1. Introduction
Particle size is one of the most important physicochemical properties of nanoparticles and plays a decisive role in determining their functionality and application. For example, catalyst particles used in electrolytic hydrogen production can reach micrometer-scale dimensions [
1], whereas nanomedicines designed for tumor therapy typically require particle sizes below 100 nm to ensure favorable biodistribution and cellular uptake [
2]. Consequently, precise control over nanoparticle size and size distribution is essential.
The polydispersity index (PDI) is widely used to evaluate the breadth of particle size distributions. A PDI value ≤ 0.1 generally indicates a very narrow distribution, while values below 0.3 are considered acceptable for drug delivery systems and suggest a relatively homogeneous particle population [
3]. In both polymeric and inorganic particulate systems, improved size homogeneity is often associated with enhanced stability and reproducibility. Regulatory guidelines, such as the U.S. Food and Drug Administration’s Guidance for Industry, emphasize particle size and size distribution as critical quality attributes for particulate drug products [
4]. Therefore, developing synthesis strategies that yield nanoparticles with narrow size distributions and low PDI values remains an important goal.
In practice, however, nanoparticle synthesis protocols vary widely across laboratories and production environments due to differences in experience, equipment, and operational preferences. For silica nanoparticles synthesized by the Stöber method, agitation is commonly applied to promote homogeneous hydrolysis and condensation processes [
5], and mixing can be achieved using magnetic stirring or other laboratory devices such as shakers and rotators [
6,
7,
8]. In addition, some studies have introduced ultrasonic irradiation, including water-bath and probe sonication [
9,
10], to assist the Stöber reaction, whereas static conditions are also occasionally adopted for simplicity [
11]. Despite the widespread use of these different approaches, their effects on particle size distribution and reproducibility have not been systematically compared. As a result, it remains unclear whether different oscillation modes are functionally equivalent or whether certain methods inherently compromise size control. Addressing this gap is essential for improving the reproducibility and transferability of nanoparticle synthesis protocols across laboratories.
In this study, silica nanoparticles were selected as a model system due to their low cost, facile synthesis, spherical morphology, and widespread use in both academic and industrial settings [
12]. Their well-defined shape enables reliable particle size analysis by transmission electronic microscope (TEM) and hydrodynamic size measurement by dynamic light scattering (DLS). Five commonly available laboratory devices—magnetic stirrer, shaker, rotator, ultrasonic bath, and probe sonicator—were employed to generate seven distinct oscillation modes: stirring, horizontal shaking, vertical shaking, rotating, vertical shaking combined with rotating, water bath sonication, and probe sonication. A static condition without external agitation was included as a control. By quantitatively comparing particle size and PDI across these modes, this work aims to identify equivalent and non-recommended oscillation strategies for reproducible silica nanoparticle synthesis.
2. Materials and Methods
2.1. Materials and Apparatus
Ethanol, tetraethyl orthosilicate (TEOS), and aqueous ammonia solution (30 wt%) were purchased from Sigma-Aldrich (St. Louis, MO, USA). Deionized water was obtained from a laboratory water purification system. A magnetic stirrer and hot plate (Isotemp™ Hot Plate Stirrer, ambient to 400 °C, ceramic surface) were purchased from Fisher Scientific (Waltham, MA, USA). An orbital shaker (AC/DC input, 115 V AC) was obtained from Sigma-Aldrich. A rotator (ROTO-THERM™ Plus incubated rotator with variable speed and tube holders) was purchased from Benchmark Scientific (Sayreville, NJ, USA). An ultrasonic bath (Branson, 5.5 gal tank capacity) was obtained from Marshall Scientific (Hampton, NH, USA). A probe-type ultrasonic processor (130 W) was purchased from Sigma-Aldrich. Transmission electron microscopy (TEM) images were acquired using a Hitachi H-600 transmission electron microscope (Hitachi High-Technologies, Tokyo, Japan) operated at an accelerating voltage of 75 keV. Particle size distributions were analyzed from TEM images using ImageJ software (version 1.53t). Dynamic light scattering (DLS) measurements were performed using a Malvern Zetasizer (ZEN3690, Malvern Instruments, Malvern, UK) at a silica nanoparticle concentration of 10 μg/mL.
