1. Introduction
As global warming worsens, the interest in energy-efficient and hydrogen-based sustainable processes is growing. According to the International Energy Agency (IEA), the iron and steel sector accounted for 7% of global CO
2 emissions, derived from the total energy system, including industrial processes [
1]. Among the heavy industries, it produced the most CO
2 emissions and consumed the second-largest amount of energy, making the steel industry a representative carbon- and energy-intensive sector [
1]. Therefore, transitioning to low-carbon processes in the iron and steel industry is crucial.
Iron and steel manufacturing is categorized into distinct routes based on the process configurations, raw materials, and reducing agents utilized. Over time, technological advancements in these processes have focused on improving energy efficiency, valorizing off-gas into chemicals, and transitioning to alternative reductants or feedstocks. Specifically, in the traditional blast furnace–basic oxygen furnace (BF–BOF) route, which consumes significant energy using iron ore and coke, studies have explored sustainable alternatives. For example, the utilization of biomass-derived coke has been investigated to mitigate CO
2 emissions, demonstrating a potential reduction of up to 42% in annual emissions, depending on carbon pricing scenarios [
2,
3].
To overcome the limitations of the BF route, smelting reduction processes were developed. The COREX
® route mitigates pollution by eliminating the coking process and utilizing non-coking coal, which reduces dust, SO
2, and NO
x emissions by 54%, 17%, and 97%, respectively, compared to the BF route [
4]. However, the COREX route employs a shaft furnace for reduction. Similar to other shaft furnace-based processes for direct reduced iron (DRI) production, this configuration necessitates high-grade pellets. This dependency presents a challenge due to the tightening global supply of high-quality pellets for DRI production.
In response, the FINEX
® route was introduced to utilize the abundant iron ore fines directly by replacing the shaft furnace with fluidized bed reactors (FBRs). While this configuration primarily secures raw material flexibility, it also demonstrates a reduced environmental footprint compared to the conventional BF route, resulting in lower dust, SO
x, and NO
x emissions by 41%, 94%, and 91%, respectively [
5].
Despite these advancements in smelting reduction utilizing coal, these processes still inevitably emit CO
2 due to the use of carbon-based reductants. Therefore, to achieve carbon neutrality, hydrogen-based reduction technology is emerging as the ultimate decarbonization strategy. By substituting fossil fuels with green hydrogen, this approach fundamentally eliminates CO
2 emissions, producing only H
2O as a byproduct. Globally, efforts are underway to transition existing natural gas-based processes into hydrogen-based systems. For instance, shaft furnace technologies such as MIDREX
® and ENERGIRON
® are being adapted to utilize hydrogen as the primary reducing agent instead of natural gas [
6]. While ENERGIRON offers the advantage of in situ reforming within the furnace [
6], these shaft furnace-based routes still face the constraint of requiring high-grade pellets [
7].
In contrast, research in South Korea, led by POSCO
®, focuses on hydrogen-based reduction utilizing an FBR technology [
8]. This process leverages the technology of the FINEX route to utilize hydrogen as the reducing agent, while maintaining the capability to operate directly with iron ore fines. In this system, iron ore fines are reduced in a multi-stage FBR to produce DRI, which is subsequently melted in an electric arc furnace (EAF) powered by renewable energy [
5,
6]. This technological pathway is expected to play a pivotal role in realizing a carbon-neutral steel industry by securing both raw material flexibility and environmental sustainability.
This study develops a simulation model of the hydrogen-based DRI production process via multi-stage FBRs and investigates the feasibility of the proposed simulation model. Methodologies for process simulation, techno-economic, and environmental assessment are described in
Section 2. The simulation results, feasibility assessment, and the impact of carbon pricing are discussed in
Section 3. Finally, concluding remarks are presented in
Section 4.
2. Methodology
2.1. Framework
A steady-state simulation model for the hydrogen-based DRI production process through the multi-stage FBRs was developed using a commercial simulation tool, Aspen Plus V14. The simulation boundary is presented in
Figure 1.
As shown in the figure, the simulation boundary is defined to the ironmaking process, explicitly excluding the steelmaking stage via EAF. To evaluate the potential for future renewable energy integration, hydrogen and electricity were defined as the primary reductant and energy source, respectively.
The thermodynamic method used the Peng–Robinson equation of state with Boston–Mathias modifications [
9]. The chemical reactions considered within the simulation boundary are as follows. For the simulation of DRI production, side reactions, such as coke generation, were assumed to be neglected.
