Biocontrol Methods in Avoidance and Downsizing of Mycotoxin Contamination of Food Crops
Abstract
1. Introduction
2. The Main Mycotoxins, Their Occurrence, and the Producers
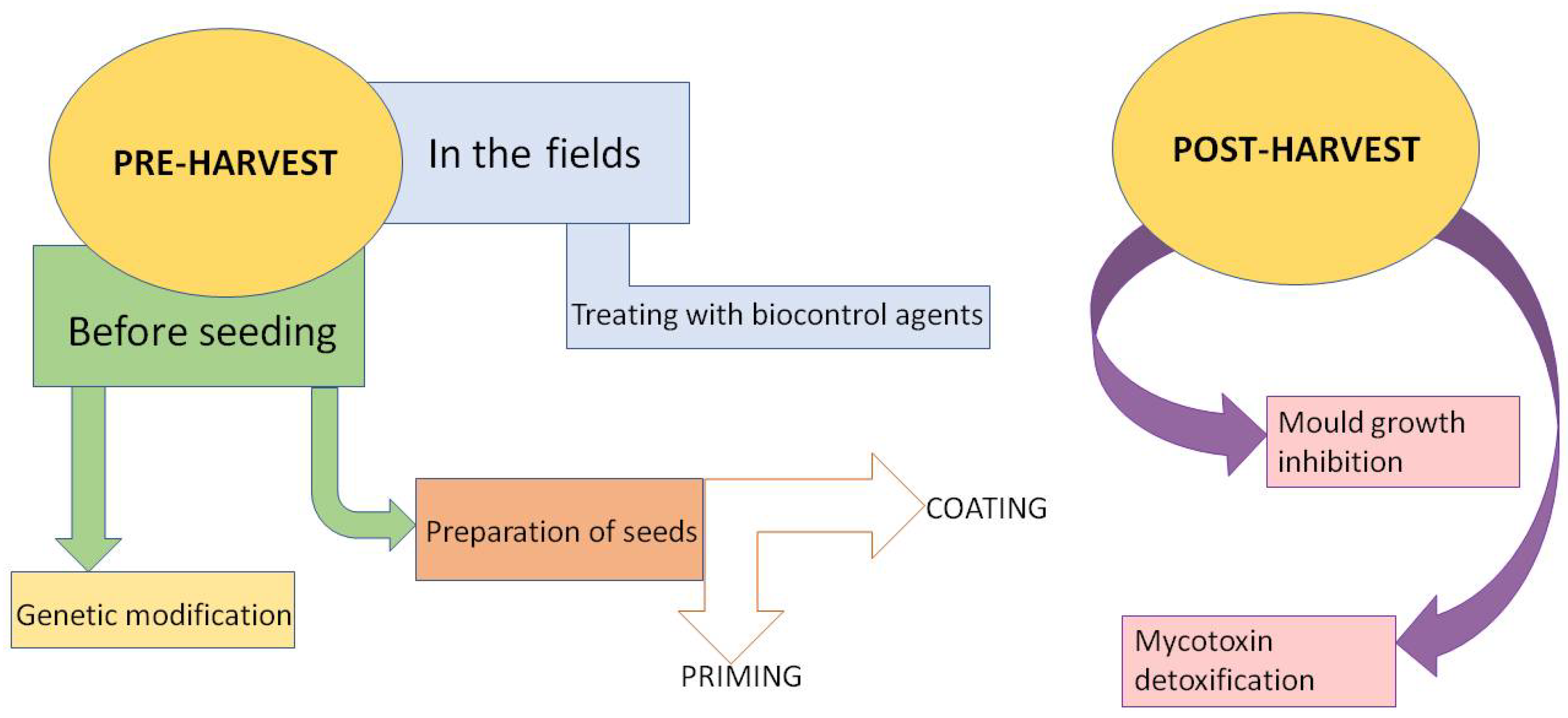
3. Pre-Harvesting Biocontrol Methods
3.1. Before Seeding
3.1.1. Genetic Base of Natural Resistance
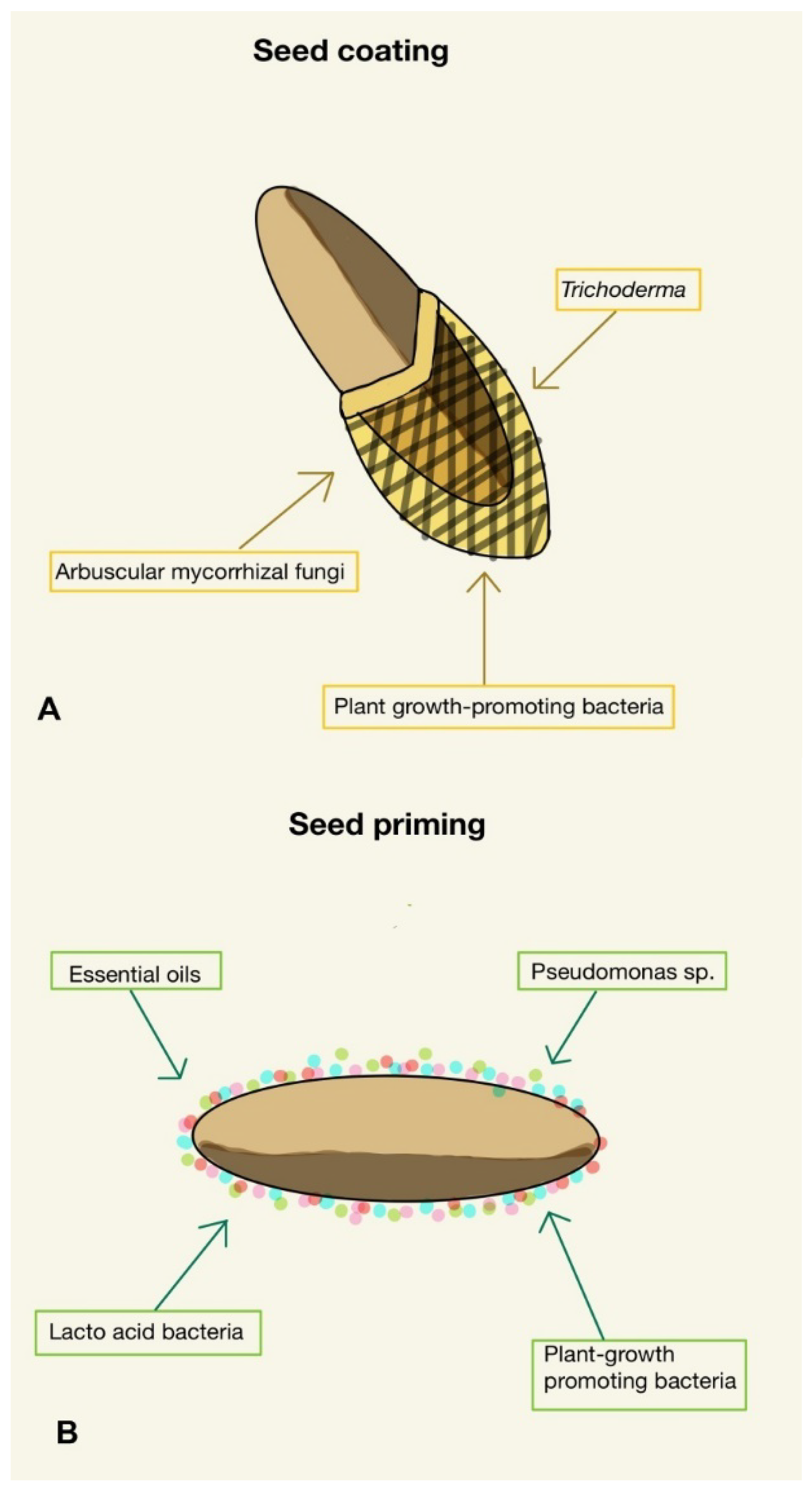
3.1.2. Preparation of Seeds before Sowing
3.2. In the Fields
4. Post-Harvest Biocontrol Methods
4.1. The Inhibition of Mold Growth
4.1.1. Microorganisms and Their Metabolites
4.1.2. Plant Extracts
4.2. Biological Degradation of Mycotoxins
Lactic Acid Bacteria
4.3. Adsorption by Microorganisms
5. Conclusions and Perspective
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- FAO. Save and Grow. In A Policymaker‘s Guide to the Sustainable Intensification of Smallholder Crop Production; Food and Agriculture Organization: Rome, Italy, 2011; ISBN 978-92-5-106871-7. [Google Scholar]
- Hussain, S.S.; Kayani, M.A.; Amjad, M. Transcription factors as tools to engineer enhanced drought stress tolerance in plants. Biotechnol. Prog. 2011, 27, 297–306. [Google Scholar] [CrossRef] [PubMed]
- Chassy, B.M. Food safety risk and consumer health. New Biotechnol. 2010, 27, 534–544. [Google Scholar] [CrossRef] [PubMed]
- Luo, S.; Du, H.; Kebede, H.; Liu, Y.; Xing, F. Contamination status of major mycotoxins in agricultural product and foodstuff in Europe. Food Control 2021, 127, 108120. [Google Scholar] [CrossRef]
- Grusie, T.; Cowan, V.; Singh, J.; McKinnon, J.; Blakley, B. Proportions of predominant Ergot alkaloids (Claviceps purpurea) detected in Western Canadian grains from 2014 to 2016. World Mycotoxin J. 2018, 11, 259–264. [Google Scholar] [CrossRef]
- Escrivá, L.; Oueslati, S.; Font, G.; Manyes, L. Alternaria Mycotoxins in Food and Feed: An Overview. J. Food Quality 2017, 1, 569748. [Google Scholar] [CrossRef]
- Windham, G.L.; Williams, W.P.; Mylroie, J.E.; Reid, C.X.; Womack, E.D. A histological study of Aspergillus flavus colonization of wound inoculated maize kernels of resistant and susceptible maize hybrids in the field. Front. Microbiol. 2018, 9, 799. [Google Scholar] [CrossRef]
- Matumba, L.; Namaumbo, S.; Ngoma, T.; Meleke, N.; De Boevre, M.; Logrieco, A.F.; De Saeger, S. Five keys to prevention and control of mycotoxins in grains: A proposal. Glob. Food Secur. 2021, 30, 100562. [Google Scholar] [CrossRef]
- Atanda, S.A.; Pessu, P.O.; Aina, J.A.; Agoda, S.; Adekalu, O.A.; Ihionu, G.C. Mycotoxin Management in Agriculture. Greener J. Agric. Sci. 2013, 3, 176–184. [Google Scholar]
- Majumder, D.; Thangaswamy, R.; Suting, E.G.; Debbarma, A. Detection of seed borne pathogens in wheat: Recent trends. Aust. J. Crop Sci. 2013, 7, 500–507. [Google Scholar]
- Singh, H.; Rupinder, K.J.; Kang, J.S.; Sandhu, S.S.; Kang, H.; Grewal, K. Seed priming techniques in field crops—A review. Agric. Rev. 2015, 36, 251–264. [Google Scholar] [CrossRef]
- Eskola, M.; Kos, G.; Elliott, C.T.; Hajšlová, J.; Mayar, S.; Krska, R. Worldwide contamination of food-crops with mycotoxins: Validity of the widely cited ‘FAO estimate’ of 25%. Crit. Rev. Food Sci. Nutr. 2020, 60, 2773–2789. [Google Scholar] [CrossRef] [PubMed]
- Schmidt-Heydt, M.; Stoll, D.; Geisen, R. Fungicides effectively used for growth inhibition of several fungi could induce mycotoxin biosynthesis in toxigenic species. Int. J. Food Microbiol. 2013, 166, 407–412. [Google Scholar] [CrossRef] [PubMed]
- Daou, R.; Joubrane, K.; Maroun, R.G.; Khabbaz, L.R.; Ismail, A. Mycotoxins: Factors influencing production and control strategies. AIMS Agric. Food 2021, 6, 416–447. [Google Scholar] [CrossRef]
- Choudhary, A.K.; Kumari, P. Management of mycotoxin contamination in preharvest and post harvest crops: Present status and future prospects. J. Phytpath. 2010, 2, 37–52. [Google Scholar]
- Gonçalves, A.; Gkrillas, J.L.; Dorne, C.; Dall’Asta, R.; Palumbo, N.; Lima, P.; Battilani, A.; Giorni, V.P. Pre- and Postharvest Strategies to Minimize Mycotoxin Contamination in the Rice Food Chain. Compr. Rev. Food Sci. F 2019, 18, 441–454. [Google Scholar] [CrossRef]
- FAO. Manual on the Application of the HACCP System in Mycotoxin Prevention and Control; Food and Nutrition Paper 73; Food and Agriculture Organization: Rome, Italy, 2001; ISBN 92-5-104611-5. [Google Scholar]
- Kent-Jones, D. Cereal Farming. Encyclopedia Britannica. 2017. Available online: https://www.britannica.com/topic/cereal-farming (accessed on 20 January 2022).
- Saavoss, M.; Capehart, T.; McBride, W.; Effland, A. Trends in Production Practices and Costs of the U.S. Corn Sector; ERR-294; U.S. Department of Agriculture, Economic Research Service: Washington, DC, USA, 2021.
- Santos Pereira, C.; Cunha, C.S.; Fernandes, J.O. Prevalent Mycotoxins in Animal Feed: Occurrence and Analytical Methods. Toxins 2019, 11, 290. [Google Scholar] [CrossRef]
- Ferreira, M.R.; Sereia, M.J.; Poliseli, F.H.; Formigoni, M.; Rigobello, E.S.; Beneti, S.C.; Reitz, F.A.; Marchi, L.B.; da Silva, C.G.; Milani, P.G.; et al. Growth of Fungal Cells and the Production of Mycotoxins. In Cell Growth; Vikas, B., Fasullo, M., Eds.; IntechOpen: London, UK, 2019. [Google Scholar] [CrossRef]
- Kochiieru, Y.; Mankevičienė, A.; Cesevičienė, J.; Semaškienė, R.; Dabkevičius, Z.; Sigita, J. The influence of harvesting time and meteorological conditions on the occurrence of Fusarium species and mycotoxin contamination of spring cereals. J. Sci. Food Agric. 2020, 100, 2999–3006. [Google Scholar] [CrossRef]
- Karlovsky, P.; Suman, M.; Berthiller, F.; De Meester, J.; Eisenbrand, G.; Perrin, I.; Oswald, I.P.; Speijers, G.; Chiodini, A.; Recker, T.; et al. Impact of food processing and detoxification treatments on mycotoxin contamination. Mycotoxin Res. 2016, 32, 179–205. [Google Scholar] [CrossRef]
- Andrade, P.D.; Caldas, E.D. Aflatoxins in cereals: Worldwide occurrence and dietary risk assessment. World Mycotoxin J. 2015, 8, 415–431. [Google Scholar] [CrossRef]
- Majeed, M.; Asghar, A.; Randhawa, M.A.; Shahzad, M.A.; Sohaib, M.; Abdullah, D. Ochratoxin A in Cereal Products, Potential Hazards and Prevention strategies: A Review. Pak. J. Food Sci. 2013, 23, 52–61. [Google Scholar]
- Pleadin, J.; Zadravec, M.; Lešić, T.; Vahčić, N.; Frece, J.; Mitak, M.; Markov, K. Co-occurrence of ochratoxin A and citrinin in unprocessed cereals established during a three-year investigation period. Food Addit. Contam. B 2018, 11, 20–25. [Google Scholar] [CrossRef] [PubMed]
- Pleadin, J.; Staver, M.; Markov, K.; Frece, J.; Zadravec, M.; Jaki, V.; Krupić, I.; Vahčić, N. Mycotoxins in organic and conventional cereals and cereal products grown and marketed in Croatia. Mycotoxin Res. 2017, 33, 219–227. [Google Scholar] [CrossRef] [PubMed]
- Pleadin, J.; Vahčić, N.; Perši, N.; Ševelj, D.; Markov, K.; Frece, J. Fusarium mycotoxins’ occurrence in cereals harvested from Croatian fields. Food Control 2013, 32, 49–54. [Google Scholar] [CrossRef]
- Golge, O.; Kabak, B. Occurrence of deoxynivalenol and zearalenone in cereals and cereal products from Turkey. Food Control 2020, 110, 106982. [Google Scholar] [CrossRef]
