Glow Discharge Plasma Processing for the Improvement of Pasteurized Orange Juice’s Aroma and Off-Flavor
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Plasma Processing
2.3. GC-MS Analysis
2.4. Odor Profile
2.5. Statistical Analysis
3. Results and Discussion
3.1. Identification of the Volatile Profile
3.2. Modifications in the Volatile Profile
3.3. Modifications in the Aroma Profile
3.4. Modifications in the Off-Flavor Concentration
3.5. Further Considerations Regarding the Technology
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- TetraPak. The Orange Book; Tetra Pak International S.A.: Pully, Switzerland, 2017. [Google Scholar]
- Umair, M.; Jabbar, S.; Lin, Y.; Nasiru, M.M.; Zhang, J.; Abid, M.; Murtaza, M.A.; Zhao, L. Comparative Study: Thermal and Non-thermal Treatment on Enzyme Deactivation and Selected Quality Attributes of Fresh Carrot Juice. Int. J. Food Sci. Technol. 2022, 57, 827–841. [Google Scholar] [CrossRef]
- Kruszewski, B.; Zawada, K.; Karpiński, P. Impact of High-Pressure Homogenization Parameters on Physicochemical Characteristics, Bioactive Compounds Content, and Antioxidant Capacity of Blackcurrant Juice. Molecules 2021, 26, 1802. [Google Scholar] [CrossRef]
- Silva, E.K.; Bargas, M.A.; Arruda, H.S.; Vardanega, R.; Pastore, G.M.; Meireles, M.A.A. Supercritical CO2 Processing of a Functional Beverage Containing Apple Juice and Aqueous Extract of Pfaffia Glomerata Roots: Fructooligosaccharides Chemical Stability after Non-Thermal and Thermal Treatments. Molecules 2020, 25, 3911. [Google Scholar] [CrossRef] [PubMed]
- Putnik, P.; Kresoja, Z.; Bosiljkov, T.; Jambrak, A.R.; Barba, F.J.; Lorenzo, J.M.; Roohinejad, S.; Granato, D.; Zuntar, I.; Kovacevic, D.B. Comparison the Effects of Thermal and Non-Thermal Technologies on Pomegranate Juice Quality: A Review. Food Chem. 2018, 279, 150–161. [Google Scholar] [CrossRef]
- Campelo, P.H.; Alves Filho, E.G.; Silva, L.M.A.; de Brito, E.S.; Rodrigues, S.; Fernandes, F.A.N. Modulation of Aroma and Flavor Using Glow Discharge Plasma Technology. Innov. Food Sci. Emerg. Technol. 2020, 62, 102363. [Google Scholar] [CrossRef]
