Life Cycle Assessment of Laboratory Analytical Workflows for Microplastics Quantification in Environmental Matrices: Sargassum and Seagrass Approach
Abstract
1. Introduction
2. Materials and Methods
2.1. Goal and Scope
2.1.1. Limitations
- The study adopts a gate-to-gate approach focused exclusively on the processes associated with quantifying microplastic retention by biofilms. As recommended by the ILCD Handbook, such a restricted boundary may omit upstream or downstream environmental burdens that could be relevant in a broader assessment [21]. Consequently, the results should not be interpreted as the total environmental impact of microplastic pollution, nor as the environmental performance of the ecosystems studied, but solely as the impact of the analytical quantification process.
- The natural process of microplastic absorption by seagrass meadows and sargassum is not modelled in terms of environmental impact, as it is an uncontrolled and non-anthropogenic phenomenon. Only the mass of microplastics retained—used as the functional output—is considered. According to ISO 14040 [22] and ISO 14044 [23], excluding biological processes beyond human control is acceptable when they are not part of the technosphere; however, this introduces uncertainty regarding comparability across different environmental conditions [22,23].
- The quantification of microplastics depends on laboratory methods that may vary in recovery efficiency, detection limits, analytical sensitivity, and accuracy. Studies have shown that FTIR and Raman spectroscopy may under-detect small particles (<20 µm), while digestion protocols can partially degrade certain polymers, leading to underestimation (e.g., [24,25]). Although standard quality-control measures (blanks, replicates, calibration) reduce these uncertainties, they cannot be eliminated.
- Data availability for some inventory flows—particularly energy consumption of analytical equipment and specific emission factors for laboratory reagents—may require the use of secondary datasets. As emphasised by the ILCD data quality guidelines, reliance on secondary data introduces uncertainty related to technological, geographical, and temporal representativeness [21]. Sensitivity analysis is recommended to evaluate the robustness of results to these uncertainties.
- The study does not include a cradle-to-grave assessment of the microplastics themselves. Their production, release into the ocean, environmental transport, fragmentation, and fate are excluded. Reviews on LCA of plastics and microplastic pollution highlight that excluding life cycle emissions of polymers prevents capturing the full environmental burden associated with the presence of microplastics in marine environments [26]. This limitation is consistent with the study’s objective but restricts interpretability.