- Farhadi, A.; Fakhri, Y.; Kachuei, R.; Vasseghian, Y.; Huseyn, E.; Khaneghah, A.M. Prevalence and concentration of fumonisins in cereal-based foods: A global systematic review and meta-analysis study. Environ. Sci. Pollut. R 2021, 28, 20998–21008. [Google Scholar] [CrossRef]
- Marques, A.; Buijs, G.; Ligterink, W.; Hilhorst, H. Evolutionary ecophysiology of seed desiccation sensitivity. Funct. Plant Biol. 2018, 45, 1083. [Google Scholar] [CrossRef]
- Smolikova, G.; Leonova, T.; Vashurina, N.; Frolov, A.; Medvedev, S. Desiccation Tolerance as the Basis of Long-Term Seed Viability. Int. J. Mol. Sci. 2020, 22, 101. [Google Scholar] [CrossRef]
- Cardarelli, M.; Woo, S.L.; Rouphael, Y.; Colla, G. Seed Treatments with Microorganisms Can Have a Biostimulant Effect by Influencing Germination and Seedling Growth of Crops. Plants 2022, 11, 259. [Google Scholar] [CrossRef]
- Rajasekaran, K.; Sickler, C.M.; Brown, R.L.; Cary, J.W.; Bhatnagar, D. Evaluation of resistance to aflatoxin contamination in kernels of maize genotypes using a GFP-expressing Aspergillus flavus strain. World Mycotoxin J. 2013, 6, 151–158. [Google Scholar] [CrossRef]
- Liu, F.; Zhang, H.; Wu, G.; Sun, J.; Hao, L.; Ge, X.; Yu, J.; Wang, W. Sequence variation and expression analysis of seed dormancy- and germination-associated ABA- and GA-related genes in rice cultivars. Front. Plant Sci. 2011, 2, 17. [Google Scholar] [CrossRef]
- Rae, G.M.; Karine, D.; Wood, M. The Dormancy Marker DRM1/ARP Associated with Dormancy but a Broader Role In Planta. J. Dev. Biol. 2013, 2013, 632524. [Google Scholar] [CrossRef]
- Naoto, S.; Rajjou, L.; North, H.M. Lost in Translation: Physiological Roles of Stored mRNAs in Seed Germination. Plants 2020, 9, 347. [Google Scholar] [CrossRef]
- Nazarul, H.; Choudhary, S.; Naaz, N.; Sharma, N.; Laskar, R.A. Recent advancements in molecular marker-assisted selection and applications in plant breeding programmes. J. Genet. Eng. Biotechnol. 2021, 19, 128. [Google Scholar] [CrossRef]
- Sanz, M.J.; Loarce, Y.; Fominaya, A.; Vossen, J.H.; Ferrer, E. Identification of RFLP and NBS/PK profiling markers for disease resistance loci in genetic maps of oats. Theor. Appl. Genet. 2013, 126, 203–218. [Google Scholar] [CrossRef] [PubMed]
- Vossen, J.H.; Dezhsetan, S.; Esselink, D.; Arens, M.; Sanz, M.J.; Verweij, W.; Verzaux, E.; van der Linden, C.G. Novel applications of motif-directed profiling to identify disease resistance genes in plants. Plant Methods 2013, 9, 37. [Google Scholar] [CrossRef] [PubMed]
- Ganal, M.W.; Pliesk, J.; Hohmeyer, A.; Polley, A.; Röder, M.S. High-throghput genotyping for cereals research and breeding. In Applications of Genetic and Genomic Research in Cereals; Miedaner, T., Korzon, V., Eds.; Woodhead Publishing: Sawston, UK, 2019; pp. 3–15. [Google Scholar]
- Collinge, B.D.; Sarrocco, S. Transgenic approaches for plant disease control: Status and prospects 2021. Plant Pathol. 2021, 71, 207–225. [Google Scholar] [CrossRef]
- Majumdar, R.; Rajasekaran, K.; Cary, J.W. RNA Interference (RNAi) as a Potential Tool for Control of Mycotoxin Contamination in Crop Plants: Concepts and Considerations. Front. Plant Sci. 2017, 8, 200. [Google Scholar] [CrossRef]
- Machado, A.K.; Brown, N.A.; Urban, M.; Kanyuka, K.; Hammond-Kosack, K.E. RNAi as an emerging approach to control Fusarium head blight disease and mycotoxin contamination in cereals. Pest Manag. Sci. 2018, 74, 790–799. [Google Scholar] [CrossRef]
- Grover, A. Plant chitinases: Genetic diversity and physiological roles. Crit. Rev. Plant Sci. 2012, 31, 57–73. [Google Scholar] [CrossRef]
- Ojiambo, P.S.; Battilani, P.; Cary, J.W.; Blum, B.H.; Carbone, I. Cultural and Genetic Approaches to Manage Aflatoxin Contamination: Recent Insights Provide Opportunities for Improved Control. Phytopathology 2018, 108, 1024–1037. [Google Scholar] [CrossRef]
- Thakare, D.; Zhang, J.; Wing, R.A.; Cotty, P.J.; Schmidt, M.A. Aflatoxin-free transgenic maize using host-induced gene silencing. Sci. Adv. 2017, 10, e1602382. [Google Scholar] [CrossRef] [PubMed]
- Masanga, J.O.; Matheka, J.M.; Omer, R.A.; Ommeh, S.C.; Monda, E.O.; Alakonya, A.E. Downregulation of transcription factor aflR in Aspergillus flavus confers reduction to aflatoxin accumulation in transgenic maize with alteration of host plant architecture. Plant Cell Rep. 2015, 34, 1379–1387. [Google Scholar] [CrossRef] [PubMed]
- Koch, A.; Kumar, N.; Weber, L.; Keller, H.; Imani, J.; Kogel, K.H. Host-induced gene silencing of cytochrome P450 lanosterol C14α-demethylase– encoding genes confers strong resistance to Fusarium species. Proc. Natl. Acad. Sci. USA 2013, 110, 19324–19329. [Google Scholar] [CrossRef] [PubMed]
- Cheng, W.; Song, X.S.; Li, H.P.; Cao, L.H.; Sun, K.; Qiu, X.L.; Xu, Y.B.; Yang, P.; Huang, T.; Zhang, J.B.; et al. Host-induced gene silencing of an essential chitin synthase gene confers durable resistance to Fusarium head blight and seedling blight in wheat. Plant Biotechnol. J. 2015, 13, 1335–1345. [Google Scholar] [CrossRef] [PubMed]
- McLaughlin, J.E.; Darwish, N.I.; Garcia-Sanchez, J.; Tyagi, N.; Trick, H.N.; McCormick, S.; Dill-Macky, R.; Tumer, N.T. A lipid transfer protein has antifungal and antioxidant activity and suppresses fusarium head blight disease and DON accumulation in transgenic wheat. Phytopathology 2021, 111, 671–683. [Google Scholar] [CrossRef] [PubMed]
- Shin, S.; Mackintosh, C.A.; Lewis, J.; Heinen, S.J.; Radmer, L.; Dill-Macky, R.; Baldridge, G.D.; Zeyen, R.J.; Muehlbauer, G.J. Transgenic wheat expressing a barley class II chitinase gene has enhanced resistance against Fusarium graminearum. J. Exp. Bot. 2008, 59, 2371–2378. [Google Scholar] [CrossRef]
- Li, P.; Pei, Y.; Sang, X.; Ling, Y.; Yang, Z.; He, G. Transgenic indica rice expressing a bitter melon (Momordica charantia) class I chitinase gene (McCHIT1) confers enhanced resistance to Magnaporthe grisea and Rhizoctonia solani. Eur. J. Plant Pathol. 2009, 125, 533–543. [Google Scholar] [CrossRef]
- Munkvold, G.P. Cultural and genetic approaches to managing mycotoxins in maize. Annu. Rev. Phytopathol. 2003, 41, 99–116. [Google Scholar] [CrossRef]
- Aaron, J. Gassmann Field-evolved resistance by western corn rootworm to multiple Bacillus thuringiensis toxins in transgenic maize. J. Invertebr. Pathol. 2012, 110, 287–293. [Google Scholar]
- Abbas, M.S.T. Genetically engineered (modified) crops (Bacillus thuringiensis crops) and the world controversy on their safety. Egypt. J. Biol. Pest Control 2018, 28, 52. [Google Scholar] [CrossRef]
- Zhang, J.; Khan, S.A.; Heckel, D.G.; Bock, R. Next-Generation Insect-Resistant Plants: RNAi-Mediated Crop Protection. Trends Biotechnol. 2017, 35, 871–882. [Google Scholar] [CrossRef] [PubMed]
- Yousaf, Z.; Saleh, N.; Ramazan, A.; Aftab, A. Postharvesting Techniques and Maintenance of Seed Quality. In New Challenges in Seed Biology—Basic and Translational Research Driving Seed Technology; Araújo, S., Ed.; ItechOpen: London, UK, 2016. [Google Scholar]