Reduction by carbon dioxide:
Dolomite half-calcination:
Although the proposed process is described as hydrogen-based, a complete reaction set including CO reduction was retained because the reducing gas contains both H
2 and CO within the simulation boundary. This also allows the model to represent the coupled CO/CO
2 and H
2/H
2O equilibria governed by the water gas shift reaction and to maintain thermodynamic as well as mass and energy balance consistency. Accordingly, the proposed hydrogen-based DRI production is defined as an H
2-dominant reduction environment, without assuming a strictly CO-free reducing gas. The contribution of CO-based reduction is quantified and discussed in
Section 3.
The FBR was modeled following the hydrogen direct reduction kinetics developed by Lee et al. [
10]. A two-phase model was employed to describe gas–solid interactions within the reactor, consisting of the emulsion and bubble phases. According to this model, in the bubble phase, gases bypass in the form of bubbles, and reactions rarely occur. Conversely, the emulsion phase represents the region where solid particles and gases are uniformly mixed, and reactions primarily occur [
11]. Consequently, local concentration gradients within the emulsion phase were neglected based on the assumption of perfect mixing, which is supported by prior comparisons showing only minor deviations between two-phase and well-mixed predictions under industrially relevant operating ranges [
11]. Based on this, the FBR was modeled as a continuous stirred tank reactor (CSTR), with the reactor volume defined as equivalent to the volume of the emulsion phase.
Within this framework, the reaction kinetics were developed using a modified grain model. This model incorporates the assumptions that internal gas diffusion is not the rate-limiting step and that both the grain size and gas concentration within a particle remain uniform [
12,
13,
14]. The kinetic parameters were adjusted by fitting the hydrogen direct reduction kinetic model to experimental data provided by a commercial steel manufacturer [
8] under operating conditions spanning 714–1031 K and 4.32–5.61 bar, following the conditions reported by Lee et al. [
10]. The estimated kinetic parameters for the four stage FBR simulation model were summarized in
Table 1, while the parameters for the driving force and adsorption terms were adopted directly from the provided dataset.
2.2. Process Description
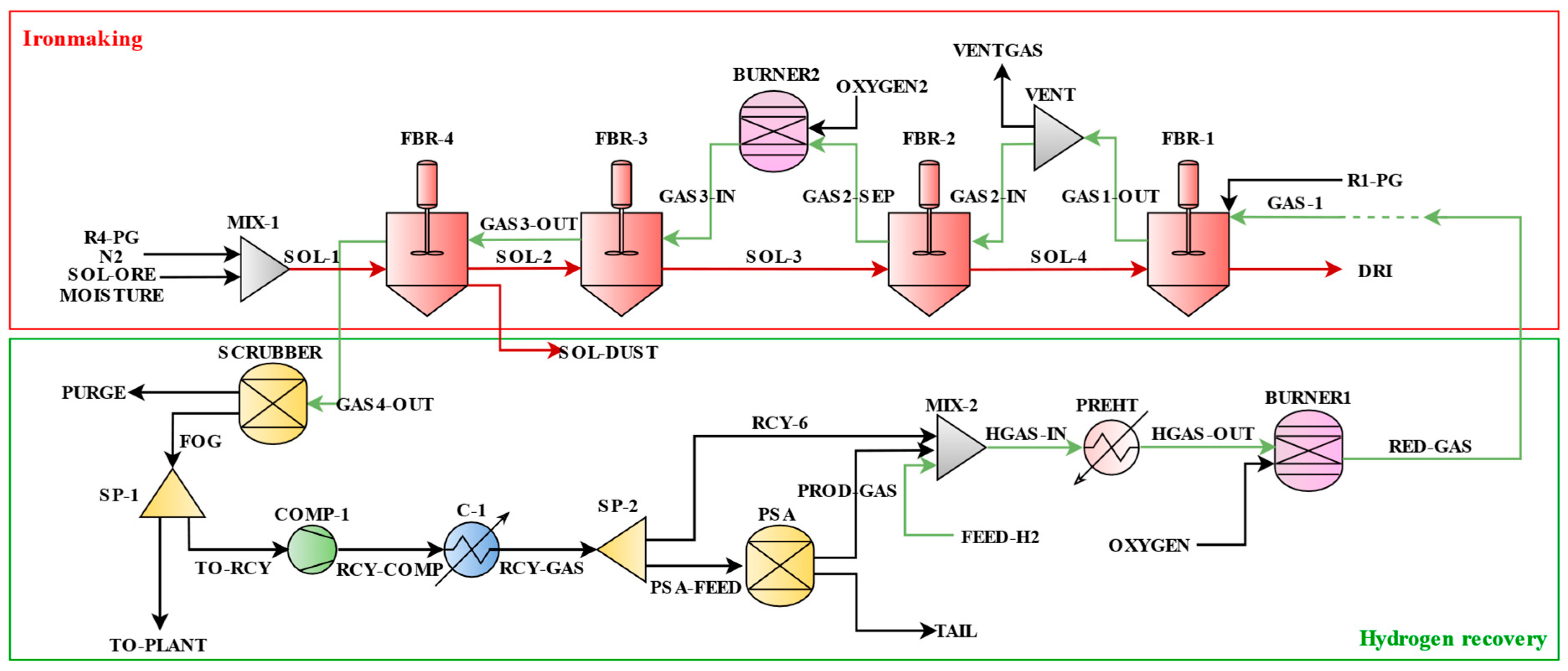
The hydrogen-based DRI production process is divided into two sections: the ironmaking process and the hydrogen recovery process.
