Clinoptilolite-Supported ZnO and TiO2 Composites for High-Efficiency Adsorption of Methylene Blue
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Instruments and Equipment Used
2.3. Preparation and Characterization of the Adsorbent
2.4. Adsorption Experiments
- qe: amount of MB adsorbed per unit mass of adsorbent (mg/g).
- C0: initial concentration of MB (mg/L).
- Ce: equilibrium concentration of MB remaining in the solution after adsorption (mg/L).
- w: amount of ZnO- and TiO2-coated zeolite used as the adsorbent (g).
- V: denotes the volume of the MB solution used in each experiment.
2.5. Desorption Studies
- Cd: The equilibrium concentration in the desorption process of MB (mg/L).
- Ca: The equilibrium concentration in the adsorption process of MB (mg/L).
2.6. Adsorption Isotherms
2.7. Characterization Studies
3. Results and Discussion
3.1. Characterization
3.2. Effects of pH on the Adsorption of MB
3.3. Adsorption Isotherms
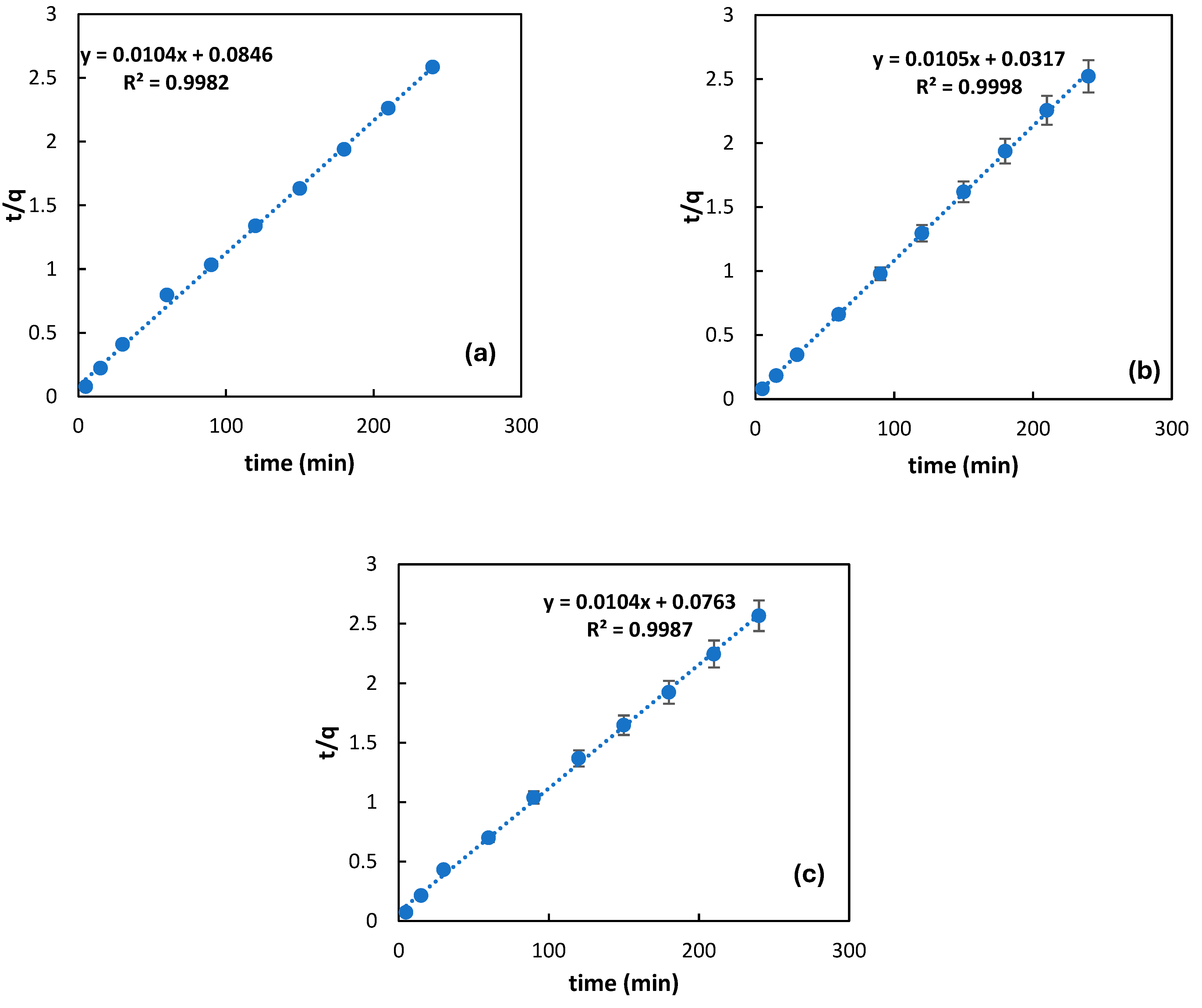
3.4. Adsorption Kinetics
3.5. Thermodynamic Investigation of Adsorption
3.6. Desorption and Reusability
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Kishor, R.; Saratale, G.D.; Saratale, R.G.; Fernando, L.; Ferreira, R.; Bilal, M.; Iqbal, H.M.N.; Bharagava, R.N. Efficient degradation and detoxification of methylene blue dye by a newly isolated ligninolytic enzyme producing bacterium Bacillus albus MW407057. Colloids Surf. B Biointerfaces 2021, 206, 111947. [Google Scholar] [CrossRef]
- Pinheiro, M.F.; Rodrigues, G.S.; Junior, J.A.; de Sousa, R.d.C.; da Costa, A.R. Analysis of the adsorptive capacity of arabic coffee straw using blue methylene dye. Braz. J. Dev. 2020, 6, 2861–2868. [Google Scholar] [CrossRef]
- Galvan, J.; Borsoi, M.X.; Julek, L.; Bordin, D.; Cabral, L.P.A.; Pomini, M.C.; Alves, F.B.T. Methylene Blue for the treatment of health conditions: A scoping review. Braz. Arch. Biol. Technol. 2021, 64, e21200266. [Google Scholar] [CrossRef]
- El-Kady, M.; El-Aassar, M.R.; El Batrawy, O.; Salem, M.; Hassan, H.S.; Fakhry, H. Equilibrium and Kinetic Behaviors of Cationic Dye Decolorization Using Poly (AN-co-Py)/ZrO2 Novel Nanopolymeric Composites. Adv. Polym. Technol. 2018, 37, 740–752. [Google Scholar] [CrossRef]
- Gureev, A.P.; Shaforostova, E.A.; Popov, V.N.; Starkov, A.A. Methylene blue does not bypass complex k in mouse brain mitochondria. FEBS Lett. 2019, 593, 499–503. [Google Scholar] [CrossRef] [PubMed]
- Altintig, E.; Ozcelik, O.T.; Aydemeir, Z.; Bozdag, D.; Kılıc, E.; Yalcıner, Y.A. Modeling of methylene blue removal on Fe3O4 modified activated carbon with artificial neural network (ANN). Int. J. Phytoremed. 2023, 25, 1714–1732. [Google Scholar] [CrossRef]
- Lou, T.; Cui, G.P.; Xun, J.J.; Wang, X.J.; Feng, N.Y.; Zhang, J. Synthesis of a terpolymer based on chitosan and lignin as an effective flocculant for dye removal. Colloids Surf. A 2018, 537, 149–154. [Google Scholar] [CrossRef]
