Study on the Ignition Characteristics of Ammonia Blended with C1–C4 Small-Molecule Alkanes
Abstract
1. Introduction
2. Calculation Method
3. Results and Discussion
3.1. Study on the Ignition Characteristics of NH3/CH4 Mixtures
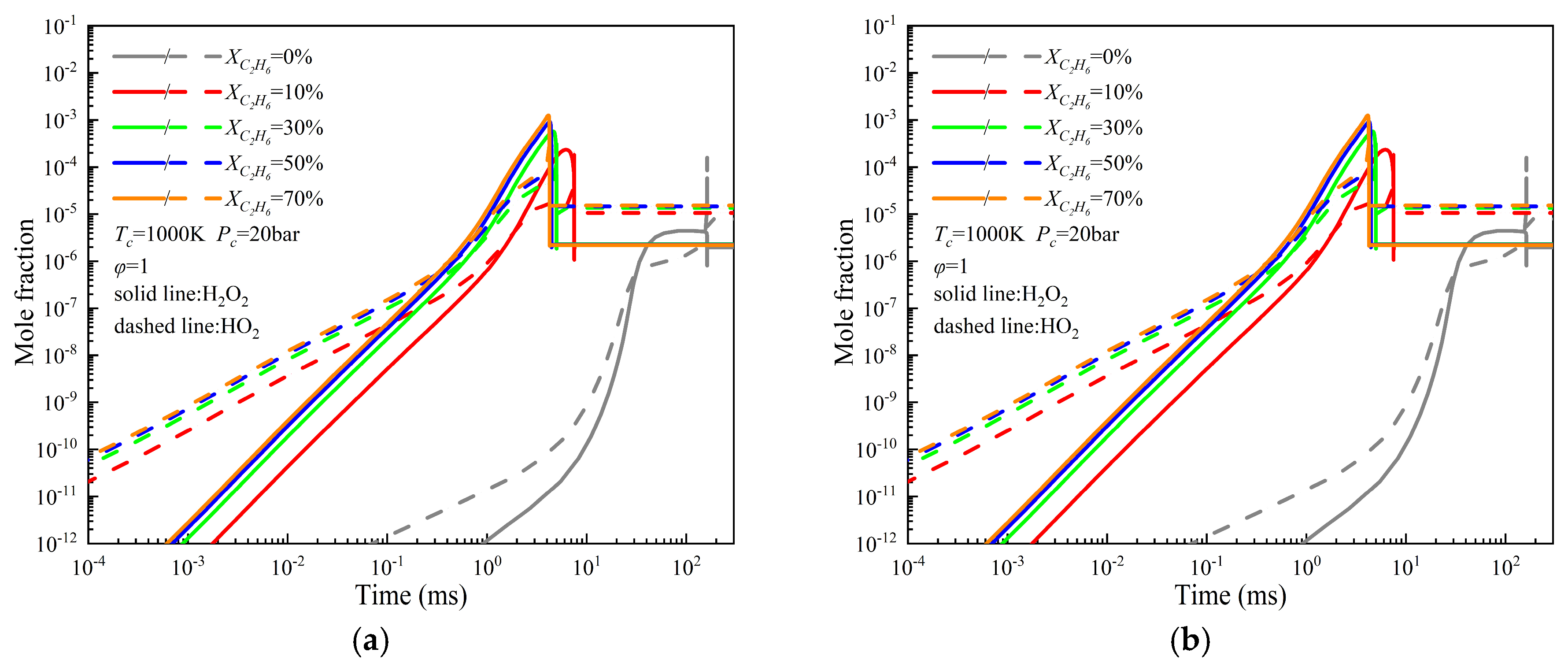
3.2. Study on the Ignition Characteristics of NH3/C2H6 Mixtures
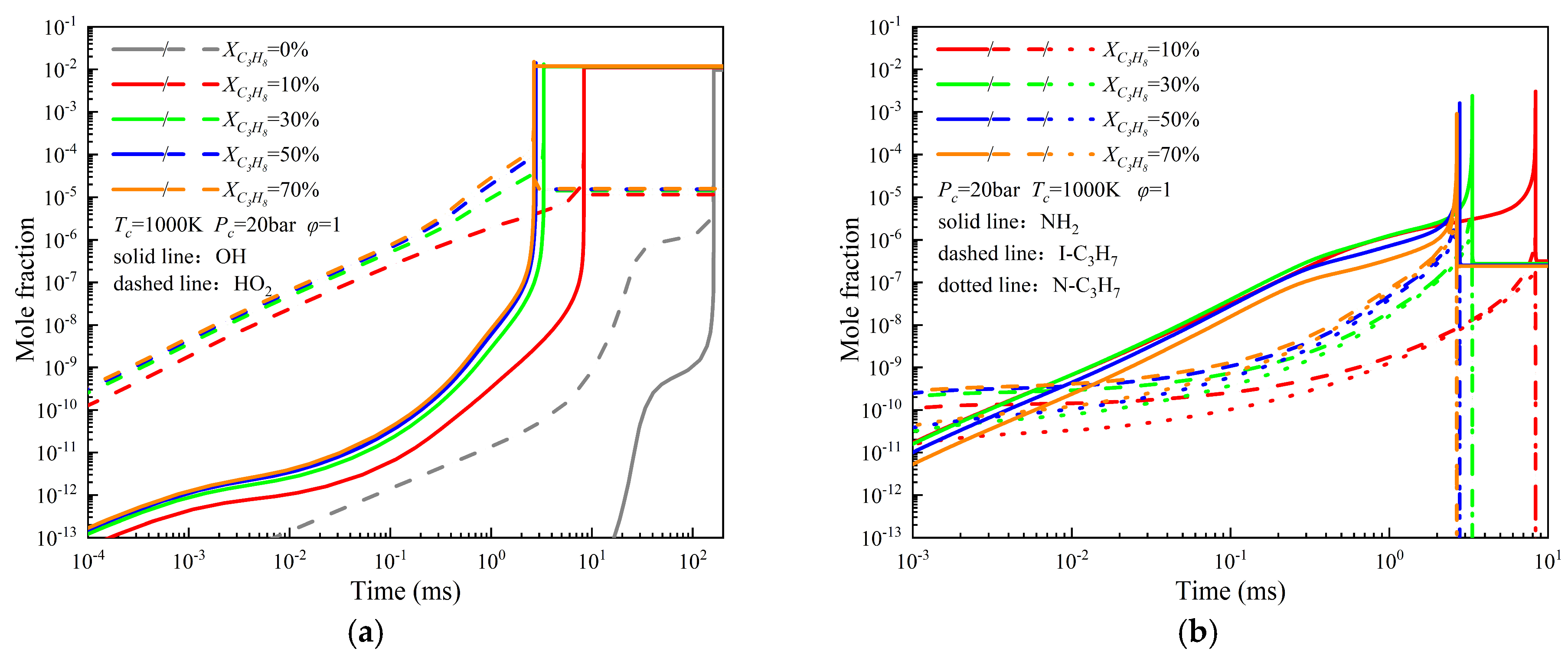
3.3. Study of the Ignition Characteristics of NH3/C3H8 Mixtures
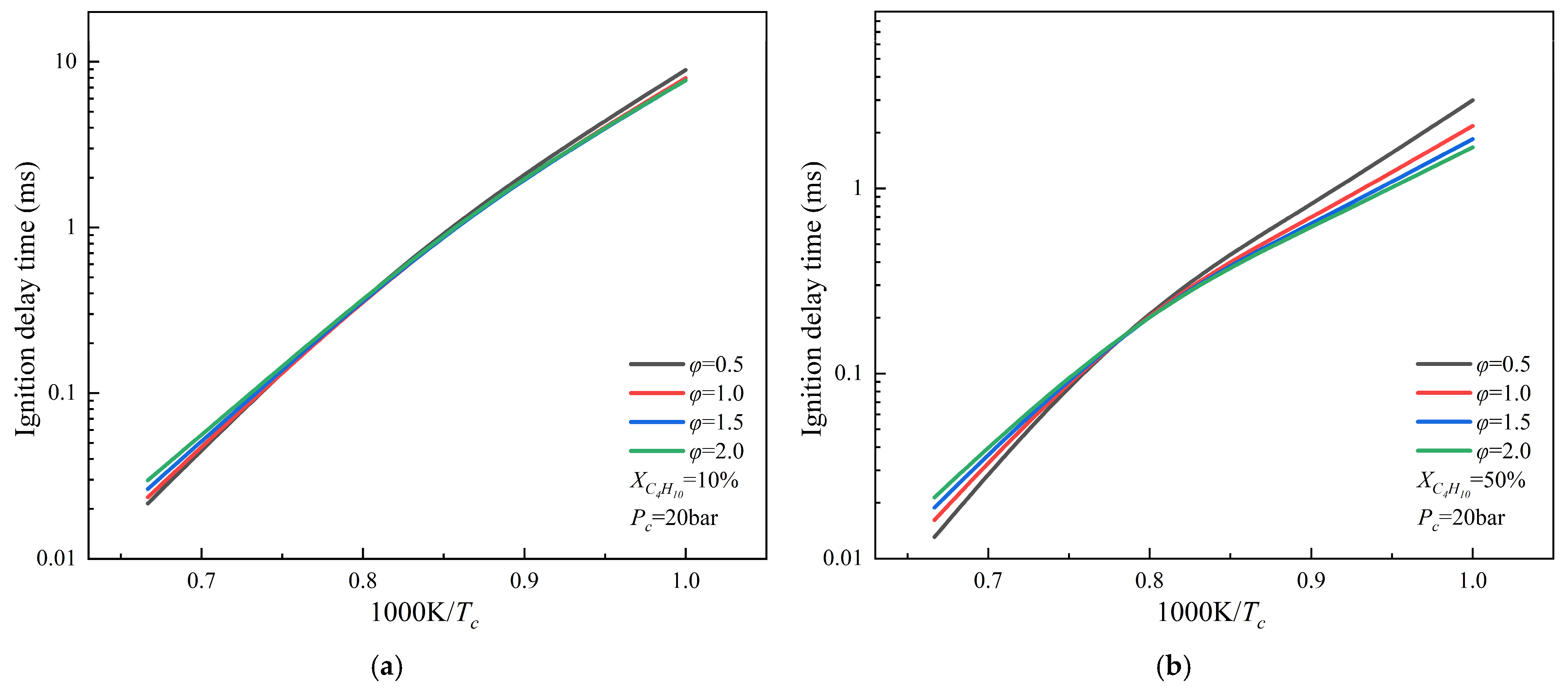
3.4. Study of the Ignition Characteristics of NH3/C4H10 Mixtures
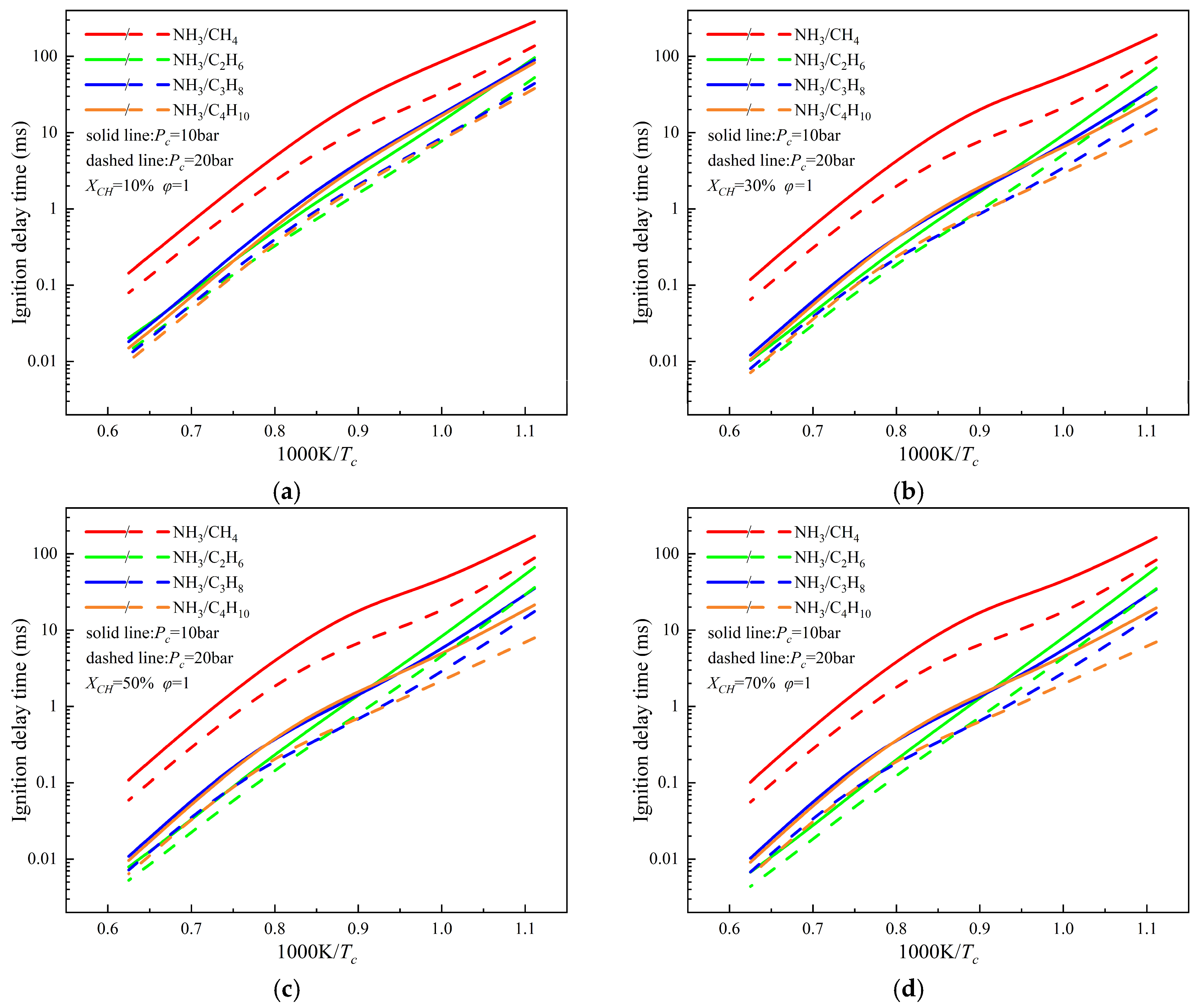
3.5. Comparison of Ignition Characteristics of NH3/C1–C4 Alkane Mixtures
4. Conclusions
