Effect of Clay Minerals and Rock Fabric on Hydrocarbon Generation and Retention by Thermal Pyrolysis of Maoming Oil Shale
Abstract
1. Introduction
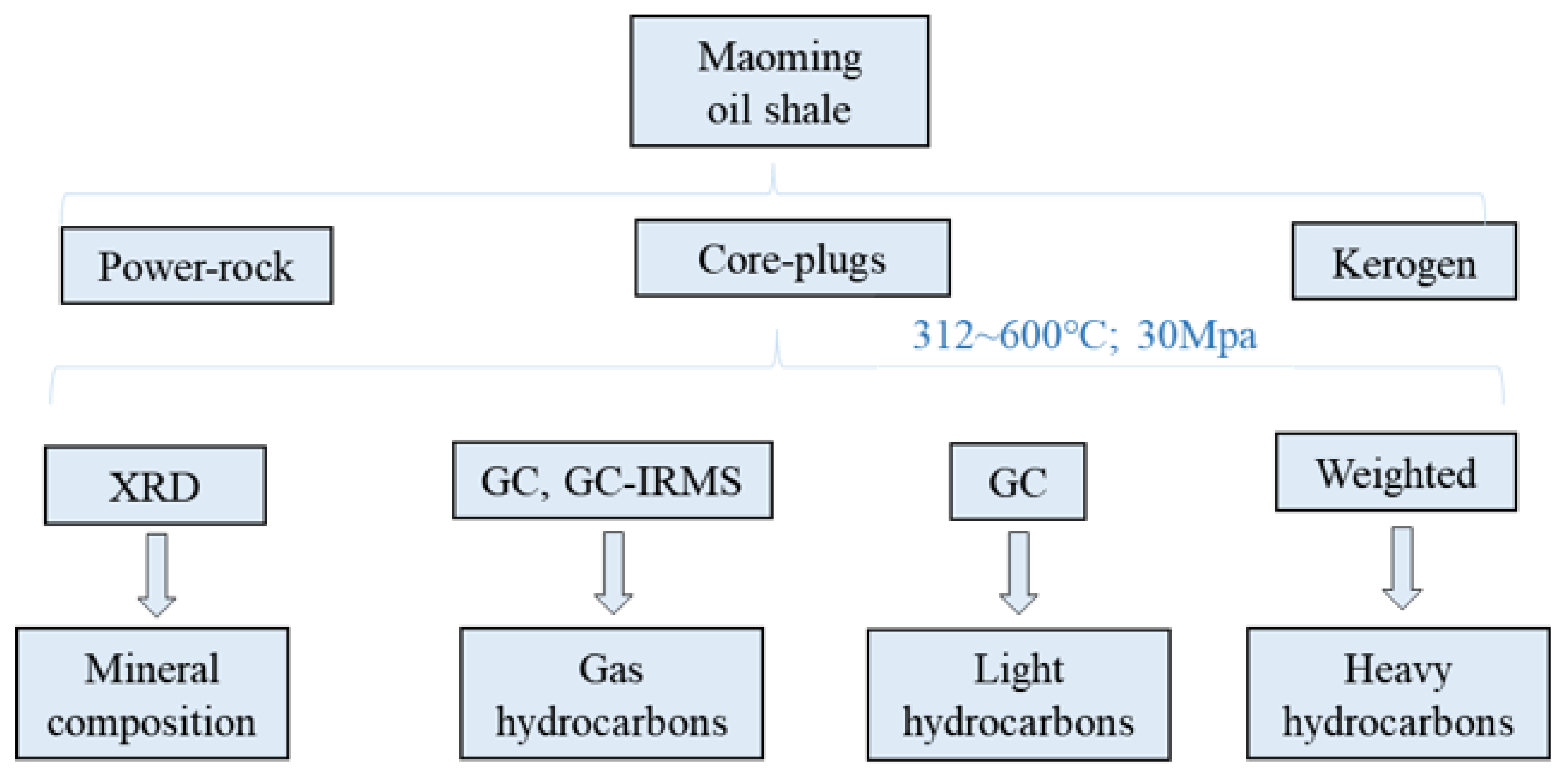
2. Experimental Procedures
2.1. Samples
2.2. Pyrolysis Apparatus
2.3. Heating Procedure
2.4. Gaseous Hydrocarbons and Inorganic Gas Analysis
2.5. Light and Heavy Hydrocarbons
3. Experimental Result
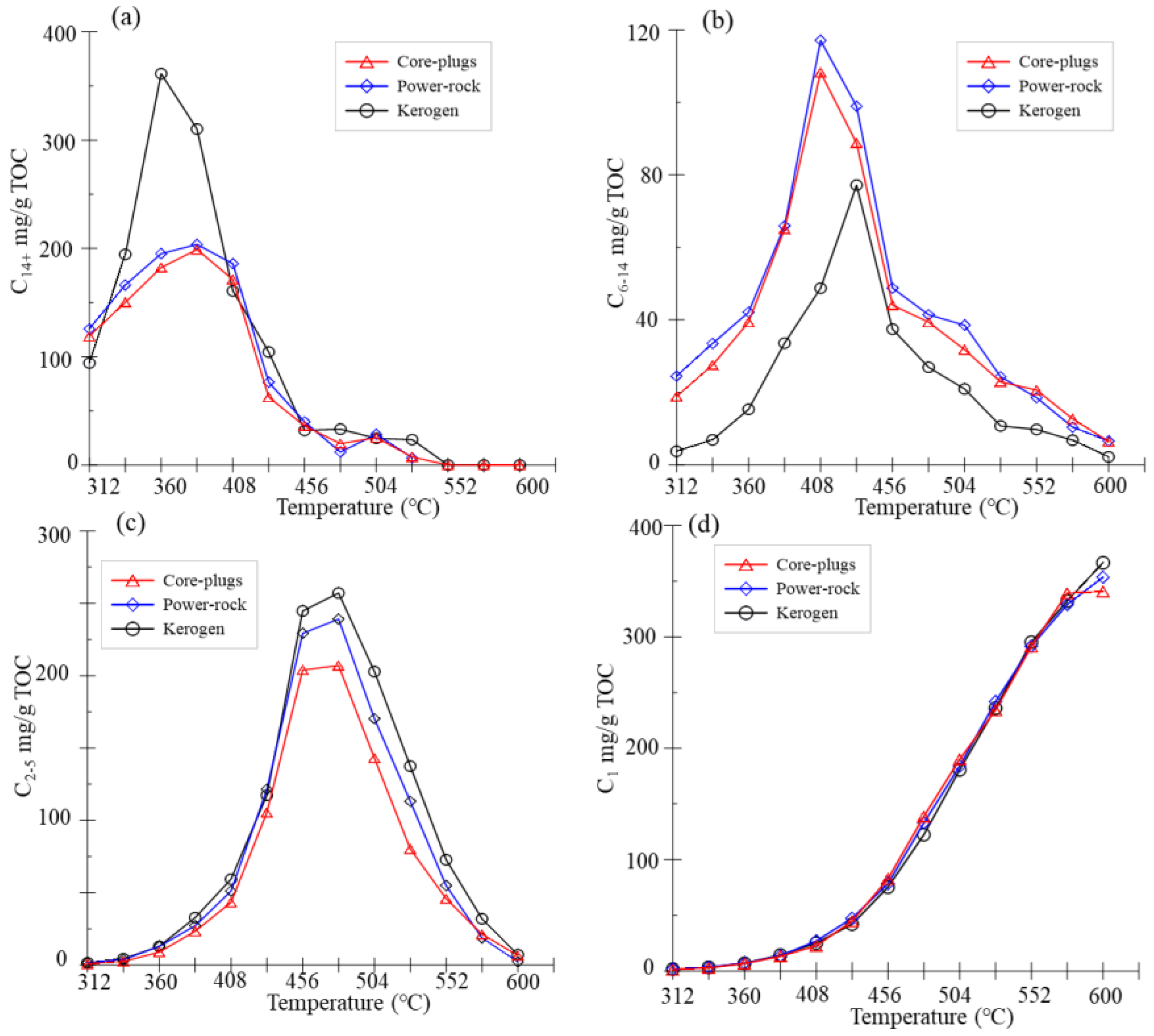
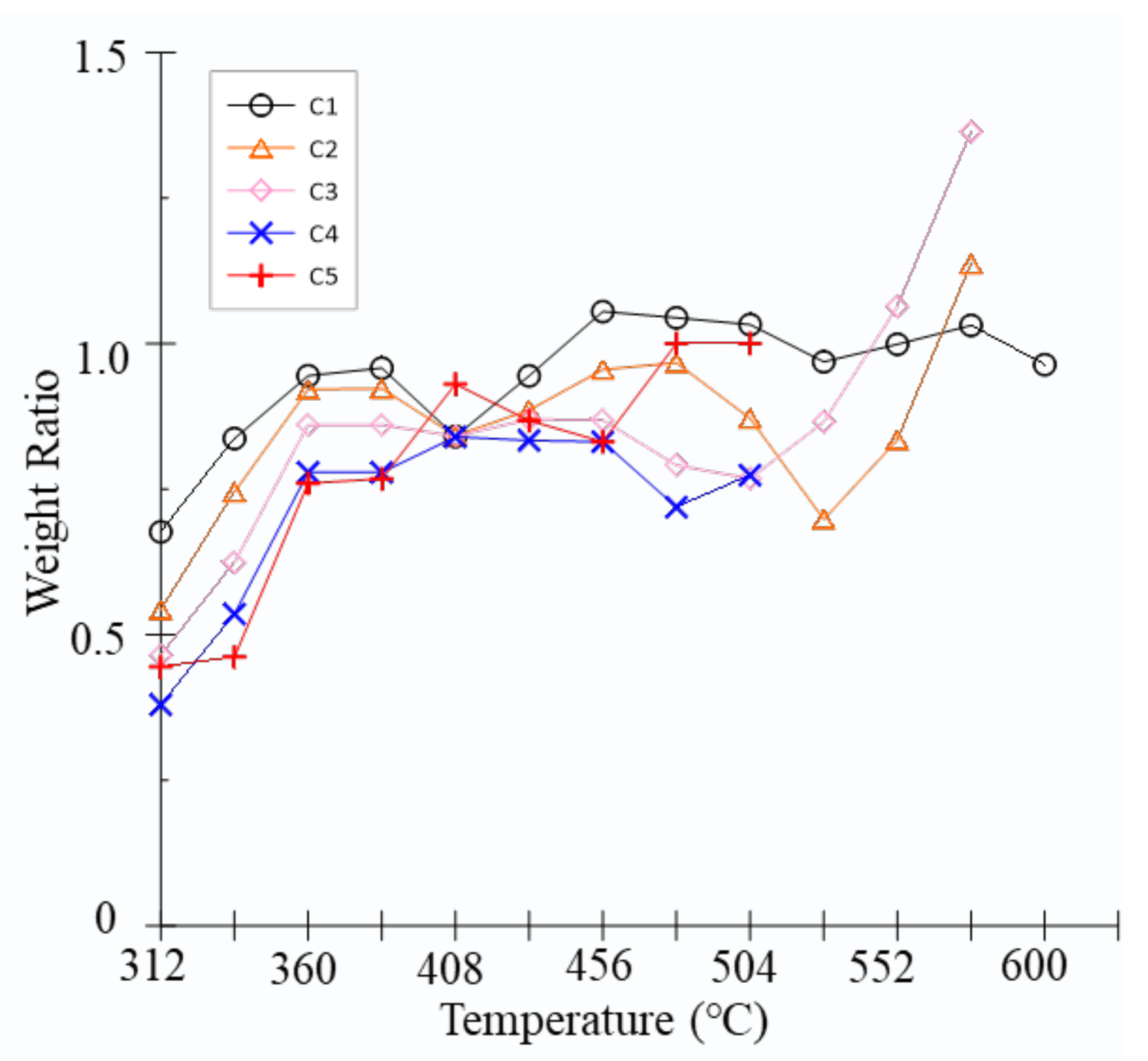
3.1. Hydrocarbon Yields
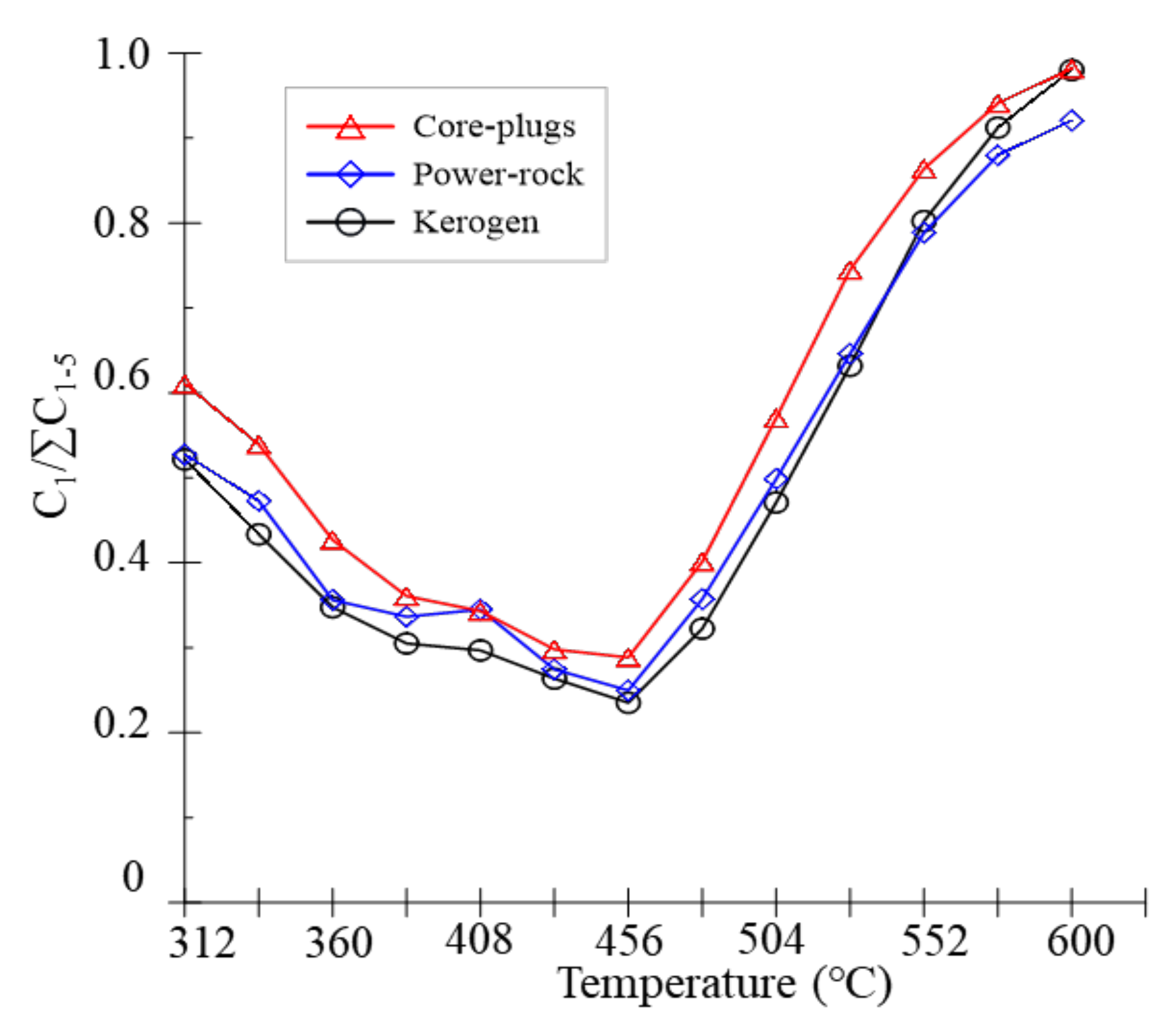
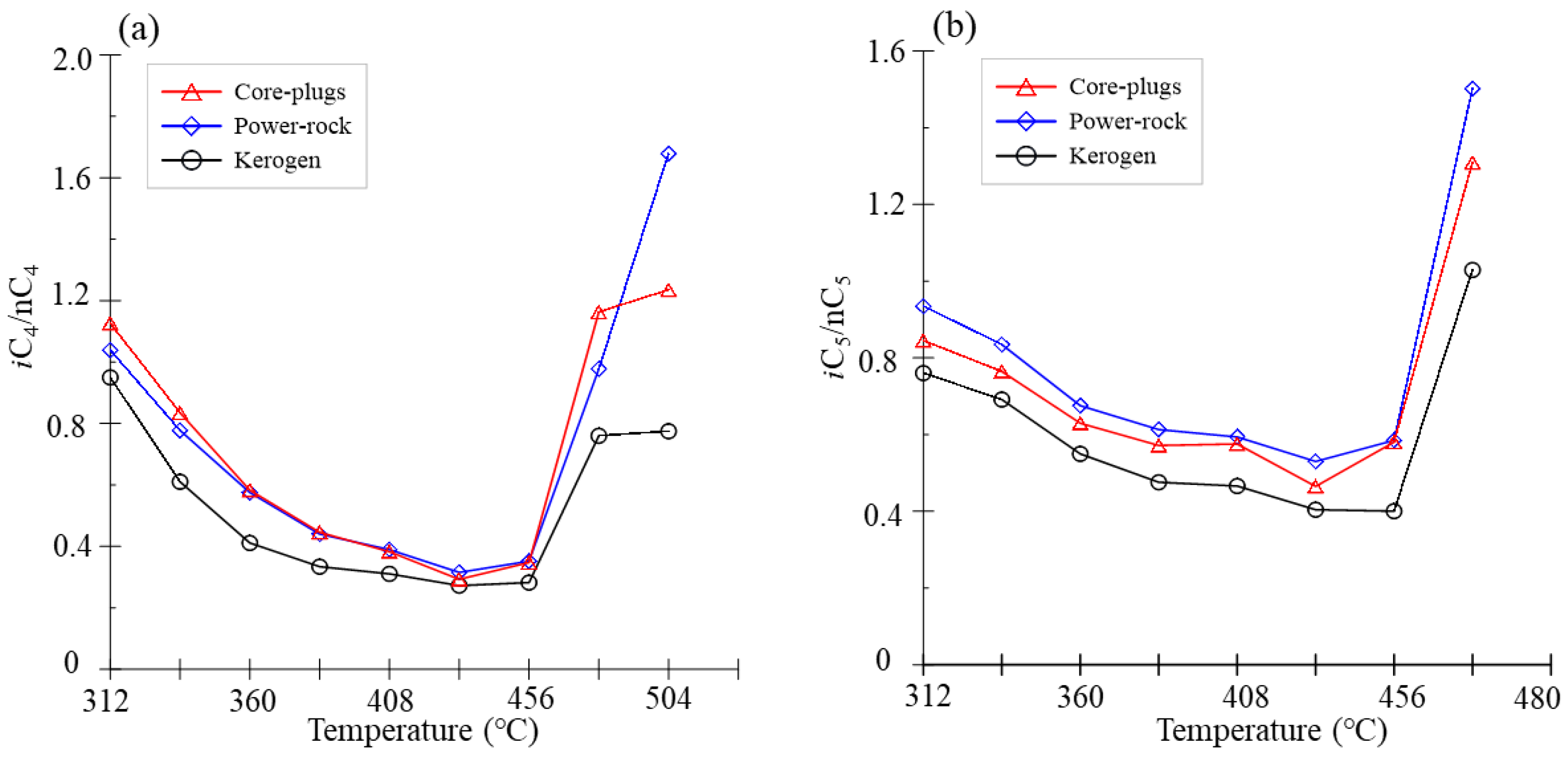
3.2. Gas Parameter Ratio
4. Discussion
4.1. Retention Effects of Clay Minerals
4.2. Effect of Rock Fabric on Gaseous Hydrocarbons
4.3. Effect of Extractable Bitumen on the Generation of Light Hydrocarbons
4.4. Implications for the Natural System
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Tissot, B.; Durand, B.; Espitalie, J.; Combaz, A. Influence of nature and diagenesis of organic matter in formation of petroleum. AAPG Bull. 1974, 58, 499–506. [Google Scholar]
- Ko, L.T.; Loucks, R.G.; Zhang, T.W.; Ruppel, S.C. Pore and pore network evolution of Upper Cretaceous Boquillas (Eagle Ford–equivalent) mudrocks: Results from gold tube pyrolysis experiments. AAPG Bull. 2016, 100, 1693–1722. [Google Scholar] [CrossRef]
- Ko, L.T.; Ruppel, S.C.; Loucks, R.G.; Hackley, P.C.; Zhang, T.W.; Shao, D.Y. Pore-types and pore-network evolution in Upper Devonian-Lower Mississippian Woodford and Mississippian Barnett mudstones: Insights from laboratory thermal maturation and organic petrology. Int. J. Coal Geol. 2018, 190, 3–28. [Google Scholar] [CrossRef]
- Hunt, J.M.; Lewan, M.D.; Hennet, R.J.C. Modeling oil generation with time-temperature index graphs based on the Arrhenius equation. AAPG Bull. 1991, 75, 795–807. [Google Scholar]
- Zhao, Z.F.; Feng, Q.; Liu, X.R.; Peng, P.A.; Liu, J.Z.; HSU, C.S. Petroleum Maturation Processes Simulated by High-Pressure Pyrolysis and Kinetic Modeling of Low-Maturity Type I Kerogen. Energy Fuels 2022, 36, 1882–1893. [Google Scholar] [CrossRef]
- Lewan, M.D. Evaluation of petroleum generation by hydrous pyrolysis experimentation. Philos. Trans. A Math. Phys. Sci. 1985, 315, 123–134. [Google Scholar]
- Schoell, M. Genetic characterization of natural gases. AAPG Bull. 1983, 67, 2225–2238. [Google Scholar]
- Galimov, E.M. Sources and mechanisms of formation of gaseous hydrocarbons in sedimentary rocks. Chem. Geol. 1988, 71, 77–95. [Google Scholar] [CrossRef]
- Lewan, M.D.; Williams, J.A. Evaluation of petroleum generation from resinites by hydrous pyrolysis. AAPG Bull. 1987, 71, 207–214. [Google Scholar]
