Release of Dissolved Organic Matter from Sludge Biochar and Its Spectral Characteristics in Different Environmental Media
Abstract
1. Introduction
2. Materials and Methods
2.1. Sludge Source and Biochar Preparation
2.2. Preparation of Extraction Solutions
2.3. DOM Extraction Experiment
2.4. Analytical Method
2.5. Statistical Analysis
3. Results and Discussion
3.1. Release and Characteristic Indices of DOM in Sludge Biochar at Different Pyrolysis Temperatures
3.2. Fluorescence Characteristics of DOM in Sludge Biochar at Different Pyrolysis Temperatures
3.2.1. Released DOM Components Analysis of Biochar
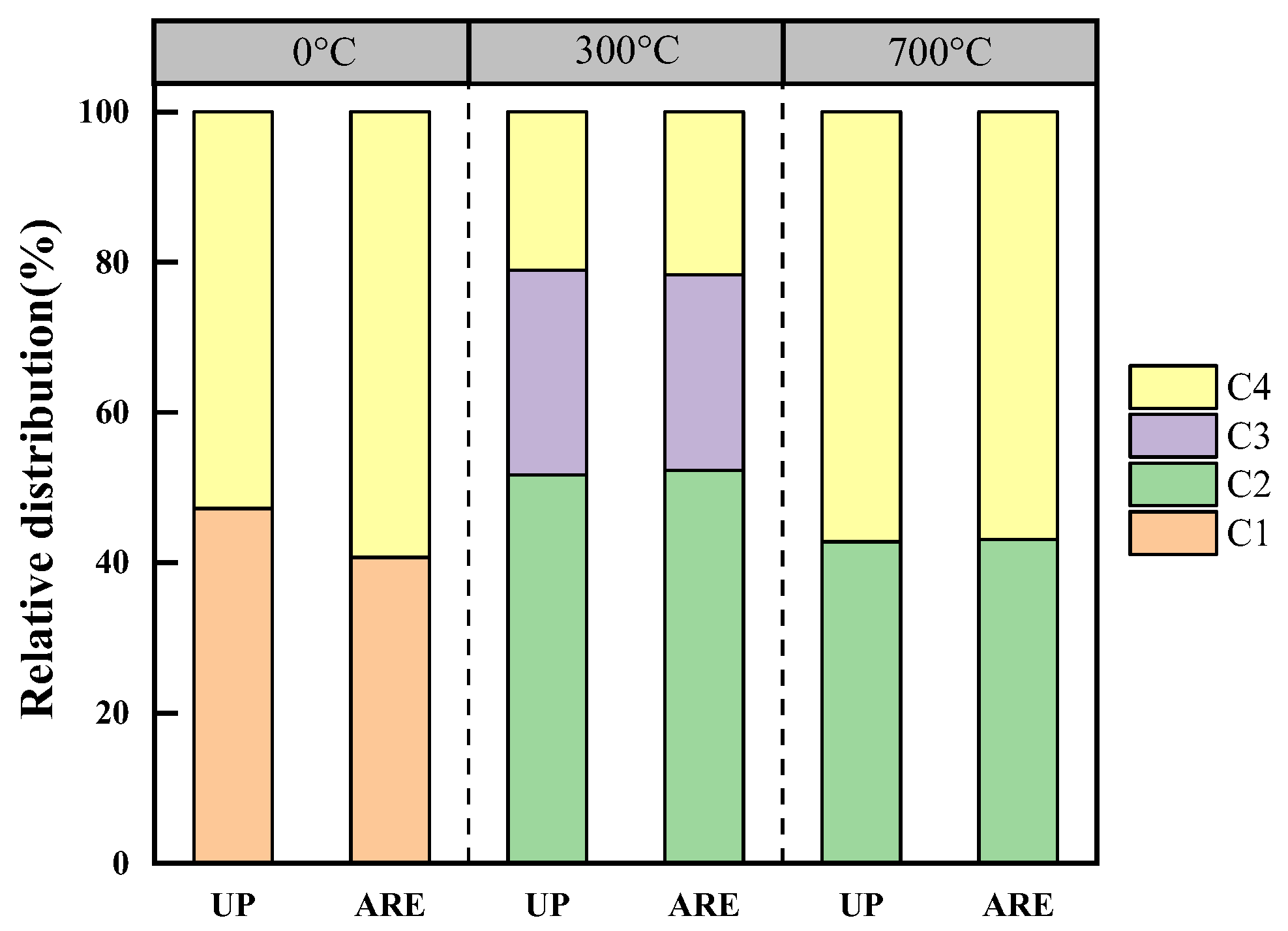
3.2.2. Distribution of DOM Components in Biochar at Different Pyrolysis Temperatures
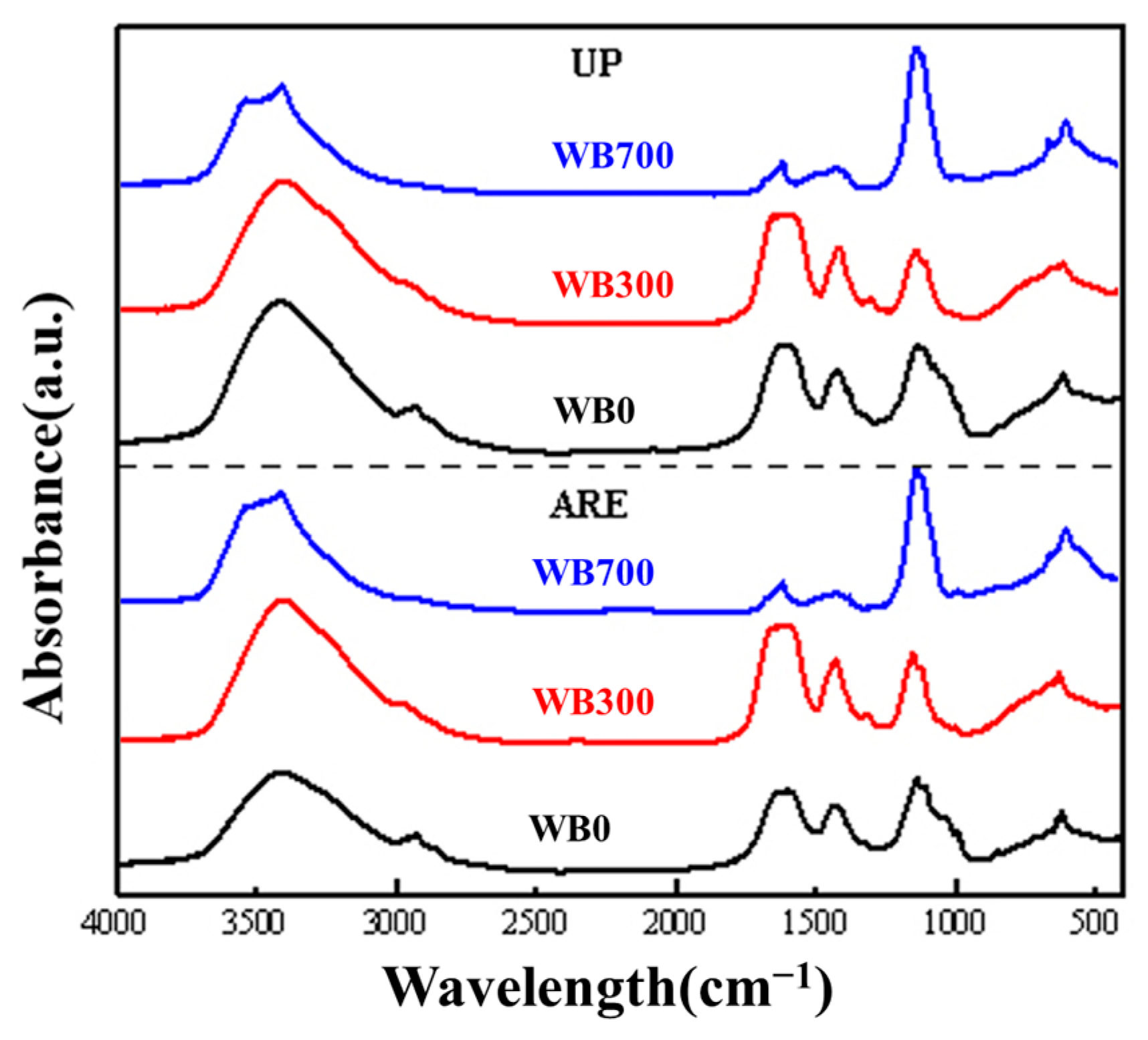
3.3. FTIR Spectroscopy and 2D-COS Analysis
3.4. Ultraviolet-Visible Spectroscopy Analysis of DOM in Sludge Biochar
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Li, M.; Zhang, A.F.; Wu, H.M.; Liu, H.; Lv, J.L. Predicting potential release of dissolved organic matter from biochars derived from agricultural residues using fluorescence and ultraviolet absorbance. J. Hazard. Mater. 2017, 334, 86–92. [Google Scholar] [CrossRef]
- Bolan, N.S.; Adriano, D.C.; Kunhikrishnan, A.; James, T.; McDowell, R.; Senesi, N. Dissolved organic matter: Biogeochemistry, dynamics, and environmental significance in soils. In Advances in Agronomy; Sparks, D.L., Ed.; Elsevier Academic Press Inc.: San Diego, CA, USA, 2011; Volume 110, pp. 1–75. [Google Scholar]
- Zhang, S.Q.; Wang, M.X.; Liu, J.; Tian, S.Y.; Yang, X.L.; Xiao, G.Q.; Xu, G.M.; Jiang, T.; Wang, D.Y. Biochar affects methylmercury production and bioaccumulation in paddy soils: Insights from soil-derived dissolved organic matter. J. Environ. Sci. 2022, 119, 68–77. [Google Scholar] [CrossRef]
- Li, J.E.; Li, Q.A.; Liang, S.Q.; Zhang, B.Y.; Wang, J.Z.; Yu, Y.X.; Xia, L.Z.; Zhang, Y.; Wang, Y.F. Aging mechanism of biochar based on fluorescence spectroscopy: Assessing soil dissolved organic matter (DOM) dynamics and Cd bioavailability. Chem. Eng. J. 2025, 505, 11. [Google Scholar] [CrossRef]
