Thermal Treatment and High-Intensity Ultrasound Processing to Evaluate the Chemical Profile and Antioxidant Activity of Amazon Fig Juices
Abstract
1. Introduction
2. Materials and Methods
2.1. Fruits Collecting and Juices Preparation
2.2. Heat Treatment and Ultrasound Processing
2.3. Soluble Solids, pH, and Titratable Acidity
2.4. Color Parameters
2.5. Antioxidant Capacity (DPPH and ABTS·+)
2.6. Total Phenolic Content (TPC)
2.7. Carotenoids Content
2.8. Chemical Profile
2.9. Principal Components Analysis (PCA) and Partial Least Squares-Discriminant Analysis (PLS-DA)
2.10. Statistical Analysis
3. Results and Discussion
3.1. Physicochemical Characterization
3.2. Antioxidant Capacity, Phenolic and Carotenoids Contents
3.3. 1H NMR and HPLC-DAD Analysis
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kinupp, V.F.; Lorenzi, H. Plantas Alimentícias Não Convencionais (PANC) No Brasil: Guia de Identificação, Aspectos Nutricionais e Receitas Ilustradas; Instituto Plantarum De Estudos Da Flora: São Paulo, Brazil, 2014. [Google Scholar]
- Mar, J.M.; da Silva, L.S.; Lira, A.C.; Kinupp, V.F.; Yoshida, M.I.; Moreira, W.P.; Bruginski, E.; Campos, F.R.; Machado, M.B.; de Souza, T.P.; et al. Bioactive Compounds-Rich Powders: Influence of Different Carriers and Drying Techniques on the Chemical Stability of the Hibiscus Acetosella Extract. Powder Technol. 2020, 360, 383–391. [Google Scholar] [CrossRef]
- Mar, J.M.; Silva, L.S.; Rabelo, M.D.; Muniz, M.P.; Nunomura, S.M.; Correa, R.F.; Kinupp, V.F.; Campelo, P.H.; Bezerra, J.D.; Sanches, E.A. Encapsulation of Amazonian Blueberry Juices: Evaluation of Bioactive Compounds and Stability. Lwt 2020, 124, 109152. [Google Scholar] [CrossRef]
- Cruz, J.M.d.A.; Corrêa, R.F.; Lamarão, C.V.; Kinupp, V.F.; Sanches, E.A.; Campelo, P.H.; Bezerra, J.d.A. Ficus spp. Fruits: Bioactive Compounds and Chemical, Biological and Pharmacological Properties. Food Res. Int. 2022, 152, 110928. [Google Scholar] [CrossRef] [PubMed]
- Chemat, F.; Rombaut, N.; Sicaire, A.-G.; Meullemiestre, A.; Fabiano-Tixier, A.-S.; Abert-Vian, M. Ultrasound Assisted Extraction of Food and Natural Products. Mechanisms, Techniques, Combinations, Protocols and Applications. A Review. Ultrason. Sonochem. 2017, 34, 540–560. [Google Scholar] [CrossRef] [PubMed]
- Rojas, M.L.; Kubo, M.T.; Miano, A.C.; Augusto, P.E. Ultrasound Processing to Enhance the Functionality of Plant-Based Beverages and Proteins. Curr. Opin. Food Sci. 2022, 48, 100939. [Google Scholar] [CrossRef]
- Illera, A.E.; Sanz, M.T.; Benito-Román, O.; Varona, S.; Beltrán, S.; Melgosa, R.; Solaesa, A.G. Effect of Thermosonication Batch Treatment on Enzyme Inactivation Kinetics and Other Quality Parameters of Cloudy Apple Juice. Innov. Food Sci. Emerg. Technol. 2018, 47, 71–80. [Google Scholar] [CrossRef]
- Dias, D.d.R.C.; Barros, Z.M.P.; de Carvalho, C.B.O.; Honorato, F.A.; Guerra, N.B.; Azoubel, P.M. Effect of Sonication on Soursop Juice Quality. Lwt 2015, 62, 883–889. [Google Scholar] [CrossRef]
- Abid, M.; Jabbar, S.; Wu, T.; Hashim, M.M.; Hu, B.; Lei, S.; Zhang, X.; Zeng, X. Effect of Ultrasound on Different Quality Parameters of Apple Juice. Ultrason. Sonochem. 2013, 20, 1182–1187. [Google Scholar] [CrossRef]
- de Souza Carvalho, L.M.; Lemos, M.C.M.; Sanches, E.A.; da Silva, L.S.; de Araújo Bezerra, J.; Aguiar, J.P.L.; das Chagas do Amaral Souza, F.; Alves Filho, E.G.; Campelo, P.H. Improvement of the Bioaccessibility of Bioactive Compounds from Amazon Fruits Treated Using High Energy Ultrasound. Ultrason. Sonochem. 2020, 67, 105148. [Google Scholar] [CrossRef]
- Instituto Adolfo Lutz. 1a Edição Digital, 4th ed.; Instituto Adolfo Lutz: São Paulo, Brazil, 2008.