- Li, R.; Chen, L.; Wu, Y.; Zhang, R.; Baskin, C.C.; Baskin, J.M.; Hu, X. Effects of Cultivar and Maternal Environment on Seed Quality in Vicia sativa. Front. Plant Sci. 2017, 8, 1411. [Google Scholar] [CrossRef] [PubMed]
- Xiaoqing, S.; Cordero, T.; Garrigues, S.; Marcos, J.F.; Daros, J.-A.; Coca, M. Efficient production of antifungal proteins in plants using a new transient expression vector derived from tobacco mosaic virus. Plant Biotechnol. J. 2019, 17, 1069–1080. [Google Scholar] [CrossRef]
- Chacón-Orozco, J.G.; Bueno, C.J.; Shapiro-Ilan, D.I.; Hazir, S.; Leite, L.G.; Harakava, R. Antifungal activity of Xenorhabdu spp. and Photorhabdus spp. against the soybean pathogenic Sclerotinia sclerotiorum. Sci. Rep. 2020, 10, 20649. [Google Scholar] [CrossRef]
- Rocha, I.; Ma, Y.; Souza-Alonso, P.; Vosátka, M.; Freitas, H.; Oliveira, R.S. Seed Coating: A Tool for Delivering Beneficial Microbes to Agricultural Crops. Front. Plant Sci. 2019, 10, 1357. [Google Scholar] [CrossRef]
- Malusa, E.; Sas-Paszt, L.; Ciesielska, J. Technologies for Beneficial Microorganisms Inocula Used as Biofertilizers. Sci. World J. 2012, 2012, 491206. [Google Scholar] [CrossRef]
- Nadeem, S.M.; Ahmad, M.; Zahir, Z.A.; Javaid, A.; Ashraf, M. The role of mycorrhizae and plant growth promoting rhizobacteria (PGPR) in improving crop productivity under stressful environments. Biotechnol. Adv. 2014, 32, 429–448. [Google Scholar] [CrossRef]
- Hayat, R.; Ali, S.; Amara, U.; Khalid, R.; Ahmed, I. Soil beneficial bacteria and their role in plant growth promotion: A review. Ann. Micro 2010, 60, 579–598. [Google Scholar] [CrossRef]
- Li, T.; Lin, G.; Zhang, X.; Chen, Y.; Zhang, S.; Chen, B. Relative importance of an arbuscular mycorrhizal fungus (Rhizophagus intraradices) and root hairs in plant drought tolerance. Mycorrhiza 2014, 24, 595–602. [Google Scholar] [CrossRef]
- Vurukonda, S.S.K.P.; Vardharajula, S.; Shrivastava, M.; Skz, A. Enhancement of drought stress tolerance in crops by plant growth promoting rhizobacteria. Microbiol. Res. 2016, 184, 13–24. [Google Scholar] [CrossRef]
- Kaymak, H.C. Potential of PGPR in Agricultural Innovations. Plant Growth and Health Promoting Bacteria; Springer: Berlin/Heidelberg, Germany, 2010; pp. 45–79. [Google Scholar] [CrossRef]
- Timmusk, S.; Behers, L.; Muthoni, J.; Muraya, A.; Aronsson, A.C. Perspectives and challenges of microbial application for crop improvement. Front. Plant Sci. 2017, 8, 49. [Google Scholar] [CrossRef] [PubMed]
- Colla, G.; Rouphael, Y.; Bonini, P.; Cardarelli, M. Coating seeds with endophytic fungi enhances growth, nutrient uptake, yield and grain quality of winter wheat. Int. J. Plant Prod. 2015, 9, 171–190. [Google Scholar]
- Oliveira, R.S.; Rocha, I.; Ma, Y.; Vosátka, M.; Freitas, H. Seed coating with arbuscular mycorrhizal fungi as an ecotechnological approach for sustainable agricultural production of common wheat (Triticum aestivum L.). J. Toxicol. Environ. Health A 2016, 79, 329–337. [Google Scholar] [CrossRef] [PubMed]
- Rocha, I.; Ma, Y.; Carvalho, M.F.; Magalhães, C.; Janoušková, M.; Vosátka, M.; Freitasi, H.; Oliveira, R.S. Seed coating with inocula of arbuscular mycorrhizal fungi and plant growth promoting rhizobacteria for nutritional enhancement of maize under different fertilization regimes. Arch. Agron. Soil Sci. 2019, 65, 31–43. [Google Scholar] [CrossRef]
- Perelló, A.E.; Dal Bello, G.M. Suppression of tan spot and plant growth promotion of wheat by synthetic and biological inducers under field conditions. Ann. Appl. Biol. 2011, 158, 267–274. [Google Scholar] [CrossRef]
- Mahmood, Y.; Khan, M.A.; Javed, N.; Arif, M.J. Comparative efficacy of fungicides and biological control agents for the management of chickpea wilt caused by Fusarium oxysporum f. sp. ciceris. J. Anim. Plant. Sci. 2015, 25, 1063–1071. [Google Scholar]
- Shahzad, S.; Khan, M.Y.; Zahir, Z.A.; Asghar, H.N.; Chaudhry, U.K. Comparative effectiveness of different carriers to improve the efficacy of bacterial consortium for enhancing wheat production under salt affected field conditions. Pak. J. Bot 2017, 49, 1523–1530. [Google Scholar]
- Khan, N.; Mishra, A.; Chauhan, P.S.; Nautiyal, C.S. Induction of Paenibacillus lentimorbus biofilm by sodium alginate and CaCl2 alleviates drought stress in chickpea. Ann. Appl. Biol. 2011, 159, 372–386. [Google Scholar] [CrossRef]
- Harley, J.L.; Smith, S.E. Mycorrhizal symbiosis. In Experimental Agriculture; Academic Press: London, UK; New York, NY, USA, 1983; p. 483. [Google Scholar] [CrossRef]
- Sikes, B.A. When do arbuscular mycorrhizal fungi protect plant roots from pathogens? Plant Signal. Behav. 2010, 5, 763–765. [Google Scholar] [CrossRef]
- Bücking, H.; Kafle, A. Role of Arbuscular Mycorrhizal Fungi in the Nitrogen Uptake of Plants: Current Knowledge and Research Gaps. Agronomy 2015, 5, 587–612. [Google Scholar] [CrossRef]
- Oliveira, R.S.; Ma, Y.; Rocha, I.; Carvalho, M.F.; Vosátka, M.; Freitas, H. Arbuscular mycorrhizal fungi are an alternative to the application of chemical fertilizer in the production of the medicinal and aromatic plant Coriandrum sativum L. J. Toxicol. Environ. Health A 2016, 79, 320–328. [Google Scholar] [CrossRef] [PubMed]
- Celebi, S.Z.; Demir, S.; Celebi, R.; Durak, E.D.; Yilmaz, I.H. The effect of arbuscular mycorrhizal fungi (AMF) applications on the silage maize (Zea mays L.) yield in different irrigation regimes. Eur. J. Soil Biol. 2010, 46, 302–305. [Google Scholar] [CrossRef]
- Abdul Latef, A.A.H.A.; Chaoxing, H. Effect of arbuscular mycorrhizal fungi on growth, mineral nutrition, antioxidant enzymes activity and fruit yield of tomato grown under salinity stress. Sci. Hortic. 2011, 127, 228–233. [Google Scholar] [CrossRef]
- Alizadeh, O.; Zare, M.; Nasr, A.H. Evaluation effect of mycorrhiza inoculate under drought stress condition on grain yield of sorghum (Sorghum bicolor). Adv. Environ. Biol. 2011, 5, 2361–2364. [Google Scholar]
- Oliveira, R.S.; Carvalho, P.; Marques, G.; Ferreira, L.; Pereira, S.; Nunes, M.; Rocha, I.; Ma, Y.; Carvalho, M.F.; Vosátka, M.; et al. Improved grain yield of cowpea (Vigna unguiculata) under water deficit after inoculation with Bradyrhizobium elkanii and Rhizophagus irregularis. Crop. Pasture Sci. 2017, 68, 1052–1059. [Google Scholar] [CrossRef]
- Chun, S.C.; Chandrasekaran, M. Proline accumulation influenced by osmotic stress in arbuscular mycorrhizal symbiotic plants. Front. Microbiol 2018, 9, 2525. [Google Scholar] [CrossRef]
- Contreras-Cornejo, H.A.; Macías-Rodríguez, L.; del-Val, E.; Larsen, J. Ecological functions of trichoderma spp. and their secondary metabolites in the rhizosphere: Interactions with plants. FEMS Microbiol. Ecol. 2016, 92, fiw036. [Google Scholar] [CrossRef]
- Lutts, S.; Benincasa, P.; Wojtyla, L.; Kubala, S.S.; Pace, R.; Lechowska, K.; Quinet, M.; Garnczarska, M. Seed Priming: New Comprehensive Approaches for an Old Empirical Technique. In New Challenges in Seed Biology—Basic and Translational Research Driving Seed Technology; IntechOpen: London, UK, 2016. [Google Scholar] [CrossRef]
