Emulsion Prepared with Auricularia polytricha (Mont.) Sacc. As a Direct Emulsifier for β-Carotene Encapsulation: Stability and Digestibility
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals and Apparatus
2.2. Preparation of the Emulsion
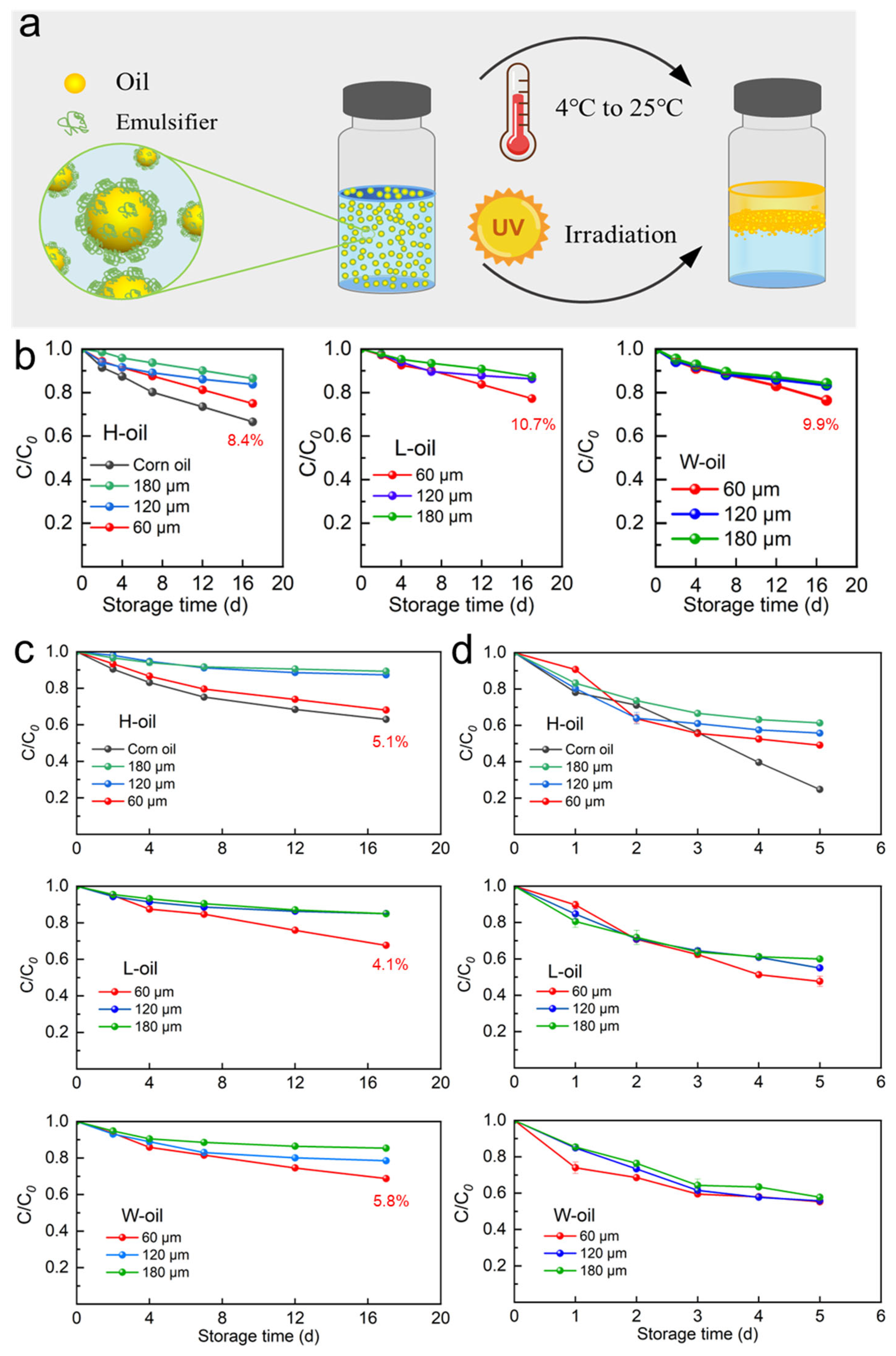
2.3. Stability Assessment of the Emulsion
2.3.1. Emulsifying Activity Index (EAI) and Emulsifying Stability Index (ESI)
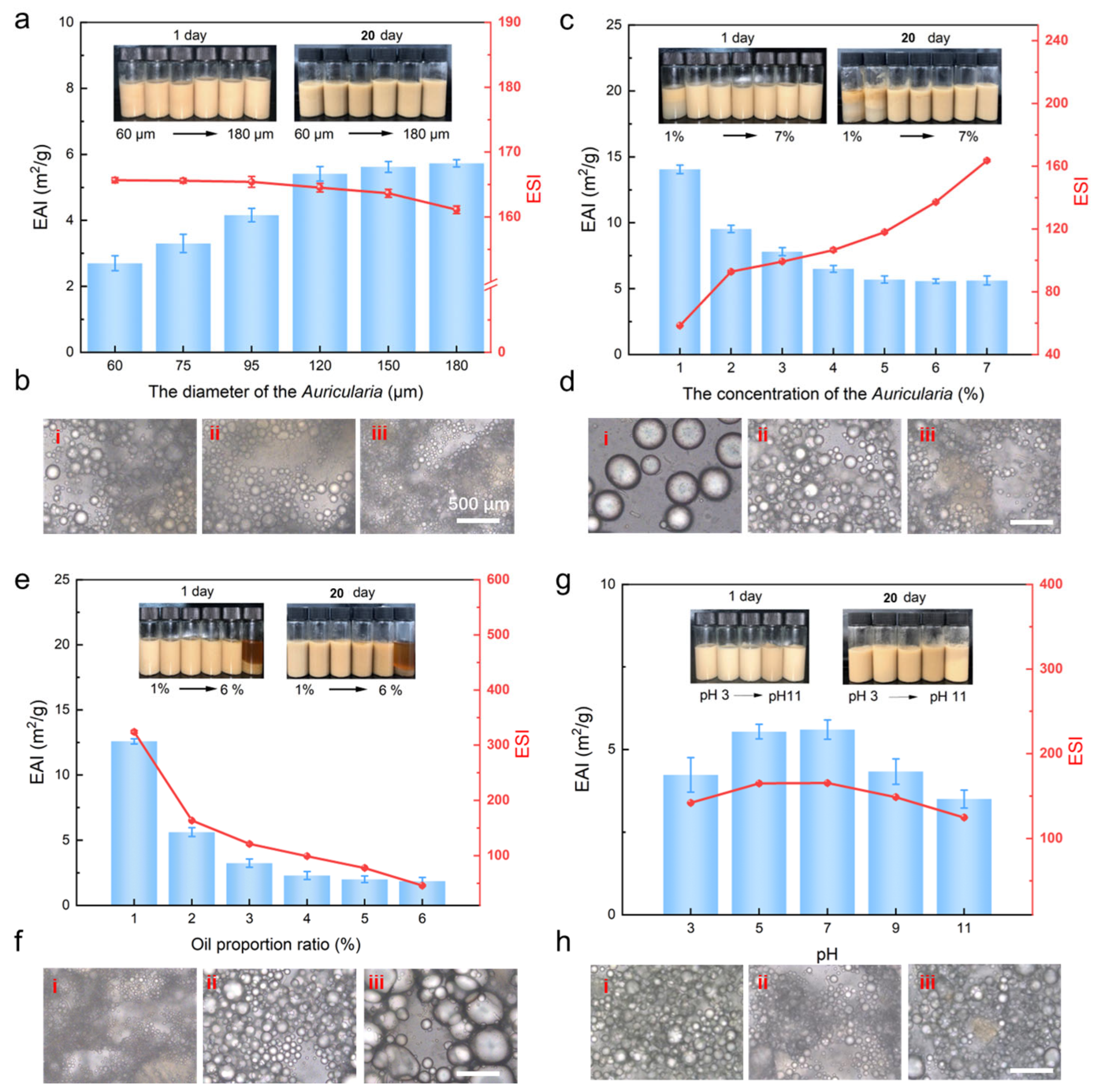
2.3.2. Effects of pH
2.4. Texture Determination of the Emulsion
2.5. Determination of the Retention Rate of β-Carotene
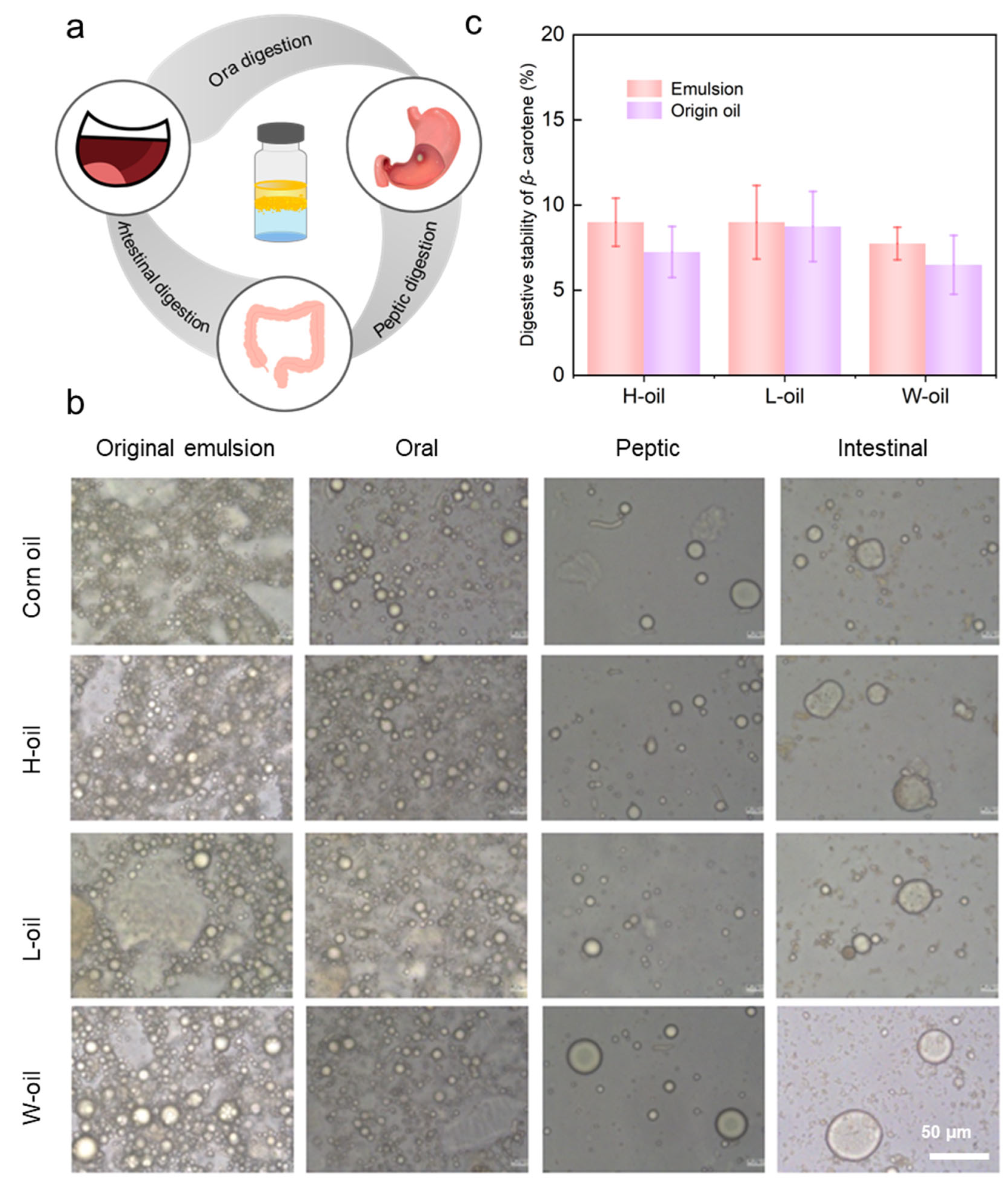
2.6. In Vitro Static Gastrointestinal Digestion Simulation
3. Results and Discussion
3.1. Optimization of Emulsion Preparation for the Particle Size
3.2. Optimization of Emulsion Preparation for the Concentration
3.3. Optimization of Emulsion Preparation for the Oil Proportion
3.4. Optimization of Emulsion Preparation for the Oil Species
3.5. Stability of β-Carotene-Loaded Emulsions
3.6. Digestive Stability of β-Carotene-Loaded Emulsions
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Shen, Z.; Ding, Y.; Xu, Y. Recent advances of protein modification strategies to enhance the freezing stability of food emulsions: Principles, applications, and prospects. Compr. Rev. Food Sci. Food Saf. 2025, 24, e70135. [Google Scholar] [CrossRef]
- Wang, S.; Wang, X.; Luo, Y.; Liang, Y. A comprehensive review of conventional and stimuli-responsive delivery systems for bioactive peptides: From food to biomedical applications. Adv. Compos. Hybrid Mater. 2024, 8, 12. [Google Scholar] [CrossRef]
- Bai, L.; Huan, S.; Rojas, O.J.; McClements, D.J. Recent Innovations in Emulsion Science and Technology for Food Applications. J. Agric. Food Chem. 2021, 69, 8944–8963. [Google Scholar] [CrossRef]
