Iron and Hydrogen Peroxidation-Induced Post-Treatment Improvement of Municipal Mesophilic Digestate in an Alkaline Environment and Its Impact on Biosolids Quality
Abstract
:1. Introduction
2. Materials and Method
2.1. Sampling of MD
2.2. Methodology
2.3. Experimental Set-Up
2.4. Analytical Methods
3. Results and Discussion
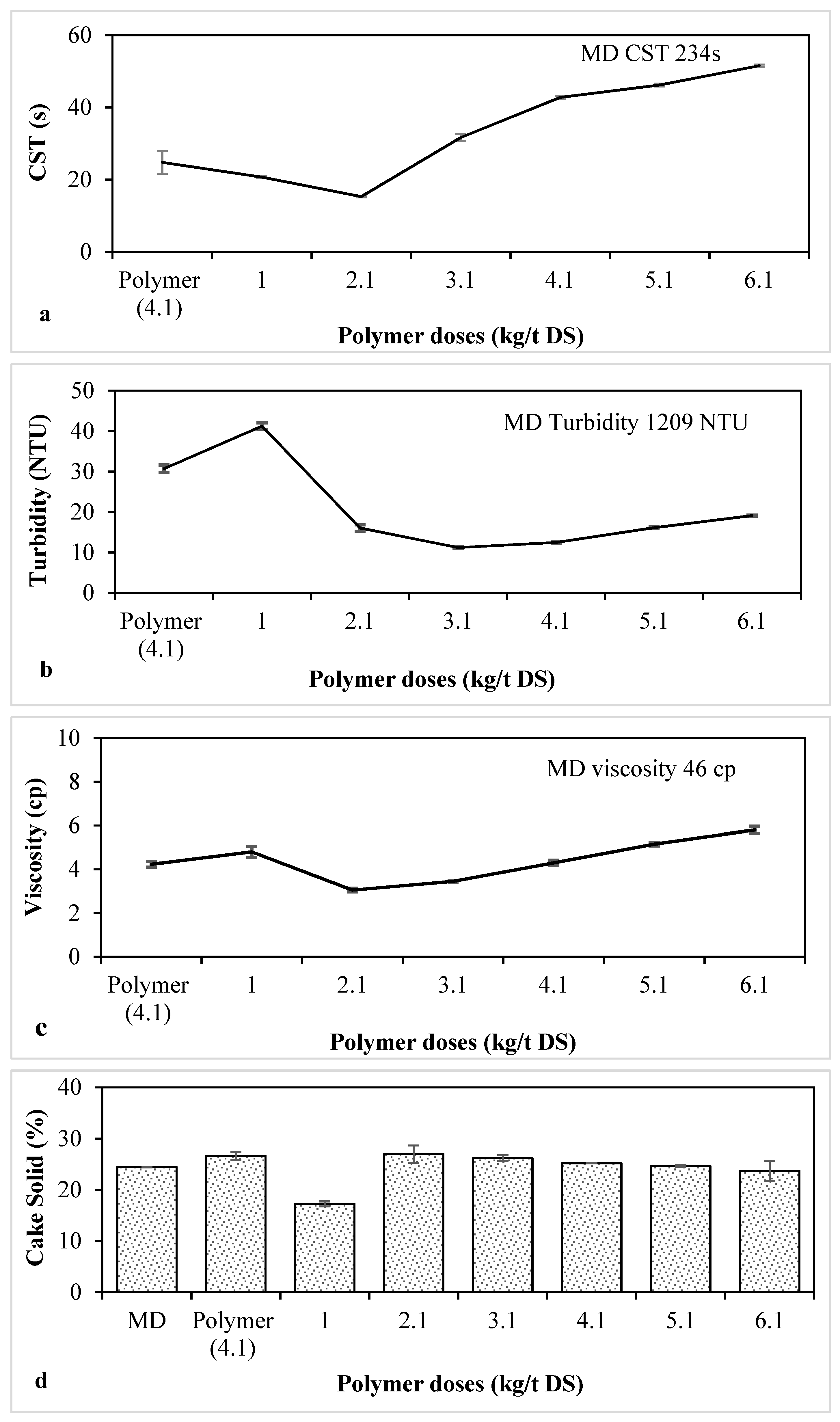
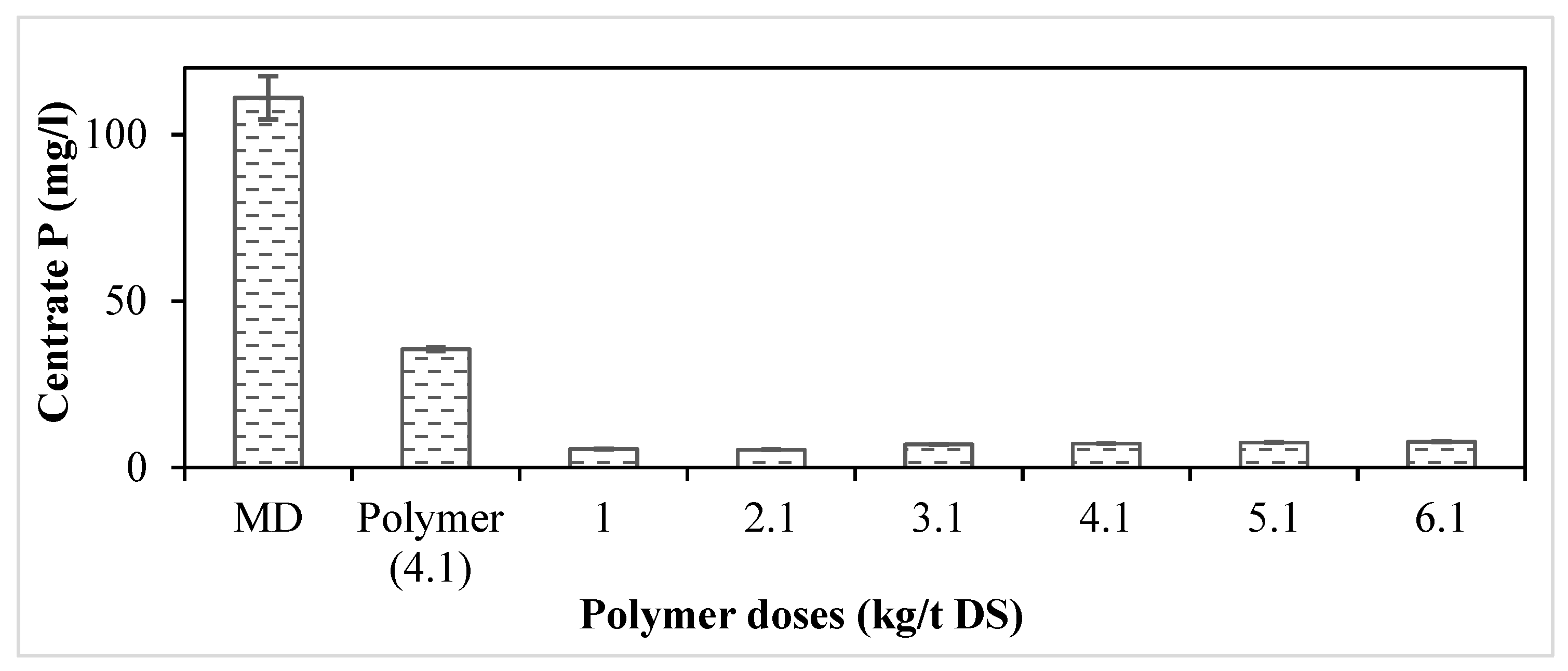
3.1. Determination of the Optimum Polymer Dose for MD
3.2. Dual Conditioning of MD with FeCl3 and Polymer with pH Adjustment
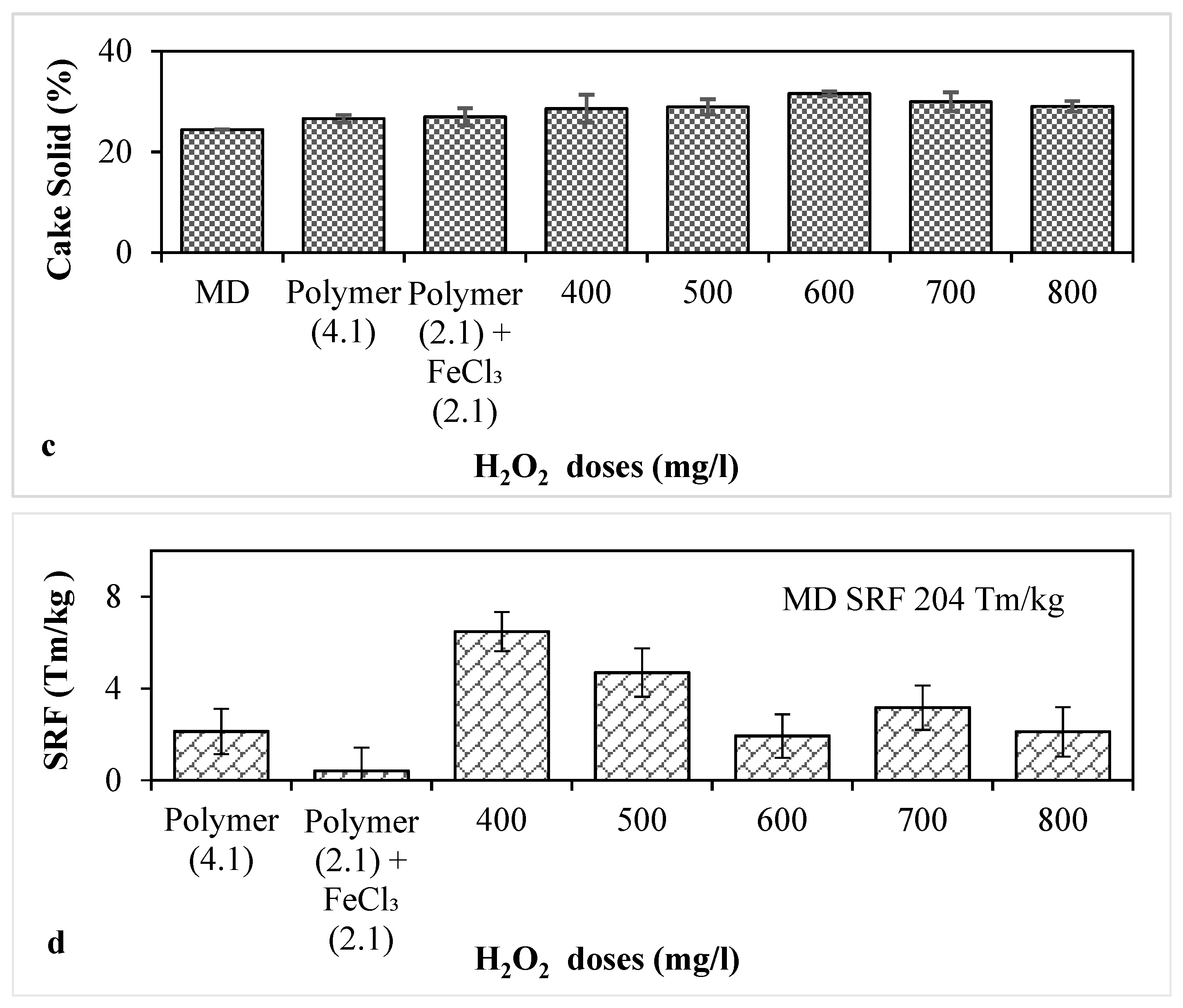
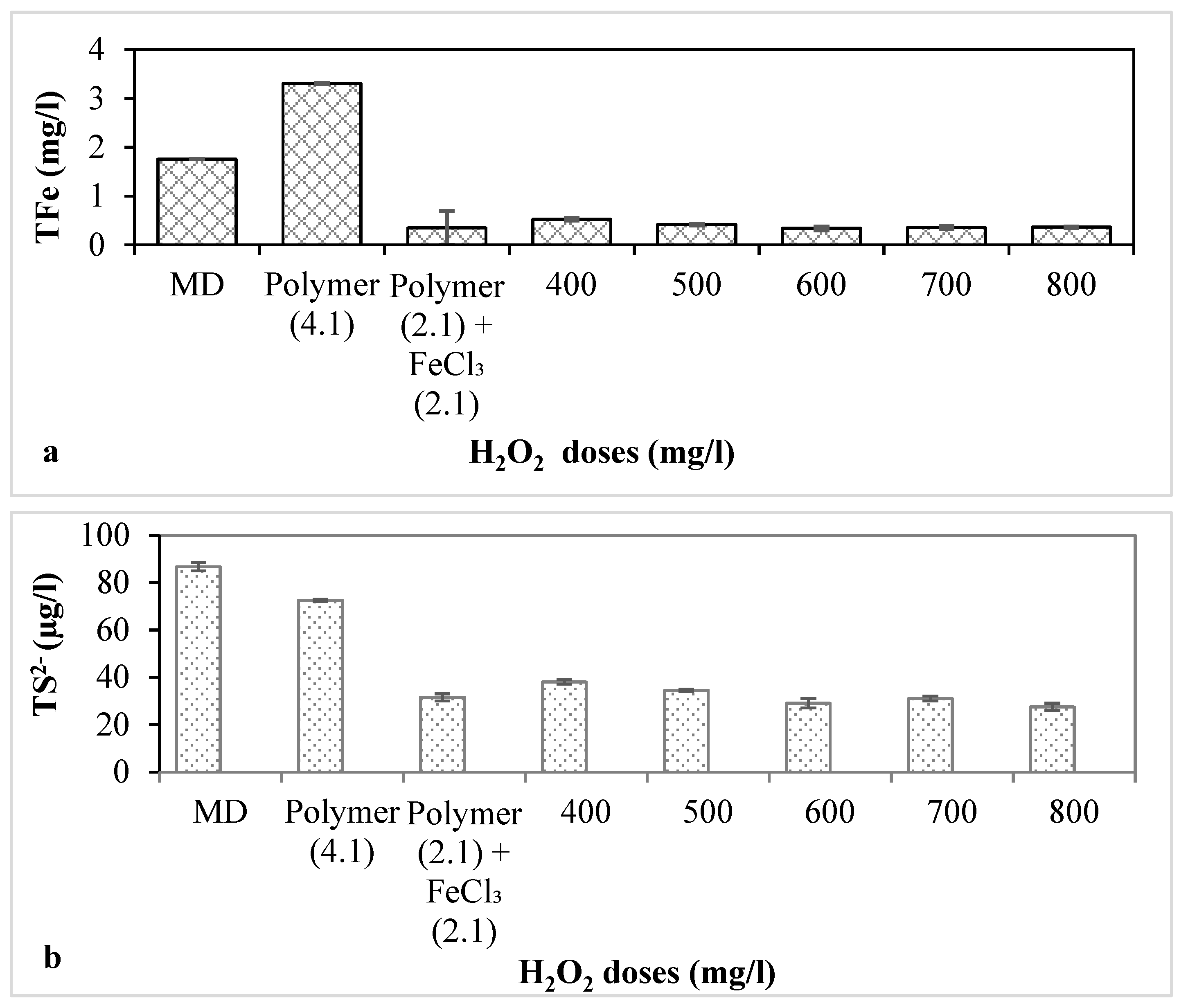