Figure 2 shows the overall process flow.
The iron ore fines (SOL-ORE), containing moisture (MOISTURE), enter along with transport and purge nitrogen (N2, R4-PG). The mixed ore fines (SOL-1) enter reactor 4 (FBR-4) and are reduced by the gas stream (GAS3-OUT). The reduced ore fines (SOL-2) enter the next reactor (FBR-3). Likewise, the solid stream moves from reactor 4 to 1 as the reduction reaction progresses in the order of hematite, magnetite, wüstite, and iron. Finally, the product (DRI) is produced from reactor 1 (FBR-1).
On the other hand, reducing gases (GAS-1) also enter reactor 1 (FBR-1) with nitrogen (R1-PG) and drive a counter-current type of iron reduction reaction. The reacted gases (GAS1-OUT) are partially bent and enter reactor 2 (GAS2-IN). The GAS2-SEP stream flowing out of the reactor is heated in the O2 burner (BURNER2) to go to the next reactor. The heated stream (GAS3-IN) passes through reactors 3 and 4 (FBR-3, FBR-4). Finally, it exits the multi-stage FBR process. Then, the last stream (GAS4-OUT) from the reactor enters the hydrogen recovery process.
Before hydrogen recovery, gases are partially purged (SCRUBBER) to remove impurities. Then, the off-gases (FOG) are separated (SP-1) into a flow to the power plant (TO-PLANT) and a flow sent to the recovery process (TO-RCY). The stream going to the recovery process is pressurized (COMP-1) and cooled (C-1) to meet the temperature and pressure conditions. The recovery gas stream (RCY-GAS) is then separated (SP-2), and one of them enters the PSA as the feed stream (PSA-FEED). Components such as CO and H2O are removed as tail gas (TAIL) from the PSA. The PSA product gas, recovered as high-purity H2 (PROD-GAS), is mixed (MIX-2) with the recycled streams (RCY-6) and green H2 (FEED-H2) for make-up. The hydrogen-rich mixed gas (HGAS-IN) undergoes the pre-heating process (PREHT) and then enters the O2 burner (BURNER1), where it is heated to approximately 1073.15 K. Finally, the reducing gases (RED-GAS) are connected to the feed into the FBR-1.
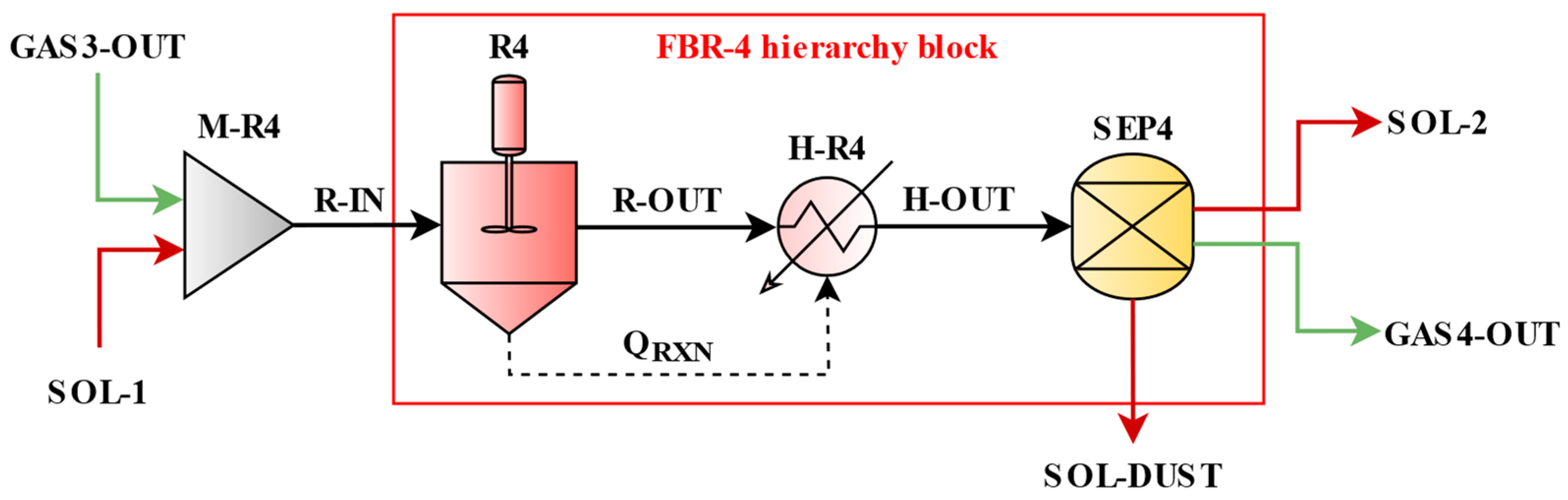
Figure 3 shows a detailed FBR process. The FBR hierarchy block consists of an isothermal CSTR (R4) used to evaluate the reaction extent, a heater unit (H-R4) for thermal compensation of the reaction heat effect, and a separator (SEP4) as a solid separation cyclone. The gas and solid streams entering the hierarchy block are mixed (M-R4) and fed into R4, where the mixed stream is heated to the reaction temperature and undergoes a reaction. Because R4 is modeled under isothermal conditions, the heat duty derived from the reaction results is interpreted as the reaction heat effect (Q
RXN) and transferred to the downstream heater (H-R4). As a result, the product stream (R-OUT) enters the heater unit and reflects the temperature change due to the reaction. In the separator, the stream (H-OUT) is separated into solids (SOL-2) and gases (GAS4-OUT) and then flows to the next reactor, respectively. In reactor 4, solids exclude part of the dust (SOL-DUST).
2.3. Techno-Economic Assessment
Techno-economic feasibility studies of the hydrogen-based DRI production through multi-stage FBRs were performed based on mass and energy balance results derived from the process simulation results.
Table 2 summarizes the key economic parameters and unit costs used to calculate the unit production cost (UPC).
General plant parameters, including plant uptime, plant lifetime, and annual interest rate, were adopted from standard design and economic assumptions reported by Turton et al. (2018) [