- Sun, Y.Q.; Xiong, X.N.; He, M.J.; Xu, Z.B.; Hou, D.Y.; Zhang, W.H.; Ok, Y.S.; Rinklebe, J.; Wang, L.L.; Tsang, D.C.W. Roles of biochar-derived dissolved organic matter in soil amendment and environmental remediation: A critical review. Chem. Eng. J. 2021, 424, 14. [Google Scholar] [CrossRef]
- Kim, H.B.; Kim, J.G.; Alessi, D.S.; Baek, K. Mechanistic understanding of copper (Cu) mobility in aged biochar: Influence of dissolved organic matters release and oxygen-containing groups. J. Hazard. Mater. 2025, 495, 10. [Google Scholar] [CrossRef]
- Zimmerman, A.R.; Ouyang, L. Priming of pyrogenic C (biochar) mineralization by dissolved organic matter and vice versa. Soil Biol. Biochem. 2019, 130, 105–112. [Google Scholar] [CrossRef]
- Devault, M.; Jing, L.; Arkoun, M.; Bauerle, T.; Kessler, A.; Lehmann, J. Biochar shifts balance between hydrophilic and lipophilic molecules in root exudates. Bioresour. Technol. 2025, 427, 5. [Google Scholar] [CrossRef]
- He, C.J.; He, X.W.; Li, J.J.; Luo, Y.; Li, J.C.; Pei, Y.; Jiang, J.Y. The spectral characteristics of biochar-derived dissolved organic matter at different pyrolysis temperatures. J. Environ. Chem. Eng. 2021, 9, 9. [Google Scholar] [CrossRef]
- Intani, K.; Latif, S.; Kabir, A.; Müller, J. Effect of self-purging pyrolysis on yield of biochar from maize cobs, husks and leaves. Bioresour. Technol. 2016, 218, 541–551. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.R.; Chen, W.P.; Fang, S.; Xu, Z.H.; Weng, H.F.; Zhang, X.D. The Influence of Pyrolysis Temperature and Feedstocks on the Characteristics of Biochar-Derived Dissolved Organic Matter: A Systematic Assessment. Clean Technol. 2024, 6, 1314–1325. [Google Scholar] [CrossRef]
- Hur, J.; Lee, B.M. Characterization of binding site heterogeneity for copper within dissolved organic matter fractions using two-dimensional correlation fluorescence spectroscopy. Chemosphere 2011, 83, 1603–1611. [Google Scholar] [CrossRef]