- Herminghaus, S.; Maass, C.C.; Krüger, C.; Thutupalli, S.; Goehring, L.; Bahr, C. Interfacial mechanisms in active emulsions. Soft Matter 2014, 10, 7008–7022. [Google Scholar] [CrossRef]
- Badmus, S.O.; Amusa, H.K.; Oyehan, T.A.; Saleh, T.A. Environmental risks and toxicity of surfactants: Overview of analysis, assessment, and remediation techniques. Environ. Sci. Pollut. Res. 2021, 28, 62085–62104. [Google Scholar] [CrossRef]
- Pinto, R.T.; Feu, K.S.; Dalmaschio, C.J.; Nascimento, A.; Lacerda, V., Jr. Oil Recovery Improvements Based on Pickering Emulsions Stabilized by Cellulose Nanoparticles and Their Underlying Mechanisms: A Review. ACS Omega 2025, 10, 3262–3281. [Google Scholar] [CrossRef]
- Velandia, S.F.; Marchal, P.; Sadtler, V.; Arnoux, P.; Bonn, D.; Roques-Carmes, T. Globular proteins as Pickering emulsion stabilizers: Particles or surfactants? Colloids Surf. A Physicochem. Eng. Asp. 2025, 704, 135469. [Google Scholar] [CrossRef]
- Neu, B.; Meiselman, H.J. The Role of Macromolecules in Stabilization and De-Stabilization of Biofluids. In Bioengineering in Cell and Tissue Research; Artmann, G.M., Chien, S., Eds.; Springer: Berlin/Heidelberg, Germany, 2008; pp. 393–414. [Google Scholar]
- Du, P.; Cui, B.-K.; Dai, Y.-C. High genetic diversity in wild culinary-medicinal wood ear mushroom, Auricularia polytricha (Mont.) Sacc., in tropical China revealed by ISSR analysis. Int. J. Med. Mushrooms 2011, 13, 289–298. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Wang, Y.; Gong, Z.; Yang, S.; Jia, F. Comparison of the Nutritional Properties and Transcriptome Profiling Between the Two Different Harvesting Periods of Auricularia polytricha. Front. Nutr. 2021, 8, 771757. [Google Scholar] [CrossRef] [PubMed]
