In Vitro Effect of Molasses Concentration, pH, and Time on Chromium Removal by Trichoderma spp. from the Effluents of a Peruvian Tannery
Abstract
1. Introduction
2. Materials and Methods
2.1. Research Design
2.2. Samples
2.3. Trichoderma Culture and Preparation of Inoculum
2.4. Obtaining Molasses
2.5. Treatments
2.6. Total Chromium and Chromium Removal
- -
- C0: Initial concentration (mg/mL)
- -
- C1: Final concentration (mg/mL)
2.7. Analysis of Data
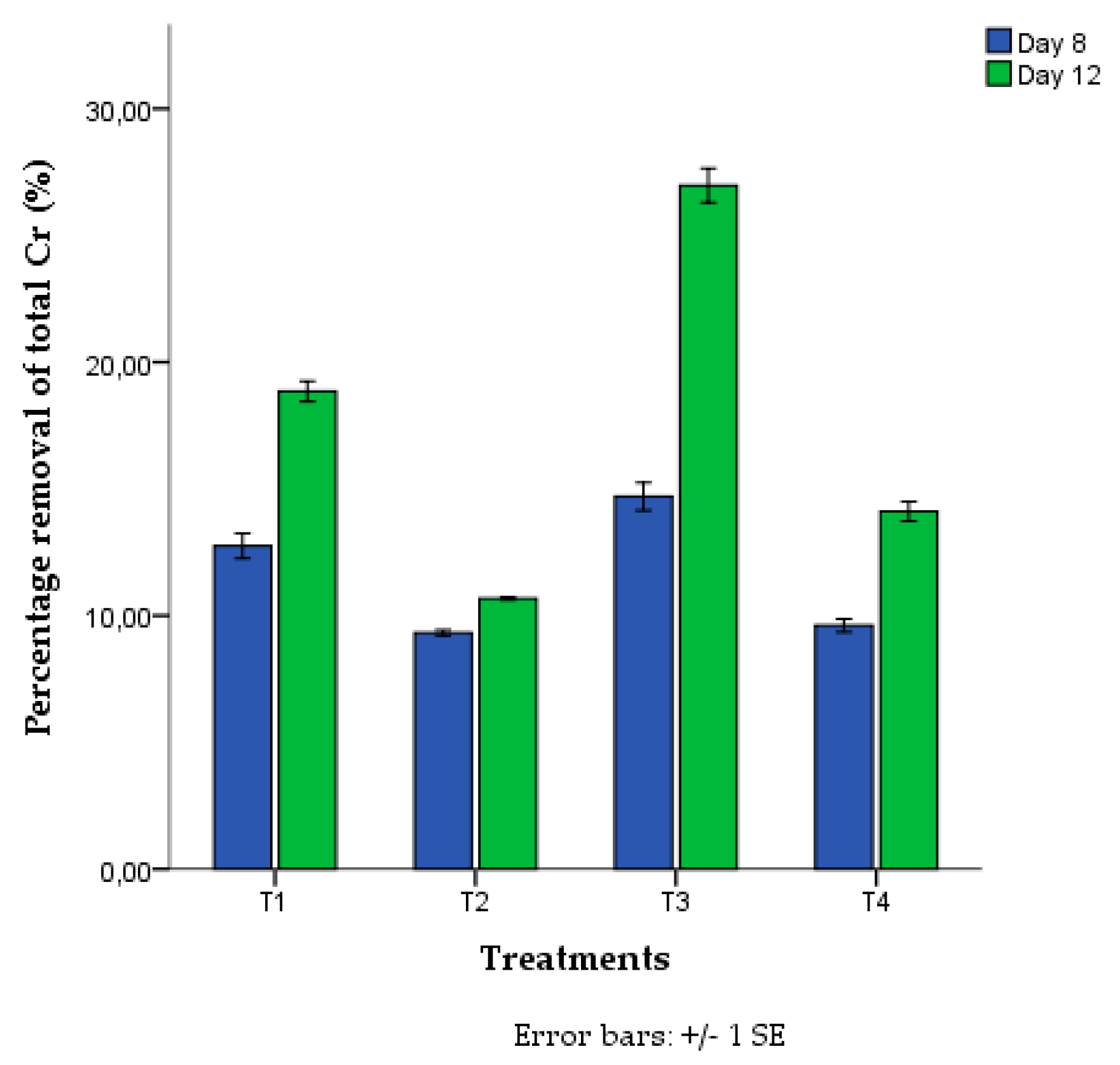
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References and Note
- Sarfraz, S.; Abid, A.J.; Javed, M.; Iqbal, S.; Aljazzar, S.O.; Zahra, M.; Alrbyawi, H.; Elkaeed, E.B.; Somaily, H.H.; Pashameah, R.A.; et al. Chromium (III) Ions Were Extracted from Wastewater Effluent Using a Synergistic Green Membrane with a BinaryCombination of D2EHPA and Kerosene. Catalysts 2022, 12, 1220. [Google Scholar] [CrossRef]
- Mitra, S.; Chakraborty, A.J.; Tareq, A.M.; Emran, T.B.; Nainu, F.; Khusro, A.; Idris, A.M.; Khandaker, M.U.; Osman, H.; Alhumaydhi, F.A.; et al. Impact of Heavy Metals on the Environment and Human Health: Novel Therapeutic Insights to Counter the Toxicity. J. King Saud Univ. Sci. 2022, 34, 101865. [Google Scholar] [CrossRef]
- De Borja Ojembarrena, F.; Sammaraie, H.; Campano, C.; Blanco, A.; Merayo, N.; Negro, C. Hexavalent Chromium Removal from Industrial Wastewater by Adsorption and Reduction onto Cationic Cellulose Nanocrystals. Nanomaterials 2022, 12, 4172. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Wang, M.; Fahad, S.; Qayyum, A.; Chen, Y.; Zhu, G. Chromium Induces Toxicity at Different Phenotypic, Physiological, Biochemical, and Ultrastructural Levels in Sweet Potato (Ipomoea batatas L). Plants. Int. J. Mol. Sci. 2022, 23, 13496. [Google Scholar] [CrossRef] [PubMed]
- Barzin, G.; Safari, F.; Bishehkolaei, R. Beneficial role of methyl jasmonate on morphological, physiological and phytochemical responses of Calendula officinalis L. under Chromium toxicity. Physiol. Mol. Biol. Plants 2022, 28, 1453–1466. [Google Scholar] [CrossRef]
- Wani, K.I.; Naeem, M.; Aftab, T. Chromium in Plant-Soil Nexus: Speciation, Uptake, Transport and Sustainable Remediation Techniques. Environ. Pollut. 2022, 315, 120350. [Google Scholar] [CrossRef]
- Den Braver-Sewradj, S.P.; van Benthem, J.; Staal, Y.C.M.; Ezendam, J.; Piersma, A.H.; Hessel, E.V.S. Occupational Exposure to Hexavalent Chromium. Part II. Hazard Assessment of Carcinogenic Effects. Regul. Toxicol. Pharmacol. 2021, 126, 105045. [Google Scholar] [CrossRef]
- Deng, Y.; Wang, M.; Tian, T.; Lin, S.; Xu, P.; Zhou, L.; Dai, C.; Hao, Q.; Wu, Y.; Zhai, Z.; et al. The Effect of Hexavalent Chromium on the Incidence and Mortality of Human Cancers: A Meta-Analysis Based on Published Epidemiological Cohort Studies. Front. Oncol. 2019, 9, 24. [Google Scholar] [CrossRef]
