Process Differences in Phosphorus Release Between Wetland and River Sediments in a Plain River Network
Abstract
1. Introduction
2. Methods and Materials
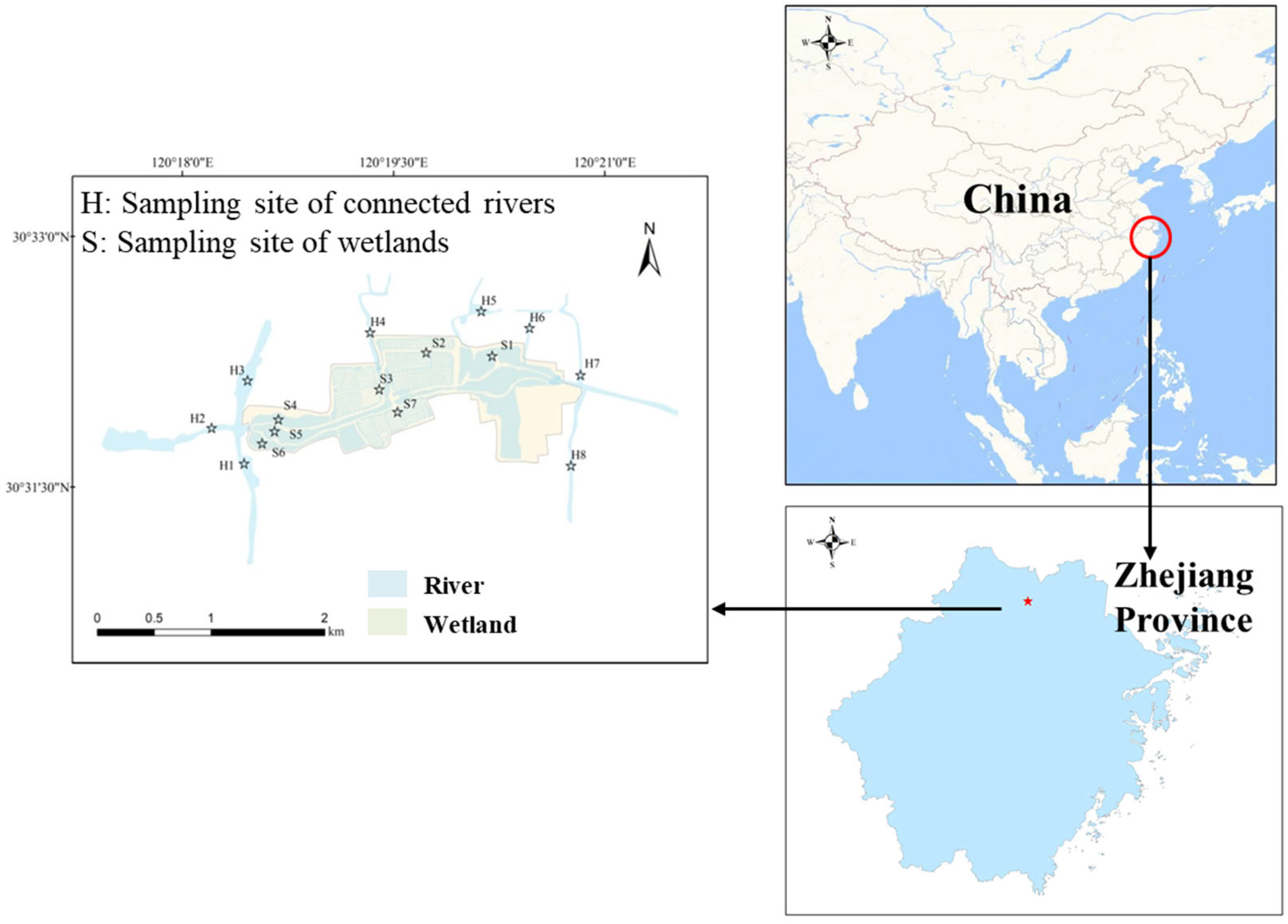
2.1. Sampling Sites and Physicochemical Analysis
2.2. Experimental Methods
2.3. Data Analysis
2.4. Microbial Sequencing and Data Analysis
3. Results and Discussion
3.1. P Spatiotemporal Distribution Characteristics of the Overlying Water and Sediments from Wetlands and Rivers
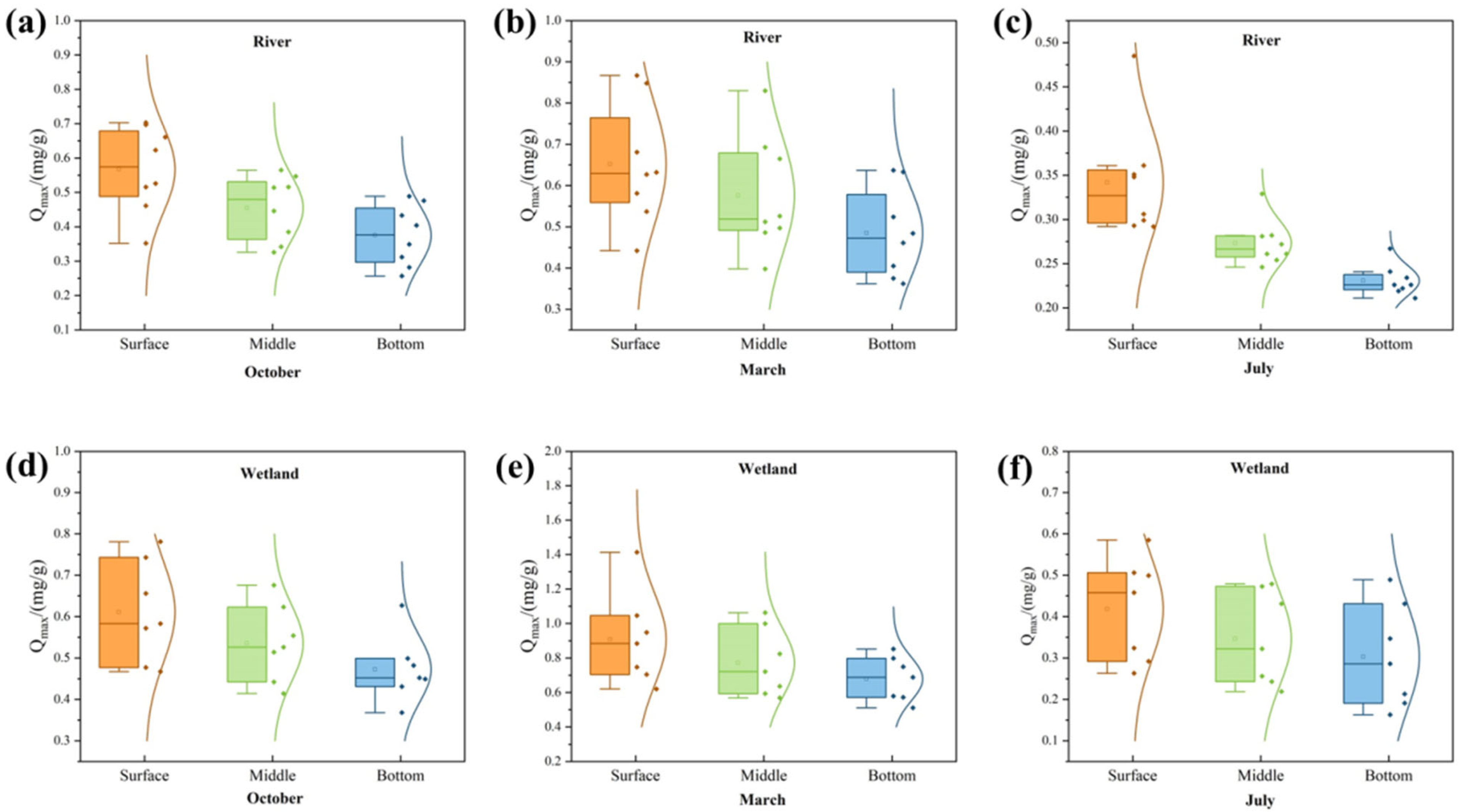
3.2. P Sorption Characteristics of the Sediments from Wetland and Rivers
3.3. P Source and Sink in SS and SH
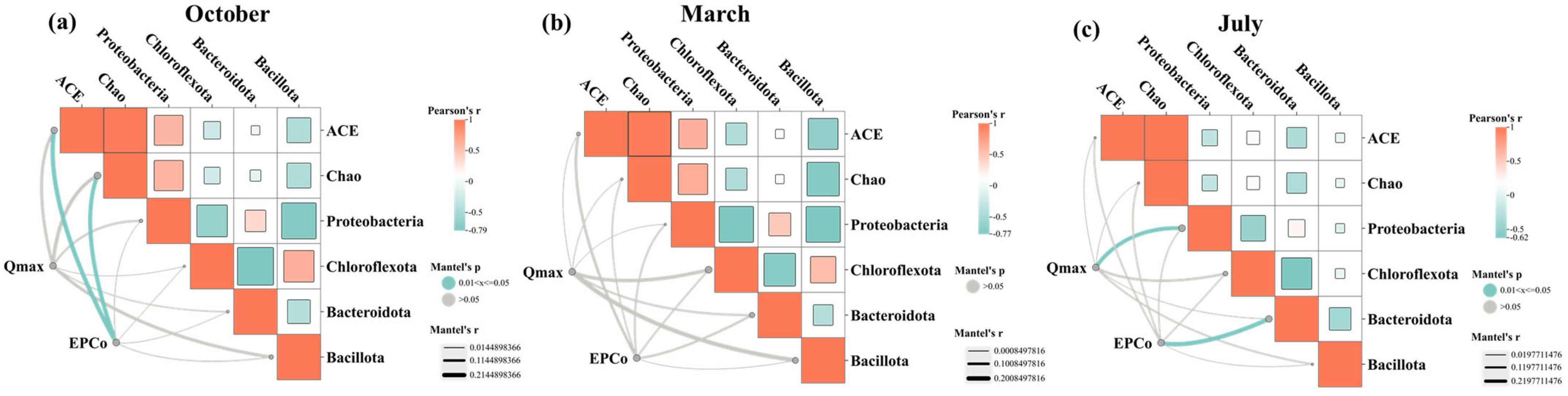
3.4. Response Mechanism of the Microbial Community in P Source and Sink Status
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Yang, H.; Chang, Y.; Dong, X.; Wang, S.; Che, F.; Huang, W. The coupled effect of sediment resuspension and microbiota on phosphorus release and transformation in a simulated aquatic ecosystem. J. Water Process Eng. 2024, 57, 104653. [Google Scholar] [CrossRef]
- Wang, S.; Chang, Y.; Yang, D.; Huang, W.; Che, F. Driving effects of dissolved organic carbon on cadmium release of the sediments by cyanobacterial decomposition. J. Water Process Eng. 2024, 59, 105058. [Google Scholar] [CrossRef]
- Chang, Y.; Dong, X.; Yang, X.; Chen, H.; Yang, H.; Huang, W. Temporal and Spatial Characterization of Sediment Bacterial Communities from Lake Wetlands in a Plain River Network Region. Separations 2023, 10, 535. [Google Scholar] [CrossRef]
- Tu, C.; Jin, Z.; Che, F.; Cao, X.; Song, X.; Lu, C.; Huang, W. Characterization of phosphorus sorption and microbial community in lake sediments during overwinter and recruitment periods of cyanobacteria. Chemosphere 2022, 307, 135777. [Google Scholar] [CrossRef]
- Wang, K.; Yang, H.; Chang, Y.; Huang, W.; Jiang, X. Phosphorus release and distribution in sediment resuspension systems under disturbing conditions. Chemosphere 2024, 359, 142386. [Google Scholar] [CrossRef]