15]. Variable costs were estimated based on utility consumption rates obtained directly from the simulation outputs, whereas unit costs for utilities and raw materials were sourced from the relevant literature and market data. Hydrogen costs were differentiated across gray, blue, and green supply pathways, as summarized in
Table 2.
The UPC was defined as the total annual cost divided by the annual production rate of DRI. The total annual cost consists of the annualized capital cost and the total operating cost (TOC). The capital cost component was derived from the total capital investment (TCI), consisting of the fixed capital investment (FCI) and working capital [
23].
In this study, the FCI was estimated based on the TCI of a structurally analogous process rather than being directly derived from purchased equipment costs, using the ratio factor adopted from Turton et al. [
15]. Specifically, capital cost data of the FINEX process, which employs a multi-stage fluidized reduction in iron ore fines and exhibits a comparable process configuration, were used as the basis for estimation [
3]. As the reactor train and solid handling units are major capital cost elements, FINEX data provide a reasonable reference basis. The reported TCI was adjusted to the base year of this study using the chemical engineering plant cost index (CEPCI).
However, inherent limitations exist because hydrogen-based operation requires distinct provisions for thermal management, reactor internals, and hydrogen handling and infrastructure, as well as specialized safety systems such as leak detection and explosion protection. Therefore, the absolute FCI estimated here should be interpreted as an early-stage estimate. Following AACE International Recommended Practice 18R-97, this estimate is associated with a typical accuracy range of −30% to +50% [
24]. Accordingly, the economic discussion emphasizes relative differences across scenarios under a consistent costing basis, and cross-technology UPC comparisons should be interpreted with caution given the proxy-based scaling assumption.
The TOC included variable costs such as raw materials, utilities, and waste treatment, as well as fixed costs, including operating labor and maintenance expenses estimated using the ratio factor as provided by Peters et al. [
23]. The capital recovery factor (CRF) was applied to convert the initial TCI into an equivalent annual capital cost over the plant lifetime. The UPC was calculated by dividing the total annual cost by the annual production rate, as defined in Equations (9)–(11):
where
,
,
, and
are operating costs for operating labor, utility, waste treatment, and raw material, respectively (USD/yr).
is the annual interest rate (-);
is the plant life (years).
In the process simulation, the pressure swing adsorption (PSA) unit was simplified as a component separator block. A fixed hydrogen recovery of 85.5% was applied to ensure that the recycled gas composition remained consistent with the calibration basis of the kinetics-calibrated reactor model. The PSA energy usage was not derived from the simulation results. Instead, the energy consumption was calculated using the formula obtained from Abd et al., as presented in Equation (12) [
25]:
where
is the energy consumption of the PSA (kJ/s),
is the ratio of specific heats (-),
is the gas constant (J/mol-K),
is temperature (K),
is pressure (bar),
is feed molar flow rate (mol/s),
is compressor efficiency (-), which is 0.8, and
is the mole ratio of purged gas to the total regenerated gas (-).
