1. Introduction
Menstrual protection methods have evolved significantly to meet women’s needs for dependable, discreet, and comfortable hygiene management. Sanitary pads, in particular, offer a variety of forms and features among contemporary disposable feminine care products. Their environmental sustainability and safety, however, continue to be serious concerns [
1]. The multilayered architecture of conventional sanitary napkins includes non-biodegradable materials such as polyethylene, polypropylene, superabsorbent polymers, and polymeric films [
2]. The widespread use of petroleum-derived polymers has resulted in a substantial carbon footprint. These materials contribute to air pollution and climate change because they remain in the environment for many hundreds of years and when disposed of release harmful pollutants including dioxins and methane [
3].
The use of disposable sanitary napkins has expanded significantly in India, as a result of increased health literacy, societal awareness, and government-led menstrual hygiene initiatives. Even if only 10% of Indian women use disposable pads, an estimated 16,180 tons of sanitary waste are produced each month, placing tremendous strain on the country’s existing waste treatment facilities [
4]. Disposable sanitary products are vital for women’s health and dignity; however, because of their poor biodegradability and inappropriate disposal methods, their environmental impact has become more apparent.
When exposed to heat, UV light, and mechanical stress, synthetic fibers such as polyester and polypropylene, which are used in traditional sanitary napkins, break down into microplastics and hinder natural degradation processes [
5]. Because they are consumed by organisms at various trophic levels, these microplastics pose serious ecological hazards, especially in aquatic habitats, where they can cause oxidative stress, physical harm, and altered metabolism [
6]. Additionally, microplastics pose a major threat to public health because they contaminate freshwater and terrestrial ecosystems, damage soil structure, impede plant growth, and enter the human food chain through food products, drinking water, and airborne exposure [
7,
8].
Recent research has focused on substituting biodegradable, plant-based materials for synthetic absorbents to address these issues. Natural fibers such as bamboo, banana fiber, corn husk, and agricultural residues have shown promising absorbent properties, lower carbon footprints, and improved end-of-life degradation profiles [
9,
10]. However, many commercially available plant-based sanitary pads emphasize biodegradability and absorbency, whereas active antimicrobial functionality—critical for controlling microbial proliferation, odor generation, and urogenital infections—remains insufficiently explored.
Water hyacinth (
Eichhornia crassipes), an invasive aquatic weed, is one of the emerging bioresources that represents both a serious environmental concern and a unique opportunity for sustainable material innovation. Due to its high biomass accumulation and rapid vegetative reproduction, water hyacinth, which is native to the Amazon Basin, has rapidly expanded throughout freshwater bodies worldwide, especially in tropical and subtropical regions [
11,
12]. Its unchecked growth creates dense floating mats that obstruct waterways, hinder navigation, reduce light penetration, and drastically lower dissolved oxygen levels, thereby disrupting aquatic ecosystems and leading to widespread fish mortality [
13,
14,
15].
Additionally, by trapping sediments and nutrients, water hyacinth infestation accelerates eutrophication, encourages mosquito reproduction, and facilitates the spread of vector-borne diseases, including dengue and malaria [
16,
17]. From an economic standpoint, infestations result in significant financial burdens due to increased river management expenses, blocked irrigation systems, decreased hydropower generation efficiency, and losses in agriculture and fisheries [
18,
19]. In India alone, millions of rupees are spent every year on the chemical control and mechanical removal of water hyacinth, with little long-term success [
20]. The necessity for sustainable utilization-based management solutions is highlighted by the fact that chemical herbicides exacerbate water contamination and endanger non-target organisms.
Despite these challenges, water hyacinth is a desirable option for value-added applications because of its cellulose-rich fiber structure, high porosity, and rapid renewability. Recent studies have demonstrated its potential in biocomposites, wastewater remediation, natural fiber reinforcement, bioenergy generation, and sustainable polymer systems. Its application has also been extended to phytoremediation for the removal of heavy metals and organic and inorganic pollutants in water bodies [
21,
22,
23]. Notably, the hydrophilic cellulose matrix and porous morphology of water hyacinth fibers enhance liquid absorption and retention, rendering them particularly suitable for absorbent hygiene products.
In this study, sanitary pads were fabricated at a laboratory scale, using processed water hyacinth sheets and finely milled water hyacinth fiber powder as the primary absorbent components. All experimental evaluations were conducted under controlled laboratory conditions to assess the functional performance, pH compatibility, and microbiological safety of the developed sanitary pads.
The principal innovation of this work lies in the dual use of water hyacinth fibers as both an absorbent material and an intrinsically antibacterial component for sanitary napkin production. Water hyacinth reportedly contains bioactive phytochemicals with antibacterial activity against common pathogenic microbes, including phenolics, flavonoids, and alkaloids [
24,
25]. These characteristics have been effectively utilized in water purification systems, wound dressings, and antimicrobial mats, demonstrating inhibitory activity against Escherichia coli, Staphylococcus aureus, and other clinically significant bacteria [
26,
27]. By incorporating these naturally occurring antimicrobial fibers into sanitary napkins, user safety can be improved without the need for synthetic chemical additives as bacterial growth, odor development, and the risk of infections during extended use may be reduced.