- Zhang, J.; Jiang, F.; Lu, Y.; Wei, S.; Xu, H.; Zhang, J.; Ge, Y.; Li, Z. Lignin microparticles-reinforced cellulose filter paper for simultaneous removal of emulsified oils and dyes. Int. J. Biol. Macromol. 2023, 230, 123120. [Google Scholar] [CrossRef]
- Khan, H.; Hussain, S.; Din, M.A.U.; Arshad, M.; Wahab, F.; Hassan, U.; Khan, A. Multiple design and modelling approaches for the optimisation of carbon felt electro-Fenton treatment of dye laden wastewater. Chemosphere 2023, 338, 139510. [Google Scholar] [CrossRef]
- Busarello, P.; de Quadros, S.; Zimmermann, L.M.; Neiva, E.G.C. Graphene oxide/ZnO nanocomposites applied in photocatalysis of dyes: Tailoring aqueous stability of quantum dots. Colloids Surf. A Physicochem. Eng. Asp. 2023, 675, 132026. [Google Scholar] [CrossRef]
- Epelle, E.I.; Macfarlane, A.; Cusack, M.; Burns, A.; Okolie, J.A.; Mackay, W.; Rateb, M.; Yaseen, M. Ozone application in different industries: A review of recent developments. Chem. Eng. J. 2023, 454, 140188. [Google Scholar] [CrossRef] [PubMed]
- Aziz, G.M.; Hussein, S.I.; M-Ridha, M.J.; Mohammed, S.J.; Abed, K.M.; Muhamad, M.H.; Hasan, H.A. Activity of laccase enzyme extracted from Malva parviflora and its potential for degradation of reactive dyes in aqueous solution. Biocatal. Agric. Biotechnol. 2023, 50, 102671. [Google Scholar] [CrossRef]
- Das, S.; Cherwoo, L.; Singh, R. Decoding dye degradation: Microbial remediation of textile industry effluents. Biotechnol. Notes 2023, 4, 64–76. [Google Scholar] [CrossRef]
- Sukmana, H.; Ballai, G.; Gyulavári, T.; Illés, E.; Kozma, G.; Kónya, Z.; Hodúr, C. Hungarian and Indonesian rice husk as bioadsorbents for binary biosorption of cationic dyes from aqueous solutions: A factorial design analysis. Heliyon 2023, 9, e17154. [Google Scholar] [CrossRef] [PubMed]
- Weber, A.C.; da Silva, B.E.; Cordeiro, S.G.; Henn, G.S.; Costa, B.; dos Santos, J.S.H.; Corbellini, V.A.; Ethur, E.M.; Hoehne, L. Immobilization of commercial horseradish peroxidase in calcium alginate-starch hybrid support and its application in the biodegradation of phenol red dye. Int. J. Biol. Macromol. 2023, 15, 125723. [Google Scholar] [CrossRef]
- Li, H.; Budarin, V.L.; Clark, J.H.; North, M.; Wu, X. Rapid and efficient adsorption of methylene blue dye from aqueous solution by hierarchically porous, activated starbons®: Mechanism and porosity dependence. J. Hazard. Mater. 2022, 436, 129174. [Google Scholar] [CrossRef]
- Naseem, T.; Durrani, T. The role of some important metal oxide nanoparticles for wastewater and antibacterial applications: A review. Environ. Chem. Ecotoxicol. 2021, 3, 59–75. [Google Scholar] [CrossRef]
- Canpolat, G.; Dolak, I.; Keçili, R.; Hussain, C.G.; Amiri, A.; Hussain, C.M. Conductive polymer-based nanocomposites as powerful sorbents: Design, preparation and extraction applications. Crit. Rev. Anal. Chem. 2023, 53, 1419–1432. [Google Scholar] [CrossRef]