- The spatial and temporal representativeness is constrained to the sampling locations and experimental conditions selected. Microplastic concentrations, hydrodynamics, biofilm structure, and vegetation density can vary substantially across regions and seasons [27]. Therefore, extrapolation of the results to other ecosystems should be performed with caution.
2.1.2. System Boundary
2.2. LCA Inventory
2.3. LCAs
2.4. LCA Interpretation
3. Results
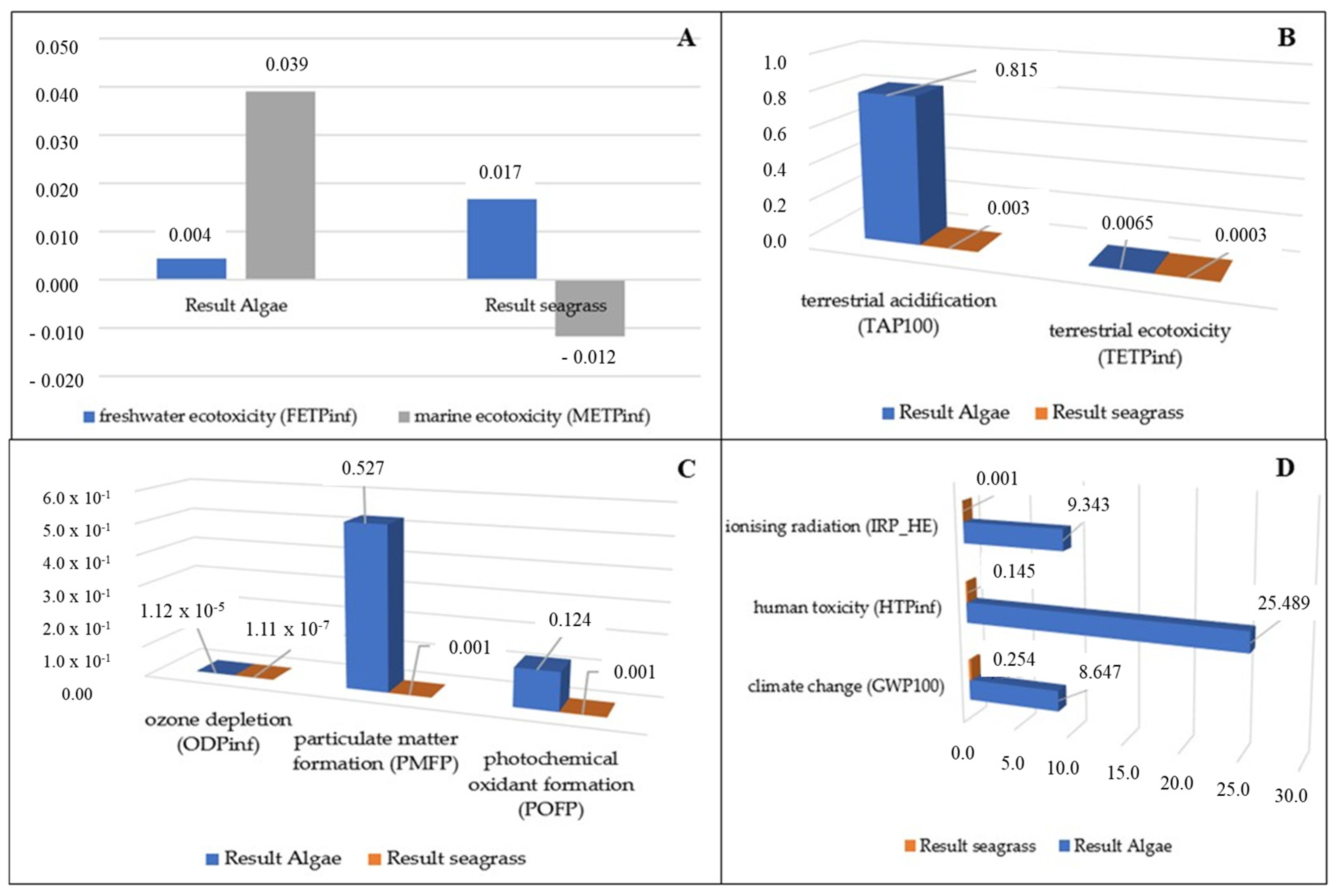
3.1. Water Impacts
3.2. Soil Impacts
3.3. Air Impacts
3.4. Other Impact Categories
4. Discussion
5. Perspectives and Future Work Regarding LCA of Microplastic Quantification
6. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| LCA | Life Cycle Assessment |
| OzLCI2019, ELCD and USDA | Free Databases for LCA and Sustainability Data |
| FTIR and µ-FTIR | Fourier Transform Infrared Spectroscopy, and Micro-FTIR |
| GWP | Climate Change |
| FETPinf | Freshwater Ecotoxicity |
| FEP | Freshwater Eutrophication |
| HTPinf | Human Toxicity |
| IRP_HE | Ionising Radiation |
| METPinf | Marine Ecotoxicity |
| MEP | Marine Eutrophication |
| ODPinf | Ozone Depletion |
| PMFP | Particulate Matter Formation |
| POFP | Photochemical Oxidant Formation |
| TAP100 | Terrestrial Acidification |
| TETPinf | Terrestrial Ecotoxicity |
| CFCs | Chlorofluorocarbons |
| UV | Ultraviolet |
| NOx | The Gases Nitric Oxide and Nitrogen Dioxide |
| VOCs | Volatile Organic Compounds |
| DNA | Deoxyribonucleic Acid |
| ILCD | International Life Cycle Data |
| EPSs | Extracellular Polymeric Substances |
References
- Greenshields, J.; Irving, A.D.; Anastasi, A.; Capper, A. Sediment composition influences microplastic trapping in seagrass meadows. Environ. Pollut. 2025, 373, 126090. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.; He, Y.; Yan, Y.; Junaid, M.; Wang, J. Characteristics, Toxic Effects, and Analytical Methods of Microplastics in the Atmosphere. Nanomaterials 2021, 11, 2747. [Google Scholar] [CrossRef] [PubMed]
- Lebreton, L.; Slat, B.; Ferrari, F.; Sainte-Rose, B.; Aitken, J.; Marthouse, R.; Hajbane, S.; Cunsolo, S.; Schwarz, A.; Levivier, A.; et al. Evidence that the Great Pacific Garbage Patch is rapidly accumulating plastic. Sci. Rep. 2018, 8, 4666. [Google Scholar] [CrossRef]
- Pal, D.; Prabhakar, R.; Barua, V.B.; Zekker, I.; Burlakovs, J.; Krauklis, A.; Hogland, W.; Vincevica-Gaile, Z. Microplastics in aquatic systems: A comprehensive review of its distribution, environmental interactions, and health risks. Environ. Sci. Pollut. Res. 2024, 32, 56–88. [Google Scholar] [CrossRef] [PubMed]
- Thin, Z.S.; Chew, J.; Ong, T.Y.Y.; Raja Ali, R.A.; Gew, L.T. Impact of microplastics on the human gut microbiome: A systematic review of microbial composition, diversity, and metabolic disruptions. BMC Gastroenterol. 2025, 25, 583. [Google Scholar] [CrossRef]