- Dai, L.; Wang, Q.; Wang, L.; Huang, Q.; Hu, B. Using Commercial Bio-Functional Fungal Polysaccharides to Construct Emulsion Systems by Associating with SPI. Foods 2025, 14, 215. [Google Scholar] [CrossRef] [PubMed]
- Qayum, A.; Rashid, A.; Liang, Q.; Wu, Y.; Cheng, Y.; Kang, L.; Liu, Y.; Zhou, C.; Hussain, M.; Ren, X.; et al. Ultrasonic and homogenization: An overview of the preparation of an edible protein–polysaccharide complex emulsion. Compr. Rev. Food Sci. Food Saf. 2023, 22, 4242–4281. [Google Scholar] [CrossRef]
- Vašková, J.; Stupák, M.; Vidová Ugurbaş, M.; Židzik, J.; Mičková, H. Therapeutic Uses of Retinol and Retinoid-Related Antioxidants. Molecules 2025, 30, 2191. [Google Scholar] [CrossRef] [PubMed]
- Tufail, T.; Bader Ul Ain, H.; Noreen, S.; Ikram, A.; Arshad, M.T.; Abdullahi, M.A. Nutritional Benefits of Lycopene and Beta-Carotene: A Comprehensive Overview. Food Sci. Nutr. 2024, 12, 8715–8741. [Google Scholar] [CrossRef] [PubMed]
- Zhao, T.; He, X.; Yan, X.; Xi, H.; Li, Y.; Yang, X. Recent advances in the extraction, synthesis, biological activities, and stabilisation strategies for β-carotene: A review. Int. J. Food Sci. Technol. 2024, 59, 2136–2147. [Google Scholar] [CrossRef]
- Roy, S.; Dora, K.C.; Kumar, S.; Saklani, P.; Muthukumar, A.; Ozogul, F.; Harisankar, K.C.; Mutum, R.D.; Mary, S.C.H.; Surasani, V.K.R.; et al. A critical review on technical advances and multifaceted role of carotenoids in human health with special emphasis on metabolic diseases. Phytochem. Rev. 2025, 25, 1067–1092. [Google Scholar] [CrossRef]
