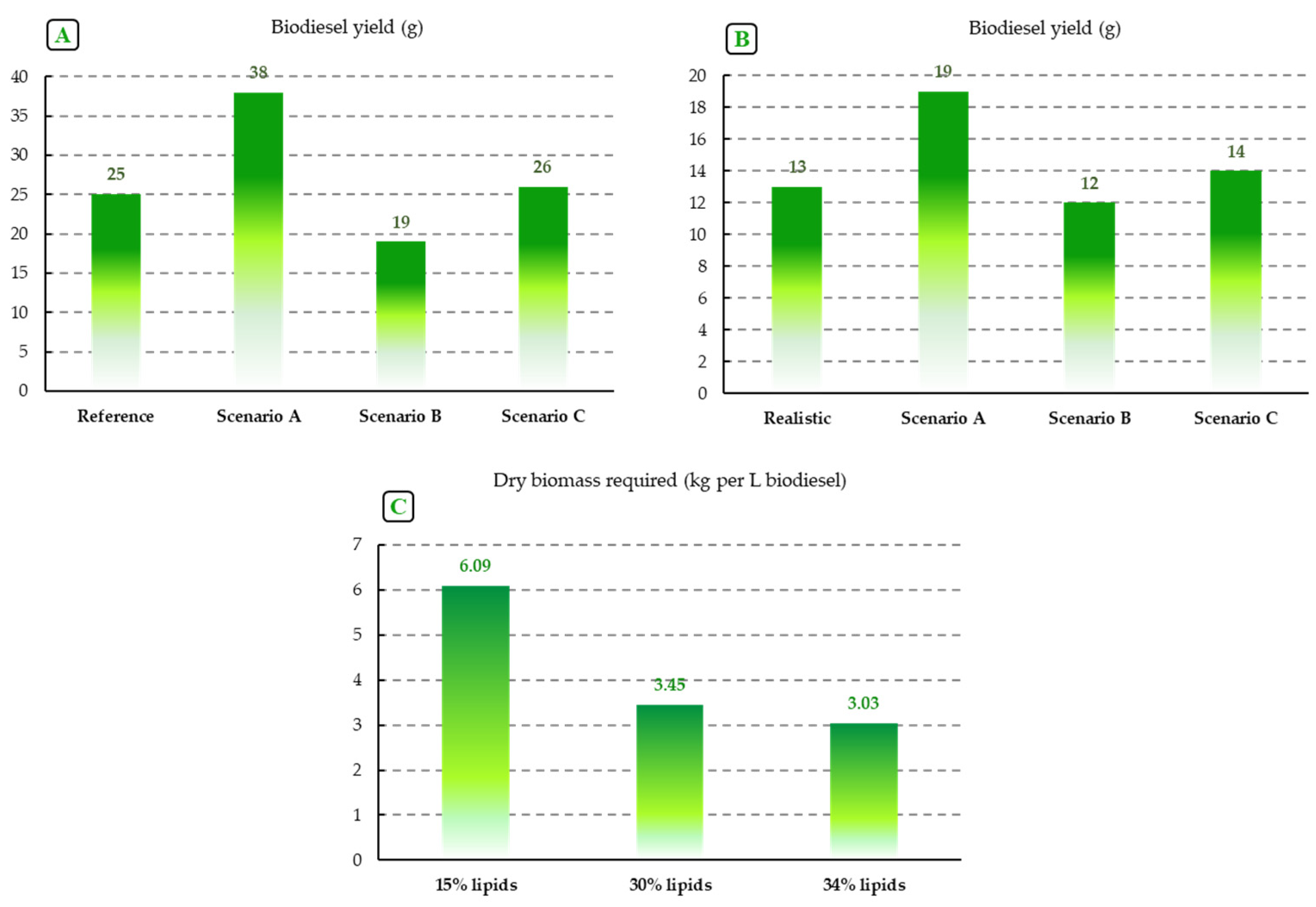
4.2. The Challenge of the Biomass Productivity–Lipid Productivity Trade-Off?
| Microalgae Species | Lipid Content by Weight (%) | Biomass Productivity (t ha−1 Year−1) | Lipid Yield (t ha−1 Year−1) |
|---|---|---|---|
| B. braunii | 25–35 | 20–30 a | 5.00–22.50 b |
| Chlamydomonas reinhardtii | 6–7 | 20–30 a | 1.20–2.10 b |
| Chlorella minutissima | 14–15 | 20–30 a | 2.80–4.50 b |
| Chlorella pyrenoidosa | 38 | 20–30 a | 7.60–11.40 b |
| C. sorokiniana | 22–24 | 20–30 a | 4.40–7.20 b |
| Chlorella sp. | 28–32 | 20–30 a | 5.60–9.60 b |
| C. vulgaris | 49 | 20–30 a | 9.80–15.60 b |
| Crypthecodinium cohnii | 20 | 20–30 a | 4.00–6.00 b |
| Cylindrotheca sp. | 16–37 | 20–30 a | 3.20–11.10 b |
| Dunaliella bioculata | 8 | 20–30 a | 1.60–2.40 b |
| Dunaliella primolecta | 23 | 20–30 a | 4.60–6.90 b |
| Dunaliella salina | 6–25 | 20–30 a | 1.20–7.50 b |
| Dunaliella tertiolecta | 11–16 | 20–30 a | 2.20–4.80 b |
| Nannochloropsis granulata | 28.5 | 20–30 a | 5.70–8.55 b |
| Nannochloropsis oculata | 45 | 20–30 a | 9.00–13.50 b |
| Nannochloropsis sp. | 20–30 | 20–30 a | 4.00–9.00 b |
| Neochloris oleoabundans | 35–40 | 20–30 a | 7.00–19.5 b |
| Nitzschia sp. | 45 | 20–30 a | 9.00–14.10 b |
| P. tricornutum | 20–30 | 20–30 a | 4.00–9.00 b |
| Schizochytrium sp. | 50 | 20–30 a | 10.00–21.00 b |
| Scenedesmus dimorphus | 46 | 20–30 a | 9.20–14.10 b |
| Scenedesmus obliquus | 30–35 | 20–30 a | 6.00–15.00 b |
| Scenedesmus quadricauda | 1.9 | 20–30 a | 0.38–0.57 b |
| Selenastrum minutum | 30–40 | 20–30 a | 6.00–12.00 b |
| Tetraselmis sp. | 20–40 | 20–30 a | 4.00–12.00 b |

4.3. The Battle for the Oil Drop: Extraction as a Second Critical Bottleneck?
4.4. Are Current Microalgae Lipid Profiles Enough for High-Performance Microalgae Biodiesel?
4.5. Does Microalgae Biodiesel Offer a Climate Advantage?
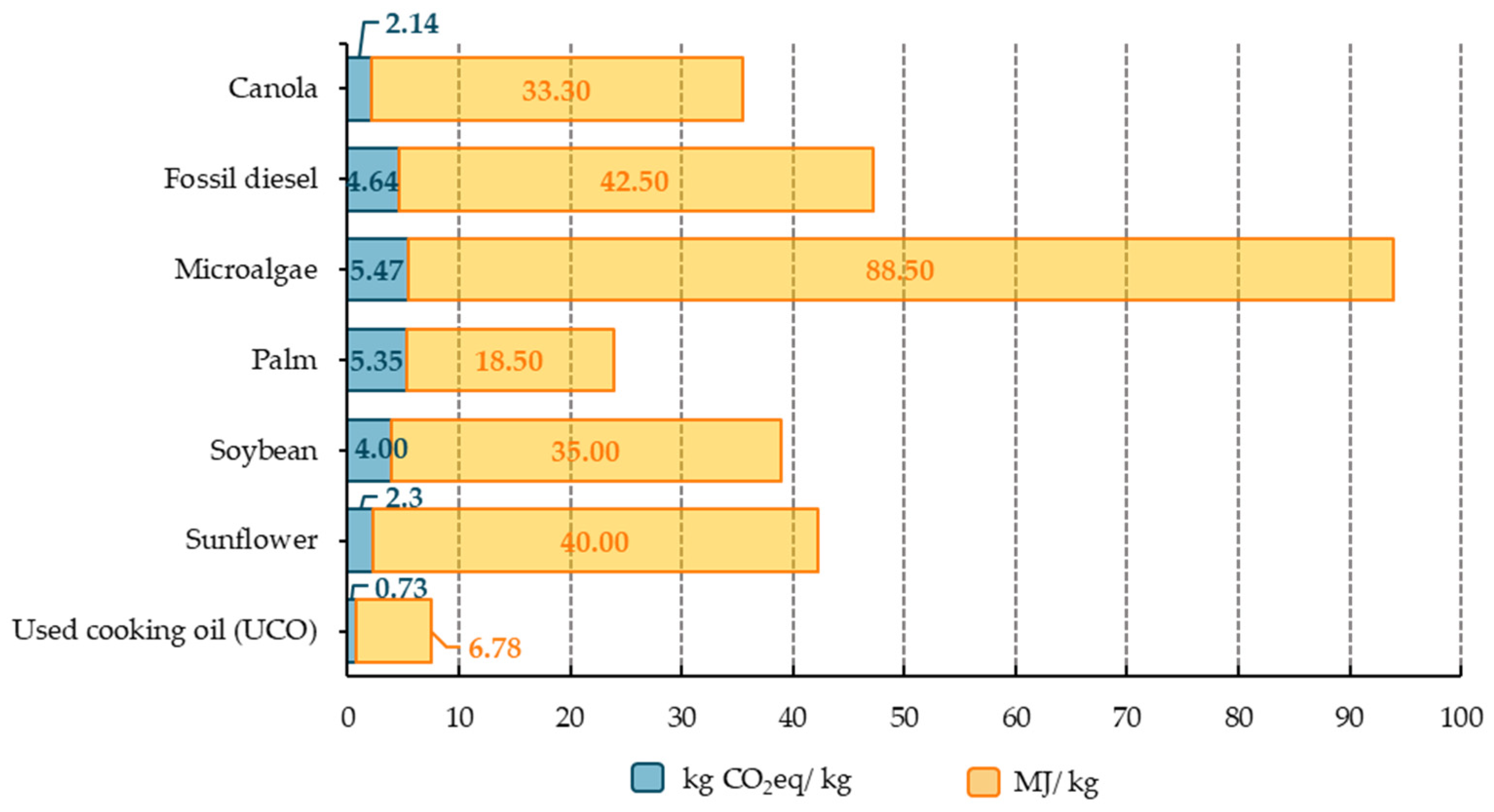
4.6. Proposed Solutions and Their Lack of Scalability
- Genetic engineering: Directs metabolism towards greater lipid accumulation, but compromises cell growth and reduces biomass productivity.