- Huang, Y.; Jiang, L.; Wu, B.; Liu, J.; Liu, Y.; Xie, L.; Zhou, M.; Deng, L.; Wang, W.; Wang, L. Effect of suspended solids from anaerobic digested wastewater on performance and microbial community of autotrophic nitrogen removal process. J. Clean. Prod. 2024, 450, 141973. [Google Scholar] [CrossRef]
- Dong, X.; Chen, H.; Chang, Y.; Yang, X.; Yang, H.; Huang, W. Exploration of Phosphorus Release Characteristics in Sediments from the Plains River Network: Vertical Distribution and the Response of Phosphorus and Microorganisms. Water 2025, 17, 2196. [Google Scholar] [CrossRef]
- Hou, X.; Huang, W.; Song, X.; Liu, Y.; Xiao, Y.; Zhao, X.; An, D. Enhancing manganese redox-driven nitrogen removal by integrating manganese ore with microelectrolysis into constructed wetlands. Bioresour. Technol. 2025, 429, 132467. [Google Scholar] [CrossRef]
- Xing, J.; Zhou, Y.; He, D.; Guo, F.; Li, P.; Zhang, Y.; Lv, R.; Xue, R.; Liu, C.; Xiao, Y. Sediment amplifies organic matter cycling and nutrients feedback in eutrophic lake zones. Water Res. 2025, 286, 124164. [Google Scholar] [CrossRef] [PubMed]
- Weigelhofer, G.; Ramião, J.P.; Pitzl, B.; Bondar-Kunze, E.; O’Keeffe, J. Decoupled water-sediment interactions restrict the phosphorus buffer mechanism in agricultural streams. Sci. Total Environ. 2018, 628–629, 44–52. [Google Scholar] [CrossRef]
- Huang, W.; Lu, Y.; Li, J.H.; Zheng, Z.; Zhang, J.; Jiang, X. Effect of ionic strength on phosphorus sorption in different sediments from eutrophic plateau lake. RSC Adv. 2015, 5, 79607–79615. [Google Scholar] [CrossRef]
- Huang, X.L.; Zhang, J.Z. Spatial Variation in Sediment-Water Exchange of Phosphorus in Florida Bay: AMP As a Model Organic Compound. Environ. Sci. Technol. 2010, 44, 7790–7795. [Google Scholar] [CrossRef]
- Zhang, H.; Xin, M.; Wang, B.D.; Wang, J.; Lin, C.; Gu, X.; Ouyang, W.; Liu, X.; He, M. Spatiotemporal variations in phosphorus concentrations in the water and sediment of Jiaozhou Bay and sediment phosphorus release potential. Sci. Total Environ. 2022, 806, 150540. [Google Scholar] [CrossRef]
- Ningning, J.; Yong, L.; Shengrui, W.; Zhihao, W.; Hong, L. Buffering effect of suspended particulate matter on phosphorus cycling during transport from rivers to lakes. Water Res. 2022, 216, 118350. [Google Scholar] [CrossRef]