- Espitalie, J.; Madec, M.; Tissot, B. Role of mineral matrix in kerogen pyrolysis: Influence on petroleum generation and migration. AAPG Bull. 1980, 64, 59–66. [Google Scholar]
- Kelemen, S.R.; Sansone, M.; Walters, C.C.; Kwiatek, P.J.; Bolin, T. Thermal transformations of organic and inorganic sulfur in Type II kerogen quantified by S-XANES. Geochim. Cosmochim. Acta 2012, 83, 61–78. [Google Scholar] [CrossRef]
- Mango, F.D. Transition metal catalysis in the generation of petroleum and natural gas. Geochim. Cosmochim. Acta 1992, 56, 553–555. [Google Scholar] [CrossRef]
- Mango, F.D.; Hightower, J.W.; James, A.T. Role of transition-metal catalysis in the formation of natural gas. Nature 1994, 368, 536–538. [Google Scholar] [CrossRef]
- Mango, F.D.; Hightower, J. The catalytic decomposition of petroleum into natural gas. Geochim. Cosmochim. Acta 1997, 61, 5347–5350. [Google Scholar] [CrossRef]
- Lewan, M.D.; Maynard, J.B. Factors controlling enrichment of vanadium and nickel in the bitumen of organic sedimentary rocks. Geochim. Cosmochim. Acta 1982, 46, 2547–2560. [Google Scholar] [CrossRef]
- Gao, J.; Liu, J.; Ni, Y. Gas generation and its isotope composition during coal pyrolysis: The catalytic effect of nickel and magnetite. Fuel 2018, 222, 74–82. [Google Scholar] [CrossRef]
- Gao, J.; Zou, C.; Li, W.; Ni, Y.; Yuan, Y. Transition metal catalysis in natural gas generation: Evidence from nonhydrous pyrolysis experiment. Mar. Pet. Geol. 2020, 115, 104280. [Google Scholar] [CrossRef]
- Medina, J.C.; Butala, S.J.; Bartholomew, C.H.; Lee, M.L. Low temperature iron-and nickel-catalyzed reactions leading to coalbed gas formation. Geochim. Cosmochim. Acta 2000, 64, 643–649. [Google Scholar] [CrossRef]
- Seewald, J.S.; Eglinton, L.B.; Ong, Y.L. An experimental study of organic-inorganic interactions during vitrinite maturation. Geochim. Cosmochim. Acta 2000, 64, 1577–1591. [Google Scholar] [CrossRef]
- Lewan, M.D. Experiments on the role of water in petroleum formation. Geochim. Cosmochim. Acta 1997, 61, 3691–3723. [Google Scholar] [CrossRef]
- Seewald, J.S. Organic–inorganic interactions in petroleum-producing sedimentary basins. Nature 2003, 426, 327–333. [Google Scholar] [CrossRef] [PubMed]
- Hill, R.J.; Jarvie, D.M.; Zumberge, J.; Henry, M.; Pollastro, R.M. Oil and gas geochemistry and petroleum systems of the Fort Worth Basin. AAPG Bull. 2007, 91, 445–473. [Google Scholar] [CrossRef]
- Jarvie, D.M.; Hill, R.J.; Ruble, T.E.; Pollastro, R.M. Unconventional shale-gas systems: The Mississippian Barnett Shale of north-central Texas as one model for thermogenic shale-gas assessment. AAPG Bull. 2007, 91, 475–499. [Google Scholar] [CrossRef]