- Sivaram, N.M.; Barik, D. Toxic Waste from Leather Industries. In Energy from Toxic Organic Waste for Heat and Power Generation; Barik, D., Ed.; Elsevier: Amsterdam, The Netherlands, 2019; pp. 55–67. ISBN 9780081025284. [Google Scholar]
- Hedberg, Y.S.; Lidén, C.; Odnevall Wallinder, I. Chromium Released from Leather—I: Exposure Conditions That Govern the Release of Chromium(III) and Chromium(VI): Chromium Released from Leather—I. Contact Dermat. 2015, 72, 206–215. [Google Scholar] [CrossRef]
- Famielec, S. Chromium Concentrate Recovery from Solid Tannery Waste in a Thermal Process. Materials 2020, 13, 1533. [Google Scholar] [CrossRef]
- Kokkinos, E.; Proskynitopoulou, V.; Zouboulis, A. Chromium and Energy Recovery from Tannery Wastewater Treatment Waste: Investigation of Major Mechanisms in the Framework of Circular Economy. J. Environ. Chem. Eng. 2019, 7, 103307. [Google Scholar] [CrossRef]
- Das, S. (Ed.) Review of Restricted Substances in Apparel. In Product Safety and Restricted Substances in Apparel; Elsevier: Amsterdam, The Netherlands, 2013; pp. 14–28. ISBN 9780857098245. [Google Scholar]
- Yang, L.; Yang, L.; Ding, L.; Deng, F.; Luo, X.-B.; Luo, S.-L. Principles for the Application of Nanomaterials in Environmental Pollution Control and Resource Reutilization. In Nanomaterials for the Removal of Pollutants and Resource Reutilization; Luo, X., Deng, F., Eds.; Elsevier: Amsterdam, The Netherlands, 2019; pp. 1–23. ISBN 9780128148372. [Google Scholar]
- Sarfraz, S.; Abid, A.J.; Javed, M.; Iqbal, S.; Aljazzar, S.O.; Zahra, M.; Alrbyawi, H.; Elkaeed, E.B.; Somaily, H.H.; Pashameah, R.A. Effects of Chromium Toxicity on Physiological Performance and Nutrient Uptake in Two Grapevine Cultivars (Vitis vinifera L.) Growing on Own Roots or Grafted onto Different Rootstocks. In Analysis Based on Published Epidemiological Cohort Studies. Frontiers Oncology; Ezendam, Y.C.M., Piersma, J., Hessel, A.H., Eds.; Elsevier: Amsterdam, The Netherlands, 2013; Volume 12, pp. 14–28. [Google Scholar]
- Younas, F.; Bibi, I.; Afzal, M.; Al-Misned, F.; Niazi, N.K.; Hussain, K.; Shahid, M.; Shakil, Q.; Ali, F.; Wang, H. Unveiling Distribution, Hydrogeochemical Behavior and Environmental Risk of Chromium in Tannery Wastewater. Water 2023, 15, 391. [Google Scholar] [CrossRef]
- Ayele, A.; Godeto, Y.G. Bioremediation of Chromium by Microorganisms and Its Mechanisms Related to Functional Groups. J. Chem. 2021, 2021, 7694157. [Google Scholar] [CrossRef]
- Fernandez, M.; Callegari, E.A.; Paez, M.D.; González, P.S.; Agostini, E. Functional Response of Acinetobacter Guillouiae Sfc 500-1a to Tannery Wastewater as Revealed by a Complementary Proteomic Approach. Available at SSRN 4264168. [CrossRef]