- de Castro, D.R.G.; Mar, J.M.; da Silva, L.S.; da Silva, K.A.; Sanches, E.A.; de Araújo Bezerra, J.; Rodrigues, S.; Fernandes, F.A.N.; Campelo, P.H. Dielectric Barrier Atmospheric Cold Plasma Applied on Camu-Camu Juice Processing: Effect of the Excitation Frequency. Food Res. Int. 2020, 131, 109044. [Google Scholar] [CrossRef]
- Pereira, G.A.; Silva, E.K.; Peixoto Araujo, N.M.; Arruda, H.S.; Meireles, M.A.A.; Pastore, G.M. Obtaining a Novel Mucilage from Mutamba Seeds Exploring Different High-Intensity Ultrasound Process Conditions. Ultrason. Sonochem. 2019, 55, 332–340. [Google Scholar] [CrossRef]
- Molyneux, P. The Use of the Stable Free Radical Diphenylpicryl-Hydrazyl (DPPH) for Estimating Anti-Oxidant Activity. Songklanakarin J. Sci. Technol. 2004, 26, 211–219. [Google Scholar]
- Re, R.; Pellegrini, N.; Proteggente, A.; Ananth, P.; Yng, M.; Rice-Evans, C. Antioxidant Activity Applying an Improved ABTS Radical Cation Decolorization Assay. Free Radic. Biol. Med. 1999, 26, 1231–1237. [Google Scholar] [CrossRef] [PubMed]
- Velioglu, Y.S.; Mazza, G.; Gao, L.; Oomah, B.D. Antioxidant Activity and Total Phenolics in Selected Fruits, Vegetables, and Grain Products. J. Agric. Food Chem. 1998, 46, 4113–4117. [Google Scholar] [CrossRef]
- Fernandes, F.A.N.; Santos, V.O.; Rodrigues, S. Effects of Glow Plasma Technology on Some Bioactive Compounds of Acerola Juice. Food Res. Int. 2019, 115, 16–22. [Google Scholar] [CrossRef]
- Ramos, A.S.; Mar, J.M.; Laiane, S.; Acho, L.D.R.; Janaína, B.; Silva, P.; Lima, E.S.; Campelo, P.H.; Sanches, E.A.; Bezerra, J.A.; et al. Pedra-Ume Caá Fruit: An Amazon Cherry Rich in Phenolic Compounds with Antiglycant and Antioxidant Properties. Food Res. Int. 2019, 123, 674–683. [Google Scholar] [CrossRef]