- Saleh, A.K.; El-Gendi, H.; Ray, J.B.; Taha, T.H. A low-cost effective media from starch kitchen waste for bacterial cellulose production and its application as simultaneous absorbance for methylene blue dye removal. Biomass Convers. Biorefinery 2023, 13, 12437–12449. [Google Scholar] [CrossRef]
- Thillainayagam, B.P.; Nagalingam, R.; Saravanan, P. Batch and column studies on removal of methylene blue dye by microalgae biochar. Biomass Convers. Biorefinery 2023, 13, 10327–10342. [Google Scholar] [CrossRef]
- Jawad, A.H.; Abdulhameed, A.S. A Mesoporous Iraqi red kaolin clay as an efficient adsorbent for methylene blue dye: Adsorption kinetics, isotherm, and mechanism study. Surf. Interfaces 2020, 18, 100422. [Google Scholar] [CrossRef]
- Jawad, A.H.; Saud, A.; Mohd, A. Acid-factionalized biomass material for methylene blue dye removal: A comprehensive adsorption and mechanism study. J. Taibah Univ. Sci. 2020, 14, 305–313. [Google Scholar] [CrossRef]
- Ahmad, R.; Ejaz, M.O. Synthesis of new alginate-silver nanoparticles/mica (Alg-AgNPs/MC) bionanocomposite for enhanced adsorption of dyes from aqueous solution. Chem. Eng. Res. Des. 2023, 197, 355–371. [Google Scholar] [CrossRef]
- Indira, V.; Abhitha, K. A review on recent developments in Zeolite A synthesis for improved carbon dioxide capture: Implications for the water-energy nexus. Energy Nexus 2022, 7, 100095. [Google Scholar] [CrossRef]
- Algieri, C.; Drioli, E. Zeolite Membranes: Synthesis and Applications. Sep. Purif. Technol. 2021, 278, 119295. [Google Scholar] [CrossRef]
- Zhang, Y.; Yan, J. Recent advances in the synthesis of defective TiO2 nanofibers and their applications in energy and catalysis. Chem. Eng. J. 2023, 472, 144831. [Google Scholar] [CrossRef]
- Agustina, T.E.; Anakotta, A.R.; Widhaningtyas, H.; Gayatri, R. The effect of weight ratio of ZnO and natural zeolite on the performance of ZnO-natural zeolite nanocomposites for synthetic dyes degradation. Mater. Today 2022, 63, 66–72. [Google Scholar] [CrossRef]
- Li, N.; Xu, Z.; Zheng, S.; Dai, H.; Wang, L.; Tian, Y.; Dong, Z.; Jiang, L. Superamphiphilic TiO2 composite surface for protein antifouling. Adv. Mater. 2021, 33, 2003559. [Google Scholar] [CrossRef]
- Rousseau, R.; Glezakou, V.A.; Selloni, A. Theoretical insights into the surface physics and chemistry of redox-active oxides. Nat. Rev. Mater. 2020, 5, 460–475. [Google Scholar] [CrossRef]
- Wang, J.; Yuan, L.; Zhang, C.; Li, S.; Wang, G.; Wan, J.; Liu, C.; Yu, C. Metal-Organic Frameworks Derived Titanium Oxides via Soft Interface Adaptive Transformation. Adv. Funct. Mater. 2021, 31, 2107260. [Google Scholar] [CrossRef]
- Wang, Y.; Wang, C.; Wang, L.; Wang, L.; Xiao, F.S. Zeolite fixed metal nanoparticles: New perspective in catalysis. Acc. Chem. Res. 2021, 54, 2579–2590. [Google Scholar] [CrossRef]
- Isa, E.D.M.; Shameli, K.; Jusoh, N.W.C.; Sukri, S.N.A.M. Photocatalytic Degradation with Green Synthesized Metal Oxide Nanoparticles—A Mini Review. J. Res. Nanosci. Nanotechnol. 2021, 2, 70–81. [Google Scholar] [CrossRef]