- Use of effluents: Reduces nutrient costs and reinforces the narrative of the circular economy, but imposes high environmental variability, contamination risks, and physiological instability of microalgae.
- Controlled photoperiods and lighting systems: Increase biomass productivity and cultivation control, but raise energy consumption and operating costs.
- Biorefinery: Expands biomass utilization through the production of higher-value-added co-products, but shifts the system to limited market niches and adds separation, purification, and regulatory compliance steps, increasing complexity and costs.
- Industrial CO2 supplementation: Favors microalgal growth and lipid yield, but requires complex infrastructure for CO2 capture, compression, and injection, in addition to increasing the system’s energy demand, raising costs.
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- IEA Bioenergy. International Energy Agency Bioenergy TCP. In IEA Bioenergy: Countries’ Report—Update 2024 (December 2024); IEA Bioenergy: Paris, France, 2024; Available online: https://www.iea.org/energy-system/renewables/bioenergy (accessed on 23 December 2025).
- IEA. Renewables 2024; International Energy Agency: Paris, France, 2024; Available online: https://www.iea.org/reports/renewables-2024/renewable-fuels (accessed on 23 December 2025).
- Chaudhary, S.; Bano, K.S.; Mohanty, T.; Kumari, A.; Mehdi, M.M. Exploring microalgae-based biodiesel as an alternative fuel: Development, production techniques and environmental impacts. Biomass Bioenergy 2025, 202, 108249. [Google Scholar] [CrossRef]
- Pandey, S.; Narayanan, I.; Selvaraj, R.; Varadavenkatesan, T.; Vinayagam, R. Biodiesel production from microalgae: A comprehensive review on influential factors, transesterification processes, and challenges. Fuel 2024, 367, 131547. [Google Scholar] [CrossRef]
- Silva Machado, R.L.; Dias, R.R.; Deprá, M.C.; Zepka, L.Q.; Jacob-Lopes, E. Using Photovoltaics in Methanol Production (Efficiency, Enviromental Impact, and Economical Performance). Compr. Methanol Sci. 2025, 2, 872–890. [Google Scholar] [CrossRef]
- Abdullah, B.; Syed Muhammad, S.A.F.; Shokravi, Z.; Ismail, S.; Kassim, K.A.; Mahmood, A.N.; Aziz, M.M.A. Fourth generation biofuel: A review on risks and mitigation strategies. Renew. Sustain. Energy Rev. 2019, 107, 37–50. [Google Scholar] [CrossRef]
- Rahman, A.; Fares, A.; Veettil, A.V.; Mohtar, R.; Awal, R. A critical review of the microalgae and cyanobacteria-based biofertilizers: An insight into the cost effectiveness of different algae cultivation strategies. Environ. Technol. Innov. 2025, 40, 104480. [Google Scholar] [CrossRef]
- Ashokkumar, V.; Rengasamy, R.; Gopinath, K.P.; Kumar, V.; Rajasekar, A.; Kumar, G.; Chen, W.-H. Cutting-edge advances in alga Botryococcus for eco-friendly biofuels and high-value bioproducts—A critical review. Algal Res. 2024, 83, 103676. [Google Scholar] [CrossRef]
- Food and Agriculture Organization of the United Nations (FAO). Agricultural Production Statistics 2010–2023 (FAOSTAT Analytical Briefs No. 96); FAO: Rome, Italy, 2024; Available online: https://openknowledge.fao.org/handle/20.500.14283/cd3755en (accessed on 27 December 2025).
- Murphy, D.J. Agronomy and Environmental Sustainability of the Four Major Global Vegetable Oil Crops: Oil Palm, Soybean, Rapeseed, and Sunflower. Agronomy 2025, 15, 1465. [Google Scholar] [CrossRef]
- Deprá, M.C.; Severo, I.A.; dos Santos, A.M.; Zepka, L.Q.; Jacob-Lopes, E. Environmental impacts on commercial microalgae-based products: Sustainability metrics and indicators. Algal Res. 2020, 51, 102056. [Google Scholar] [CrossRef]
- Bradley, T.; Rajaeifar, M.A.; Kenny, A.; Hainsworth, C.; del Pino, V.; del Valle Inclán, Y.; Póvoa, I.; Mendonça, P.; Brown, L.; Smallbone, A.; et al. Life cycle assessment of microalgae-derived biodiesel. Int. J. Life Cycle Assess. 2023, 28, 590–609. [Google Scholar] [CrossRef]
- Slegers, P.M.; Lösing, M.B.; Wijffels, R.H.; van Straten, G.; van Boxtel, A.J.B. Scenario evaluation of open pond microalgae production. Algal Res. 2013, 2, 358–368. [Google Scholar] [CrossRef]
- Quinn, J.C.; Davis, R. The potentials and challenges of algae-based biofuels: A review of the techno-economic, life cycle, and resource assessment modeling. Bioresour. Technol. 2015, 184, 444–452. [Google Scholar] [CrossRef] [PubMed]
- Davis, R.; Laurens, L.M.L. Algal Biomass Production via Open Pond Algae Farm Cultivation: 2019 State of Technology and Future Research; NREL/TP-5100-76569; National Renewable Energy Laboratory: Golden, CO, USA, 2020. Available online: https://www.nrel.gov/docs/fy20osti/76569.pdf (accessed on 29 December 2025).
- Montoya-Vallejo, C.; Guzmán Duque, F.L.; Quintero Díaz, J.C. Biomass and lipid production by the native green microalgae Chlorella sorokiniana in response to nutrients, light intensity, and carbon dioxide: Experimental and modeling approach. Front. Bioeng. Biotechnol. 2023, 11, 1149762. [Google Scholar] [CrossRef] [PubMed]
- Davis, R.; Coleman, A.; Hawkins, T.R.; Klein, B.; Zhang, J.; Zhu, Y.; Gao, S.; Singh, U.; Ou, L.; Wiatrowski, M.; et al. Chapter 7.1: Microalgae. In 2023 Billion-Ton Report; Oak Ridge National Laboratory: Oak Ridge, TN, USA, 2024. [Google Scholar] [CrossRef]
- GlobeNewswire. Microalgae Strategic Business Report 2024: Market to Grow by $600 Million by 2030—Emerging into the Spotlight as the Biofuel of the Future. 2024. Available online: https://www.globenewswire.com/news-release/2024/08/01/2922862/28124/en/Microalgae-Strategic-Business-Report-2024-Market-to-Grow-by-600-Million-by-2030-Emerging-Into-the-Spotlight-as-the-Biofuel-of-the-Future.html (accessed on 29 December 2025).
- Research and Markets. Microalgae—Global Strategic Business Report. 2024. Available online: https://www.researchandmarkets.com/reports/5140359/microalgae-global-strategic-business-report (accessed on 29 December 2025).
- Quiroz, D.; Greene, J.M.; Limb, B.J.; Quinn, J.C. Global Life Cycle and Techno-Economic Assessment of Algal-Based Biofuels. Environ. Sci. Technol. 2023, 57, 11541–11551. [Google Scholar] [CrossRef]
- Deprá, M.C.; Dias, R.R.; Zepka, L.Q.; Jacob-Lopes, E. Building cleaner production: How to anchor sustainability in the food production chain? Environ. Adv. 2022, 9, 100295. [Google Scholar] [CrossRef]
- Valdovinos-García, E.M.; Petriz-Prieto, M.A.; Olán-Acosta, M.d.l.Á.; Barajas-Fernández, J.; Guzmán-López, A.; Bravo-Sánchez, M.G. Production of Microalgal Biomass in Photobioreactors as Feedstock for Bioenergy and Other Uses: A Techno-Economic Study of Harvesting Stage. Appl. Sci. 2021, 11, 4386. [Google Scholar] [CrossRef]
- Neupane, D. Biofuels from Renewable Sources, a Potential Option for Biodiesel Production. Bioengineering 2022, 10, 29. [Google Scholar] [CrossRef]
- Okeke, E.S.; Ejeromedoghene, O.; Okoye, C.O.; Ezeorba, T.P.C.; Nyaruaba, R.; Ikechukwu, C.K.; Oladipo, A.; Orege, J.I. Microalgae biorefinery: An integrated route for the sustainable production of high-value-added products. Energy Convers. Manag. X 2022, 16, 100323. [Google Scholar] [CrossRef]
- Sarma, S.; Sharma, S.; Rudakiya, D.; Upadhyay, J.; Rathod, V.; Patel, A.; Narra, M. Valorization of microalgae biomass into bioproducts promoting circular bioeconomy: A holistic approach of bioremediation and biorefinery. 3 Biotech 2021, 11, 378. [Google Scholar] [CrossRef]
- Sharma, A.K.; Jaryal, S.; Sharma, S.; Dhyani, A.; Tewari, B.S.; Mahato, N. Biofuels from Microalgae: A Review on Microalgae Cultivation, Biodiesel Production Techniques and Storage Stability. Processes 2025, 13, 488. [Google Scholar] [CrossRef]
- Geng, Y.; Shaukat, A.; Azhar, W.; Raza, Q.-U.-A.; Tahir, A.; Abideen, M.Z.U.; Zia, M.A.B.; Bashir, M.A.; Rehim, A. Microalgal biorefineries: A systematic review of technological trade-offs and innovation pathways. Biotechnol. Biofuels Bioprod. 2025, 18, 93. [Google Scholar] [CrossRef] [PubMed]
- Bošnjaković, M.; Sinaga, N. The Perspective of Large-Scale Production of Algae Biodiesel. Appl. Sci. 2020, 10, 8181. [Google Scholar] [CrossRef]
- Organization for Economic Co-Operation and Development (OECD); Food and Agriculture Organization of the United Nations (FAO). OECD-FAO Agricultural Outlook 2025–2034; FAO: Rome, Italy, 2025. [Google Scholar] [CrossRef]
- US Department of Energy. Alternative Fuels Data Center: Alternative Fuel Price Report, National Average Fuel Prices (October 2025); U.S. Department of Energy, Office of Energy Efficiency and Renewable Energy: Washington, DC, USA, 2025. Available online: https://afdc.energy.gov/fuels/prices.html (accessed on 3 January 2026).