- Yu, W.; Wang, L.; Ma, X.; Li, J.; Li, Z.; Wang, H.; Li, D.; Fan, S.; Liu, C.; Yu, D. Restoration of submerged vegetation modulates microbial communities to decrease nitrogen and phosphorus loads in sediment-water systems. Water Res. 2025, 269, 122835. [Google Scholar] [CrossRef]
- Yin, H.; Yang, P.; Kong, M. Effects of different chemical agents on changes in sediment phosphorus composition and the response of sediment microbial community. J. Environ. Manage 2023, 342, 118321. [Google Scholar] [CrossRef]
- Mauro, B.d.T.; Helen, M.B. Variability of sedimentary phosphorus composition across Canadian lakes. Environ. Res. 2023, 236, 116654. [Google Scholar] [CrossRef]
- Huang, W.; Dong, X.; Tu, C.; Yang, H.; Chang, Y.; Yang, X.; Chen, H.; Che, F. Response mechanism of sediment endogenous phosphorus release to functional microorganisms and its cyanobacterial growth and disappearance effects. Sci. Total Environ. 2024, 906, 167676. [Google Scholar] [CrossRef]
- Ruban, V.; Lopez-Sanchez, J.F.; Pardo, P.; Rauret, G.; Muntau, H.; Quevauviller, P. Harmonized protocol and certified reference material for the determination of extractable contents of phosphorus in freshwater sediments—A synthesis of recent works. Fresenius J. Anal. Chem. 2001, 370, 224–228. [Google Scholar] [CrossRef]
- Liu, B.; Liu, Z.; Wu, H.; Pan, S.; Sun, Y.; Xu, Y. Insight into simultaneous selective removal of nitrogen and phosphorus species by lanthanum-modified porous polymer: Performance, mechanism and application. Chem. Eng. J. 2021, 415, 129026. [Google Scholar] [CrossRef]
- Majd, M.M.; Kordzadeh-Kermani, V.; Ghalandari, V.; Askari, A.; Sillanpää, M. Adsorption isotherm models: A comprehensive and systematic review (2010–2020). Sci. Total Environ. 2022, 812, 151334. [Google Scholar] [CrossRef]
- Chang, Y.; Wang, S.; Feifei, C.; Xiaoshuang, D.; Xixi, Y.; Haojie, C.; Wei, H. Arsenic source-sink dynamics under phosphorus competition in sediments from river-lake connected systems. Environ. Sci. Process. Impacts 2025, 27, 706. [Google Scholar] [CrossRef]
