Multielectrode Advanced Oxidation Treatment of Tannery Wastewater: Mass Transfer Characterization, Process Performance, Kinetic Modeling, and Energetic Analysis
Abstract
1. Introduction
2. Materials and Methods
2.1. Simulated Tannery Wastewater Preparation
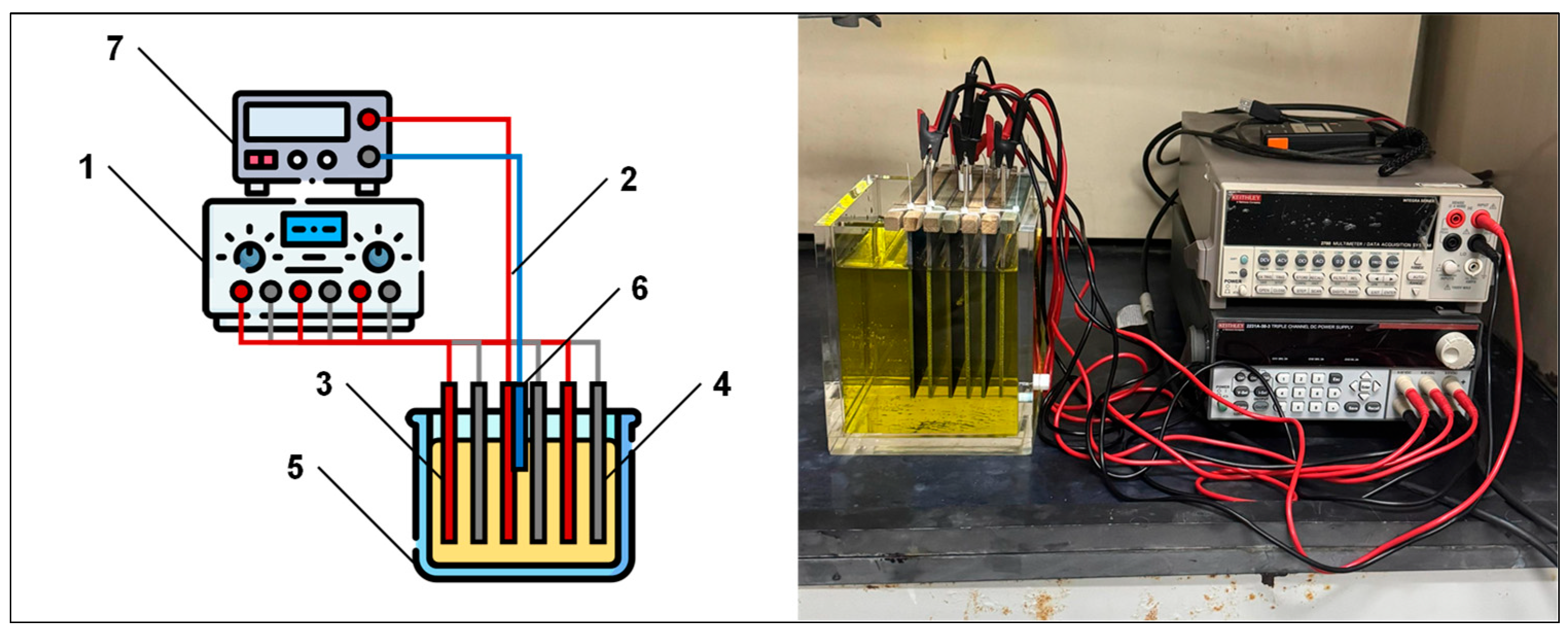
2.2. Electrochemical System
2.3. Electrochemical Characterization
2.4. Water Analyses
2.5. Kinetic Modeling
2.6. Energy and Costs Calculation
3. Results and Discussion
3.1. Electrochemical Behavior Studies
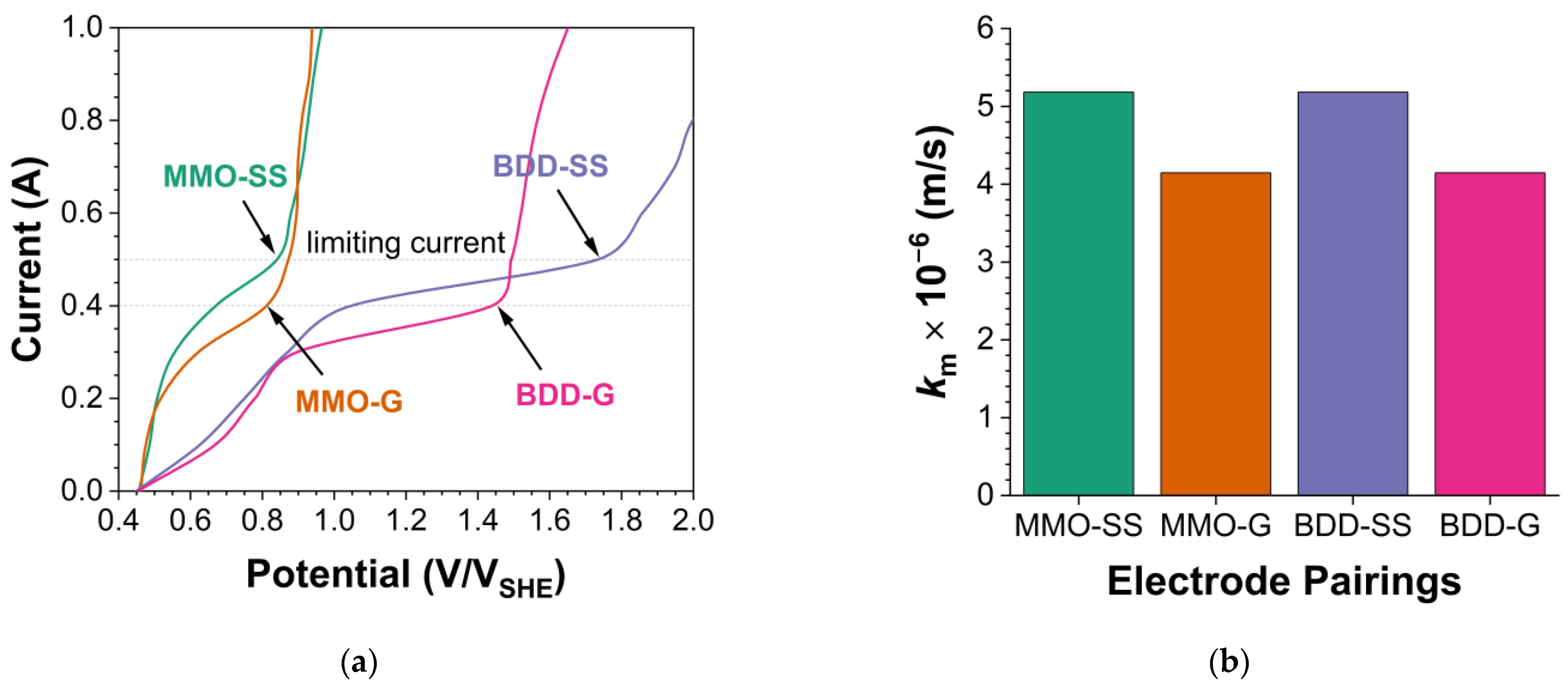
3.2. Mass Transfer Characterization
3.3. Role of Current Density
3.4. Role of Electrode Pairing
3.5. Kinetic Modeling of Pollutant Removal
3.6. Energy and Costs Consumption
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
| BDD | Boron-doped diamond |
| CER | Chlorine evolution reaction |
| CP | Chronopotentiometry |
| CV | Cyclic voltammetry |
| EC | Energy consumption |
| EO | Electro-oxidation |
| HER | Hydrogen evolution reaction |
| LSV | Linear scan voltammetry |
| MMO | Mixed-metal oxide |
| OC | Operational cost |
| OER | Oxygen evolution reaction |
| RCS | Reactive chlorine species |
| SS | Stainless steel |
| STW | Simulated tannery wastewater |
References
- Ali, H.Q.; Yasir, M.U.; Farooq, A.; Khan, M.; Salman, M.; Waqar, M. Tanneries impact on groundwater quality: A case study of Kasur city in Pakistan. Environ. Monit. Assess. 2022, 194, 823. [Google Scholar] [CrossRef]
- Rai, D.; Sinha, S. Impact of different anode materials on electro-Fenton process and tannery wastewater treatment using sequential electro-Fenton and electrocoagulation. Chemosphere 2023, 336, 139225. [Google Scholar] [CrossRef]
- Bhardwaj, A.; Kumar, S.; Singh, D. Tannery effluent treatment and its environmental impact: A review of current practices and emerging technologies. Water Qual. Res. J. 2023, 58, 128–152. [Google Scholar] [CrossRef]
- Zhao, J.; Wu, Q.; Tang, Y.; Zhou, J.; Guo, H. Tannery wastewater treatment: Conventional and promising processes, an updated 20-year review. J. Leather Sci. Eng. 2022, 4, 10. [Google Scholar] [CrossRef]
- He, Q.; Yao, K.; Sun, D.; Shi, B. Biodegradability of tannin-containing wastewater from leather industry. Biodegradation 2007, 18, 465–472. [Google Scholar] [CrossRef] [PubMed]
- Lofrano, G.; Meric, S. A review on occurrence, measurement, toxicity and tannin removal processes from wastewaters. Environ. Eng. Manag. J. 2019, 18, 109–123. [Google Scholar] [CrossRef]
- Garcia-Segura, S.; Ocon, J.D.; Chong, M.N. Electrochemical oxidation remediation of real wastewater effluents—A review. Process Saf. Environ. Prot. 2018, 113, 48–67. [Google Scholar] [CrossRef]
- Feng, Y.; Yang, L.; Liu, J.; Logan, B.E. Electrochemical technologies for wastewater treatment and resource reclamation. Environ. Sci. Water Res. Technol. 2016, 2, 800–831. [Google Scholar] [CrossRef]
- Martínez-Huitle, C.A.; Ferro, S. Electrochemical oxidation of organic pollutants for the wastewater treatment: Direct and indirect processes. Chem. Soc. Rev. 2006, 35, 1324–1340. [Google Scholar] [CrossRef]
- Adnan, F.H.; Pons, M.N.; Mousset, E. Thin film microfluidic reactors in electrochemical advanced oxidation processes for wastewater treatment: A review on influencing parameters, scaling issues, and engineering considerations. Electrochem. Sci. Adv. 2023, 3, e2100210. [Google Scholar] [CrossRef]
- Qin, T.; Yao, B.; Zhou, Y.Y.; Wu, C.C.; Li, C.W.; Ye, Z.Y.; Zhi, D.; Lam, S.S. The three-dimensional electrochemical processes for water and wastewater remediations: Mechanisms, affecting parameters, and applications. J. Clean. Prod. 2023, 408, 14. [Google Scholar] [CrossRef]
- Buso, A.; Balbo, L.; Giomo, M.; Farnia, G.; Sandonà, G. Electrochemical Removal of Tannins from Aqueous Solutions. Ind. Eng. Chem. Res. 2000, 39, 494–499. [Google Scholar] [CrossRef]