Furthermore, transforming an environmental liability into a high-value sustainable product through the valorization of water hyacinth for menstrual hygiene applications aligns with the principles of the circular economy and supports the management of invasive species. This approach integrates sustainability, functional antimicrobial performance, and invasive biomass utilization into a single hygiene product platform, in contrast to current biodegradable sanitary pads that primarily emphasize environmental compatibility. Sanitary napkins made from water hyacinth fibers are therefore anticipated to exhibit improved absorbency, biodegradability, and antibacterial activity, providing a practical and environmentally sustainable option for menstrual hygiene management.
3. Results and Discussion
Different materials are used in the manufacture of sanitary napkins to achieve optimal absorbency and comfort. Conventional materials such as cotton are valued for their soft texture and high absorbent capacity, making them comfortable for prolonged wear. Biodegradable materials, including water hyacinth paper and fibers, have recently emerged as environmentally friendly alternatives to plastic absorbent cores. Combining cotton with these biodegradable materials enables the production of high-quality sanitary napkins that provide reliable menstrual protection. This combination offers period hygiene products that are safe, efficient, and environmentally sustainable.
The biodegradable sanitary pad developed in this study was designed with a multilayer configuration consisting of a top sheet, an absorbent core, and a barrier layer. The top sheet functions to rapidly absorb menstrual fluid and transfer it to the underlying layers. Commercial sanitary pads predominantly use polypropylene-based nonwoven fabrics; however, such materials are non-biodegradable and environmentally persistent. In this study, a water hyacinth fiber-based nonwoven material was used as a sustainable alternative because of its softness, breathability, skin compatibility, and biodegradability [
4,
28].
The petiole region of
Eichhornia crassipes was chosen as the primary raw material for the production of sanitary pads because of its favorable fiber structure and relatively higher cellulose content compared with leaves and roots [
29,
30]. Since each plant normally produces 7–12 petioles, 5–6 fully developed petioles were consistently selected to ensure consistency and repeatability of the absorbent material (
Figure 3a). Mature petioles were selected to optimize fiber strength, hydrophilicity, and processing consistency, all of which are critical for hygienic applications.
After being carefully cleaned to remove attached contaminants, the collected petioles were cut into sections approximately 1–3 inches long (
Figure 3b). As documented for other natural lignocellulosic fibers, this size reduction improved chemical penetration during pulping and enabled efficient fiber liberation during subsequent processing steps [
3,
30]. Chemical pulping was performed using sodium hydroxide (2–5%
w/
v) for 60 min at 90 °C. The alkali treatment effectively disrupted the lignin and hemicellulose matrix, producing cellulose-rich fibers suitable for absorbent applications [
11,
31]. After cooling, repeated washing followed by pH neutralization with diluted acetic acid ensured the removal of residual alkali and restored a near-neutral pH, which is essential for material safety in menstrual hygiene products (
Figure 3c).
To create a uniform pulp, the fibers were mechanically ground after neutralization (
Figure 4a). The pulp’s soft texture and uniform dispersion enabled effective sheet formation without the addition of bleaching agents, binders, or synthetic additives. Naturally colored paper sheets were produced through handsheet formation and subsequently dried in sunlight (
Figure 4b,c). The high cellulose content of water hyacinth petioles provided the sheets with sufficient mechanical integrity and absorbency, indicating their suitability as biodegradable absorbent components [
19,
20,
21].
To improve absorbent performance, alkali-treated water hyacinth fibers were further processed into a fine powder in addition to sheet formation (
Figure 4d). The enlarged surface area and relatively short fiber length of the powder enhanced capillary action and packing density, thereby increasing the liquid absorption capacity. Previous studies have shown that alkali-treated water hyacinth fibers exhibit absorption capacities ranging from 5 to 11 times their dry weight, indicating their effectiveness in absorbent hygiene applications [
11,
46].
The multilayer design of the biodegradable sanitary pad created in this study included a top sheet, an absorbent core, and a barrier layer. Water hyacinth fiber-based nonwoven material, a sustainable substitute for conventional polypropylene nonwovens, was used to fabricate the top sheet. The softness, breathability, skin compatibility, and biodegradability of this layer addressed the environmental and comfort concerns associated with synthetic polymers [
4,
28]. The absorbent core consisted of cotton (approximately 8 g), fiber powder, and water hyacinth paper. The addition of water hyacinth fibers substantially improved fluid uptake, retention capacity, and antibacterial performance because of their hydrophilic cellulose matrix and intrinsic bioactive properties, whereas cotton provided softness and baseline absorbency [
3,
11,
25,
30].
In place of traditional polyethylene or polyurethane films, a biodegradable cornstarch-based sheet was used as the barrier layer to prevent leakage. Canvas paper was incorporated as a supporting layer to enhance structural stability while maintaining overall biodegradability [
47]. After aligning the top sheet, absorbent core, and barrier layer, the three layers were assembled through localized heat sealing at the margins. This assembly technique maintained the eco-friendly characteristics of the product while eliminating the need for synthetic adhesives [
9].