- Under representative conditions (Pc = 10, 20 bar, φ = 1.0, and XCH ≥ 30%), the ignition delay times of NH3/C2–C4 alkane mixtures exhibit similar trends and are consistently shorter than those of the NH3/CH4 mixture. At low initial temperatures, the ignition delay time decreases in the following order: NH3/CH4 > NH3/C2H6 > NH3/C3H8 > NH3/C4H10. In contrast, under high temperature conditions, the ignition delay time follows the order NH3/CH4 > NH3/C3H8 > NH3/C4H10 > NH3/C2H6. These results indicate that, at low temperatures, ignition performance improves progressively with increasing carbon chain length of the blended alkane. In addition, variations in equivalence ratio exert only a minor influence on ignition delay time compared with temperature and fuel composition effects.
- The ignition delay under all investigated conditions is primarily governed by highly reactive radicals, particularly O, H, and OH. Increases in the initial temperature, initial pressure, and alkane blending ratio promote the earlier formation and higher production rates of these key radicals. At an initial temperature of 1000 K, the peak molar concentrations of radicals follow the order NH3/C4H10 > NH3/C3H8 > NH3/C2H6 > NH3/CH4. When the initial temperature is increased to 1500 K, the corresponding order becomes NH3/C2H6 > NH3/C4H10 > NH3/C3H8 > NH3/CH4. The generation rates of reactive radicals and the timing of their peak concentrations are consistent with the observed ignition delay trends under the same conditions. Among all reactions, reaction R10 (O2 + H <=> OH + O) contributes the most significantly to OH radical production.
- Temperature-dependent reaction pathway characteristics of NH3: Reaction pathway analysis reveals that at lower initial temperatures, NH3 dehydrogenation is dominated by reactions involving OH radicals. As the carbon number of the blended alkane increases, the contribution of OH to NH3 dehydrogenation increases, while the involvement of NH2 radicals in alkane dehydrogenation decreases, resulting in an enhanced forward reaction rate in the initial dehydrogenation step. At higher initial temperatures, a larger fraction of NH3 participates in NO reduction reactions, which reduces the proportion of NH3 involved in dehydrogenation pathways.