- Ramos, A.S.; Souza, R.O.S.; Paula, A.; Boleti, D.A.; Bruginski, E.R.D.; Lima, E.S.; Campos, F.R.; Machado, M.B. Chemical Characterization and Antioxidant Capacity of the Araçá-Pera (Psidium acutangulum): An Exotic Amazon Fruit. Food Res. Int. J. 2015, 75, 315–327. [Google Scholar] [CrossRef]
- Choo, Y.X.; Teh, L.K.; Tan, C.X. Effects of Sonication and Thermal Pasteurization on the Nutritional, Antioxidant, and Microbial Properties of Noni Juice. Molecules 2022, 28, 313. [Google Scholar] [CrossRef]
- Ferraz, R.A.; Leonel, S.; Souza, J.M.A.; Modesto, J.H.; Ferreira, R.B.; de Souza Silva, M. Agronomical and Quality Differences of Four Fig Cultivars Grown in Brazil. Semin. Agrar. 2021, 42, 619–634. [Google Scholar] [CrossRef]
- Gençdağ, E.; Görgüç, A.; Okuroğlu, F.; Yılmaz, F.M. The Effects of Power—Ultrasound, Peroxyacetic Acid and Sodium Chloride Washing Treatments on the Physical and Chemical Quality Characteristics of Dried Figs. J. Food Process. Preserv. 2021, 45, e15009. [Google Scholar] [CrossRef]
- Rodríguez-Rico, D.; Sáenz-Esqueda, M.D.L.Á.; Meza-Velázquez, J.A.; Martínez-García, J.J.; Quezada-Rivera, J.J.; Umaña, M.M.; Minjares-Fuentes, R. High-Intensity Ultrasound Processing Enhances the Bioactive Compounds, Antioxidant Capacity and Microbiological Quality of Melon (Cucumis melo) Juice. Foods 2022, 11, 2648. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Wang, J.; Vanga, S.K.; Raghavan, V. High-Intensity Ultrasound Processing of Kiwifruit Juice: Effects on the Microstructure, Pectin, Carbohydrates and Rheological Properties. Food Chem. 2020, 313, 126121. [Google Scholar] [CrossRef] [PubMed]