- Sanjurjo, C.; Oulego, P.; Bartolomé, M.; Rodríguez, E.; Gonzalez, R.; Hernández Battez, A. Biodiesel production from the microalgae Nannochloropsis gaditana: Optimization of the transesterification reaction and physicochemical characterization. Biomass Bioenergy 2024, 185, 107240. [Google Scholar] [CrossRef]
- Liyanaarachchi, V.C.; Premaratne, M.; Ariyadasa, T.U.; Nimarshana, P.H.V.; Malik, A. Two-stage cultivation of microalgae for production of high-value compounds and biofuels: A review. Algal Res. 2021, 57, 102353. [Google Scholar] [CrossRef]
- Rafa, N.; Ahmed, S.F.; Badruddin, I.A.; Mofijur, M.; Kamangar, S. Strategies to Produce Cost-Effective Third-Generation Biofuel From Microalgae. Front. Energy Res. 2021, 9, 749968. [Google Scholar] [CrossRef]
- Machado, R.L.S.; Dutra, D.A.; Schneider, A.T.; Dias, R.R.; Zepka, L.Q.; Jacob-Lopes, E. Recent Advances in Algal Nexus for Circular Economy. In Algae as a Natural Solution for Challenges in Water-Food-Energy Nexus; Kurniawan, T.A., Anouzla, A., Eds.; Environmental Science and Engineering; Springer: Singapore, 2024; pp. 61–77. [Google Scholar] [CrossRef]
- Verma, R.; Kumari, K.V.L.K.; Srivastava, A.; Kumar, A. Photoautotrophic, mixotrophic, and heterotrophic culture media optimization for enhanced microalgae production. J. Environ. Chem. Eng. 2020, 8, 104149. [Google Scholar] [CrossRef]
- Schneider, A.T.; Machado, R.L.S.; Dutra, D.A.; Machado, E.F.; Dias, R.R.; Deprá, M.C.; Zepka, L.Q.; Jacob-Lopes, E. Microalgae biotechnology and its role in sustainable and healthy food design. Front. Bioeng. Biotechnol. 2025, 13, 1716473. [Google Scholar] [CrossRef] [PubMed]
- Goh, B.H.H.; Ong, H.C.; Cheah, M.Y.; Chen, W.H.; Yu, K.L.; Mahlia, T.M.I. Sustainability of direct biodiesel synthesis from microalgae biomass: A critical review. Renew. Sustain. Energy Rev. 2019, 107, 59–74. [Google Scholar] [CrossRef]
- Kumar, L.; Anand, R.; Shah, M.P.; Bharadvaja, N. Microalgae biodiesel: A sustainable source of energy, unit operations, technological challenges, and solutions. J. Hazard. Mater. Adv. 2022, 8, 100145. [Google Scholar] [CrossRef]
- Choi HIl Sung, Y.J.; Hong, M.E.; Han, J.; Min, B.K.; Sim, S.J. Reconsidering the potential of direct microalgal biomass utilization as end-products: A review. Renew. Sustain. Energy Rev. 2022, 155, 111930. [Google Scholar] [CrossRef]
- Barros, A.I.; Gonçalves, A.L.; Simões, M.; Pires, J.C.M. Harvesting techniques applied to microalgae: A review. Renew. Sustain. Energy Rev. 2015, 41, 1489–1500. [Google Scholar] [CrossRef]
- Deepa, P.; Sowndhararajan, K.; Kim, S. A Review of the Harvesting Techniques of Microalgae. Water 2023, 15, 3074. [Google Scholar] [CrossRef]
- Zhu, Z.; Sun, J.; Fa, Y.; Liu, X.; Lindblad, P. Enhancing microalgal lipid accumulation for biofuel production. Front. Microbiol. 2022, 13, 1024441. [Google Scholar] [CrossRef]
- Jothibasu, K.; Muniraj, I.; Jayakumar, T.; Ray, B.; Dhar, D.W.; Karthikeyan, S.; Rakesh, S. Impact of microalgal cell wall biology on downstream processing and nutrient removal for fuels and value-added products. Biochem. Eng. J. 2022, 187, 108642. [Google Scholar] [CrossRef]
- Li, S.; Hu, T.; Xu, Y.; Wang, J.; Chu, R.; Yin, Z.; Mo, F.; Zhu, L. A review on flocculation as an efficient method to harvest energy microalgae: Mechanisms, performances, influencing factors and perspectives. Renew. Sustain. Energy Rev. 2020, 131, 110005. [Google Scholar] [CrossRef]
- McGrath, S.J.; Laamanen, C.A.; Senhorinho, G.N.A.; Scott, J.A. Microalgal harvesting for biofuels—Options and associated operational costs. Algal Res. 2024, 77, 103343. [Google Scholar] [CrossRef]
- Muhammad, G.; Xu, J.; Li, Z.; Zhao, L.; Zhang, X. Current progress and future perspective of microalgae biomass pretreatment using deep eutectic solvents. Sci. Total Environ. 2024, 924, 171547. [Google Scholar] [CrossRef] [PubMed]
- Lu, W.; Alam, M.d.A.; Pan, Y.; Wu, J.; Wang, Z.; Yuan, Z. A new approach of microalgal biomass pretreatment using deep eutectic solvents for enhanced lipid recovery for biodiesel production. Bioresour. Technol. 2016, 218, 123–128. [Google Scholar] [CrossRef]
- Karim, A.; Islam, M.A.; Khalid, Z.B.; Faizal, C.K.M.; Khan, M.d.M.R.; Yousuf, A. Microalgal Cell Disruption and Lipid Extraction Techniques for Potential Biofuel Production. In Microalgae Cultivation for Biofuels Production; Elsevier: Amsterdam, The Netherlands, 2020; pp. 129–147. [Google Scholar] [CrossRef]
- Wan Osman, W.N.A.; Rosli, M.H.; Mazli, W.N.A.; Samsuri, S. Comparative review of biodiesel production and purification. Carbon Capture Sci. Technol. 2024, 13, 100264. [Google Scholar] [CrossRef]
- Anand, K.; Ranjan, A.; Mehta, P.S. Estimating the Viscosity of Vegetable Oil and Biodiesel Fuels. Energy Fuels 2010, 24, 664–672. [Google Scholar] [CrossRef]
- Gonçalves, M.D.; da Silva, F.C.; Lopes Maria, A.C.; de Souza, L.A.; de Oliveira, P.C.O. Production and Characterization of Biodiesel Produced with Single Oils and Binary Blends. Braz. J. Environ. Sci. 2019, 53, 33–50. [Google Scholar]
- Chen, M.; Chen, Y.; Zhang, Q. A Review of Energy Consumption in the Acquisition of Bio-Feedstock for Microalgae Biofuel Production. Sustainability 2021, 13, 8873. [Google Scholar] [CrossRef]
- Chaos-Hernández, D.; Reynel-Ávila, H.E.; Bonilla-Petriciolet, A.; Villalobos-Delgado, F.J. Extraction methods of algae oils for the production of third generation biofuels—A review. Chemosphere 2023, 341, 139856. [Google Scholar] [CrossRef]
- Thawechai, T.; Cheirsilp, B.; Louhasakul, Y.; Boonsawang, P.; Prasertsan, P. Mitigation of carbon dioxide by oleaginous microalgae for lipids and pigments production: Effect of light illumination and carbon dioxide feeding strategies. Bioresour. Technol. 2016, 219, 139–149. [Google Scholar] [CrossRef]
- Xin, Y.; Wu, S.; Miao, C.; Xu, T.; Lu, Y. Towards Lipid from Microalgae: Products, Biosynthesis, and Genetic Engineering. Life 2024, 14, 447. [Google Scholar] [CrossRef] [PubMed]
- Vijayan, J.; Alvarez, S.; Naldrett, M.J.; Morse, W.; Maliva, A.; Wase, N.; Riekhof, W.R. Nitrogen starvation leads to TOR kinase-mediated downregulation of fatty acid synthesis in the algae Chlorella sorokiniana and Chlamydomonas reinhardtii. BMC Plant Biol. 2024, 24, 753. [Google Scholar] [CrossRef] [PubMed]
- de Jesus, S.S.; Filho, R.M. Potential of algal biofuel production in a hybrid photobioreactor. Chem. Eng. Sci. 2017, 171, 282–292. [Google Scholar] [CrossRef]
- Borella, L.; Diotto, D.; Barbera, E.; Fiorimonte, D.; Sforza, E.; Trivellin, N. Application of flashing blue-red LED to boost microalgae biomass productivity and energy efficiency in continuous photobioreactors. Energy 2022, 259, 125087. [Google Scholar] [CrossRef]
- Leong, W.H.; Lim, J.W.; Rawindran, H.; Liew, C.S.; Lam, M.K.; Ho, Y.C.; Khoo, K.S.; Kusakabe, K.; Abdelghani, H.T.M.; Ho, C.-D.; et al. Energy balance and life cycle assessments in producing microalgae biodiesel via a continuous microalgal-bacterial photobioreactor loaded with wastewater. Chemosphere 2023, 341, 139953. [Google Scholar] [CrossRef]
- Alishah Aratboni, H.; Rafiei, N.; Garcia-Granados, R.; Alemzadeh, A.; Morones-Ramírez, J.R. Biomass and lipid induction strategies in microalgae for biofuel production and other applications. Microb. Cell Fact. 2019, 18, 178. [Google Scholar] [CrossRef]
- Song, X.; Liu, B.F.; Kong, F.; Song, Q.; Ren, N.Q.; Ren, H.Y. New insights into rare earth element-induced microalgae lipid accumulation: Implication for biodiesel production and adsorption mechanism. Water Res. 2024, 251, 121134. [Google Scholar] [CrossRef]
- Breuer, G.; Lamers, P.P.; Janssen, M.; Wijffels, R.H.; Martens, D.E. Opportunities to improve the areal oil productivity of microalgae. Bioresour. Technol. 2015, 186, 294–302. [Google Scholar] [CrossRef]
- Kenny, P.; Flynn, K.J. Physiology limits commercially viable photoautotrophic production of microalgal biofuels. J. Appl. Phycol. 2017, 29, 2713–2727. [Google Scholar] [CrossRef] [PubMed]
- Rawat, I.; Ranjith Kumar, R.; Mutanda, T.; Bux, F. Biodiesel from microalgae: A critical evaluation from laboratory to large scale production. Appl. Energy 2013, 103, 444–467. [Google Scholar] [CrossRef]
- Breuer, G.; Lamers, P.P.; Martens, D.E.; Draaisma, R.B.; Wijffels, R.H. The impact of nitrogen starvation on the dynamics of triacylglycerol accumulation in nine microalgae strains. Bioresour. Technol. 2012, 124, 217–226. [Google Scholar] [CrossRef] [PubMed]
- Themistoklis Neokosmidis. Algae to Liquid Biofuels-State of Industry and Technology Literature Review. 2025. Available online: https://www.concawe.eu/publication/algae-to-liquid-biofuels-state-of-industry-and-technology-literature-review/ (accessed on 21 March 2026).