- Vendruscolo, F.; da Rocha Ferreira, G.L.; Antoniosi Filho, N.R. Biosorption of Hexavalent Chromium by Microorganisms. Int. Biodeterior. Biodegrad. 2017, 119, 87–95. [Google Scholar] [CrossRef]
- Garg, S.K.; Tripathi, M.; Srinath, T. Strategies for Chromium Bioremediation of Tannery Effluent. Rev. Environ. Contam. Toxicol. 2012, 217, 75–140. [Google Scholar] [CrossRef]
- Zapana-Huarache, S.V.; Romero-Sánchez, C.K.; Gonza, A.P.D.; Torres-Huaco, F.D.; Rivera, A.M.L. Chromium (VI) Bioremediation Potential of Filamentous Fungi Isolated from Peruvian Tannery Industry Effluents. Braz. J. Microbiol. 2020, 51, 271–278. [Google Scholar] [CrossRef]
- Nongmaithem, N.; Roy, A.; Bhattacharya, P.M. Screening of Trichoderma Isolates for Their Potential of Biosorption of Nickel and Cadmium. Braz. J. Microbiol. 2016, 47, 305–313. [Google Scholar] [CrossRef]
- Sharma, S.; Malaviya, P. Bioremediation of Tannery Wastewater by Chromium Resistant Novel Fungal Consortium. Ecol. Eng. 2016, 91, 419–425. [Google Scholar] [CrossRef]
- Sharma, S.; Malaviya, P. Bioremediation of Tannery Wastewater by Chromium Resistant Fungal Isolate Fusarium Chlamydosporium SPFS2-g. Curr. World Environ. 2014, 9, 721–727. [Google Scholar] [CrossRef]
- Zafra, G.; Cortés-Espinosa, D.V. Biodegradation of Polycyclic Aromatic Hydrocarbons by Trichoderma Species: A Mini Review. Environ. Sci. Pollut. Res. Int. 2015, 22, 19426–19433. [Google Scholar] [CrossRef] [PubMed]
- Elkhateeb, W.A.; Elnahas, M.O.; Daba, G.M.; Zohri, A.-N.A. Biotechnology and Environmental Applications of Trichoderma spp. Res. J. Pharmacogn. Phytochem. 2021, 13, 149–157. [Google Scholar] [CrossRef]
- Ayad, F.; Matallah-Boutiba, A.; Rouane-Hacene, O.; Bouderbala, M.; Boutiba, Z. Tolerance of Trichoderma Sp. to Heavy Metals and Its Antifungal Activity in Algerian Marine Environment. J. Pure Appl. Microbiol. 2018, 12, 855–870. [Google Scholar] [CrossRef]
- Zhang, D.; Yin, C.; Abbas, N.; Mao, Z.; Zhang, Y. Multiple Heavy Metal Tolerance and Removal by an Earthworm Gut Fungus Trichoderma Brevicompactum QYCD-6. Sci. Rep. 2020, 10, 6940. [Google Scholar] [CrossRef] [PubMed]
- Morales-Barrera, L.; Cristiani-Urbina, E. Hexavalent Chromium Removal by a Trichoderma Inhamatum Fungal Strain Isolated from Tannery Effluent. Water Air Soil Pollut. 2007, 187, 327–336. [Google Scholar] [CrossRef]
- American Water Works Association; Water Environment Federation. 3500-Cr Chromium. In Standard Methods for the Examination of Water and Wastewater; Lipps, W.C., Baxter, T.E., Braun-Howland, E., Eds.; APHA Press: Washington, DC, USA, 2018. [Google Scholar] [CrossRef]
- Decreto Supremo Que Aprueba el Reglamento de Valores Máximos Admisibles (VMA) para las Descargas de Aguas Residuales No Domésticas en el Sistema de Alcantarillado Sanitario-DECRETO SUPREMO-N° 010-2019-VIVIENDA. Available online: https://busquedas.elperuano.pe/normaslegales/decreto-supremo-que-aprueba-el-reglamento-de-valores-maximos-decreto-supremo-n-010-2019-vivienda-1748339-3/ (accessed on 23 March 2023).