2.2. Synthesis of SiO2 Nanoparticles
Silica nanoparticles were synthesized via a modified Stöber method [
13]. Briefly, 8 mL of ethanol was mixed with 1 mL of ammonia to form solution A, while 0.1 mL of TEOS was dissolved in 0.9 mL of ethanol to form solution B. Solution B was added to solution A in one portion, followed by mixing under the specified oscillation mode.
For stirring, a magnetic stirrer was operated at 400 rpm. Horizontal shaking was performed using an orbital shaker at 100 rpm. Vertical shaking and rotating were conducted using a rotator in rocking and rotating modes, respectively, at a rotational speed of 20 rpm. Vertical shaking combined with rotating was achieved using a mixed oscillation mode. For water bath sonication, the reaction vessel was placed in an ultrasonic bath at 40 kHz and ~200 W, and the temperature was maintained near room temperature by adding ice as needed. For probe sonication, the reaction vessel was immersed in an ice-water bath, and sonication was applied in a pulsed mode (3 s on, 3 s off) at 20 kHz and 130 W while monitoring temperature. A static condition without external agitation was used as a control.
All reactions were carried out at room temperature for 30 min. The resulting nanoparticles were collected by centrifugation at 10,000 rpm for 10 min and washed twice with ethanol and once with deionized water. After each washing step, the particles were redispersed by vortex mixing. Each synthesis was performed in triplicate under identical conditions.
2.3. TEM Images Analysis
Particle size distributions were obtained from TEM images using ImageJ according to a previously reported protocol [
14]. Particles in contact with the image boundary or overlapping aggregates were excluded to ensure accurate statistical analysis.
3. Results
Representative TEM images of SiO
2 nanoparticles synthesized under different oscillation modes are shown in
Figure 1. In all cases, spherical silica nanoparticles were obtained, indicating that the modified Stöber reaction is robust with respect to the applied oscillation mode. However, quantitative analysis reveals that particle size uniformity and dispersion are strongly dependent on the specific mixing condition.
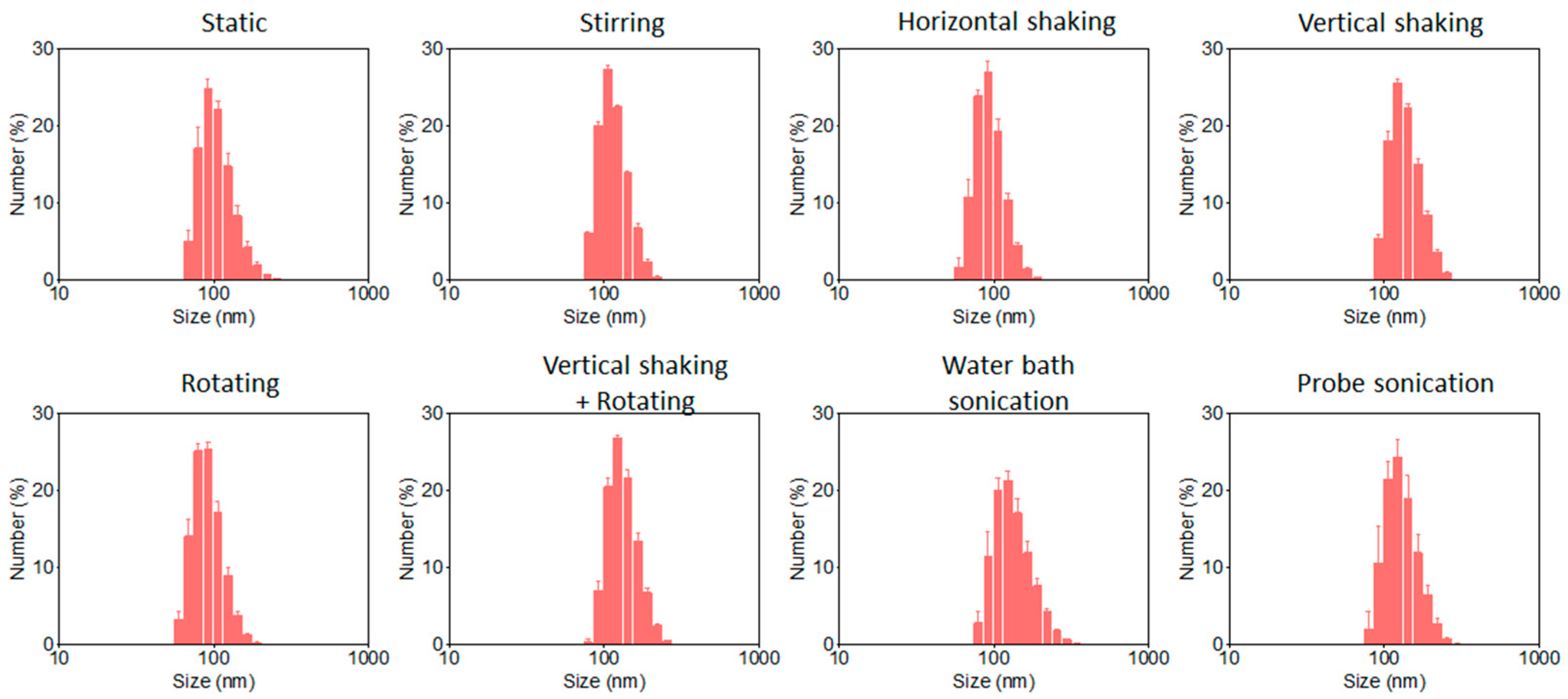
The particle size distributions derived from TEM images are summarized in
Figure 2, and the corresponding average particle sizes are listed in