- Klein, B.; Davis, R.; Wiatrowski, M. Algal Biomass Production via Open Pond Algae Farm Cultivation: 2023 State of Technology and Future Research. 2024. Available online: https://www.nrel.gov/docs/fy24osti/88802.pdf (accessed on 3 January 2026).
- Yousuf, A.; Unc, A.; Bergerson, J.; De la Hoz Siegler, H. Unveiling the factors shaping variability in biomass productivity: Meta-analysis of outdoor pilot-scale microalgal cultures. Bioresour. Technol. 2025, 430, 132566. [Google Scholar] [CrossRef]
- Chisti, Y. Biodiesel from microalgae. Biotechnol. Adv. 2007, 25, 294–306. [Google Scholar] [CrossRef]
- Sun, H.; Zhao, W.; Mao, X.; Li, Y.; Wu, T.; Chen, F. High-value biomass from microalgae production platforms: Strategies and progress based on carbon metabolism and energy conversion. Biotechnol. Biofuels 2018, 11, 227. [Google Scholar] [CrossRef]
- Neag, E.; Stupar, Z.; Maicaneanu, S.A.; Roman, C. Advances in Biodiesel Production from Microalgae. Energies 2023, 16, 1129. [Google Scholar] [CrossRef]
- Vázquez-Romero, B.; Perales, J.A.; Pereira, H.; Barbosa, M.; Ruiz, J. Techno-economic assessment of microalgae production, harvesting and drying for food, feed, cosmetics, and agriculture. Sci. Total Environ. 2022, 837, 155742. [Google Scholar] [CrossRef]
- Vasistha, S.; Khanra, A.; Clifford, M.; Rai, M.P. Current advances in microalgae harvesting and lipid extraction processes for improved biodiesel production: A review. Renew. Sustain. Energy Rev. 2021, 137, 110498. [Google Scholar] [CrossRef]
- Oh, Y.K.; Kim, S.; Ilhamsyah, D.P.A.; Lee, S.G.; Kim, J.R. Cell disruption and lipid extraction from Chlorella species for biorefinery applications: Recent advances. Bioresour. Technol. 2022, 366, 128183. [Google Scholar] [CrossRef] [PubMed]
- Demarco, A.; Gibon, V. Overview of the soybean process in the crushing industry. OCL 2020, 27, 60. [Google Scholar] [CrossRef]
- Khoo, K.S.; Chew, K.W.; Yew, G.Y.; Leong, W.H.; Chai, Y.H.; Show, P.L.; Chen, W.-H. Recent advances in downstream processing of microalgae lipid recovery for biofuel production. Bioresour. Technol. 2020, 304, 122996. [Google Scholar] [CrossRef]
- Wijffels, R.H.; Barbosa, M.J. An Outlook on Microalgal Biofuels. Science (1979) 2010, 329, 796–799. [Google Scholar] [CrossRef]
- Gonzalez, D.I.; Ynalvez, R.A. Comparison of the effects of nitrogen-, sulfur- and combined nitrogen- and sulfur-deprivations on cell growth, lipid bodies and gene expressions in Chlamydomonas reinhardtii cc5373-sta6. BMC Biotechnol. 2023, 23, 35. [Google Scholar] [CrossRef]
- Ramesh Kumar, B.; Deviram, G.; Mathimani, T.; Duc, P.A.; Pugazhendhi, A. Microalgae as rich source of polyunsaturated fatty acids. Biocatal. Agric. Biotechnol. 2019, 17, 583–588. [Google Scholar] [CrossRef]
- Mulgund, A. Increasing lipid accumulation in microalgae through environmental manipulation, metabolic and genetic engineering: A review in the energy NEXUS framework. Energy Nexus 2022, 5, 100054. [Google Scholar] [CrossRef]
- Simionato, D.; Block, M.A.; La Rocca, N.; Jouhet, J.; Maréchal, E.; Finazzi, G.; Morosinotto, T. The Response of Nannochloropsis gaditana to Nitrogen Starvation Includes De Novo Biosynthesis of Triacylglycerols, a Decrease of Chloroplast Galactolipids, and Reorganization of the Photosynthetic Apparatus. Eukaryot. Cell 2013, 12, 665–676. [Google Scholar] [CrossRef]
- Branco-Vieira, M.; Mata, T.M.; Martins, A.A.; Freitas, M.A.V.; Caetano, N.S. Economic analysis of microalgae biodiesel production in a small-scale facility. Energy Rep. 2020, 6, 325–332. [Google Scholar] [CrossRef]
- Sharma, V.; Hossain, A.K.; Duraisamy, G.; Griffiths, G. Microalgal Biodiesel: A Challenging Route toward a Sustainable Aviation Fuel. Fermentation 2023, 9, 907. [Google Scholar] [CrossRef]
- Sivaramakrishnan, R.; Suresh, S.; Kanwal, S.; Ramadoss, G.; Ramprakash, B.; Incharoensakdi, A. Microalgal Biorefinery Concepts’ Developments for Biofuel and Bioproducts: Current Perspective and Bottlenecks. Int. J. Mol. Sci. 2022, 23, 2623. [Google Scholar] [CrossRef]
- Santin, A.; Russo, M.T.; Ferrante, M.I.; Balzano, S.; Orefice, I.; Sardo, A. Highly Valuable Polyunsaturated Fatty Acids from Microalgae: Strategies to Improve Their Yields and Their Potential Exploitation in Aquaculture. Molecules 2021, 26, 7697. [Google Scholar] [CrossRef]
- Braun, J.C.A.; Colla, L.M. Use of Microalgae for the Development of Biofertilizers and Biostimulants. Bioenergy Res. 2023, 16, 289–310. [Google Scholar] [CrossRef]
- Machado, R.L.S.; Dutra, D.A.; Schneider, A.T.; Dias, R.R.; Deprá, M.C.; Zepka, L.Q.; Jacob-Lopes, E. Future prospect of development of integrated wastewater and algal biorefineries and its impact on biodiversity and environment. In Algal Biorefinery; Elsevier: Amsterdam, The Netherlands, 2025; pp. 371–383. [Google Scholar] [CrossRef]
- Chhandama, M.V.L.; Ruatpuia, J.V.L.; Ao, S.; Chetia, A.C.; Satyan, K.B.; Rokhum, S.L. Microalgae as a sustainable feedstock for biodiesel and other production industries: Prospects and challenges. Energy Nexus 2023, 12, 100255. [Google Scholar] [CrossRef]
- Shivakumar, S.; Serlini, N.; Esteves, S.M.; Miros, S.; Halim, R. Cell Walls of Lipid-Rich Microalgae: A Comprehensive Review on Characterisation, Ultrastructure, and Enzymatic Disruption. Fermentation 2024, 10, 608. [Google Scholar] [CrossRef]
- Tang, D.Y.Y.; Khoo, K.S.; Chew, K.W.; Tao, Y.; Ho, S.H.; Show, P.L. Potential utilization of bioproducts from microalgae for the quality enhancement of natural products. Bioresour. Technol. 2020, 304, 122997. [Google Scholar] [CrossRef]
- Khan, M.I.; Shin, J.H.; Kim, J.D. The promising future of microalgae: Current status, challenges, and optimization of a sustainable and renewable industry for biofuels, feed, and other products. Microb. Cell Fact. 2018, 17, 36. [Google Scholar] [CrossRef] [PubMed]
- Khoo, K.S.; Ahmad, I.; Chew, K.W.; Iwamoto, K.; Bhatnagar, A.; Show, P.L. Enhanced microalgal lipid production for biofuel using different strategies including genetic modification of microalgae: A review. Prog. Energy Combust. Sci. 2023, 96, 101071. [Google Scholar] [CrossRef]
- Zapata-Boada, S.; Gonzalez-Miquel, M.; Jobson, M.; Cuéllar-Franca, R.M. Techno-economic and Environmental Analysis of Algae Biodiesel Production via Lipid Extraction Using Alternative Solvents. Ind. Eng. Chem. Res. 2022, 61, 18030–18044. [Google Scholar] [CrossRef]
- Oliva, G.; Buonerba, A.; Grassi, A.; Hasan, S.W.; Korshin, G.V.; Zorpas, A.A.; Belgiorno, V.; Naddeo, V.; Zarra, T. Microalgae to biodiesel: A novel green conversion method for high-quality lipids recovery and in-situ transesterification to fatty acid methyl esters. J. Environ. Manag. 2024, 357, 120830. [Google Scholar] [CrossRef] [PubMed]
- Ischebeck, T.; Krawczyk, H.E.; Mullen, R.T.; Dyer, J.M.; Chapman, K.D. Lipid droplets in plants and algae: Distribution, formation, turnover and function. Semin. Cell Dev. Biol. 2020, 108, 82–93. [Google Scholar] [CrossRef]
- Dutra, D.A.; Deprá, M.C.; Dias, R.R.; Zepka, L.Q.; Jacob-Lopes, E. Ensuring nutrition and food safety within planetary boundaries: The role of microalgae-based ingredients in sustainable food chain. J. Appl. Phycol. 2024, 36, 2635–2654. [Google Scholar] [CrossRef]
- Xu, K.; Zou, W.; Peng, B.; Guo, C.; Zou, X. Lipid Droplets from Plants and Microalgae: Characteristics, Extractions, and Applications. Biology 2023, 12, 594. [Google Scholar] [CrossRef] [PubMed]