- Lin, H.; Guo, L.D. Variations in Colloidal DOM Composition with Molecular Weight within Individual Water Samples as Characterized by Flow Field-Flow Fractionation and EEM-PARAFAC Analysis. Environ. Sci. Technol. 2020, 54, 1657–1667. [Google Scholar] [CrossRef]
- He, W.; Hur, J. Conservative behavior of fluorescence EEM-PARAFAC components in resin fractionation processes and its applicability for characterizing dissolved organic. Water Res. 2015, 83, 217–226. [Google Scholar] [CrossRef]
- Cui, H.Y.; Xie, L.N.; Zhang, G.G.; Zhao, Y.; Wei, Z.M. Revealing the Inner Changes of Component Composition Derived from DOM PARAFAC Based on Two-Dimensional Correlation Spectroscopy. Molecules 2022, 27, 7316. [Google Scholar] [CrossRef]
- Saison, C.; Perrin-Ganier, C.; Amellal, S.; Morel, J.L.; Schiavon, M. Effect of metals on the adsorption and extractability of 14C-phenanthrene in soils. Chemosphere 2004, 55, 477–485. [Google Scholar] [CrossRef]
- Yan, C.X.; Wang, W.Y.; Nie, M.H.; Ding, M.J.; Wang, P.; Zhang, H.; Huang, G.X. Characterization of copper binding to biochar-derived dissolved organic matter: Effects of pyrolysis temperature and natural wetland plants. J. Hazard. Mater. 2023, 442, 13. [Google Scholar] [CrossRef] [PubMed]
- Song, F.H.; Li, T.T.; Shi, Q.; Guo, F.; Bai, Y.C.; Wu, F.C.; Xing, B.S. Novel Insights into the Molecular-Level Mechanism Linking the Chemical Diversity and Copper Binding Heterogeneity of Biochar-Derived Dissolved Black Carbon and Dissolved Organic Matter. Environ. Sci. Technol. 2021, 55, 11624–11636. [Google Scholar] [CrossRef]
- Xing, J.; Li, L.C.; Li, G.B.; Xu, G.R. Feasibility of sludge-based biochar for soil remediation: Characteristics and safety performance of heavy metals influenced by pyrolysis temperatures. Ecotox. Environ. Safe. 2019, 180, 457–465. [Google Scholar] [CrossRef] [PubMed]
- Ling, W.T.; Ren, L.L.; Gao, Y.Z.; Zhu, X.Z.; Sun, B.Q. Impact of low-molecular-weight organic acids on the availability of phenanthrene and pyrene in soil. Soil Biol. Biochem. 2009, 41, 2187–2195. [Google Scholar] [CrossRef]