Table 1. For stirring, horizontal shaking, vertical shaking, and rotating modes, the silica nanoparticles exhibited comparable average TEM diameters, ranging from approximately 87 to 92 nm, with relatively narrow size distributions. The corresponding PDI values obtained from DLS measurements were all below 0.03 (
Table 1 and
Figure 3), indicating highly uniform particle populations. Within experimental uncertainty, no statistically significant differences in average particle size or PDI were observed among these four oscillation modes, suggesting that they provide functionally equivalent mixing environments under the present synthesis conditions.
In contrast, samples synthesized under static conditions displayed a smaller average TEM particle size of approximately 78 nm, but with a markedly broader size distribution. This is reflected in a substantially higher PDI value of ~0.105 (
Table 1). These results indicate that insufficient mixing leads to heterogeneous growth conditions, likely due to locally non-uniform supersaturation, temporally distributed nucleation events, and diffusion-limited mass transport [
15,
16,
17], resulting in poor size uniformity despite the smaller mean particle size.
More pronounced deviations were observed for the vertical shaking combined with rotating mode as well as for water bath sonication and probe sonication. As summarized in
Table 1, these samples exhibited significantly larger average TEM particle sizes, typically exceeding 110 nm, accompanied by elevated PDI values. In particular, probe sonication at 20 kHz and 130 W produced the largest particles, with an average TEM diameter of approximately 144 nm and a PDI of ~0.083. Similarly, water bath sonication operated at ~40 kHz and ~200 W resulted in broadened size distributions. The corresponding size distributions (
Figure 2) were not strictly bimodal but instead showed broadened profiles with extended tails toward larger particle sizes, indicating less controlled nucleation and growth processes. In contrast, moderate agitation modes, including magnetic stirring (400 rpm), horizontal shaking (100 rpm), vertical shaking (20 rpm), and rotating (20 rpm), provided more homogeneous mixing environments and narrower size distributions. These observations suggest that excessive or complex energy input, particularly under high-frequency and high-power ultrasonic conditions, can disrupt the balance between nucleation and growth during the Stöber reaction, leading to reduced size control.