- Walayat, N.; Yurdunuseven-Yıldız, A.; Kumar, M.; Goksen, G.; Öztekin, S.; Lorenzo, J.M. Oxidative stability, quality, and bioactive compounds of oils obtained by ultrasound and microwave-assisted oil extraction. Crit. Rev. Food Sci. Nutr. 2024, 64, 9974–9991. [Google Scholar] [CrossRef]
- Reuscher, S.; Klock, G.; Kersten, A.; Schabel, S.; Graf, R. Improved pre-treatment for lipid staining of microalgae with a rigid cell wall. J. Microbiol. Methods 2025, 236, 107205. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Guo, Y.; Wang, S.; Xie, L.; Lu, Q.; Wang, X. Effect of steam explosion pretreatment on the aqueous extraction efficiency of soybean oil. Innov. Food Sci. Emerg. Technol. 2025, 104, 104080. [Google Scholar] [CrossRef]
- Farooq, W.; Mishra, S.K.; Moon, M.; Suh, W.I.; Shrivastav, A.; Kumar, K.; Kwon, J.H.; Park, M.S.; Yang, J.W. Energy efficient process for microalgae cell disruption for oil recovery using triiodide resin. Algal Res. 2016, 13, 102–108. [Google Scholar] [CrossRef]
- Rahman, M.; Rasul, M.; Hassan, N.; Hyde, J. Prospects of Biodiesel Production from Macadamia Oil as an Alternative Fuel for Diesel Engines. Energies 2016, 9, 403. [Google Scholar] [CrossRef]
- Pataro, G.; Eslami, E.; Pignataro, F.; Procentese, A. Downstream Processes in a Microalgae Biorefinery: Cascaded Enzymatic Hydrolysis and Pulsed Electric Field as Green Solution. Processes 2025, 13, 1629. [Google Scholar] [CrossRef]
- Domozych, D.S.; Ciancia, M.; Fangel, J.U.; Mikkelsen, M.D.; Ulvskov, P.; Willats, W.G.T. The Cell Walls of Green Algae: A Journey through Evolution and Diversity. Front. Plant Sci. 2012, 3, 82. [Google Scholar] [CrossRef]
- Gaurav, K.; Neeti, K.; Singh, R. Microalgae-based biodiesel production and its challenges and future opportunities: A review. Green Technol. Sustain. 2024, 2, 100060. [Google Scholar] [CrossRef]
- Makareviciene, V.; Sendzikiene, E. Application of Microalgae Biomass for Biodiesel Fuel Production. Energies 2022, 15, 4178. [Google Scholar] [CrossRef]
- Mahata, C.; Das, P.; Khan, S.; Thaher, M.I.A.; Abdul Quadir, M.; Annamalai, S.N.; Al Jabri, H. The Potential of Marine Microalgae for the Production of Food, Feed, and Fuel (3F). Fermentation 2022, 8, 316. [Google Scholar] [CrossRef]
- Jeon, B.-H.; Choi, J.-A.; Kim, H.-C.; Hwang, J.-H.; Abou-Shanab, R.A.I.; Dempsey, B.A.; Regan, J.M.; Kim, J.R. Ultrasonic disintegration of microalgal biomass and consequent improvement of bioaccessibility/bioavailability in microbial fermentation. Biotechnol. Biofuels 2013, 6, 37. [Google Scholar] [CrossRef]
- Nguyen, T.T.; Lam, M.K.; Cheng, Y.W.; Uemura, Y.; Mansor, N.; Lim, J.W.; Show, P.L.; Tan, I.S.; Lim, S. Reaction kinetic and thermodynamics studies for in-situ transesterification of wet microalgae paste to biodiesel. Chem. Eng. Res. Des. 2021, 169, 250–264. [Google Scholar] [CrossRef]
- Kumar, M.; Ansari, N.A.; Gautam, R. Algae biodiesel as a alternative green fuel: A futuristic scope. Clean. Chem. Eng. 2025, 11, 100178. [Google Scholar] [CrossRef]
- ASTM D6751; Standard Specification for Biodiesel Fuel Blend Stock (B100) for Middle Distillate Fuels. ASTM International: West Conshohocken, PA, USA, 2020. Available online: https://www.astm.org/d6751-20a.html (accessed on 20 December 2025).
- EN 14214; Biodiesel Fuel Standards. CEN: Brussels, Belgium, 2024. Available online: https://dieselnet.com/standards/eu/fuel_biodiesel.php (accessed on 20 December 2025).
- National Agency of Petroleum NG and B (ANP). ANP 45/2014; ANP Resolution No. 45/2014: Biodiesel Specification. 2014. Available online: https://www.gov.br/anp/pt-br/assuntos/producao-e-fornecimento-de-biocombustiveis/biodiesel/especificacao-do-biodiesel (accessed on 20 December 2025).
- AS 3570:1998; Automotive Diesel Fuel. Standards Australia: Sydney, Australia, 1998. Available online: https://img.antpedia.com/standard/files/pdfs_ora/20230612/as/AS/AS%203570-1998.pdf (accessed on 20 December 2025).
- Francisco, É.C.; Neves, D.B.; Jacob-Lopes, E.; Franco, T.T. Microalgae as feedstock for biodiesel production: Carbon dioxide sequestration, lipid production and biofuel quality. J. Chem. Technol. Biotechnol. 2010, 85, 395–403. [Google Scholar] [CrossRef]
- Pradana, Y.S.; Makertihartha, I.G.B.N.; Indarto, A.; Prakoso, T.; Soerawidjaja, T.H. A Review of Biodiesel Cold Flow Properties and Its Improvement Methods: Towards Sustainable Biodiesel Application. Energies 2024, 17, 4543. [Google Scholar] [CrossRef]
- Hawrot-Paw, M.; Ratomski, P.; Koniuszy, A.; Golimowski, W.; Teleszko, M.; Grygier, A. Fatty Acid Profile of Microalgal Oils as a Criterion for Selection of the Best Feedstock for Biodiesel Production. Energies 2021, 14, 7334. [Google Scholar] [CrossRef]
- Tizvir, A.; Shojaeefard, M.H.; Molaeimanesh, G.R.; Zahedi, A.; Kanani, B. Sustainable production and application of microalgae-based biodiesel using a renewable hybrid energy system: Enhancing emission reduction and engine performance in cold start conditions for greener transportation. Biofuels 2026, 17, 35–56. [Google Scholar] [CrossRef]
- Zheng, F.; Cho, H.M. The Effect of Different Mixing Proportions and Different Operating Conditions of Biodiesel Blended Fuel on Emissions and Performance of Compression Ignition Engines. Energies 2024, 17, 344. [Google Scholar] [CrossRef]
- Dhairiyasamy, R.; Dixit, S.; Singh, S.; Gabiriel, D. A comprehensive analysis of combustion efficiency and emissions in biodiesel blends for sustainable energy solutions. Int. J. Thermofluids 2025, 29, 101357. [Google Scholar] [CrossRef]
- de Oliveira, D.T.; de Jesus Paiva, R.; de Mescouto, V.A.; da Silva, S.R.F.; Da Costa, A.A.F.; Santos, A.V.; Gonçalves, E.C.; da Rocha Filho, G.N.; Noronha, R.C.R.; Nascimento, L.A.S.D. The potential of third-generation biodiesel from Tolypothrix sp. CACIAM22 as a feedstock. Heliyon 2024, 10, e36343. [Google Scholar] [CrossRef]
- Saber, H.; Galal, H.R.; Abo-Eldahab, M.; Alwaleed, E. Enhancing the biodiesel production in the green alga Chlorella vulgaris by heavy metal stress and prediction of fuel properties from fatty acid profiles. Environ. Sci. Pollut. Res. 2024, 31, 35952–35968. [Google Scholar] [CrossRef]
- Hasnain, M.; Munir, N.; Abideen, Z.; Macdonald, H.; Hamid, M.; Abbas, Z.; El-Keblawy, A.; Mancinelli, R.; Radicetti, E. Prospects for Biodiesel Production from Emerging Algal Resource: Process Optimization and Characterization of Biodiesel Properties. Agriculture 2023, 13, 407. [Google Scholar] [CrossRef]
- Trivedi, J.; Agrawal, D.; Atray, N.; Ray, A. Enhanced lipid production in Scenedesmus obliquus via nitrogen starvation in a two-stage cultivation process and evaluation for biodiesel production. Fuel 2022, 316, 123418. [Google Scholar] [CrossRef]
- Ariyanti, D.; Sasongko, N.A.; Fitriana, E.L.; Nugroho, R.A.; Arfiyanti. comparison of the physicochemical properties of microalgae biodiesel with other oilseed feedstocks for sustainable energy production: A meta-analysis. J. Appl. Res. Technol. 2024, 22, 432–440. [Google Scholar] [CrossRef]
- Díez-Valbuena, G.; García Tuero, A.; Rodríguez, E.; Fernández-González, A.; Hernández Battez, A. Modified oil from Schizochytrium sp. microalgae as a lubricity improver for diesel. Biomass Bioenergy 2026, 208, 108883. [Google Scholar] [CrossRef]
- Sanford, S.D.; White, J.M.; Shah, P.S.; Wee, C.; Valverde, M.A.; Meier, G.R. Feedstock and Biodiesel Characteristics Report; Renewable Energy Group, Inc.: Ames, IA, USA, 2009; Available online: https://biodieseleducation.org/Feedstock/documents/Feedstock%20and%20Biodiesel%20Characteristics%20Report%20(1).pdf (accessed on 29 November 2025).