- Shamim, S. Biosorption of Heavy Metals. In Biosorption; Derco, J., Vrana, B., Eds.; InTech: London, UK, 2018; ISBN 9781789234725. [Google Scholar]
- Benila Smily, J.R.M.; Sumithra, P.A. Optimization of Chromium Biosorption by Fungal Adsorbent, Trichoderma sp. BSCR02 and Its Desorption Studies. Hayati 2017, 24, 65–71. [Google Scholar] [CrossRef]
- Narolkar, S.; Mishra, A. Biosorption of Chromium by Fungal Strains Isolated from Industrial Effluent Contaminated Area. Pollution 2022, 8, 159–168. [Google Scholar] [CrossRef]
- Abed Ali, W.J.; Walli, H.A. Bioremediation of Pollutant Waters by Trichoderma Harzianum Using EDX and Laboratory Examination. J. Phys. Conf. Ser. 2020, 1664, 012138. [Google Scholar] [CrossRef]
- Chang, F.; Tian, C.; Liu, S.; Ni, J. Discrepant Hexavalent Chromium Tolerance and Detoxification by Two Strains of Trichoderma Asperellum with High Homology. Chem. Eng. J. 2016, 298, 75–81. [Google Scholar] [CrossRef]
- Vankar, P.S.; Bajpai, D. Phyto-Remediation of Chrome-VI of Tannery Effluent by Trichoderma Species. Desalination 2008, 222, 255–262. [Google Scholar] [CrossRef]
- Rodríguez-León, J.A.; Domenech, F.; León, M.; Méndez, T.; Rodríguez, D.E.; Pandey, A. Production of Spores of Trichoderma Harzianum on Sugar Cane Molasses and Bagasse Pith in Solid State Fermentation for Biocontrol. Braz. Arch. Biol. Technol. 1999, 42, 1–21. [Google Scholar] [CrossRef]
- Kumar, V. Optimal Physical Parameters for Growth of Trichoderma Species at Varying PH, Temperature and Agitation. Virol. Mycol. 2013, 3, 1000127. [Google Scholar] [CrossRef]
- Mendoza, J.C.; Perea, Y.S.; Pretin, C.; Silveti, A. Biosorción de Cromo, Arsénico y Plomo de soluciones acuosas por cultivos bacterianos en suspensión. Rev. Latinoam. Ambiente Cienc. 2010, 1, 67–73. [Google Scholar]
- Aquise, A.; Kent, L. Biosorción de Metales Pesados por Hongos Filamentosos, Aislados de Cuerpos de Agua Altoandinos Contaminados Con Relaves Mineros de la Sierra Central del Perú; Universidad Nacional Mayor de San Marcos: Lima, Peru, 2019. [Google Scholar]
- Shukla, D.; Vankar, P.S. Role of Trichoderma Species in Bioremediation Process. In Biotechnology and Biology of Trichoderma; Gupta, V.K., Schmoll, M., Herrera-Estrella, A., Upadhyay, R.S., Druzhinina, I., Tuohy, M.G., Eds.; Elsevier: Amsterdam, The Netherlands, 2014; pp. 405–412. ISBN 9780444595768. [Google Scholar]
- Otiniano, N.M.; De La Cruz-Noriega, M.; Cabanillas-Chirinos, L.; Rojas-Flores, S.; Muñoz-Ríos, M.A.; Rojas-Villacorta, W.; Robles-Castillo, H. Arsenic Biosorption by the Macroalgae Chondracanthus Chamissoi and Cladophora sp. Processes 2022, 10, 1967. [Google Scholar] [CrossRef]