2.4. Environmental Assessment
To evaluate the environmental sustainability of the hydrogen-based DRI production process, the CO2 equivalent (CO2e) emissions were quantified on an hourly basis. The calculation was based on the energy consumption data derived from the mass and energy balance simulation result.
The total CO
2e emissions were calculated as the sum of direct and indirect emissions. Direct emissions refer to the emitted CO
2 directly within the system boundaries, whereas indirect emissions account for the GHGs associated with the consumption of utilities. To quantify indirect impacts, specific global warming impact (GWI) factors were applied to the utility consumption rates [
26]. The formula used the following Equations (13) and (14):
where
denotes the consumption rate of utility
(Gcal/h), and
is the specific GWI factor for that utility source (tCO
2e/Gcal).
In this study, the environmental impact was analyzed under two distinct electricity source scenarios: (1) utilizing the conventional natural gas-based grid (NG-based scenario) and (2) integrating renewable energy sources such as wind power (RE-based scenario).
2.5. Impact of Carbon Pricing
Although the integration of renewable energy significantly mitigates carbon emissions, it typically incurs higher UPCs due to the elevated price of renewable electricity compared to fossil fuel-based power. To evaluate the economic feasibility of the RE-based scenario under carbon regulations, a scenario analysis was conducted considering the potential carbon revenue within the framework of the Korea Emission Trading System (K-ETS). The analysis range for the carbon price was set from 0 to 26 USD/tCO
2e, referencing the market data from 2018, where the average price was approximately 20.66 USD/tCO
2e [
27].
The potential carbon credit revenue was calculated based on the emission reduction achieved by replacing the conventional BF process with the RE-based scenario. This benefit is defined in Equation (15):
where
represents the carbon credit revenue (USD/ton),
and
denote the specific CO
2e emissions of the conventional BF process and the RE-based scenario, respectively (tCO
2e/h).
is the K-ETS allowance price (USD/tCO
2e).
Finally, to assess the cost competitiveness, the relative cost ratio compared to the NG-based scenario was calculated using Equation (16):
where
and
represent the UPCs of the RE-based and NG-based scenarios, respectively (USD/ton). This equation evaluates the extent to which the carbon revenue can offset the increased cost of renewable electricity.
3. Results
3.1. Simulation Validation
To ensure the reliability of the developed simulation model for the hydrogen-based DRI production process, the simulation results were validated against the experimental values. Specifically, the prediction accuracy of the simulation model was evaluated by comparing the solid and gas composition profiles across the multi-stage FBRs. To quantitatively assess the correlation between the experimental values and the simulation outputs, the coefficient of determination (R
2) was calculated using Equation (17) [
28]:
where
is the experimental data of
,
is the simulation data, and
is the mean of the experimental data.
Figure 4 illustrates the parity plots comparing the experimental and simulation values for solid and gas phase compositions. As shown in the figure, the data points are tightly clustered around the y = x line, indicating a high degree of consistency between the model prediction results and the experimental values. The calculated R
2 values for the solid and gas compositions were 1.000 and 0.999, respectively. These high correlations indicate strong agreement between the model predictions obtained with calibrated kinetic parameters and experimental data, supporting its use for representing the steady-state performance of the multi-stage FBRs system under the studied conditions. Consequently, the developed simulation model is considered suitable to serve as the basis for techno-economic and environmental assessments.
Based on the validated model, the relative contribution of CO-based reduction reactions was quantified against the total reduction rate, excluding the water gas shift reaction. The analysis indicated that CO-based reduction contributes a minor fraction under the hydrogen-based reduction, confirming a H2-dominant reduction environment. This result helps clarify the role of CO reactions within the overall reduction pathways.
3.2. Techno-Economic Assessment Results
The economic feasibility of the hydrogen-based DRI production process was initially evaluated based on the gray hydrogen scenario, which serves as the baseline reflecting the current market conditions. In this scenario, hydrogen is assumed to be produced via conventional steam methane reforming (SMR). The UPC was estimated at 455.52 USD per ton of DRI. To identify the primary cost drivers, the distribution of the UPC was analyzed, as illustrated in
Figure 5.