- Mahmood, A.; Turgay, O.C.; Farooq, M.; Hayat, R. Seed biopriming with plant growth promoting rhizobacteria: A review. FEMS Microbiol. Ecol. 2016, 92, fiw112. [Google Scholar] [CrossRef]
- Reddy, P.P. Bio-priming of seeds. In Recent Advances in Crop Protection; Reddy, P.P., Ed.; Springer: New Delhi, India, 2013; pp. 83–90. [Google Scholar] [CrossRef]
- Glick, B.R. Plant Growth-Promoting Bacteria: Mechanisms and Applications. Scientifica 2012, 2012, 963401. [Google Scholar] [CrossRef]
- Timmusk, S.; Abd El-Daim, I.A.; Copolovici, L.; Tanilas, T.; Kännaste, A.; Behers, L.; Nevo, E.; Seisenbaeva, G.; Stenström, E.; Niinemets, Ü. Drought-tolerance of wheat improved by rhizosphere bacteria from harsh environments: Enhanced biomass production and reduced emissions of stress volatiles. PLoS ONE 2014, 9, 96086. [Google Scholar] [CrossRef]
- Raj, N.S.; Shetty, N.P.; Shetty, H.S. Seed bio-priming with Pseudomonas fluorescens isolates enhances growth of pearl millet plants and induces resistance against downy mildew. Int. J. Pest Manag. 2004, 50, 41–48. [Google Scholar] [CrossRef]
- Suproniene, S.; Semaskiene, R.; Juodeikiene, G.; Mankeviciene, A.; Cizeikiene, D.; Vidmantiene, D.; Basinskiene, L.; Sakalauskas, S. Seed treatment with lactic acid bacteria against seed-borne pathogens of spring wheat. Biocont. Sci. Technol. 2015, 25, 144–154. [Google Scholar] [CrossRef]
- de Almeida Møller, C.O.; Freire, L.; Rosim, R.E.; Margalho, L.P.; Balthazar, C.F.; Franco, L.T.; Sant’Ana, A.S.; Corassin, C.H.; Rattray, F.P.; de Oliveira, C.A.F. Effect of Lactic Acid Bacteria Strains on the Growth and Aflatoxin Production Potential of Aspergillus parasiticus, and Their Ability to Bind Aflatoxin B1, Ochratoxin A, and Zearalenone in vitro. Front. Microbiol. 2021, 12, 655386. [Google Scholar] [CrossRef] [PubMed]
- Tinivella, F.; Hirata, L.M.; Celan, M.A.; Wright, S.A.I.; Amein, T.; Schmitt, A.; Koch, E.; van der Wolf, J.M.; Groot, S.P.C.; Stephan, D.; et al. Control of seed-borne pathogens on legumes by microbial and other alternative seed treatments. Eur. J. Plant Pathol. 2009, 123, 139–151. [Google Scholar] [CrossRef]
- Özcan, M.M.; Chalchat, J.-C. Chemical composition and antifungal activity of rosemary (Rosmarinus officinalis L.) oil from Turkey. Int. J. Food Sci. Nutr. 2008, 59, 691–698. [Google Scholar] [CrossRef]
- Fraternale, D.; Ricci, D. Chemical Composition and Antifungal Activity of the Essential Oil of Cotinus coggygria from Marche Region (Italy). Nat. Prod. Commun 2018, 13, 1175–1178. [Google Scholar] [CrossRef]
- Achimóna, F.; Brito, V.D.; Pizzolitto, R.P.; Ramirez Sanchez, A.; Gómez, E.A.; Zygadlo, J.A. Chemical composition and antifungal properties of commercial essential oils against the maize phytopathogenic fungus Fusarium verticillioides. Rev. Argent. Microbiol. 2021, 53, 292–303. [Google Scholar] [CrossRef]
- Marinelli, E.; Orzali, L.; Lotti, E.; Riccioni, L. Activity of Some Essential Oils against Pathogenic Seed Borne Fungi on Legumes. Asian J. Plant Pathol. 2012, 6, 66–74. [Google Scholar] [CrossRef][Green Version]
- Atak, M.; Mavi, K.; Uremis, I. Bio-Herbicidal Effects of Oregano and Rosemary Essential Oils on Germination and Seedling Growth of Bread Wheat Cultivars and Weeds. Rom. Biotechnol. Lett. 2016, 21, 11149–11159. [Google Scholar]
- Ben-Jabeur, M.; Vicente, R.; López-Cristoffanini, C.; Alesami, N.; Djébali, N.; Gracia-Romero, A.; Serret, M.D.; López-Carbonell, M.; Araus, J.L.; Hamada, W. A Novel Aspect of Essential Oils: Coating Seeds with Thyme Essential Oil induces Drought Resistance in Wheat. Plants 2019, 8, 371. [Google Scholar] [CrossRef]
- Velluti, A.; Sanchis, V.; Ramos, A.J.; Marin, S. Effect of essential oils of Cinnamon, Clove, Lemon Grass, Oregano, and Palmarosa on growth of and fumonisin B1 production by Fusarium verticillioides in maize. J. Sci. Food Agr. 2004, 84, 1141–1146. [Google Scholar] [CrossRef]
- Simic, A.; Sokovic, M.D.; Ristic, M.; Grujic-Jovanovic, S.; Vukojevic, J.; Marin, P.D. The chemical composition of some lauraceae essential oils and their antifungal activities. Phytother. Res. 2004, 18, 713–717. [Google Scholar] [CrossRef] [PubMed]
- Chatterjee, D. Inhibition of fungal growth and infection in maize grains by spice oils. Lett. Appl. Microbiol. 1990, 11, 148–151. [Google Scholar] [CrossRef]
- Tsitsigiannis, D.I.; Dimakopoulou, M.; Polymnia, P.A.; Tjamos, E.C. Biological control strategies of mycotoxigenic fungi and associated mycotoxins in mediterranean basis crops. Phytopathol. Mediterr. 2012, 51, 158–174. [Google Scholar] [CrossRef]
- Dorner, J.W. Biological control of aflatoxin contamination in corn using a non-toxigenic strain of Aspergillus flavus. J. Food Protect. 2009, 72, 801–804. [Google Scholar] [CrossRef]
- Pitt, J.; Manthong, C.; Siriacha, P.; Chotechaunmanirat, S.; Markwell, P. Studies on the biocontrol of aflatoxin in maize in Thailand. Biocontrol Sci. Technol. 2015, 25, 1070–1091. [Google Scholar] [CrossRef]
- Spadaro, D.; Gullino, M.L. State of the art and future prospects of biological control of postharvest fruit diseases. Int. J. Food Microbiol. 2004, 91, 185–194. [Google Scholar] [CrossRef]
- Fapohunda, S.O.; Esan, A.O.; Anjorin, T.S. Biological Control of Mycotoxins: An Update. World Vet. J. 2017, 7, 117–127. [Google Scholar]
- Garber, N.; Cotty, P.J. Timing of Herbicide Applications May Influence Efficacy of Aflatoxin Biocontrol. In Proceedings of the Beltwide Cotton Conferences, San Antonio, TX, USA, 3–6 January 2006; 2006; p. 11. [Google Scholar]
- Abbas, H.K.; Accinelli, C.; Shier, W.T. Biological Control of Aflatoxin Contamination in U.S. Crops and the Use of Bioplastic Formulations of Aspergillus flavus Biocontrol Strains to Optimize Application Strategies. J. Agric. Food Chem. 2017, 65, 7081–7087. [Google Scholar] [CrossRef]
- Liu, Y.; Galani Yamdeu, J.H.; Gong, Y.Y.; Orfila, C. A review of postharvest approaches to reduce fungal and mycotoxin contamination of foods. Compr. Rev. Food Sci. Food Saf. 2020, 19, 1521–1560. [Google Scholar] [CrossRef]
- Weaver, M.A.; Abbas, H.K. Field Displacement of Aflatoxigenic Aspergillus flavus Strains Through Repeated Biological Control Applications. Front. Microbiol. 2019, 10, 1788. [Google Scholar] [CrossRef] [PubMed]
- Bandyopadhyay, R.; Cotty, P.J. Biological controls for aflatoxin reduction. In Aflatoxins—Finding Solutions for Improved Food Safety; Unnevehr, L., Grace, D., Eds.; International Food Policy Research Institute: Washington, DC, USA, 2013; pp. 16–17. [Google Scholar]
- Atehnkeng, J.; Ojiambo, P.S.; Cotty, P.J.; Bandyopadhyay, R. Field efficacy of a mixture of atoxigenic Aspergillus flavus Link: Fr vegetative compatibility groups in preventing aflatoxin contamination in maize (Zea mays L.). Biol. Control 2014, 72, 62–70. [Google Scholar] [CrossRef]