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Joung, T.; Kang, S.; Lee, J.; Ahn, J. The IMO initial strategy for reducing Greenhouse Gas (GHG) emissions, and its follow-up actions towards 2050. J. Int. Marit. Saf. Environ. Aff. Shipp. 2020, 4, 1–7. [Google Scholar] [CrossRef]
- Brainy, J.R.V.J.; Narayanamoorthy, S.; Sandra, M.; Pamucar, D.; Kang, D. An unified fuzzy decision strategy for analysing green fuel alternatives: A road to long-term development. Eng. Appl. Artif. Intell. 2024, 130, 107733. [Google Scholar] [CrossRef]
- Wouters, C.; Burkardt, P.; Steeger, F.; Fleischmann, M.; Pischinger, S. Comprehensive assessment of methanol as an alternative fuel for spark-ignition engines. Fuel 2023, 340, 127627. [Google Scholar] [CrossRef]
- Kishore, K.; Kurien, C.; Mittal, M. Experimental and numerical analysis of engine characteristics of an ammonia-substituted dual-fuel CRDI diesel engine. Fuel 2024, 366, 13154. [Google Scholar] [CrossRef]
- Aghamiri, A.R.; Lahijani, P. Catalytic conversion of biomass and plastic waste to alternative aviation fuels: A review. Biomass Bioenergy 2024, 183, 107120. [Google Scholar] [CrossRef]
- Tripathi, A.; Valera, H.; Agarwal, A.K. Dimethyl ether fuelled genset engine development and optimisation of novel fuel injection equipment for improved performance and emissions. Fuel 2024, 367, 131300. [Google Scholar] [CrossRef]
- Kalghatgi, G.; Levinsky, H.; Colket, M. Future transportation fuels. Prog. Energy Combust. Sci. 2018, 69, 103–105. [Google Scholar] [CrossRef]
- Zhou, F.; Yu, J.; Wu, C.; Fu, J.; Liu, J.; Duan, X. The application prospect and challenge of the alternative methanol fuel in the internal combustion engine. Sci. Total Environ. 2024, 913, 169708. [Google Scholar] [CrossRef]
- Stančin, H.; Mikulčić, H.; Wang, X.; Duić, N. A review on alternative fuels in future energy system. Renew. Sustain. Energy Rev. 2020, 128, 109927. [Google Scholar] [CrossRef]
- Oumer, A.N.; Hasan, M.M.; Baheta, A.T.; Mamat, R.; Abdullah, A.A. Bio-based liquid fuels as a source of renewable energy: A review. Renew. Sustain. Energy Rev. 2018, 88, 82–98. [Google Scholar] [CrossRef]
- Zamfirescu, C.; Dincer, I. Ammonia as a green fuel and hydrogen source for vehicular applications. Fuel Process. Technol. 2009, 90, 729–737. [Google Scholar] [CrossRef]
- Yousefi Rizi, H.A.; Shin, D. Green Hydrogen Production Technologies from Ammonia Cracking. Energies 2022, 15, 8246. [Google Scholar] [CrossRef]
- Cai, T.; Tang, A.; Li, C. Experimental and kinetic analyses on the flame dynamics and stabilization of ammonia/hydrogen-air mixtures in a micro-planar combustor. Chem. Eng. J. 2023, 477, 147038. [Google Scholar] [CrossRef]
- Cai, T.; Becker, S.M.; Cao, F.; Wang, B.; Tang, A.; Fu, J.; Han, L.; Sun, Y.; Zhao, D. NOx emission performance assessment on a perforated plate-implemented premixed ammonia-oxygen micro-combustion system. Chem. Eng. J. 2021, 417, 128033. [Google Scholar] [CrossRef]
- MacFarlane, D.R.; Cherepanov, P.V.; Choi, J.; Suryanto, B.H.R.; Hodgetts, R.Y.; Bakker, J.M.; Vallana, F.M.F.; Simonov, A.N. A Roadmap to the Ammonia Economy. Joule 2020, 4, 1186–1205. [Google Scholar] [CrossRef]