- Farias, T.R.B.; Alves Filho, E.G.; Silva, L.M.A.; De Brito, E.S.; Rodrigues, S.; Fernandes, F.A.N. NMR Evaluation of Apple Cubes and Apple Juice Composition Subjected to Two Cold Plasma Technologies. LWT Food Sci. Technol. 2021, 150, 112062. [Google Scholar] [CrossRef]
- Campelo, P.H.; Alves Filho, E.G.; Silva, L.M.A.; de Brito, E.S.; Rodrigues, S.; Fernandes, F.A.N. Modulation of Aroma and Flavor Using Dielectric Barrier Discharge Plasma Technology in a Juice Rich in Terpenes and Sesquiterpenes. LWT 2020, 130, 109644. [Google Scholar] [CrossRef]
- Moshonas, M.G.; Shaw, P.E. Flavor and Chemical Comparison of Pasteurized and Fresh Valencia Orange Juices. J. Food Qual. 1997, 20, 31–40. [Google Scholar] [CrossRef]
- Marcotte, M.; Stewart, B.; Fustier, P. Abused Thermal Treatment Impact on Degradation Products of Chilled Pasteurized Orange Juice. J. Agric. Food Chem. 1998, 46, 1991–1996. [Google Scholar] [CrossRef]
- Alves Filho, E.G.; Silva, L.M.A.; de Brito, E.S.; Wurlitzer, N.J.; Fernandes, F.A.N.; Rabelo, M.C.; Fonteles, T.V.; Rodrigues, S. Evaluation of Thermal and Non-Thermal Processing Effect on Non-Prebiotic and Prebiotic Acerola Juices Using1H QNMR and GC–MS Coupled to Chemometrics. Food Chem. 2018, 265, 23–31. [Google Scholar] [CrossRef] [Green Version]
- Alves Filho, E.G.; Rodrigues, T.H.S.; Fernandes, F.A.N.; de Brito, E.S.; Cullen, P.J.; Frias, J.M.; Bourke, P.; Cavalcante, R.S.; Almeida, F.D.L.; Rodrigues, S. An Untargeted Chemometric Evaluation of Plasma and Ozone Processing Effect on Volatile Compounds in Orange Juice. Innov. Food Sci. Emerg. Technol. 2019, 53, 63–69. [Google Scholar] [CrossRef]
- Rodríguez, Ó.; Gomes, W.F.; Rodrigues, S.; Fernandes, F.A.N. Effect of Indirect Cold Plasma Treatment on Cashew Apple Juice (Anacardium Occidentale L.). LWT Food Sci. Technol. 2017, 84, 457–463. [Google Scholar] [CrossRef]