3.3. Combined Conditioning with Polymer, FeCl3 and H2O2 for MD with pH Adjusted at 8.0
3.4. Biosolids Quality Analysis
3.5. Comparison of Different Doses
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Mazhar, S.; Ditta, A.; Bulgariu, L.; Ahmad, I.; Ahmed, M.; Nadiri, A.A. Sequential treatment of paper and pulp industrial wastewater: Prediction of water quality parameters by Mamdani Fuzzy Logic model and phytotoxicity assessment. Chemosphere 2019, 227, 256–268. [Google Scholar] [CrossRef]
- Tetteh, E.K.; Rathilal, S.; Tetteh, E.K.; Rathilal, S. Application of Organic Coagulants in Water and Wastewater Treatment. In Organic Polymers; IntechOpen: London, UK, 2019. [Google Scholar] [CrossRef]
- Christensen, M.L.; Keiding, K.; Nielsen, P.H.; Jørgensen, M.K. Dewatering in biological wastewater treatment: A review. Water Res. 2015, 82, 14–24. [Google Scholar] [CrossRef] [PubMed]
- Al Momani, F.A.; Örmeci, B. Optimization of polymer dose based on residual polymer concentration in dewatering supernatant. Water Air Soil Pollut. 2014, 225, 2154. [Google Scholar] [CrossRef]
- Nie, E.; He, P.; Zhang, H.; Hao, L.; Shao, L.; Lü, F. How does temperature regulate anaerobic digestion? Renew. Sustain. Energy Rev. 2021, 150, 111453. [Google Scholar] [CrossRef]
- Spinosa, L.; Vesilind, P.A. Sludge into Biosolids—Processing, Disposal, Utilization 6; IWA Publishing: London, UK, 2007. [Google Scholar] [CrossRef]
- Wilson, C.; Murthy, S.; Fang, Y.; Novak, J. The Effect of Digester Temperature on the Production of Volatile Organic Sulfur Compounds Associated with Thermophilic Anaerobic Biosolids. Proc. Water Environ. Fed. 2006, 2006, 6830–6847. [Google Scholar] [CrossRef]
- Bay, W.A. Ashbridges Bay Wastewater Treatment Plant. 2021 Annual Report. Available online: https://www.toronto.ca/wp-content/uploads/2022/05/903e-2021-TAB-Annual-Report-FINAL.pdf (accessed on 17 August 2023).