- Panizza, M.; Cerisola, G. Electrochemical oxidation as a final treatment of synthetic tannery wastewater. Environ. Sci. Technol. 2004, 38, 5470–5475. [Google Scholar] [CrossRef]
- Costa, C.R.; Montilla, F.; Morallón, E.; Olivi, P. Electrochemical oxidation of synthetic tannery wastewater in chloride-free aqueous media. J. Hazard. Mater. 2010, 180, 429–435. [Google Scholar] [CrossRef]
- Alsaffar, M.A.; Rahman, M.A.; Mageed, A.K.; Ali, S.A.K.; Lutfee, T.; Adnan, S.W.; Shakir, H.A.A. Electrochemical removal of dye from a tanning process industrial wastewater. Chem. Pap. 2023, 77, 6311–6318. [Google Scholar] [CrossRef]
- Zou, J.; Peng, X.; Li, M.; Xiong, Y.; Wang, B.; Dong, F.; Wang, B. Electrochemical oxidation of COD from real textile wastewaters: Kinetic study and energy consumption. Chemosphere 2017, 171, 332–338. [Google Scholar] [CrossRef] [PubMed]
- Doumbi, R.T.; Bertrand Noumi, G.; Ngobtchok, B.; Domga. Tannery wastewater treatment by electro-Fenton and electro-persulfate processes using graphite from used batteries as free-cost electrode materials. Case Stud. Chem. Environ. Eng. 2022, 5, 100190. [Google Scholar] [CrossRef]
- Martínez-Huitle, C.A.; dos Santos, E.V.; de Araújo, D.M.; Panizza, M. Applicability of diamond electrode/anode to the electrochemical treatment of a real textile effluent. J. Electroanal. Chem. 2012, 674, 103–107. [Google Scholar] [CrossRef]
- Du, X.; Oturan, M.A.; Zhou, M.; Belkessa, N.; Su, P.; Cai, J.; Trellu, C.; Mousset, E. Nanostructured electrodes for electrocatalytic advanced oxidation processes: From materials preparation to mechanisms understanding and wastewater treatment applications. Appl. Catal. B Environ. 2021, 296, 120332. [Google Scholar] [CrossRef]
- Kusmierek, E. Semiconductor electrode materials applied in photoelectrocatalytic wastewater treatment—An overview. Catalysts 2020, 10, 439. [Google Scholar] [CrossRef]
- Karthick, K.; Sam Sankar, S.; Kundu, S. Chapter 16—Transition metal-based nitrides for energy applications. In Nanostructured, Functional, and Flexible Materials for Energy Conversion and Storage Systems; Pandikumar, A., Rameshkumar, P., Eds.; Elsevier: Amsterdam, The Netherlands, 2020; pp. 493–515. [Google Scholar]
- Faye, G.; Sibali, L.L. A Review on Characterization, Treatment, and Impact of Tannery Wastewater in Ethiopia. Environ. Qual. Manag. 2025, 34, e70015. [Google Scholar] [CrossRef]
- Bravo-Yumi, N.; Villaseñor-Basulto, D.L.; Pérez-Segura, T.; Pacheco-Álvarez, M.A.; Picos-Benitez, A.; Rodriguez-Narvaez, O.M.; Peralta-Hernández, J.M. Comparison and statistical analysis for post-tanning synthetic wastewater degradation using different electrochemical processes. Chem. Eng. Process.—Process Intensif. 2021, 159, 108244. [Google Scholar] [CrossRef]
- Maisuria, K.J.; Shah, K.A.; Rana, J.K. Removal of tannic acid and COD from synthetic tannery wastewater. IOP Conf. Ser. Earth Environ. Sci. 2022, 1086, 012035. [Google Scholar] [CrossRef]
- Cañizares, P.; Martínez, L.; Paz, R.; Sáez, C.; Lobato, J.; Rodrigo, M.A. Treatment of Fenton-refractory olive oil mill wastes by electrochemical oxidation with boron-doped diamond anodes. J. Chem. Technol. Biotechnol. 2006, 81, 1331–1337. [Google Scholar] [CrossRef]
- Anglada, Á.; Urtiaga, A.M.; Ortiz, I. Laboratory and pilot plant scale study on the electrochemical oxidation of landfill leachate. J. Hazard. Mater. 2010, 181, 729–735. [Google Scholar] [CrossRef]
- Adnan, F.H.; Pons, M.-N.; Mousset, E. Mass transport evolution in microfluidic thin film electrochemical reactors: New correlations from millimetric to submillimetric interelectrode distances. Electrochem. Commun. 2021, 130, 107097. [Google Scholar] [CrossRef]
- LaRowe, D.E.; Van Cappellen, P. Degradation of natural organic matter: A thermodynamic analysis. Geochim. Et Cosmochim. Acta 2011, 75, 2030–2042. [Google Scholar] [CrossRef]