- Hlihor, R.M.; Roşca, M.; Drăgoi, E.N.; Simion, I.M.; Favier, L.; Gavrilescu, M. New Insights into the Application of Fungal Biomass for Chromium(VI) Bioremoval from Aqueous Solutions Using Design of Experiments and Differential Evolution Based Neural Network Approaches. Chem. Eng. Res. Des. 2023, 190, 233–254. [Google Scholar] [CrossRef]

| Treatments | Independent Variables * | |||
|---|---|---|---|---|
| pH | Molasses Concentration | Time (Days) | Combinations | |
| 1 | P1 | M1 | T1 | P1M1 T1 |
| 2 | P1 | M1 | T2 | P1M1 T2 |
| 3 | P1 | M2 | T1 | P1M2 T1 |
| 4 | P1 | M2 | T2 | P1M2 T2 |
| 5 | P2 | M1 | T1 | P2M1 T1 |
| 6 | P2 | M1 | T2 | P2M1 T2 |
| 7 | P2 | M2 | T1 | P2M2 T1 |
| 8 | P2 | M2 | T2 | P2M2 T2 |
| Total combinations | 2(P) | 2(M) | 2(T) | 2X2X2 |
| Parameter | Value |
|---|---|
| Degree °Brix | 88.40 |
| Specific weight (g/cm) | 1.47 |
| Sucrose % | 36.41 |
| Reducing sugars % | 9.09 |
| Total sugars % | 45.50 |
| Do not sugar % | 42.90 |
| Water % | 11.60 |
| Treatments | Effluent 20% (mL) | Molasses (%) a | Trichoderma Inoculum (%) b | pH | Evaluation Time |
|---|---|---|---|---|---|
| T0 | 500 | --- | --- | --- | --- |
| T1 | 500 | 1.0 | 10 | 6 | 8 |
| 12 | |||||
| T2 | 500 | 0.5 | 10 | 6 | 8 |
| 12 | |||||
| T3 | 500 | 1.0 | 10 | 4 | 8 |
| 12 | |||||
| T4 | 500 | 0.5 | 10 | 4 | 8 |
| 12 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tello-Galarreta, F.A.; Durand-Paz, J.H.; Rojas-Villacorta, W.; Cabanillas-Chirinos, L.; De La Cruz-Noriega, M.; Nazario-Naveda, R.; Benites, S.M.; Rojas-Flores, S. In Vitro Effect of Molasses Concentration, pH, and Time on Chromium Removal by Trichoderma spp. from the Effluents of a Peruvian Tannery. Processes 2023, 11, 1557. https://doi.org/10.3390/pr11051557
Tello-Galarreta FA, Durand-Paz JH, Rojas-Villacorta W, Cabanillas-Chirinos L, De La Cruz-Noriega M, Nazario-Naveda R, Benites SM, Rojas-Flores S. In Vitro Effect of Molasses Concentration, pH, and Time on Chromium Removal by Trichoderma spp. from the Effluents of a Peruvian Tannery. Processes. 2023; 11(5):1557. https://doi.org/10.3390/pr11051557
Chicago/Turabian StyleTello-Galarreta, Fabricio A., Juan H. Durand-Paz, Walter Rojas-Villacorta, Luis Cabanillas-Chirinos, Magaly De La Cruz-Noriega, Renny Nazario-Naveda, Santiago M. Benites, and Segundo Rojas-Flores. 2023. "In Vitro Effect of Molasses Concentration, pH, and Time on Chromium Removal by Trichoderma spp. from the Effluents of a Peruvian Tannery" Processes 11, no. 5: 1557. https://doi.org/10.3390/pr11051557
APA StyleTello-Galarreta, F. A., Durand-Paz, J. H., Rojas-Villacorta, W., Cabanillas-Chirinos, L., De La Cruz-Noriega, M., Nazario-Naveda, R., Benites, S. M., & Rojas-Flores, S. (2023). In Vitro Effect of Molasses Concentration, pH, and Time on Chromium Removal by Trichoderma spp. from the Effluents of a Peruvian Tannery. Processes, 11(5), 1557. https://doi.org/10.3390/pr11051557







