Theranostic Iron Oxide Nanoparticles for Controlled Oxaliplatin Release Under Simulated Circulation and Cytotoxicity Evaluation in Colorectal Cancer Cell Lines
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals and Materials
2.2. Preparation of Mesoporous Fe3O4@MSN-NH2 Nanoparticles
2.3. Preparation of Fe3O4@MSN-NH2/FA in the Dark
2.4. Drug Loading (Fe3O4@MSN-NH2/OXA and Fe3O4@MSN-NH2/FA/OXA)
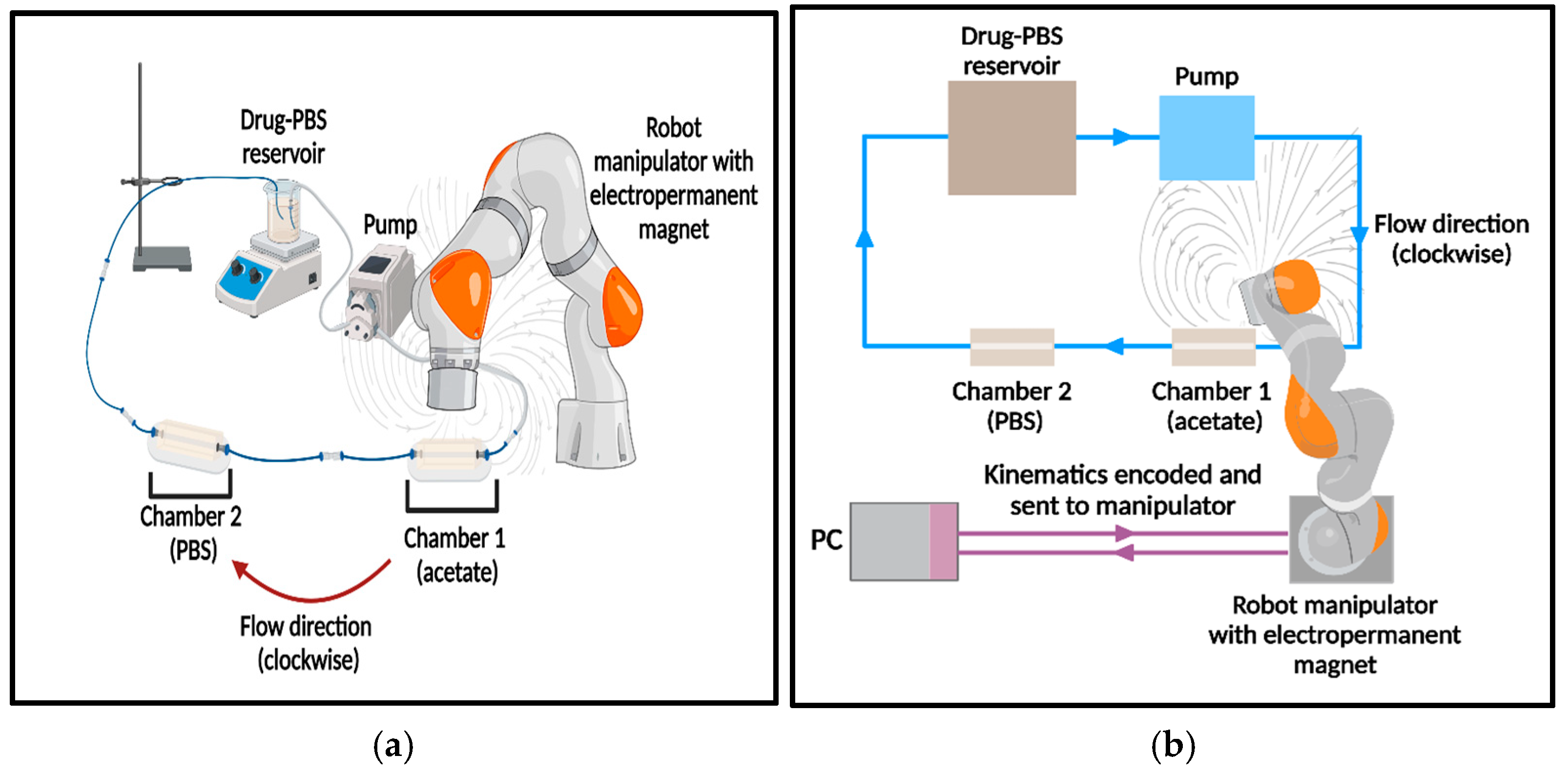
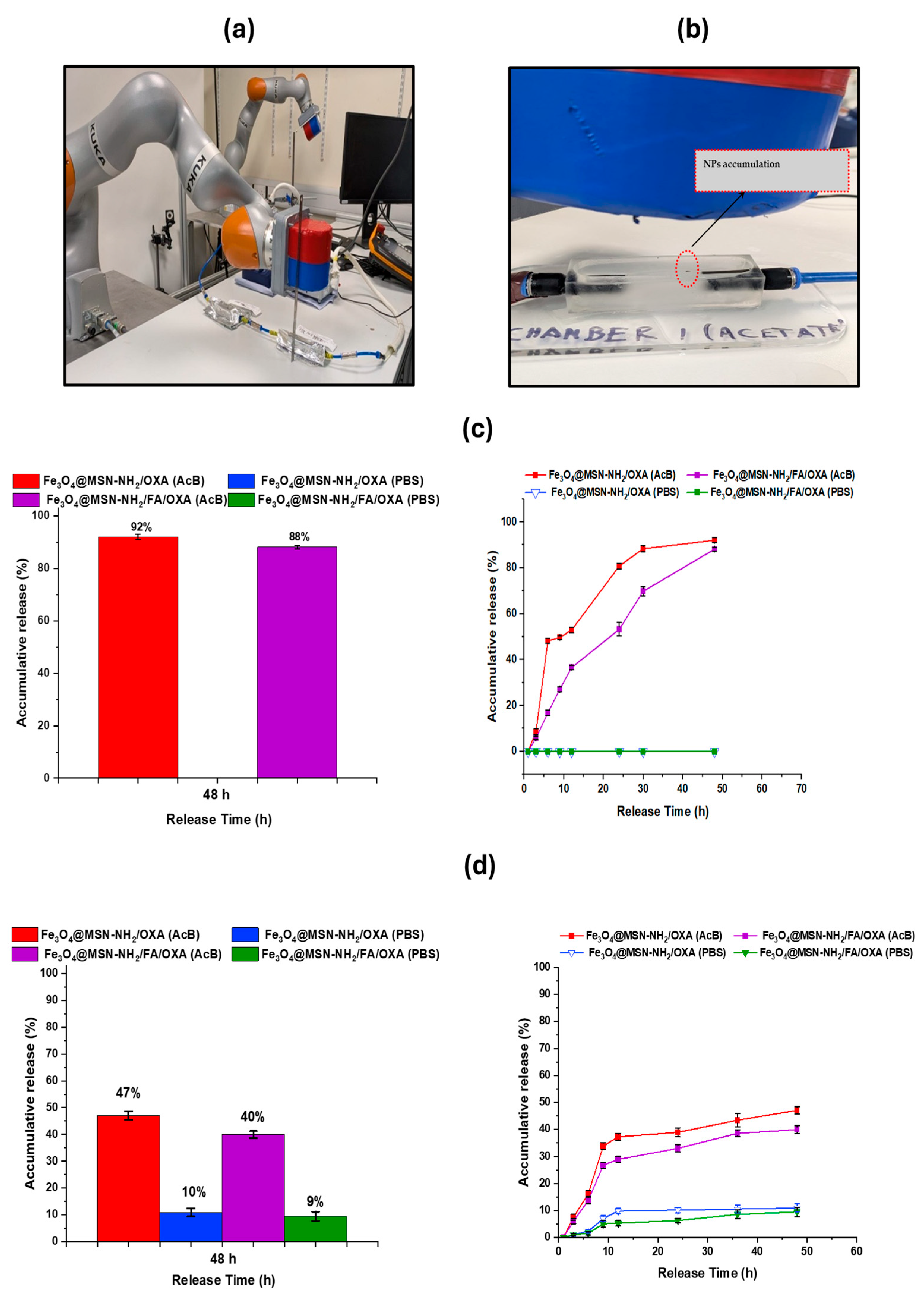
2.5. External Magnetic Robot-Assisted Localized Release of Oxaliplatin
2.6. Characterizations
2.7. FOLR1 Gene Expression Analysis
2.8. MTT Cytotoxicity Assay
3. Results and Discussion
3.1. Physicochemical Characterization of Nanoparticles
3.1.1. FT-IR Spectroscopy
3.1.2. Powder X-Ray Diffraction (PXRD)
3.1.3. Thermogravimetric and Derivative Thermogravimetric Analysis
3.1.4. X-Ray Photoelectron Spectroscopy
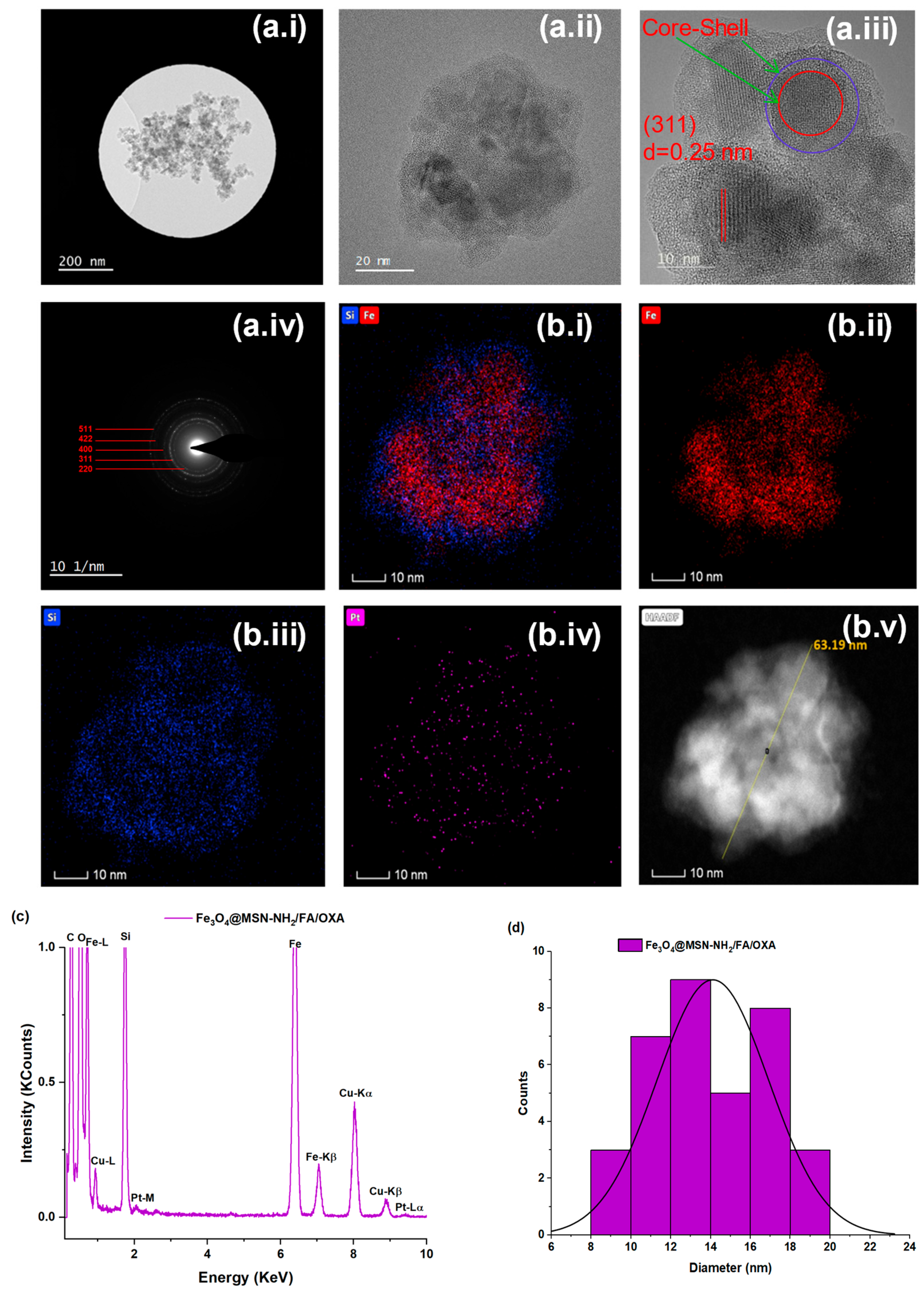
3.1.5. Transmission Electron Microscopy
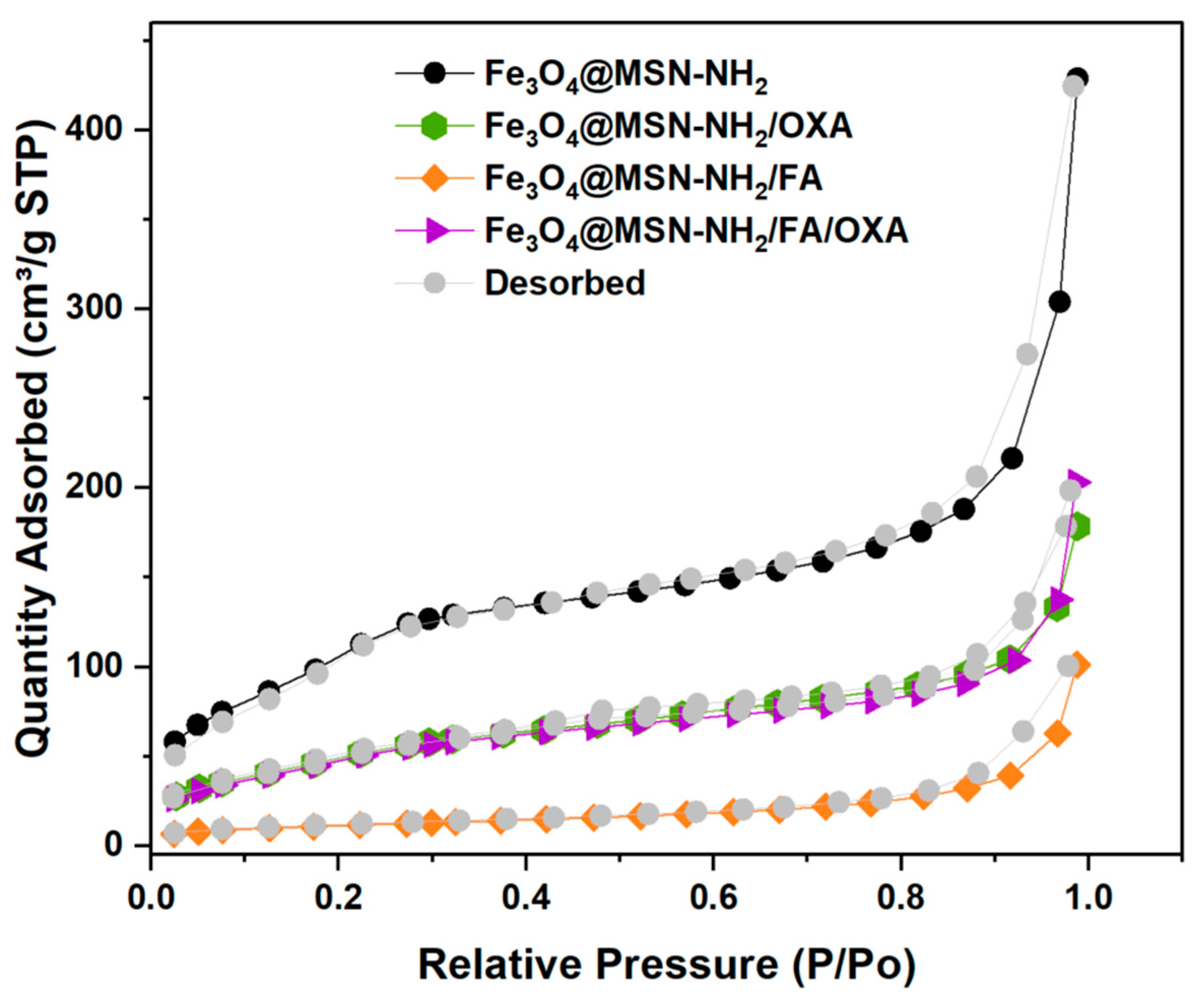
3.1.6. Brunauer–Emmett–Teller Surface Area and Porosity Analysis
3.1.7. Dispersion Stability and Colloidal Properties
3.1.8. Dynamic Light Scattering and Zeta Potential
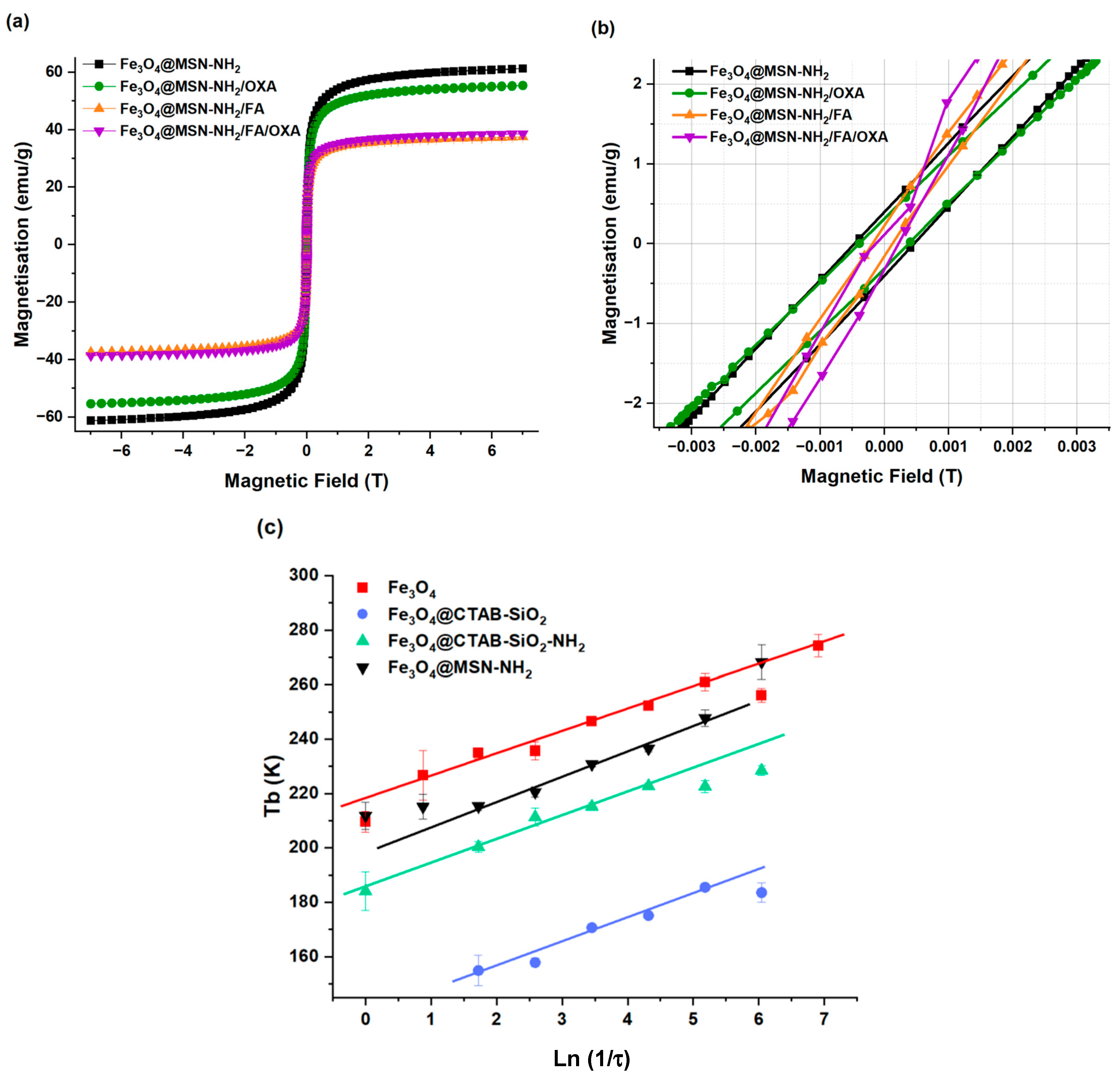
3.2. Magnetic Properties
3.2.1. Magnetization and Superparamagnetic Properties
3.2.2. Frequency Dependence of the Blocking Temperature
3.3. MRI Analysis
3.4. Drug Loading Capacity
3.5. Drug Release with Magnetic Robot Actuated/Without Magnetic Robot
3.5.1. Overview of Release Profile and System Setup
| Plastic tubing (D = 0.005 m) | A ≈ 1.96 × 10−5 m2, v = Q/A ≈ 0.204 m s−1, Re = 1020 |
| Dialysis section (D = 0.007 m) | A ≈ 3.85 × 10−5 m2, v = Q/A ≈ 0.104 m s−1, Re = 728 |
3.5.2. MRP-Assisted Release Profiles
3.5.3. Release Profiles Without MRP
3.5.4. Mechanistic Interpretation
3.5.5. Contribution of MRP Navigation
3.5.6. Comparative Analysis with Literature
3.6. Proof of Principle of Targeted Cytotoxicity In Vitro
3.6.1. FOLR1 Gene Expression in Colorectal Cancer Cell Lines
3.6.2. Cytotoxicity of Functionalized Nanoparticles
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AcB | Acetate buffer |
| APTES | 3-aminopropyltriethoxysilane |
| ACX″ | Imaginary component of AC susceptibility |
| ATR | Attenuated total reflection |
| BET | Brunauer–Emmett–Teller |
| Tb | Blocking temperature |
| Caco2 | Cancer Coli, colon cancer |
| CDR | Controlled drug release |
| CTAB | Cetyltrimethylammonium bromide |
| DAD | Diode array detector |
| DH | Hydrodynamic size |
| DL | Drug release |
| DLS | Dynamic light scattering |
| DMSO | Dimethyl sulfoxide |
| DOX | Doxorubicin |
| DTG | Derivative thermogravimetry |
| EDCI | 1-ethyl-3-(3-dimethylaminopropyl) carbodiimide |
| EDX | Energy dispersive x-ray spectroscopy |
| EE | Encapsulation efficiency |
| EPM | Electro-permanent magnetic |
| EtOH | Ethanol |
| FA | Folic acid |
| FCS | Fetal calf serum |
| FOLR1high | Folate receptor alpha high |
| FOLR1low | Folate receptor alpha low |
| FR | Folate receptors |
| FT-IR | Fourier transfer inferred spectroscopy |
| FWHM | Full width at half maximum |
| HAADF | High annular dark-field imaging |
| HPLCW | HPLC grade water |
| HPLC-UV | High-performance liquid chromatography with ultraviolet detection |
| HR-TEM | High-resolution transmission electron microscopy |
| ICP-OES | inductively coupled plasma optical emission spectrometer |
| MRP | Magnetic robot actuated platform |
| MNP | Magnetic nanoparticle |
| MRI | Magnetic resonance imaging |
| MSN | Mesoporous silica nanoparticles |
| NH2 | Amine groups |
| NH4NO3 | Ammonium nitrate |
| NHS | N-hydroxysuccinimide |
| NMP | N-Methyl-2-pyrrolidone |
| NPs | Nanoparticles |
| OXA | Oxaliplatin |
| PBS | Phosphate-buffered solution |
| Pt | Platinum |
| PXRD | Powder X-ray diffraction |
| RES | Reticuloendothelial system |
| ROI | Region-of-interest |
| RT-qPCR | Reverse Transcription Quantitative Polymerase Chain Reaction |
| RT | Retention time |
| S | Section |
| SAED | Selected area diffraction pattern |
| SI | Supplementary Information |
| SiO2 | Silicon dioxide |
| SQID-VSM | Superconducting quantum interference device vibrating sample magnetometer |
| T | Tesla |
| TDD | Targeted drug delivery |
| TEOS | Tetraethyl orthosilicate |