- Steffen, A.; Pawliszyn, J. Analysis of Flavor Volatiles Using Headspace Solid-Phase Microextraction. J. Agric. Food Chem. 1996, 44, 2187–2193. [Google Scholar] [CrossRef]
- Gómez-Ariza, J.L.; García-Barrera, T.; Lorenzo, F. Determination of Flavour and Off-Flavour Compounds in Orange Juice by on-Line Coupling of a Pervaporation Unit to Gas Chromatography–Mass Spectrometry. J. Chromatogr. A 2004, 1047, 313–317. [Google Scholar] [CrossRef]
- Company, T.G.S. The Good Scents Company Database. Available online: http://www.thegoodscentscompany.com/ (accessed on 5 March 2022).
- Grosch, W. Evaluation of the Key Odorants of Foods by Dilution Experiments, Aroma Models and Omission. Chem. Senses 2001, 26, 533–545. [Google Scholar] [CrossRef]
- Usami, A.; Ono, T.; Marumoto, S.; Miyazawa, M. Comparison of Volatile Compounds with Characteristic Odor in Flowers and Leaves of Nojigiku (Chrysanthemum Japonense). J. Oleo Sci. 2013, 62, 631–636. [Google Scholar] [CrossRef]
- Padrayuttawat, A.; Yoshizawa, T.; Tamura, H.; Tokunaga, T. Optical Isomers and Odor Thresholds of Volatile Constituents in Citrus Sudachi. Food Sci. Technol. Int. Tokyo 1997, 3, 402–408. [Google Scholar] [CrossRef]
- Xiao, Z.; Chen, J.; Niu, Y.; Chen, F. Characterization of the Key Odorants of Fennel Essential Oils of Different Regions Using GC–MS and GC–O Combined with Partial Least Squares Regression. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2017, 1063, 226–234. [Google Scholar] [CrossRef]
- Fazzalari, F.A. Compilation of Odor and Taste Threshold Values Data; American Society for Testing and Materials: Philadelphia, PA, USA, 1978. [Google Scholar]
- Buttery, R.G.; Ling, L.C.; Light, D.M. Tomato Leaf Volatile Aroma Components. J. Agric. Food Chem. 1987, 35, 1039–1042. [Google Scholar] [CrossRef]
- Qian, M.C.; Wang, Y. Seasonal Variation of Volatile Composition and Odor Activity Value of “Marion” (Rubus Spp. Hyb) and “Thornless Evergreen” (R. Laciniatus L.) Blackberries. J. Food Sci. 2005, 70, 13–20. [Google Scholar] [CrossRef]