- Kreiling, R.M.; Perner, P.M.; Breckner, K.J.; Williamson, T.N.; Bartsch, L.A.; Hood, J.M.; Manning, N.F.; Johnson, L.T. Watershed- and reach-scale drivers of phosphorus retention and release by streambed sediment in a western Lake Erie watershed during summer. Sci. Total Environ. 2023, 863, 160804. [Google Scholar] [CrossRef]
- Huang, W.; Yang, X.; Chen, H.; Lu, C.; Che, F. Promoting effect of submerged plants on phosphorus source and sink in sediments: Response of phosphorus release and microbial community to Vallisneria natans and Potamogeton malaianus growth. Environ. Technol. Innov. 2024, 36, 103899. [Google Scholar] [CrossRef]
- Song, C.; Cao, X.; Zhou, Y.; Azzaro, M.; Monticelli, L.S.; Maimone, G.; Azzaro, F.; La Ferla, R.; Caruso, G. Nutrient regeneration mediated by extracellular enzymes in water column and interstitial water through a microcosm experiment. Sci Total Environ. 2019, 670, 982–992. [Google Scholar] [CrossRef]
- Wang, T.; Zhu, B.; Zhou, M.H. Ecological ditch system for nutrient removal of rural domestic sewage in the hilly area of the central Sichuan Basin, China. J. Hydrol. 2019, 570, 839–849. [Google Scholar] [CrossRef]
- Xu, X.G.; Zhou, Y.W.; Han, R.M.; Song, K.; Zhou, X.H.; Wang, G.X.; Wang, Q.L. Eutrophication triggers the shift of nutrient absorption pathway of submerged macrophytes: Implications for the phytoremediation of eutrophic waters. J. Environ. Manag. 2019, 239, 376–384. [Google Scholar] [CrossRef]
- Chao, Y.; Rui, X.; Yan, C.; Lixin, J.; Xiaoyu, L.; Yingze, Y.; Qiang, H.; Kai, Z.; Lina, L.; Hao, L. Endogenous phosphorus release from plateau lakes responds significantly to temperature variability over the last 50 years. J. Environ. Manag. 2024, 371, 123259. [Google Scholar] [CrossRef]
- Qu, Y.H.; Zhao, L.; Jin, Z.H.; Yang, H.R.; Tu, C.Q.; Che, F.F.; Russel, M.; Song, X.S. Study on the management efficiency of lanthanum/iron co-modified attapulgite on sediment phosphorus load. Chemosphere 2023, 313, 137315. [Google Scholar] [CrossRef]
- Samanta, S.; Debnath, D.; Maitra, N.; Banerjee, M.; Chowdhury, A.N.; Sharma, A.P.; Manna, S.K. Sediment phosphorus forms and levels in two tropical floodplain wetlands. Aquat. Ecosyst. Health 2015, 18, 467–474. [Google Scholar] [CrossRef]