| TGA/DSC | Thermogravimetric and differential scanning calorimetry |
| TME | Tumor microenvironment |
| TFA | Trifluoracetic acid |
| WET | WET colloidal suspension |
| XPS | X-ray photoelectron spectroscopy |
| UHV | Ultra-high Vacuum |
| ZP | Zeta potential |
References
- Yu, C.; Wang, Z.; Sun, Z.; Zhang, L.; Zhang, W.; Xu, Y.; Zhang, J.J. Platinum-based combination therapy: Molecular rationale, current clinical uses, and future perspectives. J. Med. Chem. 2020, 63, 13397–13412. [Google Scholar] [CrossRef]
- Anand, U.; Dey, A.; Singh Chandel, A.K.; Sanyal, R.; Mishra, A.; Pandey, D.K.; De Falco, V.; Upadhyay, A.P.; Kandimalla, R.; Chaudhary, A.; et al. Cancer chemotherapy and beyond: Current status, drug candidates, associated risks and progress in targeted therapeutics. Genes Dis. 2022, 10, 1367–1401. [Google Scholar] [CrossRef]
- Rothenberg, M.L.; Oza, A.M.; Bigelow, R.H.; Berlin, J.D.; Marshall, J.L.; Ramanathan, R.K.; Hart, L.L.; Gupta, S.; Garay, C.A.; Burger, B.; et al. Superiority of oxaliplatin and fluorouracil-leucovorin compared with either therapy alone in patients with progressive colorectal cancer after irinotecan and fluorouracil-leucovorin: Interim results of a phase III trial. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2003, 21, 2059–2069. [Google Scholar] [CrossRef]
- Alcindor, T.; Beauger, N. Oxaliplatin: A review in the era of molecularly targeted therapy. Curr. Oncol. 2011, 18, 18–25. [Google Scholar] [CrossRef]
- Yothers, G.; O’connell, M.J.; Allegra, C.J.; Kuebler, J.P.; Colangelo, L.H.; Petrelli, N.J.; Wolmark, N. Oxaliplatin as adjuvant therapy for colon cancer: Updated results of NSABP C-07 trial, including survival and subset analyses. J. Clin. Oncol. 2011, 29, 3768–3774. [Google Scholar] [CrossRef]
- Mauri, G.; Gori, V.; Bonazzina, E.; Amatu, A.; Tosi, F.; Bencardino, K.; Ruggieri, L.; Patelli, G.; Arena, S.; Bardelli, A.; et al. Oxaliplatin retreatment in metastatic colorectal cancer: Systematic review and future research opportunities. Cancer Treat. Rev. 2020, 91, 102112. [Google Scholar] [CrossRef]
- Grolleau, F.; Gamelin, L.; Boisdron-Celle, M.; Lapied, B.; Pelhate, M.; Gamelin, E.C. A possible explanation for a neurotoxic effect of the anticancer agent oxaliplatin on neuronal voltage-gated sodium channels. J. Neurophysiol. 2001, 85, 2293–2297. [Google Scholar] [CrossRef]
- Webster, R.G.; Brain, K.L.; Wilson, R.H.; Grem, J.L.; Vincent, A. Oxaliplatin induces hyperexcitability at motor and autonomic neuromuscular junctions through effects on voltage-gated sodium channels. Br. J. Pharmacol. 2005, 146, 1027–1039. [Google Scholar] [CrossRef]
- Han, C.H.; Khwaounjoo, P.; Hill, A.G.; Miskelly, G.M.; McKeage, M.J. Predicting effects on oxaliplatin clearance: In vitro, kinetic and clinical studies of calcium- and magnesium-mediated oxaliplatin degradation. Sci. Rep. 2017, 7, 4073. [Google Scholar] [CrossRef]
- Gan, L.; Liu, Z.; Sun, C. Obesity linking to hepatocellular carcinoma: A global view. Biochim. Biophys. Acta (BBA) Rev. Cancer 2018, 1869, 97–102. [Google Scholar] [CrossRef]
- Wu, Y.; Liang, Y.; Liu, Y.D.; Hao, Y.; Tao, N.; Li, J.; Sun, X.; Zhou, M.; Liu, Y. A Bi2S3-embedded gellan gum hydrogel for localized tumor photothermal/antiangiogenic therapy. J. Mater. Chem. B 2021, 9, 3224–3234. [Google Scholar] [CrossRef] [PubMed]
- Zafar, H.; Raza, F.; Ma, S.; Wei, Y.; Zhang, J.; Shen, Q. Recent progress on nanomedicine-induced ferroptosis for cancer therapy. Biomater. Sci. 2021, 9, 5092–5115. [Google Scholar] [CrossRef]
- Sun, Q.; Zhou, Z.; Qiu, N.; Shen, Y. Rational Design of Cancer Nanomedicine: Nanoproperty Integration and Synchronization. Adv. Mater. 2017, 29, 1606628. [Google Scholar] [CrossRef]
- Feng, C.; Wang, Y.; Xu, J.; Zheng, Y.; Zhou, W.; Wang, Y.; Luo, C. Precisely Tailoring Molecular Structure of Doxorubicin Prodrugs to Enable Stable Nanoassembly, Rapid Activation, and Potent Antitumor Effect. Pharmaceutics 2024, 16, 1582. [Google Scholar] [CrossRef]
- Wang, Y.; Xu, Y.; Song, J.; Liu, X.; Liu, S.; Yang, N.; Wang, L.; Liu, Y.; Zhao, Y.; Zhou, W.; et al. Tumor Cell-Targeting and Tumor Microenvironment–Responsive Nanoplatforms for the Multimodal Imaging-Guided Photodynamic/Photothermal/Chemodynamic Treatment of Cervical Cancer. Int. J. Nanomed. 2024, 19, 5837–5858. [Google Scholar] [CrossRef]