- Huang, Y.; Xiao, X.; Effiong, K.; Xu, C.; Su, Z.; Hu, J.; Jiao, S.; Holmer, M. New Insights into the Microplastic Enrichment in the Blue Carbon Ecosystem: Evidence from Seagrass Meadows and Mangrove Forests in Coastal South China Sea. Environ. Sci. Technol. 2021, 55, 4804–4812. [Google Scholar] [CrossRef]
- Seng, N.; Lai, S.; Fong, J.; Saleh, M.F.; Cheng, C.; Cheok, Z.Y.; Todd, P.A. Early evidence of microplastics on seagrass and macroalgae. Mar. Freshw. Res. 2020, 71, 922–928. [Google Scholar] [CrossRef]
- Feng, Z.; Zhang, T.; Shi, H.; Gao, K.; Huang, W.; Xu, J.; Wang, J.; Wang, R.; Li, J.; Gao, G. Microplastics in bloom-forming macroalgae: Distribution, characteristics and impacts. J. Hazard. Mater. 2020, 397, 122752. [Google Scholar] [CrossRef]
- Goss, H.; Jaskiel, J.; Rotjan, R. Thalassia testudinum as a potential vector for incorporating microplastics into benthic marine food webs. Mar. Pollut. Bull. 2018, 135, 1085–1089. [Google Scholar] [CrossRef]
- Zhou, Q.; Tu, C.; Fu, C.; Li, Y.; Zhang, H.; Xiong, K.; Zhao, X.; Li, L.; Waniek, J.J.; Luo, Y. Characteristics and distribution of microplastics in the coastal mangrove sediments of China. Sci. Total Environ. 2020, 703, 134807. [Google Scholar] [CrossRef]
- Zhao, Y.; Lv, H.; Qasim, W.; Wan, L.; Wang, Y.; Lian, X.; Liu, Y.; Hu, J.; Wang, Z.; Li, G.; et al. Drip fertigation with straw incorporation significantly reduces N2O emission and N leaching while maintaining high vegetable yields in solar greenhouse production. Environ. Pollut. 2021, 273, 116521. [Google Scholar] [CrossRef]
- da Cunha, A.C.; de Abreu, C.H.M.; Crizanto, J.L.P.; Cunha, H.F.A.; Brito, A.U.; Pereira, N.N. Modeling pollutant dispersion scenarios in high vessel-traffic areas of the Lower Amazon River. Mar. Pollut. Bull. 2021, 168, 112404. [Google Scholar] [CrossRef]
- Hu, Y.; Chen, N.; Liu, T.; Feng, C.; Ma, L.; Chen, S.; Li, M. The mechanism of nitrate-Cr(VI) reduction mediated by microbial under different initial pHs. J. Hazard. Mater. 2020, 393, 122434. [Google Scholar] [CrossRef]
- Yang, L.; Wang, N.; Liu, S.; Xiao, Q.; Geng, G.; Zhang, X.; Li, H.; Zheng, Y.; Guo, F.; Li, Q.; et al. The PM2.5 concentration reduction improves survival rate of lung cancer in Beijing. Sci. Total Environ. 2023, 858, 159857. [Google Scholar] [CrossRef]
- Ventura, E.; Marín, A.; Gámez-Pérez, J.; Cabedo, L. Recent advances in the relationships between biofilms and microplastics in natural environments. World J. Microbiol. Biotechnol. 2024, 40, 220. [Google Scholar] [CrossRef] [PubMed]
- Oberbeckmann, S.; Labrenz, M. Marine Microbial Assemblages on Microplastics: Diversity, Adaptation, and Role in Degradation. Annu. Rev. Mar. Sci. 2020, 12, 209–232. [Google Scholar] [CrossRef] [PubMed]
- Colmenares-Quintero, R.F.; Corredor-Muñoz, L.S.; Piedrahita-Rodriguez, S. Valorisation Pathways Analysis of Marine and Coastal Resources for Renewable Energy Carriers and High Value Bioproducts in La Guajira, Colombia. Energies 2025, 18, 6459. [Google Scholar] [CrossRef]