- Yang, C.; Yang, P.; Geng, J.; Yin, H.; Chen, K. Sediment internal nutrient loading in the most polluted area of a shallow eutrophic lake (Lake Chaohu, China) and its contribution to lake eutrophication. Environ. Pollut. 2020, 262, 114292. [Google Scholar] [CrossRef] [PubMed]
- Zhan, Q.; Teurlincx, S.; van Herpen, F.; Raman, N.; Lürling, M.; Waajen, G.; de Senerpont Domis, L. Towards climate-robust water quality management: Testing the efficacy of different eutrophication control measures during a heatwave in an urban canal. Sci. Total Environ. 2022, 828, 154421. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Liang, Z.L.; Callier, M.D.; d’Orbcastel, E.R.; Sun, G.X.; Ma, X.N.; Li, X.; Wang, S.K.; Liu, Y.; Song, X.F. Nutrients removal and substrate enzyme activities in vertical subsurface flow constructed wetlands for mariculture wastewater treatment: Effects of ammonia nitrogen loading rates and salinity levels. Mar. Pollut. Bull. 2018, 131, 142–150. [Google Scholar] [CrossRef] [PubMed]
- Yin, H.; Yan, X.; Gu, X. Evaluation of thermally-modified calcium-rich attapulgite as a low-cost substrate for rapid phosphorus removal in constructed wetlands. Water Res. 2017, 115, 329–338. [Google Scholar] [CrossRef]
- Chen, X.; Jiang, X.; Huang, W. Evaluation and mechanism of ammonia nitrogen removal using sediments from a malodorous river. R. Soc. Open Sci. 2018, 5, 172257. [Google Scholar] [CrossRef]
- Cai, O.; Xiong, Y.; Yang, H.; Liu, J.; Wang, H. Phosphorus transformation under the influence of aluminum, organic carbon, and dissolved oxygen at the water-sediment interface: A simulative study. Front. Environ. Sci. Eng. 2020, 14, 12. [Google Scholar] [CrossRef]
- O’Day, P.A.; Nwosu, U.G.; Barnes, M.E.; Hart, S.C.; Berhe, A.A.; Christensen, J.N.; Williams, K.H. Phosphorus Speciation in Atmospherically Deposited Particulate Matter and Implications for Terrestrial Ecosystem Productivity. Environ. Sci. Technol. 2020, 54, 4984–4994. [Google Scholar] [CrossRef]
- Pant, H.K.; Reddy, K.R. Phosphorus sorption characteristics of estuarine sediments under different redox conditions. J. Environ. Qual. 2001, 30, 1474–1480. [Google Scholar] [CrossRef]