- Curtis, J.B. Fractured shale-gas systems. AAPG Bull. 2002, 86, 1921–1938. [Google Scholar]
- Li, E.T.; Pan, C.C.; Yu, S.; Jin, X.D.; Liu, J.Z. Hydrocarbon generation from coal, extracted coal and bitumen rich coal in confined pyrolysis experiments. Org. Geochem. 2013, 64, 58–75. [Google Scholar] [CrossRef]
- Jüntgen, H.; Van Heek, K.H. An update of German non-isothermal coal pyrolysis work. Fuel Process. Technol. 1979, 2, 261–293. [Google Scholar] [CrossRef]
- Jüntgen, H. Review of the kinetics of pyrolysis and hydropyrolysis in relation to the chemical constitution of coal. Fuel 1984, 63, 731–737. [Google Scholar] [CrossRef]
- Wagner, R.; Wanzl, W.; van Heek, K.H. Influence of transport effects on pyrolysis reaction of coal at high heating rates. Fuel 1985, 64, 571–573. [Google Scholar] [CrossRef]
- Inan, S.; Yalçin, M.N.; Mann, U. Expulsion of oil from petroleum source rocks: Inferences from pyrolysis of samples of unconventional grain size. Org. Geochem. 1998, 29, 45–61. [Google Scholar] [CrossRef]
- Inan, S. Gaseous hydrocarbons generated during pyrolysis of petroleum source rocks using unconventional grain-size: Implications for natural gas composition. Org. Geochem. 2000, 31, 1409–1418. [Google Scholar] [CrossRef]
- Lewan, M.D. Laboratory simulation of petroleum formation: Hydrous pyrolysis. In Organic Geochemistry-Principles and Applications; Engel, M., Macko, S., Eds.; Plenum Press: New York, NY, USA, 1993; pp. 419–442. [Google Scholar]
- Shao, D.Y.; Ellis, G.S.; Li, Y.F.; Zhang, T.W. Experimental investigation of the role of rock fabric in gas generation and expulsion during thermal maturation: Anhydrous closed-system pyrolysis of a bitumen-rich Eagle Ford Shale. Org. Geochem. 2018, 119, 22–35. [Google Scholar] [CrossRef]
- Liu, Y.K.; Xiong, Y.Q.; Li, Y.; Peng, P.A. Effects of oil expulsion and pressure on nanopore development in highly mature shale: Evidence from a pyrolysis study of the Eocene Maoming oil shale, south China. Mar. Pet. Geol. 2017, 86, 526–536. [Google Scholar] [CrossRef]
- Li, K.; Lu, H.; Zhao, Z.F.; Peng, P.A.; Hsu, C.S. Hydroconversion for Hydrocarbon Generation of Highly Overmature Kerogens under Fischer–Tropsch Synthesis Conditions. Energy Fuels 2021, 35, 7808–7818. [Google Scholar] [CrossRef]
- Ma, X.X.; Zheng, G.D.; Sajjad, W.; Wang, X.; Fan, Q.H.; Zheng, J.J.; Xia, Y.Q. Influence of minerals and iron on natural gases generation during pyrolysis of type-III kerogen. Mar. Pet. Geol. 2018, 89, 216–224. [Google Scholar] [CrossRef]
- Kissin, Y.V. Catagenesis and composition of petroleum: Origin of n-alkanes and isoalkanes in petroleum crude. Geochim. Cosmochim. Acta 1987, 51, 2445–2457. [Google Scholar] [CrossRef]