- Yuan, H.; Chen, Y.; Hu, Y.; Li, Y.; Zhang, H.; Zhang, S.; Chen, Q.; Zhou, W.; Sun, J.; He, Z.; et al. Disulfide bond-driven nanoassembly of lipophilic epirubicin prodrugs for breast cancer therapy. J. Pharm. Investig. 2025, 55, 889–902. [Google Scholar] [CrossRef]
- Dadfar, S.M.; Roemhild, K.; Drude, N.I.; von Stillfried, S.; Knüchel, R.; Kiessling, F.; Lammers, T. Iron oxide nanoparticles: Diagnostic, therapeutic and theranostic applications. Adv. Drug Deliv. Rev. 2019, 138, 302–325. [Google Scholar] [CrossRef]
- Zhao, J.; Li, X.; Wang, X.; Wang, X. Fabrication of Hybrid Nanostructures Based on Fe3O4 Nanoclusters as Theranostic Agents for Magnetic Resonance Imaging and Drug Delivery. Nanoscale Res. Lett. 2019, 14, 200. [Google Scholar] [CrossRef] [PubMed]
- Mahmoudi, R.; Esmaeili, A.; Nematollahzadeh, A. Preparation of Fe3O4/Au/Ag3VO4 nanocomposite coated with Caerophyllum macropodum extract modified with oleic acid for theranostics agent in medical imaging. J. Photochem. Photobiol. A Chem. 2021, 425, 113724. [Google Scholar] [CrossRef]
- Divband, B.; Gharehaghaji, N.; Hassani, S. Fe3O4/Graphene-Based Nanotheranostics for Bimodal Magnetic Resonance/Fluorescence Imaging and Cancer Therapy. J. Inorg. Organomet. Polym. Mater. 2022, 32, 4443–4460. [Google Scholar] [CrossRef]
- Shen, Z.; Liu, T.; Li, Y.; Lau, J.; Yang, Z.; Fan, W.; Zhou, Z.; Shi, C.; Ke, C.; Bregadze, V.I.; et al. Fenton-Reaction-Acceleratable Magnetic Nanoparticles for Ferroptosis Therapy of Orthotopic Brain Tumors. ACS Nano 2018, 12, 11355–11365. [Google Scholar] [CrossRef]
- Maeda, H. Tumor-selective delivery of macromolecular drugs via the EPR effect: Background and future prospects. Bioconjugate Chem. 2010, 21, 797–802. [Google Scholar] [CrossRef]
- Akhtar, M.J.; Ahamed, M.; Alhadlaq, H.A.; Alrokayan, S.A.; Kumar, S. Targeted anticancer therapy: Overexpressed receptors and nanotechnology. Clin. Chim. Acta 2014, 436, 78–92. [Google Scholar] [CrossRef]
- Muhamad, N.; Plengsuriyakarn, T.; Na-Bangchang, K. Application of active targeting nanoparticle delivery system for chemotherapeutic drugs and traditional/herbal medicines in cancer therapy: A systematic review. Int. J. Nanomed. 2018, 13, 3921–3935. [Google Scholar] [CrossRef]
- Nosrati, H.; Salehiabar, M.; Sefidi, N.; Javani, S.; Davaran, S.; Danafar, H. Target Delivery of Iron Oxide Magnetic Nanoparticles for Imaging and Treatment. In Magnetic Nanoheterostructures: Diagnostic, Imaging and Treatment; Sharma, S.K., Javed, Y., Eds.; Springer: Cham, Switzerland, 2020; pp. 267–285. [Google Scholar]
- Yoo, J.; Park, C.; Yi, G.; Lee, D.; Koo, H. Active Targeting Strategies Using Biological Ligands for Nanoparticle Drug Delivery Systems. Cancers 2019, 11, 640. [Google Scholar] [CrossRef]
- Salehnia, Z.; Shahbazi-Gahrouei, D.; Akbarzadeh, A.; Baradaran, B.; Farajnia, S.; Naghibi, M. Synthesis and characterisation of iron oxide nanoparticles conjugated with epidermal growth factor receptor (EGFR) monoclonal antibody as MRI contrast agent for cancer detection. IET Nanobiotechnol. 2019, 13, 400–406. [Google Scholar] [CrossRef]
- Unnikrishnan, B.S.; Sen, A.; Preethi, G.U.; Joseph, M.M.; Maya, S.; Shiji, R.; Anusree, K.S.; Sreelekha, T.T. Folic acid-appended galactoxyloglucan-capped iron oxide nanoparticles as a biocompatible nanotheranostic agent for tumor-targeted delivery of doxorubicin. Int. J. Biol. Macromol. 2020, 168, 130–142. [Google Scholar] [CrossRef]
- Sanchez, C.; El Hajj Diab, D.; Connord, V.; Clerc, P.; Meunier, E.; Pipy, B.; Payre, B.; Tan, R.P.; Gougeon, M.; Carrey, J.; et al. Targeting a G-protein-coupled receptor overexpressed in endocrine tumors by magnetic nanoparticles to induce cell death. ACS Nano 2014, 8, 1350–1363. [Google Scholar] [CrossRef]
- Pineux, F.; Federico, S.; Klotz, K.-N.; Kachler, S.; Michiels, C.; Sturlese, M.; Prato, M.; Spalluto, G.; Moro, S.; Bonifazi, D. Targeting G Protein-Coupled Receptors with Magnetic Carbon Nanotubes: The Case of the A3 Adenosine Receptor. ChemMedChem 2020, 15, 1909–1920. [Google Scholar] [CrossRef]
- Lopez, S.F.; Hallali, N.; Lalatonne, Y.; Hillion, A.; Antunes, J.C.; Serhan, N.; Clerc, P.; Fourmy, D.; Motte, L.; Carrey, J.; et al. Magneto-mechanical destruction of cancer-associated fibroblasts using ultra-small iron oxide nanoparticles and low frequency rotating magnetic fields. Nanoscale Adv. 2021, 4, 421–436. [Google Scholar] [CrossRef]