- Yuen, P.K.; Lau, C.M.D. Application of stoichiometric hydrogen atoms for balancing organic combustion reactions. Chem. Teach. Int. 2021, 3, 313–323. [Google Scholar] [CrossRef]
- Mousset, E.; Puce, M.; Pons, M.-N. Advanced electro-oxidation with boron-doped diamond for acetaminophen removal from real wastewater in a microfluidic reactor: Kinetics and mass-transfer studies. ChemElectroChem 2019, 6, 2908–2916. [Google Scholar] [CrossRef]
- Alagesan, J.; Jaisankar, M.; Muthuramalingam, S.; Mousset, E.; Chellam, P.V. Influence of number of azo bonds and mass transport limitations towards the elimination capacity of continuous electrochemical process for the removal of textile industrial dyes. Chemosphere 2021, 262, 128381. [Google Scholar] [CrossRef]
- Panizza, M.; Kapalka, A.; Comninellis, C. Oxidation of organic pollutants on BDD anodes using modulated current electrolysis. Electrochim. Acta 2008, 53, 2289–2295. [Google Scholar] [CrossRef]
- Panizza, M.; Michaud, P.A.; Cerisola, G.; Comninellis, C. Anodic oxidation of 2-naphthol at boron-doped diamond electrodes. J. Electroanal. Chem. 2001, 507, 206–214. [Google Scholar] [CrossRef]
- Panizza, M.; Martinez-Huitle, C.A. Role of electrode materials for the anodic oxidation of a real landfill leachate—Comparison between Ti–Ru–Sn ternary oxide, PbO2 and boron-doped diamond anode. Chemosphere 2013, 90, 1455–1460. [Google Scholar] [CrossRef]
- Adnan, F.H.; Pontvianne, S.; Pons, M.-N.; Mousset, E. Role of anodically electrogenerated hydroxyl radicals in minimizing mineral cathodic electroprecipitation in the presence of hard water. Electrochem. Commun. 2023, 150, 107493. [Google Scholar] [CrossRef]
- Abdel-Aziz, A.B.; Heakal, F.E.-T.; El Nashar, R.M.; Ghayad, I.M. Green synthesis and characterization of binary, ternary, and quaternary Ti/MMO anodes for chlorine and oxygen evolution reactions. Sci. Rep. 2024, 14, 9821. [Google Scholar] [CrossRef]
- Liu, Y.-J.; Hu, C.-Y.; Lo, S.-L. Direct and indirect electrochemical oxidation of amine-containing pharmaceuticals using graphite electrodes. J. Hazard. Mater. 2019, 366, 592–605. [Google Scholar] [CrossRef]
- Özbay, İ.; Polat, N. Electro-oxidation of woodworking wastewater by using boron-doped diamond electrode. Int. J. Environ. Anal. Chem. 2022, 102, 7659–7672. [Google Scholar] [CrossRef]
- Groenen Serrano, K. Chapter 6—Indirect electrochemical oxidation using hydroxyl radical, active chlorine, and peroxodisulfate. In Electrochemical Water and Wastewater Treatment; Martínez-Huitle, C.A., Rodrigo, M.A., Scialdone, O., Eds.; Butterworth-Heinemann: Oxford, UK, 2018; pp. 133–164. [Google Scholar]
- Yao, J.; Mei, Y.; Xia, G.; Lu, Y.; Xu, D.; Sun, N.; Wang, J.; Chen, J. Process optimization of electrochemical oxidation of ammonia to nitrogen for actual dyeing wastewater treatment. Int. J. Environ. Res. Public Health 2019, 16, 2931. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Guo, X.; Li, J.; Yang, Y.; Lei, Z.; Zhang, Z. Efficient electrochemical removal of ammonia with various cathodes and Ti/RuO2-Pt anode. Open J. Appl. Sci. 2012, 02, 241–247. [Google Scholar] [CrossRef]
- De Laat, J.; Truong Le, G.; Legube, B. A comparative study of the effects of chloride, sulfate and nitrate ions on the rates of decomposition of H2O2 and organic compounds by Fe(II)/H2O2 and Fe(III)/H2O2. Chemosphere 2004, 55, 715–723. [Google Scholar] [CrossRef]
- Huang, L.; Li, L.; Dong, W.; Liu, Y.; Hou, H. Removal of ammonia by OH radical in aqueous phase. Environ. Sci. Technol. 2008, 42, 8070–8075. [Google Scholar] [CrossRef] [PubMed]
- Adnan, F.H.; Olorounto, O.-G.; Pontvianne, S.; Pons, M.-N.; Mousset, E. Importance of organic- and inorganic-based electro-precipitation on the advanced electro-oxidation efficiency towards reclaimed wastewater treatment in thin-film reactor—Mechanistic and modeling studies. J. Environ. Chem. Eng. 2024, 12, 114908. [Google Scholar] [CrossRef]