Figure 5 illustrates the completed biodegradable sanitary napkin.
Overall, the findings show that water hyacinth-based materials can successfully replace synthetic components in sanitary napkins while still providing sufficient absorbency, comfort, and hygienic performance. By reducing dependence on petroleum-based polymers and increasing the value of invasive biomass, this strategy offers dual sustainability benefits.
3.1. Absorbance Examination
Absorbency is a critical feature of menstrual pads and is essential for ensuring comfort and security during use. Effective absorbency ensures that menstrual flow is managed quickly and efficiently. To measure this property, an absorbency test is performed. During this test, a specified volume of aqueous coloring solution (dye) is applied to the surface of the pad, and the time required for the fluid to be completely absorbed is recorded [
31]. This procedure evaluates the pad’s ability to manage menstrual flow promptly and effectively.
In the present study, absorbency tests were performed on different types of menstrual pads, including water hyacinth-based pads, cloth (cotton) pads, and commercial sanitary pads (
Figure 6). The comparative results are presented in
Table 1.
The data indicate that both the cotton and water hyacinth pads absorbed a larger volume of liquid compared with the commercial product, which absorbed only 71 mL. In contrast, the cotton and water hyacinth pads each absorbed 111 mL under the same test conditions. These findings suggest that the absorbent capacity of these natural materials may exceed that of some commercial sanitary pads, likely because of the high water affinity of cellulose-rich fibers [
31].
The absorbent performance of natural fibers has been well documented in previous studies. For example, cotton fibers have a hydrophilic structure and high cellulose content, enabling them to absorb 24–27 times their own weight in water, although this capacity can vary with weave and fabric form [
24,
48]. Similarly, studies on natural and eco-friendly menstrual pad materials have shown that plant-derived fibers such as bamboo can outperform conventional cotton in fluid uptake, with absorbency several times greater than that of commercial pads in controlled tests [
49].
Earlier research specifically addressing water hyacinth fiber highlights its excellent moisture absorbency. Water hyacinth (
Eichhornia crassipes) is a cellulose-rich aquatic plant whose fibers exhibit significant water uptake capability, often comparable to or exceeding that of untreated cotton fibers after appropriate processing [
50]. This high absorbency performance is attributed to the hydrophilic nature of cellulose chains and the porous structure of the fibers, which facilitates fluid penetration and retention. In this context, the absorbent properties of water hyacinth fibers have attracted attention as sustainable alternatives for absorbent core materials in hygiene products. The current experimental results align with these observations, showing that the combination of water hyacinth fiber with cotton not only preserves but may also enhance liquid retention capacity relative to conventional commercial pads. This effect likely arises from the complementary absorbent characteristics of cotton and water hyacinth fibers, with cotton providing established comfort and wicking properties and water hyacinth contributing additional hydrophilic sites and structural porosity.
Additionally, previous studies emphasize the importance of material selection and treatment in determining overall absorbency performance. For instance, surface treatments, fiber processing methods, and composite configurations play a significant role in optimizing water uptake and retention in natural fibers, which directly affect their suitability as absorbent media in sanitary products [
51].
These findings and the supporting literature indicate that natural plant fibers such as cotton and water hyacinth can provide competitive or improved absorbency compared with conventional commercial products while also offering ecological benefits such as biodegradability and sustainability.
3.2. pH Assessment: Considerations for Sanitary Pads and Implications for Vaginal Health
The pH of sanitary pads is a critical parameter influencing vaginal health, comfort, and microbiological balance. The healthy human vagina maintains a naturally acidic environment, typically within a pH range of 3.5–4.5. This condition is primarily due to the dominance of
Lactobacillus species, which produce lactic acid as a metabolic byproduct. This acidic environment plays a protective role by inhibiting the growth of pathogenic microorganisms that thrive in neutral or alkaline conditions, thereby reducing the risk of infections such as bacterial vaginosis and vulvovaginal candidiasis [
52,
53].
Sanitary pads come into prolonged contact with the vulvar region during menstruation, and the materials used in their construction can influence the local pH microenvironment. Prolonged exposure to products with unsuitable pH levels may disrupt the natural acidic barrier, potentially leading to irritation, itching, dermatitis, or increased susceptibility to microbial infections [
54]. Previous studies have shown that sanitary products manufactured from natural fibers such as cotton, cellulose, or other plant-based materials generally exhibit pH values closer to physiological compatibility compared with pads containing predominantly synthetic polymers such as polypropylene or polyethylene [
7,
11]. This material-dependent pH variation directly influences wearer comfort and overall genital health.
In India, the safety and quality of sanitary napkins are regulated by IS 5405:2019 [
55], which specifies that the pH of aqueous extracts of sanitary napkins should fall within the range of 6.0–8.5. This range is considered acceptable for minimizing toxicological risks, including skin irritation, allergic reactions, and microbial growth during extended use [
34]. Although this standard pH range is higher than the native vaginal pH, it is intended to ensure dermal safety at the vulvar interface rather than directly replicating the vaginal environment.