- Peixoto Araujo, N.M.; Silva, E.K.; Arruda, H.S.; Rodrigues de Morais, D.; Angela, A.; Meireles, M.; Pereira, G.A.; Pastore, G.M. Recovering Phenolic Compounds from Eugenia Calycina Cambess Employing High-Intensity Ultrasound Treatments: A Comparison among Its Leaves, Fruit Pulp, and Seed as Promising Sources of Bioactive Compounds. Sep. Purif. Technol. 2021, 272, 118920. [Google Scholar] [CrossRef]
- Mohd Dom, N.S.; Yahaya, N.; Adam, Z.; Nik Mohd, N.M.A.; Hamid, M. Antiglycation and Antioxidant Properties of Ficus Deltoidea Varieties. Evid. Based Complement. Altern. Med. 2020, 2020, 6374632. [Google Scholar] [CrossRef] [PubMed]
- Tewari, D.; Zengin, G.; Ak, G.; Sinan, K.I.; Cziáky, Z.; Mishra, S.T.; Jekő, J. Phenolic Profiling, Antioxidants, Multivariate, and Enzyme Inhibitory Properties of Wild Himalayan Fig (Ficus palmata Forssk.): A Potential Candidate for Designing Innovative Nutraceuticals and Related Products. Anal. Lett. 2021, 54, 1439–1456. [Google Scholar] [CrossRef]
- Debib, A.; Dueñas, M.; Boumediene, M.; Mothana, R.A.; Latifa, A.; Tir-Touil, M.A. Synergetic Hepatoprotective Effect of Phenolic Fractions Obtained from Ficus Carica Dried Fruit and Extra Virgin Olive Oil on CCL 4-Induced Oxidative Stress and Hepatotoxicity in Rats. J. Food Biochem. 2016, 40, 507–516. [Google Scholar] [CrossRef]
- Palmeira, L.; Pereira, C.; Dias, M.I.; Abreu, R.M.V.; Corrêa, R.C.G.; Pires, T.C.S.P.; Alves, M.J.; Barros, L.; Ferreira, I.C.F.R. Nutritional, Chemical and Bioactive Profiles of Different Parts of a Portuguese Common Fig (Ficus carica L.) Variety. Food Res. Int. 2019, 126, 108572. [Google Scholar] [CrossRef]