- Alaniz Zanon, M.S.; Barros, G.G.; Chulze, S.N. Non-aflatoxigenic Aspergillus flavus as potential biocontrol agents to reduce aflatoxin contamination in peanuts harvested in northern Argentina. Int. J. Food Microbiol. 2016, 231, 63–68. [Google Scholar] [CrossRef] [PubMed]
- Mallikarjunaiah, N.H.; Jayapala, N.; Puttaswamy, H.; Ramachandrappa, N.S. Characterization of non-aflatoxigenic strains of Aspergillus flavus as potential biocontrol agent for the management of aflatoxin contamination in groundnut. Microbiol. Pathog. 2016, 102, 21–28. [Google Scholar] [CrossRef]
- Shifa, H.; Tasneem, S.; Gopalakrishnan, C.; Velazhahan, R. Biological control of pre-harvest aflatoxin contamination in groundnut (Arachis hypogaea L.) with Bacillus subtilis G1. Arch. Phytopathol. Plant Prot. 2016, 49, 137–148. [Google Scholar] [CrossRef]
- Pereira, P.; Nesci, A.; Castillo, C.; Etcheverry, M. Impact of bacterial biological control agents on fumonisin B1 content and Fusarium verticillioides infection of field-grown maize. Biol. Control 2010, 53, 258–266. [Google Scholar] [CrossRef]
- Sartori, M.; Nesci, A.; Castillo, C.; Etcheverry, M. Biological control of fumonisins production in maize at field level. Int. J. Agric. Policy Res. 2013, 1, 188–196. [Google Scholar]
- Alberts, J.F.; van Zyl, W.H.; Gelderblom, W.C.A. Biologically based methods for control of fumonisin-producing Fusarium species and reduction of the fumonisins. Front. Microbiol. 2016, 7, 548. [Google Scholar] [CrossRef]
- De Souza, M.L.; Passamani, F.R.F.; da Silva, Á.C.L.; Batista, L.R.; Schwan, R.F.; Silva, C.F. Use of wild yeasts as a biocontrol agent against toxigenic fungi and OTA production. Acta Scientiarum 2017, 39, 349–358. [Google Scholar] [CrossRef]
- Ponsone, M.L.; Chiotta, M.L.; Combina, M.; Chulze, S. Biocontrol as a strategy to reduce the impact of ochratoxin A and Aspergillus section Nigri in grapes. Int. J. Food Microbiol. 2011, 151, 70–77. [Google Scholar] [CrossRef]
- Vekiru, E.; Hametner, C.; Mitterbauer, R.; Rechthaler, J.; Adam, G.; Schatzmayr, G.; Krska, R.; Schuhmacher, R. Cleavage of zearalenone by Trichosporon mycotoxinivorans to a novel nonestrogenic metabolite. Appl. Environ. Microb. 2010, 76, 2353–2359. [Google Scholar] [CrossRef] [PubMed]
- Brodehl, A.; Moller, A.; Kunte, H.-J.; Koch, M.; Maul, R. Biotransformation of the mycotoxin zearalenone by fungi of the genera Rhizopus and Aspergillus. FEMS Microbiol. Lett. 2014, 359, 124–130. [Google Scholar] [CrossRef] [PubMed]
- Kosawang, C.; Karlsson, M.; Jensen, B.; Vélëz, H.; Rasmussen, P.H.; Collinge, D.B.; Jensen, D.F. Detoxification of the Fusarium mycotoxin zearalenone is an important trait of Clonostachys rosea in biocontrol of Fusarium foot rot of barley. In Working Group―Biological Control of Fungal and Bacterial Plant Pathogens, Proceedings of the Meeting at Reims; Pertot, I., Elad, Y., Barka, E.A., Clemente, C., Eds.; DTU Orbit: Reims, France, 2013; pp. 133–136. [Google Scholar]
- Rose, L.J.; Okoth, S.; Flett, B.C.; van Rensburg, B.J.; Viljoen, A. Preharvest Management Strategies and Their Impact on Mycotoxigenic Fungi and Associated Mycotoxins. In Mycotoxins—Impact and Management Strategies; Berka Njobeh, P., Stepman, F., Eds.; IntechOpen: London, UK, 2018. [Google Scholar] [CrossRef]
- Sarrocco, S.; Vannacci, G. Preharvest application of beneficial fungi as a strategy to prevent postharvest mycotoxin contamination: A review. Crop Prot. 2018, 110, 160–170. [Google Scholar] [CrossRef]
- Sarrocco, S.; Mauro, A.; Battilani, P. Use of Competitive Filamentous Fungi as an Alternative Approach for Mycotoxin Risk Reduction in Staple Cereals: State of Art and Future Perspectives. Toxins 2019, 11, 701. [Google Scholar] [CrossRef] [PubMed]
- Nešić, K.; Habschied, K.; Mastanjević, K. Possibilities for the Biological Control of Mycotoxins in Food and Feed. Toxins 2021, 13, 198. [Google Scholar] [CrossRef] [PubMed]
- Peles, F.; Sipos, P.; Kovács, S.; Győri, Z.; Pócsi, I.; Pusztahelyi, T. Biological Control and Mitigation of Aflatoxin Contamination in Commodities. Toxins 2021, 13, 104. [Google Scholar] [CrossRef]
- Cheli, F.; Pinotti, L.; Rossi, L.; Dell’Orto, V. Effect of milling procedures on mycotoxin distribution in wheat fractions: A review. LWT-Food Sci. Technol. 2013, 54, 307–314. [Google Scholar] [CrossRef]
- Janić, H.E.; Mastilović, J.; Bagi, F.; Orčić, D.; Budakov, D.; Kos, J.; Savić, Z. Effect of Wheat Milling Process on the Distribution of Alternaria Toxins. Toxins 2019, 11, 139. [Google Scholar] [CrossRef]
- Pleadin, J.; Perši, N.; Kovačević, D.; Vulić, A.; Frece, J.; Markov, K. Ochratoxin A reduction in meat sausages using processing methods practiced in households. Food Addit. Contam. B 2014, 7, 239–246. [Google Scholar] [CrossRef]
- Pleadin, J.; Markov, K.; Frece, J.; Vulić, A.; Perši, N. Bio-Prevalence, Determination and Reduction of Aflatoxin B1 in Cereals. In Aflatoxins: Food Sources, Occurrence and Toxicological Effects; Faulkner, A.G., Ed.; Nova Science Publishers: Hauppauge, NY, USA, 2014; pp. 1–34. [Google Scholar]
- Delgado, J.; Acosta, R.; Rodríguez-Martín, A.; Bermúdez, E.; Núñez, F.; Asensio, M.A. Growth inhibition and stability of PgAFP from Penicillium chrysogenum against fungi common on dry-ripened meat products. Int. J. Food Microbiol. 2015, 205, 23–29. [Google Scholar] [CrossRef]
- Delgado, J.; Owens, R.A.; Doyle, S.; Asensio, M.A.; Nunez, F. Antifungal proteins from moulds: Analytical tools and potential application to dry-ripened foods. Appl. Microbiol. Biot. 2016, 100, 6991–7000. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B.B.; Xie, C.J.; Yang, X.Y. A novel small antifungal peptide from Bacillus strain B-TL2 isolated from tobacco stems. Peptides 2008, 29, 350–355. [Google Scholar] [CrossRef]
- Devi, M.S.; Sashidhar, R.B. Antiaflatoxigenic effects of selected antifungal peptides. Peptides 2019, 115, 15–26. [Google Scholar] [CrossRef] [PubMed]
- Luz, C.; Saladino, F.; Luciano, F.B.; Mañes, J.; Meca, G. In vitro antifungal activity of bioactive peptides produced by Lactobacillus plantarum against Aspergillus parasiticus and Penicillium expansum. LWT-Food Sci. Technol. 2017, 81, 128–135. [Google Scholar] [CrossRef]
- Chen, Y.; Kong, Q.; Liang, Y. Three newly identified peptides from Bacillus megaterium strongly inhibit the growth and aflatoxin B1 production of Aspergillus flavus. Food Control 2019, 95, 41–49. [Google Scholar] [CrossRef]
- Dalié, D.; Deschamps, A.; Richard-Forget, F. Lactic acid bacteria–Potential for control of mould growth and mycotoxins: A review. Food Control 2010, 21, 370–380. [Google Scholar] [CrossRef]
- Gerez, C.L.; Torino, M.I.; Rollan, G.; Font de Valdez, G. Prevention of bread mould spoilage by using lactic acid bacteria with antifungal properties. Food Control 2009, 20, 144–148. [Google Scholar] [CrossRef]
- Romanens, E.; Freimuller Leischtfeld, S.; Volland, A.; Stevens, M.J.A.; Krahenmann, U.; Isele, D.; Volland, A.; Fischer, B.; Meile, L.; Schwenninger Miescher, S. Screening of lactic acid bacteria and yeast strains to select adapted anti-fungal co-cultures for cocoa bean fermentation. Int. J. Food Microbiol. 2019, 290, 262–272. [Google Scholar] [CrossRef] [PubMed]