In the present study, the pH values obtained for the aqueous extracts of the developed water hyacinth-based sanitary pad are summarized in
Table 2.
Statistical analysis revealed minimal variation among triplicate measurements (SD < 0.05), indicating good reproducibility and uniformity of the pad material. The measured pH value of 6.87 lies well within the permissible range specified by IS 5405:1980 [
32], confirming dermal safety for sanitary napkin applications.
The developed water hyacinth-based sanitary pad is classified as compliant because the measured pH value (6.87 ± 0.04) lies well within the acceptable range of 6.0–8.5 specified by IS 5405:1980 for sanitary napkins. This range is prescribed to ensure dermal safety and minimize the risk of skin irritation, allergic reactions, or adverse dermatological effects during prolonged contact with the vulvar area. The low standard deviation indicates consistent pH values across replicates, confirming material uniformity. Since the pH does not exceed the upper safety limit or fall below the minimum specified value, the product meets the regulatory requirements and is considered safe and suitable for hygienic use.
Although the physiological vaginal pH is acidic (3.5–4.5), sanitary pads are primarily intended for contact with the vulvar skin, where a near-neutral pH is desirable to prevent irritation and dermatitis. Pads with an excessively alkaline pH have been associated with skin barrier disruption and increased susceptibility to microbial infections [
52,
54]. The observed pH value suggests that the lignocellulosic composition of
Eichhornia crassipes fibers helps maintains a skin-compatible microenvironment.
Recent investigations into biodegradable and reusable sanitary products have highlighted the importance of optimizing pH alongside absorbency and mechanical performance. For instance, a reusable cloth pad fabricated from corn husk fibers exhibited a pH value of 7.45, which lies toward the upper limit of the recommended IS standard but remains within the permissible safety range [
56]. Similarly, studies evaluating natural fiber–based sanitary napkins have reported that plant-derived cellulose materials tend to maintain neutral to mildly acidic pH levels, thereby reducing the likelihood of irritation during prolonged wear [
19,
26]. The pH result obtained in this study is comparable to those reported for reusable cloth pads (pH 7.2–7.5) and remains significantly below the upper safety threshold (8.5), suggesting a reduced risk of irritation during prolonged menstrual use.
In general, preserving vulvovaginal health requires sanitary pad materials to be pH-compatible, especially when biodegradable and plant-based substitutes are developed as sustainable alternatives to petroleum-derived polymers. The results of this study suggest that water hyacinth-based sanitary pads meet established safety criteria and offer a promising eco-friendly alternative without compromising pH-related user safety.
3.3. Bioburden Assessment
Bioburden testing is a critical microbiological quality control parameter for sanitary products, as it evaluates the total viable microbial load present on a product prior to use. Excessive microbial contamination may lead to skin irritation, allergic reactions, and an increased risk of urogenital infections, particularly during prolonged contact with sensitive skin areas. Therefore, maintaining a low microbial bioburden is essential to ensure product safety, hygienic integrity, and user comfort [
57,
58].
According to IS 5405:2019 [
55] and ISO 11737-1:2018 [
34] (Amd.1:2021), sanitary napkins are required to meet stringent microbiological limits. A product is considered microbiologically safe when the total bacterial count does not exceed 1000 CFU/g and the total yeast and mold count remains below 10 CFU/g [
34]. These limits are established to minimize the risk of microbial growth during storage and use, thereby ensuring consumer safety.
The bioburden results obtained for the developed water hyacinth-based sanitary pad are presented in
Table 3. The total bacterial count was found to be 360 CFU/g, while the total yeast and mold count was <1 CFU/g, both of which fall well within the permissible limits specified by the standards. Therefore, the sample complied with the established bioburden requirements.
The total aerobic bacterial load was 360 CFU/g, which is significantly lower than the maximum permissible limit of 1000 CFU/g. Yeast and mold counts were below the detectable limit (<1 CFU/g). One-way ANOVA confirmed no statistically significant variation among replicate samples (p > 0.05), demonstrating microbiological consistency of the fabricated pads.
The developed water hyacinth-based sanitary pad meets the bioburden requirements because the observed microbial counts are well below the maximum permissible limits specified by the relevant standards. The total bacterial count of 360 ± 22 CFU/g is substantially lower than the allowable limit of ≤1000 CFU/g prescribed under IS 5405:2019 (Clause 7.3.1), indicating effective hygienic processing and minimal microbial contamination. Similarly, the total yeast and mold count was below the detectable limit (<1 CFU/g), which is far lower than the specified maximum of ≤10 CFU/g as defined in ISO 11737-1:2018 (Amd.1:2021) [
34]. These results confirm that the product meets microbiological safety criteria, poses minimal risk of infection or irritation, and is suitable for safe hygienic use, thereby justifying the Pass compliance status.