- Vause, D.; Heaney, N.; Lin, C.X. Differential release of sewage sludge biochar-borne elements by common low-molecular-weight organic acids. Ecotox. Environ. Safe. 2018, 165, 219–223. [Google Scholar] [CrossRef]
- Sun, Y.H.; Guan, F.; Yang, W.W.; Wang, F.Y. Removal of Chromium from a Contaminated Soil Using Oxalic Acid, Citric Acid, and Hydrochloric Acid: Dynamics, Mechanisms, and Concomitant Removal of Non-Targeted Metals. Int. J. Environ. Res. Public Health 2019, 16, 2771. [Google Scholar] [CrossRef]
- McBeath, A.V.; Wurster, C.M.; Bird, M.I. Influence of feedstock properties and pyrolysis conditions on biochar carbon stability as determined by hydrogen pyrolysis. Biomass Bioenerg. 2015, 73, 155–173. [Google Scholar] [CrossRef]
- Murtaza, G.; Usman, M.; Iqbal, J.; Hyder, S.; Solangi, F.; Iqbal, R.; Okla, M.K.; Al-Ghamdi, A.A.; Elsalahy, H.H.; Tariq, W.; et al. Liming potential and characteristics of biochar produced from woody and non-woody biomass at different pyrolysis temperatures. Sci. Rep. 2024, 14, 12. [Google Scholar] [CrossRef]
- Wu, H.M.; Qi, Y.S.; Dong, L.; Zhao, X.; Liu, H. Revealing the impact of pyrolysis temperature on dissolved organic matter released from the biochar prepared from Typha orientalis. Chemosphere 2019, 228, 264–270. [Google Scholar] [CrossRef] [PubMed]
- Du, Z.L.; Hu, A.B.; Wang, Q.D.; Ai, J.; Zhang, W.J.; Liang, Y.; Cao, M.X.; Wu, H.J.; Wang, D.S. Molecular composition and biotoxicity effects of dissolved organic matters in sludge-based carbon: Effects of pyrolysis temperature. J. Hazard. Mater. 2022, 424, 11. [Google Scholar] [CrossRef]
- Wang, Y.L.; Yang, C.M.; Zou, L.M.; Cui, H.Z. Spatial Distribution and Fluorescence Properties of Soil Dissolved Organic Carbon Across a Riparian Buffer Wetland in Chongming Island, China. Pedosphere 2015, 25, 220–229. [Google Scholar] [CrossRef]
- Zhang, A.F.; Zhou, X.; Li, M.; Wu, H.M. Impacts of biochar addition on soil dissolved organic matter characteristics in a wheat-maize rotation system in Loess Plateau of China. Chemosphere 2017, 186, 986–993. [Google Scholar] [CrossRef] [PubMed]