- Burgos-Arcos, C.; Caicedo-Concha, D.M.; Coz, A.; Llano, T.; Colmenares-Quintero, J.C.; Colmenares-Quintero, R.F. Assessment of the Potential for Biogas Production in Post-Conflict Rural Areas in Colombia Using Cocoa Residues. Energies 2025, 18, 3091. [Google Scholar] [CrossRef]
- Colmenares-Quintero, R.F.; Rojas, N.; Colmenares-Quintero, J.C.; Stansfield, K.E.; Villar-Villar, S.S.; Albericci-Avendaño, S.E. Design of an Integral Simulation Model for Solar-Powered Seawater Desalination in Coastal Communities: A Case Study in Manaure, La Guajira, Colombia. Sustainability 2025, 17, 1505. [Google Scholar] [CrossRef]
- Colmenares-Quintero, R.F.; Caicedo-Concha, D.M.; Corredor-Muñoz, L.S.; Piedrahita-Rodríguez, S.; Coz, A.; Colmenares-Quintero, J.C. A Critical Review of Life Cycle Assessments of Cocoa: Environmental Impacts and Methodological Challenges for Sustainable Production. AgriEngineering 2025, 7, 419. [Google Scholar] [CrossRef]
- Wolf, M.; Chomkhamsri, K.; Brandao, M.; Pant, R.; Ardente, F.; Pennington, D.; Manfredi, S.; De Camillis, C.; Goralczyk, M. International Reference Life Cycle Data System (ILCD) Handbook-General Guide for Life Cycle Assessment-Detailed Guidance; EUR 24708 EN; Publications Office of the European Union: Luxembourg, 2010. [Google Scholar]
- ISO 14040:2006; Environmental Management—Life Cycle Assessment—Principles and Framework. ISO: Geneva, Switzerland, 2006.
- ISO 14044:2006; Environmental Management—Life Cycle Assessment—Requirements and Guidelines. ISO: Geneva, Switzerland, 2006.
- Prata, J.C.; da Costa, J.P.; Duarte, A.C.; Rocha-Santos, T. Methods for sampling and detection of microplastics in water and sediment: A critical review. Trends Anal. Chem. 2019, 110, 150–159. [Google Scholar] [CrossRef]
- Qian, J.; Zhang, M.; Jing, R.; Bai, L.; Zhou, B.; Zhao, M.; Pei, X.; Wei, L.; Chen, G.H. Thiosulfate as the electron acceptor in Sulfur Bioconversion-Associated Process (SBAP) for sewage treatment. Water Res. 2019, 163, 114850. [Google Scholar] [CrossRef]
- Horton, A.A.; Walton, A.; Spurgeon, D.J.; Lahive, E.; Svendsen, C. Microplastics in freshwater and terrestrial environments: Evaluating the current understanding to identify the knowledge gaps and future research priorities. Sci. Total Environ. 2017, 586, 127–141. [Google Scholar] [CrossRef]
- Zhang, Y.; Gao, T.; Kang, S.; Allen, S.; Luo, X.; Allen, D. Microplastics in glaciers of the Tibetan Plateau: Evidence for the long-range transport of microplastics. Sci. Total Environ. 2021, 758, 143634. [Google Scholar] [CrossRef]
- Sanchez-Vidal, A.; Canals, M.; de Haan, W.P.; Romero, J.; Veny, M. Seagrasses provide a novel ecosystem service by trapping marine plastics. Sci. Rep. 2021, 11, 254. [Google Scholar] [CrossRef]
- GreenDelta. OpenLCA 2.0—The World’s Leading Open Source LCA Software; GreenDelta GmbH: Berlin, Germany, 2023; Available online: https://www.openlca.org (accessed on 8 December 2025).