- Perez-Cacho, P.R.; Rouseff, R. Processing and Storage Effects on Orange Juice Aroma: A Review. J. Agric. Food Chem. 2008, 56, 9785–9796. [Google Scholar] [CrossRef]
- Elss, S.; Kleinhenz, S.; Schreier, P. Odor and Taste Thresholds of Potential Carry-over/off-Flavor Compounds in Orange and Apple Juice. LWT Food Sci. Technol. 2007, 40, 1826–1831. [Google Scholar] [CrossRef]
- Ito, T.; Uchida, G.; Nakajima, A.; Takenaka, K.; Setsuhara, Y. Control of Reactive Oxygen and Nitrogen Species Production in Liquid by Nonthermal Plasma Jet with Controlled Surrounding Gas. Jpn. J. Appl. Phys. 2017, 56, 01AC06. [Google Scholar] [CrossRef]
- Takahashi, K.; Satoh, K.; Itoh, H.; Kawaguchi, H.; Timoshkin, I.; Given, M.; MacGregor, S. Production Characteristics of Reactive Oxygen/Nitrogen Species in Water Using Atmospheric Pressure Discharge Plasmas. Jpn. J. Appl. Phys. 2016, 55, 07FL01. [Google Scholar] [CrossRef]
- Chen, F.; Al-Ahmad, H.; Joyce, B.; Zhao, N.; Kollner, T.G.; Degenhardt, J.; Stewart, C.N., Jr. Within-Plant Distribution and Emission of Sesquiterpenes from Copaifera Officinalis. Plant Physiol. Biochem. 2009, 47, 1017–1023. [Google Scholar] [CrossRef]
- Agger, S.; Lopez-Gallego, F.; Schmidt-Dannert, C. Diversity of Sesquiterpene Synthases in the Basidiomycete Coprinus Cinereus. Mol. Microbiol. 2009, 72, 1181–1195. [Google Scholar] [CrossRef]
- Konen, P.P.; Wust, M. Analysis of Sesquiterpene Hydrocarbons in Grape Berry Exocarp (Vitis Vinifera L.) Using in Vivo-Labeling and Comprehensive Two-Dimensional Gas Chromatography-Mass Spectrometry (GCxGC-MS). Beilstein J. Org. Chem. 2019, 15, 1945–1961. [Google Scholar] [CrossRef]
- Kumeta, Y.; Ito, M. Characterization of A-Humulene Synthases Responsible for the Production of Sesquiterpenes Induced by Methyl Jasmonate in Aquilaria Cell Culture. J. Nat. Med. 2016, 70, 452–459. [Google Scholar] [CrossRef]
- Dauben, W.G.; Hubbell, J.P.; Vietmeyer, N.D. Acid-Catalyzed Rearrangements of Humulene. J. Org. Chem. 1975, 40, 479–485. [Google Scholar] [CrossRef]