- Liao, L.L.; Wang, Y.; Chen, C.S.; Shi, S.Y.; Deng, R. Kinetic study of marine and lacustrine shale grains using Rock-Eval pyrolysis: Implications to hydrocarbon generation, retention and expulsion. Mar. Pet. Geol. 2018, 89, 164–173. [Google Scholar] [CrossRef]
- Gao, Y.P.; Long, Q.L.; Su, J.S.; He, J.Y.; Guo, P. Approaches to improving the porosity and permeability of Maoming oil shale, South China. Oil Shale 2016, 33, 216–227. [Google Scholar] [CrossRef]
- Tannenbaum, E.; Kaplan, I.R. Low-Mr hydrocarbons generated during hydrous and dry pyrolysis of kerogen. Nature 1985, 317, 708–709. [Google Scholar] [CrossRef]
- Tannenbaum, E.; Kaplan, I.R. Role of minerals in the thermal alteration of organic matter—I: Generation of gases and condensates under dry condition. Geochim. Cosmochim. Acta 1985, 49, 2589–2604. [Google Scholar] [CrossRef]
- Pan, C.C.; Geng, A.S.; Zhong, N.N.; Liu, J.Z.; Yu, L.P. Kerogen pyrolysis in the presence and absence of water and minerals. 1. Gas components. Energy Fuels 2008, 22, 416–427. [Google Scholar] [CrossRef]
- Rahman, H.M.; Kennedy, M.; Löhr, S.; Dewhurst, D.N. Clay-organic association as a control on hydrocarbon generation in shale. Org. Geochem. 2017, 105, 42–55. [Google Scholar] [CrossRef]
- Javadpour, F.; Fisher, D.; Unsworth, M. Nanoscale gas flow in shale gas sediments. J. Can. Pet. Technol. 2007, 46, PETSOC-07-10-06. [Google Scholar] [CrossRef]
- Javadpour, F. Nanopores and Apparent Permeability of Gas Flow in Mudrocks (Shales and Siltstone). J. Can. Pet. Technol. 2009, 48, 16–21. [Google Scholar] [CrossRef]
- Ross, D.; Bustin, R.M. The importance of shale composition and pore structure upon gas storage potential of shale gas reservoirs. Mar. Pet. Geol. 2009, 26, 916–927. [Google Scholar] [CrossRef]
- Zhang, T.; Ellis, G.S.; Ruppel, S.C. Effect of organic-matter type and thermal maturity on methane adsorption in shale-gas systems. Org. Geochem. 2012, 47, 120–131. [Google Scholar] [CrossRef]
- Zargari, S.; Canter, K.L.; Prasad, M. Porosity evolution in oil-prone source rocks. Fuel 2015, 153, 110–117. [Google Scholar] [CrossRef]
- Guo, H.; Jia, W.; He, R. Distinct evolution trends of nanometer-scale pores displayed by the pyrolysis of organic matter-rich lacustrine shales: Implications for the pore development mechanisms. Mar. Pet. Geol. 2020, 121, 104622. [Google Scholar] [CrossRef]
- Behar, F.; Kressmann, S.; Rudkiewicz, J.L.; Vandenbrcoucke, M. Experimental simulation in a confined system and kinetic modelling of kerogen and oil cracking. Org. Geochem. 1992, 19, 173–189. [Google Scholar] [CrossRef]