The microbiological assessment is further illustrated in
Figure 7 and
Figure 8, depicting the total bacterial count and total yeast and mold count, respectively. In both cases, the developed sanitary pad sample showed negligible microbial growth compared with the positive control, while the results were comparable to those of the negative control, confirming satisfactory microbiological quality.
Low bioburden levels are particularly important for maintaining vaginal and perineal health, as microbial imbalance in sanitary materials may contribute to infections and discomfort [
54]. The results of the present study demonstrate that the water hyacinth sanitary pad is microbiologically safe and suitable for hygienic use.
Comparative studies on natural fiber-based sanitary napkins have reported higher microbial loads in certain formulations. For instance, sanitary pads developed using 50% corn husk fiber exhibited fungal bioburden levels of approximately 400 CFU/mL and bacterial loads of 510 CFU/mL, indicating relatively higher microbial presence [
59]. Similarly, pads incorporated with neem extract showed a markedly elevated bacterial count of 12 × 10
3 CFU/mL, possibly due to processing- or storage-related factors despite neem’s known antimicrobial properties [
57]. In contrast, the water hyacinth-based pad developed in this study demonstrated substantially lower microbial counts, highlighting its superior microbiological safety profile.
Overall, the low bacterial and fungal counts observed in the present investigation confirm that the developed sanitary pad meets international and national microbiological safety standards, thereby ensuring reduced infection risk and improved user safety.
3.4. Skin Irritation Assessment
Skin irritation and microbiological safety are critical parameters in evaluating sanitary napkins, as prolonged skin contact under moist conditions can promote microbial growth and lead to infections. Skin irritation tests are routinely performed to assess the presence of pathogenic microorganisms that may compromise dermal and vaginal health. Among these,
Staphylococcus aureus is of particular concern because of its role in skin and soft tissue infections, bacteremia, endocarditis, and toxin-mediated diseases such as toxic shock syndrome (TSS) [
54]. This bacterium is also frequently associated with irritation, inflammation, and secondary infections in users of contaminated hygiene products.
Previous studies have established that
S. aureus colonization in menstrual hygiene products can disrupt the normal skin microflora and increase susceptibility to infections, particularly among adolescent and reproductive-age women [
60]. Consequently, regulatory standards emphasize the complete absence of specified pathogens in sanitary products. According to ISO 11737-1:2018 (Amd.1:2021) and IS 5405:2019 [
34,
40], sanitary napkins must be free from
Staphylococcus aureus to ensure consumer safety and hygienic acceptability.
In the present study, the developed water hyacinth-based sanitary pads were evaluated for the presence of
Staphylococcus aureus using standard microbiological test methods (
Table 4). The results demonstrated the complete absence of
S. aureus in the tested samples, indicating excellent microbiological safety. The sample successfully met the specified requirement of “absence per gram,” thereby fulfilling the skin irritation and pathogen safety criteria.
No detectable growth of Staphylococcus aureus was observed in the developed sanitary pad sample. The absence of this pathogen satisfies mandatory regulatory requirements and confirms excellent microbiological safety.
The developed water hyacinth-based sanitary pad is rated as Pass for pathogen safety because Staphylococcus aureus was not detected in the sample. According to IS 5405:2019 and ISO 11737-1:2018 (Amd.1:2021), sanitary napkins must be completely free from specified pathogenic microorganisms, including Staphylococcus aureus, to ensure user safety. The absence of this pathogen per gram of the product indicates that the sanitary pad does not pose a risk of skin irritation, infection, or toxin-mediated conditions such as TSS. Therefore, the product fully complies with the regulatory microbiological safety requirements, thereby justifying the Pass compliance status.
These findings were further confirmed by
Figure 9, showing no detectable microbial growth in the developed sanitary pad sample compared with the positive and negative controls. The absence of
S. aureus strongly suggests that the water hyacinth-based material does not promote pathogenic bacterial colonization and is suitable for prolonged skin contact.
The use of natural and sustainable fibers in hygiene products has gained increasing attention because of their favorable antimicrobial properties, breathability, and reduced risk of irritation [
26]. Several studies have reported similar outcomes in sanitary pads fabricated using plant-based fibers such as corn husk, banana fiber, and other lignocellulosic materials, where no
Staphylococcus aureus growth was detected [
4,
39]. In another study, sanitary pads incorporating 50% corn husk fibers were also found to be free from
S. aureus, supporting the antimicrobial suitability of agro-waste-derived fibers [
61,
62].
Overall, the absence of Staphylococcus aureus in the developed water hyacinth sanitary pads highlights their potential as a safe, hygienic, and eco-friendly alternative to conventional synthetic products. These findings reinforce the suitability of water hyacinth fibers for sustainable menstrual hygiene applications, with minimal risk of skin irritation or bacterial infection.