- Gomes, A.; Costa, A.L.R.; Rodrigues, P.D.; de Castro, R.J.S.; Silva, E.K. Sonoprocessing of Freshly Squeezed Orange Juice: Ascorbic Acid Content, Pectin Methylesterase Activity, Rheological Properties and Cloud Stability. Food Control 2022, 131, 108391. [Google Scholar] [CrossRef]
- Wang, S.; Liu, Z.; Zhao, S.; Zhang, L.; Li, C.; Liu, S. Effect of Combined Ultrasonic and Enzymatic Extraction Technique on the Quality of Noni (Morinda citrifolia L.) Juice. Ultrason. Sonochem. 2023, 92, 106231. [Google Scholar] [CrossRef]
- Esbensen, K.H.; Swarbrick, B. Multivariate Data Analysis: An Introduction to Multivariate Analysis, Process Analytical Technology and Quality by Design; 6a; Camo Software AS: Oslo, Norway, 2018; ISBN 978-8269110401. [Google Scholar]
- Mehaya, F.M.; Mohammad, A.A. Thermostability of Bioactive Compounds during Roasting Process of Coffee Beans. Heliyon 2020, 6, e05508. [Google Scholar] [CrossRef]
- Carlos, A.L.E.; Priego-Capote, F.; De Castro, M.D.L. Comparative Study of the Effect of Sample Pretreatment and Extraction on the Determination of Flavonoids from Lemon (Citrus limon). PLoS ONE 2016, 11, e0148056. [Google Scholar] [CrossRef]
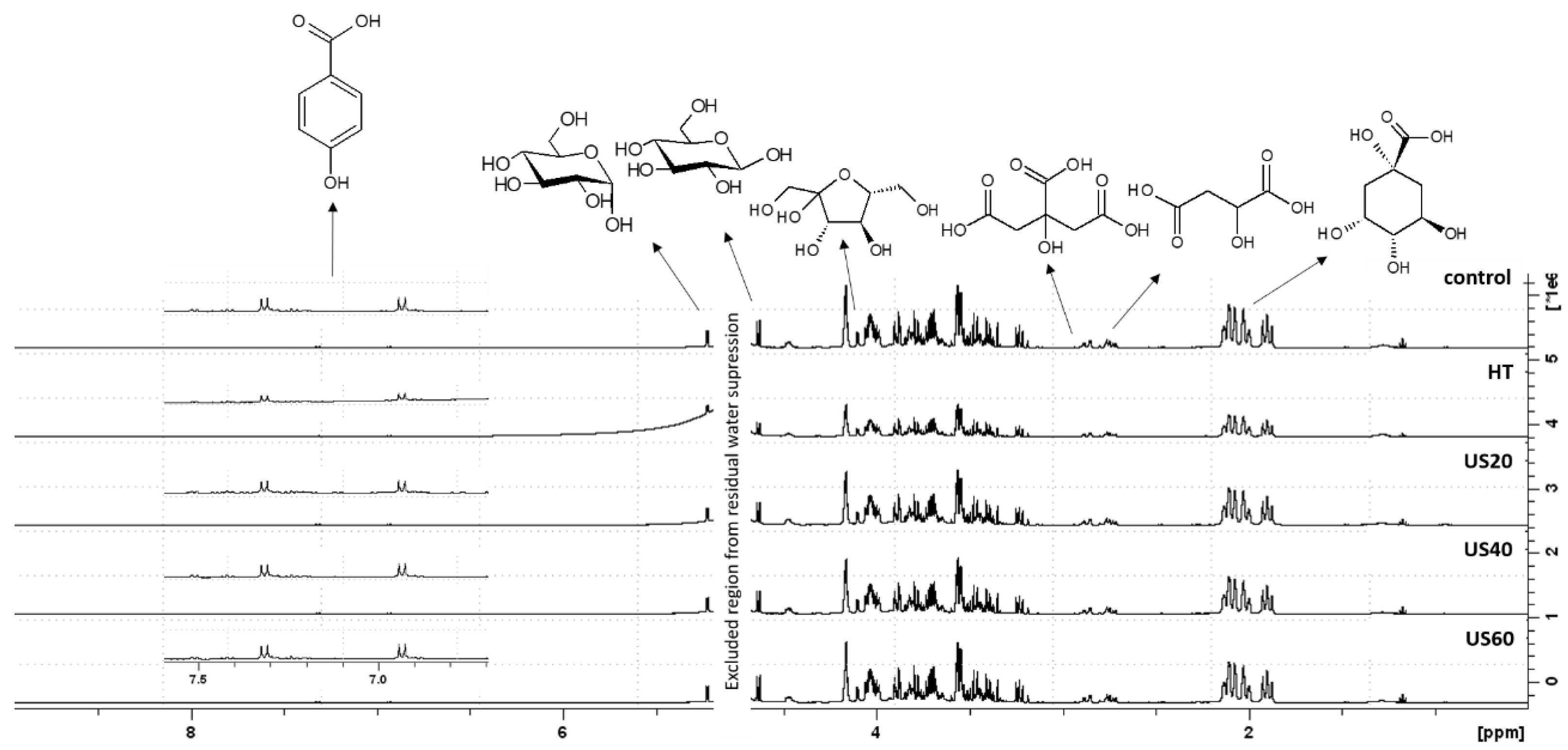

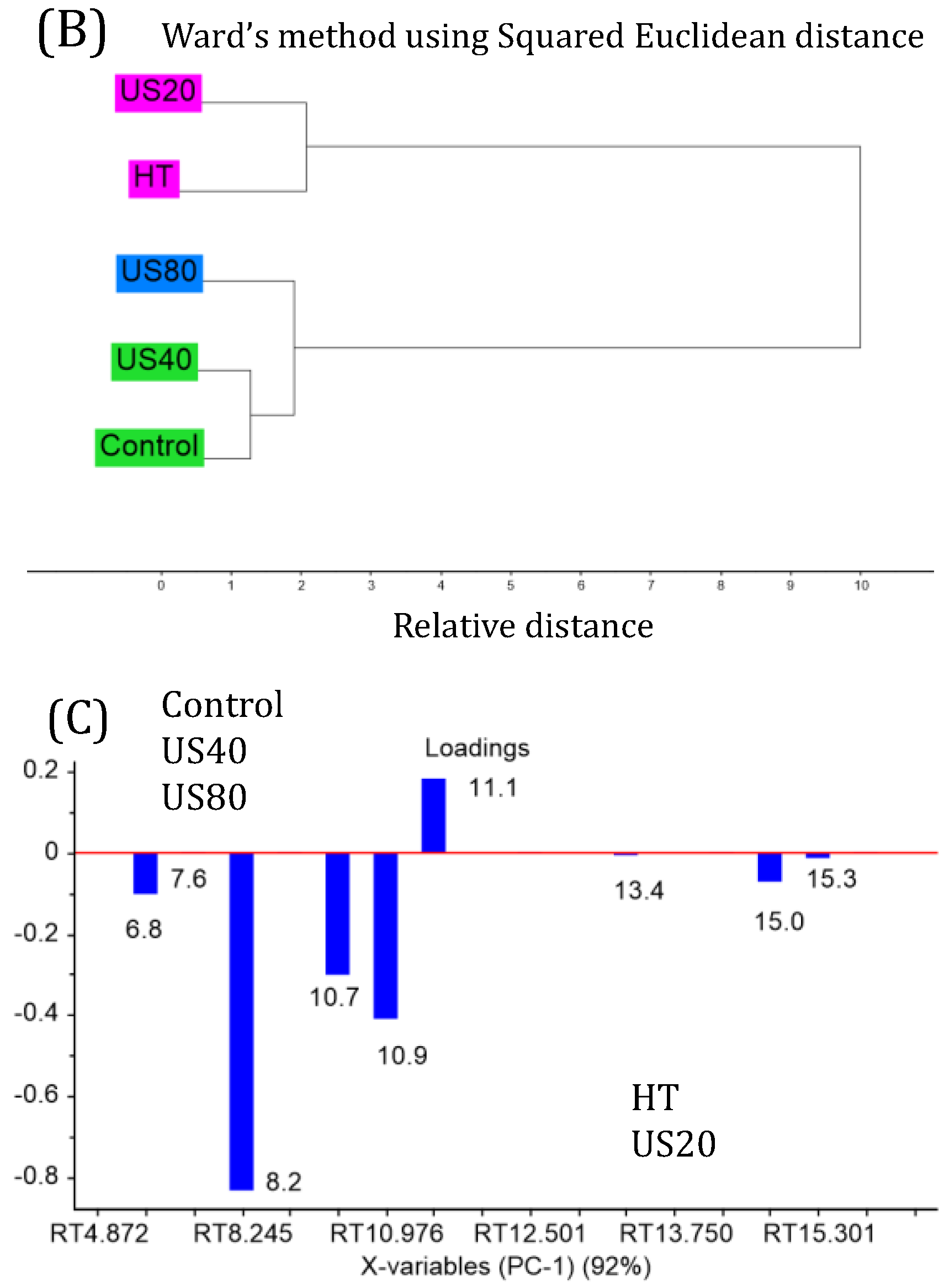
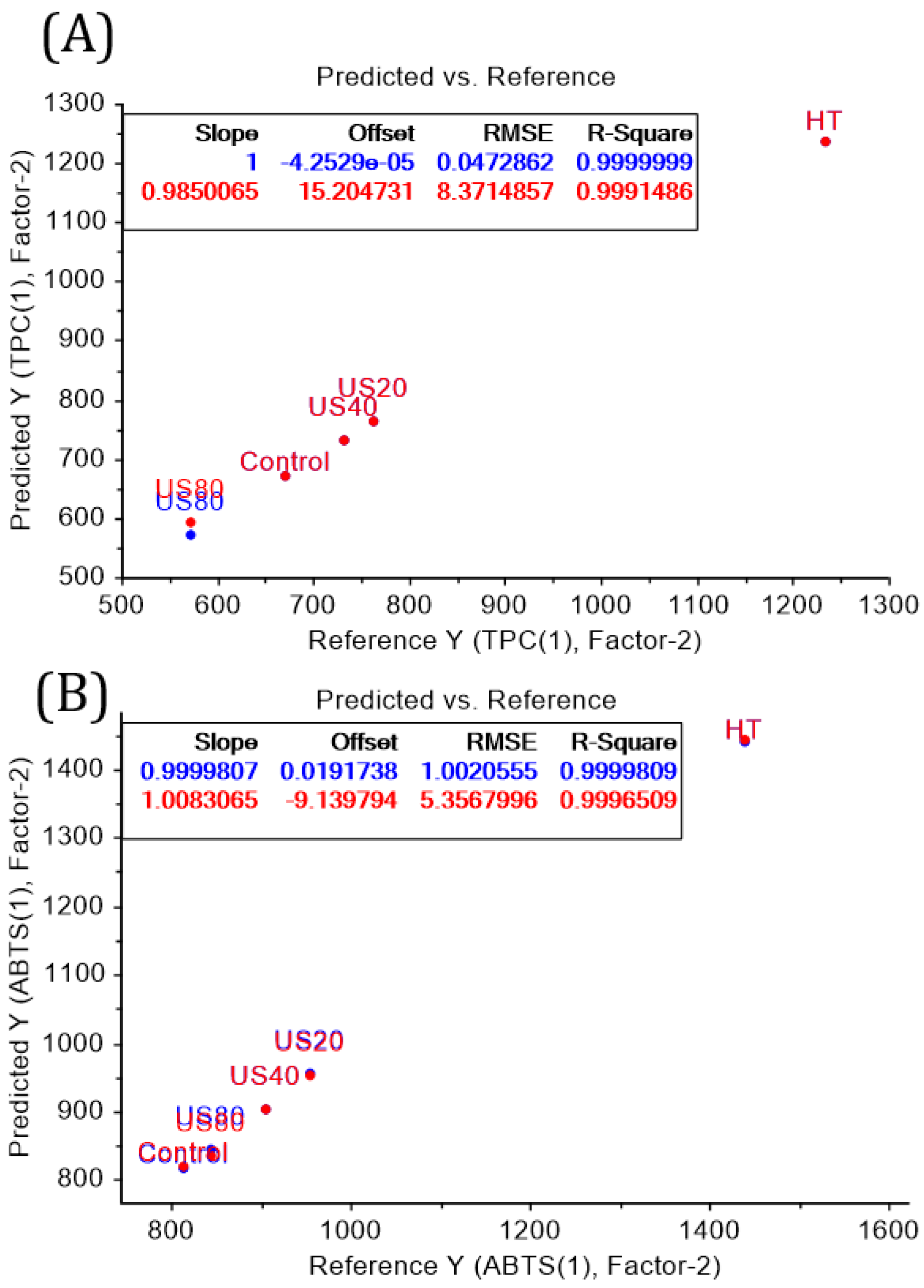
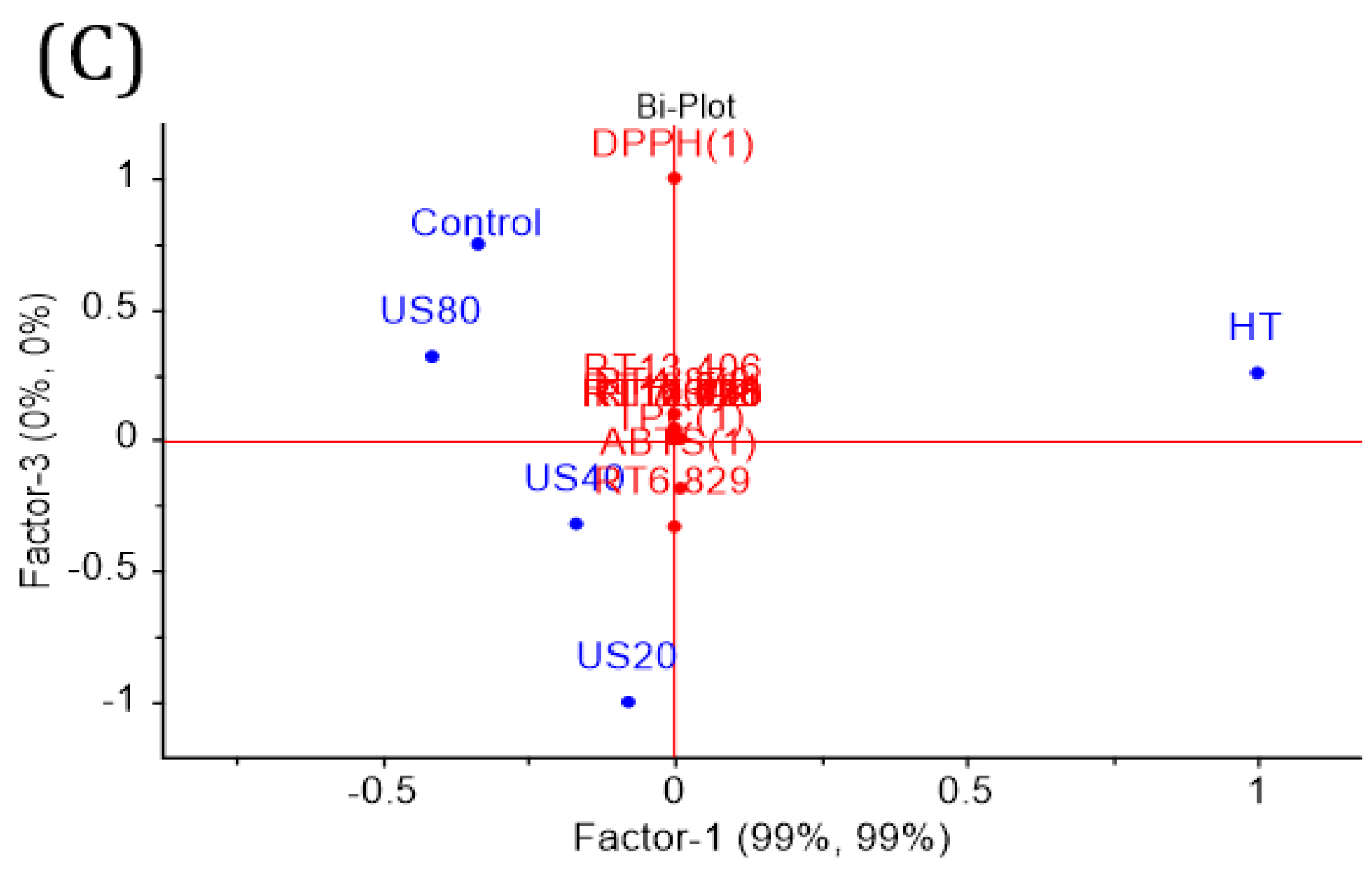
| Treatment | Power (W) | Time (min) | Initial Temperature (°C) | Final Temperature (°C) |
|---|---|---|---|---|
| HT | – | 10 | 21.27 ± 0.06 | 77.87 ± 0.21 |