- Schafer, H.; Bernard, L.; Courties, C.; Lebaron, P.; Servais, P.; Pukall, R.; Stackebrandt, E.; Troussellier, M.; Guindulain, T.; Vives-Rego, J.; et al. Microbial community dynamics in Mediterranean nutrient-enriched seawater mesocosms: Changes in the genetic diversity of bacterial populations. FEMS Microbiol. Ecol. 2001, 34, 243–253. [Google Scholar] [CrossRef]
- Vezzulli, L.; Fabiano, M. Sediment biochemical and microbial variables for the evaluation of trophic status along the Italian and Albanian continental shelves. J. Mar. Biol. Assoc. UK 2006, 86, 27–37. [Google Scholar] [CrossRef]
- Li, Y.H.; Zhao, S.H.; Wang, Y.Q. Microbial desulfurization of ground tire rubber by Thiobacillus ferrooxidans. Polym. Degrad. Stab. 2011, 96, 1662–1668. [Google Scholar] [CrossRef]
- Sun, Z.; Li, G.; Wang, C.; Jing, Y.; Zhu, Y.; Zhang, S.; Liu, Y. Community dynamics of prokaryotic and eukaryotic microbes in an estuary reservoir. Sci. Rep. 2014, 4, 6966. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.; Jiang, X. Profiling of Sediment Microbial Community in Dongting Lake before and after Impoundment of the Three Gorges Dam. Int. J. Environ. Res. Public Health 2016, 13, 617. [Google Scholar] [CrossRef] [PubMed]



Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Liu, Y.; Xu, X.; Cui, J.; Tang, D.; Zhao, S. Process Differences in Phosphorus Release Between Wetland and River Sediments in a Plain River Network. Processes 2026, 14, 877. https://doi.org/10.3390/pr14050877
Liu Y, Xu X, Cui J, Tang D, Zhao S. Process Differences in Phosphorus Release Between Wetland and River Sediments in a Plain River Network. Processes. 2026; 14(5):877. https://doi.org/10.3390/pr14050877
Chicago/Turabian StyleLiu, Yinan, Xin Xu, Jianglong Cui, Dongya Tang, and Shanshan Zhao. 2026. "Process Differences in Phosphorus Release Between Wetland and River Sediments in a Plain River Network" Processes 14, no. 5: 877. https://doi.org/10.3390/pr14050877
APA StyleLiu, Y., Xu, X., Cui, J., Tang, D., & Zhao, S. (2026). Process Differences in Phosphorus Release Between Wetland and River Sediments in a Plain River Network. Processes, 14(5), 877. https://doi.org/10.3390/pr14050877