- Chopra, L. Photo-degradation of dyes and drugs using aloe vera synthesized zinc oxide nanoparticles—A review. Mater. Today Proc. 2023, 72, 1613–1617. [Google Scholar] [CrossRef]
- Mahamud, M.; Taddesse, A.M.; Bogale, Y.; Bezu, Z. Zeolite supported CdS/TiO2/CeO2 composite: Synthesis, characterization and photocatalytic activity for methylene blue dye degradation. Mater. Res. Bull. 2023, 161, 112176. [Google Scholar] [CrossRef]
- AlMohamadi, H.; Awad, S.A.; Sharma, A.K.; Fayzullaev, N.; Távara-Aponte, A.; Chiguala-Contreras, L.; Amari, A.; Rodriguez-Benites, C.; Tahoon, M.A.; Esmaeili, H. Photocatalytic Activity of Metal- and Non-Metal-Anchored ZnO and TiO2 Nanocatalysts for Advanced Photocatalysis: Comparative Study. Catalysts 2024, 14, 420. [Google Scholar] [CrossRef]
- Sun, Q.; Wang, N.; Yu, J. Advances in catalytic applications of zeolite-supported metal catalysts. Adv. Mater. 2021, 33, 2104442. [Google Scholar] [CrossRef]
- Lanjwani, M.F.; Tuzen, M.; Khuhawar, M.Y.; Saleh, T.A. Trends in photocatalytic degradation of organic dye pollutants using nanoparticles: A review. Inor. Chem. Commun. 2024, 159, 111613. [Google Scholar] [CrossRef]
- Hassani, A.; Khataeec, A.; Fathinia, M.; Karaca, S. Photocatalytic ozonation of ciprofloxacin from aqueous solution using TiO2/MMT nanocomposite: Nonlinear modeling and optimization of the process via artificial neural network integrated genetic algorithm. Process Saf. Environ. Prot. 2018, 116, 365–376. [Google Scholar] [CrossRef]
- Tunç, I.D.; Erol, M.; Güneş, F.; Sütçü, M. Growth of ZnO nanowires on carbon fibers for photocatalytic degradation of methylene blue aqueous solutions: An investigation on the optimization of processing parameters through response surface methodology/central composite design. Ceram. Int. 2020, 46, 7459–7474. [Google Scholar] [CrossRef]
- Parra, M.R.; Haque, F.Z. Aqueous chemical route synthesis and the effect of calcination temperature on the structural and optical properties of ZnO nanoparticles. J. Mater. Res. Technol. 2014, 3, 363–369. [Google Scholar] [CrossRef]
- Torkian, N.; Bahrami, A.; Hosseini-Abari, A.; Momeni, M.M.; Abdolkarimi-Mahabadi, M.; Bayat, A.; Hajipour, P.; Rourani, H.A.; Abbasi, M.S.; Torkian, S.; et al. Synthesis and characterization of Ag-ion-exchanged zeolite/TiO2 nanocomposites for antibacterial applications and photocatalytic degradation of antibiotics. Environ. Res. 2022, 207, 112157. [Google Scholar] [CrossRef] [PubMed]
- Huang, Z.; Li, Y.; Chen, W.; Shi, J.; Zhang, N.; Wang, X.; Li, Z.; Gao, L.; Zhang, Y. Modified bentonite adsorption of organic pollutants of dye wastewater. Mater. Chem. Phys. 2017, 202, 266–276. [Google Scholar] [CrossRef]
- Rashid, J.; Tehreem, F.; Rehman, A.; Kumar, R. Synthesis using natural functionalization of activated carbon from pumpkin peels for decolourization of aqueous methylene blue. Sci. Total Environ. 2019, 671, 369–376. [Google Scholar] [CrossRef] [PubMed]
- Unaldı, T.; Mızrak, I.; Kadir, S. Physicochemical characterisation of natural K-clinoptilolite and heavy-metal forms from Gördes (Manisa, western Turkey). J. Mol. Struct. 2023, 1054–1055, 349–358. [Google Scholar] [CrossRef]