| US20 | 150 | 16.6 | 25.33 ± 0.58 | 64.67 ± 0.58 |
| US40 | 300 | 8.3 | 25.67 ± 0.58 | 72.33 ± 0.58 |
| US80 | 600 | 4.1 | 25.67 ± 0.58 | 62.33 ± 0.58 |
| Treatment | Control | HT | US20 | US40 | US80 |
|---|---|---|---|---|---|
| pH | 3.46 ± 0.05 a | 3.50 ± 0.05 a | 3.44 ± 0.05 a | 3.47 ± 0.05 a | 3.47 ± 0.05 a |
| TA (%) | 0.044 ± 0.001 a | 0.044 ± 0.001 a | 0.046 ± 0.001 a | 0.048 ± 0.001 a | 0.046 ± 0.001 a |
| SS (°Brix) | 2.34 ± 0.05 a | 2.35 ± 0.05 a | 2.35 ± 0.05 a | 2.40 ± 0.05 a | 2.37 ± 0.04 a |
| L* | 11.9 ± 0.5 d | 14.2 ± 0.3 c | 15.6 ± 0.4 b | 16.8 ± 0.2 a | 16.4 ± 0.2 ab |
| a* | 10.7 ± 0.6 a | 9.4 ± 0.3 b | 10.0 ± 0.3 ab | 10.0 ± 0.3 ab | 10.1 ± 0.5 ab |
| b* | 8.3 ± 0.1 a | 8.8 ± 0.1 a | 9.4 ± 0.7 a | 8.6 ± 0.7 a | 9.0 ± 0.2 a |
| C* | 13.6 ± 0.4 a | 12.9 ± 0.2 a | 13.7 ± 0.6 a | 13.2 ± 0.4 a | 13.5 ± 0.2 a |
| h* | 38.0 ± 2.0 a | 43.2 ± 1.2 a | 43.1 ± 1.8 a | 40.7 ± 2.8 a | 41.7 ± 2.0 a |
| ΔE | – | 2.6 ± 0.1 b | 4.0 ± 0.4 a | 4.9 ± 0.6 a | 4.6 ± 0.4 a |
| Treatment | DPPH | ABTS | TPC | TCC |
|---|---|---|---|---|
| Control | 672 ± 6 d | 814 ± 6 d | 207 ± 1 c | ND |
| HT | 1235 ± 11 a | 1440 ± 13 a | 312 ± 5 a | ND |
| US20 | 762 ± 11 b | 956 ± 10 b | 215 ± 1 b | ND |
| US40 | 732 ± 8 c | 904 ± 4 c | 212 ± 1 bc | ND |
| US80 | 572 ± 7 e | 844 ± 10 d | 209 ± 2 bc | ND |
| RT (min) | Compound | λ (nm) | Control | HT | US20 | US40 | US80 | R2 | %RSD |
|---|---|---|---|---|---|---|---|---|---|
| mg/mL | |||||||||
| 6.82 | Gallic acid derivative | 271 | ND | ND | ND | ND | ND | ND | ND |
| 7.69 | Gallic acid | 271 | 0.10 ± 0.02 b | 0.26 ± 0.10 a | 0.10 ± 0.04 b | 0.06 ± 0.01 bc | 0.10 ± 0.01 b | 0.998 | 19.9 |