- Heisler, J.; Glibert, P.; Burkholder, J.; Anderson, D.; Cochlan, W.; Dennison, W.; Dortch, Q.; Gobler, C.; Heil, C.; Humphries, E.; et al. Eutrophication and Harmful Algal Blooms: A Scientific Consensus. Harmful Algae 2008, 8, 3–13. [Google Scholar] [CrossRef]
- Zander, Z.; Newton, D.; Scaglione, H.; Reiber, A.; Agarwal, P. Microbial susceptibility of various polymers and evaluation of thermoplastic elastomers with antimicrobial additives. Polym. Eng. Sci. 2021, 61, 3029–3036. [Google Scholar] [CrossRef]
- Dai, Y.; Huang, S.; Liang, J.; Zhang, S.; Sun, S.; Tang, B.; Xu, Q. Role of organic compounds from different EPS fractions and their effect on sludge dewaterability by combining anaerobically mesophilic digestion pre-treatment and Fenton’s reagent/lime. Chem. Eng. J. 2017, 321, 123–138. [Google Scholar] [CrossRef]
- Huang, L.; Jin, Y.; Zhou, D.; Liu, L.; Huang, S.; Zhao, Y.; Chen, Y. A Review of the Role of Extracellular Polymeric Substances (EPS) in Wastewater Treatment Systems. Int. J. Environ. Res. Public Heal. 2022, 19, 12191. [Google Scholar] [CrossRef]
- Hyder, U.S.; Kakar, F.; Okoye, F.; Elbeshbishy, E. Management of digestate from anaerobic digestion of municipal sludge. In Anaerobic Digestate Management; IWA Publishing: London, UK, 2022; pp. 75–110. [Google Scholar] [CrossRef]
- Yu, W.; Wan, Y.; Wang, Y.; Zhu, Y.; Tao, S.; Xu, Q.; Xiao, K.; Liang, S.; Liu, B.; Hou, H.; et al. Enhancing waste activated sludge dewaterability by reducing interaction energy of sludge flocs. Environ. Res. 2021, 196, 110328. [Google Scholar] [CrossRef]
- Liang, J.; Zhou, Y. Iron-based advanced oxidation processes for enhancing sludge dewaterability: State of the art, challenges, and sludge reuse. Water Res. 2022, 218, 118499. [Google Scholar] [CrossRef] [PubMed]
- Asensi, E.; Alemany, E.; Duque-Sarango, P.; Aguado, D. Assessment and modelling of the effect of precipitated ferric chloride addition on the activated sludge settling properties. Chem. Eng. Res. Des. 2019, 150, 14–25. [Google Scholar] [CrossRef]
- Sanin, F.D.; Clarkson, W.W.; Vesilind, P.A. Sludge Engineering: The Treatment and Disposal of Wastewater Sludges; DEStech Publications, Inc.: Lancaster, PA, USA, 2011. [Google Scholar]
- Yan, Y.; Sun, X.; Ma, F.; Li, J.; Shen, J.; Han, W.; Liu, X.; Wang, L. Removal of phosphate from wastewater using alkaline residue. J. Environ. Sci. 2014, 26, 970–980. [Google Scholar] [CrossRef]
- Siami, S.; Aminzadeh, B.; Karimi, R.; Hallaji, S.M. Process optimization and effect of thermal, alkaline, H2O2 oxidation and combination pretreatment of sewage sludge on solubilization and anaerobic digestion. BMC Biotechnol. 2020, 20, 21. [Google Scholar] [CrossRef] [PubMed]
- Yu, W.; Yang, J.; Shi, Y.; Song, J.; Shi, Y.; Xiao, J.; Li, C.; Xu, X.; He, S.; Liang, S.; et al. Roles of iron species and pH optimization on sewage sludge conditioning with Fenton’s reagent and lime. Water Res. 2016, 95, 124–133. [Google Scholar] [CrossRef] [PubMed]
- Nepfumbada, C.; Tavengwa, N.T.; Masindi, V.; Foteinis, S.; Chatzisymeon, E. Recovery of phosphate from municipal wastewater as calcium phosphate and its subsequent application for the treatment of acid mine drainage. Resour. Conserv. Recycl. 2023, 190, 106779. [Google Scholar] [CrossRef]
- Martin, B.D.; Parsons, S.A.; Jefferson, B. Removal and recovery of phosphate from municipal wastewaters using a polymeric anion exchanger bound with hydrated ferric oxide nanoparticles. Water Sci. Technol. 2009, 60, 2637–2645. [Google Scholar] [CrossRef]
- CCME. Canadian Water Quality Guidelines for the Protection of Aquatic Life—Phosphorus: Canadian Guidance Framework for the Management of Freshwater Systems; Canadian Council of Ministers of the Environment: Winnipeg, MB, Canada, 2004. Available online: https://www.ccme.ca/ (accessed on 17 August 2023).