Dynamic light scattering measurements further support these observations. As shown in
Figure 4 and summarized in
Table 1, the hydrodynamic diameters measured by DLS were consistently larger than the particle sizes determined by TEM, with values typically in the range of 110–170 nm, depending on the oscillation mode. This difference is mainly attributed to the hydrodynamic nature of DLS measurements, which includes the solvation layer surrounding the particles, bias toward larger particles (I ∝ d
6) [
18,
19,
20]. In addition, occasional weakly associated clusters observed in TEM images may further contribute to this discrepancy, even when relatively low PDI values are obtained. Importantly, although the absolute sizes differed between TEM and DLS measurements, the relative trends across different oscillation modes were consistent. Oscillation modes that produced uniform particles in TEM analysis also exhibited low PDI values and narrower hydrodynamic size distributions in DLS measurements, confirming the robustness of the observed trends.
Taken together, the TEM-based size analysis, DLS measurements, and PDI evaluation (
Table 1) demonstrate that the oscillation mode plays a critical role in determining particle size uniformity during silica nanoparticle synthesis. Moderate and continuous oscillation modes provide reproducible and uniform particle growth, whereas insufficient or overly intense mixing leads to broader size distributions and reduced reproducibility.
4. Conclusions
In this study, the effect of oscillation mode on the synthesis of SiO2 nanoparticles via a modified Stöber method was systematically evaluated using TEM and DLS characterization. While spherical silica nanoparticles were successfully obtained under all tested conditions, quantitative analysis revealed clear differences in particle size and dispersion behavior depending on the applied oscillation mode.
Stirring, horizontal shaking, vertical shaking, and rotating produced silica nanoparticles with comparable average TEM sizes of approximately 87–92 nm and consistently low PDI values below 0.03, indicating that these oscillation modes provide equivalent and reliable mixing conditions for reproducible particle synthesis. In contrast, static conditions resulted in poorer size uniformity, as evidenced by a higher PDI of ~0.105, despite yielding smaller average particle sizes. More complex or intense oscillation modes—including vertical shaking combined with rotating, water bath sonication, and probe sonication—led to significantly larger particles (typically >110 nm) and broader size distributions, with probe sonication producing the largest particles and one of the highest PDI values.
In the present study, spherical silica nanoparticles with an average diameter of approximately 100 nm were selected as a representative and well-established model system. The observed influence of oscillation modes is primarily governed by mixing efficiency, local concentration homogeneity, and hydrodynamic shear during nucleation and growth, which are fundamental factors in many colloidal synthesis processes. Therefore, similar qualitative trends may also be expected for silica nanoparticles with different size ranges and for other nanoparticle systems synthesized via solution-based routes. Nevertheless, quantitative outcomes may vary depending on particle composition, surface chemistry, growth kinetics, and target size regime. For example, larger particles may be more sensitive to sedimentation and secondary aggregation, whereas smaller particles may be more strongly affected by early-stage nucleation dynamics. Further systematic investigations on different particle sizes, morphologies, and material systems will be required to fully establish the general applicability of the present findings.
These results demonstrate that increasing mixing intensity does not necessarily improve size control in silica nanoparticle synthesis. Instead, moderate and continuous oscillation modes are sufficient to achieve uniform particle growth, whereas insufficient or excessive energy input can disrupt controlled nucleation and growth processes. The quantitative comparisons summarized in
Table 1 provide practical process-level guidance for selecting appropriate oscillation modes in laboratory-scale silica nanoparticle synthesis when particle size uniformity and reproducibility are critical considerations.