- Hassan, Y.I.; Zhou, T.; Bullerman, L.B. Sourdough lactic acid bacteria as antifungal and mycotoxin-controlling agents. Food Sci. Technol. Int. 2015, 22, 79–90. [Google Scholar] [CrossRef]
- Bourdichon, F.; Casaregola, S.; Farrokh, C.; Frisvad, J.C.; Gerds, M.L.; Hammes, W.P.; Harnett, J.; Huys, G.; Laulund, S.; Ouwehand, A.; et al. Food fermentations: Microorganisms with technological beneficial use. Int. J. Food Microbiol. 2012, 154, 87–97. [Google Scholar] [CrossRef]
- Prakash, B.; Kedia, A.; Mishra, P.K.; Dubey, N.K. Plant essential oils as food preservatives to control moulds, mycotoxin contamination and oxidative deterioration of agri-food commodities—Potentials and challenges. Food Control 2015, 47, 381–391. [Google Scholar] [CrossRef]
- Bakkali, F.; Averbeck, S.; Averbeck, D.; Idaomar, M. Biological effects of essential oils: A review. Food Chem. Toxicol. 2008, 46, 446–475. [Google Scholar] [CrossRef] [PubMed]
- Zuzarte, M.; Vale-Silva, L.; Gonçalves, M.J.; Cavaleiro, C.; Vaz, S.; Canhoto, J.; Pinto, E.; Salgueiro, L. Antifungal activity of phenolic-rich Lavandula multifida L. essential oil. Eur. J. Clin. Microbiol. Infect. Dis. 2012, 31, 1359–1366. [Google Scholar] [CrossRef] [PubMed]
- Prakash, B.; Shukla, R.; Singh, P.; Mishra, P.K.; Dubey, N.K.; Kharwar, R.N. Efficacy of chemically characterized Ocimum gratissimum L. essential oil as an antioxidant and a safe plant based antimicrobial against fungal and aflatoxin B1 contamination of spices. Food Res. Int. 2011, 44, 385–390. [Google Scholar] [CrossRef]
- Prakash, B.; Singh, P.; Kedia, A.; Dubey, N.K. Assessment of some essential oils as food preservatives based on antifungal antiaflatoxin, antioxidant activities and in vivo efficacy in food system. Food Res. Int. 2012, 49, 201–208. [Google Scholar] [CrossRef]
- Perczak, A.; Gwiazdowska, D.; Marchwińska, K.; Juś, K.; Gwiazdowski, R.; Waśkiewicz, A. Antifungal activity of selected essential oils against Fusarium culmorum and F. graminearum and their secondary metabolites in wheat seeds. Arch. Microbiol. 2019, 201, 1085–1097. [Google Scholar] [CrossRef] [PubMed]
- Perczak, A.; Gwiazdowska, D.; Gwiazdowski, R.; Juś, K.; Marchwińska, K.; Waśkiewicz, A. The Inhibitory Potential of Selected Essential Oils on Fusarium spp. Growth and Mycotoxins Biosynthesis in Maize Seeds. Pathogens 2020, 9, 23. [Google Scholar] [CrossRef] [PubMed]
- Park, S.C.; Cheong, M.S.; Kim, E.J.; Kim, J.H.; Chi, Y.H.; Jang, M.K. Antifungal effect of Arabidopsis SGT1 proteins via mitochondrial reactive oxygen species. J. Agric. Food Chem. 2017, 65, 8340–8347. [Google Scholar] [CrossRef]
- Subramanyam, K.; Arun, M.; Mariashibu, T.S.; Theboral, J.; Rajesh, M.; Singh, N.K.; Manickavasagam, M.; Ganapathi, A. Overexpression of tobacco osmotin (Tbosm) in soybean conferred resistance to salinity stress and fungal infections. Planta 2012, 236, 1909–1925. [Google Scholar] [CrossRef]
- Yan, J.; Yuan, S.S.; Jiang, L.L.; Ye, X.J.; Ng, T.B.; Wu, Z.J. Plant antifungal proteins and their applications in agriculture. Appl. Microbiol. Biot. 2015, 99, 4961–4981. [Google Scholar] [CrossRef]
- Chan, Y.S.; Ng, T.B. Northeast red beans produce a thermostable and pH-stable defensin-like peptide with potent antifungal activity. Cell Biochem. Biophys. 2013, 66, 637–648. [Google Scholar] [CrossRef]
- Paiva, A.L.; Vasconcelos, I.M.; Oliveira, J.T.A. Purification, characterization and antifungal activity of LPA a lectin from Pachira aquatica seeds: Their possible role in plant defence. BMC Proc. 2014, 8, 134. [Google Scholar] [CrossRef]
- Bouarab-Chibane, L.; Forquet, V.; Lantéri, P.; Clément, Y.; Léonard-Akkari, L.; Oulahal, N.; Degraeve, P.; Bordes, C. Antibacterial Properties of Polyphenols: Characterization and QSAR (Quantitative Structure-Activity Relationship) Models. Front. Microbiol. 2019, 10, 829. [Google Scholar] [CrossRef] [PubMed]
- Ilboudo, O.; Bonzi, S.; Tapsoba, I.; Somda, I.; Bonzi-Coulibaly, Y.L. In vitro antifungal activity of flavonoid diglycosides of Mentha piperita and their oxime derivatives against two cereals fungi. Comptes Rendus Chimie 2016, 19, 857–862. [Google Scholar] [CrossRef]
- Diaz-Gomez, J.; Marin, S.; Nogareda, C.; Sanchis, V.; Ramos, A.J. The effect of enhanced carotenoid content of transgenic maize grain on fungal colonization and mycotoxin content. Mycotoxin Res. 2016, 32, 221–228. [Google Scholar] [CrossRef] [PubMed]
- Suwarno, W.B.; Hannok, P.; Palacios-Rojas, N.; Windham, G.; Crossa, J.; Pixley, K.V. Provitamin A carotenoids in grain reduce aflatoxin contamination of maize while combating vitamin A deficiency. Front. Plant Sci. 2019, 10, 132. [Google Scholar] [CrossRef]
- Atanasova-Penichon, V.; Barreau, C.; Richard-Forget, F. Antioxidant secondary metabolites in cereals: Potential involvement in resistance to Fusarium and mycotoxin accumulation. Front. Microbiol. 2016, 7, 566. [Google Scholar] [CrossRef] [PubMed]
- Akabassi, G.C.; Padonou, E.A.; Yao, E.J.K.; Nakpalo, S.; Palanga, K.K.; Eliaet, B. Extract yield, dilution methods and antifungal potential of fruits of Picralima nitida (Stapf.) T. A. Durand & H. Durand. J. Saudi Soc. Agric. Sci. 2021, in press. [Google Scholar] [CrossRef]
- Hathout, S.A.; Soher, E.A. Biological detoxification of mycotoxins: A review. Ann. Microbiol. 2014, 64, 905–919. [Google Scholar] [CrossRef]
- Markov, K.; Frece, J.; Pleadin, J.; Bevardi, M.; Barišić, L.; Gajdoš Kljusurić, J.; Vulić, A.; Jakopović, Ž.; Mrvčić, J. Gluconobacter oxydans—Potential biological agent for binding or biotransformation of mycotoxins. World Mycotoxin J. 2019, 12, 153–161. [Google Scholar] [CrossRef]
- Muhialdin, B.J.; Saari, N.; Meor Hussin, A.S. Review on the biological detoxification of mycotoxins using lactic acid bacteria to enhance the sustainability of foods supply. Molecules 2020, 25, 2655. [Google Scholar] [CrossRef] [PubMed]
- Guan, S.; Ji, C.; Zhou, T.; Li, J.; Ma, Q.; Niu, T. Aflatoxin B1 degradation by Stenotrophomonas maltophilia and other microbes selected using coumarin medium. Int. J. Mol. Sci. 2008, 9, 1489–1503. [Google Scholar] [CrossRef] [PubMed]
- Sayed, Y.R. Aflatoxin detoxification by manganese peroxidase purified from Pleurotus ostreatus. Braz. J. Microbiol. 2014, 45, 127–133. [Google Scholar]
- Ji, C.; Fan, Y.; Zhao, L. Review on biological degradation of mycotoxins. Anim. Nutr. 2016, 2, 127–133. [Google Scholar] [CrossRef]
- Alberts, J.F.; Gelderblom, W.C.; Botha, A.; van Zyl, W.H. Degradation of aflatoxin B(1) by fungal laccase enzymes. Int. J. Food Microbiol. 2009, 135, 47–52. [Google Scholar] [CrossRef]
- Zhang, Q.; Xiong, K.; Tatsumi, E.; Li, L.T.; Liu, H.J. Elimination of aflatoxin B1 in peanuts by acidic electrolyzed oxidizing water. Food Control 2012, 27, 16–20. [Google Scholar] [CrossRef]
- Sato, I.; Ito, M.; Ishizaka, M.; Ikunaga, Y.; Sato, Y.; Yoshida, S.; Koitabashi, M.; Tsushima, S. Thirteen novel deoxynivalenol-degrading bacteria are classified within two genera with distinct degradation mechanisms. FEMS Microbiol. Lett. 2012, 327, 110–117. [Google Scholar] [CrossRef] [PubMed]