- Loucks, R.G.; Reed, R.M.; Ruppel, S.C.; Jarvie, D.M. Morphology, genesis, and distribution of nanometer-scale pores in siliceous mudstones of the Mississippian Barnett Shale. J. Sediment. Res. 2009, 79, 848–861. [Google Scholar] [CrossRef]
- Slatt, R.M.; O’Brien, N.R. Pore types in the Barnett and Woodford gas shales: Contribution to understanding gas storage and migration pathways in fine-grained rocks. AAPG Bull. 2011, 95, 2017–2030. [Google Scholar] [CrossRef]
- Krooss, B.M. Experimental investigation of the molecular migration of C1–C6 hydrocarbons: Kinetics of hydrocarbon release from source rocks. Org. Geochem. 1988, 13, 513–523. [Google Scholar] [CrossRef]
- Clayton, C. Carbon isotope fractionation during natural gas generation from kerogen. Mar. Pet. Geol. 1991, 8, 232–240. [Google Scholar] [CrossRef]
- Price, L.C. Thermal stability of hydrocarbons in nature: Limits, evidence, characteristics, and possible controls. Geochim. Cosmochim. Acta 1993, 57, 3261–3280. [Google Scholar] [CrossRef]
- Whiticar, M.J. Stable isotope geochemistry of coals, humic kerogens and related natural gases. Int. J. Coal Geol. 1996, 32, 191–215. [Google Scholar] [CrossRef]
- Rice, D.D.; Threlkeld, C.N.; Vuletich, A.K. Character, origin and occurrence of natural gases in the Anadarko basin, southwestern Kansas, western Oklahoma and Texas Panhandle, USA. Chem. Geol. 1988, 71, 149–157. [Google Scholar] [CrossRef]
- Palacas, J.G.; Anders, D.E.; King, J.D. South Florida basin—A prime example of carbonate source rocks of petroleum. AAPG Bull. 1984, 18, 71–96. [Google Scholar]
- Horsfield, B. Pyrolysis studies and petroleum exploration. Adv. Pet. Geochem. 1984, 1, 247–298. [Google Scholar]
- Tiem, V.T.A.; Horsfield, B.; Sykes, R. Influence of in-situ bitumen on the generation of gas and oil in New Zealand coals. Org. Geochem. 2008, 39, 1606–1619. [Google Scholar]
- Mansuy, L.; Landais, P.; Ruau, O. Importance of the reacting medium in artificial maturation of a coal by confined pyrolysis. 1. Hydrocarbons and polar compounds. Energy Fuels 1995, 9, 691–703. [Google Scholar] [CrossRef]
- Li, E.T.; Pan, C.C.; Yu, S.; Jin, X.D.; Liu, J.Z. Interaction of coal and oil in confined pyrolysis experiments: Insight from the yields and carbon isotopes of gas and liquid hydrocarbons. Mar. Pet. Geol. 2016, 69, 13–37. [Google Scholar] [CrossRef]
- Price, L.C.; Schoell, M. Constraints on the origins of hydrocarbon gas from compositions of gases at their site of origin. Nature 1995, 378, 368–371. [Google Scholar] [CrossRef] [PubMed]

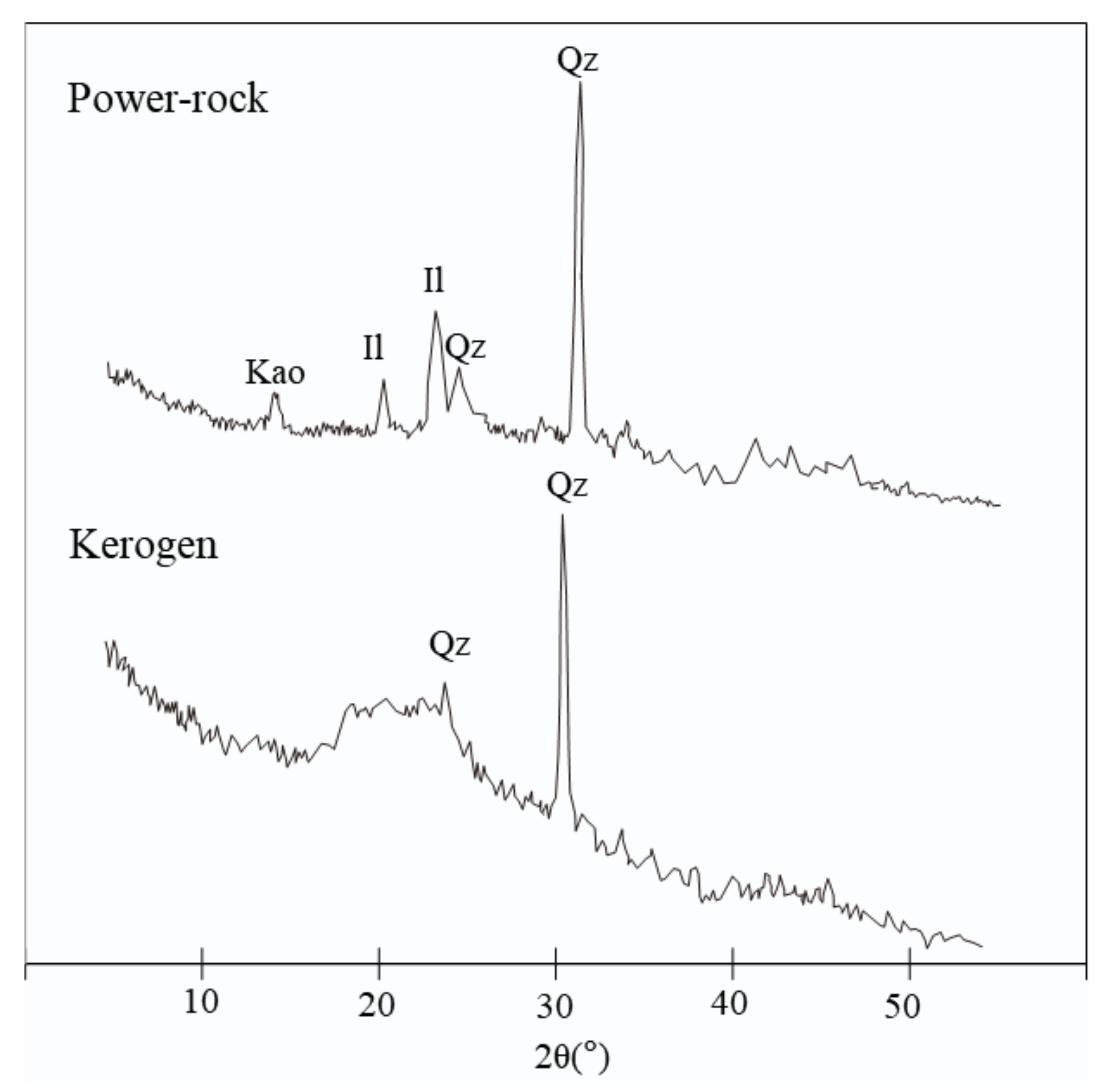


| TOC (wt%) | HI (mg/g TOC) | Tmax (°C) | S1 (mg/g Sample) | S2 (mg/g Sample) | S3 (mg/g Sample) | Quartz (%) | Illite (%) | Kaolite (%) | |
|---|---|---|---|---|---|---|---|---|---|