- Gonzalez, T.; Muminovic, M.; Nano, O.; Vulfovich, M. Folate Receptor Alpha—A Novel Approach to Cancer Therapy. Int. J. Mol. Sci. 2024, 25, 1046. [Google Scholar] [CrossRef]
- Elwood, P.C. Molecular cloning and characterization of the human folate-binding protein cDNA from placenta and malignant tissue culture (KB) cells. J. Biol. Chem. 1989, 264, 14893–14901. [Google Scholar] [CrossRef]
- Zayed, A.L.; Bustami, R.T.; Alabsi, W.; El-Elimat, T. Development and Validation of a Rapid High-Performance Liquid Chromatography–Tandem Mass Spectrometric Method for Determination of Folic Acid in Human Plasma. Pharmaceuticals 2018, 11, 52. [Google Scholar] [CrossRef]
- Cal, P.M.S.D.; Frade, R.F.M.; Chudasama, V.; Cordeiro, C.; Caddick, S.; Gois, P.M.P. Targeting cancer cells with folic acid-iminoboronate fluorescent conjugates. Chem. Commun. 2014, 50, 5261–5263. [Google Scholar] [CrossRef]
- Slastnikova, T.A.; Rosenkranz, A.A.; Zalutsky, M.R.; Sobolev, A.S. Modular nanotransporters for targeted intracellular delivery of drugs: Folate receptors as potential targets. Curr. Pharm. Des. 2015, 21, 1227–1238. [Google Scholar] [CrossRef][Green Version]
- Shiji, R.; Joseph, M.M.; Anitha, S.D.C.; Pillai, K.R.; Unnikrishnan, B.S.; Sreelekha, T.T. Galactomannan armed superparamagnetic iron oxide nanoparticles as a folate receptor targeted multi-functional theranostic agent in the management of cancer. Int. J. Biol. Macromol. 2022, 219, 740–753. [Google Scholar] [CrossRef]
- Berg, K.C.G.; Eide, P.W.; Eilertsen, I.A.; Johannessen, B.; Bruun, J.; Danielsen, S.A.; Bjørnslett, M.; Meza-Zepeda, L.A.; Eknæs, M.; Lind, G.E.; et al. Multi-omics of 34 colorectal cancer cell lines—A resource for biomedical studies. Mol. Cancer 2017, 16, 116. [Google Scholar] [CrossRef]
- Veiseh, O.; Gunn, J.W.; Zhang, M. Design and fabrication of magnetic nanoparticles for targeted drug delivery and imaging. Adv. Drug Deliv. Rev. 2010, 62, 284–304. [Google Scholar] [CrossRef]
- Samadi, M.; Shokrollahi, H.; Zamanian, A. The magnetic-field-assisted synthesis of the Co-ferrite nanoparticles via reverse co-precipitation and their magnetic and structural properties. Mater. Chem. Phys. 2018, 215, 355–359. [Google Scholar] [CrossRef]
- Chen, Y.; Hou, S. Recent progress in the effect of magnetic iron oxide nanoparticles on cells and extracellular vesicles. Cell Death Discov. 2023, 9, 195. [Google Scholar] [CrossRef]
- Mohammed, L.; Gomaa, H.; Ragab, D.M.; Zhu, J. Magnetic nanoparticles for environmental and biomedical applications: A review. Particuology 2017, 30, 1–14. [Google Scholar] [CrossRef]
- Aram, E.; Moeni, M.; Abedizadeh, R.; Sabour, D.; Sadeghi-Abandansari, H.; Gardy, J.; Hassanpour, A. Smart and Multi-Functional Magnetic Nanoparticles for Cancer Treatment Applications: Clinical Challenges and Future Prospects. Nanomaterials 2022, 12, 3567. [Google Scholar] [CrossRef]
- Gao, M.; Li, W.; Dong, J.; Zhang, Z.R.; Yang, B. Synthesis and Characterization of Superparamagnetic Fe3O4@SiO2 Core-Shell Composite Nanoparticles. World J. Condens. Matter Phys. 2011, 1, 49–54. [Google Scholar] [CrossRef]
- Cheng, J.; Tan, G.; Li, W.; Li, J.; Wang, Z.; Jin, Y.J.R.A. Preparation, characterization and in vitro photodynamic therapy of a pyropheophorbide-a-conjugated Fe3O4 multifunctional magnetofluorescence photosensitizer. RSC Adv. 2016, 6, 37610–37620. [Google Scholar] [CrossRef]
- Cheng, J.; Tan, G.; Li, W.; Zhang, H.; Li, S.; Wang, Z.; Jin, Y. Preparation, Characterization, and Spectral Property Studies on Methotrexate Conjugated Multifunctional Fe3O4-Pyropheophorbide-a Fluorescence Nanoparticles. J. Nanosci. Nanotechnol. 2017, 17, 4461–4470. [Google Scholar]
- Sobhanardakani, S.; Jafari, A.; Zandipak, R.; Meidanchi, A. Removal of heavy metal (Hg(II) and Cr(VI)) ions from aqueous solutions using Fe2O3@SiO2 thin films as a novel adsorbent. Process Saf. Environ. Prot. 2018, 120, 348–357. [Google Scholar] [CrossRef]
- Lerma, T.A.; Garcés, V.; Palencia, M. Novel multi- and bio-functional hybrid polymer hydrogels based on bentonite-poly(acrylic acid) composites and sorbitol polyesters: Structural and functional characterization. Eur. Polym. J. 2020, 128, 109627. [Google Scholar]