3.5. Characterization of Water Hyacinth Fibrous Polymer
3.5.1. SEM Analysis
The surface morphology of water hyacinth fibers before and after pretreatment was examined using SEM. In the untreated fiber sample, the surface features are not clearly distinguishable in the SEM micrograph (
Figure 10a). The arrangement of individual fibers is also unclear, as the fiber surfaces and inter-fiber spaces are largely covered by components such as lignin, pectin, hemicellulose, and other soluble substances. In contrast, SEM analysis of the treated water hyacinth fibers agrees with observations reported in earlier studies [
63]. As shown in
Figure 10b, the fiber surface becomes noticeably rougher after treatment, which can be attributed to the removal of surface-coating components by the NaOH solution.
The micrographs indicate that impurities were successfully removed after treatment. The results of chemical composition analysis further demonstrate that the percentages of lignin and hemicellulose decreased following treatment. The removal of the lignin layer results in a rougher fiber surface, which may enhance inter-fiber interactions [
63].
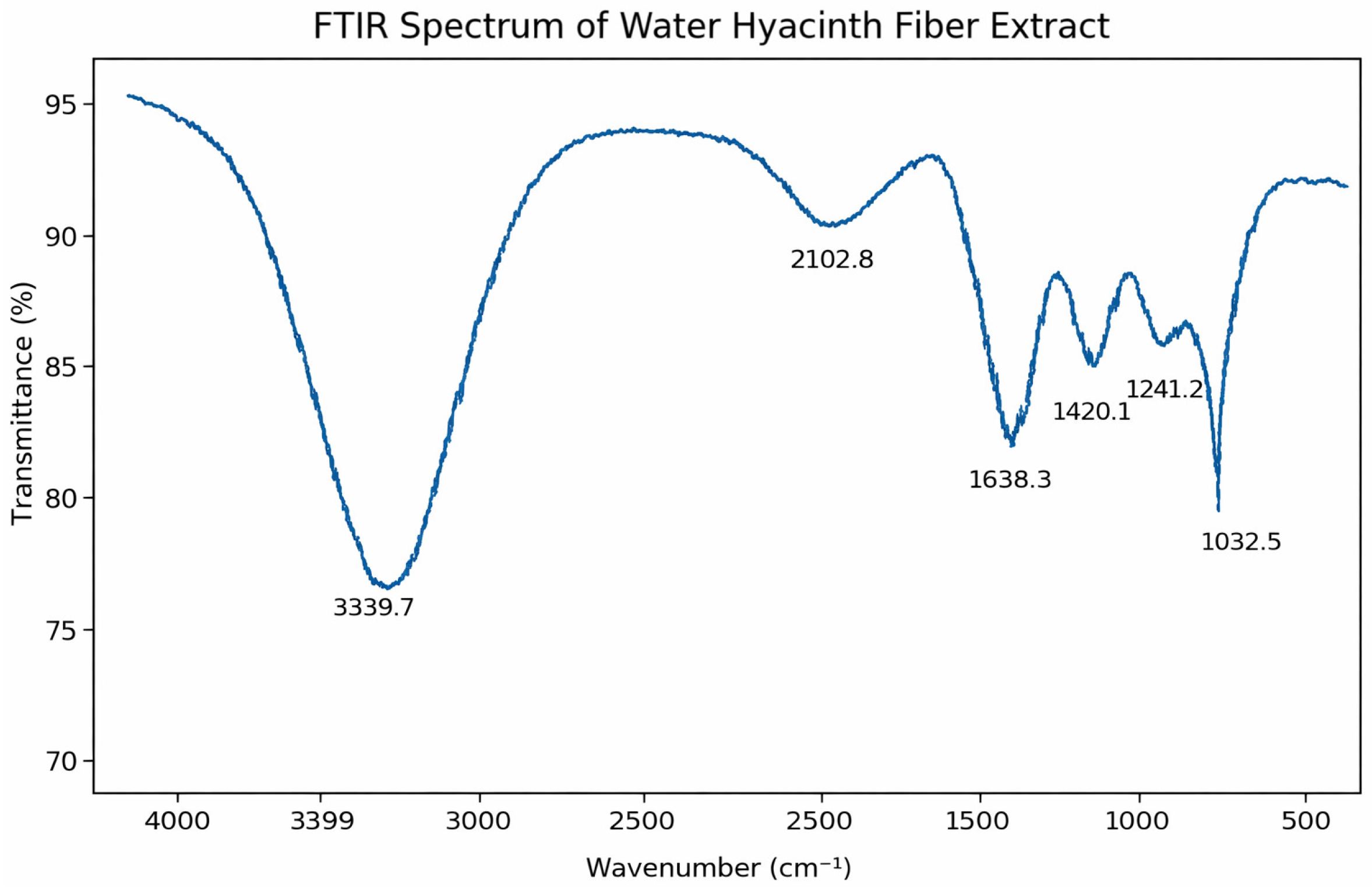
3.5.2. FTIR Spectroscopy
To determine the functional groups, present and evaluate how effectively bleaching removed non-cellulosic components such as hemicellulose and lignin, the FTIR spectrum of bleached water hyacinth cellulose was examined (
Figure 11).