| Kerogen | 68 | 667 | 435 | 3.56 | 453.87 | 12.95 | 100 | 0 | 0 |
| Powdered rock | 22.89 | 605 | 428 | 1.53 | 138.41 | 4.3 | 19.6 | 65.2 | 15.2 |
| Core plug | 23.02 | 600 | 427 | 1.61 | 138.20 | 4.1 | 21.2 | 64 | 14.8 |
| Series | C1 | C2H6 | C2H4 | C3H8 | C3H6 | iC4 | nC4 | iC5 | nC5 | H2 | CO2 | H2S | ∑C1-5 | ∑C2-5 | ∑C6-14 | C14+ | C1/∑C1-5 | iC4/ nC4 | iC5/nC5 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| MK-312 | 1.68 | 0.61 | 0.00 | 0.52 | 0.01 | 0.14 | 0.15 | 0.04 | 0.06 | 0.05 | 61.88 | 0.85 | 3.22 | 1.54 | 3.72 | 94.13 | 0.52 | 0.95 | 0.76 |
| MK-336 | 3.34 | 1.73 | 0.00 | 1.49 | 0.02 | 0.30 | 0.50 | 0.13 | 0.19 | 0.05 | 74.26 | 1.43 | 7.71 | 4.37 | 6.89 | 194.43 | 0.43 | 0.61 | 0.69 |
| MK-360 | 6.89 | 5.21 | 0.01 | 4.32 | 0.03 | 0.68 | 1.67 | 0.36 | 0.66 | 0.04 | 92.38 | 2.91 | 19.82 | 12.93 | 15.30 | 361.27 | 0.35 | 0.41 | 0.55 |
| MK-384 | 14.38 | 12.90 | 0.01 | 10.71 | 0.06 | 1.56 | 4.68 | 0.92 | 1.93 | 0.09 | 113.48 | 5.95 | 47.15 | 32.77 | 33.57 | 310.18 | 0.31 | 0.33 | 0.48 |
| MK-408 | 25.05 | 22.10 | 0.01 | 19.48 | 0.13 | 2.84 | 9.13 | 1.79 | 3.84 | 0.16 | 115.31 | 7.04 | 84.38 | 59.33 | 48.73 | 160.75 | 0.30 | 0.31 | 0.47 |
| MK-432 | 42.02 | 38.64 | 0.03 | 38.95 | 0.41 | 5.64 | 20.70 | 3.74 | 9.24 | 0.24 | 119.23 | 7.09 | 159.38 | 117.36 | 77.16 | 104.32 | 0.26 | 0.27 | 0.40 |
| MK-456 | 75.26 | 78.91 | 0.09 | 86.67 | 0.81 | 12.64 | 44.80 | 5.96 | 14.88 | 0.35 | 128.25 | 9.08 | 320.00 | 244.74 | 37.42 | 31.94 | 0.24 | 0.28 | 0.40 |
| MK-480 | 122.36 | 112.45 | 0.13 | 102.08 | 0.69 | 16.74 | 22.02 | 1.52 | 1.48 | 0.53 | 130.31 | 8.48 | 379.48 | 257.12 | 26.89 | 33.02 | 0.32 | 0.76 | 1.03 |
| MK-504 | 180.59 | 124.46 | 0.19 | 66.29 | 0.50 | 4.61 | 5.95 | 0.40 | 0.39 | 0.77 | 136.92 | 8.15 | 383.39 | 202.80 | 20.93 | 24.60 | 0.47 | 0.77 | \ |
| MK-528 | 235.98 | 118.52 | 0.14 | 17.21 | 0.19 | 0.72 | 0.55 | 0.05 | 0.08 | 1.00 | 142.03 | 7.84 | 373.45 | 137.47 | 10.72 | 23.42 | 0.63 | \ | \ |
| MK-552 | 295.25 | 71.54 | 0.06 | 0.99 | 0.03 | 0.02 | 0.09 | 0.00 | 0.01 | 1.36 | 142.44 | 7.33 | 368.00 | 72.75 | 9.74 | 0.00 | 0.80 | \ | \ |
| MK-576 | 332.18 | 31.50 | 0.04 | 0.33 | 0.01 | 0.00 | 0.02 | 0.00 | 0.00 | 1.54 | 147.44 | 6.66 | 364.09 | 31.91 | 6.79 | 0.00 | 0.91 | \ | \ |
| MK-600 | 366.51 | 6.55 | 0.02 | 0.42 | 0.01 | 0.07 | 0.06 | 0.00 | 0.00 | 1.62 | 146.78 | 7.22 | 373.64 | 7.13 | 2.20 | 0.00 | 0.98 | \ | \ |
| MP-312 | 1.70 | 0.58 | 0.01 | 0.54 | 0.02 | 0.15 | 0.14 | 0.04 | 0.05 | 0.00 | 79.90 | 0.00 | 3.22 | 1.52 | 24.44 | 125.63 | 0.53 | 0.04 | 0.93 |
| MP-336 | 3.37 | 1.44 | 0.01 | 1.30 | 0.03 | 0.31 | 0.40 | 0.12 | 0.14 | 0.00 | 86.07 | 0.00 | 7.13 | 3.76 | 33.46 | 166.20 | 0.47 | 0.78 | 0.83 |