- Huijbregts, M.A.J.; Steinmann, Z.J.N.; Elshout, P.M.F.; Stam, G.; Verones, F.; Vieira, M.; Zijp, M.; Hollander, A.; van Zelm, R. ReCiPe2016: A harmonised life cycle impact assessment method at midpoint and endpoint level. Int. J. Life Cycle Assess. 2017, 22, 138–147. [Google Scholar] [CrossRef]
- Raccary, B.; Loubet, P.; Peres, C.; Sonnemann, G. Evaluating the environmental impacts of analytical chemistry methods: From a critical review towards a proposal using a life cycle approach. TrAC Trends Anal. Chem. 2022, 147, 116525. [Google Scholar] [CrossRef]
- Rodríguez-Alegre, R.; Durán-Videra, S.; Carmona-Fernández, D.; Pérez Megías, L.; Andecochea Saiz, C.; You, X. Comparative Assessment of Protocols for Microplastic Quantification in Wastewater. Microplastics 2025, 4, 49. [Google Scholar] [CrossRef]
- Zea Cobos, A.G.; Amón, J.; León, E.; Caballero, P. Standardization of FTIR-Based Methodologies for Microplastics Detection in Drinking Water: A Meta-Analysis Indeed and Practical Approach. Water 2024, 16, 3170. [Google Scholar] [CrossRef]
- Cowger, W.; Gray, A.; Christiansen, S.H.; DeFrond, H.; Deshpande, A.D.; Hemabessiere, L.; Lee, E.; Mill, L.; Munno, K.; Ossmann, B.E.; et al. Critical Review of Processing and Classification Techniques for Images and Spectra in Microplastic Research. Appl. Spectrosc. 2020, 74, 989–1010. [Google Scholar] [CrossRef] [PubMed]
- Eckelman, M.J.; Sherman, J. Environmental Impacts of the U.S. Health Care System and Effects on Public Health. PLoS ONE 2016, 11, e0157014. [Google Scholar] [CrossRef] [PubMed]
- Dahl, M.; Bergman, S.; Björk, M.; Diaz-Almela, E.; Granberg, M.; Gullström, M.; Leiva-Dueñas, C.; Magnusson, K.; Marco-Méndez, C.; Piñeiro-Juncal, N.; et al. A temporal record of microplastic pollution in Mediterranean seagrass soils. Environ. Pollut. 2021, 273, 116451. [Google Scholar] [CrossRef] [PubMed]
- Horton, A.A.; Dixon, S.J. Microplastics: An introduction to environmental transport processes. WIREs Water 2018, 5, e1268. [Google Scholar] [CrossRef]
- Koelmans, A.A.; Bakir, A.; Burton, G.A.; Janssen, C.R. Microplastic as a Vector for Chemicals in the Aquatic Environment: Critical Review and Model-Supported Reinterpretation of Empirical Studies. Environ. Sci. Technol. 2016, 50, 3315–3326. [Google Scholar] [CrossRef]


| Inputs | Outputs | ||||
|---|---|---|---|---|---|
| Seagrass biomass | 1 | kg | Seagrass biomass | 1 | g |
| Microplastics in seawater | 0.016 | kg | Microplastics retained by biomass | 0.016 | kg |
| 0.01 | kg | * | 0.01 | kg | |
| 0.02 | kg | * | 0.02 | kg | |
| Ethanol | 0.005 | kg | Ethanol ** | 0.005 | kg |
| Energy derived from equipment | 0.072 | MJ | |||
| Inputs | Outputs | ||||
|---|---|---|---|---|---|
| Algae biomass | 1 | kg | Algae biomass | 1 | kg |
| Microplastics in seawater | 0.022 | kg | Microplastics retained by biomass | 0.022 | kg |
| 0.09 | kg | * | 0.09 | kg | |
| 0.02 | kg | * | 0.02 | kg | |
| 1 | kg | * | 1 | kg | |
| Energy derived from equipment | 6.84 | MJ | |||
| Impact Category | Keywords | Reference Unit |
|---|---|---|
| climate change (GWP100) | Greenhouse gases (GHG), CO2 equivalent, emissions, global warming, radiative forcing (100 years). | kg CO2-Eq |