- Bagastyo, A.Y.; Batstone, D.J.; Rabaey, K.; Radjenovic, J. Electrochemical oxidation of electrodialysed reverse osmosis concentrate on Ti/Pt–IrO2, Ti/SnO2–Sb and boron-doped diamond electrodes. Water Res. 2013, 47, 242–250. [Google Scholar] [CrossRef]
- Bergmann, M.E.H.; Koparal, A.S.; Iourtchouk, T. Electrochemical Advanced Oxidation Processes, Formation of Halogenate and Perhalogenate Species: A Critical Review. Crit. Rev. Environ. Sci. Technol. 2014, 44, 348–390. [Google Scholar] [CrossRef]
- Azizi, O.; Hubler, D.; Schrader, G.; Farrell, J.; Chaplin, B.P. Mechanism of Perchlorate Formation on Boron-Doped Diamond Film Anodes. Environ. Sci. Technol. 2011, 45, 10582–10590. [Google Scholar] [CrossRef]
- Bergmann, M.E.H.; Rollin, J. Product and by-product formation in laboratory studies on disinfection electrolysis of water using boron-doped diamond anodes. Catal. Today 2007, 124, 198–203. [Google Scholar] [CrossRef]
- Bergmann, M.E.H.; Rollin, J.; Iourtchouk, T. The occurrence of perchlorate during drinking water electrolysis using BDD anodes. Electrochim. Acta 2009, 54, 2102–2107. [Google Scholar] [CrossRef]
- Lin, Z.; Yao, W.; Wang, Y.; Yu, G.; Deng, S.; Huang, J.; Wang, B. Perchlorate formation during the electro-peroxone treatment of chloride-containing water: Effects of operational parameters and control strategies. Water Res. 2016, 88, 691–702. [Google Scholar] [CrossRef]
- DOE. Environmental Quality (Industrial Effluent) Regulations 2009; Ministry of Natural Resources And Environmental Sustainability: Putrajaya, Malaysia, 2025. [Google Scholar]
- Santacruz, W.; Faria, J.; De Mello, R.; Boldrin, M.V.; Motheo, A.d.J. Comparative study of MMO and BDD anodes for electrochemical degradation of diuron in methanol medium. Chemosphere 2024, 366, 143517. [Google Scholar] [CrossRef] [PubMed]
- Muddemann, T.; Neuber, R.; Haupt, D.; Graßl, T.; Issa, M.; Bienen, F.; Enstrup, M.; Möller, J.; Matthée, T.; Sievers, M.; et al. Improving the Treatment Efficiency and Lowering the Operating Costs of Electrochemical Advanced Oxidation Processes. Processes 2021, 9, 1482. [Google Scholar] [CrossRef]
- Sopaj, F.; Oturan, N.; Pinson, J.; Podvorica, F.I.; Oturan, M.A. Effect of cathode material on electro-Fenton process efficiency for electrocatalytic mineralization of the antibiotic sulfamethazine. Chem. Eng. J. 2020, 384, 123249. [Google Scholar] [CrossRef]
- Horváth-Gönczi, N.; Bagi, Z.; Szuhaj, M.; Rákhely, G.; Kovacs, K. Bioelectrochemical systems (BES) for biomethane production—Review. Fermentation 2023, 9, 610. [Google Scholar] [CrossRef]
- Dihrab, S.; Sopian, K.; Zaharim, A. Membrane and bipolar plates materials for regenerative fuel cells. In Proceedings of the 8th WSEAS International Conference on Simulation, Modelling and Optimization (SMO ’08), Santander, Spain, 23–25 September 2008; pp. 183–188. [Google Scholar]
- Wei, R.; Pei, S.; Yu, Y.; Zhang, J.; Liu, Y.; You, S. Water Flow-Driven Coupling Process of Anodic Oxygen Evolution and Cathodic Oxygen Activation for Water Decontamination and Prevention of Chlorinated Byproducts. Environ. Sci. Technol. 2023, 57, 17404–17414. [Google Scholar] [CrossRef]
- Shao, D.; Zhang, Y.; Lyu, W.; Zhang, X.; Tan, G.; Xu, H.; Yan, W. A modular functionalized anode for efficient electrochemical oxidation of wastewater: Inseparable synergy between OER anode and its magnetic auxiliary electrodes. J. Hazard. Mater. 2020, 390, 122174. [Google Scholar] [CrossRef]
- Snowdon, M.R.; Rathod, S.; Fattahi, A.; Khan, A.; Bragg, L.M.; Liang, R.; Zhou, N.; Servos, M.R. Water purification and electrochemical oxidation: Meeting different targets with BDD and MMO anodes. Environments 2022, 9, 135. [Google Scholar] [CrossRef]


 : 2 mA/cm2,
: 2 mA/cm2,  : 4 mA/cm2,
: 4 mA/cm2,  : 6 mA/cm2).