| 8.24 | Not Indentified | 323 | ND | ND/↑ | ND | ND | ND | ND | ND |
| 10.19 | Protocatechuic acid | 293 | 0.07 ± 0.00 c | 0.10 ± 0.07 a | 0.07 ± 0.01 c | 0.04 ± 0.00 d | 0.08 ± 0.00 b | 0.998 | 19.85 |
| 10.79 | Not Indentified | 323 | ND | ND | ND/↑ | ND | ND | ND | ND |
| 10.97 | Not Indentified | 319 | ND | ND/↑ | ND | ND | ND | ND | ND |
| 11.17 | Not Indentified | 324 | ND/↑ | ND | ND/↑ | ND | ND | ND | ND |
| 11.73 | Cyanidin 3-O-glucoside | 527 | 0.26 ± 0.01 c | 0.27 ± 0.00 b | 0.24 ± 0.01 d | 0.16 ± 0.00 e | 0.35 ± 0.01 a | 0.998 | 18.8 |
| 12.50 | p-hydroxybenzoic acid | 324 | <LOQ | 0.19 ± 0.04 a | 0.02 ± 0.01 c | 0.01 ± 0.00 c | 0.04 ± 0.01 b | 0.997 | 17.7 |
| 12.67 | Cyanidin | 519 | 0.03 ± 0.01 a | 0.02 ± 0.00 b | 0.02 ± 0.00 b | 0.01 ± 0.00 c | 0.02 ± 0.00 b | 0.996 | 16.9 |
| 13.14 | Delphinidin | 526 | <LOQ | <LOQ | <LOQ | <LOQ | <LOQ | 0.988 | 19.5 |
| 13.40 | Syringic acid | 267 | 1.44 ± 0.46 b | 2.22 ± 0.19 a | 0.89 ± 0.04 c | 0.58 ± 0.04 d | 1.98 ± 0.14 b | 0.988 | 18.4 |
| 13.75 | Caffeic acid | 324 | 0.08 ± 0.01 a | 0.06 ± 0.01 ab | 0.05 ± 0.01 ab | 0.03 ± 0.00 c | 0.06 ± 0.01 ab | 0.992 | 19.8 |
| 14.01 | Epicatechin | 290 | 0.18 ± 0.04 b | 0.23 ± 0.04 a | 0.16 ± 0.02 bc | 0.10 ± 0.01 d | 0.18 ± 0.03 b | 0.987 | 18.1 |
| 15.01 | Epicatechin derivative | 297 | <LOQ | <LOQ | <LOQ | <LOQ | <LOQ | ||
| 15.95 | Flavonol derivative | 344 | <LOQ | <LOQ | <LOQ | <LOQ | <LOQ | ||
| 15.30 | Catechin derivative | 297 | <LOQ | <LOQ | <LOQ | <LOQ | <LOQ | ||
| 16.43 | p-coumaric acid | 309 | 0.04 ± 0.02 b | 0.08 ± 0.04 a | 0.00 ± 0.00 d | 0.00 ± 0.00 d | 0.03 ± 0.02 c | 0.995 | 17.8 |