- Hu, C.J.; Li, D.; Ma, Y.X.; Zhang, W.; Lin, C.; Zheng, X.Q.; Liang, Y.R.; Lu, J.L. Formation Mechanism of the Oolong Tea Characteristic Aroma during Bruising and Withering Treatment. Food Chem. 2018, 269, 202–211. [Google Scholar] [CrossRef]
- Sampedro, F.; Fan, X. Inactivation Kinetics and Photoreactivation of Vegetable Oxidative Enzymes after Combined UV-C and Thermal Processing. Innov. Food Sci. Emerg. Technol. 2014, 23, 107–113. [Google Scholar] [CrossRef]
- Nordby, H.E.; Nagy, S. Fatty Acid Profiles of Orange and Tangor Juice Sac Lipids. Phytochemistry 1971, 10, 615–619. [Google Scholar] [CrossRef]
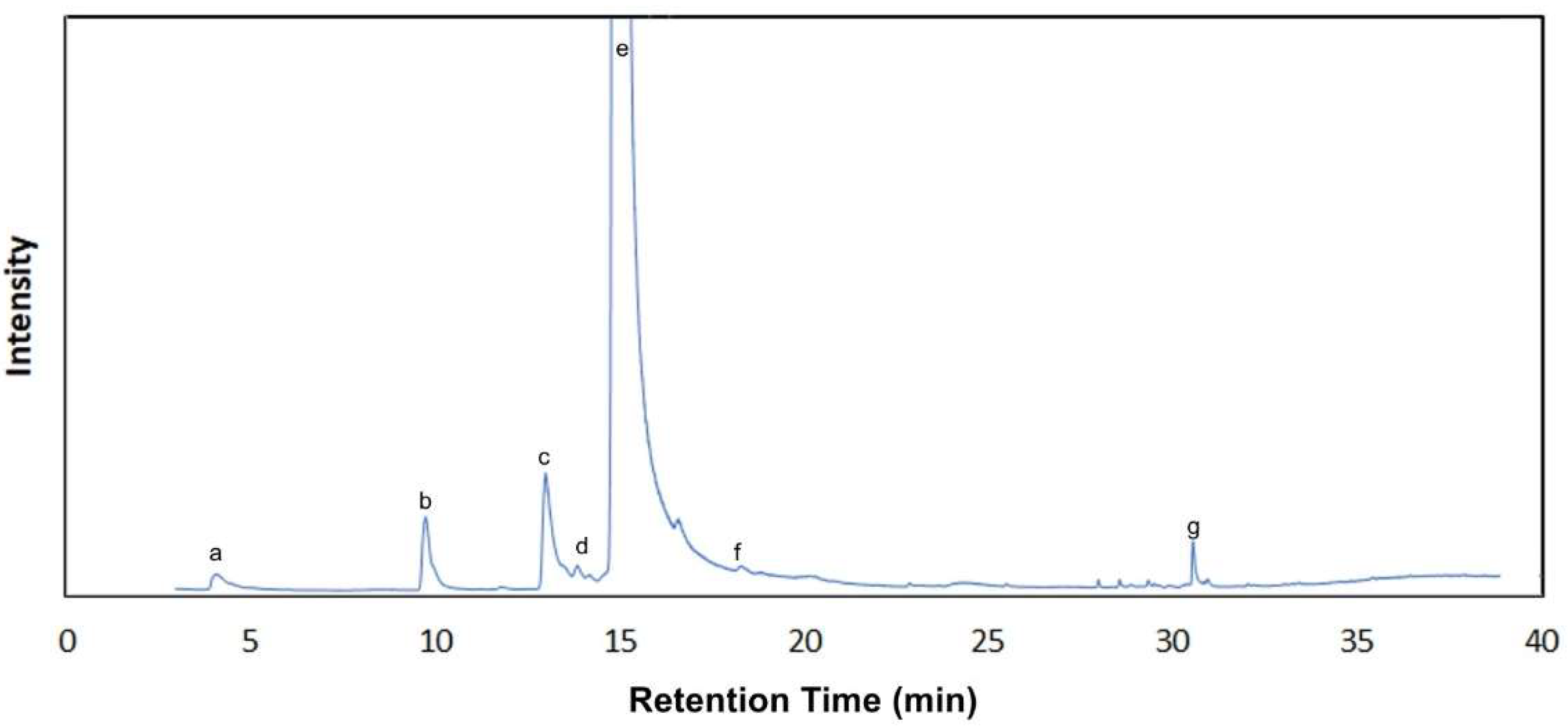

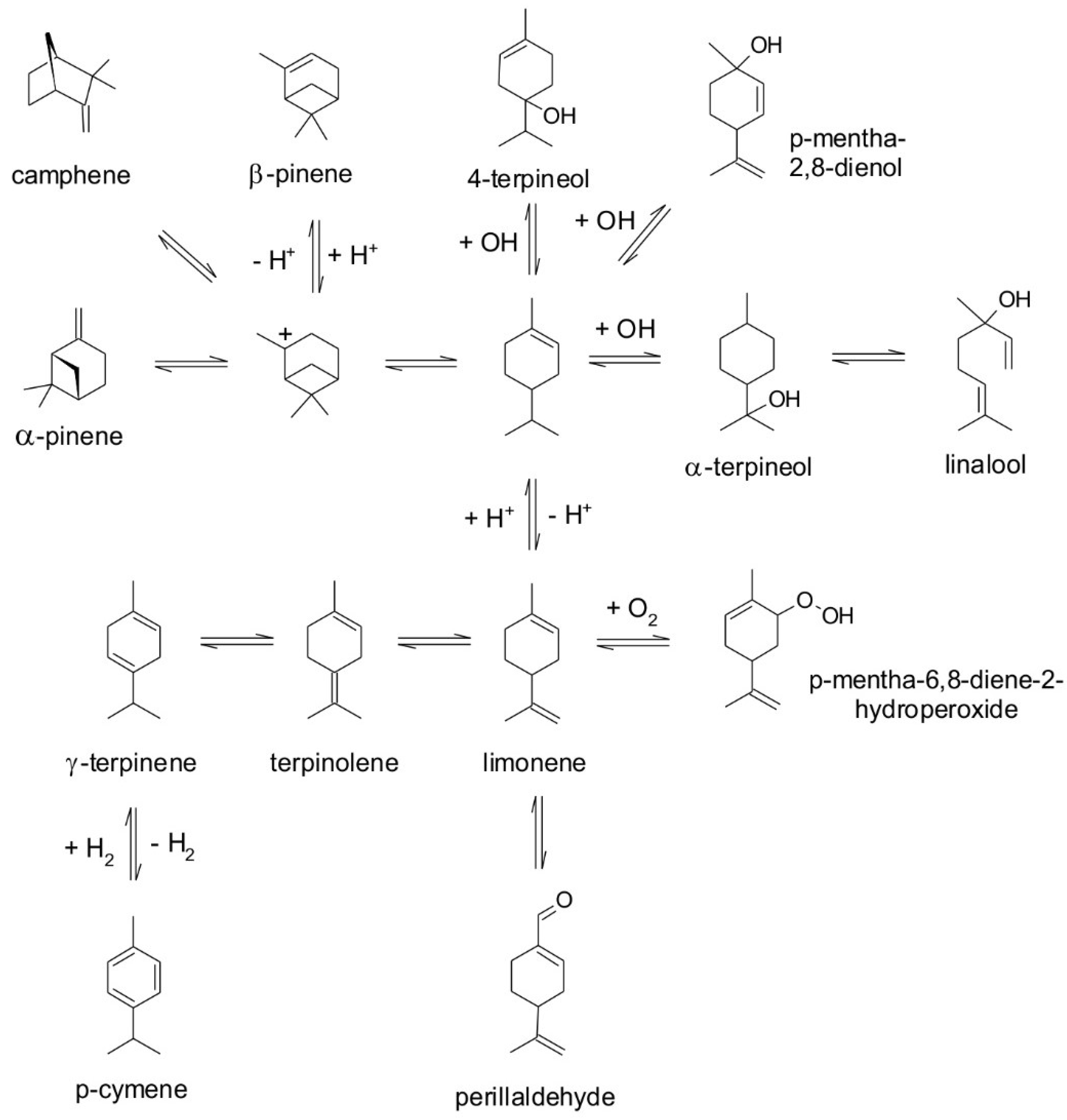

| Compound | Retention Time (min) | Kovats Index | m/z | Odor Threshold in Water (mg/L) | Odor Description |
|---|---|---|---|---|---|
| Alcohols | |||||
| 1-Octanol | 17.97 | 1084 | 56.0; 41.0; 70.0 | 0.875 | Waxy |
| α-Linalool | 19.10 | 1107 | 71.0; 93.0; 55.0 | 0.006 | Floral |
| p-Mentha-2,8-dienol | 20.40 | 1133 | 73.0; 91.0; 117.0 | -- | -- |
| 4-Terpineol | 23.11 | 1188 | 71.0; 111.0; 43.0 | 6.400 | Spicy |
| α-Terpineol | 23.83 | 1204 | 59.0; 93.0; 136.0 | 0.330 | Woody |
| Aldehydes | |||||
| Octanal | 13.79 | 999 | 43.0; 41.0; 56.0 | 0.0069 | Aldehydic |
| Nonanal | 19.52 | 1115 | 41.0; 43.0; 57.0 | 0.001 | Aldehydic |
| Decanal | 24.30 | 1221 | 41.0; 43.0; 55.0 | 0.030 | Aldehydic |
| Perillaldehyde | 26.91 | 1311 | 68.0; 79.0; 107.0 | 0.030 | Herbal |
| Dodecanal | 29.84 | 1425 | 41.0; 43.0; 57.0 | 0.002 | Aldehydic |
| Terpenoids | |||||
| α-Pinene | 9.56 | 914 | 93.0; 92.0; 77.0 | 0.062 | Herbal |
| Camphene | 10.36 | 930 | 93.0; 121.0; 41.0 | 1.980 | Terpenic |
| Sabinene | 11.86 | 960 | 93.0; 91.0; 77.0 | 0.037 | Woody |
| β-Pinene | 12.69 | 977 | 93.0; 41.0; 69.0 | 2.540 | Herbal |
| 3-Carene | 13.53 | 994 | 93.0; 91.0; 77.0 | 0.044 | Citrus |
| Terpinolene | 14.11 | 1006 | 93.0; 121.0; 136.0 | 0.200 | Herbal |
| Limonene | 14.97 | 1023 | 93.0; 68.0; 136.0 | 1.040 | Citrus |
| γ-Terpinene | 16.48 | 1054 | 93.0; 91.0; 136.0 | 0.260 | Terpenic |