| freshwater ecotoxicity (FETPinf) | Toxic substances, chemical spills, damage to aquatic organisms, alteration of freshwater ecosystems. | kg 1,4-DCB-Eq |
| freshwater eutrophication (FEP) | Excessive nutrients (phosphorus), water enrichment, algae growth, decomposition, oxygen depletion. | kg P-Eq |
| human toxicity (HTPinf) | Toxic substances in air, water or soil. Industrial emissions. Risk of cancer, chronic diseases, risk to human health. | kg 1,4-DCB-Eq |
| ionising radiation (IRP_HE) | Radioactive isotopes (U235), X-rays/Gamma/Alpha/Beta rays, DNA damage, cell mutations, cancer. | kg U235-Eq |
| marine ecotoxicity (METPinf) | Release of chemicals into the ocean, bioaccumulation, marine mortality, damage to ocean biodiversity. | kg 1,4-DB-Eq |
| marine eutrophication (MEP) | Excessive nutrients (nitrogen), agricultural/residential runoff, algal blooms (red tide), areas of hypoxia/anoxia. | kg N-Eq |
| ozone depletion (ODPinf) | CFCs (chlorofluorocarbons), anthropogenic chemicals, ozone depletion, harmful UV radiation. | kg CFC-11-Eq |
| particulate matter formation (PMFP) | Precursor gases (SO2, NOx, NH3), particulate matter, respiratory/cardiovascular diseases, mortality/morbidity. | kg PM10-Eq |
| photochemical oxidant formation (POFP) | NOx + VOCs + Sunlight, vehicle emissions, photochemical smog, tropospheric ozone, respiratory irritation. | kg NMVOC-Eq |
| terrestrial acidification (TAP100) | Sulphur and nitrogen oxides, acid deposition, soil pH decrease, aluminium leaching, root damage. | kg SO2-Eq |
| terrestrial ecotoxicity (TETPinf) | Soil contaminants, persistent chemicals, damage to soil organisms (microorganisms, plants, fungi), loss of biological fertility. | kg 1,4-DCB-Eq |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Colmenares-Quintero, R.F.; Corredor-Muñoz, L.S.; Colmenares-Quintero, J.C.; Piedrahita-Rodriguez, S. Life Cycle Assessment of Laboratory Analytical Workflows for Microplastics Quantification in Environmental Matrices: Sargassum and Seagrass Approach. Processes 2026, 14, 258. https://doi.org/10.3390/pr14020258
Colmenares-Quintero RF, Corredor-Muñoz LS, Colmenares-Quintero JC, Piedrahita-Rodriguez S. Life Cycle Assessment of Laboratory Analytical Workflows for Microplastics Quantification in Environmental Matrices: Sargassum and Seagrass Approach. Processes. 2026; 14(2):258. https://doi.org/10.3390/pr14020258
Chicago/Turabian StyleColmenares-Quintero, Ramón Fernando, Laura Stefania Corredor-Muñoz, Juan Carlos Colmenares-Quintero, and Sara Piedrahita-Rodriguez. 2026. "Life Cycle Assessment of Laboratory Analytical Workflows for Microplastics Quantification in Environmental Matrices: Sargassum and Seagrass Approach" Processes 14, no. 2: 258. https://doi.org/10.3390/pr14020258
APA StyleColmenares-Quintero, R. F., Corredor-Muñoz, L. S., Colmenares-Quintero, J. C., & Piedrahita-Rodriguez, S. (2026). Life Cycle Assessment of Laboratory Analytical Workflows for Microplastics Quantification in Environmental Matrices: Sargassum and Seagrass Approach. Processes, 14(2), 258. https://doi.org/10.3390/pr14020258