- Wang, L.; Han, C.; Nadagouda, M.N.; Dionysiou, D.D. An innovative zinc oxide-coated zeolite adsorbent for removal of humic acid. J. Hazard. Mater. 2016, 313, 283–290. [Google Scholar] [CrossRef]
- Gayatri, R.; Agustina, T.E.; Bahrin, D.; Moeksin, R.; Gustini, G. Preparation and characterization of ZnO-zeolite nanocomposite for photocatalytic degradation by ultraviolet light. J. Ecol. Eng. 2021, 22, 178–186. [Google Scholar] [CrossRef]
- Ahmadi, Y.; Mansouri, M.; Pourafshary, P. Application of ZnO–TiO2/zeolite nanocomposites to enhance oil recovery from sandstone formations. Pet. Sci. Technol. 2024, 42, 4932–4945. [Google Scholar] [CrossRef]
- Al-Taweel, S.S.; Saud, H.R. New route for synthesis of pure anatase TiO2 nanoparticles via utrasound-assisted sol-gel method. J. Chem. Pharm. Res. 2016, 8, 620–626. [Google Scholar]
- Adam, M.R.; Othman, M.H.D.; Hubadillah, S.K.; Abd Aziz, M.H.; Jamalludin, M.R. Application of natural zeolite clinoptilolite for the removal of ammonia in wastewater. Mater. Today Proc. 2023, 1–6. [Google Scholar] [CrossRef]
- Moradi, M.; Karimzadeh, R.; Moosavi, E.S. Modified and ion exchanged clinoptilolite for the adsorptive removal of sulfur compounds in a model fuel: New adsorbents for desulfurization. Fuel 2018, 217, 467–477. [Google Scholar] [CrossRef]
- Nazarkovsky, M.; Czech, B.; Żmudka, A.; Bogatyrov, V.M.; Artiushenko, O.; Zaitsev, V.; Saint-Pierre, T.D.; Rocha, R.C.; Kai, J.; Xing, Y.; et al. Structural, optical and catalytic properties of ZnO-SiO2 colored powders with the visible light-driven activity. J. Photochem. Photobiol. A 2021, 421, 113532. [Google Scholar] [CrossRef]
- Elghniji, K.; Elaloui, E.; Moussaoui, Y. Coating of anatase titania on clinoptilolite by metal organic chemical vapor deposition method: Enhanced mesoporosity and photocatalytic activity. Chem. Pap. 2018, 72, 1159–1168. [Google Scholar] [CrossRef]
- Charoensri, K.; Rodwihok, C.; Ko, S.H.; Wongratanaphisan, D.; Park, H.J. Enhanced antimicrobial and physical properties of poly (butylene adipate-co-terephthalate)/zinc oxide/reduced graphene oxide ternary nanocomposite films. Mater. Today Commun. 2021, 28, 102586. [Google Scholar] [CrossRef]
- Cao, J.; Wang, P.; Shen, J.; Sun, Q. Core-shell Fe3O4 @zeolite NaA as an adsorbent for Cu2+. Materials 2020, 13, 5047. [Google Scholar] [CrossRef]
- Znad, H.; Abbas, K.; Hena, S.; Awual, M.R. Synthesis a novel multilamellar mesoporous TiO2/ZSM-5 for photo-catalytic degradation of methyl orange dye in aqueous media. J. Environ. Chem. Eng. 2018, 6, 218–227. [Google Scholar] [CrossRef]
- Arunkumar, M.; Samson Nesaraj, A. Photocatalytic degradation of malachite green dye using NiAl2O4 and Co doped NiAl2O4 nanophotocatalysts prepared by simple one pot wet chemical synthetic route. Iran. J. Catal. 2020, 10, 235–245. Available online: https://oiccpress.com/ijc/article/view/3566 (accessed on 31 August 2025).