| p-Cymene | 18.54 | 1095 | 119.0; 134.0; 91.0 | 0.120 | Terpenic |
| Other Compounds | |||||
| 3-Amino-2,3-dihydrobenzoic acid | 2.67 | 774 | 166.0; 150.0; 147.0 | -- | -- |
| Ethyl butanoate | 4.56 | 812 | 71.0; 43.0; 88.0 | 0.100 | Fruity |
| p-Mentha-6,8-diene-2-hydroperoxide | 28.97 | 1383 | 109.0; 93.0; 107.0 | -- | -- |
| Germacrene D | 30.04 | 1438 | 119.0; 161.0; 91.0 | -- | -- |
| Eremophila-1(10),11-diene | 31.09 | 1506 | 107.0; 93.0; 161.0 | -- | -- |
| Compound | Control Sample (Untreated) | Plasma Treatment 10 mL/min 10 min | Plasma Treatment 10 mL/min 20 min | Plasma Treatment 10 mL/min 30 min | Plasma Treatment 20 mL/min 10 min | Plasma Treatment 20 mL/min 20 min | Plasma Treatment 20 mL/min 30 min | Plasma Treatment 30 mL/min 10 min | Plasma Treatment 30 mL/min 20 min | Plasma Treatment 30 mL/min 30 min |
|---|---|---|---|---|---|---|---|---|---|---|
| 3-Amino-2,3-DBA | 0.03 ± 0.01 bc | 0.03 ± 0.01 bc | 0.00 ± 0.00 d | 0.06 ± 0.01 a | 0.03 ± 0.01 bc | 0.02 ± 0.01 cd | 0.04 ± 0.01 ab | 0.04 ± 0.01 ab | 0.03 ± 0.01 bc | 0.01 ± 0.01 cd |
| Ethyl butanoate | 0.21 ± 0.02 c | 0.24 ± 0.03 b | 0.21 ± 0.02 c | 0.24 ± 0.03 ab | 0.25 ± 0.03 ab | 0.25 ± 0.03 ab | 0.30 ± 0.03 a | 0.24 ± 0.02 b | 0.23 ± 0.02 bc | 0.23 ± 0.02 bc |
| α-Pinene | 0.94 ± 0.04 d | 0.79 ± 0.03 f | 1.55 ± 0.05 a | 1.32 ± 0.05 b | 0.82 ± 0.03 ef | 1.27 ± 0.05 b | 0.65 ± 0.03 g | 1.06 ± 0.05 c | 0.86 ± 0.03 e | 1.53 ± 0.05 a |
| Camphene | 0.07 ± 0.01 cd | 0.06 ± 0.01 d | 0.14 ± 0.02 a | 0.13 ± 0.02 ab | 0.07 ± 0.01 cd | 0.10 ± 0.02 bc | 0.06 ± 0.01 d | 0.10 ± 0.02 bc | 0.05 ± 0.01 d | 0.13 ± 0.02 ab |
| Sabinene | 0.02 ± 0.01 bc | 0.01 ± 0.01 c | 0.03 ± 0.01 ab | 0.02 ± 0.01 bc | 0.02 ± 0.01 bc | 0.01 ± 0.01 c | 0.05 ± 0.01 a | 0.02 ± 0.01 bc | 0.01 ± 0.01 c | 0.02 ± 0.01 bc |
| β-Pinene | 2.21 ± 0.05 d | 1.75 ± 0.05 e | 3.09 ± 0.05 a | 2.49 ± 0.05 c | 1.77 ± 0.05 e | 2.15 ± 0.05 d | 1.19 ± 0.05 g | 2.31 ± 0.05 b | 1.37 ± 0.05 f | 2.61 ± 0.05 b |
| 3-Carene | 0.46 ± 0.03 c | 0.32 ± 0.03 d | 0.61 ± 0.03 a | 0.54 ± 0.03 b | 0.31 ± 0.03 d | 0.45 ± 0.03 c | 0.26 ± 0.03 d | 0.46 ± 0.03 c | 0.29 ± 0.02 d | 0.54 ± 0.03 b |