- Guan, R.; Yuan, X.; Wu, Z.; Jiang, L.; Li, Y.; Zeng, G. Principle and application of hydrogen peroxide based advanced oxidation processes in activated sludge treatment: A review. Chem. Eng. J. 2018, 339, 519–530. [Google Scholar] [CrossRef]
- Ateeq, F. Chemical Removal of Total Phosphorus from Wastewater to Low Levels and Its Analysis. Master’s Thesis, Wilfrid Laurier University, Waterloo, ON, Canada, 2016. [Google Scholar]
- Petrucci, E.; Di Palma, L.; Merli, C. Oxidation of phosphorus compounds by Fenton’s reagent. Ann. Chim. 2003, 93, 935–945. [Google Scholar]
- Mansour, Y.H.; Othmani, B.; Rebah, F.B.; Mnif, W.; Saoudi, M.; Khadhraoui, M. Could Plant-Based Flocculants Substitute the Conventional Synthetic Chemicals in the Sludge Dewatering Process? Water 2023, 15, 2602. [Google Scholar] [CrossRef]
- Li, Y.; Xu, Q.; Liu, X.; Wang, Y.; Wang, D.; Yang, G.; Yuan, X.; Yang, F.; Huang, J.; Wu, Z. Peroxide/Zero-valent iron (Fe0) pretreatment for promoting dewaterability of anaerobically digested sludge: A mechanistic study. J. Hazard. Mater. 2020, 400, 123112. [Google Scholar] [CrossRef]
- Liang, J.; Zhang, S.; Ye, M.; Huang, J.; Yang, X.; Li, S.; Huang, S.; Sun, S. Improving sewage sludge dewaterability with rapid and cost-effective in-situ generation of Fe2+ combined with oxidants. Chem. Eng. J. 2020, 380, 122499. [Google Scholar] [CrossRef]
- Ola, S.; Miklas, S. Modeling the Relationship between Capillary Suction Time and Specific Resistance to Filtration. J. Environ. Eng. 2010, 136, 983–991. [Google Scholar] [CrossRef]
- Gray, N.F. Chapter Seventeen—Capillary Suction Time (CST). In Progress in Filtration and Separation; Tarleton, S., Ed.; Academic Press: Oxford, UK, 2015; pp. 659–670. [Google Scholar] [CrossRef]
- Sawalha, O.; Scholz, M. Assessment of Capillary Suction Time (CST) Test Methodologies. Environ. Technol. 2007, 28, 1377–1386. [Google Scholar] [CrossRef] [PubMed]
- Lipps, W.C.; Braun-Howland, E.B.; Baxter, T.E. Standard Methods for the Examination of Water and Wastewater, 23rd ed.; American Public Health Association: Washington, DC, USA, 2022; Available online: https://books.google.ca/books?id=b_eazwEACAAJ (accessed on 17 August 2023).
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef] [PubMed]
- Richards, C.; O’Connor, N.; Jose, D.; Barrett, A.; Regan, F. Selection and optimization of protein and carbohydrate assays for the characterization of marine biofouling. Anal. Methods 2020, 12, 2228–2236. [Google Scholar] [CrossRef]
- USEPA. Method 1680: Fecal Coliforms in Sewage Sludge (Biosolids) by Multiple-Tube Fermentation Using Lauryl Tryptose Broth (LTB) and EC Medium; no. EPA-821-R-14-009; USEPA: Washington, DC, USA, 2014.
- Allen, M.E. MacConkey Agar Plates Protocols; American Society for Microbiology: Washington, DC, 2005; pp. 1–4. [Google Scholar]
- Ferasat, Z.; Panahi, R.; Mokhtarani, B. Natural polymer matrix as safe flocculant to remove turbidity from kaolin suspension: Performance and governing mechanism. J. Environ. Manag. 2020, 255, 109939. [Google Scholar] [CrossRef]
- Abu-Orf, M.M.; Örmeci, B. Measuring Sludge Network Strength Using Rheology and Relation to Dewaterability, Filtration, and Thickening—Laboratory and Full-Scale Experiments. J. Environ. Eng. 2005, 131, 1139–1146. [Google Scholar] [CrossRef]
- Ranjbar, F.; Karrabi, M.; Danesh, S.; Gheibi, M. Improvement of wastewater sludge dewatering using ferric chloride, aluminum sulfate, and calcium oxide (experimental investigation and descriptive statistical analysis). Water Environ. Res. 2021, 93, 1138–1149. [Google Scholar] [CrossRef]
- Inam, M.; Khan, R.; Park, D.; Lee, Y.-W.; Yeom, I. Removal of Sb(III) and Sb(V) by Ferric Chloride Coagulation: Implications of Fe Solubility. Water 2018, 10, 418. [Google Scholar] [CrossRef]