- Ullah, R.; Sun, J.; Gul, A.; Munir, T.; Wu, X. Evaluations of physico-chemical properties of TiO2/clinoptilolite synthesized via three methods on photocatalytic degradation of crystal violet. Chin. J. Chem. Eng. 2021, 33, 181–189. [Google Scholar] [CrossRef]
- Javier, F.; Miriam, M.; Sanchez, R.; Navlani, G.; Angel, B.; Diego, C. Synthesis of TiO2/nanozeolite composites for highly efficient photocatalytic oxidation of propene in the gas phase. ACS Omega 2020, 5, 31323–31331. [Google Scholar] [CrossRef]
- Sonmez, B.; Baser, E.; Gel, O.Y. Photodecolourization of methylene blue by Fe- and Cd-incorporated titania-supported zeolite clinoptilolite. Microporous Mesoporous Mater. 2022, 340, 112001. [Google Scholar] [CrossRef]
- Saberian, M.; Nezamzadeh-Ejhieh, A. Synergistic photocatalytic degraded tetracycline upon supported CuO clinoptilolite nanoparticles. Solid State Sci. 2024, 147, 107381. [Google Scholar] [CrossRef]
- Akhter, S.M.H.; Siddiqui, V.U.; Ahmad, S.; Husain, D.; Naeem, S.; Alam, M.T. Sustainable synthesis of zinc oxide nanoparticles using Terminalia chebula extract: Effect of concentration and temperature on properties and antibacterial efficacy. Nano-Struct. Nano-Objects 2024, 38, 101158. [Google Scholar] [CrossRef]
- Ori, K.J.; Okocha, B.I. Biosynthesis and antimicrobial studies of zinc oxide nanoparticles of Vernonia amygdalina Leaf with varying concentration of zinc oxide. Inor. Chem. Commun. 2024, 162, 112178. [Google Scholar] [CrossRef]
- Chen, J.; Shu, C.; Wang, N.; Feng, J.; Ma, H.; Yan, W. Adsorbent synthesis of polypyrrole/TiO2 for effective fluoride removal from aqueous solution for drinking water purification: Adsorbent characterization and adsorption mechanism. J. Colloid Interface Sci. 2017, 495, 44–52. [Google Scholar] [CrossRef] [PubMed]
- Bayram, O.; Moral, E.; Göde, F. Removal of Crystal Violet Dye from Aqueous Solution Using Biochar Obtained from Oleaster Seeds. J. Inst. Sci. Technol. 2023, 13, 448–457. [Google Scholar]
- Naghipour, D.; Taghavi, K.; Hasanzadeh, D.; Ashrafi, S.D.; Moslemzadeh, M. Remove safranin dye from the aqueous solution using ZnO stabilized on zeolite in the presence of ultraviolet light. Des. Water Treat. 2023, 304, 238–246. [Google Scholar] [CrossRef]
- Mirzaei, N.; Ghaffari, H.R.; Sharafi, K.; Velayati, A.; HoseinDost, G.; Rezaei, S.; Mahvi, A.H.; Azari, A.; Dindarloo, K. Modified natural zeolite using ammonium quaternary-based material for Acid red 18 removal from aqueous solution. J. Environ. Chem. 2017, 5, 3151–3160. [Google Scholar] [CrossRef]
- Song, H.; Yuan, H.; Chen, H.; Tang, A.; Xu, G.; Liu, L.; Zhang, Z.; Kuang, Q. Synthesis of TiO2/S@PPy composite for chemisorption of polysulfides in high performance Li-S batteries. J. Solid State Electrochem. 2020, 24, 997–1006. [Google Scholar] [CrossRef]
- Setthaya, N.; Chindaprasirt, P.; Yin, S.; Pimraks, K. TiO2-zeolite photocatalysts made of metakaolin and rice husk ash for removal of methylene blue dye. Powder Technol. 2017, 313, 417–426. [Google Scholar] [CrossRef]
- Kakhki, R.M.; Karimian, A.; Hasan-nejad, H.; Ahsani, F. Zinc Oxide–Nanoclinoptilolite as a Superior Catalyst for Visible Photo-Oxidation of Dyes and Green Synthesis of Pyrazole Derivatives. J. Inorg. Organomet. Polym. Mater. 2019, 29, 1358–1367. [Google Scholar] [CrossRef]
- Gowthaman, K.; Gowthaman, P.; Venkatachalam, M.; Saroja, M.; Kutraleeswaran, M.; Dhinesh, S. Design and synthesis of TiO2/ZnO nanocomposite with enhanced oxygen vacancy: Better photocatalytic removal of MB dye under visible light-driven condition. Inorg. Chem. Commun. 2022, 146, 110197. [Google Scholar] [CrossRef]