: 6 mA/cm2).
 : 2 mA/cm2,
: 2 mA/cm2,  : 4 mA/cm2,
: 4 mA/cm2,  : 6 mA/cm2).
: 6 mA/cm2).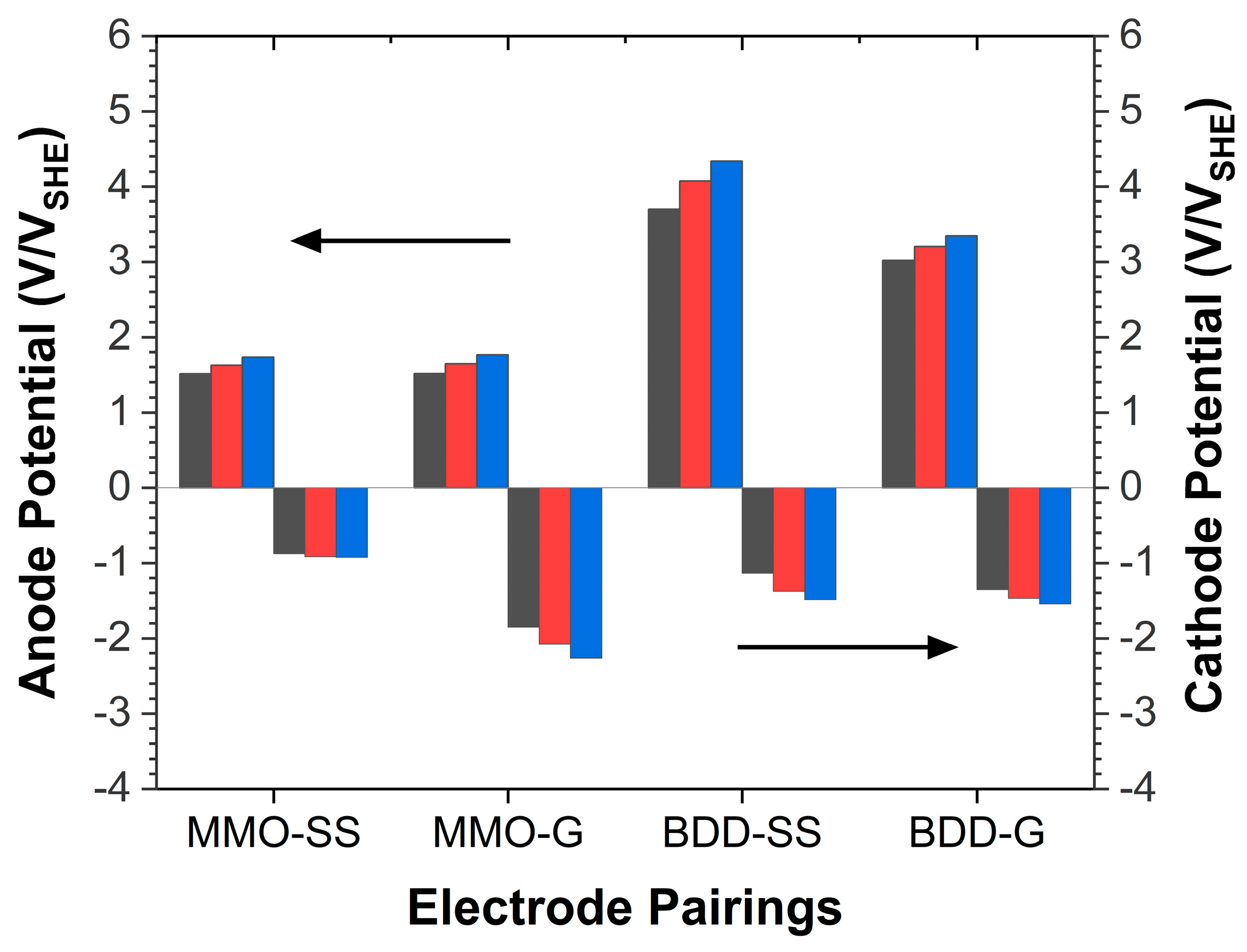

 ,
,  : 2 mA/cm2;
: 2 mA/cm2;  ,
,  : 4 mA/cm2;
: 4 mA/cm2;  ,
,  : 6 mA/cm2) for different electrode pairings. The scatter plots represent the experimental data, while the dotted lines indicate the theoretical model curves.
: 6 mA/cm2) for different electrode pairings. The scatter plots represent the experimental data, while the dotted lines indicate the theoretical model curves.
 ,
,  : 2 mA/cm2;
: 2 mA/cm2;  ,
,  : 4 mA/cm2;
: 4 mA/cm2;  ,
,  : 6 mA/cm2) for different electrode pairings. The scatter plots represent the experimental data, while the dotted lines indicate the theoretical model curves.