- Iddir, M.; Vahid, F.; Merten, D.; Larondelle, Y.; Bohn, T. Influence of Proteins on the Absorption of Lipophilic Vitamins, Carotenoids and Curcumin—A Review. Mol. Nutr. Food Res. 2022, 66, 2200076. [Google Scholar] [CrossRef]
- Sun, Y.; Tang, W.; Pu, C.; Li, R.; Sun, Q.; Wang, H. Improved stability of liposome-stabilized emulsions as a co-encapsulation delivery system for vitamin B2, vitamin E and β-carotene. Food Funct. 2022, 13, 2966–2984. [Google Scholar] [CrossRef]
- Li, Y.; Fu, J.; Peng, L.; Sun, X.; Wang, G.; Wang, Y.; Chen, L. A sustainable emulsion for separation and Raman identification of microplastics and nanoplastics. Chem. Eng. J. 2023, 469, 143992. [Google Scholar] [CrossRef]
- Zhao, L.; Li, J.; Yin, K.; Ding, Y.; Sun, L. Emulsion gels prepared from Longzhua mushroom polysaccharides with self-gelling properties as β-carotene carriers: Stability and in vitro digestibility of β-carotene. Int. J. Biol. Macromol. 2024, 276, 134110. [Google Scholar] [CrossRef]
- Tian, L.; Roos, Y.H.; Gómez-Mascaraque, L.G.; Lu, X.; Miao, S. Tremella fuciform Polysaccharides: Extraction, Physicochemical, and Emulsion Properties at Different pHs. Polymers 2023, 15, 1771. [Google Scholar] [CrossRef] [PubMed]
- Shao, P.; Qiu, Q.; Xiao, J.; Zhu, Y.; Sun, P. Chemical Stability and in vitro release properties of β-carotene in emulsions stabilized by Ulva fasciata polysaccharide. Int. J. Biol. Macromol. 2017, 102, 225–231. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Qiao, F.; He, J.; Fu, J.; Liu, T.; Yi, H.; Sheng, Q.; Zhang, L.; Lin, K. Phospholipid Remodeling and Tri-Layer Membrane Reconstruction Mediate Cognitive Effects of Humanized Milk Fat Globules in Neonatal Rats. Adv. Sci. 2025, 12, e07926. [Google Scholar] [CrossRef]