- Cai, X.L.; Lei, S.H.; Li, Y.M.; Li, J.Z.; Xu, J.; Lyu, H.; Li, J.D.; Dong, X.Z.; Wang, G.L.; Zeng, S. Humification levels of dissolved organic matter in the eastern plain lakes of China based on long-term satellite observations. Water Res. 2024, 250, 14. [Google Scholar] [CrossRef]
- Fan, J.X.; Duan, T.; Zou, L.; Sun, J.X. Characteristics of dissolved organic matter composition in biochar: Effects of feedstocks and pyrolysis temperatures. Environ. Sci. Pollut. Res. 2023, 30, 85139–85153. [Google Scholar] [CrossRef] [PubMed]
- Gui, X.Y.; Liu, C.; Li, F.Y.; Wang, J.F. Effect of pyrolysis temperature on the composition of DOM in manure-derived biochar. Ecotox. Environ. Safe. 2020, 197, 10. [Google Scholar] [CrossRef]
- Yang, L.; Chang, S.W.; Shin, H.S.; Hur, J. Tracking the evolution of stream DOM source during storm events using end member mixing analysis based on DOM quality. J. Hydrol. 2015, 523, 333–341. [Google Scholar] [CrossRef]
- Helms, J.R.; Stubbins, A.; Ritchie, J.D.; Minor, E.C.; Kieber, D.J.; Mopper, K. Absorption spectral slopes and slope ratios as indicators of molecular weight, source, and photobleaching of chromophoric dissolved organic matter. Limnol. Oceanogr. 2008, 53, 955–969. [Google Scholar] [CrossRef]
- Osburn, C.L.; Stedmon, C.A. Linking the chemical and optical properties of dissolved organic matter in the Baltic-North Sea transition zone to differentiate three allochthonous inputs. Mar. Chem. 2011, 126, 281–294. [Google Scholar] [CrossRef]
- Chen, M.L.; Price, R.M.; Yamashita, Y.; Jaffé, R. Comparative study of dissolved organic matter from groundwater and surface water in the Florida coastal Everglades using multi-dimensional spectrofluorometry combined with multivariate statistics. Appl. Geochem. 2010, 25, 872–880. [Google Scholar] [CrossRef]
- Zhang, X.X.; Xu, Z.B.; Sun, Y.Q.; Mohanty, S.K.; Lei, H.W.; Khan, E.; Tsang, D.C.W. Implications of Pyrolytic Gas Dynamic Evolution on Dissolved Black Carbon Formed During Production of Biochar from Nitrogen-Rich Feedstock. Environ. Sci. Technol. 2025, 59, 2699–2710. [Google Scholar] [CrossRef] [PubMed]