- Giddey, S.; Badwal, S.P.S.; Munnings, C.; Dolan, M. Ammonia as a Renewable Energy Transportation Media. ACS Sustain. Chem. Eng. 2017, 5, 10231–10239. [Google Scholar] [CrossRef]
- Lee, K.; Liu, X.; Vyawahare, P.; Sun, P.; Elgowainy, A.; Wang, M. Techno-economic performances and life cycle greenhouse gas emissions of various ammonia production pathways including conventional, carbon-capturing, nuclear-powered, and renewable production. Green Chem. 2022, 24, 4830–4844. [Google Scholar] [CrossRef]
- Boretti, A. Novel dual fuel diesel-ammonia combustion system in advanced TDI engines. Int. J. Hydrogen Energy 2017, 42, 7071–7076. [Google Scholar] [CrossRef]
- Hayakawa, A.; Arakawa, Y.; Mimoto, R.; Somarathne, K.D.K.A.; Kudo, T.; Kobayashi, H. Experimental investigation of stabilization and emission characteristics of ammonia/air premixed flames in a swirl combustor. Int. J. Hydrogen Energy 2017, 42, 14010–14018. [Google Scholar] [CrossRef]
- Hayakawa, A.; Goto, T.; Mimoto, R.; Arakawa, Y.; Kudo, T.; Kobayashi, H. Laminar burning velocity and Markstein length of ammonia/air premixed flames at various pressures. Fuel 2015, 159, 98–106. [Google Scholar] [CrossRef]
- Dimitriou, P.; Javaid, R. A review of ammonia as a compression ignition engine fuel. Int. J. Hydrogen Energy 2020, 45, 7098–7118. [Google Scholar] [CrossRef]
- Cellek, M.S. The decreasing effect of ammonia enrichment on the combustion emission of hydrogen, methane, and propane fuels. Int. J. Hydrogen Energy 2022, 47, 19916–19934. [Google Scholar] [CrossRef]
- Peng, Y.; Ranjan, D.; Sun, W. A shock tube study of fuel concentration effect on high-pressure autoignition delay of ammonia. Appl. Energy Combust. Sci. 2023, 16, 100202. [Google Scholar] [CrossRef]
- Nadiri, S.; Shu, B.; Goldsmith, C.F.; Fernandes, R. Development of comprehensive kinetic models of ammonia/methanol ignition using Reaction Mechanism Generator (RMG). Combust. Flame 2023, 251, 112710. [Google Scholar] [CrossRef]
- Xiao, H.; Lai, S.; Valera-Medina, A.; Li, J.; Liu, J.; Fu, H. Experimental and modeling study on ignition delay of ammonia/methane fuels. Int. J. Energy Res. 2020, 44, 6939–6949. [Google Scholar] [CrossRef]
- Liao, W.; Wang, Y.; Chu, Z.; Yang, B. Investigating auto-ignition characteristics and kinetic modeling of NH3/CH4 mixtures using an RCM. Combust. Flame 2024, 260, 113257. [Google Scholar] [CrossRef]
- Dai, L.; Gersen, S.; Glarborg, P.; Mokhov, A.; Levinsky, H. Autoignition studies of NH3/CH4 mixtures at high pressure. Combust. Flame 2020, 218, 19–26. [Google Scholar] [CrossRef]
- Li, M.; He, X.; Zhu, D.; Fernandes, R.; Moshammer, K. Experimental and kinetic modeling study of the autoignition and oxidation of ammonia/ethane mixtures in a rapid compression machine and a jet-stirred reactor. Combust. Flame 2023, 256, 112931. [Google Scholar] [CrossRef]
- Jiang, Z.; Dong, S.; Gao, W.; Wang, B.; Ding, H.; Xie, J.; Zou, C.; Cheng, X. Probing the effect of fuel components on the auto-ignition behavior of ammonia/natural gas blends: A case study of ethane addition. Combust. Flame 2024, 259, 113186. [Google Scholar] [CrossRef]
- Zhang, Q.; Zhang, R.; Qi, Y.; Wang, Z. Ignition characteristics of ammonia-methanol blended fuel in a rapid compression machine. Fuel 2024, 368, 131636. [Google Scholar] [CrossRef]