- Brodkorb, A.; Egger, L.; Alminger, M.; Alvito, P.; Assunção, R.; Ballance, S.; Bohn, T.; Bourlieu-Lacanal, C.; Boutrou, R.; Carrière, F.; et al. INFOGEST static in vitro simulation of gastrointestinal food digestion. Nat. Protoc. 2019, 14, 991–1014. [Google Scholar] [CrossRef]
- Ma, H.; Zhang, L.; Niu, X.; Zhang, Y.; Yang, X.; Li, L. Soy protein-gellan gum noncovalent complexes stabilized emulsion: Effect of heating and pH on emulsion stability. Int. J. Biol. Macromol. 2025, 301, 140067. [Google Scholar] [CrossRef]
- Li, X.; Yue, X.a.; Wang, Z.; Yan, R.; Guo, Y. Role of Emulsification and Interfacial Tension of a Surfactant for Oil Film Displacement. Energy Fuels 2021, 35, 3032–3041. [Google Scholar] [CrossRef]
- Shafiei, M.; Kazemzadeh, Y.; Martyushev, D.A.; Dai, Z.; Riazi, M. Effect of chemicals on the phase and viscosity behavior of water in oil emulsions. Sci. Rep. 2023, 13, 4100. [Google Scholar] [CrossRef]
- Xu, H.; Tian, H.; Deng, J.; Zhuo, Q.; Cui, J.; Wang, J.; Yin, Y.; Yu, P. Review of influence of steric effect on aggregation behavior of fine particles. Miner. Eng. 2023, 203, 108304. [Google Scholar] [CrossRef]
- Fu, Y.; McClements, D.J.; Luo, S.; Ye, J.; Liu, C. Degradation kinetics of rutin encapsulated in oil-in-water emulsions: Impact of particle size. J. Sci. Food Agric. 2023, 103, 770–778. [Google Scholar] [CrossRef] [PubMed]
- Anjali, T.G.; Basavaraj, M.G. Influence of pH and Salt Concentration on Pickering Emulsions Stabilized by Colloidal Peanuts. Langmuir 2018, 34, 13312–13321. [Google Scholar] [CrossRef]