- Ali, E.N.; Tay, C.I. Characterization of Biodiesel Produced from Palm Oil via Base Catalyzed Transesterification. Procedia Eng. 2013, 53, 7–12. [Google Scholar] [CrossRef]
- Anwar, M.; Rasul, M.G.; Ashwath, N.; Rahman, M.M. Optimisation of Second-Generation Biodiesel Production from Australian Native Stone Fruit Oil Using Response Surface Method. Energies 2018, 11, 2566. [Google Scholar] [CrossRef]
- Abd El Baky, H.; El Baroty, G. Cultivation of Pseudochlorella pringsheimii for biodiesel production in a scalable indoor photobioreactor: Case studies from Egypt. J. Genet. Eng. Biotechnol. 2023, 21, 25. [Google Scholar] [CrossRef]
- Kipkoech, R.; Takase, M.; Ahogle, A.M.A.; Ocholla, G. Analysis of properties of biodiesel and its development and promotion in Ghana. Heliyon 2024, 10, e39078. [Google Scholar] [CrossRef]
- Uebe, J.; Lekaviciute, E.; Kryzevicius, Z.; Zukauskaite, A. Comparison of Antioxidants to Increase the Oxidation Stability of Pyrolysis Oils of Three Plastics Using Iodine Value. Processes 2024, 12, 638. [Google Scholar] [CrossRef]
- Nayak, C.; Bhatlu, L.D.; Chelluboyana, V.R.; Sahoo, S.P.; Behera, S.K.; Bhaisal, T.; Samal, B.; Sahoo, B.; Sahoo, K.S.S.; Arunkumar, P.; et al. An in-depth comparative analysis of fuel evolution: Tracking physico-chemical characteristics in conventional diesel, e-diesel, biodiesel, and aged biodiesel. Biofuels 2025, 1–47. [Google Scholar] [CrossRef]
- Srinivasa Rao, Y.; Getachew Alenka, T. Performance and Emission Analysis of Common Rail Diesel Engine with Microalgae Biodiesel. J. Eng. 2022, 2022, 7441659. [Google Scholar] [CrossRef]
- Thomassen, G.; Van Dael, M.; Van Passel, S. The potential of microalgae biorefineries in Belgium and India: An environmental techno-economic assessment. Bioresour. Technol. 2018, 267, 271–280. [Google Scholar] [CrossRef]
- Kirana Fadilah Achmad, A.; Fathurrahman, N.A.; Kunarti, E.S.; Wibowo, C.S.; Khalil, M.; Al-Saadi, A.A. Optimizing cold-flow properties and oxidation stability of B40 biodiesel blend with turpentine oil and ethanol: Experimental and quantum chemical approach. Fuel 2025, 381, 133258. [Google Scholar] [CrossRef]
- Venu, H.; Appavu, P. Effect of nano additives in Spirulina microalgae biodiesel–diesel blends in a direct injection diesel engine. Int. J. Ambient. Energy 2022, 43, 6167–6174. [Google Scholar] [CrossRef]
- Tiwari, C.; Verma, T.N.; Dwivedi, G.; Verma, P. Energy-Exergy Analysis of Diesel Engine Fueled with Microalgae Biodiesel-Diesel Blend. Appl. Sci. 2023, 13, 1857. [Google Scholar] [CrossRef]
- Çamur, H.; Al-Ani, A.M.R. Prediction of Oxidation Stability of Biodiesel Derived from Waste and Refined Vegetable Oils by Statistical Approaches. Energies 2022, 15, 407. [Google Scholar] [CrossRef]
- Alcocer-García, H.; Sánchez-Ramírez, E.; García-García, E.; Ramírez-Márquez, C.; Ponce-Ortega, J.M. Unlocking the Potential of Biomass Resources: A Review on Sustainable Process Design and Intensification. Resources 2025, 14, 143. [Google Scholar] [CrossRef]
- Huang, R.; Li, J.; Tang, Y.; Song, W.; Yu, Y.; Yang, W.; Zheng, R. Comparative life-cycle assessment of microalgal biodiesel production via various emerging wet scenarios: Energy conversion characteristics and environmental impacts. Energy Convers. Manag. 2022, 257, 115427. [Google Scholar] [CrossRef]
- Ali Rajaeifar, M.; Ghobadian, B.; Davoud Heidari, M.; Fayyazi, E. Energy consumption and greenhouse gas emissions of biodiesel production from rapeseed in Iran. J. Renew. Sustain. Energy 2013, 5, 063134. [Google Scholar] [CrossRef]
- Severo, I.A.; Siqueira, S.F.; Deprá, M.C.; Maroneze, M.M.; Zepka, L.Q.; Jacob-Lopes, E. Biodiesel facilities: What can we address to make biorefineries commercially competitive? Renew. Sustain. Energy Rev. 2019, 112, 686–705. [Google Scholar] [CrossRef]
- Patel, K.; Singh, S.K. Assessing the Sustainability of Jatropha and Rapeseed Biodiesel: An LCA Approach. In Recent Developments in Energy and Environmental Engineering; Springer: Singapore, 2023; pp. 191–200. [Google Scholar] [CrossRef]
- Leonidovna, K.G.; Sharipovish, S.Z.; Tatyana, A.P.; Viktorovna, S.N.; Fedorovna, M.E. Comprehensive Assessment of Soybean Biodiesel Environmental and Combustion Characteristics. Tikrit J. Eng. Sci. 2025, 32, 1–9. [Google Scholar] [CrossRef]
- Szczepaniak, I.; Olech, I.; Szymańska, E.J. The Use of Canola for Biofuel Production in the Context of Energy Security—A Systematic Literature Review. Energies 2025, 18, 2410. [Google Scholar] [CrossRef]
- Patel, K.; Singh, S.K. Energy and Environmental Life Cycle Analysis of Biodiesel Production from Palm Stearin. In Recent Advances in Geomatics, Water Resources and Environmental Engineering (TRACE 2024 2024); Reddy, K.R., Nagabhatla, N., Singh, S.K., Soundharajan, B., Kumari, M., Jain, S.K., Eds.; Lecture Notes in Civil Engineering; Springer: Singapore, 2026; Volume 727, pp. 183–195. [Google Scholar] [CrossRef]
- Patel, K.; Singh, S.K. Comparative Life Cycle Assessment of Palm Oil-, UCO-, and Microalgae-Based Biodiesel Production. In Advances in Clean Energy Technologies (ICET 2023); Dwivedi, G., Verma, P., Shende, V., Eds.; Springer Proceedings in Energy; Springer: Singapore, 2025; pp. 311–319. [Google Scholar] [CrossRef]
- Wahyono, Y.; Hadiyanto, H.; Gheewala, S.H.; Budihardjo, M.A.; Adiansyah, J.S. Evaluating the environmental impacts of the multi-feedstock biodiesel production process in Indonesia using life cycle assessment (LCA). Energy Convers. Manag. 2022, 266, 115832. [Google Scholar] [CrossRef]
- Singh, D.; Sharma, D.; Soni, S.L.; Inda, C.S.; Sharma, S.; Sharma, P.K.; Jhalani, A. A comprehensive review of biodiesel production from waste cooking oil and its use as fuel in compression ignition engines: 3rd generation cleaner feedstock. J. Clean. Prod. 2021, 307, 127299. [Google Scholar] [CrossRef]
- Saranya, G.; Ramachandra, T.V. Life cycle assessment of biodiesel from estuarine microalgae. Energy Convers. Manag. X 2020, 8, 100065. [Google Scholar] [CrossRef]
- Liu, Y.; Xia, R.; Wang, Y.; Zhao, Y.; Ju, M. Life cycle environmental impact assessment of microalgae biodiesel derived from different processes. Ind. Crops Prod. 2025, 233, 121381. [Google Scholar] [CrossRef]
- Rafiq, A.; Morris, C.; Schudel, A.; Gheewala, S. Life Cycle Assessment of Microalgae-Based Products for Carbon Dioxide Utilization in Thailand: Biofertilizer, Fish Feed, and Biodiesel. F1000Research 2025, 13, 1503. [Google Scholar] [CrossRef]
- Schade, S.; Meier, T. Distinct microalgae species for food—Part 1: A methodological (top-down) approach for the life cycle assessment of microalgae cultivation in tubular photobioreactors. J. Appl. Phycol. 2020, 32, 2977–2995. [Google Scholar] [CrossRef]
- Wei, C.; Xu, Y.; Li, Y.; Wei, W.; Feng, Y.; Li, Z.; Xu, L.; Zhang, Y.; Liu, H.; Wang, J.; et al. Life-cycle assessment of microalgae liquid biofuel production in biofilm cultivation system via conversion technologies of transesterification, hydrothermal liquefaction and pyrolysis. J. Clean. Prod. 2024, 436, 140559. [Google Scholar] [CrossRef]
- Álvarez-González, A.; Castro, I.M.P.; Ortiz, A.; Díez-Montero, R.; Passos, F.; Garfí, M.; Uggetti, E. Environmental and economic benefits of using microalgae grown in wastewater as biofertilizer for lettuce cultivation. Bioresour. Technol. 2025, 424, 132230. [Google Scholar] [CrossRef]
- Naseema Rasheed, R.; Pourbakhtiar, A.; Mehdizadeh Allaf, M.; Baharlooeian, M.; Rafiei, N.; Alishah Aratboni, H.; Morones-Ramirez, J.R.; Winck, F.V. Microalgal co-cultivation -recent methods, trends in omic-studies, applications, and future challenges. Front. Bioeng. Biotechnol. 2023, 11, 1193424. [Google Scholar] [CrossRef] [PubMed]
- Pérez-López, P.; Rajaonison, A.; Zebian, B.; Bouallou, C.; González-Fernández, C.; Greses, S.; Gómez-Serrano, C.; Acién Fernández, F.G. Exploiting outcomes of life cycle costing to conduct coherent screening social life cycle assessments of emerging systems: A case study of microalgae biorefineries. Int. J. Life Cycle Assess. 2025, 30, 770–791. [Google Scholar] [CrossRef]
- Dammak, I.; Fersi, M.; Hachicha, R.; Abdelkafi, S. Current Insights into Growing Microalgae for Municipal Wastewater Treatment and Biomass Generation. Resources 2023, 12, 119. [Google Scholar] [CrossRef]