- Boushehrian, M.M.; Esmaeili, H.; Foroutan, R. Ultrasonic assisted synthesis of Kaolin/CuFe2O4 nanocomposite for removing cationic dyes from aqueous media. J. Environ. Chem. Eng. 2020, 8, 103869. [Google Scholar] [CrossRef]
- Langmuir, I. The constitution and fundamental properties of solids and liquids. Part I. Solids. J. Am. Chem. Soc. 1916, 38, 2221–2295. [Google Scholar] [CrossRef]
- Tonk, S.; Aradi, L.E.; Kovacs, G.; Turza, A.; Rapo, E. Effectiveness and characterization of novel mineral clay in Cd2+ adsorption process: Linear and nonlinear isotherm regression analysis. Water 2020, 14, 279. [Google Scholar] [CrossRef]
- Freundlich, H.M.F. Over the adsorption in solution. J. Phys. Chem. 1906, 57, 385–471. [Google Scholar]
- Vigdorowitsch, M.; Pchelintsev, A.; Tsygankova, L.; Tanygina, E. Freundlich isotherm: An adsorption model complete framework. Appl. Sci. 2021, 11, 8078. [Google Scholar] [CrossRef]
- Kalam, S.; Abu-Khamsin, S.A.; Kamal, M.S.; Patil, S. Surfactant adsorption isotherms: A review. ACS Omega 2021, 6, 32342–32348. [Google Scholar] [CrossRef]
- Nassar, M.Y.; Abdelrahman, E.A.; Aly, A.A.; Mohamed, T.Y. A facile synthesis of mordenite zeolite nanostructures for efficient bleaching of crude soybean oil and removal of methylene blue dye from aqueous media. J. Mol. Liq. 2017, 248, 302–313. [Google Scholar] [CrossRef]
- Chaabna, I.; Delimi, R.; Testas, R. Removal of methylene blue using magnetic Na-bentonite composite in aqueous Solutions. Desalin. Water Treat. 2024, 318, 100353. [Google Scholar] [CrossRef]
- Daneshgar, H.; Sojdeh, S.; Salehi, G.; Edrisi, M.; Bagherzadeh, M.; Rabiee, N. Comparative study of synthesis methods and pH-dependent adsorption of methylene blue dye on UiO-66 and NH2-UiO-66. Chemosphere 2024, 353, 141543. [Google Scholar] [CrossRef]
- Wu, K.H.; Huang, W.C.; Hung, W.C.; Tsai, C.W. Modified expanded graphite/Fe3O4 composite as an adsorbent of methylene blue: Adsorption kinetics and isotherms. Mater. Sci. Eng. B 2021, 266, 115068. [Google Scholar] [CrossRef]
- Barakat, K. Thermodynamic study of (pantothenic acid) in different solvents and temperatures: Treatment by theoretical study. NTU J. Pure Sci. 2025, 4, 9–21. [Google Scholar] [CrossRef]
- Shi, T.; Xie, Z.; Zhu, Z.; Shi, W.; Liu, Y.; Liu, M. Highly efficient and selective adsorption of heavy metal ions by hydrazide-modified sodium alginate. Carbohydr. Polym. 2022, 276, 118797. [Google Scholar] [CrossRef]
- Yang, X.; Chen, L.; He, G.; Yang, M.; Geng, S.; Sun, Y.; Feng, X.; Ma, C.; Wei, Q.; Zhao, H.; et al. Chitosan-loaded coconut fiber for highly enhanced heavy metal adsorption from wastewater. Sep. Pur. Technol. 2025, 368, 133105. [Google Scholar] [CrossRef]
- Muedas-Taipe, G.; Maza Mejía, I.M.; Santillan, F.A.; Vel’asquez, C.J.; Asencios, Y.J.O. Removal of azo dyes in aqueous solutions using magnetized and chemically modified chitosan beads. Mater. Chem. Phys. 2020, 256, 123595. [Google Scholar] [CrossRef]
- Yahya, N.A.A.; Samir, O.M.; Al-Ariki, S.; Ahmed, A.A.M.; Swillam, M.A. Synthesis of novel antibacterial nanocomposite CuO/Ag-modified zeolite for removal of MB dye. Sci. Rep. 2023, 13, 14948. [Google Scholar] [CrossRef] [PubMed]
- Majid, Z.; AbdulRazak, A.A.; Noori, W.A.H. Modification of Zeolite by Magnetic Nanoparticles for Organic Dye Removal. Arabian J. Sci. Eng. 2019, 44, 5457–5474. [Google Scholar] [CrossRef]