As shown in
Figure 5, the cost structure is dominated by TOCs, particularly raw materials, rather than TCI. Specifically, raw material costs account for approximately 62% of the UPC. A detailed breakdown reveals that hydrogen feedstock cost constitutes the largest portion of raw material costs, significantly overshadowing the cost of iron ore fines. This cost disparity highlights the fundamental economic challenge of replacing relatively inexpensive coal-based reductants with hydrogen. The utility costs, primarily electricity, also represent a notable share of the TCI. Unlike conventional BF, which utilizes the chemical energy of coal for heat, the proposed FBR process relies on electrical energy for pre-heating the feed mixture to the reaction temperature. While this electrification strategy is essential for decarbonization, it inevitably leads to higher operating costs compared to fossil fuel-based heating.
To reflect realistic industrial operations and mitigate utility costs, this study incorporated industrially reported energy recovery performance into the base case estimation. Industrial operation data indicate that internally recovered byproduct gases can supply approximately 69% of the total electricity demand in integrated steelworks, with the remaining electricity purchased externally [
29]. Accordingly, 69% of electricity demand was assumed to be internally supplied, and only the remaining 31% was considered as purchased electricity in the utility cost calculation of the ironmaking system. This assumption was used solely to adjust utility cost estimation within the ironmaking system boundary, without explicitly modeling plant-wide power generation facilities. Under these conditions, internal energy recovery reduces the UPC by approximately 9.6%, as reflected in the UPC distribution results in
Figure 5.
To evaluate the economic feasibility of the transition toward a low-carbon steel industry, the analysis was extended to the blue and green hydrogen scenarios. These scenarios correspond to hydrogen produced via SMR coupled with carbon capture and storage and water electrolysis, respectively.
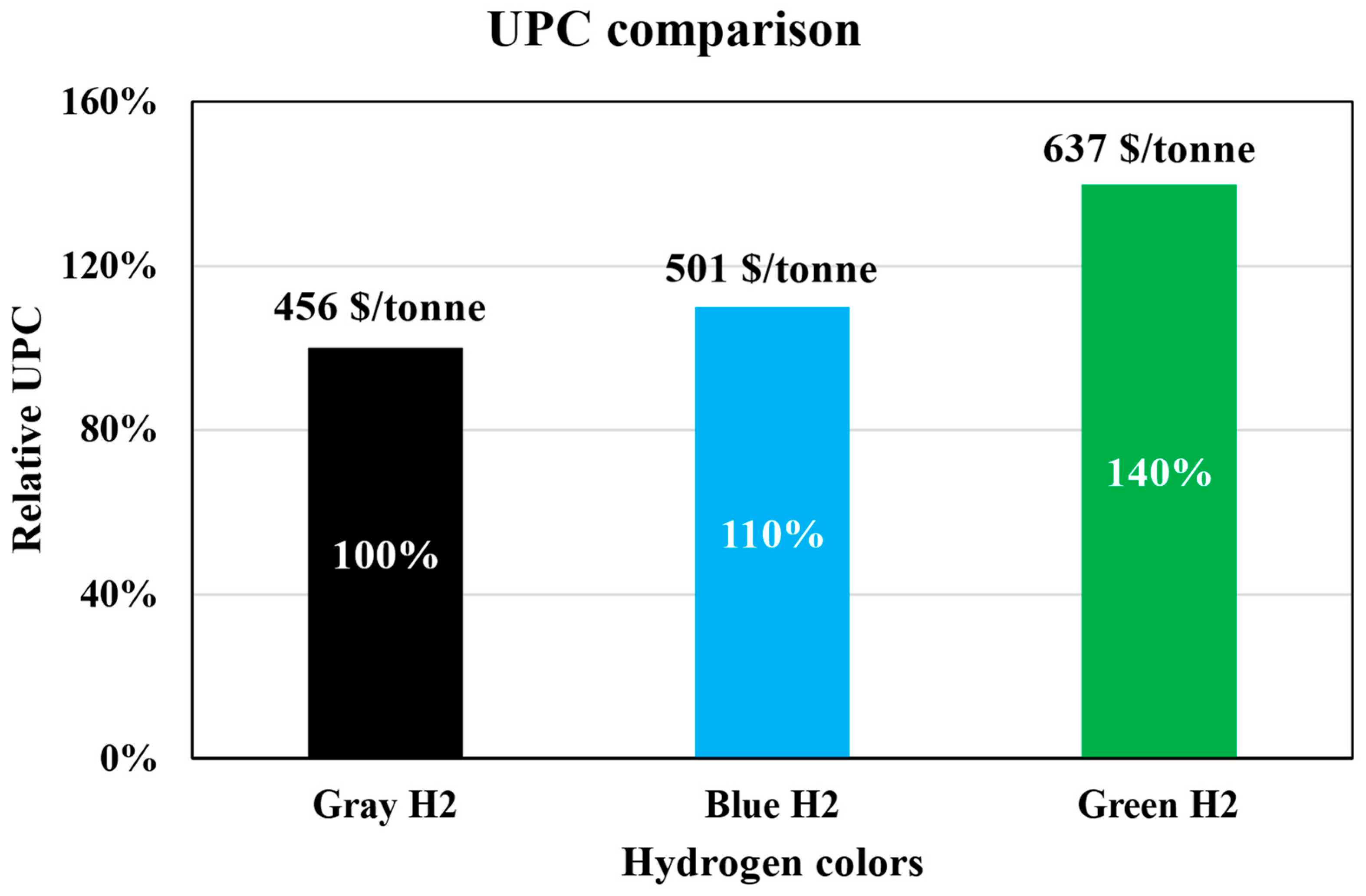
Figure 6 shows the comparison of the UPCs across these three pathways.