- Chaurasia, A.K.; Thorat, N.D.; Tandon, A.; Kim, J.-H.; Park, S.H.; Kim, K.K. Coupling of radiofrequency with magnetic nanoparticles treatment as an alternative physical antibacterial strategy against multiple drug resistant bacteria. Sci. Rep. 2016, 6, 33662. [Google Scholar] [CrossRef]
- Chen, Y.; Li, J.; Yuan, Z.; Feng, J.; Chen, Z. Metabolic fate and subchronic biological effects of core–shell structured Fe3O4@SiO2-NH2 nanoparticles. Nanotoxicology 2018, 12, 621–636. [Google Scholar]
- Fang, Z.; Li, X.; Xu, Z.; Du, F.; Wang, W.; Shi, R.; Gao, D. Hyaluronic acid-modified mesoporous silica-coated superparamagnetic Fe3O4 nanoparticles for targeted drug delivery. Int. J. Nanomed. 2019, 14, 5785–5797. [Google Scholar] [CrossRef]
- Camacho-Fernández, J.C.; González-Quijano, G.K.; Séverac, C.; Dague, E.; Gigoux, V.; Santoyo-Salazar, J.; Martinez-Rivas, A. Nanobiomechanical behavior of Fe3O4@SiO2 and Fe3O4@SiO2–NH2 nanoparticles over HeLa cells interfaces. Nanotechnology 2021, 32, 385702. [Google Scholar] [CrossRef]
- Tabasi, H.; Mosavian, M.T.H.; Sabouri, Z.; Khazaei, M.; Darroudi, M. pH-responsive and CD44-targeting by Fe3O4/MSNs-NH2 nanocarriers for Oxaliplatin loading and colon cancer treatment. Inorg. Chem. Commun. 2021, 125, 108430. [Google Scholar] [CrossRef]
- Tabasi, H.; Mosavian, M.T.H.; Darroudi, M.; Khazaei, M.; Hashemzadeh, A.; Sabouri, Z. Synthesis and characterization of amine-functionalized Fe3O4/Mesoporous Silica Nanoparticles (MSNs) as potential nanocarriers in drug delivery systems. J. Porous Mater. 2022, 29, 1817–1828. [Google Scholar] [CrossRef]
- Hussain, A.; Hussain, M.; Alam, M.S.; Yasir, M.; Mohammad Khan, S.; Ud Din, S.; Hussain, N.; Hussain, E. Biogenic PAG-FeNPs from Prunus armeniaca (Gum) as robust colorimetric sensors for methyl orange detection. Results Chem. 2024, 11, 101789. [Google Scholar] [CrossRef]
- Ciuti, G.; Donlin, R.; Valdastri, P.; Arezzo, A.; Menciassi, A.; Morino, M.; Dario, P. Robotic versus manual control in magnetic steering of an endoscopic capsule. Endoscopy 2009, 42, 148–152. [Google Scholar] [CrossRef]
- Koleoso, M.; Feng, X.; Xue, Y.; Li, Q.; Munshi, T.; Chen, X. Micro/nanoscale magnetic robots for biomedical applications. Mater. Today Bio 2020, 8, 100085. [Google Scholar] [CrossRef]
- Dupont, P.E.; Nelson, B.J.; Goldfarb, M.; Hannaford, B.; Menciassi, A.; O’Malley, M.K.; Simaan, N.; Valdastri, P.; Yang, G.-Z. A decade retrospective of medical robotics research from 2010 to 2020. Sci. Robot. 2021, 6, eabi8017. [Google Scholar] [CrossRef]
- Yang, Z.; Zhang, L. Magnetic Actuation Systems for Miniature Robots: A Review. Adv. Intell. Syst. 2020, 2, 2000082. [Google Scholar] [CrossRef]
- Moeni, M.; Edokali, M.; Rogers, M.; Cespedes, O.; Tliba, L.; Habib, T.; Menzel, R.; Hassanpour, A. Effect of reaction and post-treatment conditions on physico-chemical properties of magnetic iron oxide nano-particles. Particuology 2024, 91, 155–167. [Google Scholar] [CrossRef]
- Moeni, M.; Edokali, M.; Rogers, M.; Cespedes, O.; Abedizadeh, R.; Khorasani, H.R.; Aram, E.; Sadeghi-Abandansari, H.; Gardy, J.; Sabour, D.; et al. Engineering Triphenyl Phosphonium Conjugated Iron Oxide Nanoparticles for Oxaliplatin loading: Application in Cancer Treatment. Chem. Eng. Res. Des. 2025, 220, 482–499. [Google Scholar] [CrossRef]
- Ngo, M.T.; Lee, U.Y.; Ha, H.; Jin, N.; Chung, G.H.; Kwak, Y.-G.; Jung, J.; Kwak, H.S. Comparison of Hemodynamic Visualization in Cerebral Arteries: Can Magnetic Resonance Imaging Replace Computational Fluid Dynamics? J. Pers. Med. 2021, 11, 253. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Guo, X.; Wei, K.; Wang, L.; Yang, D.; Lai, L.; Cheng, M.; Liu, Q. Synthesis and drug-loading properties of folic acid-modified superparamagnetic Fe3O4 hollow microsphere core/mesoporous SiO2 shell composite particles. J. Nanoparticle Res. 2013, 16, 2210. [Google Scholar] [CrossRef]
- Rana, S.; Shetake, N.G.; Barick, K.C.; Pandey, B.N.; Salunke, H.G.; Hassan, P.A. Folic acid conjugated Fe3O4 magnetic nanoparticles for targeted delivery of doxorubicin. Dalton Trans. 2016, 45, 17401–17408. [Google Scholar] [CrossRef]