: 6 mA/cm2) for different electrode pairings. The scatter plots represent the experimental data, while the dotted lines indicate the theoretical model curves.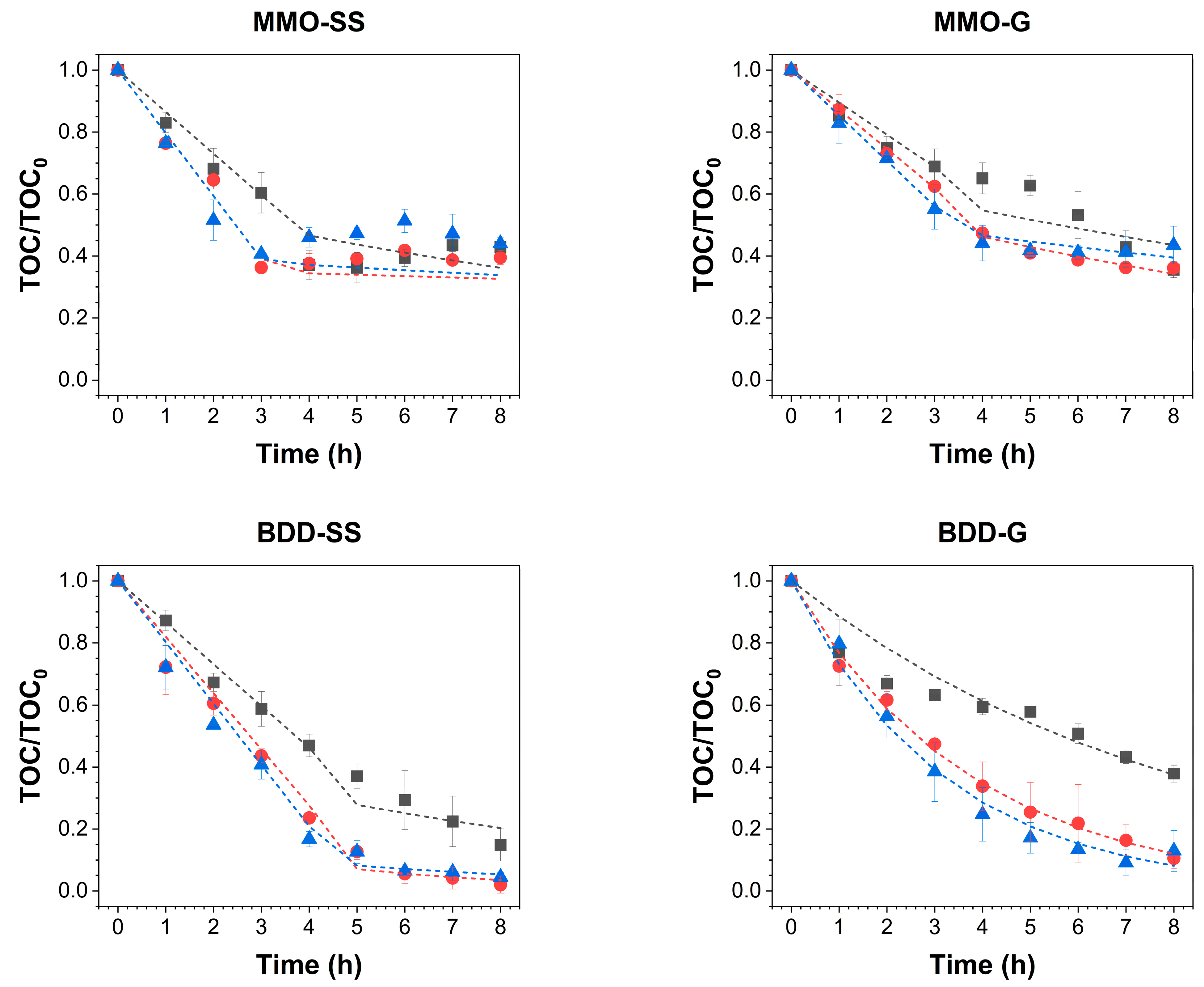
 : 2 mA/cm2,
: 2 mA/cm2,  : 4 mA/cm2,
: 4 mA/cm2,  : 6 mA/cm2) using different electrode pairings.
: 6 mA/cm2) using different electrode pairings.
 : 2 mA/cm2,
: 2 mA/cm2,  : 4 mA/cm2,
: 4 mA/cm2,  : 6 mA/cm2) using different electrode pairings.
: 6 mA/cm2) using different electrode pairings.
 : 2 mA/cm2,
: 2 mA/cm2,  : 4 mA/cm2,
: 4 mA/cm2,  : 6 mA/cm2) using different electrode pairings.
: 6 mA/cm2) using different electrode pairings.
 : 2 mA/cm2,
: 2 mA/cm2,  : 4 mA/cm2,
: 4 mA/cm2,  : 6 mA/cm2) using different electrode pairings.
: 6 mA/cm2) using different electrode pairings.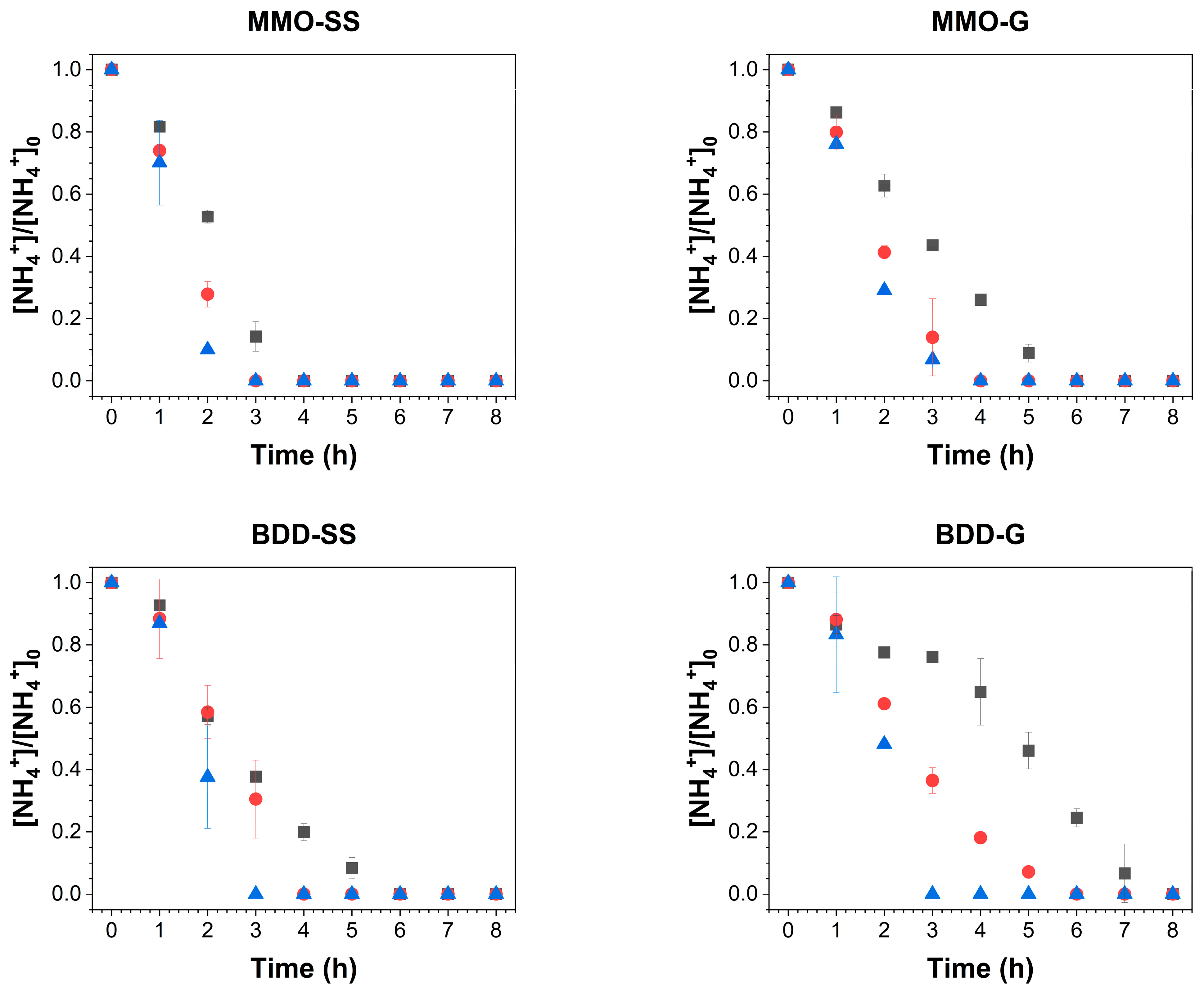
 : MMO-SS,
: MMO-SS,  : MMO-G,
: MMO-G,  : BDD-SS,
: BDD-SS,  : BDD-G). The scatter plots represent the experimental data, while the dotted lines indicate the theoretical model curves.
: BDD-G). The scatter plots represent the experimental data, while the dotted lines indicate the theoretical model curves.
 : MMO-SS,
: MMO-SS,  : MMO-G,
: MMO-G,  : BDD-SS,
: BDD-SS,  : BDD-G). The scatter plots represent the experimental data, while the dotted lines indicate the theoretical model curves.