- Li, X.; Chu, X.; Ma, Z.; Jin, Y.; Wang, X.; Xi, Z.; Hu, S.; Chen, H. An experimental and kinetic modeling study on the ignition characteristics of ammonia/ethanol at high temperatures. Fuel 2023, 352, 129074. [Google Scholar] [CrossRef]
- Maffei, L.; Langer, R.; Murakami, Y.; Wagnon, S.; Wang, P. Modeling combustion chemistry using C3MechV4.0, An extension to mixtures of hydrogen, ammonia, alkanes, and cycloalkanes. Appl. Energy Combust. Sci. 2025, 24, 100385. [Google Scholar] [CrossRef]
- Chen, J.; Mohamed, A.; Wang, P.; Zhai, Y. A comprehensive experimental and kinetic modeling study of methyl tert-butyl ether combustion. Proc. Combust. Inst. 2024, 40, 105685. [Google Scholar] [CrossRef]
- Mohamed, A.A.E.S.; Sahu, A.B.; Panigrahy, S.; Baigmohammadi, M.; Bourque, G.; Curran, H. The effect of the addition of nitrogen oxides on the oxidation of propane: An experimental and modeling study. Combust. Flame 2022, 245, 112306. [Google Scholar] [CrossRef]
- Dong, S.; Wang, B.; Jiang, Z.; Li, Y.; Gao, W.; Wang, Z.; Cheng, X.; Curran, H. An experimental and kinetic modeling study of ammonia/n-heptane blends. Combust. Flame 2022, 246, 112428. [Google Scholar] [CrossRef]
- Fang, R.; Saggese, C.; Wagnon, S.; Sahu, A. Effect of nitric oxide and exhaust gases on gasoline surrogate autoignition: Iso-octane experiments and modeling. Combust. Flame 2021, 236, 111807. [Google Scholar] [CrossRef]
- Liu, B.; Zhou, M.; Zhang, Z.; Mi, X.; Belal, B. Ignition Delay Times and Chemical Reaction Kinetic Analysis for the Ammonia-Natural Gas Blends. Energy Fuels 2024, 38, 1373–1382. [Google Scholar] [CrossRef]
- Liu, J.; Liu, D.; Dai, L.; Zou, C.; Lin, Q. The effects of C/N cross-reactions on the NH3/C3H8 combustion: A shock-tube and modeling study. Combust. Flame 2024, 270, 113716. [Google Scholar] [CrossRef]
- Michael, P.; Justin, J.; Ramees, K.; Christopher, W. Ignition Delay Times and Chemical Kinetic Model Validation for Hydrogen and Ammonia Blending with Natural Gas at Gas Turbine Relevant Conditions. J. Eng. Gas Turbines Power 2024, 146, 061010. [Google Scholar] [CrossRef]















Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Zhao, G.; Yao, B.; Zhou, G.; Liang, J.; Li, F.; Yu, Y.; Wang, J.; Zhao, W.; Wu, J.; Zhang, H. Study on the Ignition Characteristics of Ammonia Blended with C1–C4 Small-Molecule Alkanes. Processes 2026, 14, 484. https://doi.org/10.3390/pr14030484
Zhao G, Yao B, Zhou G, Liang J, Li F, Yu Y, Wang J, Zhao W, Wu J, Zhang H. Study on the Ignition Characteristics of Ammonia Blended with C1–C4 Small-Molecule Alkanes. Processes. 2026; 14(3):484. https://doi.org/10.3390/pr14030484
Chicago/Turabian StyleZhao, Guohao, Baofeng Yao, Gongxi Zhou, Jia Liang, Fei Li, Yusong Yu, Jiawei Wang, Weijie Zhao, Jiale Wu, and Hongguang Zhang. 2026. "Study on the Ignition Characteristics of Ammonia Blended with C1–C4 Small-Molecule Alkanes" Processes 14, no. 3: 484. https://doi.org/10.3390/pr14030484
APA StyleZhao, G., Yao, B., Zhou, G., Liang, J., Li, F., Yu, Y., Wang, J., Zhao, W., Wu, J., & Zhang, H. (2026). Study on the Ignition Characteristics of Ammonia Blended with C1–C4 Small-Molecule Alkanes. Processes, 14(3), 484. https://doi.org/10.3390/pr14030484