| MP-360 | 7.06 | 4.06 | 0.01 | 3.52 | 0.06 | 0.73 | 1.26 | 0.30 | 0.45 | 0.02 | 132.34 | 0.00 | 19.82 | 12.93 | 42.09 | 195.10 | 0.36 | 0.58 | 0.68 |
| MP-384 | 13.76 | 10.33 | 0.02 | 9.14 | 0.16 | 1.63 | 3.70 | 0.82 | 1.33 | 0.10 | 160.65 | 1.05 | 40.89 | 27.13 | 65.90 | 203.61 | 0.34 | 0.44 | 0.61 |
| MP-408 | 26.92 | 20.76 | 0.04 | 17.51 | 0.34 | 2.57 | 6.60 | 1.23 | 2.07 | 0.20 | 180.15 | 1.53 | 78.05 | 51.13 | 117.09 | 185.81 | 0.34 | 0.39 | 0.59 |
| MP-432 | 47.38 | 43.88 | 0.07 | 42.56 | 0.71 | 5.97 | 18.95 | 3.22 | 6.07 | 0.33 | 207.87 | 2.19 | 172.59 | 121.42 | 98.92 | 76.40 | 0.27 | 0.32 | 0.53 |
| MP-456 | 78.41 | 81.81 | 0.16 | 84.92 | 1.25 | 12.61 | 35.84 | 4.68 | 8.00 | 0.48 | 202.30 | 0.00 | 313.96 | 229.27 | 48.75 | 39.79 | 0.25 | 0.35 | 0.58 |
| MP-480 | 132.86 | 111.49 | 0.20 | 95.31 | 0.86 | 14.78 | 15.12 | 0.82 | 0.54 | 0.63 | 220.52 | 2.56 | 371.99 | 239.13 | 41.40 | 11.92 | 0.36 | 0.98 | 0.50 |
| MP-504 | 184.01 | 117.47 | 0.38 | 45.30 | 0.78 | 3.80 | 2.26 | 0.17 | 0.12 | 0.87 | 219.64 | 2.62 | 369.00 | 170.28 | 38.51 | 28.45 | 0.50 | 0.68 | \ |
| MP-528 | 241.82 | 106.33 | 0.12 | 6.31 | 0.08 | 0.16 | 0.14 | 0.01 | 0.01 | 1.39 | 248.65 | 2.60 | 374.31 | 113.15 | 24.30 | 6.63 | 0.65 | \ | \ |
| MP-552 | 291.73 | 54.31 | 0.05 | 0.46 | 0.01 | 0.01 | 0.02 | 0.00 | 0.00 | 1.80 | 265.14 | 2.54 | 369.93 | 54.87 | 18.53 | \ | 0.79 | \ | \ |
| MP-576 | 328.96 | 18.55 | 0.03 | 0.11 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 2.05 | 281.53 | 2.14 | 373.97 | 18.70 | 10.37 | \ | 0.88 | \ | \ |
| MP-600 | 353.50 | 2.19 | 0.01 | 0.01 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 2.79 | 287.56 | 0.00 | 384.00 | 2.21 | 6.55 | \ | 0.92 | \ | \ |
| MC-312 | 1.15 | 0.32 | 0.00 | 0.25 | 0.01 | 0.06 | 0.05 | 0.02 | 0.02 | 0.01 | 60.18 | 0.00 | 1.88 | 0.73 | 18.83 | 118.89 | 0.61 | 1.13 | 0.84 |
| MC-336 | 2.82 | 1.07 | 0.01 | 0.81 | 0.02 | 0.17 | 0.21 | 0.05 | 0.07 | 0.00 | 79.39 | 0.00 | 5.22 | 2.41 | 27.52 | 150.11 | 0.54 | 0.83 | 0.76 |
| MC-360 | 6.67 | 3.74 | 0.01 | 3.02 | 0.06 | 0.57 | 0.98 | 0.22 | 0.35 | 0.02 | 123.01 | 0.00 | 15.62 | 8.95 | 39.34 | 182.33 | 0.43 | 0.58 | 0.63 |
| MC-384 | 13.18 | 9.53 | 0.02 | 7.82 | 0.18 | 1.28 | 2.87 | 0.60 | 1.05 | 0.10 | 149.32 | 1.55 | 36.52 | 23.35 | 65.01 | 198.94 | 0.36 | 0.45 | 0.57 |
| MC-408 | 22.62 | 17.46 | 0.05 | 14.59 | 0.42 | 2.13 | 5.57 | 1.12 | 1.95 | 0.21 | 163.03 | 2.46 | 65.92 | 43.29 | 108.26 | 171.19 | 0.34 | 0.38 | 0.58 |
| MC-432 | 44.75 | 38.81 | 0.12 | 36.40 | 1.23 | 4.70 | 16.07 | 2.56 | 5.50 | 0.29 | 178.66 | 4.41 | 150.14 | 105.39 | 88.75 | 62.67 | 0.30 | 0.29 | 0.46 |