: BDD-G). The scatter plots represent the experimental data, while the dotted lines indicate the theoretical model curves.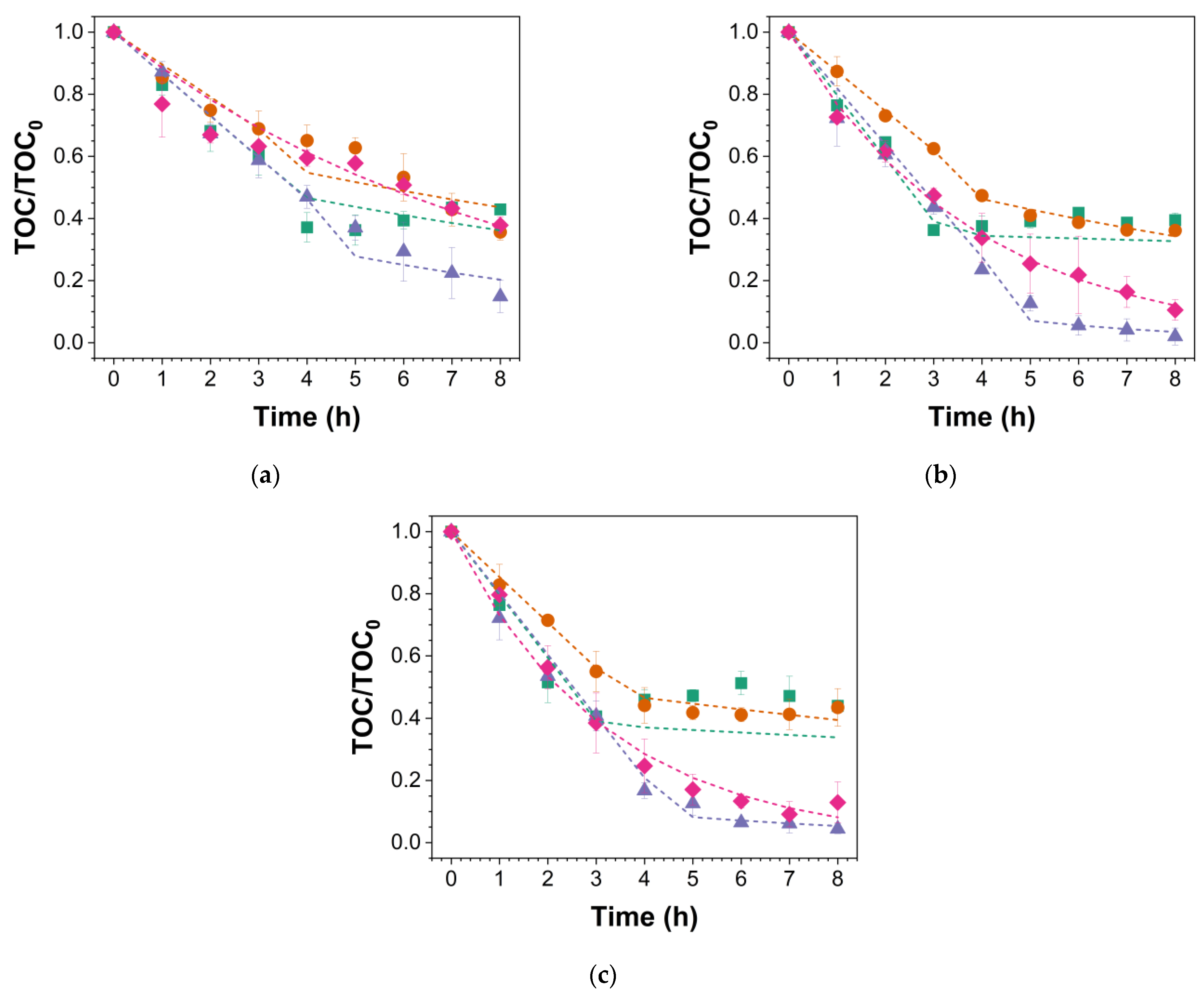
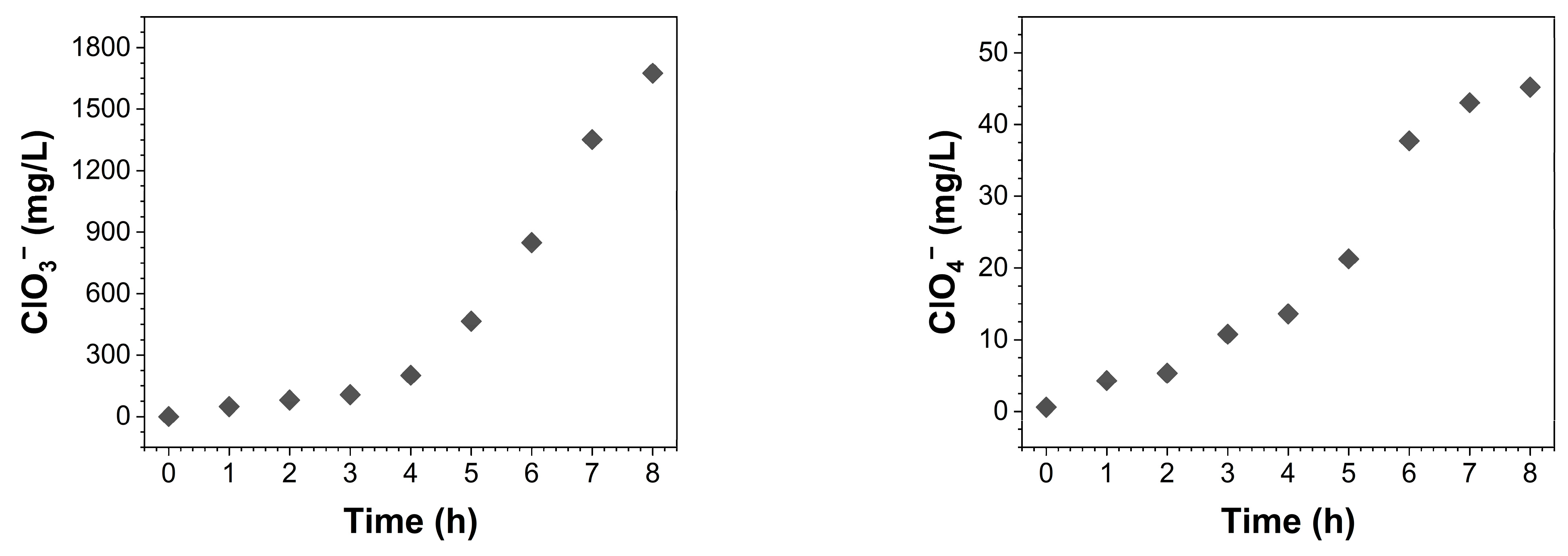
 : 2 mA/cm2,
: 2 mA/cm2,  : 4 mA/cm2,
: 4 mA/cm2,  : 6 mA/cm2).
: 6 mA/cm2).
 : 2 mA/cm2,
: 2 mA/cm2,  : 4 mA/cm2,
: 4 mA/cm2,  : 6 mA/cm2).
: 6 mA/cm2).
| Parameter | Simulated Tannery Wastewater |
|---|---|
| pH | 4.29 ± 0.12 |
| Electrical conductivity (mS/cm) | 23.6 ± 0.9 |
| Temperature (°C) | 27.1 ± 0.7 |
| Chloride, Cl− (mg/L) | 5687 ± 221 |
| Sulfate, SO42− (mg/L) | 5415 ± 74 |
| Ammonium, NH4+ (mg/L) | 720 ± 33 |
| Total Nitrogen, TN (mg/L) | 503 ± 40 |
| Total Organic Carbon, TOC (mg/L) | 489 ± 77 |
| Electrode Pairings | Tariff Costs (USD/m3) | Anode Costs (USD/m3) | Cathode Costs (USD/m3) | Total Operational Costs (USD/m3) |
|---|---|---|---|---|
| MMO-SS | 1.42 | 45 | 45 | 91.42 |
| MMO-G | 1.56 | 45 | 90 | 136.56 |
| BDD-SS | 4.40 | 600 | 45 | 649.40 |
| BDD-G | 3.61 | 600 | 90 | 693.61 |
| Electrode Pairings | Current OC (USD/m3) | Alternative OC (USD/m3) |
|---|---|---|
| MMO-SS | 91.42 | 91.42 |
| MMO-G | 136.56 | 54.36 |
| BDD-SS | 649.40 | 296.90 |