- He, D.; Zhang, Y.-J.; He, C.; Yu, H.-Q. Changing profiles of bound water content and distribution in the activated sludge treatment by NaCl addition and pH modification. Chemosphere 2017, 186, 702–708. [Google Scholar] [CrossRef] [PubMed]
- Brandt, M.J.; Johnson, K.M.; Elphinston, A.J.; Ratnayaka, D.D. Chapter 8—Storage, Clarification and Chemical Treatment. In Twort’s Water Supply, 7th ed.; Brandt, M.J., Johnson, K.M., Elphinston, A.J., Ratnayaka, D.D., Eds.; Butterworth-Heinemann: Boston, MS, USA, 2017; pp. 323–366. [Google Scholar] [CrossRef]
- Oliveira, I.M.D.B. Evaluation of Sewage Digested Sludge Conditioning and Dewatering through Qualitative and Quantitative Laboratory Methodologies towards Process Monitoring; University of South Wales: Newport, UK, 2017. [Google Scholar]
- Chen, C.; Zhang, T.; Lv, L.; Chen, Y.; Tang, W.; Tang, S. Destroying the structure of extracellular polymeric substance to improve the dewatering performance of waste activated sludge by ionic liquid. Water Res. 2021, 199, 117161. [Google Scholar] [CrossRef] [PubMed]
- Xiao, K.; Chen, Y.; Jiang, X.; Tyagi, V.K.; Zhou, Y. Characterization of key organic compounds affecting sludge dewaterability during ultrasonication and acidification treatments. Water Res. 2016, 105, 470–478. [Google Scholar] [CrossRef] [PubMed]
- Hu, Z.; Zhang, T.; Lv, L.; Chen, Y.; Zhong, B.; Tang, S. Extraction performance and mechanism of TBP in the separation of Fe3+ from wet-processing phosphoric acid. Sep. Purif. Technol. 2021, 272, 118822. [Google Scholar] [CrossRef]
- Millero, F.J.; Woosley, R. The hydrolysis of Al (III) in NaCl solutions-A model for Fe (III). Environ. Sci. Technol. 2009, 43, 1818–1823. [Google Scholar] [CrossRef] [PubMed]
- Liang, J.; Zhang, S.; Huang, J.; Huang, S.; Zheng, L.; Sun, S.; Zhong, Z.; Zhang, X.; Yu, X. Comprehensive insights into the inorganic coagulants on sludge dewatering: Comparing aluminium and iron salts. J. Chem. Technol. Biotechnol. 2019, 94, 1534–1550. [Google Scholar] [CrossRef]
- Dean, J.A. Lange’s Handbook of Chemistry; McGraw-Hill: New York, NY, USA, 1999. [Google Scholar]
- Bień, B.; Bień, J.D. Influence of Digested Sludge Conditioning on the Dewatering Processes and the Quality of Sludge Liquid. Ecol. Chem. Eng. S 2020, 27, 151–164. [Google Scholar] [CrossRef]
- Kamizela, T.; Kowalczyk, M.; Zawieja, I. The Use of Chemical Methods and Magnetic Field in Conditioning and Dewatering of Digested Sewage Sludge. Water 2020, 12, 1642. [Google Scholar] [CrossRef]
- Liang, J.; Huang, J.; Zhang, L.; Sun, F.; Ye, M.; Liao, X.; Huang, S.; Sun, S. High-level waste activated sludge dewaterability using Fenton-like process based on pretreated zero valent scrap iron as an in-situ cycle iron donator. J. Hazard. Mater. 2020, 391, 122219. [Google Scholar] [CrossRef]
- Pani, N.; Tejani, V.; Anantha-Singh, T.S.; Kandya, A. Simultaneous removal of COD and Ammoniacal Nitrogen from dye intermediate manufacturing Industrial Wastewater using Fenton oxidation method. Appl. Water Sci. 2020, 10, 66. [Google Scholar] [CrossRef]
- Zhang, W.; Dai, X.; Dong, B.; Dai, L. New insights into the effect of sludge proteins on the hydrophilic/hydrophobic properties that improve sludge dewaterability during anaerobic digestion. Water Res. 2020, 173, 115503. [Google Scholar] [CrossRef] [PubMed]
- Luo, H.; Sun, Y.; Yin, Y.; Wang, Z.-W. Odor generation, identification, and control during anaerobic digestion of organic solid wastes. Adv. Bioenergy 2022, 7, 139–165. [Google Scholar] [CrossRef]
- Novak, J.T.; Verma, N.; Muller, C.D. The role of iron and aluminum in digestion n and odor formation. Water Sci. Technol. 2007, 56, 59–65. [Google Scholar] [CrossRef] [PubMed]