- Noori, M.; Tahmasebpoor, M.; Foroutan, R. Enhanced adsorption capacity of low-cost magnetic clinoptilolite powders/beads for the effective removal of methylene blue: Adsorption and desorption studies. Mater. Chem. Phys. 2022, 278, 125655. [Google Scholar] [CrossRef]
- Jaramillo-Fierro, X.; González, S.; Montesdeoca-Mendoza, F.; Medina, F. Structuring of ZnTiO3/TiO2 Adsorbents for the Removal of Methylene Blue, Using Zeolite Precursor Clays as Natural Additives. Nanomaterials 2021, 11, 898. [Google Scholar] [CrossRef]













| Structural Parameters | C | ZnO-C | TiO2-C |
|---|---|---|---|
| BET surface area (SBET) (m2/g) | 10.21 | 11.41 | 10.80 |
| Micropore area (m2/g) | 9.08 | 10.70 | 10.28 |
| Langmuir surface area (m2/g) | 12.65 | 12.22 | 11.74 |
| BJH average pore width (4 V/A), nm | 420.65 | 864.22 | 467.54 |
| t-plot external surface area (m2/g) | 1.13 | 0.72 | 0.52 |
| 298 K | Langmuir Isotherms | Freundlich Isotherms | ||||
|---|---|---|---|---|---|---|
| qmax (mg/g) | KL × 10 (L/mg) | R2 | KF (mg/g) | n | R2 | |
| C | 38.5 ± 1.9 | 0.215 ± 0.090 | 0.951 | 9.25 ± 0.42 | 2.45 ± 0.12 | 0.952 |
| ZnO-C | 56.0 ± 2.4 | 0.130 ± 0.006 | 0.997 | 8.08 ± 0.36 | 1.66 ± 0.09 | 0.978 |
| TiO2-C | 106.0 ± 4.8 | 0.290 ± 0.014 | 0.990 | 45.6 ± 2.1 | 5.05 ± 0.27 | 0.817 |
| C0 100 (mg/L) | qe exp | Pseudo-First-Order | Pseudo-Second-Order | ||||
|---|---|---|---|---|---|---|---|
| k1 × 10 (h−1) | qe cal (mg/g) | R2 | k2 × 103 (g/mg·h) | qe cal (mg/g) | R2 | ||
| C | 92.8 ± 1.4 | 0.017 ± 0.001 | 29.5 ± 1.8 | 0.908 | 0.140 ± 0.010 | 96.2 ± 2.3 | 0.998 |
| ZnO-C | 93.08 ± 1.6 | 0.015 ± 0.001 | 9.0 ± 0.7 | 0.601 | 0.120 ± 0.010 | 96.2 ± 2.1 | 0.998 |
| TiO2-C | 93.49 ± 1.6 | 0.015 ± 0.001 | 28.2 ± 1.5 | 0.952 | 0.330 ± 0.020 | 95.2 ± 2.6 | 0.998 |
| Sample | T (K) | ∆G° (kJ/mol) | ∆S° (kJ/mol K) | ∆H° (kJ/mol) |
|---|---|---|---|---|
| 298 | −7.3 ± 0.5 | 0.193 ± 0.014 | 50.2 ± 3.2 | |
| ZnO-C | 303 | −8.36 ± 0.51 | ||
| 308 | −9.37 ± 0.84 | |||
| 313 | −10.17 ± 0.97 | |||
| 298 | −4.36 ± 0.38 | 0.095 ± 0.010 | 24.0 ± 2.1 | |
| TiO2-C | 303 | −4.81 ± 0.41 | ||
| 308 | −5.29 ± 0.48 | |||
| 313 | −5.80 ± 0.51 |
| Adsorbent | pH | Temperature (K) | Time (min) | MB Concentration mg/L | Adsorption Capacity (mg g−1) | Reference |
|---|---|---|---|---|---|---|
| Magnetic Na-bentonite | 7 | 298 | 60 | 100 | 86.2 | [78] |
| Clinoptilolite/Fe3O4 | - | - | 30 | 50 | 70.4 | [85] |
| Zeolite HY modified by magnetic nanoparticles (powder) | 9 | 326.91 | 3 | 10.23 | 28.41 | [86] |
| CuO/Ag nanocomposite/zeolite | 10 | 298 | 60 | 10 | 45.662 | [87] |
| ZnTiO3/TiO2 | 7 | 303 | 180 | 5 | 46.36 | [88] |
| C | 298 | 60 | 100 | 38.46 | This Study | |
| ZnO-C | 298 | 60 | 100 | 56 | This Study | |
| TiO2-C | 298 | 60 | 100 | 106 | This Study |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Altintig, E.; Kabadayi, O. Clinoptilolite-Supported ZnO and TiO2 Composites for High-Efficiency Adsorption of Methylene Blue. Processes 2026, 14, 575. https://doi.org/10.3390/pr14030575
Altintig E, Kabadayi O. Clinoptilolite-Supported ZnO and TiO2 Composites for High-Efficiency Adsorption of Methylene Blue. Processes. 2026; 14(3):575. https://doi.org/10.3390/pr14030575
Chicago/Turabian StyleAltintig, Esra, and Onur Kabadayi. 2026. "Clinoptilolite-Supported ZnO and TiO2 Composites for High-Efficiency Adsorption of Methylene Blue" Processes 14, no. 3: 575. https://doi.org/10.3390/pr14030575
APA StyleAltintig, E., & Kabadayi, O. (2026). Clinoptilolite-Supported ZnO and TiO2 Composites for High-Efficiency Adsorption of Methylene Blue. Processes, 14(3), 575. https://doi.org/10.3390/pr14030575