- Gao, J.K.; Shi, Z.Y.; Wu, H.M.; Lv, J.L. Fluorescent characteristics of dissolved organic matter released from biochar and paddy soil incorporated with biochar. RSC Adv. 2020, 10, 5785–5793. [Google Scholar] [CrossRef]
- Smebye, A.; Ailing, V.; Vogt, R.D.; Gadmar, T.C.; Mulder, J.; Cornelissen, G.; Hale, S.E. Biochar amendment to soil changes dissolved organic matter content and composition. Chemosphere 2016, 142, 100–105. [Google Scholar] [CrossRef]
- Zhang, H.Y.; Ni, J.Z.; Qian, W.; Yu, S.H.; Xiang, Y.; Yang, L.M.; Chen, W.F. Pyrolysis atmospheres and temperatures co-mediated spectral variations of biochar-derived dissolved organic carbon: Quantitative prediction and self-organizing maps analysis. Molecules 2023, 28, 2247. [Google Scholar] [CrossRef] [PubMed]





| Component | EEM Wavelength (nm) | EEM Equipotential Surface | Spectrum Characteristic |
|---|---|---|---|
| C1 | Ex: 235/275 Em: 416 |  | 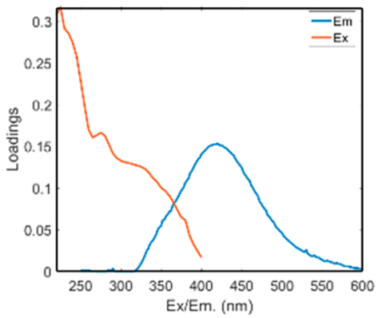 |
| C2 | Ex: 225/320 Em: 390 |  |  |
| C3 | Ex: 225/365 Em: 438 |  |  |
| C4 | Ex: 225/280 Em: 304 |  |  |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Li, B.; Liao, J.; Wen, H.; Ma, L.; Li, B.; Song, W.; Fu, C. Release of Dissolved Organic Matter from Sludge Biochar and Its Spectral Characteristics in Different Environmental Media. Processes 2026, 14, 595. https://doi.org/10.3390/pr14040595
Li B, Liao J, Wen H, Ma L, Li B, Song W, Fu C. Release of Dissolved Organic Matter from Sludge Biochar and Its Spectral Characteristics in Different Environmental Media. Processes. 2026; 14(4):595. https://doi.org/10.3390/pr14040595
Chicago/Turabian StyleLi, Bowen, Jianjun Liao, Hao Wen, Lincheng Ma, Bin Li, Wei Song, and Caixia Fu. 2026. "Release of Dissolved Organic Matter from Sludge Biochar and Its Spectral Characteristics in Different Environmental Media" Processes 14, no. 4: 595. https://doi.org/10.3390/pr14040595
APA StyleLi, B., Liao, J., Wen, H., Ma, L., Li, B., Song, W., & Fu, C. (2026). Release of Dissolved Organic Matter from Sludge Biochar and Its Spectral Characteristics in Different Environmental Media. Processes, 14(4), 595. https://doi.org/10.3390/pr14040595