O–H stretching vibrations of hydroxyl groups, which are typical of cellulose and arise from significant intra- and intermolecular hydrogen bonding, are responsible for the broad and strong absorption band observed at 3339.7 cm
−1 [
64].
Polysaccharide-rich materials frequently exhibit overtone or combination vibrations, which are represented by the weak band at 2102.8 cm
−1. Aromatic skeletal vibrations associated with residual lignin structures and/or H–O–H bending vibrations of adsorbed moisture are attributed to the absorption peak at 1638.3 cm [
65].
A clearly defined cellulosic backbone is indicated by the band at 1420.1 cm
−1, which is attributed to CH
2 bending vibrations. The signal detected at 1241.2 cm
−1 indicates trace levels of lignin or hemicellulose and is associated with C–O stretching vibrations. The prominent band at 1032.5 cm
−1 corresponds to C–O–C stretching vibrations of polysaccharides, a characteristic feature of cellulose [
66].
The FTIR results confirm the lignocellulosic nature of the water hyacinth fibers and indicate that bleaching successfully increased the cellulose content while substantially reducing hemicellulose and lignin impurities, thereby preparing the material for use in biodegradable and sustainable products.
3.5.3. X-Ray Diffraction Analysis
CrI% was calculated from the XRD spectra, as shown in
Figure 12. Both samples exhibited distinctive diffraction peaks indicative of the crystalline structure of cellulose I. With respect to the (200) and (110) crystallographic planes of native cellulose, the untreated water hyacinth showed a prominent diffraction peak centered around 2θ = 22° and a less intense peak near 2θ ≈ 16°. However, the peaks appeared broader and less intense, indicating the presence of amorphous components such as hemicellulose and lignin within the lignocellulosic matrix.
In contrast, the cellulose isolated from water hyacinth showed stronger and sharper diffraction peaks at similar 2θ positions, indicating that crystalline cellulose domains were enriched following chemical treatment. In the extracted cellulose, CrI increased from 53.21% in raw water hyacinth to 62.56%. This increase in crystallinity is attributed to the removal of amorphous components, particularly hemicellulose and lignin, during alkali and bleaching treatments. Alkali treatment breaks ester bonds and solubilizes non-cellulosic fractions, thereby improving the ordering of cellulose chains. Similar increases in crystallinity following chemical purification of lignocellulosic biomass have been extensively documented in the literature [
67,
68,
69].
The crystallinity index (CrI%) was calculated from the XRD patterns using the Segal method, where the maximum intensity of the crystalline peak at 2θ ≈ 22° (I002) and the amorphous intensity at 2θ ≈ 18° (Iam) were used. Both samples exhibited characteristic diffraction peaks corresponding to cellulose I, with peaks near 16° and 22° representing the (110) and (200) planes. The untreated water hyacinth showed broader and less intense peaks due to the presence of amorphous components such as hemicellulose and lignin. After chemical treatment, the extracted cellulose displayed sharper diffraction peaks, indicating improved crystalline ordering. Consequently, the crystallinity index increased from 53.21% to 62.56%, confirming the removal of amorphous constituents during alkali and bleaching treatments.
Stronger intermolecular hydrogen bonding and improved structural order within the cellulose microfibrils are suggested by the increased crystallinity of the isolated cellulose. Increased crystallinity is typically associated with improved mechanical strength, enhanced thermal stability, and reduced water solubility in biodegradable absorbent materials [
70]. The CrI value of 62.56% obtained in this study is consistent with previously reported crystallinity indices for cellulose derived from aquatic and agricultural biomass sources, which often range between 55% and 70% depending on extraction conditions [
68,
71].
The relatively lower crystallinity index of raw water hyacinth (53.21%) reflects the composite nature of the lignocellulosic structure, in which amorphous lignin and hemicellulose hinder the regular packing of cellulose chains. The observed increase in peak sharpness and intensity after treatment confirms the effectiveness of the extraction protocol in isolating cellulose-rich fractions suitable for biodegradable product development.
3.6. Biodegradability Test
The biodegradation profile of the sanitary pad made from water hyacinth was determined over a 60-day period. The results indicated that degradation occurred progressively over time, with clear visual evidence of structural breakdown caused by natural environmental factors (
Figure 13 and
Figure 14).
The data revealed that by Day 10 of the study, approximately 15% degradation had already occurred, suggesting rapid microbial colonization and enzymatic activity on the hydrophilic polysaccharide matrix of water hyacinth. During this phase, cellulose and hemicellulose were degraded as a result of microbial colonization and enzymatic activity, thereby significantly reducing their contribution to the structural integrity of the sanitary pad [
72,
73]. Between Days 20 and 30, the degree of degradation increased significantly to between 35% and 55%. This increase corresponds to accelerated polymer breakdown caused by an increase in the surface area and porosity of the fiber matrix, which enhances microbial access to the material and improves water retention. Similar patterns have been reported in previous studies examining the degradation of lignocellulosic fibrous waste materials used in biodegradable absorbent products [
60,
74].