As the hydrogen source transitions from gray to green, a progressive increase in UPC is observed. The estimated UPC for the gray, blue, and green hydrogen scenarios is calculated to be 455.52 USD/ton, 500.85 USD/ton, and 636.83 USD/ton, respectively. Notably, the adoption of green hydrogen leads to a substantial cost increase of approximately 40% compared to the gray hydrogen scenario. This significant disparity indicates that the economic competitiveness of the eco-friendly process is dependent on the market price of hydrogen feedstock. Consequently, securing a low-cost hydrogen supply and optimizing hydrogen utilization efficiency within the reactor system are identified as critical prerequisites for the commercialization of green steel production.
To assess the economic feasibility of the process under projected future market conditions, the IEA’s Net Zero Emission (NZE) 2050 scenario was examined. According to this roadmap, the price of green hydrogen is projected to decline to 1.0 USD/kg, driven by technological advancements and renewable energy expansion [
30]. Applying this target price to the green hydrogen scenario reveals that the UPC would significantly decrease by 35.9% from the current green hydrogen scenario, reaching an estimated 410.19 USD/ton. This cost is 9.95% lower than that of the gray hydrogen scenario. However, achieving a green hydrogen cost level of 1.0 USD/kg is region-dependent. In Korea, constraints such as limited domestic renewable availability, grid integration requirements, and potential import dependence may introduce additional costs and uncertainty. Reaching this level would require either expanded low-cost renewable supply with supportive grid conditions, or access to competitively priced imports. Overall, this projection suggests that while the process currently faces a significant economic barrier, gradual technological advancements aligned with global net zero targets can eventually secure the economic viability of the hydrogen reduction process.
3.3. Environmental Assessment Results
To assess the environmental sustainability of the proposed process, the CO
2e emissions were quantified and compared against the conventional BF ironmaking process.
Table 3 shows the specific CO
2e emissions for the three ironmaking processes: the conventional BF process, the NG-based scenario, and the RE-based scenario. Both scenarios represent the green hydrogen scenario implemented with distinct electricity sources. For a fair comparison, the emission data for the BF process are restricted to the ironmaking boundary, excluding the downstream BOF steelmaking section to ensure a consistent comparison basis [
31].
As presented in
Table 3, the transition from the coal-based BF process to the hydrogen-based reduction process yields a significant reduction in carbon emissions under the hydrogen-based DRI production process. The specific CO
2e emissions per 1 ton of hematite decreases from 1937 kg in the BF process to 338.8 kg in the NG-based scenario, representing a reduction of approximately 82.5%.
Furthermore, the RE-based scenario, integrated with renewable electricity, demonstrates the most substantial decarbonization potential. By eliminating the indirect emissions associated with fossil fuel-based power generation, the RE-based scenario achieves a specific emission rate of 36.8 kg/tHM. This corresponds to a reduction of 1900 kg/tHM (98.1%) compared to the BF and an additional decrease of 302 kg/tHM (89.1%) relative to the NG-based scenario. This quantitative result confirms that integrating clean energy is imperative for maximizing the environmental benefits of the hydrogen reduction process.
To further investigate the impact of the electricity source on the emission profile, the distribution of CO
2e emissions for each scenario was analyzed.
Figure 7 shows the contribution of each specific emission source to the total environmental impact.
As shown in
Figure 7, the energy-intensive equipment, particularly the pre-heater and FBRs, account for the vast majority (92.2%) of total emissions due to the carbon intensity of the natural gas-based grid. Consequently, direct emissions represent a negligible fraction, accounting for only 2.6% of the total emissions.
Conversely, in the RE-based scenario, the emission profile undergoes a structural change. While the total emissions decreased due to the decarbonized electricity supply, the absolute amount of direct emissions remained constant. As a result, the relative contribution of direct emissions notably increased to 23.9%, becoming a significant source of the remaining carbon footprint. This distributional shift suggests that once the electricity supply is fully decarbonized, further environmental improvements should focus on optimizing the process to minimize direct gas losses from purge and vent streams, for instance, by utilizing hydrogen-rich purge gas as a supplementary fuel through heat integration for process heating and temperature control.