- Ni, Y.; Deng, P.; Yin, R.; Zhu, Z.; Ling, C.; Ma, M.; Wang, J.; Li, S.; Liu, R. Effect and mechanism of paclitaxel loaded on magnetic Fe3O4@mSiO2-NH2-FA nanocomposites to MCF-7 cells. Drug Deliv. 2022, 30, 64–82. [Google Scholar] [CrossRef]
- Gong, Y.; Liu, G.; Wang, Q.; Zhu, A.; Liu, P.; Wu, Q. Synthesis of a novel mesoporous Fe3O4@SiO2/CTAB-SiO2 composite material and its application in the efficient removal of bisphenol A from water. Colloid Polym. Sci. 2021, 299, 807–822. [Google Scholar] [CrossRef]
- Kim, Y.S.; An, G.S. Surface Engineering of Fe3O4@SiO2 Core–Shell Nanoparticles: Role of CTAB/TEOS Ratio. Ceram. Int. 2024, 51, 379–385. [Google Scholar] [CrossRef]
- Yang, Y.; Huang, M.; Qian, J.; Gao, D.; Liang, X. Tunable Fe3O4 Nanorods for Enhanced Magnetic Hyperthermia Performance. Sci. Rep. 2020, 10, 8331. [Google Scholar] [CrossRef]
- Dawn, R.; Zzaman, M.; Faizal, F.; Kiran, C.; Kumari, A.; Shahid, R.; Panatarani, C.; Joni, I.M.; Verma, V.K.; Sahoo, S.; et al. Origin of Magnetization in Silica-coated Fe3O4 Nanoparticles Revealed by Soft X-ray Magnetic Circular Dichroism. Braz. J. Phys. 2022, 52, 99. [Google Scholar] [CrossRef]
- Ulu, A.; Ozcan, I.; Koytepe, S.; Ateş, B. Design of epoxy-functionalized Fe3O4@MCM-41 core-shell nanoparticles for enzyme immobilization. Int. J. Biol. Macromol. 2018, 115, 1122–1130. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Shu, Y.; Zhang, P. Solid-state CTAB-assisted synthesis of mesoporous Fe3O4 and Au@Fe3O4 by mechanochemistry. Chin. J. Catal. 2019, 40, 1078–1084. [Google Scholar] [CrossRef]
- Jia, Y.-p.; Shi, K.; Liao, J.; Peng, J.; Hao, Y.; Qu, Y.; Chen, L.J.; Liu, L.; Yuan, X.; Qian, Z.; et al. Effects of Cetyltrimethylammonium Bromide on the Toxicity of Gold Nanorods Both In Vitro and In Vivo: Molecular Origin of Cytotoxicity and Inflammation. Small Methods 2020, 4, 1900799. [Google Scholar] [CrossRef]
- Kim, K.-S.; Winograd, N.; Davis, R.E. Electron spectroscopy of platinum-oxygen surfaces and application to electrochemical studies. J. Am. Chem. Soc. 1971, 93, 6296–6297. [Google Scholar] [CrossRef]
- Nefedov, V.I. X-ray photoelectron study of ligands in coordination compounds. J. Mol. Struct. 1978, 46, 251–268. [Google Scholar] [CrossRef]
- Drawdy, J.E.; Hoflund, G.B.; Gardner, S.D.; Yngvadóttir, E.; Schryer, D.R. Effect of pretreatment on a platinized tin oxide catalyst used for low-temperature Co oxidation. Surf. Interface Anal. 1990, 16, 369–374. [Google Scholar] [CrossRef]
- Mishra, S.; Manna, K.; Kayal, U.; Saha, M.; Chatterjee, S.; Chandra, D.; Hara, M.; Datta, S.; Bhaumik, A.; Das Saha, K. Folic acid-conjugated magnetic mesoporous silica nanoparticles loaded with quercetin: A theranostic approach for cancer management. RSC Adv. 2020, 10, 23148–23164. [Google Scholar] [CrossRef]
- Pochapski, D.J.; Carvalho dos Santos, C.; Leite, G.W.; Pulcinelli, S.H.; Santilli, C.V. Zeta Potential and Colloidal Stability Predictions for Inorganic Nanoparticle Dispersions: Effects of Experimental Conditions and Electrokinetic Models on the Interpretation of Results. Langmuir 2021, 37, 13379–13389. [Google Scholar] [CrossRef]
- Sadat, M.E.; Bud’ko, S.L.; Ewing, R.C.; Xu, H.; Pauletti, G.M.; Mast, D.B.; Shi, D. Effect of Dipole Interactions on Blocking Temperature and Relaxation Dynamics of Superparamagnetic Iron-Oxide (Fe3O4) Nanoparticle Systems. Materials 2023, 16, 496. [Google Scholar]
- Noguchi, S.; Trisnanto, S.B.; Yamada, T.; Ota, S.; Takemura, Y. AC Magnetic Susceptibility of Magnetic Nanoparticles Measured Under DC Bias Magnetic Field. J. Magn. Soc. Jpn. 2022, 46, 42–48. [Google Scholar] [CrossRef]
- Kushwaha, P.; Chauhan, P. Influence of different surfactants on morphological, structural, optical, and magnetic properties of α-Fe2O3 nanoparticles synthesized via co-precipitation method. Appl. Phys. A 2021, 128, 18. [Google Scholar]
- Rajan, A.; Kaczmarek-Szczepańskac, B.; Sahu, N.K. Magneto-thermal response of Fe3O4@CTAB nanoparticles for cancer hyperthermia applications. Mater. Today Commun. 2021, 28, 102583. [Google Scholar] [CrossRef]
- Meng, C.; Zhikun, W.; Qiang, L.; Chunling, L.; Shuangqing, S.; Songqing, H. Preparation of amino-functionalized Fe3O4@mSiO2 core-shell magnetic nanoparticles and their application for aqueous Fe3+ removal. J. Hazard. Mater. 2018, 341, 198–206. [Google Scholar] [CrossRef]