- Smiley, R.D.; Draughon, F.A. Preliminary evidence that degradation of aflatoxin B1 by Flavobacterium aurantiacum is enzymatic. J. Food Prot. 2000, 63, 415–418. [Google Scholar] [CrossRef]
- Sangare, L.; Zhao, Y.; Folly, Y.M.; Chang, J.; Li, J.; Selvaraj, J.N.; Xing, F.; Zhou, L.; Wang, Y.; Liu, Y. Aflatoxin B₁ degradation by a Pseudomonas strain. Toxins 2014, 6, 3028–3040. [Google Scholar] [CrossRef] [PubMed]
- Farzaneh, M.; Shi, Z.Q.; Ghassempour, A.; Sedaghat, N.; Ahmadzadeh, M.; Mirabolfathy, M.; Javan-Nikkhah, M. Aflatoxin B1 degradation by Bacillus subtilis UTBSP1 isolated from pistachio nuts of Iran. Food Control 2012, 23, 100–106. [Google Scholar] [CrossRef]
- Schatzmayr, G.; Streit, E. Global occurrence of mycotoxins in the food and feed chain: Facts and toxins. World Mycotoxin J. 2013, 6, 213–222. [Google Scholar] [CrossRef]
- Ahad, R.; Zhou, T.; Lepp, D.; Pauls, K.P. Microbial detoxification of eleven food and feed contaminating trichothecene mycotoxins. BMC Biotechnol. 2017, 17, 30. [Google Scholar] [CrossRef] [PubMed]
- Jard, G.; Liboz, T.; Mathieu, F.; Guyonvarc’h, A.; Lebrihi, A. Review of mycotoxin reduction in food and feed: From prevention in the field to detoxification by adsorption or transformation. Food Addit. Contam. 2011, 28, 1590–1609. [Google Scholar] [CrossRef] [PubMed]
- He, J.W.; Bondy, G.S.; Zhou, T.; Caldwell, D.; Boland, G.J.; Scott, P.M. Toxicology of 3-epi-deoxynivalenol, a deoxynivalenol-transformation product by Devosia mutans 17–2-E-8. Food Chem. Toxicol. 2015, 84, 250–259. [Google Scholar] [CrossRef]
- Schatzmayr, G.; Zehner, F.; Täubel, M.; Schatzmayr, D.; Klimitsch, A.; Loibner, A.P.; Binder, E.M. Microbiologicals for deactivating mycotoxins. Mol. Nutr. Food Res. 2006, 50, 543–551. [Google Scholar] [CrossRef]
- Foroud, N.A.; Baines, D.; Gagkaeva, T.Y.; Thakor, N.; Badea, A.; Steiner, B.; Bürstmayr, M.; Bürstmayr, H. Trichothecenes in Cereal Grains—An Update. Toxins 2019, 11, 634. [Google Scholar] [CrossRef]
- Hassan, Y.I.; Zhou, T. Addressing the mycotoxin deoxynivalenol contamination with soil-derived bacterial and enzymatic transformations targeting the C3 carbon. World Mycotoxin J. 2018, 11, 101–112. [Google Scholar] [CrossRef]
- Gao, X.; Mu, P.; Wen, J.; Sun, Y.; Chen, Q.; Deng, Y. Detoxification of trichothecene mycotoxins by a novel bacterium, Eggerthella sp. DII-9. Food Chem. Toxicol. 2018, 112, 310–319. [Google Scholar] [CrossRef]
- Luo, Y.; Liu, X.; Li, J. Updating techniques on controlling mycotoxins—A review. Food Control 2018, 89, 123–132. [Google Scholar] [CrossRef]
- Zohri, A.A.; Abdel-Kareem, M.M. Four strains of yeasts: As effective biocontrol agents against both growth and mycotoxins formation by selected 11 toxigenic fungi. Glob. Adv. Res. J. Microbiol. 2018, 7, 132–135. [Google Scholar]
- Liu, Y.; Chang, J.; Wang, P.; Yin, Q.; Huang, W.; Liu, C.; Bai, X.; Zhu, Q.; Gao, T.; Zhou, P. Effects of Saccharomyces cerevisiae on alleviating cytotoxicity of porcine jejunal epithelia cells induced by deoxynivalenol. AMB Express 2019, 9, 137. [Google Scholar] [CrossRef] [PubMed]
- Jakopović, Ž.; Čiča, K.H.; Mrvčić, J.; Pucić, I.; Čanak, I.; Frece, J.; Pleadin, J.; Stanzer, D.; Zjalic, S.; Markov, K. Properties and Fermentation Activity of Industrial Yeasts Saccharomyces cerevisiae, S. uvarum, Candida utilis and Kluyveromyces marxianus Exposed to AFB1, OTA and ZEA. Food Technol. Biotechnol. 2018, 56, 208–217. [Google Scholar] [CrossRef]
- Yang, Q.; Wang, J.; Zhang, H.; Li, C.; Zhang, X. Ochratoxin A is degraded by Yarrowia lipolytica and generates non-toxic degradation products. World Mycotoxin J. 2016, 9, 269–278. [Google Scholar] [CrossRef]
- Hackbart, H.C.S.; Machado, A.R.; Christ-Ribeiro, A.; Prietto, L.; Badiale-Furlong, E. Reduction of aflatoxins by Rhizopus oryzae and Trichoderma reesei. Mycotoxin Res. 2014, 30, 141–149. [Google Scholar] [CrossRef] [PubMed]
- Utermark, J.; Karlovsky, P. Role of Zearalenone Lactonase in Protection of Gliocladium roseum from Fungitoxic Effects of the Mycotoxin Zearalenone. Appl. Environ. Microbiol. 2006, 73, 637–642. [Google Scholar] [CrossRef] [PubMed]
- Wu, Q.; Jezkova, A.; Yuan, Z.; Pavlikova, L.; Dohnal, V.; Kuca, K. Biological degradation of aflatoxins. Drug Metab. Rev. 2009, 41, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Nichea, M.J.; Palacios, S.A.; Chiacchiera, S.M.; Sulyok, M.; Krska, R.; Chulze, S.N.; Torres, A.M.; Ramirez, M.L. Presence of Multiple Mycotoxins and Other Fungal Metabolites in Native Grasses from a Wetland Ecosystem in Argentina Intended for Grazing Cattle. Toxins 2015, 7, 3309–3329. [Google Scholar] [CrossRef] [PubMed]
- Gerez, C.L.; Cuezzo, S.; Rollán, G.; Font de Valdez, G. Lactobacillus reuteri CRL 1100 as starter culture for wheat dough fermentation. Food Microbiol. 2008, 25, 253–259. [Google Scholar] [CrossRef]
- Abrunhosa, L.; Paterson, R.R.M.; Venancio, A. Biodegradation of ochratoxin A for food and Feed Decontamination. Toxins 2010, 2, 1078–1099. [Google Scholar] [CrossRef] [PubMed]
- Juodeikiene, G.; Salomskiene, J.; Eidukonyte, D.; Vidmantiene, D.; Narbutaite, V.; Vaiciulyte-Funk, L. The impact of novel fermented products containing extruded wheat material on the quality of wheat bread. Food Technol. Biotechnol. 2011, 49, 502–510. [Google Scholar]
- Chapot-Chartier, M.P.; Kulakauskas, S. Cell wall structure and function in lactic acid bacteria. Microb. Cell Factories 2014, 13, S9. [Google Scholar] [CrossRef] [PubMed]
- Luz, C.; Ferrer, J.; Mañes, J.; Meca, G. Toxicity reduction of ochratoxin A by lactic acid bacteria. Food Chem. Toxicol. 2018, 112, 60–66. [Google Scholar] [CrossRef] [PubMed]
- Perczak, A.; Goliński, P.; Bryła, M.; Waśkiewicz, A. The efficiency of lactic acid bacteria against pathogenic fungi and mycotoxins. Arh. Hig Rada Toksikol. 2018, 69, 32–45. [Google Scholar] [CrossRef] [PubMed]
- El-Nezami, H.; Polychronaki, N.; Salminen, S.; Mykkanen, H. Binding rather than metabolism may explain the interaction of two food-grade Lactobacillus strains with zearalenone and its derivative ά-zearalenol. Appl. Environ. Microb. 2002, 68, 3545–3549. [Google Scholar] [CrossRef] [PubMed]
- Turbic, A.; Ahokas, J.T.; Haskard, C.A. Selective in vitro binding of dietary mutagens, individually or in combination, by lactic acid bacteria. Food Addit. Contam. 2002, 19, 144–152. [Google Scholar] [CrossRef] [PubMed]
- Vosough, P.R.; Sani, A.M.; Mehraban, M.; Karazhyan, R. In vitro effect of Lactobacillus rhamnosus GG on reduction of aflatoxin B1. Nutr. Food Sci. 2014, 44, 32–40. [Google Scholar] [CrossRef]
- Kuharić, Ž.; Jakopović, Ž.; Čanak, I.; Frece, J.; Bošnir, J.; Ivešić, M.; Markov, K. Removing aflatoxin M1 from milk with native lactic acid bacteria, centrifugation, and filtration. Arh. Hig. Rada Toksikol. 2018, 69, 334–339. [Google Scholar] [CrossRef]
- Sreekumar, O.; Hosono, A. The heterocyclic amine binding receptors of Lactobacillus gasseri cells. Mutat Res. 1998, 421, 65–72. [Google Scholar] [CrossRef]