| BDD-G | 693.61 | 258.91 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Nafiat, N.; Mohd Junaidi, M.U.; Hussain, M.A.; Rabuni, M.F.; Chua, A.S.M.; Adnan, F.H. Multielectrode Advanced Oxidation Treatment of Tannery Wastewater: Mass Transfer Characterization, Process Performance, Kinetic Modeling, and Energetic Analysis. Processes 2026, 14, 184. https://doi.org/10.3390/pr14020184
Nafiat N, Mohd Junaidi MU, Hussain MA, Rabuni MF, Chua ASM, Adnan FH. Multielectrode Advanced Oxidation Treatment of Tannery Wastewater: Mass Transfer Characterization, Process Performance, Kinetic Modeling, and Energetic Analysis. Processes. 2026; 14(2):184. https://doi.org/10.3390/pr14020184
Chicago/Turabian StyleNafiat, Niswah, Mohd Usman Mohd Junaidi, Mohd Azlan Hussain, Mohamad Fairus Rabuni, Adeline Seak May Chua, and Faidzul Hakim Adnan. 2026. "Multielectrode Advanced Oxidation Treatment of Tannery Wastewater: Mass Transfer Characterization, Process Performance, Kinetic Modeling, and Energetic Analysis" Processes 14, no. 2: 184. https://doi.org/10.3390/pr14020184
APA StyleNafiat, N., Mohd Junaidi, M. U., Hussain, M. A., Rabuni, M. F., Chua, A. S. M., & Adnan, F. H. (2026). Multielectrode Advanced Oxidation Treatment of Tannery Wastewater: Mass Transfer Characterization, Process Performance, Kinetic Modeling, and Energetic Analysis. Processes, 14(2), 184. https://doi.org/10.3390/pr14020184