| Octanal | 0.14 ± 0.02 cd | 0.11 ± 0.02 d | 0.18 ± 0.02 ab | 0.18 ± 0.02 ab | 0.10 ± 0.02 d | 0.16 ± 0.02 bc | 0.20 ± 0.02 a | 0.11 ± 0.02 d | 0.17 ± 0.02 ab | 0.19 ± 0.02 a |
| Terpinolene | 0.11 ± 0.02 a | 0.08 ± 0.02 ab | 0.11 ± 0.02 a | 0.10 ± 0.02 a | 0.06 ± 0.02 bc | 0.08 ± 0.02 ab | 0.06 ± 0.02 bc | 0.09 ± 0.02 ab | 0.05 ± 0.02 c | 0.10 ± 0.02 a |
| Limonene | 92.08 ± 0.09 b | 93.57 ± 0.10 a | 89.21 ± 0.08 e | 90.46 ± 0.10 d | 93.38 ± 0.10 a | 91.62 ± 0.08 c | 93.62 ± 0.10 a | 91.54 ± 0.08 c | 93.35 ± 0.10 a | 90.10 ± 0.10 d |
| γ-Terpinene | 0.34 ± 0.03 ab | 0.25 ± 0.03 cd | 0.40 ± 0.03 a | 0.34 ± 0.03 ab | 0.28 ± 0.03 ac | 0.31 ± 0.03 b | 0.21 ± 0.03 d | 0.33 ± 0.03 b | 0.21 ± 0.03 d | 0.38 ± 0.03 ab |
| 1-Octanol | 0.21 ± 0.02 cd | 0.14 ± 0.01 e | 0.35 ± 0.02 a | 0.28 ± 0.02 b | 0.17 ± 0.01 d | 0.22 ± 0.02 c | 0.14 ± 0.01 e | 0.24 ± 0.02 bc | 0.13 ± 0.02 e | 0.31 ± 0.02 a |
| p-Cymenene | 2.44 ± 0.08 d | 1.72 ± 0.05 f | 3.24 ± 0.07 a | 2.66 ± 0.07 c | 1.74 ± 0.05 f | 2.18 ± 0.06 e | 1.09 ± 0.05 h | 2.68 ± 0.05 c | 1.25 ± 0.05 g | 2.83 ± 0.05 b |
| α-Linalool | 0.20 ± 0.03 bc | 0.26 ± 0.03 b | 0.17 ± 0.02 a | 0.21 ± 0.03 bc | 0.22 ± 0.03 bc | 0.22 ± 0.03 bc | 0.37 ± 0.03 a | 0.21 ± 0.03 bc | 0.34 ± 0.03 a | 0.20 ± 0.03 bc |
| Nonanal | 0.02 ± 0.01 bc | 0.01 ± 0.01 c | 0.03 ± 0.01 bc | 0.05 ± 0.01 a | 0.01 ± 0.01 c | 0.03 ± 0.01 b | 0.04 ± 0.01 ab | 0.01 ± 0.01 c | 0.03 ± 0.01 bc | 0.04 ± 0.01 ab |
| p-m-2,8-dienol | 0.01 ± 0.01 b | 0.06 ± 0.01 b | 0.01 ± 0.01 b | 0.08 ± 0.01 b | 0.14 ± 0.01 b | 0.09 ± 0.01 b | 0.28 ± 0.01 b | 0.01 ± 0.01 b | 0.32 ± 0.01 b | 0.01 ± 0.01 b |
| 4-Terpineol | 0.06 ± 0.02 ab | 0.06 ± 0.01 ab | 0.04 ± 0.01 b | 0.05 ± 0.01 b | 0.04 ± 0.01 b | 0.05 ± 0.01 b | 0.08 ± 0.01 a | 0.05 ± 0.01 b | 0.08 ± 0.01 a | 0.05 ± 0.01 b |
| α-Terpineol | 0.15 ± 0.02 bc | 0.13 ± 0.03 bc | 0.08 ± 0.02 d | 0.16 ± 0.02 bc | 0.13 ± 0.02 c | 0.13 ± 0.02 c | 0.22 ± 0.04 a | 0.13 ± 0.02 c | 0.20 ± 0.03 ab | 0.11 ± 0.02 cd |