- Fajardo, C.; De Donato, M.; Carrasco, R.; Martínez-Rodríguez, G.; Mancera, J.M.; Fernández-Acero, F.J. Advances and challenges in genetic engineering of microalgae. Rev. Aquac. 2020, 12, 365–381. [Google Scholar] [CrossRef]
- Onn, S.M.; Koh, G.J.; Yap, W.H.; Teoh, M.L.; Low, C.F.; Goh, B.H. Recent advances in genetic engineering of microalgae: Bioengineering strategies, regulatory challenges and future perspectives. J. Appl. Phycol. 2025, 37, 247–264. [Google Scholar] [CrossRef]
- Grama, S.B.; Liu, Z.; Li, J. Emerging Trends in Genetic Engineering of Microalgae for Commercial Applications. Mar. Drugs 2022, 20, 285. [Google Scholar] [CrossRef]
- Barati, B.; Zeng, K.; Baeyens, J.; Wang, S.; Addy, M.; Gan, S.Y.; Abomohra, A.E.F. Recent progress in genetically modified microalgae for enhanced carbon dioxide sequestration. Biomass Bioenergy 2021, 145, 105927. [Google Scholar] [CrossRef]
- Kumar, V.; Sharma, N.; Jaiswal, K.K.; Vlaskin, M.S.; Nanda, M.; Tripathi, M.K.; Kumar, S. Microalgae with a truncated light-harvesting antenna to maximize photosynthetic efficiency and biomass productivity: Recent advances and current challenges. Process Biochem. 2021, 104, 83–91. [Google Scholar] [CrossRef]
- Godbole, V.; Pal, M.K.; Gautam, P. A critical perspective on the scope of interdisciplinary approaches used in fourth-generation biofuel production. Algal Res. 2021, 58, 102436. [Google Scholar] [CrossRef]
- Trovão, M.; Schüler, L.M.; Machado, A.; Bombo, G.; Navalho, S.; Barros, A.; Pereira, H.; Silva, J.; Freitas, F.; Varela, J. Random Mutagenesis as a Promising Tool for Microalgal Strain Improvement towards Industrial Production. Mar. Drugs 2022, 20, 440. [Google Scholar] [CrossRef] [PubMed]
- Xue, J.; Balamurugan, S.; Li, T.; Cai, J.-X.; Chen, T.-T.; Wang, X.; Yang, W.-D.; Li, H.-Y. Biotechnological approaches to enhance biofuel producing potential of microalgae. Fuel 2021, 302, 121169. [Google Scholar] [CrossRef]
- Sebesta, J.; Xiong, W.; Guarnieri, M.T.; Yu, J. Biocontainment of Genetically Engineered Algae. Front. Plant Sci. 2022, 13, 839446. [Google Scholar] [CrossRef]
- Rafeeq, H.; Afsheen, N.; Rafique, S.; Arshad, A.; Intisar, M.; Hussain, A.; Bilal, M.; Iqbal, H.M.N. Genetically engineered microorganisms for environmental remediation. Chemosphere 2023, 310, 136751. [Google Scholar] [CrossRef] [PubMed]
- de Castro, A.P.M.; Braga, M.Q.; Araujo, P.V.; da Gama, R.C.N.; Pereira, A.S.A.P.; Silva, T.A.; Henriques, B.S.; Lorentz, J.F.; Avelar, N.V.; Calijuri, M.L. Emerging trends in wastewater-grown microalgae: A techno-economic and environmental perspective. Biomass Bioenergy 2026, 208, 108838. [Google Scholar] [CrossRef]
- Han, W.; Jin, W.; Li, Z.; Wei, Y.; He, Z.; Chen, C.; Qin, C.; Chen, Y.; Tu, R.; Zhou, X. Cultivation of microalgae for lipid production using municipal wastewater. Process Saf. Environ. Prot. 2021, 155, 155–165. [Google Scholar] [CrossRef]
- Sharma, R.; Mishra, A.; Pant, D.; Malaviya, P. Recent advances in microalgae-based remediation of industrial and non-industrial wastewaters with simultaneous recovery of value-added products. Bioresour. Technol. 2022, 344, 126129. [Google Scholar] [CrossRef]
- Dias, R.R.; Deprá, M.C.; de Menezes, C.R.; Zepka, L.Q.; Jacob-Lopes, E. Microalgae Cultivation in Wastewater: How Realistic Is This Approach for Value-Added Product Production? Processes 2025, 13, 2052. [Google Scholar] [CrossRef]
- Olguín, E.J.; Sánchez-Galván, G.; Arias-Olguín, I.I.; Melo, F.J.; González-Portela, R.E.; Cruz, L.; De Philippis, R.; Adessi, A. Microalgae-Based Biorefineries: Challenges and Future Trends to Produce Carbohydrate Enriched Biomass, High-Added Value Products and Bioactive Compounds. Biology 2022, 11, 1146. [Google Scholar] [CrossRef] [PubMed]
- Machado, E.F.; Schneider, A.T.; Machado, R.L.S.; Dutra, D.A.; Grutzmacher, A.J.L.; Dias, R.R.; Deprá, M.C.; Zepka, L.Q.; Jacob-Lopes, E. Photobioreactor Systems and Influencing Factors for Uptake of Nutrient Resources from Industrial Wastewater by Microalgae. In Phycoremediation. Interdisciplinary Biotechnological Advances; Verma, P., Dadheech, P.K., Eds.; Springer: New York, NY, USA, 2026; pp. 193–211. [Google Scholar] [CrossRef]
- Singh, A.K.; Srivastava, R.K.; Pal, P.; Mandal, S.; Sahoo, U.K.; Prakash, A.; Sridhar, K.; Sharma, M.; Sarangi, P.K.; Inbaraj, B.S.; et al. Microalgal biorefinery as a sustainable and cost-effective platform for co-production of high-value-added products/metabolites: An insight into emerging trends, challenges, and opportunities. Biocatal. Agric. Biotechnol. 2024, 58, 103192. [Google Scholar] [CrossRef]
- Barkia, I.; Saari, N.; Manning, S.R. Microalgae for High-Value Products Towards Human Health and Nutrition. Mar. Drugs 2019, 17, 304. [Google Scholar] [CrossRef]
- Wiatrowski, M.; Klein, B.C.; Davis, R.W.; Quiroz-Arita, C.; Tan, E.C.D.; Hunt, R.W.; Davis, R.E. Techno-economic assessment for the production of algal fuels and value-added products: Opportunities for high-protein microalgae conversion. Biotechnol. Biofuels Bioprod. 2022, 15, 8. [Google Scholar] [CrossRef]
- Figueroa-Torres, G.M.; Theodoropoulos, C. Techno-economic analysis of a microalgae-based biorefinery network for biofuels and value-added products. Bioresour. Technol. Rep. 2023, 23, 101524. [Google Scholar] [CrossRef]
- Makepa, D.C.; Chihobo, C.H. Barriers to commercial deployment of biorefineries: A multi-faceted review of obstacles across the innovation chain. Heliyon 2024, 10, e32649. [Google Scholar] [CrossRef]
- Santos, B.; Freitas, F.; Sobral, A.J.F.N.; Encarnação, T. Microalgae and circular economy: Unlocking waste to resource pathways for sustainable development. Int. J. Sustain. Eng. 2025, 18, 2501488. [Google Scholar] [CrossRef]
- Wiatrowski, M.; Davis, R.; Kruger, J. Algal Biomass Conversion to Fuels via Combined Algae Processing (CAP): 2021 State of Technology and Future Research. 2021. Available online: https://www.nrel.gov/docs/fy22osti/82502.pdf (accessed on 20 December 2025).
- Gurreri, L.; Rindina, M.C.; Luciano, A.; Falqui, L.; Mancini, G. Life Cycle Assessment Based on Primary Data of an Industrial Plant for Microalgae Cultivation. Chem. Eng. Trans. 2024, 109, 493–498. [Google Scholar]
- Chen, H.; Jiang, Y.; Zhu, K.; Yang, J.; Fu, Y.; Wang, S. A Review on Industrial CO2 Capture through Microalgae Regulated by Phytohormones and Cultivation Processes. Energies 2023, 16, 897. [Google Scholar] [CrossRef]
- Li, G.; Yao, J. A Review of Algae-Based Carbon Capture, Utilization, and Storage (Algae-Based CCUS). Gases 2024, 4, 468–503. [Google Scholar] [CrossRef]
- AlSalal, A.M.; Almomani, F.; Alkanaimsh, S. Harnessing microalgae for a synergistic approach to CO2 capture, wastewater treatment, and biodiesel production: A review. J. Environ. Chem. Eng. 2025, 13, 117465. [Google Scholar] [CrossRef]
- Llamas, B.; Suárez-Rodríguez, M.C.; González-López, C.V.; Mora, P.; Acién, F.G. Techno-economic analysis of microalgae related processes for CO2 bio-fixation. Algal Res. 2021, 57, 102339. [Google Scholar] [CrossRef]
- Singh, U.; Naaz, F.; Hawkins, T.R.; Weinberg, E.; Banerjee, S.; Mroz, R.; Ohri, N.; Zhang, J.; Lee, Y.-Y.; Chen, F.; et al. Direct flue gas capture for algae cultivation and subsequent valorization: Evaluating life cycle emissions and costs. Sustain. Energy Fuels 2025, 9, 4392–4403. [Google Scholar] [CrossRef]
- Zhang, Z.; Sang, Y.; Liu, S.; Kai, T.; Song, C. Comparative techno-economic and life cycle assessment of microalgae-based CO2 capture technologies for coal-fired power plants under different carbon supply modes. Energy 2025, 336, 138588. [Google Scholar] [CrossRef]
- Ashour, M.; Mansour, A.T.; Alkhamis, Y.A.; Elshobary, M. Usage of Chlorella and diverse microalgae for CO2 capture—Towards a bioenergy revolution. Front. Bioeng. Biotechnol. 2024, 12, 1387519. [Google Scholar] [CrossRef]
- Zieliński, M.; Dębowski, M.; Kazimierowicz, J.; Świca, I. Microalgal Carbon Dioxide (CO2) Capture and Utilization from the European Union Perspective. Energies 2023, 16, 1446. [Google Scholar] [CrossRef]
- European Parliament; Council of the European Union. Directive (EU) 2023/2413 of the European Parliament and of the Council of 18 October 2023 Amending Directive (EU) 2018/2001, Regulation (EU) 2018/1999 and Directive 98/70/EC as Regards the Promotion of Energy from Renewable Sources (RED III). 2023. Available online: https://eur-lex.europa.eu/eli/dir/2023/2413/oj (accessed on 20 December 2025).