- Akgul, D.; Abbott, T.; Eskicioglu, C. Assessing iron and aluminum-based coagulants for odour and pathogen reductions in sludge digesters and enhanced digestate dewaterability. Sci. Total. Environ. 2017, 598, 881–888. [Google Scholar] [CrossRef]
- Novak, J.T. Effect of Aluminum and Iron on Odors, Digestion Efficiency, and Dewatering Properties; IWA Publishing: London, UK, 2010. [Google Scholar] [CrossRef]
- Chen, Q.; Liu, H.; Yang, Z.; Tan, D. Regeneration performance of spent granular activated carbon for tertiary treatment of dyeing wastewater by Fenton reagent and hydrogen peroxide. J. Mater. Cycles Waste Manag. 2017, 19, 256–264. [Google Scholar] [CrossRef]





| Parameters | Values |
|---|---|
| Temperature (°C) | 25 ± 1.9 |
| Viscosity (cp) | 46 ± 1.2 |
| Turbidity (NTU) | 1209 ± 2.9 |
| CST (s) | 234 ± 10 |
| SRF (Tm/kg) | 204.2 ± 5.8 |
| Cake solid content (%) | 24 ± 0 |
| Centrate P (mg/L) | 111 ± 6.6 |
| Cake P (mg/L) | 328 ± 2 |
| Soluble chemical oxygen demand (sCOD) (mg/L) | 1247 ± 106.7 |
| NH3-N (mg/L) | 509.1 ± 0.9 |
| PN/PS ratio | 44 ± 4 |
| Alkalinity (mg/L Ca CO3) | 2116.5 ± 12.5 |
| Centrate total iron (TFe) (mg/L) | 1.8 ± 0 |
| Centrate total sulfide (TS2−) (µg/L) | 86.7 ± 1.7 |
| MD Cake Fecal Coliform Count | MPN Index | Fecal Coliform (MPN/mL) | Fecal Coliform (MPN/g DS) |
|---|---|---|---|
| MD fecal coliform | |||
| 5-5-5 | >160.9 | >1.61 × 107 | >5.72 × 107 |
| Chemically conditioned MD fecal coliform | |||
| 1-0-0 | 0.2 | 2000 | 6728 |
| Chemically conditioned MD fecal coliform (Regrowth potential after two weeks) | |||
| 2-0-0 | 0.4 | 4000 | 13,455 |
| Types of Heat Treatment of Combined Chemically Conditioned Biosolids | Chemically Conditioned | After Two Weeks Regrowth |
|---|---|---|
| E. coli (CFU/g DS) | E. coli (CFU/g DS) | |
| MD cake (Untreated) | 1.85 × 108 | 1.85 × 108 |
| Combined chemically conditioned at 20 °C | 3.07 × 104 | 1.7 × 105 |
| 40 °C Heat treated | 1.7 × 104 | 1.6 × 105 |
| 60 °C Heat treated | 1.02 × 104 | 1.23 × 105 |
| 80 °C Heat treated | 2.39 × 103 | 1.77 × 105 |
| 100 °C Heat treated | NG | NG |
| Conditioned Chemical Doses, (Polymer & FeCl3 in kg/t DS, H2O2 mg/L) | MD | Polymer (4.1) | Polymer (2.1) + FeCl3 (2.1) at pH 8.0 | Polymer (2.1) + FeCl3 (2.1) + H2O2 (600) at pH 8.0 |
|---|---|---|---|---|
| CST (s) | 234 ± 10 | 25 ± 3.1 | 15 ± 0.2 | 8 ± 0.1 |
| Turbidity (NTU) | 1209 ± 2.9 | 31 ± 0.9 | 16 ± 0.8 | 9 ± 0.3 |
| Cake solid (%) | 25.6 ± 0 | 27 ± 0.8 | 27 ± 1.7 | 32 ± 0.5 |
| Centrate P (mg/L) | 111 ± 6.6 | 36 ± 0.5 | 7 ± 0 | 1.6 ± 1.6 |
| TS2− (µg/L) | 87 ± 1.7 | 73 ± 0.5 | 32 ± 1.5 | 29 ± 2 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hyder, U.S.; AlSayed, A.; Elbeshbishy, E.; McPhee, J.; Misir, R. Iron and Hydrogen Peroxidation-Induced Post-Treatment Improvement of Municipal Mesophilic Digestate in an Alkaline Environment and Its Impact on Biosolids Quality. Processes 2023, 11, 2752. https://doi.org/10.3390/pr11092752
Hyder US, AlSayed A, Elbeshbishy E, McPhee J, Misir R. Iron and Hydrogen Peroxidation-Induced Post-Treatment Improvement of Municipal Mesophilic Digestate in an Alkaline Environment and Its Impact on Biosolids Quality. Processes. 2023; 11(9):2752. https://doi.org/10.3390/pr11092752
Chicago/Turabian StyleHyder, Umme Sharmeen, Ahmed AlSayed, Elsayed Elbeshbishy, Joseph McPhee, and Reshmi Misir. 2023. "Iron and Hydrogen Peroxidation-Induced Post-Treatment Improvement of Municipal Mesophilic Digestate in an Alkaline Environment and Its Impact on Biosolids Quality" Processes 11, no. 9: 2752. https://doi.org/10.3390/pr11092752
APA StyleHyder, U. S., AlSayed, A., Elbeshbishy, E., McPhee, J., & Misir, R. (2023). Iron and Hydrogen Peroxidation-Induced Post-Treatment Improvement of Municipal Mesophilic Digestate in an Alkaline Environment and Its Impact on Biosolids Quality. Processes, 11(9), 2752. https://doi.org/10.3390/pr11092752