The degradation percentages of the water hyacinth-based pad increased steadily over 40 days, reaching nearly 70% degradation at the end of this period and approximately 95% degradation after a total of 60 days. This result reflects the predominance of cellulose and hemicellulose, which are compostable components, over lignin in the pretreated water hyacinth fiber and explains the high biodegradation observed compared with other studies on green composites and natural fiber-based hygiene products that reported over 80% biodegradation within 2–3 months [
72,
75]. Commercial sanitary pads commonly available in the market do not readily biodegrade in the environment and are known to persist in landfills for hundreds of years, with only minimal degradation (<15% after several months) [
60]. The significantly faster biodegradation rate of the water hyacinth-based product demonstrates its superior environmental performance compared with conventional products and highlights its suitability as a sustainable alternative sanitary pad. In addition, as an aquatic invasive species, the use of Eichhornia crassipes provides an environmental benefit by converting problematic biomass into an eco-friendly value-added product. These advantages align with the principles of sustainable waste management strategies and the circular bioeconomy model and confirm that water hyacinth fibers have strong potential as a raw material for manufacturing biodegradable menstrual products [
72,
74].
3.7. Statistical Study
Statistical evaluation (mean ± SD; one-way ANOVA, p < 0.05) confirmed that the developed water hyacinth-based sanitary pad demonstrated skin-compatible pH, very low bioburden levels, complete absence of pathogenic bacteria, and compliance with IS and ISO safety standards.
These findings collectively establish the hygienic safety, dermal compatibility, and suitability of Eichhornia crassipes fibers for sustainable menstrual hygiene applications. The results strongly support the potential of water hyacinth-based products as an eco-friendly alternative to petroleum-derived sanitary products without compromising user safety.
4. Conclusions
This study demonstrates the strong potential of water hyacinth (Eichhornia crassipes) fibers as a sustainable, biodegradable, and environmentally responsible alternative material for sanitary napkin core development. The findings suggest that the use of natural lignocellulosic fibers in menstrual hygiene products can help alleviate user safety concerns and reduce the growing environmental burden associated with traditional sanitary pads. Commercial sanitary pads are primarily composed of non-biodegradable materials such as polyethylene and polymeric films. Large quantities of these products contribute to solid waste accumulation, microplastic pollution, and hazardous by-products during disposal, whereas water hyacinth fibers, as a renewable natural material, could significantly reduce the overall environmental impact of menstrual hygiene products.
Comprehensive material characterization demonstrated that water hyacinth fibers are suitable for use in absorbent hygiene products. Alkali treatment of the fibers, as observed through SEM analysis, resulted in the removal of surface contaminants (i.e., lignin, hemicellulose, and pectin) and produced a rougher and more exposed fiber surface. These morphological changes improved fiber–fiber bonding and liquid absorption capacity, which are essential properties for hygienic performance. The removal of non-cellulosic components was further confirmed by FTIR spectroscopy through the identification of characteristic cellulose peaks, indicating that chemical treatment increased the cellulose content of the extracted fibers. The crystallinity index of the raw and treated fibers was determined using XRD, increasing from 53.21% in raw fibers to 62.56% in extracted cellulose. This increase indicates the improved structural ordering, stronger intermolecular hydrogen bonding, and enhanced mechanical strength and thermal stability of the material—properties that are desirable for biodegradable absorbent applications.
Performance evaluation of the handmade sanitary napkin indicated that combining cotton with water hyacinth fibers improved liquid absorption and distribution, thereby enhancing comfort and protection. The measured pH value of 6.87 falls within the acceptable limit specified by the Indian Standard IS 5405:1980, indicating good skin compatibility and a low risk of irritation or disruption of the natural vulvovaginal flora. The bioburden level was low at the time of analysis and Staphylococcus aureus was absent, indicating a minimal risk of infection during use.
In the biodegradability testing, the sanitary pads demonstrated significant environmental benefits. Soil burial testing showed that approximately 70% degradation occurred after 40 days and nearly 95% degradation after 60 days, whereas traditional sanitary napkins show little or no measurable biodegradation even after several months. The biodegradability of the treated fibers was attributed to the high proportion of cellulose and hemicellulose in the material and the successful removal of lignin. Furthermore, since water hyacinth is an aquatic invasive weed, converting its biomass into useful products provides an additional environmental benefit by supporting circular bioeconomy principles and sustainable waste management.
This study has shown that sanitary napkins made from water hyacinth have strong potential as a next-generation solution that balances performance, safety, and sustainability. Future research should emphasize improving fiber processing techniques to enhance pad performance, exploring alternative constructions using advanced layering strategies or bio-based superabsorbents, and incorporating natural antimicrobial or odor-control agents into the pads. To ensure that the production of water hyacinth-based sanitary napkins can be implemented at an industrial scale while delivering environmental benefits, further scale-up studies, long-term field assessments, life-cycle assessments, and techno-economic analyses are required before commercial deployment. These findings further support the transition of the hygiene products market toward technologies that are safer, greener, and more socially responsible.