- Zhao Zhou, F.; Qi, Y.; Dziugan, P.; Bai, F.; Walczak, P.; Zhang, B. Screening of Lactobacillus strains for their ability to bind benzo(a)pyrene and the mechanism of the process. Food Chem. Toxicol. 2013, 59, 67–71. [Google Scholar] [CrossRef]
- Zou, Z.-Y.; He, Z.-F.; Li, H.-J.; Han, P.-F.; Meng, X.; Zhang, Y.; Zhou, F.; Ouyang, K.-P.; Chen, X.-Y.; Tang, J. In vitro removal of deoxynivalenol and T-2 toxin by lactic acid bacteria. Food Sci. Biotechechnol. 2012, 21, 1677–1683. [Google Scholar] [CrossRef]
- Jakopović, Z.; Mrvčić, J.; Pleadin, J.; Barišić, L.; Frece, J.; Čanak, I.; Markov, K. FT-IR spectroscopy as a method to detected cell surface components of Gluconobacter oxydans involved in the bacteria–OTA interaction. J. Biotechnol. 2017, 256, S66. [Google Scholar] [CrossRef]
- Haskard, C.; Binnion, C.; Ahokas, J. Factors affecting the sequestration of aflatoxin by Lactobacillus rhamnosus strain GG. Chem.-Biol. Interact. 2000, 128, 39–49. [Google Scholar] [CrossRef]
- Petruzzi, L.; Sinigaglia, M.; Corbo, M.R.; Beneduce, L.; Bevilacqua, A. Ochratoxin A removal by Saccharomyces cerevisiae strains: Effect of wine-related physicochemical factors. J. Sci. Food Agric. 2012, 93, 2110–2115. [Google Scholar] [CrossRef]
- Caridi, A.; Galvano, F.; Tafuri, A.; Ritieni, A. Ochratoxin A removal during winemaking. Enzyme Microb Technol. 2006, 40, 122–126. [Google Scholar] [CrossRef]
- Fernández Juri, M.G.; Dalcero, A.M.; Magnoli, C.E. In vitro aflatoxin B1 binding capacity by two Enterococcus faecium strains isolated from healthy dog faeces. J. Appl. Microbiol. 2015, 118, 574–582. [Google Scholar] [CrossRef] [PubMed]
- Assaf, J.C.; El Khoury, A.; Atoui, A.; Louka, N.; Chokr, A. A novel technique for aflatoxinM1 detoxification using chitin or treated shrimp shells: In vitro effect of physical and kinetic parameters on the binding stability. Appl. Microbiol. Biot 2018, 102, 6687–6697. [Google Scholar] [CrossRef] [PubMed]


| Mycotoxin | Commodity | Main Producers | Toxic Effects | Reference | |
|---|---|---|---|---|---|
| Aflatoxins (AFs) | B1, B2 G1, G2 M1, M2 | Wheat Maize | A. flavus, A. parasiticus, A. nominus, A. pseudotamarii | Carcinogenic, liver cancer, immune suppressive | [24] |
| Ochratoxins | A, B | Wheat Barley Maize | A. ochraceus, A. carbonarius, P. verrucosum | Neurotoxic, nephrotoxic, kidney damage, and cancer; immune suppressive | [25,26] |
| Type B Trichothecenes | DON (Vomitoxin) ADON, NIV, ANIV | Wheat Barley Maize | F. graminearum, F. culmorum, F. acuminatum, F. crookwellense, F. avenaceum, F. equiseti | ATA (alimentary toxic leukopenia), immunotoxic, acute toxicity | [27] |
| Type A Trichothecenes | T-2 HT-2 | Wheat Barley Oats Maize Ray | F. poae, F. sporotrichioides | Acute toxicity, linked to ATA, immunotoxic, immune system and hematological disorders | [28] |
| Zearalenone (ZEA) | Wheat Maize | F. graminearum, F. culmorum, F. cerealis, F. avenaceum, F. equiseti | Estrogenic effects, reproductive disorders, affects endocrine system | [29] | |
| Fumonisin (FUM) | FB1 FB2 | Wheat Barley Oat Maize | F. verticillioides, F. proliferatum | Esophageal cancer, sphingolipid metabolism disruption, immune suppression | [30] |
| Ergot alkaloids | Ergotamine, ergometrine, ergosine, ergocristine, ergocryptine, ergocornine | Wheat Ray Barley Maize | Claviceps purpurea, Claviceps fusiformis | Necrosis of limbs—St Anthony‘s fire, vasoconstrictive properties, gangrenous and convulsive ergotism | [5] |
| Alternaria mycotoxins | ALT, AS, ALN, TeA, ALS, ATXs, alterperylenol or alteichin, ALTCH, STE | Wheat Oat Barley Maize | A. alternata, A. tenuissima, A. arborescens, A.radicina, A. brassicae, A. brassicicola, and A. infectoria. | cytotoxicity, fetotoxicity, and teratogenicity, hematological disorders, esophageal cancer, mutagenic, clastogenic, and estrogenic in microbial and mammalian cell systems and tumorigenic in rats | [6] |
| Strategy | Gene/Construct | Cereal | Pathogen | Phenotype | Reference |
|---|---|---|---|---|---|
| Host-induced gene silencing (HIGS) | aflC aflatoxin biosynthetic gene | Maize (Zea Mays) | A flavus | Reduced aflatoxin levels | [47] |
| aflR aflatoxin biosynthetic gene | Maize (Zea Mays) | A flavus | Reduced aflatoxin levels | [48] | |
| Cytochrome P450 lanosterol C-14α- demethylases using a double and inverted CaMV 35S promoter | Barley (Hordeum vulgare) | F. graminearum | Strong resistence | [49] | |
| Chitin synthase(Chs)3b | Spring wheat (Triticum aestivum L.) | F graminearum | Reduction in DON accumulation | [50] | |
| Nonspecific lipid transfer protein (nsLTP) | AtLTP4.4 using maize ubiquitin promoter | Spring wheat (T. aestivum) | F. graminearum (FHB) | Reduction in DON accumulation | [51] |
| Antimicrobial protein (chitinase) | Class II chitinase using maize ubiquitin promoter | Spring wheat (T. aestivum) cv. Bobwhite | Fusarium graminearum (FHB) | Reduced infection and deoxynivalenol (DON) levels | [52] |
| Class I chitinase (McCHIT1) using maize ubiquitin promoter | Rice (Oryza. sativa var. indica) | Magnaporthe grisea and Rhizoctonia solani | Reduced infection and DON levels | [53] |
| Microogranisms | Mixotoxin | Reference |
|---|---|---|
| Bacteriae | ||
| Flavobacterium aurantiacum | AFB, OTA, ZEA | [181] |
| Phenylobacterium immobile | ||
| Gliocladium roseum | ||
| Eubacterium BBSH 797 | DON | [182] |
| Devosia genus | DON | [183] |
| Eggerthella spp. DII-9 | DON, T-2, HT-2 | [184] |
| Bacillus | AFB, ZEA, DON, OTA | [185] |
| Brevibacterium | ||
| Yeasts | ||
| Candida krusei AUMC 8161 | AFB, OTA, ZEN, alternariol | [186] |
| Pichia anomala AUMC 2674 | ||
| Pichia guilliermondii AUMC 2663 | ||
| Saccharomyces cerevisiae AUMC 3875 | ||
| Saccharomyces cerevisiae | DON | [187] |
| Candida utilis | AFB, OTA, ZEA | [188] |
| Yarrowia lipolytica | OTA | [189] |
| Fungi | ||
| Rhizopus oryzae | AFB1, AFB2, AFG1, AFG2 | [122] |
| Trichoderma reesei | ||
| Clonostachys rosea | ZEA | [190] |
| Trichosporon mycotoxinivorans | ZEA | [191] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zadravec, M.; Markov, K.; Lešić, T.; Frece, J.; Petrović, D.; Pleadin, J. Biocontrol Methods in Avoidance and Downsizing of Mycotoxin Contamination of Food Crops. Processes 2022, 10, 655. https://doi.org/10.3390/pr10040655
Zadravec M, Markov K, Lešić T, Frece J, Petrović D, Pleadin J. Biocontrol Methods in Avoidance and Downsizing of Mycotoxin Contamination of Food Crops. Processes. 2022; 10(4):655. https://doi.org/10.3390/pr10040655
Chicago/Turabian StyleZadravec, Manuela, Ksenija Markov, Tina Lešić, Jadranka Frece, Danijela Petrović, and Jelka Pleadin. 2022. "Biocontrol Methods in Avoidance and Downsizing of Mycotoxin Contamination of Food Crops" Processes 10, no. 4: 655. https://doi.org/10.3390/pr10040655
APA StyleZadravec, M., Markov, K., Lešić, T., Frece, J., Petrović, D., & Pleadin, J. (2022). Biocontrol Methods in Avoidance and Downsizing of Mycotoxin Contamination of Food Crops. Processes, 10(4), 655. https://doi.org/10.3390/pr10040655