| Decanal | 0.08 ± 0.01 c | 0.08 ± 0.01 c | 0.18 ± 0.02 b | 0.21 ± 0.03 b | 0.08 ± 0.01 c | 0.19 ± 0.02 b | 0.30 ± 0.02 a | 0.09 ± 0.01 c | 0.23 ± 0.02 b | 0.23 ± 0.03 b |
| Perillaldehyde | 0.00 ± 0.00 f | 0.06 ± 0.01 d | 0.04 ± 0.00 e | 0.07 ± 0.01 d | 0.14 ± 0.01 c | 0.08 ± 0.01 d | 0.23 ± 0.02 b | 0.00 ± 0.00 f | 0.29 ± 0.02 a | 0.01 ± 0.00 f |
| p-m-6,8-d-2-h | 0.01 ± 0.00 d | 0.02 ± 0.00 c | 0.04 ± 0.01 b | 0.05 ± 0.01 ab | 0.02 ± 0.00 c | 0.05 ± 0.01 ab | 0.06 ± 0.01 a | 0.02 ± 0.00 c | 0.05 ± 0.01 ab | 0.06 ± 0.01 a |
| Dodecanal | 0.01 ± 0.00 b | 0.02 ± 0.01 ab | 0.02 ± 0.00 ab | 0.02 ± 0.00 ab | 0.01 ± 0.00 b | 0.02 ± 0.01 ab | 0.03 ± 0.01 a | 0.01 ± 0.00 b | 0.03 ± 0.01 a | 0.02 ± 0.00 ab |
| Germacrene D | 0.01 ± 0.00 b | 0.01 ± 0.00 b | 0.02 ± 0.01 b | 0.02 ± 0.00 ab | 0.01 ± 0.00 b | 0.02 ± 0.01 ab | 0.03 ± 0.01 a | 0.01 ± 0.00 b | 0.02 ± 0.00 ab | 0.02 ± 0.01 ab |
| Er-1(10),11-diene | 0.19 ± 0.02 d | 0.23 ± 0.02 cd | 0.24 ± 0.02 cd | 0.27 ± 0.02 bc | 0.22 ± 0.02 d | 0.30 ± 0.03 b | 0.49 ± 0.03 a | 0.22 ± 0.02 d | 0.42 ± 0.04 a | 0.30 ± 0.03 b |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rodrigues, S.; Fernandes, F.A.N. Glow Discharge Plasma Processing for the Improvement of Pasteurized Orange Juice’s Aroma and Off-Flavor. Processes 2022, 10, 1812. https://doi.org/10.3390/pr10091812
Rodrigues S, Fernandes FAN. Glow Discharge Plasma Processing for the Improvement of Pasteurized Orange Juice’s Aroma and Off-Flavor. Processes. 2022; 10(9):1812. https://doi.org/10.3390/pr10091812
Chicago/Turabian StyleRodrigues, Sueli, and Fabiano Andre Narciso Fernandes. 2022. "Glow Discharge Plasma Processing for the Improvement of Pasteurized Orange Juice’s Aroma and Off-Flavor" Processes 10, no. 9: 1812. https://doi.org/10.3390/pr10091812
APA StyleRodrigues, S., & Fernandes, F. A. N. (2022). Glow Discharge Plasma Processing for the Improvement of Pasteurized Orange Juice’s Aroma and Off-Flavor. Processes, 10(9), 1812. https://doi.org/10.3390/pr10091812