| MC-456 | 82.72 | 78.08 | 0.23 | 73.03 | 1.81 | 10.38 | 29.89 | 3.87 | 6.67 | 0.42 | 192.60 | 3.15 | 286.68 | 203.95 | 44.03 | 36.04 | 0.29 | 0.35 | 0.58 |
| MC-480 | 138.75 | 107.74 | 0.27 | 74.79 | 1.28 | 11.56 | 9.94 | 0.77 | 0.59 | 0.56 | 195.42 | 2.84 | 345.71 | 206.95 | 39.36 | 19.75 | 0.40 | 1.16 | 1.31 |
| MC-504 | 190.01 | 102.36 | 0.33 | 34.60 | 0.79 | 2.59 | 2.10 | 0.15 | 0.14 | 0.65 | 200.32 | 1.86 | 333.07 | 143.06 | 31.77 | 25.24 | 0.57 | \ | \ |
| MC-528 | 234.26 | 74.10 | 0.19 | 5.33 | 0.20 | 0.20 | 0.20 | 0.00 | 0.00 | 0.91 | 210.58 | 3.12 | 314.48 | 80.22 | 22.89 | 7.40 | 0.74 | \ | \ |
| MC-552 | 291.39 | 45.28 | 0.08 | 0.47 | 0.03 | 0.01 | 0.02 | 0.00 | 0.00 | 1.30 | 231.89 | 1.80 | 337.27 | 45.88 | 20.61 | 0.00 | 0.86 | \ | \ |
| MC-576 | 339.23 | 21.09 | 0.04 | 0.14 | 0.01 | 0.00 | 0.00 | 0.00 | 0.00 | 1.98 | 249.37 | 2.98 | 360.51 | 21.28 | 12.68 | 0.00 | 0.94 | \ | \ |
| MC-600 | 340.69 | 6.40 | 0.02 | 0.03 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 2.35 | 275.25 | 1.27 | 347.14 | 6.45 | 6.39 | 0.00 | 0.98 | \ | \ |
| Sample Form | C14+ Retained | Retention Proportion | Sample Form | C14+ Retained | Retention Proportion |
|---|---|---|---|---|---|
| MP-312 | / | / | MC-312 | / | / |
| MP-336 | 28.23 | 14.52 | MC-336 | 44.32 | 22.79 |
| MP-360 | 166.17 | 46.00 | MC-360 | 178.94 | 49.53 |
| MP-384 | 106.57 | 34.36 | MC-384 | 111.24 | 35.86 |
| MP-408 | / | / | MC-408 | / | / |
| MP-432 | 27.92 | 26.76 | MC-432 | 41.65 | 39.93 |
| MP-456 | / | / | MC-456 | / | / |
| MP-480 | 21.10 | 63.90 | MC-480 | 13.27 | 40.19 |
| MP-504 | / | / | MC-504 | / | / |
| MP-528 | 16.79 | 71.69 | MC-528 | 16.02 | 68.40 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, K.; Wang, Q.; Ma, H.; Huang, H.; Lu, H.; Peng, P. Effect of Clay Minerals and Rock Fabric on Hydrocarbon Generation and Retention by Thermal Pyrolysis of Maoming Oil Shale. Processes 2023, 11, 894. https://doi.org/10.3390/pr11030894
Li K, Wang Q, Ma H, Huang H, Lu H, Peng P. Effect of Clay Minerals and Rock Fabric on Hydrocarbon Generation and Retention by Thermal Pyrolysis of Maoming Oil Shale. Processes. 2023; 11(3):894. https://doi.org/10.3390/pr11030894
Chicago/Turabian StyleLi, Kang, Qiang Wang, Hongliang Ma, Huamei Huang, Hong Lu, and Ping’an Peng. 2023. "Effect of Clay Minerals and Rock Fabric on Hydrocarbon Generation and Retention by Thermal Pyrolysis of Maoming Oil Shale" Processes 11, no. 3: 894. https://doi.org/10.3390/pr11030894
APA StyleLi, K., Wang, Q., Ma, H., Huang, H., Lu, H., & Peng, P. (2023). Effect of Clay Minerals and Rock Fabric on Hydrocarbon Generation and Retention by Thermal Pyrolysis of Maoming Oil Shale. Processes, 11(3), 894. https://doi.org/10.3390/pr11030894