- Álvarez, R.; Giménez, B.; Mackie, A.; Torcello-Gómez, A.; Quintriqueo, A.; Oyarzun-Ampuero, F.; Robert, P. Influence of the particle size of encapsulated chia oil on the oil release and bioaccessibility during in vitro gastrointestinal digestion. Food Funct. 2022, 13, 1370–1379. [Google Scholar] [CrossRef]
- Chen, L.; Yokoyama, W.; Liang, R.; Zhong, F. Enzymatic degradation and bioaccessibility of protein encapsulated β-carotene nano-emulsions during in vitro gastro-intestinal digestion. Food Hydrocoll. 2020, 100, 105177. [Google Scholar] [CrossRef]
- Wilde, P.J.; Chu, B.S. Interfacial & colloidal aspects of lipid digestion. Adv. Colloid Interface Sci. 2011, 165, 14–22. [Google Scholar] [CrossRef] [PubMed]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Fu, J.; Wei, J.; Yan, T.; Zhu, X.; Chen, Y.; Hao, Z. Emulsion Prepared with Auricularia polytricha (Mont.) Sacc. As a Direct Emulsifier for β-Carotene Encapsulation: Stability and Digestibility. Processes 2026, 14, 1017. https://doi.org/10.3390/pr14061017
Fu J, Wei J, Yan T, Zhu X, Chen Y, Hao Z. Emulsion Prepared with Auricularia polytricha (Mont.) Sacc. As a Direct Emulsifier for β-Carotene Encapsulation: Stability and Digestibility. Processes. 2026; 14(6):1017. https://doi.org/10.3390/pr14061017
Chicago/Turabian StyleFu, Jianxin, Jing Wei, Tingxia Yan, Xing Zhu, Yuhang Chen, and Zhenghong Hao. 2026. "Emulsion Prepared with Auricularia polytricha (Mont.) Sacc. As a Direct Emulsifier for β-Carotene Encapsulation: Stability and Digestibility" Processes 14, no. 6: 1017. https://doi.org/10.3390/pr14061017
APA StyleFu, J., Wei, J., Yan, T., Zhu, X., Chen, Y., & Hao, Z. (2026). Emulsion Prepared with Auricularia polytricha (Mont.) Sacc. As a Direct Emulsifier for β-Carotene Encapsulation: Stability and Digestibility. Processes, 14(6), 1017. https://doi.org/10.3390/pr14061017