| 17.12 | trans-ferulic acid | 322 | 0.039 ± 0.02 a | 0.04 ± 0.02 a | 0.02 ± 0.00 bc | 0.01 ± 0.00 d | 0.03 ± 0.01 ab | 0.998 | 16.9 |
| 17.71 | Vitexin | 338 | 0.10 ± 0.04 b | 0.11 ± 0.04 a | 0.04 ± 0.00 cd | 0.04 ± 0.00 cd | 0.08 ± 0.04 c | 0.999 | 17.4 |
| ANOVA | * | * | * | * | * | * | * | ||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cruz, J.M.d.A.; Ramos, A.S.; Corrêa, R.F.; Sanches, E.A.; Campelo, P.H.; Kinupp, V.F.; Bezerra, J.d.A. Thermal Treatment and High-Intensity Ultrasound Processing to Evaluate the Chemical Profile and Antioxidant Activity of Amazon Fig Juices. Processes 2023, 11, 408. https://doi.org/10.3390/pr11020408
Cruz JMdA, Ramos AS, Corrêa RF, Sanches EA, Campelo PH, Kinupp VF, Bezerra JdA. Thermal Treatment and High-Intensity Ultrasound Processing to Evaluate the Chemical Profile and Antioxidant Activity of Amazon Fig Juices. Processes. 2023; 11(2):408. https://doi.org/10.3390/pr11020408
Chicago/Turabian StyleCruz, Josias M. dos A., Andrezza S. Ramos, Renilto F. Corrêa, Edgar A. Sanches, Pedro H. Campelo, Valdely F. Kinupp, and Jaqueline de A. Bezerra. 2023. "Thermal Treatment and High-Intensity Ultrasound Processing to Evaluate the Chemical Profile and Antioxidant Activity of Amazon Fig Juices" Processes 11, no. 2: 408. https://doi.org/10.3390/pr11020408
APA StyleCruz, J. M. d. A., Ramos, A. S., Corrêa, R. F., Sanches, E. A., Campelo, P. H., Kinupp, V. F., & Bezerra, J. d. A. (2023). Thermal Treatment and High-Intensity Ultrasound Processing to Evaluate the Chemical Profile and Antioxidant Activity of Amazon Fig Juices. Processes, 11(2), 408. https://doi.org/10.3390/pr11020408