- Nguyen, M.D.; Tran, H.-V.; Xu, S.; Lee, T.R. Fe3O4 Nanoparticles: Structures, Synthesis, Magnetic Properties, Surface Functionalization, and Emerging Applications. Appl. Sci. 2021, 11, 11301. [Google Scholar] [CrossRef]
- Goto, H.; Futagawa, M.; Takemura, Y.; Ota, S. Effects of Néel and Brownian relaxations on dynamic magnetization empirically characterized in single-core and multicore structures of magnetic nanoparticles. Nanoscale 2025, 17, 12817–12825. [Google Scholar] [CrossRef]
- Simon, G.H.; Bauer, J.S.; Saborovski, O.; Fu, Y.; Corot, C.; Wendland, M.F.; Daldrup-Link, H.E. T1 and T2 relaxivity of intracellular and extracellular USPIO at 1.5T and 3T clinical MR scanning. Eur. Radiol. 2006, 16, 738–745. [Google Scholar]
- Lindegaard, K.F.; Lundar, T.; Wiberg, J.; Sjøberg, D.I.K.; Aaslid, R.; Nornes, H. Variations in middle cerebral artery blood flow investigated with noninvasive transcranial blood velocity measurements. Stroke 1987, 18, 1025–1030. [Google Scholar] [CrossRef] [PubMed]
- Pian, Q.; Alfadhel, M.A.H.; Tang, J.; Lee, G.V.; Li, B.; Fu, B.; Ayata, Y.; Yaseen, M.A.; Boas, D.A.; Secomb, T.W.; et al. Cortical microvascular blood flow velocity mapping by combining dynamic light scattering optical coherence tomography and two-photon microscopy. J. Biomed. Opt. 2023, 28, 076003. [Google Scholar] [CrossRef] [PubMed]
- Contreras-Sánchez, H.A.; Zabel, W.J.; Flueraru, C.; Lilge, L.; Taylor, E.; Vitkin, A. Characterizing Normal and Tumour Blood Microcirculatory Systems Using Optical Coherence Tomography. Photonics 2024, 11, 891. [Google Scholar] [CrossRef]
- Trịnh, K.T. On The Critical Reynolds Number For Transition From Laminar To Turbulent Flow. arXiv 2010, arXiv:1007.0810. [Google Scholar] [CrossRef]
- Papaioannou, T.G.; Stefanadis, C. Vascular wall shear stress: Basic principles and methods. Hell. J. Cardiol. 2005, 46, 9–15. [Google Scholar]
- Uhlén, M.; Zhang, C.; Lee, S.; Sjöstedt, E.; Fagerberg, L.; Bidkhori, G.; Benfeitas, R.; Arif, M.; Liu, Z.; Edfors, F.; et al. A pathology atlas of the human cancer transcriptome. Science 2017, 357, eaan2507. [Google Scholar] [CrossRef] [PubMed]
- Luo, C.; Li, X.; Yan, H.; Guo, Q.; Liu, J.; Li, Y. Iron oxide nanoparticles induce ferroptosis under mild oxidative stress in vitro. Sci. Rep. 2024, 14, 31383. [Google Scholar] [CrossRef] [PubMed]
- Meng, Y.Q.; Shi, Y.N.; Zhu, Y.P.; Liu, Y.Q.; Gu, L.W.; Liu, D.D.; Ma, A.; Xia, F.; Guo, Q.; Xu, C.C.; et al. Recent trends in preparation and biomedical applications of iron oxide nanoparticles. J. Nanobiotechnol. 2024, 22, 24. [Google Scholar] [CrossRef] [PubMed]
- Yadav, R.K.; Verma, S.; Ramakant, R.; Singh, A.K.; Pandey, V. Iron Oxide Nanoparticle Genotoxicity, ROS Production, and Epigenetic Alterations: A Comprehensive Review. Int. J. For. Anim. Fish. Res. 2025, 9, 1–16. [Google Scholar] [CrossRef]


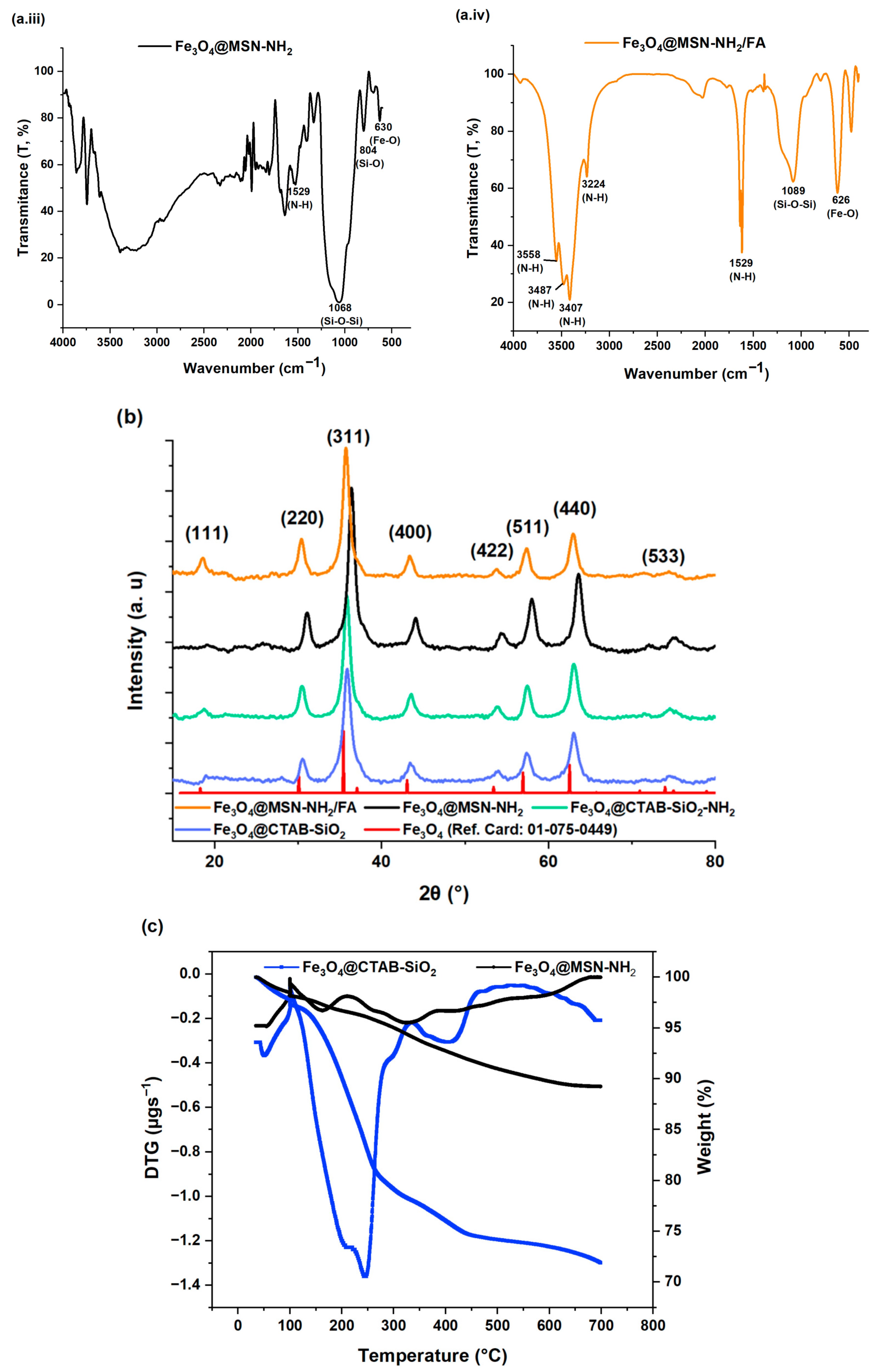





| Samples | Functional Group | Wavenumber (cm−1) | Assignment | Ref. |
|---|---|---|---|---|
| Fe3O4@MSN-NH2/FA | N-H | 3558 | Primary amine stretching of FA pteridine ring | [63,64,65] |
| N-H | 3487 | |||
| N-H | 3407 | N-H stretching of FA pteridine ring (H-bonding) | ||
| N-H | 3224 | Secondary amide N-H stretching | ||
| Fe3O4@CTAB-SiO2, Fe3O4@CTAB-SiO2-NH2 | C-H | 2925, 2854 | Asymmetric and symmetric C-H stretching of CTAB alkyl chains | [65,66] |
| Fe3O4@CTAB-SiO2 | H-O-H | 1637 | Scissoring/bending vibration of adsorbed water | [67] |
| Fe3O4@MSN-NH2/FA | C=O | 1616 | Primary amide band (C=O stretching) overlapping with FA primary amine bending | [65] |
| Fe3O4@CTAB-SiO2-NH2, Fe3O4@MSN-NH2 | N-H | 1554–1527 | N-H bending of surface primary amines | [61,63,65,67,68] |
| Fe3O4@CTAB-SiO2 | C-H | 1473 | Alkyl group bending from CTAB | [63,66] |
| All samples | Si-O-Si | 1083–1039 | Asymmetric stretching | [63,69] |
| All samples | Si-O | 800 | Symmetric stretching | [61,69] |
| All samples | Fe-O | 630 | Stretching vibration of the octahedral Fe-O in Fe3O4 | [60] |
| Sample Name | Instability Index (4 °C) | Instability Index (25 °C) | Instability Index (36 °C) | Instability Index (43 °C) |
|---|---|---|---|---|
| Fe3O4@MSN-NH2 | 0.94 ± 0.07 | 0.79 ± 0.06 | 0.88 ± 0.05 | 0.57 ± 0.05 |
| Fe3O4@MSN-NH2/OXA | 0.82 ± 0.05 | 0.90 ± 0.03 | 0.99 ± 0.06 | 0.91 ± 0.03 |