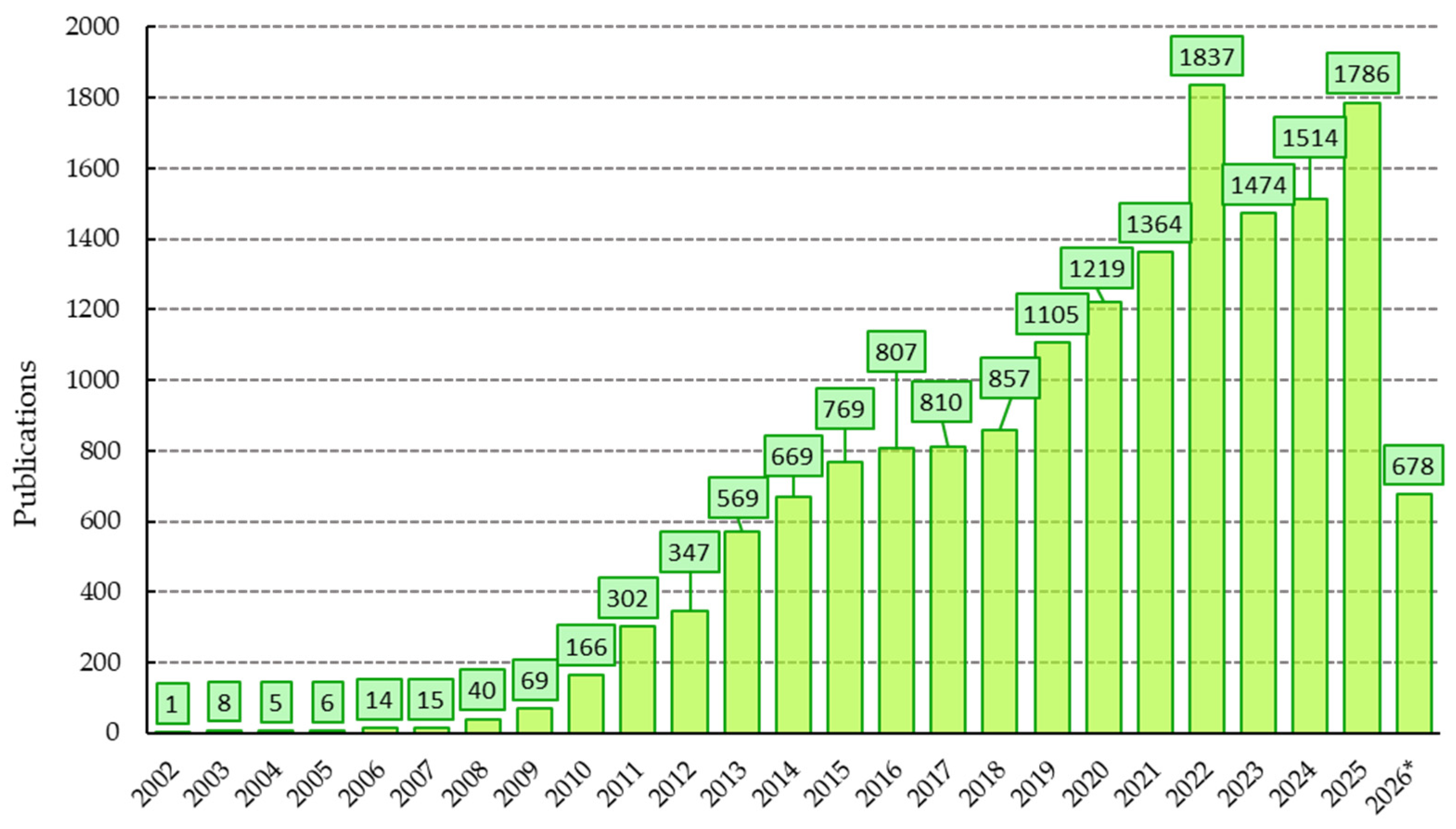

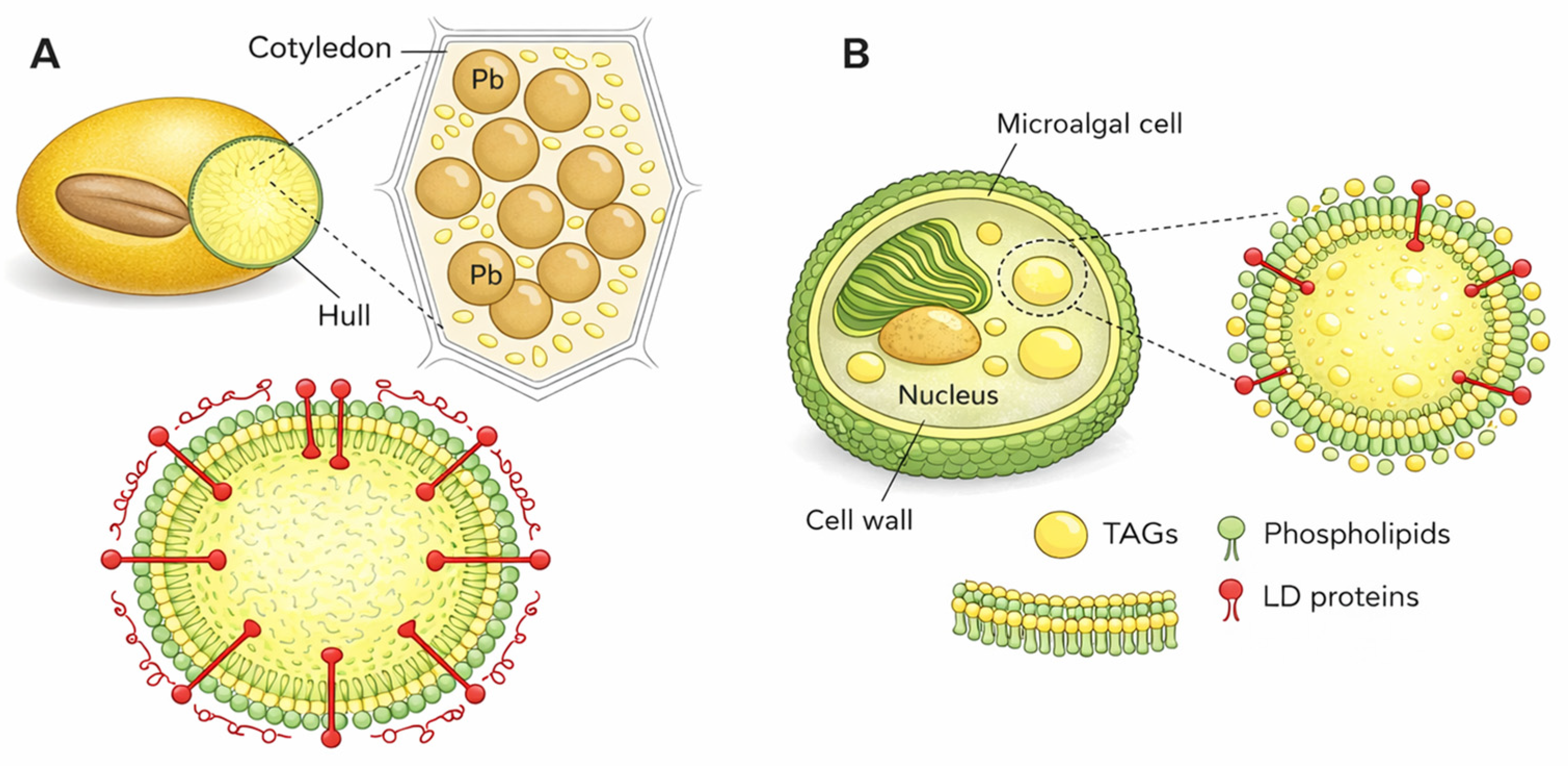
| Production System | Microalgae Species | Biomass Productivity (g m3 Day) |
|---|---|---|
| Bioreactors | Euglena gracilis | 0.09 |
| Membrane bioreactors | Chlorella vulgaris | 0.10–0.14 |
| Membrane bioreactors | Nannochloropsis sp. | 0.07–0.43 |
| Membrane photobioreactor | Arthospira platensis | 0.92 |
| Tubular photobioreactor | Porphyridium cmentum | 1.5 |
| Tubular photobioreactor | Phaeodactylum tricornutum | 1.2 |
| Tubular photobioreactor | P. tricornutum | 1.9 |
| Inclined tubular photobioreactor | Chlorella sorokiniana | 1.47 |
| Undular row tubular photobioreactor | A. platensis | 2.7 |
| Outdoor helical tubular photobioreactor | P. tricornutum | 1.4 |
| Parallel tubular photobioreactor | Haematococcus pluvialis | 0.05 |
| Bubble column photobioreactor | H. pluvialis | 0.06 |
| Tubular photobioreactor | H. pluvialis | 0.41 |
| Tubular photobioreactor | A. platensis | 0.42 |
| Flat-plate photobioreactor | A. platensis | 1.15 |
| Flat-plate photobioreactor | Nannochloropsis spp. | 0.27 |
| Flat-plate photobioreactor | Chlorella spp. | 3.8 |
| Flat-plate photobioreactor | Chlorella spp. | 3.2 |
| Column photobioreactor | Tetraselmis | 0.42 |
| Parabola photobioreactor | Chlorococcum | 0.09 |
| Dome photobioreactor | Chlorococcum | 0.1 |
| Open pond reactors | A. platensis | 0.18–0.32 |
| Method | Advantages | Disadvantages |
|---|---|---|
| Mechanical rupture |
|
|
| Chemical disruption |
|
|
| Biological disruption |
|
|
| Source | EC (wt %) | CN | SV | IV (g I2/100 g−1) | DU (wt %) | LCSF (wt %) | CFPP (°C) |
|---|---|---|---|---|---|---|---|
| Aphanothece sp. | 99.9 | 55.8 | 225.1 | 65.4 | 70.6 | 3.8 | −4.6 |
| Chlorella sp. | 99.8 | 56.7 | 217.8 | 65.0 | 74.1 | 6.7 | 4.5 |
| Dunaliella sp. | 99.8 | 52.2 | 220.8 | 83.8 | 98.0 | 2.6 | −8.4 |
| Phaeodactylum sp. | 99.9 | 53.7 | 266.1 | 58.7 | 52.7 | 1.3 | −12.3 |
| Phormidium sp. | 99.8 | 54.6 | 217.9 | 74.5 | 76.3 | 6.6 | 4.4 |
| Scenedesmus sp. | 99.8 | 56.1 | 217.5 | 68.2 | 67.8 | 11.9 | 20.8 |
| Soybean (FAME) | 96.9 | 49 | 195 | 128 | 150 | 3.0 | −5 |
| Palm (FAME) | 97.7 | 61 | 207 | 57 | 65.0 | 6.7 | −10 |
| Petroleum diesel | 0 | 45 | 0 | <10 | 0 | 0 | −12.5 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Silva Machado, R.L.; Deprá, M.C.; Dutra, D.A.; Schneider, A.T.; Machado, E.F.; Zepka, L.Q.; Jacob-Lopes, E. Why Does Microalgae Biodiesel Not Work? Processes 2026, 14, 1046. https://doi.org/10.3390/pr14071046
Silva Machado RL, Deprá MC, Dutra DA, Schneider AT, Machado EF, Zepka LQ, Jacob-Lopes E. Why Does Microalgae Biodiesel Not Work? Processes. 2026; 14(7):1046. https://doi.org/10.3390/pr14071046
Chicago/Turabian StyleSilva Machado, Richard Luan, Mariany Costa Deprá, Darissa Alves Dutra, Adriane Terezinha Schneider, Eduarda Funari Machado, Leila Queiroz Zepka, and Eduardo Jacob-Lopes. 2026. "Why Does Microalgae Biodiesel Not Work?" Processes 14, no. 7: 1046. https://doi.org/10.3390/pr14071046
APA StyleSilva Machado, R. L., Deprá, M. C., Dutra, D. A., Schneider, A. T., Machado, E. F., Zepka, L. Q., & Jacob-Lopes, E. (2026). Why Does Microalgae Biodiesel Not Work? Processes, 14(7), 1046. https://doi.org/10.3390/pr14071046