3.4. Impact of Carbon Pricing Results
While the RE-based scenario achieves substantial environmental benefits, its economic feasibility is initially constrained by the high cost of renewable electricity, resulting in a UPC of 640.37 USD/ton in the absence of carbon pricing. However,
Figure 8 illustrates that considering the potential carbon revenue offsets increased the UPC derived from renewable electricity. The figure presents the sensitivity of the relative cost ratio with respect to K-ETS allowance prices, evaluating the economic competitiveness against the NG-based scenario.
The analysis identifies the specific allowance price where the UPC of the RE-based scenario becomes lower than that of the NG-based scenario. As shown in
Figure 8, the cost competitiveness against the NG-based scenario is secured at a relatively low ETS price of 1.44 USD/tCO
2e. This low threshold is primarily attributed to the substantial emission reduction achieved relative to the conventional BF baseline, which forms the basis for carbon credit calculation. Because this large reduction potential amplifies carbon credit revenue, even a minimal carbon price generates sufficient revenue to offset the cost difference between the RE- and NG-based scenarios. At this point, the UPC of the RE-based scenario decreases to 636.83 USD/ton, reaching parity with the NG-based scenario. This indicates that even a minimal ETS price is sufficient to bridge the economic gap, driven by the massive emission reduction achieved by the green hydrogen process.
Furthermore, when applying the 2018 average K-ETS price of 20.66 USD/tCO2e, the economic competitiveness is significantly enhanced. Under this market condition, the UPC of the RE-based scenario decreases to 589.76 USD/ton. This represents a 7.90% reduction compared to its initial UPC and is 7.39% lower than the NG-based scenario. These results confirm that under realistic carbon pricing mechanisms, the proposed process secures a distinct economic advantage over the grid-based case.
4. Conclusions
This study developed a process simulation model for the hydrogen-based DRI production via multi-stage FBRs and performed a comprehensive techno-economic and environmental assessment. The analysis focuses on the ironmaking process and excludes downstream steelmaking routes such as the EAF. To accurately predict the reactor performance, the FBR system was modeled as a CSTR, incorporating hydrogen reduction kinetics with parameters estimated from the experimental data. The simulation results were validated against the experimental data, yielding R2 of 1.000 for the solid phase and 0.999 for the gas phase. These high correlations confirm the reliability of the developed model as a solid foundation for subsequent feasibility studies.
The techno-economic assessment initially highlighted the economic challenges of the green hydrogen transition. The UPC for the green hydrogen scenario was estimated at 636.83 USD/ton, which is approximately 40% higher than the gray hydrogen baseline. This disparity is primarily driven by the high market price of hydrogen feedstock. However, the sensitivity analysis based on the IEA’s NZE 2050 scenario suggests a promising outlook. If the green hydrogen price drops to 1.0 USD/kg due to technological advancements, the UPC would decrease to 410.19 USD/ton. This figure is 9.95% lower than the current gray hydrogen cost, indicating that the process can achieve intrinsic economic viability through the maturation of the hydrogen economy.
In the environmental assessment, the proposed process demonstrated remarkable potential for decarbonization. The RE-based scenario achieved specific CO2e emissions of 36.8 kg/tHM. This represents a drastic reduction of 98.1% compared to the conventional BF process and an 89.1% reduction relative to the NG-based scenario. The analysis of the emission distribution revealed that as indirect emissions were effectively eliminated in the RE-based scenario, the relative contribution of direct emissions increased to 23.9%. This distributional shift implies that future environmental optimization should prioritize minimizing direct gas losses from purge and vent streams.
Finally, the scenario analysis incorporating the K-ETS confirmed that carbon pricing can effectively bridge the economic gap before hydrogen prices stabilize. The results identified that cost competitiveness against the NG-based scenario is secured at a carbon price threshold of only 1.44 USD/tCO2e. Furthermore, under the 2018 average K-ETS price of 20.66 USD/tCO2e, the RE-based scenario secured a distinct economic advantage, lowering the UPC to 589.76 USD/ton, which is 7.39% lower than the fossil fuel grid-based alternative.
In conclusion, this study demonstrates that the hydrogen-based DRI production process is a technically feasible and environmentally superior alternative to conventional ironmaking. Although the process currently faces economic barriers due to high energy costs, the convergence of declining hydrogen prices and the implementation of carbon pricing policies provides a clear pathway toward commercial viability, positioning it as a promising technology for a carbon-neutral steel industry.