| Fe3O4@MSN-NH2/FA | 0.83 ± 0.07 | 1.03 ± 0.09 | 0.87 ± 0.06 | 0.76 ± 0.07 |
| Fe3O4@MSN-NH2/FA/OXA | 0.80 ± 0.06 | 0.81 ± 0.05 | 0.88 ± 0.07 | 0.51 ± 0.07 |
| Parameters | Fe3O4@MSN-NH2 | Fe3O4@MSN-NH2/OXA | Fe3O4@MSN-NH2/FA | Fe3O4@MSN-NH2/FA/OXA |
|---|---|---|---|---|
| Hydrodynamic size (nm) | 69 ± 7 | 60 ± 5 | 80 ± 9 | 93 ± 15 |
| Zeta potential (mV) | +17.45 ± 2 | −32.6 ± 1.3 | −18.76 ± 1.8 | −51.2 ± 1.5 |
| PDI | 0.116 | 0.101 | 0.119 | 0.113 |
| Samples | Fe3O4 | Fe3O4@CTAB-SiO2 | Fe3O4@CTAB-SiO2-NH2 | Fe3O4@MSN-NH2 | Fe3O4@MSN-NH2/OXA | Fe3O4@MSN-NH2/FA | Fe3O4@MSN-NH2/FA/OXA |
|---|---|---|---|---|---|---|---|
| Hc (T) | 0.00060 (2) | 0.00142 (2) | 0.00040 (2) | 0.00048 (2) | 0.00038 (2) | 0.00012 (2) | 0.00023 (2) |
| Mr (emu/g) | 0.664 | 0.548 | 0.385 | 0.398 | 0.316 | 0.229 | 0.1098 |
| Ms (emu/g) | 78.50 | 27.52 | 58.85 | 61.24 | 55.35 | 37.48 | 38.55 |
| Mr/Ms | 0.00846 | 0.01991 | 0.00654 | 0.00650 | 0.00571 | 0.00610 | 0.00495 |
| ROI | Samples | [µg/mL] | T1/[s] | T2/[s] |
|---|---|---|---|---|
| 1 | Fe3O4@MSN-NH2/FA | 50 | 2.254± 0.047 | 0.013107 ± 0.00049 |
| 2 | Fe3O4@MSN-NH2/OXA | 50 | 2.368 ± 0.050 | 0.03400 ± 0.00084 |
| 3 | Fe3O4@MSN-NH2 | 50 | 2.615 ± 0.064 | 0.0688 ± 0.0017 |
| 4 | Fe3O4@MSN-NH2/FA | 5 | 2.818 ± 0.048 | 0.05624 ± 0.00067 |
| 5 | Fe3O4@MSN-NH2/FA/OXA | 5 | 2.835 ± 0.055 | 0.05848 ± 0.00064 |
| 6 | Fe3O4@MSN-NH2/OXA | 5 | 2.819 ± 0.045 | 0.08001 ± 0.00079 |
| 7 | Fe3O4@MSN-NH2 | 5 | 2.852 ± 0.054 | 0.09014 ± 0.00085 |
| 8 | Fe3O4@MSN-NH2/FA/OXA | 50 | 2.382 ± 0.054 | 0.01544 ± 0.00050 |
| 9 | Agarose | - | 2.826 ± 0.051 | 0.07295 ± 0.00063 |
| Formulations | Payload | Loading Metric and (Quantification Method) | Release Profile (48 h) | Type of Release Study | Targeting Modality | Ref. |
|---|---|---|---|---|---|---|
| Fe3O4@MSN-NH2/FA/OXA | OXA | DL: 29.73% (ICP-OES) | pH 5.0: 88.0%, pH 7.4: ~0.0% (MRP) pH 5.0: ~40.0%, pH 7.4: <10.0% (no MRP) | Dynamic circulation ± MRP | FA-FOLR1 (active) + MRP | This study |
| Fe3O4@MSN-NH2/OXA | OXA | DL: 10.33% (ICP-OES) | pH 5.0: ~94.0% (MRP), pH 7.4: ~0.0% (MRP) pH 5.0: ~47.0%, pH 7.4: <10.0% (no MRP) | Dynamic circulation/flow ± MRP | NH2 surface + MRP | This study |
| Fe3O4@MSN-NH2/TPP/OXA | OXA | DL: 16.29% (ICP-OES) | pH 5.0: ~94.3% pH 7.4: 9.0% | Static-no flow | TPP mitochondrial targeting | [61] |
| Fe3O4@MSN-NH2/OXA | OXA | DL: 10.33% (ICP-OES) | pH 5.0: ~88.4% pH 7.4: <10.0% | Static-no flow | NH2 surface | [61] |
| Fe3O4@MSN/OXA | OXA | EE: 18.50% (ICP-OES) | pH 5.0: >93.0% pH 7.4: <10.0% | Static-no flow | Unmodified MSN | [53] |
| Fe3O4@MSN-NH2/OXA | OXA | EE: 55.00% (ICP-OES) | pH 5: >93.0% pH 7.4: <10.0% | Static-no flow | NH2 surface | [53] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Moeni, M.; Edokali, M.; Bacchetti, A.; Davy, J.; Sun, H.; Rogers, M.; Cespedes, O.; Aslam, Z.; Britton, A.; Khazin, L.; et al. Theranostic Iron Oxide Nanoparticles for Controlled Oxaliplatin Release Under Simulated Circulation and Cytotoxicity Evaluation in Colorectal Cancer Cell Lines. Processes 2026, 14, 597. https://doi.org/10.3390/pr14040597
Moeni M, Edokali M, Bacchetti A, Davy J, Sun H, Rogers M, Cespedes O, Aslam Z, Britton A, Khazin L, et al. Theranostic Iron Oxide Nanoparticles for Controlled Oxaliplatin Release Under Simulated Circulation and Cytotoxicity Evaluation in Colorectal Cancer Cell Lines. Processes. 2026; 14(4):597. https://doi.org/10.3390/pr14040597
Chicago/Turabian StyleMoeni, Masome, Mohamed Edokali, Alistair Bacchetti, Joshua Davy, Hanyang Sun, Matthew Rogers, Oscar Cespedes, Zabeada Aslam, Andrew Britton, Leah Khazin, and et al. 2026. "Theranostic Iron Oxide Nanoparticles for Controlled Oxaliplatin Release Under Simulated Circulation and Cytotoxicity Evaluation in Colorectal Cancer Cell Lines" Processes 14, no. 4: 597. https://doi.org/10.3390/pr14040597
APA StyleMoeni, M., Edokali, M., Bacchetti, A., Davy, J., Sun, H., Rogers, M., Cespedes, O., Aslam, Z., Britton, A., Khazin, L., Schneider, J. E., Valdastri, P., Menzel, R., Volpato, M., & Hassanpour, A. (2026). Theranostic Iron Oxide Nanoparticles for Controlled Oxaliplatin Release Under Simulated Circulation and Cytotoxicity Evaluation in Colorectal Cancer Cell Lines. Processes, 14(4), 597. https://doi.org/10.3390/pr14040597






